Radiomics Models for Predicting Microvascular Invasion in Hepatocellular Carcinoma: A Systematic Review and Radiomics Quality Score Assessment
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
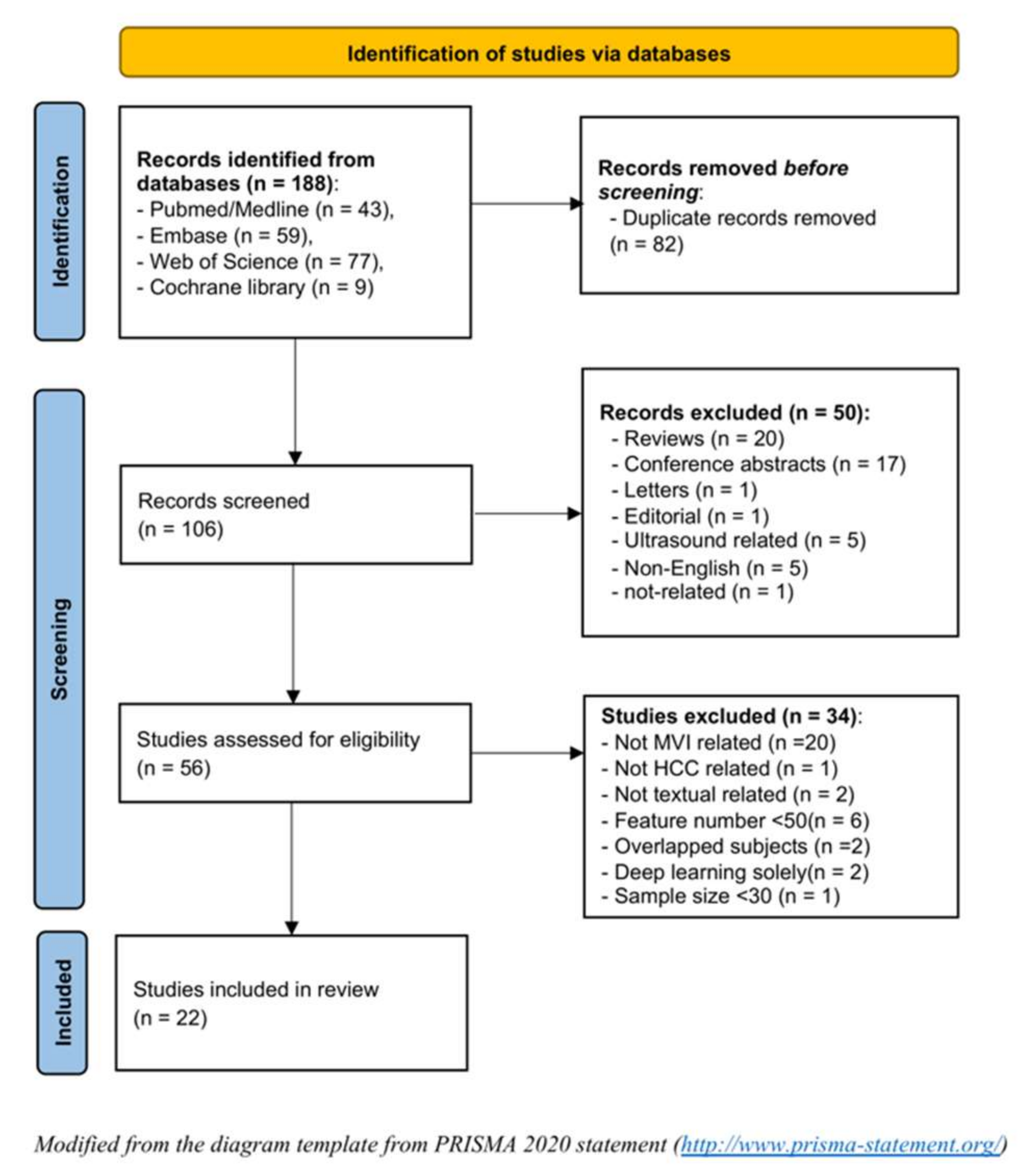
2.1. Literature Research and Study Selection
2.2. Data Extraction
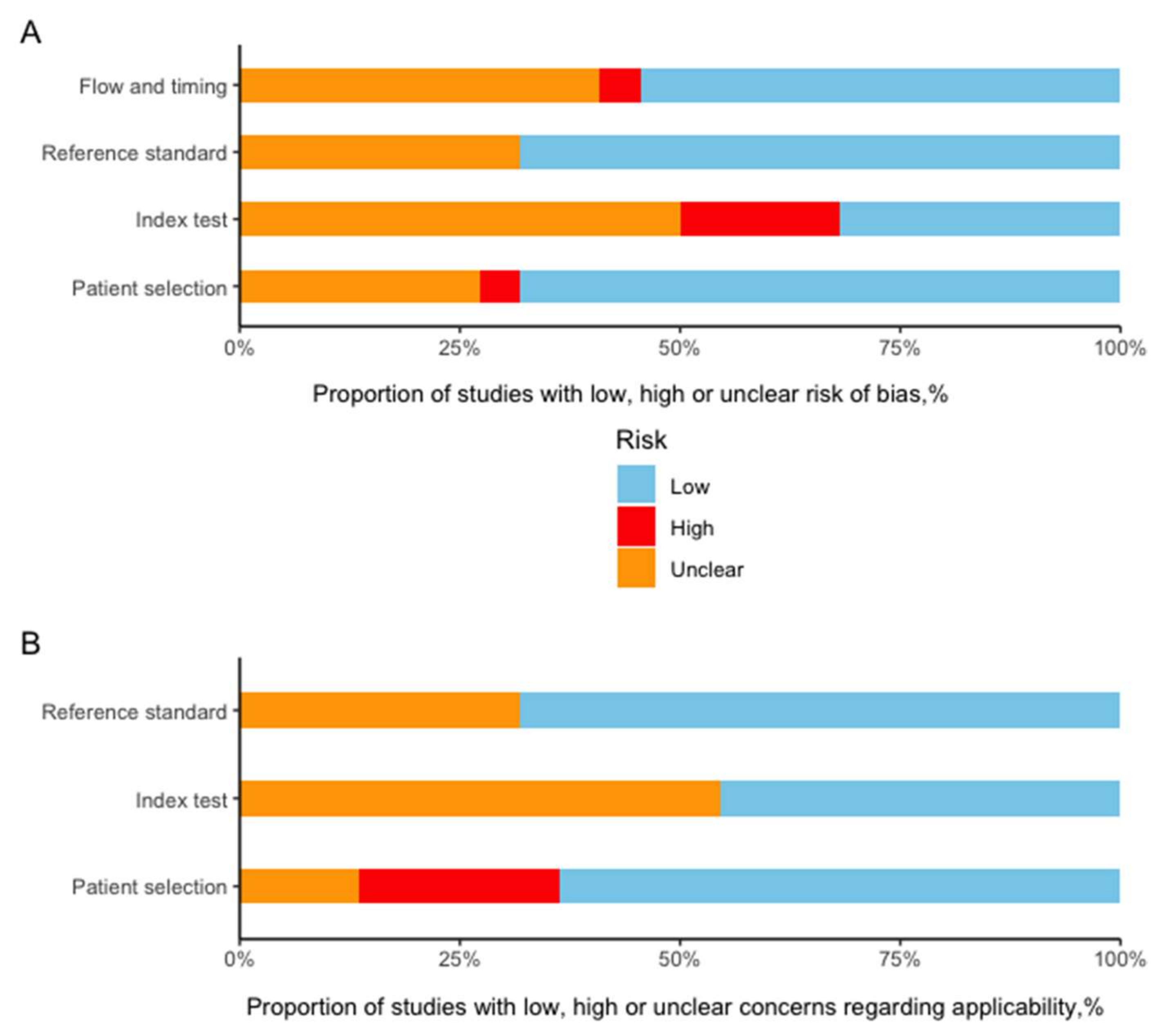
2.3. Assessment of Radiomics Quality Score, Risk of Bias, and Research Type
3. Results
3.1. Literature Selection
3.2. General Characteristics and the Incidence of MVI
3.3. RQS and Risk of Bias Assessment
3.4. Study Characteristics
3.4.1. Imaging Acquisition
3.4.2. Tumor Segmentation
3.4.3. Imaging Preprocessing and Feature Extraction
3.4.4. Feature Selection and Modelling
3.4.5. Performance of the Prediction Model
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sheng, X.; Ji, Y.; Ren, G.P.; Lu, C.L.; Yun, J.P.; Chen, L.H.; Meng, B.; Qu, L.J.; Duan, G.J.; Sun, Q.; et al. A standardized pathological proposal for evaluating microvascular invasion of hepatocellular carcinoma: A multicenter study by LCPGC. Hepatol. Int. 2020, 14, 1034–1047. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.B.; Choi, S.H.; Kim, S.Y.; Shim, J.H.; Lee, S.S.; Byun, J.H.; Park, S.H.; Kim, K.W.; Kim, S.; Lee, N.K. MRI Features for Predicting Microvascular Invasion of Hepatocellular Carcinoma: A Systematic Review and Meta-Analysis. Liver Cancer 2021, 10, 94–106. [Google Scholar] [CrossRef] [PubMed]
- Lei, Z.; Li, J.; Wu, D.; Xia, Y.; Wang, Q.; Si, A.; Wang, K.; Wan, X.; Lau, W.Y.; Wu, M.; et al. Nomogram for Preoperative Estimation of Microvascular Invasion Risk in Hepatitis B Virus-Related Hepatocellular Carcinoma Within the Milan Criteria. JAMA Surg. 2016, 151, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, S.H.; Lee, J.E.; Sinn, D.H.; Park, C.K. Preoperative gadoxetic acid-enhanced MRI for predicting microvascular invasion in patients with single hepatocellular carcinoma. J. Hepatol. 2017, 67, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Pandey, G.; Xu, L.; Chen, W.; Gu, L.; Wu, Y.; Chen, X. The Value of TTPVI in Prediction of Microvascular Invasion in Hepatocellular Carcinoma. Cancer Manag. Res. 2020, 12, 4097–4105. [Google Scholar] [CrossRef] [PubMed]
- Centonze, L.; Di Sandro, S.; Lauterio, A.; De Carlis, R.; Frassoni, S.; Rampoldi, A.; Tuscano, B.; Bagnardi, V.; Vanzulli, A.; De Carlis, L. Surgical Resection vs. Percutaneous Ablation for Single Hepatocellular Carcinoma: Exploring the Impact of Li-RADS Classification on Oncological Outcomes. Cancers 2021, 13, 1617. [Google Scholar] [CrossRef]
- Centonze, L.; Di Sandro, S.; Lauterio, A.; De Carlis, R.; Sgrazzutti, C.; Ciulli, C.; Vella, I.; Vicentin, I.; Incarbone, N.; Bagnardi, V.; et al. A retrospective single-centre analysis of the oncological impact of LI-RADS classification applied to Metroticket 2.0 calculator in liver transplantation: Every nodule matters. Transpl. Int. 2021, 34, 1712–1721. [Google Scholar] [CrossRef]
- Cunha, G.M.; Tamayo-Murillo, D.E.; Fowler, K.J. LI-RADS and transplantation: Challenges and controversies. Abdom Radiol. 2021, 46, 29–42. [Google Scholar] [CrossRef]
- Zhou, H.; Sun, J.; Jiang, T.; Wu, J.; Li, Q.; Zhang, C.; Zhang, Y.; Cao, J.; Sun, Y.; Jiang, Y.; et al. A Nomogram Based on Combining Clinical Features and Contrast Enhanced Ultrasound LI-RADS Improves Prediction of Microvascular Invasion in Hepatocellular Carcinoma. Front. Oncol. 2021, 11, 699290. [Google Scholar] [CrossRef]
- Chen, J.; Zhou, J.; Kuang, S.; Zhang, Y.; Xie, S.; He, B.; Deng, Y.; Yang, H.; Shan, Q.; Wu, J.; et al. Liver Imaging Reporting and Data System Category 5: MRI Predictors of Microvascular Invasion and Recurrence After Hepatectomy for Hepatocellular Carcinoma. AJR Am. J. Roentgenol. 2019, 213, 821–830. [Google Scholar] [CrossRef]
- Yang, L.; Gu, D.; Wei, J.; Yang, C.; Rao, S.; Wang, W.; Chen, C.; Ding, Y.; Tian, J.; Zeng, M. A Radiomics Nomogram for Preoperative Prediction of Microvascular Invasion in Hepatocellular Carcinoma. Liver Cancer 2019, 8, 373–386. [Google Scholar] [CrossRef]
- Lambin, P.; Rios-Velazquez, E.; Leijenaar, R.; Carvalho, S.; van Stiphout, R.G.; Granton, P.; Zegers, C.M.; Gillies, R.; Boellard, R.; Dekker, A.; et al. Radiomics: Extracting more information from medical images using advanced feature analysis. Eur. J. Cancer 2012, 48, 441–446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gillies, R.J.; Kinahan, P.E.; Hricak, H. Radiomics: Images Are More than Pictures, They Are Data. Radiology 2016, 278, 563–577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomaszewski, M.R.; Gillies, R.J. The Biological Meaning of Radiomic Features. Radiology 2021, 299, E256. [Google Scholar] [CrossRef]
- Collins, G.S.; Reitsma, J.B.; Altman, D.G.; Moons, K.G. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): The TRIPOD Statement. BMC Med. 2015, 13, g7594. [Google Scholar] [CrossRef] [Green Version]
- Lambin, P.; Leijenaar, R.T.H.; Deist, T.M.; Peerlings, J.; de Jong, E.E.C.; van Timmeren, J.; Sanduleanu, S.; Larue, R.; Even, A.J.G.; Jochems, A.; et al. Radiomics: The bridge between medical imaging and personalized medicine. Nat. Rev. Clin. Oncol. 2017, 14, 749–762. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.; Hu, Y.; Si, L.; Jia, G.; Xing, Y.; Zhang, H.; Yao, W. A systematic review of radiomics in osteosarcoma: Utilizing radiomics quality score as a tool promoting clinical translation. Eur. Radiol. 2021, 31, 1526–1535. [Google Scholar] [CrossRef]
- Park, J.E.; Kim, D.; Kim, H.S.; Park, S.Y.; Kim, J.Y.; Cho, S.J.; Shin, J.H.; Kim, J.H. Quality of science and reporting of radiomics in oncologic studies: Room for improvement according to radiomics quality score and TRIPOD statement. Eur. Radiol. 2020, 30, 523–536. [Google Scholar] [CrossRef]
- Whiting, P.F.; Rutjes, A.W.; Westwood, M.E.; Mallett, S.; Deeks, J.J.; Reitsma, J.B.; Leeflang, M.M.; Sterne, J.A.; Bossuyt, P.M.; Group, Q. QUADAS-2: A revised tool for the quality assessment of diagnostic accuracy studies. Ann. Intern. Med. 2011, 155, 529–536. [Google Scholar] [CrossRef]
- Zheng, J.; Chakraborty, J.; Chapman, W.C.; Gerst, S.; Gonen, M.; Pak, L.M.; Jarnagin, W.R.; DeMatteo, R.P.; Do, R.K.G.; Simpson, A.L.; et al. Preoperative Prediction of Microvascular Invasion in Hepatocellular Carcinoma Using Quantitative Image Analysis. J. Am. Coll. Surg. 2017, 225, 778–788. [Google Scholar] [CrossRef]
- Peng, J.; Zhang, J.; Zhang, Q.; Xu, Y.; Zhou, J.; Liu, L. A radiomics nomogram for preoperative prediction of microvascular invasion risk in hepatitis B virus-related hepatocellular carcinoma. Diagn. Interv. Radiol. 2018, 24, 121–127. [Google Scholar] [CrossRef] [Green Version]
- Ma, X.; Wei, J.; Gu, D.; Zhu, Y.; Feng, B.; Liang, M.; Wang, S.; Zhao, X.; Tian, J. Preoperative radiomics nomogram for microvascular invasion prediction in hepatocellular carcinoma using contrast-enhanced CT. Eur. Radiol. 2019, 29, 3595–3605. [Google Scholar] [CrossRef]
- Feng, S.T.; Jia, Y.; Liao, B.; Huang, B.; Zhou, Q.; Li, X.; Wei, K.; Chen, L.; Li, B.; Wang, W.; et al. Preoperative prediction of microvascular invasion in hepatocellular cancer: A radiomics model using Gd-EOB-DTPA-enhanced MRI. Eur. Radiol. 2019, 29, 4648–4659. [Google Scholar] [CrossRef]
- Ni, M.; Zhou, X.; Lv, Q.; Li, Z.; Gao, Y.; Tan, Y.; Liu, J.; Liu, F.; Yu, H.; Jiao, L.; et al. Radiomics models for diagnosing microvascular invasion in hepatocellular carcinoma: Which model is the best model? Cancer Imaging 2019, 19, 60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, R.; Xu, L.; Wen, X.; Zhang, J.; Yang, P.; Zhang, L.; Xue, X.; Wang, X.; Huang, Q.; Guo, C.; et al. A nomogram based on bi-regional radiomics features from multimodal magnetic resonance imaging for preoperative prediction of microvascular invasion in hepatocellular carcinoma. Quant. Imaging Med. Surg. 2019, 9, 1503–1515. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.J.; Feng, B.; Wang, S.; Wang, L.M.; Wu, J.F.; Ma, X.H.; Zhao, X.M. Model-based three-dimensional texture analysis of contrast-enhanced magnetic resonance imaging as a potential tool for preoperative prediction of microvascular invasion in hepatocellular carcinoma. Oncol. Lett. 2019, 18, 720–732. [Google Scholar] [CrossRef] [PubMed]
- Nebbia, G.; Zhang, Q.; Arefan, D.; Zhao, X.; Wu, S. Pre-operative Microvascular Invasion Prediction Using Multi-parametric Liver MRI Radiomics. J. Digit. Imaging 2020, 33, 1376–1386. [Google Scholar] [CrossRef]
- Liu, Q.P.; Xu, X.; Zhu, F.P.; Zhang, Y.D.; Liu, X.S. Prediction of prognostic risk factors in hepatocellular carcinoma with transarterial chemoembolization using multi-modal multi-task deep learning. EClinicalMedicine 2020, 23, 100379. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Ruan, S.; Xiao, W.; Shao, J.; Tian, W.; Liu, W.; Zhang, Z.; Wan, D.; Huang, J.; Huang, Q.; et al. Contrast-enhanced CT radiomics for preoperative evaluation of microvascular invasion in hepatocellular carcinoma: A two-center study. Clin. Transl. Med. 2020, 10, e111. [Google Scholar] [CrossRef]
- Jiang, Y.Q.; Cao, S.E.; Cao, S.; Chen, J.N.; Wang, G.Y.; Shi, W.Q.; Deng, Y.N.; Cheng, N.; Ma, K.; Zeng, K.N.; et al. Preoperative identification of microvascular invasion in hepatocellular carcinoma by XGBoost and deep learning. J. Cancer Res. Clin. Oncol. 2021, 147, 821–833. [Google Scholar] [CrossRef]
- He, M.; Zhang, P.; Ma, X.; He, B.; Fang, C.; Jia, F. Radiomic Feature-Based Predictive Model for Microvascular Invasion in Patients With Hepatocellular Carcinoma. Front. Oncol. 2020, 10, 574228. [Google Scholar] [CrossRef]
- Chong, H.H.; Yang, L.; Sheng, R.F.; Yu, Y.L.; Wu, D.J.; Rao, S.X.; Yang, C.; Zeng, M.S. Multi-scale and multi-parametric radiomics of gadoxetate disodium-enhanced MRI predicts microvascular invasion and outcome in patients with solitary hepatocellular carcinoma ≤ 5 cm. Eur. Radiol. 2021, 31, 4824–4838. [Google Scholar] [CrossRef]
- Chen, Y.; Xia, Y.; Tolat, P.P.; Long, L.; Jiang, Z.; Huang, Z.; Tang, Q. Comparison of Conventional Gadoxetate Disodium-Enhanced MRI Features and Radiomics Signatures With Machine Learning for Diagnosing Microvascular Invasion. AJR Am. J. Roentgenol. 2021, 216, 1510–1520. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Y.; Fang, Q.; Zhang, X.; Hou, P.; Wu, H.; Wang, X. Radiomics analysis of [(18)F]FDG PET/CT for microvascular invasion and prognosis prediction in very-early- and early-stage hepatocellular carcinoma. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2599–2614. [Google Scholar] [CrossRef]
- Song, D.; Wang, Y.; Wang, W.; Wang, Y.; Cai, J.; Zhu, K.; Lv, M.; Gao, Q.; Zhou, J.; Fan, J.; et al. Using deep learning to predict microvascular invasion in hepatocellular carcinoma based on dynamic contrast-enhanced MRI combined with clinical parameters. J. Cancer Res. Clin. Oncol. 2021, 147, 3757–3767. [Google Scholar] [CrossRef]
- Dai, H.; Lu, M.; Huang, B.; Tang, M.; Pang, T.; Liao, B.; Cai, H.; Huang, M.; Zhou, Y.; Chen, X.; et al. Considerable effects of imaging sequences, feature extraction, feature selection, and classifiers on radiomics-based prediction of microvascular invasion in hepatocellular carcinoma using magnetic resonance imaging. Quant. Imaging Med. Surg. 2021, 11, 1836–1853. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Tan, X.Z.; Zhang, T.; Gu, Q.B.; Mao, X.H.; Li, Y.C.; He, Y.Q. Prediction of microvascular invasion in solitary hepatocellular carcinoma ≤ 5 cm based on computed tomography radiomics. World J. Gastroenterol. 2021, 27, 2015–2024. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Xu, G.; Duan, C.; Zhou, X.; Wang, X.; Yu, H.; Yu, L.; Li, Z.; Gao, Y.; Zhao, R.; et al. Radiomics Analysis of MR Imaging with Gd-EOB-DTPA for Preoperative Prediction of Microvascular Invasion in Hepatocellular Carcinoma: Investigation and Comparison of Different Hepatobiliary Phase Delay Times. Biomed Res. Int. 2021, 2021, 6685723. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Yang, R.; Liang, F.; Liu, G.; Chen, A.; Wu, H.; Lai, S.; Ding, W.; Wei, X.; Zhen, X.; et al. Prediction of Microvascular Invasion in Hepatocellular Carcinoma With a Multi-Disciplinary Team-Like Radiomics Fusion Model on Dynamic Contrast-Enhanced Computed Tomography. Front. Oncol. 2021, 11, 660629. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.P.; Wang, Y.C.; Zhou, J.Y.; Yu, Q.; Lu, C.Q.; Xia, C.; Tang, T.Y.; Xu, J.; Sun, K.; Xiao, W.; et al. Comparison of MRI and CT for the Prediction of Microvascular Invasion in Solitary Hepatocellular Carcinoma Based on a Non-Radiomics and Radiomics Method: Which Imaging Modality Is Better? J. Magn. Reson. Imaging 2021, 54, 526–536. [Google Scholar] [CrossRef]
- Zhang, Y.; Shu, Z.; Ye, Q.; Chen, J.; Zhong, J.; Jiang, H.; Wu, C.; Yu, T.; Pang, P.; Ma, T.; et al. Preoperative Prediction of Microvascular Invasion in Hepatocellular Carcinoma via Multi-Parametric MRI Radiomics. Front. Oncol. 2021, 11, 633596. [Google Scholar] [CrossRef]
- Wakabayashi, T.; Ouhmich, F.; Gonzalez-Cabrera, C.; Felli, E.; Saviano, A.; Agnus, V.; Savadjiev, P.; Baumert, T.F.; Pessaux, P.; Marescaux, J.; et al. Radiomics in hepatocellular carcinoma: A quantitative review. Hepatol. Int. 2019, 13, 546–559. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Segal, E.; Sirlin, C.B.; Ooi, C.; Adler, A.S.; Gollub, J.; Chen, X.; Chan, B.K.; Matcuk, G.R.; Barry, C.T.; Chang, H.Y.; et al. Decoding global gene expression programs in liver cancer by noninvasive imaging. Nat. Biotechnol. 2007, 25, 675–680. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Wang, D.S.; Kim, H.J.; Sirlin, C.B.; Chan, M.G.; Korn, R.L.; Rutman, A.M.; Siripongsakun, S.; Lu, D.; Imanbayev, G.; et al. A computed tomography radiogenomic biomarker predicts microvascular invasion and clinical outcomes in hepatocellular carcinoma. Hepatology 2015, 62, 792–800. [Google Scholar] [CrossRef]
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riley, R.D.; Ensor, J.; Snell, K.I.E.; Harrell, F.E., Jr.; Martin, G.P.; Reitsma, J.B.; Moons, K.G.M.; Collins, G.; van Smeden, M. Calculating the sample size required for developing a clinical prediction model. BMJ 2020, 368, m441. [Google Scholar] [CrossRef] [Green Version]
- Mongan, J.; Moy, L.; Kahn, C.E., Jr. Checklist for Artificial Intelligence in Medical Imaging (CLAIM): A Guide for Authors and Reviewers. Radiol. Artif. Intell. 2020, 2, e200029. [Google Scholar] [CrossRef] [Green Version]


| First Author | Year | Study Design | No. of Patients (Train vs. Test Cohort) | Independent Validation Cohort | Age (Mean/Median) | Gender (M/F, %) | Indication | MVI Incidence |
|---|---|---|---|---|---|---|---|---|
| Jian Zheng [20] | 2017 | R# | 120 (NA) | No | 70 | 73/27 | HCC | 44% |
| Jie Peng [21] | 2018 | R | 304 (184:120) | No | 53 vs. 55 † | 85/15 | HCC (solitary) | 66% |
| Xiaohong Ma [22] | 2018 | R | 157 (110:47) | No | 53 vs. 55 † | 85/15 | HCC (≤6 cm, solitary) | 35% |
| ShiTing Feng [23] | 2019 | R | 160 (110:50) | No | 54.8 | 91/9 | HCC | 38.8% |
| Ming Ni [24] | 2019 | R | 206 (148:58) | No | 57 vs. 59 † | NA | HCC (>1 cm) | 42.7% |
| Rui Zhang [25] | 2019 | R | 267 (194:73) | No | 57.9 | 86/14 | HCC (solitary) | 33.7% |
| Yong-Jian Zhu [26] | 2019 | R | 142 (99:43) | No | 57 | 87/13 | HCC (<5 cm, solitary) | 37.3% |
| Giacomo Nebbia [27] | 2020 | R | 99 (NA) | No | 51 vs. 54 (MVI vs. non-MVI) | 84/16 | HCC | 61.6% |
| Qiu-ping Liu [28] | 2020 | R | 494 (346:148) | No | NA | 84/16 | HCC | 30.2% |
| Xiuming Zhang [29] | 2020 | R | 637 (451:111) | Yes (75, external) | 57.5 vs. 56.2 vs. 60.7 § | 86/14 | HCC | 40% |
| Yi-quan Jiang [30] | 2020 | R | 405 (324:81) | No | 48.5 | 85/15 | HCC | 54.3% |
| Mu He [31] | 2020 | R | 163 (101:44) | Yes (18, internal) | 50.0 vs. 47.5 vs. 52.0 § | 82/18 | HCC | 67.5% |
| Huan-Huan Chong [32] | 2021 | R | 356 (250:106) | No | 54.2 | 85/15 | HCC (≤5 cm) | 25.3% |
| Yidi Chen [33] | 2021 | R | 269 (188:81) | No | 51.5 | 81/19 | HCC | 41.3% |
| Youcai Li [34] | 2021 | R | 80 (50:30) | No | NA | 91/9 | HCC (BCLC 0/A) | 45% |
| Danjun Song [35] | 2021 | R | 601 (461:140) | No | 56.5 | 82/18 | HCC (solitary) | 37.40% |
| Houjiao Dai [36] | 2021 | R | 69 (LOOCV) | No | 52.7 | 96/4 | HCC (solitary) | 42.0% |
| Peng Liu [37] | 2021 | R | 185 (124:61) | No | 54 vs. 52 † | 84/26 | HCC (≤5 cm, solitary) | 34.1% |
| Shuai Zhang [38] | 2021 | R | 130 (91:39) | No | 57.8 vs. 58.6 † | 68/32 | HCC (>1 cm) | 61.5% |
| Wanli Zhang [39] | 2021 | R | 111 (88:23) | No | NA | 88/12 | HCC | 51.4% |
| Xiang-pan Meng [40] | 2021 | R | 402 (300:102) | No | 57 vs. 57 † | 85/15 | HCC (solitary) | 40% |
| Yang Zhang [41] | 2021 | R | 195 (136:59) | No | 57.7 | 88/12 | HCC (≤5 cm) | 56.4% |
| Study ID | Imaging Modality | Phase/Sequence | Segmentation | Extension of VOI | Feature Selection | Number of Imaging Features Included in the Model | Event/Feature Ratio during Model Development | Clinical Variables for Modeling | AUC in Test Cohort |
|---|---|---|---|---|---|---|---|---|---|
| Zheng 2017 | CE-CT | PVP | 2D; auto | Yes (5-pixel) | univariable logistic regression | 21 | 1.6 | AFP, tumor size, hepatitis | NA |
| Peng 2018 | CE-CT | AP, PVP, DP | 3D; semi-auto | No | LASSO | 8 | 15.9 | AFP, non-smooth tumor margin, internal arteries, hypoattenuating halos | 0.84 |
| Ma 2018 | CE-CT | AP, PVP, DP | 3D; manually | No | ICC/CCC, LASSO | 7 | 5.3 | Age, tumor size, hepatitis B | 0.80 |
| Feng 2019 | Gd-EOB-DTPA MRI | HBP | 3D; manually | Yes (10 mm) | LASSO | 10 | 4.2 | NA | 0.84 |
| Ni 2019 | CE-CT | PVP | 2D; manually | No | LASSO, neighbourhood rough set, PCA | Unclear | NA | NA | NA |
| R. Zhang 2019 | Multimodel MRI | AP, PVP, EP, T1, T2, DWI | 3D; manually | Yes (10 mm) | mRMR | 12 | 5.3 | AFP, arterial peritumoral enhancement | 0.86 |
| Zhu 2019 | CE-MRI | AP, PVP | 3D; manually | No | Kruskal-Wallis test; Pearson correlation | 4 | 9.3 | AFP, tumor size, differentiation | 0.79 |
| Nebbia 2020 | CE-MRI | AP, PVP, T1, T2, DWI | 3D; manually | Yes (10 pixel) | LASSO | 17 | 3.6 | NA | NA |
| Q. Liu 2020 | CE-CT | AP, PVP | 3D;semiauto | No | ICC, RF | 28 | 5.3 | NA | 0.79 |
| X. Zhang 2020 | CE-CT | DP | 3D; manually | Yes (10 mm) | LASSO | 44 | 4.0 | Age, AFP | 0.80 (0.80 in the validation cohort) |
| Jiang 2020 | CE-CT | AP, PVP, DP | 3D; manually | Yes (10 mm) | Xgboost/3D-CNN | Unclear | NA | AFP | 0.91 |
| He 2020 | CE-CT | PVP | 3D; auto | No | LASSO | 2 | 35.5 | AFP, neutrophilic granulocytes, hemoglobin | 0.71 (0.84 in the validation cohort) |
| Chong 2021 | Gd-EOB-DTPA MRI | AP, PVP, HBP, DWI | 3D; manually | Yes (5 mm,10 mm, 50%) | ICC, LASSO, Univariate Feature Selection | 74 | 0.9 | AFP, TBIL, capsule enhancement, peritumoral enhancement | 0.92 |
| Chen 2021 | Gd-EOB-DTPA MRI | AP, PVP, HBP, T1, T2, DWI | 3D; manually | No | LASSO | 21 | NA | NA | 0.94 |
| Li 2021 | PET/CT | [18F]FDG PET/CT | 3D; semiauto | No | LASSO | 11 | 0.7 | NA | 0.69 |
| Song 2021 | CE-MRI | AP, PVP, DP, T1,T2, ADC, DWI | 3D; manually | No | PCA, analysis of variance | Unclear | NA | NA | 0.73 |
| Dai 2021 | Gd-EOB-DTPA MRI | AP, PVP, HBP, T1 | 3D; manually | No | LASSO-RFE, LASSO, mRMR, SVM-RFE | 5 | 5.8 | NA | 0.90 (LOOCV) |
| P. Liu 2021 | CE-CT | AP | 3D; manually | No | ICC, LASSO | 10 | 4.2 | NA | 0.75 |
| Sh. Zhang 2021 | Gd-EOB-DTPA MRI | HBP (5, 10, 15 min) | 3D; manually | No | LASSO | 14 | NA | NA | NA |
| W. Zhang 2021 | CE-CT | EAP, LAP, PVP, EP | 3D; manually | 2, 4, 6, 8, 10, 12, 14 mm | 15 methods (Fisher score, t score, etc.) | Unclear | NA | NA | 0.81 |
| Meng 2021 | Multiparametric MRI & multiphasic CT | AP, PVP, T2, DWI (MRI)/AP, PVP (CT) | 3D; manually | Yes (3 mm) | ICC, univariate analysis, feature reduction, LASSO | 16/16 | 8.1/1.9 # | NA | 0.80 |
| Y. Zhang 2021 | Multiparametric MRI | AP, PVP, DP, T2, DWI, ADC | 3D; manually | No | ICC, analysis of variance, Mann-Whitney U-test, correlation analysis, LASSO | 47 | 1.6 | Age, AFP, tumor size | 0.84 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Q.; Li, C.; Zhang, J.; Hu, X.; Fan, Y.; Ma, K.; Sparrelid, E.; Brismar, T.B. Radiomics Models for Predicting Microvascular Invasion in Hepatocellular Carcinoma: A Systematic Review and Radiomics Quality Score Assessment. Cancers 2021, 13, 5864. https://doi.org/10.3390/cancers13225864
Wang Q, Li C, Zhang J, Hu X, Fan Y, Ma K, Sparrelid E, Brismar TB. Radiomics Models for Predicting Microvascular Invasion in Hepatocellular Carcinoma: A Systematic Review and Radiomics Quality Score Assessment. Cancers. 2021; 13(22):5864. https://doi.org/10.3390/cancers13225864
Chicago/Turabian StyleWang, Qiang, Changfeng Li, Jiaxing Zhang, Xiaojun Hu, Yingfang Fan, Kuansheng Ma, Ernesto Sparrelid, and Torkel B. Brismar. 2021. "Radiomics Models for Predicting Microvascular Invasion in Hepatocellular Carcinoma: A Systematic Review and Radiomics Quality Score Assessment" Cancers 13, no. 22: 5864. https://doi.org/10.3390/cancers13225864
APA StyleWang, Q., Li, C., Zhang, J., Hu, X., Fan, Y., Ma, K., Sparrelid, E., & Brismar, T. B. (2021). Radiomics Models for Predicting Microvascular Invasion in Hepatocellular Carcinoma: A Systematic Review and Radiomics Quality Score Assessment. Cancers, 13(22), 5864. https://doi.org/10.3390/cancers13225864






