Development and Psychometric Evaluation of Healthcare Access Measures among Women with Ovarian Cancer
Abstract
Simple Summary
Abstract: Introduction
1. Introduction
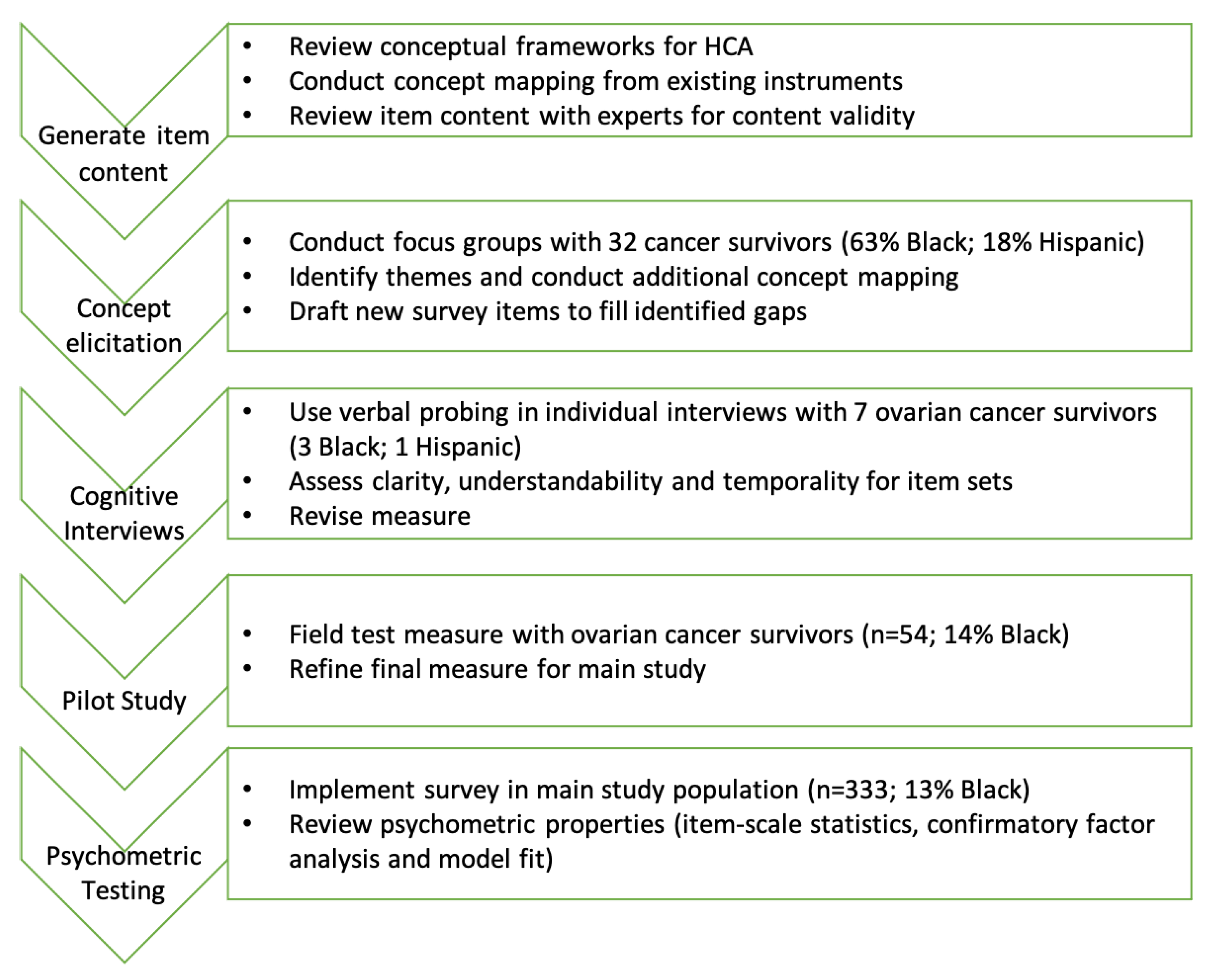
2. Methods
2.1. A. Concept Elicitation
2.2. B. Cognitive Interviews and Pilot Testing
2.3. C. Psychometric Evaluation
3. Results
3.1. A. Concept Elicitation
3.1.1. Dimension 1: Acceptability
3.1.2. Dimension 2: Accommodation
3.1.3. Dimension 3: Availability
3.1.4. Dimension 4: Affordability
3.1.5. Dimension 5: Accessibility
3.1.6. Emergent Codes: Facilitators and Barriers to Treatment
3.2. B. Cognitive Interviews and Pilot Testing
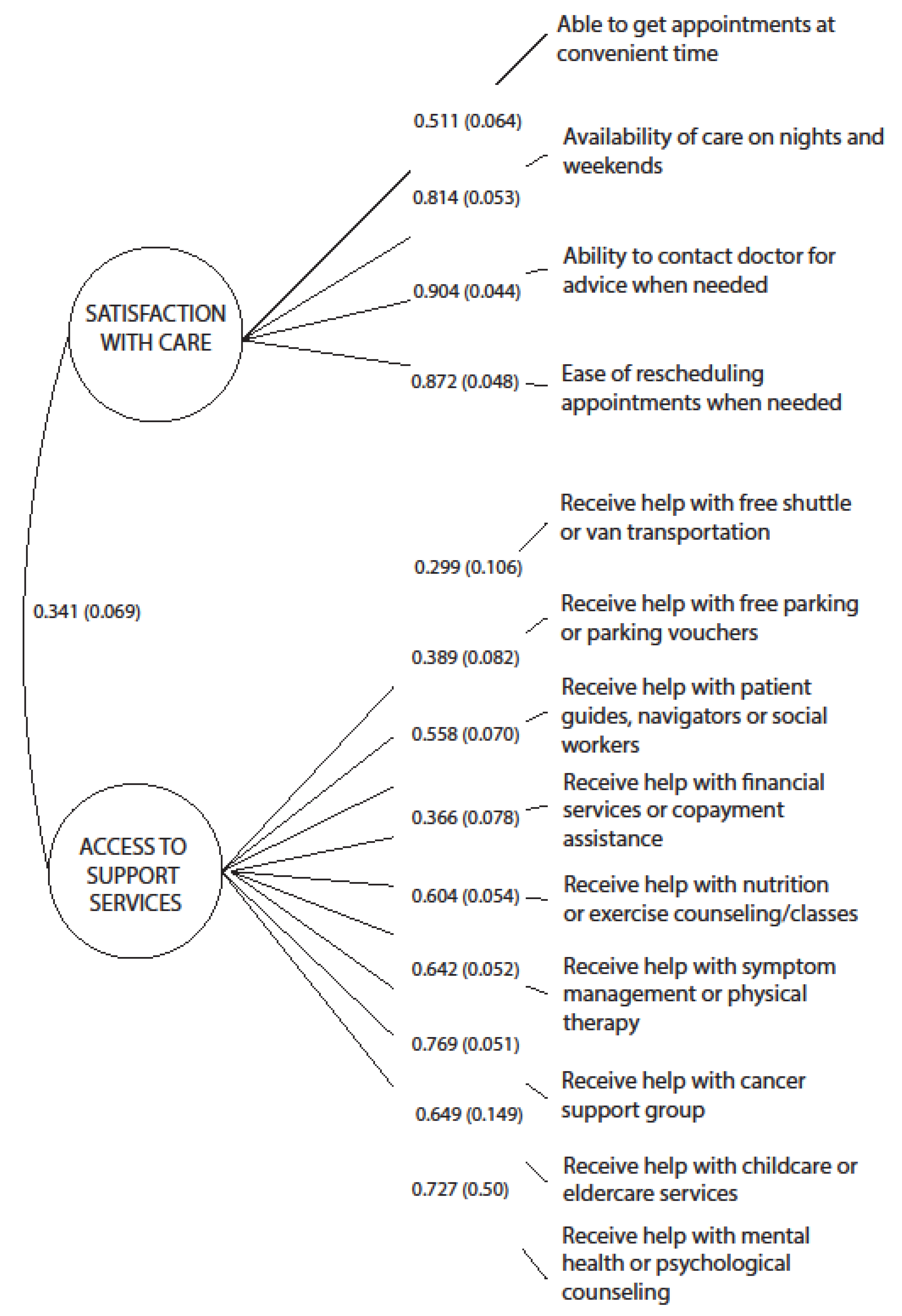
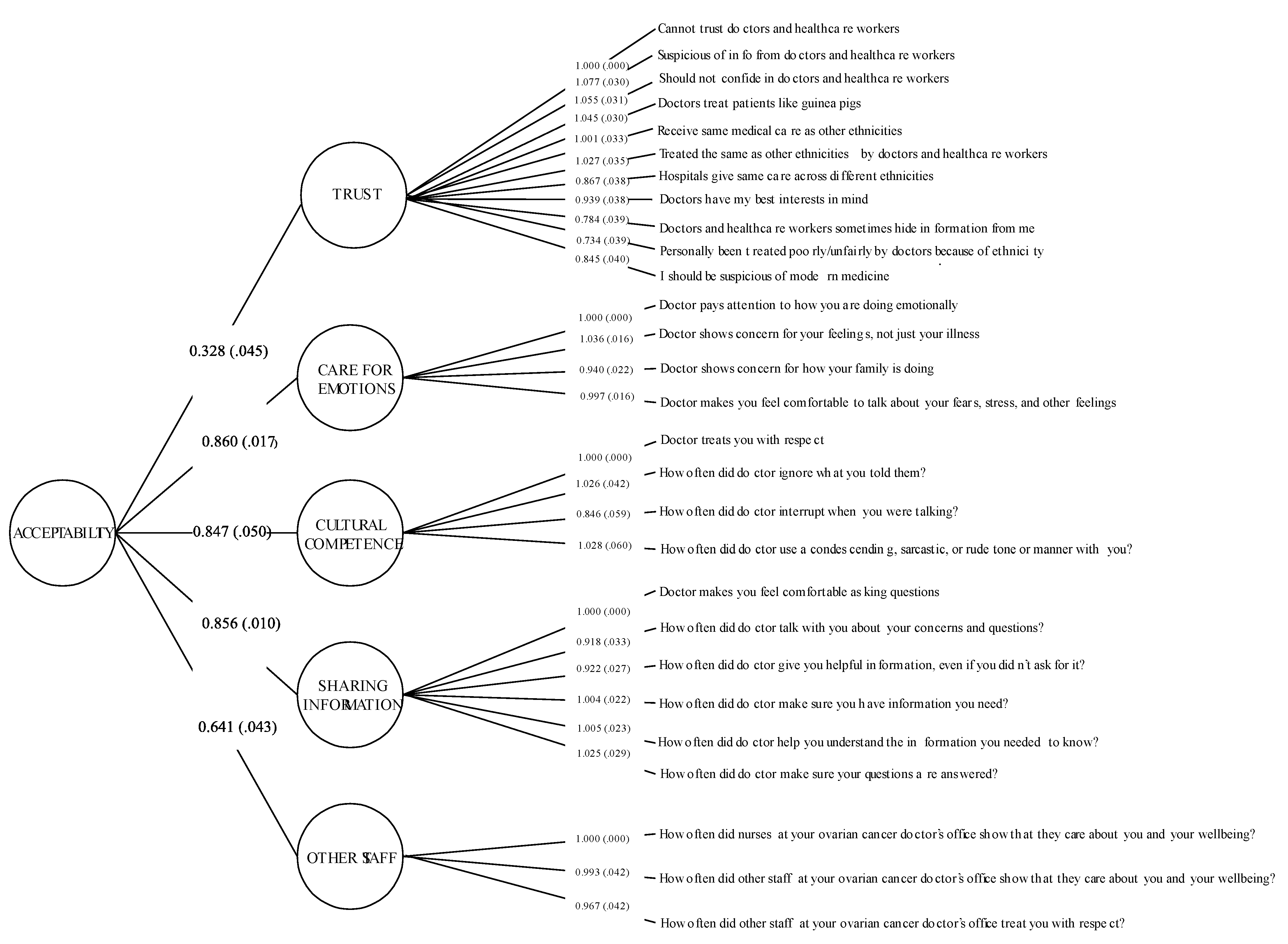
3.3. C. Psychometric Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Disclosures
References
- Aday, L.A.; Andersen, R. A Framework for the Study of Access to Medical Care. Health Serv. Res. 1974, 9, 208–220. [Google Scholar] [PubMed]
- Andersen, R.; Aday, L.A. Access to Medical Care in the U.S.: Realized and Potential. Med. Care 1978, 16, 533–546. [Google Scholar] [CrossRef] [PubMed]
- Penchansky, R.; Thomas, J.W. The concept of access: Definition and relationship to consumer satisfaction. Med. Care 1981, 19, 127–140. [Google Scholar] [CrossRef] [PubMed]
- Thomas, J.W.; Penchansky, R. Relating Satisfaction with Access to Utilization of Services. Med. Care 1984, 22, 553–568. [Google Scholar] [CrossRef] [PubMed]
- Zeybek, B.; Webster, E.; Pogosian, N.; Tymon-Rosario, J.; Balch, A.; Altwerger, G.; Clark, M.; Menderes, G.; Huang, G.; Azodi, M.; et al. Financial toxicity in patients with gynecologic malignancies: A cross sectional study. J. Gynecol. Oncol. 2021, 32, e87. [Google Scholar] [CrossRef]
- Harrison, R.F.; Fu, S.; Sun, C.C.; Zhao, H.; Lu, K.H.; Giordano, S.H.; Meyer, L.A. Patient cost sharing during poly(adenosine diphosphate-ribose) polymerase inhibitor treatment in ovarian cancer. Am. J. Obstet. Gynecol. 2021, 225, 68.e1–68.e11. [Google Scholar] [CrossRef]
- Karpinskyj, C.; Burnell, M.; Gonzalez-Izquierdo, A.; Ryan, A.; Kalsi, J.; Jacobs, I.; Parmar, M.; Menon, U.; Gentry-Maharaj, A. Socioeconomic Status and Ovarian Cancer Stage at Diagnosis: A Study Nested within UKCTOCS. Diagnostics 2020, 10, 89. [Google Scholar] [CrossRef]
- Akinyemiju, T.; Deveaux, A.; Wilson, L.; Gupta, A.; Joshi, A.; Bevel, M.; Omeogu, C.; Ohamadike, O.; Huang, B.; Pisu, M.; et al. Ovarian Cancer Epidemiology, Healthcare Access and Disparities (ORCHiD): Methodology for a population-based study of black, Hispanic and white patients with ovarian cancer. BMJ Open 2021, 11, e052808. [Google Scholar] [CrossRef]
- Flick, U. Designing Qualitative Research; SAGE Publications: London, UK, 2007. [Google Scholar]
- Safran, D.G.; Karp, M.; Coltin, K.; Chang, H.; Li, A.; Ogren, J.; Rogers, W.H. Measuring patients’ experiences with individual primary care physicians. Results of a statewide demonstration project. J. Gen. Intern. Med. 2006, 21, 13–21. [Google Scholar] [CrossRef]
- Centers for Medicare & Medicaid Services. Medicare Current Beneficiary Survey (MCBS). Available online: https://www.cms.gov/Research-Statistics-Data-and-Systems/Research/MCBS (accessed on 4 October 2022).
- Thompson, H.S.; Valdimarsdottir, H.B.; Winkel, G.; Jandorf, L.; Redd, W. The Group-Based Medical Mistrust Scale: Psychometric properties and association with breast cancer screening. Prev. Med. 2003, 38, 209–218. [Google Scholar] [CrossRef]
- Reeve, B.B.; Thissen, D.M.; Bann, C.M.; Mack, N.; Treiman, K.; Sanoff, H.K.; Roach, N.; Magnus, B.E.; He, J.; Wagner, L.; et al. Psychometric evaluation and design of patient-centered communication measures for cancer care settings. Patient Educ. Couns. 2017, 100, 1322–1328. [Google Scholar] [CrossRef] [PubMed]
- Agency for Healthcare Research and Quality. CAHPS Surveys and Tools to Advance Patient-Centered Care. Available online: https://www.ahrq.gov/cahps/index.html (accessed on 4 October 2022).
- Thompson, B. Confirmatory factor analysis decision sequence. In Exploratory and Confirmatory Factor Analysis: Understanding Concepts and Applications; American Psychological Association: Washington, DC, USA, 2004. [Google Scholar]
- Brown, T.A. Confirmatory Factor Analysis for Applied Research; Guilford Publications: New York, NY, USA, 2006. [Google Scholar]
- Brown, T.A. Confirmatory Factor Analysis for Applied Research; Guilford Publications: New York, NY, USA, 2015. [Google Scholar]
- Browne, M.W.; Cudeck, R. Alternative ways of assessing model fit. Sociol. Methods Res. 1992, 21, 230–258. [Google Scholar] [CrossRef]
- Tucker, L.R.; Lewis, C. A reliability coefficient for maximum likelihood factor analysis. Psychometrika 1973, 38, 1–10. [Google Scholar] [CrossRef]
- Bentler, P.M. Comparative Fit Indexes in Structural Models. Psychol. Bull. 1990, 107, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Brunner, M.; Süß, H.-M. Analyzing the Reliability of Multidimensional Measures: An Example from Intelligence Research. Educ. Psychol. Meas. 2005, 65, 227–240. [Google Scholar] [CrossRef]
- Hair, J.; Hult, T.; Ringle, C.; Sarstedt, M. A Primer on Partial Least Squares Structural Equation Modeling (PLS-SEM); Sage Publications, Inc.: Thousand Oaks, CA, USA, 2014. [Google Scholar]
- Fornell, C.; Larcker, D.F. Evaluating Structural Equation Models with Unobservable Variables and Measurement Error. J. Mark. Res. 1981, 18, 39–50. [Google Scholar] [CrossRef]
- E Lasser, K.; Ayanian, J.Z.; Fletcher, R.H.; Good, M.-J.D. Barriers to colorectal cancer screening in community health centers: A qualitative study. BMC Fam. Pract. 2008, 9, 15. [Google Scholar] [CrossRef]
- Sharf, B.F.; Stelljes, L.A.; Gordon, H.S. ‘A little bitty spot and I’m a big man’: Patients’ perspectives on refusing diagnosis or treatment for lung cancer. Psycho-oncology 2005, 14, 636–646. [Google Scholar] [CrossRef]
- Kinlock, B.L.; Parker, L.J.; Bowie, J.V.; Howard, D.L.; LaVeist, T.A.; Thorpe, J.R.J. High Levels of Medical Mistrust are Associated with Low Quality of Life among Black and White Men with Prostate Cancer. Cancer Control 2017, 24, 72–77. [Google Scholar] [CrossRef]
- Schwei, R.J.; Kadunc, K.; Nguyen, A.L.; Jacobs, E.A. Impact of sociodemographic factors and previous interactions with the health care system on institutional trust in three racial/ethnic groups. Patient Educ. Couns. 2014, 96, 333–338. [Google Scholar] [CrossRef]
- Jacobs, E.; Mendenhall, E.; Scheck-McAlearney, A.; Rolle, I.; Whitaker, E.; Warnecke, R.; Ferrans, C.E. An Exploratory Study of How Trust in Health Care Institutions Varies across African American, Hispanic and white Populations. Commun. Med. 2011, 8, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Ayers, S.L.; Kronenfeld, J.J. The association between perceived provider discrimination, healthcare utilization and health status in racial and ethnic minorities. Ethn. Dis. 2009, 19, 330–337. [Google Scholar] [PubMed]
- Freedman, T.G. Prescriptions for health providers: From cancer patients. Cancer Nurs. 2003, 26, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Katapodi, M.C.; Facione, N.C.; Humphreys, J.C.; Dodd, M.J. Perceived breast cancer risk: Heuristic reasoning and search for a dominance structure. Soc. Sci. Med. 2005, 60, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Lacey, M.D. The Experience of Using Decisional Support Aids by Patients with Breast Cancer. Oncol. Nurs. Forum 2002, 29, 1491–1497. [Google Scholar] [CrossRef]
- E Goldman, R.; Risica, P.M. Perceptions of breast and cervical cancer risk and screening among Dominicans and Puerto Ricans in Rhode Island. Ethn. Dis. 2004, 14, 32–42. [Google Scholar]
- Guan, M.; Han, J.Y.; Shah, D.V.; Gustafson, D.H. Exploring the Role of Social Support in Promoting Patient Participation in Health Care among Women with Breast Cancer. Health Commun. 2020, 36, 1581–1589. [Google Scholar] [CrossRef]
- Cohen, S.D.; Sharma, T.; Acquaviva, K.; Peterson, R.A.; Patel, S.S.; Kimmel, P.L. Social Support and Chronic Kidney Disease: An Update. Adv. Chronic Kidney Dis. 2007, 14, 335–344. [Google Scholar] [CrossRef]
- Arora, N.K.; Rutten, L.J.F.; Gustafson, D.H.; Moser, R.; Hawkins, R.P. Perceived helpfulness and impact of social support provided by family, friends, and health care providers to women newly diagnosed with breast cancer. Psycho-Oncology 2007, 16, 474–486. [Google Scholar] [CrossRef]
- White-Means, S.; Dapremont, J.; Davis, B.D.; Thompson, T. Who Can Help Us on This Journey? African American Woman with Breast Cancer: Living in a City with Extreme Health Disparities. Int. J. Environ. Res. Public Health 2020, 17, 1126. [Google Scholar] [CrossRef]
- Ashing-Giwa, K.T.; Padilla, G.; Tejero, J.; Kraemer, J.; Wright, K.; Coscarelli, A.; Clayton, S.; Williams, I.; Hills, D. Understanding the breast cancer experience of women: A qualitative study of African American, Asian American, Latina and Caucasian cancer survivors. Psycho-Oncology 2003, 13, 408–428. [Google Scholar] [CrossRef]
- Mollica, M.; Nemeth, L. Transition from Patient to Survivor in African American Breast Cancer Survivors. Cancer Nurs. 2015, 38, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Gates, M.F.; Lackey, N.R.; Brown, G. Caring demands and delay in seeking care in African American women newly diagnosed with breast cancer: An ethnographic, photographic study. Oncol. Nurs. Forum 2001, 28, 529–537. [Google Scholar] [PubMed]
- Henderson, P.D.; Gore, S.V.; Davis, B.L.; Condon, E.H. African American Women Coping with Breast Cancer: A Qualitative Analysis. Oncol. Nurs. Forum 2003, 30, 641–647. [Google Scholar] [CrossRef] [PubMed]
- Patt, D.; Gordan, L.; Diaz, M.; Okon, T.; Grady, L.; Harmison, M.; Markward, N.; Sullivan, M.; Peng, J.; Zhou, A. Impact of COVID-19 on Cancer Care: How the Pandemic Is Delaying Cancer Diagnosis and Treatment for American Seniors. JCO Clin. Cancer Inform. 2020, 4, 1059–1071. [Google Scholar] [CrossRef]
- Du, S.; Carfang, L.; Restrepo, E.; Benjamin, C.; Epstein, M.M.; Fairley, R.; Roudebush, L.; Hertz, C.; Eshraghi, L.; Warner, E.T. Patient-Reported Experiences of Breast Cancer Screening, Diagnosis, and Treatment Delay, and Telemedicine Adoption during COVID-19. Curr. Oncol. 2022, 29, 5919–5932. [Google Scholar] [CrossRef]
- Richards, M.; Anderson, M.; Carter, P.; Ebert, B.L.; Mossialos, E. The impact of the COVID-19 pandemic on cancer care. Nat. Rev. Cancer 2020, 1, 565–567. [Google Scholar] [CrossRef]
| HCA Dimension | Operational Definition 1 | Examples |
|---|---|---|
| Acceptability | Patient’s attitude to personal and practice characteristics of healthcare provider | Empathy; compassion; provider’s respect for faith and beliefs; patient–provider communication |
| Accessibility | The physical location of medical professionals and treatment(s) in relation to the patient | Location and distance; transportation available; convenience of parking |
| Accommodation | Organization of healthcare resources in relation to patients’ convenience and ability to accommodate such services | Hospital/clinic schedule; wait times; ease of scheduling/rescheduling; language Accessibility/interpreter |
| Affordability | Pricing, willingness, and ability to pay for treatment and other forms of supportive and/or follow-up care | Income; insurance; insurance co-pays; missed hours of work/pay or forced to quit job |
| Availability | Type, quality, and volume of healthcare services in need relation to patient need | Number of doctors/hospitals, provider specialty and training; hospital/provider volume |
| Emergent Code | Operational Definition 2 | Examples |
| Support | Factor or characteristic that supports one’s treatment journey | Attitude; faith; self-advocacy; support system |
| Challenges | Factor or characteristic that negatively impacts one’s treatment journey | Fear, inadequate support system, mental and emotional wellness, role conflict |
| # Groups Mentioned 1 | # Total Mentions 2 | % Total Mentions | |
|---|---|---|---|
| HCA Dimension | |||
| Acceptability | 7 | 108 | 41% |
| Accessibility | 5 | 28 | 11% |
| Accommodation | 7 | 54 | 20% |
| Affordability | 7 | 37 | 14% |
| Availability | 7 | 38 | 14% |
| Facilitators | |||
| Attitude | 5 | 5 | 5% |
| Faith | 6 | 31 | 28% |
| Self-advocacy | 4 | 9 | 8% |
| Support system | 7 | 65 | 59% |
| Barriers | |||
| Attitude | 2 | 12 | 12% |
| Fear | 7 | 46 | 46% |
| Inadequate support system | 5 | 15 | 15% |
| Mental and emotional awareness | 4 | 18 | 18% |
| Role conflict | 4 | 9 | 9% |
| Fit Statistic | Hypothesized Model Structures | ||
|---|---|---|---|
| Accommodation | 1-Factor First-Order Model | 3-Factor First-Order Model | 2-Factor First-Order Model |
| χ2 | 273.091 * | 92.687 | 77.452 |
| Df | 65 | 74 | 64 |
| P | 0.000 | 0.070 | 0.120 |
| RMSEA | 0.098 | 0.028 | 0.025 |
| CFI | 0.780 | 0.980 | 0.986 |
| TLI | 0.736 | 0.976 | 0.983 |
| SRMR | 0.142 | 0.080 | 0.082 |
| Acceptability | 1-Factor First-Order Model | 5-Factor First-Order Model | 5-Factor Higher-Order Model |
| χ2 | 3536.770 * | 1124.280 * | 1119.895 * |
| Df | 350 | 340 | 346 |
| P | 0.0000 | 0.0000 | 0.0000 |
| RMSEA | 0.166 | 0.083 | 0.082 |
| CFI | 0.825 | 0.957 | 0.958 |
| TLI | 0.812 | 0.952 | 0.954 |
| SRMR | 0.218 | 0.087 | 0.093 |
| Multi-Item Scales (# of Items) | Latent Factor’s Composite Reliability (Ω) | Latent Factor’s Average Variance Extracted (AVE) |
|---|---|---|
| Accommodation (# items: 14) | ||
| Satisfaction with care (# items: 5) | 0.80 | 0.51 |
| Access to support services (# items: 9) | 0.82 | 0.36 |
| Acceptability (# items: 28) | 0.89 | 0.65 |
| Trust (# items: 11) | 0.93 | 0.56 |
| Care for emotions (# items: 4) | 0.94 | 0.79 |
| Cultural competence (# items: 4) | 0.91 | 0.71 |
| Sharing information (# items: 6) | 0.94 | 0.74 |
| Other staff (# items: 3) | 0.92 | 0.80 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akinyemiju, T.; Joshi, A.; Deveaux, A.; Wilson, L.E.; Chen, D.; Meernik, C.; Bevel, M.; Gathings, J.; Fish, L.; Barrett, N.; et al. Development and Psychometric Evaluation of Healthcare Access Measures among Women with Ovarian Cancer. Cancers 2022, 14, 6266. https://doi.org/10.3390/cancers14246266
Akinyemiju T, Joshi A, Deveaux A, Wilson LE, Chen D, Meernik C, Bevel M, Gathings J, Fish L, Barrett N, et al. Development and Psychometric Evaluation of Healthcare Access Measures among Women with Ovarian Cancer. Cancers. 2022; 14(24):6266. https://doi.org/10.3390/cancers14246266
Chicago/Turabian StyleAkinyemiju, Tomi, Ashwini Joshi, April Deveaux, Lauren E. Wilson, Dandan Chen, Clare Meernik, Malcolm Bevel, Jen Gathings, Laura Fish, Nadine Barrett, and et al. 2022. "Development and Psychometric Evaluation of Healthcare Access Measures among Women with Ovarian Cancer" Cancers 14, no. 24: 6266. https://doi.org/10.3390/cancers14246266
APA StyleAkinyemiju, T., Joshi, A., Deveaux, A., Wilson, L. E., Chen, D., Meernik, C., Bevel, M., Gathings, J., Fish, L., Barrett, N., Worthy, V., Boyce, X., Martin, K., Robinson, C., Pisu, M., Liang, M., Potosky, A., Huang, B., Ward, K., ... Reeve, B. B. (2022). Development and Psychometric Evaluation of Healthcare Access Measures among Women with Ovarian Cancer. Cancers, 14(24), 6266. https://doi.org/10.3390/cancers14246266







