Prognostic Value of Plasma hPG80 (Circulating Progastrin) in Metastatic Renal Cell Carcinoma
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
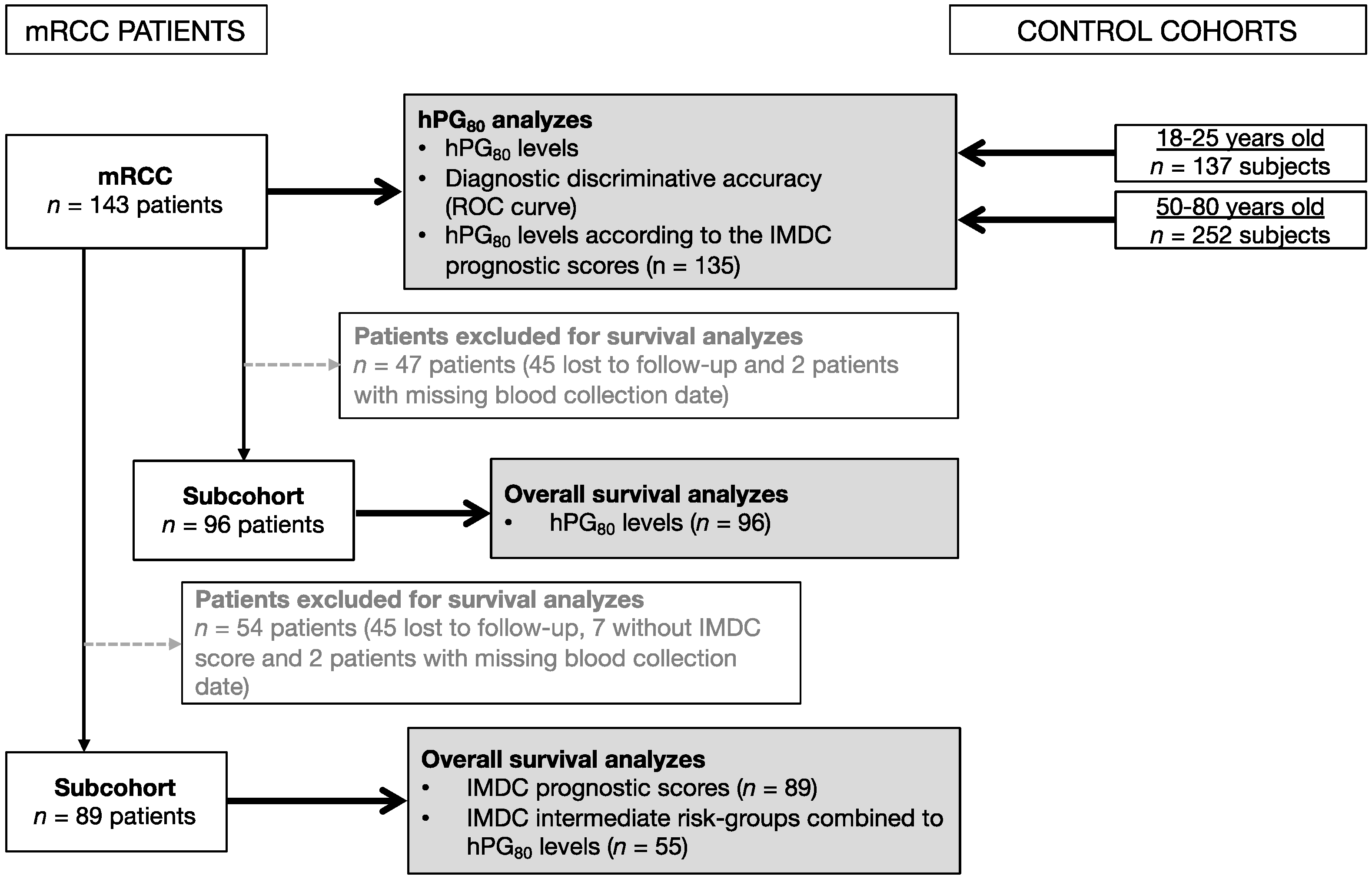
2.1. Patients and Control Cohorts
2.1.1. mRCC Patient Cohort
2.1.2. RCC Patient Cohort
2.1.3. Non-Cancer Age and Non Age Matched Control Cohorts
2.2. hPG80 Level Measurements in the Blood Samples
2.3. Statistical Analyses
3. Results
3.1. Characteristics of the Study Population
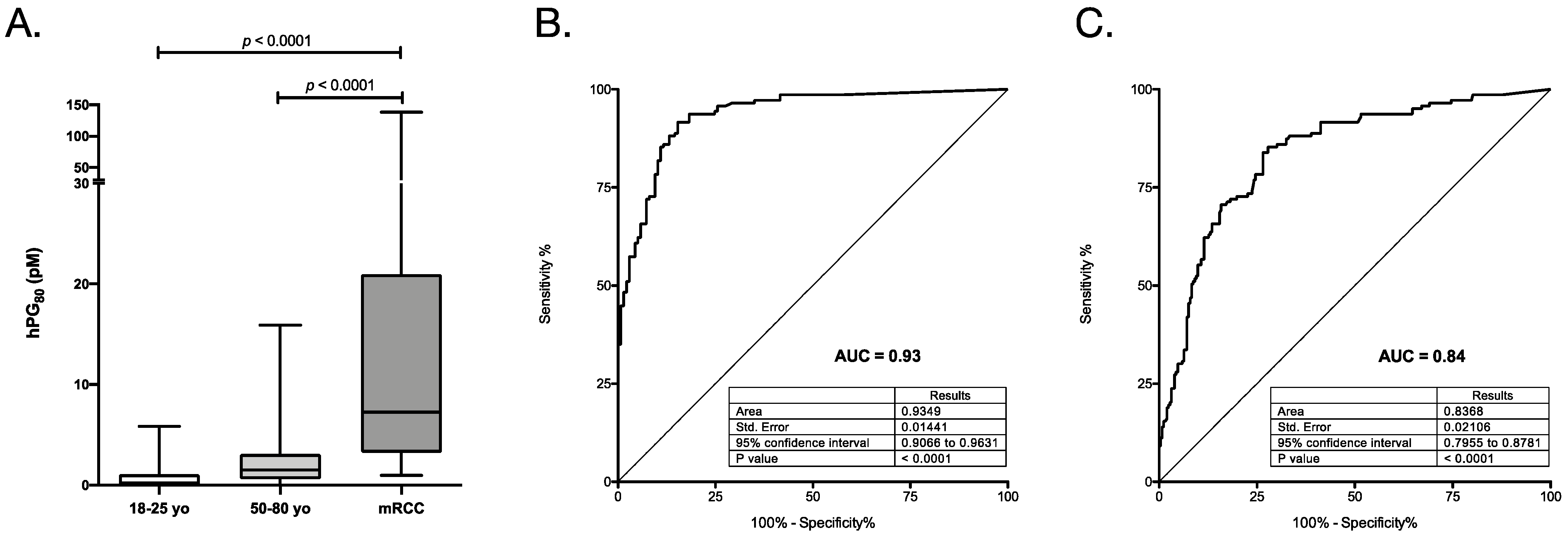
3.2. Diagnostic Performance of hPG80 in mRCC Patients
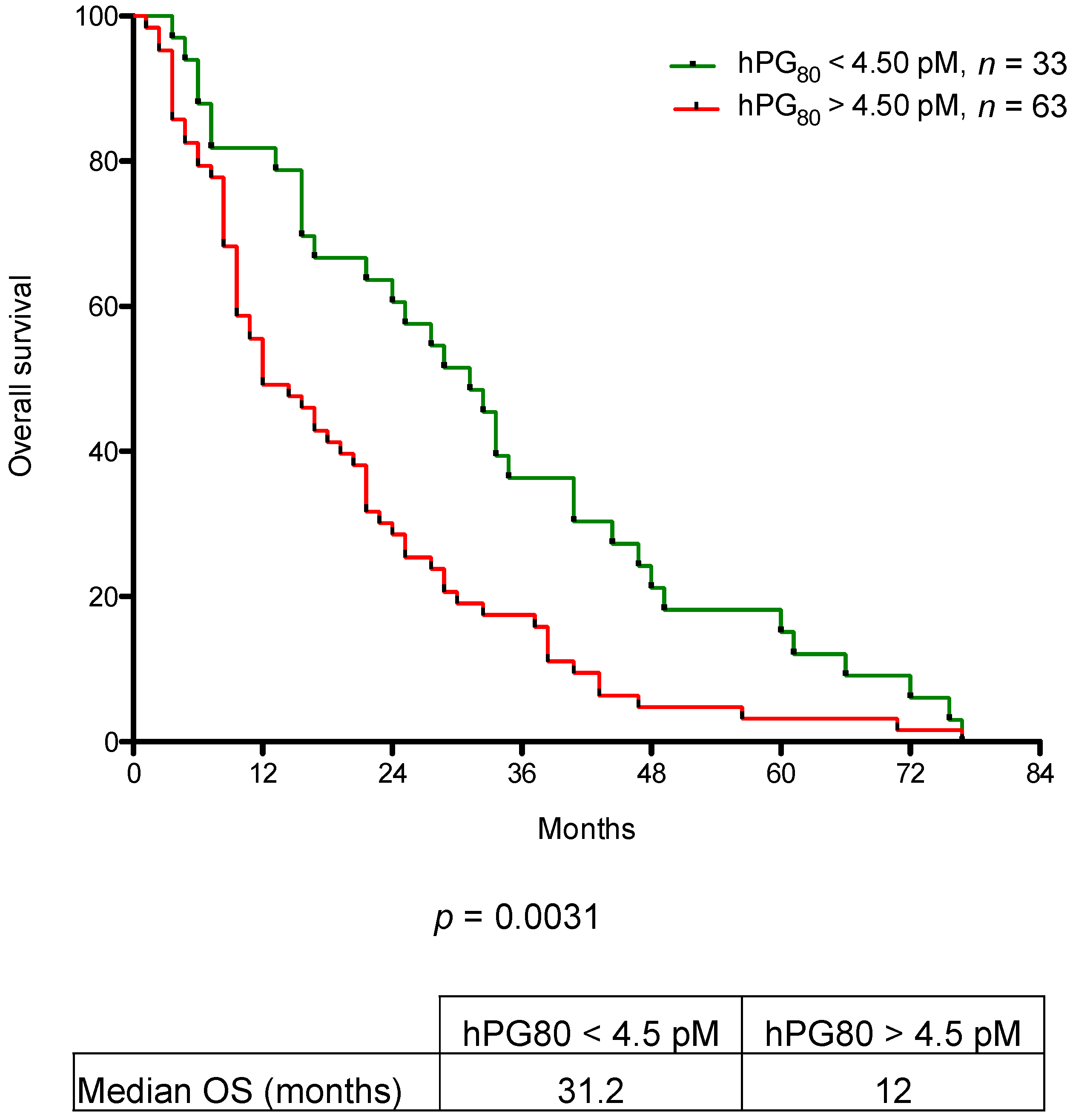
3.3. OS for Patients with mRCC Stratified Using hPG80 Levels
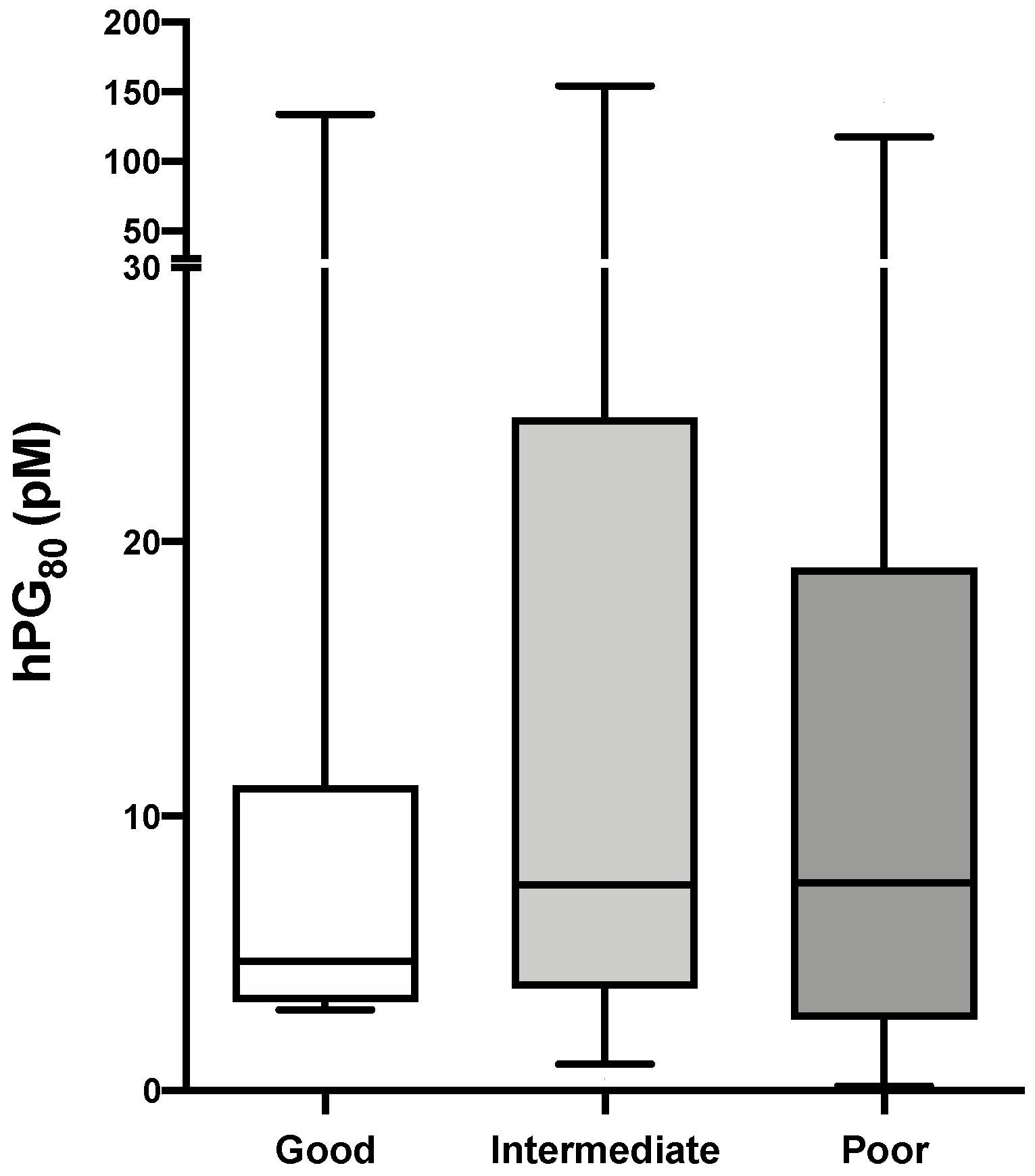
3.4. hPG80 Levels According to IMDC Prognostic Group
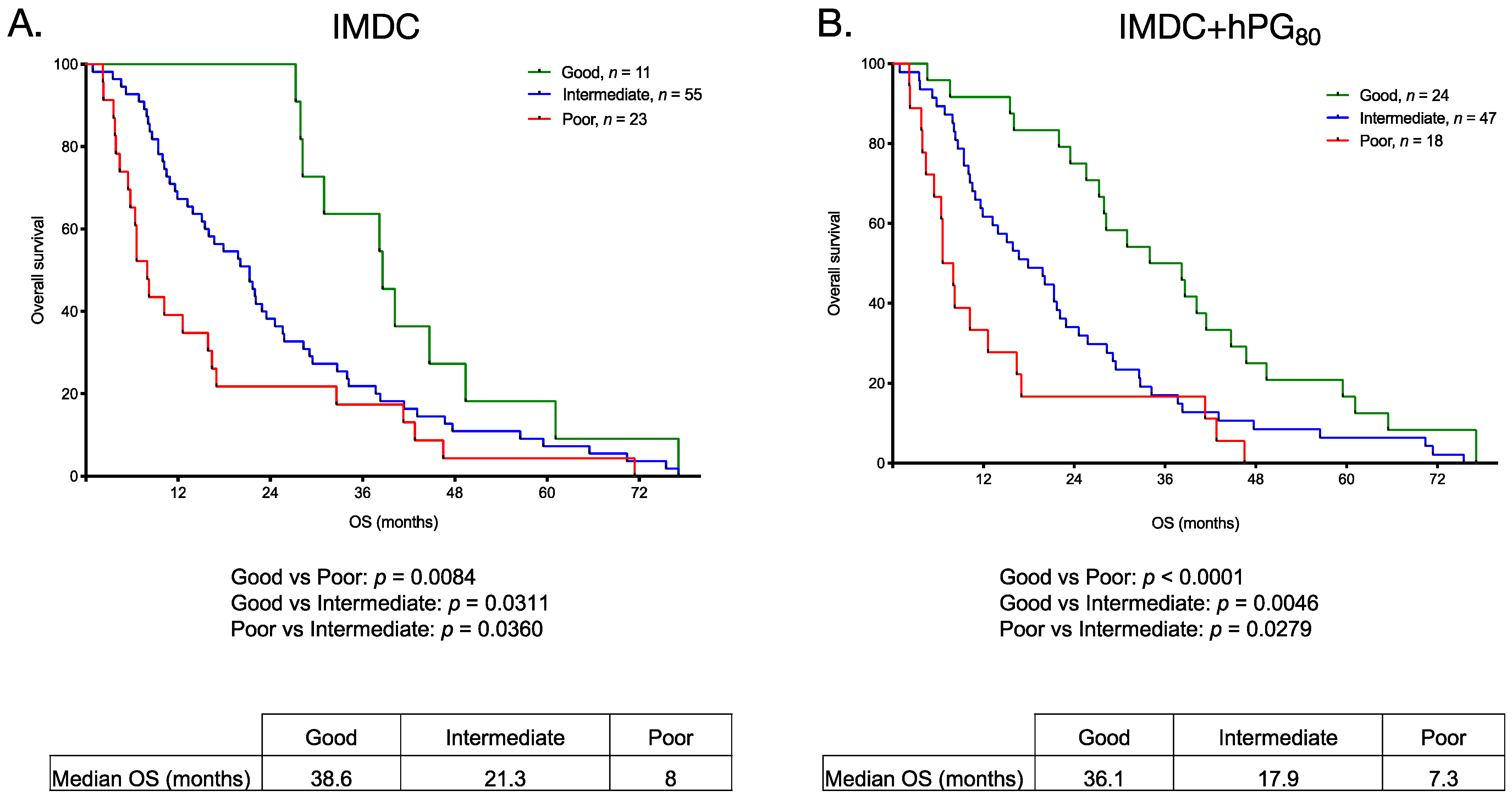
3.5. OS for Patients with mRCC Stratified Using the IMDC Risk Classification
3.6. OS for Patients with mRCC Stratified Using the IMDC Risk Classification and High and Low hPG80 Levels
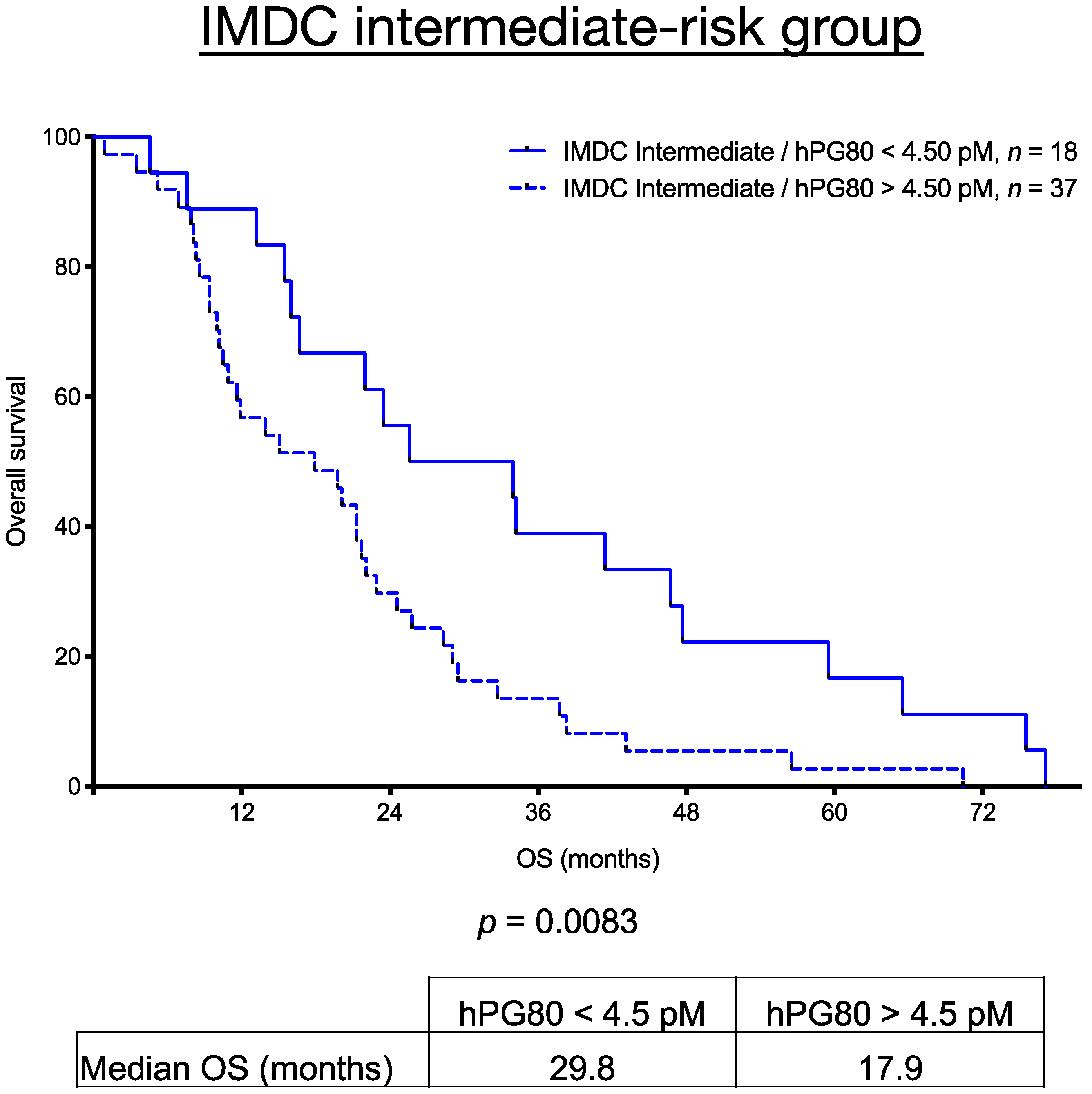
3.7. OS in The IMDC Intermediate-Risk Group Stratified Using hPG80 Levels
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rini, B.I.; Campbell, S.C.; Escudier, B. Renal cell carcinoma. Lancet 2009, 373, 1119–1132. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Barrisford, G.W.; Singer, E.A.; Rosner, I.L.; Linehan, W.M.; Bratslavsky, G. Familial renal cancer: Molecular genetics and surgical management. Int. J. Surg. Oncol. 2011, 2011, 658767. [Google Scholar] [CrossRef] [PubMed]
- Gerlinger, M.; Rowan, A.J.; Horswell, S.; Math, M.; Larkin, J.; Endesfelder, D.; Gronroos, E.; Martinez, P.; Matthews, N.; Stewart, A.; et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N. Engl. J. Med. 2012, 366, 883–892. [Google Scholar] [CrossRef] [PubMed]
- Escudier, B.; Eisen, T.; Stadler, W.M.; Szczylik, C.; Oudard, S.; Siebels, M.; Negrier, S.; Chevreau, C.; Solska, E.; Desai, A.A.; et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N. Engl. J. Med. 2007, 356, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Beisland, C.; Johannesen, T.B.; Klepp, O.; Axcrona, U.; Torgersen, K.M.; Kowalski, J.; Solli, O.; Sandin, R.; Oldenburg, J. Overall survival in renal cell carcinoma after introduction of targeted therapies: A Norwegian population-based study. Onco. Targets Ther. 2017, 10, 371–385. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Labriola, M.K.; Batich, K.A.; Zhu, J.; McNamara, M.A.; Harrison, M.R.; Armstrong, A.J.; George, D.J.; Zhang, T. Immunotherapy Is Changing First-Line Treatment of Metastatic Renal-Cell Carcinoma. Clin. Genitourin. Cancer 2019, 17, e513–e521. [Google Scholar] [CrossRef] [PubMed]
- Brannon, A.R.; Reddy, A.; Seiler, M.; Arreola, A.; Moore, D.T.; Pruthi, R.S.; Wallen, E.M.; Nielsen, M.E.; Liu, H.; Nathanson, K.L.; et al. Molecular Stratification of Clear Cell Renal Cell Carcinoma by Consensus Clustering Reveals Distinct Subtypes and Survival Patterns. Genes Cancer 2010, 1, 152–163. [Google Scholar] [CrossRef]
- Beuselinck, B.; Job, S.; Becht, E.; Karadimou, A.; Verkarre, V.; Couchy, G.; Giraldo, N.; Rioux-Leclercq, N.; Molinie, V.; Sibony, M.; et al. Molecular subtypes of clear cell renal cell carcinoma are associated with sunitinib response in the metastatic setting. Clin. Cancer Res. 2015, 21, 1329–1339. [Google Scholar] [CrossRef]
- Hakimi, A.A.; Ostrovnaya, I.; Reva, B.; Schultz, N.; Chen, Y.B.; Gonen, M.; Liu, H.; Takeda, S.; Voss, M.H.; Tickoo, S.K.; et al. Adverse outcomes in clear cell renal cell carcinoma with mutations of 3p21 epigenetic regulators BAP1 and SETD2: A report by MSKCC and the KIRC TCGA research network. Clin. Cancer Res. 2013, 19, 3259–3267. [Google Scholar] [CrossRef]
- Choueiri, T.K.; Halabi, S.; Sanford, B.L.; Hahn, O.; Michaelson, M.D.; Walsh, M.K.; Feldman, D.R.; Olencki, T.; Picus, J.; Small, E.J.; et al. Cabozantinib Versus Sunitinib As Initial Targeted Therapy for Patients With Metastatic Renal Cell Carcinoma of Poor or Intermediate Risk: The Alliance A031203 CABOSUN Trial. J. Clin. Oncol. 2017, 35, 591–597. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Tannir, N.M.; McDermott, D.F.; Aren Frontera, O.; Melichar, B.; Choueiri, T.K.; Plimack, E.R.; Barthelemy, P.; Porta, C.; George, S.; et al. Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carcinoma. N. Engl. J. Med. 2018, 378, 1277–1290. [Google Scholar] [CrossRef] [PubMed]
- Rini, B.I.; Plimack, E.R.; Stus, V.; Gafanov, R.; Hawkins, R.; Nosov, D.; Pouliot, F.; Alekseev, B.; Soulieres, D.; Melichar, B.; et al. Pembrolizumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N. Engl. J. Med. 2019, 380, 1116–1127. [Google Scholar] [CrossRef] [PubMed]
- Heng, D.Y.; Xie, W.; Regan, M.M.; Warren, M.A.; Golshayan, A.R.; Sahi, C.; Eigl, B.J.; Ruether, J.D.; Cheng, T.; North, S.; et al. Prognostic factors for overall survival in patients with metastatic renal cell carcinoma treated with vascular endothelial growth factor-targeted agents: Results from a large, multicenter study. J. Clin. Oncol. 2009, 27, 5794–5799. [Google Scholar] [CrossRef]
- Jonasch, E. NCCN Guidelines Updates: Management of Metastatic Kidney Cancer. J. Natl. Compr. Cancer Netw. 2019, 17, 587–589. [Google Scholar] [CrossRef]
- Motzer, R.J.; Jonasch, E.; Michaelson, M.D.; Nandagopal, L.; Gore, J.L.; George, S.; Alva, A.; Haas, N.; Harrison, M.R.; Plimack, E.R.; et al. NCCN Guidelines Insights: Kidney Cancer, Version 2.2020. J. Natl. Compr. Cancer Netw. 2019, 17, 1278–1285. [Google Scholar] [CrossRef]
- You, B.; Mercier, F.; Assenat, E.; Langlois-Jacques, C.; Glehen, O.; Soule, J.; Payen, L.; Kepenekian, V.; Dupuy, M.; Belouin, F.; et al. The oncogenic and druggable hPG80 (Progastrin) is overexpressed in multiple cancers and detected in the blood of patients. EBioMedicine 2019, 51, 102574. [Google Scholar] [CrossRef]
- Rehfeld, J.F.; Zhu, X.; Norrbom, C.; Bundgaard, J.R.; Johnsen, A.H.; Nielsen, J.E.; Vikesaa, J.; Stein, J.; Dey, A.; Steiner, D.F.; et al. Prohormone convertases 1/3 and 2 together orchestrate the site-specific cleavages of progastrin to release gastrin-34 and gastrin-17. Biochem. J. 2008, 415, 35–43. [Google Scholar] [CrossRef]
- Varro, A.; Voronina, S.; Dockray, G.J. Pathways of processing of the gastrin precursor in rat antral mucosa. J. Clin. Invest. 1995, 95, 1642–1649. [Google Scholar] [CrossRef]
- Siddheshwar, R.K.; Gray, J.C.; Kelly, S.B. Plasma levels of progastrin but not amidated gastrin or glycine extended gastrin are elevated in patients with colorectal carcinoma. Gut 2001, 48, 47–52. [Google Scholar] [CrossRef]
- Prieur, A.; Cappellini, M.; Habif, G.; Lefranc, M.P.; Mazard, T.; Morency, E.; Pascussi, J.M.; Flaceliere, M.; Cahuzac, N.; Vire, B.; et al. Targeting the Wnt Pathway and Cancer Stem Cells with Anti-progastrin Humanized Antibodies as a Potential Treatment for K-RAS-Mutated Colorectal Cancer. Clin. Cancer Res. 2017, 23, 5267–5280. [Google Scholar] [CrossRef] [PubMed]
- Koh, T.J.; Chen, D. Gastrin as a growth factor in the gastrointestinal tract. Regul. Pept. 2000, 93, 37–44. [Google Scholar] [CrossRef]
- Xu, Q.; Krause, M.; Samoylenko, A.; Vainio, S. Wnt Signaling in Renal Cell Carcinoma. Cancers 2016, 8, 57. [Google Scholar] [CrossRef] [PubMed]
- Hollande, F.; Lee, D.J.; Choquet, A.; Roche, S.; Baldwin, G.S. Adherens junctions and tight junctions are regulated via different pathways by progastrin in epithelial cells. J. Cell Sci. 2003, 116, 1187–1197. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Owlia, A.; Varro, A.; Dai, B.; Rajaraman, S.; Wood, T. Gastrin gene expression is required for the proliferation and tumorigenicity of human colon cancer cells. Cancer Res. 1996, 56, 4111–4115. [Google Scholar] [PubMed]
- Wu, H.; Owlia, A.; Singh, P. Precursor peptide progastrin(1-80) reduces apoptosis of intestinal epithelial cells and upregulates cytochrome c oxidase Vb levels and synthesis of ATP. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 285, G1097–G1110. [Google Scholar] [CrossRef]
- Giraud, J.; Failla, L.M.; Pascussi, J.M.; Lagerqvist, E.L.; Ollier, J.; Finetti, P.; Bertucci, F.; Ya, C.; Gasmi, I.; Bourgaux, J.F.; et al. Autocrine Secretion of Progastrin Promotes the Survival and Self-Renewal of Colon Cancer Stem-like Cells. Cancer Res. 2016, 76, 3618–3628. [Google Scholar] [CrossRef] [PubMed]
- Najib, S.; Kowalski-Chauvel, A.; Do, C.; Roche, S.; Cohen-Jonathan-Moyal, E.; Seva, C. Progastrin a new pro-angiogenic factor in colorectal cancer. Oncogene 2015, 34, 3120–3130. [Google Scholar] [CrossRef]
- Du, M.; Giridhar, K.V.; Tian, Y.; Tschannen, M.R.; Zhu, J.; Huang, C.C.; Kilari, D.; Kohli, M.; Wang, L. Plasma exosomal miRNAs-based prognosis in metastatic kidney cancer. Oncotarget 2017, 8, 63703–63714. [Google Scholar] [CrossRef]
- Giridhar, K.V.; Sosa, C.P.; Hillman, D.W.; Sanhueza, C.; Dalpiaz, C.L.; Costello, B.A.; Quevedo, F.J.; Pitot, H.C.; Dronca, R.S.; Ertz, D.; et al. Whole Blood mRNA Expression-Based Prognosis of Metastatic Renal Cell Carcinoma. Int. J. Mol. Sci. 2017, 18, 2326. [Google Scholar] [CrossRef]
- White, M.C.; Holman, D.M.; Boehm, J.E.; Peipins, L.A.; Grossman, M.; Henley, S.J. Age and cancer risk: A potentially modifiable relationship. Am. J. Prev. Med. 2014, 46, S7–S15. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, N.; Obara, W.; Tatsugami, K.; Naito, S.; Kamba, T.; Takahashi, M.; Murai, S.; Abe, T.; Oba, K.; Naito, S. Prognosis of Japanese patients with previously untreated metastatic renal cell carcinoma in the era of molecular-targeted therapy. Cancer Sci. 2015, 106, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, Y.; Iguchi, T.; Tamada, S.; Yasuda, S.; Kato, M.; Ninomiya, N.; Yamasaki, T.; Nakatani, T. Oncological outcomes classified according to metastatic lesions in the era of molecular targeted drugs for metastatic renal cancer. Mol. Clin. Oncol. 2018, 8, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Sella, A.; Michaelson, M.D.; Matczak, E.; Simantov, R.; Lin, X.; Figlin, R.A. Heterogeneity of Patients With Intermediate-Prognosis Metastatic Renal Cell Carcinoma Treated With Sunitinib. Clin. Genitourin. Cancer 2017, 15, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Beuselinck, B.; Vano, Y.A.; Oudard, S.; Wolter, P.; De Smet, R.; Depoorter, L.; Teghom, C.; Karadimou, A.; Zucman-Rossi, J.; Debruyne, P.R.; et al. Prognostic impact of baseline serum C-reactive protein in patients with metastatic renal cell carcinoma (RCC) treated with sunitinib. BJU Int. 2014, 114, 81–89. [Google Scholar] [CrossRef]
- Teishima, J.; Inoue, S.; Hayashi, T.; Matsubara, A. Current status of prognostic factors in patients with metastatic renal cell carcinoma. Int. J. Urol. 2019, 26, 608–617. [Google Scholar] [CrossRef]
- de Velasco, G.; Culhane, A.C.; Fay, A.P.; Hakimi, A.A.; Voss, M.H.; Tannir, N.M.; Tamboli, P.; Appleman, L.J.; Bellmunt, J.; Kimryn Rathmell, W.; et al. Molecular Subtypes Improve Prognostic Value of International Metastatic Renal Cell Carcinoma Database Consortium Prognostic Model. Oncologist 2017, 22, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Escudier, B.; Porta, C.; Schmidinger, M.; Rioux-Leclercq, N.; Bex, A.; Khoo, V.; Grunwald, V.; Gillessen, S.; Horwich, A.; ESMO Guidelines Committee. Renal cell carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-updagger. Ann. Oncol. 2019, 30, 706–720. [Google Scholar] [CrossRef]
- Atashzar, M.R.; Baharlou, R.; Karami, J.; Abdollahi, H.; Rezaei, R.; Pourramezan, F.; Zoljalali Moghaddam, S.H. Cancer stem cells: A review from origin to therapeutic implications. J. Cell. Physiol. 2020, 235, 790–803. [Google Scholar] [CrossRef]
- Bussolati, B.; Dekel, B.; Azzarone, B.; Camussi, G. Human renal cancer stem cells. Cancer Lett. 2013, 338, 141–146. [Google Scholar] [CrossRef]
- Varna, M.; Gapihan, G.; Feugeas, J.P.; Ratajczak, P.; Tan, S.; Ferreira, I.; Leboeuf, C.; Setterblad, N.; Duval, A.; Verine, J.; et al. Stem cells increase in numbers in perinecrotic areas in human renal cancer. Clin. Cancer Res. 2015, 21, 916–924. [Google Scholar] [CrossRef] [PubMed]
| Cohorts | mRCC | Controls | ||
|---|---|---|---|---|
| 18–25 Years Old | 50–80 Years Old | |||
| N (%) | N (%) | N (%) | ||
| n = 143 | n = 137 | n =252 | ||
| Age, years | Median (range) | 63 (41–85) | 21 (18–25) | 55 (50–80) |
| Gender | Male | 107 (74.8%) | 79 (57.7%) | 99 (39.3%) |
| Female | 36 (25.2%) | 58 (42.3%) | 153 (60.7%) | |
| Histology | Clear cell | 110 | NA | |
| Chromophobe | 8 | |||
| Papillary | 14 | |||
| Other | 11 | |||
| Fuhrman Grade (G) | G1 | 3 | ||
| G2 | 45 | |||
| G3 | 45 | |||
| G4 | 32 | |||
| Not applicable | 0 | |||
| Unspecified | 18 | |||
| Sarcomatoid differentiation | Present | 16 | ||
| Absent | 78 | |||
| Unspecified | 49 | |||
| Necrosis | Present | 66 | ||
| Absent | 77 | |||
| IMDC prognostic score | Good | 28 | ||
| Intermediate | 81 | |||
| Poor | 26 | |||
| Unspecified | 8 | |||
| Clinical stage | I | 0 | ||
| II | 0 | |||
| III | 0 | |||
| IV | 143 | |||
| Unspecified | 0 | |||
| Total lines of systemic therapy | 0 | 24 | ||
| 1 | 55 | |||
| 2 | 30 | |||
| 3 | 19 | |||
| 4 | 8 | |||
| 5 or more | 7 | |||
| Characteristics | N | hPG80 | ||
|---|---|---|---|---|
| Median (IQR), pM | Mean (SE), pM | |||
| Patients | mRCC | 143 | 7.23 (3.24–20.61) | 24.50 (3.75) |
| Control cohorts | 18–25 years old | 137 | <LoD | <LoD |
| 50–80 years old | 252 | 1.5 * (0.0–3.09) | 3.82 (0.55) | |
| Characteristics | hPG80 | |||
|---|---|---|---|---|
| N (%) | Median (IQR), pM | Mean (SE), pM | ||
| IMDC prognostic score | Good | 28 (20.7%) | 4.72 (3.24–11.14) | 20.00 (7.18) |
| Intermediate | 81 (60.0%) | 7.49 (3.73–24.55) | 28.83 (5.79) | |
| Poor | 26 (19.3%) | 7.58 (2.58–19.05) | 19.13 (6.11) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kohli, M.; Tan, W.; Vire, B.; Liaud, P.; Blairvacq, M.; Berthier, F.; Rouison, D.; Garnier, G.; Payen, L.; Cousin, T.; et al. Prognostic Value of Plasma hPG80 (Circulating Progastrin) in Metastatic Renal Cell Carcinoma. Cancers 2021, 13, 375. https://doi.org/10.3390/cancers13030375
Kohli M, Tan W, Vire B, Liaud P, Blairvacq M, Berthier F, Rouison D, Garnier G, Payen L, Cousin T, et al. Prognostic Value of Plasma hPG80 (Circulating Progastrin) in Metastatic Renal Cell Carcinoma. Cancers. 2021; 13(3):375. https://doi.org/10.3390/cancers13030375
Chicago/Turabian StyleKohli, Manish, Winston Tan, Bérengère Vire, Pierre Liaud, Mélina Blairvacq, Frederic Berthier, Daniel Rouison, George Garnier, Léa Payen, Thierry Cousin, and et al. 2021. "Prognostic Value of Plasma hPG80 (Circulating Progastrin) in Metastatic Renal Cell Carcinoma" Cancers 13, no. 3: 375. https://doi.org/10.3390/cancers13030375
APA StyleKohli, M., Tan, W., Vire, B., Liaud, P., Blairvacq, M., Berthier, F., Rouison, D., Garnier, G., Payen, L., Cousin, T., Joubert, D., & Prieur, A. (2021). Prognostic Value of Plasma hPG80 (Circulating Progastrin) in Metastatic Renal Cell Carcinoma. Cancers, 13(3), 375. https://doi.org/10.3390/cancers13030375






