DPP9: Comprehensive In Silico Analyses of Loss of Function Gene Variants and Associated Gene Expression Signatures in Human Hepatocellular Carcinoma
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Protein Sequences
2.2. Public Databases for Accessing Loss of Function Variants
2.3. Loss of Function Variants
2.4. Intronic Variants
2.5. Genetic Variant Tolerance Parameters
2.6. Gene Correlation Analysis
2.7. Survival Analysis
2.8. Reactome Pathways Enrichment Analysis
2.9. Data and Statistical Analyses
3. Results
3.1. DPP9 Loss of Function (LoF) Variants and Disease Associations
3.2. Intronic SNPs in DPP9
3.3. Genetic Variation Tolerance in DPP9 and the DPP4 Gene Family
3.4. Association of DPP9 Expression with Patient Survival in HCC and UCEC
3.5. Association of DPP9 Expression with Clinical Parameters and Survival Outcome in HCC
3.6. Association of Expression of DPPs with Poor Survival of Patients with HCC
3.7. Genes Correlated with DPPs Significantly Associated with Poor Prognosis in HCC and Multiple Oncogenic and Epigenetic Pathways
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, H.; Chen, Y.; Keane, F.M.; Gorrell, M.D. Advances in understanding the expression and function of dipeptidyl peptidase 8 and 9. Mol. Cancer Res. 2013, 11, 1487–1496. [Google Scholar] [CrossRef]
- Yu, D.M.; Ajami, K.; Gall, M.G.; Park, J.; Lee, C.S.; Evans, K.A.; McLaughlin, E.A.; Pitman, M.R.; Abbott, C.A.; McCaughan, G.W.; et al. The in vivo expression of dipeptidyl peptidases 8 and 9. J. Histochem. Cytochem. 2009, 57, 1025–1040. [Google Scholar] [CrossRef]
- Yao, T.W.; Kim, W.S.; Yu, D.M.; Sharbeen, G.; McCaughan, G.W.; Choi, K.Y.; Xia, P.; Gorrell, M.D. A novel role of dipeptidyl peptidase 9 in epidermal growth factor signaling. Mol. Cancer Res. 2011, 9, 948–959. [Google Scholar] [CrossRef] [PubMed]
- Okondo, M.C.; Rao, S.D.; Taabazuing, C.Y.; Chui, A.J.; Poplawski, S.E.; Johnson, D.C.; Bachovchin, D.A. Inhibition of Dpp8/9 activates the Nlrp1b inflammasome. Cell Chem. Biol. 2018, 25, 262–267.e265. [Google Scholar] [CrossRef] [PubMed]
- Okondo, M.C.; Johnson, D.C.; Sridharan, R.; Go, E.B.; Chui, A.J.; Wang, M.S.; Poplawski, S.E.; Wu, W.; Liu, Y.; Lai, J.H.; et al. DPP8 and DPP9 inhibition induces pro-caspase-1-dependent monocyte and macrophage pyroptosis. Nat. Chem. Biol. 2017, 13, 46–53. [Google Scholar] [CrossRef]
- Justa-Schuch, D.; Silva-Garcia, M.; Pilla, E.; Engelke, M.; Kilisch, M.; Lenz, C.; Möller, U.; Nakamura, F.; Urlaub, H.; Geiss-Friedlander, R. DPP9 is a novel component of the N-end rule pathway targeting the tyrosine kinase Syk. eLife 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Li, J.; Shen, Q.; Feng, J.; Liu, H.; Wang, W.; Xu, L.; Shi, G.; Ye, X.; Ge, M.; et al. Contribution of upregulated dipeptidyl peptidase 9 (DPP9) in promoting tumoregenicity, metastasis and the prediction of poor prognosis in non-small cell lung cancer (NSCLC). Int. J. Cancer 2017, 140, 1620–1632. [Google Scholar] [CrossRef]
- Yokobori, T. Editorial Comment on: DPP9 Increases Chemoresistance and is an Indicator of Poor Prognosis in Colorectal Cancer. Ann. Surg. Oncol. 2020, 27, 4084–4085. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.Q.; Zhao, M.; Huang, G.Z.; Zheng, Z.N.; Chen, Y.; Zeng, W.S.; Lv, X.Z. Fibroblast Activation Protein (FAP) Overexpression Induces Epithelial-Mesenchymal Transition (EMT) in Oral Squamous Cell Carcinoma by Down-Regulating Dipeptidyl Peptidase 9 (DPP9). Onco Targets Ther. 2020, 13, 2599–2611. [Google Scholar] [CrossRef]
- Chowdhury, S.; Chen, Y.; Yao, T.W.; Ajami, K.; Wang, X.M.; Popov, Y.; Schuppan, D.; Bertolino, P.; McCaughan, G.W.; Yu, D.M.; et al. Regulation of dipeptidyl peptidase 8 and 9 expression in activated lymphocytes and injured liver. World J. Gastroenterol. 2013, 19, 2883–2893. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Chen, Y.; Wadham, C.; McCaughan, G.W.; Keane, F.M.; Gorrell, M.D. Dipeptidyl peptidase 9 subcellular localization and a role in cell adhesion involving focal adhesion kinase and paxillin. Biochim. Biophys. Acta 2015, 1853, 470–480. [Google Scholar] [CrossRef]
- Badvie, S. Hepatocellular carcinoma. Postgrad Med. J. 2000, 76, 4–11. [Google Scholar] [CrossRef]
- Forner, A.; Reig, M.; Bruix, J. Hepatocellular carcinoma. Lancet 2018, 391, 1301–1314. [Google Scholar] [CrossRef]
- El-Serag, H.B. Hepatocellular carcinoma: An epidemiologic view. J. Clin. Gastroenterol. 2002, 35, S72–S78. [Google Scholar] [CrossRef]
- El–Serag, H.B.; Rudolph, K.L. Hepatocellular Carcinoma: Epidemiology and Molecular Carcinogenesis. Gastroenterology 2007, 132, 2557–2576. [Google Scholar] [CrossRef] [PubMed]
- Ray, K. NAFLD-the next global epidemic. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 621. [Google Scholar] [CrossRef] [PubMed]
- Ajami, K.; Abbott, C.A.; McCaughan, G.W.; Gorrell, M.D. Dipeptidyl peptidase 9 has two forms, a broad tissue distribution, cytoplasmic localization and DPIV-like peptidase activity. Biochim. Biophys. Acta 2004, 1679, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.M.; Yao, T.W.; Chowdhury, S.; Nadvi, N.A.; Osborne, B.; Church, W.B.; McCaughan, G.W.; Gorrell, M.D. The dipeptidyl peptidase IV family in cancer and cell biology. FEBS J. 2010, 277, 1126–1144. [Google Scholar] [CrossRef]
- Dunaevsky, Y.E.; Tereshchenkova, V.F.; Oppert, B.; Belozersky, M.A.; Filippova, I.Y.; Elpidina, E.N. Human proline specific peptidases: A comprehensive analysis. Biochim. Biophys. Acta. Gen. Subj. 2020, 1864, 129636. [Google Scholar] [CrossRef]
- Ross, B.; Krapp, S.; Augustin, M.; Kierfersauer, R.; Arciniega, M.; Geiss-Friedlander, R.; Huber, R. Structures and mechanism of dipeptidyl peptidases 8 and 9, important players in cellular homeostasis and cancer. Proc. Natl. Acad. Sci. USA 2018, 115, E1437–E1445. [Google Scholar] [CrossRef]
- Justa-Schuch, D.; Möller, U.; Geiss-Friedlander, R. The amino terminus extension in the long dipeptidyl peptidase 9 isoform contains a nuclear localization signal targeting the active peptidase to the nucleus. Cell Mol. Life Sci. 2014, 71, 3611–3626. [Google Scholar] [CrossRef]
- Lee, H.J.; Chen, Y.S.; Chou, C.Y.; Chien, C.H.; Lin, C.H.; Chang, G.G.; Chen, X. Investigation of the dimer interface and substrate specificity of prolyl dipeptidase DPP8. J. Biol. Chem. 2006, 281, 38653–38662. [Google Scholar] [CrossRef]
- Pilla, E.; Möller, U.; Sauer, G.; Mattiroli, F.; Melchior, F.; Geiss-Friedlander, R. A novel SUMO1-specific interacting motif in dipeptidyl peptidase 9 (DPP9) that is important for enzymatic regulation. J. Biol. Chem. 2012, 287, 44320–44329. [Google Scholar] [CrossRef]
- Gall, M.G.; Gorrell, M.D. The multifunctional post-proline dipeptidyl peptidase, DPP9, in mice, cell biology and immunity. In Pathophysiological Aspects of Proteases; Chakraborti, S., Dhalla, N.S., Eds.; Springer: Singapore, 2017; Volume 1, pp. 23–46. [Google Scholar]
- Zhang, H.E.; Gorrell, M.D. DPP9. In Encyclopedia of Signaling Molecules, 2nd ed.; Choi, S., Ed.; Springer International Publishing: New York, NY, USA, 2018; pp. 1418–1422. [Google Scholar] [CrossRef]
- Minikel, E.V.; Karczewski, K.J.; Martin, H.C.; Cummings, B.B.; Whiffin, N.; Rhodes, D.; Alföldi, J.; Trembath, R.C.; van Heel, D.A.; Daly, M.J.; et al. Evaluating drug targets through human loss-of-function genetic variation. Nature 2020, 581, 459–464. [Google Scholar] [CrossRef]
- Plenge, R.M.; Scolnick, E.M.; Altshuler, D. Validating therapeutic targets through human genetics. Nat. Rev. Drug Discov. 2013, 12, 581–594. [Google Scholar] [CrossRef] [PubMed]
- Gelernter, J.; Kranzler, H.R.; Sherva, R.; Almasy, L.; Koesterer, R.; Smith, A.H.; Anton, R.; Preuss, U.W.; Ridinger, M.; Rujescu, D.; et al. Genome-wide association study of alcohol dependence:significant findings in African- and European-Americans including novel risk loci. Mol. Psychiatry 2014, 19, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, B.D.; Putman, R.K.; Araki, T.; Nishino, M.; Gudmundsson, G.; Gudnason, V.; Eiriksdottir, G.; Zilhao Nogueira, N.R.; Dupuis, J.; Xu, H.; et al. Overlap of Genetic Risk between Interstitial Lung Abnormalities and Idiopathic Pulmonary Fibrosis. Am. J. Respir Crit Care Med. 2019, 200, 1402–1413. [Google Scholar] [CrossRef] [PubMed]
- Fingerlin, T.E.; Murphy, E.; Zhang, W.; Peljto, A.L.; Brown, K.K.; Steele, M.P.; Loyd, J.E.; Cosgrove, G.P.; Lynch, D.; Groshong, S.; et al. Genome-wide association study identifies multiple susceptibility loci for pulmonary fibrosis. Nat. Genet. 2013, 45, 613–620. [Google Scholar] [CrossRef]
- Chan, V.; Fong, G.C.; Luk, K.D.; Yip, B.; Lee, M.K.; Wong, M.S.; Lu, D.D.; Chan, T.K. A genetic locus for adolescent idiopathic scoliosis linked to chromosome 19p13.3. Am. J. Hum. Genet. 2002, 71, 401–406. [Google Scholar] [CrossRef]
- Pairo-Castineira, E.; Clohisey, S.; Klaric, L.; Bretherick, A.D.; Rawlik, K.; Pasko, D.; Walker, S.; Parkinson, N.; Fourman, M.H.; Russell, C.D.; et al. Genetic mechanisms of critical illness in Covid-19. Nature 2020. [Google Scholar] [CrossRef]
- Wang, L.; Balmat, T.J.; Antonia, A.L.; Constantine, F.J.; Henao, R.; Burke, T.W.; Ingham, A.; McClain, M.T.; Tsalik, E.L.; Ko, E.R.; et al. An atlas connecting shared genetic architecture of human diseases and molecular phenotypes provides insight into COVID-19 susceptibility. medRxiv 2020. [Google Scholar] [CrossRef]
- Karczewski, K.J.; Francioli, L.C.; Tiao, G.; Cummings, B.B.; Alföldi, J.; Wang, Q.; Collins, R.L.; Laricchia, K.M.; Ganna, A.; Birnbaum, D.P.; et al. The mutational constraint spectrum quantified from variation in 141,456 humans. Nature 2020, 581, 434–443. [Google Scholar] [CrossRef] [PubMed]
- Grossman, R.L.; Heath, A.P.; Ferretti, V.; Varmus, H.E.; Lowy, D.R.; Kibbe, W.A.; Staudt, L.M. Toward a Shared Vision for Cancer Genomic Data. N. Engl. J. Med. 2016, 375, 1109–1112. [Google Scholar] [CrossRef] [PubMed]
- Tate, J.G.; Bamford, S.; Jubb, H.C.; Sondka, Z.; Beare, D.M.; Bindal, N.; Boutselakis, H.; Cole, C.G.; Creatore, C.; Dawson, E.; et al. COSMIC: The Catalogue Of Somatic Mutations In Cancer. Nucleic Acids Res. 2019, 47, D941–D947. [Google Scholar] [CrossRef]
- UniProt: A worldwide hub of protein knowledge. Nucleic Acids Res. 2019, 47, D506–D515. [CrossRef]
- Firth, H.V.; Richards, S.M.; Bevan, A.P.; Clayton, S.; Corpas, M.; Rajan, D.; Van Vooren, S.; Moreau, Y.; Pettett, R.M.; Carter, N.P. DECIPHER: Database of Chromosomal Imbalance and Phenotype in Humans Using Ensembl Resources. Am. J. Hum. Genet. 2009, 84, 524–533. [Google Scholar] [CrossRef]
- Stelzer, G.; Rosen, N.; Plaschkes, I.; Zimmerman, S.; Twik, M.; Fishilevich, S.; Stein, T.I.; Nudel, R.; Lieder, I.; Mazor, Y.; et al. The GeneCards Suite: From Gene Data Mining to Disease Genome Sequence Analyses. Curr. Protoc. Bioinform. 2016, 54, 1.30.1–1.30.33. [Google Scholar] [CrossRef]
- Staley, J.R.; Blackshaw, J.; Kamat, M.A.; Ellis, S.; Surendran, P.; Sun, B.B.; Paul, D.S.; Freitag, D.; Burgess, S.; Danesh, J.; et al. PhenoScanner: A database of human genotype–phenotype associations. Bioinformatics 2016, 32, 3207–3209. [Google Scholar] [CrossRef]
- Chandrashekar, D.S.; Bashel, B.; Balasubramanya, S.A.H.; Creighton, C.J.; Ponce-Rodriguez, I.; Chakravarthi, B.; Varambally, S. UALCAN: A Portal for Facilitating Tumor Subgroup Gene Expression and Survival Analyses. Neoplasia 2017, 19, 649–658. [Google Scholar] [CrossRef]
- Anaya, J. OncoRank: A pan-cancer method of combining survival correlations and its application to mRNAs, miRNAs, and lncRNAs. PeerJ Prepr. 2016, 4. [Google Scholar] [CrossRef]
- Aguirre-Gamboa, R.; Gomez-Rueda, H.; Martínez-Ledesma, E.; Martínez-Torteya, A.; Chacolla-Huaringa, R.; Rodriguez-Barrientos, A.; Tamez-Peña, J.G.; Treviño, V. SurvExpress: An Online Biomarker Validation Tool and Database for Cancer Gene Expression Data Using Survival Analysis. PLoS ONE 2013, 8, e74250. [Google Scholar] [CrossRef]
- Herwig, R.; Hardt, C.; Lienhard, M.; Kamburov, A. Analyzing and interpreting genome data at the network level with ConsensusPathDB. Nat. Protoc. 2016, 11, 1889–1907. [Google Scholar] [CrossRef] [PubMed]
- Ajami, K.; Abbott, C.A.; Obradovic, M.; Gysbers, V.; Kähne, T.; McCaughan, G.W.; Gorrell, M.D. Structural requirements for catalysis, expression, and dimerization in the CD26/DPIV gene family. Biochemistry 2003, 42, 694–701. [Google Scholar] [CrossRef] [PubMed]
- Karczewski, K.J.; Francioli, L.C.; Tiao, G.; Cummings, B.B.; Alföldi, J.; Wang, Q.; Collins, R.L.; Laricchia, K.M.; Ganna, A.; Birnbaum, D.P.; et al. Variation across 141,456 human exomes and genomes reveals the spectrum of loss-of-function intolerance across human protein-coding genes. bioRxiv 2019. [Google Scholar] [CrossRef]
- Dang, V.T.; Kassahn, K.S.; Marcos, A.E.; Ragan, M.A. Identification of human haploinsufficient genes and their genomic proximity to segmental duplications. Eur. J. Hum. Genet. 2008, 16, 1350–1357. [Google Scholar] [CrossRef]
- Itan, Y.; Shang, L.; Boisson, B.; Patin, E.; Bolze, A.; Moncada-Vélez, M.; Scott, E.; Ciancanelli, M.J.; Lafaille, F.G.; Markle, J.G.; et al. The human gene damage index as a gene-level approach to prioritizing exome variants. Proc. Natl. Acad. Sci. USA 2015, 112, 13615–13620. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef]
- Lay, A.J.; Zhang, H.E.; McCaughan, G.W.; Gorrell, M.D. Fibroblast activation protein in liver fibrosis. Front. Biosci. 2019, 24, 1–17. [Google Scholar]
- Cho, J.M.; Yang, E.H.; Quan, W.; Nam, E.H.; Cheon, H.G. Discovery of a novel fibroblast activation protein (FAP) inhibitor, BR103354, with anti-diabetic and anti-steatotic effects. Sci. Rep. 2020, 10, 21280. [Google Scholar] [CrossRef]
- Silva-Garcia, M.; Bolgi, O.; Ross, B.; Pilla, E.; Kari, V.; Killisch, M.; Stark, N.; Lenz, C.; Spitzner, M.; Gorrell, M.D.; et al. Dipeptidyl peptidase 9 triggers BRCA2 degradation by the N-degron pathway to promote DNA-damage repair. bioRxiv 2020. [Google Scholar] [CrossRef]
- Gall, M.G.; Chen, Y.; Vieira de Ribeiro, A.J.; Zhang, H.; Bailey, C.G.; Spielman, D.S.; Yu, D.M.; Gorrell, M.D. Targeted inactivation of dipeptidyl peptidase 9 enzymatic activity causes mouse neonate lethality. PLoS ONE 2013, 8, e78378. [Google Scholar] [CrossRef]
- Chen, Y.; Gall, M.G.; Zhang, H.; Keane, F.M.; McCaughan, G.W.; Yu, D.M.; Gorrell, M.D. Dipeptidyl peptidase 9 enzymatic activity influences the expression of neonatal metabolic genes. Exp. Cell Res. 2016, 342, 72–82. [Google Scholar] [CrossRef]
- Kim, M.; Minoux, M.; Piaia, A.; Kueng, B.; Gapp, B.; Weber, D.; Haller, C.; Barbieri, S.; Namoto, K.; Lorenz, T.; et al. DPP9 enzyme activity controls survival of mouse migratory tongue muscle progenitors and its absence leads to neonatal lethality due to suckling defect. Dev. Biol. 2017, 431, 297–308. [Google Scholar] [CrossRef]
- Harapas, C.R.; Robinson, K.; Lay, K.; Wong, J.; Raas-Rothschild, A.; Laohamonthonkul, P.; Boisson, B.; Drutman, S.B.; Gorrell, M.; Bonner, D.; et al. DPP9 deficiency: An inflammasomopathy which can be rescued by lowering NLRP1/IL-1 signaling. medRxiv 2021. [Google Scholar] [CrossRef]
- Duncan, B.B.; Highfill, S.L.; Qin, H.; Bouchkouj, N.; Larabee, S.; Zhao, P.; Woznica, I.; Liu, Y.; Li, Y.; Wu, W.; et al. A pan-inhibitor of DASH family enzymes induces immune-mediated regression of murine sarcoma and is a potent adjuvant to dendritic cell vaccination and adoptive T-cell therapy. J. Immunother. 2013, 36, 400–411. [Google Scholar] [CrossRef] [PubMed]
- Donahue, R.N.; Duncan, B.B.; Fry, T.J.; Jones, B.; Bachovchin, W.W.; Kiritsy, C.P.; Lai, J.H.; Wu, W.; Zhao, P.; Liu, Y.; et al. A pan inhibitor of DASH family enzymes induces immunogenic modulation and sensitizes murine and human carcinoma cells to antigen-specific cytotoxic T lymphocyte killing: Implications for combination therapy with cancer vaccines. Vaccine 2014, 32, 3223–3231. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.; Miller, G.T.; Jesson, M.I.; Watanabe, T.; Jones, B.; Wallner, B.P. PT-100, a small molecule dipeptidyl peptidase inhibitor, has potent antitumor effects and augments antibody-mediated cytotoxicity via a novel immune mechanism. Cancer Res. 2004, 64, 5471–5480. [Google Scholar] [CrossRef] [PubMed]
- Su, C.-W.; Lei, H.-J.; Chau, G.-Y.; Hung, H.-H.; Wu, J.-C.; Hsia, C.-Y.; Lui, W.-Y.; Su, Y.-H.; Wu, C.-W.; Lee, S.-D. The Effect of Age on the Long-term Prognosis of Patients with Hepatocellular Carcinoma After Resection Surgery: A Propensity Score Matching Analysis. Arch. Surg. 2012, 147, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Zhan, P.; Ji, Y.N.; Yu, L.K. TP53 mutation is associated with a poor outcome for patients with hepatocellular carcinoma: Evidence from a meta-analysis. Hepatobiliary Surg. Nutr. 2013, 2, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.H.; Indarto, D.; Doucet, A.; Pogson, L.D.; Pitman, M.R.; McNicholas, K.; Menz, R.I.; Overall, C.M.; Abbott, C.A. Identifying natural substrates for dipeptidyl peptidases 8 and 9 using terminal amine isotopic labeling of substrates (TAILS) reveals in vivo roles in cellular homeostasis and energy metabolism. J. Biol. Chem. 2013, 288, 13936–13949. [Google Scholar] [CrossRef]
- Finger, Y.; Habich, M.; Gerlich, S.; Urbanczyk, S.; van de Logt, E.; Koch, J.; Schu, L.; Lapacz, K.J.; Ali, M.; Petrungaro, C.; et al. Proteasomal degradation induced by DPP9-mediated processing competes with mitochondrial protein import. EMBO J. 2020, 39, e103889. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Maqsudi, S.; Rainczuk, A.; Duffield, N.; Lawrence, J.; Keane, F.M.; Justa-Schuch, D.; Geiss-Friedlander, R.; Gorrell, M.D.; Stephens, A.N. Identification of novel dipeptidyl peptidase 9 substrates by two-dimensional differential in-gel electrophoresis. FEBS J. 2015, 282, 3737–3757. [Google Scholar] [CrossRef] [PubMed]
- Han, R.; Wang, X.; Bachovchin, W.; Zukowska, Z.; Osborn, J.W. Inhibition of dipeptidyl peptidase 8/9 impairs preadipocyte differentiation. Sci. Rep. 2015, 5, 12348. [Google Scholar] [CrossRef] [PubMed]
- Wolk, A.; Gridley, G.; Svensson, M.; Nyrén, O.; McLaughlin, J.K.; Fraumeni, J.F.; Adami, H.-O. A prospective study of obesity and cancer risk (Sweden). Cancer Causes Control 2001, 12, 13–21. [Google Scholar] [CrossRef]
- Calle, E.E.; Rodriguez, C.; Walker-Thurmond, K.; Thun, M.J. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N. Engl. J. Med. 2003, 348, 1625–1638. [Google Scholar] [CrossRef]
- Sun, L.; Dai, J.J.; Hu, W.F.; Wang, J. Expression of toll-like receptors in hepatic cirrhosis and hepatocellular carcinoma. Genet. Mol. Res. 2016, 15. [Google Scholar] [CrossRef]
- Mohamed, F.E.; Al-Jehani, R.M.; Minogue, S.S.; Andreola, F.; Winstanley, A.; Olde Damink, S.W.; Habtesion, A.; Malagó, M.; Davies, N.; Luong, T.V.; et al. Effect of toll-like receptor 7 and 9 targeted therapy to prevent the development of hepatocellular carcinoma. Liver Int. 2015, 35, 1063–1076. [Google Scholar] [CrossRef]
- Seki, E.; Park, E.; Fujimoto, J. Toll-like receptor signaling in liver regeneration, fibrosis and carcinogenesis. Hepatol. Res. 2011, 41, 597–610. [Google Scholar] [CrossRef]
- Weinstein, J.N.; Collisson, E.A.; Mills, G.B.; Shaw, K.R.; Ozenberger, B.A.; Ellrott, K.; Shmulevich, I.; Sander, C.; Stuart, J.M. The Cancer Genome Atlas Pan-Cancer analysis project. Nat. Genet. 2013, 45, 1113–1120. [Google Scholar] [CrossRef]

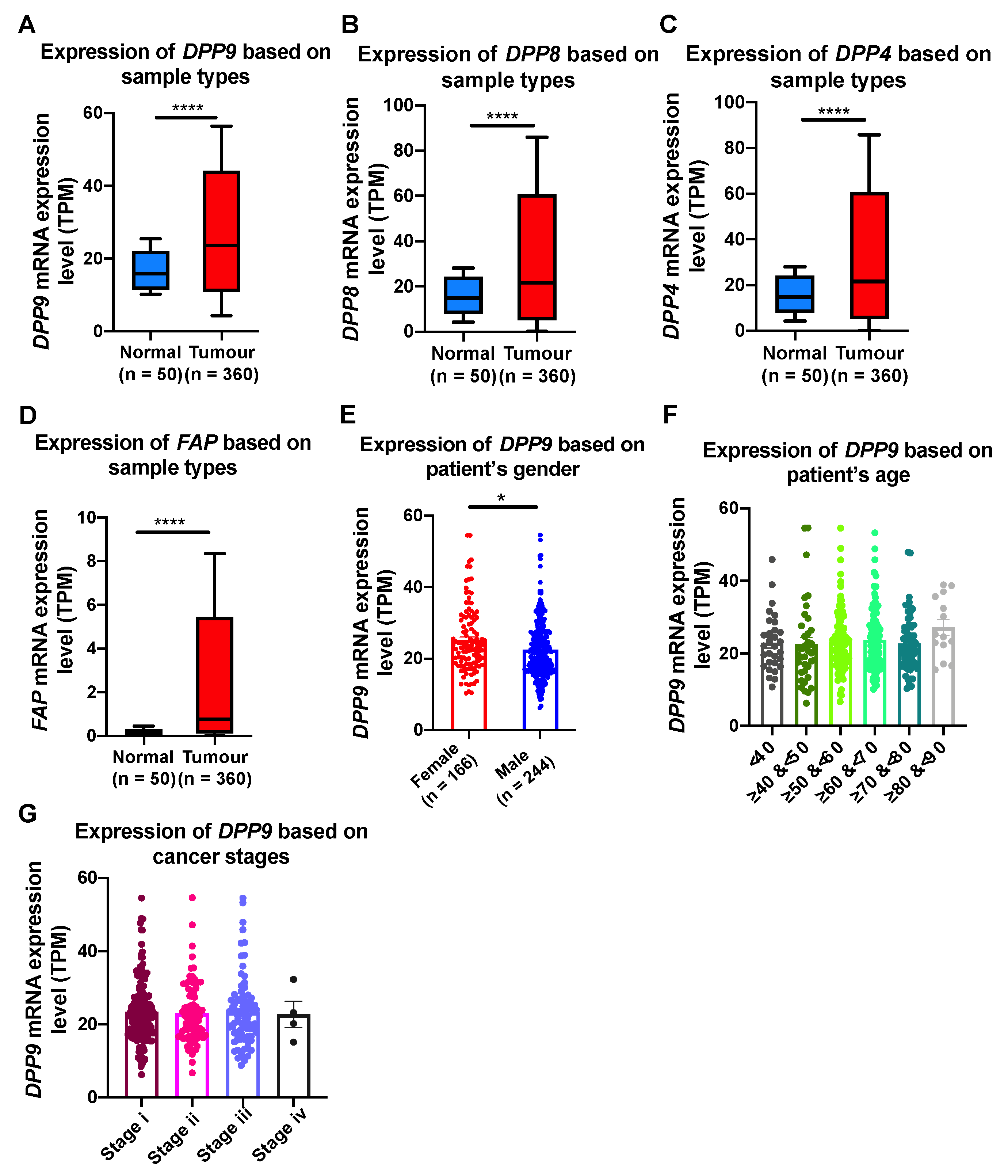
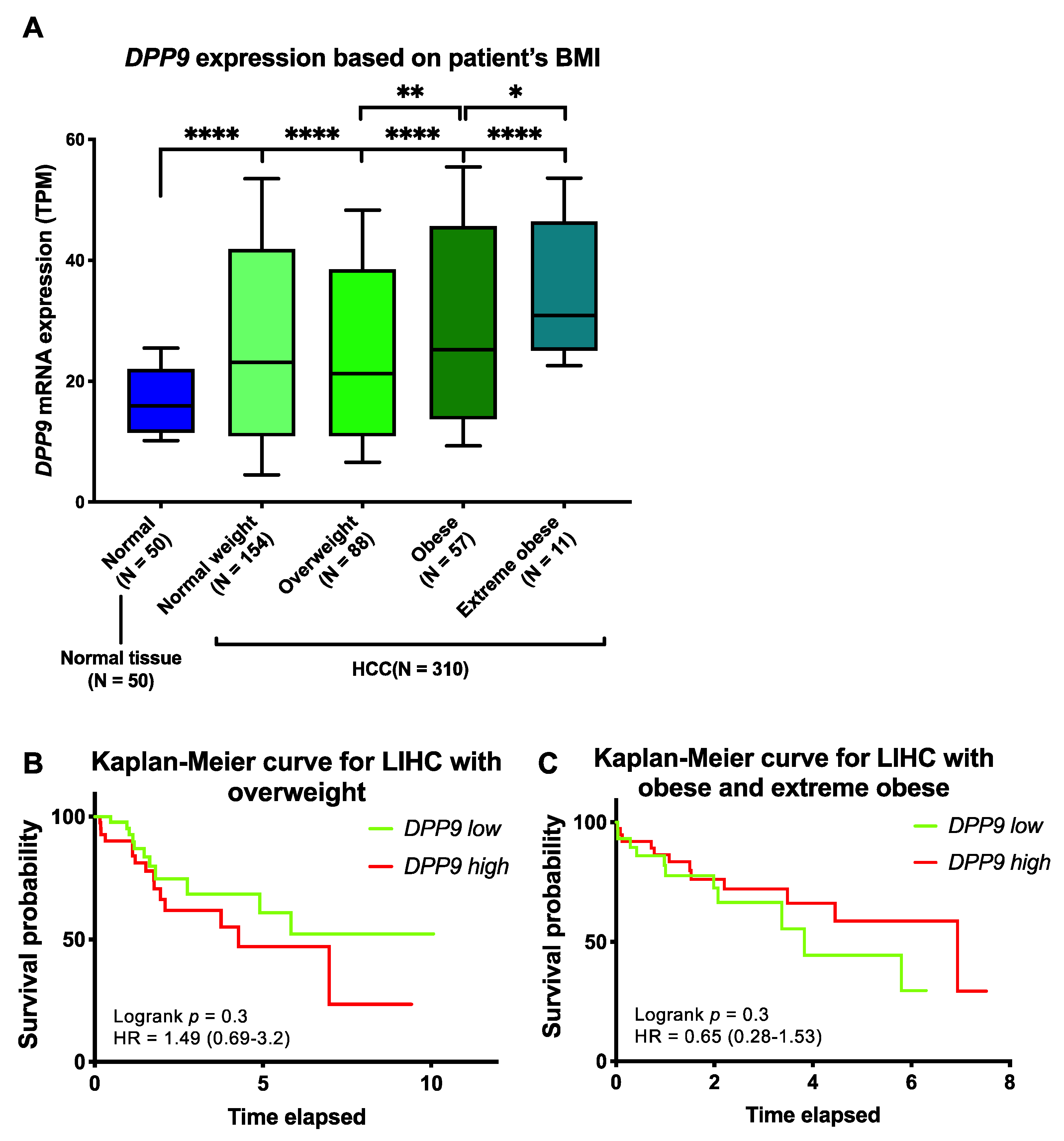
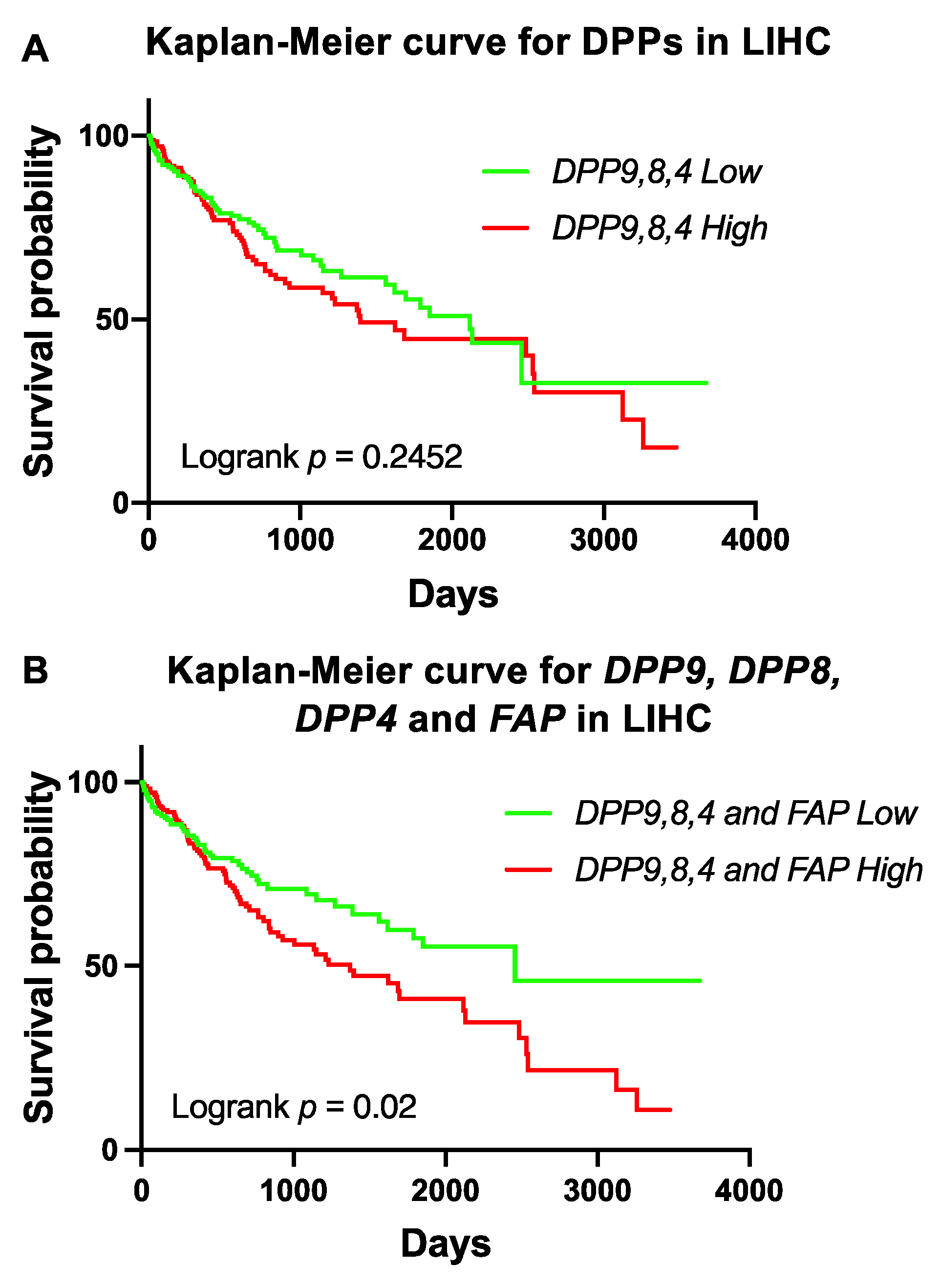
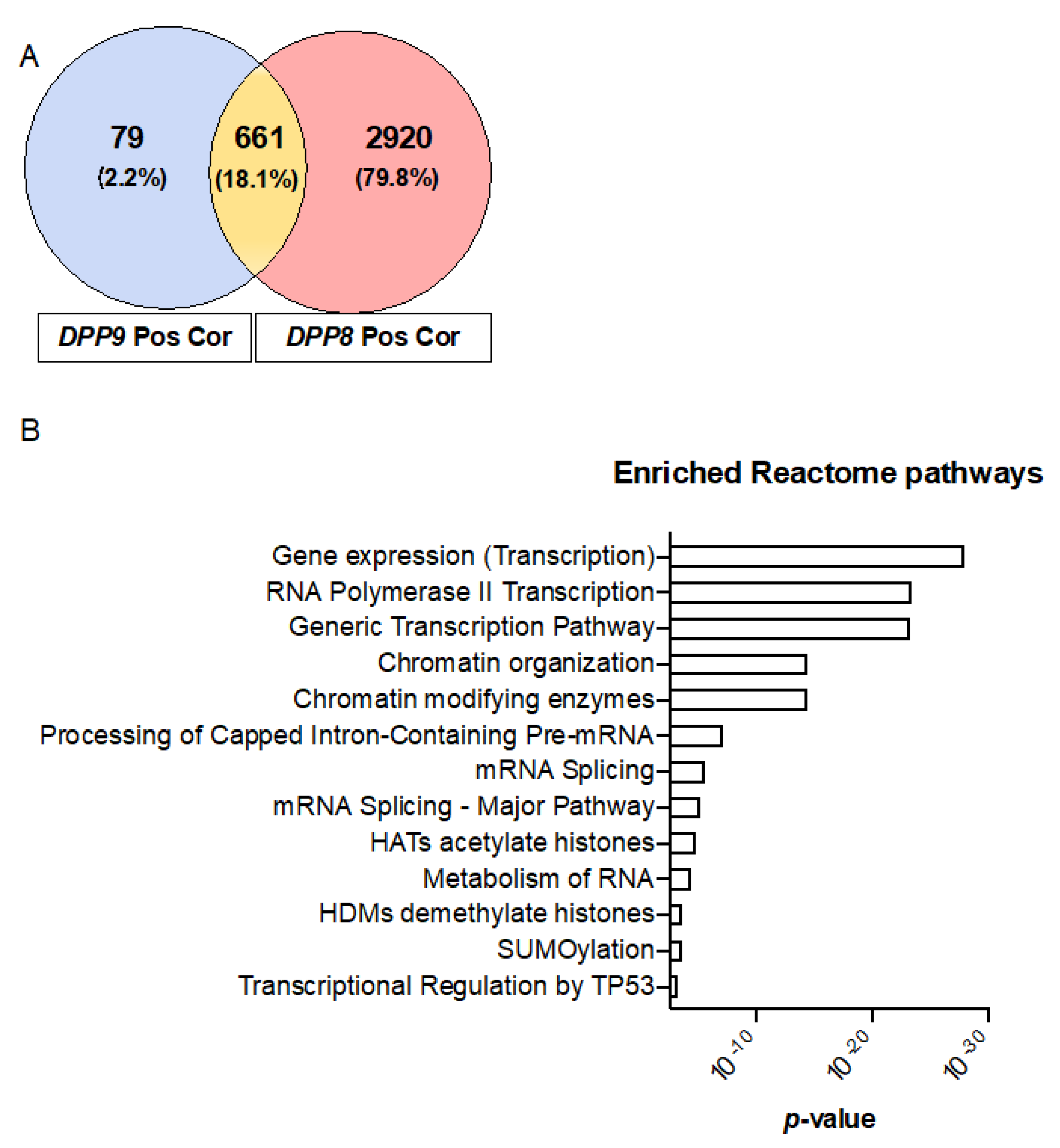

| Databases | URL | Data Obtained Date | Citation |
|---|---|---|---|
| The Genome Aggregation database (gnomAD) | https://gnomad.broadinstitute.org/ | 5 April 2020 | [34] |
| The Cancer Genome Atlas (TCGA) | https://portal.gdc.cancer.gov/ | 20 April 2020 | [35] |
| Catalogue of Somatic Mutation in Cancer (COSMIC) | https://cancer.sanger.ac.uk/cosmic | 15 June 2020 | [36] |
| UniProt Consortium | https://www.uniprot.org/ | 7 April 2020 | [37] |
| DatabasE of genomiC varIation and Phenotype in Humans using Ensembl Resources (DECIPHER) | https://decipher.sanger.ac.uk/ | 10 May 2020 | [38] |
| GeneCards human gene database | https://www.genecards.org/ | 12 May 2020 | [39] |
| Status | BMI Range |
|---|---|
| Normal weight | 18.5–25 |
| Overweight | 25–30 |
| Obese | 30–40 |
| Extreme obese | >40 |
| Position | rsID | Reference | Alternate | Protein Consequence | Annotation | Allele Frequency |
|---|---|---|---|---|---|---|
| 4682801 | rs766329416 | CCT | C | p.Gly794Val * 3 | frameshift variant | 7.89 × 10−6 |
| 4694820 | rs1360075526 | AGAAG | A | p.Phe456Ile * 34 | frameshift variant | 4.02 × 10−6 |
| 4714106 | rs1162979581 | GC | G | p.Arg100Pro * 58 | frameshift variant | 4.1 × 10−6 |
| 4683566 | rs1199678192 | G | A | p.Arg752 * | Stop gained | 4.02 × 10−6 |
| 4684713 | rs1165785000 | G | A | p.Arg714 * | Stop gained | 4.1 × 10−6 |
| 4676614 | rs1479652400 | C | A | p.Glu881 * | Stop gained | 1.25 × 10−5 |
| 4694808 | rs1365336720 | G | A | p.Gln461 * 1 | Stop gained | 4.02 × 10−6 |
| 4694825 | rs1418069794 | A | ATCTTCTATC | p.Asp454Ile455insArg * Lys | Stop gained | 4.02 × 10−6 |
| 4702672 | rs1466943995 | G | A | p.Gln276 * | Stop gained | 7.53 × 10−6 |
| 4705965 | rs779854175 | G | A | p.Arg111 * 1 | Stop gained | 1.2 × 10−5 |
| Position | Reference | Alternate | Protein Consequence | Annotation | Disease | Number of SSM Affected Cases and Frequency |
|---|---|---|---|---|---|---|
| 4685761_4685762insG | p.Asp633Gly * 51 | Frameshift | Stomach Adenocarcinoma | 1/440 (0.23%) | ||
| 4684670delT | p.Asn724Thr * 27 | Frameshift | Stomach Adenocarcinoma | 1/440 (0.23%) | ||
| 4679893delA | p.Phe843Ser * 10 | Frameshift | Colon Adenocarcinoma | 1/400 (0.25%) | ||
| 4676645_4676646insT | p.Asn866Lys * 16 | Frameshift | Uterine Corpus Endometrial Carcinoma | 1/530 (0.19%) | ||
| 4702132 | G | A | p.Arg303 * | Stop gained | Uterine Corpus Endometrial Carcinoma | 2/530 (0.38%) |
| 4704271 | C | A | p.Glu154 * 1 | Stop gained | Lung adenocarcinoma | 1/567 (0.18%) |
| 4695519 | C | T | p.Trp404 * | Stop gained | Uterine Corpus Endometrial Carcinoma | 1/530 (0.19%) |
| 4685745 | C | A | p.Glu638 * | Stop gained | Uterine Corpus Endometrial Carcinoma | 1/530 (0.19%) |
| CDS Mutation | AA Mutation | Legacy Mutation ID | Type | Disease | Number of SSM Affected Cases |
|---|---|---|---|---|---|
| c.43G > T | p.Gly15 * 1 | COSM3960221 | Nonsense | Lung squamous cell carcinoma | 2 |
| c.180G > A | p.Trp60 * | COSM9150037 | Nonsense | Plasma cell myeloma | 1 |
| c.306C > G | p.Tyr102 * | COSM4982488 | Nonsense | Esophagus squamous cell carcinoma | 3 |
| c.460G > T | p.Glu154 * 1 | COSM7761095 | Nonsense | Lung adenocarcinoma | 1 |
| c.864C > A | p.Cys288 * | COSM8582330 | Nonsense | Stomach carcinoma | 1 |
| c.907C > T | p.Arg303 * | COSM998442 | Nonsense | Endometrioid carcinoma Colon adenocarcinoma | 2 |
| c.976G > T | p.Glu326 * | COSM7365414 | Nonsense | Lung adenocarcinoma | 1 |
| c.1090G > T | p.Glu364 * | COSM8089586 | Nonsense | Malignant melanoma | 1 |
| c.1385C > G | p.Ser462 * 1 | COSM7365412 | Nonsense | Lung adenocarcinoma | 1 |
| c.2584C > T | p.Gln862 * | COSM7322837 | Nonsense | Biliary tract adenocarcinoma | 1 |
| c.2668G > T | p.Glu890 * | COSM6866899 | Nonsense | Skin squamous cell carcinoma | 1 |
| Gene | o:e Ratio of LoF | pLI | Haploinsufficiency Score | Tolerance Percentile | GDI |
|---|---|---|---|---|---|
| DPP9 | 0.17 (0.1–0.3) | 0.99 | 74.03 | 7.37% | 3.14 |
| DPP8 | 0.14 (0.08–0.27) | 1 | 24.90 | 26.2% | 1.79 |
| DPP4 | 0.44 (0.31–0.62) | 0 | 8.9 | 15% | 2.81 |
| FAP | 0.86 (0.68–1.1) | 0 | 8.57 | 10.2% | 2.77 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, J.C.; Emran, A.A.; Endaya, J.M.; McCaughan, G.W.; Gorrell, M.D.; Zhang, H.E. DPP9: Comprehensive In Silico Analyses of Loss of Function Gene Variants and Associated Gene Expression Signatures in Human Hepatocellular Carcinoma. Cancers 2021, 13, 1637. https://doi.org/10.3390/cancers13071637
Huang JC, Emran AA, Endaya JM, McCaughan GW, Gorrell MD, Zhang HE. DPP9: Comprehensive In Silico Analyses of Loss of Function Gene Variants and Associated Gene Expression Signatures in Human Hepatocellular Carcinoma. Cancers. 2021; 13(7):1637. https://doi.org/10.3390/cancers13071637
Chicago/Turabian StyleHuang, Jiali Carrie, Abdullah Al Emran, Justine Moreno Endaya, Geoffrey W. McCaughan, Mark D. Gorrell, and Hui Emma Zhang. 2021. "DPP9: Comprehensive In Silico Analyses of Loss of Function Gene Variants and Associated Gene Expression Signatures in Human Hepatocellular Carcinoma" Cancers 13, no. 7: 1637. https://doi.org/10.3390/cancers13071637
APA StyleHuang, J. C., Emran, A. A., Endaya, J. M., McCaughan, G. W., Gorrell, M. D., & Zhang, H. E. (2021). DPP9: Comprehensive In Silico Analyses of Loss of Function Gene Variants and Associated Gene Expression Signatures in Human Hepatocellular Carcinoma. Cancers, 13(7), 1637. https://doi.org/10.3390/cancers13071637







