Development of Lymphopenia during Therapy with Immune Checkpoint Inhibitors Is Associated with Poor Outcome in Metastatic Cutaneous Melanoma
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Patients’ Characteristics
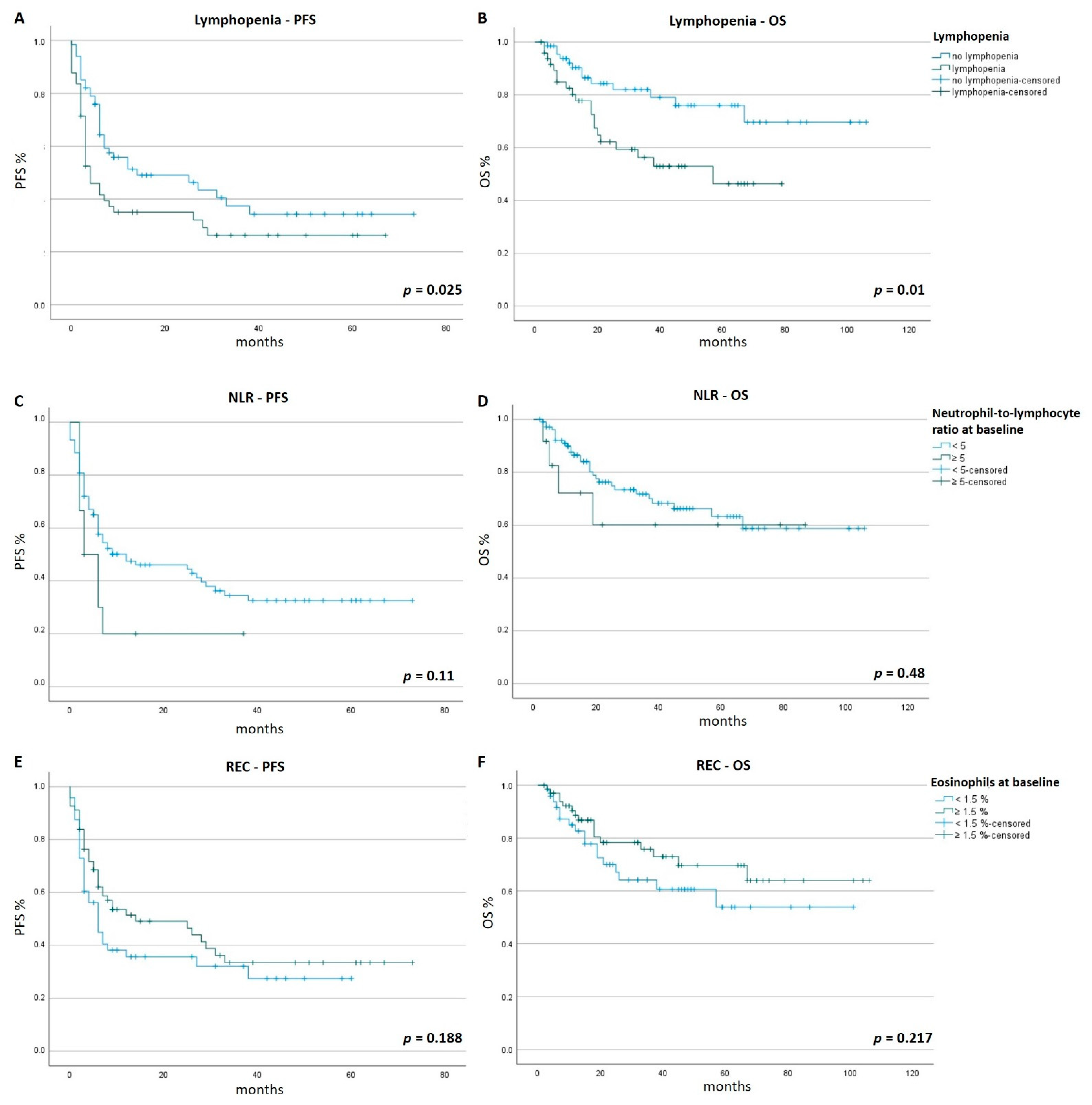
3.2. PFS and OS
3.3. Onset of Lymphopenia (Time to Lymphopenia)
3.4. Antibiotic Treatment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| NLC ≥ 5 (n = 12) | NLC < 5 (n = 104) | p-Values | |
|---|---|---|---|
| Age–y | |||
| Mean | 62.2 | 61.9 | 0.936 |
| Range | 44–85 | 22–90 | |
| Sex–no. (%) | |||
| Male | 9 (75.0) | 56 (53.8) | 0.224 |
| Female | 3 (25.0) | 48 (46.2) | 0.224 |
| ECOG performance status–no. (%) | |||
| 0 | 12 (100.0) | 97 (93.3) | 1.000 |
| 1 | 0 (0.0) | 7 (6.7) | 1.000 |
| M stage–no. (%) | |||
| M1a | 1 (8.3) | 16 (15.4) | 1.000 |
| M1b | 4 (33.3) | 22 (21.2) | 0.463 |
| M1c | 2 (16.7) | 31 (29.8) | 0.505 |
| M1d | 5 (41.7) | 35 (33.7) | 0.749 |
| Lactate dehydrogenase–no. (%) | |||
| ≤ULN | 7 (58.3) | 84 (80.8) | 0.129 |
| >ULN | 5 (41.7) | 16 (15.4) | 0.041 |
| ≥2 × ULN | 0 (0.0) | 4 (3.8) | 1.000 |
| BRAF status–no. (%) | |||
| Mutation | 4 (33.3) | 43 (41.3) | 0.759 |
| No mutation | 8 (66.7) | 61 (58.7) | 0.759 |
| NRAS status–no. (%) | |||
| Mutation | 3 (25.0) | 19 (18.3) | 0.696 |
| No mutation | 9 (75.0) | 85 (81.7) | 0.696 |
| Treatment–no. (%) | |||
| Ipilimumab | 1 (8.3) | 8 (7.7) | 1.000 |
| Nivolumab | 2 (16.7) | 20 (19.2) | 1.000 |
| Pembrolizumab | 4 (33.3) | 22 (21.2) | 0.463 |
| Ipilimumab + Nivolumab | 5 (41.7) | 54 (51.9) | 0.555 |
| REC < 1.5 % (n = 48) | REC ≥ 1.5 % (n = 68) | p-Values | |
|---|---|---|---|
| Age–y | |||
| Mean | 61.6 | 62.2 | 0.832 |
| Range | 22–85 | 33–90 | |
| Sex–no. (%) | |||
| Male | 32 (66.7) | 33 (48.5) | 0.060 |
| Female | 16 (33.4) | 35 (51.5) | 0.060 |
| ECOG performance status–no. (%) | |||
| 0 | 46 (95.8) | 63 (92.6) | 0.124 |
| 1 | 2 (4.2) | 5 (7.4) | 0.124 |
| M stage–no. (%) | |||
| M1a | 8 (16.7) | 9 (13.2) | 0.607 |
| M1b | 12 (25.0) | 14 (20.6) | 0.653 |
| M1c | 10 (20.8) | 23 (33.8) | 0.147 |
| M1d | 18 (37.5) | 22 (32.4) | 0.692 |
| Lactate dehydrogenase–no. (%) | |||
| ≤ULN | 39 (81.3) | 52 (76.5) | 0.649 |
| >ULN | 9 (18.8) | 12 (17.6) | 1.000 |
| ≥2 × ULN | 0 (0.0) | 4 (5.9) | 0.141 |
| BRAF status–no. (%) | |||
| Mutation | 18 (37.5) | 29 (42.6) | 0.701 |
| No mutation | 30 (62.5) | 39 (57.4) | 0.701 |
| NRAS status–no. (%) | |||
| Mutation | 10 (20.8) | 12 (17.6) | 0.811 |
| No mutation | 38 (79.2) | 56 (82.4) | 0.811 |
| Treatment–no. (%) | |||
| Ipilimumab | 6 (12.5) | 3 (4.4) | 0.159 |
| Nivolumab | 9 (18.8) | 13 (19.1) | 1.000 |
| Pembrolizumab | 8 (16.7) | 18 (26.5) | 0.262 |
| Ipilimumab + Nivolumab | 25 (52.1) | 34 (50.0) | 0.852 |
References
- Wolchok, J.D.; Chiarion-Sileni, V.; Gonzalez, R.; Rutkowski, P.; Grob, J.J.; Cowey, C.L.; Lao, C.D.; Wagstaff, J.; Schadendorf, D.; Ferrucci, P.F.; et al. Overall Survival with Combined Nivolumab and Ipilimumab in Advanced Melanoma. N. Engl. J. Med. 2017, 377, 1345–1356. [Google Scholar] [CrossRef] [PubMed]
- Gide, T.N.; Wilmott, J.S.; Scolyer, R.A.; Long, G.V. Primary and Acquired Resistance to Immune Checkpoint Inhibitors in Metastatic Melanoma. Clin. Cancer Res. 2018, 24, 1260–1270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Larkin, J.; Chiarion-Sileni, V.; Gonzalez, R.; Grob, J.J.; Cowey, C.L.; Lao, C.D.; Schadendorf, D.; Dummer, R.; Smylie, M.; Rutkowski, P.; et al. Combined Nivolumab and Ipilimumab or Monotherapy in Untreated Melanoma. N. Engl. J. Med. 2015, 373, 23–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fischer, G.M.; Carapeto, F.C.L.; Joon, A.Y.; Haydu, L.E.; Chen, H.; Wang, F.; Van Arnam, J.S.; McQuade, J.L.; Wani, K.; Kirkwood, J.M.; et al. Molecular and immunological associations of elevated serum lactate dehydrogenase in metastatic melanoma patients: A fresh look at an old biomarker. Cancer Med. 2020, 9, 8650–8661. [Google Scholar] [CrossRef]
- Thomas, N.E.; Edmiston, S.N.; Alexander, A.; Groben, P.A.; Parrish, E.; Kricker, A.; Armstrong, B.K.; Anton-Culver, H.; Gruber, S.B.; From, L.; et al. Association between NRAS and BRAF Mutational Status and Melanoma-Specific Survival among Patients with Higher-Risk Primary Melanoma. JAMA Oncol. 2015, 1, 359–368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gutzmer, R.; Vordermark, D.; Hassel, J.C.; Krex, D.; Wendl, C.; Schadendorf, D.; Sickmann, T.; Rieken, S.; Pukrop, T.; Höller, C.; et al. Melanoma brain metastases—Interdisciplinary management recommendations 2020. Cancer Treat. Rev. 2020, 89, 102083. [Google Scholar] [CrossRef] [PubMed]
- Weide, B.; Martens, A.; Hassel, J.C.; Berking, C.; Postow, M.A.; Bisschop, K.; Simeone, E.; Mangana, J.; Schilling, B.; Di Giacomo, A.M.; et al. Baseline Biomarkers for Outcome of Melanoma Patients Treated with Pembrolizumab. Clin. Cancer Res. 2016, 22, 5487–5496. [Google Scholar] [CrossRef] [Green Version]
- Diehl, A.; Yarchoan, M.; Hopkins, A.; Jaffee, E.; Grossman, S.A. Relationships between lymphocyte counts and treatment-related toxicities and clinical responses in patients with solid tumors treated with PD-1 checkpoint inhibitors. Oncotarget 2017, 8, 114268–114280. [Google Scholar] [CrossRef] [Green Version]
- Pinato, D.J.; Howlett, S.; Ottaviani, D.; Urus, H.; Patel, A.; Mineo, T.; Brock, C.; Power, D.; Hatcher, O.; Falconer, A.; et al. Association of Prior Antibiotic Treatment with Survival and Response to Immune Checkpoint Inhibitor Therapy in Patients with Cancer. JAMA Oncol. 2019, 5, 1774–1778. [Google Scholar] [CrossRef]
- Moreira, A.; Leisgang, W.; Schuler, G.; Heinzerling, L. Eosinophilic count as a biomarker for prognosis of melanoma patients and its importance in the response to immunotherapy. Immunotherapy 2017, 9, 115–121. [Google Scholar] [CrossRef]
- Karantanos, T.; Karanika, S.; Seth, B.; Gignac, G. The absolute lymphocyte count can predict the overall survival of patients with non-small cell lung cancer on nivolumab: A clinical study. Clin. Transl. Oncol. 2019, 21, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Ménétrier-Caux, C.; Ray-Coquard, I.; Blay, J.; Caux, C. Lymphopenia in Cancer Patients and its Effects on Response to Immunotherapy: An opportunity for combination with Cytokines? J. Immunother. Cancer 2019, 7, 85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robert, C.; Schachter, J.; Long, G.V.; Arance, A.; Grob, J.J.; Mortier, L.; Daud, A.; Carlino, M.S.; McNeil, C.; Lotem, M.; et al. Pembrolizumab versus Ipilimumab in Advanced Melanoma. N. Engl. J. Med. 2015, 372, 2521–2532. [Google Scholar] [CrossRef] [PubMed]
- Topalian, S.L.; Sznol, M.; McDermott, D.F.; Kluger, H.M.; Carvajal, R.D.; Sharfman, W.H.; Brahmer, J.R.; Lawrence, D.P.; Atkins, M.B.; Powderly, J.D.; et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J. Clin. Oncol. 2014, 32, 1020–1030. [Google Scholar] [CrossRef]
- Ott, P.A.; Bang, Y.J.; Piha-Paul, S.A.; Razak, A.R.A.; Bennouna, J.; Soria, J.C.; Rugo, H.S.; Cohen, R.B.; O’Neil, B.H.; Mehnert, J.M.; et al. T-Cell-Inflamed Gene-Expression Profile, Programmed Death Ligand 1 Expression, and Tumor Mutational Burden Predict Efficacy in Patients Treated with Pembrolizumab across 20 Cancers: KEYNOTE-028. J. Clin. Oncol. 2019, 37, 318–327. [Google Scholar] [CrossRef]
- Nigro, O.; Pinotti, G.; De Galitiis, F.; Di Pietro, F.R.; Giusti, R.; Filetti, M.; Bersanelli, M.; Lazzarin, A.; Bordi, P.; Catino, A.; et al. Late immune-related adverse events in long-term responders to PD-1/PD-L1 checkpoint inhibitors: A multicentre study. Eur. J. Cancer 2020, 134, 19–28. [Google Scholar] [CrossRef]
- Tang, S.Q.; Tang, L.L.; Mao, Y.P.; Li, W.F.; Chen, L.; Zhang, Y.; Guo, Y.; Liu, Q.; Sun, Y.; Xu, C.; et al. The Pattern of Time to Onset and Resolution of Immune-Related Adverse Events Caused by Immune Checkpoint Inhibitors in Cancer: A Pooled Analysis of 23 Clinical Trials and 8436 Patients. Cancer Res. Treat. 2021, 53, 339–354. [Google Scholar] [CrossRef]
- Warny, M.; Helby, J.; Nordestgaard, B.G.; Birgens, H.; Bojesen, S.E. Lymphopenia and risk of infection and infection-related death in 98,344 individuals from a prospective Danish population-based study. PLoS Med. 2018, 15, e1002685. [Google Scholar] [CrossRef] [Green Version]
- Kramer, R.; Zaremba, A.; Moreira, A.; Ugurel, S.; Johnson, D.B.; Hassel, J.C.; Salzmann, M.; Gesierich, A.; Weppler, A.; Spain, L.; et al. Hematological immune related adverse events after treatment with immune checkpoint inhibitors. Eur. J. Cancer 2021, 147, 170–181. [Google Scholar] [CrossRef]
- Ménard, C.; Ghiringhelli, F.; Roux, S.; Chaput, N.; Mateus, C.; Grohmann, U.; Caillat-Zucman, S.; Zitvogel, L.; Robert, C. Ctla-4 blockade confers lymphocyte resistance to regulatory T-cells in advanced melanoma: Surrogate marker of efficacy of tremelimumab? Clin. Cancer Res. 2008, 14, 5242–5249. [Google Scholar] [CrossRef] [Green Version]
- Zhu, J.; Powis de Tenbossche, C.G.; Cané, S.; Colau, D.; van Baren, N.; Lurquin, C.; Schmitt-Verhulst, A.M.; Liljeström, P.; Uyttenhove, C.; Van den Eynde, B.J. Resistance to cancer immunotherapy mediated by apoptosis of tumor-infiltrating lymphocytes. Nat. Commun. 2017, 8, 1404. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thommen, D.S.; Schumacher, T.N. T Cell Dysfunction in Cancer. Cancer Cell 2018, 33, 547–562. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gros, A.; Parkhurst, M.R.; Tran, E.; Pasetto, A.; Robbins, P.F.; Ilyas, S.; Prickett, T.D.; Gartner, J.J.; Crystal, J.S.; Roberts, I.M.; et al. Prospective identification of neoantigen-specific lymphocytes in the peripheral blood of melanoma patients. Nat. Med. 2016, 22, 433–438. [Google Scholar] [CrossRef] [PubMed]
| Lymphopenia (n = 49) | No Lymphopenia (n = 67) | p-Values | |
|---|---|---|---|
| Age–y | |||
| Mean | 60.6 | 62.9 | 0.350 |
| Range | 37–81 | 22–90 | |
| Sex–no. (%) | |||
| Male | 34 (69.4) | 31 (46.3) | 0.015 |
| Female | 15 (30.6) | 36 (53.7) | 0.015 |
| ECOG performance status–no. (%) | |||
| 0 | 47 (95.9) | 62 (92.5) | 0.697 |
| 1 | 2 (4.1) | 5 (7.5) | 0.697 |
| M stage–no. (%) | |||
| M1a | 7 (14.3) | 10 (14.9) | 1.000 |
| M1b | 7 (14.3) | 19 (28.4) | 0.114 |
| M1c | 14 (28.6) | 19 (28.4) | 1.000 |
| M1d | 21 (42.9) | 19 (28.4) | 0.117 |
| Lactate dehydrogenase–no. (%) | |||
| ≤ULN | 37 (75.5) | 54 (80.6) | 0.648 |
| >ULN | 10 (20.4) | 11 (16.4) | 0.630 |
| ≥2 × ULN | 2 (4.1) | 2 (3.0) | 1.000 |
| BRAF status–no. (%) | |||
| Mutation | 25 (51.0) | 22 (32.8) | 0.057 |
| No mutation | 24 (49.0) | 45 (67.2) | 0.057 |
| NRAS status–no. (%) | |||
| Mutation | 10 (20.4) | 12 (17.9) | 0.812 |
| No mutation | 39 (79.6) | 55 (82.1) | 0.812 |
| Treatment–no. (%) | |||
| Ipilimumab | 5 (10.2) | 4 (6.0) | 0.490 |
| Nivolumab | 7 (14.3) | 15 (22.4) | 0.341 |
| Pembrolizumab | 6 (12.2) | 20 (29.9) | 0.026 |
| Ipilimumab + Nivolumab | 31 (63.3) | 28 (41.8) | 0.025 |
| Early (0–4 Months) n = 16 | Delayed (4–12 Months) n = 15 | Late (>12 Months) n = 15 | p-Values | |
|---|---|---|---|---|
| PFS (months) | 11.4 (range 0–50) | 17.1 (range 0–67) | 11.9 (range 0–44) | 0.727 |
| OS (months) | 24.4 (range 2–62) | 29.8 (range 3–70) | 30.1 (range 3–66) | 0.878 |
| No Antibiotic Treatment | Antibiotic Treatment | p-Values | |||
|---|---|---|---|---|---|
| No Lymphopenia | n = 59 | PFS 17.2 months (range 1–73) | n = 8 | PFS 14.8 months (range 2–54) | 0.322 |
| OS 37.1 months (range 5–106) | OS 34.3 months (range 4–104) | 0.512 | |||
| Lymphopenia | n = 37 | PFS 14.8 months (range 0–67) | n = 12 | PFS 8.6 months (range 0–50) | 0.976 |
| OS 26.6 months (range 2–70) | OS 33.2 months (range 3–79) | 0.730 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomsitz, D.; Schlaak, M.; Zierold, S.; Pesch, G.; Schulz, T.U.; Müller, G.; Zecha, C.; French, L.E.; Heinzerling, L. Development of Lymphopenia during Therapy with Immune Checkpoint Inhibitors Is Associated with Poor Outcome in Metastatic Cutaneous Melanoma. Cancers 2022, 14, 3282. https://doi.org/10.3390/cancers14133282
Tomsitz D, Schlaak M, Zierold S, Pesch G, Schulz TU, Müller G, Zecha C, French LE, Heinzerling L. Development of Lymphopenia during Therapy with Immune Checkpoint Inhibitors Is Associated with Poor Outcome in Metastatic Cutaneous Melanoma. Cancers. 2022; 14(13):3282. https://doi.org/10.3390/cancers14133282
Chicago/Turabian StyleTomsitz, Dirk, Max Schlaak, Sarah Zierold, Giulia Pesch, Thomas U. Schulz, Genoveva Müller, Christine Zecha, Lars E. French, and Lucie Heinzerling. 2022. "Development of Lymphopenia during Therapy with Immune Checkpoint Inhibitors Is Associated with Poor Outcome in Metastatic Cutaneous Melanoma" Cancers 14, no. 13: 3282. https://doi.org/10.3390/cancers14133282
APA StyleTomsitz, D., Schlaak, M., Zierold, S., Pesch, G., Schulz, T. U., Müller, G., Zecha, C., French, L. E., & Heinzerling, L. (2022). Development of Lymphopenia during Therapy with Immune Checkpoint Inhibitors Is Associated with Poor Outcome in Metastatic Cutaneous Melanoma. Cancers, 14(13), 3282. https://doi.org/10.3390/cancers14133282






