Dual-Wavelength Fluorescence Monitoring of Photodynamic Therapy: From Analytical Models to Clinical Studies
Abstract
:Simple Summary
Abstract
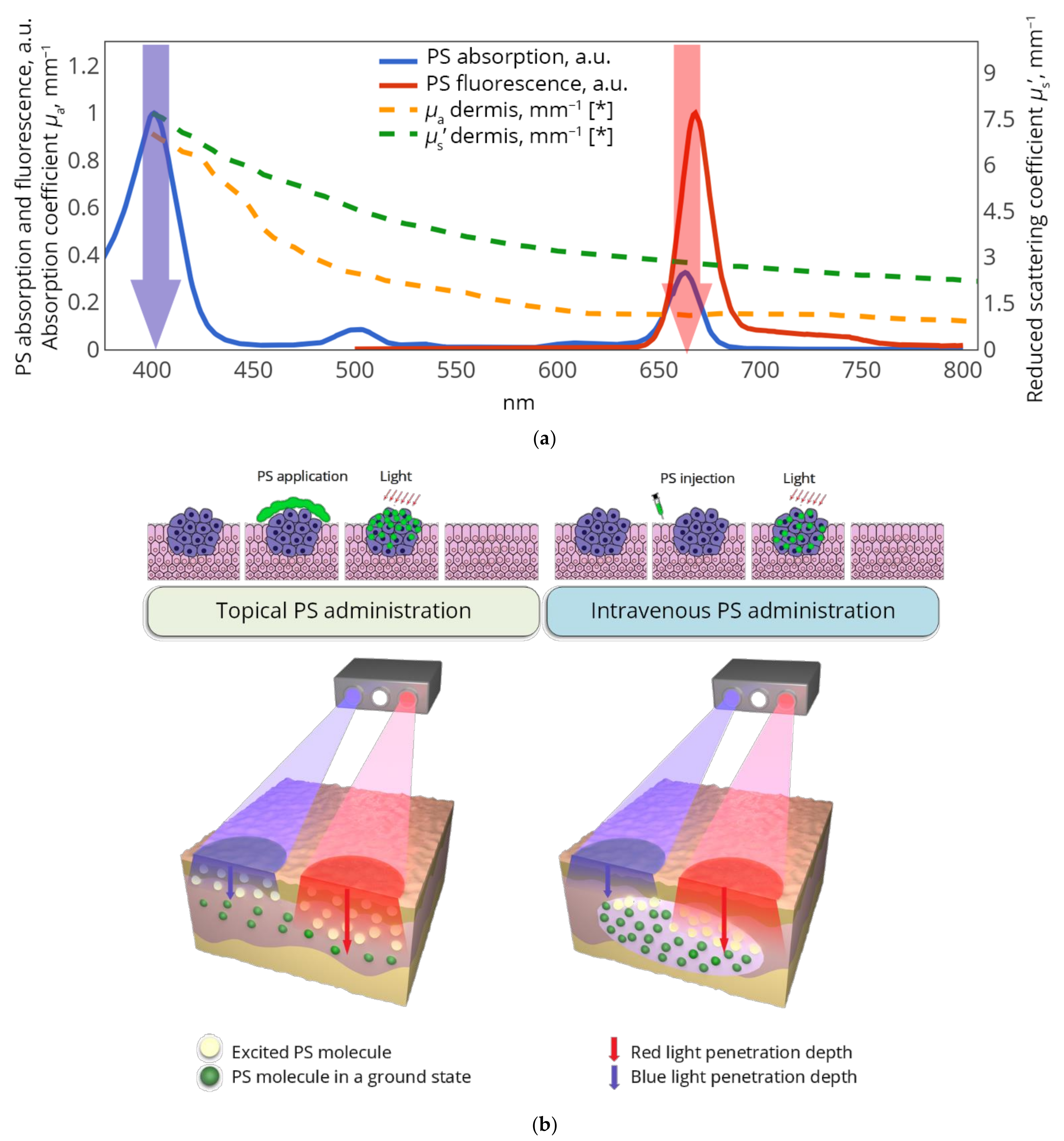
1. Introduction
2. Materials and Methods
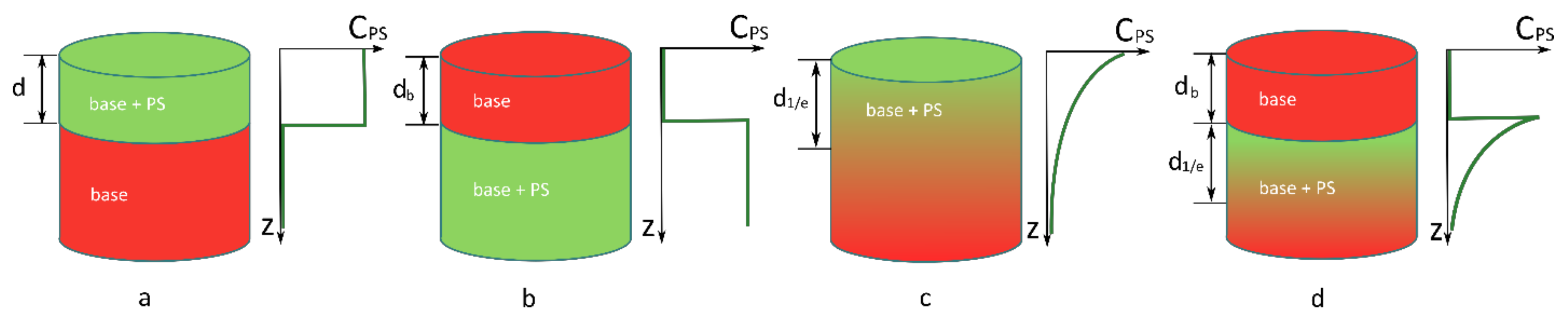
2.1. Analytical Model of Dual-Wavelength Fluorescence Sensing in Biotissues with Different Distributions of Fluorophores
2.1.1. PS Is Distributed Uniformly in the Upper Layer of a Biotissue of Thickness d
2.1.2. PS Is Distributed Uniformly within the Semispace below the Biotissue Layer of Thickness db
2.1.3. PS Concentration Exponentially Decreases In-depth with the 1/e Decay Scale d1/e
2.1.4. A Layer with an Exponentially Decaying PS Concentration with the 1/e Scale Is Located below the PS-Free Biotissue Layer of Thickness
2.1.5. Estimation of the PS Depth from the Ratio
2.2. Dual-Wavelength Fluorescence Imaging Setup
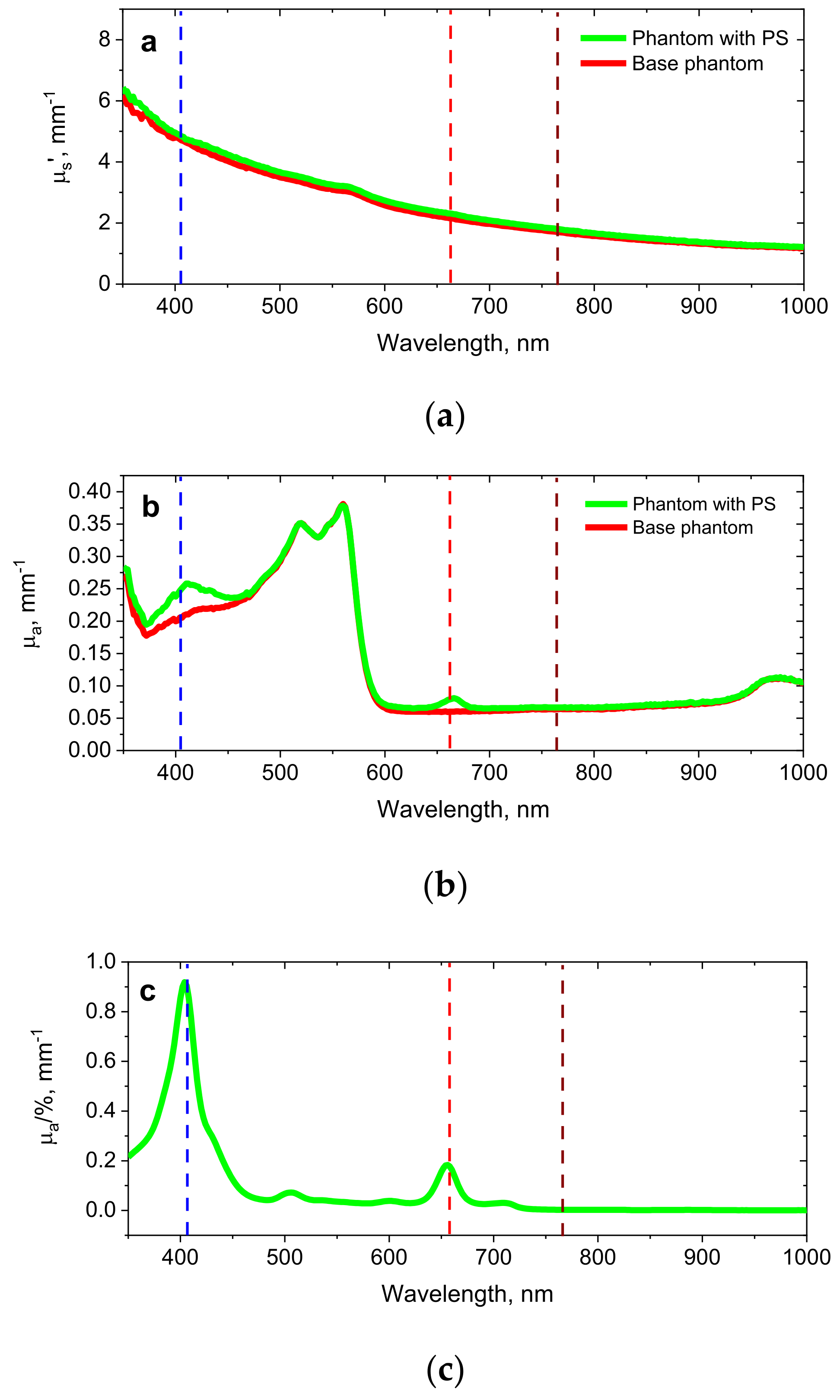
2.3. Model Experiment on Biotissue Phantoms
2.4. Monte Carlo Simulations
2.5. Photosensitizers
2.6. Animal Studies
2.6.1. Intact Tissue Study
2.6.2. Tumor Study
2.7. In Vivo Experiment on Volunteers
2.8. Clinical Studies
2.8.1. Actinic Keratosis Study
2.8.2. Basal Cell Carcinoma Study
3. Results
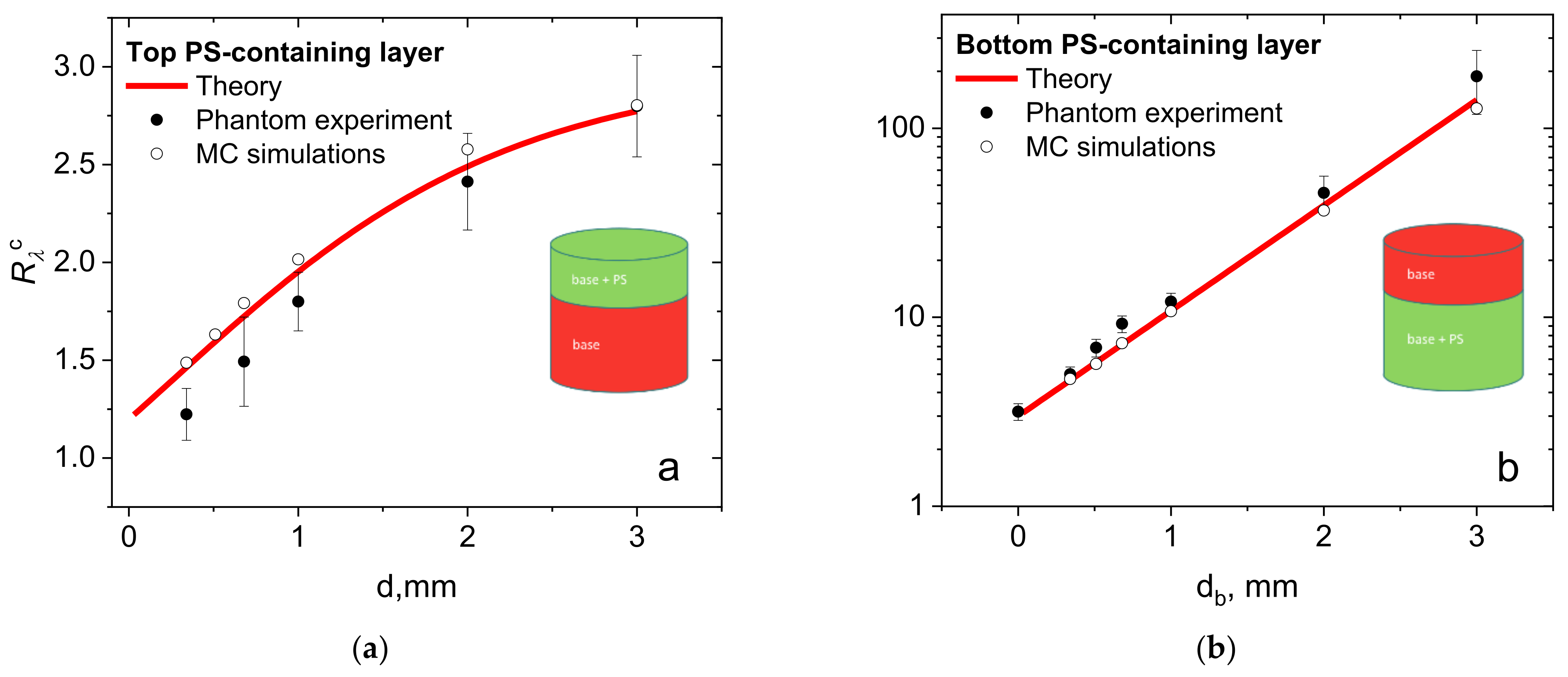
3.1. Comparison of the Analytical Model and Numerical Simulations
3.2. Phantom Experiments
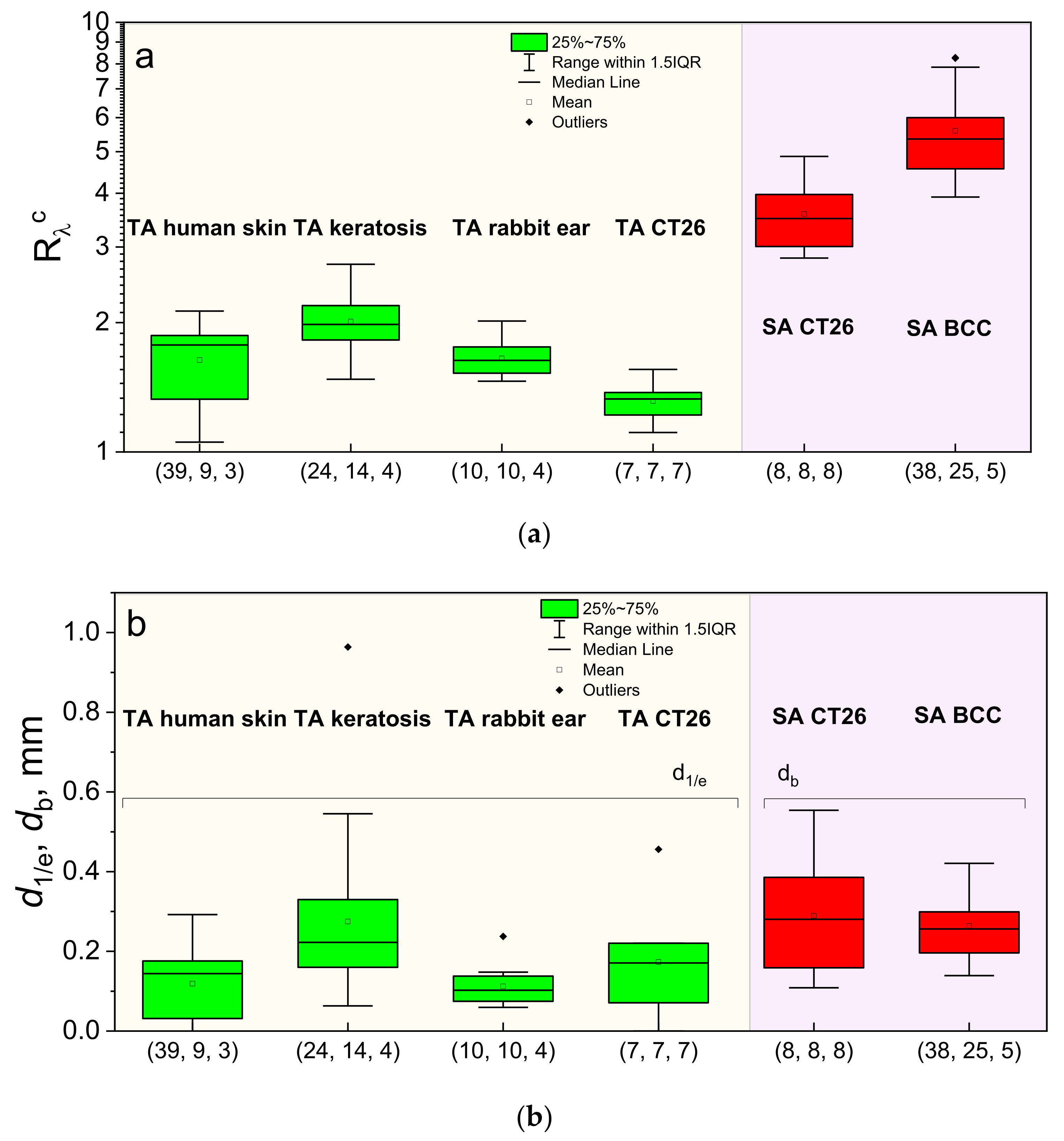
3.3. Reconstruction of the Fluorophore Localization Depth from Dual-Wavelength Measurements
3.4. In Vivo Estimations of the PS Accumulation Depth
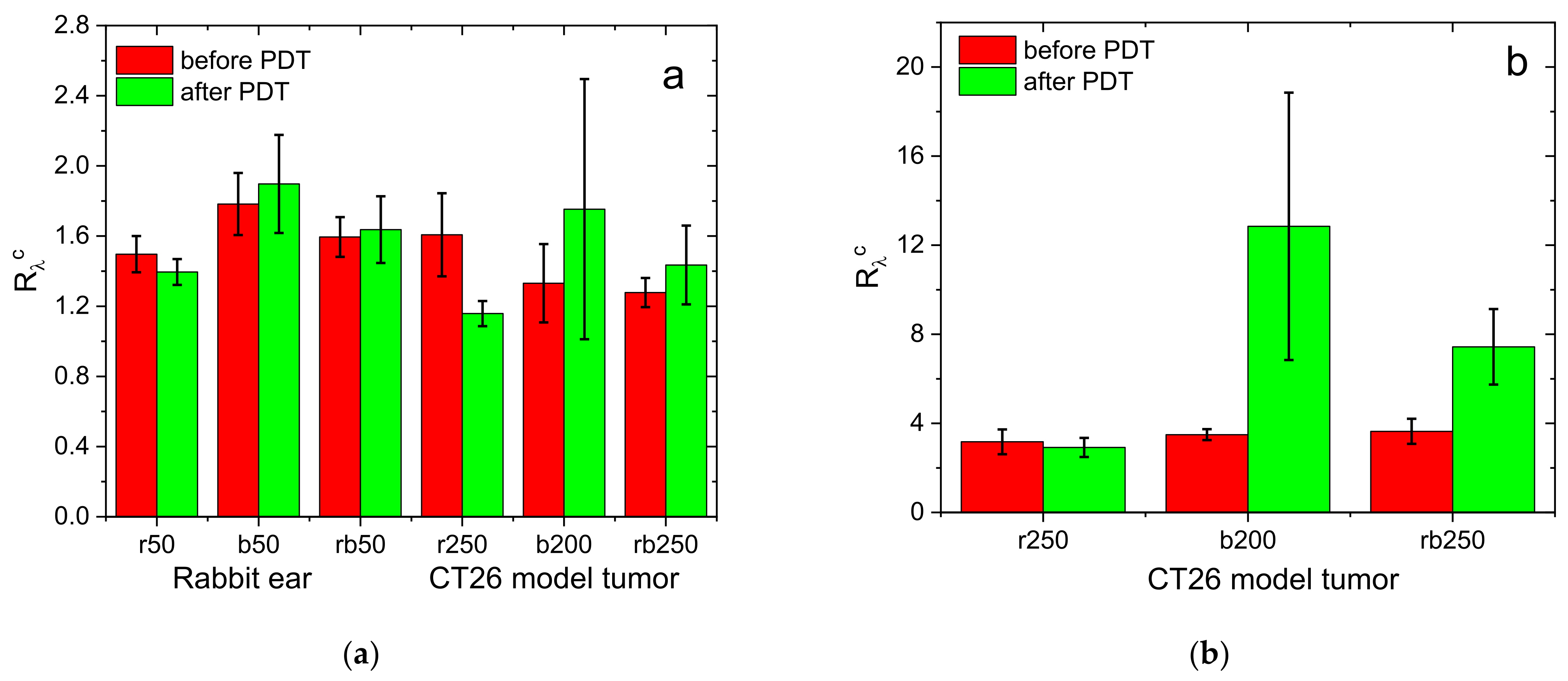
3.5. Monitoring of PDT in Animal Studies
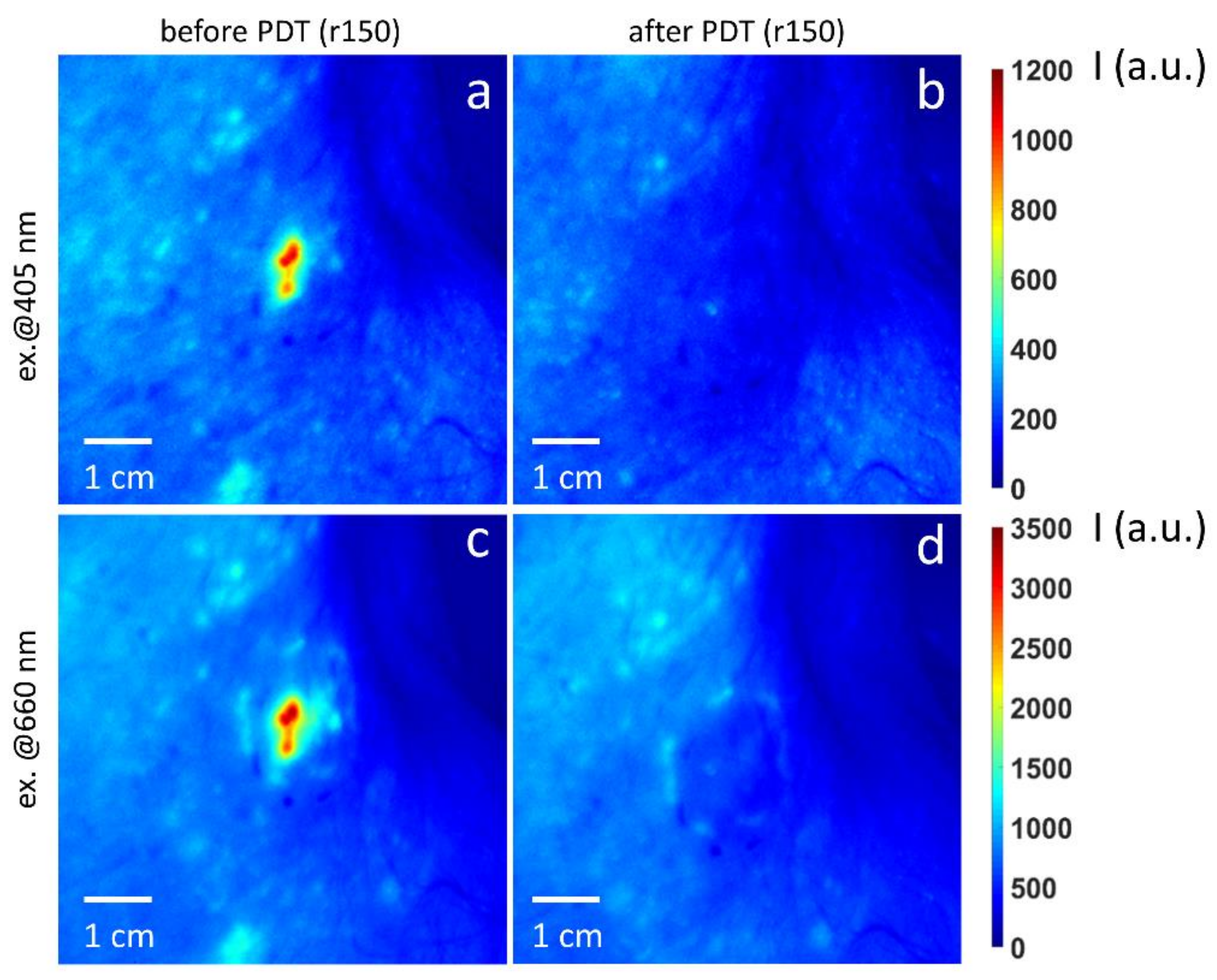
3.6. Clinical PDT Monitoring
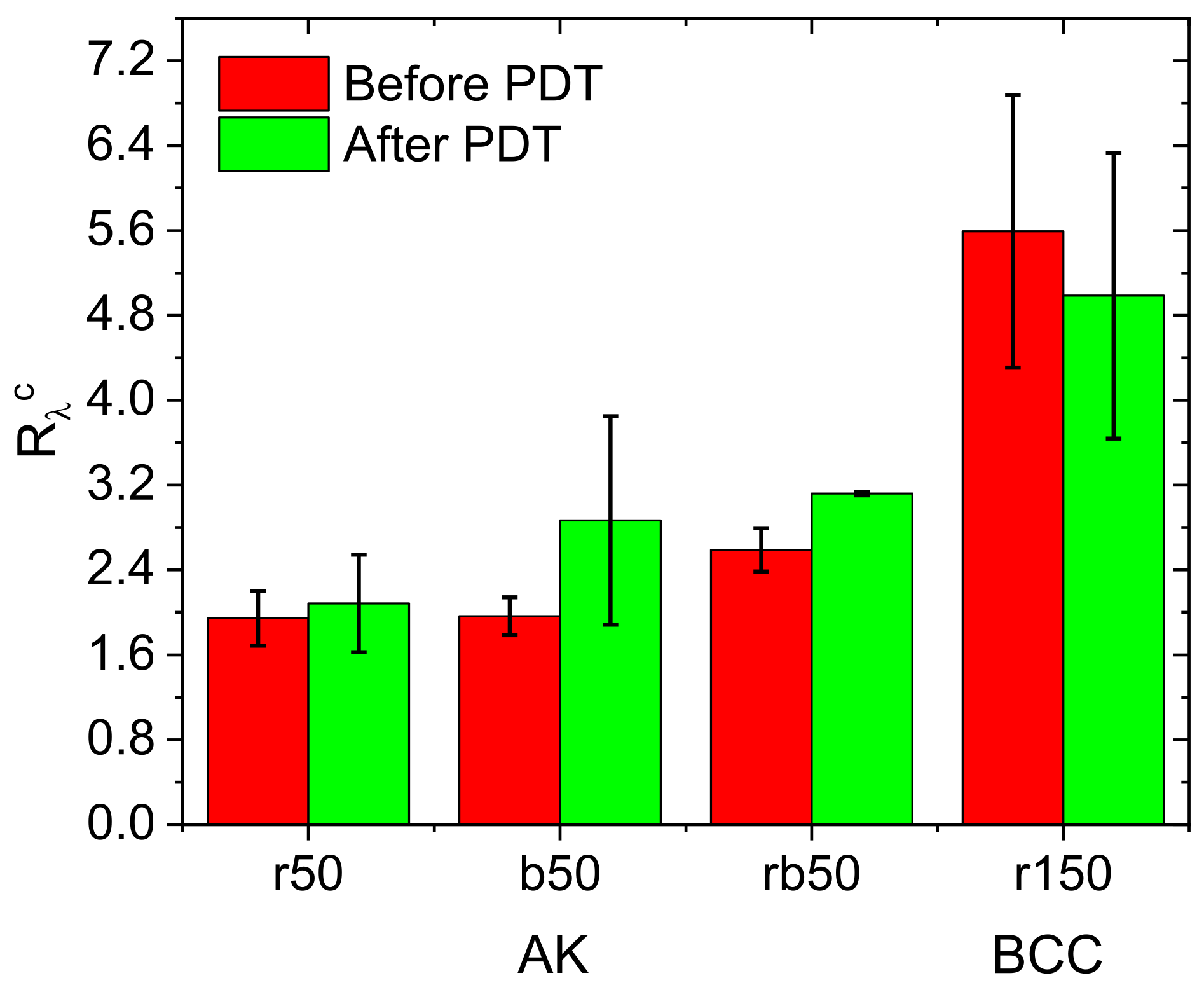
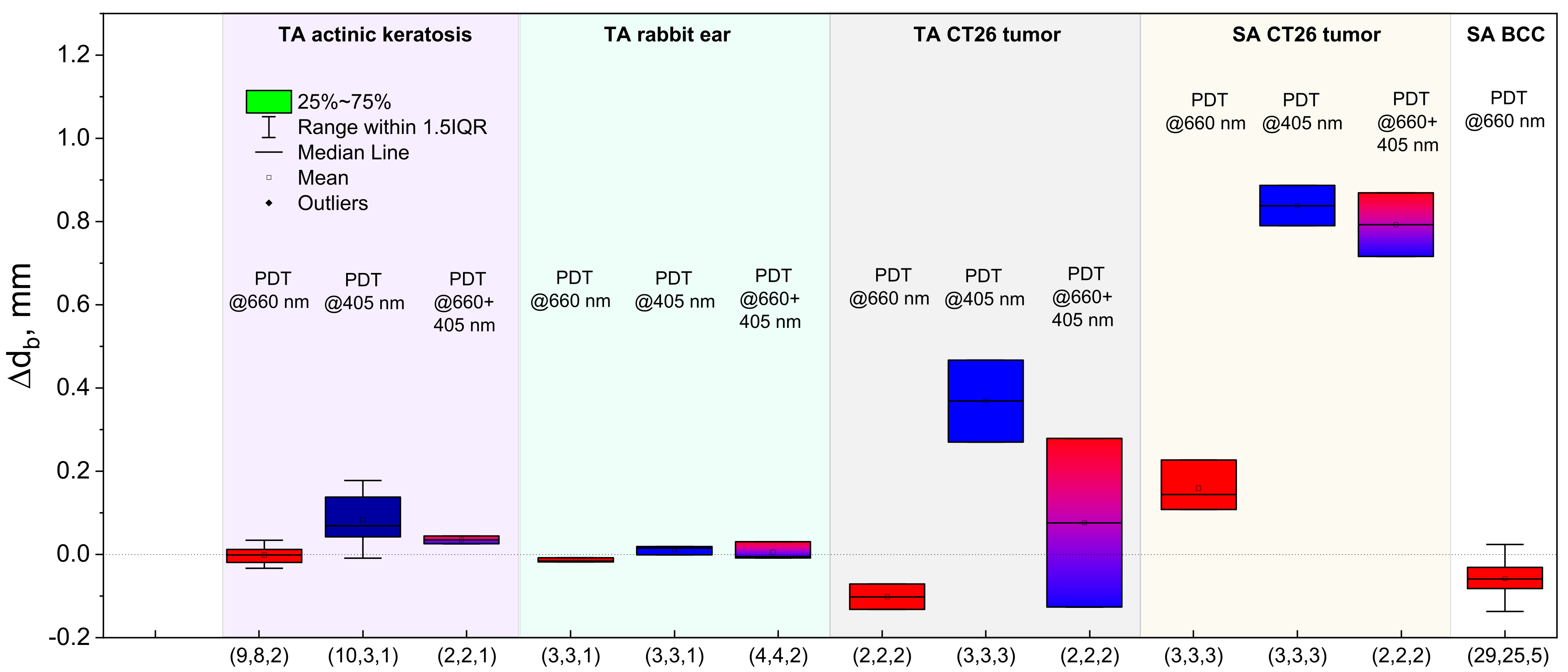
3.7. Estimation of the PS Localization Depth Variation as a Result of a PDT Procedure
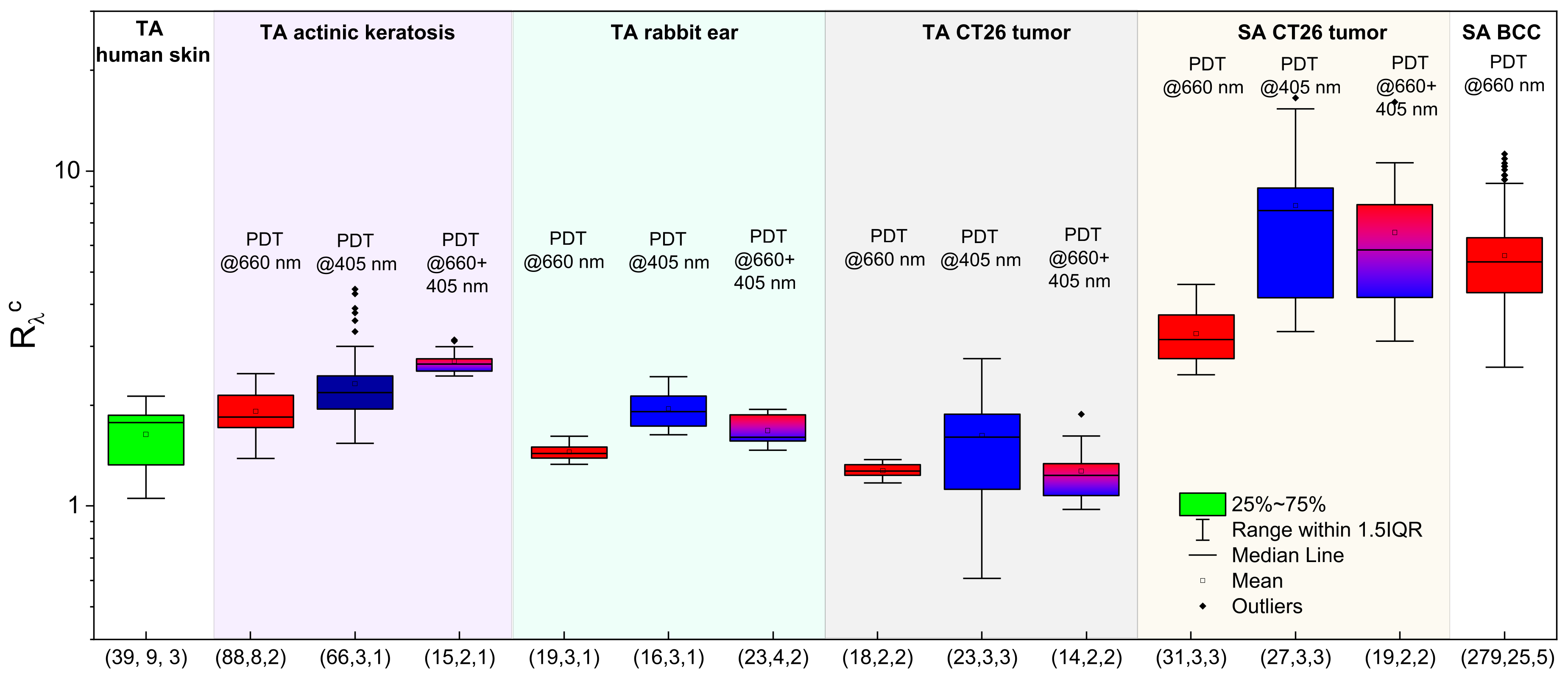
3.8. Fluorescence Signal Ratio: Overview
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Andersson-Engels, S.; af Klinteberg, C.; Svanberg, K.; Svanberg, S. In vivo fluorescence imaging for tissue diagnostics. Phys. Med. Biol. 1997, 42, 815. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frangioni, J.V. In vivo near-infrared fluorescence imaging. Curr. Opin. Chem. Biol. 2003, 7, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Yuan, Y.; Chen, J.; Zhang, B.; Li, D.; Zhou, D.; Jing, P.; Xu, G.; Wang, Y.; Holá, K. In vivo theranostics with near-infrared-emitting carbon dots—highly efficient photothermal therapy based on passive targeting after intravenous administration. Light Sci. Appl. 2018, 7, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, S.; Song, J.; Qu, J.; Cheng, Z. Crucial breakthrough of second near-infrared biological window fluorophores: Design and synthesis toward multimodal imaging and theranostics. Chem. Soc. Rev. 2018, 47, 4258–4278. [Google Scholar] [CrossRef]
- Celli, J.P.; Spring, B.Q.; Rizvi, I.; Evans, C.L.; Samkoe, K.S.; Verma, S.; Pogue, B.W.; Hasan, T. Imaging and photodynamic therapy: Mechanisms, monitoring, and optimization. Chem. Rev. 2010, 110, 2795–2838. [Google Scholar] [CrossRef] [Green Version]
- Akopov, A.L.; Rusanov, A.A.; Papayan, G.V.; Kazakov, N.V.; Gerasin, A.V.; Urtenova, M.A. Endobronchial photodynamic therapy under fluorescence control: Photodynamic theranostics. Photodiagn. Photodyn. Ther. 2017, 19, 73–77. [Google Scholar] [CrossRef]
- Weston, M.A.; Patterson, M.S. Monitoring oxygen concentration during photodynamic therapy using prompt photosensitizer fluorescence. Phys. Med. Biol. 2013, 58, 7039. [Google Scholar] [CrossRef] [Green Version]
- Sheng, C.; Jack Hoopes, P.; Hasan, T.; Pogue, B.W. Photobleaching-based dosimetry predicts deposited dose in ALA-PPIX PDT of rodent esophagus. Photochem. Photobiol. 2007, 83, 738–748. [Google Scholar] [CrossRef]
- Jacques, S.L.; Joseph, R.; Gofstein, G. How photobleaching affects dosimetry and fluorescence monitoring of PDT in turbid media. In Optical Methods for Tumor Treatment and Detection: Mechanisms and Techniques in Photodynamic Therapy II; International Society for Optics and Photonics: Bellingham, WA, USA, 1993; pp. 168–179. [Google Scholar] [CrossRef]
- Warren, C.B.; Lohser, S.; Wene, L.C.; Pogue, B.W.; Bailin, P.; Maytin, E. Noninvasive fluorescence monitoring of protoporphyrin ix production and clinical outcomes in actinic keratoses following short-contact application of 5-aminolevulinate. J. Biomed. Opt. 2010, 15, 051607. [Google Scholar] [CrossRef]
- Piffaretti, F.; Zellweger, M.; Kasraee, B.; Barge, J.; Salomon, D.; Van Den Bergh, H.; Wagnières, G. Correlation between protoporphyrin IX fluorescence intensity, photobleaching, pain and clinical outcome of actinic keratosis treated by photodynamic therapy. Dermatology 2013, 227, 214–225. [Google Scholar] [CrossRef]
- Gamayunov, S.; Turchin, I.; Fiks, I.; Korchagina, K.; Kleshnin, M.; Shakhova, N. Fluorescence imaging for photodynamic therapy of non-melanoma skin malignancies–a retrospective clinical study. Photonics Lasers Med. 2016, 5, 101–111. [Google Scholar] [CrossRef]
- Corlu, A.; Choe, R.; Durduran, T.; Rosen, M.A.; Schweiger, M.; Arridge, S.R.; Schnall, M.D.; Yodh, A.G. Three-dimensional in vivo fluorescence diffuse optical tomography of breast cancer in humans. Opt. Express 2007, 15, 6696–6716. [Google Scholar] [CrossRef]
- Ntziachristos, V. Fluorescence molecular imaging. Annu. Rev. Biomed. Eng. 2006, 8, 1–33. [Google Scholar] [CrossRef] [Green Version]
- van den Berg, N.S.; Buckle, T.; KleinJan, G.H.; van der Poel, H.G.; van Leeuwen, F.W. Multispectral fluorescence imaging during robot-assisted laparoscopic sentinel node biopsy: A first step towards a fluorescence-based anatomic roadmap. Eur. Urol. 2017, 72, 110–117. [Google Scholar] [CrossRef]
- Pu, H.; He, W.; Zhang, G.; Zhang, B.; Liu, F.; Zhang, Y.; Luo, J.; Bai, J. Separating structures of different fluorophore concentrations by principal component analysis on multispectral excitation-resolved fluorescence tomography images. Biomed. Opt. Express 2013, 4, 1829–1845. [Google Scholar] [CrossRef] [Green Version]
- Kleshnin, M.; Turchin, I. Fluorescence diffuse tomography technique with autofluorescence removal based on dispersion of biotissue optical properties. Laser Phys. Lett. 2013, 10, 075601. [Google Scholar] [CrossRef]
- Swartling, J.; Svensson, J.; Bengtsson, D.; Terike, K.; Andersson-Engels, S. Fluorescence spectra provide information on the depth of fluorescent lesions in tissue. Appl. Opt. 2005, 44, 1934–1941. [Google Scholar] [CrossRef] [Green Version]
- Miller, J.P.; Maji, D.; Lam, J.; Tromberg, B.J.; Achilefu, S. Noninvasive depth estimation using tissue optical properties and a dual-wavelength fluorescent molecular probe in vivo. Biomed. Opt. Express 2017, 8, 3095–3109. [Google Scholar] [CrossRef] [Green Version]
- Khilov, A.; Kirillin, M.Y.; Loginova, D.; Turchin, I. Estimation of chlorin-based photosensitizer penetration depth prior to photodynamic therapy procedure with dual-wavelength fluorescence imaging. Laser Phys. Lett. 2018, 15, 126202. [Google Scholar] [CrossRef]
- Khilov, A.V.; Kurakina, D.; Turchin, I.V.; Kirillin, M.Y. Monitoring of chlorin-based photosensitiser localisation with dual-wavelength fluorescence imaging: Numerical simulations. Quantum Electron. 2019, 49, 63. [Google Scholar] [CrossRef]
- Khilov, A.V.; Sergeeva, E.; Kurakina, D.; Turchin, I.V.; Kirillin, M.Y. Analytical model of fluorescence intensity for the estimation of fluorophore localisation in biotissue with dual-wavelength fluorescence imaging. Quantum Electron. 2021, 51, 95–103. [Google Scholar] [CrossRef]
- Salomatina, E.V.; Jiang, B.; Novak, J.; Yaroslavsky, A.N. Optical properties of normal and cancerous human skin in the visible and near-infrared spectral range. J. Biomed. Opt. 2006, 11, 064026. [Google Scholar] [CrossRef]
- Kirillin, M.; Kurakina, D.; Khilov, A.; Orlova, A.; Shakhova, M.; Orlinskaya, N.; Sergeeva, E. Red and blue light in antitumor photodynamic therapy with chlorin-based photosensitizers: A comparative animal study assisted by optical imaging modalities. Biomed. Opt. Express 2021, 12, 872–892. [Google Scholar] [CrossRef]
- Jacques, S.L. How tissue optics affect dosimetry of photodynamic therapy. J. Biomed. Opt. 2010, 15, 051608. [Google Scholar] [CrossRef] [Green Version]
- Haskell, R.C.; Svaasand, L.O.; Tsay, T.-T.; Feng, T.-C.; McAdams, M.S.; Tromberg, B.J. Boundary conditions for the diffusion equation in radiative transfer. JOSA A 1994, 11, 2727–2741. [Google Scholar] [CrossRef] [Green Version]
- Kim, A.D.; Ishimaru, A. Optical diffusion of continuous-wave, pulsed, and density waves in scattering media and comparisons with radiative transfer. Appl. Opt. 1998, 37, 5313–5319. [Google Scholar] [CrossRef]
- Svaasand, L.O.; Wyss, P.; Wyss, M.T.; Tadir, Y.; Tromberg, B.J.; Berns, M.W. Dosimetry model for photodynamic therapy with topically administered photosensitizers. Lasers Surg. Med. Off. J. Am. Soc. Laser Med. Surg. 1996, 18, 139–149. [Google Scholar] [CrossRef]
- Kleshnin, M.; Fiks, I.; Plekhanov, V.; Gamayunov, S.; Turchin, I. Compact and fully automated system for monitoring photodynamic therapy, based on two LEDs and a single CCD. Laser Phys. Lett. 2015, 12, 115602. [Google Scholar] [CrossRef]
- Shakhova, M.; Loginova, D.; Meller, A.; Sapunov, D.; Orlinskaya, N.; Shakhov, A.; Khilov, A.; Kirillin, M. Photodynamic therapy with chlorin-based photosensitizer at 405 nm: Numerical, morphological, and clinical study. J. Biomed. Opt. 2018, 23, 091412. [Google Scholar] [CrossRef]
- Wang, L.; Jacques, S.L.; Zheng, L. MCML—Monte Carlo modeling of light transport in multi-layered tissues. Comput. Methods Programs Biomed. 1995, 47, 131–146. [Google Scholar] [CrossRef]
- Svaasand, L.; Tromberg, B.; Wyss, P.; Wyss-Desserich, M.-T.; Tadir, Y.; Berns, M. Light and drug distribution with topically administered photosensitizers. Lasers Med. Sci. 1996, 11, 261–265. [Google Scholar] [CrossRef]
- Kamuhabwa, A.R.; Roelandts, R.; de Witte, P.A. Skin photosensitization with topical hypericin in hairless mice. J. Photochem. Photobiol. B: Biol. 1999, 53, 110–114. [Google Scholar] [CrossRef]
- Gallardo-Villagrán, M.; Leger, D.Y.; Liagre, B.; Therrien, B. Photosensitizers used in the photodynamic therapy of rheumatoid arthritis. Int. J. Mol. Sci. 2019, 20, 3339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calixto, G.M.F.; Bernegossi, J.; De Freitas, L.M.; Fontana, C.R.; Chorilli, M. Nanotechnology-based drug delivery systems for photodynamic therapy of cancer: A review. Molecules 2016, 21, 342. [Google Scholar] [CrossRef]
- Ali-Seyed, M.; Bhuvaneswari, R.; Soo, K.C.; Olivo, M. Photolon™-photosensitization induces apoptosis via ROS-mediated cross-talk between mitochondria and lysosomes. Int. J. Oncol. 2011, 39, 821–831. [Google Scholar] [CrossRef]
- Istomin, Y.P.; Kaplan, M.A.; Shliakhtsin, S.V.; Lapzevich, T.P.; Cerkovsky, D.A.; Marchanka, L.N.; Fedulov, A.S.; Trukhachova, T.V. Immediate and long-term efficacy and safety of photodynamic therapy with Photolon (Fotolon):A seven-year clinical experience. Proc. SPIE 2009, 7380, 73806V. [Google Scholar] [CrossRef]
- Trukhachova, T. Safety and efficacy of photosensitizer Photolon (Fotolon) in photodynamic therapy. Proc. SPIE 2019, 11070, 1107037. [Google Scholar] [CrossRef]
- Correa, J.; Bagnato, V.; Imasato, H.; Perussi, J. Previous illumination of a water soluble chlorine photosensitizer increases its cytotoxicity. Laser Phys. 2012, 22, 1387–1394. [Google Scholar] [CrossRef]
- Kurakina, D.; Khilov, A.; Shakhova, M.; Orlinskaya, N.; Sergeeva, E.; Meller, A.; Turchin, I.; Kirillin, M. Comparative analysis of single- and dual-wavelength photodynamic therapy regimes with chlorin-based photosensitizers: Animal study. J. Biomed. Opt. 2019, 25, 063804. [Google Scholar] [CrossRef] [Green Version]
- Shliakhtsin, S.; Trukhachova, T.; Isakau, H.; Istomin, Y. Pharmacokinetics and biodistribution of Photolon®(Fotolon®) in intact and tumor-bearing rats. Photodiagn. Photodyn. Ther. 2009, 6, 97–104. [Google Scholar] [CrossRef]
- Paul, S.; Heng, P.W.S.; Chan, L.W. Optimization in solvent selection for chlorin e6 in photodynamic therapy. J. Fluoresc. 2013, 23, 283–291. [Google Scholar] [CrossRef]
- Isakau, H.; Parkhats, M.; Knyukshto, V.; Dzhagarov, B.; Petrov, E.; Petrov, P. Toward understanding the high PDT efficacy of chlorin e6–polyvinylpyrrolidone formulations: Photophysical and molecular aspects of photosensitizer–polymer interaction in vitro. J. Photochem. Photobiol. B Biol. 2008, 92, 165–174. [Google Scholar] [CrossRef]
- Mesradi, M.; Genoux, A.; Cuplov, V.; Abi-Haidar, D.; Jan, S.; Buvat, I.; Pain, F. Experimental and analytical comparative study of optical coefficient of fresh and frozen rat tissues. J. Biomed. Opt. 2013, 18, 117010. [Google Scholar] [CrossRef] [Green Version]
- Wei, J.C.; Edwards, G.A.; Martin, D.J.; Huang, H.; Crichton, M.L.; Kendall, M.A. Allometric scaling of skin thickness, elasticity, viscoelasticity to mass for micro-medical device translation: From mice, rats, rabbits, pigs to humans. Sci. Rep. 2017, 7, 1–16. [Google Scholar] [CrossRef]
- Tan, J.; Lambie, D.; Sinnya, S.; Sahebian, A.; Soyer, H.; Prow, T.; Ardigò, M. Histopathology and reflectance confocal microscopy features of photodamaged skin and actinic keratosis. J. Eur. Acad. Dermatol. Venereol. 2016, 30, 1901–1911. [Google Scholar] [CrossRef]
- Heerfordt, I.M.; Nissen, C.V.; Poulsen, T.; Philipsen, P.A.; Wulf, H.C. Thickness of actinic keratosis does not predict dysplasia severity or p53 expression. Sci. Rep. 2016, 6, 1–6. [Google Scholar] [CrossRef]
- Fernández-Figueras, M.; Saenz-Sardà, X.; Vargas, P.; Thompson, C.; Carrato, C.; Puig, L.; Ferrándiz, C.; Ariza, A. The depth of follicular extension in actinic keratosis correlates with the depth of invasion in squamous cell carcinoma: Implication for clinical treatment. J. Eur. Acad. Dermatol. Venereol. 2018, 32, 1657–1661. [Google Scholar] [CrossRef]
- Sabino, C.P.; Deana, A.M.; Yoshimura, T.M.; da Silva, D.F.; França, C.M.; Hamblin, M.R.; Ribeiro, M.S. The optical properties of mouse skin in the visible and near infrared spectral regions. J. Photochem. Photobiol. B Biol. 2016, 160, 72–78. [Google Scholar] [CrossRef] [Green Version]
- Ntziachristos, V.; Ripoll, J.; Wang, L.V.; Weissleder, R. Looking and listening to light: The evolution of whole-body photonic imaging. Nat. Biotechnol. 2005, 23, 313–320. [Google Scholar] [CrossRef]
- Davis, S.C.; Tichauer, K.M. Small-animal imaging using diffuse fluorescence tomography. In In Vivo Fluorescence Imaging; Springer: Berlin/Heidelberg, Germany, 2016; pp. 123–137. [Google Scholar] [CrossRef]
- Nagaya, T.; Nakamura, Y.A.; Choyke, P.L.; Kobayashi, H. Fluorescence-guided surgery. Front. Oncol. 2017, 7, 314. [Google Scholar] [CrossRef]
- Hurley, B.R.; Regillo, C.D. Fluorescein angiography: General principles and interpretation. In Retinal Angiography and Optical Coherence Tomography; Springer: Berlin/Heidelberg, Germany, 2009; pp. 27–42. [Google Scholar] [CrossRef]
- Hamblin, M.R.; Huang, Y. Imaging in Photodynamic Therapy; CRC Press: Boca Raton, FL, USA, 2017; pp. 27–42. [Google Scholar]
- Fukuhara, H.; Yamamoto, S.; Karashima, T.; Inoue, K. Photodynamic diagnosis and therapy for urothelial carcinoma and prostate cancer: New imaging technology and therapy. Int. J. Clin. Oncol. 2021, 26, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Leblond, F.; Davis, S.C.; Valdés, P.A.; Pogue, B.W. Pre-clinical whole-body fluorescence imaging: Review of instruments, methods and applications. J. Photochem. Photobiol. B Biol. 2010, 98, 77–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kosaka, N.; McCann, T.E.; Mitsunaga, M.; Choyke, P.L.; Kobayashi, H. Real-time optical imaging using quantum dot and related nanocrystals. Nanomedicine 2010, 5, 765–776. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lavaud, J.; Henry, M.; Gayet, P.; Fertin, A.; Vollaire, J.; Usson, Y.; Coll, J.-L.; Josserand, V. Noninvasive monitoring of liver metastasis development via combined multispectral photoacoustic imaging and fluorescence diffuse optical tomography. Int. J. Biol. Sci. 2020, 16, 1616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, C.T.; Svenmarker, P.; Liu, H.; Wu, X.; Messing, M.E.; Wallenberg, L.R.; Andersson-Engels, S. High-resolution fluorescence diffuse optical tomography developed with nonlinear upconverting nanoparticles. ACS Nano 2012, 6, 4788–4795. [Google Scholar] [CrossRef]
- Zhu, Q.; Dehghani, H.; Tichauer, K.; Holt, R.; Vishwanath, K.; Leblond, F.; Pogue, B. A three-dimensional finite element model and image reconstruction algorithm for time-domain fluorescence imaging in highly scattering media. Phys. Med. Biol. 2011, 56, 7419. [Google Scholar] [CrossRef] [Green Version]
- Deliolanis, N.C.; Dunham, J.; Wurdinger, T.; Figueiredo, J.-L.; Bakhos, T.; Ntziachristos, V. In-vivo imaging of murine tumors using complete-angle projection fluorescence molecular tomography. J. Biomed. Opt. 2009, 14, 030509. [Google Scholar] [CrossRef] [Green Version]
- Fiks, I.I.; Turchin, I.V. Reconstruction of fluorophore concentration distribution in diffuse fluorescence tomography based on Tikhonov regularisation and nonnegativity condition. Quantum Electron. 2021, 51, 400. [Google Scholar] [CrossRef]
- Ong, Y.H.; Kim, M.M.; Zhu, T.C. Photodynamic therapy explicit dosimetry. In Photodynamic Therapy Explicit Dosimetry; Nova Science Publishers: Hauppauge, NY, USA, 2018; pp. 45–72. [Google Scholar]
- Zhu, W.; Gao, Y.-H.; Song, C.-H.; Lu, Z.-B.; Namulinda, T.; Han, Y.-P.; Yan, Y.-J.; Wang, L.-X.; Chen, Z.-L. Synthesis and evaluation of new 5-aminolevulinic acid derivatives as prodrugs of protoporphyrin for photodynamic therapy. Photochem. Photobiol. Sci. 2017, 16, 1623–1630. [Google Scholar] [CrossRef]
- Chen, H.; Yang, Z.; Zou, X.; Wang, J.; Zhu, J.; Gu, Y. Retinal injury thresholds for 532, 578, and 630 nm lasers in connection to photodynamic therapy for choroidal neovascularization. Lasers Surg. Med. 2014, 46, 54–60. [Google Scholar] [CrossRef]


| λ(nm) | μa PS, mm−1 | M0 | μa BT, mm−1 | μs BT, mm−1 | gBT | μs‘BT, mm−1 |
|---|---|---|---|---|---|---|
| 405 | 0.1 | 0.02 | 0.96 | 38 | 0.8 | 7.6 |
| 660 | 0.02 | 0.004 | 0.15 | 14 | 0.8 | 2.8 |
| 760 | 0 | 0 | 0.13 | 12 | 0.8 | 2.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kirillin, M.; Khilov, A.; Kurakina, D.; Orlova, A.; Perekatova, V.; Shishkova, V.; Malygina, A.; Mironycheva, A.; Shlivko, I.; Gamayunov, S.; et al. Dual-Wavelength Fluorescence Monitoring of Photodynamic Therapy: From Analytical Models to Clinical Studies. Cancers 2021, 13, 5807. https://doi.org/10.3390/cancers13225807
Kirillin M, Khilov A, Kurakina D, Orlova A, Perekatova V, Shishkova V, Malygina A, Mironycheva A, Shlivko I, Gamayunov S, et al. Dual-Wavelength Fluorescence Monitoring of Photodynamic Therapy: From Analytical Models to Clinical Studies. Cancers. 2021; 13(22):5807. https://doi.org/10.3390/cancers13225807
Chicago/Turabian StyleKirillin, Mikhail, Aleksandr Khilov, Daria Kurakina, Anna Orlova, Valeriya Perekatova, Veronika Shishkova, Alfia Malygina, Anna Mironycheva, Irena Shlivko, Sergey Gamayunov, and et al. 2021. "Dual-Wavelength Fluorescence Monitoring of Photodynamic Therapy: From Analytical Models to Clinical Studies" Cancers 13, no. 22: 5807. https://doi.org/10.3390/cancers13225807
APA StyleKirillin, M., Khilov, A., Kurakina, D., Orlova, A., Perekatova, V., Shishkova, V., Malygina, A., Mironycheva, A., Shlivko, I., Gamayunov, S., Turchin, I., & Sergeeva, E. (2021). Dual-Wavelength Fluorescence Monitoring of Photodynamic Therapy: From Analytical Models to Clinical Studies. Cancers, 13(22), 5807. https://doi.org/10.3390/cancers13225807







