In Vivo Targeting of CXCR4—New Horizons
Abstract
:Simple Summary
Abstract
1. Introduction
2. CXCR4-Targeted Theranostics in Cancer and Its Limitations
2.1. CXCR4-Targeted PET Imaging
2.2. CXCR4-Targeted Radioligand Therapy
3. Beyond Cancer: CXCR4-Targeted Imaging of Immune Cell Infiltrates
3.1. Cardiovascular Diseases
3.2. Inflammation Imaging
3.3. Infection Imaging
4. Imaging of CXCR4-Positive Immune Cells in Preclinical Models
5. CXCR4-Targeted Imaging Agents: An Ever-Evolving Field
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Alluri, S.R.; Higashi, Y.; Kil, K.E. PET Imaging Radiotracers of Chemokine Receptors. Molecules 2021, 26, 5174. [Google Scholar] [CrossRef]
- Buck, A.K.; Stolzenburg, A.; Hanscheid, H.; Schirbel, A.; Luckerath, K.; Schottelius, M.; Wester, H.J.; Lapa, C. Chemokine Receptor—Directed Imaging and Therapy. Methods 2017, 130, 63–71. [Google Scholar] [CrossRef]
- Fakhari, A.; Aghanejad, A.; Jalilian, A.R.; Gharepapagh, E. Recent Developments in Targeted Imaging of CXCR4-Chemokine Receptor. J. Radioanal. Nucl. Chem. 2018, 317, 1–14. [Google Scholar] [CrossRef]
- Kircher, M.; Lapa, C. Infection and Inflammation Imaging: Beyond FDG. PET Clin. 2020, 15, 215–229. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.; Mayer, I.A.; Walenkamp, A.M.E.; Lapa, C.; Andreeff, M.; Bobirca, A. At the Bedside: Profiling and Treating Patients with CXCR4-Expressing Cancers. J. Leukoc. Biol. 2021, 109, 953–967. [Google Scholar] [CrossRef] [PubMed]
- Walenkamp, A.M.E.; Lapa, C.; Herrmann, K.; Wester, H.J. CXCR4 Ligands: The Next Big Hit? J. Nucl. Med. 2017, 58, 77s–82s. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuyumcu, S.; Isik, E.G.; Tiryaki, T.O.; Has-Simsek, D.; Sanli, Y.; Buyukkaya, F.; Ozkan, Z.G.; Kalayoglu-Besisik, S.; Unal, S.N. Prognostic Significance of (68)Ga-Pentixafor PET/CT in Multiple Myeloma Recurrence: A Comparison to (18)F-FDG PET/CT and Laboratory Results. Ann. Nucl. Med. 2021, 35, 1147–1156. [Google Scholar] [CrossRef]
- Lapa, C.; Schreder, M.; Schirbel, A.; Samnick, S.; Kortum, K.M.; Herrmann, K.; Kropf, S.; Einsele, H.; Buck, A.K.; Wester, H.J.; et al. [(68)Ga]Pentixafor-PET/CT for Imaging of Chemokine Receptor CXCR4 Expression in Multiple Myeloma—Comparison to [(18)F]FDG and Laboratory Values. Theranostics 2017, 7, 205–212. [Google Scholar] [CrossRef]
- Pan, Q.; Cao, X.; Luo, Y.; Li, J.; Feng, J.; Li, F. Chemokine Receptor-4 targeted PET/CT with (68)Ga-Pentixafor in Assessment of Newly Diagnosed Multiple Myeloma: Comparison to (18)F-FDG PET/CT. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 537–546. [Google Scholar] [CrossRef]
- Lapa, C.; Schirbel, A.; Samnick, S.; Luckerath, K.; Kortum, K.M.; Knop, S.; Wester, H.J.; Buck, A.K.; Schreder, M. The Gross Picture: Intraindividual Tumour Heterogeneity in a Patient with Nonsecretory Multiple Myeloma. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1097–1098. [Google Scholar] [CrossRef]
- Lapa, C.; Luckerath, K.; Kircher, S.; Hanscheid, H.; Grigoleit, G.U.; Rosenwald, A.; Stolzenburg, A.; Kropf, S.; Einsele, H.; Wester, H.J.; et al. Potential Influence of Concomitant Chemotherapy on CXCR4 Expression in Receptor Directed Endoradiotherapy. Br. J. Haematol. 2019, 184, 440–443. [Google Scholar] [CrossRef] [Green Version]
- Herhaus, P.; Habringer, S.; Philipp-Abbrederis, K.; Vag, T.; Gerngross, C.; Schottelius, M.; Slotta-Huspenina, J.; Steiger, K.; Altmann, T.; Weisser, T.; et al. Targeted Positron Emission Tomography Imaging of CXCR4 Expression in Patients with Acute Myeloid Leukemia. Haematologica 2016, 101, 932–940. [Google Scholar] [CrossRef] [PubMed]
- Mayerhoefer, M.E.; Jaeger, U.; Staber, P.; Raderer, M.; Wadsak, W.; Pfaff, S.; Kornauth, C.; Senn, D.; Weber, M.; Wester, H.J.; et al. [68Ga]Ga-Pentixafor PET/MRI for CXCR4 Imaging of Chronic Lymphocytic Leukemia: Preliminary Results. Invest Radiol. 2018, 53, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Duell, J.; Krummenast, F.; Schirbel, A.; Klassen, P.; Samnick, S.; Rauert-Wunderlich, H.; Rasche, L.; Buck, A.K.; Wester, H.J.; Rosenwald, A.; et al. Improved Primary Staging of Marginal-Zone Lymphoma by Addition of CXCR4-Directed PET/CT. J. Nucl. Med. 2021, 62, 1415–1421. [Google Scholar] [CrossRef]
- Herhaus, P.; Habringer, S.; Vag, T.; Steiger, K.; Slotta-Huspenina, J.; Gerngross, C.; Wiestler, B.; Wester, H.J.; Schwaiger, M.; Keller, U. Response Assessment with the CXCR4-Directed Positron Emission Tomography Tracer [(68)Ga]Pentixafor in a Patient with Extranodal Marginal Zone Lymphoma of the Orbital Cavities. EJNMMI Res. 2017, 7, 51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mayerhoefer, M.; Raderer, M.; Lamm, W.; Weber, M.; Kiesewetter, B.; Rohrbeck, J.; Simonitsch-Klupp, I.; Hacker, M.; Leisser, A.; Nics, L.; et al. CXCR4 PET/MRI for Follow-Up of Gastric Mucosa-Associated Lymphoid Tissue Lymphoma after First-Line H. Pylori Eradication. Blood 2021. [Google Scholar] [CrossRef] [PubMed]
- Mayerhoefer, M.E.; Raderer, M.; Lamm, W.; Pichler, V.; Pfaff, S.; Weber, M.; Kiesewetter, B.; Hacker, M.; Kazianka, L.; Staber, P.B.; et al. CXCR4 PET Imaging of Mantle Cell Lymphoma Using [(68)Ga]Pentixafor: Comparison with [(18)F]FDG-PET. Theranostics 2021, 11, 567–578. [Google Scholar] [CrossRef]
- Kraus, S.; Dierks, A.; Rasche, L.; Kertels, O.; Kircher, M.; Schirbel, A.; Zovko, J.; Steinbrunn, T.; Tibes, R.; Wester, H.J.; et al. (68)Ga-Pentixafor-PET/CT Imaging Represents a Novel Approach to Detect Chemokine Receptor CXCR4 Expression in Myeloproliferative Neoplasms. J. Nucl. Med. 2021. [Google Scholar] [CrossRef]
- Herhaus, P.; Lipkova, J.; Lammer, F.; Yakushev, I.; Vag, T.; Slotta-Huspenina, J.; Habringer, S.; Lapa, C.; Pukrop, T.; Hellwig, D.; et al. CXCR4-Targeted PET Imaging of Central Nervous System B-Cell Lymphoma. J. Nucl. Med. 2020, 61, 1765–1771. [Google Scholar] [CrossRef]
- Luo, Y.; Cao, X.; Pan, Q.; Li, J.; Feng, J.; Li, F. (68)Ga-pentixafor PET/CT for Imaging of Chemokine Receptor-4 Expression in Waldenstrom Macroglobulinemia/Lymphoplasmacytic Lymphoma: Comparison to (18)F-FDG PET/CT. J. Nucl. Med. 2019, 60, 1724–1729. [Google Scholar] [CrossRef]
- Luo, Y.; Pan, Q.; Feng, J.; Cao, X.; Li, F. Chemokine Receptor CXCR4-Targeted PET/CT With 68Ga-Pentixafor Shows Superiority to 18F-FDG in a Patient with Waldenstrom Macroglobulinemia. Clin. Nucl. Med. 2018, 43, 548–550. [Google Scholar] [CrossRef]
- Pan, Q.; Cao, X.; Luo, Y.; Li, J.; Li, F. Chemokine Receptor 4-Targeted 68Ga-Pentixafor PET/CT in Response Assessment of Waldenstrom Macroglobulinemia/Lymphoplasmacytic Lymphoma: Comparison to 18F-FDG PET/CT. Clin. Nucl. Med. 2021, 46, 732–737. [Google Scholar] [CrossRef]
- Domanska, U.M.; Kruizinga, R.C.; Nagengast, W.B.; Timmer-Bosscha, H.; Huls, G.; de Vries, E.G.; Walenkamp, A.M. A Review on CXCR4/CXCL12 Axis in Oncology: No Place to Hide. Eur. J. Cancer 2013, 49, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Guo, L.; Zhao, H.; Zhao, J.; Weng, H.; Zhao, B. CXCR4 Over-Expression and Survival in Cancer: A System Review and Meta-Analysis. Oncotarget 2015, 6, 5022–5040. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Werner, R.A.; Weich, A.; Higuchi, T.; Schmid, J.S.; Schirbel, A.; Lassmann, M.; Wild, V.; Rudelius, M.; Kudlich, T.; Herrmann, K.; et al. Imaging of Chemokine Receptor 4 Expression in Neuroendocrine Tumors—A Triple Tracer Comparative Approach. Theranostics 2017, 7, 1489–1498. [Google Scholar] [CrossRef] [PubMed]
- Vag, T.; Gerngross, C.; Herhaus, P.; Eiber, M.; Philipp-Abbrederis, K.; Graner, F.P.; Ettl, J.; Keller, U.; Wester, H.J.; Schwaiger, M. First Experience with Chemokine Receptor CXCR4-Targeted PET Imaging of Patients with Solid Cancers. J. Nucl. Med. 2016, 57, 741–746. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vag, T.; Steiger, K.; Rossmann, A.; Keller, U.; Noske, A.; Herhaus, P.; Ettl, J.; Niemeyer, M.; Wester, H.J.; Schwaiger, M. PET Imaging of Chemokine Receptor CXCR4 in Patients with Primary and Recurrent Breast Carcinoma. EJNMMI Res. 2018, 8, 90. [Google Scholar] [CrossRef]
- Werner, R.A.; Kircher, S.; Higuchi, T.; Kircher, M.; Schirbel, A.; Wester, H.J.; Buck, A.K.; Pomper, M.G.; Rowe, S.P.; Lapa, C. CXCR4-Directed Imaging in Solid Tumors. Front Oncol. 2019, 9, 770. [Google Scholar] [CrossRef] [Green Version]
- Jacobs, S.M.; Wesseling, P.; de Keizer, B.; Tolboom, N.; Ververs, F.F.T.; Krijger, G.C.; Westerman, B.A.; Snijders, T.J.; Robe, P.A.; van der Kolk, A.G. CXCR4 Expression in Glioblastoma Tissue and the Potential for PET Imaging and Treatment with [(68)Ga]Ga-Pentixafor /[(177)Lu]Lu-Pentixather. Eur. J. Nucl. Med. Mol. Imaging 2021. [Google Scholar] [CrossRef]
- Stumpf, C.; Kaemmerer, D.; Neubauer, E.; Sanger, J.; Schulz, S.; Lupp, A. Somatostatin and CXCR4 Expression Patterns in Adenocarcinoma and Squamous Cell Carcinoma of the Lung Relative to Small Cell Lung Cancer. J. Cancer Res. Clin. Oncol. 2018, 144, 1921–1932. [Google Scholar] [CrossRef]
- Lapa, C.; Luckerath, K.; Rudelius, M.; Schmid, J.S.; Schoene, A.; Schirbel, A.; Samnick, S.; Pelzer, T.; Buck, A.K.; Kropf, S.; et al. [68Ga]Pentixafor-PET/CT for Imaging of Chemokine Receptor 4 Expression in Small Cell Lung Cancer—Initial Experience. Oncotarget 2016, 7, 9288–9295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watts, A.; Singh, B.; Basher, R.; Singh, H.; Bal, A.; Kapoor, R.; Arora, S.K.; Wester, H.J.; Mittal, B.R.; Behera, D. 68Ga-Pentixafor PET/CT Demonstrating Higher CXCR4 Density in Small Cell Lung Carcinoma than in Non-Small Cell Variant. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 909–910. [Google Scholar] [CrossRef]
- Bluemel, C.; Hahner, S.; Heinze, B.; Fassnacht, M.; Kroiss, M.; Bley, T.A.; Wester, H.J.; Kropf, S.; Lapa, C.; Schirbel, A.; et al. Investigating the Chemokine Receptor 4 as Potential Theranostic Target in Adrenocortical Cancer Patients. Clin. Nucl. Med. 2017, 42, e29–e34. [Google Scholar] [CrossRef]
- Schottelius, M.; Osl, T.; Poschenrieder, A.; Herrmann, K.; Lapa, C.; Hoffmann, F.; Schwaiger, M.; Lassmann, M.; Buck, A.; Wester, H. (177) Lu-pentixather: Preclinical and First Patient Results with a Highly Promising CXCR4-Directed Endoradiotherapeutic Agent. J. Nucl. Med. 2015, 56, 339. [Google Scholar]
- Habringer, S.; Lapa, C.; Herhaus, P.; Schottelius, M.; Istvanffy, R.; Steiger, K.; Slotta-Huspenina, J.; Schirbel, A.; Hanscheid, H.; Kircher, S.; et al. Dual Targeting of Acute Leukemia and Supporting Niche by CXCR4-Directed Theranostics. Theranostics 2018, 8, 369–383. [Google Scholar] [CrossRef] [Green Version]
- Maurer, S.; Herhaus, P.; Lippenmeyer, R.; Hanscheid, H.; Kircher, M.; Schirbel, A.; Maurer, H.C.; Buck, A.K.; Wester, H.J.; Einsele, H.; et al. Side Effects of CXC-Chemokine Receptor 4-Directed Endoradiotherapy with Pentixather before Hematopoietic Stem Cell Transplantation. J. Nucl. Med. 2019, 60, 1399–1405. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herrmann, K.; Schottelius, M.; Lapa, C.; Osl, T.; Poschenrieder, A.; Haenscheid, H.; Lueckerath, K.; Schreder, M.; Bluemel, C.; Knott, M. First-In-Man Experience of CXCR4-Directed Endoradiotherapy with 177Lu-And 90Y-Labelled Pentixather in Advanced Stage Multiple Myeloma with Extensive Intra- and Extramedullary Disease. J. Nucl. Med. 2015, 56, 14. [Google Scholar]
- Lapa, C.; Herrmann, K.; Schirbel, A.; Hanscheid, H.; Luckerath, K.; Schottelius, M.; Kircher, M.; Werner, R.A.; Schreder, M.; Samnick, S.; et al. CXCR4-Directed Endoradiotherapy Induces High Response Rates in Extramedullary Relapsed Multiple Myeloma. Theranostics 2017, 7, 1589–1597. [Google Scholar] [CrossRef] [Green Version]
- Lapa, C.; Hanscheid, H.; Kircher, M.; Schirbel, A.; Wunderlich, G.; Werner, R.A.; Samnick, S.; Kotzerke, J.; Einsele, H.; Buck, A.K.; et al. Feasibility of CXCR4-Directed Radioligand Therapy in Advanced Diffuse Large B-Cell Lymphoma. J. Nucl. Med. 2019, 60, 60–64. [Google Scholar] [CrossRef]
- Kircher, M.; Herhaus, P.; Schottelius, M.; Buck, A.K.; Werner, R.A.; Wester, H.J.; Keller, U.; Lapa, C. CXCR4-Directed Theranostics in Oncology and Inflammation. Ann. Nucl. Med. 2018, 32, 503–511. [Google Scholar] [CrossRef] [Green Version]
- Borchert, T.; Beitar, L.; Langer, L.B.N.; Polyak, A.; Wester, H.J.; Ross, T.L.; Hilfiker-Kleiner, D.; Bengel, F.M.; Thackeray, J.T. Dissecting the Target Leukocyte Subpopulations of Clinically Relevant Inflammation Radiopharmaceuticals. J. Nucl. Cardiol. 2019, 28, 1636–1645. [Google Scholar] [CrossRef]
- Thackeray, J.T.; Derlin, T.; Haghikia, A.; Napp, L.C.; Wang, Y.; Ross, T.L.; Schafer, A.; Tillmanns, J.; Wester, H.J.; Wollert, K.C.; et al. Molecular Imaging of the Chemokine Receptor CXCR4 After Acute Myocardial Infarction. JACC Cardiovasc. Imaging 2015, 8, 1417–1426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lapa, C.; Reiter, T.; Werner, R.A.; Ertl, G.; Wester, H.J.; Buck, A.K.; Bauer, W.R.; Herrmann, K. [(68)Ga]Pentixafor-PET/CT for Imaging of Chemokine Receptor 4 Expression after Myocardial Infarction. JACC Cardiovasc. Imaging 2015, 8, 1466–1468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reiter, T.; Kircher, M.; Schirbel, A.; Werner, R.A.; Kropf, S.; Ertl, G.; Buck, A.K.; Wester, H.J.; Bauer, W.R.; Lapa, C. Imaging of C-X-C Motif Chemokine Receptor CXCR4 Expression after Myocardial Infarction with [(68)Ga]Pentixafor-PET/CT in Correlation with Cardiac MRI. JACC Cardiovasc. Imaging 2018, 11, 1541–1543. [Google Scholar] [CrossRef] [PubMed]
- Hess, A.; Derlin, T.; Koenig, T.; Diekmann, J.; Wittneben, A.; Wang, Y.; Wester, H.J.; Ross, T.L.; Wollert, K.C.; Bauersachs, J.; et al. Molecular Imaging-Guided Repair after Acute Myocardial Infarction by Targeting the Chemokine Receptor CXCR4. Eur. Heart J. 2020, 41, 3564–3575. [Google Scholar] [CrossRef]
- Wang, Y.; Dembowsky, K.; Chevalier, E.; Stuve, P.; Korf-Klingebiel, M.; Lochner, M.; Napp, L.C.; Frank, H.; Brinkmann, E.; Kanwischer, A.; et al. C-X-C Motif Chemokine Receptor 4 Blockade Promotes Tissue Repair after Myocardial Infarction by Enhancing Regulatory T Cell Mobilization and Immune-Regulatory Function. Circulation 2019, 139, 1798–1812. [Google Scholar] [CrossRef] [PubMed]
- Rieckmann, M.; Delgobo, M.; Gaal, C.; Buchner, L.; Steinau, P.; Reshef, D.; Gil-Cruz, C.; Horst, E.N.T.; Kircher, M.; Reiter, T.; et al. Myocardial Infarction Triggers Cardioprotective Antigen-Specific T Helper Cell Responses. J. Clin. Invest. 2019, 129, 4922–4936. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Yu, W.; Wollenweber, T.; Lu, X.; Wei, Y.; Beitzke, D.; Wadsak, W.; Kropf, S.; Wester, H.J.; Haug, A.R.; et al. [(68)Ga]Pentixafor PET/MR imaging of Chemokine Receptor 4 Expression in the Human Carotid Artery. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1616–1625. [Google Scholar] [CrossRef] [Green Version]
- Hyafil, F.; Pelisek, J.; Laitinen, I.; Schottelius, M.; Mohring, M.; Doring, Y.; van der Vorst, E.P.; Kallmayer, M.; Steiger, K.; Poschenrieder, A.; et al. Imaging the Cytokine Receptor CXCR4 in Atherosclerotic Plaques with the Radiotracer (68)Ga-Pentixafor for PET. J. Nucl. Med. 2017, 58, 499–506. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Heber, D.; Leike, T.; Beitzke, D.; Lu, X.; Zhang, X.; Wei, Y.; Mitterhauser, M.; Wadsak, W.; Kropf, S.; et al. [68Ga]Pentixafor-PET/MRI for the Detection of Chemokine Receptor 4 Expression in Atherosclerotic Plaques. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 558–566. [Google Scholar] [CrossRef]
- Weiberg, D.; Thackeray, J.T.; Daum, G.; Sohns, J.M.; Kropf, S.; Wester, H.J.; Ross, T.L.; Bengel, F.M.; Derlin, T. Clinical Molecular Imaging of Chemokine Receptor CXCR4 Expression in Atherosclerotic Plaque Using (68)Ga-Pentixafor PET: Correlation with Cardiovascular Risk Factors and Calcified Plaque Burden. J. Nucl. Med. 2018, 59, 266–272. [Google Scholar] [CrossRef] [Green Version]
- Derlin, T.; Sedding, D.G.; Dutzmann, J.; Haghikia, A.; Konig, T.; Napp, L.C.; Schutze, C.; Owsianski-Hille, N.; Wester, H.J.; Kropf, S.; et al. Imaging of Chemokine Receptor CXCR4 Expression in Culprit and Nonculprit Coronary Atherosclerotic Plaque Using Motion-Corrected [(68)Ga]pentixafor PET/CT. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 1934–1944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Kemmer, L.; Zhang, X.; Kircher, M.; Buck, A.K.; Wester, H.J.; Hacker, M.; Lapa, C. Anti-Inflammatory Effects on Atherosclerotic Lesions Induced by CXCR4-Directed Endoradiotherapy. J. Am. Coll. Cardiol. 2018, 72, 122–123. [Google Scholar] [CrossRef] [PubMed]
- Pawig, L.; Klasen, C.; Weber, C.; Bernhagen, J.; Noels, H. Diversity and Inter-Connections in the CXCR4 Chemokine Receptor/Ligand Family: Molecular Perspectives. Front. Immunol. 2015, 6, 429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van der Vorst, E.P.; Doring, Y.; Weber, C. MIF and CXCL12 in Cardiovascular Diseases: Functional Differences and Similarities. Front. Immunol. 2015, 6, 373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Merckelbach, S.; van der Vorst, E.P.C.; Kallmayer, M.; Rischpler, C.; Burgkart, R.; Doring, Y.; de Borst, G.J.; Schwaiger, M.; Eckstein, H.H.; Weber, C.; et al. Expression and Cellular Localization of CXCR4 and CXCL12 in Human Carotid Atherosclerotic Plaques. Thromb. Haemost. 2018, 118, 195–206. [Google Scholar] [CrossRef]
- Kircher, M.; Tran-Gia, J.; Kemmer, L.; Zhang, X.; Schirbel, A.; Werner, R.A.; Buck, A.K.; Wester, H.J.; Hacker, M.; Lapa, C.; et al. Imaging Inflammation in Atherosclerosis with CXCR4-directed (68)Ga-Pentixafor PET/CT—Correlation with (18)F-FDG PET/CT. J. Nucl. Med. 2020, 61, 751–756. [Google Scholar] [CrossRef]
- Derlin, T.; Jaeger, B.; Jonigk, D.; Apel, R.M.; Freise, J.; Shin, H.O.; Weiberg, D.; Warnecke, G.; Ross, T.L.; Wester, H.J.; et al. Clinical Molecular Imaging of Pulmonary CXCR4 Expression to Predict Outcome of Pirfenidone Treatment in Idiopathic Pulmonary Fibrosis. Chest 2021, 159, 1094–1106. [Google Scholar] [CrossRef]
- Bouter, C.; Meller, B.; Sahlmann, C.O.; Staab, W.; Wester, H.J.; Kropf, S.; Meller, J. (68)Ga-Pentixafor PET/CT Imaging of Chemokine Receptor CXCR4 in Chronic Infection of the Bone: First Insights. J. Nucl. Med. 2018, 59, 320–326. [Google Scholar] [CrossRef] [Green Version]
- Bouter, Y.; Meller, B.; Sahlmann, C.O.; Wolf, B.J.; Langer, L.; Bankstahl, J.P.; Wester, H.J.; Kropf, S.; Meller, J.; Bouter, C. Immunohistochemical Detection of Chemokine Receptor 4 Expression in Chronic Osteomyelitis Confirms Specific Uptake in 68Ga-Pentixafor-PET/CT. Nuklearmedizin 2018, 57, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Derlin, T.; Gueler, F.; Brasen, J.H.; Schmitz, J.; Hartung, D.; Herrmann, T.R.; Ross, T.L.; Wacker, F.; Wester, H.J.; Hiss, M.; et al. Integrating MRI and Chemokine Receptor CXCR4-Targeted PET for Detection of Leukocyte Infiltration in Complicated Urinary Tract Infections After Kidney Transplantation. J. Nucl. Med. 2017, 58, 1831–1837. [Google Scholar] [CrossRef] [Green Version]
- Osl, T.; Schmidt, A.; Schwaiger, M.; Schottelius, M.; Wester, H.J. A New Class of PentixaFor- and PentixaTher-Based Theranostic Agents with Enhanced CXCR4-Targeting Efficiency. Theranostics 2020, 10, 8264–8280. [Google Scholar] [CrossRef] [PubMed]
- Glasenapp, A.; Derlin, K.; Gutberlet, M.; Hess, A.; Ross, T.L.; Wester, H.J.; Bengel, F.M.; Thackeray, J.T. Molecular Imaging of Inflammation and Fibrosis in Pressure Overload Heart Failure. Circ. Res. 2021, 129, 369–382. [Google Scholar] [CrossRef]
- Hess, A.; Wittneben, A.; Hueper, K.; Ross, T.L.; Wester, H.; Bengel, F.; Thackeray, J. Imaging of Chemokine Receptor Type 4 (CXCR4) Upregulation Early after Acute Myocardial Infarction Predicts Subsequent Left Ventricular Remodeling in Mice. J. Nucl. Med. 2017, 58 (Suppl. S1), 163. [Google Scholar]
- Hess, A.; Wittneben, A.; Kropf, S.; Wester, H.; Bengel, F.; Thackeray, J. CXCR4-Targeted Imaging of Leukocyte Mobilization after Myocardial Infarction. J. Nucl. Med. 2018, 59 (Suppl. S1), 37. [Google Scholar]
- Werner, R.A.; Hess, A.; Koenig, T.; Diekmann, J.; Derlin, T.; Melk, A.; Thackeray, J.T.; Bauersachs, J.; Bengel, F.M. Molecular Imaging of Inflammation Crosstalk along the Cardio-Renal Axis Following Acute Myocardial Infarction. Theranostics 2021, 11, 7984–7994. [Google Scholar] [CrossRef]
- Meester, E.J.; de Blois, E.; Krenning, B.J.; van der Steen, A.F.W.; Norenberg, J.P.; van Gaalen, K.; Bernsen, M.R.; de Jong, M.; van der Heiden, K. Autoradiographical Assessment of Inflammation-Targeting Radioligands for Atherosclerosis Imaging: Potential for Plaque Phenotype Identification. EJNMMI Res. 2021, 11, 27. [Google Scholar] [CrossRef]
- Baba, O.; Huang, L.H.; Elvington, A.; Szpakowska, M.; Sultan, D.; Heo, G.S.; Zhang, X.; Luehmann, H.; Detering, L.; Chevigne, A.; et al. CXCR4-Binding Positron Emission Tomography Tracers Link Monocyte Recruitment and Endothelial Injury in Murine Atherosclerosis. Arter. Thromb Vasc Biol. 2021, 41, 822–836. [Google Scholar] [CrossRef]
- Burke, B.P.; Miranda, C.S.; Lee, R.E.; Renard, I.; Nigam, S.; Clemente, G.S.; D’Huys, T.; Ruest, T.; Domarkas, J.; Thompson, J.A.; et al. (64)Cu PET Imaging of the CXCR4 Chemokine Receptor Using a Cross-Bridged Cyclam Bis-Tetraazamacrocyclic Antagonist. J. Nucl. Med. 2020, 61, 123–128. [Google Scholar] [CrossRef]
- Brickute, D.; Braga, M.; Kaliszczak, M.A.; Barnes, C.; Lau, D.; Carroll, L.; Stevens, E.; Trousil, S.; Alam, I.S.; Nguyen, Q.D.; et al. Development and Evaluation of an (18)F-Radiolabeled Monocyclam Derivative for Imaging CXCR4 Expression. Mol. Pharm. 2019, 16, 2106–2117. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; You, L.; Chen, S.; Gao, M.; Guo, Z.; Du, J.; Lu, J.; Zhang, X. Development of a Novel (99m) Tc-Labeled Small Molecular Antagonist for CXCR4 Positive Tumor Imaging. J. Labelled Comp. Radiopharm. 2018, 61, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Amor-Coarasa, A.; Kelly, J.; Ponnala, S.; Vedvyas, Y.; Nikolopoulou, A.; Williams, C.; Jr Jin, M.M.; David Warren, J.; Babich, J.W. [(18)F]RPS-544: A PET Tracer for Imaging the Chemokine Receptor CXCR4. Nucl. Med. Biol. 2018, 60, 37–44. [Google Scholar] [CrossRef]
- Zhang, H.; Maeda, M.; Shindo, M.; Ko, M.; Mane, M.; Grommes, C.; Weber, W.; Blasberg, R. Imaging CXCR4 Expression with Iodinated and Brominated Cyclam Derivatives. Mol. Imaging Biol. 2020, 22, 1184–1196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oum, Y.H.; Shetty, D.; Yoon, Y.; Liang, Z.; Voll, R.J.; Goodman, M.M.; Shim, H. A Benzenesulfonamide Derivative as a Novel PET Radioligand for CXCR4. Bioorg. Med. Chem. 2020, 28, 115240. [Google Scholar] [CrossRef]
- Schottelius, M.; Ludescher, M.; Richter, F.; Kapp, T.G.; Kessler, H.; Wester, H.J. Validation of [(125)I]CPCR4.3 as an Investigative Tool for the Sensitive and Specific Detection of hCXCR4 and mCXCR4 Expression In Vitro and In Vivo. EJNMMI Res. 2019, 9, 75. [Google Scholar] [CrossRef]
- Fang, H.Y.; Munch, N.S.; Schottelius, M.; Ingermann, J.; Liu, H.; Schauer, M.; Stangl, S.; Multhoff, G.; Steiger, K.; Gerngross, C.; et al. CXCR4 Is a Potential Target for Diagnostic PET/CT Imaging in Barrett’s Dysplasia and Esophageal Adenocarcinoma. Clin. Cancer Res. 2018, 24, 1048–1061. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marcazzan, S.; Braz Carvalho, M.J.; Konrad, M.; Strangmann, J.; Tenditnaya, A.; Baumeister, T.; Schmid, R.M.; Wester, H.-J.; Ntziachristos, V.; Gorpas, D.; et al. CXCR4 Peptide-Based Fluorescence Endoscopy in a Mouse Model of Barrett’s Esophagus. EJNMMI Res. 2021; submitted for publication. [Google Scholar]
- Konrad, M.; Lapa, C.; Wester, H.-J.; Schottelius, M. [99mTc]PentixaSPECT—Expanding the Pentixafor/Pentixather-based Theranostic Concept Towards High-Contrast SPECT Imaging of CXCR4 Expression In Vivo. Unpublished work. 2021. [Google Scholar]
- Avila-Sanchez, M.; Ferro-Flores, G.; Jimenez-Mancilla, N.; Ocampo-Garcia, B.; Bravo-Villegas, G.; Luna-Gutierrez, M.; Santos-Cuevas, C.; Azorin-Vega, E.; Aranda-Lara, L.; Isaac-Olive, K.; et al. Synthesis and Preclinical Evaluation of the Tc-99m-/Lu-177-CXCR4-L Theranostic Pair for In Vivo Chemokine-4 Receptor-Specific Targeting. J. Radioanal. Nucl. Chem. 2020, 324, 21–32. [Google Scholar] [CrossRef]
- Trotta, A.M.; Aurilio, M.; D’Alterio, C.; Ierano, C.; Di Martino, D.; Barbieri, A.; Luciano, A.; Gaballo, P.; Santagata, S.; Portella, L.; et al. Novel Peptide-Based PET Probe for Non-Invasive Imaging of C-X-C Chemokine Receptor Type 4 (CXCR4) in Tumors. J. Med. Chem. 2021, 64, 3449–3461. [Google Scholar] [CrossRef]
- Suzuki, K.; Ui, T.; Nagano, A.; Hino, A.; Arano, Y. C-Terminal-Modified LY2510924: A Versatile Scaffold for Targeting C-X-C Chemokine Receptor Type 4. Sci. Rep. 2019, 9, 15284. [Google Scholar] [CrossRef] [Green Version]
- Lau, J.; Kwon, D.; Rousseau, E.; Zhang, Z.; Zeisler, J.; Uribe, C.F.; Kuo, H.T.; Zhang, C.; Lin, K.S.; Benard, F. [(68)Ga]Ga/[(177)Lu]Lu-BL01, a Novel Theranostic Pair for Targeting C-X-C Chemokine Receptor 4. Mol. Pharm. 2019, 16, 4688–4695. [Google Scholar] [CrossRef] [PubMed]
- Kwon, D.; Lozada, J.; Zhang, Z.; Zeisler, J.; Poon, R.; Zhang, C.; Roxin, A.; Lin, K.S.; Perrin, D.; Benard, F. High-Contrast CXCR4-Targeted (18)F-PET Imaging Using a Potent and Selective Antagonist. Mol. Pharm. 2021, 18, 187–197. [Google Scholar] [CrossRef]
- Peng, T.; Wang, X.; Li, Z.; Bi, L.; Gao, J.; Yang, M.; Wang, Y.; Yao, X.; Shan, H.; Jin, H. Preclinical Evaluation of [(64)Cu]NOTA-CP01 as a PET Imaging Agent for Metastatic Esophageal Squamous Cell Carcinoma. Mol. Pharm. 2021, 18, 3638–3648. [Google Scholar] [CrossRef]
- Turnbull, W.L.; Yu, L.; Murrell, E.; Milne, M.; Charron, C.L.; Luyt, L.G. A Dual Modality (99m)Tc/Re(i)-labelled T140 Analogue for Imaging of CXCR4 Expression. Org. Biomol. Chem. 2019, 17, 598–608. [Google Scholar] [CrossRef]
- Lesniak, W.G.; Aboye, T.; Chatterjee, S.; Camarero, J.A.; Nimmagadda, S. In vivo Evaluation of an Engineered Cyclotide as Specific CXCR4 Imaging Reagent. Chemistry 2017, 23, 14469–14475. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, X.; Wu, H.Y.; Sun, L.; Ma, Y.; Xu, J.; Lin, Q.; Zeng, D. (64)Cu-Labeled Ubiquitin for PET Imaging of CXCR4 Expression in Mouse Breast Tumor. ACS Omega. 2019, 4, 12432–12437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hartimath, S.V.; Draghiciu, O.; Daemen, T.; Nijman, H.W.; van Waarde, A.; Dierckx, R.; de Vries, E.F.J. Therapy-Induced Changes in CXCR4 Expression in Tumor Xenografts Can Be Monitored Noninvasively with N-[(11)C]Methyl-AMD3465 PET. Mol. Imaging Biol. 2020, 22, 883–890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Clercq, E.; Yamamoto, N.; Pauwels, R.; Balzarini, J.; Witvrouw, M.; De Vreese, K.; Debyser, Z.; Rosenwirth, B.; Peichl, P.; Datema, R.; et al. Highly Potent and Selective Inhibition of Human Immunodeficiency Virus by the Bicyclam Derivative JM3100. Antimicrob Agents Chemother. 1994, 38, 668–674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamamura, H.; Xu, Y.; Hattori, T.; Zhang, X.; Arakaki, R.; Kanbara, K.; Omagari, A.; Otaka, A.; Ibuka, T.; Yamamoto, N.; et al. A Low-Molecular-Weight Inhibitor Against the Chemokine Receptor CXCR4: A Strong Anti-HIV Peptide T140. Biochem. Biophys. Res. Commun. 1998, 253, 877–882. [Google Scholar] [CrossRef]
- Muller, A.; Homey, B.; Soto, H.; Ge, N.; Catron, D.; Buchanan, M.E.; McClanahan, T.; Murphy, E.; Yuan, W.; Wagner, S.N.; et al. Involvement of Chemokine Receptors in Breast Cancer Metastasis. Nature 2001, 410, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Kuil, J.; Buckle, T.; van Leeuwen, F.W. Imaging Agents for the Chemokine Receptor 4 (CXCR4). Chem. Soc. Rev. 2012, 41, 5239–5261. [Google Scholar] [CrossRef]
- Weiss, I.D.; Jacobson, O. Molecular Imaging of Chemokine Receptor CXCR4. Theranostics 2013, 3, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Wan, Q.; Cheng, Z.; Chen, Y. Radionuclide-Labeled Peptides for Imaging and Treatment of CXCR4- Overexpressing Malignant Tumors. Curr. Top. Med. Chem. 2019, 19, 17–32. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, M.; Wang, L.; Wang, S.; Kang, F.; Li, G.; Jacobson, O.; Niu, G.; Yang, W.; Wang, J.; et al. Prospective Study of (68)Ga-NOTA-NFB: Radiation Dosimetry in Healthy Volunteers and First Application in Glioma Patients. Theranostics 2015, 5, 882–889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herrmann, K.; Lapa, C.; Wester, H.J.; Schottelius, M.; Schiepers, C.; Eberlein, U.; Bluemel, C.; Keller, U.; Knop, S.; Kropf, S.; et al. Biodistribution and radiation dosimetry for the chemokine receptor CXCR4-targeting probe 68Ga-pentixafor. J. Nucl. Med. 2015, 56, 410–416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, Y.; Lin, Y.; Lin, X.; Sun, X.; Luo, K. Combination of PET and CXCR4-Targeted Peptide Molecule Agents for Noninvasive Tumor Monitoring. J. Cancer 2019, 10, 3420–3426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilson, G.C.; Freeman, C.M.; Kuethe, J.W.; Quillin, R.C.; Nojima, H., 3rd; Schuster, R.; Blanchard, J.; Edwards, M.J.; Caldwell, C.C.; Lentsch, A.B. CXC Chemokine Receptor-4 Signaling Limits Hepatocyte Proliferation after Hepatic Ischemia-Reperfusion in Mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 308, G702–G709. [Google Scholar] [CrossRef] [Green Version]
- Jacobson, O.; Weiss, I.D.; Szajek, L.; Farber, J.M.; Kiesewetter, D.O. 64Cu-AMD3100—A Novel Imaging Agent for Targeting Chemokine Receptor CXCR4. Bioorg. Med. Chem. 2009, 17, 1486–1493. [Google Scholar] [CrossRef] [Green Version]
- Weiss, I.D.; Huff, L.M.; Evbuomwan, M.O.; Xu, X.; Dang, H.D.; Velez, D.S.; Singh, S.P.; Zhang, H.H.; Gardina, P.J.; Lee, J.H.; et al. Screening of Cancer Tissue Arrays Identifies CXCR4 on Adrenocortical Carcinoma: Correlates with Expression and Quantification on Metastases Using (64)Cu-plerixafor PET. Oncotarget 2017, 8, 73387–73406. [Google Scholar] [CrossRef] [Green Version]
- Di Maro, S.; Di Leva, F.S.; Trotta, A.M.; Brancaccio, D.; Portella, L.; Aurilio, M.; Tomassi, S.; Messere, A.; Sementa, D.; Lastoria, S.; et al. Structure-Activity Relationships and Biological Characterization of a Novel, Potent, and Serum Stable C-X-C Chemokine Receptor Type 4 (CXCR4) Antagonist. J. Med. Chem. 2017, 60, 9641–9652. [Google Scholar] [CrossRef]
- Wester, H.J.; Keller, U.; Schottelius, M.; Beer, A.; Philipp-Abbrederis, K.; Hoffmann, F.; Šimeček, J.; Gerngross, C.; Lassmann, M.; Herrmann, K. Disclosing the CXCR4 Expression in Lymphoproliferative Diseases by Targeted Molecular Imaging. Theranostics 2015, 5, 618. [Google Scholar] [CrossRef]
- Bosma, S.E.; van Driel, P.B.; Hogendoorn, P.C.; Dijkstra, P.S.; Sier, C.F. Introducing Fluorescence Guided Surgery into Orthopedic Oncology: A Systematic Review of Candidate Protein Targets for Ewing Sarcoma. J. Surg. Oncol. 2018, 118, 906–914. [Google Scholar] [CrossRef] [Green Version]
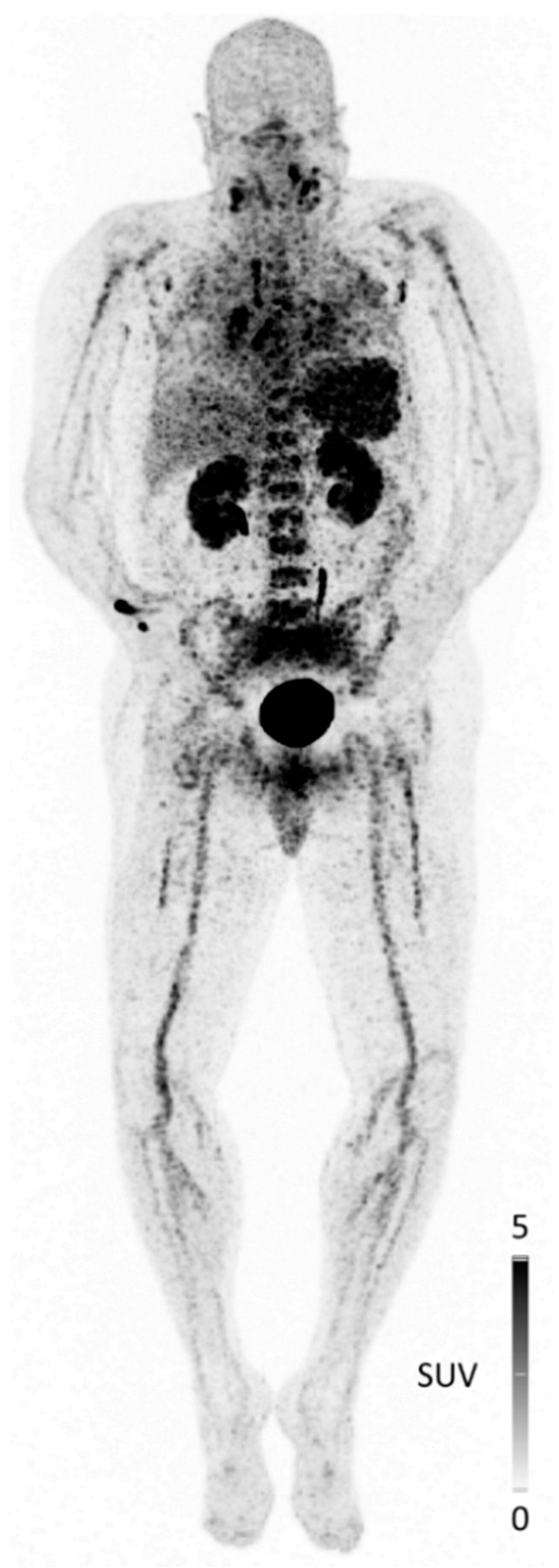
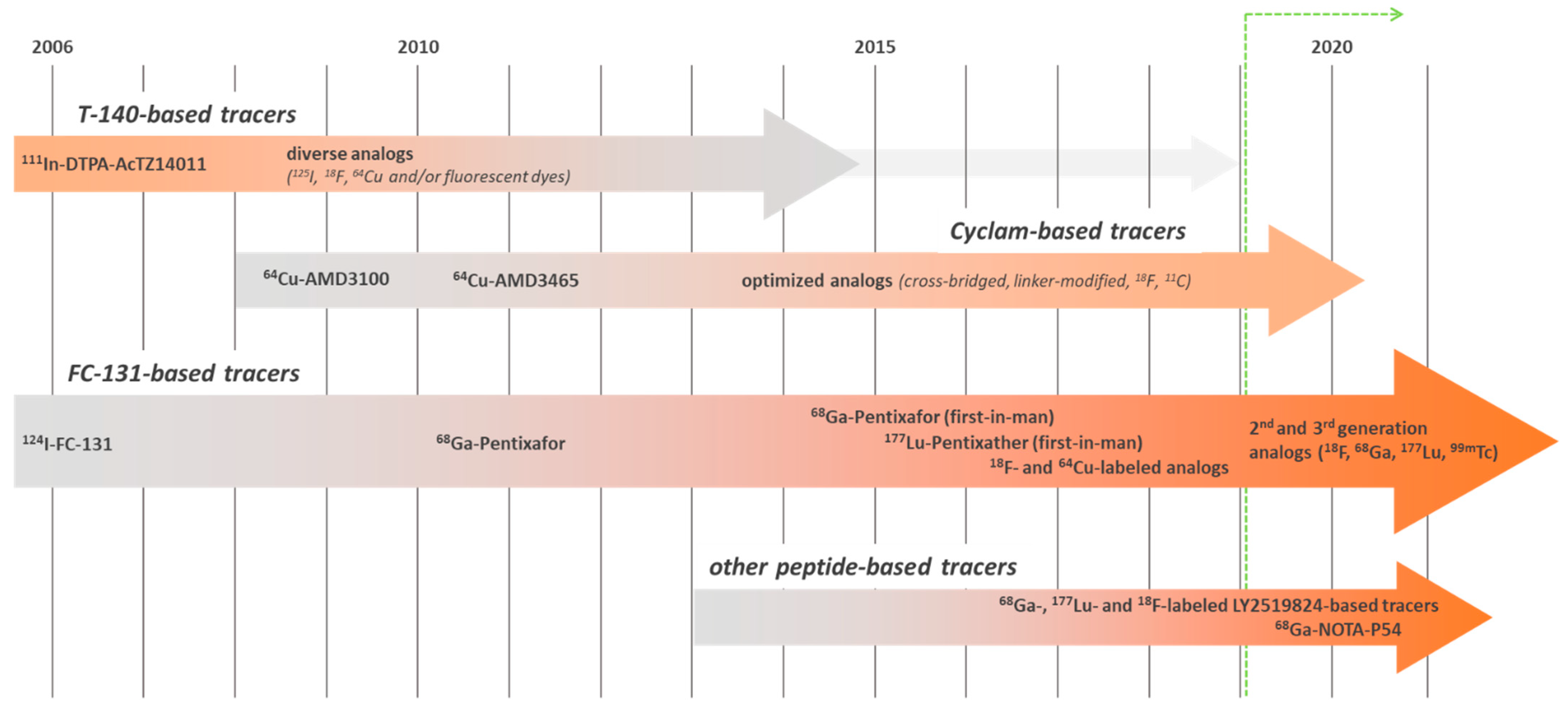
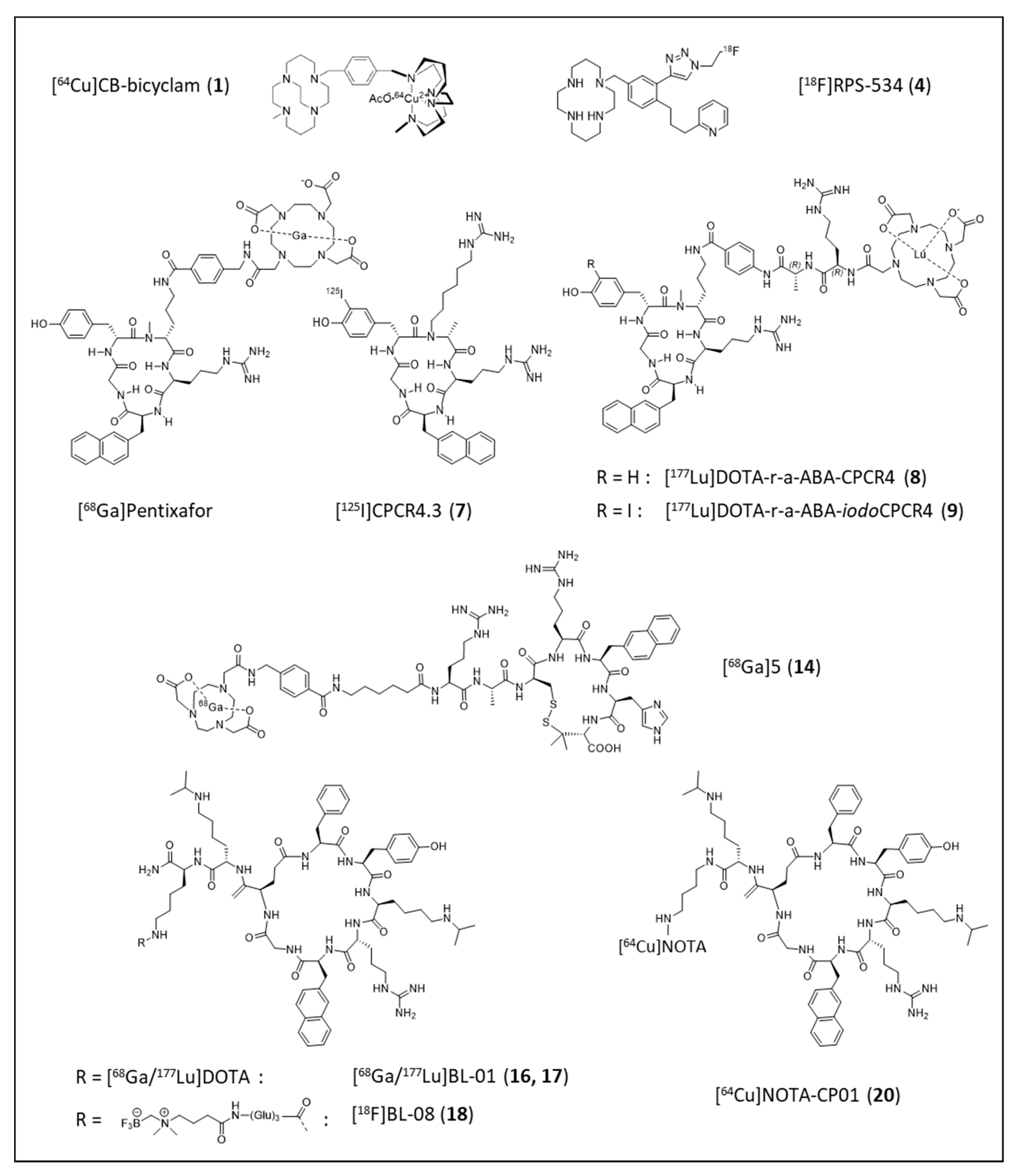
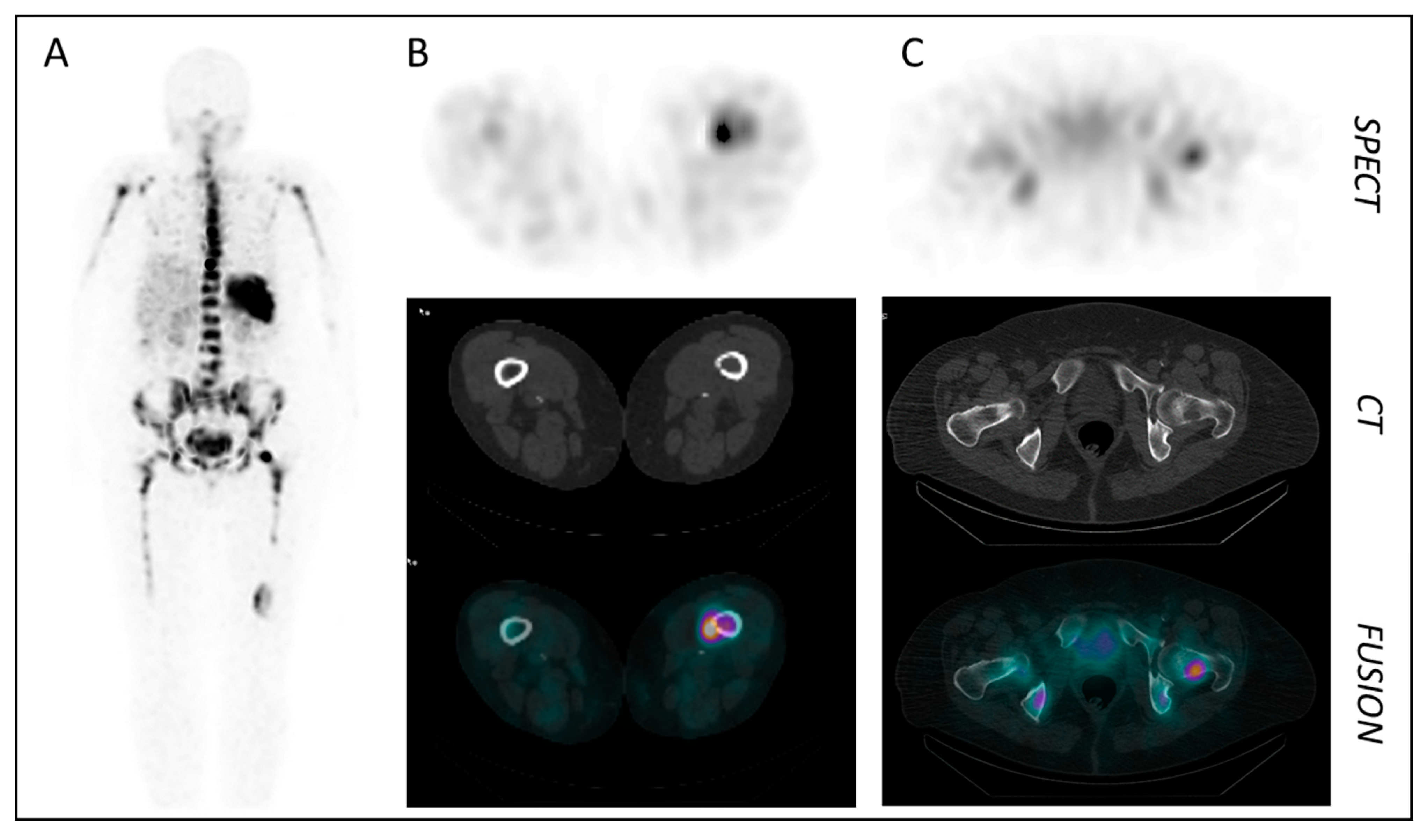
| Entry | Compound | Affinity [IC50, nM] | Internalized [% of Total Cellular Activity] | Tumor Uptake [%iD/g] | Liver [%iD/g] | T/Muscle Ratio | Ref. |
|---|---|---|---|---|---|---|---|
| 1 | [64Cu]CB-bicyclam | 8 (hCXCR4) b | n.d. | SUVmax: 7.4 ± 1.8 (U87.CXCR4) | 13.8 | 23.6 ± 2.7 | [69] |
| 2 (mCXCR4) a | SUVmax: 0.8 ± 0.1 (U87) | 3.0 ± 0.5 | |||||
| 2 | [18F]MCFB | 111 ± 4 a | ~40 | 3.3 ± 0.9 (U2932) | 63 ± 5 | 4.0 ± 0.8 | [70] |
| 1.8 ± 0.1 (SUDHL-8) | 52 ± 3 | 2.1 ± 0.3 | |||||
| 3 | [99mTc]AMD3465 | n.d. | n.d. | 2.1 ± 0.4 (MCF-7) | 25 ± 7 | 4.7 | [71] |
| 4 | [18F]RPS-534 | 218 ± 38 c | ~7 (2 h) | 7.2 ± 0.3 (PC3-CXCR4) | 19.1 ± 0.4 | 42.4 ± 0.1 | [72] |
| 5 | [76Br]HZ270-1 | 6.7 ± 0.7 c | n.d. | 9.5 ± 1.3 (U87.CXCR4, 24 h) | 7.6 ± 1.3 (24 h) | n.d. | [73] |
| 6 | [18F]5 | 6.9 d | n.d. | 4.0 ± 0.3 (SCCHN, 1.5 h) | 1.2 ± 0.1 (90 min) | 25 | [74] |
| 7 | [125I]CPCR4.3 | 5.4 ± 1.5 (hCXCR4) e | 68 ± 3 (MCF-7) | n.d. | 21.2 ± 2.9 | n.d. | [75,76] |
| 4.9 ± 1.7 (mCXCR4) f | |||||||
| 8 | [177Lu]DOTA-r-a-ABA-CPCR4 | 1.5 ± 0.1 (hCXCR4) e | 65 ± 6 (Chem1) | 18.3 ± 3.7 (Daudi) | 11.9 ± 1.6 | 413 ± 100 | [62] |
| 182 ± 26 (mCXCR4) f | |||||||
| 9 | [177Lu]DOTA-r-a-ABA-iodoCPCR4 | 1.7 ± 0.6 (hCXCR4) e | 91 ± 4 (Chem1) | 17.2 ± 2.0 (Daudi) | 27.1 ± 1.9 | 226 ± 36 | |
| 49 ± 1 (mCXCR4) f | |||||||
| 10 | [125I]MK007 | 10.2 ± 4.0 e | n.d. | 1.1 ± 0.1 (Jurkat) | 35.3 ± 1.0 | ~2 | [77] |
| 11 | [99mTc]PentixaSPECT | 10.2 ± 2.4 g | 95 (Chem1) | 8.6 ± 1.3 (Jurkat) | 7.7 ± 0.7 | 29 ± 6 | [78] |
| 12 | [99mTc]HYNIC-L | Kd 3.3 ± 0.4 (DU-4475) | ~9 | 3.2 ± 0.9 (DU-4475) | 2.0 ± 0.4 | n.d. | [79] |
| 13 | [177Lu]DOTA-HYNIC-L | Kd 3.2 ± 0.4 (DU-4475) | ~9 | 4.2 ± 1.1 (DU-4475) | 2.3 ± 0.5 | n.d. | |
| 14 | [68Ga]5 | 15.6 ± 4.2 e | 91 (Chem1) | 7.9 ± 1.4 (Daudi) | 0.36 ± 0.01 | 115 ± 48 | [80] |
| 15 | [67Ga]FRM001 | 2.3 ± 0.5 a | ~15 | 12.0 ± 2.0 (CCRF-CEM, 4 h) | 16.1 ± 2.7 (4h) | 112 | [81] |
| 16 | [68Ga]BL-01 | 21.2 ± 16 a | n.d. | 10.2 ± 2.6 (Daudi) | 7.1 ± 1.3 | 23 ± 4 | [82] |
| 17 | [177Lu]BL-01 | 7.1 ± 1.7 a | n.d. | 14.0 ± 1.1 (Daudi) | 10.3 ± 0.9 | 32 ± 3 | |
| 18 | [18F]BL-08 | 11.6 ± 7.0 a | n.d. | 7.6 ± 1.4 (Daudi) | 0.62 ± 0.02 | 108 ± 25 | [83] |
| 19 | [18F]BL-09 | 13.4 ± 2.3 a | n.d. | 6.6 ± 2.1 (Daudi) | 0.56 ± 0.09 | 83 ± 19 | |
| 20 | [64Cu]NOTA-CP01 | 1.6 ± 1.0 h | n.d. | SUVmax: 1.3 ± 0.2 (EC109, 6 h) | SUVmax: ~3.5 (6 h) | 15.4 ± 3.0 (6 h) | [84] |
| 21 | [99mTc]T140 analog | 1.9 a | negligible | 0.5 ± 0.1 (U87.CXCR4, 2 h) | 22.7 ± 5.0 (2 h) | ~2 (2 h) | [85] |
| 22 | [64Cu]DOTA-vMIP-II | n.d. | n.d. | ~4.5 (aortic plaque) | n.d. | ~3 | [68] |
| 23 | [64Cu]MCo-CVX-6D | 0.07 ± 0.02 (FRET) | n.d. | 5.7 ± 0.5 (U87.CXCR4, 24 h) | 23.3 ± 2.1 (24 h) | 19.9 ± 4.7 | [86] |
| 24 | [64Cu]Ubiquitin | n.d. | n.d. | 1.6 ± 0.6 (4T1, 2 h) | ~4.8 (2 h) | 8.5 ± 2.3 (2 h) | [87] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schottelius, M.; Herrmann, K.; Lapa, C. In Vivo Targeting of CXCR4—New Horizons. Cancers 2021, 13, 5920. https://doi.org/10.3390/cancers13235920
Schottelius M, Herrmann K, Lapa C. In Vivo Targeting of CXCR4—New Horizons. Cancers. 2021; 13(23):5920. https://doi.org/10.3390/cancers13235920
Chicago/Turabian StyleSchottelius, Margret, Ken Herrmann, and Constantin Lapa. 2021. "In Vivo Targeting of CXCR4—New Horizons" Cancers 13, no. 23: 5920. https://doi.org/10.3390/cancers13235920
APA StyleSchottelius, M., Herrmann, K., & Lapa, C. (2021). In Vivo Targeting of CXCR4—New Horizons. Cancers, 13(23), 5920. https://doi.org/10.3390/cancers13235920






