Diagnostic Value of Bronchoscopy for Peripheral Metastatic Lung Tumors
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Equipment and Procedures
2.3. Variables
2.4. Statistical Analyses
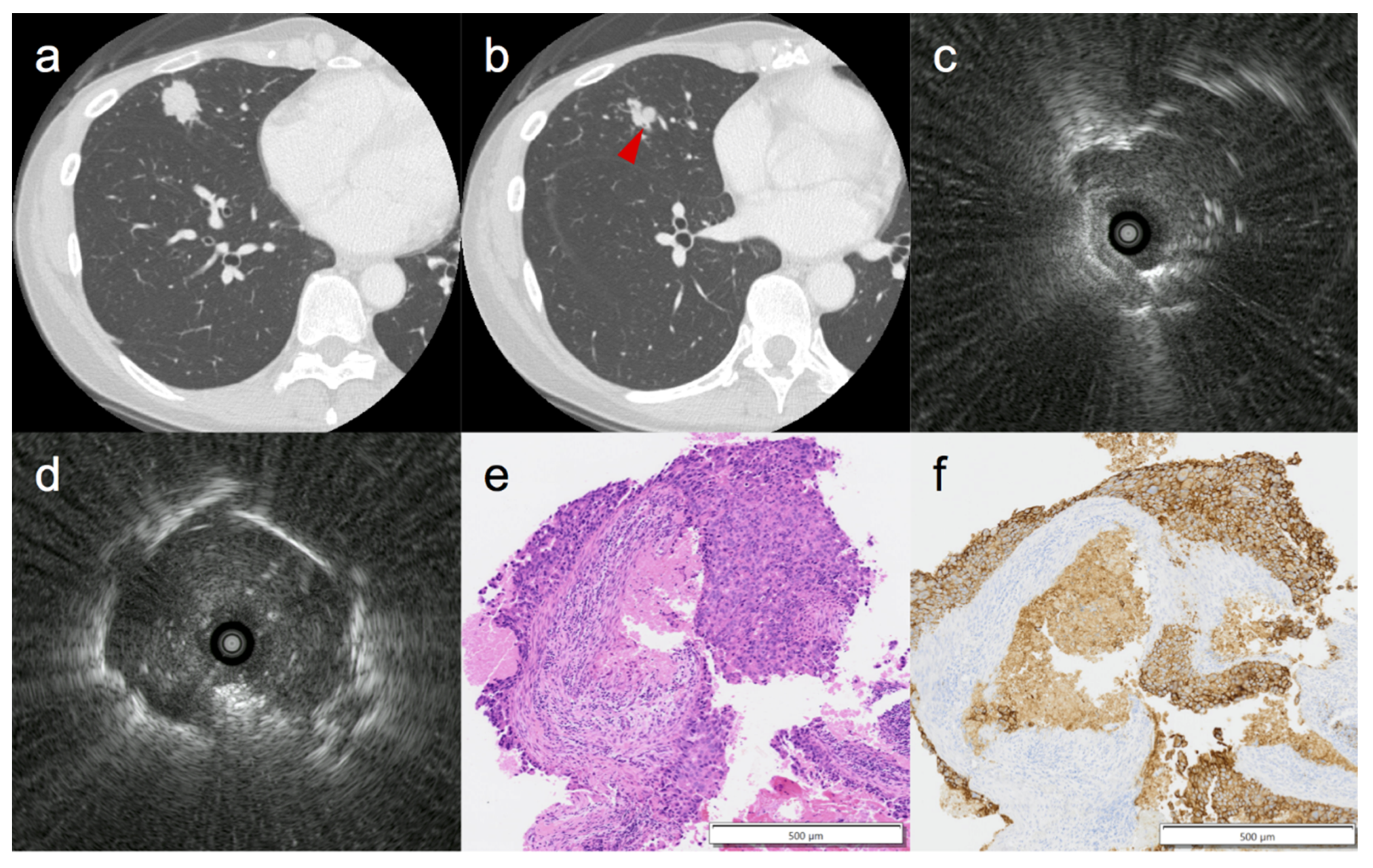
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Debois, J.M. TxNxM1: The Anatomy and Clinics of Metastatic Cancer; Kluwer Academic Publishers: Boston, MA, USA, 2002. [Google Scholar]
- Zhao, X.; Wen, X.; Wei, W.; Chen, Y.; Zhu, J.; Wang, C. Clinical Characteristics and Prognoses of Patients Treated Surgically for Metastatic Lung Tumors. Oncotarget 2017, 8, 46491–46497. [Google Scholar] [CrossRef] [PubMed]
- Libshitz, H.I.; North, L.B. Pulmonary Metastases. Radiol. Clin. N. Am. 1982, 20, 437–451. [Google Scholar] [PubMed]
- Gross, B.H.; Glazer, G.M.; Bookstein, F.L. Multiple Pulmonary Nodules Detected by Computed Tomography: Diagnostic Implications. J. Comput. Assist. Tomogr. 1985, 9, 880–885. [Google Scholar] [CrossRef] [PubMed]
- Regnard, J.F.; Grunenwald, D.; Spaggiari, L.; Girard, P.; Elias, D.; Ducreux, M.; Baldeyrou, P.; Levasseur, P. Surgical Treatment of Hepatic and Pulmonary Metastases From Colorectal Cancers. Ann. Thorac. Surg. 1998, 66, 214–218; discussion 218. [Google Scholar] [CrossRef]
- Wolff, A.C.; Hammond, M.E.H.; Allison, K.H.; Harvey, B.E.; Mangu, P.B.; Bartlett, J.M.S.; Bilous, M.; Ellis, I.O.; Fitzgibbons, P.; Hanna, W.; et al. Human Epidermal Growth Factor receptor 2 Testing in Breast Cancer: American Society of Clinical Oncology/College of American Pathologists Clinical Practice Guideline Focused Update. J. Clin. Oncol. 2018, 36, 2105–2122. [Google Scholar] [CrossRef] [PubMed]
- Singarayer, R.; Mete, O.; Perrier, L.; Thabane, L.; Asa, S.L.; Van Uum, S.; Ezzat, S.; Goldstein, D.P.; Sawka, A.M. A Systematic Review and Meta-Analysis of the Diagnostic Performance of BRAF V600E Immunohistochemistry in Thyroid Histopathology. Endocr. Pathol. 2019, 30, 201–218. [Google Scholar] [CrossRef]
- Wiener, R.S.; Schwartz, L.M.; Woloshin, S.; Welch, H.G. Population-Based Risk for Complications after Transthoracic Needle Lung Biopsy of a Pulmonary Nodule: An Analysis of Discharge Records. Ann. Intern. Med. 2011, 155, 137–144. [Google Scholar] [CrossRef]
- Tomiyama, N.; Yasuhara, Y.; Nakajima, Y.; Adachi, S.; Arai, Y.; Kusumoto, M.; Eguchi, K.; Kuriyama, K.; Sakai, F.; Noguchi, M.; et al. CT-Guided Needle Biopsy of Lung Lesions: A Survey of Severe Complication Based on 9783 Biopsies in Japan. Eur. J. Radiol. 2006, 59, 60–64. [Google Scholar] [CrossRef]
- Poe, R.H.; Ortiz, C.; Israel, R.H.; Marin, M.G.; Qazi, R.; Dale, R.C.; Greenblatt, D.G. Sensitivity, Specificity, and Predictive Values of Bronchoscopy in Neoplasm Metastatic to Lung. Chest 1985, 88, 84–88. [Google Scholar] [CrossRef]
- Díaz, G.; Jiménez, D.; Domínguez-Reboiras, S.; Carrillo, F.; Pérez-Rodríguez, E. Yield of Bronchoscopy in the Diagnosis of Neoplasm Metastatic to Lung. Respir. Med. 2003, 97, 27–29. [Google Scholar] [CrossRef][Green Version]
- Zavala, D.C. Diagnostic Fiberoptic Bronchoscopy: Techniques and Results of Biopsy in 600 Patients. Chest 1975, 68, 12–19. [Google Scholar] [CrossRef]
- Rivera, M.P.; Mehta, A.C.; Wahidi, M.M. Establishing the diagnosis of lung cancer: Diagnosis and management of lung cancer, 3rd ed.: American college of chest physicians evidence-based clinical practice guidelines. Chest 2013, 143, e142S–e165S. [Google Scholar] [CrossRef]
- Ali, M.S.; Trick, W.; Mba, B.I.; Mohananey, D.; Sethi, J.; Musani, A.I. Radial Endobronchial Ultrasound for the Diagnosis of Peripheral Pulmonary Lesions: A Systematic Review and Meta-Analysis. Respirology 2017, 22, 443–453. [Google Scholar] [CrossRef]
- Casutt, A.; Prella, M.; Beigelman-Aubry, C.; Fitting, J.W.; Nicod, L.; Koutsokera, A.; Lovis, A. Fluoroscopic-Guided Radial Endobronchial Ultrasound without Guide Sheath for Peripheral Pulmonary Lesions: A Safe and Efficient Combination. Arch. Bronconeumol. 2015, 51, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.H.; Cheng, W.C.; Wu, B.R.; Chen, C.Y.; Chen, W.C.; Hsia, T.C.; Liao, W.C.; Tu, C.Y.; Shih, C.M.; Hsu, W.H.; et al. Improved Diagnostic Yield of Bronchoscopy in Peripheral Pulmonary Lesions: Combination of Radial Probe Endobronchial Ultrasound and Rapid On-Site Evaluation. J. Thorac. Dis. 2015, 7 (Suppl. 4), S418–S425. [Google Scholar] [PubMed]
- Yoshikawa, M.; Sukoh, N.; Yamazaki, K.; Kanazawa, K.; Fukumoto, S.; Harada, M.; Kikuchi, E.; Munakata, M.; Nishimura, M.; Isobe, H. Diagnostic Value of Endobronchial Ultrasonography With a Guide Sheath for Peripheral Pulmonary Lesions without X-Ray Fluoroscopy. Chest 2007, 131, 1788–1793. [Google Scholar] [CrossRef] [PubMed]
- Baaklini, W.A.; Reinoso, M.A.; Gorin, A.B.; Sharafkaneh, A.; Manian, P. Diagnostic Yield of Fiberoptic Bronchoscopy in Evaluating Solitary Pulmonary Nodules. Chest 2000, 117, 1049–1054. [Google Scholar] [CrossRef] [PubMed]
- Gaeta, M.; Pandolfo, I.; Volta, S.; Russi, E.G.; Bartiromo, G.; Girone, G.; La Spada, F.; Barone, M.; Casablanca, G.; Minutoli, A. Bronchus sign on CT in peripheral carcinoma of the lung: Value in predicting results of transbronchial biopsy. AJR Am. J. Roentgenol. 1991, 157, 1181–1185. [Google Scholar] [CrossRef] [PubMed]
- Braman, S.S.; Whitcomb, M.E. Endobronchial Metastasis. Arch. Intern. Med. 1975, 135, 543–547. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, W.A.; Mark, J.B.D. Metastatic Malignancies from Distant Sites to the Tracheobronchial Tree. J. Thorac. Cardiovasc. Surg. 1980, 79, 499–503. [Google Scholar] [CrossRef]
- Asano, F.; Shinagawa, N.; Ishida, T.; Tsuzuku, A.; Tachihara, M.; Kanazawa, K.; Yamada, N.; Oizumi, S.; Moriya, H. Virtual Bronchoscopic Navigation Improves the Diagnostic Yield of Radial-Endobronchial Ultrasound for Peripheral Pulmonary Lesions With Involved Bronchi on CT. Intern. Med. 2015, 54, 1021–1025. [Google Scholar] [CrossRef]
- Ali, M.S.; Sethi, J.; Taneja, A.; Musani, A.; Maldonado, F. Computed Tomography Bronchus Sign and the Diagnostic Yield of Guided Bronchoscopy for Peripheral Pulmonary Lesions. A Systematic Review and Meta-Analysis. Ann. Am. Thorac. Soc. 2018, 15, 978–987. [Google Scholar] [CrossRef] [PubMed]
- Guvenc, C.; Yserbyt, J.; Testelmans, D.; Zanca, F.; Carbonez, A.; Ninane, V.; De Wever, W.; Dooms, C. Computed Tomography Characteristics Predictive for Radial EBUS-Miniprobe-Guided Diagnosis of Pulmonary Lesions. J. Thorac. Oncol. 2015, 10, 472–478. [Google Scholar] [CrossRef] [PubMed]
- Minezawa, T.; Okamura, T.; Yatsuya, H.; Yamamoto, N.; Morikawa, S.; Yamaguchi, T.; Morishita, M.; Niwa, Y.; Takeyama, T.; Mieno, Y.; et al. Bronchus Sign on Thin-Section Computed Tomography Is a Powerful Predictive Factor for Successful Transbronchial Biopsy Using Endobronchial Ultrasound with a Guide Sheath for Small Peripheral Lung Lesions: A Retrospective Observational Study. BMC Med. Imaging 2015, 15, 21. [Google Scholar] [CrossRef]
- Evison, M.; Crosbie, P.A.J.; Morris, J.; Martin, J.; Barber, P.V.; Booton, R. Can Computed Tomography Characteristics Predict Outcomes in Patients Undergoing Radial Endobronchial Ultrasound-Guided Biopsy of Peripheral Lung Lesions? J. Thorac. Oncol. 2014, 9, 1393–1397. [Google Scholar] [CrossRef] [PubMed]
- Döme, B.; Hendrix, M.J.C.; Paku, S.; Tóvári, J.; Tímár, J. Alternative Vascularization Mechanisms in Cancer: Pathology and Therapeutic Implications. Am. J. Pathol. 2007, 170, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Hayama, M.; Izumo, T.; Chavez, C.; Matsumoto, Y.; Tsuchida, T.; Sasada, S. Additional Transbronchial Needle Aspiration Through a Guide Sheath for Peripheral Pulmonary Lesions That Cannot Be Detected by Radial EBUS. Clin. Respir. J. 2017, 11, 757–764. [Google Scholar] [CrossRef]
- Gasparini, S.; Bonifazi, M.; Wang, K.P. Transbronchial Needle Aspirations vs. Percutaneous Needle Aspirations. J. Thorac. Dis. 2015, 7 (Suppl. 4), S300–S303. [Google Scholar]
- Chao, T.Y.; Chien, M.T.; Lie, C.H.; Chung, Y.H.; Wang, J.L.; Lin, M.C. Endobronchial Ultrasonography-Guided Transbronchial Needle Aspiration Increases the Diagnostic Yield of Peripheral Pulmonary Lesions: A Randomized Trial. Chest 2009, 136, 229–236. [Google Scholar] [CrossRef]
- Kho, S.S.; Chan, S.K.; Yong, M.C.; Tie, S.T. Performance of Transbronchial Cryobiopsy in Eccentrically and Adjacently Orientated Radial Endobronchial Ultrasound Lesions. ERJ Open Res. 2019, 5, 00135–02019. [Google Scholar] [CrossRef]
- Takeshita, J.; Masago, K.; Kato, R.; Hata, A.; Kaji, R.; Fujita, S.; Katakami, N. CT-Guided Fine-Needle Aspiration and Core Needle Biopsies of Pulmonary Lesions: A Single-Center Experience With 750 Biopsies in Japan. AJR Am. J. Roentgenol. 2015, 204, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Saji, H.; Nakamura, H.; Tsuchida, T.; Tsuboi, M.; Kawate, N.; Konaka, C.; Kato, H. The Incidence and the Risk of Pneumothorax and Chest Tube Placement after Percutaneous CT-Guided Lung Biopsy: The Angle of the Needle Trajectory Is a Novel Predictor. Chest 2002, 121, 1521–1526. [Google Scholar] [CrossRef]
- Pastorino, U.; Buyse, M.; Friedel, G.; Ginsberg, R.J.; Girard, P.; Goldstraw, P.; Johnston, M.; McCormack, P.; Pass, H.; Putnam, J.B.; et al. Long-Term Results of Lung Metastasectomy: Prognostic Analyses Based on 5206 Cases. J. Thorac. Cardiovasc. Surg. 1997, 113, 37–49. [Google Scholar] [CrossRef]
- Blackmon, S.H.; Shah, N.; Roth, J.A.; Correa, A.M.; Vaporciyan, A.A.; Rice, D.C.; Hofstetter, W.; Walsh, G.L.; Benjamin, R.; Pollock, R.; et al. Resection of Pulmonary and Extrapulmonary Sarcomatous Metastases Is Associated With Long-Term Survival. Ann. Thorac. Surg. 2009, 88, 877–884; discussion 884. [Google Scholar] [CrossRef] [PubMed]
- Younes, R.N.; Fares, A.L.; Gross, J.L. Pulmonary Metastasectomy: A Multivariate Analysis of 440 Patients Undergoing Complete Resection. Interact. Cardiovasc. Thorac. Surg. 2012, 14, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, A.; Arce, N.; Bernardo, J.; Eugénio, L.; Antunes, M.J. Surgical Resection of Lung Metastases From Epithelial Tumors. Ann. Thorac. Surg. 2004, 77, 431–437. [Google Scholar] [CrossRef]
| Variable | N (%) or Median (Range) |
|---|---|
| Age (years) | 66 (27–86) |
| ≤65 | 109 (46.4) |
| >65 | 126 (53.6) |
| Sex | |
| Male | 112 (47.7) |
| Female | 123 (52.3) |
| Lesion size (mm) | 18.8 (6.0–93.4) |
| Small (≤20) | 130 (55.3) |
| Large (>20) | 105 (44.7) |
| Lobe | |
| Upper | 100 (42.5) |
| Middle or lingular | 42 (17.9) |
| Lower | 93 (39.6) |
| Distance from the costal pleura (mm) | 14.2 (0–65.0) |
| Near (≤10) | 99 (42.1) |
| Far (>10) | 136 (57.9) |
| Location | |
| Outer 1/3 | 123 (52.3) |
| Inner 2/3 | 112 (47.7) |
| Bronchus sign | |
| Positive | 126 (53.6) |
| Negative | 105 (44.7) |
| Visibility on radiography | |
| Visible | 178 (75.7) |
| Invisible | 57 (24.3) |
| Number of pulmonary lesions | |
| Single | 116 (49.4) |
| Multiple | 119 (50.6) |
| Navigation | |
| Used | 204 (86.8) |
| Not used | 31 (13.2) |
| Needle | |
| Used | 107 (45.5) |
| Not used | 128 (54.5) |
| Guide sheath | |
| Used | 146 (62.1) |
| Not used | 89 (37.9) |
| Variable | Diagnostic Cases, N (%) | Nondiagnostic Cases, N (%) | Univariable | Multivariable | |
|---|---|---|---|---|---|
| p-Value | p-Value | Adjusted OR (95% CI) | |||
| Overall | 180 (76.6) | 55 (23.4) | - | - | - |
| Age (years) | 0.122 | 0.228 | 1.54 (0.76–3.14) | ||
| ≤65 | 78 (71.6) | 31 (28.4) | |||
| >65 | 102 (81.0) | 24 (19.1) | |||
| Sex | 1.000 | 0.771 | 1.11 (0.54–2.27) | ||
| Male | 86 (76.8) | 26 (23.2) | |||
| Female | 94 (76.4) | 29 (23.6) | |||
| Lesion size (mm) | < 0.001 | 0.043 | 2.26 (1.01–5.04) | ||
| Small (≤20) | 88 (67.7) | 42 (32.3) | |||
| Large (>20) | 92 (87.6) | 13 (12.4) | |||
| Lobe | 0.398 | NA | NA | ||
| Upper | 79 (79.0) | 21 (21.0) | |||
| Middle or lingular | 34 (81.0) | 8 (19.0) | |||
| Lower | 67 (72.0) | 26 (28.0) | |||
| Distance from the costal pleura (mm) | 0.086 | NA | NA | ||
| Near (≤10) | 70 (70.7) | 29 (29.3) | |||
| Far (>10) | 110 (80.9) | 26 (19.1) | |||
| Location | 0.005 | 0.004 | 2.79 (1.36–5.70) | ||
| Outer 1/3 | 85 (69.1) | 38 (30.9) | |||
| Inner 2/3 | 95 (84.8) | 17 (15.2) | |||
| Bronchus sign | 0.063 | 0.207 | 1.58 (0.77–3.22) | ||
| Positive | 103 (81.8) | 23 (18.3) | |||
| Negative | 77 (70.6) | 32 (29.4) | |||
| Visibility on radiography | < 0.001 | 0.015 | 3.29 (1.57–6.91) | ||
| Visible | 148 (83.2) | 30 (16.9) | |||
| Invisible | 32 (56.1) | 25 (43.9) | |||
| Number of pulmonary lesions | 0.090 | 0.148 | 1.64 (0.84–3.22) | ||
| Single | 83 (71.6) | 33 (28.5) | |||
| Multiple | 97 (81.5) | 22 (18.5) | |||
| Navigation | 0.254 | NA | NA | ||
| Used | 159 (77.9) | 45 (22.1) | |||
| Not used | 21 (67.7) | 10 (32.3) | |||
| Needle | 0.359 | NA | NA | ||
| Used | 85 (79.4) | 22 (20.6) | |||
| Not used | 95 (74.2) | 33 (25.8) | |||
| Guide sheath | 0.017 | NA | NA | ||
| Used | 104 (71.2) | 42 (28.8) | |||
| Not used | 76 (85.4) | 13 (14.6) | |||
| Tumor Type | Diagnostic Cases, N (%) |
|---|---|
| Breast cancer | 46/52 (88.5) |
| Colorectal cancer | 33/38 (86.8) |
| Uterine cancer | 15/23 (65.2) |
| Pancreatic cancer | 8/13 (61.5) |
| Lung cancer | 7/13 (53.8) |
| Oral cancer | 7/10 (70.0) |
| Gastric cancer | 7/8 (87.5) |
| Esophageal cancer | 7/8 (87.5) |
| Laryngeal cancer | 6/8 (75.0) |
| Urothelial cancer | 6/7 (85.7) |
| Biliary tract cancer | 6/7 (85.7) |
| Renal cancer | 5/7 (71.4) |
| Prostate cancer | 5/6 (75.0) |
| Melanoma | 4/4 (100.0) |
| Liposarcoma | 1/4 (25.0) |
| Uterine leiomyosarcoma | 2/3 (66.7) |
| Thymic cancer | 2/3 (66.7) |
| Sarcoma, NOS | 0/3 (0.0) |
| Solitary fibrous tumor | 2/2 (100.0) |
| Uterine carcinosarcoma | 2/2 (100.0) |
| Epithelioid sarcoma | 2/2 (100.0) |
| Chondrosarcoma | 2/2 (100.0) |
| Hepatocellular cancer | 2/2 (100.0) |
| Ovarian cancer | 1/2 (50.0) |
| Skin cancer | 1/1 (100.0) |
| Angiosarcoma | 1/1 (100.0) |
| Osteosarcoma | 0/1 (0.0) |
| Testicular sarcoma | 0/1 (0.0) |
| Follicular dendritic cell sarcoma | 0/1 (0.0) |
| Germ cell tumor | 0/1 (0.0) |
| R-EBUS Finding | Diagnostic Cases, N (%) | p-Value |
|---|---|---|
| Within the lesion | 92/97 (94.9) | <0.001 |
| Adjacent to the lesion | 85/115 (73.9) | |
| Invisible lesion | 3/23 (13.0) |
| Bronchus Sign | Initial R-EBUS Finding | Best R-EBUS Finding | ||||
|---|---|---|---|---|---|---|
| Within the Lesion, N (%) | Adjacent to the Lesion, N (%) | Invisible Lesion, N (%) | Within the Lesion, N (%) | Adjacent to the Lesion, N (%) | Invisible Lesion, N (%) | |
| Positive | 52 (41.3) | 59 (46.8) | 15 (11.9) | 68 (54.0) | 47 (37.3) | 11 (8.7) |
| Negative | 10 (9.2) | 75 (68.8) | 24 (22.0) | 29 (26.6) | 68 (62.4) | 12 (11.0) |
| Improved *, N (%) | Not Improved, N (%) | p-Value | |
|---|---|---|---|
| Needle | 0.002 | ||
| Used | 31 (29.0) | 76 (71.0) | |
| Not used | 16 (12.5) | 112 (87.5) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsujimoto, Y.; Matsumoto, Y.; Tanaka, M.; Imabayashi, T.; Uchimura, K.; Tsuchida, T. Diagnostic Value of Bronchoscopy for Peripheral Metastatic Lung Tumors. Cancers 2022, 14, 375. https://doi.org/10.3390/cancers14020375
Tsujimoto Y, Matsumoto Y, Tanaka M, Imabayashi T, Uchimura K, Tsuchida T. Diagnostic Value of Bronchoscopy for Peripheral Metastatic Lung Tumors. Cancers. 2022; 14(2):375. https://doi.org/10.3390/cancers14020375
Chicago/Turabian StyleTsujimoto, Yoshie, Yuji Matsumoto, Midori Tanaka, Tatsuya Imabayashi, Keigo Uchimura, and Takaaki Tsuchida. 2022. "Diagnostic Value of Bronchoscopy for Peripheral Metastatic Lung Tumors" Cancers 14, no. 2: 375. https://doi.org/10.3390/cancers14020375
APA StyleTsujimoto, Y., Matsumoto, Y., Tanaka, M., Imabayashi, T., Uchimura, K., & Tsuchida, T. (2022). Diagnostic Value of Bronchoscopy for Peripheral Metastatic Lung Tumors. Cancers, 14(2), 375. https://doi.org/10.3390/cancers14020375






