Progesterone Receptor Membrane Component (PGRMC)1 and PGRMC2 and Their Roles in Ovarian and Endometrial Cancer
Abstract
Simple Summary
Abstract
1. Introduction
2. PGRMC1 and PGRMC2 in Ovarian and Endometrial Cancer
2.1. PGRMC1 and PGRMC2 as Mediators of P4′s Actions
2.2. Overview of the Morbidity and Mortality of Ovarian and Endometrial Cancers
3. Expression and Subcellular Localization of PGRMC1 and PGRMC2 in Neoplastic Cells of the Ovary and Endometrium
3.1. PGRMC1 and PGRMC2 Subcellular Localizations
3.2. Cellular Localization and Expression Levels within Neoplastic Ovary
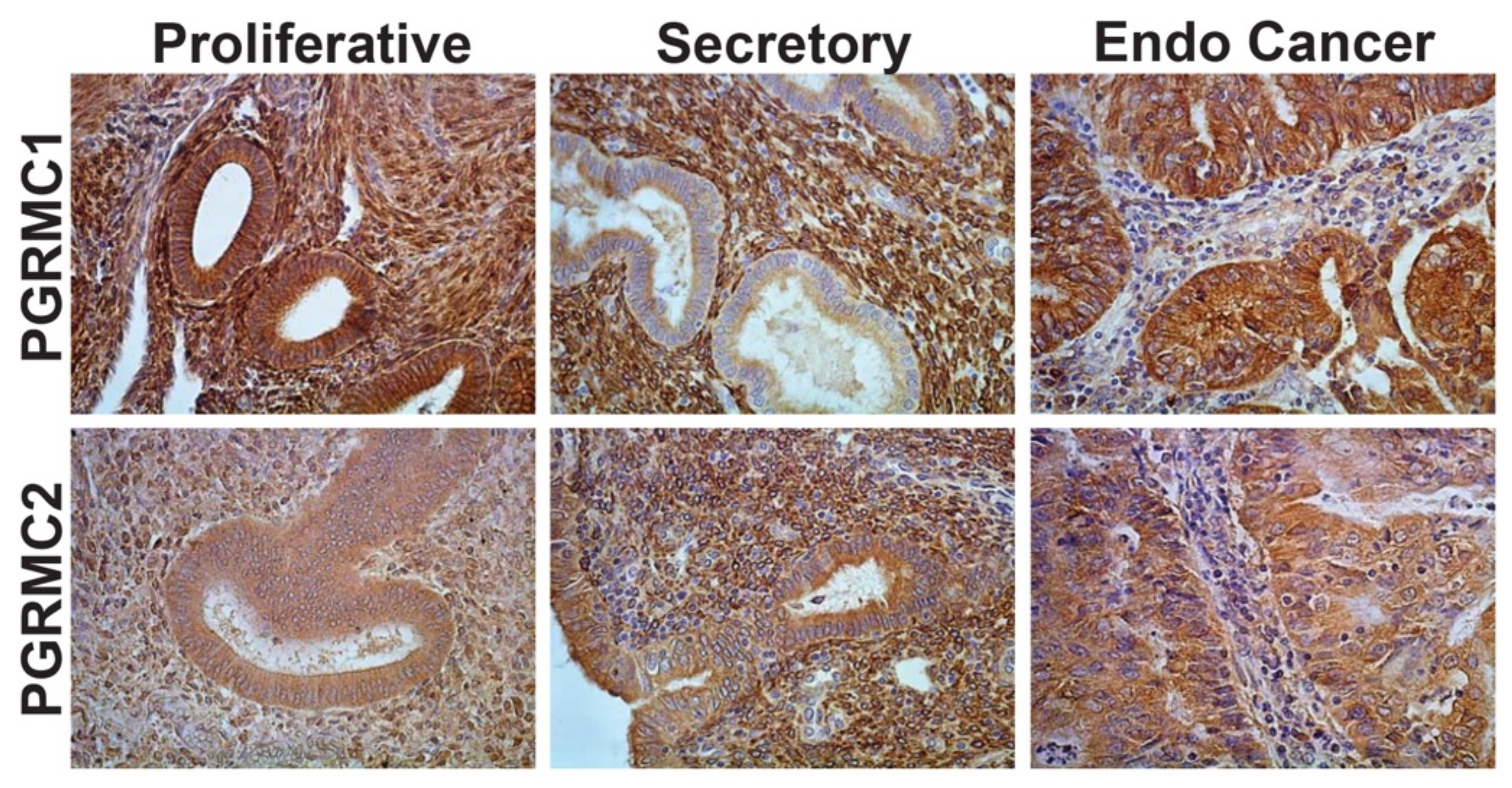
3.3. Cellular Localization and Expression Levels within Neoplastic Endometrium
4. Regulation of PGRMC1 and PGRMC2 Expression in Ovarian and Endometrial Cancers
4.1. Hormonal Regulation of PGRMC1 and PGRMC2
4.2. Environmental Regulators of PGRMC1 and PGRMC2
5. PGRMC1 and PGRMC2 as Regulators of Tumor Growth
5.1. In Vivo Studies Using Human Ovarian and Endometrial Cancer Xenografts
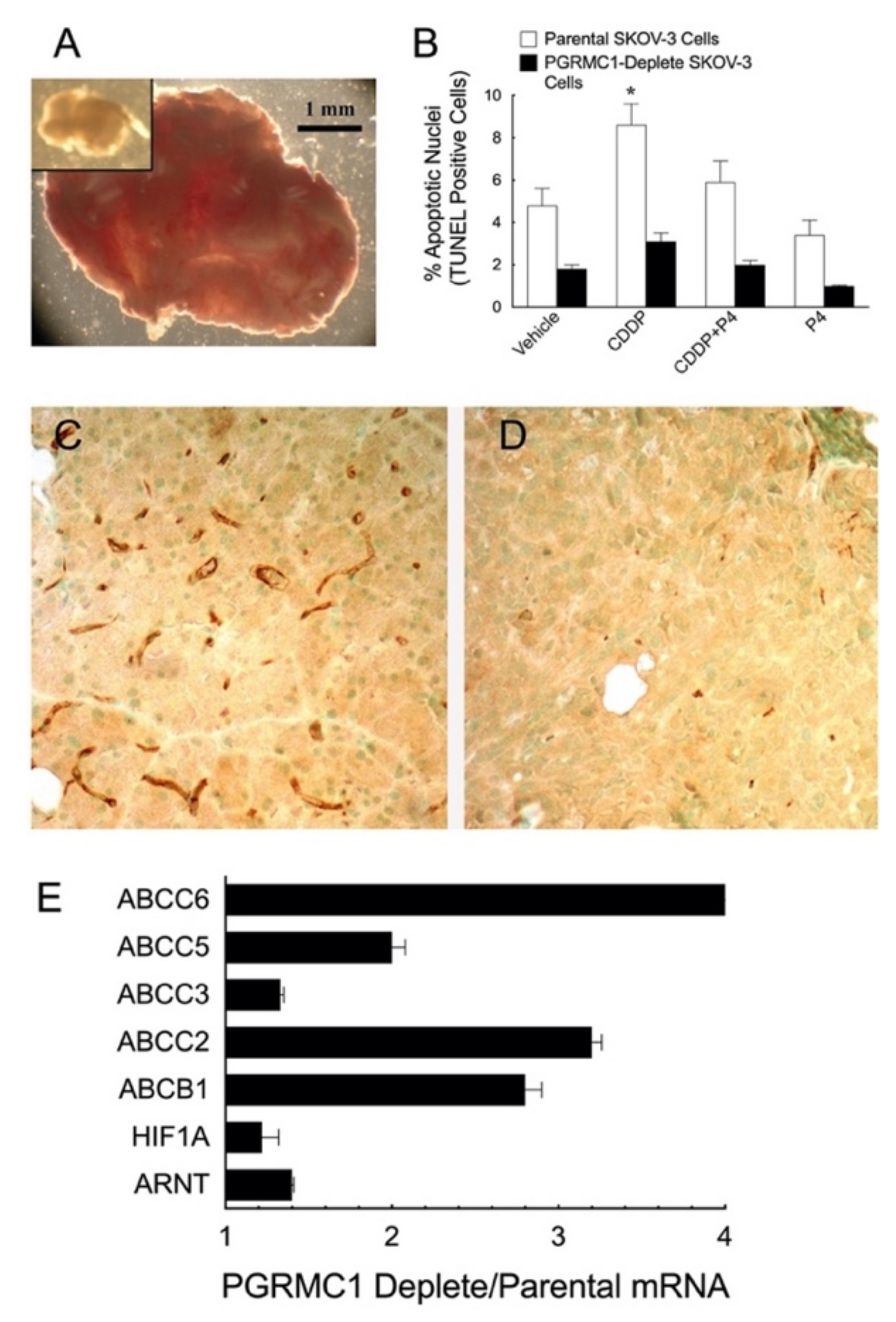
5.1.1. Ovarian Cancer Xenografts
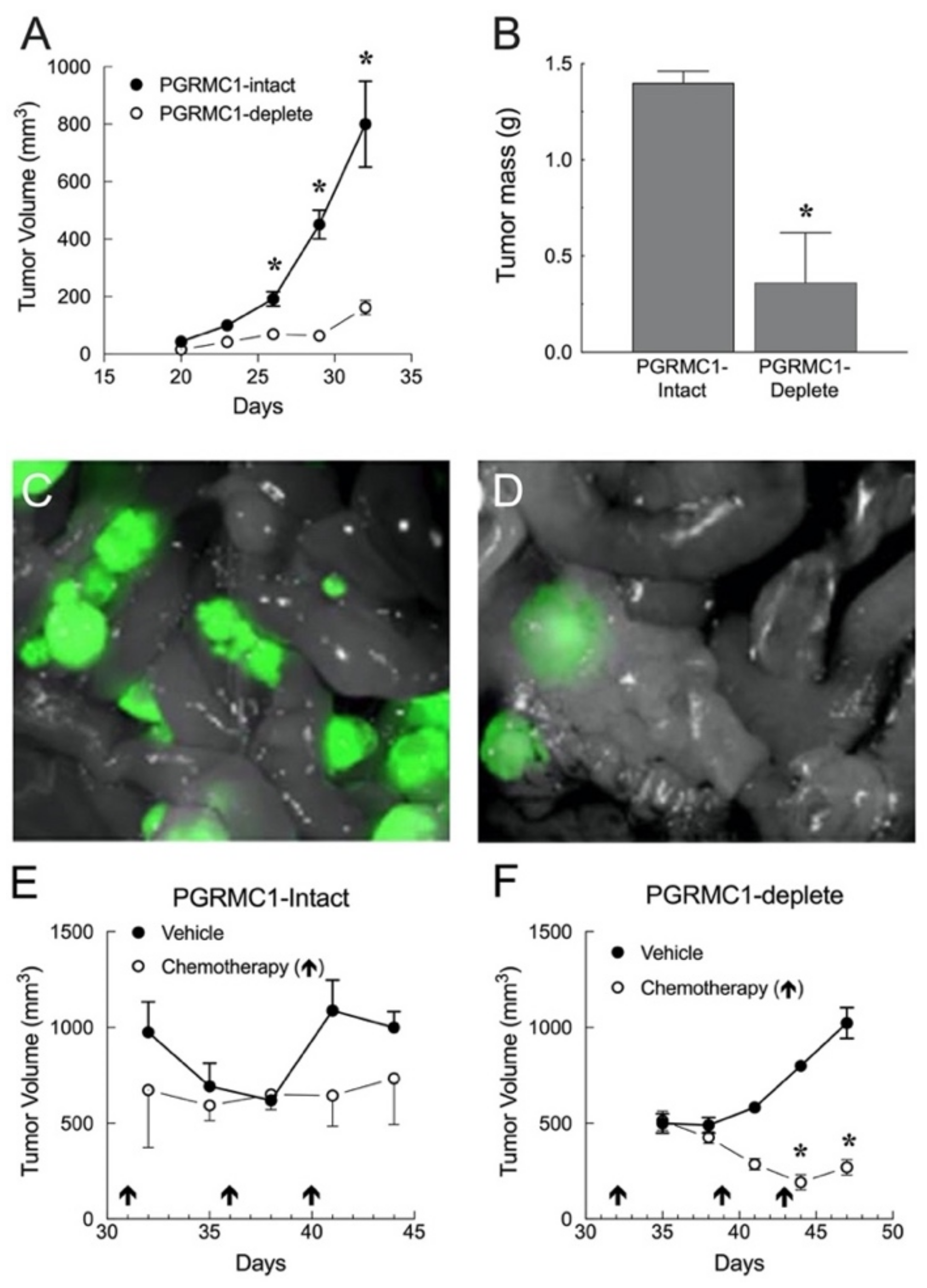
5.1.2. Endometrial Cancer Xenografts
6. Biological Functions of PGRMC1 and PGRMC2
6.1. PGRMC1 as a Regulator of Mitosis and Apoptosis
6.2. PGRMC1 and Chemoresistance
6.2.1. PGRMCs and Preserving Responsiveness to Cisplatin
6.2.2. PGRMCs and Cancer Stem Cells
6.2.3. PGRMCs and their Role in Cell Migration and Metastasis
7. PGRMCs and the Regulation of RNA and Protein Synthesis
7.1. PGRMC1 and its Nuclear Site of Action
7.2. PGRMCs and Ribosomal Protein Interaction
8. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Shih, I.-M.; Wang, Y.; Wang, T.-L. The Origin of Ovarian Cancer Species and Precancerous Landscape. Am. J. Pathol. 2020, 191, 26–39. [Google Scholar] [CrossRef] [PubMed]
- Cahill, M.A. Progesterone receptor membrane component 1: An integrative review. J. Steroid Biochem. Mol. Biol. 2007, 105, 16–36. [Google Scholar] [CrossRef] [PubMed]
- Friel, A.; Zhang, L.; Pru, C.; Clark, N.; McCallum, M.; Blok, L.; Shioda, T.; Peluso, J.; Rueda, B.; Pru, K.; et al. Progesterone receptor membrane component 1 deficiency attenuates growth while promoting chemosensitivity of human endometrial xenograft tumors. Cancer Lett. 2015, 356, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Peluso, J.J.; Liu, X.; Saunders, M.M.; Claffey, K.P.; Phoenix, K. Regulation of Ovarian Cancer Cell Viability and Sensitivity to Cisplatin by Progesterone Receptor Membrane Component-1. J. Clin. Endocrinol. Metab. 2008, 93, 1592–1599. [Google Scholar] [CrossRef]
- Ponikwicka-Tyszko, D.; Chrusciel, M.; Stelmaszewska, J.; Bernaczyk, P.; Chrusciel, P.; Sztachelska, M.; Scheinin, M.; Bidzinski, M.; Szamatowicz, J.; Huhtaniemi, I.T.; et al. Molecular mechanisms underlying mifepristone’s agonistic action on ovarian cancer progression. EBioMedicine 2019, 47, 170–183. [Google Scholar] [CrossRef]
- Meyer, C.; Schmid, R.; Scriba, P.C.; Wehling, M. Purification and Partial Sequencing of High-Affinity Progesterone-Binding Site(s) from Porcine Liver Membranes. JBIC J. Biol. Inorg. Chem. 1996, 239, 726–731. [Google Scholar] [CrossRef]
- Clark, N.C.; Pru, C.A.; Yee, S.-P.; Lydon, J.P.; Peluso, J.J.; Pru, J.K. Conditional Ablation of Progesterone Receptor Membrane Component 2 Causes Female Premature Reproductive Senescence. Endocrinology 2016, 158, 640–651. [Google Scholar] [CrossRef]
- McCallum, M.L.; Pru, C.A.; Niikura, Y.; Yee, S.-P.; Lydon, J.P.; Peluso, J.J.; Pru, J.K. Conditional Ablation of Progesterone Receptor Membrane Component 1 Results in Subfertility in the Female and Development of Endometrial Cysts. Endocrinology 2016, 157, 3309–3319. [Google Scholar] [CrossRef] [PubMed]
- Kaluka, D.; Batabyal, D.; Chiang, B.-Y.; Poulos, T.L.; Yeh, S.-R. Spectroscopic and Mutagenesis Studies of Human PGRMC1. Biochemistry 2015, 54, 1638–1647. [Google Scholar] [CrossRef] [PubMed]
- Cahill, M.A. The evolutionary appearance of signaling motifs in PGRMC1. Biosci. Trends 2017, 11, 179–192. [Google Scholar] [CrossRef][Green Version]
- Hehenberger, E.; Eitel, M.; Fortunato, S.A.; Miller, D.J.; Keeling, P.J.; Cahill, M.A. Early eukaryotic origins and metazoan elaboration of MAPR family proteins. Mol. Phylogenet. Evol. 2020, 148, 106814. [Google Scholar] [CrossRef] [PubMed]
- Thornton, J.W. Evolution of vertebrate steroid receptors from an ancestral estrogen receptor by ligand exploitation and serial genome expansions. Proc. Natl. Acad. Sci. USA 2001, 98, 5671–5676. [Google Scholar] [CrossRef] [PubMed]
- Thornton, J.W.; Need, E.; Crews, D. Resurrecting the Ancestral Steroid Receptor: Ancient Origin of Estrogen Signaling. Science 2003, 301, 1714–1717. [Google Scholar] [CrossRef] [PubMed]
- Reid, B.M.; Permuth, J.B.; Sellers, T.A. Epidemiology of ovarian cancer: A review. Cancer Biol. Med. 2017, 14, 9–32. [Google Scholar] [PubMed]
- Willibald, M.; Bayer, G.; Stahlhut, V.; Poschmann, G.; Stühler, K.; Gierke, B.; Pawlak, M.; Seeger, H.; Mueck, A.O.; Niederacher, D.; et al. Progesterone receptor membrane component 1 is phosphorylated upon progestin treatment in breast cancer cells. Oncotarget 2017, 8, 72480–72493. [Google Scholar] [CrossRef]
- Ruan, X.; Zhang, Y.; Mueck, A.O.; Willibald, M.; Seeger, H.; Fehm, T.; Brucker, S.; Neubauer, H. Increased expression of progesterone receptor membrane component 1 is associated with aggressive phenotype and poor prognosis in ER-positive and negative breast cancer. Menopause 2017, 24, 203–209. [Google Scholar] [CrossRef]
- Schneck, H.; Ruan, X.; Seeger, H.; Cahill, M.A.; Fehm, T.; Mueck, A.O.; Neubauer, H. Membrane-receptor initiated proliferative effects of dienogest in human breast cancer cells. Gynecol. Endocrinol. 2012, 29, 160–162. [Google Scholar] [CrossRef]
- Stewart, C.; Ralyea, C.; Lockwood, S. Ovarian Cancer: An Integrated Review. Semin. Oncol. Nurs. 2019, 35, 151–156. [Google Scholar] [CrossRef]
- Keyvani, V.; Farshchian, M.; Esmaeili, S.-A.; Yari, H.; Moghbeli, M.; Nezhad, S.-R.K.; Abbaszadegan, M.R. Ovarian cancer stem cells and targeted therapy. J. Ovarian Res. 2019, 12, 120. [Google Scholar] [CrossRef]
- La Vecchia, C. Ovarian cancer: Epidemiology and risk factors. Eur. J. Cancer Prev. 2017, 26, 55–62. [Google Scholar] [CrossRef]
- Webb, P.M.; Jordan, S.J. Epidemiology of epithelial ovarian cancer. Best Pract. Res. Clin. Obstet. Gynaecol. 2017, 41, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Trabert, B.; Tworoger, S.S.; O’Brien, K.M.; Townsend, M.K.; Fortner, R.T.; Iversen, E.S.; Hartge, P.; White, E.; Amiano, P.; Arslan, A.; et al. The Risk of Ovarian Cancer Increases with an Increase in the Lifetime Number of Ovulatory Cycles: An Analysis from the Ovarian Cancer Cohort Consortium (OC3). Cancer Res. 2020, 80, 1210–1218. [Google Scholar] [CrossRef]
- Adani, G.; Filippini, T.; Wise, L.A.; Halldorsson, T.I.; Blaha, L.; Vinceti, M. Dietary Intake of Acrylamide and Risk of Breast, En-do-metrial, and Ovarian Cancers: A Systematic Review and Dose-Response Meta-analysis. Cancer Epidemiol. Biomark. Prev. 2020, 29, 1095–1106. [Google Scholar] [CrossRef]
- Anastasi, E.; Filardi, T.; Tartaglione, S.; Lenzi, A.; Angeloni, A.; Morano, S. Linking type 2 diabetes and gynecological cancer: An introductory overview. Clin. Chem. Lab. Med. 2018, 56, 1413–1425. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Goding Sauer, A.; Fedewa, S.A.; Butterly, L.F.; Anderson, J.C.; Cercek, A.; Smith, R.A.; Jemal, A. Colorectal cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 145–164. [Google Scholar] [CrossRef]
- Levine, D.A. Integrated genomic characterization of endometrial carcinoma. Nature 2013, 497, 67–73. [Google Scholar] [CrossRef]
- Gadducci, A.; Cosio, S.; Genazzani, A.R. Tissue and serum biomarkers as prognostic variables in endometrioid-type endometrial cancer. Crit. Rev. Oncol. 2011, 80, 181–192. [Google Scholar] [CrossRef]
- Lee, I.I.; Kim, J.J. Influence of AKT on progesterone action in endometrial diseases. Biol. Reprod. 2014, 91, 63. [Google Scholar] [CrossRef] [PubMed]
- McConechy, M.K.; Ding, J.; Cheang, M.C.U.; Wiegand, K.C.; Senz, J.; A Tone, A.; Yang, W.; Prentice, L.M.; Tse, K.; Zeng, T.; et al. Use of mutation profiles to refine the classification of endometrial carcinomas. J. Pathol. 2012, 228, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Adambekov, S.; Yi, Y.; Fabio, A.; Miljkovic, I.; Edwards, R.P.; Lopa, S.; Linkov, F. Metabolic Syndrome in Endometrial Cancer Patients: Systematic Review. Metab. Syndr. Relat. Disord. 2019, 17, 241–249. [Google Scholar] [CrossRef]
- Constantine, G.D.; Kessler, G.; Graham, S.; Goldstein, S.R. Increased Incidence of Endometrial Cancer Following the Women’s Health Initiative: An Assessment of Risk Factors. J. Womens Heal. 2019, 28, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Mackintosh, M.L.; Crosbie, E.J. Obesity-driven endometrial cancer: Is weight loss the answer? BJOG 2013, 120, 791–794. [Google Scholar] [CrossRef]
- Meczekalski, B.; Perez-Roncero, G.R.; Lopez-Baena, M.T.; Chedraui, P.; Perez-Lopez, F.R. The polycystic ovary syndrome and gy-necological cancer risk. Gynecol. Endocrinol. 2020, 36, 289–293. [Google Scholar] [CrossRef]
- Mortimer, J.; Urban, J.H. Long-term toxicities of selective estrogen-receptor modulators and antiaromatase agents. Oncology 2003, 17, 652–659. [Google Scholar]
- Navaratnarajah, R.; Pillay, O.; Hardiman, P. Polycystic Ovary Syndrome and Endometrial Cancer. Semin. Reprod. Med. 2008, 26, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Peluso, J.J.; Lodde, V.; Liu, X. Progesterone Regulation of Progesterone Receptor Membrane Component 1 (PGRMC1) Sumoylation and Transcriptional Activity in Spontaneously Immortalized Granulosa Cells. Endocrinology 2012, 153, 3929–3939. [Google Scholar] [CrossRef]
- Sueldo, C.; Liu, X.; Peluso, J.J. Progestin and AdipoQ Receptor 7, Progesterone Membrane Receptor Component 1 (PGRMC1), and PGRMC2 and Their Role in Regulating Progesterone’s Ability to Suppress Human Granulosa/Luteal Cells from Entering into the Cell Cycle. Biol. Reprod. 2015, 93, 63. [Google Scholar] [CrossRef] [PubMed]
- Cahill, M.A.; Jazayeri, J.A.; Catalano, S.M.; Toyokuni, S.; Kovacevic, Z.; Richardson, D.R. The emerging role of progesterone receptor membrane component 1 (PGRMC1) in cancer biology. Biochim. Biophys. Acta (BBA)-Bioenerg. 2016, 1866, 339–349. [Google Scholar] [CrossRef]
- Cahill, M.A.; Jazayeri, J.A.; Kovacevic, Z.; Richardson, D.R. PGRMC1 regulation by phosphorylation: Potential new insights in controlling biological activity. Oncotarget 2016, 7, 50822–50827. [Google Scholar] [CrossRef]
- Papatheodorou, I.; A Fonseca, N.; Keays, M.; Tang, A.; Barrera, E.; Bazant, W.; Burke, M.; Füllgrabe, A.; Fuentes, A.M.-P.; George, N.; et al. Expression Atlas: Gene and protein expression across multiple studies and organisms. Nucleic Acids Res. 2017, 46, D246–D251. [Google Scholar] [CrossRef]
- Hampton, K.K.; Stewart, R.; Napier, D.; Claudio, P.P.; Craven, R.J. PGRMC1 Elevation in Multiple Cancers and Essential Role in Stem Cell Survival. Adv. Lung Cancer 2015, 04, 37–51. [Google Scholar] [CrossRef]
- Oliveira, D.V.N.P.; Prahm, K.P.; Christensen, I.J.; Hansen, A.; Høgdall, C.K.; Høgdall, E.V. Gene expression profile association with poor prognosis in epithelial ovarian cancer patients. Sci. Rep. 2021, 11, 5438. [Google Scholar] [CrossRef]
- Hatano, Y.; Hatano, K.; Tamada, M.; Morishige, K.-I.; Tomita, H.; Yanai, H.; Hara, A. A Comprehensive Review of Ovarian Serous Carcinoma. Adv. Anat. Pathol. 2019, 26, 329–339. [Google Scholar] [CrossRef]
- Peluso, J.J.; Gawkowska, A.; Liu, X.; Shioda, T.; Pru, J.K. Progesterone Receptor Membrane Component-1 Regulates the Development and Cisplatin Sensitivity of Human Ovarian Tumors in Athymic Nude Mice. Endocrinology 2009, 150, 4846–4854. [Google Scholar] [CrossRef]
- Modugno, F.; Laskey, R.; Smith, A.L.; Andersen, C.L.; Haluska, P.; Oesterreich, S. Hormone response in ovarian cancer: Time to reconsider as a clinical target? Endocr. Relat. Cancer 2012, 19, R255–R279. [Google Scholar] [CrossRef]
- Peluso, J.J.; Romak, J.; Liu, X. Progesterone Receptor Membrane Component-1 (PGRMC1) Is the Mediator of Progesterone’s Antiapoptotic Action in Spontaneously Immortalized Granulosa Cells As Revealed by PGRMC1 Small Interfering Ribonucleic Acid Treatment and Functional Analysis of PGRMC1 Mutations. Endocrinology 2007, 149, 534–543. [Google Scholar] [CrossRef] [PubMed]
- Thieffry, C.; Van Wynendaele, M.; Aynaci, A.; Maja, M.; Dupuis, C.; Loriot, A.; Marbaix, E.; Henriet, P. AG-205 Upregulates Enzymes Involved in Cholesterol Biosynthesis and Steroidogenesis in Human Endometrial Cells Independently of PGRMC1 and Re-lated MAPR Proteins. Biomolecules 2021, 11, 1472. [Google Scholar] [CrossRef]
- Wang-Eckhardt, L.; Eckhardt, M. A progesterone receptor membrane component 1 antagonist induces large vesicles in-de-pendent of progesterone receptor membrane component 1 expression. Biol. Chem. 2020, 401, 1093–1099. [Google Scholar] [CrossRef] [PubMed]
- Kao, L.C.; Tulac, S.; Lobo, S.; Imani, B.; Yang, J.P.; Germeyer, A.; Osteen, K.; Taylor, R.N.; Lessey, B.A.; Giudice, L.C. Global gene profiling in human endometrium during the window of implantation. Endocrinology 2002, 143, 2119–2138. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.I.-C.; Hannan, N.; Mak, Y.; Nicholls, P.K.; Zhang, J.; Rainczuk, A.; Stanton, P.; Robertson, D.M.; Salamonsen, L.; Stephens, A.N. Proteomic Characterization of Midproliferative and Midsecretory Human Endometrium. J. Proteome Res. 2009, 8, 2032–2044. [Google Scholar] [CrossRef] [PubMed]
- Peluso, J.J.; Pappalardo, A.; Losel, R.; Wehling, M. Progesterone membrane receptor component 1 expression in the immature rat ovary and its role in mediating progesterone’s antiapoptotic action. Endocrinology 2006, 147, 3133–3140. [Google Scholar] [CrossRef][Green Version]
- Xiong, S.; Mhawech-Fauceglia, P.; Tsao-Wei, D.; Roman, L.; Gaur, R.K.; Epstein, A.L.; Pinski, J. Expression of the luteinizing hormone receptor (LHR) in ovarian cancer. BMC Cancer 2019, 19, 1114. [Google Scholar] [CrossRef]
- Richards, J.S. From Follicular Development and Ovulation to Ovarian Cancers: An Unexpected Journey. Vitam. Horm 2018, 107, 453–472. [Google Scholar] [CrossRef] [PubMed]
- Krebs, C.J.; Jarvis, E.D.; Chan, J.; Lydon, J.P.; Ogawa, S.; Pfaff, D.W. A membrane-associated progesterone-binding protein, 25-Dx, is regulated by progesterone in brain regions involved in female reproductive behaviors. Proc. Natl. Acad. Sci. USA 2000, 97, 12816–12821. [Google Scholar] [CrossRef] [PubMed]
- Lösel, R.M.; Besong, D.; Peluso, J.J.; Wehling, M. Progesterone receptor membrane component 1—Many tasks for a versatile protein. Steroids 2008, 73, 929–934. [Google Scholar] [CrossRef]
- Stepanova, M.; Lin, F.; Lin, V.C. In silico modelling of hormone response elements. BMC Bioinform. 2006, 7 (Suppl. 4). [Google Scholar] [CrossRef]
- Zhang, L.; Kanda, Y.; Roberts, D.J.; Ecker, J.L.; Losel, R.; Wehling, M.; Peluso, J.J.; Pru, J.K. Expression of progesterone receptor membrane component 1 and its partner serpine 1 mRNA binding protein in uterine and placental tissues of the mouse and human. Mol. Cell. Endocrinol. 2008, 287, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Keator, C.S.; Mah, K.; Slayden, O.D. Alterations in progesterone receptor membrane component 2 (PGRMC2) in the endometrium of macaques afflicted with advanced endometriosis. Mol. Hum. Reprod. 2012, 18, 308–319. [Google Scholar] [CrossRef]
- Kleine, W.; Maier, T.; Geyer, H.; Pfleiderer, A. Estrogen and progesterone receptors in endometrial cancer and their prognostic relevance. Gynecol. Oncol. 1990, 38, 59–65. [Google Scholar] [CrossRef]
- Panda, H.; Chuang, T.-D.; Luo, X.; Chegini, N. Endometrial miR-181a and miR-98 expression is altered during transition from normal into cancerous state and target PGR, PGRMC1, CYP19A1, DDX3X, and TIMP3. J. Clin. Endocrinol. Metab. 2012, 97, E1316–E1326. [Google Scholar] [CrossRef]
- Ali Syeda, Z.; Langden, S.S.S.; Munkhzul, C.; Lee, M.; Song, S.J. Regulatory Mechanism of MicroRNA Expression in Cancer. Int. J. Mol. Sci. 2020, 21, 1723. [Google Scholar] [CrossRef]
- Wendler, A.; Keller, D.; Albrecht, C.; Peluso, J.J.; Wehling, M. Involvement of let-7/miR-98 microRNAs in the regulation of pro-gesterone receptor membrane component 1 expression in ovarian cancer cells. Oncol. Rep. 2011, 25, 273–279. [Google Scholar] [PubMed]
- Chirshev, E.; Oberg, K.C.; Ioffe, Y.J.; Unternaehrer, J.J. Let-7as biomarker, prognostic indicator, and therapy for precision medicine in cancer. Clin. Transl. Med. 2019, 8, 24. [Google Scholar] [CrossRef]
- Liu, N.; Zhou, C.; Zhao, J.; Chen, Y. Reversal of Paclitaxel Resistance in Epithelial Ovarian Carcinoma Cells by a MUC1 Aptamer-let-7i Chimera. Cancer Investig. 2012, 30, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Thammaiah, C.K.; Jayaram, S. Role of let-7 family microRNA in breast cancer. Non-Coding RNA Res. 2016, 1, 77–82. [Google Scholar] [CrossRef]
- Cavallini, A.; Lippolis, C.; Vacca, M.; Nardelli, C.; Castegna, A.; Arnesano, F.; Carella, N.; DePalo, R. The Effects of Chronic Lifelong Activation of the AHR Pathway by Industrial Chemical Pollutants on Female Human Reproduction. PLoS ONE 2016, 11, e0152181. [Google Scholar] [CrossRef] [PubMed]
- Selmin, O.; Lucier, G.W.; Clark, G.C.; Tritscher, A.M.; Heuvel, J.P.; Gastel, J.A.; Walker, N.; R-Sutter, T.; Bell, D. Isolation and characterization of a novel gene induced by 2, 3, 7, 8-tetrachlorodibenzo-p-dioxin in rat liver. Carcinogenesis 1996, 17, 2609–2615. [Google Scholar] [CrossRef]
- Huang, Q.; Chen, Q. Mediating Roles of PPARs in the Effects of Environmental Chemicals on Sex Steroids. PPAR Res 2017, 2017, 3203161. [Google Scholar] [CrossRef]
- Zhang, G.Y.; Ahmed, N.; Riley, C.; Oliva, K.; Barker, G.; Quinn, M.A.; Rice, G.E. Enhanced expression of peroxisome prolif-era-tor-activated receptor gamma in epithelial ovarian carcinoma. Br. J. Cancer 2005, 92, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Tong, B.J.; Tan, J.; Tajeda, L.; Das, S.K.; Chapman, J.A.; DuBois, R.N.; Dey, S.K. Heightened expression of cyclooxygenase-2 and perox-isome proliferator-activated receptor-delta in human endometrial adenocarcinoma. Neoplasia 2000, 2, 483–490. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Furuhata, R.; Kabe, Y.; Kanai, A.; Sugiura, Y.; Tsugawa, H.; Sugiyama, E.; Hirai, M.; Yamamoto, T.; Koike, I.; Yoshikawa, N.; et al. Progesterone receptor membrane associated component 1 enhances obesity progression in mice by fa-cili-tating lipid accumulation in adipocytes. Commun. Biol. 2020, 3, 479. [Google Scholar] [CrossRef] [PubMed]
- Magnotti, E.; Marasco, W.A. The latest animal models of ovarian cancer for novel drug discovery. Expert. Opin. Drug Discov. 2018, 13, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Materna, V.; Stege, A.; Surowiak, P.; Priebsch, A.; Lage, H. RNA interference-triggered reversal of ABCC2-dependent cisplatin resistance in human cancer cells. Biochem. Biophys. Res. Commun. 2006, 348, 153–157. [Google Scholar] [CrossRef]
- Sabbir, M.G. Progesterone induced Warburg effect in HEK293 cells is associated with post-translational modifications and proteasomal degradation of progesterone receptor membrane component 1. J. Steroid Biochem. Mol. Biol. 2019, 191, 105376. [Google Scholar] [CrossRef] [PubMed]
- Damia, G.; Broggini, M. Platinum Resistance in Ovarian Cancer: Role of DNA Repair. Cancers 2019, 11, 119. [Google Scholar] [CrossRef]
- Lodde, V.; Peluso, J.J. A novel role for progesterone and progesterone receptor membrane component 1 in regulating spindle microtubule stability during rat and human ovarian cell mitosis. Biol. Reprod. 2011, 84, 715–722. [Google Scholar] [CrossRef]
- Peluso, J.J.; Griffin, D.; Liu, X.; Horne, M. Progesterone receptor membrane component-1 (PGRMC1) and PGRMC-2 interact to suppress entry into the cell cycle in spontaneously immortalized rat granulosa cells. Biol. Reprod. 2014, 91, 104. [Google Scholar] [CrossRef]
- Griffin, D.; Liu, X.; Pru, C.; Pru, J.K.; Peluso, J.J. Expression of Progesterone Receptor Membrane Component-2 Within the Immature Rat Ovary and Its Role in Regulating Mitosis and Apoptosis of Spontaneously Immortalized Granulosa Cells1. Biol. Reprod. 2014, 91, 36. [Google Scholar] [CrossRef]
- Peluso, J.J.; Pru, C.A.; Liu, X.; Kelp, N.C.; Pru, J.K. Progesterone receptor membrane component 1 and 2 regulate granulosa cell mitosis and survival through a NFKappaB-dependent mechanism. Biol. Reprod. 2019, 100, 1571–1580. [Google Scholar] [CrossRef]
- Thomas, P.; Pang, Y.; Dong, J. Enhancement of cell surface expression and receptor functions of membrane progestin receptor alpha (mPRalpha) by progesterone receptor membrane component 1 (PGRMC1): Evidence for a role of PGRMC1 as an adaptor protein for steroid receptors. Endocrinology 2014, 155, 1107–1119. [Google Scholar] [CrossRef] [PubMed]
- Mir, S.U.R.; Jin, L.; Craven, R.J. Neutrophil Gelatinase-associated Lipocalin (NGAL) Expression Is Dependent on the Tumor-associated Sigma-2 Receptor S2RPgrmc1. J. Biol. Chem. 2012, 287, 14494–14501. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Kim, S.Y.; Choi, H.S.; Kim, M.K.; Lee, H.M.; Jang, Y.J.; Ryu, C.J. Progesterone Receptor Membrane Component 1 suppresses the p53 and Wnt/beta-catenin pathways to promote human pluripotent stem cell self-renewal. Sci. Rep. 2018, 8, 3048. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, I.S.; Rohe, H.J.; Twist, K.E.; Craven, R.J. Pgrmc1 (Progesterone Receptor Membrane Component 1) Associates with Epidermal Growth Factor Receptor and Regulates Erlotinib Sensitivity. J. Biol. Chem. 2010, 285, 24775–24782. [Google Scholar] [CrossRef]
- Brasseur, K.; Gevry, N.; Asselin, E. Chemoresistance and targeted therapies in ovarian and endometrial cancers. Oncotarget 2017, 8, 4008–4042. [Google Scholar] [CrossRef]
- Deng, J.; Wang, L.; Chen, H.; Hao, J.; Ni, J.; Chang, L.; Duan, W.; Graham, P.; Li, Y. Targeting epithelial-mesenchymal transition and cancer stem cells for chemoresistant ovarian cancer. Oncotarget 2016, 7, 55771–55788. [Google Scholar] [CrossRef]
- Klemba, A.; Bodnar, L.; Was, H.; Brodaczewska, K.K.; Wcislo, G.; Szczylik, C.A.; Kieda, C. Hypoxia-Mediated Decrease of Ovarian Cancer Cells Reaction to Treatment: Significance for Chemo- and Immunotherapies. Int. J. Mol. Sci. 2020, 21, 9492. [Google Scholar] [CrossRef] [PubMed]
- Reijnen, C.; ENITEC-consortium; Van Weelden, W.J.; Arts, M.S.J.P.; Peters, J.P.; Rijken, P.F.; Van De Vijver, K.; Santacana, M.; Bronsert, P.; Bulten, J.; et al. Poor outcome in hypoxic endometrial carcinoma is related to vascular density. Br. J. Cancer 2019, 120, 1037–1044. [Google Scholar] [CrossRef]
- Muz, B.; de la Puente, P.; Azab, F.; Azab, A.K. The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia 2015, 3, 83–92. [Google Scholar] [CrossRef]
- Li, S.-S.; Ma, J.; Wong, A.S.T. Chemoresistance in ovarian cancer: Exploiting cancer stem cell metabolism. J. Gynecol. Oncol. 2018, 29, e32. [Google Scholar] [CrossRef]
- Giannone, G.; Attademo, L.; Scotto, G.; Genta, S.; Ghisoni, E.; Tuninetti, V.; Aglietta, M.; Pignata, S.; Valabrega, G. Endometrial Cancer Stem Cells: Role, Characterization and Therapeutic Implications. Cancers 2019, 11, 1820. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kim, S.Y.; Choi, H.S.; An, S.; Ryu, C.J. Epitope mapping of anti-PGRMC1 antibodies reveals the non-conventional mem-brane topology of PGRMC1 on the cell surface. Sci. Rep. 2019, 9, 653. [Google Scholar] [CrossRef] [PubMed]
- Peluso, J.J.; Liu, X.; Uliasz, T.; Pru, C.A.; Kelp, N.C.; Pru, J.K. PGRMC1/2 promotes luteal vascularization and maintains the primordial follicles of mice. Reproduction 2018, 156, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; He, M.; Cui, L.; Gao, M.; Zhang, M.; Yue, F.; Shi, T.; Yang, X.; Pan, Y.; Zheng, X.; et al. Chemotherapy exacerbates ovarian cancer cell migration and cancer stem cell-like characteristics through GLI1. Br. J. Cancer 2020, 122, 1638–1648. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, X.; Yuan, M.; Xian, S.; Zhang, L.; Yang, D.; Cheng, Y. Promotion of ovarian cancer cell invasion, migration and colony formation by the miR21/Wnt/CD44v6 pathway. Oncol. Rep. 2019, 42, 91–102. [Google Scholar]
- Albrecht, C.; Huck, V.; Wehling, M.; Wendler, A. In vitro inhibition of SKOV-3 cell migration as a distinctive feature of proges-terone receptor membrane component type 2 versus type 1. Steroids 2012, 77, 1543–1550. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Hidalgo, N.R.; Ramirez, P.T.; Ngo, B.; Perez-Hoyos, S.; Coreas, N.; Sanchez-Iglesias, J.L.; Cabrera, S.; Franco, S.; Benavente, A.P.; Gil-Moreno, A. Oncologic impact of micrometastases or isolated tumor cells in sentinel lymph nodes of patients with en-dometrial cancer: A meta-analysis. Clin. Transl. Oncol. 2020, 22, 1272–1279. [Google Scholar] [CrossRef] [PubMed]
- Hirai, Y.; Utsugi, K.; Takeshima, N.; Kawamata, Y.; Furuta, R.; Kitagawa, T.; Kawaguchi, T.; Hasumi, K.; Noda, S.T. Putative gene loci associated with carcinogenesis and metastasis of endocervical adenocarcinomas of uterus determined by conventional and array-based CGH. Am. J. Obstet. Gynecol. 2004, 191, 1173–1182. [Google Scholar] [CrossRef]
- Gao, Y.; Dai, M.; Liu, H.; He, W.; Lin, S.; Yuan, T.; Chen, H.; Dai, S. Diagnostic value of circulating miR-21: An update meta-analysis in various cancers and validation in endometrial cancer. Oncotarget 2016, 7, 68894–68908. [Google Scholar] [CrossRef]
- Kabe, Y.; Nakane, T.; Koike, I.; Yamamoto, T.; Sugiura, Y.; Harada, E.; Sugase, K.; Shimamura, T.; Ohmura, M.; Muraoka, K.; et al. Haem-dependent dimerization of PGRMC1/Sigma-2 receptor facilitates cancer proliferation and chemo-resistance. Nat. Commun. 2016, 7, 11030. [Google Scholar] [CrossRef] [PubMed]
- Peluso, J.J.; Liu, X.; Gawkowska, A.; Lodde, V.; Wu, C.A. Progesterone inhibits apoptosis in part by PGRMC1-regulated gene ex-pression. Mol. Cell. Endocrinol. 2010, 320, 153–161. [Google Scholar] [CrossRef]
- Peluso, J.J.; DeCerbo, J.; Lodde, V. Evidence for a genomic mechanism of action for progesterone receptor membrane com-po-nent-1. Steroids 2012, 77, 1007–1012. [Google Scholar] [CrossRef] [PubMed]
- Deforges, J.; Locker, N.; Sargueil, B. mRNAs that specifically interact with eukaryotic ribosomal subunits. Biochimie 2015, 114, 48–57. [Google Scholar] [CrossRef]
- Korobeinikova, A.V.; Garber, M.B.; Gongadze, G.M. Ribosomal proteins: Structure, function, and evolution. Biochemistry (Moscow) 2012, 77, 562–574. [Google Scholar] [CrossRef] [PubMed]
- Sauert, M.; Temmel, H.; Moll, I. Heterogeneity of the translational machinery: Variations on a common theme. Biochimie 2015, 114, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Mauro, V.P.; Edelman, G.M. The ribosome filter hypothesis. Proc. Natl. Acad. Sci. USA 2002, 99, 12031–12036. [Google Scholar] [CrossRef]
- Gry, M.; Rimini, R.; Strömberg, S.; Asplund, A.; Pontén, F.; Uhlén, M.; Nilsson, P. Correlations between RNA and protein expression profiles in 23 human cell lines. BMC Genom. 2009, 10, 365. [Google Scholar] [CrossRef] [PubMed]
- Maier, T.; Guell, M.; Serrano, L. Correlation of mRNA and protein in complex biological samples. FEBS Lett. 2009, 583, 3966–3973. [Google Scholar] [CrossRef]

| PGRMC1 | PGRMC2 | |
|---|---|---|
| Protein Name | % Coverage | % Coverage |
| ribosomal protein S4 | 14 | 10 |
| ribosomal protein S26 | 31 | ND |
| ribosomal protein S27 | 24 | 38 |
| ribosomal protein L3 | 25 | ND |
| ribosomal protein L4 | 17 | 28 |
| ribosomal protein L12 | 18 | 32 |
| ribosomal protein L27 | ND | 40 |
| ribosomal protein L30 | 30 | 70 |
| ribosomal protein L34 | 24 | 21 |
| ribosomal protein L38 | ND | 50 |
| PGRMC1 | PGRMC1-∆PO4 | |
|---|---|---|
| Protein Name | % Coverage | % Coverage |
| ribosomal protein S4 | 54 | 47 |
| ribosomal protein S26 | ND | 44 |
| ribosomal protein S27 | ND | 39 |
| ribosomal protein L3 | ND | 45 |
| ribosomal protein L4 | 46 | 51 |
| ribosomal protein L12 | ND | 53 |
| ribosomal protein L30 | 70 | 70 |
| ribosomal protein L34 | 38 | ND |
| Ribosomal Protein Interaction with PGRMC1 (% coverage) | |||
|---|---|---|---|
| RPS25 (30%) | RPL22 (30%) | RPS12 (54%) | RPL4 (24%) |
| RPS4X (40%) | RPS10 (15%) | RPS18 (20%) | RPS20 (19%) |
| RPS14 (32%) | RPL7A (28%) | RPS19 (51%) | RPSA (23%) |
| RPS17 (56%) | RPL5 (22%) | RPS23A (41%) | RPS3 (19%) |
| RPS15A (18%) | RPL18 (49%) | RPS13 (35%) | RPS11 (42%) |
| RPL13A (11%) | RPL38 (47%) | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peluso, J.J.; Pru, J.K. Progesterone Receptor Membrane Component (PGRMC)1 and PGRMC2 and Their Roles in Ovarian and Endometrial Cancer. Cancers 2021, 13, 5953. https://doi.org/10.3390/cancers13235953
Peluso JJ, Pru JK. Progesterone Receptor Membrane Component (PGRMC)1 and PGRMC2 and Their Roles in Ovarian and Endometrial Cancer. Cancers. 2021; 13(23):5953. https://doi.org/10.3390/cancers13235953
Chicago/Turabian StylePeluso, John J., and James K. Pru. 2021. "Progesterone Receptor Membrane Component (PGRMC)1 and PGRMC2 and Their Roles in Ovarian and Endometrial Cancer" Cancers 13, no. 23: 5953. https://doi.org/10.3390/cancers13235953
APA StylePeluso, J. J., & Pru, J. K. (2021). Progesterone Receptor Membrane Component (PGRMC)1 and PGRMC2 and Their Roles in Ovarian and Endometrial Cancer. Cancers, 13(23), 5953. https://doi.org/10.3390/cancers13235953






