Image-Guided Brachytherapy for Rectal Cancer: Reviewing the Past Two Decades of Clinical Investigation
Abstract
Simple Summary
Abstract
1. Introduction
2. Pre-Operative HDREBT
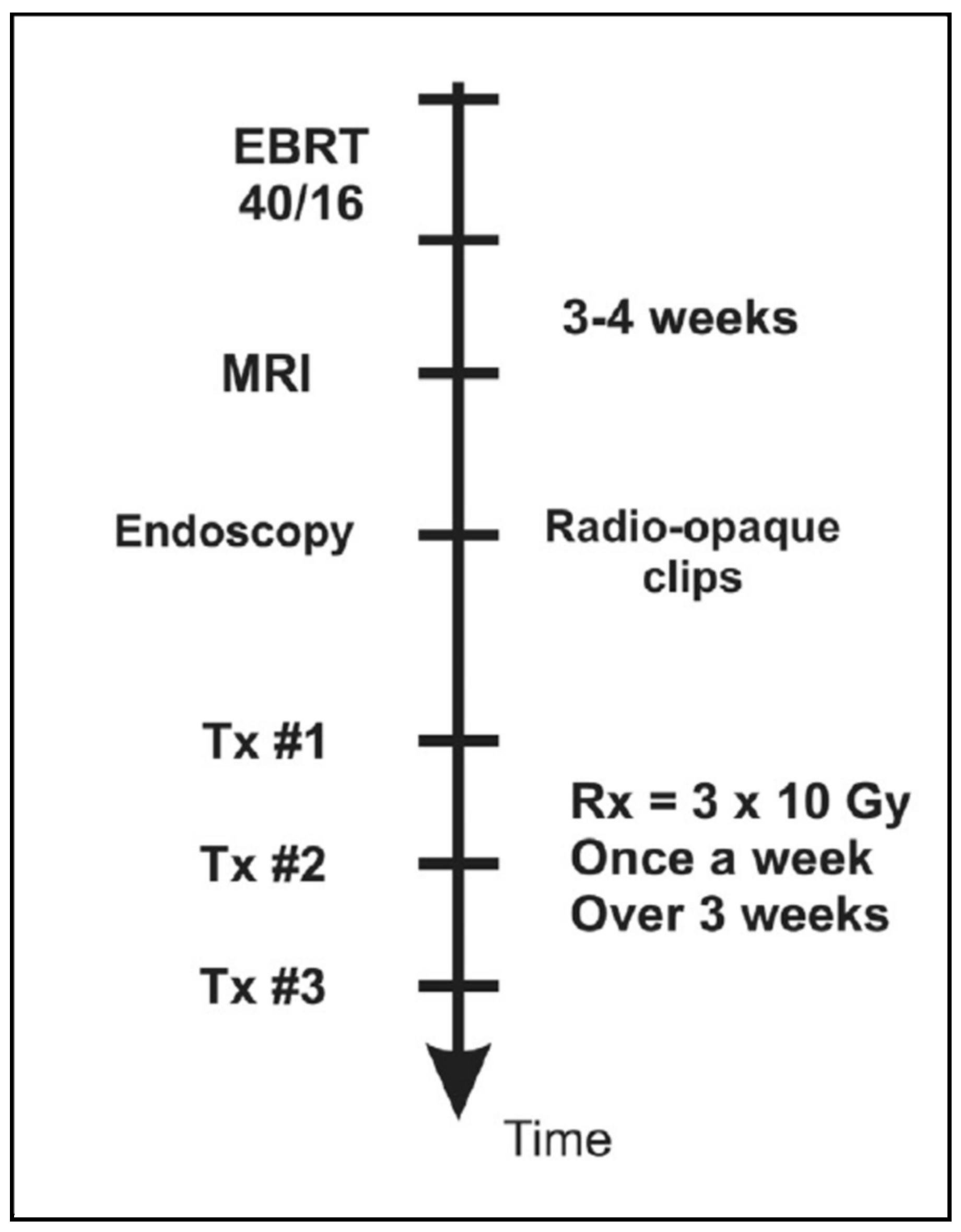
2.1. Clinical and Dosimetric Aspects
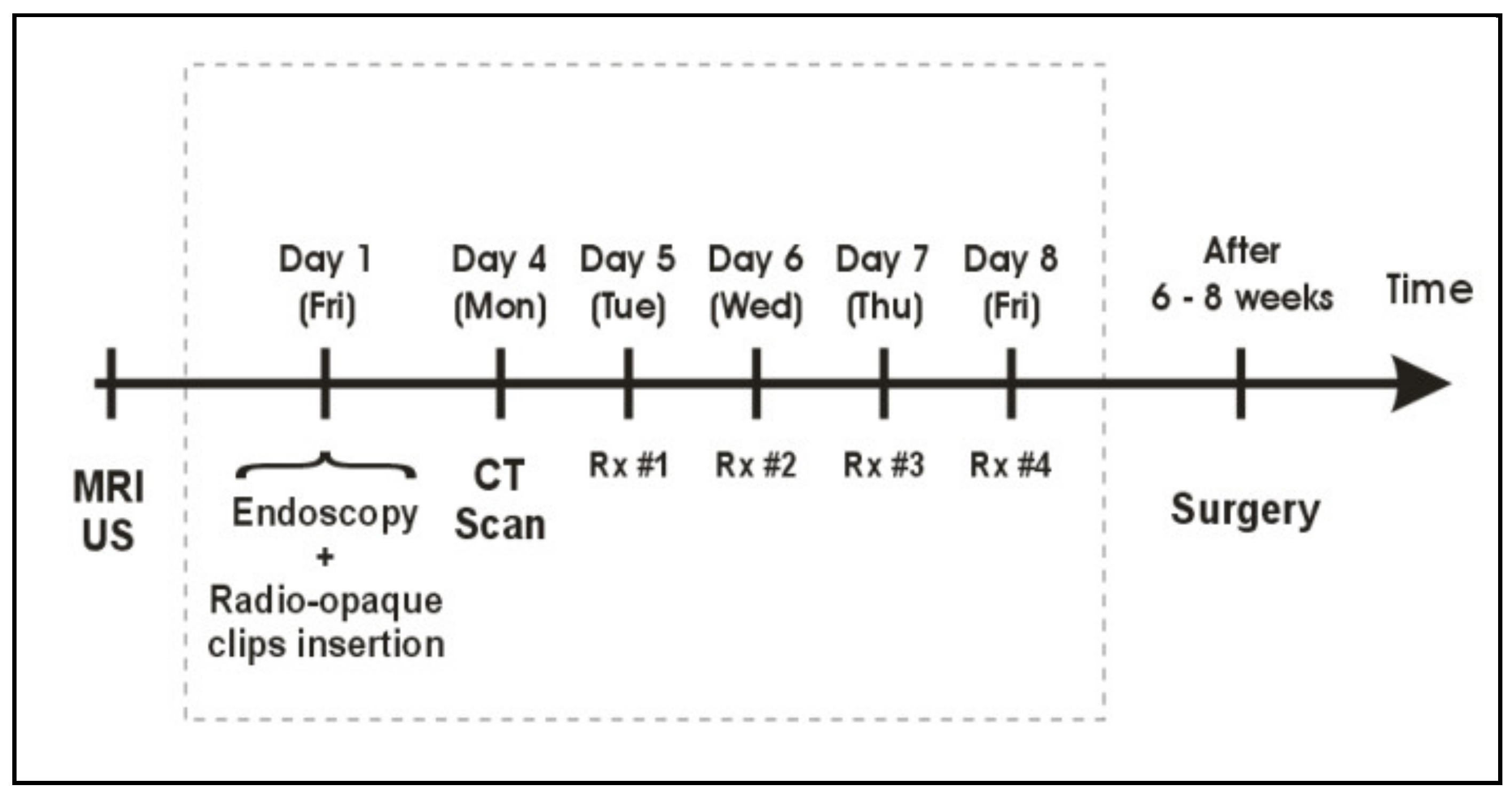
2.2. Locally Advanced, Stage III Rectal Cancer: The KIR Trial
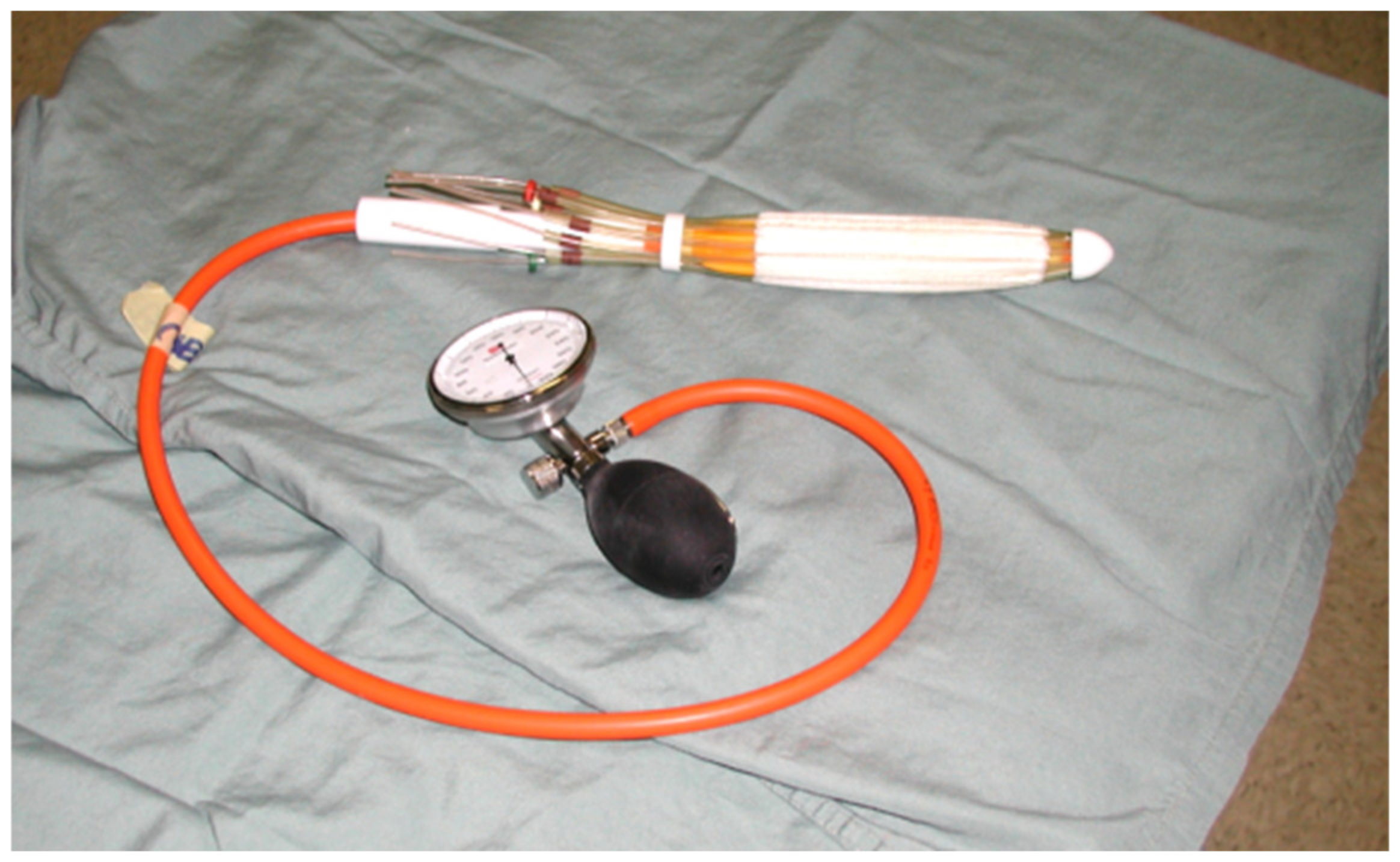
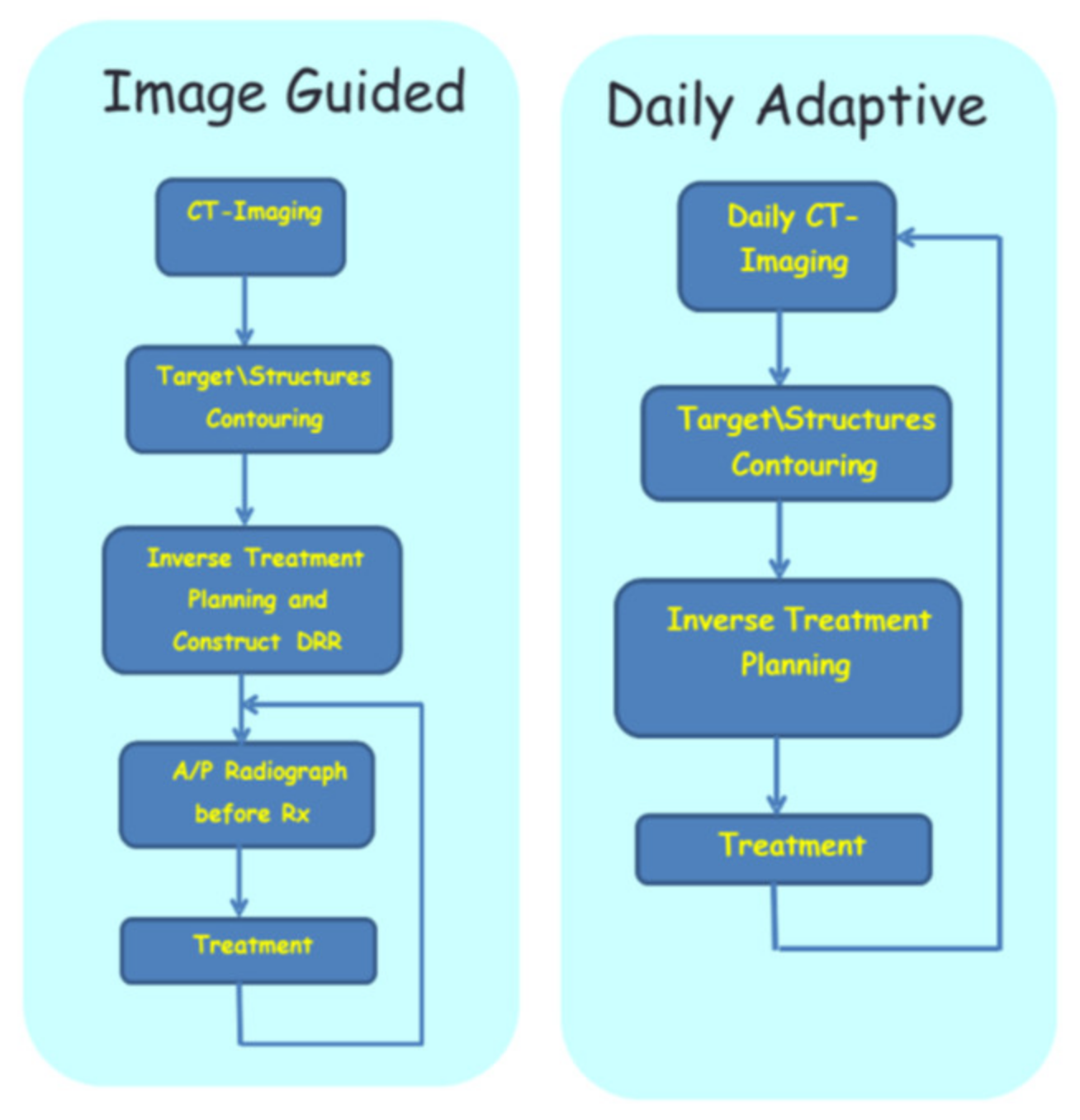
2.3. Technical Aspects
3. Boost HDREBT
3.1. Clinical Aspects
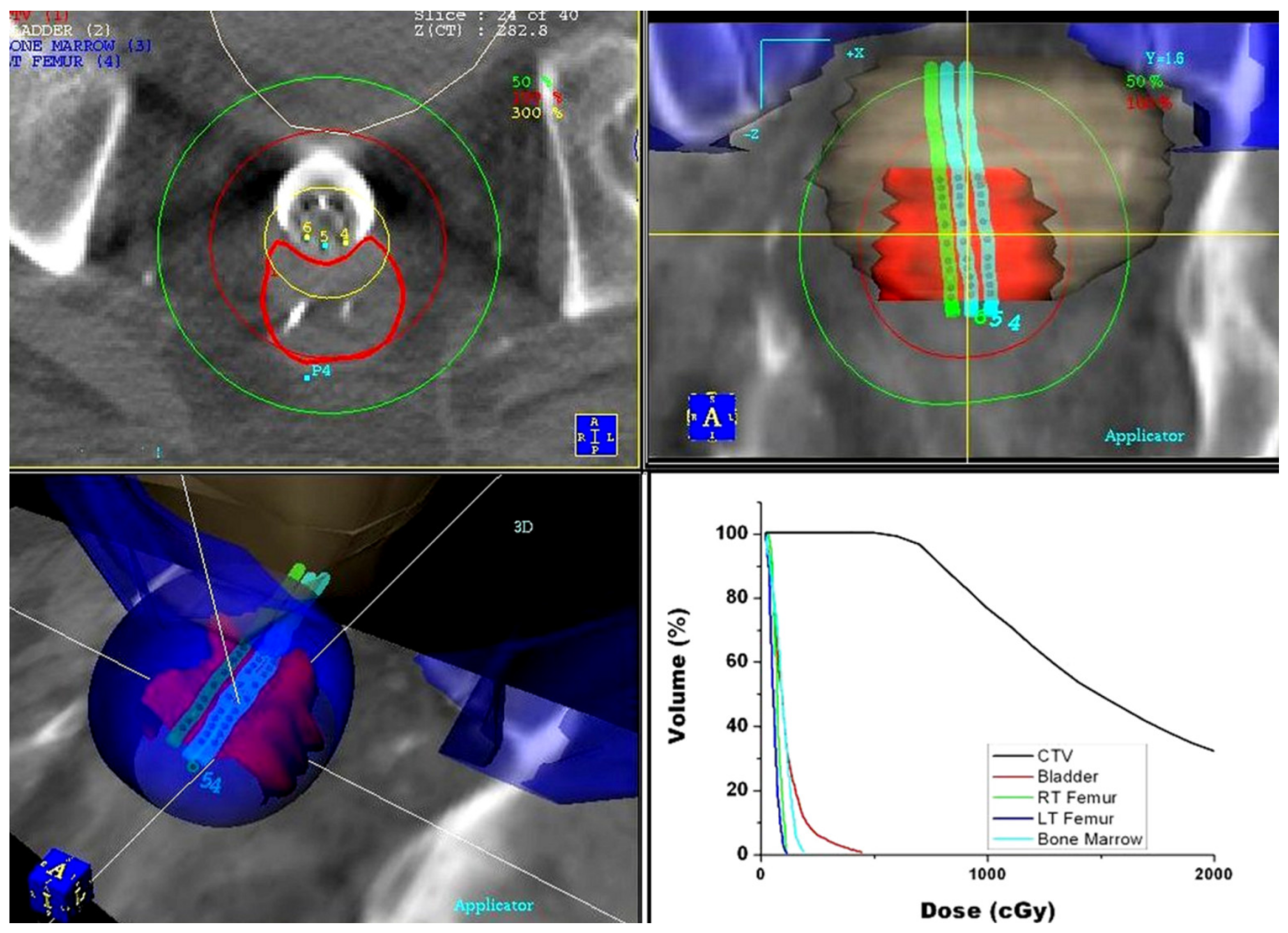
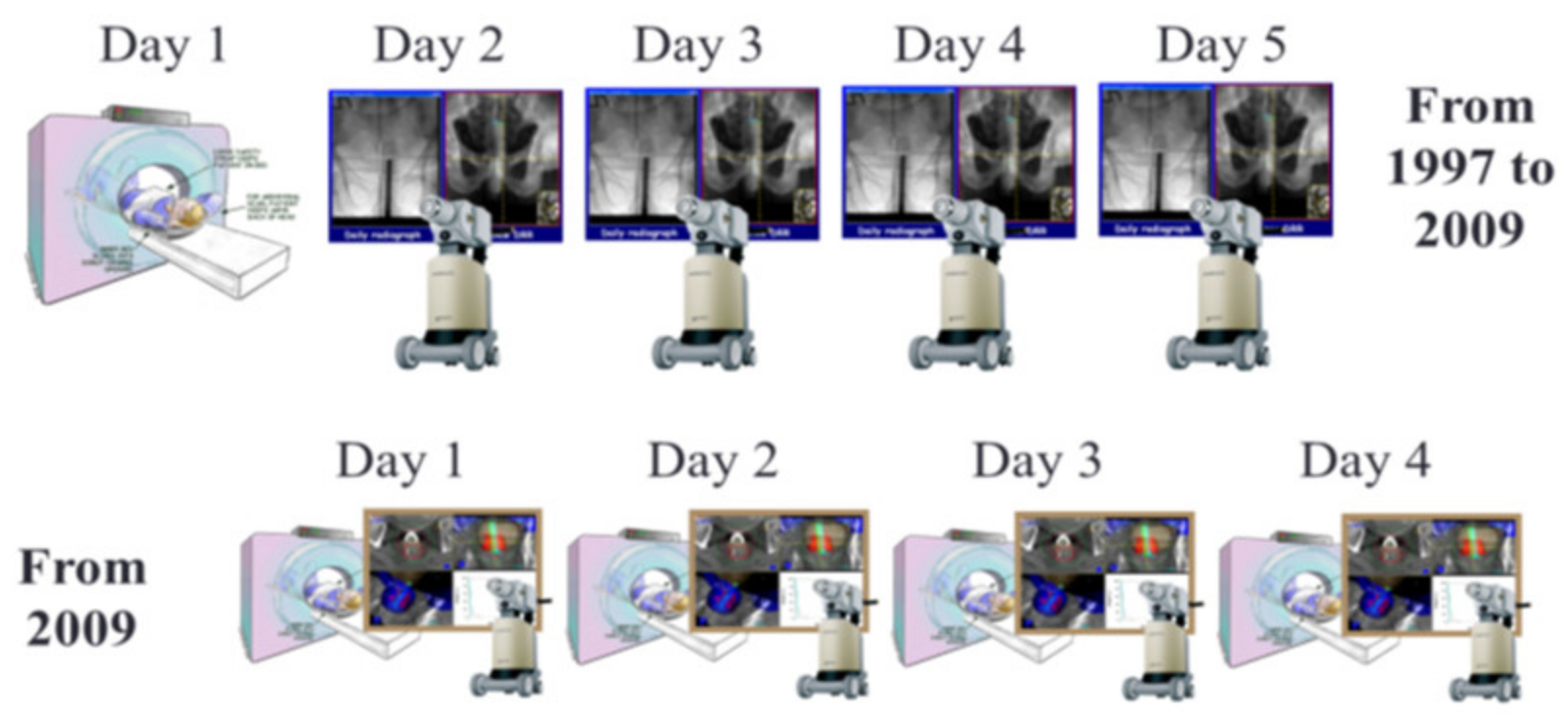
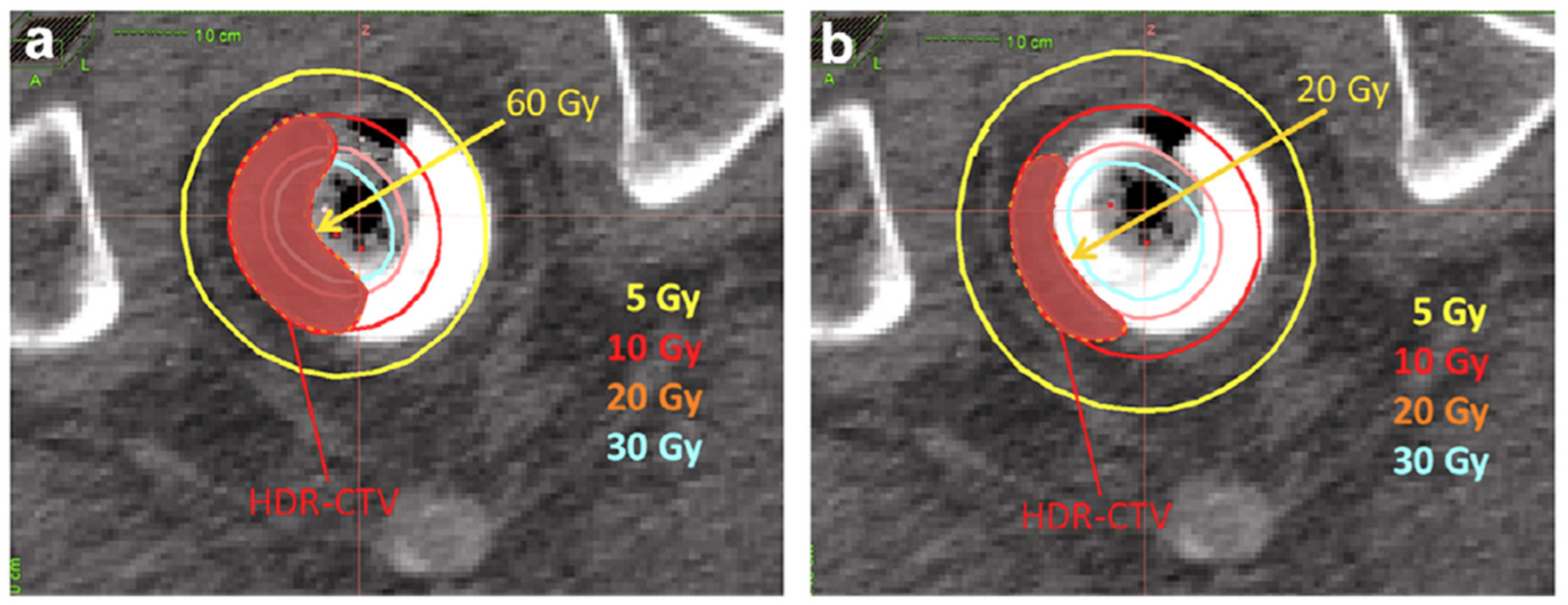
3.2. Technical Aspects
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Heald, R.J. The ‘Holy Plane’ of rectal surgery. J. R. Soc. Med. 1988, 81, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, J.K.; Ryall, R.D.; Heald, R.J. Mesorectal excision for rectal cancer. Lancet 1993, 341, 457–460. [Google Scholar] [CrossRef]
- Martling, A.L.; Holm, T.; Rutqvist, L.E.; Moran, B.J.; Heald, R.J.; Cedermark, B. Effects of a surgical training program on outcome of rectal cancer in the County of Stockholm. Lancet 2000, 356, 93–96. [Google Scholar] [CrossRef]
- Dahlberg, M.; Glimelius, B.; Pahlman, L. Changing strategy for rectal cancer associated with improved outcome. Br. J. Surg. 1999, 86, 379–384. [Google Scholar] [CrossRef]
- Kapiteijn, E.; Marijnen, C.A.; Nagtegaal, I.D.; Putter, H.; Steup, W.H.; Wiggers, T.; Van De Velde, C.J. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N. Engl. J. Med. 2001, 345, 638–646. [Google Scholar] [CrossRef]
- Cammà, C.; Giunta, M.; Fiorica, F.; Pagliaro, L.; Craxì, A.; Cottone, M. Preoperative Radiotherapy for Resectable Rectal Cancer. JAMA 2000, 284, 1008–1015. [Google Scholar] [CrossRef]
- Vuong, T.; Devic, S.; Moftah, B.; Evans, M.; Podgorsak, E.B. High dose rate endorectal brachytherapy in the treatment of locally advanced rectal carcinoma: Technical aspects. Brachytherapy 2005, 4, 230–235. [Google Scholar] [CrossRef]
- Vuong, T.; Belliveau, P.J.; Michel, R.P.; Moftah, B.A.; Parent, J.; Trudel, J.L.; Reinhold, C.; Souhami, L. Conformal Preoperative Endorectal Brachytherapy Treatment for Locally Advanced Rectal Cancer. Dis. Colon Rectum 2002, 45, 1486–1495. [Google Scholar] [CrossRef]
- Vuong, T.; Richard, C.; Niazi, T.; Liberman, S.; Letellier, F.; Morin, N.; Hu, K.; Anderson, D.; Devic, S. High Dose Rate Endorectal Brachytherapy for Patients With Curable Rectal Cancer. Semin. Colon Rectal Surg. 2010, 21, 115–119. [Google Scholar] [CrossRef]
- Breugom, A.J.; Vermeer, T.A.; van den Broek CB, M.; Vuong, T.; Bastiaannet, E.; Azoulay, L.; van de Velde, C.J.H. Effect of preoperative treatment strategies on the outcome of patients with clinical T3, non-metastasized rectal cancer: A comparison between Dutch and Canadian expert centers. Eur. J. Surg. Oncol. 2015, 41, 1039–1044. [Google Scholar] [CrossRef]
- Hesselager, C.; Vuong, T.; Påhlman, L.; Richard, C.; Liberman, S.; Letellier, F.; Folkesson, J. Short-term outcomes after neoadjuvant high dose rate endorectal brachytherapy versus or short-course external beam radiotherapy in resectable rectal cancer. Color. Dis. 2013, 15, 662–666. [Google Scholar] [CrossRef] [PubMed]
- Garant, A.; Kavan, P.; Martin, A.-G.; Azoulay, L.; Vendrely, V.; Lavoie, C.; Vasilevsky, C.-A.; Boutros, M.; Faria, J.; Nguyen, T.N.; et al. Optimizing treatment sequencing of chemotherapy for patients with rectal cancer: The KIR randomized phase II trial. Radiother. Oncol. 2020, 155, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Parker, W.; Patrocinio, H.; Vuong, T.; Roman, T.; Fallone, B.; Pla, C. 37 Virtual simulation for brachytherapy treatment planning. Radiother. Oncol. 1998, 47 (Supp. 1), S10. [Google Scholar] [CrossRef]
- Nath, R.; Anderson, L.; Luxton, G.; Weaver, K.A.; Williamson, J.F.; Meigooni, A.S. Dosimetry of interstitial brachytherapy sources: Recommendations of the AAPM Radiation Therapy Committee Task Group No. Med. Phys. 1995, 22, 209–234. [Google Scholar] [CrossRef]
- Devic, S.; Vuong, T.; Moftah, B.; Evans, M.; Podgorsak, E.B.; Poon, E.; Verhaegen, F. Image-guided high dose rate endorectal brachytherapy. Med. Phys. 2007, 34, 4451–4458. [Google Scholar] [CrossRef] [PubMed]
- Devic, S.; Bekerat, H.; Garant, A.; Vuong, T. Optimization of HDRBT boost dose delivery for patients with rectal cancer. Brachytherapy 2019, 18, 559–563. [Google Scholar] [CrossRef]
- Nout, R.A.; Devic, S.; Niazi, T.; Wyse, J.; Boutros, M.; Pelsser, V.; Vuong, T. CT-based adaptive high-dose-rate endorectal brachytherapy in the preoperative treatment of locally advanced rectal cancer, Technical and practical aspects. Brachytherapy 2016, 15, 477–484. [Google Scholar] [CrossRef]
- Garant, A.; Magnan, S.; Devic, S.; Martin, A.G.; Boutros, M.; Vasilevsky, C.A.; Vuong, T. Image guided adaptive endorectal brachytherapy in the non-operative management of patients with rectal cancer. Int. J. Radiat. Oncol. Biol. Phys. 2019, 105, 1005–1011. [Google Scholar] [CrossRef]
- Wang, Y.; Cummings, B.; Catton, P.; Dawson, L.; Kim, J.; Ringash, J.; Brierley, J. Primary radical external beam radiotherapy of rectal adenocarcinoma: Long term outcome of 271 patients. Radiother. Oncol. 2005, 77, 126–132. [Google Scholar] [CrossRef]
- Hall, M.D.; Schultheiss, T.E.; Smith, D.D.; Fakih, M.G.; Wong, J.Y.; Chen, Y.-J. Effect of increasing radiation dose on pathologic complete response in rectal cancer patients treated with neoadjuvant chemoradiation therapy. Acta Oncol. 2016, 55, 1392–1399. [Google Scholar]
- Sanghera, P.; Wong, D.; McConkey, C.; Geh, J.; Hartley, A. Chemoradiotherapy for Rectal Cancer: An Updated Analysis of Factors Affecting Pathological Response. Clin. Oncol. 2008, 20, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Appelt, A.L.; Pløen, J.; Vogelius, I.R.; Bentzen, S.M.; Jakobsen, A. Radiation Dose-Response Model for Locally Advanced Rectal Cancer After Preoperative Chemoradiation Therapy. Int. J. Radiat. Oncol. 2013, 85, 74–80. [Google Scholar] [CrossRef]
- Bentzen, S.M.; Constine, L.S.; Deasy, J.O.; Eisbruch, A.; Jackson, A.; Marks, L.B.; Yorke, E.D. Quantitative analyses of normal tissue effects in the clinic (QUANTEC): An introduction to the scientific issues. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, S3–S9. [Google Scholar] [CrossRef] [PubMed]
- Michalski, J.M.; Moughan, J.; Purdy, J.; Bosch, W.; Bruner, D.W.; Bahary, J.P.; Sandler, H. Effect of standard vs dose- escalated radiation therapy for patients with intermediate-risk prostate cancer: The NRG oncology RTOG 0126 randomized clinical trial. JAMA Oncol. 2018, 4, e180039. [Google Scholar] [CrossRef] [PubMed]
- Rijkmans, E.; van Triest, B.; Nout, R.; Kerkhof, E.; Buijsen, J.; Rozema, T.; Franssen, J.; Velema, L.; Laman, M.; Cats, A.; et al. Evaluation of clinical and endoscopic toxicity after external beam radiotherapy and endorectal brachytherapy in elderly patients with rectal cancer treated in the HERBERT study. Radiother. Oncol. 2018, 126, 417–423. [Google Scholar] [CrossRef]
- Appelt, A.L.; Pløen, J.; Harling, H.; Jensen, F.S.; Jensen, L.H.; Jørgensen, J.C.R.; Lindebjerg, J.; Rafaelsen, S.R.; Jakobsen, A. High-dose chemoradiotherapy and watchful waiting for distal rectal cancer: A prospective observational study. Lancet Oncol. 2015, 16, 919–927. [Google Scholar] [CrossRef]
- Garant, A.; Vasilevsky, C.; Boutros, M.; Khosrow-Khavar, F.; Kavan, P.; Diec, H.; Desgroseilliers, S.; Faria, J.; Ferland, E.; Pelsser, V.; et al. OC-0621 Interim Safety Analysis and Preliminary Results of the Morpheus Phase III Study. Radiother. Oncol. 2021, 161, S486–S487. [Google Scholar] [CrossRef]
- Myint, A.S.; Thamphya, B.; Gerard, J.P. Does non-TME surgery of rectal cancer compromise the chance of cure? Preliminary surgical salvage data from OPERA phase III randomized trial. J. Clin. Oncol. 2021, 39, 12. [Google Scholar] [CrossRef]
- Poon, E.; Reniers, B.; Devic, S.; Vuong, T.; Verhaegen, F. Dosimetric characterization of a novel intracavitary mould applicator for 192Ir high dose rate endorectal brachytherapy treatment. Med. Phys. 2006, 33, 4515–4526. [Google Scholar] [CrossRef]
- Garant, A.; Vasilevsky, C.A.; Boutros, M.; Khosrow-Khavar, F.; Kavan, P.; Diec, H.; Vuong, T. MORPHEUS Phase II-III Study: A Pre-Planned Interim Safety Analysis and Preliminary Results. Cancers 2022, 14, 3665. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vuong, T.; Garant, A.; Vendrely, V.; Nout, R.; Martin, A.-G.; Enger, S.A.; Podgorsak, E.; Moftah, B.; Devic, S. Image-Guided Brachytherapy for Rectal Cancer: Reviewing the Past Two Decades of Clinical Investigation. Cancers 2022, 14, 4846. https://doi.org/10.3390/cancers14194846
Vuong T, Garant A, Vendrely V, Nout R, Martin A-G, Enger SA, Podgorsak E, Moftah B, Devic S. Image-Guided Brachytherapy for Rectal Cancer: Reviewing the Past Two Decades of Clinical Investigation. Cancers. 2022; 14(19):4846. https://doi.org/10.3390/cancers14194846
Chicago/Turabian StyleVuong, Te, Aurelie Garant, Veronique Vendrely, Remi Nout, André-Guy Martin, Shirin A. Enger, Ervin Podgorsak, Belal Moftah, and Slobodan Devic. 2022. "Image-Guided Brachytherapy for Rectal Cancer: Reviewing the Past Two Decades of Clinical Investigation" Cancers 14, no. 19: 4846. https://doi.org/10.3390/cancers14194846
APA StyleVuong, T., Garant, A., Vendrely, V., Nout, R., Martin, A.-G., Enger, S. A., Podgorsak, E., Moftah, B., & Devic, S. (2022). Image-Guided Brachytherapy for Rectal Cancer: Reviewing the Past Two Decades of Clinical Investigation. Cancers, 14(19), 4846. https://doi.org/10.3390/cancers14194846






