Telomerase Regulation: A Role for Epigenetics
Simple Summary
Abstract
1. Telomeres and Telomerase Regulation
2. Transcription Factors and Regulation of TERT Promoter
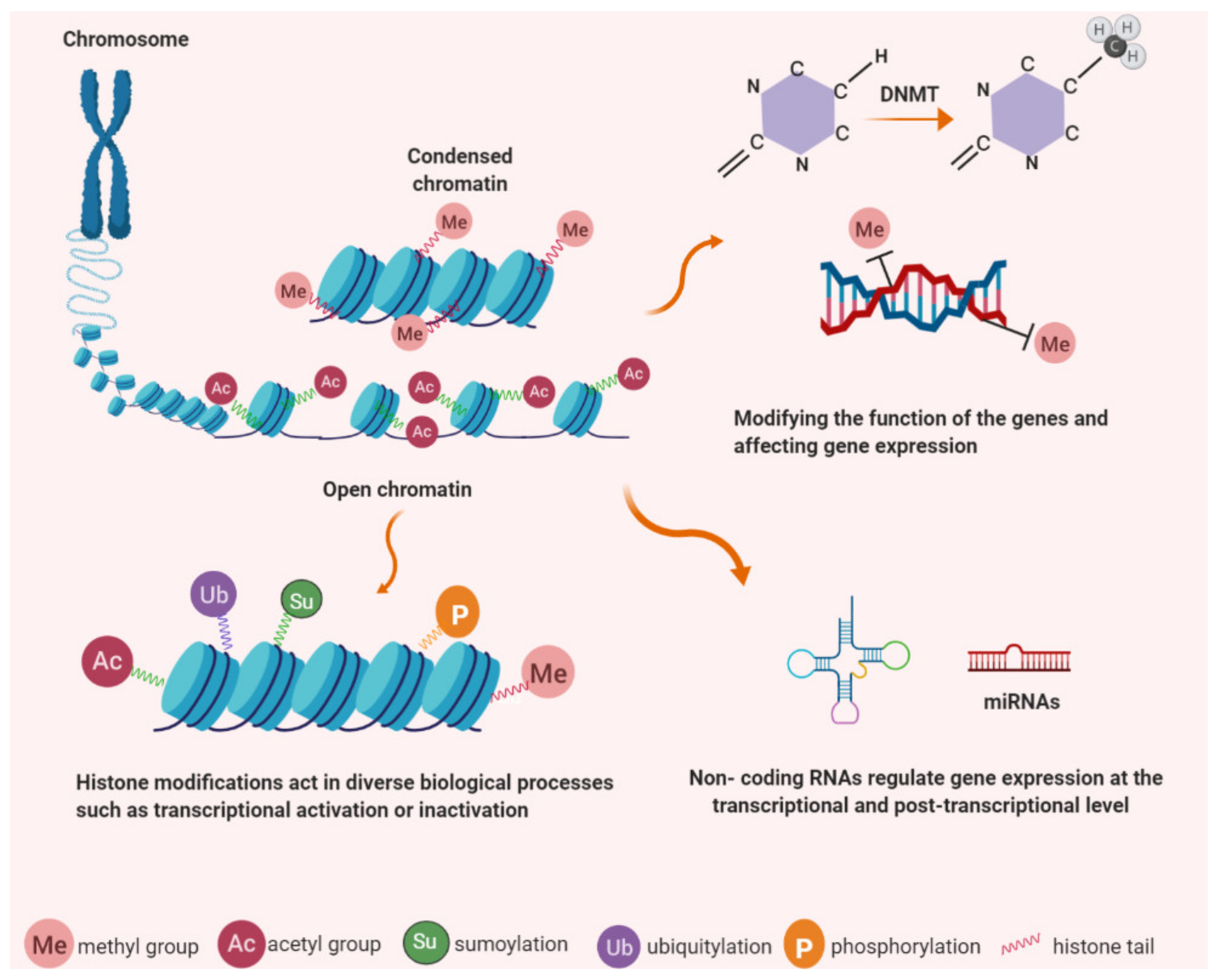
3. Epigenetic Control
4. TERT Expression Regulation by Epigenetic Mechanisms
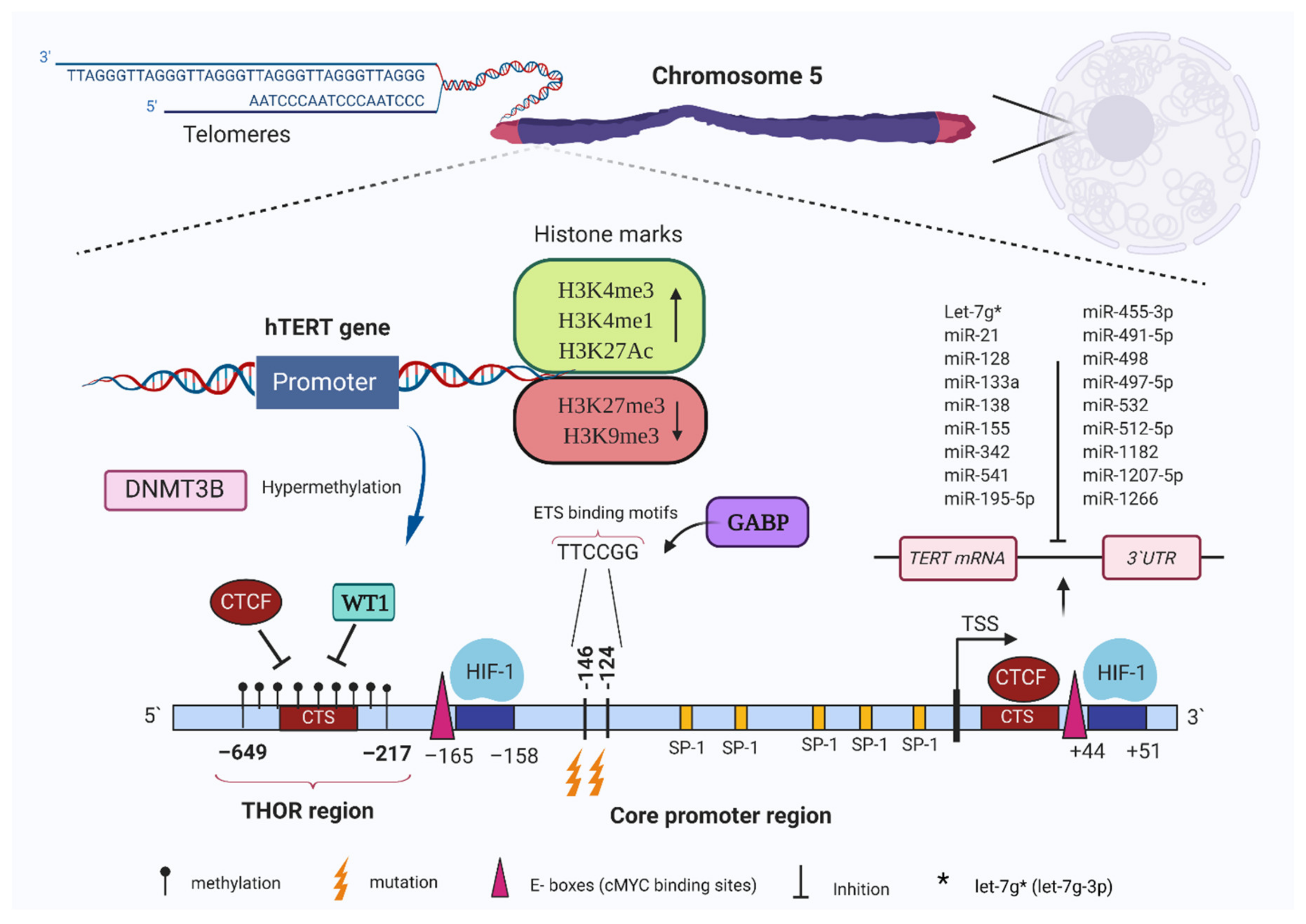
5. TERT Promoter Methylation in Cancer Cells
6. Transcriptional Regulation of Telomerase by Histone Modifications
| Histone Modification | Associated Mechanism | Cell Type | Reference |
|---|---|---|---|
| H3K4Me3, H3K9Ac, H3Ac H4Ac (Active marks) H3K9Me3, H4K20Me (Repressive) | The lowest expression of repressive marks but the highest levels of active marks in the cell lines expressing high levels of TERT Higher levels of repressive marks at the promoter in ALT cell lines | WI38, SUSM-1, KMST-6, and WI38-SV40 (ALT cells lines) C33a, A2780, and 5637 (high TERT expression) | [163] |
| H3K4Me2/3 (Active marks) | Increase occupancy of MYC and SP1 on the TERT promoter | HCT116 and L1236 cells | [156] |
| H3K9Ac, H3K4Me2 (Active marks) H3K9me3, H3K27me3 (Repressive) | Both active and inactive chromatin signs present across the TERT promoter | RKO, SW480, HCT 116, MCF7, and VA13 cells | [136] |
| H3K9Ac (Active marks) H3K9Me3 (Repressive) | Depletion of SIRT1 induces H3K9Ac and reduce H3K9Me3 at TERT | Hepatocellular carcinoma | [168] |
| H3K4Me2/3 (Active marks) H3K27me3 (Repressive) | Mutant TERT promoters exhibit the H3K4me2/3 and recruit the GABPA/B1 transcription factor in multiple cancer cell lines, while the wild-type allele exhibits the H3K27me3 | HepG2, SNU-475, SNU-423, UMUC3 and T24 | [76] |
| H3K4Me3, H3K9Ac (Active marks) | Enrichment of both H3K4Me3 and H3K9Ac in the proximal TERT promoter region in mutant cell lines | BLM, A375, T98G (bearing -146 C>T mutation), and U251 (bearing −124 C>T mutation) | [161] |
| H3K4Me3 (Active mark) | Increased H3K4Me3 allows MYC/MAX binding to E-boxes in the TERT promoter | E6E7 IDH1mut pre- and postcrisis cells | [160] |
| H3K27Me1, H3K27ac (Active marks) H3K27me3 (Repressive) | Enrichment of active marks in the juxtaposed to the TERT promoter | NIH Roadmap Epigenomics Consortium (111 epigenomes) | [15] |
| H3K4me3, H4Ac (Active marks) H3K9Me3 H3K27me3 (Repressive) | TERT silencing during differentiation accompanied by increases of repressive marks | Human embryonic stem cell | [159] |
| H3K4me3 (Active mark) H3K27me3, HP-1α (Repressive) | H3K4me3 is enriched in iPSCs, whereas H3K27me3 and HP-1α levels are higher in somatic cells | Fetal lung fibroblast (MRC) and MRC-iPSCs | [135] |
| H3K4Me3, H3Ac (Active marks) H3K27Me3 (Repressive) | Active marks are enriched at TERT core promoter | Acute promyelocytic leukemia cells NB4-LR1 and NB4-LR1 | [169] |
7. Regulation of TERT via Non-Coding RNAs
| miRNAs | Tissue Type | Mode of Action | Assay | References |
|---|---|---|---|---|
| let-7g * | Idiopathic pulmonary fibrosis | Interaction with 3′UTR of TERT to reduce telomerase expression | Reporter Assays | [185] |
| miR-21 | Glioblastoma | Decreases TERT through a STAT3-dependent manner | Reporter Assays and CHIP | [59] |
| miR-128 | HeLa, lung cancer, colon cancer and pancreatic cancer cells | Interacts directly with the coding sequence of TERT and reduces the mRNA and protein levels | Reporter Assays, Immunopurification strategy | [178] |
| miR-133a, miR-342, miR-541 | Jurkat cells | Interaction with the 3′UTR of TERT to reduce expression | Reporter Assays | [186] |
| miR-138 | Thyroid carcinoma cell lines | Interacts with 3′UTR of TERT and reduces protein expression | Reporter Assays | [187] |
| miR-498 | Ovarian and breast cancers | Targets the 3′UTR of TERT mRNA and decreased its expression | Ribonuclease Protection Assays, Reporter Assay | [188,189] |
| miR-1182 | Bladder, ovarian and gastric cancers | Targets 3’UTR and ORF1 of TERT and reduces protein expression | Reporter Assay Western blotting | [190,191,192] |
| miR-1207-5p, miR-1266 | Gastric cancer | Interacts with the 3’ UTR of TERT and suppresses TERT | Reporter Assay | [193] |
| miR-497-5p, miR-195-5p, miR-455-3p | Melanoma cells | Targets TERT and decrease expression at both mRNA and protein levels | Dual-luciferase reporter assay | [194] |
| miR-155 | Bladder cancer | Association of lower expression of miR-155 and higher expression of TERT | RT-PCR | [195] |
| miR-512-5p | Head and neck squamous cell carcinoma | Targets 3’ UTR and downregulates TERT expression and telomerase activity | Dual-luciferase reporter assay | [196] |
| miR-661 | Glioma cells | Targets 3’ UTR and silence TERT | Reporter Assay | [197] |
| miR-491-5p | Cervical cancer | Targets 3’ UTR of TERT and decreases protein expression | Dual-luciferase reporter assay | [176] |
8. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| hESC | Human embryonic stem cell |
| hiPSC | Human-induced pluripotent stem cells |
| TERC | telomerase RNA component |
| TERT | telomerase reverse transcriptase |
| CpG | Cytosines followed by guanine residues |
| 5mC | 5-methylcytosine |
| HIF1 | Hypoxia-inducible factor 1 |
| CTCF | CCCTC-binding factor |
| DNMT | DNA methyltransferase |
| HAT | Histone acetyltransferase |
| HDAC | Histone deacetylase |
| SMYD3 | SET and MYND domain-containing protein 3 |
| H3K4 | Lysine 4 on histone 3 |
| miRNA | MicroRNA |
| TRAP | Telomeric repeat amplification protocol |
| EBs | Embryoid bodies |
| HCC | Hepatocellular carcinoma |
| TERRA | Telomere repeat-containing RNA |
| TRF2 | Telomeric repeat-binding factors 2 |
| ALT | Alternative lengthening of telomeres |
| UTSS | Upstream of the transcription start site |
| THOR | TERT hypermethylated oncological region |
| Tet1 | Ten-eleven translocation methylcytosine dioxygenase 1 |
| SP1 | Specificity Protein |
| NF-κB | Nuclear Factor κB |
| STAT3 | Signal Transducer and Activator of Transcription 3 |
| Pax | Paired Box Proteins |
| ER | Oestrogen Receptor |
| WT | Wilms’ tumour 1 suppressor |
References
- Muller, H.J. The Remaking of Chromosomes. Collect. Net. 1938, 13, 181–198. [Google Scholar]
- McClintock, B. The Stability of Broken Ends of Chromosomes in Zea Mays. Genetics 1941, 26, 234–282. [Google Scholar] [PubMed]
- Blackburn, E.H.; Gall, J.G. A Tandemly Repeated Sequence at the Termini of the Extrachromosomal Ribosomal RNA Genes in Tetrahymena. J. Mol. Biol. 1978, 120, 33–53. [Google Scholar] [CrossRef]
- Greider, C.W.; Blackburn, E.H. Identification of a Specific Telomere Terminal Transferase Activity in Tetrahymena Extracts. Cell 1985, 43, 405–413. [Google Scholar] [CrossRef]
- Morin, G.B. The Human Telomere Terminal Transferase Enzyme Is a Ribonucleoprotein That Synthesizes TTAGGG Repeats. Cell 1989, 59, 521–529. [Google Scholar] [CrossRef]
- Harrington, L.; McPhail, T.; Mar, V.; Zhou, W.; Oulton, R.; Bass, M.B.; Arruda, I.; Robinson, M.O. A Mammalian Telomerase-Associated Protein. Science 1997, 275, 973–977. [Google Scholar] [CrossRef] [PubMed]
- Holt, S.E.; Aisner, D.L.; Baur, J.; Tesmer, V.M.; Dy, M.; Ouellette, M.; Trager, J.B.; Morin, G.B.; Toft, D.O.; Shay, J.W.; et al. Functional Requirement of P23 and Hsp90 in Telomerase Complexes. Genes Dev. 1999, 13, 817–826. [Google Scholar] [CrossRef]
- Nakamura, T.M.; Morin, G.B.; Chapman, K.B.; Weinrich, S.L.; Andrews, W.H.; Lingner, J.; Harley, C.B.; Cech, T.R. Telomerase Catalytic Subunit Homologs from Fission Yeast and Human. Science 1997, 277, 955–959. [Google Scholar] [CrossRef] [PubMed]
- Autexier, C.; Lue, N.F. The Structure and Function of Telomerase Reverse Transcriptase. Annu. Rev. Biochem. 2006, 75, 493–517. [Google Scholar] [CrossRef]
- Chai, W.; Ford, L.P.; Lenertz, L.; Wright, W.E.; Shay, J.W. Human Ku70/80 Associates Physically with Telomerase through Interaction with HTERT. J. Biol. Chem. 2002, 277, 47242–47247. [Google Scholar] [CrossRef]
- Ting, N.S.Y.; Yu, Y.; Pohorelic, B.; Lees-Miller, S.P.; Beattie, T.L. Human Ku70/80 Interacts Directly with HTR, the RNA Component of Human Telomerase. Nucleic Acids Res. 2005, 33, 2090–2098. [Google Scholar] [CrossRef] [PubMed]
- Avilion, A.A.; Piatyszek, M.A.; Gupta, J.; Shay, J.W.; Bacchetti, S.; Greider, C.W. Human Telomerase RNA and Telomerase Activity in Immortal Cell Lines and Tumor Tissues. Cancer Res. 1996, 56, 645–650. [Google Scholar] [PubMed]
- Ito, H.; Kyo, S.; Kanaya, T.; Takakura, M.; Koshida, K.; Namiki, M.; Inoue, M.; Gynecology, O. Detection of Human Telomerase Reverse Transcriptase Messenger RNA in Voided Urine Samples as a Useful Diagnostic Tool for Bladder Cancer. Clin. Cancer Res. 1998, 4, 2807–2810. [Google Scholar]
- Huang, F.W.; Hodis, E.; Xu, M.J.; Kryukov, G.V.; Chin, L.; Garraway, L.A. Highly Recurrent TERT Promoter Mutations in Human Melanoma. Science 2013, 339, 957–959. [Google Scholar] [CrossRef]
- Barthel, F.P.; Wei, W.; Tang, M.; Martinez-Ledesma, E.; Hu, X.; Amin, S.B.; Akdemir, K.C.; Seth, S.; Song, X.; Wang, Q.; et al. Systematic Analysis of Telomere Length and Somatic Alterations in 31 Cancer Types. Nat. Genet. 2017, 49, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, M.; Kojima, M.; Ueda, Y.; Kurihara, S.; Hiyama, E. Telomere Biology Including TERT Rearrangements in Neuroblastoma: A Useful Indicator for Surgical Treatments. J. Pediatr. Surg. 2016, 51, 2080–2085. [Google Scholar] [CrossRef] [PubMed]
- Guilleret, I.; Yan, P.; Grange, F.; Braunschweig, R.; Bosman, F.T.; Benhattar, J. Hypermethylation of the Human Telomerase Catalytic Subunit (HTERT) Gene Correlates with Telomerase Activity. Int. J. Cancer 2002, 101, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Lewis, K.A.; Tollefsbol, T.O. Regulation of the Telomerase Reverse Transcriptase Subunit through Epigenetic Mechanisms. Front. Genet. 2016, 7, 1–12. [Google Scholar] [CrossRef]
- Cong, Y.-S.; Wright, W.E.; Shay, J.W. Human Telomerase and Its Regulation. Microbiol. Mol. Biol. Rev. 2002, 66, 407–425. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Ludlow, A.T.; Min, J.; Robin, J.D.; Stadler, G.; Mender, I.; Lai, T.P.; Zhang, N.; Wright, W.E.; Shay, J.W. Regulation of the Human Telomerase Gene TERT by Telomere Position Effect—Over Long Distances (TPE-OLD): Implications for Aging and Cancer. PLoS Biol. 2016, 14. [Google Scholar] [CrossRef]
- Leão, R.; Apolónio, J.D.; Lee, D.; Figueiredo, A.; Tabori, U.; Castelo-Branco, P. Mechanisms of Human Telomerase Reverse Transcriptase (HTERT) Regulation: Clinical Impacts in Cancer. J. Biomed. Sci. 2018, 25, 22. [Google Scholar] [CrossRef]
- Moyzis, R.K.; Buckingham, J.M.; Cram, L.S.; Dani, M.; Deaven, L.L.; Jones, M.D.; Meyne, J.; Ratliff, R.L.; Wu, J.R. A Highly Conserved Repetitive DNA Sequence, (TTAGGG)n, Present at the Telomeres of Human Chromosomes. Proc. Natl. Acad. Sci. USA 1988, 85, 6622–6626. [Google Scholar] [CrossRef] [PubMed]
- Greider, C.W. Telomeres Do D-Loop-T-Loop. Cell. 1999, 97, 419–422. [Google Scholar] [CrossRef]
- Griffith, J.D.; Comeau, L.; Rosenfield, S.; Stansel, R.M.; Bianchi, A.; Moss, H.; De Lange, T. Mammalian Telomeres End in a Large Duplex Loop. Cell 1999, 97, 503–514. [Google Scholar] [CrossRef]
- De Lange, T. Shelterin: The Protein Complex That Shapes and Safeguards Human Telomeres. In Genes and Development; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2005; pp. 2100–2110. [Google Scholar] [CrossRef]
- Rodriguez, R.; Müller, S.; Yeoman, J.A.; Trentesaux, C.; Riou, J.-F.; Balasubramanian, S. A Novel Small Molecule That Alters Shelterin Integrity and Triggers a DNA-Damage Response at Telomeres. J. Am. Chem. Soc. 2008, 130, 15758–15759. [Google Scholar] [CrossRef]
- Jones, M.; Bisht, K.; Savage, S.A.; Nandakumar, J.; Keegan, C.E.; Maillard, I. The Shelterin Complex and Hematopoiesis. J. Clin. Investig. 2016, 1621–1629. [Google Scholar] [CrossRef]
- Palm, W.; de Lange, T. How Shelterin Protects Mammalian Telomeres. Annu. Rev. Genet. 2008, 42, 301–334. [Google Scholar] [CrossRef] [PubMed]
- Klapper, W.; Parwaresch, R.; Krupp, G. Telomere Biology in Human Aging and Aging Syndromes. Mech. Ageing Dev. 2001, 122, 695–712. [Google Scholar] [CrossRef]
- Takubo, K.; Izumiyama-Shimomura, N.; Honma, N.; Sawabe, M.; Arai, T.; Kato, M.; Oshimura, M.; Nakamura, K.-I. Telomere Lengths Are Characteristic in Each Human Individual. Exp. Gerontol. 2002, 37, 523–531. [Google Scholar] [CrossRef]
- Lingner, J.; Cooper, J.P.; Cech, T.R. Telomerase and DNA End Replication: No Longer a Lagging Strand Problem? Science 1995, 269, 1533–1534. [Google Scholar] [CrossRef] [PubMed]
- Hayflick, L. The Limited in Vitro Lifetime of Human Diploid Cell Strains. Exp. Cell Res. 1965, 37, 614–636. [Google Scholar] [CrossRef]
- Allday, M.J.; Inman, G.J.; Crawford, D.H.; Farrell, P.J. DNA Damage in Human B Cells Can Induce Apoptosis, Proceeding from G1/S When P53 Is Transactivation Competent and G2/M When It Is Transactivation Defective. EMBO J. 1995, 14, 4994–5005. [Google Scholar] [CrossRef] [PubMed]
- Canman, C.E.; Lim, D.S.; Cimprich, K.A.; Taya, Y.; Tamai, K.; Sakaguchi, K.; Appella, E.; Kastan, M.B.; Siliciano, J.D. Activation of the ATM Kinase by Ionizing Radiation and Phosphorylation of P53. Science 1998, 281, 1677–1679. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, G.I.; Edwards, C.D.; Ewen, M.E.; Rollins, B.J. P16 INK4A Participates in a G 1 Arrest Checkpoint in Response to DNA Damage. Mol. Cell. Biol. 1998, 18, 378–387. [Google Scholar] [CrossRef]
- Janknecht, R. On the Road to Immortality: HTERT Upregulation in Cancer Cells. FEBS Lett. 2004, 564, 9–13. [Google Scholar] [CrossRef]
- Januszkiewicz, D.; Wysoki, J.; Lewandowski, K.; Pernak, M.; Nowicka, K.; Rembowska, J.; Nowak, J. Lack of Correlation between Telomere Length and Telomerase Activity and Expression in Leukemic Cells. Int. J. Mol. Med. 2003, 12, 935–938. [Google Scholar] [CrossRef] [PubMed]
- Hornsby, P.J. Telomerase and the Aging Process. Exp. Gerontol. 2007, 42, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.W.; Piatyszek, M.A.; Prowse, K.R.; Harley, C.B.; West, M.D.; Ho, P.L.; Coviello, G.M.; Wright, W.E.; Weinrich, S.L.; Shay, J.W. Specific Association of Human Telomerase Activity with Immortal Cells and Cancer. Science 1994, 266, 2011–2015. [Google Scholar] [CrossRef] [PubMed]
- Bryan, T.M.; Englezou, A.; Gupta, J.; Bacchetti, S.; Reddel, R.R. Telomere Elongation in Immortal Human Cells without Detectable Telomerase Activity. EMBO J. 1995, 14, 4240–4248. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.M.; Brenneman, M.A.; Goodwin, E.H. Frequent Recombination in Telomeric DNA May Extend the Proliferative Life of Telomerase-Negative Cells. Nucleic Acids Res. 2004, 32, 3743–3751. [Google Scholar] [CrossRef] [PubMed]
- Grobelny, J.V.; Godwin, A.K.; Broccoli, D. ALT-Associated PML Bodies Are Present in Viable Cells and Are Enriched in Cells in the G2/M Phase of the Cell Cycle. J. Cell Sci. 2000, 113, 4577–4585. [Google Scholar] [PubMed]
- Yeager, T.R.; Neumann, A.A.; Englezou, A.; Huschtscha, L.I.; Noble, J.R.; Reddel, R.R. Telomerase-Negative Immortalized Human Cells Contain a Novel Type of Promyelocytic Leukemia (PML) Body. Cancer Res. 1999, 59, 4175–4179. [Google Scholar] [PubMed]
- Liu, L.; Bailey, S.M.; Okuka, M.; Muñoz, P.; Li, C.; Zhou, L.; Wu, C.; Czerwiec, E.; Sandler, L.; Seyfang, A.; et al. Telomere Lengthening Early in Development. Nat. Cell Biol. 2007, 9, 1436–1441. [Google Scholar] [CrossRef]
- Shay, J.W.; Wright, W.E. Role of Telomeres and Telomerase in Cancer. Semin. Cancer Biol. 2011, 21, 349–353. [Google Scholar] [CrossRef]
- Leem, S.H.; Londoño-Vallejo, J.A.; Kim, J.H.; Bui, H.; Tubacher, E.; Solomon, G.; Park, J.E.; Horikawa, I.; Kouprina, N.; Barrett, J.C.; et al. The Human Telomerase Gene: Complete Genomic Sequence and Analysis of Tandem Repeat Polymorphisms in Intronic Regions. Oncogene 2002, 21, 769–777. [Google Scholar] [CrossRef][Green Version]
- Robin, J.D.; Ludlow, A.T.; Batten, K.; Magdinier, F.; Stadler, G.; Wagner, K.R.; Shay, J.W.; Wright, W.E. Telomere Position Effect: Regulation of Gene Expression with Progressive Telomere Shortening over Long Distances. Genes Dev. 2014, 28, 2464–2476. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Shay, J.W. Long-Range Telomere Regulation of Gene Expression: Telomere Looping and Telomere Position Effect over Long Distances (TPE-OLD). Differentiation 2018, 99, 1–9. [Google Scholar] [CrossRef]
- Horikawa, I.; Cable, P.L.; Afshari, C.; Barrett, J.C. Cloning and Characterization of the Promoter Region of Human Telomerase Reverse Transcriptase Gene. Cancer Res. 1999, 59, 826–830. [Google Scholar]
- Cong, Y.S.; Wen, J.; Bacchetti, S. The Human Telomerase Catalytic Subunit HTERT: Organization of the Gene and Characterization of the Promoter. Hum. Mol. Genet. 1999, 8, 137–142. [Google Scholar] [CrossRef]
- Ramlee, M.K.; Wang, J.; Toh, W.X.; Li, S. Transcription Regulation of the Human Telomerase Reverse Transcriptase (HTERT) Gene. Genes 2016, 7, 50. [Google Scholar] [CrossRef]
- Grandori, C.; Cowley, S.M.; James, L.P.; Eisenman, R.N. The Myc/Max/Mad Network and the Transcriptional Control of Cell Behavior. Annu. Rev. Cell Dev. Biol. 2000, 16, 653–699. [Google Scholar] [CrossRef] [PubMed]
- Kyo, S.; Takakura, M.; Taira, T.; Kanaya, T.; Itoh, H.; Yutsudo, M.; Ariga, H.; Inoue, M. Sp1 Cooperates with C-Myc to Activate Transcription of the Human Telomerase Reverse Transcriptase Gene (HTERT). Nucleic Acids Res. 2000, 28, 669–677. [Google Scholar] [CrossRef]
- Yu, J.; Yuan, X.; Sjöholm, L.; Liu, T.; Kong, F.; Ekström, T.J.; Björkholm, M.; Xu, D. Telomerase Reverse Transcriptase Regulates DNMT3B Expression/Aberrant DNA Methylation Phenotype and AKT Activation in Hepatocellular Carcinoma. Cancer Lett. 2018, 434, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Won, J.; Yim, J.; Kim, T.K. Sp1 and Sp3 Recruit Histone Deacetylase to Repress Transcription of Human Telomerase Reverse Transcriptase (HTERT) Promoter in Normal Human Somatic Cells. J. Biol. Chem. 2002, 277, 38230–38238. [Google Scholar] [CrossRef]
- Oh, S.; Song, Y.H.; Yim, J.; Kim, T.K. Identification of Mad as a Repressor of the Human Telomerase (HTERT) Gene. Oncogene 2000, 19, 1485–1490. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.H.; Liu, B.R.; Qu, B.; Xing, H.; Gao, S.L.; Yin, J.M.; Wang, X.F.; Cheng, Y.Q. Silencing STAT3 May Inhibit Cell Growth through Regulating Signaling Pathway, Telomerase, Cell Cycle, Apoptosis and Angiogenesis in Hepatocellular Carcinoma: Potential Uses for Gene Therapy. Neoplasma 2011, 58, 158–164. [Google Scholar] [CrossRef]
- Chung, S.S.; Aroh, C.; Vadgama, J.V. Constitutive Activation of STAT3 Signaling Regulates HTERT and Promotes Stem Cell-like Traits in Human Breast Cancer Cells. PLoS ONE 2013, 8, 83971. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Y.; Sun, G.; Luo, H.; Wang, X.F.; Lan, F.M.; Yue, X.; Fu, L.S.; Pu, P.Y.; Kang, C.S.; Liu, N.; et al. Mir-21 Modulates Htert through a Stat3-Dependent Manner on Glioblastoma Cell Growth. CNS Neurosci. Ther. 2012, 18, 722–728. [Google Scholar] [CrossRef]
- Yamada, O.; Ozaki, K.; Akiyama, M.; Kawauchi, K. JAK-STAT and JAK-PI3K-MTORC1 Pathways Regulate Telomerase Transcriptionally and Posttranslationally in ATL Cells. Mol. Cancer Ther. 2012, 11, 1112–1121. [Google Scholar] [CrossRef] [PubMed]
- Yamada, O.; Ozaki, K.; Furukawa, T.; Machida, M.; Wang, Y.H.; Motoji, T.; Mitsuishi, T.; Akiyama, M.; Yamada, H.; Kawauchi, K.; et al. Activation of STAT5 Confers Imatinib Resistance on Leukemic Cells through the Transcription of TERT and MDR1. Cell. Signal. 2011, 23, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Sinha-Datta, U.; Horikawa, I.; Michishita, E.; Datta, A.; Sigler-Nicot, J.C.; Brown, M.; Kazanji, M.; Barrett, J.C.; Nicot, C. Transcriptional Activation of HTERT through the NF-ΚB Pathway in HTLV-I-Transformed Cells. Blood 2004, 104, 2523–2531. [Google Scholar] [CrossRef] [PubMed]
- Kyo, S.; Takakura, M.; Kanaya, T.; Zhuo, W.; Fujimoto, K.; Nishio, Y.; Orimo, A.; Inoue, M. Estrogen Activates Telomerase. Cancer Res. 1999, 59, 5917–5921. [Google Scholar] [PubMed]
- Misiti, S.; Nanni, S.; Fontemaggi, G.; Cong, Y.-S.; Wen, J.; Hirte, H.W.; Piaggio, G.; Sacchi, A.; Pontecorvi, A.; Bacchetti, S.; et al. Induction of HTERT Expression and Telomerase Activity by Estrogens in Human Ovary Epithelium Cells. Mol. Cell. Biol. 2000, 20, 3764–3771. [Google Scholar] [CrossRef]
- Semenza, G.L. Oxygen Sensing, Homeostasis, and Disease. N. Engl. J. Med. 2011, 365, 537–547. [Google Scholar] [CrossRef] [PubMed]
- Nishi, H.; Nakada, T.; Kyo, S.; Inoue, M.; Shay, J.W.; Isaka, K. Hypoxia-Inducible Factor 1 Mediates Upregulation of Telomerase (HTERT). Mol. Cell. Biol. 2004, 24, 6076–6083. [Google Scholar] [CrossRef]
- Kyo, S.; Takakura, M.; Fujiwara, T.; Inoue, M. Understanding and Exploiting HTERT Promoter Regulation for Diagnosis and Treatment of Human Cancers. Cancer Sci. 2008, 99, 1528–1538. [Google Scholar] [CrossRef]
- Lou, F.; Chen, X.; Jalink, M.; Zhu, Q.; Ge, N.; Zhao, S.; Fang, X.; Fan, Y.; Bjö, M.; Liu, Z.; et al. The Opposing Effect of Hypoxia-Inducible Factor-2A on Expression of Telomerase Reverse Transcriptase. Mol. Cell. Biol. 2007. [Google Scholar] [CrossRef]
- Lai, S.R.; Phipps, S.; Liu, L.; Andrews, L.; Tollefsbol, T. Epigenetic Control of Telomerase and Modes of Telomere Maintenance in Aging and Abnormal Systems. Front. Biosci. 2005, 5, 1779–1796. [Google Scholar] [CrossRef] [PubMed]
- Shats, I.; Milyavsky, M.; Tang, X.; Stambolsky, P.; Erez, N.; Brosh, R.; Kogan, I.; Braunstein, I.; Tzukerman, M.; Ginsberg, D.; et al. P53-Dependent down-Regulation of Telomerase Is Mediated by P21 Waf1. J. Biol. Chem. 2004, 279, 50976–50985. [Google Scholar] [CrossRef]
- Kanaya, T.; Kyo, S.; Hamada, K.; Takakura, M.; Kitagawa, Y.; Harada, H.; Inoue, M. Adenoviral Expression of P53 Represses Telomerase Activity through Down-Regulation of Human Telomerase Reverse Transcriptase Transcription. Clin. Cancer Res. 2000, 6, 1239–1247. [Google Scholar] [PubMed]
- Takakura, M.; Kyo, S.; Kanaya, T.; Hirano, H.; Takeda, J.; Yutsudo, M.; Inoue, M. Cloning of Human Telomerase Catalytic Subunit (HTERT) Gene Promoter and Identification of Proximal Core Promoter Sequences Essential for Transcriptional Activation in Immortalized and Cancer Cells. Cancer Res. 1999, 59, 551–557. [Google Scholar] [PubMed]
- Horn, S.; Figl, A.; Rachakonda, P.S.; Fischer, C.; Sucker, A.; Gast, A.; Kadel, S.; Moll, I.; Nagore, E.; Hemminki, K.; et al. TERT Promoter Mutations in Familial and Sporadic Melanoma. Science 2013, 339, 959–961. [Google Scholar] [CrossRef] [PubMed]
- Borah, S.; Xi, L.; Zaug, A.J.; Powell, N.M.; Dancik, G.M.; Cohen, S.B.; Costello, J.C.; Theodorescu, D.; Cech, T.R. Cancer. TERT Promoter Mutations and Telomerase Reactivation in Urothelial Cancer. Science 2015, 347, 1006–1010. [Google Scholar] [CrossRef]
- Zehir, A.; Benayed, R.; Shah, R.H.; Syed, A.; Middha, S.; Kim, H.R.; Srinivasan, P.; Gao, J.; Chakravarty, D.; Devlin, S.M.; et al. Mutational Landscape of Metastatic Cancer Revealed from Prospective Clinical Sequencing of 10,000 Patients. Nat. Med. 2017, 23, 703–713. [Google Scholar] [CrossRef]
- Stern, J.L.; Theodorescu, D.; Vogelstein, B.; Papadopoulos, N.; Cech, T.R. Mutation of the TERT Promoter, Switch to Active Chromatin, and Monoallelic TERT Expression in Multiple Cancers. Genes Dev. 2015, 29, 2219–2224. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Shen, J.; Cao, J.; Zhou, G.; Lei, T.; Sun, Y.; Gao, H.; Ding, Y.; Xu, W.; Zhan, Z.; et al. Alternative Splicing of Human Telomerase Reverse Transcriptase in Gliomas and Its Modulation Mediated by CX-5461. J. Exp. Clin. Cancer Res. 2018, 37. [Google Scholar] [CrossRef]
- Ulaner, G.A.; Hu, J.F.; Vu, T.H.; Giudice, L.C.; Hoffman, A.R. Tissue-Specific Alternate Splicing of Human Telomerase Reverse Transcriptase (HTERT) Influences Telomere Lengths during Human Development. Int. J. Cancer 2001, 91, 644–649. [Google Scholar] [CrossRef]
- Hrdlickova, R.; Nehyba, J.; Bose, H.R. Alternatively Spliced Telomerase Reverse Transcriptase Variants Lacking Telomerase Activity Stimulate Cell Proliferation. Mol. Cell. Biol. 2012, 32, 4283–4296. [Google Scholar] [CrossRef]
- Wong, M.S.; Wright, W.E.; Shay, J.W. Alternative Splicing Regulation of Telomerase: A New Paradigm? Trends Genet. 2014, 30, 430–438. [Google Scholar] [CrossRef]
- Yi, X. Quantitation of Telomerase Components and HTERT MRNA Splicing Patterns in Immortal Human Cells. Nucleic Acids Res. 2001, 29, 4818–4825. [Google Scholar] [CrossRef]
- Yi, X.; White, D.M.; Aisner, D.L.; Baur, J.A.; Wright, W.E.; Shay, J.W. An Alternate Splicing, Variant of the Human Telomerase Catalytic Subunit Inhibits Telomerase Activity. Neoplasia 2000, 2, 433–440. [Google Scholar] [CrossRef]
- Colgin, L.M.; Wilkinson, C.; Englezou, A.; Kilian, A.; Robinson, M.O.; Reddel, R.R. The HTERTα Splice Variant Is a Dominant Negative Inhibitor of Telomerase Activity. Neoplasia 2000, 2, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Ulaner, G.A.; Hu, J.-F.; Vu, T.H.; Giudice, L.C.; Hoffman, A.R. Telomerase Activity in Human Development Is Regulated by Human Telomerase Reverse Transcriptase (HTERT) Transcription and by Alternate Splicing of HTERT Transcripts1. Cancer Res. 1998, 58, 4168–4172. [Google Scholar] [PubMed]
- Liu, X.; Wang, Y.; Chang, G.; Wang, F.; Wang, F.; Geng, X. Alternative Splicing of HTERT Pre-MRNA: A Potential Strategy for the Regulation of Telomerase Activity. Int. J. Mol. Sci. 2017, 18, 567. [Google Scholar] [CrossRef] [PubMed]
- Waddington, C.H. The Epigenotype. Int. J. Epidemiol. 2012, 41, 10–13. [Google Scholar] [CrossRef]
- Sharma, S.; Kelly, T.K.; Jones, P.A. Epigenetics in Cancer. Carcinogenesis 2010, 31, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Mattick, J.S.; Makunin, I.V. Non-Coding RNA. Hum. Mol. Genet. 2006, 15 (Suppl. 1), R17–R29. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.A.; Laird, P.W. Cancer-Epigenetics Comes of Age. Nat. Genet. 1999, 21, 163–167. [Google Scholar] [CrossRef]
- Feil, R.; Fraga, M.F. Epigenetics and the Environment: Emerging Patterns and Implications. Nat. Rev. Genet. 2012, 13, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Shen, Y.; Xue, Z.; Bibikova, M.; April, C.; Liu, Z.; Cheng, L.; Nagy, A.; Pellegrini, M.; Fan, J.-B.; et al. A Panel of CpG Methylation Sites Distinguishes Human Embryonic Stem Cells and Induced Pluripotent Stem Cells. Stem Cell Rep. 2014, 2, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Bird, A. Perceptions of Epigenetics. Nature 2007, 447, 396–398. [Google Scholar] [CrossRef]
- Moore, L.D.; Le, T.; Fan, G. DNA Methylation and Its Basic Function. Neuropsychopharmacology 2013, 38, 23–38. [Google Scholar] [CrossRef] [PubMed]
- Ting, A.H.; Jair, K.; Schuebel, K.E.; Baylin, S.B. Differential Requirement for DNA Methyltransferase 1 in Maintaining Human Cancer Cell Gene Promoter Hypermethylation. Cancer Res. 2006, 66, 729–735. [Google Scholar] [CrossRef]
- Hermann, A.; Goyal, R.; Jeltsch, A. The Dnmt1 DNA-(Cytosine-C5)-Methyltransferase Methylates DNA Processively with High Preference for Hemimethylated Target Sites. J. Biol. Chem. 2004, 279, 48350–48359. [Google Scholar] [CrossRef] [PubMed]
- Li, E. Chromatin Modification and Epigenetic Reprogramming in Mammalian Development. Nat. Rev. Genet. 2002, 3, 662–673. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, N.; Morin, S.; Chute, I.C.; Robert, M.-F.; Nguyen, H.; MacLeod, A.R. An Essential Role for DNA Methyltransferase DNMT3B in Cancer Cell Survival. J. Biol. Chem. 2002, 277, 28176–28181. [Google Scholar] [CrossRef]
- Ostler, K.R.; Davis, E.M.; Payne, S.L.; Gosalia, B.B.; Expósito-Céspedes, J.; Beau, M.M.L.; Godley, L.A. Cancer Cells Express Aberrant DNMT3B Transcripts Encoding Truncated Proteins. Oncogene 2007, 26, 5553–5563. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-Y.; Pu, M.-T.; Hirasawa, R.; Li, B.-Z.; Huang, Y.-N.; Zeng, R.; Jing, N.-H.; Chen, T.; Li, E.; Sasaki, H.; et al. Synergistic Function of DNA Methyltransferases Dnmt3a and Dnmt3b in the Methylation of Oct4 and Nanog. Mol. Cell. Biol. 2007, 27, 8748–8759. [Google Scholar] [CrossRef]
- Liao, J.; Karnik, R.; Gu, H.; Ziller, M.J.; Clement, K.; Tsankov, A.M.; Akopian, V.; Gifford, C.A.; Donaghey, J.; Galonska, C.; et al. Targeted Disruption of DNMT1, DNMT3A and DNMT3B in Human Embryonic Stem Cells. Nat. Genet. 2015, 47, 469–478. [Google Scholar] [CrossRef]
- Ito, S.; Shen, L.; Dai, Q.; Wu, S.C.; Collins, L.B.; Swenberg, J.A.; He, C.; Zhang, Y. Tet Proteins Can Convert 5-Methylcytosine to 5-Formylcytosine and 5-Carboxylcytosine. Science 2011, 333, 1300–1303. [Google Scholar] [CrossRef]
- Globisch, D.; Münzel, M.; Müller, M.; Michalakis, S.; Wagner, M.; Koch, S.; Brückl, T.; Biel, M.; Carell, T. Tissue Distribution of 5-Hydroxymethylcytosine and Search for Active Demethylation Intermediates. PLoS ONE 2010, 5, e15367. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Chavez, L.; Chang, X.; Wang, X.; Pastor, W.A.; Kang, J.; Zepeda-Martínez, J.A.; Pape, U.J.; Jacobsen, S.E.; Peters, B.; et al. Distinct Roles of the Methylcytosine Oxidases Tet1 and Tet2 in Mouse Embryonic Stem Cells. Proc. Natl. Acad. Sci. USA 2014, 111, 1361–1366. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Dalessio, A.C.; Taranova, O.V.; Hong, K.; Sowers, L.C.; Zhang, Y. Role of Tet Proteins in 5mC to 5hmC Conversion, ES-Cell Self-Renewal and Inner Cell Mass Specification. Nature 2010, 466, 1129–1133. [Google Scholar] [CrossRef] [PubMed]
- Tsagaratou, A.; González-Avalos, E.; Rautio, S.; Scott-Browne, J.P.; Togher, S.; Pastor, W.A.; Rothenberg, E.V.; Chavez, L.; Lähdesmäki, H.; Rao, A. TET Proteins Regulate the Lineage Specification and TCR-Mediated Expansion of INKT Cells. Nat. Immunol. 2017, 18, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Ficz, G.; Gribben, J.G. Loss of 5-Hydroxymethylcytosine in Cancer: Cause or Consequence? Genomics 2014, 104, 352–357. [Google Scholar] [CrossRef]
- Ficz, G.; Branco, M.R.; Seisenberger, S.; Santos, F.; Krueger, F.; Hore, T.A.; Marques, C.J.; Andrews, S.; Reik, W. Dynamic Regulation of 5-Hydroxymethylcytosine in Mouse ES Cells and during Differentiation. Nature 2011, 473, 398–404. [Google Scholar] [CrossRef] [PubMed]
- Xhemalce, B.; Dawson, M.A.; Bannister, A.J. Histone Modifications. In Encyclopedia of Molecular Cell Biology and Molecular Medicine; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2011. [Google Scholar] [CrossRef]
- Seto, E.; Yoshida, M. Erasers of Histone Acetylation: The Histone Deacetylase Enzymes. Cold Spring Harb. Perspect. Biol. 2014, 6, a018713. [Google Scholar] [CrossRef]
- Bannister, A.J.; Kouzarides, T. Regulation of Chromatin by Histone Modifications. Cell Res. 2011, 21, 381–395. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.S.; Yue, W.W.; Oppermann, U.; Klose, R.J. Dynamic Protein Methylation in Chromatin Biology. Cell. Mol. Life Sci. 2009, 66, 407–422. [Google Scholar] [CrossRef]
- Bedford, M.T.; Clarke, S.G. Protein Arginine Methylation in Mammals: Who, What, and Why. Mol. Cell 2009, 33, 1–13. [Google Scholar] [CrossRef]
- Rice, J.C.; Briggs, S.D.; Ueberheide, B.; Barber, C.M.; Shabanowitz, J.; Hunt, D.F.; Shinkai, Y.; Allis, C.D. Histone Methyltransferases Direct Different Degrees of Methylation to Define Distinct Chromatin Domains. Mol. Cell 2003, 12, 1591–1598. [Google Scholar] [CrossRef]
- Guil, S.; Esteller, M. DNA Methylomes, Histone Codes and MiRNAs: Tying It All Together. Int. J. Biochem. Cell Biol. 2009, 87–95. [Google Scholar] [CrossRef]
- Whitehead, J.; Pandey, G.K.; Kanduri, C. Regulation of the Mammalian Epigenome by Long Noncoding RNAs. Biochim. Biophys. Acta Gen. Subj. 2009, 936–947. [Google Scholar] [CrossRef] [PubMed]
- Ørom, U.A.; Derrien, T.; Beringer, M.; Gumireddy, K.; Gardini, A.; Bussotti, G.; Lai, F.; Zytnicki, M.; Notredame, C.; Huang, Q.; et al. Long Noncoding RNAs with Enhancer-like Function in Human Cells. Cell 2010, 143, 46–58. [Google Scholar] [CrossRef]
- Zhao, X.; Tian, X.; Kajigaya, S.; Cantilena, C.R.; Strickland, S.; Savani, B.N.; Mohan, S.; Feng, X.; Keyvanfar, K.; Dunavin, N.; et al. Epigenetic Landscape of the TERT Promoter: A Potential Biomarker for High Risk AML/MDS. Br. J. Haematol. 2016, 175, 427–439. [Google Scholar] [CrossRef]
- Stern, J.L.; Paucek, R.D.; Huang, F.W.; Ghandi, M.; Nwumeh, R.; Costello, J.C.; Cech, T.R. Allele-Specific DNA Methylation and Its Interplay with Repressive Histone Marks at Promoter-Mutant TERT Genes. Cell Rep. 2017, 21, 3700–3707. [Google Scholar] [CrossRef]
- Hanada, M.; Delia, D.; Aiello, A.; Stadtmauer, E.; Reed, J.C. Bcl-2 Gene Hypomethylation and High-Level Expression in B-Cell Chronic Lymphocytic Leukemia. Blood 1993, 82, 1820–1828. [Google Scholar] [CrossRef] [PubMed]
- Peifer, M.; Hertwig, F.; Roels, F.; Dreidax, D.; Gartlgruber, M.; Menon, R.; Krämer, A.; Roncaioli, J.L.; Sand, F.; Heuckmann, J.M.; et al. Telomerase Activation by Genomic Rearrangements in High-Risk Neuroblastoma. Nature 2015, 526, 700–704. [Google Scholar] [CrossRef] [PubMed]
- Eitsuka, T.; Nakagawa, K.; Kato, S.; Ito, J.; Otoki, Y.; Takasu, S.; Shimizu, N.; Takahashi, T.; Miyazawa, T. Modulation of Telomerase Activity in Cancer Cells by Dietary Compounds: A Review. Int. J. Mol. Sci. 2018, 19, 478. [Google Scholar] [CrossRef] [PubMed]
- Albanell, J.; Han, W.; Mellado, B.; Gunawardane, R.; Scher, H.I.; Dmitrovsky, E.; Moore, M.A. Telomerase Activity Is Repressed during Differentiation of Maturation-Sensitive but Not Resistant Human Tumor Cell Lines. Cancer Res. 1996, 56, 1503–1508. [Google Scholar] [PubMed]
- Phipps, S.M.O.; Love, W.K.; Mott, T.E.; Andrews, L.G.; Tollefsbol, T.O. Differential Expression of Epigenetic Modulators during Human Embryonic Stem Cell Differentiation. Mol. Biotechnol. 2009, 41, 201–207. [Google Scholar] [CrossRef]
- Devereux, T.R.; Horikawa, I.; Anna, C.H.; Annab, L.A.; Afshari, C.A.; Barrett, J.C. DNA Methylation Analysis of the Promoter Region of the Human Telomerase Reverse Transcriptase (HTERT) Gene. Cancer Res. 1999, 59, 6087–6090. [Google Scholar] [PubMed]
- Skowronski, K.; Dubey, S.; Rodenhiser, D.I.; Coomber, B. Ischemia Dysregulates DNA Methyltransferases and P16INK4a Methylation in Human Colorectal Cancer Cells. Epigenetics 2010, 5, 547–556. [Google Scholar] [CrossRef] [PubMed]
- Shahrzad, S.; Bertrand, K.; Minhas, K.; Coomber, B.L. Induction of DNA Hypomethylation by Tumor Hypoxia. Epigenetics 2007, 2, 119–125. [Google Scholar] [CrossRef]
- Dessain, S.K.; Yu, H.; Reddel, R.R.; Beijersbergen, R.L.; Weinberg, R.A. Methylation of the Human Telomerase Gene CpG Island. Cancer Res. 2000, 60, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Castelo-Branco, P.; Choufani, S.; Mack, S.; Gallagher, D.; Zhang, C.; Lipman, T.; Zhukova, N.; Walker, E.J.; Martin, D.; Merino, D.; et al. Methylation of the TERT Promoter and Risk Stratification of Childhood Brain Tumours: An Integrative Genomic and Molecular Study. Lancet Oncol. 2013, 14, 534–542. [Google Scholar] [CrossRef]
- Lee, D.D.; Leão, R.; Komosa, M.; Gallo, M.; Zhang, C.H.; Lipman, T.; Remke, M.; Heidari, A.; Nunes, N.M.; Apolónio, J.D.; et al. DNA Hypermethylation within TERT Promoter Upregulates TERT Expression in Cancer. J. Clin. Invest. 2019, 129, 223–229. [Google Scholar] [CrossRef]
- Renaud, S.; Loukinov, D.; Bosman, F.T.; Lobanenkov, V.; Benhattar, J. CTCF Binds the Proximal Exonic Region of HTERT and Inhibits Its Transcription. Nucleic Acids Res. 2005, 33, 6850–6860. [Google Scholar] [CrossRef] [PubMed]
- Renaud, S.; Loukinov, D.; Abdullaev, Z.; Guilleret, I.; Bosman, F.T.; Lobanenkov, V.; Benhattar, J. Dual Role of DNA Methylation inside and Outside of CTCF-Binding Regions in the Transcriptional Regulation of the Telomerase HTERT Gene. Nucleic Acids Res. 2007, 35, 1245–1256. [Google Scholar] [CrossRef]
- Meeran, S.M.; Patel, S.N.; Tollefsbol, T.O. Sulforaphane Causes Epigenetic Repression of HTERT Expression in Human Breast Cancer Cell Lines. PLoS ONE 2010, 5, e11457. [Google Scholar] [CrossRef] [PubMed]
- Sitaram, R.T.; Degerman, S.; Ljungberg, B.; Andersson, E.; Oji, Y.; Sugiyama, H.; Roos, G.; Li, A. Wilms’ Tumour 1 Can Suppress HTERT Gene Expression and Telomerase Activity in Clear Cell Renal Cell Carcinoma via Multiple Pathways. Br. J. Cancer 2010, 103, 1255–1262. [Google Scholar] [CrossRef]
- Wu, Y.; Li, G.; He, D.; Yang, F.; He, G.; He, L.; Zhang, H.; Deng, Y.; Fan, M.; Shen, L.; et al. Telomerase Reverse Transcriptase Methylation Predicts Lymph Node Metastasis and Prognosis in Patients with Gastric Cancer. OncoTargets Ther. 2016, 9, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Takasawa, K.; Arai, Y.; Yamazaki-Inoue, M.; Toyoda, M.; Akutsu, H.; Umezawa, A.; Nishino, K. DNA Hypermethylation Enhanced Telomerase Reverse Transcriptase Expression in Human-Induced Pluripotent Stem Cells. Hum. Cell 2018, 31, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Zinn, R.L.; Pruitt, K.; Eguchi, S.; Baylin, S.B.; Herman, J.G. HTERT Is Expressed in Cancer Cell Lines despite Promoter DNA Methylation by Preservation of Unmethylated DNA and Active Chromatin around the Transcription Start Site. Cancer Res. 2007, 67, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Iliopoulos, D.; Satra, M.; Drakaki, A.; Poultsides, G.A.; Tsezou, A. Epigenetic Regulation of HTERT Promoter in Hepatocellular Carcinomas. Int. J. Oncol. 2009, 34, 391–399. [Google Scholar] [CrossRef]
- Zhang, H.; Weng, X.; Ye, J.; He, L.; Zhou, D.; Liu, Y. Promoter Hypermethylation of TERT Is Associated with Hepatocellular Carcinoma in the Han Chinese Population. Clin. Res. Hepatol. Gastroenterol. 2015, 39, 600–609. [Google Scholar] [CrossRef]
- Wang, N.; Kjellin, H.; Sofiadis, A.; Fotouhi, O.; Juhlin, C.C.; Bäckdahl, M.; Zedenius, J.; Xu, D.; Lehtiö, J.; Larsson, C. Genetic and Epigenetic Background and Protein Expression Profiles in Relation to Telomerase Activation in Medullary Thyroid Carcinoma. Oncotarget 2016, 7, 21332–21346. [Google Scholar] [CrossRef] [PubMed]
- Castelo-Branco, P.; Leão, R.; Lipman, T.; Campbell, B.; Lee, D.; Price, A.; Zhang, C.; Heidari, A.; Stephens, D.; Boerno, S.; et al. A Cancer Specific Hypermethylation Signature of the TERT Promoter Predicts Biochemical Relapse in Prostate Cancer: A Retrospective Cohort Study. Oncotarget 2016, 7, 57726–57736. [Google Scholar] [CrossRef]
- Fan, Y.; Lee, S.; Wu, G.; Easton, J.; Yergeau, D.; Dummer, R.; Vogel, P.; Kirkwood, J.M.; Barnhill, R.L.; Pappo, A.; et al. Telomerase Expression by Aberrant Methylation of the TERT Promoter in Melanoma Arising in Giant Congenital Nevi. J. Invest. Dermatol. 2016, 136, 339–342. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Wang, W.; Zhao, J.; Li, T.; Kang, X. Aberrant HTERT Promoter Methylation Predicts Prognosis in Chinese Patients with Acral and Mucosal Melanoma: A CONSORT-Compliant Article. Medicine 2019, 98. [Google Scholar] [CrossRef]
- Faleiro, I.; Apolónio, J.D.; Price, A.J.; De Mello, R.A.; Roberto, V.P.; Tabori, U.; Castelo-Branco, P. The TERT Hypermethylated Oncologic Region Predicts Recurrence and Survival in Pancreatic Cancer. Futur. Oncol. 2017, 13, 2045–2051. [Google Scholar] [CrossRef] [PubMed]
- Leão, R.; Lee, D.; Figueiredo, A.; Hermanns, T.; Wild, P.; Komosa, M.; Lau, I.; Mistry, M.; Nunes, N.M.; Price, A.J.; et al. Combined Genetic and Epigenetic Alterations of the TERT Promoter Affect Clinical and Biological Behavior of Bladder Cancer. Int. J. Cancer 2019, 144, 1676–1684. [Google Scholar] [CrossRef]
- Minasi, S.; Baldi, C.; Pietsch, T.; Donofrio, V.; Pollo, B.; Antonelli, M.; Massimino, M.; Giangaspero, F.; Buttarelli, F.R. Telomere Elongation via Alternative Lengthening of Telomeres (ALT) and Telomerase Activation in Primary Metastatic Medulloblastoma of Childhood. J. Neurooncol. 2019, 142, 435–444. [Google Scholar] [CrossRef]
- Avin, B.A.; Wang, Y.; Gilpatrick, T.; Workman, R.E.; Lee, I.; Timp, W.; Umbricht, C.B.; Zeiger, M.A. Characterization of Human Telomerase Reverse Transcriptase Promoter Methylation and Transcription Factor Binding in Differentiated Thyroid Cancer Cell Lines. Genes Chromosom. Cancer 2019, 58, 530–540. [Google Scholar] [CrossRef]
- Love, W.; Berletch, J.; Andrews, L.; Tollefsbol, T. Epigenetic Regulation of Telomerase in Retinoid-Induced Differentiation of Human Leukemia Cells. Int. J. Oncol. 2008, 32, 625–631. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Majid, S.; Kikuno, N.; Nelles, J.; Noonan, E.; Tanaka, Y.; Kawamoto, K.; Hirata, H.; Li, L.C.; Zhao, H.; Okino, S.T.; et al. Genistein Induces the P21WAF1/CIP1 and P16INK4a Tumor Suppressor Genes in Prostate Cancer Cells by Epigenetic Mechanisms Involving Active Chromatin Modification. Cancer Res. 2008, 68, 2736–2744. [Google Scholar] [CrossRef]
- Kikuno, N.; Shiina, H.; Urakami, S.; Kawamoto, K.; Hirata, H.; Tanaka, Y.; Majid, S.; Igawa, M.; Dahiya, R. Retracted: Genistein Mediated Histone Acetylation and Demethylation Activates Tumor Suppressor Genes in Prostate Cancer Cells. Int. J. Cancer 2008, 123, 552–560. [Google Scholar] [CrossRef]
- Li, Y.; Liu, L.; Andrews, L.G.; Tollefsbol, T.O. Genistein Depletes Telomerase Activity through Cross-Talk between Genetic and Epigenetic Mechanisms. Int. J. Cancer 2009, 125, 286–296. [Google Scholar] [CrossRef]
- Berletch, J.B.; Liu, C.; Love, W.K.; Andrews, L.G.; Katiyar, S.K.; Tollefsbol, T.O. Epigenetic and Genetic Mechanisms Contribute to Telomerase Inhibition by EGCG. J. Cell. Biochem. 2008, 103, 509–519. [Google Scholar] [CrossRef]
- Yuan, X.; Xu, D. Molecular Sciences Telomerase Reverse Transcriptase (TERT) in Action: Cross-Talking with Epigenetics. Int. J. Mol. Sci. 2019, 20, 3338. [Google Scholar] [CrossRef] [PubMed]
- Karlić, R.; Chung, H.-R.; Lasserre, J.; Vlahoviček, K.; Vingron, M. Histone Modification Levels Are Predictive for Gene Expression. Proc. Natl. Acad. Sci. USA 2010, 107, 2926–2931. [Google Scholar] [CrossRef]
- Seynnaeve, B.; Lee, S.; Borah, S.; Park, Y.; Pappo, A.; Kirkwood, J.M.; Bahrami, A. Genetic and Epigenetic Alterations of TERT Are Associated with Inferior Outcome in Adolescent and Young Adult Patients with Melanoma. Sci. Rep. 2017, 7, 45704. [Google Scholar] [CrossRef]
- Liu, T.; Yuan, X.; Xu, D. Cancer-Specific Telomerase Reverse Transcriptase (TERT) Promoter Mutations: Biological and Clinical Implications. Genes 2016, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Fang, X.; Ge, Z.; Jalink, M.; Kyo, S.; Bjorkholm, M.; Gruber, A.; Sjoberg, J.; Xu, D. The Telomerase Reverse Transcriptase (HTERT) Gene Is a Direct Target of the Histone Methyltransferase SMYD3. Cancer Res. 2007, 67, 2626–2631. [Google Scholar] [CrossRef] [PubMed]
- Guccione, E.; Martinato, F.; Finocchiaro, G.; Luzi, L.; Tizzoni, L.; Dall’ Olio, V.; Zardo, G.; Nervi, C.; Bernard, L.; Amati, B. Myc-Binding-Site Recognition in the Human Genome Is Determined by Chromatin Context. Nat. Cell Biol. 2006, 8, 764–770. [Google Scholar] [CrossRef] [PubMed]
- Strahl, B.D.; Allis, C.D. The Language of Covalent Histone Modifications. Nature 2000, 403, 41–45. [Google Scholar] [CrossRef]
- Cheng, D.; Wang, S.; Jia, W.; Zhao, Y.; Zhang, F.; Kang, J.; Zhu, J. Regulation of Human and Mouse Telomerase Genes by Genomic Contexts and Transcription Factors during Embryonic Stem Cell Differentiation. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Ohba, S.; Mukherjee, J.; Johannessen, T.C.; Mancini, A.; Chow, T.T.; Wood, M.; Jones, L.; Mazor, T.; Marshall, R.E.; Viswanath, P.; et al. Mutant IDH1 Expression Drives TERT Promoter Reactivation as Part of the Cellular Transformation Process. Cancer Res. 2016, 76, 6680–6689. [Google Scholar] [CrossRef]
- Akıncılar, S.C.; Khattar, E.; Boon, P.L.S.; Unal, B.; Fullwood, M.J.; Tergaonkar, V. Long-Range Chromatin Interactions Drive Mutant TERT Promoter Activation. Cancer Discov. 2016, 6, 1276–1292. [Google Scholar] [CrossRef]
- Sui, X.; Kong, N.; Wang, Z.; Pan, H. Epigenetic Regulation of the Human Telomerase Reverse Transciptase Gene: A Potential Therapeutic Target for the Treatment of Leukemia (Review). Oncol. Lett. 2013, 6, 317–322. [Google Scholar] [CrossRef][Green Version]
- Atkinson, S.P.; Hoare, S.F.; Glasspool, R.M.; Keith, W.N. Lack of Telomerase Gene Expression in Alternative Lengthening of Telomere Cells Is Associated with Chromatin Remodeling of the HTR and HTERT Gene Promoters. Cancer Res. 2005, 65, 7585–7590. [Google Scholar] [CrossRef] [PubMed]
- Takakura, M.; Kyo, S.; Sowa, Y.; Wang, Z.; Yatabe, N.; Maida, Y.; Tanaka, M.; Inoue, M. Telomerase Activation by Histone Deacetylase Inhibitor in Normal Cells. Nucleic Acids Res. 2001, 29, 3006–3011. [Google Scholar] [CrossRef] [PubMed]
- Qing, H.; Aono, J.; Findeisen, H.M.; Jones, K.L.; Heywood, E.B.; Bruemmer, D. Differential Regulation of Telomerase Reverse Transcriptase Promoter Activation and Protein Degradation by Histone Deacetylase Inhibition. J. Cell. Physiol. 2016, 231, 1276–1282. [Google Scholar] [CrossRef]
- Khaw, A.K.; Silasudjana, M.; Banerjee, B.; Suzuki, M.; Baskar, R.; Hande, M.P. Inhibition of Telomerase Activity and Human Telomerase Reverse Transcriptase Gene Expression by Histone Deacetylase Inhibitor in Human Brain Cancer Cells. Mutat. Res. Mol. Mech. Mutagen. 2007, 625, 134–144. [Google Scholar] [CrossRef]
- Zhu, K.; Qu, D.; Sakamoto, T.; Fukasawa, I.; Hayashi, M.; Inaba, N. Telomerase Expression and Cell Proliferation in Ovarian Cancer Cells Induced by Histone Deacetylase Inhibitors. Arch. Gynecol. Obstet. 2007, 277, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Chen, J.; Cheng, A.S.L.; Ko, B.C.B. Depletion of Sirtuin 1 (SIRT1) Leads to Epigenetic Modifications of Telomerase (TERT) Gene in Hepatocellular Carcinoma Cells. PLoS ONE 2014, 9, e84931. [Google Scholar] [CrossRef]
- Garsuault, D.; Bouyer, C.; Nguyen, E.; Kandhari, R.; Prochazkova-Carlotti, M.; Chevret, E.; Forgez, P.; Ségal-Bendirdjian, E. Complex Context Relationships between DNA Methylation and Accessibility, Histone Marks, and HTERT Gene Expression in Acute Promyelocytic Leukemia Cells: Perspectives for All-Trans Retinoic Acid in Cancer Therapy. Mol. Oncol. 2020, 14, 1310–1326. [Google Scholar] [CrossRef] [PubMed]
- Santambrogio, F.; Gandellini, P.; Cimino-Reale, G.; Zaffaroni, N.; Folini, M. MicroRNA-Dependent Regulation of Telomere Maintenance Mechanisms: A Field as Much Unexplored as Potentially Promising. Curr. Pharm. Des. 2014, 20, 6404–6421. [Google Scholar] [CrossRef]
- Gaur, A.; Jewell, D.A.; Liang, Y.; Ridzon, D.; Moore, J.H.; Chen, C.; Ambros, V.R.; Israel, M.A. Characterization of MicroRNA Expression Levels and Their Biological Correlates in Human Cancer Cell Lines. Cancer Res. 2007, 67, 2456–2468. [Google Scholar] [CrossRef]
- Grady, W.M.; Tewari, M. The next Thing in Prognostic Molecular Markers: MicroRNA Signatures of Cancer. Gut 2010, 706–708. [Google Scholar] [CrossRef]
- Brennecke, J.; Stark, A.; Russell, R.B.; Cohen, S.M. Principles of MicroRNA–Target Recognition. PLoS Biol. 2005, 3, e85. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Lu, J.; Kulbokas, E.J.; Golub, T.R.; Mootha, V.; Lindblad-Toh, K.; Lander, E.S.; Kellis, M. Systematic Discovery of Regulatory Motifs in Human Promoters and 3′ UTRs by Comparison of Several Mammals. Nature 2005, 434, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. MicroRNAs: Target Recognition and Regulatory Functions. Cell 2009, 215–233. [Google Scholar] [CrossRef]
- Zhao, Q.; Zhai, Y.X.; Liu, H.Q.; Shi, Y.A.; Li, X.B. MicroRNA-491-5p Suppresses Cervical Cancer Cell Growth by Targeting HTERT. Oncol. Rep. 2015, 34, 979–986. [Google Scholar] [CrossRef] [PubMed]
- Zhou, N.; Fei, D.; Zong, S.; Zhang, M.; Yue, Y. MicroRNA-138 Inhibits Proliferation, Migration and Invasion through Targeting HTERT in Cervical Cancer. Oncol. Lett. 2016, 12, 3633–3639. [Google Scholar] [CrossRef]
- Guzman, H.; Sanders, K.; Idica, A.; Bochnakian, A.; Jury, D.; Daugaard, I.; Zisoulis, D.G.; Pedersen, I.M. MiR-128 Inhibits Telomerase Activity by Targeting TERT MRNA. Oncotarget 2018, 9, 13244–13253. [Google Scholar] [CrossRef]
- Li, C.J.; Xiao, Y.; Yang, M.; Su, T.; Sun, X.; Guo, Q.; Huang, Y.; Luo, X.H. Long Noncoding RNA Bmncr Regulates Mesenchymal Stem Cell Fate during Skeletal Aging. J. Clin. Invest. 2018, 5219–5221. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Li, Y.; Gong, R.; Gao, M.; Feng, C.; Liu, T.; Sun, Y.; Jin, M.; Wang, D.; Yuan, Y.; et al. The Long Non-Coding RNA-ORLNC1 Regulates Bone Mass by Directing Mesenchymal Stem Cell Fate. Mol. Ther. 2019, 27, 394–410. [Google Scholar] [CrossRef]
- Cesana, M.; Cacchiarelli, D.; Legnini, I.; Santini, T.; Sthandier, O.; Chinappi, M.; Tramontano, A.; Bozzoni, I. A Long Noncoding RNA Controls Muscle Differentiation by Functioning as a Competing Endogenous RNA. Cell 2011, 147, 358–369. [Google Scholar] [CrossRef] [PubMed]
- Lü, M.H.; Tang, B.; Zeng, S.; Hu, C.J.; Xie, R.; Wu, Y.Y.; Wang, S.M.; He, F.T.; Yang, S.M. Long Noncoding RNA BC032469, a Novel Competing Endogenous RNA, Upregulates HTERT Expression by Sponging MIR-1207-5p and Promotes Proliferation in Gastric Cancer. Oncogene 2016, 35, 3524–3534. [Google Scholar] [CrossRef]
- Zong, Z.H.; Du, Y.P.; Guan, X.; Chen, S.; Zhao, Y. CircWHSC1 Promotes Ovarian Cancer Progression by Regulating MUC1 and HTERT through Sponging MiR-145 and MiR-1182. J. Exp. Clin. Cancer Res. 2019, 38, 437. [Google Scholar] [CrossRef]
- Zhu, L.-Y.; Zhu, Y.-R.; Dai, D.-J.; Wang, X.; Jin, H.-C. Epigenetic Regulation of Alternative Splicing. Am. J. Cancer Res. 2018, 8, 2346–2358. [Google Scholar] [PubMed]
- Singh, M.K.; Tzfati, Y.; Chensny, L.J.; Benos, P.V.; Kaminski, N. Regulation Telomerase and Telomere Length In IPF By MicroRNAs. In Proceedings of the American Thoracic Society International 2010 Conference, New Orleans, LA, USA, 14–19 May 2010; American Thoracic Society: New Orleans, LA, USA, 2010; p. A2297. [Google Scholar] [CrossRef]
- Hrdličková, R.; Nehyba, J.; Bargmann, W.; Bose, H.R. Multiple Tumor Suppressor MicroRNAs Regulate Telomerase and TCF7, an Important Transcriptional Regulator of the Wnt Pathway. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Mitomo, S.; Maesawa, C.; Ogasawara, S.; Iwaya, T.; Shibazaki, M.; Yashima-Abo, A.; Kotani, K.; Oikawa, H.; Sakurai, E.; Izutsu, N.; et al. Downregulation of MiR-138 Is Associated with Overexpression of Human Telomerase Reverse Transcriptase Protein in Human Anaplastic Thyroid Carcinoma Cell Lines. Cancer Sci. 2008, 99, 280–286. [Google Scholar] [CrossRef]
- Kasiappan, R.; Shen, Z.; Tse, A.K.W.; Jinwal, U.; Tang, J.; Lungchukiet, P.; Sun, Y.; Kruk, P.; Nicosia, S.V.; Zhang, X.; et al. 1,25-Dihydroxyvitamin D3 Suppresses Telomerase Expression and Human Cancer Growth through MicroRNA-498. J. Biol. Chem. 2012, 287, 41297–41309. [Google Scholar] [CrossRef] [PubMed]
- Kasiappan, R.; Sun, Y.; Lungchukiet, P.; Quarni, W.; Zhang, X.; Bai, W. Vitamin D Suppresses Leptin Stimulation of Cancer Growth through MicroRNA. Cancer Res. 2014, 74, 6194–6204. [Google Scholar] [CrossRef]
- Zhang, D.; Xiao, Y.F.; Zhang, J.W.; Xie, R.; Hu, C.J.; Tang, B.; Wang, S.M.; Wu, Y.Y.; Hao, N.B.; Yang, S.M. MiR-1182 Attenuates Gastric Cancer Proliferation and Metastasis by Targeting the Open Reading Frame of HTERT. Cancer Lett. 2015, 360, 151–159. [Google Scholar] [CrossRef]
- Zhou, J.; Dai, W.; Song, J. MIR-1182 Inhibits Growth and Mediates the Chemosensitivity of Bladder Cancer by Targeting HTERT. Biochem. Biophys. Res. Commun. 2016, 470, 445–452. [Google Scholar] [CrossRef]
- Hou, X.S.; Han, C.Q.; Zhang, W. MiR-1182 Inhibited Metastasis and Proliferation of Ovarian Cancer by Targeting HTERT. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 1622–1628. [Google Scholar] [CrossRef]
- Chen, L.; Lü, M.H.; Zhang, D.; Hao, N.B.; Fan, Y.H.; Wu, Y.Y.; Wang, S.M.; Xie, R.; Fang, D.C.; Zhang, H.; et al. MiR-1207-5p and MiR-1266 Suppress Gastric Cancer Growth and Invasion by Targeting Telomerase Reverse Transcriptase. Cell Death Dis. 2014, 5. [Google Scholar] [CrossRef]
- Chai, L.; Kang, X.J.; Sun, Z.Z.; Zeng, M.F.; Yu, S.R.; Ding, Y.; Liang, J.Q.; Li, T.T.; Zhao, J. MiR-497-5p, MiR-195-5p and MiR-455-3p Function as Tumor Suppressors by Targeting HTERT in Melanoma A375 Cells. Cancer Manag. Res. 2018, 10, 989–1003. [Google Scholar] [CrossRef] [PubMed]
- Abdelgawad, A.; Mosbah, A.; Eissa, L.A. Expression of MicroRNA-155 and Human Telomerase Reverse Transcriptase in Patients with Bladder Cancer. Egypt. J. Basic Appl. Sci. 2020, 7, 315–322. [Google Scholar] [CrossRef]
- Li, J.; Lei, H.; Xu, Y.; Tao, Z.Z. MiR-512-5p Suppresses Tumor Growth by Targeting HTERT in Telomerase Positive Head and Neck Squamous Cell Carcinoma in Vitro and in Vivo. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Li, Z.; Liu, Y.H.; Diao, H.Y.; Ma, J.; Yao, Y.L. MiR-661 Inhibits Glioma Cell Proliferation, Migration and Invasion by Targeting HTERT. Biochem. Biophys. Res. Commun. 2015, 468, 870–876. [Google Scholar] [CrossRef]
- Liu, L.; Liu, C.; Fotouhi, O.; Fan, Y.; Wang, K.; Xia, C.; Shi, B.; Zhang, G.; Wang, K.; Kong, F.; et al. TERT Promoter Hypermethylation in Gastrointestinal Cancer: A Potential Stool Biomarker. Oncologist 2017, 22, 1178–1188. [Google Scholar] [CrossRef]
- Nemunaitis, J.; Tong, A.W.; Nemunaitis, M.; Senzer, N.; Phadke, A.P.; Bedell, C.; Adams, N.; Zhang, Y.-A.; Maples, P.B.; Chen, S.; et al. A Phase I Study of Telomerase-Specific Replication Competent Oncolytic Adenovirus (Telomelysin) for Various Solid Tumors. Mol. Ther. 2010, 18, 429–434. [Google Scholar] [CrossRef]
- Xu, X.; Tao, Y.; Gao, X.; Zhang, L.; Li, X.; Zou, W.; Ruan, K.; Wang, F.; Xu, G.L.; Hu, R. A CRISPR-Based Approach for Targeted DNA Demethylation. Cell Discov. 2016, 2, 1–12. [Google Scholar] [CrossRef]
- Spiegel, J.; Adhikari, S.; Balasubramanian, S. The Structure and Function of DNA G-Quadruplexes. Trends Chem. 2020, 123–136. [Google Scholar] [CrossRef]
- Mukherjee, A.K.; Sharma, S.; Chowdhury, S. Non-Duplex G-Quadruplex Structures Emerge as Mediators of Epigenetic Modifications. Trends Genet. 2019, 35. [Google Scholar] [CrossRef] [PubMed]
- Li, P.T.; Wang, Z.F.; Chu, I.T.; Kuan, Y.M.; Li, M.H.; Huang, M.C.; Chiang, P.C.; Chang, T.C.; Chen, C.T. Expression of the Human Telomerase Reverse Transcriptase Gene Is Modulated by Quadruplex Formation in Its First Exon Due to DNA Methylation. J. Biol. Chem. 2017, 292, 20859–20870. [Google Scholar] [CrossRef] [PubMed]
- Naderlinger, E.; Holzmann, K. Epigenetic Regulation of Telomere Maintenance for Therapeutic Interventions in Gliomas. Genes 2017, 8, 145. [Google Scholar] [CrossRef]
- Carew, J.S.; Giles, F.J.; Nawrocki, S.T. Histone Deacetylase Inhibitors: Mechanisms of Cell Death and Promise in Combination Cancer Therapy. Cancer Lett. 2008, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.; Eom, G.H. HDAC and HDAC Inhibitor: From Cancer to Cardiovascular Diseases. Chonnam Med. J. 2016, 52, 1. [Google Scholar] [CrossRef] [PubMed]
- Mann, B.S.; Johnson, J.R.; Cohen, M.H.; Justice, R.; Pazdur, R. FDA Approval Summary: Vorinostat for Treatment of Advanced Primary Cutaneous T-Cell Lymphoma. Oncologist 2007, 12, 1247–1252. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, S.J.; Demierre, M.F.; Kim, E.J.; Rook, A.H.; Lerner, A.; Duvic, M.; Scarisbrick, J.; Reddy, S.; Robak, T.; Becker, J.C.; et al. Final Results from a Multicenter, International, Pivotal Study of Romidepsin in Refractory Cutaneous T-Cell Lymphoma. J. Clin. Oncol. 2010, 28, 4485–4491. [Google Scholar] [CrossRef]
- Coiffier, B.; Pro, B.; Prince, H.M.; Foss, F.; Sokol, L.; Greenwood, M.; Caballero, D.; Borchmann, P.; Morschhauser, F.; Wilhelm, M.; et al. Results from a Pivotal, Open-Label, Phase II Study of Romidepsin in Relapsed or Refractory Peripheral T-Cell Lymphoma after Prior Systemic Therapy. J. Clin. Oncol. 2012, 30, 631–636. [Google Scholar] [CrossRef]
- Poole, R.M. Belinostat: First Global Approval. Drugs 2014, 74, 1543–1554. [Google Scholar] [CrossRef]
- Oki, Y.; Buglio, D.; Fanale, M.; Fayad, L.; Copeland, A.; Romaguera, J.; Kwak, L.W.; Pro, B.; De Castro Faria, S.; Neelapu, S.; et al. Phase i Study of Panobinostat plus Everolimus in Patients with Relapsed or Refractory Lymphoma. Clin. Cancer Res. 2013, 19, 6882–6890. [Google Scholar] [CrossRef] [PubMed]
- Li, C.-T.; Hsiao, Y.-M.; Wu, T.-C.; Lin, Y.; Yeh, K.-T.; Ko, J.-L. Vorinostat, SAHA, Represses Telomerase Activity via Epigenetic Regulation of Telomerase Reverse Transcriptase in Non-Small Cell Lung Cancer Cells. J. Cell. Biochem. 2011, 112, 3044–3053. [Google Scholar] [CrossRef]
- Kretzner, L.; Scuto, A.; Dino, P.M.; Kowolik, C.M.; Wu, J.; Ventura, P.; Jove, R.; Forman, S.J.; Yen, Y.; Kirschbaum, M.H. Combining Histone Deacetylase Inhibitor Vorinostat with Aurora Kinase Inhibitors Enhances Lymphoma Cell Killing with Repression of C-Myc, HTERT, and MicroRNA Levels. Cancer Res. 2011, 71, 3912–3920. [Google Scholar] [CrossRef]
- Rahman, R.; Osteso-Ibanez, T.; Hirst, R.A.; Levesley, J.; Kilday, J.-P.; Quinn, S.; Peet, A.; O’callaghan, C.; Coyle, B.; Grundy, R.G. Histone Deacetylase Inhibition Attenuates Cell Growth with Associated Telomerase Inhibition in High Grade Childhood Brain Tumor Cells. Mol. Cancer Ther. 2010, 9, 2568–2581. [Google Scholar] [CrossRef] [PubMed]
- Tsurutani, J.; Soda, H.; Oka, M.; Suenaga, M.; Doi, S.; Nakamura, Y.; Nakatomi, K.; Shiozawa, K.; Yamada, Y.; Kamihira, S.; et al. Antiproliferative Effects of the Histone Deacetylase Inhibitor FR901228 on Small-Cell Lung Cancer Lines and Drug-Resistant Sublines. Int. J. Cancer 2003, 104, 238–242. [Google Scholar] [CrossRef] [PubMed]
- Suenaga, M.; Soda, H.; Oka, M.; Yamaguchi, A.; Nakatomi, K.; Shiozawa, K.; Kawabata, S.; Kasai, T.; Yamada, Y.; Kamihira, S.; et al. Histone Deacetylase Inhibitors Suppress Telomerase Reverse Transcriptase MRNA Expression in Prostate Cancer Cells. Int. J. Cancer 2002, 97, 621–625. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Min, N.Y.; Park, J.; Kim, J.H.; Park, S.H.; Ko, Y.J.; Kang, Y.; Moon, Y.J.; Rhee, S.; Ham, S.W.; et al. TSA-Induced DNMT1 down-Regulation Represses HTERT Expression via Recruiting CTCF into Demethylated Core Promoter Region of HTERT in HCT116. Biochem. Biophys. Res. Commun. 2010, 391, 449–454. [Google Scholar] [CrossRef]
- Jamiruddin, M.R.; Kaitsuka, T.; Hakim, F.; Fujimura, A.; Wei, F.Y.; Saitoh, H.; Tomizawa, K. HDAC9 Regulates the Alternative Lengthening of Telomere (ALT) Pathway via the Formation of ALT-Associated PML Bodies. Biochem. Biophys. Res. Commun. 2016, 481, 25–30. [Google Scholar] [CrossRef]
| Cell Types | Region Tested | Methylation Status | Reference |
|---|---|---|---|
| SUSM-1, CMV, SiHA, MDA-231/435, Calu 1/3/6, HTB 57/178/182, SW480, HTC 116 (Telomerase +) | From −500 UTSS to +50 first exon (72 CpG sites) | Partial or total methylation | [124] |
| A549, HTB183 (Telomerase +) NHF, MRC-5/p27 (Telomerase −) | From −500 UTSS to +50 first exon (72 CpG sites) | Unmethylated | [124] |
| U2OS, telomerase-negative breast carcinoma, VA13 GM847 | Promoter region | Partial or total methylation | [127] |
| CT1485 (Telomerase +) WI38, HA-1 pre-crisis cell strain, JFCF-6T/5K pre-crisis cell strain, IMR90, BJ fibroblast, adrenal carcinoma (Telomerase −) | Promoter region | Unmethylated | [127] |
| J82, T24, MCF-7, A431, HeLa, Co115, HT29, SW480, H520, SW2, PC-3, | 27 CpG sites −441 to −218 relative to UTSS | Partial or total hypermethylation and correlation with TERT mRNA expression and telomerase activity | [17] |
| Tumour tissues from brain, breast, bladder, colon, kidney, lung, soft tissue | |||
| Saos-2, U2-OS (Telomerase −) | 27 CpG sites −441 to −218 relative to UTSS | Partial or total hypermethylation | [17] |
| Normal tissues; skin, brain, bladder, muscle, kidney, heart, placenta, testis, colon | Unmethylation and telomerase negative | ||
| HeLa, SW480, Tumour tissues (breast, bladder and cervix) (Telomerase +) | −100 to +100 | 75 to 100% methylation | [131] |
| −165 to −100 | 0 to 55% methylation | ||
| BJ fibroblasts (Telomerase −) | −165 to +100 relative to UTSS | 3 to 23% hypomethylation | [131] |
| Caco-2, HCT116, RKO, SW480, MCF7, MDA-MB-231, MDA-MB-435S, MDA-MB-453, H82, H157, H209, H146, H358, H417, H549, H747, H1299, U1752, DMS53, HL-60, KG-1a, Jurkat, Raji, LCL | −600 bp relative to UTSS | Partial or total methylation and TERT expression | [136] |
| −150 to +150 relative to UTSS | Partial or unmethylation | ||
| Hepatocarcinoma cell lines HepG2, SNU-182, SNU-398, HCC Tissue (Telomerase +) | −165 to +49 relative to UTSS | 10 HCC clones and HepG2 Hypo/unmethylated 12 HCC clones and SNU-182, SNU-398 hypermethylated and reduced TERT | [137] |
| Normal liver tissue (Telomerase −) | −165 to +49 relative to UTSS | Hypermethylated and reduced TERT | [137] |
| Malignant pediatric brain tumours | 5 CpG sites located UTSS | Hypermethylation TERT expression | [128] |
| Neuroblastoma | UTSS | Highly methylated | [120] |
| Hepatocellular carcinoma | −452 to −667 and −974 to −1419 relative to UTSS | Hypermethylation and high TERT expression | [138] |
| Gastric cancer | 25 CpG sites located −555 from UTSS | Hypermethylation and high TERT expression | [134] |
| Haematopoietic cell lines including Jurkat, THP1, K562, cervical cancer and embryonic kidney cells | −60 to +20 relative to ATG | Hypermethylation and TERT expression | [117] |
| Normal peripheral blood cell populations (granulocytes, T cells, B cells and monocytes) | −60 to +20 relative to ATG | Unmethylated | [117] |
| Acute myeloid leukaemia, myelodysplastic syndrome | −520 to −400 relative to ATG | Hypermethylation | [117] |
| Thyroid cancer | −541 to −578 relative to ATG | Hypermethylation | [139] |
| Prostate cancer | 52 CpG –140 to –572 relative to the UTSS | Hypermethylation | [140] |
| Melanoma | 26 CpG –482 to –667, relative to the ATG | Hypermethylation and positively correlated with the TERT expression | [141] |
| Melanoma | −945 to −669 bp relative to UTSS | Hypermethylation and positively correlated with the TERT expression | [142] |
| Pancreatic cancer | Position –575 relative to the UTSS | Hypermethylation | [143] |
| Colon, blood, breast, prostate, brain, lungs, bladder, ovaries, bone, skin cancers | 52 CpG sites −100 to −600 located UTSS | Hypermethylation and upregulated TERT expression | [129] |
| Bladder cancers | 5 CpG sites in –140 to –572 region relative to UTSS | Hypermethylation and higher TERT expression | [132,144] |
| Primary metastatic medulloblastoma | 5 CpG sites localized −600 bp UTSS region | Hypermethylation | [145] |
| Thyroid Cancer Cell Lines | −662 to +174 relative to UTSS | Hypermethylation of upstream promoter and correlated with TERT expression | [146] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dogan, F.; Forsyth, N.R. Telomerase Regulation: A Role for Epigenetics. Cancers 2021, 13, 1213. https://doi.org/10.3390/cancers13061213
Dogan F, Forsyth NR. Telomerase Regulation: A Role for Epigenetics. Cancers. 2021; 13(6):1213. https://doi.org/10.3390/cancers13061213
Chicago/Turabian StyleDogan, Fatma, and Nicholas R. Forsyth. 2021. "Telomerase Regulation: A Role for Epigenetics" Cancers 13, no. 6: 1213. https://doi.org/10.3390/cancers13061213
APA StyleDogan, F., & Forsyth, N. R. (2021). Telomerase Regulation: A Role for Epigenetics. Cancers, 13(6), 1213. https://doi.org/10.3390/cancers13061213







