Feasibility, SAR Distribution, and Clinical Outcome upon Reirradiation and Deep Hyperthermia Using the Hypercollar3D in Head and Neck Cancer Patients
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Population
2.2. Radiotherapy Treatment
2.3. Hyperthermia Treatment
2.4. Collection of Patient and Follow-Up Data
2.5. Hyperthermia Treatment Parameters
2.6. Statistical Analysis
3. Results
3.1. General Characteristics of Patients, Treatment, and Follow-Up
3.2. Hyperthermia Treatment Feasibility
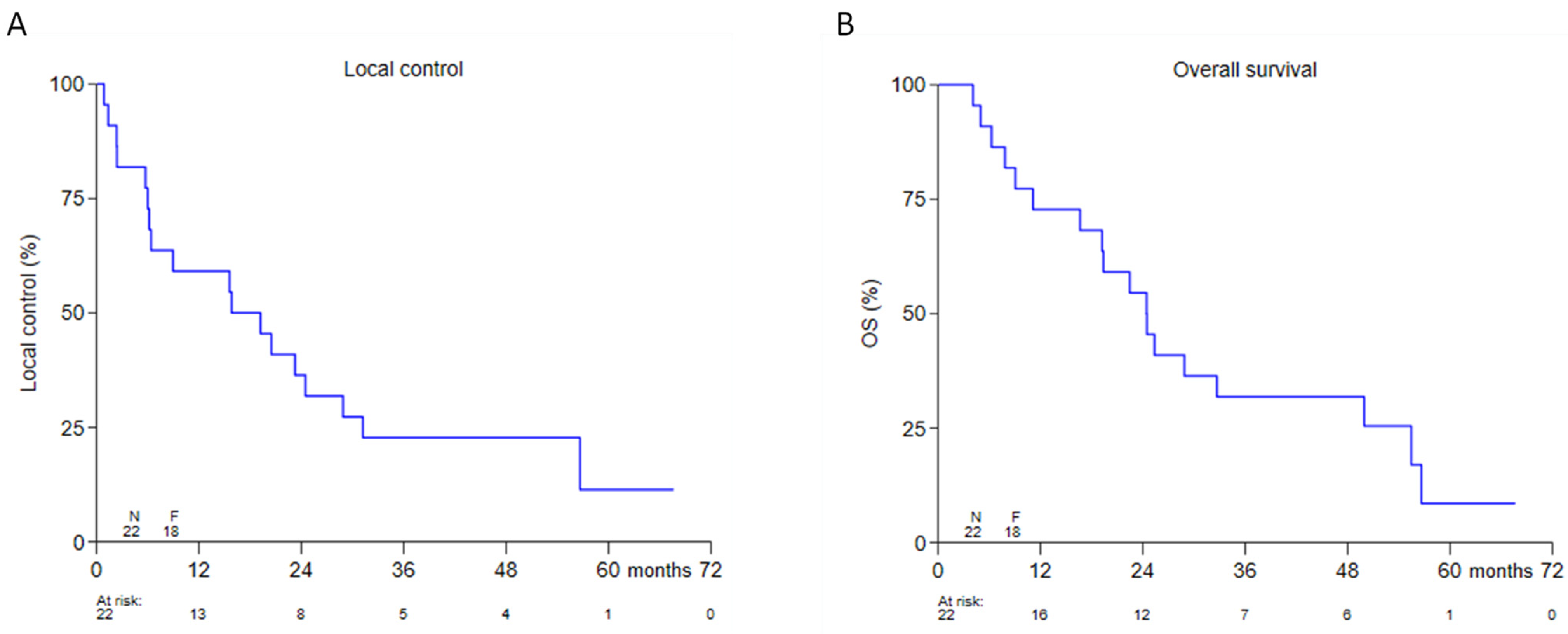
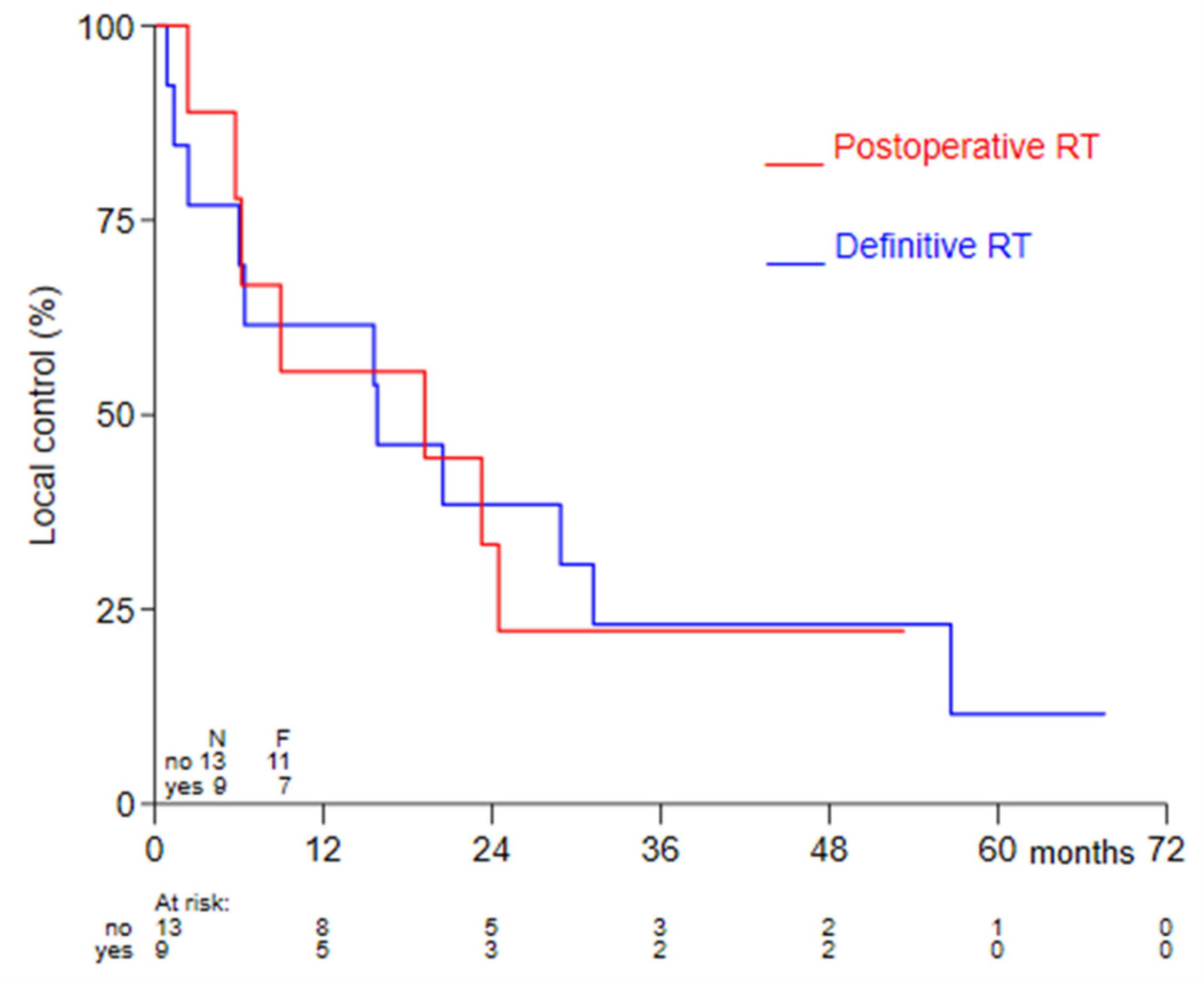
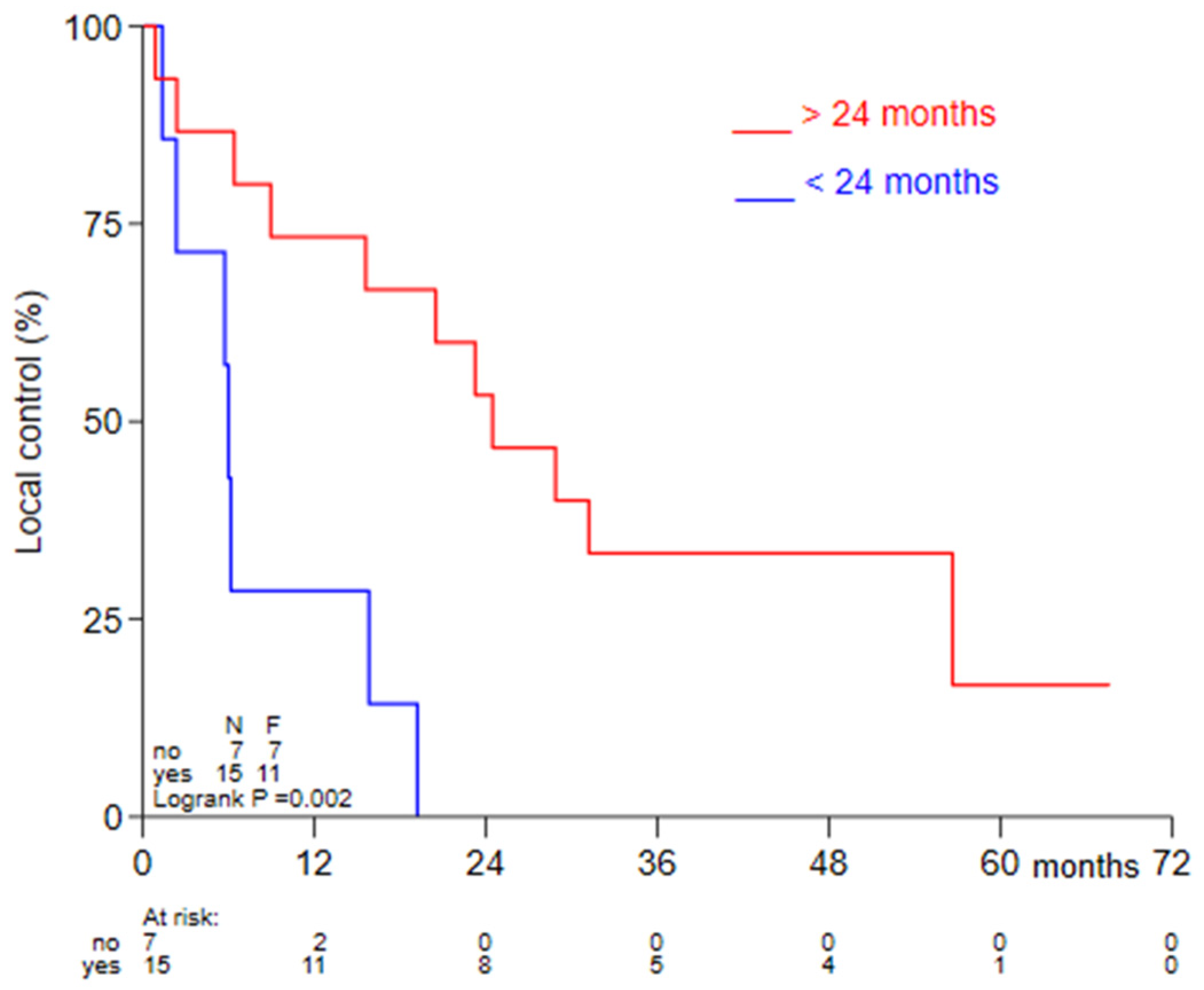
3.3. Oncological Outcome and Relation with Interval with Previous Radiotherapy
3.4. Toxicity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ang, K.K.; Zhang, Q.; Rosenthal, D.I.; Nguyen-Tan, P.F.; Sherman, E.J.; Weber, R.S.; Galvin, J.M.; Bonner, J.A.; Harris, J.; El-Naggar, A.K. Randomized phase III trial of concurrent accelerated radiation plus cisplatin with or without cetuximab for stage III to IV head and neck carcinoma: RTOG 0522. J. Clin. Oncol. 2014, 32, 2940–2950. [Google Scholar] [CrossRef]
- Adelstein, D.J.; Li, Y.; Adams, G.L.; Wagner, H., Jr.; Kish, J.A.; Ensley, J.F.; Schuller, D.E.; Forastiere, A.A. An intergroup phase III comparison of standard radiation therapy and two schedules of concurrent chemoradiotherapy in patients with unresectable squamous cell head and neck cancer. J. Clin. Oncol. 2003, 21, 92–98. [Google Scholar] [CrossRef] [Green Version]
- Forastiere, A.A.; Zhang, Q.; Weber, R.S.; Maor, M.H.; Goepfert, H.; Pajak, T.F.; Morrison, W.; Glisson, B.; Trotti, A.; Ridge, J.A.; et al. Long-term results of RTOG 91-11: A comparison of three nonsurgical treatment strategies to preserve the larynx in patients with locally advanced larynx cancer. J. Clin. Oncol. 2013, 31, 845–852. [Google Scholar] [CrossRef]
- Vargo, J.A.; Ward, M.C.; Caudell, J.J.; Riaz, N.; Dunlap, N.E.; Isrow, D.; Zakem, S.J.; Dault, J.; Awan, M.J.; Higgins, K.A.; et al. A Multi-institutional Comparison of SBRT and IMRT for Definitive Reirradiation of Recurrent or Second Primary Head and Neck Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2018, 100, 595–605. [Google Scholar] [CrossRef] [PubMed]
- Ward, M.C.; Riaz, N.; Caudell, J.J.; Dunlap, N.E.; Isrow, D.; Zakem, S.J.; Dault, J.; Awan, M.J.; Vargo, J.A.; Heron, D.E.; et al. Refining Patient Selection for Reirradiation of Head and Neck Squamous Carcinoma in the IMRT Era: A Multi-institution Cohort Study by the MIRI Collaborative. Int. J. Radiat. Oncol. Biol. Phys. 2018, 100, 586–594. [Google Scholar] [CrossRef]
- De Crevoisier, R.; Bourhis, J.; Domenge, C.; Wibault, P.; Koscielny, S.; Lusinchi, A.; Mamelle, G.; Janot, F.; Julieron, M.; Leridant, A.M.; et al. Full-dose reirradiation for unresectable head and neck carcinoma: Experience at the Gustave-Roussy Institute in a series of 169 patients. J. Clin. Oncol. 1998, 16, 3556–3562. [Google Scholar] [CrossRef]
- Spencer, S.A.; Harris, J.; Wheeler, R.H.; Machtay, M.; Schultz, C.; Spanos, W.; Rotman, M.; Meredith, R.; Ang, K.K. Final report of RTOG 9610, a multi-institutional trial of reirradiation and chemotherapy for unresectable recurrent squamous cell carcinoma of the head and neck. Head Neck 2008, 30, 281–288. [Google Scholar] [CrossRef]
- Sulman, E.P.; Schwartz, D.L.; Le, T.T.; Ang, K.K.; Morrison, W.H.; Rosenthal, D.I.; Ahamad, A.; Kies, M.; Glisson, B.; Weber, R.; et al. IMRT reirradiation of head and neck cancer-disease control and morbidity outcomes. Int. J. Radiat. Oncol. Biol. Phys. 2009, 73, 399–409. [Google Scholar] [CrossRef]
- Datta, N.R.; Rogers, S.; Ordonez, S.G.; Puric, E.; Bodis, S. Hyperthermia and radiotherapy in the management of head and neck cancers: A systematic review and meta-analysis. Int. J. Hyperth. 2016, 32, 31–40. [Google Scholar] [CrossRef]
- Verduijn, G.M.; de Wee, E.M.; Rijnen, Z.; Togni, P.; Hardillo, J.A.U.; Ten Hove, I.; Franckena, M.; van Rhoon, G.C.; Paulides, M.M. Deep hyperthermia with the HYPERcollar system combined with irradiation for advanced head and neck carcinoma—A feasibility study. Int. J. Hyperth. 2018, 34, 994–1001. [Google Scholar] [CrossRef]
- Zschaeck, S.; Weingartner, J.; Ghadjar, P.; Wust, P.; Mehrhof, F.; Kalinauskaite, G.; Ehrhardt, V.H.; Hartmann, V.; Tinhofer, I.; Heiland, M.; et al. Fever range whole body hyperthermia for re-irradiation of head and neck squamous cell carcinomas: Final results of a prospective study. Oral Oncol. 2021, 116, 105240. [Google Scholar] [CrossRef]
- Paulides, M.M.; Verduijn, G.M.; Van Holthe, N. Status quo and directions in deep head and neck hyperthermia. Radiat. Oncol. 2016, 11, 21. [Google Scholar] [CrossRef] [Green Version]
- Paulides, M.M.; Bakker, J.F.; Neufeld, E.; van der Zee, J.; Jansen, P.P.; Levendag, P.C.; van Rhoon, G.C. Winner of the "New Investigator Award" at the European Society of Hyperthermia Oncology Meeting 2007. The HYPERcollar: A novel applicator for hyperthermia in the head and neck. Int. J. Hyperth. 2007, 23, 567–576. [Google Scholar] [CrossRef]
- Verhaart, R.F.; Verduijn, G.M.; Fortunati, V.; Rijnen, Z.; van Walsum, T.; Veenland, J.F.; Paulides, M.M. Accurate 3D temperature dosimetry during hyperthermia therapy by combining invasive measurements and patient-specific simulations. Int. J. Hyperth. 2015, 31, 686–692. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rijnen, Z.; Bakker, J.F.; Canters, R.A.; Togni, P.; Verduijn, G.M.; Levendag, P.C.; Van Rhoon, G.C.; Paulides, M.M. Clinical integration of software tool VEDO for adaptive and quantitative application of phased array hyperthermia in the head and neck. Int. J. Hyperth. 2013, 29, 181–193. [Google Scholar] [CrossRef]
- Cappiello, G.; Drizdal, T.; Mc Ginley, B.; O’Halloran, M.; Glavin, M.; van Rhoon, G.C.; Jones, E.; Paulides, M.M. The potential of time-multiplexed steering in phased array microwave hyperthermia for head and neck cancer treatment. Phys. Med. Biol. 2018, 63, 135023. [Google Scholar] [CrossRef]
- Verhaart, R.F.; Fortunati, V.; Verduijn, G.M.; van Walsum, T.; Veenland, J.F.; Paulides, M.M. CT-based patient modeling for head and neck hyperthermia treatment planning: Manual versus automatic normal-tissue-segmentation. Radiother. Oncol. 2014, 111, 158–163. [Google Scholar] [CrossRef]
- Perez, C.A.; Pajak, T.; Emami, B.; Hornback, N.B.; Tupchong, L.; Rubin, P. Randomized phase III study comparing irradiation and hyperthermia with irradiation alone in superficial measurable tumors. Final report by the Radiation Therapy Oncology Group. Am. J. Clin. Oncol. 1991, 14, 133–141. [Google Scholar] [CrossRef]
- Datta, N.R.; Bose, A.K.; Kapoor, H.K.; Gupta, S. Head and neck cancers: Results of thermoradiotherapy versus radiotherapy. Int. J. Hyperth. 1990, 6, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Linthorst, M.; Baaijens, M.; Wiggenraad, R.; Creutzberg, C.; Ghidey, W.; van Rhoon, G.C.; van der Zee, J. Local control rate after the combination of re-irradiation and hyperthermia for irresectable recurrent breast cancer: Results in 248 patients. Radiother. Oncol. J. Eur. Soc. Ther. Radiol. Oncol. 2015, 117, 217–222. [Google Scholar] [CrossRef]
- Gabriele, P.; Ferrara, T.; Baiotto, B.; Garibaldi, E.; Marini, P.G.; Penduzzu, G.; Giovannini, V.; Bardati, F.; Guiot, C. Radio hyperthermia for re-treatment of superficial tumours. Int. J. Hyperth. 2009, 25, 189–198. [Google Scholar] [CrossRef]
- Janot, F.; de Raucourt, D.; Benhamou, E.; Ferron, C.; Dolivet, G.; Bensadoun, R.J.; Hamoir, M.; Gery, B.; Julieron, M.; Castaing, M.; et al. Randomized trial of postoperative reirradiation combined with chemotherapy after salvage surgery compared with salvage surgery alone in head and neck carcinoma. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2008, 26, 5518–5523. [Google Scholar] [CrossRef]
- Hurria, A.; Togawa, K.; Mohile, S.G.; Owusu, C.; Klepin, H.D.; Gross, C.P.; Lichtman, S.M.; Gajra, A.; Bhatia, S.; Katheria, V.; et al. Predicting chemotherapy toxicity in older adults with cancer: A prospective multicenter study. J. Clin. Oncol. 2011, 29, 3457–3465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pignon, J.P.; le Maitre, A.; Maillard, E.; Bourhis, J.; Mach-Nc Collaborative Group. Meta-analysis of chemotherapy in head and neck cancer (MACH-NC): An update on 93 randomised trials and 17,346 patients. Radiother. Oncol. 2009, 92, 4–14. [Google Scholar] [CrossRef]
- Szturz, P.; Vermorken, J.B. Treatment of Elderly Patients with Squamous Cell Carcinoma of the Head and Neck. Front. Oncol. 2016, 6, 199. [Google Scholar] [CrossRef] [Green Version]
- Machtay, M.; Moughan, J.; Trotti, A.; Garden, A.S.; Weber, R.S.; Cooper, J.S.; Forastiere, A.; Ang, K.K. Factors associated with severe late toxicity after concurrent chemoradiation for locally advanced head and neck cancer: An RTOG analysis. J. Clin. Oncol. 2008, 26, 3582–3589. [Google Scholar] [CrossRef]
- Lassen, P.; Eriksen, J.G.; Hamilton-Dutoit, S.; Tramm, T.; Alsner, J.; Overgaard, J. Effect of HPV-associated p16INK4A expression on response to radiotherapy and survival in squamous cell carcinoma of the head and neck. J. Clin. Oncol. 2009, 27, 1992–1998. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, K.; Hisamatsu, K.; Suzui, N.; Hara, A.; Tomita, H.; Miyazaki, T. A Review of HPV-Related Head and Neck Cancer. J. Clin. Med. 2018, 7, 241. [Google Scholar] [CrossRef] [Green Version]
- Chen, A.M.; Felix, C.; Wang, P.C.; Hsu, S.; Basehart, V.; Garst, J.; Beron, P.; Wong, D.; Rosove, M.H.; Rao, S.; et al. Reduced-dose radiotherapy for human papillomavirus-associated squamous-cell carcinoma of the oropharynx: A single-arm, phase 2 study. Lancet Oncol. 2017, 18, 803–811. [Google Scholar] [CrossRef] [Green Version]
- Huilgol, N.G.; Gupta, S.; Sridhar, C.R. Hyperthermia with radiation in the treatment of locally advanced head and neck cancer: A report of randomized trial. J. Cancer Res. Ther 2010, 6, 492–496. [Google Scholar] [CrossRef]
- Kroesen, M.; Mulder, H.T.; van Holthe, J.M.L.; Aangeenbrug, A.A.; Mens, J.W.M.; van Doorn, H.C.; Paulides, M.M.; Oomen-de Hoop, E.; Vernhout, R.M.; Lutgens, L.C.; et al. The Effect of the Time Interval Between Radiation and Hyperthermia on Clinical Outcome in 400 Locally Advanced Cervical Carcinoma Patients. Front. Oncol. 2019, 9, 134. [Google Scholar] [CrossRef] [PubMed]
- Kroesen, M.; Mulder, H.T.; van Holthe, J.M.L.; Aangeenbrug, A.A.; Mens, J.W.M.; van Doorn, H.C.; Paulides, M.M.; Oomen-de Hoop, E.; Vernhout, R.M.; Lutgens, L.C.; et al. Confirmation of thermal dose as a predictor of local control in cervical carcinoma patients treated with state-of-the-art radiation therapy and hyperthermia. Radiother. Oncol. 2019, 140, 150–158. [Google Scholar] [CrossRef]

| Characteristic | Categories | Value |
|---|---|---|
| Patient/tumor characteristics | ||
| Age (years) | Years (median) | 67.0 (IQR 59.5–71.5) |
| Sex | Male | 16 (73.0%) |
| Female | 6 (27.0%) | |
| Prior surgery (primary tumor) | Yes | 13 (59.0%) |
| No | 9 (41.0%) | |
| Prior systemic therapy (primary tumor) | Yes | 7 (32.0%) |
| No | 15 (68.0%) | |
| Recurrent/SP HNC | Recurrent tumor | 14 (64.0%) |
| Second primary tumor | 8 (36.0%) | |
| Tumor site (recurrent or SP tumor) | Nasopharynx | 2 (9.0%) |
| Oropharynx | 12 (55.0%) | |
| Oral cavity | 2 (9.0%) | |
| Salivary gland | 2 (9.0%) | |
| Hypopharynx | 1 (5.0%) | |
| Larynx | 3 (14.0%) | |
| Histology (recurrent or SP tumor) | Squamous cell carcinoma | 19 (86.0%) |
| Other | 3 (14.0%) | |
| Tumor stage (recurrent or SP tumor) | T0 | 8 (36.0%) |
| T1 | 1 (5.0%) | |
| T2 | 6 (27.0%) | |
| T3 | 2 (9.0%) | |
| T4 | 4 (18.0%) | |
| Unknown | 1 (5.0%) | |
| Nodal stage (recurrent or SP tumor) | N0 | 9 (41.0%) |
| N1 | 3 (14.0 %) | |
| N2 | 8 (36.0%) | |
| N3 | 1 (5.0%) | |
| Unknown | 1 (5.0%) | |
| Postoperative/definitivereirradiation + hyperthermia | Postoperative | 9 (41.0%) |
| Definitive | 13 (59.0%) | |
| Fractionation radiotherapy | 6 × 5.5 Gy | 7 (31.8%) |
| 10 × 2.0 Gy | 1 (4.5%) | |
| 25 × 2.0 Gy | 2 (9.0%) | |
| 28 × 1.8 Gy | 1 (4.5%) | |
| 30 × 2.0 Gy | 9 (40.9%) | |
| 33 × 1.8 Gy | 2 (9.0%) | |
| Technique radiotherapy | IMRT | 10 (44.5%) |
| VMAT | 5 (22.7%) | |
| Cyberknife | 7 (31.8%) | |
| Radiation field | Tumor | 10 (45.5%) |
| Neck | 7 (31.8%) | |
| Both | 5 (22,7%) | |
| Time from previous radiotherapy treatment | Months (median) | 51.5 (IQR 17.5–122.0) |
| Number of planned hyperthermia treatments | Number of treatments per patient | Number of patients |
| 3 | 7 (31.8%) | |
| 4 | 1 (4.5%) | |
| 5 | 2 (9.1%) | |
| 6 | 11 (50%) | |
| 7 | 1 (4.5%) | |
| Total number of all treatments | 108 | 22 (100%) |
| Complete clinical response 12 weeks post-treatment for definitive radiotherapy | Yes | 9 (81.8%) |
| No | 2 (18.2%) | |
| Characteristic | Categories | Value |
|---|---|---|
| Hyperthermia treatment characteristics | ||
| HT treatments | n | 107 |
| HTV volume | milliliters | 40.8 mL (2.8–108.9) |
| Treatment planning | ||
| TC25 | % | 90 (44–99) |
| TC50 | % | 58 (5–80) |
| TC75 | % | 12 (0–48) |
| THQ_1% | - | 1.28 (0.38–3.83) |
| Mean applied power * | Watts | All 1–22: 134.9 (49.9–353.0) |
| Pat 1–5: 278.8 (179.3–353.0) | ||
| Pat 6–22: 92.5 (49.9–123.1) | ||
| Mean estimated cf-SAR tumor * (applied power * predicted cf-SAR * efficiency) | W/kg | All 1–22: 104.2 (36.5–314.8) |
| Pat 1–5: 185.0 (69.4–314.8) | ||
| Pat 6–22: 80.5 (36.5–145.1) | ||
| Target temperature | n (%) | 5 (4.6) |
| Patient reference | A B C D E | |
| Maximum | °C | 38.3, 43.9, 40.9, 42.2, 38.0 |
| Median | °C | 37.8, 40.8, 40.5, 41.9, 37.2 |
| Minimum | °C | 36.5, 36.6, 35.8, 35.7, 34.3 |
| Maximum normal tissue temperature | n (%) | 56 (52.3) |
| Median | °C | 40.1 (35.0–42.8) |
| Toxicity | Baseline N = 22 | End RT N = 18 | 3–4 Months Post-Treatment N = 18 | 12 Months Sost-Treatment N = 10 | |
|---|---|---|---|---|---|
| Grade | Number (%) | Number (%) | Number (%) | Number (%) | |
| Xerostomia | 2 | 0 | 1 (6%) | 1 (6%) | 0 |
| 3 | 0 | 1 (6%) | 1 (6%) | 1 (10%) | |
| Altered taste | 2 | 0 | 2 (11%) | 1 (6%) | 0 |
| 3 | 0 | 0 | 0 | 0 | |
| Dysphagia | 2 | 6 (27%) | 6 (33%) | 5 (28%) | 5 (50%) |
| 3 | 3 (14%) | 5 (28%) | 3 (17%) | 2 (20%) | |
| Edema face | 2 | 0 | 2 (11%) | 0 | 1 (10%) |
| 3 | 0 | 0 | 0 | 0 | |
| Erythema skin | 2 | 0 | 0 | 0 | 0 |
| 3 | 0 | 0 | 0 | 0 | |
| Ulcus skin | 2 | 0 | 1 (6%) | 2 (11%) | 2 (20%) |
| 3 | 0 | 0 | 0 | 1 (10%) | |
| Trimus | 2 | 2 (9%) | 5 (28%) | 4 (22%) | 3 (30%) |
| 3 | 1 (6%) | 1 (10%) | |||
| Osteoradionecrosis | Yes | 0 | 0 | 3 (17%) | 1 (10%) |
| No | 22 (100%) | 18 (100%) | 15 (83%) | 9 (90%) | |
| Burn wound | Yes | 0 | 0 | 0 | 0 |
| No | 22 (100%) | 18 (100%) | 18 (100%) | 10 (100%) | |
| Vertigo | Yes | 0 | 0 | 0 | 0 |
| No | 22 (100%) | 18 (100%) | 18 (100%) | 10 (100%) | |
| Tube feeding | Yes | 2 (9%) | 6 (33%) | 3 (17%) | 2 (20%) |
| No | 20 (91%) | 12 (67%) | 15 (83%) | 8 (80%) | |
| Opioid use | Yes | 4 (18%) | 9 (50%) | 6 (33%) | 1 (10%) |
| No | 18 (82%) | 9 (50%) | 12 (66%) | 9 (90%) | |
| Other grade 3 or higher toxicity | Yes | 1 (5%) | 1 (6%) | 1 (6%) | 1 (10%) |
| No | 21 (95%) | 17 (94%) | 17 (94%) | 9 (90%) | |
| Tracheostoma | Yes | 4 (18%) | 4 (22%) | 1 (6%) | 1 (10%) |
| No | 18 (82%) | 13 (72%) | 17 (94%) | 9 (90%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kroesen, M.; van Holthe, N.; Sumser, K.; Chitu, D.; Vernhout, R.; Verduijn, G.; Franckena, M.; Hardillo, J.; van Rhoon, G.; Paulides, M. Feasibility, SAR Distribution, and Clinical Outcome upon Reirradiation and Deep Hyperthermia Using the Hypercollar3D in Head and Neck Cancer Patients. Cancers 2021, 13, 6149. https://doi.org/10.3390/cancers13236149
Kroesen M, van Holthe N, Sumser K, Chitu D, Vernhout R, Verduijn G, Franckena M, Hardillo J, van Rhoon G, Paulides M. Feasibility, SAR Distribution, and Clinical Outcome upon Reirradiation and Deep Hyperthermia Using the Hypercollar3D in Head and Neck Cancer Patients. Cancers. 2021; 13(23):6149. https://doi.org/10.3390/cancers13236149
Chicago/Turabian StyleKroesen, Michiel, Netteke van Holthe, Kemal Sumser, Dana Chitu, Rene Vernhout, Gerda Verduijn, Martine Franckena, Jose Hardillo, Gerard van Rhoon, and Margarethus Paulides. 2021. "Feasibility, SAR Distribution, and Clinical Outcome upon Reirradiation and Deep Hyperthermia Using the Hypercollar3D in Head and Neck Cancer Patients" Cancers 13, no. 23: 6149. https://doi.org/10.3390/cancers13236149
APA StyleKroesen, M., van Holthe, N., Sumser, K., Chitu, D., Vernhout, R., Verduijn, G., Franckena, M., Hardillo, J., van Rhoon, G., & Paulides, M. (2021). Feasibility, SAR Distribution, and Clinical Outcome upon Reirradiation and Deep Hyperthermia Using the Hypercollar3D in Head and Neck Cancer Patients. Cancers, 13(23), 6149. https://doi.org/10.3390/cancers13236149










