Comparison of Oncologic Outcomes between Transduodenal Ampullectomy and Pancreatoduodenectomy in Ampulla of Vater Cancer: Korean Multicenter Study
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection and Evaluation
2.2. Statistical Analysis
3. Results
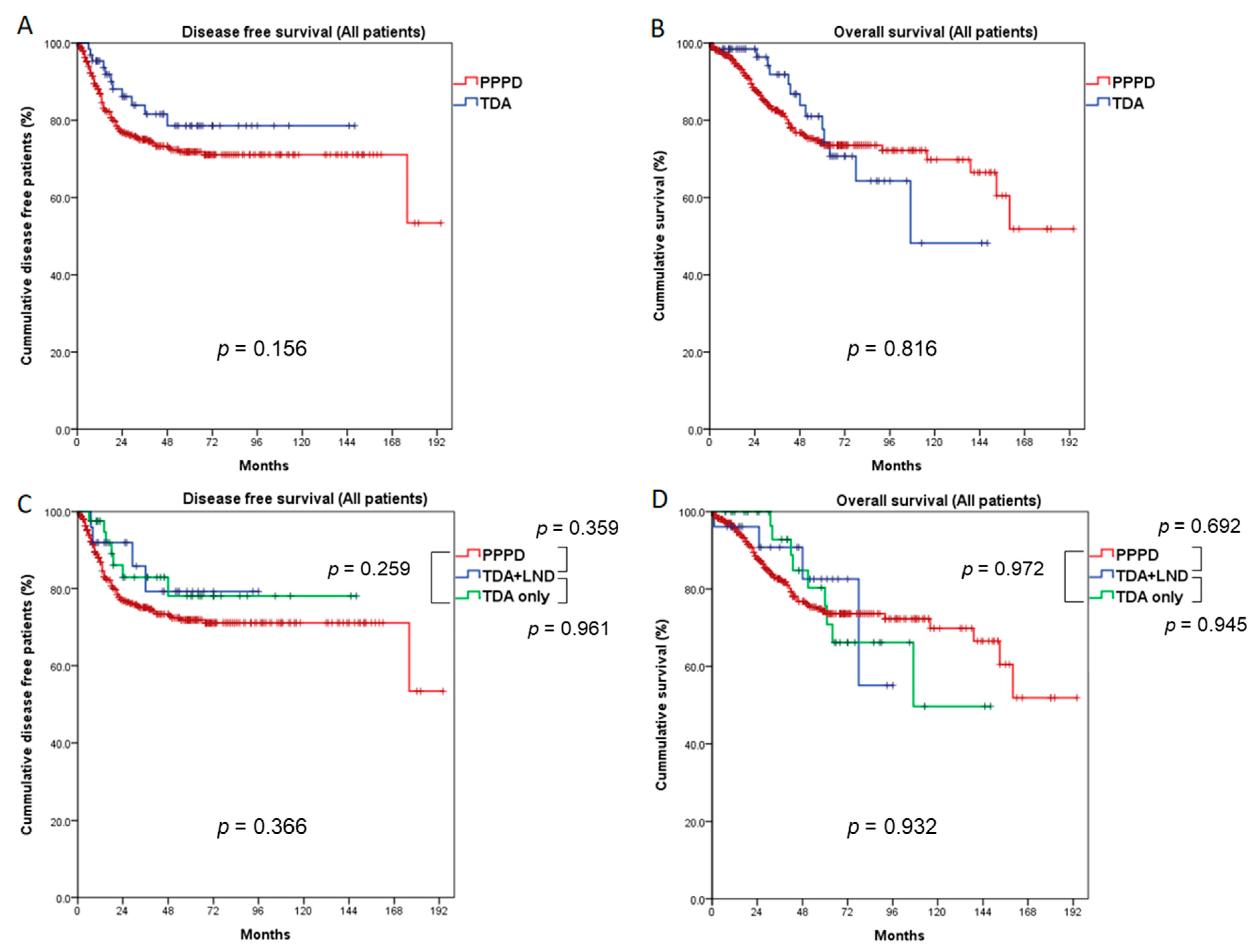
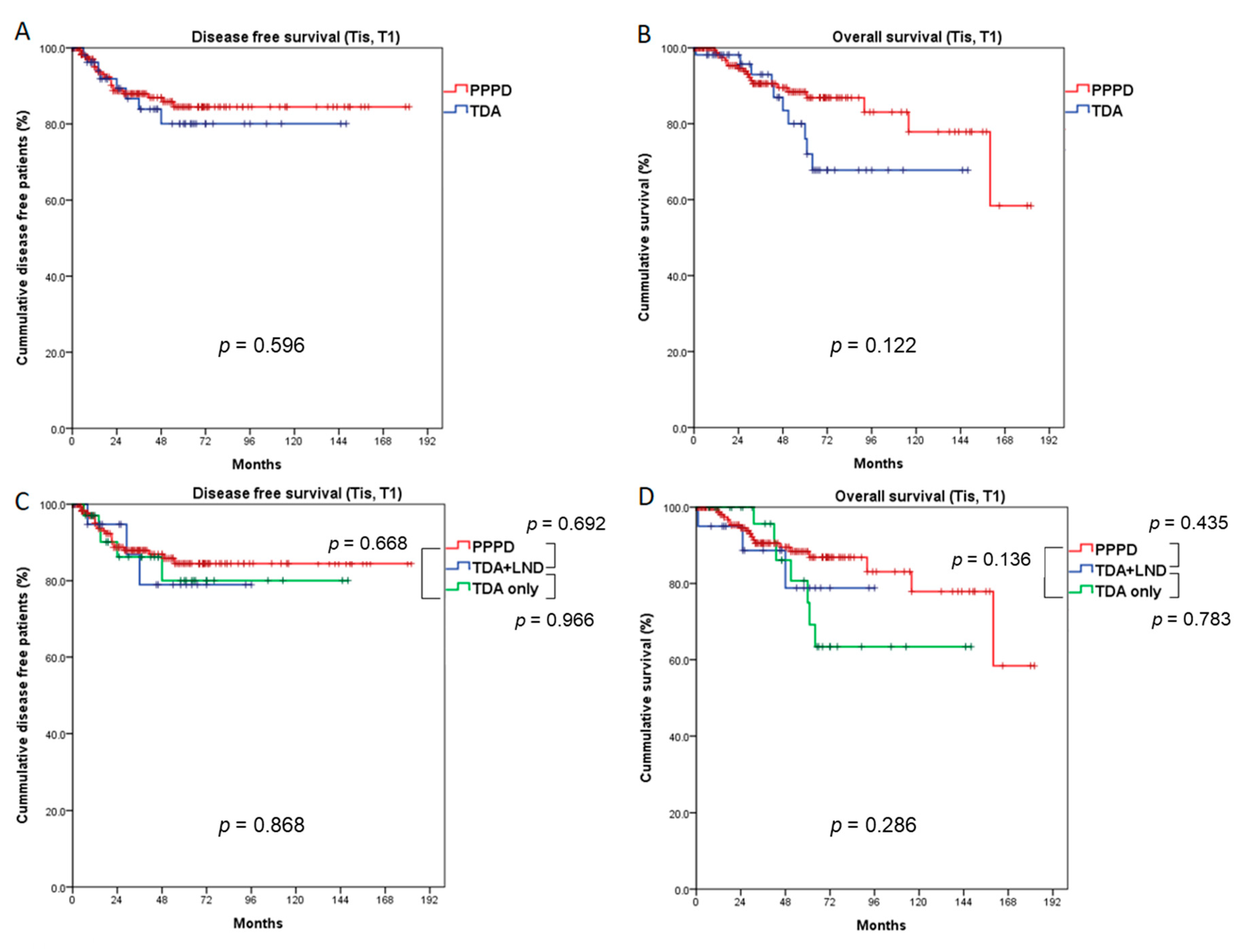
3.1. Clinicopathologic Characteristics of the PPPD and TDA Groups according to T Stage
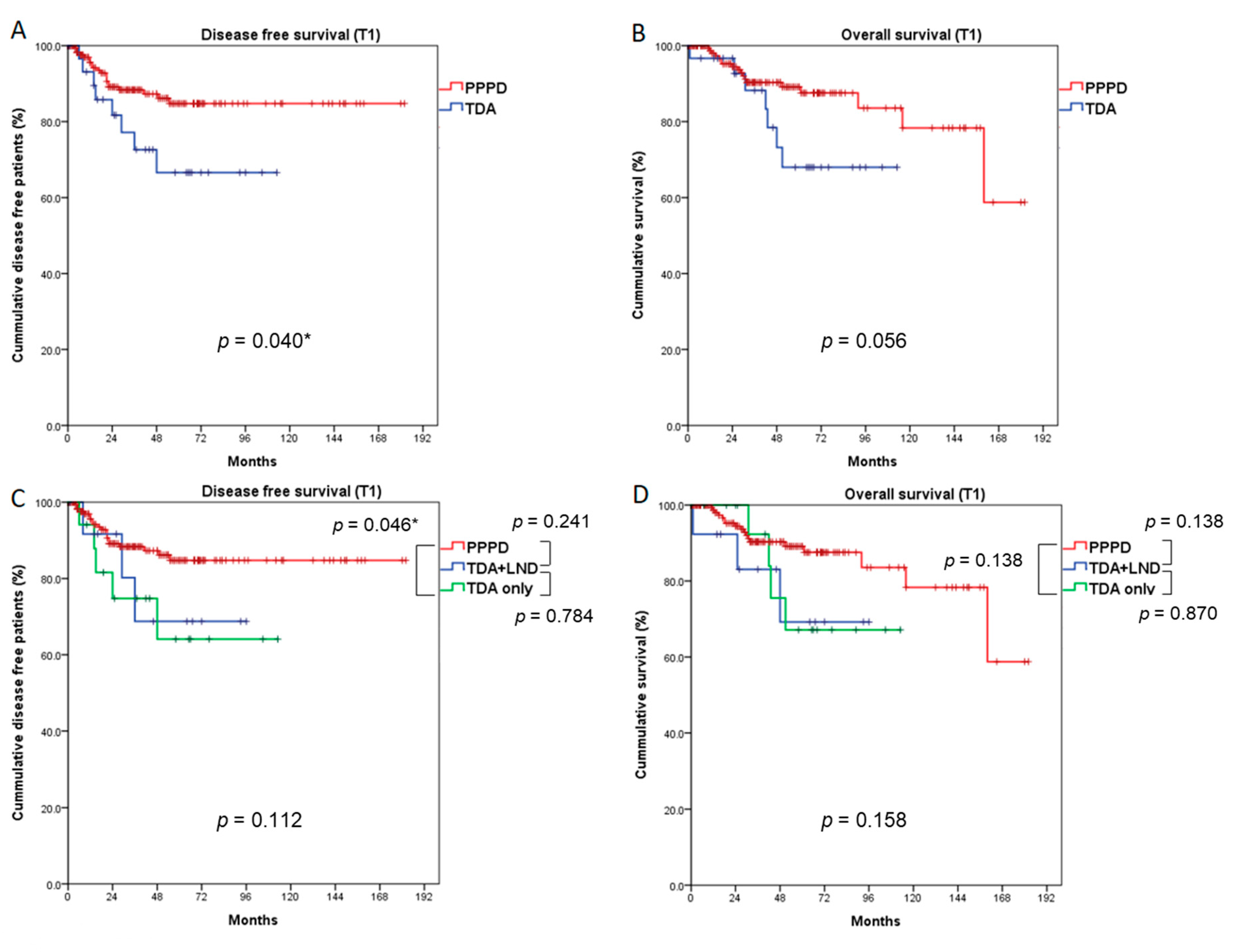
3.2. Clinicopathologic Characteristics of the PPPD, TDA+LND, and TDA-Only Groups with T1 Stage Disease
3.3. Comparison of Survival Outcomes
3.4. Propensity Score Matching Analysis
3.5. Analysis of Risk Factors That Affect Survival Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jemal, A.; Siegel, R.; Ward, E.; Hao, Y.; Xu, J.; Murray, T.; Thun, M.J. Cancer statistics, 2008. CA Cancer J. Clin. 2008, 58, 71–96. [Google Scholar] [CrossRef]
- Howard, J.M.; Jordan, G.L. Surgical Diseases of the Pancreas; Pitman Medical Publishing: London, UK, 1960. [Google Scholar]
- Sommerville, C.A.; Limongelli, P.; Pai, M.; Ahmad, R.; Stamp, G.; Habib, N.A.; Williamson, R.C.; Jiao, L.R. Survival analysis after pancreatic resection for ampullary and pancreatic head carcinoma: An analysis of clinicopathological factors. J. Surg. Oncol. 2009, 100, 651–656. [Google Scholar] [CrossRef]
- Yeo, C.J.; Cameron, J.L.; Sohn, T.A.; Lillemoe, K.D.; Pitt, H.A.; Talamini, M.A.; Hruban, R.H.; Ord, S.E.; Sauter, P.K.; Coleman, J.; et al. Six hundred fifty consecutive pancreaticoduodenectomies in the 1990s: Pathology, complications, and outcomes. Ann. Surg. 1997, 226, 248–257. [Google Scholar] [CrossRef]
- Bouvet, M.; Gamagami, R.A.; Gilpin, E.A.; Romeo, O.; Sasson, A.; Easter, D.W.; Moossa, A.R. Factors influencing survival after resection for periampullary neoplasms. Am. J. Surg. 2000, 180, 13–17. [Google Scholar] [CrossRef]
- Ahmad, S.R.; Adler, D.G. Cancer of the ampulla of vater: Current evaluation and therapy. Hosp. Pract. 2014, 42, 45–61. [Google Scholar] [CrossRef]
- Ahn, D.H.; Bekaii-Saab, T. Ampullary cancer: An overview. Am. Soc. Clin. Oncol. Educ. Book 2014. [Google Scholar] [CrossRef]
- Kobayashi, A.; Konishi, M.; Nakagohri, T.; Takahashi, S.; Kinoshita, T. Therapeutic approach to tumors of the ampulla of Vater. Am. J. Surg. 2006, 192, 161–164. [Google Scholar] [CrossRef]
- O’Connell, J.B.; Maggard, M.A.; Manunga, J., Jr.; Tomlinson, J.S.; Reber, H.A.; Ko, C.Y.; Hines, O.J. Survival after resection of ampullary carcinoma: A national population-based study. Ann. Surg. Oncol. 2008, 15, 1820–1827. [Google Scholar] [CrossRef]
- Kim, A.L.; Choi, Y.I. Safety of duodenal ampullectomy for benign periampullary tumors. Ann. Hepato-Biliary-Pancreat. Surg. 2017, 21, 146–150. [Google Scholar] [CrossRef]
- Jung, Y.K.; Paik, S.S.; Choi, D.; Lee, K.G. Transduodenal ampullectomy for ampullary tumor. Asian J. Surg. 2021. [Google Scholar] [CrossRef]
- Park, J.S.; Yoon, D.S.; Park, Y.N.; Lee, W.J.; Chi, H.S.; Kim, B.R. Transduodenal local resection for low-risk group ampulla of vater carcinoma. J. Laparoendosc Adv. Surg. Tech. A 2007, 17, 737–742. [Google Scholar] [CrossRef]
- Dittrick, G.W.; Mallat, D.B.; Lamont, J.P. Management of ampullary lesions. Curr. Treat. Options Gastroenterol. 2006, 9, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Sauvanet, A.; Dokmak, S.; Cros, J.; Cazals-Hatem, D.; Ponsot, P.; Palazzo, M. Surgical Ampullectomy with Complete Resection of the Common Bile Duct: A New Procedure for Radical Resection of Non-invasive Ampulloma with Biliary Extension. J. Gastrointest. Surg. 2017, 21, 1533–1539. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Liu, H.; Li, Z.; Yang, C.; Sun, Y.; Wang, C. Long-term prognosis of surgical treatment for early ampullary cancers and implications for local ampullectomy. BMC Surg. 2015, 15, 32. [Google Scholar] [CrossRef] [PubMed]
- Winter, J.M.; Cameron, J.L.; Olino, K.; Herman, J.M.; de Jong, M.C.; Hruban, R.H.; Wolfgang, C.L.; Eckhauser, F.; Edil, B.H.; Choti, M.A.; et al. Clinicopathologic analysis of ampullary neoplasms in 450 patients: Implications for surgical strategy and long-term prognosis. J. Gastrointest. Surg. 2010, 14, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Zhu, Y.; Huang, X.; Wang, H.; Huang, X.; Yuan, Z. Transduodenal ampullectomy provides a less invasive technique to cure early ampullary cancer. BMC Surg. 2016, 16, 36. [Google Scholar] [CrossRef]
- Lee, H.; Park, J.Y.; Kwon, W.; Heo, J.S.; Choi, D.W.; Choi, S.H. Transduodenal Ampullectomy for the Treatment of Early-Stage Ampulla of Vater Cancer. World J. Surg. 2016, 40, 967–973. [Google Scholar] [CrossRef]
- Halsted, W.S. Contributions to the Surgery of the Bile Passages, Especially of the Common Bile-Duct. Boston Med. Surg. J. 1899, 141, 645–654. [Google Scholar] [CrossRef]
- Kausch, W. Das Carcinom der Papilla duodeni und seine radikale Entfernung. Beitr. Klin. Chir. 1912, 78, 439–486. [Google Scholar]
- Tenani, O. Contributo alla chirurgia della papilla del Vater. Policlinico 1922, 29, 291–333. [Google Scholar]
- Whipple, A.O.; Parsons, W.B.; Mullins, C.R. Treatment of carcinoma of the ampulla of Vater. Ann. Surg. 1935, 102, 763–779. [Google Scholar] [CrossRef]
- Brunschwig, A. Resection of head of pancreas and duodenum for carcinoma-pancreatoduodenectomy. Surg. Gynecol. Obs. 1937, 65, 681–684. [Google Scholar] [CrossRef]
- Whipple, A. Observations on radical surgery for lesions of the pancreas. Surg. Gynecol. Obs. 1946, 82, 623–631. [Google Scholar]
- Yoon, Y.S.; Kim, S.W.; Park, S.J.; Lee, H.S.; Jang, J.Y.; Choi, M.G.; Kim, W.H.; Lee, K.U.; Park, Y.H. Clinicopathologic analysis of early ampullary cancers with a focus on the feasibility of ampullectomy. Ann. Surg. 2005, 242, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.M.; Kim, M.-H.; Kim, M.J.; Jang, S.J.; Lee, T.Y.; Kwon, S.; Oh, H.-C.; Lee, S.S.; Seo, D.W.; Lee, S.K. Focal early stage cancer in ampullary adenoma: Surgery or endoscopic papillectomy? Gastrointest. Endosc. 2007, 66, 701–707. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.-H.; Shyr, Y.-M.; Wang, S.-E. Ampullectomy versus pancreaticoduodenectomy for ampullary tumors. J. Chin. Med. Assoc. 2015, 78, 339–344. [Google Scholar] [CrossRef][Green Version]
- Demetriades, H.; Zacharakis, E.; Kirou, I.; Pramateftakis, M.-G.; Sapidis, N.; Kanellos, I.; Betsis, D. Local excision as a treatment for tumors of ampulla of Vater. World J. Surg. Oncol. 2006, 4, 14. [Google Scholar] [CrossRef][Green Version]
- Beger, H.G.; Treitschke, F.; Gansauge, F.; Harada, N.; Hiki, N.; Mattfeldt, T. Tumor of the Ampulla of Vater: Experience With Local or Radical Resection in 171 Consecutively Treated Patients. Arch. Surg. 1999, 134, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Cheng, J.-L.; Cui, J.; Xu, Z.-Z.; Fu, Z.; Liu, J.; Tian, H. Surgical method choice and coincidence rate of pathological diagnoses in transduodenal ampullectomy: A retrospective case series study and review of the literature. World J. Clin. Cases 2019, 7, 717–726. [Google Scholar] [CrossRef]
- Park, J.S.; Yoon, D.S.; Kim, K.S.; Choi, J.S.; Lee, W.J.; Chi, H.S.; Kim, B.R. Factors influencing recurrence after curative resection for ampulla of Vater carcinoma. J. Surg. Oncol. 2007, 95, 286–290. [Google Scholar] [CrossRef]
- Westgaard, A.; Tafjord, S.; Farstad, I.N.; Cvancarova, M.; Eide, T.J.; Mathisen, O.; Clausen, O.P.F.; Gladhaug, I.P. Pancreatobiliary versus intestinal histologic type of differentiation is an independent prognostic factor in resected periampullary adenocarcinoma. BMC Cancer 2008, 8, 170. [Google Scholar] [CrossRef] [PubMed]
- Westgaard, A.; Pomianowska, E.; Clausen, O.P.F.; Gladhaug, I.P. Intestinal-type and Pancreatobiliary-type Adenocarcinomas: How Does Ampullary Carcinoma Differ from Other Periampullary Malignancies? Ann. Surg. Oncol. 2013, 20, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.E.; Kim, M.-J.; Park, M.-S.; Choi, J.Y.; Kim, H.; Kim, S.K.; Lee, M.; Kim, H.J.; Choi, J.-S.; Song, S.Y.; et al. Differential Features of Pancreatobiliary- and Intestinal-type Ampullary Carcinomas at MR Imaging. Radiology 2010, 257, 384–393. [Google Scholar] [CrossRef] [PubMed]

| Tis, T1, and T2 | Tis and T1 | T1 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| PPPD (418) | TDA (68) | p-Value | PPPD (194) | TDA (55) | p-Value | PPPD (188) | TDA (31) | p-Value | |
| Age (years) | 62.5 ± 9.5 | 61.2 ± 12.7 | 0.405 | 63.2 ± 9.1 | 60.1 ± 12.9 | 0.105 | 63.1 ± 9.0 | 60.6 ± 14.3 | 0.348 |
| Male (%) | 213 (51.0%) | 36 (52.9%) | 0.761 | 89 (45.9%) | 28 (50.9%) | 0.509 | 86(45.7%) | 15(48.4%) | 0.785 |
| Size (cm) | 1.9 ± 1.0 | 1.6 ± 0.8 | 0.034 | 1.8 ± 1.1 | 1.6 ± 0.8 | 0.249 | 1.8 ± 1.1 | 1.44 ± 0.8 | 0.076 |
| T staging | <0.001 | <0.001 | |||||||
| Tis | 6 (1.4%) | 24 (35.3%) | 6 (3.1%) | 24 (43.6%) | |||||
| T1 | 188 (45.0%) | 31 (45.6%) | 188 (96.9%) | 31 (56.4%) | |||||
| T2 | 224 (53.6%) | 13 (19.1%) | |||||||
| N staging | <0.001 | <0.001 | <0.001 | ||||||
| Nx | 10 (2.4%) | 42 (61.8%) | 6 (3.1%) | 35 (63.6%) | 6 (3.2%) | 18 (58.1%) | |||
| N0 | 303 (72.5%) | 24 (35.3%) | 167 (86.1%) | 19 (34.5%) | 161 (85.6%) | 12(38.7%) | |||
| N1 | 105 (25.1%) | 2 (2.9%) | 21 (10.8%) | 1 (1.85) | 21(11.2%) | 1(3.2%) | |||
| Differentiation | <0.001 | <0.001 | 0.023 | ||||||
| Well | 184 (45.0%) | 28 (54.9%) | 114 (61.0%) | 24 (58.5%) | 113(62.1%) | 22(78.6%) | |||
| Moderate | 189 (46.2%) | 8 (15.7%) | 61 (32.6%) | 4 (9.8%) | 61(33.5%) | 4(14.3%) | |||
| Poorly | 29 (7.1%) | 1 (2.0%) | 6 (3.2%) | 0 (0.0%) | 6(3.3%) | 0 | |||
| Undiff. | 2 (0.5%) | 0 (0.0%) | 1 (0.5%) | 0 (0.0%) | 0 | 0 | |||
| Etc. | 5 (1.2%) | 14 (27.5%) | 5 (2.7%) | 13 (31.7%) | 2(1.1%) | 2(7.1%) | |||
| LVI (+) | 128 (36.2%) | 0 (0.0%) | 0.002 | 35 (22.6%) | 0 (0.0%) | 0.077 | 35(22.9%) | 0 | 0.123 |
| PNI (+) | 71 (20.2%) | 1 (4.8%) | 0.092 | 22 (14.3%) | 0 (0.0%) | 0.230 | 22(14.4%) | 0 | 0.220 |
| Adj. Tx | 104 (24.9%) | 7 (10.3%) | 0.008 | 18 (9.3%) | 1 (1.8%) | 0.084 | 18(9.6%) | 1(3.2%) | 0.487 |
| R status | <0.001 | 0.010 | 0.019 | ||||||
| R0 | 417 (99.8%) | 62 (91.2%) | 194 (100%) | 52 (94.5%) | 188(100%) | 29(93.5%) | |||
| R1 | 1 (0.2%) | 6 (8.8%) | 0 | 3 (5.5%) | 0 | 2(6.5%) | |||
| Recurrence | 93 (22.2%) | 11 (16.2%) | 0.258 | 21(10.8%) | 8 (14.5%) | 0.448 | 20 (10.6%) | 8 (25.8%) | 0.036 |
| Recurrence pattern | 0.012 | 0.033 | 0.015 | ||||||
| Local | 23 (5.5%) | 7 (10.3%) | 2 (1.0%) | 4 (7.3%) | 1 (0.5%) | 4 (12.9%) | |||
| Systemic | 70 (16.7%) | 4 (5.9%) | 19 (9.8%) | 4 (7.3%) | 19 (10.1%) | 4 (12.9%) | |||
| Peritoneal seeding | 7 (1.7%) | 1 (1.5%) | 1.000 | ||||||
| Operation | p-Value | ||||||
|---|---|---|---|---|---|---|---|
| 1. PPPD (188) | 2. TDA + LND (13) | 3. TDA-Only (18) | 1 vs. 2 | 1 vs. 3 | 2 vs. 3 | Total | |
| Age (years) | 63.07 ± 9.03 | 62.15 ± 11.66 | 59.39 ± 16.17 | 0.729 | 0.354 | 0.584 | 0.319 |
| Male (%) | 86 (45.7%) | 6 (46.2%) | 9 (50.0%) | 0.977 | 0.729 | 0.833 | 0.942 |
| Size (cm) | 1.81 ± 1.09 | 1.76 ± 0.68 | 1.20 ± 0.79 | 0.866 | 0.025 | 0.049 | 0.073 |
| N staging | |||||||
| Nx | 6 (3.2%) | 18 (100.0%) | 1.000 | <0.001 | <0.001 | <0.001 | |
| N0 | 161 (85.6%) | 12 (92.3%) | |||||
| N1 | 21 (11.2%) | 1 (7.7%) | |||||
| Differentiation | |||||||
| Well | 113 (62.1%) | 8 (72.7%) | 14 (82.4%) | 0.845 | 0.008 | 0.253 | 0.017 |
| Moderate | 61 (33.5%) | 3 (27.3%) | 1 (5.9%) | ||||
| Poorly | 6 (3.3%) | 0 (0.0%) | 0 (0.0%) | ||||
| Undiff. | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | ||||
| Etc. | 2 (1.1%) | 0 (0.0%) | 2 (11.8%) | ||||
| LVI (+) | 35 (22.9%) | 0 (0.0%) | 0 (0.0%) | 0.349 | 0.576 | − | 0.202 |
| PNI (+) | 22 (14.4%) | 0 (0.0%) | 0 (0.0%) | 0.595 | 1.000 | − | 0.340 |
| Adj. Tx | 18 (9.6%) | 1 (7.7%) | 0 (0.0%) | 1.000 | 0.377 | 0.419 | 0.383 |
| R status | |||||||
| R0 | 188 (100.0%) | 11 (84.6%) | 18 (100.0%) | 0.004 | − | 0.168 | <0.001 |
| R1 | 0 (0.0%) | 2 (15.4%) | 0 (0.0%) | ||||
| Recurrence | 20 (10.6%) | 3 (23.1%) | 5 (27.8%) | 0.174 | 0.050 | 1.000 | 0.019 |
| Recurrence pattern | 0.249 | 0.016 | 1.000 | 0.004 | |||
| Local | 1 (0.5%) | 1 (7.7%) | 3 (16.7%) | ||||
| Systemic | 19 (10.1%) | 2 (15.4%) | 2 (11.1%) | ||||
| Disease Free Survival | Overall Survival | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Univariate | Multivariate | Univariate | Multivariate | |||||||||
| OR | 95% CI | p | OR | 95% CI | p | OR | 95% CI | p | OR | 95% CI | p | |
| Age ≥65 years | 1.502 | 1.024–2.203 | 0.038 | 1.976 | 1.317–2.967 | <0.001 | ||||||
| Male | 1.132 | 0.772–1.659 | 0.525 | 1.336 | 0.888–2.012 | 0.165 | ||||||
| Size ≥1.0 cm | 1.481 | 0.748–2.933 | 0.260 | 1.828 | 0.799–4.183 | 0.153 | ||||||
| Op method | ||||||||||||
| PPPD | ||||||||||||
| TDA + LND | 0.630 | 0.232–1.713 | 0.365 | 0.826 | 0.302–2.256 | 0.709 | ||||||
| TDA-only | 0.646 | 0.300–1.393 | 0.265 | 0.990 | 0.497–1.973 | 0.978 | ||||||
| T stage | ||||||||||||
| Tis | ||||||||||||
| T1 | 3.892 | 0.530–28.611 | 0.182 | 0.827 | 0.287–2.377 | 0.724 | ||||||
| T2 | 10.869 | 1.512–78.153 | 0.018 | 827.4 | 0–∞ | 0.899 | 2.115 | 0.770–5.807 | 0.146 | |||
| N stage | ||||||||||||
| N0 | ||||||||||||
| Nx | 1.195 | 0.605–2.359 | 0.608 | 1.855 | 0.967–3.559 | 0.063 | ||||||
| N1 | 3.841 | 2.566–5.751 | <0.001 | 2.030 | 1.263–3.262 | 0.003 | 5.396 | 3.483–8.361 | <0.001 | 3.204 | 1.806–5.062 | <0.001 |
| Differentiation | ||||||||||||
| Well | ||||||||||||
| Mod. | 2.574 | 1.643–4.032 | <0.001 | 1.778 | 1.023–3.089 | 0.041 | 2.750 | 1.701–4.445 | <0.001 | 2.397 | 1.309–4.388 | 0.005 |
| Poorly | 4.002 | 2.065–7.755 | <0.001 | 2.059 | 0.974–4.354 | 0.059 | 3.523 | 1.632–7.605 | <0.001 | 2.343 | 0.991–5.540 | 0.052 |
| LVI+ | 2.580 | 1.709–3.895 | <0.001 | 1.394 | 0.865–2.246 | 0.172 | 3.449 | 2.179–5.460 | <0.001 | 1.727 | 1.026–2.908 | 0.040 |
| PNI+ | 1.621 | 1.028–2.555 | 0.038 | 1.206 | 0.752–1.935 | 0.437 | 1.442 | 0.854–2.435 | 0.170 | |||
| Disease Free Survival | Overall Survival | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Univariate | Multivariate | Univariate | Multivariate | |||||||||
| OR | 95% CI | p | OR | 95% CI | p | OR | 95% CI | p | OR | 95% CI | p | |
| Age ≥65 | 1.696 | 0.807–3.567 | 0.163 | 2.249 | 1.008–5.018 | 0.048 | ||||||
| Male | 0.537 | 0.243–1.187 | 0.124 | 1.021 | 0.466–2.238 | 0.959 | ||||||
| Size ≥1.0 cm | 1.315 | 0.456–3.789 | 0.613 | 2.047 | 0.479–8.751 | 0.334 | ||||||
| Op method | ||||||||||||
| PPPD | ||||||||||||
| TDA + LND | 2.033 | 0.604–6.844 | 0.252 | 2.473 | 0.720–8.493 | 0.150 | ||||||
| TDA-only | 2.500 | 0.938–6.664 | 0.067 | 2.211 | 0.739–6.617 | 0.156 | ||||||
| N stage | ||||||||||||
| N0 | ||||||||||||
| Nx | 3.045 | 1.157–8.014 | 0.024 | − | − | − | 4.228 | 1.56611.413 | 0.004 | 2.863 | 0.526–15.584 | 0.224 |
| N1 | 7.920 | 3.362–18.655 | <0.001 | 2.148 | 0.645–7.146 | 0.213 | 13.214 | 5.157–33.859 | <0.001 | 3.898 | 1.024–14.849 | 0.046 |
| Differentiation | ||||||||||||
| Well | ||||||||||||
| Mod. | 3.035 | 1.278–7.204 | 0.012 | 1.924 | 0.576–6.425 | 0.287 | 1.313 | 0.543–3.178 | 0.545 | |||
| Poorly | 11.104 | 2.948–41.821 | <0.001 | 5.446 | 1.073–27.650 | 0.041 | 8.613 | 1.829–40.557 | 0.006 | 1.303 | 0.222–7.641 | 0.770 |
| LVI+ | 4.108 | 1.617–10.434 | 0.003 | 1.725 | 0.546–5.446 | 0.353 | 11.960 | 3.729–38.362 | <0.001 | 6.933 | 1.594–30.160 | 0.010 |
| PNI+ | 4.124 | 1.683–10.107 | 0.002 | 2.953 | 1.016–8.578 | 0.047 | 2.613 | 0.803–8.502 | 0.111 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hong, S.-S.; Han, S.-S.; Kwon, W.; Jang, J.-Y.; Kim, H.-J.; Cho, C.-K.; Ahn, K.-S.; Yang, J.-D.; Park, Y.; Min, S.-K.; et al. Comparison of Oncologic Outcomes between Transduodenal Ampullectomy and Pancreatoduodenectomy in Ampulla of Vater Cancer: Korean Multicenter Study. Cancers 2021, 13, 2038. https://doi.org/10.3390/cancers13092038
Hong S-S, Han S-S, Kwon W, Jang J-Y, Kim H-J, Cho C-K, Ahn K-S, Yang J-D, Park Y, Min S-K, et al. Comparison of Oncologic Outcomes between Transduodenal Ampullectomy and Pancreatoduodenectomy in Ampulla of Vater Cancer: Korean Multicenter Study. Cancers. 2021; 13(9):2038. https://doi.org/10.3390/cancers13092038
Chicago/Turabian StyleHong, Seung-Soo, Sung-Sik Han, Wooil Kwon, Jin-Young Jang, Hee-Joon Kim, Chol-Kyoon Cho, Keun-Soo Ahn, Jae-Do Yang, Youngmok Park, Seog-Ki Min, and et al. 2021. "Comparison of Oncologic Outcomes between Transduodenal Ampullectomy and Pancreatoduodenectomy in Ampulla of Vater Cancer: Korean Multicenter Study" Cancers 13, no. 9: 2038. https://doi.org/10.3390/cancers13092038
APA StyleHong, S.-S., Han, S.-S., Kwon, W., Jang, J.-Y., Kim, H.-J., Cho, C.-K., Ahn, K.-S., Yang, J.-D., Park, Y., Min, S.-K., Moon, J.-I., Roh, Y.-H., Lee, S.-E., Park, J.-S., Kim, S.-G., Jeong, C.-Y., Heo, J.-S., & Hwang, H.-K. (2021). Comparison of Oncologic Outcomes between Transduodenal Ampullectomy and Pancreatoduodenectomy in Ampulla of Vater Cancer: Korean Multicenter Study. Cancers, 13(9), 2038. https://doi.org/10.3390/cancers13092038






