Relationship between Serum Bortezomib Concentration and Emergence of Diarrhea in Patients with Multiple Myeloma and/or AL Amyloidosis
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design, Ethics and Patients
2.2. Outcomes Measures
2.3. Development of Serum Bortezomib Concentration Measurement Method
2.4. Statistical Analysis
3. Results
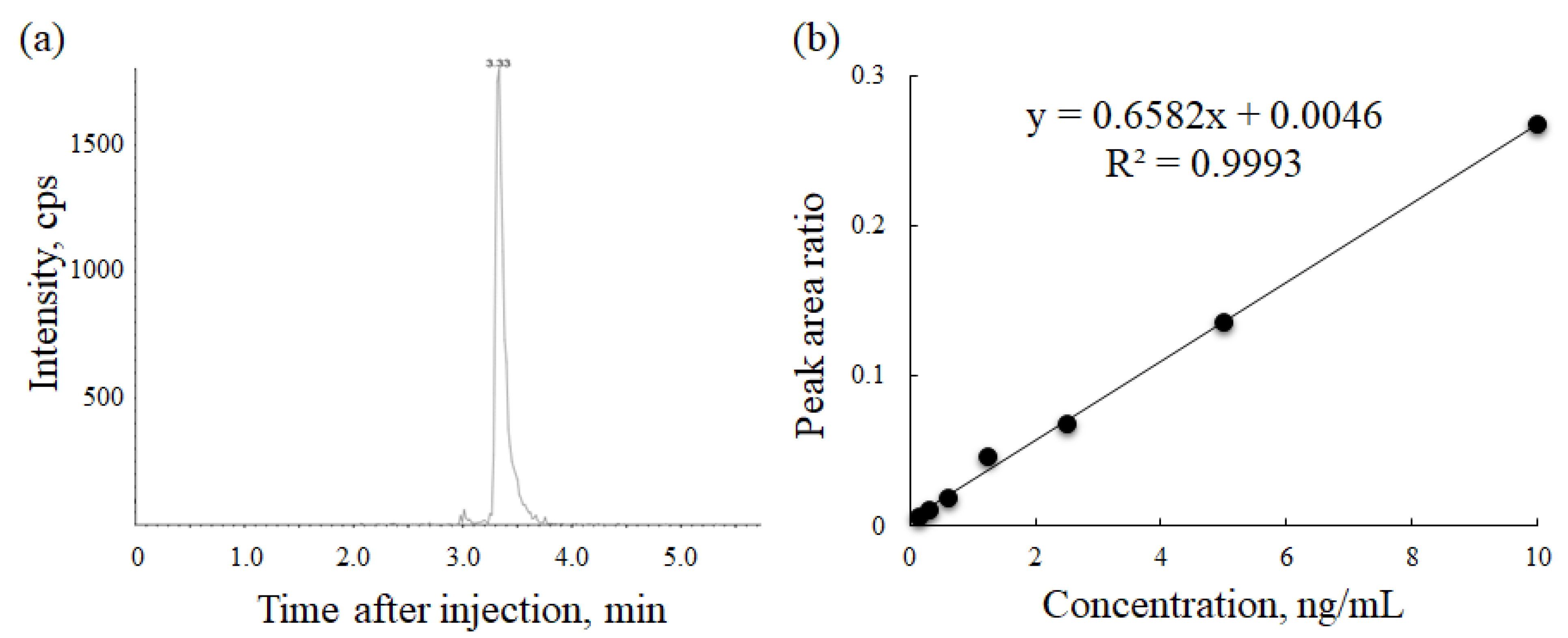
3.1. Development of Serum Bortezomib Concentration Measurement Method
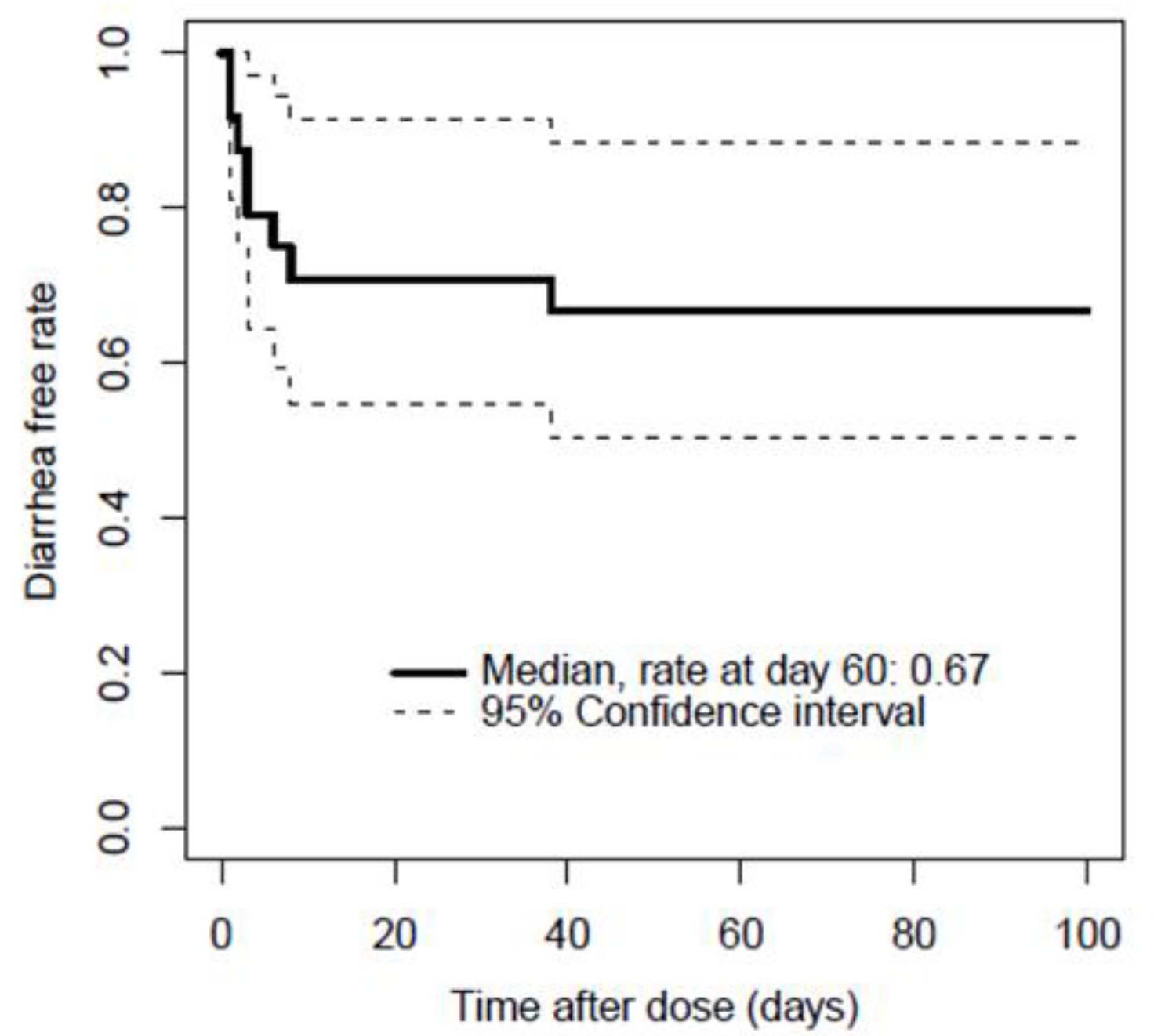
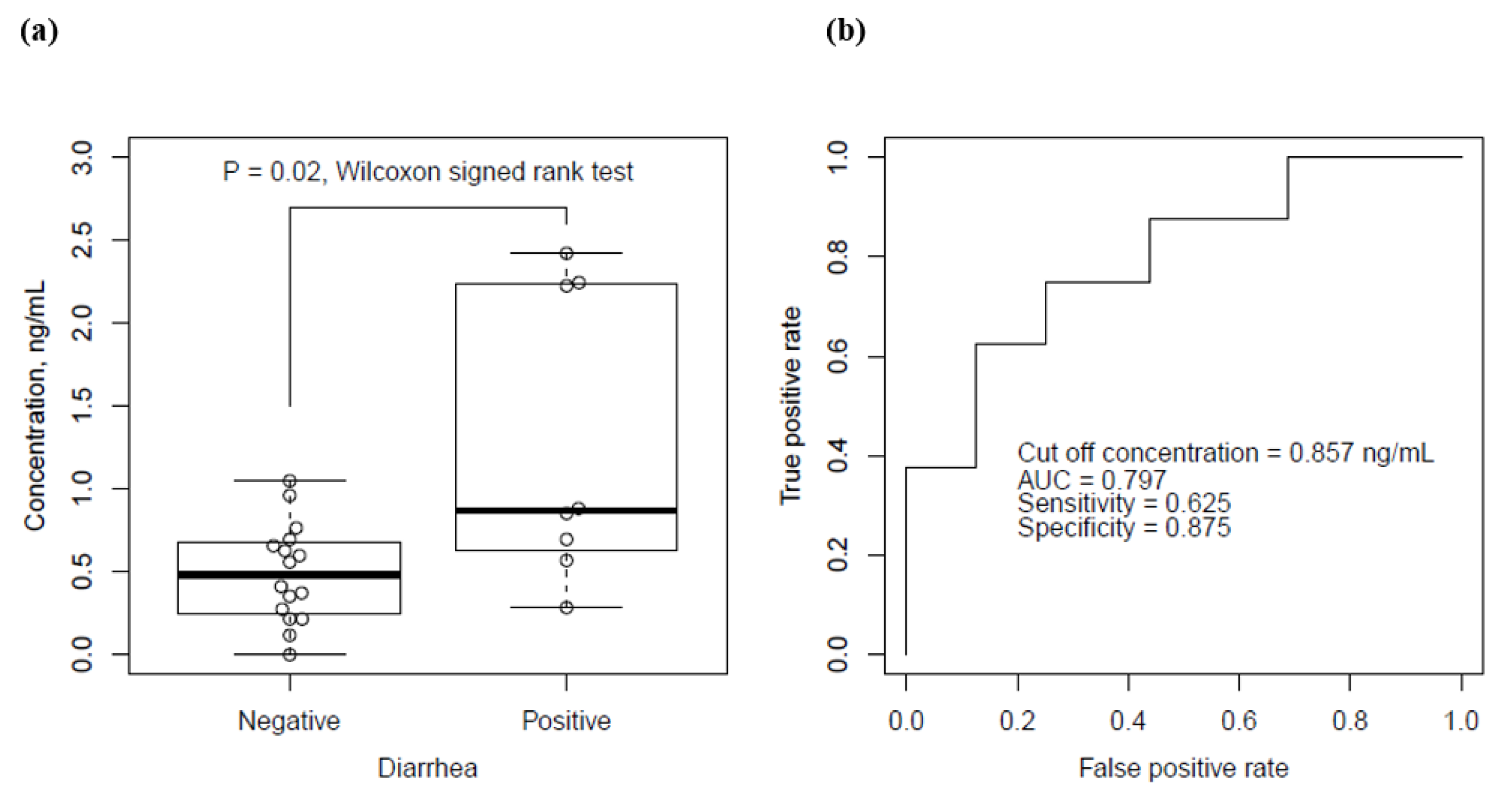
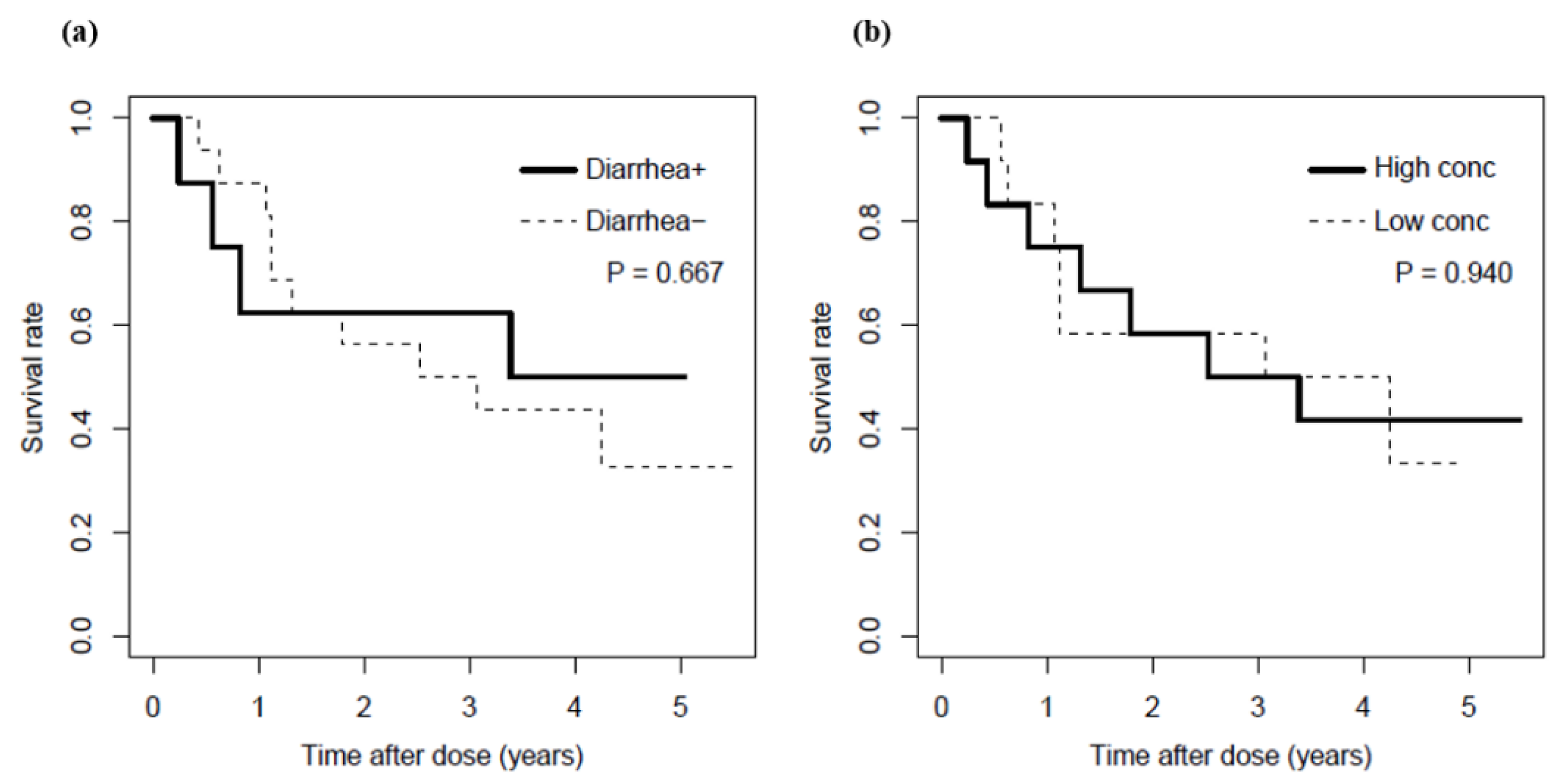
3.2. Relationship between Serum Bortezomib Concentration and Emergence of Diarrhea
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rajkumar, S.V.; Dimopoulos, M.A.; Palumbo, A.; Blade, J.; Merlini, G.; Mateos, M.-V.; Kumar, S.; Hillengass, J.; Kastritis, E.; Richardson, P.; et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014, 15, e538–e548. [Google Scholar] [CrossRef]
- Rajkumar, S.V. Multiple myeloma: Every year a new standard? Hematol. Oncol. 2019, 37 (Suppl. S1), 62–65. [Google Scholar] [CrossRef]
- Kazandjian, D. Multiple myeloma epidemiology and survival: A unique malignancy. Semin. Oncol. 2016, 43, 676–681. [Google Scholar] [CrossRef]
- Scott, K.; Hayden, P.J.; Will, A.; Wheatley, K.; Coyne, I. Bortezomib for the treatment of multiple myeloma. Cochrane Database Syst. Rev. 2016, 4, CD010816. [Google Scholar] [CrossRef]
- Kumar, S.K.; Rajkumar, S.V.; Dispenzieri, A.; Lacy, M.Q.; Hayman, S.R.; Buadi, F.K.; Zeldenrust, S.R.; Dingli, D.; Russell, S.J.; Lust, J.A.; et al. Improved survival in multiple myeloma and the impact of novel therapies. Blood 2008, 111, 2516–2520. [Google Scholar] [CrossRef]
- Singhal, S.; Mehta, J.; Desikan, R.; Ayers, D.; Roberson, P.; Eddlemon, P.; Munshi, N.; Anaissie, E.; Wilson, C.; Dhodapkar, M.; et al. Antitumor Activity of Thalidomide in Refractory Multiple Myeloma. N. Engl. J. Med. 1999, 341, 1565–1571. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, S.V.; Gupta, V.; Fonseca, R.; Dispenzieri, A.; Gonsalves, W.I.; Larson, D.; Ketterling, R.; Lust, J.A.; Kyle, R.A.; Kumar, S.K. Impact of primary molecular cytogenetic abnormalities and risk of progression in smoldering multiple myeloma. Leukemia 2013, 27, 1738–1744. [Google Scholar] [CrossRef] [PubMed]
- Sonneveld, P.; Goldschmidt, H.; Rosiñol, L.; Bladé, J.; Lahuerta, J.J.; Cavo, M.; Tacchetti, P.; Zamagni, E.; Attal, M.; Lokhorst, H.M.; et al. Bortezomib-Based versus Nonbortezomib-Based Induction Treatment before Autologous Stem-Cell Transplantation in Patients with Previously Untreated Multiple Myeloma: A Meta-Analysis of Phase III Randomized, Controlled Trials. J. Clin. Oncol. 2013, 31, 3279–3287. [Google Scholar] [CrossRef]
- Wang, J.; Huang, J.; Zeng, Q. Network meta-analysis of targeted therapies for diffuse large B cell lymphoma. BMC Cancer 2020, 20, 1218. [Google Scholar] [CrossRef]
- Kumar, S.K.; Harrison, S.J.; Cavo, M.; de la Rubia, J.; Popat, R.; Gasparetto, C.; Hungria, V.; Salwender, H.; Suzuki, K.; Kim, I.; et al. Venetoclax or placebo in com-bination with bortezomib and dexamethasone in patients with relapsed or refractory multiple myeloma (BELLINI): A ran-domised, double-blind, multicentre, phase 3 trial. Cells 2020, 21, 1630–1642. [Google Scholar]
- Seval, G.C.; Beksac, M. The safety of bortezomib for the treatment of multiple myeloma. Expert. Opin. Drug Saf. 2018, 17, 953–962. [Google Scholar] [CrossRef]
- Stansborough, R.; Gibson, R. Proteasome inhibitor-induced gastrointestinal toxicity. Curr. Opin. Support. Palliat. Care 2017, 11, 133–137. [Google Scholar] [CrossRef]
- Richardson, P.G.; Barlogie, B.; Berenson, J.; Singhal, S.; Jagannath, S.; Irwin, D.; Rajkumar, S.V.; Srkalovic, G.; Alsina, M.; Alexanian, R.; et al. A Phase 2 Study of Bortezomib in Relapsed, Refractory Myeloma. N. Engl. J. Med. 2003, 348, 2609–2617. [Google Scholar] [CrossRef] [PubMed]
- Kane, R.C.; Bross, P.F.; Farrell, A.T.; Pazdur, R. Velcade®: U.S. FDA Approval for the Treatment of Multiple Myeloma Progressing on Prior Therapy. Oncology 2003, 8, 508–513. [Google Scholar] [CrossRef]
- Orlowski, R.Z.; Stinchcombe, T.E.; Mitchell, B.S.; Shea, T.C.; Baldwin, A.S.; Stahl, S.; Adams, J.; Esseltine, D.-L.; Elliott, P.J.; Pien, C.S.; et al. Phase I Trial of the Proteasome Inhibitor PS-341 in Patients with Refractory Hematologic Malignancies. J. Clin. Oncol. 2002, 20, 4420–4427. [Google Scholar] [CrossRef]
- Adams, J.; Palombella, V.J.; Sausville, E.A.; Johnson, J.; Destree, A.; Lazarus, D.D.; Maas, J.; Pien, C.S.; Prakash, S.; Elliott, P.J. Proteasome inhibitors: A novel class of potent and effective antitumor agents. Cancer Res. 1999, 59, 2615–2622. [Google Scholar] [PubMed]
- Drug Name: Bortezomib. Available online: http://www.bccancer.bc.ca/drug-database-site/Drug%20Index/Bortezomib_monograph.pdf (accessed on 17 October 2021).
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. STROBE Initiative. The strengthening the re-porting of observational studies in epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Epidemiol. 2008, 61, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, T.M.; Ryan, S.J.; Bennett, A.V.; Stover, A.; Saracino, R.M.; Rogak, L.J.; Jewell, S.; Matsoukas, K.; Li, Y.; Basch, E. The association between clinician-based common terminology criteria for adverse events (CTCAE) and patient-reported outcomes (PRO): A systematic review. Support. Care Cancer 2016, 24, 3669–3676. [Google Scholar] [CrossRef]
- Mandrekar, J.N. Receiver Operating Characteristic Curve in Diagnostic Test Assessment. J. Thorac. Oncol. 2010, 5, 1315–1316. [Google Scholar] [CrossRef]
- Reece, D.E.; Sullivan, D.; Lonial, S.; Mohrbacher, A.F.; Chatta, G.; Shustik, C.; Burris, H., III.; Venkatakrishnan, K.; Neuwirth, R.; Riordan, W.J.; et al. Pharmacokinetic and pharmacodynamic study of two doses of bortezomib in patients with relapsed multiple myeloma. Cancer Chemother. Pharmacol. 2011, 67, 57–67. [Google Scholar] [CrossRef][Green Version]
- Hanley, M.J.; Mould, D.R.; Taylor, T.J.; Gupta, N.; Suryanarayan, K.; Neuwirth, R.; Esseltine, D.L.; Horton, T.M.; Aplenc, R.; Alonzo, T.A.; et al. Population pharmacokinetic analysis of bortezomib in pediatric leukemia patients: Mod-el-based support for body surface area-based dosing over the 2- to 16-year age range. J. Clin. Pharmacol. 2017, 57, 1183–1193. [Google Scholar] [CrossRef]
- Tan, C.R.C.; Abdul-Majeed, S.; Cael, B.; Barta, S.K. Clinical Pharmacokinetics and Pharmacodynamics of Bortezomib. Clin. Pharmacokinet. 2019, 58, 157–168. [Google Scholar] [CrossRef]
- LoRusso, P.M.; Venkatakrishnan, K.; Ramanathan, R.K.; Sarantopoulos, J.; Mulkerin., D.; Shibata., S.I.; Hamilton., A.; Dowlati, A.; Mani, S.; Rudek, M.A.; et al. Pharmacokinetics and safety of bortezomib in patients with advanced malignancies and varying degrees of liver dysfunction: Phase I NCI Organ Dysfunction Working Group Study NCI-6432. Clin. Cancer Res. 2012, 18, 2954–2963. [Google Scholar] [CrossRef]
- Kurose, K.; Sugiyama, E.; Saito, Y. Population differences in major functional polymorphisms of pharmacokinetics/pharmacodynamics-related genes in Eastern Asians and Europeans: Implications in the clinical trials for novel drug development. Drug Metab. Pharmacokinet. 2012, 27, 9–54. [Google Scholar] [CrossRef] [PubMed]
- Ota, T.; Kamada, Y.; Hayashida, M.; Iwao-Koizumi, K.; Murata, S.; Kinoshita, K. Combination Analysis in Genetic Polymorphisms of Drug-Metabolizing Enzymes CYP1A2, CYP2C9, CYP2C19, CYP2D6 and CYP3A5 in the Japanese Population. Int. J. Med Sci. 2015, 12, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Quinn, D.I.; Nemunaitis, J.; Fuloria, J.; Britten, C.D.; Gabrail, N.; Yee, L.; Acharya, M.; Chan, K.; Cohen, N.; Dudov, A. Effect of the cytochrome P450 2C19 inhibitor omeprazole on the pharmacokinetics and safety profile of bortezomib in patients with advanced solid tumours, non-Hodgkin’s lymphoma or multiple myeloma. Clin. Pharmacokinet. 2009, 48, 199–209. [Google Scholar] [CrossRef]
- Zhou, W.; An, G.; Jian, Y.; Guo, H.; Chen, W. Effect of CYP2C19 and CYP3A4 gene polymorphisms on the efficacy of borte-zomib-based regimens in patients with multiple myeloma. Oncol. Lett. 2015, 10, 1171–1175. [Google Scholar] [CrossRef] [PubMed]
- Hellmann, A.; Rule, S.; Walewski, J.; Shpilberg, O.; Feng, H.; van de Velde, H.; Patel, H.; Skee, D.M.; Girgis, S.; Louw, V.J. Effect of cytochrome P450 3A4 inducers on the pharmacokinetic, pharmacodynamic and safety profiles of bortezomib in patients with multiple myeloma or non-Hodgkin’s lymphoma. Clin. Pharmacokinet. 2011, 50, 781–791. [Google Scholar] [CrossRef]
- Iwamoto, T.; Ishibashi, M.; Fujieda, A.; Masuya, M.; Katayama, N.; Okuda, M. Drug Interaction Between Itraconazole and Bortezomib: Exacerbation of Peripheral Neuropathy and Thrombocytopenia Induced by Bortezomib. Pharmacotherapy 2010, 30, 661–665. [Google Scholar] [CrossRef]
- Mateos, M.V.; Richardson, P.G.; Dimopoulos, M.A.; Palumbo, A.; Anderson, K.C.; Shi, H.; Elliott, J.; Dow, E.; van de Velde, H.; Niculescu, L.; et al. Effect of cumulative bortezomib dose on survival in multiple myeloma patients receiving borte-zomib-melphalanprednisone in the phase III VISTA study. Am. J. Hematol. 2015, 90, 314–319. [Google Scholar] [CrossRef]
| Character | Patients without Diarrhea | Patients with Diarrhea | p Value | |||
|---|---|---|---|---|---|---|
| Baseline characteristics | ||||||
| Male/female (male %) | 11/5 (68.8%) | 4/4 (50%) | 0.412 | |||
| Age, years | 77 | (49–83) | 62 | (54–71) | 0.008 * | |
| Height, cm | 160.9 | (145.1–171.2) | 162.1 | (140.0–167.4) | 0.811 | |
| Weight, kg | 57.4 | (43–78.4) | 60.4 | (38.6–71.2) | 0.893 | |
| Serum albumin, g/dL | 3.3 | (1.3–5.9) | 3.8 | (2.5–4.6) | 0.579 | |
| Blood urea nitrogen, mg/dL | 19.3 | (11.4–70.4) | 12.8 | (6.8–23.1) | 0.033 | |
| Serum creatinine, mg/dL | 0.97 | (0.42–8.01) | 0.74 | (0.47–1.33) | 0.229 | |
| Total bilirubin, mg/dL | 0.55 | (0.30–1.00) | 0.65 | (0.40–2.30) | 0.195 | |
| Alanine transaminase, IU/L | 19 | (11–68) | 22.5 | (14–66) | 0.475 | |
| Aspartate transaminase, IU/L | 22 | (9–90) | 17 | (10–54) | 0.696 | |
| γ-glutamyl transpeptidase, IU/L | 24 | (7–125) | 29 | (11–343) | 0.427 | |
| Cholinesterase, IU/L | 237 | (102–451) | 244 | (113–321) | 0.634 | |
| White blood cell, counts × 103/μL | 5 | (2.9–8.7) | 3.5 | (1.6–10.1) | 0.387 | |
| Red blood cell, counts × 106/μL | 3.7 | (2.7–5.0) | 3.5 | (2.8–4.9) | 0.773 | |
| Hemoglobin, g/dL | 11.8 | (8.6–15.4) | 9.9 | (0.8–15.7) | 0.377 | |
| Hematocrit, % | 36.1 | (27.4–43.7) | 33.7 | (22.9–47.7) | 0.993 | |
| Platelet, counts × 103/μL | 190 | (56–315) | 159 | (53–246) | 0.424 | |
| Prothrombin time, % | 94 | (11–134) | 90 | (61–137) | 0.605 | |
| Prothrombin time-international normalized ratio | 1.02 | (0.89–1.76) | 1.05 | (0.89–1.26) | 0.610 | |
| Comorbidities | ||||||
| Hypertension (%) | 7 (43.8%) | 3 (37.5%) | 1.000 | |||
| Chronic kidney disease (%) | 6 (37.5%) | 0 (0.0%) | 0.066 | |||
| Diabetes mellitus (%) | 2 (12.5%) | 2 (25.0%) | 0.578 | |||
| Solid carcinoma (%) | 3 (18.8%) | 1 (12.5%) | 1.000 | |||
| Heart failure (%) | 7 (43.8%) | 2 (25.0%) | 0.657 | |||
| Liver disiease (%) | 7 (43.8%) | 2 (25.0%) | 0.657 | |||
| Alcohol consumption | 5 (31.3%) | 3 (37.5%) | 1.000 | |||
| Multiple myeloma type | 0.674 | |||||
| IgGκ (%) | 6 (37.5%) | 4 (25.0%) | ||||
| IgGλ (%) | 0 (0.0%) | 1 (6.3%) | ||||
| IgAκ (%) | 1 (6.3%) | 1 (6.3%) | ||||
| IgAλ (%) | 2 (12.5%) | 0 (0%) | ||||
| AL amyloidosis (%) | 4 (25.0%) | 1 (6.3%) | ||||
| Ohters (%) | 3 (18.8%) | 1 (6.3%) | ||||
| Recurrence (%) | 4 (25.0%) | 4 (50.0%) | 0.363 | |||
| Gastrointestinal amyloidosis | 3 (18.8%) | 0 (0%) | 0.526 | |||
| Disease stage | ||||||
| I | 7 (43.8%) | 7 (87.5%) | 0.079 | |||
| II | 5 (31.3%) | 1 (12.5%) | 0.621 | |||
| III | 4 (25.0%) | 0 (0.0%) | 0.262 | |||
| Exposure | ||||||
| Measured concentration, ng/mL | 0.48 | (0.26–0.67) | 0.87 | (0.66–2.23) | 0.040 * | |
| Measurement after second dose | 4 (25.0%) | 3 (37.5%) | 0.866 | |||
| Dose, mg/m2 | 0.657 | |||||
| 1.3 mg/m2 (%) | 9 (56.2%) | 6 (75.0%) | ||||
| 1.0 mg/m2 (%) | 7 (43.8%) | 2 (25.0%) | ||||
| Body surface area, m2 | 1.6 | (1.3–1.9) | 1.6 | (1.3–1.8) | 1.000 | |
| Dose, mg | 1.7 | (1.5–2.4) | 1.9 | (1.6–2.2) | 0.433 | |
| Concentration/dose, (ng/mL)/(mg/m2) | 0.5 | (0.0–1.0) | 0.8 | (0.3–1.9) | 0.020 | |
| Injection route | ||||||
| Subcutaneous injection (%) | 15 (93.8%) | 8 (100%) | 1.000 | |||
| Intravenous injection (%) | 1 (6.3%) | 0 (0.0%) | 1.000 | |||
| Concomitant CYP 2C19 inhibitors | ||||||
| Lansoprazole (%) | 7 (43.8%) | 2 (25.0%) | 0.657 | |||
| Esomeprazole (%) | 5 (31.3%) | 2 (25.0%) | 1.000 | |||
| Rabeprazole (%) | 1 (6.3%) | 3 (37.5%) | 0.091 | |||
| Vonoprazan (%) | 1 (6.3%) | 0 (0.0%) | 1.000 | |||
| Fluconazole (%) | 1 (6.3%) | 0 (0.0%) | 1.000 | |||
| Concomitant CYP 3A4 inhibitors | ||||||
| Dexamethasone (%) | 15 (93.8%) | 7 (87.5%) | 1.000 | |||
| Concomitant CYP 3A4 inducers | ||||||
| Amlodipine (%) | 1 (6.3%) | 3 (37.5%) | 0.091 | |||
| Fluconazole (%) | 1 (6.3%) | 0 (0.0%) | 1.000 | |||
| Others (%) | 3 (18.8%) | 1 (12.5%) | 1.000 | |||
| Regimens | ||||||
| Bor + CPA + DEX (%) | 12 (75.0%) | 5 (62.5%) | 0.647 | |||
| Bor + DEX (%) | 2 (12.5%) | 1 (12.5%) | 0.333 | |||
| Others (%) | 2 (12.5%) | 2 (25.0%) | 0.578 | |||
| Outcomes | ||||||
| Peripheral neuropathy (%) | 4 (25.0%) | 3 (37.5%) | 0.647 | |||
| Fatigue (%) | 1 (6.3%) | 1 (12.5%) | 1.000 | |||
| Nausea or vomiting (%) | 1 (6.3%) | 0 (0.0%) | 1.000 | |||
| Constipation (%) | 9 (56.3%) | 1 (12.5%) | 0.079 | |||
| Thrombocytopenia (<75,000 counts/μL%) | 8 (50%) | 5 (62.5%) | 0.679 | |||
| Fever (>38 °C) (%) | 1 (6.3%) | 1 (12.5%) | 1.000 | |||
| Anemia (Hb <10.0 g/dL) (%) | 12 (75%) | 6 (75%) | 1.000 | |||
| Leukopenia (<3,000 counts/μL)% | 8 (50%) | 3 (37.5%) | 0.680 | |||
| Response rate (%) | 12 (75.0%) | 4 (50.0%) | 0.363 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fujimoto, Y.; Ueno, S.; Oda, K.; Gunda, N.; Shimomura, Y.; Nishimura, Y.; Yamaguchi, A.; Kuwano, A.; Ito, Y.; Baba, Y.; et al. Relationship between Serum Bortezomib Concentration and Emergence of Diarrhea in Patients with Multiple Myeloma and/or AL Amyloidosis. Cancers 2021, 13, 5674. https://doi.org/10.3390/cancers13225674
Fujimoto Y, Ueno S, Oda K, Gunda N, Shimomura Y, Nishimura Y, Yamaguchi A, Kuwano A, Ito Y, Baba Y, et al. Relationship between Serum Bortezomib Concentration and Emergence of Diarrhea in Patients with Multiple Myeloma and/or AL Amyloidosis. Cancers. 2021; 13(22):5674. https://doi.org/10.3390/cancers13225674
Chicago/Turabian StyleFujimoto, Yuki, Shikiko Ueno, Kazutaka Oda, Nao Gunda, Yumi Shimomura, Yuka Nishimura, Ayami Yamaguchi, Akari Kuwano, Yuki Ito, Yusuke Baba, and et al. 2021. "Relationship between Serum Bortezomib Concentration and Emergence of Diarrhea in Patients with Multiple Myeloma and/or AL Amyloidosis" Cancers 13, no. 22: 5674. https://doi.org/10.3390/cancers13225674
APA StyleFujimoto, Y., Ueno, S., Oda, K., Gunda, N., Shimomura, Y., Nishimura, Y., Yamaguchi, A., Kuwano, A., Ito, Y., Baba, Y., Nishigaki, A., Michiwaki, N., Uchino, S., Kurogi, K., Kawano, Y., Matsuoka, M., Saito, H., Okuno, Y., & Jono, H. (2021). Relationship between Serum Bortezomib Concentration and Emergence of Diarrhea in Patients with Multiple Myeloma and/or AL Amyloidosis. Cancers, 13(22), 5674. https://doi.org/10.3390/cancers13225674







