Predictive Factors for RAI-Refractory Disease and Short Overall Survival in PDTC
Abstract
Simple Summary
Abstract
1. Introduction
2. Methods
2.1. RAI Avidity
2.2. Response Evaluation
2.3. Predictive Factors for Overall Survival
2.4. Statistical Analysis
3. Results
3.1. Imaging and Treatment Protocol
3.2. Study Cohort
3.3. Initial RAI Avidity
3.4. Therapy Response
3.5. Survival Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sanders, E.M., Jr.; Livolsi, V.A.; Brierley, J.; Shin, J.; Randolph, G.W. An evidence-based review of poorly differentiated thyroid cancer. World J. Surg. 2007, 31, 934–945. [Google Scholar] [PubMed]
- Volante, M.; Landolfi, S.; Chiusa, L.; Palestini, N.; Motta, M.; Codegone, A.; Torchio, B.; Papotti, M.G. Poorly differentiated carcinomas of the thyroid with trabecular, insular, and solid patterns. Cancer 2004, 100, 950–957. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, A.; Kasai, N.; Sugano, H. Poorly differentiated carcinoma of the thyroid. A clinicopathologic entity for a high-risk group of papillary and follicular carcinomas. Cancer 1983, 52, 1849–1855. [Google Scholar] [CrossRef]
- de la Fouchardière, C.; Decaussin-Petrucci, M.; Berthiller, J.; Descotes, F.; Lopez, J.; Lifante, J.-C.; Peix, J.-L.; Giraudet, A.-L.; Delahaye, A.; Masson, S.; et al. Predictive factors of outcome in poorly differentiated thyroid carcinomas. Eur. J. Cancer 2018, 92, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Volante, M.; Bussolati, G.; Papotti, M. The story of poorly differentiated thyroid carcinoma: From Langhans’ description to the Turin proposal via Juan Rosai. Semin. Diagn. Pathol. 2016, 33, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Underwood, H.J.; Shaha, A.R.; Patel, K.N. Variable response to radioactive iodine treatment in poorly differentiated thyroid carcinoma. Gland. Surg. 2019, 8, 589–590. [Google Scholar] [CrossRef]
- Hiltzik, D.; Carlson, D.L.; Tuttle, R.M.; Chuai, S.; Ishill, N.; Shaha, A.; Shah, J.P.; Singh, B.; Ghossein, R.A. Poorly differentiated thyroid carcinomas defined on the basis of mitosis and necrosis: A clinicopathologic study of 58 patients. Cancer 2006, 106, 1286–1295. [Google Scholar] [CrossRef] [PubMed]
- Shah, D.R.; Shah, R.R.; Morganroth, J. Tyrosine Kinase Inhibitors: Their on-target toxicities as potential indicators of efficacy. Drug Saf. 2013, 36, 413–426. [Google Scholar] [CrossRef]
- Shah, R.R.; Morganroth, J.; Shah, D.R. Cardiovascular safety of tyrosine kinase inhibitors: With a special focus on cardiac repolarisation (QT interval). Drug Saf. 2013, 36, 295–316. [Google Scholar] [CrossRef]
- Kreissl, M.C.; Janssen, M.J.; Nagarajah, J. Current Treatment Strategies in Metastasized Differentiated Thyroid Cancer. J. Nucl. Med. 2019, 60, 9–15. [Google Scholar] [CrossRef]
- Manohar, P.M.; Beesley, L.J.; Bellile, E.L.; Worden, F.P.; Avram, A.M. Prognostic Value of FDG-PET/CT Metabolic Parameters in Metastatic Radioiodine-Refractory Differentiated Thyroid Cancer. Clin. Nucl. Med. 2018, 43, 641–647. [Google Scholar] [CrossRef]
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef]
- Haugen, B.R. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: What is new and what has changed? Cancer 2017, 123, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Jentzen, W.; Hoppenbrouwers, J.; van Leeuwen, P.; van der Velden, D.; van de Kolk, R.; Poeppel, T.D.; Nagarajah, J.; Brandau, W.; Bockisch, A.; Rosenbaum-Krumme, S. Assessment of lesion response in the initial radioiodine treatment of differentiated thyroid cancer using 124I PET imaging. J. Nucl. Med. 2014, 55, 1759–1765. [Google Scholar] [CrossRef] [PubMed]
- Maxon, H.R., 3rd; Englaro, E.E.; Thomas, S.R.; Hertzberg, V.S.; Hinnefeld, J.D.; Chen, L.S.; Smith, H.; Cummings, D.; Aden, M.D. Radioiodine-131 therapy for well-differentiated thyroid cancer--a quantitative radiation dosimetric approach: Outcome and validation in 85 patients. J. Nucl. Med. 1992, 33, 1132–1136. [Google Scholar]
- Maxon, H.R.; Thomas, S.R.; Hertzberg, V.S.; Kereiakes, J.G.; Chen, I.-W.; Sperling, M.I.; Saenger, E.L. Relation between effective radiation dose and outcome of radioiodine therapy for thyroid cancer. N. Engl. J. Med. 1983, 309, 937–941. [Google Scholar] [CrossRef]
- Jung, T.S.; Kim, T.Y.; Kim, K.W.; Oh, Y.L.; Park, D.J.; Cho, B.Y.; Shong, Y.K.; Kim, W.B.; Park, Y.J.; Jung, J.H.; et al. Clinical features and prognostic factors for survival in patients with poorly differentiated thyroid carcinoma and comparison to the patients with the aggressive variants of papillary thyroid carcinoma. Endocr. J. 2007, 54, 265–274. [Google Scholar] [CrossRef]
- Bazan, J.G.; Koong, A.C.; Kapp, D.S.; Quon, A.; Graves, E.E.; Loo, B.W.; Chang, D.T. Metabolic tumor volume predicts disease progression and survival in patients with squamous cell carcinoma of the anal canal. J. Nucl. Med. 2013, 54, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Paidpally, V.; Chirindel, A.; Chung, C.H.; Richmon, J.; Koch, W.; Quon, H.; Subramaniam, R.M. FDG volumetric parameters and survival outcomes after definitive chemoradiotherapy in patients with recurrent head and neck squamous cell carcinoma. Am. J. Roentgenol. 2014, 203, W139–W145. [Google Scholar] [CrossRef][Green Version]
- Weber, M.; Kersting, D.; Umutlu, L.; Schäfers, M.; Rischpler, C.; Fendler, W.P.; Buvat, I.; Herrmann, K.; Seifert, R. Just another “Clever Hans”? Neural networks and FDG PET-CT to predict the outcome of patients with breast cancer. Eur. J. Nucl. Med. Mol. Imaging 2021. [Google Scholar] [CrossRef]
- Soydal, Ç.; Yüksel, C.B.; Küçük, N.Ö.; Ökten, I.; Özkan, E.; Erdoğan, B.D. Prognostic Value of Metabolic Tumor Volume Measured by 18F-FDG PET/CT in Esophageal Cancer Patients. Mol. Imaging Radionucl. Ther. 2014, 23, 12–15. [Google Scholar] [CrossRef] [PubMed]
- Hyun, S.H.; Choi, J.Y.; Shim, Y.M.; Kim, K.; Lee, S.J.; Cho, Y.S.; Lee, J.Y.; Lee, K.-H.; Kim, B.-T. Prognostic value of metabolic tumor volume measured by 18f-fluorodeoxyglucose positron emission tomography in patients with esophageal carcinoma. Ann. Surg. Oncol. 2009, 17, 115–122. [Google Scholar] [CrossRef] [PubMed]
- La, T.H.; Filion, E.J.; Turnbull, B.B.; Chu, J.N.; Lee, P.; Nguyen, K.; Maxim, P.; Quon, A.; Graves, E.E.; Loo, B.W.; et al. Metabolic tumor volume predicts for recurrence and death in head-and-neck cancer. Int. J. Radiat. Oncol. 2009, 74, 1335–1341. [Google Scholar] [CrossRef] [PubMed]
- Chun, Y.J.; Jeung, H.-C.; Park, H.S.; Park, J.S.; Rha, S.Y.; Choi, H.J.; Lee, J.-H.; Jeon, T.J. Significance of Metabolic Tumor Volume and Total Lesion Glycolysis Measured Using (18)F-FDG PET/CT in Locally Advanced and Metastatic Gallbladder Carcinoma. Yonsei Med. J. 2019, 60, 604–610. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Baba, S.; Endo, M.; Setsu, N.; Iida, K.; Fukushi, J.-I.; Kawaguchi, K.; Okada, S.; Bekki, H.; Isoda, T.; et al. Metabolic Tumor Volume by (18)F-FDG PET/CT Can Predict the Clinical Outcome of Primary Malignant Spine/Spinal Tumors. BioMed Res. Int. 2017, 2017, 8132676. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, X.; Li, H.; Li, X.; Lin, Y. Quantitative thyroglobulin response to radioactive iodine treatment in predicting radioactive iodine-refractory thyroid cancer with pulmonary metastasis. PLoS ONE 2017, 12, e0179664. [Google Scholar] [CrossRef]
- Tuttle, R.M.; Grewal, R.K.; Larson, S.M. Radioactive iodine therapy in poorly differentiated thyroid cancer. Nat. Clin. Pract. Oncol. 2007, 4, 665–668. [Google Scholar] [CrossRef]
- Moon, H.J.; Kim, E.-K.; Chung, W.Y.; Yoon, J.H.; Kwak, J.Y. Minimal extrathyroidal extension in patients with papillary thyroid microcarcinoma: Is it a real prognostic factor? Ann. Surg. Oncol. 2011, 18, 1916–1923. [Google Scholar] [CrossRef]
- Radowsky, J.S.; Howard, R.S.; Burch, H.B.; Stojadinovic, A. Impact of degree of extrathyroidal extension of disease on papillary thyroid cancer outcome. Thyroid 2014, 24, 241–244. [Google Scholar] [CrossRef]
- Kist, J.W.; De Keizer, B.; Van Der Vlies, M.; Brouwers, A.H.; Huysmans, D.A.; Van Der Zant, F.M.; Hermsen, R.; Stokkel, M.P.; Hoekstra, O.S.; Vogel, W.V.; et al. 124I PET/CT to Predict the Outcome of Blind 131I Treatment in Patients with Biochemical Recurrence of Differentiated Thyroid Cancer: Results of a Multicenter Diagnostic Cohort Study (THYROPET). J. Nucl. Med. 2015, 57, 701–707. [Google Scholar] [CrossRef]

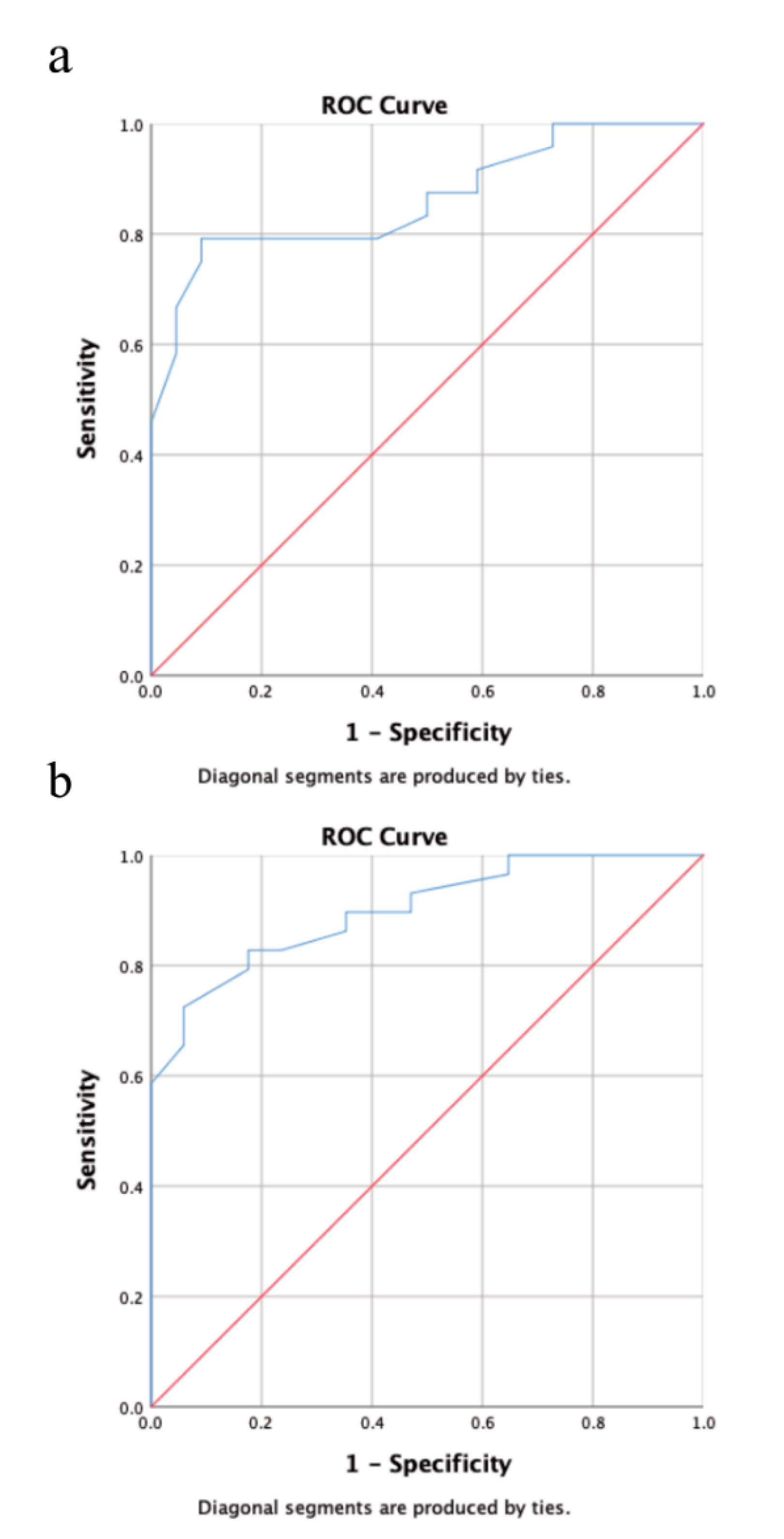
| Age | Mean (Range) | 58.5 (15–87) |
|---|---|---|
| >55 years, n (%) | 30 (59) | |
| Sex | Male, n (%) | 23 (45) |
| Female, n (%) | 28 (55) | |
| Size of primary | >40 mm, n (%) | 31 (61) |
| ≤40 mm, n (%) | 15 (29) | |
| Tx, n (%) | 2 (4) | |
| n/a, n (%) | 3 (6) | |
| ETE | Present, n (%) | 28 (55) |
| Absent, n (%) | 21 (41) | |
| Tx, n (%) | 2 (4) | |
| Stage | N0M0, n (%) | 19 (37) |
| N1M0, n (%) | 10 (20) | |
| N0M1, n (%) | 9 (18) | |
| N1M1, n (%) | 13 (25) | |
| Tg | Initial, mean (range) | 1734.6 (0–49,362) |
| Early Tg progression n/a, n (%) | 8 (16) | |
| Early Tg progression, n (%) | 15 (29) | |
| No early Tg progression, n (%) | 28 (55) | |
| Med. OS, early Tg progression, months | 57.8 | |
| Med. OS, no early Tg progression, months | nr | |
| Category | RAI-R TC, n (%) | 28 (55) |
| Radioavid TC, n (%) | 7 (14) | |
| Disease-free, n (%) | 16 (31) | |
| Progression to RAI-R TC | At initial diagnosis, n (%) | 25 (49) |
| ≤12 months, n (%) | 3 (6) | |
| >12 months, n (%) | 6 (12) | |
| Not observed, n (%) | 17 (33) | |
| AJCC TNM stage | I | 12 (24) |
| II | 3 (6) | |
| III | 8 (16) | |
| IV | 28 (55) |
| Age | Mean (Range) | 58.5 (13–87) |
|---|---|---|
| Sex | Male, n (%) | 24 (43) |
| Female, n (%) | 32 (57) | |
| Stage | N0M0, n (%) | 25 (45) |
| N1M0, n (%) | 8 (14) | |
| N0M1, n (%) | 11 (20) | |
| N1M1, n (%) | 12 (21) | |
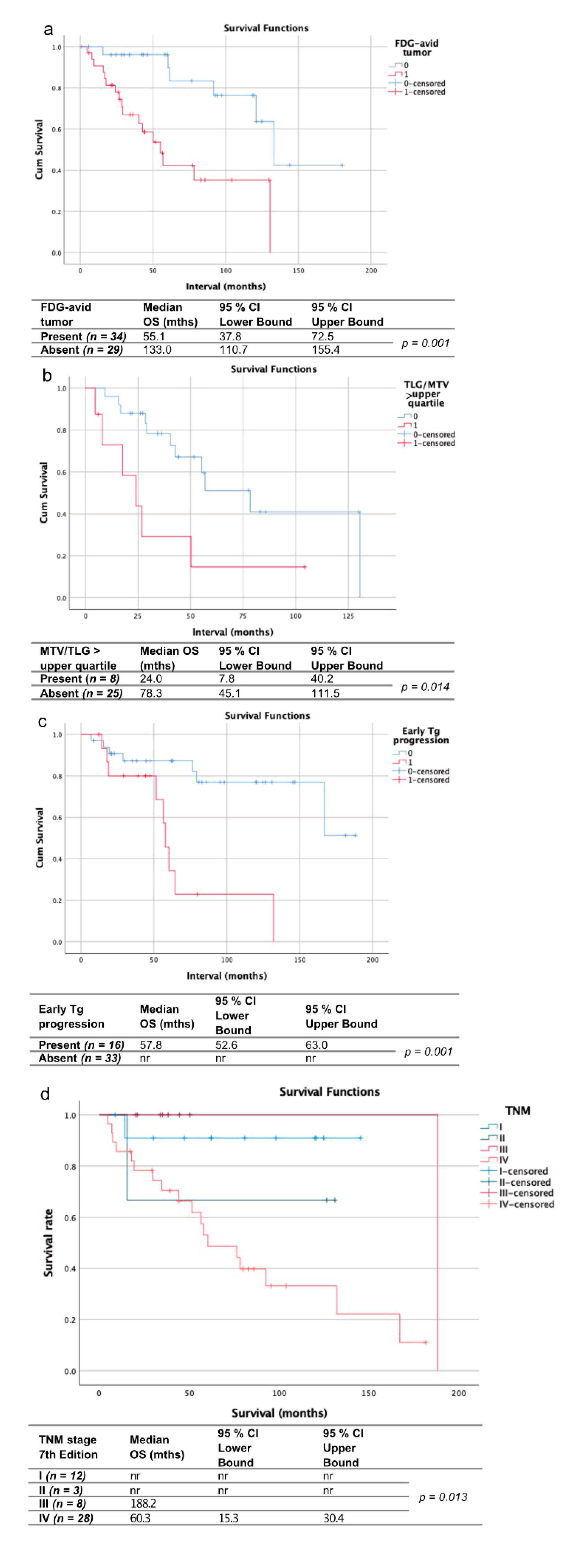
| FDG-avid tumor | Present, n (%) | 31 (55) |
| Absent, n (%) | 25 (45) | |
| Med. Survival FDG-avid tumor present, months | 50.2 | |
| Med. Survival FDG-avid tumor absent, months | 133.0 | |
| MTV | Mean (range), mL | 58.1 (0.2–468.9) |
| Upper quartile, mL | 229.0 | |
| Med. Survival MTV > upper quartile, months | 29.0 | |
| Med. Survival MTV < upper quartile, months | 56.9 | |
| TLG | Mean (range), mL | 1159.3 (2.3–15,175.7) |
| Upper quartile, mL | 49.0 | |
| Med. Survival MTV > upper quartile, months | 29.0 | |
| Med. Survival MTV < upper quartile, months | 56.9 | |
| Follow-up | Overall survival, mean ± SD | 57.0 ± 43.9 |
| RAI-R TC Occurrence | ETE | No ETE | Significance |
|---|---|---|---|
| Initial RAI-R TC | 75.0% | 28.6% | p = 0.001 |
| Ever RAI-R TC | 89.3% | 33.3% | p < 0.001 |
| Primary > 40 mm | Primary ≤ 40 mm | Significance | |
| Initial RAI-R TC | 64.5% | 26.7% | p = 0.017 |
| Ever RAI-R TC | 80.6% | 26.7% | p = 0.001 |
| Age > 55 | Age ≤ 55 | Significance | |
| Initial RAI-R TC | 66.7% | 38.1% | p = 0.041 |
| Ever RAI-R TC | 83.3% | 42.9% | p = 0.003 |
| (a) | ||||
| Risk factors | p | Hazard Ratio | 95% CI for Hazard Ratio | |
| CI Lower | CI Upper | |||
| ETE | 0.016 | 5.171 | 1.354 | 19.747 |
| Primary size >40 mm | 0.081 | 3.669 | 0.85 | 15.838 |
| (b) | ||||
| p | Hazard Ratio | 95% CI for Hazard Ratio | ||
| CI Lower | CI Upper | |||
| ETE | 0.003 | 14.821 | 2.526 | 86.943 |
| Primary size >40 mm | 0.007 | 11.596 | 1.932 | 69.612 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kersting, D.; Seifert, R.; Kessler, L.; Herrmann, K.; Theurer, S.; Brandenburg, T.; Dralle, H.; Weber, F.; Umutlu, L.; Führer-Sakel, D.; et al. Predictive Factors for RAI-Refractory Disease and Short Overall Survival in PDTC. Cancers 2021, 13, 1728. https://doi.org/10.3390/cancers13071728
Kersting D, Seifert R, Kessler L, Herrmann K, Theurer S, Brandenburg T, Dralle H, Weber F, Umutlu L, Führer-Sakel D, et al. Predictive Factors for RAI-Refractory Disease and Short Overall Survival in PDTC. Cancers. 2021; 13(7):1728. https://doi.org/10.3390/cancers13071728
Chicago/Turabian StyleKersting, David, Robert Seifert, Lukas Kessler, Ken Herrmann, Sarah Theurer, Tim Brandenburg, Henning Dralle, Frank Weber, Lale Umutlu, Dagmar Führer-Sakel, and et al. 2021. "Predictive Factors for RAI-Refractory Disease and Short Overall Survival in PDTC" Cancers 13, no. 7: 1728. https://doi.org/10.3390/cancers13071728
APA StyleKersting, D., Seifert, R., Kessler, L., Herrmann, K., Theurer, S., Brandenburg, T., Dralle, H., Weber, F., Umutlu, L., Führer-Sakel, D., Görges, R., Rischpler, C., & Weber, M. (2021). Predictive Factors for RAI-Refractory Disease and Short Overall Survival in PDTC. Cancers, 13(7), 1728. https://doi.org/10.3390/cancers13071728






