No Impact of NRAS Mutation on Features of Primary and Metastatic Melanoma or on Outcomes of Checkpoint Inhibitor Immunotherapy: An Italian Melanoma Intergroup (IMI) Study
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
2.1. Demographics
2.2. Characteristics of Primary Melanoma, Disease-Free Interval and Metastatic Disease
2.3. Treatments Used for Metastatic Disease
2.4. Response to Treatment
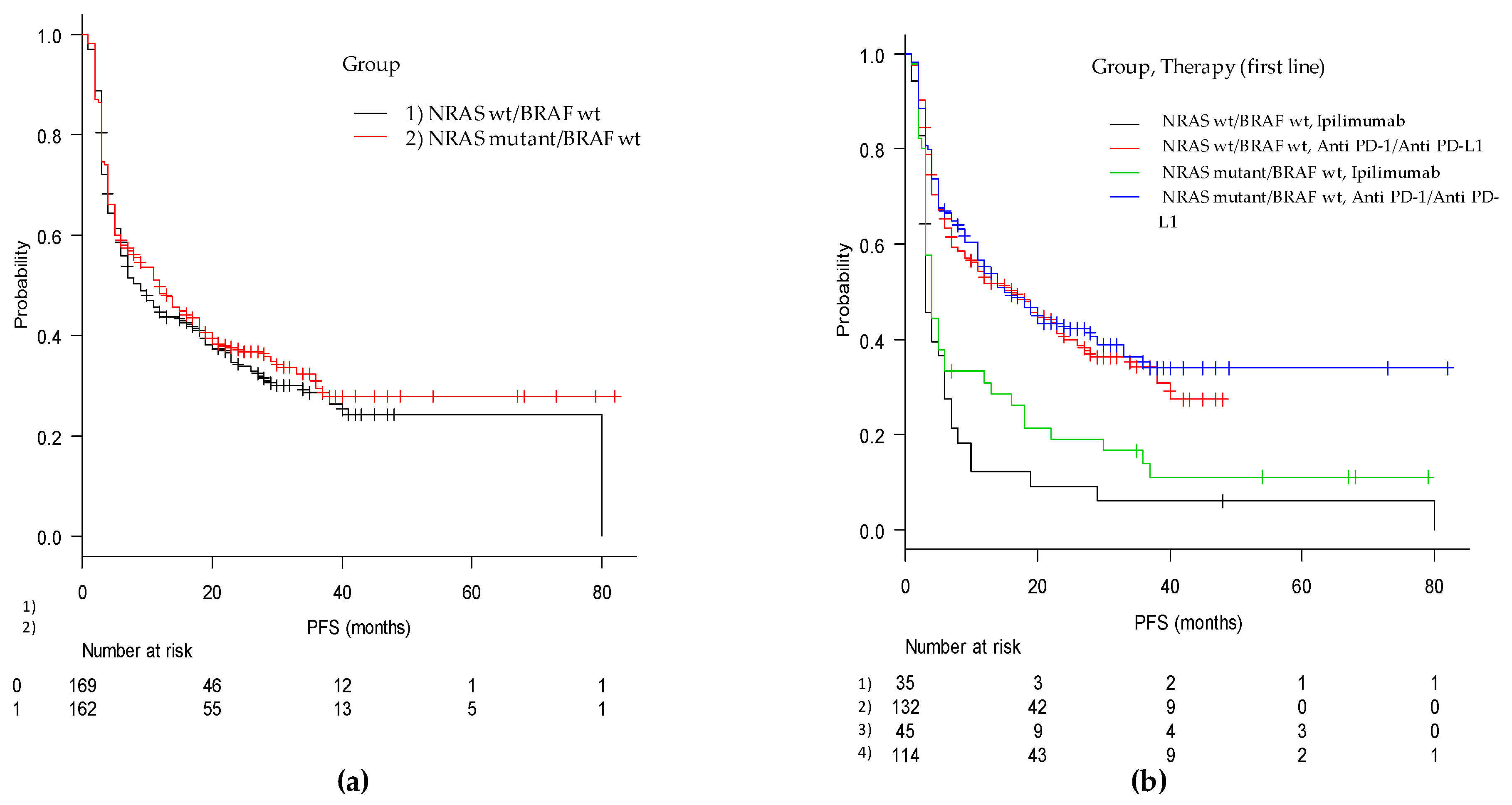
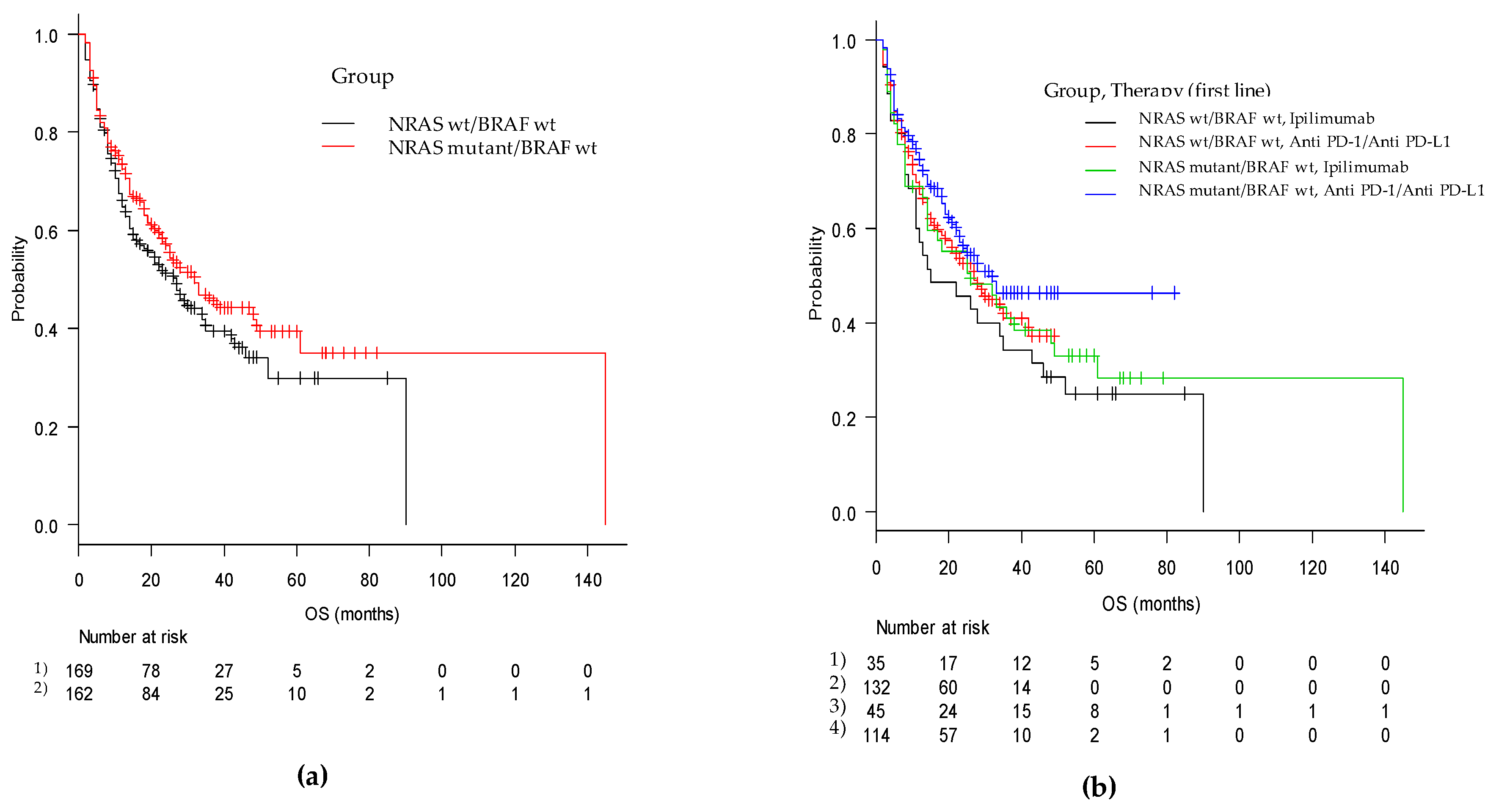
2.5. Progression-Free Survival and Overall Survival
3. Discussion
4. Materials and Methods
4.1. Patients and Study Design
4.2. Genetic Analysis
4.3. Treatment and Clinical Outcomes
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shain, A.H.; Yeh, I.; Kovalyshyn, I.; Sriharan, A.; Talevich, E.; Gagnon, A.; Dummer, R.; North, J.; Pincus, L.; Ruben, B.; et al. The genetic Evolution of Melanoma from precursor lesions. N. Engl. J. Med. 2015, 373, 1926–1936. [Google Scholar] [CrossRef] [PubMed]
- Long, G.V.; Eroglu, Z.; Infante, J.; Patel, S.; Daud, A.; Johnson, D.B.; Gonzalez, R.; Kefford, R.; Hamid, O.; Schuchter, L.; et al. Long-term outcomes in patients with Braf V600-mutant metastatic melanoma who received dabrafenib combined with trametinib. J. Clin. Oncol. 2018, 36, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Hodi, F.S.; O’Day, S.J.; McDermott, D.F.; Patel, S.; Daud, A.; Johnson, D.B.; Gonzalez, R.; Kefford, R.; Hamid, O.; Schuchter, L.; et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 2010, 363, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Larkin, J.; Chiarion-Sileni, V.; Gonzalez, R.; Grob, J.J.; Rutkowski, P.; Lao, C.D.; Cowey, C.L.; Schadendorf, D.; Wagstaff, J.; Dummer, R.; et al. Five-year survival with combined nivolumab and ipilimumab in advanced melanoma. N. Engl. J. Med. 2019, 381, 1535–1546. [Google Scholar] [CrossRef] [PubMed]
- Devitt, B.; Liu, W.; Salemi, R.; Wolfe, R.; Kelly, J.; Tzen, C.Y.; Dobrovic, A.; McArthur, G. Clinical outcome and pathological features associated with NRAS mutation in cutaneous melanoma. Pigment. Cell. Melanoma Res. 2011, 24, 666–672. [Google Scholar] [CrossRef]
- Jakob, J.A.; Bassett, R.L.; Ng, C.S.; Curry, J.L.; Joseph, R.W.; Alvarado, G.C.; Rohlfs, M.L.; Richard, J.; Gershenwald, J.E.; Kim, K.B.; et al. NRAS mutation status is an independent prognostic factor in metastatic melanoma. Cancer 2012, 118, 4014–4023. [Google Scholar] [CrossRef]
- Bertoli, E.; Giavarra, M.; Vitale, M.G.; Minisini, A.M. Neuroblastoma rat sarcoma mutated melanoma: That’s what we got so far. Pigment. Cell. Melanoma Res. 2019, 32, 744–752. [Google Scholar] [CrossRef]
- Wu, S.; Kuo, H.; Li, W.Q.; Canales, A.L.; Han, J.; Qureshi, A.A. Association between BRAFV600E and NRASQ61R mutations and clinicopathologic characteristics, risk factors and clinical outcome of primary invasive cutaneous melanoma. Cancer Causes Control 2014, 25, 1379–1386. [Google Scholar] [CrossRef]
- Dummer, R.; Schadendorf, D.; Ascierto, P.A.; Arance, A.; Dutriaux, C.; Di Giacomo, A.M.; Rutkowski, P.; Del Vecchio, M.; Gutzmer, R.; Mandala, M.; et al. Binimetinib versus dacarbazine in patients with advanced NRAS-mutant melanoma (NEMO): A multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2017, 18, 435–445. [Google Scholar] [CrossRef]
- Robert, C.; Long, G.V.; Brady, B.; Dutriaux, C.; Maio, M.; Mortier, L.; Hassel, J.C.; Rutkowski, P.; McNeil, C.; Kalinka-Warzocha, E.; et al. Nivolumab in previously untreated melanoma without BRAF mutation. N. Engl. J. Med. 2015, 372, 320–330. [Google Scholar] [CrossRef]
- Joseph, R.W.; Sullivan, R.J.; Harrell, R.; Stemke-Hale, K.; Panka, D.; Manoukian, G.; Percy, A.; Bassett, R.L.; Ng, C.S.; Radvanyi, L.; et al. Correlation of NRAS mutations with clinical response to high-dose IL-2 in patients with advanced melanoma. J. Immun. 2012, 35, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.B.; Lovly, C.M.; Flavin, M.; Panageas, K.S.; Ayers, G.D.; Zhao, Z.; Iams, W.T.; Colgan, M.; DeNoble, S.; Terry, C.R.; et al. Impact of NRAS mutations for patients with advanced melanoma treated with immune therapies. Cancer Immunol. Res. 2015, 3, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Kirchberger, M.C.; Ugurel, S.; Mangana, J.; Heppt, M.V.; Eigentler, T.K.; Berking, C.; Schadendorf, D.; Schuler, G.; Dummer, R.; Heinzerling, L. MEK inhibition may increase survival of NRAS-mutated melanoma patients treated with checkpoint blockade: Results of a retrospective multicentre analysis of 364 patients. Eur. J. Cancer 2018, 98, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Lo, S.K.; Li, I.T.; Tsou, T.S.; See, L. Non-significant in univariate but significant in multivariate analysis: A discussion with examples. Chang. Yi Xue Za Zhi 1995, 18, 95–101. [Google Scholar]
- Heppt, M.V.; Siepmann, T.; Engel, J.; Schubert-Fritschle, G.; Eckel, R.; Mirlach, L.; Kirchner, T.; Jung, A.; Gesierich, A.; Ruzicka, T.; et al. Prognostic significance of BRAF and NRAS mutations in melanoma: A German study from routine care. BMC Cancer 2017, 17, 536. [Google Scholar] [CrossRef]
- Carlino, M.S.; Haydu, L.E.; Kakavand, H.; Menzies, A.M.; Hamilton, A.L.; Yu, B.; Ng, C.C.; Cooper, W.A.; Thompson, J.F.; Kefford, R.F.; et al. Correlation of BRAF and NRAS mutation status with outcome, site of distant metastasis and response to chemotherapy in metastatic melanoma. Br. J. Cancer 2014, 111, 292–299. [Google Scholar] [CrossRef]
- Bersanelli, M.; Leonetti, A.; Buti, S.; Michiara, M. “Finding NEMO” in NRAS-mutant melanoma: A step towards a sequential strategy? Lancet Oncol. 2017, 18, e298. [Google Scholar] [CrossRef][Green Version]
- Snyder, A.; Makarov, V.; Merghoub, T.; Yuan, J.; Zaretsky, J.M.; Desrichard, A.; Walsh, L.A.; Postow, M.A.; Wong, P.; Ho, T.S.; et al. Genetic basis for clinical response to CTLA-4 blockade in melanoma. N. Engl. J. Med. 2014, 371, 2189–2199. [Google Scholar] [CrossRef]
- Atefi, M.; Avramis, E.; Lassen, A.; Wong, D.J.L.; Robert, L.; Foulad, D.; Cerniglia, M.; Titz, B.; Chodon, T.; Graeber, T.G.; et al. Effects of MAPK and PI3K pathways on PD-L1 expression in melanoma. Clin. Cancer Res. 2014, 20, 3446–3457. [Google Scholar] [CrossRef]
- Mangana, J.; Cheng, P.F.; Schindler, K.; Weide, B.; Held, U.; Frauchiger, A.L.; Romano, E.; Kähler, K.C.; Rozati, S.; Rechsteiner, M.; et al. Analysis of BRAF and NRAS muta- tion status in advanced melanoma patients treated with anti-CTLA-4 antibodies: Association with overall survival? PLoS ONE 2015, 10, e0139438. [Google Scholar] [CrossRef]
- Ribas, A.; Puzanov, I.; Dummer, R.; Schadendorf, D.; Hamid, O.; Robert, C.; Hodi, F.S.; Schachter, J.; Pavlick, A.C.; Lewis, K.D.; et al. Pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory melanoma (KEYNOTE-002): A randomised, controlled, phase 2 trial. Lancet Oncol. 2015, 16, 908–918. [Google Scholar] [CrossRef]
- Ferrucci, P.F.; Ascierto, P.A.; Pigozzo, J.; Del Vecchio, M.; Maio, M.; Antonini Cappellini, G.C.; Guidoboni, M.; Queirolo, P.; Savoia, P.; Mandalà, M.; et al. Baseline neutrophils and derived neutrophil-to-lymphocyte ratio: Prognostic relevance in metastatic melanoma patients receiving ipilimumab. Ann. Oncol. 2016, 27, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Capone, M.; Giannarelli, D.; Mallardo, D.; Madonna, G.; Festino, L.; Grimaldi, A.M.; Vanella, V.; Simeone, E.; Paone, M.; Palmieri, G.; et al. Baseline neutrophil-to-lymphocyte ratio (NLR) and derived NLR could predict overall survival in patients with advanced melanoma treated with nivolumab. J. Immunother. Cancer 2018, 6, 74. [Google Scholar] [CrossRef] [PubMed]
- Guida, M.; Bartolomeo, N.; De Risi, I.; Fucci, L.; Armenio, A.; Filannino, R.; Ruggieri, E.; Macina, F.; Traversa, M.; Nardone, A.; et al. The management of oligoprogression in the landscape of new therapies for metastatic melanoma. Cancers 2019, 11, 1559. [Google Scholar] [CrossRef]
- Bartlett, E.K.; Flynn, J.R.; Panageas, K.S.; Ferraro, R.A.; Sta Cruz, J.M.; Postow, M.A.; Coit, D.G.; Ariyan, C.E. High neutrophil-to-lymphocyte ratio (NLR) is associated with treatment failure and death in patients who have melanoma treated with PD-1 inhibitor monotherapy. Cancer 2020, 126, 76–85. [Google Scholar] [CrossRef]
- Zhang, F.; Gorg, W. Prognostic value of the platelet-to-lymphocyte ratio in patients with melanoma: A meta-analysis. Front. Oncol. 2020, 10, 1116. [Google Scholar] [CrossRef]
- Ma, W.; Zhang, P.; Qi, J.; Gu, L.; Zang, M.; Yao, H.; Shi, X.; Wang, C.; Jiang, Y. Prognostic value of platelet to lymphocyte ratio in hepatocellular carcinoma: A meta-analysis. Sci. Rep. 2016, 6, 35378. [Google Scholar] [CrossRef]
- Gasparyan, A.Y.; Ayvazyan, L.; Mikhailidis, D.P.; Kitas, G.D. Mean platelet volume: A link between thrombosis and inflammation? Curr. Pharm. Des. 2011, 17, 47–58. [Google Scholar] [CrossRef]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised recist guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef]
- Cortellini, A.; Palumbo, P.; Porzio, G.; Verna, L.; Giordano, A.V.; Masciocchi, C.; Parisi, A.; Cannita, K.; Ficorella, C.; Bozzetti, F. Single-institution study of correlations between skeletal muscle mass, its density, and clinical outcomes in non-small cell lung cancer patients treated with first-line chemotherapy. Thorac. Cancer 2018, 9, 1623–1630. [Google Scholar] [CrossRef]
- Kanda, Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transp. 2013, 48, 452–458. [Google Scholar] [CrossRef] [PubMed]
| Clinical Features | NRAS Mutant/BRAF wt n (%) | NRAS wt/BRAF wt n (%) | p Value * |
|---|---|---|---|
| Total | 162 | 169 | |
| Origin of melanoma | |||
| Cutaneous Unknown | 137 (85) 25 (15) | 147 (87) 22 (13) | 0.53 |
| Gender | |||
| Female Male | 58 (36) 104 (64) | 69 (41) 100 (59) | 0.35 |
| Adjuvant therapy | |||
| Yes No | 22 (14) 140 (86) | 13 (8) 156 (92) | 0.08 |
| Therapy (first line) | |||
| Anti PD-1 Ipilimumab Anti PD-1 + Ipilimumab | 114 (70) 45 (28) 3 (2) | 132 (78) 35 (21) 2 (1) | 0.27 |
| Therapy (second line) | |||
| Yes No | 56 (35) 106 (65) | 61 (36) 108 (64) | 0.77 |
| Therapy (third line) | |||
| Yes No | 17 (10) 145 (90) | 10 (6) 159 (94) | 0.13 |
| Disease Features | NRAS Mutant/BRAF wt | NRAS wt/BRAF wt | p Value |
|---|---|---|---|
| Age at diagnosis, years | 0.86 * | ||
| Median (IQR) | 63.4 (53.3–73.8) | 65 (54.0–73.0) | |
| Site (%) | < 0.001 ** | ||
| Head & neck | 11 (7) | 36 (21) | |
| Trunk | 65 (40) | 50 (30) | |
| Upper limbs | 20 (12) | 16 (9) | |
| Lower limbs | 41 (25) | 37 (22) | |
| Other | 0 | 8 (5) | |
| Unknown | 25 (15) | 22 (13) | |
| Thickness (%) | 0.76 ** | ||
| pT1 | 10 (7) | 10 (6) | |
| pT2 | 25 (17) | 21 (13) | |
| pT3 | 35 (23) | 38 (24) | |
| pT4 | 56 (37) | 67 (42) | |
| Unknown | 24 (16) | 22 (14) | |
| Ulceration (%) | 0.03 ** | ||
| yes | 71(44) | 91 (54) | |
| No | 51 (31) | 32 (19) | |
| Unknown | 40 (25) | 46 (27) | |
| Lymph node status 1 (%) | 0.48 ** | ||
| Positive | 70 (55) | 68 (51) | |
| Negative | 57 (45) | 66 (49) | |
| DFI (months) | 0.97 * | ||
| Median (IQR) | 15.4 (4–36) | 15 (3–37) |
| Clinical Features | NRAS Mutant/BRAF wt | NRAS wt/BRAF wt | p Value |
|---|---|---|---|
| Age at metastatic disease, years | |||
| Median (IQR) | 68 (54–76) | 68.7 (56–76) | 0.94 * |
| Serum LDH, n (%) | |||
| Normal | 92 (57) | 90 (53) | 0.67 ** |
| Elevated | 47 (29) | 51 (30) | |
| Unspecified | 22 (14) | 28 (17) | |
| Blood cellular count (IQR) | |||
| WBC (103/μL) | 6.7 (5.67–8.20) | 6.8 (5.55–7.88) | 0.77 * |
| Lymphocytes (103/μL) | 1.66 (1.31–2.06) | 1.65 (1.31–2.1) | 0.99 * |
| N/L | 2.51 (1.89–3.60) | 2.51 (1.67–3.6) | 0.89 * |
| Platelets (103/μL) | 229 (194.7–297.2) | 236.5 (181–288.2) | 0.82 * |
| Site of disease, n (%) ^ | |||
| Skin/Soft tissue | 90 (56) | 77 (46) | 0.07 ** |
| Lymph node | 112 (69) | 102 (60) | 0.09 ** |
| Lung | 75 (46) | 103 (61) | <0.01 ** |
| Liver | 34 (21) | 29 (17) | 0.37 ** |
| Brain | 12 (7) | 28 (17) | 0.01 ** |
| Bone | 20 (12) | 16 (9) | 0.40 ** |
| Other | 33 (20) | 16 (9) | <0.01 ** |
| N. of metastatic sites, n (%) | |||
| <3 | 94 (58) | 108 (64) | 0.27 ** |
| ≥3 | 68 (42) | 61 (36) | |
| Stage at metastatic disease, n (%) | |||
| III | 1 (1) | 2 (1) | <0.01 ** |
| IVA | 46 (28) | 38 (22) | |
| IVB | 41(25) | 56 (33) | |
| IVC | 64 (39) | 45 (27) | |
| IVD | 10 (6) | 28 (17) | |
| Brain progression, n (%) | |||
| Yes | 17 (19) | 35 (37) | <0.01 ** |
| No | 72 (81) | 59(63) | |
| ECOG 1 PS, n (%) | |||
| 0–1 | 123 (76.4) | 129 (76.3) | 0.91 ** |
| 2 | 36 (22.4) | 37 (21.9) | |
| 3 | 2 (1.2) | 3 (1.8) |
| Therapy | Response | NRAS Mutant/BRAF wt n (%) | NRAS wt/BRAF wt n (%) | p Value * |
|---|---|---|---|---|
| All patients | ORR | 68 (42) | 63 (37) | 0.38 |
| DCR | 97 (60) | 100 (59) | 0.90 | |
| Anti-PD-1 | ORR | 49 (43) | 56 (42) | 0.93 |
| DCR | 78 (68) | 88 (67) | 0.77 | |
| Ipilimumab | ORR | 16 (36) | 6 (17) | 0.07 |
| DCR | 16 (36) | 11 (31) | 0.70 |
| NRAS Mutation | Frequency n (%) | ORR n (%) | p Value * | PFS, Median, Months (IQR) | p Value ** | OS, Median, Months (IQR) | p Value ** |
|---|---|---|---|---|---|---|---|
| Q61K | 60 (37) | 24 (40) | 0.41 | 7.5 (4–20) | 0.35 | 26 (14–48) | 0.39 |
| Q61R | 56 (35) | 25 (45) | 12 (5–20) | 28 (18-NA) | |||
| Q61L | 17 (10) | 8 (47) | 18 (3-NA) | NA (5-NA) | |||
| Q61H | 9 (6) | 4 (44) | 33 (3-NA) | NA (15-NA) | |||
| Not Q61 | 8 (5) | 5 (62) | 17 (4-NA) | 22 (8-NA) | |||
| Unspecified | 12 (7) | 2(17) | 5 (2–20) | 33 (3-NA) |
| Parameter | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| OR | 95% CI | p Value | OR | 95% CI | p Value | |
| Sex (M versus F) | 1.09 | (0.69–1.71) | 0.72 | - | - | - |
| Age (+1) | 1.00 | (0.99–1.02) | 0.81 | - | - | - |
| NRAS status (mut versus wt) | 1.03 | (0.66–1.60) | 0.90 | 1.95 | (1.07–3.54) | 0.03 |
| Site of primary melanoma | <0.001 | 0.05 | ||||
| Head & neck versus unknown | 1.11 | (0.45–2.71) | 1.43 | (0.42–4.85) | ||
| Trunk versus unknown | 0.53 | (0.26–1.10) | 0.50 | (0.20–1.25) | ||
| Upper limbs versus unknown | 1.10 | (0.42–2.88) | 0.90 | (0.28–2.92) | ||
| Lower limbs versus unknown | 0.31 | (0.14–0.67) | 0.37 | (0.14–0.98) | ||
| Other versus unknown | 2.97 | (0.33–26.4) | 3.18 | (0.29–35.2) | ||
| AntiPD-1 versus ipilimumab | 4.07 | (2.39–6.95) | <0.001 | 5.81 | (2.78–12.1) | <0.001 |
| N. metastatic sites (<3 versus ≥3) | 1.96 | (1.25–3.07) | <0.01 | 2.64 | (1.43–4.88) | <0.01 |
| WBC (+1000) | 0.95 | (0.90–1.01) | 0.10 | - | - | - |
| Lymphocytes (+1000) | 0.91 | (0.77–1.08) | 0.30 | 0.78 | (0.55–1.11) | 0.16 |
| N/L ratio (+1) | 0.88 | (0.77–1.00) | 0.05 | 0.91 | (0.79–1.04) | 0.17 |
| Platelet (+100) | 0.66 | (0.50–0.87) | <0.01 | 0.67 | (0.47–0.96) | 0.03 |
| LDH (<ULM versus >ULM) | 1.73 | (1.04–2.86) | 0.03 | - | - | - |
| ECOG PS (2–3 versus 0–1) | 0.40 | (0.24–0.67) | <0.01 | 0.40 | (0.20–0.82) | 0.01 |
| Parameter | Univariate | Multivariable | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p Value | HR | 95% CI | p Value | |
| Sex (M versus F) | 0.97 | (0.74–1.27) | 0.81 | 1.39 | (0.99–1.96) | 0.05 |
| Age (+1) | 1.01 | (0.99–1.02) | 0.34 | - | - | - |
| NRAS status (mut versus wt) | 0.92 | (0.70–1.20) | 0.53 | 0.73 | (0.53–1.00) | 0.05 |
| Site of primary melanoma | 0.01 | 0.03 | ||||
| Head & neck versus unknown | 0.63 | (0.37–1.06) | 0.53 | (0.27–1.04) | ||
| Trunk versus unknown | 1.02 | (0.68–1.54) | 1.12 | (0.70–1.81) | ||
| Upper limbs versus unknown | 0.57 | (0.32–1.03) | 0.58 | (0.30–1.14) | ||
| Lower limbs versus unknown | 1.30 | (0.84–2.00) | 1.16 | (0.68–1.97) | ||
| Other versus unknown | 0.73 | (0.28–1.88) | 0.50 | (0.17–1.51) | ||
| AntiPD-1 versus ipilimumab | 0.46 | (0.34–0.61) | <0.0001 | 0.41 | (0.29–0.60) | <0.0001 |
| N. metastatic sites (<3 versus ≥3) | 0.63 | (0.49–0.83) | <0.01 | 0.60 | (0.44–0.84) | <0.01 |
| WBC (+1000) | 1.01 | (1.00–1.03) | 0.15 | - | - | - |
| Lymphocytes (+1000) | 1.03 | (0.92–1.16) | 0.61 | 1.10 | - | - |
| N/L ratio (+1) | 1.04 | (1.00–1.09) | 0.06 | 1.05 | - | - |
| Platelet (+100) | 1.14 | (0.96–1.35) | 0.14 | 1.21 | (1.01–1.44) | 0.04 |
| LDH (<ULM versus >ULM) | 0.65 | (0.48–0.89) | <0.01 | 0.78 | (0.56–1.09) | 0.15 |
| ECOG PS (2–3 versus 0–1) | 2.20 | (1.65–2.93) | <0.0001 | 2.02 | (1.42–2.86) | <0.0001 |
| Parameter | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p Value | HR | 95% CI | p Value | |
| Sex (M versus F) | 1.01 | (0.75–1.37) | 0.94 | 1.48 | (1.00–2.18) | 0.05 |
| Age (+1) | 1.00 | (0.99–1.02) | 0.64 | - | - | - |
| NRAS status (mut versus wt) | 0.82 | (0.61–1.11) | 0.20 | 0.72 | (0.51–1.03) | 0.07 |
| Site of primary melanoma | 0.03 | 0.01 | ||||
| Head & neck versus unknown | 0.77 | (0.43–1.40) | 0.79 | (0.34–1.82) | ||
| Trunk versus unknown | 1.11 | (0.69–1.80) | 1.62 | (0.90–2.92) | ||
| Upper limbs versus unknown | 0.66 | (0.34–1.28) | 0.79 | (0.34–1.82) | ||
| Lower limbs versus unknown | 1.45 | (0.89–2.37) | 2.11 | (1.13–3.93) | ||
| Other versus unknown | 0.43 | (0.10–1.82) | 0.67 | (0.15–3.04) | ||
| AntiPD-1 versus ipilimumab | 0.82 | (0.59–1.13) | 0.23 | - | - | - |
| N. metastatic sites (<3 versus ≥3) | 0.65 | (0.48–0.87) | <.01 | - | - | - |
| WBC (+1000) | 1.01 | (1.00–1.04) | 0.03 | 1.02 | (1.00–1.04) | 0.04 |
| Lymphocytes (+1000) | 1.00 | (0.89–1.11) | 0.97 | - | - | - |
| N/L ratio (+1) | 1.06 | (1.02–1.11) | <0.01 | 1.05 | (0.99–1.11) | 0.09 |
| Platelet (+100) | 1.26 | (1.05–1.51) | 0.01 | 1.31 | (1.07–1.59) | <0.01 |
| LDH_ (<ULM versus >ULM) | 0.50 | (0.36–0.69) | <0.001 | 0.57 | (0.39–0.82) | <0.01 |
| ECOG PS (2–3 versus 0–1) | 1.77 | (1.27–2.45) | <0.001 | 1.52 | (1.02–2.25) | 0.04 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guida, M.; Bartolomeo, N.; Quaglino, P.; Madonna, G.; Pigozzo, J.; Di Giacomo, A.M.; Minisini, A.M.; Tucci, M.; Spagnolo, F.; Occelli, M.; et al. No Impact of NRAS Mutation on Features of Primary and Metastatic Melanoma or on Outcomes of Checkpoint Inhibitor Immunotherapy: An Italian Melanoma Intergroup (IMI) Study. Cancers 2021, 13, 475. https://doi.org/10.3390/cancers13030475
Guida M, Bartolomeo N, Quaglino P, Madonna G, Pigozzo J, Di Giacomo AM, Minisini AM, Tucci M, Spagnolo F, Occelli M, et al. No Impact of NRAS Mutation on Features of Primary and Metastatic Melanoma or on Outcomes of Checkpoint Inhibitor Immunotherapy: An Italian Melanoma Intergroup (IMI) Study. Cancers. 2021; 13(3):475. https://doi.org/10.3390/cancers13030475
Chicago/Turabian StyleGuida, Michele, Nicola Bartolomeo, Pietro Quaglino, Gabriele Madonna, Jacopo Pigozzo, Anna M. Di Giacomo, Alessandro M. Minisini, Marco Tucci, Francesco Spagnolo, Marcella Occelli, and et al. 2021. "No Impact of NRAS Mutation on Features of Primary and Metastatic Melanoma or on Outcomes of Checkpoint Inhibitor Immunotherapy: An Italian Melanoma Intergroup (IMI) Study" Cancers 13, no. 3: 475. https://doi.org/10.3390/cancers13030475
APA StyleGuida, M., Bartolomeo, N., Quaglino, P., Madonna, G., Pigozzo, J., Di Giacomo, A. M., Minisini, A. M., Tucci, M., Spagnolo, F., Occelli, M., Ridolfi, L., Queirolo, P., De Risi, I., Quaresmini, D., Gambale, E., Chiaron Sileni, V., Ascierto, P. A., Stigliano, L., Strippoli, S., & on behalf of the Italian Melanoma Intergroup (IMI) Study. (2021). No Impact of NRAS Mutation on Features of Primary and Metastatic Melanoma or on Outcomes of Checkpoint Inhibitor Immunotherapy: An Italian Melanoma Intergroup (IMI) Study. Cancers, 13(3), 475. https://doi.org/10.3390/cancers13030475










