Development of a Radiomic-Based Model Predicting Lymph Node Involvement in Prostate Cancer Patients
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Population
2.2. Prostate MRI
2.3. Clinical and Radiomics Features
2.4. Model Building
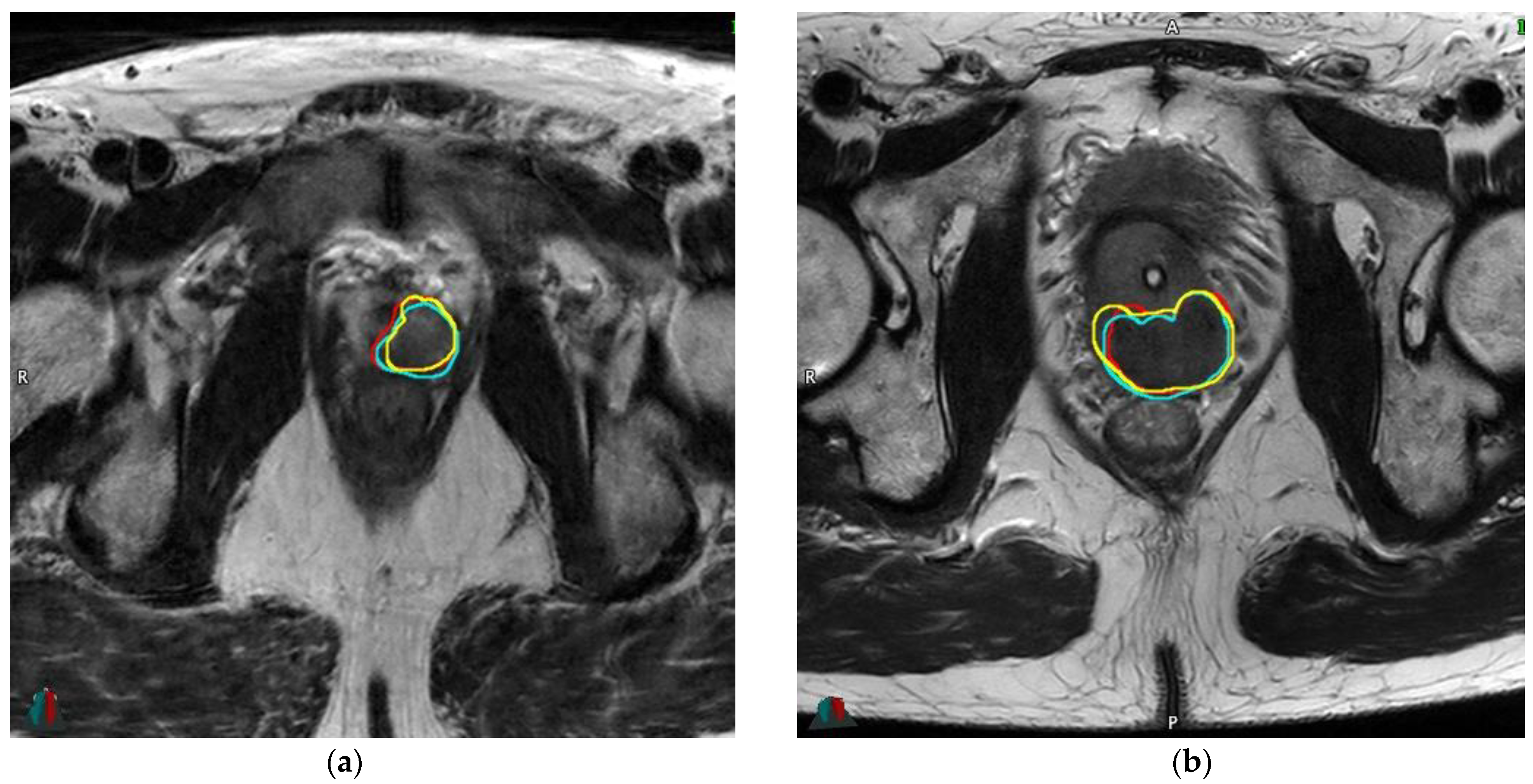
2.5. Inter-Reader Variability
2.6. Ethics Committee
3. Results
3.1. Population
3.2. Feature Set Reduction
3.3. Training Set
3.4. Testing Set
3.5. Inter-Reader Variability
3.6. NCCN Risk Classification
3.7. Subgroup with Targeted Biopsies
3.8. Radiomics Quality Score
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- EU. ECIS: European Cancer Information System. Available online: https://ecis.jrc.ec.europa.eu (accessed on 1 June 2021).
- Abdollah, F.; Suardi, N.; Cozzarini, C.; Gallina, A.; Capitanio, U.; Bianchi, M.; Sun, M.; Fossati, N.; Passoni, N.M.; Fiorino, C.; et al. Selecting the Optimal Candidate for Adjuvant Radiotherapy after Radical Prostatectomy for Prostate Cancer: A Long-term Survival Analysis. Eur. Urol. 2012, 63, 998–1008. [Google Scholar] [CrossRef]
- Loeb, S.; Partin, A.W.; Schaeffer, E.M. Complications of pelvic lymphadenectomy: Do the risks outweigh the benefits? Rev. Urol. 2010, 12, 20–24. [Google Scholar] [PubMed]
- Briganti, A.; Chun, F.; Salonia, A.; Suardi, N.; Gallina, A.; Da Pozzo, L.F.; Roscigno, M.; Zanni, G.; Valiquette, L.; Rigatti, P.; et al. Complications and Other Surgical Outcomes Associated with Extended Pelvic Lymphadenectomy in Men with Localized Prostate Cancer. Eur. Urol. 2006, 50, 1006–1013. [Google Scholar] [CrossRef] [PubMed]
- Mazzone, E.; Preisser, F.; Nazzani, S.; Tian, Z.; Bandini, M.; Gandaglia, G.; Fossati, N.; Montorsi, F.; Graefen, M.; Shariat, S.; et al. The Effect of Lymph Node Dissection in Metastatic Prostate Cancer Patients Treated with Radical Prostatectomy: A Contemporary Analysis of Survival and Early Postoperative Outcomes. Eur. Urol. Oncol. 2018, 2, 541–548. [Google Scholar] [CrossRef]
- Fossati, N.; Willemse, P.-P.M.; Broeck, T.V.D.; Bergh, R.C.V.D.; Yuan, Y.; Briers, E.; Bellmunt, J.; Bolla, M.; Cornford, P.; De Santis, M.; et al. The Benefits and Harms of Different Extents of Lymph Node Dissection during Radical Prostatectomy for Prostate Cancer: A Systematic Review. Eur. Urol. 2017, 72, 84–109. [Google Scholar] [CrossRef] [PubMed]
- Touijer, K.A.; Sjoberg, D.D.; Benfante, N.; Laudone, V.P.; Ehdaie, B.; Eastham, J.A.; Scardino, P.T.; Vickers, A. Limited versus Extended Pelvic Lymph Node Dissection for Prostate Cancer: A Randomized Clinical Trial. Eur. Urol. Oncol. 2021, 4, 532–539. [Google Scholar] [CrossRef]
- Mottet, N.; Cornford, P.; van den bergh, E.; Briers, E.; De Santis, M.; Fanti, S.; Gillessen, S.; Grummet, J.P.; Henry, A.M.; Lam, T.B.; et al. EAU Guidelines: Prostate Cancer 2021. Available online: https://uroweb.org/guideline/prostate-cancer/ (accessed on 1 June 2021).
- Tosoian, J.J.; Chappidi, M.; Feng, Z.; Humphreys, E.B.; Han, M.; Pavlovich, C.P.; Epstein, J.I.; Partin, A.W.; Trock, B.J. Prediction of pathological stage based on clinical stage, serum prostate-specific antigen, and biopsy Gleason score: Partin Tables in the contemporary era. BJU Int. 2016, 119, 676–683. [Google Scholar] [CrossRef] [Green Version]
- Roach, M.; Marquez, C.; Yuo, H.-S.; Narayan, P.; Coleman, L.; Nseyo, U.O.; Navvab, Z.; Carroll, P.R. Predicting the risk of lymph node involvement using the pre-treatment prostate specific antigen and gleason score in men with clinically localized prostate cancer. Int. J. Radiat. Oncol. 1994, 28, 33–37. [Google Scholar] [CrossRef]
- Yu, J.B.; Makarov, D.; Gross, C. A New Formula for Prostate Cancer Lymph Node Risk. Int. J. Radiat. Oncol. 2011, 80, 69–75. [Google Scholar] [CrossRef]
- Briganti, A.; Larcher, A.; Abdollah, F.; Capitanio, U.; Gallina, A.; Suardi, N.; Bianchi, M.; Sun, M.; Freschi, M.; Salonia, A.; et al. Updated Nomogram Predicting Lymph Node Invasion in Patients with Prostate Cancer Undergoing Extended Pelvic Lymph Node Dissection: The Essential Importance of Percentage of Positive Cores. Eur. Urol. 2011, 61, 480–487. [Google Scholar] [CrossRef]
- Gandaglia, G.; Fossati, N.; Zaffuto, E.; Bandini, M.; Dell’Oglio, P.; Bravi, C.A.; Fallara, G.; Pellegrino, F.; Nocera, L.; Karakiewicz, P.I.; et al. Development and Internal Validation of a Novel Model to Identify the Candidates for Extended Pelvic Lymph Node Dissection in Prostate Cancer. Eur. Urol. 2017, 72, 632–640. [Google Scholar] [CrossRef]
- Godoy, G.; Chong, K.T.; Cronin, A.; Vickers, A.; Laudone, V.; Touijer, K.; Guillonneau, B.; Eastham, J.A.; Scardino, P.T.; Coleman, J. Extent of Pelvic Lymph Node Dissection and the Impact of Standard Template Dissection on Nomogram Prediction of Lymph Node Involvement. Eur. Urol. 2011, 60, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Ahdoot, M.; Wilbur, A.R.; Reese, S.E.; Lebastchi, A.H.; Mehralivand, S.; Gomella, P.; Bloom, J.; Gurram, S.; Siddiqui, M.; Pinsky, P.; et al. MRI-Targeted, Systematic, and Combined Biopsy for Prostate Cancer Diagnosis. N. Engl. J. Med. 2020, 382, 917–928. [Google Scholar] [CrossRef] [PubMed]
- Gandaglia, G.; Ploussard, G.; Valerio, M.; Mattei, A.; Fiori, C.; Fossati, N.; Stabile, A.; Beauval, J.-B.; Malavaud, B.; Roumiguié, M.; et al. A Novel Nomogram to Identify Candidates for Extended Pelvic Lymph Node Dissection among Patients with Clinically Localized Prostate Cancer Diagnosed with Magnetic Resonance Imaging-targeted and Systematic Biopsies. Eur. Urol. 2018, 75, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Kasivisvanathan, V.; Rannikko, A.; Borghi, M.; Panebianco, V.; Mynderse, L.A.; Vaarala, M.; Briganti, A.; Budäus, L.; Hellawell, G.; Hindley, R.G.; et al. MRI-Targeted or Standard Biopsy for Prostate-Cancer Diagnosis. N. Engl. J. Med. 2018, 378, 1767–1777. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Rosenkrantz, A.; Huang, R.; Deng, F.-M.; Wysock, J.S.; Bjurlin, M.A.; Huang, W.; Lepor, H.; Taneja, S.S. The Institutional Learning Curve of Magnetic Resonance Imaging-Ultrasound Fusion Targeted Prostate Biopsy: Temporal Improvements in Cancer Detection in 4 Years. J. Urol. 2018, 200, 1022–1029. [Google Scholar] [CrossRef]
- Bourbonne, V.; Fournier, G.; Vallières, M.; Lucia, F.; Doucet, L.; Tissot, V.; Cuvelier, G.; Hue, S.; Du, H.L.P.; Perdriel, L.; et al. External Validation of an MRI-Derived Radiomics Model to Predict Biochemical Recurrence after Surgery for High-Risk Prostate Cancer. Cancers 2020, 12, 814. [Google Scholar] [CrossRef] [Green Version]
- Bourbonne, V.; Vallières, M.; Lucia, F.; Doucet, L.; Visvikis, D.; Tissot, V.; Pradier, O.; Hatt, M.; Schick, U. MRI-Derived Radiomics to Guide Post-operative Management for High-Risk Prostate Cancer. Front. Oncol. 2019, 9, 807. [Google Scholar] [CrossRef]
- Yasaka, K.; Akai, H.; Kunimatsu, A.; Kiryu, S.; Abe, O. Deep learning with convolutional neural network in radiology. Jpn. J. Radiol. 2018, 36, 257–272. [Google Scholar] [CrossRef] [PubMed]
- Heidenreich, A.; Ohlmann, C.H.; Polyakov, S. Anatomical Extent of Pelvic Lymphadenectomy in Patients Undergoing Radical Prostatectomy. Eur. Urol. 2007, 52, 29–37. [Google Scholar] [CrossRef]
- Turkbey, B.; Rosenkrantz, A.B.; Haider, M.A.; Padhani, A.; Villeirs, G.; Macura, K.J.; Tempany, C.M.; Choyke, P.L.; Cornud, F.; Margolis, D.J.; et al. Prostate Imaging Reporting and Data System Version 2.1: 2019 Update of Prostate Imaging Reporting and Data System Version 2. Eur. Urol. 2019, 76, 340–351. [Google Scholar] [CrossRef]
- Fortin, J.-P.; Cullen, N.; Sheline, Y.I.; Taylor, W.D.; Aselcioglu, I.; Cook, P.A.; Adams, P.; Cooper, C.; Fava, M.; McGrath, P.J.; et al. Harmonization of cortical thickness measurements across scanners and sites. NeuroImage 2017, 167, 104–120. [Google Scholar] [CrossRef] [PubMed]
- Johnson, W.; Li, C.; Rabinovic, A. Adjusting batch effects in microarray expression data using empirical Bayes methods. Biostatistics 2006, 8, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Lambin, P.; Leijenaar, R.T.; Deist, T.M.; Peerlings, J.; De Jong, E.E.; van Timmeren, J.; Sanduleanu, S.; LaRue, R.T.; Even, A.J.; Jochems, A.; et al. Radiomics: The bridge between medical imaging and personalized medicine. Nat. Rev. Clin. Oncol. 2017, 14, 749–762. [Google Scholar] [CrossRef] [PubMed]
- Peilleron, N.; Seigneurin, A.; Herault, C.; Verry, C.; Bolla, M.; Rambeaud, J.-J.; Descotes, J.-L.; Long, J.-A.; Fiard, G. External evaluation of the Briganti nomogram to predict lymph node metastases in intermediate-risk prostate cancer patients. World J. Urol. 2020, 39, 1489–1497. [Google Scholar] [CrossRef]
- Cornud, F.; Roumiguié, M.; De Longchamps, N.B.; Ploussard, G.; Bruguière, E.; Portalez, D.; Malavaud, B. Precision Matters in MR Imaging–targeted Prostate Biopsies: Evidence from a Prospective Study of Cognitive and Elastic Fusion Registration Transrectal Biopsies. Radiology 2018, 287, 534–542. [Google Scholar] [CrossRef]
- Wibmer, A.; Hricak, H.; Gondo, T.; Matsumoto, K.; Veeraraghavan, H.; Fehr, D.; Zheng, J.; Goldman, D.; Moskowitz, C.; Fine, S.W.; et al. Haralick texture analysis of prostate MRI: Utility for differentiating non-cancerous prostate from prostate cancer and differentiating prostate cancers with different Gleason scores. Eur. Radiol. 2015, 25, 2840–2850. [Google Scholar] [CrossRef] [PubMed]
- Zwanenburg, A.; Vallières, M.; Abdalah, M.A.; Aerts, H.J.W.L.; Andrearczyk, V.; Apte, A.; Ashrafinia, S.; Bakas, S.; Beukinga, R.J.; Boellaard, R.; et al. The Image Biomarker Standardization Initiative: Standardized Quantitative Radiomics for High-Throughput Image-based Phenotyping. Radiology 2020, 295, 328–338. [Google Scholar] [CrossRef] [Green Version]
- Dalela, D.; Loppenberg, B.; Sood, A.; Sammon, J.; Abdollah, F. Contemporary Role of the Decipher(R) Test in Prostate Cancer Management: Current Practice and Future Perspectives. Rev. Urol. 2016, 18, 1–9. [Google Scholar] [PubMed]
- Clark, T.; Nyberg, E. Creating the Black Box: A Primer on Convolutional Neural Network Use in Image Interpretation. Curr. Probl. Diagn. Radiol. 2020, 49, 365–367. [Google Scholar] [CrossRef]
- Gandaglia, G.; Martini, A.; Ploussard, G.; Fossati, N.; Stabile, A.; De Visschere, P.; Borgmann, H.; Heidegger, I.; Steinkohl, F.; Kretschmer, A.; et al. External Validation of the 2019 Briganti Nomogram for the Identification of Prostate Cancer Patients Who Should Be Considered for an Extended Pelvic Lymph Node Dissection. Eur. Urol. 2020, 78, 138–142. [Google Scholar] [CrossRef]
- Diamand, R.; Oderda, M.; Albisinni, S.; Fourcade, A.; Fournier, G.; Benamran, D.; Iselin, C.; Fiard, G.; Descotes, J.-L.; Assenmacher, G.; et al. External validation of the Briganti nomogram predicting lymph node invasion in patients with intermediate and high-risk prostate cancer diagnosed with magnetic resonance imaging-targeted and systematic biopsies: A European multicenter study. Urol. Oncol. Semin. Orig. Investig. 2020, 38, 847.e9–847.e16. [Google Scholar] [CrossRef]
- Pommier, P.; Chabaud, S.; Lagrange, J.-L.; Richaud, P.; Le Prise, E.; Wagner, J.-P.; Azria, D.; Beckendorf, V.; Suchaud, J.-P.; Bernier, V.; et al. Is There a Role for Pelvic Irradiation in Localized Prostate Adenocarcinoma? Update of the Long-Term Survival Results of the GETUG-01 Randomized Study. Int. J. Radiat. Oncol. 2016, 96, 759–769. [Google Scholar] [CrossRef] [Green Version]
- Sandler, K.A.; Cook, R.; Ciezki, J.P.; Ross, A.E.; Pomerantz, M.M.; Nguyen, P.L.; Shaikh, T.; Tran, P.T.; Stock, R.G.; Merrick, G.S.; et al. Prostate-only Versus Whole-pelvis Radiation with or without a Brachytherapy Boost for Gleason Grade Group 5 Prostate Cancer: A Retrospective Analysis. Eur. Urol. 2019, 77, 3–10. [Google Scholar] [CrossRef]
- Murthy, V.; Maitre, P.; Kannan, S.; Panigrahi, G.; Krishnatry, R.; Bakshi, G.; Prakash, G.; Pal, M.; Menon, S.; Phurailatpam, R.; et al. Prostate-Only versus Whole-Pelvic Radiation Therapy in High-Risk and Very High-Risk Prostate Cancer (POP-RT): Outcomes from Phase III Randomized Controlled Trial. J. Clin. Oncol. 2021, 39, 1234–1242. [Google Scholar] [CrossRef]

| Training Set | Testing Set | p | ||||
|---|---|---|---|---|---|---|
| n = 168 (60.0%) | n = 112 (40.0%) | |||||
| Age (median, range) | 65.0 | 45.3–79.6 | 66.7 | 50.8–77.8 | 0.10 | |
| PSA level (median, range) | 7.7 | 1.5–63.0 | 7.4 | 1.0–49.9 | 0.16 | |
| Clinical Tumour Stage (nb, %) | T1c | 90 | 53.6 | 71 | 63.4 | 0.06 |
| T2a | 34 | 20.2 | 22 | 19.6 | ||
| T2b | 30 | 17.9 | 14 | 12.5 | ||
| T2c | 9 | 5. | 3 | 2.7 | ||
| T3a | 1 | 0.6 | 1 | 0.9 | ||
| T3b | 4 | 2.4 | 1 | 0.9 | ||
| mpMRI Tumour Stage (nb, %) | T2a | 0 | 0.0 | 0 | 0.0 | 0.60 |
| T2b | 71 | 42.3 | 45 | 40.2 | ||
| T2c | 66 | 39.3 | 43 | 38.4 | ||
| T3a | 31 | 18.4 | 24 | 21.4 | ||
| ≥T3b | 0 | 0 | 0 | 0 | ||
| PIRADS (nb, %) | 3 | 12 | 7.1 | 39 | 34.8 | <0.0001 |
| 4 | 60 | 35.7 | 41 | 36.6 | ||
| 5 | 96 | 57.2 | 32 | 28.6 | ||
| Median maximum index lesion diameter on mpMRI (mm, IQR) | 13.2 | 9.7–16.2 | 13.6 | 11.6–15.7 | 0.26 | |
| Number of Lesion PIRADS ≥ 3 on mpMRI per patient (nb, %) | 1 | 153 | 91.1 | 100 | 89.3 | 0.62 |
| 2 | 15 | 8.9 | 12 | 10.7 | ||
| Type of MRI (nb, %) | Siemens 1.5T | 123 | 73.2 | 75 | 67.0 | 0.26 |
| Philips 3T | 45 | 26.8 | 37 | 33.0 | ||
| Biopsy ISUP score (nb, %) | 1 | 38 | 22.6 | 19 | 17.0 | 0.12 |
| 2 | 77 | 45.8 | 53 | 47.3 | ||
| 3 | 30 | 17.9 | 13 | 11.6 | ||
| 4 | 23 | 13.7 | 23 | 20.5 | ||
| 5 | 0 | 0.0 | 4 | 3.6 | ||
| NCCN risk classification (nb, %) | Low | 15 | 8.9 | 8 | 7.1 | 0.23 |
| Intermediate | 114 | 67.9 | 71 | 63.4 | ||
| High | 39 | 23.2 | 33 | 29.5 | ||
| Nb Cores (mean, range) | 13.7 | 6–35 | 13.3 | 8–24 | 0.37 | |
| Nb Positive Cores (mean, range) | 5.9 | 1–22 | 5.4 | 1–12 | 0.49 | |
| Ratio Positive Cores ± (mean, range) | 43.5 | 2.9–100 | 41.2 | 5.9–100 | 0.57 | |
| Percentage of positive cores with highest grade (median, range) | 30.0 | 3.2–73.9 | 29.5 | 2.6–67.3 | 0.68 | |
| Percentage of positive cores with lower grade (median, range) | 28.0 | 0.5–62.7 | 27.1 | 2.2–61.8 | 0.42 | |
| Pathological Tumour Stage (nb, %) | T2a | 3 | 1.8 | 8 | 7.1 | 0.16 |
| T2b | 3 | 1.8 | 5 | 4.5 | ||
| T2c | 67 | 39.9 | 38 | 33.9 | ||
| T3a | 59 | 35.1 | 45 | 40.2 | ||
| T3b | 34 | 20.2 | 16 | 14.3 | ||
| T4 | 2 | 1.2 | 0 | 0 | ||
| Surgical Margin (nb, %) | Positive | 88 | 52.4 | 66 | 58.9 | 0.28 |
| Negative | 80 | 47.6 | 46 | 41.1 | ||
| Pathological ISUP score (nb, %) | 1 | 17 | 10.1 | 10 | 8.9 | 0.23 |
| 2 | 56 | 33.3 | 46 | 41.1 | ||
| 3 | 50 | 29.8 | 36 | 32.1 | ||
| 4 | 10 | 6.0 | 2 | 1.8 | ||
| 5 | 35 | 20.8 | 18 | 16.1 | ||
| Nb Nodes removed (median IQR) | 15 | 12–17 | 15 | 12–18 | 0.44 | |
| Nb Nodes positive (median, range) | 1 | 1–2 | 1 | 1–2 | 0.67 | |
| Patients with LNI (nb, %) | 32 | 19.0 | 19 | 17.0 | 0.66 | |
| Model | Feature | Importance |
|---|---|---|
| New-Clinical Model | PIRADS Score | 3.6% |
| Percentage of positive biopsy cores with lowest-grade PCa | 4.2% | |
| Clinical Tumour Stage | 4.9% | |
| Percentage of positive biopsy cores | 16.3% | |
| Percentage of positive biopsy cores with highest-grade PCa | 71.0% | |
| New-Combined Model | PIRADS Score | 3.4% |
| Percentage of positive biopsy cores with lower grade Pca | 4.0% | |
| MRI Tumour Stage | 4.6% | |
| Feature 2 | 6.4% | |
| Percentage of positive biopsy cores | 15.2% | |
| Percentage of positive biopsy cores with highest-grade PCa | 66.4% | |
| Combat-Combined Model | Feature 8 | 4.6% |
| Number of positive biopsy cores | 5.7% | |
| Feature 2 | 7.0% | |
| Feature 7 | 7.2% | |
| Feature 6 | 7.7% | |
| Percentage of positive biopsy cores | 12.7% | |
| Percentage of positive biopsy cores with highest-grade PCa | 55.2% |
| Model Cut-Off | C-Index | Se | Sp | BAcc | Number of Patients, n (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Below the Cutoff (eLND not Recommended) | Above the Cutoff (eLND Recommended) | |||||||||
| Total | Without LNI | With LNI | Total | Without LNI | With LNI | |||||
| 1% | 0.88 | 100 | 76.5 | 88.3 | 104 (61.9) | 104 (100) | 0 (0) | 64 (38.1) | 32 (50.0) | 32 (50.0) |
| 2% | 0.91 | 100 | 81.6 | 90.8 | 111 (66.1) | 111 (100) | 0 (0) | 57 (33.9) | 25 (43.9) | 32 (56.1) |
| 3% | 0.93 | 100 | 86 | 93.0 | 117 (69.6) | 117 (100) | 0 (0) | 51 (30.4) | 19 (37.3) | 32 (62.7) |
| 4% | 0.95 | 100 | 89.7 | 94.9 | 122 (72.6) | 122 (100) | 0 (0) | 46 (27.4) | 14 (30.4) | 32 (69.6) |
| 5% | 0.96 | 100 | 91.9 | 96.0 | 125 (74.4) | 125 (100) | 0 (0) | 43 (25.6) | 11 (25.6) | 32 (74.4) |
| 6% | 0.97 | 100 | 94.1 | 97.1 | 128 (76.2) | 128 (100) | 0 (0) | 40 (23.8) | 8 (20.0) | 32 (80.0) |
| 7% | 0.98 | 100 | 95.6 | 97.8 | 130 (77.4) | 130 (100) | 0 (0) | 38 (22.6) | 6 (15.8) | 32 (84.2) |
| 8% | 0.98 | 100 | 96.3 | 98.2 | 131 (78.0) | 131 (100) | 0 (0) | 37 (22.0) | 5 (13.5) | 32 (86.5) |
| 9% | 0.99 | 100 | 97.8 | 98.9 | 133 (79.2) | 133 (100) | 0 (0) | 35 (20.8) | 3 (8.6) | 32 (91.4) |
| 10% | 1.00 | 100 | 99.3 | 99.7 | 135 (80.4) | 135 (100) | 0 (0) | 33 (19.6) | 1 (3.0) | 32 (97.0) |
| 11% | 1.00 | 100 | 99.3 | 99.7 | 135 (80.4) | 135 (100) | 0 (0) | 33 (19.6) | 1 (3.0) | 32 (97.0) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bourbonne, V.; Jaouen, V.; Nguyen, T.A.; Tissot, V.; Doucet, L.; Hatt, M.; Visvikis, D.; Pradier, O.; Valéri, A.; Fournier, G.; et al. Development of a Radiomic-Based Model Predicting Lymph Node Involvement in Prostate Cancer Patients. Cancers 2021, 13, 5672. https://doi.org/10.3390/cancers13225672
Bourbonne V, Jaouen V, Nguyen TA, Tissot V, Doucet L, Hatt M, Visvikis D, Pradier O, Valéri A, Fournier G, et al. Development of a Radiomic-Based Model Predicting Lymph Node Involvement in Prostate Cancer Patients. Cancers. 2021; 13(22):5672. https://doi.org/10.3390/cancers13225672
Chicago/Turabian StyleBourbonne, Vincent, Vincent Jaouen, Truong An Nguyen, Valentin Tissot, Laurent Doucet, Mathieu Hatt, Dimitris Visvikis, Olivier Pradier, Antoine Valéri, Georges Fournier, and et al. 2021. "Development of a Radiomic-Based Model Predicting Lymph Node Involvement in Prostate Cancer Patients" Cancers 13, no. 22: 5672. https://doi.org/10.3390/cancers13225672
APA StyleBourbonne, V., Jaouen, V., Nguyen, T. A., Tissot, V., Doucet, L., Hatt, M., Visvikis, D., Pradier, O., Valéri, A., Fournier, G., & Schick, U. (2021). Development of a Radiomic-Based Model Predicting Lymph Node Involvement in Prostate Cancer Patients. Cancers, 13(22), 5672. https://doi.org/10.3390/cancers13225672






