Metabolic Diseases and Risk of Head and Neck Cancer: A Cohort Study Analyzing Nationwide Population-Based Data
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
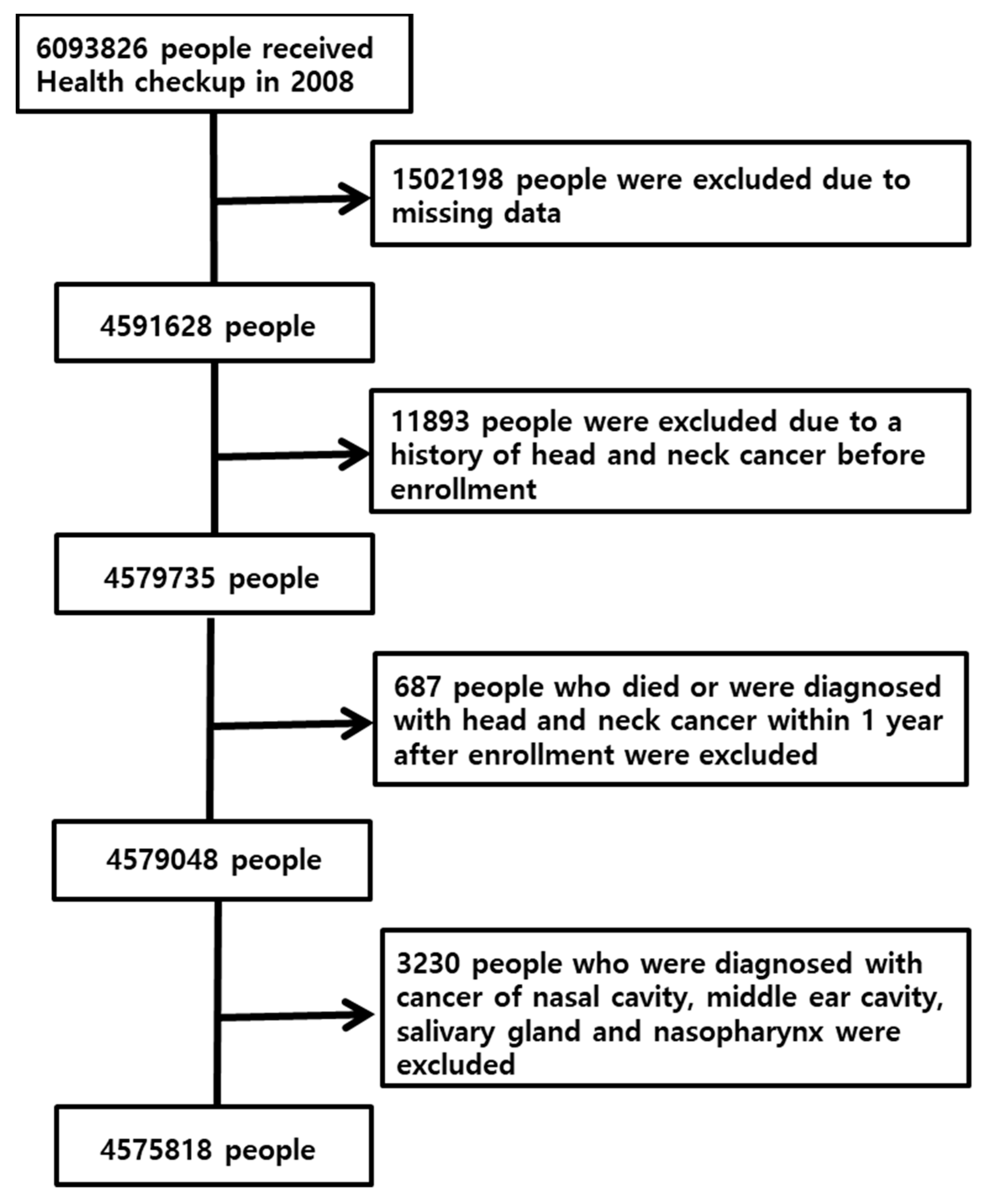
2.1. Study Population
2.2. Definition of Variables
2.3. Statistical Analysis
3. Results
3.1. Baseline Study Participant Characteristics
3.2. Impact of Each Factor on the Risk of HNC
3.3. Analysis of the HNC Risk by Sex According to Each Factor
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fitzmaurice, C.; Abate, D.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdel-Rahman, O.; Abdelalim, A.; Abdoli, A.; Abdollahpour, I.; Abdulle, A.S. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2017: A systematic analysis for the global burden of disease study. JAMA Oncol. 2019, 5, 1749–1768. [Google Scholar] [PubMed] [Green Version]
- Marur, S.; Forastiere, A.A. Head and neck cancer: Changing epidemiology, diagnosis, and treatment. In Mayo Clinic Proceedings; Elsevier: Amsterdam, The Netherlands, 2008; pp. 489–501. [Google Scholar]
- Rettig, E.M.; D’Souza, G. Epidemiology of head and neck cancer. Surg. Oncol. Clin. 2015, 24, 379–396. [Google Scholar] [CrossRef] [PubMed]
- Bellastella, G.; Scappaticcio, L.; Esposito, K.; Giugliano, D.; Maiorino, M.I. Metabolic syndrome and cancer:“The common soil hypothesis”. Diabetes Res. Clin. Pract. 2018, 143, 389–397. [Google Scholar] [CrossRef]
- Grundy, S.M.; Cleeman, J.I.; Daniels, S.R.; Donato, K.A.; Eckel, R.H.; Franklin, B.A.; Gordon, D.J.; Krauss, R.M.; Savage, P.J.; Smith Jr, S.C. Diagnosis and management of the metabolic syndrome: An American Heart Association/National Heart, Lung, and Blood Institute scientific statement. Circulation 2005, 112, 2735–2752. [Google Scholar] [CrossRef] [Green Version]
- Alberti, K.G.M.M.; Zimmet, P.Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: Diagnosis and classification of diabetes mellitus. Provisional report of a WHO consultation. Diabet. Med. 1998, 15, 539–553. [Google Scholar] [CrossRef]
- Balkau, B.; Charles, M.-A. Comment on the provisional report from the WHO consultation. European Group for the Study of Insulin Resistance (EGIR). Diabet. Med. A J. Br. Diabet. Assoc. 1999, 16, 442–443. [Google Scholar]
- Klose, G.; Beil, F.U.; Dieplinger, H.; von Eckardstein, A.; Föger, B.; Gouni-Berthold, I.; Heigl, F.; Koenig, W.; Kostner, G.M.; Landmesser, U. New AHA and ACC guidelines on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk. Wien. Klin. Wochenschr. 2014, 126, 169–175. [Google Scholar] [CrossRef]
- Einhorn, D. American College of Endocrinology position statement on the insulin resistance syndrome. Endocr. Pract. 2003, 9, 5–21. [Google Scholar] [CrossRef]
- Giovannucci, E.; Harlan, D.M.; Archer, M.C.; Bergenstal, R.M.; Gapstur, S.M.; Habel, L.A.; Pollak, M.; Regensteiner, J.G.; Yee, D. Diabetes and cancer: A consensus report. CA A Cancer J. Clin. 2010, 60, 207–221. [Google Scholar] [CrossRef] [Green Version]
- Han, H.; Guo, W.; Shi, W.; Yu, Y.; Zhang, Y.; Ye, X.; He, J. Hypertension and breast cancer risk: A systematic review and meta-analysis. Sci. Rep. 2017, 7, 44877. [Google Scholar] [CrossRef]
- Stocks, T.; Van Hemelrijck, M.; Manjer, J.; Bjørge, T.; Ulmer, H.; Hallmans, G.; Lindkvist, B.; Selmer, R.; Nagel, G.; Tretli, S. Blood pressure and risk of cancer incidence and mortality in the Metabolic Syndrome and Cancer Project. Hypertension 2012, 59, 802–810. [Google Scholar] [CrossRef] [PubMed]
- Calle, E.E.; Kaaks, R. Overweight, obesity and cancer: Epidemiological evidence and proposed mechanisms. Nat. Rev. Cancer 2004, 4, 579–591. [Google Scholar] [CrossRef] [PubMed]
- Hwang, K.-T.; Han, K.-D.; Oh, S.; Koo, B.K.; Lee, S.K.; Kim, J.; Seo, H.J.; Jung, J.; Kim, B.H.; Hur, H. Influence of Metabolic Syndrome on Risk of Breast Cancer: A Study Analyzing Nationwide Data from Korean National Health Insurance Service. Cancer Epidemiol. Prev. Biomark. 2020, 29, 2038–2047. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Lu, L.; Liu, L.; Wei, S.; He, Y.; Chang, J.; Lian, X. Blood lipids profile and lung cancer risk in a meta-analysis of prospective cohort studies. J. Clin. Lipidol. 2017, 11, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Meccariello, G.; Maniaci, A.; Bianchi, G.; Cammaroto, G.; Iannella, G.; Catalano, A.; Sgarzani, R.; De Vito, A.; Capaccio, P.; Pelucchi, S.; et al. Neck dissection and trans oral robotic surgery for oropharyngeal squamous cell carcinoma. Auris Nasus Larynx 2022, 49, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Hicks, D.F.; Bakst, R.; Doucette, J.; Kann, B.H.; Miles, B.; Genden, E.; Misiukiewicz, K.; Posner, M.; Gupta, V. Impact of obesity on outcomes for patients with head and neck cancer. Oral Oncol. 2018, 83, 11–17. [Google Scholar] [CrossRef]
- Lee, J.; Lee, J.S.; Park, S.-H.; Shin, S.A.; Kim, K. Cohort profile: The national health insurance service–national sample cohort (NHIS-NSC), South Korea. Int. J. Epidemiol. 2017, 46, e15. [Google Scholar] [CrossRef]
- Alberti, K.G.M.M.; Zimmet, P.; Shaw, J. Metabolic syndrome—a new world-wide definition. A consensus statement from the international diabetes federation. Diabet. Med. 2006, 23, 469–480. [Google Scholar] [CrossRef]
- World Health Organization. The Asia-Pacific Perspective: Redefining Obesity and Its Treatment; World Health Organization: Geneva, Switzerland, 2000.
- Cleeman, J.; Grundy, S.; Becker, D.; Clark, L. Expert panel on detection, evaluation and treatment of high blood cholesterol in adults. Executive summary of the third report of the National Cholesterol Education Program (NCEP) Adult Treatment Panel (ATP III). JAMA 2001, 285, 2486–2497. [Google Scholar]
- Flegal, K.M.; Carroll, M.D.; Ogden, C.L.; Johnson, C.L. Prevalence and trends in obesity among US adults, 1999–2000. JAMA 2002, 288, 1723–1727. [Google Scholar] [CrossRef]
- Gu, D.; Reynolds, K.; Wu, X.; Chen, J.; Duan, X.; Reynolds, R.F.; Whelton, P.K.; He, J.; Group, I.C. Prevalence of the metabolic syndrome and overweight among adults in China. Lancet 2005, 365, 1398–1405. [Google Scholar] [CrossRef]
- Esposito, K.; Chiodini, P.; Colao, A.; Lenzi, A.; Giugliano, D. Metabolic syndrome and risk of cancer: A systematic review and meta-analysis. Diabetes Care 2012, 35, 2402–2411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiu, H.M.; Lin, J.T.; Shun, C.T.; Liang, J.T.; Lee, Y.C.; Huang, S.P.; Wu, M.S. Association of metabolic syndrome with proximal and synchronous colorectal neoplasm. Clin. Gastroenterol. Hepatol. 2007, 5, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Laukkanen, J.A.; Laaksonen, D.E.; Niskanen, L.; Pukkala, E.; Hakkarainen, A.; Salonen, J.T. Metabolic syndrome and the risk of prostate cancer in Finnish men: A population-based study. Cancer Epidemiol. Prev. Biomark. 2004, 13, 1646–1650. [Google Scholar] [CrossRef]
- Lund Håheim, L.; Wisløff, T.; Holme, I.; Nafstad, P. Metabolic syndrome predicts prostate cancer in a cohort of middle-aged Norwegian men followed for 27 years. Am. J. Epidemiol. 2006, 164, 769–774. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pasanisi, P.; Berrino, F.; De Petris, M.; Venturelli, E.; Mastroianni, A.; Panico, S. Metabolic syndrome as a prognostic factor for breast cancer recurrences. Int. J. Cancer 2006, 119, 236–238. [Google Scholar] [CrossRef]
- Jiang, H.; Zhou, L.; He, Q.; Jiang, K.; Yuan, J.; Huang, X. The effect of metabolic syndrome on head and neck cancer incidence risk: A population-based prospective cohort study. Cancer Metab. 2021, 9, 25. [Google Scholar] [CrossRef]
- Pelton, K.; Freeman, M.R.; Solomon, K.R. Cholesterol and prostate cancer. Curr. Opin. Pharmacol. 2012, 12, 751–759. [Google Scholar] [CrossRef] [Green Version]
- Farwell, W.R.; Scranton, R.E.; Lawler, E.V.; Lew, R.A.; Brophy, M.T.; Fiore, L.D.; Gaziano, J.M. The association between statins and cancer incidence in a veterans population. J. Natl. Cancer Inst. 2008, 100, 134–139. [Google Scholar] [CrossRef] [Green Version]
- Pommier, A.; Alves, G.; Viennois, E.; Bernard, S.; Communal, Y.; Sion, B.; Marceau, G.; Damon, C.; Mouzat, K.; Caira, F. Liver X Receptor activation downregulates AKT survival signaling in lipid rafts and induces apoptosis of prostate cancer cells. Oncogene 2010, 29, 2712–2723. [Google Scholar] [CrossRef] [Green Version]
- Nofer, J.-R.; Junker, R.; Pulawski, E.; Fobker, M.; Levkau, B.; von Eckardstein, A.; Seedorf, U.; Assmann, G.; Walter, M. High density lipoproteins induce cell cycle entry in vascular smooth muscle cells via mitogen activated protein kinase-dependent pathway. Thromb. Haemost. 2001, 85, 730–735. [Google Scholar]
- Nofer, J.-R.; Levkau, B.; Wolinska, I.; Junker, R.; Fobker, M.; von Eckardstein, A.; Seedorf, U.; Assmann, G. Suppression of endothelial cell apoptosis by high density lipoproteins (HDL) and HDL-associated lysosphingolipids. J. Biol. Chem. 2001, 276, 34480–34485. [Google Scholar] [CrossRef] [Green Version]
- Von Eckardstein, A.; Hersberger, M.; Rohrer, L. Current understanding of the metabolism and biological actions of HDL. Curr. Opin. Clin. Nutr. Metab. Care 2005, 8, 147–152. [Google Scholar] [CrossRef]
- Gaudet, M.M.; Patel, A.V.; Sun, J.; Hildebrand, J.S.; McCullough, M.L.; Chen, A.Y.; Gapstur, S.M. Prospective studies of body mass index with head and neck cancer incidence and mortality. Cancer Epidemiol. Prev. Biomark. 2012, 21, 497–503. [Google Scholar] [CrossRef] [Green Version]
- Mendonça, F.M.; de Sousa, F.R.; Barbosa, A.L.; Martins, S.C.; Araújo, R.L.; Soares, R.; Abreu, C. Metabolic syndrome and risk of cancer: Which link? Metabolism 2015, 64, 182–189. [Google Scholar] [CrossRef]
- Hashibe, M.; Hunt, J.; Wei, M.; Buys, S.; Gren, L.; Lee, Y.C.A. Tobacco, alcohol, body mass index, physical activity, and the risk of head and neck cancer in the prostate, lung, colorectal, and ovarian (PLCO) cohort. Head Neck 2013, 35, 914–922. [Google Scholar] [CrossRef]
- Chen, Y.; Lee, Y.-C.A.; Li, S.; Li, Q.; Chen, C.-J.; Hsu, W.-L.; Lou, P.-J.; Zhu, C.; Pan, J.; Shen, H. Body mass index and the risk of head and neck cancer in the Chinese population. Cancer Epidemiol. 2019, 60, 208–215. [Google Scholar] [CrossRef]
- Etemadi, A.; O’Doherty, M.G.; Freedman, N.D.; Hollenbeck, A.R.; Dawsey, S.M.; Abnet, C.C. A prospective cohort study of body size and risk of head and neck cancers in the NIH–AARP Diet and Health Study. Cancer Epidemiol. Prev. Biomark. 2014, 23, 2422–2429. [Google Scholar] [CrossRef] [Green Version]
- Wang, K.; Yu, X.-h.; Tang, Y.-J.; Tang, Y.-l.; Liang, X.-h. Obesity: An emerging driver of head and neck cancer. Life Sci. 2019, 233, 116687. [Google Scholar] [CrossRef]
- Gaudet, M.M.; Kitahara, C.M.; Newton, C.C.; Bernstein, L.; Reynolds, P.; Weiderpass, E.; Kreimer, A.R.; Yang, G.; Adami, H.-O.; Alavanja, M.C. Anthropometry and head and neck cancer: A pooled analysis of cohort data. Int. J. Epidemiol. 2015, 44, 673–681. [Google Scholar] [CrossRef] [Green Version]
- Etemadi, A.; Islami, F.; Phillips, D.H.; Godschalk, R.; Golozar, A.; Kamangar, F.; Malekshah, A.F.T.; Pourshams, A.; Elahi, S.; Ghojaghi, F. Variation in PAH-related DNA adduct levels among non-smokers: The role of multiple genetic polymorphisms and nucleotide excision repair phenotype. Int. J. Cancer 2013, 132, 2738–2747. [Google Scholar] [CrossRef]
- Mizoue, T.; Kasai, H.; Kubo, T.; Tokunaga, S. Leanness, smoking, and enhanced oxidative DNA damage. Cancer Epidemiol. Prev. Biomark. 2006, 15, 582–585. [Google Scholar] [CrossRef] [Green Version]
- Akbartabartoori, M.; Lean, M.; Hankey, C. Relationships between cigarette smoking, body size and body shape. Int. J. Obes. 2005, 29, 236–243. [Google Scholar] [CrossRef] [Green Version]
- Canoy, D.; Wareham, N.; Luben, R.; Welch, A.; Bingham, S.; Day, N.; Khaw, K.T. Cigarette smoking and fat distribution in 21, 828 British men and women: A population-based study. Obes. Res. 2005, 13, 1466–1475. [Google Scholar] [CrossRef]
- Gaudet, M.M.; Olshan, A.F.; Chuang, S.-C.; Berthiller, J.; Zhang, Z.-F.; Lissowska, J.; Zaridze, D.; Winn, D.M.; Wei, Q.; Talamini, R. Body mass index and risk of head and neck cancer in a pooled analysis of case–control studies in the International Head and Neck Cancer Epidemiology (INHANCE) Consortium. Int. J. Epidemiol. 2010, 39, 1091–1102. [Google Scholar] [CrossRef]
- Tseng, K.-S.; Lin, C.; Lin, Y.-S.; Weng, S.-F. Risk of head and neck cancer in patients with diabetes mellitus: A retrospective cohort study in Taiwan. JAMA Otolaryngol. Head Neck Surg. 2014, 140, 746–753. [Google Scholar] [CrossRef] [Green Version]
- Stott-Miller, M.; Chen, C.; Schwartz, S.M. Type II diabetes and metabolic syndrome in relation to head and neck squamous cell carcinoma risk: A SEER-Medicare database study. Cancer Epidemiol. 2013, 37, 428–433. [Google Scholar] [CrossRef]
- Colt, J.S.; Schwartz, K.; Graubard, B.I.; Davis, F.; Ruterbusch, J.; DiGaetano, R.; Purdue, M.; Rothman, N.; Wacholder, S.; Chow, W.-H. Hypertension and risk of renal cell carcinoma among white and black Americans. Epidemiol. (Camb. Mass.) 2011, 22, 797. [Google Scholar] [CrossRef] [Green Version]
- Lindgren, A.; Nissinen, A.M.; Tuomilehto, J.; Pukkala, E. Cancer pattern among hypertensive patients in North Karelia, Finland. J. Hum. Hypertens. 2005, 19, 373–379. [Google Scholar] [CrossRef] [Green Version]
- Rubin, S.J.; Wu, K.Y.; Kirke, D.N.; Ezzat, W.H.; Truong, M.T.; Salama, A.R.; Jalisi, S. Head and Neck Cancer Complications in the Geriatric Population Based on Hospital Case Volume. Ear Nose Throat J. 2021, 100, NP62–NP68. [Google Scholar] [CrossRef] [Green Version]
| Parameter | With HNC (n = 8749) | Without HNC (n = 4,567,069) | p Value |
|---|---|---|---|
| Sex, n (%) | <0.001 * | ||
| Male | 7308 (83.53) | 2,469,600 (53,97) | |
| Female | 1441 (16,47) | 2,097,469 (45,84) | |
| Age, year | 59.53 ± 9.69 | 53.96 ± 9.32 | <0.001 * |
| Alcohol drinking, n (%) | <0.001 * | ||
| No | 4036 (46.13) | 2,564,134 (56.04) | |
| Moderate | 4200 (48.01) | 1,860,466 (40.66) | |
| Heavy | 513 (5.86) | 142,469 (3.11) | |
| Smoking status, n (%) | <0.001 * | ||
| Never smoker | 4427 (50.60) | 3,167,225 (69.22)0 | |
| Ex-smoker | 1276 (14.58) | 472,603 (10.33) | |
| Current smoker | 3046 (34.82) | 927,241 (20.26) | |
| Body mass index, kg/m2 | 23.70 ± 3.00 | 23.98 ± 3.32 | <0.001 * |
| Waist circumference, cm | 83.78 ± 8.16 | 81.68 ± 8.45 | <0.001 * |
| Total cholesterol, mg/dL | 193.35 ± 37.43 | 197.45 ± 36.95 | <0.001 * |
| Triglyceride, mg/dL | 146.24 ± 98.10 | 197.45 ± 36.95 | <0.001 * |
| LDL cholesterol, mg/dL | 113.56 ± 115.86 | 119.64 ± 79.48 | <0.001 * |
| HDL cholesterol, mg/dL | 54.71 ± 31.45 | 55.55 ± 31.89 | 0.0144 * |
| Hypertension, n (%) | <0.001 * | ||
| No | 3642 (41.63) | 2,330,154 (50.92) | |
| Yes | 5107 (58.37) | 2,236,915 (48.89) | |
| Diabetes, n | <0.001 * | ||
| No | 7260 (82.98) | 4,029,420 (88.06) | |
| Yes | 1489 (17.02) | 537,649 (11.75) | |
| Metabolic syndrome, n (%) | <0.001 * | ||
| No | 6209 (70.97) | 3,372,434 (73.84) | |
| Yes | 5540 (29.03) | 1,194,635 (26.16) |
| Parameter | n | HNC (Adjusted for Age and Sex) | |
|---|---|---|---|
| p Value | HR (95% CI) | ||
| Alcohol drinking, n | |||
| No | 4036 | 1 (reference) | |
| Moderate | 4200 | 0.0013 | 1.079 (1.030–1.131) |
| Heavy | 513 | <0.001 | 1.498 (1.363–1.646) |
| Smoking status, n | |||
| Never smoker | 4427 | 1 (reference) | |
| Ex-smoker | 1276 | <0.001 | 1.185 (1.110–1.265) |
| Current smoker | 3046 | <0.001 | 1.668 (1.586–1.754) |
| BMI, kg/m2 | |||
| <18.5 | 303 | <0.001 | 1.694 (1.505–1.906) |
| 18.5–22.9 | 3250 | 1 (reference) | |
| 23–24.9 | 2344 | <0.001 | 0.838 (0.794–0.883) |
| ≥25 | 2852 | <0.001 | 0.804 (0.765–0.846) |
| Central obesity | |||
| No | 6691 | 1 (reference) | |
| Yes | 2058 | 0.0218 | 0.943 (0.897–0.992) |
| Total cholesterol | |||
| Low | 5198 | 1 (reference) | |
| High | 3551 | <0.001 | 0.910 (0.871–0.949) |
| LDL cholesterol | |||
| Low | 3257 | 1 (reference) | |
| High | 5492 | <0.001 | 0.839 (0.803–0.876) |
| TG | |||
| Low | 5657 | 1 (reference) | |
| High | 3092 | 0.649 | 1.010 (0.967–1.056) |
| HDL cholesterol | |||
| Low | 7158 | 0.7573 | 1.009 (0.955–1.066) |
| High | 1591 | 1 (reference) | |
| Hypertension | |||
| No | 3642 | 1 (reference) | |
| Yes | 5107 | 0.1977 | 1.029 (0.985–1.075) |
| Diabetes | |||
| No | 7260 | 1 (reference) | |
| Yes | 1489 | <0.001 | 1.181 (1.116–1.249) |
| Metabolic syndrome | |||
| No | 6209 | 1 (reference) | |
| Yes | 2540 | 0.3578 | 1.022 (0.976–1.071) |
| Parameter | n | Male (n = 7308) | n | Female (n = 1441) | ||
|---|---|---|---|---|---|---|
| p Value | HR (95% CI) | p Value | HR (95% CI) | |||
| BMI, kg/m2 | ||||||
| <18.5 | 260 | <0001 | 1.710 (1.506–1.942) | 43 | 0.0042 | 1.578 (1.155–2.156) |
| 18.5–22.9 | 2751 | < | 1 (reference) | 499 | 1 (reference) | |
| 23–24.9 | 1951 | <0001 | 506 | 0.1546 | 1.101 (0.964–1.258) | |
| ≥25 | 2346 | <0001 | 393 | 0.4324 | 1.052 (0.928–1.192) | |
| Central obesity | ||||||
| No | 5435 | 1 (reference) | 1256 | 1 (reference) | ||
| Yes | 1873 | 0.016 | 0.937 (0.889–0.988) | 185 | 0.684 | 1.033 (0.883–1.208) |
| Total cholesterol | ||||||
| Low | 4458 | 1 (reference) | 740 | 1 (reference) | ||
| High | 2850 | 0.0004 | 0.918 (0.876–0.962) | 701 | 0.2519 | 0.941 (0.847–1.044) |
| LDL cholesterol | ||||||
| Low | 2862 | 1 (reference) | 395 | 1 (reference) | ||
| High | 4446 | <0001 | 0.830 (0.792–0.870) | 1046 | 0.2588 | 0.935 (0.833–1.05) |
| TG | ||||||
| Low | 4622 | 1 (reference) | 1035 | 1 (reference) | ||
| High | 2686 | 0.228 | 1.03 (0.982–1.08) | 406 | 0.8323 | 1.013 (0.901–1.138) |
| HDL cholesterol | ||||||
| Low | 6201 | 1 (reference) | 957 | 1 (reference) | ||
| High | 1107 | 0.8254 | 1.007 (0.945–1.074) | 484 | 0.6007 | 0.971 (0.87–1.084) |
| Hypertension | ||||||
| No | 2942 | 1 (reference) | 700 | 1 (reference) | ||
| Yes | 4366 | 0.0467 | 1.049 (1.001–1.101) | 741 | 0.6007 | 0.971 (0.87–1.084) |
| Diabetes | ||||||
| No | 6001 | 1 (reference) | 1259 | 1 (reference) | ||
| Yes | 1307 | <0001 | 1.183 (1.114–1.256) | 182 | 0.0307 | 1.190 (1.016–1.393) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, S.-Y.; Cheong, H.-K.; Lee, M.-K.; Kang, J.-W.; Lee, Y.-C.; Oh, I.-H.; Eun, Y.-G. Metabolic Diseases and Risk of Head and Neck Cancer: A Cohort Study Analyzing Nationwide Population-Based Data. Cancers 2022, 14, 3277. https://doi.org/10.3390/cancers14133277
Choi S-Y, Cheong H-K, Lee M-K, Kang J-W, Lee Y-C, Oh I-H, Eun Y-G. Metabolic Diseases and Risk of Head and Neck Cancer: A Cohort Study Analyzing Nationwide Population-Based Data. Cancers. 2022; 14(13):3277. https://doi.org/10.3390/cancers14133277
Chicago/Turabian StyleChoi, Soo-Young, Hyeon-Kyoung Cheong, Min-Kyeong Lee, Jeong-Wook Kang, Young-Chan Lee, In-Hwan Oh, and Young-Gyu Eun. 2022. "Metabolic Diseases and Risk of Head and Neck Cancer: A Cohort Study Analyzing Nationwide Population-Based Data" Cancers 14, no. 13: 3277. https://doi.org/10.3390/cancers14133277
APA StyleChoi, S.-Y., Cheong, H.-K., Lee, M.-K., Kang, J.-W., Lee, Y.-C., Oh, I.-H., & Eun, Y.-G. (2022). Metabolic Diseases and Risk of Head and Neck Cancer: A Cohort Study Analyzing Nationwide Population-Based Data. Cancers, 14(13), 3277. https://doi.org/10.3390/cancers14133277







