Nonreplicating Adenoviral Vectors: Improving Tropism and Delivery of Cancer Gene Therapy
Abstract
Simple Summary
Abstract
1. Overview: Structural and Molecular Features of Nonreplicating Adenoviral Vectors
2. Current Applications of Nonreplicating Adenoviral Vectors in Cancer Immunotherapy
3. Challenges of Using Adenoviral Vectors
3.1. Tissue Tropism
3.2. Pre-Existing Immunity in the Host
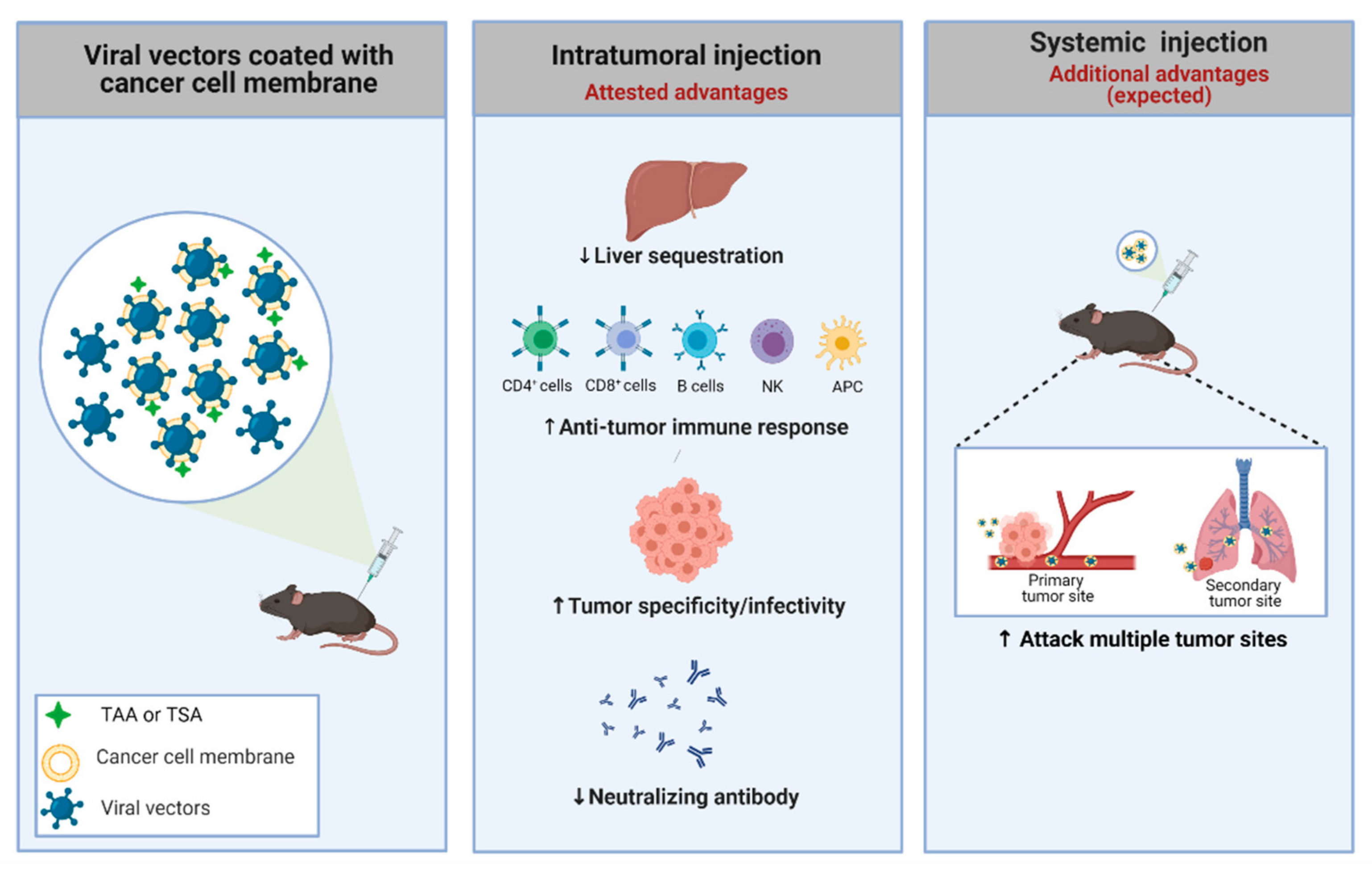
3.3. Different Administration Routes and Their Particularities
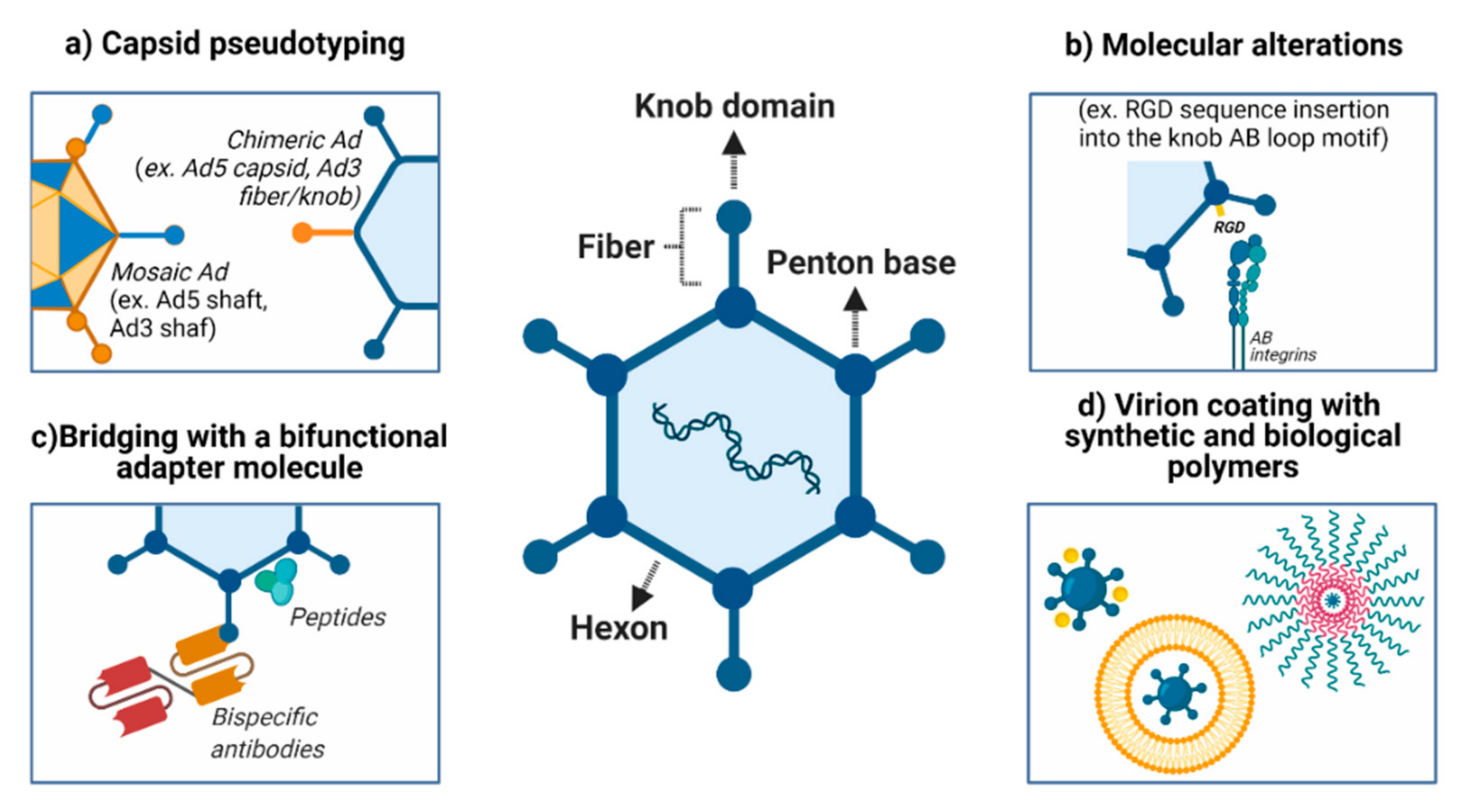
4. Strategies to Modify Adenovirus Tropism
4.1. Modifications in Viral Entry: Attachment Receptors and Virus Internalization
4.2. Pseudotyping the Capsid Using Components from Different Adenoviruses
4.3. Encapsulation of Adenovirus Using Synthetic Polymers
4.4. Cancer Cell Membrane-Coated Adenoviral Vectors
4.5. Association of Antibodies and Viral Structures
4.6. Genetic and Chemical Capsid Modifications and Association with Polymers
5. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ginn, S.L.; Amaya, A.K.; Alexander, I.E.; Edelstein, M.; Abedi, M.R. Gene therapy clinical trials worldwide to 2017: An update. J. Gene Med. 2018, 20, e3015. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.-W.; Li, L.; Li, D.; Liu, J.; Li, X.; Li, W.; Xu, X.; Zhang, M.J.; Chandler, L.A.; Lin, H.; et al. The First Approved Gene Therapy Product for Cancer Ad- P53 (Gendicine): 12 Years in the Clinic. Hum. Gene Ther. 2018, 29, 160–179. [Google Scholar] [CrossRef]
- National Library of Medicine. A Phase II Clinical Trial to Evaluate the Recombinant Vaccine for COVID-19 (Adenovirus Vector). 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04341389 (accessed on 1 April 2021).
- National Library of Medicine. ChiCTR2000030906. A Phase I Clinical Trial for Recombinant Novel Coronavirus (2019-COV) Vaccine (Adenoviral Vector). 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04313127 (accessed on 1 April 2021).
- Chinese Clinical Trial Registry (ChiCTR). ChiCTR2000031781. A Randomized, Double-Blinded, Placebo-Controlled Phase II Clinical Trial for Recombinant Novel Coronavirus (2019-NCOV) Vaccine (Adenovirus Vector). 2020. Available online: http://www.chictr.org.cn/showprojen.aspx?proj=52006 (accessed on 1 April 2021).
- National Library of Medicine. Phase I Clinical Trial of a COVID-19 Vaccine in 18–60 Healthy Adults. 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04313127 (accessed on 1 April 2021).
- National Library of Medicine. Phase I/II Clinical Trial of Recombinant Novel Coronavirus Vaccine (Adenovirus Type 5 Vector) in Canada. 2020. Available online: https://www.clinicaltrials.gov/ct2/show/NCT04398147 (accessed on 1 April 2021).
- National Library of Medicine. COVID-19 Vaccine (ChAdOx1 NCoV-19) Trial in South African Adults with and without HIV-Infection. 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04444674 (accessed on 1 April 2021).
- National Library of Medicine. A Study of a Candidate COVID-19 Vaccine (COV001). 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04324606 (accessed on 1 April 2021).
- National Library of Medicine. An Open Study of the Safety, Tolerability and Immunogenicity of “Gam-COVID-Vac Lyo” Vaccine Against COVID-19. 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04437875 (accessed on 1 April 2021).
- Gamaleya Research Institute of Epidemiology and Microbiology. An Open Study of the Safety, Tolerability and Immunogenicity of the Drug “Gam-COVID-Vac” Vaccine against COVID-19. 2020. Available online: https://www.smartpatients.com/trials/NCT04437875 (accessed on 1 April 2021).
- Coughlan, L. Factors Which Contribute to the Immunogenicity of Non-Replicating Adenoviral Vectored Vaccines. Front. Immunol. 2020, 11, 909. [Google Scholar] [CrossRef] [PubMed]
- Kumon, H.; Ariyoshi, Y.; Sasaki, K.; Sadahira, T.; Araki, M.; Ebara, S.; Yanai, H.; Watanabe, M.; Nasu, Y. Adenovirus Vector Carrying REIC/DKK-3 Gene: Neoadjuvant Intraprostatic Injection for High-Risk Localized Prostate Cancer Undergoing Radical Prostatectomy. Cancer Gene Ther. 2016, 23, 400–409. [Google Scholar] [CrossRef] [PubMed]
- Goto, Y.; Ohe, Y.; Kuribayashi, K.; Nakano, T.; Okada, M.; Toyooka, S.; Kumon, H.; Nakanishi, Y. P2.06-11 A Phase I/II Study of Intrapleural Ad-SGE-REIC Administration in Patients with Refractory Malignant Pleural Mesothelioma. J. Thorac. Oncol. 2018, 13, S746. [Google Scholar] [CrossRef]
- Lowenstein, P.R.; Orringer, D.A.; Sagher, O.; Heth, J.; Hervey-Jumper, S.L.; Mammoser, A.G.; Junck, L.; Leung, D.; Umemura, Y.; Lawrence, T.S.; et al. First-in-Human Phase I Trial of the Combination of Two Adenoviral Vectors Expressing HSV1-TK and FLT3L for the Treatment of Newly Diagnosed Resectable Malignant Glioma: Initial Results from the Therapeutic Reprogramming of the Brain Immune System. J. Clin. Oncol. 2019, 37, 2019. [Google Scholar] [CrossRef]
- Zhu, R.; Weng, D.; Lu, S.; Lin, D.; Wang, M.; Chen, D.; Lv, J.; Li, H.; Lv, F.; Xi, L.; et al. Double-Dose Adenovirus-Mediated Adjuvant Gene Therapy Improves Liver Transplantation Outcomes in Patients with Advanced Hepatocellular Carcinoma. Hum. Gene Ther. 2018, 29, 251–258. [Google Scholar] [CrossRef]
- Kieran, M.W.; Goumnerova, L.; Manley, P.; Chi, S.N.; Marcus, K.J.; Manzanera, A.G.; Polanco, M.L.S.; Guzik, B.W.; Aguilar-Cordova, E.; Diaz-Montero, C.M.; et al. Phase I Study of Gene-Mediated Cytotoxic Immunotherapy with AdV-Tk as Adjuvant to Surgery and Radiation for Pediatric Malignant Glioma and Recurrent Ependymoma. Neuro Oncol. 2019, 21, 537–546. [Google Scholar] [CrossRef]
- Behbahani, T.E.; Rosenthal, E.L.; Parker, W.B.; Sorscher, E.J. Intratumoral Generation of 2-fluoroadenine to Treat Solid Malignancies of the Head and Neck. Head Neck 2019, 41, 1979–1983. [Google Scholar] [CrossRef] [PubMed]
- Shore, N.D.; Boorjian, S.A.; Canter, D.J.; Ogan, K.; Karsh, L.I.; Downs, T.M.; Gomella, L.G.; Kamat, A.M.; Lotan, Y.; Svatek, R.S.; et al. Intravesical RAd–IFNα/Syn3 for Patients with High-Grade, Bacillus Calmette-Guerin–Refractory or Relapsed Non–Muscle-Invasive Bladder Cancer: A Phase II Randomized Study. J. Clin. Oncol. 2017, 35, 3410–3416. [Google Scholar] [CrossRef]
- Sterman, D.H.; Recio, A.; Haas, A.R.; Vachani, A.; Katz, S.I.; Gillespie, C.T.; Cheng, G.; Sun, J.; Moon, E.; Pereira, L.; et al. A Phase I Trial of Repeated Intrapleural Adenoviral-Mediated Interferon-β Gene Transfer for Mesothelioma and Metastatic Pleural Effusions. Mol. Ther. 2010, 18, 852–860. [Google Scholar] [CrossRef]
- Chiocca, E.A.; Yu, J.S.; Lukas, R.V.; Solomon, I.H.; Ligon, K.L.; Nakashima, H.; Triggs, D.A.; Reardon, D.A.; Wen, P.; Stopa, B.M.; et al. Regulatable Interleukin-12 Gene Therapy in Patients with Recurrent High-Grade Glioma: Results of a Phase 1 Trial. Sci. Transl. Med. 2019, 11, eaaw5680. [Google Scholar] [CrossRef] [PubMed]
- Buller, R.E.; Runnebaum, I.B.; Karlan, B.Y.; Horowitz, J.A.; Shahin, M.; Buekers, T.; Petrauskas, S.; Kreienberg, R.; Slamon, D.; Pegram, M. A Phase I/II Trial of RAd/P53 (SCH 58500) Gene Replacement in Recurrent Ovarian Cancer. Cancer Gene Ther. 2002, 9, 553–566. [Google Scholar] [CrossRef] [PubMed]
- Yoo, G.H.; Moon, J.; LeBlanc, M.; Lonardo, F.; Urba, S.; Kim, H.; Hanna, E.; Tsue, T.; Valentino, J.; Ensley, J.; et al. A Phase 2 Trial of Surgery with Perioperative INGN 201 (Ad5CMV-P53) Gene Therapy Followed by Chemoradiotherapy for Advanced, Resectable Squamous Cell Carcinoma of the Oral Cavity, Oropharynx, Hypopharynx, and Larynx: Report of the Southwest Oncology Group. Arch. Otolaryngol. Neck Surg. 2009, 135, 869. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.M.; Singh, G.; Lee, J.Y.; Dehghan, S.; Rajaiya, J.; Liu, E.B.; Yousuf, M.A.; Betensky, R.A.; Jones, M.S.; Dyer, D.W.; et al. Molecular Evolution of Human Adenoviruses. Sci. Rep. 2013, 3, 1812. [Google Scholar] [CrossRef]
- HAdV Working Group. Available online: http://hadvwg.gmu.edu. (accessed on 22 June 2020).
- San Martín, C. Latest Insights on Adenovirus Structure and Assembly. Viruses 2012, 4, 847–877. [Google Scholar] [CrossRef]
- Russell, W.C. Adenoviruses: Update on Structure and Function. J. Gen. Virol. 2009, 90, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.T.; Greenshields-Watson, A.; Coughlan, L.; Davies, J.A.; Uusi-Kerttula, H.; Cole, D.K.; Rizkallah, P.J.; Parker, A.L. Diversity within the Adenovirus Fiber Knob Hypervariable Loops Influences Primary Receptor Interactions. Nat. Commun. 2019, 10, 741. [Google Scholar] [CrossRef]
- Loustalot, F.; Kremer, E.J.; Salinas, S. The Intracellular Domain of the Coxsackievirus and Adenovirus Receptor Differentially Influences Adenovirus Entry. J. Virol. 2015, 89, 9417–9426. [Google Scholar] [CrossRef] [PubMed]
- Murakami, S.; Sakurai, F.; Kawabata, K.; Okada, N.; Fujita, T.; Yamamoto, A.; Hayakawa, T.; Mizuguchi, H. Interaction of Penton Base Arg-Gly-Asp Motifs with Integrins Is Crucial for Adenovirus Serotype 35 Vector Transduction in Human Hematopoietic Cells. Gene Ther. 2007, 14, 1525–1533. [Google Scholar] [CrossRef]
- Strunze, S.; Engelke, M.F.; Wang, I.-H.; Puntener, D.; Boucke, K.; Schleich, S.; Way, M.; Schoenenberger, P.; Burckhardt, C.J.; Greber, U.F. Kinesin-1-Mediated Capsid Disassembly and Disruption of the Nuclear Pore Complex Promote Virus Infection. Cell Host Microbe 2011, 10, 210–223. [Google Scholar] [CrossRef]
- Ricobaraza, A.; Gonzalez-Aparicio, M.; Mora-Jimenez, L.; Lumbreras, S.; Hernandez-Alcoceba, R. High-Capacity Adenoviral Vectors: Expanding the Scope of Gene Therapy. Int. J. Mol. Sci. 2020, 21, 3643. [Google Scholar] [CrossRef]
- Khanal, S.; Ghimire, P.; Dhamoon, A. The Repertoire of Adenovirus in Human Disease: The Innocuous to the Deadly. Biomedicines 2018, 6, 30. [Google Scholar] [CrossRef]
- Lee, C.S.; Bishop, E.S.; Zhang, R.; Yu, X.; Farina, E.M.; Yan, S.; Zhao, C.; Zeng, Z.; Shu, Y.; Wu, X.; et al. Adenovirus-Mediated Gene Delivery: Potential Applications for Gene and Cell-Based Therapies in the New Era of Personalized Medicine. Genes Dis. 2017, 4, 43–63. [Google Scholar] [CrossRef]
- Crystal, R.G. Adenovirus: The First Effective In Vivo Gene Delivery Vector. Hum. Gene Ther. 2014, 25, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Mese, K.; Bunz, O.; Ehrhardt, A. State-of-the-art Human Adenovirus Vectorology for Therapeutic Approaches. FEBS Lett. 2019, 593, 3609–3622. [Google Scholar] [CrossRef]
- McGrory, W.J.; Bautista, D.S.; Graham, F.L. A Simple Technique for the Rescue of Early Region I Mutations into Infectious Human Adenovirus Type 5. Virology 1988, 163, 614–617. [Google Scholar] [CrossRef]
- Bett, A.J.; Haddara, W.; Prevec, L.; Graham, F.L. An Efficient and Flexible System for Construction of Adenovirus Vectors with Insertions or Deletions in Early Regions 1 and 3. Proc. Natl. Acad. Sci. USA 1994, 91, 8802–8806. [Google Scholar] [CrossRef] [PubMed]
- Kovesdi, I.; Hedley, S.J. Adenoviral Producer Cells. Viruses 2010, 2, 1681–1703. [Google Scholar] [CrossRef]
- Graham, F.L.; Smiley, J.; Russell, W.C.; Nairn, R. Characteristics of a Human Cell Line Transformed by DNA from Human Adenovirus Type 5. J. Gen. Virol. 1977, 36, 59–72. [Google Scholar] [CrossRef] [PubMed]
- Lusky, M.; Christ, M.; Rittner, K.; Dieterle, A.; Dreyer, D.; Mourot, B.; Schultz, H.; Stoeckel, F.; Pavirani, A.; Mehtali, M. In Vitro and In Vivo Biology of Recombinant Adenovirus Vectors with E1, E1/E2A, or E1/E4 Deleted. J. Virol. 1998, 72, 2022–2032. [Google Scholar] [CrossRef]
- Seth, P. Vector-Mediated Cancer Gene Therapy: An Overview. Cancer Biol. Ther. 2005, 4, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Di Paolo, N.C.; Miao, E.A.; Iwakura, Y.; Murali-Krishna, K.; Aderem, A.; Flavell, R.A.; Papayannopoulou, T.; Shayakhmetov, D.M. Virus Binding to a Plasma Membrane Receptor Triggers Interleukin-1α-Mediated Proinflammatory Macrophage Response In Vivo. Immunity 2009, 31, 110–121. [Google Scholar] [CrossRef]
- Zhu, J.; Huang, X.; Yang, Y. Innate Immune Response to Adenoviral Vectors Is Mediated by Both Toll-Like Receptor-Dependent and -Independent Pathways. J. Virol. 2007, 81, 3170–3180. [Google Scholar] [CrossRef] [PubMed]
- Takaoka, A.; Wang, Z.; Choi, M.K.; Yanai, H.; Negishi, H.; Ban, T.; Lu, Y.; Miyagishi, M.; Kodama, T.; Honda, K.; et al. DAI (DLM-1/ZBP1) Is a Cytosolic DNA Sensor and an Activator of Innate Immune Response. Nature 2007, 448, 501–505. [Google Scholar] [CrossRef]
- Muruve, D.A.; Pétrilli, V.; Zaiss, A.K.; White, L.R.; Clark, S.A.; Ross, P.J.; Parks, R.J.; Tschopp, J. The Inflammasome Recognizes Cytosolic Microbial and Host DNA and Triggers an Innate Immune Response. Nature 2008, 452, 103–107. [Google Scholar] [CrossRef]
- Franchi, L.; Warner, N.; Viani, K.; Nuñez, G. Function of Nod-like Receptors in Microbial Recognition and Host Defense. Immunol. Rev. 2009, 227, 106–128. [Google Scholar] [CrossRef] [PubMed]
- Binnewies, M.; Roberts, E.W.; Kersten, K.; Chan, V.; Fearon, D.F.; Merad, M.; Coussens, L.M.; Gabrilovich, D.I.; Ostrand-Rosenberg, S.; Hedrick, C.C.; et al. Understanding the Tumor Immune Microenvironment (TIME) for Effective Therapy. Nat. Med. 2018, 24, 541–550. [Google Scholar] [CrossRef]
- Shaw, A.R.; Suzuki, M. Immunology of Adenoviral Vectors in Cancer Therapy. Mol. Ther. Methods Clin. Dev. 2019, 15, 418–429. [Google Scholar] [CrossRef] [PubMed]
- Wold, W.S.M.; Toth, K. Adenovirus Vectors for Gene Therapy, Vaccination and Cancer Gene Therapy. Curr. Gene Ther. 2013, 13, 421–433. [Google Scholar] [CrossRef] [PubMed]
- Tatsis, N.; Ertl, H.C.J. Adenoviruses as Vaccine Vectors. Mol. Ther. 2004, 10, 616–629. [Google Scholar] [CrossRef]
- Robert-Guroff, M. Replicating and Non-Replicating Viral Vectors for Vaccine Development. Curr. Opin. Biotechnol. 2007, 18, 546–556. [Google Scholar] [CrossRef] [PubMed]
- Osada, T.; Yang, X.Y.; Hartman, Z.C.; Glass, O.; Hodges, B.L.; Niedzwiecki, D.; Morse, M.A.; Lyerly, H.K.; Amalfitano, A.; Clay, T.M. Optimization of Vaccine Responses with an E1, E2b and E3-Deleted Ad5 Vector Circumvents Pre-Existing Anti-Vector Immunity. Cancer Gene Ther. 2009, 16, 673–682. [Google Scholar] [CrossRef]
- Cappuccini, F.; Stribbling, S.; Pollock, E.; Hill, A.V.S.; Redchenko, I. Immunogenicity and Efficacy of the Novel Cancer Vaccine Based on Simian Adenovirus and MVA Vectors Alone and in Combination with PD-1 MAb in a Mouse Model of Prostate Cancer. Cancer Immunol. Immunother. 2016, 65, 701–713. [Google Scholar] [CrossRef]
- Bajgelman, M.C.; Strauss, B.E. Development of an Adenoviral Vector with Robust Expression Driven by P53. Virology 2008, 371, 8–13. [Google Scholar] [CrossRef]
- Hunger, A.; Medrano, R.F.; Zanatta, D.B.; Del Valle, P.R.; Merkel, C.A.; de Almeida Salles, T.; Ferrari, D.G.; Furuya, T.K.; Bustos, S.O.; de Freitas Saito, R.; et al. Reestablishment of P53/Arf and Interferon-β Pathways Mediated by a Novel Adenoviral Vector Potentiates Antiviral Response and Immunogenic Cell Death. Cell Death Discov. 2017, 3, 17017. [Google Scholar] [CrossRef] [PubMed]
- Merkel, C.A.; da Silva Soares, R.B.; de Carvalho, A.C.V.; Zanatta, D.B.; Bajgelman, M.C.; Fratini, P.; Costanzi-Strauss, E.; Strauss, B.E. Activation of Endogenous P53 by Combined P19Arf Gene Transfer and Nutlin-3 Drug Treatment Modalities in the Murine Cell Lines B16 and C6. BMC Cancer 2010, 10, 316. [Google Scholar] [CrossRef]
- Medrano, R.F.V.; Catani, J.P.P.; Ribeiro, A.H.; Tomaz, S.L.; Merkel, C.A.; Costanzi-Strauss, E.; Strauss, B.E. Vaccination Using Melanoma Cells Treated with P19arf and Interferon Beta Gene Transfer in a Mouse Model: A Novel Combination for Cancer Immunotherapy. Cancer Immunol. Immunother. 2016, 65, 371–382. [Google Scholar] [CrossRef] [PubMed]
- Catani, J.P.P.; Medrano, R.F.V.; Hunger, A.; Del Valle, P.; Adjemian, S.; Zanatta, D.B.; Kroemer, G.; Costanzi-Strauss, E.; Strauss, B.E. Intratumoral Immunization by P19Arf and Interferon-β Gene Transfer in a Heterotopic Mouse Model of Lung Carcinoma. Transl. Oncol. 2016, 9, 565–574. [Google Scholar] [CrossRef]
- Hunger, A.; Medrano, R.F.; Strauss, B.E. Harnessing Combined P19Arf and Interferon-Beta Gene Transfer as an Inducer of Immunogenic Cell Death and Mediator of Cancer Immunotherapy. Cell Death Dis. 2017, 8, e2784. [Google Scholar] [CrossRef]
- Medrano, R.F.V.; Hunger, A.; Catani, J.P.P.; Strauss, B.E. Uncovering the Immunotherapeutic Cycle Initiated by P19Arf and Interferon-β Gene Transfer to Cancer Cells: An Inducer of Immunogenic Cell Death. OncoImmunology 2017, e1329072. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.-Q.; Beckham, C.; Brown, J.L.; Lukashev, M.; Barsoum, J. Human and Mouse IFN-β Gene Therapy Exhibits Different Anti-Tumor Mechanisms in Mouse Models. Mol. Ther. 2001, 4, 356–364. [Google Scholar] [CrossRef]
- Cerqueira, O.L.D.; Clavijo-Salomon, M.A.; Cardoso, E.C.; Citrangulo Tortelli Junior, T.; Mendonça, S.A.; Barbuto, J.A.M.; Strauss, B.E. Combined P14ARF and Interferon-β Gene Transfer to the Human Melanoma Cell Line SK-MEL-147 Promotes Oncolysis and Immune Activation. Front. Immunol. 2020, 11, 576658. [Google Scholar] [CrossRef]
- Aguilar, L.K.; Shirley, L.A.; Chung, V.M.; Marsh, C.L.; Walker, J.; Coyle, W.; Marx, H.; Bekaii-Saab, T.; Lesinski, G.B.; Swanson, B.; et al. Gene-Mediated Cytotoxic Immunotherapy as Adjuvant to Surgery or Chemoradiation for Pancreatic Adenocarcinoma. Cancer Immunol. Immunother. 2015, 64, 727–736. [Google Scholar] [CrossRef]
- Herman, J.R.; Adler, H.L.; Aguilar-Cordova, E.; Rojas-Martinez, A.; Woo, S.; Timme, T.L.; Wheeler, T.M.; Thompson, T.C.; Scardino, P.T. In Situ Gene Therapy for Adenocarcinoma of the Prostate: A Phase I Clinical Trial. Hum. Gene Ther. 1999, 10, 1239–1250. [Google Scholar] [CrossRef] [PubMed]
- Maatta, A.-M.; Samaranayake, H.; Pikkarainen, J.; Wirth, T.; Yla-Herttuala, S. Adenovirus Mediated Herpes Simplex Virus-Thymidine Kinase/Ganciclovir Gene Therapy for Resectable Malignant Glioma. Curr. Gene Ther. 2009, 9, 356–367. [Google Scholar] [CrossRef]
- Chévez-Barrios, P.; Chintagumpala, M.; Mieler, W.; Paysse, E.; Boniuk, M.; Kozinetz, C.; Hurwitz, M.Y.; Hurwitz, R.L. Response of Retinoblastoma with Vitreous Tumor Seeding to Adenovirus-Mediated Delivery of Thymidine Kinase Followed by Ganciclovir. J. Clin. Oncol. 2005, 23, 7927–7935. [Google Scholar] [CrossRef] [PubMed]
- Sterman, D.H.; Recio, A.; Vachani, A.; Sun, J.; Cheung, L.; DeLong, P.; Amin, K.M.; Litzky, L.A.; Wilson, J.M.; Kaiser, L.R.; et al. Long-Term Follow-up of Patients with Malignant Pleural Mesothelioma Receiving High-Dose Adenovirus Herpes Simplex Thymidine Kinase/Ganciclovir Suicide Gene Therapy. Clin. Cancer Res. 2005, 11, 7444–7453. [Google Scholar] [CrossRef][Green Version]
- Siddiqui, M.R.; Grant, C.; Sanford, T.; Agarwal, P.K. Current Clinical Trials in Non–Muscle Invasive Bladder Cancer. Urol. Oncol. Semin. Orig. Investig. 2017, 35, 516–527. [Google Scholar] [CrossRef]
- Boorjian, S.A.; Alemozaffar, M.; Konety, B.R.; Shore, N.D.; Gomella, L.G.; Kamat, A.M.; Bivalacqua, T.J.; Montgomery, J.S.; Lerner, S.P.; Busby, J.E.; et al. Intravesical Nadofaragene Firadenovec Gene Therapy for BCG-Unresponsive Non-Muscle-Invasive Bladder Cancer: A Single-Arm, Open-Label, Repeat-Dose Clinical Trial. Lancet Oncol. 2021, 22, 107–117. [Google Scholar] [CrossRef]
- Nemunaitis, J. Vaccines in Cancer: GVAX®, a GM-CSF Gene Vaccine. Expert Rev. Vaccines 2005, 4, 259–274. [Google Scholar] [CrossRef]
- Oosterhoff, D.; Sluijter, B.J.R.; Hangalapura, B.N.; de Gruijl, T.D. The Dermis as a Portal for Dendritic Cell-Targeted Immunotherapy of Cutaneous Melanoma. In Intradermal Immunization; Current Topics in Microbiology and Immunology; Teunissen, M.B.M., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; Volume 351, pp. 181–220. ISBN 978-3-642-23689-1. [Google Scholar]
- Butterfield, L.H.; Comin-Anduix, B.; Vujanovic, L.; Lee, Y.; Dissette, V.B.; Yang, J.-Q.; Vu, H.T.; Seja, E.; Oseguera, D.K.; Potter, D.M.; et al. Adenovirus MART-1–Engineered Autologous Dendritic Cell Vaccine for Metastatic Melanoma: J. Immunother. 2008, 31, 294–309. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Lee, M.-H.; Garon, E.; Goldman, J.W.; Salehi-Rad, R.; Baratelli, F.E.; Schaue, D.; Wang, G.; Rosen, F.; Yanagawa, J.; et al. Phase I Trial of Intratumoral Injection of CCL21 Gene–Modified Dendritic Cells in Lung Cancer Elicits Tumor-Specific Immune Responses and CD8 + T-Cell Infiltration. Clin. Cancer Res. 2017, 23, 4556–4568. [Google Scholar] [CrossRef]
- Chiappori, A.A.; Soliman, H.; Janssen, W.E.; Antonia, S.J.; Gabrilovich, D.I. INGN-225: A Dendritic Cell-Based P53 Vaccine (Ad.P53-DC) in Small Cell Lung Cancer: Observed Association between Immune Response and Enhanced Chemotherapy Effect. Expert Opin. Biol. Ther. 2010, 10, 983–991. [Google Scholar] [CrossRef]
- Wang, D.; Huang, X.F.; Hong, B.; Song, X.-T.; Hu, L.; Jiang, M.; Zhang, B.; Ning, H.; Li, Y.; Xu, C.; et al. Efficacy of Intracellular Immune Checkpoint-Silenced DC Vaccine. JCI Insight 2018, 3, e98368. [Google Scholar] [CrossRef]
- Lu, Y.-C.; Robbins, P.F. Cancer Immunotherapy Targeting Neoantigens. Semin. Immunol. 2016, 28, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Gubin, M.M.; Artyomov, M.N.; Mardis, E.R.; Schreiber, R.D. Tumor Neoantigens: Building a Framework for Personalized Cancer Immunotherapy. J. Clin. Investig. 2015, 125, 3413–3421. [Google Scholar] [CrossRef]
- Basak, S.K.; Kiertscher, S.M.; Harui, A.; Roth, M.D. Modifying Adenoviral Vectors for Use as Gene-Based Cancer Vaccines. Viral Immunol. 2004, 17, 182–196. [Google Scholar] [CrossRef]
- Goyvaerts, C.; Breckpot, K. The Journey of in Vivo Virus Engineered Dendritic Cells from Bench to Bedside: A Bumpy Road. Front. Immunol. 2018, 9, 2052. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Kumar, R.; Agrawal, B. Adenoviral Vector-Based Vaccines and Gene Therapies: Current Status and Future Prospects. In Adenoviruses; Desheva, Y., Ed.; IntechOpen: London, UK, 2019; ISBN 978-1-78984-990-5. [Google Scholar]
- Short, J.J.; Vasu, C.; Holterman, M.J.; Curiel, D.T.; Pereboev, A. Members of Adenovirus Species B Utilize CD80 and CD86 as Cellular Attachment Receptors. Virus Res. 2006, 122, 144–153. [Google Scholar] [CrossRef]
- Gaggar, A.; Shayakhmetov, D.M.; Lieber, A. CD46 Is a Cellular Receptor for Group B Adenoviruses. Nat. Med. 2003, 9, 1408–1412. [Google Scholar] [CrossRef] [PubMed]
- Lyle, C.; McCormick, F. Integrin Avβ5 Is a Primary Receptor for Adenovirus in CAR-Negative Cells. Virol. J. 2010, 7, 148. [Google Scholar] [CrossRef]
- Nemerow, G.; Flint, J. Lessons Learned from Adenovirus (1970–2019). FEBS Lett. 2019, 593, 3395–3418. [Google Scholar] [CrossRef]
- Zhang, Y.; Bergelson, J.M. Adenovirus Receptors. J. Virol. 2005, 79, 12125–12131. [Google Scholar] [CrossRef]
- Gao, J.; Zhang, W.; Ehrhardt, A. Expanding the Spectrum of Adenoviral Vectors for Cancer Therapy. Cancers 2020, 12, 1139. [Google Scholar] [CrossRef]
- Moyer, C.L.; Wiethoff, C.M.; Maier, O.; Smith, J.G.; Nemerow, G.R. Functional Genetic and Biophysical Analyses of Membrane Disruption by Human Adenovirus. J. Virol. 2011, 85, 2631–2641. [Google Scholar] [CrossRef]
- Stasiak, A.C.; Stehle, T. Human Adenovirus Binding to Host Cell Receptors: A Structural View. Med. Microbiol. Immunol. 2020, 209, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Meier, O.; Greber, U.F. Adenovirus Endocytosis. J. Gene Med. 2004, 6, S152–S163. [Google Scholar] [CrossRef] [PubMed]
- Bergelson, J.M. Isolation of a Common Receptor for Coxsackie B Viruses and Adenoviruses 2 and 5. Science 1997, 275, 1320–1323. [Google Scholar] [CrossRef]
- Thomas, C.E.; Edwards, P.; Wickham, T.J.; Castro, M.G.; Lowenstein, P.R. Adenovirus Binding to the Coxsackievirus and Adenovirus Receptor or Integrins Is Not Required to Elicit Brain Inflammation but Is Necessary to Transduce Specific Neural Cell Types. J. Virol. 2002, 76, 3452–3460. [Google Scholar] [CrossRef]
- Zhang, N.-H.; Peng, R.-Q.; Ding, Y.; Zhang, X.-S. Rejection of Adenovirus Infection Is Independent of Coxsackie and Adenovirus Receptor Expression in Cisplatin-Resistant Human Lung Cancer Cells. Oncol. Rep. 2016, 36, 715–720. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Persson, B.D.; Lenman, A.; Frängsmyr, L.; Schmid, M.; Ahlm, C.; Plückthun, A.; Jenssen, H.; Arnberg, N. Lactoferrin-Hexon Interactions Mediate CAR-Independent Adenovirus Infection of Human Respiratory Cells. J. Virol. 2020, 94, e00542-20. [Google Scholar] [CrossRef] [PubMed]
- Bots, S.T.F.; Hoeben, R.C. Non-Human Primate-Derived Adenoviruses for Future Use as Oncolytic Agents? Int. J. Mol. Sci. 2020, 21, 4821. [Google Scholar] [CrossRef] [PubMed]
- Mennechet, F.J.D.; Paris, O.; Ouoba, A.R.; Salazar Arenas, S.; Sirima, S.B.; Takoudjou Dzomo, G.R.; Diarra, A.; Traore, I.T.; Kania, D.; Eichholz, K.; et al. A Review of 65 Years of Human Adenovirus Seroprevalence. Expert Rev. Vaccines 2019, 18, 597–613. [Google Scholar] [CrossRef]
- Lin, K.-H.; Lin, Y.-C.; Chen, H.-L.; Ke, G.-M.; Chiang, C.-J.; Hwang, K.-P.; Chu, P.-Y.; Lin, J.-H.; Liu, D.-P.; Chen, H.-Y. A Two Decade Survey of Respiratory Adenovirus in Taiwan: The Reemergence of Adenovirus Types 7 and 4. J. Med. Virol. 2004, 73, 274–279. [Google Scholar] [CrossRef]
- Sumida, S.M.; Truitt, D.M.; Lemckert, A.A.C.; Vogels, R.; Custers, J.H.H.V.; Addo, M.M.; Lockman, S.; Peter, T.; Peyerl, F.W.; Kishko, M.G.; et al. Neutralizing Antibodies to Adenovirus Serotype 5 Vaccine Vectors Are Directed Primarily against the Adenovirus Hexon Protein. J. Immunol. 2005, 174, 7179–7185. [Google Scholar] [CrossRef]
- Shiver, J.W.; Emini, E.A. Recent Advances in the Development of HIV-1 Vaccines Using Replication-Incompetent Adenovirus Vectors. Annu. Rev. Med. 2004, 55, 355–372. [Google Scholar] [CrossRef]
- Abbink, P.; Lemckert, A.A.C.; Ewald, B.A.; Lynch, D.M.; Denholtz, M.; Smits, S.; Holterman, L.; Damen, I.; Vogels, R.; Thorner, A.R.; et al. Comparative Seroprevalence and Immunogenicity of Six Rare Serotype Recombinant Adenovirus Vaccine Vectors from Subgroups B and D. J. Virol. 2007, 81, 4654–4663. [Google Scholar] [CrossRef]
- Abbas, K.Z.; Lombos, E.; Duvvuri, V.R.; Olsha, R.; Higgins, R.R.; Gubbay, J.B. Temporal Changes in Respiratory Adenovirus Serotypes Circulating in the Greater Toronto Area, Ontario, during December 2008 to April 2010. Virol. J. 2013, 10, 15. [Google Scholar] [CrossRef] [PubMed]
- Heim, A.; Ebnet, C.; Harste, G.; Pring-Åkerblom, P. Rapid and Quantitative Detection of Human Adenovirus DNA by Real-Time PCR. J. Med. Virol. 2003, 70, 228–239. [Google Scholar] [CrossRef]
- Lee, W.-J.; Jung, H.-D.; Cheong, H.-M.; Kim, K. Molecular Epidemiology of a Post-Influenza Pandemic Outbreak of Acute Respiratory Infections in Korea Caused by Human Adenovirus Type 3: Post-Influenza Pandemic Outbreak of Human Adenovirus Type 3. J. Med. Virol. 2015, 87, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Scott, M.K.; Chommanard, C.; Lu, X.; Appelgate, D.; Grenz, L.; Schneider, E.; Gerber, S.I.; Erdman, D.D.; Thomas, A. Human Adenovirus Associated with Severe Respiratory Infection, Oregon, USA, 2013–2014. Emerg. Infect. Dis. 2016, 22, 1044–1051. [Google Scholar] [CrossRef]
- Richmond, S.; Burman, R.; Crosdale, E.; Cropper, L.; Longson, D.; Enoch, B.E.; Dodd, C.L. A Large Outbreak of Keratoconjunctivitis Due to Adenovirus Type 8. J. Hyg. 1984, 93, 285–291. [Google Scholar] [CrossRef]
- Tanaka-Yokogui, K.; Itoh, N.; Usui, N.; Takeuchi, S.; Uchio, E.; Aoki, K.; Usui, M.; Ohno, S. New Genome Type of Adenovirus Serotype 19 Causing Nosocomial Infections of Epidemic Keratoconjunctivitis in Japan. J. Med. Virol. 2001, 65, 530–533. [Google Scholar] [CrossRef] [PubMed]
- Jernigan, J.A.; Lowry, B.S.; Hayden, F.G.; Kyger, S.A.; Conway, B.P.; Groschel, D.H.M.; Farr, B.M. Adenovirus Type 8 Epidemic Keratoconjunctivitis in an Eye Clinic: Risk Factors and Control. J. Infect. Dis. 1993, 167, 1307–1313. [Google Scholar] [CrossRef]
- Aronson, B.; Aronson, S.; Sobel, G.; Walker, D. Pharyngoconjunctival Fever; Report of an Epidemic Outbreak. AMA J. Dis. Child. 1956, 92, 596–612. [Google Scholar]
- Harley, D.; Harrower, B.; Lyon, M.; Dick, A. A Primary School Outbreak of Pharyngoconjunctival Fever Caused by Adenovirus Type 3. Commun. Dis. Intell. 2001, 25, 9–12. [Google Scholar] [PubMed]
- Qiu, F.; Shen, X.; Li, G.; Zhao, L.; Chen, C.; Duan, S.; Guo, J.; Zhao, M.; Yan, T.; Qi, J.-J.; et al. Adenovirus Associated with Acute Diarrhea: A Case-Control Study. BMC Infect. Dis. 2018, 18, 450. [Google Scholar] [CrossRef]
- Uhnoo, I.; Wadell, G.; Svensson, L.; Johansson, M.E. Importance of Enteric Adenoviruses 40 and 41 in Acute Gastroenteritis in Infants and Young Children. J. Clin. Microbiol. 1984, 20, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Mitra, A.K.; Clarke, K. Viral Obesity: Fact or Fiction? Obes. Rev. 2010, 11, 289–296. [Google Scholar] [CrossRef]
- Shang, Q.; Wang, H.; Song, Y.; Wei, L.; Lavebratt, C.; Zhang, F.; Gu, H. Serological Data Analyses Show That Adenovirus 36 Infection Is Associated with Obesity: A Meta-Analysis Involving 5739 Subjects: Ad36 Associated with Obesity by Meta-Analysis. Obesity 2014, 22, 895–900. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, K.L.; Richardson, S.E.; MacGregor, D.; Mahant, S.; Raghuram, K.; Bitnun, A. Adenovirus-Associated Central Nervous System Disease in Children. J. Pediatr. 2019, 205, 130–137. [Google Scholar] [CrossRef]
- Xie, L.; Zhang, B.; Xiao, N.; Zhang, F.; Zhao, X.; Liu, Q.; Xie, Z.; Gao, H.; Duan, Z.; Zhong, L. Epidemiology of Human Adenovirus Infection in Children Hospitalized with Lower Respiratory Tract Infections in Hunan, China: XIE ET AL. J. Med. Virol. 2019, 91, 392–400. [Google Scholar] [CrossRef]
- Lai, C.-Y.; Lee, C.-J.; Lu, C.-Y.; Lee, P.-I.; Shao, P.-L.; Wu, E.-T.; Wang, C.-C.; Tan, B.-F.; Chang, H.-Y.; Hsia, S.-H.; et al. Adenovirus Serotype 3 and 7 Infection with Acute Respiratory Failure in Children in Taiwan, 2010–2011. PLoS ONE 2013, 8, e53614. [Google Scholar] [CrossRef]
- Binder, A.M.; Biggs, H.M.; Haynes, A.K.; Chommanard, C.; Lu, X.; Erdman, D.D.; Watson, J.T.; Gerber, S.I. Human Adenovirus Surveillance—United States, 2003–2016. MMWR Morb. Mortal. Wkly. Rep. 2017, 66, 1039–1042. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Hernández, A.M.; Duquesroix, B.; Benítez-del-Castillo, J.M. ADenoVirus Initiative Study in Epidemiology (ADVISE): Resultados de un estudio epidemiológico multicéntrico en España. Arch. Soc. Esp. Oftalmol. 2018, 93, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Mayindou, G.; Ngokana, B.; Sidibé, A.; Moundélé, V.; Koukouikila-Koussounda, F.; Christevy Vouvoungui, J.; Kwedi Nolna, S.; Velavan, T.P.; Ntoumi, F. Molecular Epidemiology and Surveillance of Circulating Rotavirus and Adenovirus in Congolese Children with Gastroenteritis: Rotavirus and Adenovirus in Congolese Children. J. Med. Virol. 2016, 88, 596–605. [Google Scholar] [CrossRef] [PubMed]
- Krause, A.; Joh, J.H.; Hackett, N.R.; Roelvink, P.W.; Bruder, J.T.; Wickham, T.J.; Kovesdi, I.; Crystal, R.G.; Worgall, S. Epitopes Expressed in Different Adenovirus Capsid Proteins Induce Different Levels of Epitope-Specific Immunity. J. Virol. 2006, 80, 5523–5530. [Google Scholar] [CrossRef] [PubMed]
- Bradley, R.R.; Maxfield, L.F.; Lynch, D.M.; Iampietro, M.J.; Borducchi, E.N.; Barouch, D.H. Adenovirus Serotype 5-Specific Neutralizing Antibodies Target Multiple Hexon Hypervariable Regions. J. Virol. 2012, 86, 1267–1272. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.G.; Cassany, A.; Gerace, L.; Ralston, R.; Nemerow, G.R. Neutralizing Antibody Blocks Adenovirus Infection by Arresting Microtubule-Dependent Cytoplasmic Transport. J. Virol. 2008, 82, 6492–6500. [Google Scholar] [CrossRef]
- Duffy, M.R.; Doszpoly, A.; Turner, G.; Nicklin, S.A.; Baker, A.H. The Relevance of Coagulation Factor X Protection of Adenoviruses in Human Sera. Gene Ther. 2016, 23, 592–596. [Google Scholar] [CrossRef]
- Xu, Z.; Qiu, Q.; Tian, J.; Smith, J.S.; Conenello, G.M.; Morita, T.; Byrnes, A.P. Coagulation Factor X Shields Adenovirus Type 5 from Attack by Natural Antibodies and Complement. Nat. Med. 2013, 19, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Tomita, K.; Sakurai, F.; Iizuka, S.; Hemmi, M.; Wakabayashi, K.; Machitani, M.; Tachibana, M.; Katayama, K.; Kamada, H.; Mizuguchi, H. Antibodies against Adenovirus Fiber and Penton Base Proteins Inhibit Adenovirus Vector-Mediated Transduction in the Liver Following Systemic Administration. Sci. Rep. 2018, 8, 12315. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Dong, J.; Wang, C.; Zhan, Y.; Zhang, H.; Wu, J.; Kong, W.; Yu, X. Characteristics of Neutralizing Antibodies to Adenovirus Capsid Proteins in Human and Animal Sera. Virology 2013, 437, 118–123. [Google Scholar] [CrossRef]
- Yang, T.C.; Dayball, K.; Wan, Y.H.; Bramson, J. Detailed Analysis of the CD8+ T-Cell Response Following Adenovirus Vaccination. J. Virol. 2003, 77, 13407–13411. [Google Scholar] [CrossRef] [PubMed]
- Olive, M.; Eisenlohr, L.; Flomenberg, N.; Hsu, S.; Flomenberg, P. The Adenovirus Capsid Protein Hexon Contains a Highly Conserved Human CD4 + T-Cell Epitope. Hum. Gene Ther. 2002, 13, 1167–1178. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, M.D.; Yamashina, S.; Froh, M.; Rusyn, I.; Thurman, R.G. Adenoviral Gene Delivery Can Inactivate Kupffer Cells: Role of Oxidants in NF-κB Activation and Cytokine Production. J. Leukoc. Biol. 2011, 69, 622–630. [Google Scholar]
- Khare, R.; Chen, Y.C.; Weaver, A.E.; Barry, A.M. Advances and Future Challenges in Adenoviral Vector Pharmacology and Targeting. Curr. Gene Ther. 2011, 11, 241–258. [Google Scholar] [CrossRef]
- Green, N.K.; Herbert, C.W.; Hale, S.J.; Hale, A.B.; Mautner, V.; Harkins, R.; Hermiston, T.; Ulbrich, K.; Fisher, K.D.; Seymour, L.W. Extended Plasma Circulation Time and Decreased Toxicity of Polymer-Coated Adenovirus. Gene Ther. 2004, 11, 1256–1263. [Google Scholar] [CrossRef]
- Barr, D.; Tubb, J.; Ferguson, D.; Scaria, A.; Lieber, A.; Wilson, C.; Perkins, J.; Kay, M.A. Strain Related Variations in Adenovirally Mediated Transgene Expression from Mouse Hepatocytes in Vivo: Comparisons between Immunocompetent and Immunodeficient Inbred Strains. Gene Ther. 1995, 2, 151–155. [Google Scholar]
- Yang, Y.; Jooss, K.U.; Su, Q.; Ertl, H.C.; Wilson, J.M. Immune Responses to Viral Antigens versus Transgene Product in the Elimination of Recombinant Adenovirus-Infected Hepatocytes in Vivo. Gene Ther. 1996, 3, 137–144. [Google Scholar]
- Chen, P.; Kovesdi, I.; Bruder, J.T. Effective Repeat Administration with Adenovirus Vectors to the Muscle. Gene Ther. 2000, 7, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Holst, P.J.; Ørskov, C.; Thomsen, A.R.; Christensen, J.P. Quality of the Transgene-Specific CD8 + T Cell Response Induced by Adenoviral Vector Immunization Is Critically Influenced by Virus Dose and Route of Vaccination. J. Immunol. 2010, 184, 4431–4439. [Google Scholar] [CrossRef] [PubMed]
- Núñez-Ochoa, L.; Madrid-Marina, V.; Gutiérrez-López, A. Evaluation of Adverse Events in Dogs with Adenoviral Therapy by Intralymphonodal Administration in Canine Spontaneous Multicentric Lymphosarcoma. Clin. Oncol. 2017, 2, 1–8. [Google Scholar]
- Yamamoto, Y.; Nagasato, M.; Yoshida, T.; Aoki, K. Recent Advances in Genetic Modification of Adenovirus Vectors for Cancer Treatment. Cancer Sci. 2017, 108, 831–837. [Google Scholar] [CrossRef] [PubMed]
- Beatty, M.S.; Curiel, D.T. Adenovirus Strategies for Tissue-Specific Targeting. In Advances in Cancer Research; Elsevier: Amsterdam, The Netherlands, 2012; Volume 115, pp. 39–67. ISBN 978-0-12-398342-8. [Google Scholar]
- Dmitriev, I.; Krasnykh, V.; Miller, C.R.; Wang, M.; Kashentseva, E.; Mikheeva, G.; Belousova, N.; Curiel, D.T. An Adenovirus Vector with Genetically Modified Fibers Demonstrates Expanded Tropism via Utilization of a Coxsackievirus and Adenovirus Receptor-Independent Cell Entry Mechanism. J. Virol. 1998, 72, 9706–9713. [Google Scholar] [CrossRef]
- Wickham, T.J.; Roelvink, P.W.; Brough, D.E.; Kovesdi, I. Adenovirus Targeted to Heparan-Containing Receptors Increases Its Gene Delivery Efficiency to Multiple Cell Types. Nat. Biotechnol. 1996, 14, 1570–1573. [Google Scholar] [CrossRef] [PubMed]
- Blackhall, F.H.; Merry, C.L.R.; Davies, E.J.; Jayson, G.C. Heparan Sulfate Proteoglycans and Cancer. Br. J. Cancer 2001, 85, 1094–1098. [Google Scholar] [CrossRef] [PubMed]
- Wickham, T.J.; Segal, D.M.; Roelvink, P.W.; Carrion, M.E.; Lizonova, A.; Lee, G.M.; Kovesdi, I. Targeted Adenovirus Gene Transfer to Endothelial and Smooth Muscle Cells by Using Bispecific Antibodies. J. Virol. 1996, 70, 6831–6838. [Google Scholar] [CrossRef]
- Miura, Y.; Yoshida, K.; Nishimoto, T.; Hatanaka, K.; Ohnami, S.; Asaka, M.; Douglas, J.T.; Curiel, D.T.; Yoshida, T.; Aoki, K. Direct Selection of Targeted Adenovirus Vectors by Random Peptide Display on the Fiber Knob. Gene Ther. 2007, 14, 1448–1460. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Sadata, A.; Zhang, W.; Saito, K.; Shinoura, N.; Hamada, H. Generation of Fiber-Mutant Recombinant Adenoviruses for Gene Therapy of Malignant Glioma. Hum. Gene Ther. 1998, 9, 2503–2515. [Google Scholar] [CrossRef]
- Krasnykh, V.; Dmitriev, I.; Mikheeva, G.; Miller, C.R.; Belousova, N.; Curiel, D.T. Characterization of an Adenovirus Vector Containing a Heterologous Peptide Epitope in the HI Loop of the Fiber Knob. J. Virol. 1998, 72, 1844–1852. [Google Scholar] [CrossRef]
- Nicklin, S.A.; Von Seggern, D.J.; Work, L.M.; Pek, D.C.K.; Dominiczak, A.F.; Nemerow, G.R.; Baker, A.H. Ablating Adenovirus Type 5 Fiber–CAR Binding and HI Loop Insertion of the SIGYPLP Peptide Generate an Endothelial Cell-Selective Adenovirus. Mol. Ther. 2001, 4, 534–542. [Google Scholar] [CrossRef]
- Nicklin, S.A.; White, S.J.; Nicol, C.G.; Von Seggern, D.J.; Baker, A.H. In Vitro Andin Vivo Characterisation of Endothelial Cell Selective Adenoviral Vectors. J. Gene Med. 2004, 6, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Joung, I.; Harber, G.; Gerecke, K.M.; Carroll, S.L.; Collawn, J.F.; Engler, J.A. Improved Gene Delivery into Neuroglial Cells Using a Fiber-Modified Adenovirus Vector. Biochem. Biophys. Res. Commun. 2005, 328, 1182–1187. [Google Scholar] [CrossRef] [PubMed]
- Miura, Y.; Yamasaki, S.; Davydova, J.; Brown, E.; Aoki, K.; Vickers, S.; Yamamoto, M. Infectivity-Selective Oncolytic Adenovirus Developed by High-Throughput Screening of Adenovirus-Formatted Library. Mol. Ther. 2013, 21, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Müller, O.J.; Kaul, F.; Weitzman, M.D.; Pasqualini, R.; Arap, W.; Kleinschmidt, J.A.; Trepel, M. Random Peptide Libraries Displayed on Adeno-Associated Virus to Select for Targeted Gene Therapy Vectors. Nat. Biotechnol. 2003, 21, 1040–1046. [Google Scholar] [CrossRef] [PubMed]
- Bhat, N.R.; Fan, F. Adenovirus Infection Induces Microglial Activation: Involvement of Mitogen-Activated Protein Kinase Pathways. Brain Res. 2002, 948, 93–101. [Google Scholar] [CrossRef]
- Tibbles, L.A.; Spurrell, J.C.L.; Bowen, G.P.; Liu, Q.; Lam, M.; Zaiss, A.K.; Robbins, S.M.; Hollenberg, M.D.; Wickham, T.J.; Muruve, D.A. Activation of P38 and ERK Signaling during Adenovirus Vector Cell Entry Lead to Expression of the C-X-C Chemokine IP-10. J. Virol. 2002, 76, 1559–1568. [Google Scholar] [CrossRef]
- Li, E.; Stupack, D.; Bokoch, G.M.; Nemerow, G.R. Adenovirus Endocytosis Requires Actin Cytoskeleton Reorganization Mediated by Rho Family GTPases. J. Virol. 1998, 72, 8806–8812. [Google Scholar] [CrossRef]
- Wickham, T.J.; Mathias, P.; Cheresh, D.A.; Nemerow, G.R. Integrins Avβ3 and Avβ5 Promote Adenovirus Internalization but Not Virus Attachment. Cell 1993, 73, 309–319. [Google Scholar] [CrossRef]
- Ranki, T.; Hemminki, A. Serotype Chimeric Human Adenoviruses for Cancer GeneTherapy. Viruses 2010, 2, 2196–2212. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, E.; Havenga, M.J.; Ophorst, O.; de Leeuw, B.; Gijsbers, L.; Gillissen, G.; Hoeben, R.C.; ter Horst, M.; Nanda, D.; Dirven, C.; et al. Human Adenovirus Type 35 Vector for Gene Therapy of Brain Cancer: Improved Transduction and Bypass of Pre-Existing Anti-Vector Immunity in Cancer Patients. Cancer Gene Ther. 2007, 14, 211–219. [Google Scholar] [CrossRef]
- Kanerva, A.; Mikheeva, G.V.; Krasnykh, V.; Coolidge, C.J.; Lam, J.T.; Mahasreshti, P.J.; Shannon, D.B.; Barker, S.D. Targeting Adenovirus to the Serotype 3 Receptor Increases Gene Transfer Efficiency to Ovarian Cancer Cells. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2002, 8, 275–280. [Google Scholar]
- Sarkioja, M.; Kanerva, A.; Salo, J.; Kangasniemi, L.; Eriksson, M.; Raki, M.; Ranki, T.; Hakkarainen, T.; Hemminki, A. Noninvasive Imaging for Evaluation of the Systemic Delivery of Capsid-Modified Adenoviruses in an Orthotopic Model of Advanced Lung Cancer. Cancer 2006, 107, 1578–1588. [Google Scholar] [CrossRef]
- Diaconu, I.; Denby, L.; Pesonen, S.; Cerullo, V.; Bauerschmitz, G.J.; Guse, K.; Rajecki, M.; Dias, J.D.; Taari, K.; Kanerva, A.; et al. Serotype Chimeric and Fiber-Mutated Adenovirus Ad5/19p-HIT for Targeting Renal Cancer and Untargeting the Liver. Hum. Gene Ther. 2009, 20, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.T.; Davies, J.A.; Bates, E.A.; Moses, E.; Mundy, R.M.; Marlow, G.; Cole, D.K.; Bliss, C.M.; Rizkallah, P.J.; Parker, A.L. The Fiber Knob Protein of Human Adenovirus Type 49 Mediates Highly Efficient and Promiscuous Infection of Cancer Cell Lines Using a Novel Cell Entry Mechanism. J. Virol. 2020, 95, e01849-20. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Jin, C.; Ramachandran, M.; Xu, J.; Nilsson, B.; Korsgren, O.; Le Blanc, K.; Uhrbom, L.; Forsberg-Nilsson, K.; Westermark, B.; et al. Adenovirus Serotype 5 Vectors with Tat-PTD Modified Hexon and Serotype 35 Fiber Show Greatly Enhanced Transduction Capacity of Primary Cell Cultures. PLoS ONE 2013, 8, e54952. [Google Scholar] [CrossRef] [PubMed]
- Ishida, T.; Harashima, H.; Kiwada, H. Liposome Clearance. Biosci. Rep. 2002, 22, 197–224. [Google Scholar] [CrossRef]
- Singh, R.; Kostarelos, K. Designer Adenoviruses for Nanomedicine and Nanodiagnostics. Trends Biotechnol. 2009, 27, 220–229. [Google Scholar] [CrossRef]
- Fisher, K.D.; Green, N.K.; Hale, A.; Subr, V.; Ulbrich, K.; Seymour, L.W. Passive Tumour Targeting of Polymer-Coated Adenovirus for Cancer Gene Therapy. J. Drug Target. 2007, 15, 546–551. [Google Scholar] [CrossRef]
- Kreppel, F.; Kochanek, S. Modification of Adenovirus Gene Transfer Vectors With Synthetic Polymers: A Scientific Review and Technical Guide. Mol. Ther. 2008, 16, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.M.; Chess, R.B. Effect of Pegylation on Pharmaceuticals. Nat. Rev. Drug Discov. 2003, 2, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Eto, Y.; Yoshioka, Y.; Mukai, Y.; Okada, N.; Nakagawa, S. Development of PEGylated Adenovirus Vector with Targeting Ligand. Int. J. Pharm. 2008, 354, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Fontana, F.; Shahbazi, M.-A.; Liu, D.; Zhang, H.; Mäkilä, E.; Salonen, J.; Hirvonen, J.T.; Santos, H.A. Multistaged Nanovaccines Based on Porous Silicon@Acetalated Dextran@Cancer Cell Membrane for Cancer Immunotherapy. Adv. Mater. 2017, 29, 1603239. [Google Scholar] [CrossRef]
- Li, R.; He, Y.; Zhang, S.; Qin, J.; Wang, J. Cell Membrane-Based Nanoparticles: A New Biomimetic Platform for Tumor Diagnosis and Treatment. Acta Pharm. Sin. B 2018, 8, 14–22. [Google Scholar] [CrossRef]
- Yang, F.; Shi, K.; Jia, Y.; Hao, Y.; Peng, J.; Qian, Z. Advanced Biomaterials for Cancer Immunotherapy. Acta Pharmacol. Sin. 2020, 41, 911–927. [Google Scholar] [CrossRef]
- Kerkar, S.P.; Restifo, N.P. Cellular Constituents of Immune Escape within the Tumor Microenvironment: Figure 1. Cancer Res. 2012, 72, 3125–3130. [Google Scholar] [CrossRef]
- Devaud, C.; John, L.B.; Westwood, J.A.; Darcy, P.K.; Kershaw, M.H. Immune Modulation of the Tumor Microenvironment for Enhancing Cancer Immunotherapy. OncoImmunology 2013, 2, e25961. [Google Scholar] [CrossRef]
- Zhuang, J.; Holay, M.; Park, J.H.; Fang, R.H.; Zhang, J.; Zhang, L. Nanoparticle Delivery of Immunostimulatory Agents for Cancer Immunotherapy. Theranostics 2019, 9, 7826–7848. [Google Scholar] [CrossRef]
- Fang, R.H.; Hu, C.-M.J.; Luk, B.T.; Gao, W.; Copp, J.A.; Tai, Y.; O’Connor, D.E.; Zhang, L. Cancer Cell Membrane-Coated Nanoparticles for Anticancer Vaccination and Drug Delivery. Nano Lett. 2014, 14, 2181–2188. [Google Scholar] [CrossRef] [PubMed]
- Fontana, F.; Fusciello, M.; Groeneveldt, C.; Capasso, C.; Chiaro, J.; Feola, S.; Liu, Z.; Mäkilä, E.M.; Salonen, J.J.; Hirvonen, J.T.; et al. Biohybrid Vaccines for Improved Treatment of Aggressive Melanoma with Checkpoint Inhibitor. ACS Nano 2019, 13, 6477–6490. [Google Scholar] [CrossRef] [PubMed]
- Kroll, A.V.; Fang, R.H.; Jiang, Y.; Zhou, J.; Wei, X.; Yu, C.L.; Gao, J.; Luk, B.T.; Dehaini, D.; Gao, W.; et al. Nanoparticulate Delivery of Cancer Cell Membrane Elicits Multiantigenic Antitumor Immunity. Adv. Mater. 2017, 29, 1703969. [Google Scholar] [CrossRef]
- Fusciello, M.; Fontana, F.; Tähtinen, S.; Capasso, C.; Feola, S.; Martins, B.; Chiaro, J.; Peltonen, K.; Ylösmäki, L.; Ylösmäki, E.; et al. Artificially Cloaked Viral Nanovaccine for Cancer Immunotherapy. Nat. Commun. 2019, 10, 5747. [Google Scholar] [CrossRef] [PubMed]
- van Erp, E.A.; Kaliberova, L.N.; Kaliberov, S.A.; Curiel, D.T. Retargeted Oncolytic Adenovirus Displaying a Single Variable Domain of Camelid Heavy-Chain-Only Antibody in a Fiber Protein. Mol. Ther. Oncolytics 2015, 2, 15001. [Google Scholar] [CrossRef]
- Bhatia, S.; O’Bryan, S.M.; Rivera, A.A.; Curiel, D.T.; Mathis, J.M. CXCL12 Retargeting of an Adenovirus Vector to Cancer Cells Using a Bispecific Adapter. Oncolytic Virother. 2016, 5, 99–113. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, B.; Mahalingam, M. The CXCR4/CXCL12 Axis in Cutaneous Malignancies with an Emphasis on Melanoma. Histol. Histopathol. 2014, 1539–1546. [Google Scholar] [CrossRef]
- Zhang, Z.; Ni, C.; Chen, W.; Wu, P.; Wang, Z.; Yin, J.; Huang, J.; Qiu, F. Expression of CXCR4 and Breast Cancer Prognosis: A Systematic Review and Meta-Analysis. BMC Cancer 2014, 14, 49. [Google Scholar] [CrossRef]
- Schmid, M.; Ernst, P.; Honegger, A.; Suomalainen, M.; Zimmermann, M.; Braun, L.; Stauffer, S.; Thom, C.; Dreier, B.; Eibauer, M.; et al. Adenoviral Vector with Shield and Adapter Increases Tumor Specificity and Escapes Liver and Immune Control. Nat. Commun. 2018, 9, 450. [Google Scholar] [CrossRef]
- Stumpp, M.T.; Binz, H.K.; Amstutz, P. DARPins: A New Generation of Protein Therapeutics. Drug Discov. Today 2008, 13, 695–701. [Google Scholar] [CrossRef]
- Dreier, B.; Honegger, A.; Hess, C.; Nagy-Davidescu, G.; Mittl, P.R.E.; Grutter, M.G.; Belousova, N.; Mikheeva, G.; Krasnykh, V.; Pluckthun, A. Development of a Generic Adenovirus Delivery System Based on Structure-Guided Design of Bispecific Trimeric DARPin Adapters. Proc. Natl. Acad. Sci. USA 2013, 110, E869–E877. [Google Scholar] [CrossRef]
- Einfeld, D.A.; Schroeder, R.; Roelvink, P.W.; Lizonova, A.; King, C.R.; Kovesdi, I.; Wickham, T.J. Reducing the Native Tropism of Adenovirus Vectors Requires Removal of Both CAR and Integrin Interactions. J. Virol. 2001, 75, 11284–11291. [Google Scholar] [CrossRef]
- Koizumi, N.; Mizuguchi, H.; Sakurai, F.; Yamaguchi, T.; Watanabe, Y.; Hayakawa, T. Reduction of Natural Adenovirus Tropism to Mouse Liverby Fiber-Shaft Exchange in Combination with Both CAR- Andαv Integrin-BindingAblation. J. Virol. 2003, 77, 13062–13072. [Google Scholar] [CrossRef]
- Yao, X.-L.; Yoshioka, Y.; Ruan, G.-X.; Chen, Y.-Z.; Mizuguchi, H.; Mukai, Y.; Okada, N.; Gao, J.-Q.; Nakagawa, S. Optimization and Internalization Mechanisms of PEGylated Adenovirus Vector with Targeting Peptide for Cancer Gene Therapy. Biomacromolecules 2012, 13, 2402–2409. [Google Scholar] [CrossRef]
- Black, P.C.; Agarwal, P.K.; Dinney, C.P.N. Targeted Therapies in Bladder Cancer—An Update. Urol. Oncol. Semin. Orig. Investig. 2007, 25, 433–438. [Google Scholar] [CrossRef]
- Bonsted, A.; Engesæter, B.Ø.; Høgset, A.; Mælandsmo, G.M.; Prasmickaite, L.; D’Oliveira, C.; Hennink, W.E.; van Steenis, J.H.; Berg, K. Photochemically Enhanced Transduction of Polymer-Complexed Adenovirus Targeted to the Epidermal Growth Factor Receptor. J. Gene Med. 2006, 8, 286–297. [Google Scholar] [CrossRef] [PubMed]
- Vetter, A.; Virdi, K.S.; Espenlaub, S.; Rödl, W.; Wagner, E.; Holm, P.S.; Scheu, C.; Kreppel, F.; Spitzweg, C.; Ogris, M. Adenoviral Vectors Coated with PAMAM Dendrimer Conjugates Allow CAR Independent Virus Uptake and Targeting to the EGF Receptor. Mol. Pharm. 2013, 10, 606–618. [Google Scholar] [CrossRef]
- Kreppel, F.; Gackowski, J.; Schmidt, E.; Kochanek, S. Combined Genetic and Chemical Capsid Modifications Enable Flexible and Efficient De- and Retargeting of Adenovirus Vectors. Mol. Ther. 2005, 12, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Corjon, S.; Wortmann, A.; Engler, T.; van Rooijen, N.; Kochanek, S.; Kreppel, F. Targeting of Adenovirus Vectors to the LRP Receptor Family with the High-Affinity Ligand RAP via Combined Genetic and Chemical Modification of the PIX Capsomere. Mol. Ther. 2008, 16, 1813–1824. [Google Scholar] [CrossRef] [PubMed]
- Prill, J.-M.; Espenlaub, S.; Samen, U.; Engler, T.; Schmidt, E.; Vetrini, F.; Rosewell, A.; Grove, N.; Palmer, D.; Ng, P.; et al. Modifications of Adenovirus Hexon Allow for Either Hepatocyte Detargeting or Targeting with Potential Evasion From Kupffer Cells. Mol. Ther. 2011, 19, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, F.; Hagedorn, C.; Kreppel, F. Combined Genetic and Chemical Capsid Modifications of Adenovirus-Based Gene Transfer Vectors for Shielding and Targeting. J. Vis. Exp. 2018, 58480. [Google Scholar] [CrossRef]
- Gujar, S.; Pol, J.G.; Kroemer, G. Heating It up: Oncolytic Viruses Make Tumors ‘Hot’ and Suitable for Checkpoint Blockade Immunotherapies. OncoImmunology 2018, e1442169. [Google Scholar] [CrossRef]
- Tran, E.; Robbins, P.F.; Rosenberg, S.A. “Final Common Pathway” of Human Cancer Immunotherapy: Targeting Random Somatic Mutations. Nat. Immunol. 2017, 18, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Peng, M.; Mo, Y.; Wang, Y.; Wu, P.; Zhang, Y.; Xiong, F.; Guo, C.; Wu, X.; Li, Y.; Li, X.; et al. Neoantigen Vaccine: An Emerging Tumor Immunotherapy. Mol. Cancer 2019, 18, 128. [Google Scholar] [CrossRef]
- Beck-Engeser, G.B.; Monach, P.A.; Mumberg, D.; Yang, F.; Wanderling, S.; Schreiber, K.; Espinosa, R.; Le Beau, M.M.; Meredith, S.C.; Schreiber, H. Point Mutation in Essential Genes with Loss or Mutation of the Second Allele. J. Exp. Med. 2001, 194, 285–300. [Google Scholar] [CrossRef] [PubMed]
- Sato-Dahlman, M.; LaRocca, C.J.; Yanagiba, C.; Yamamoto, M. Adenovirus and Immunotherapy: Advancing Cancer Treatment by Combination. Cancers 2020, 12, 1295. [Google Scholar] [CrossRef] [PubMed]
- Cohen, C.J.; Gartner, J.J.; Horovitz-Fried, M.; Shamalov, K.; Trebska-McGowan, K.; Bliskovsky, V.V.; Parkhurst, M.R.; Ankri, C.; Prickett, Toddd, D.; Crystal, J.S.; et al. Isolation of Neoantigen-Specific T Cells from Tumor and Peripheral Lymphocytes. J. Clin. Investig. 2015, 125, 3981–3991. [Google Scholar] [CrossRef]
- Prickett, T.D.; Crystal, J.S.; Cohen, C.J.; Pasetto, A.; Parkhurst, M.R.; Gartner, J.J.; Yao, X.; Wang, R.; Gros, A.; Li, Y.F.; et al. Durable Complete Response from Metastatic Melanoma after Transfer of Autologous T Cells Recognizing 10 Mutated Tumor Antigens. Cancer Immunol. Res. 2016, 4, 669–678. [Google Scholar] [CrossRef]
- Lu, Y.-C.; Yao, X.; Crystal, J.S.; Li, Y.F.; El-Gamil, M.; Gross, C.; Davis, L.; Dudley, M.E.; Yang, J.C.; Samuels, Y.; et al. Efficient Identification of Mutated Cancer Antigens Recognized by T Cells Associated with Durable Tumor Regressions. Clin. Cancer Res. 2014, 20, 3401–3410. [Google Scholar] [CrossRef]
- Snyder, A.; Makarov, V.; Merghoub, T.; Yuan, J.; Zaretsky, J.M.; Desrichard, A.; Walsh, L.A.; Postow, M.A.; Wong, P.; Ho, T.S.; et al. Genetic Basis for Clinical Response to CTLA-4 Blockade in Melanoma. N. Engl. J. Med. 2014, 371, 2189–2199. [Google Scholar] [CrossRef] [PubMed]
- Hugo, W.; Zaretsky, J.M.; Sun, L.; Song, C.; Moreno, B.H.; Hu-Lieskovan, S.; Berent-Maoz, B.; Pang, J.; Chmielowski, B.; Cherry, G.; et al. Genomic and Transcriptomic Features of Response to Anti-PD-1 Therapy in Metastatic Melanoma. Cell 2016, 165, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, N.A.; Hellmann, M.D.; Snyder, A.; Kvistborg, P.; Makarov, V.; Havel, J.J.; Lee, W.; Yuan, J.; Wong, P.; Ho, T.S.; et al. Mutational Landscape Determines Sensitivity to PD-1 Blockade in Non–Small Cell Lung Cancer. Science 2015, 348, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Powles, T.; Eder, J.P.; Fine, G.D.; Braiteh, F.S.; Loriot, Y.; Cruz, C.; Bellmunt, J.; Burris, H.A.; Petrylak, D.P.; Teng, S.; et al. MPDL3280A (Anti-PD-L1) Treatment Leads to Clinical Activity in Metastatic Bladder Cancer. Nature 2014, 515, 558–562. [Google Scholar] [CrossRef] [PubMed]
- D’Alise, A.M.; Leoni, G.; Cotugno, G.; Troise, F.; Langone, F.; Fichera, I.; De Lucia, M.; Avalle, L.; Vitale, R.; Leuzzi, A.; et al. Adenoviral Vaccine Targeting Multiple Neoantigens as Strategy to Eradicate Large Tumors Combined with Checkpoint Blockade. Nat. Commun. 2019, 10, 2688. [Google Scholar] [CrossRef]
- Yoon, A.-R.; Hong, J.; Kim, S.W.; Yun, C.-O. Redirecting Adenovirus Tropism by Genetic, Chemical, and Mechanical Modification of the Adenovirus Surface for Cancer Gene Therapy. Expert Opin. Drug Deliv. 2016, 13, 843–858. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.C.; Scully, M.A.; Day, E.S. Cancer Cell Membrane-Coated Nanoparticles for Cancer Management. Cancers 2019, 11, 1836. [Google Scholar] [CrossRef] [PubMed]
| Vector | Transgene | Cancer | Mechanism | Therapy | Phase | Clinical Trial/ Reference | Status |
|---|---|---|---|---|---|---|---|
| Ad5-SGE-REIC/Dkk3 | REIC/Dkk3 | Localized prostate cancer | Cancer cell death induction and anticancer immunity | Neoadjuvant | I/II | NCT01931046 [13] # | Active, not recruiting |
| Ad5-SGE-REIC/Dkk3 (MTG201) | REIC/Dkk3 | Relapsed malignant pleural mesothelioma | Cancer cell death induction and anticancer immunity | Combination with nivolumab | II | NCT04013334 [14] # | Active, recruiting |
| AdHSV-tk /GCV | HSV-tk Ad-hCMV-Flt3L | High-grade malignant gliomas | TK: direct tumor cell killing Flt3L: immunostimulating effects | I/II | NCT01811992 [15] # | Active, not recruiting | |
| Adv/tk (GMCI) | HSV-tk | Advanced nonmetastatic pancreatic adenocarcinoma | TK: direct tumor cell killing | Neoadjuvant plus chemoradiation | II | NCT02446093 | Active, not recruiting |
| Adv/tk | HSV-tk | Advanced hepatocellular carcinoma | TK: direct tumor cell killing | Liver transplantation | III | NCT03313596 [16] # | Active, recruiting |
| Adv/tk (GMCI) | Adv-tk | Pediatric brain tumors | Direct tumor cell killing | Combination with radiation therapy | I | NCT00634231 [17] # | Active, not recruiting |
| Adv/RSV-tk | HSV-tk | Recurrent prostate cancer | Direct tumor cell killing | Combination with brachytherapy | I/II | NCT01913106 | Active, recruiting |
| Adv/HSV-tk | HSV-tk | Metastatic nonsmall cell lung carcinoma and uveal melanoma | Direct tumor cell killing | Combination with stereotactic body radiation therapy or nivolumab | II | NCT02831933 | Terminated (Lack of funding) |
| Ad/PNP + fludarabine | PNP | Head and neck squamous cell carcinoma | PNP protein actives the second component of the therapy fludarabine phosphate | I | NCT01310179 [18] | Completed | |
| rAd-IFN/Syn-3 (Instiladrin) | IFN α-2b | High-grade nonmuscle invasive bladder cancer | Immunoregulatory effects | III | NCT02773849 [19] # | Active, not recruiting | |
| BG00001 | IFN-β | Pleural mesothelioma | Immunoregulatory effects | I | NCT00299962 [20] | Completed | |
| Ad-RTS-hIL-12 | IL-12 | Advanced or metastatic breast cancer | Proinflammatory cytokine, enhances the cytotoxic activity of T-lymphocytes and resting natural killer cells | Combination with VELEDIMEX | Ib/II | NCT02423902 | Unknown |
| Ad-RTS-hIL-12 | IL-12 | Recurrent or progressive glioblastoma | Proinflammatory cytokine, enhances the cytotoxic activity of T-lymphocytes and resting natural killer cells | Ad-RTS-hIL-12 + Veledimex in combination with Cemiplimab | II | NCT04006119 | Active, not recruiting |
| Ad-RTS-hIL-12 | IL-12 | Glioblastoma or malignant glioma | Proinflammatory cytokine, enhances the cytotoxic activity of T-lymphocytes and resting natural killer cells | Combination with Veledimex | I | NCT02026271 [21] # | Active, not recruiting |
| SCH-58500 | P53 | Primary ovarian, fallopian tube, or peritoneal cancer | Tumor suppressor gene: antitumor effect by blocking cell cycle progression at the G1/S, activating DNA repair pathways | I | NCT00002960 [22] | Completed | |
| Ad-p53 | P53 | Recurrent or metastatic head and neck squamous cell carcinoma | Tumor suppressor gene: antitumor effect by blocking cell cycle progression at the G1/S, activating DNA repair pathways | Adjuvant in combination with Anti-PD-1 or Anti-PD-L1 therapy | II | NCT03544723 | Active, recruiting |
| ADVEXIN | P53 | Squamous cell carcinoma of the oral cavity, oropharynx, hypopharynx, and larynx | Tumor suppressor gene: antitumor effect by blocking cell cycle progression at the G1/S, activating DNA repair pathways | I/II | NCT00064103 [23] | Completed |
| Classification and Tropism of Human Adenoviruses | |||
|---|---|---|---|
| Subgroup | Serotypes | Identified Receptors | Tropism |
| A | 12, 18, 31, 61 | CAR | Enteric, respiratory |
| B | 3, 7, 11, 14, 16, 21, 34, 35, 50, 55, 66,68, 76–79 | CD46, DSG2, CD80, CD86 | Renal, ocular, respiratory |
| C | 1, 2, 5, 6, 57, 89 | CAR, VCAM-1, HSPG, MHC1-a2, SR | Ocular, lymphoid, respiratory, hepatic |
| D | 8–10, 13, 15, 17, 19, 20, 22–30, 32, 33, 36–39, 42–49, 51, 53, 54, 56, 58–60, 62–65, 67, 69–75, 80–88, 90–103 | SA, CD46, CAR, GD1a | Ocular, enteric |
| E | 4 | CAR | Ocular, respiratory |
| F | 40, 41 | CAR | Enteric |
| G | 52 | CAR, AS | Enteric |
| Attachment Receptor | Tropism | Modification | Serotype Origin/ Subgroup | Results | Reference |
|---|---|---|---|---|---|
| CD46 | Glioma | Fiber replacement | Ad35, Ad16, Ad50 | Increased transduction of patient-derived cells | [156] |
| Adenovirus serotype 3 receptor | Ovarian cancer cells | Fiber knob replacement | Ad3 (modified)/B1 | Enhanced gene transfer to various cancer cell lines and primary tumor tissues | [157] |
| Adenovirus serotype 3 receptor | Lung cancer (NSCLC primary tissue) | Fiber knob replacement | Ad3 (modified)/B1 | Improved killing of NSCLC cells | [158] |
| Sialic acid, phage display for kidney | Renal cancer and detargeting the liver | Fiber knob replacement | Ad5 (modified)/19p (fiber) | Reduced liver tropism and improved gene transfer to renal cancer | [159] |
| Unidentified cellular receptor | Cancer cell lines of pancreatic, breast, lung, esophageal, and ovarian | Fiber knob replacement | Ad5 (modified)/D49 | Efficiently transduced | [160] |
| CD46 | Primary human cell cultures | Fiber replacement | Ad5PTD/F35 | Increased transduction capacity of T-cells, monocytes, macrophages, dendritic cells, pancreatic islets, mesenchymal stem cells, and tumor-initiating cells | [161] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tessarollo, N.G.; Domingues, A.C.M.; Antunes, F.; Luz, J.C.d.S.d.; Rodrigues, O.A.; Cerqueira, O.L.D.; Strauss, B.E. Nonreplicating Adenoviral Vectors: Improving Tropism and Delivery of Cancer Gene Therapy. Cancers 2021, 13, 1863. https://doi.org/10.3390/cancers13081863
Tessarollo NG, Domingues ACM, Antunes F, Luz JCdSd, Rodrigues OA, Cerqueira OLD, Strauss BE. Nonreplicating Adenoviral Vectors: Improving Tropism and Delivery of Cancer Gene Therapy. Cancers. 2021; 13(8):1863. https://doi.org/10.3390/cancers13081863
Chicago/Turabian StyleTessarollo, Nayara Gusmão, Ana Carolina M. Domingues, Fernanda Antunes, Jean Carlos dos Santos da Luz, Otavio Augusto Rodrigues, Otto Luiz Dutra Cerqueira, and Bryan E. Strauss. 2021. "Nonreplicating Adenoviral Vectors: Improving Tropism and Delivery of Cancer Gene Therapy" Cancers 13, no. 8: 1863. https://doi.org/10.3390/cancers13081863
APA StyleTessarollo, N. G., Domingues, A. C. M., Antunes, F., Luz, J. C. d. S. d., Rodrigues, O. A., Cerqueira, O. L. D., & Strauss, B. E. (2021). Nonreplicating Adenoviral Vectors: Improving Tropism and Delivery of Cancer Gene Therapy. Cancers, 13(8), 1863. https://doi.org/10.3390/cancers13081863








