Use of Antiangiogenic Therapies in Pediatric Solid Tumors
Simple Summary
Abstract
1. Introduction
Origin of Childhood Cancer
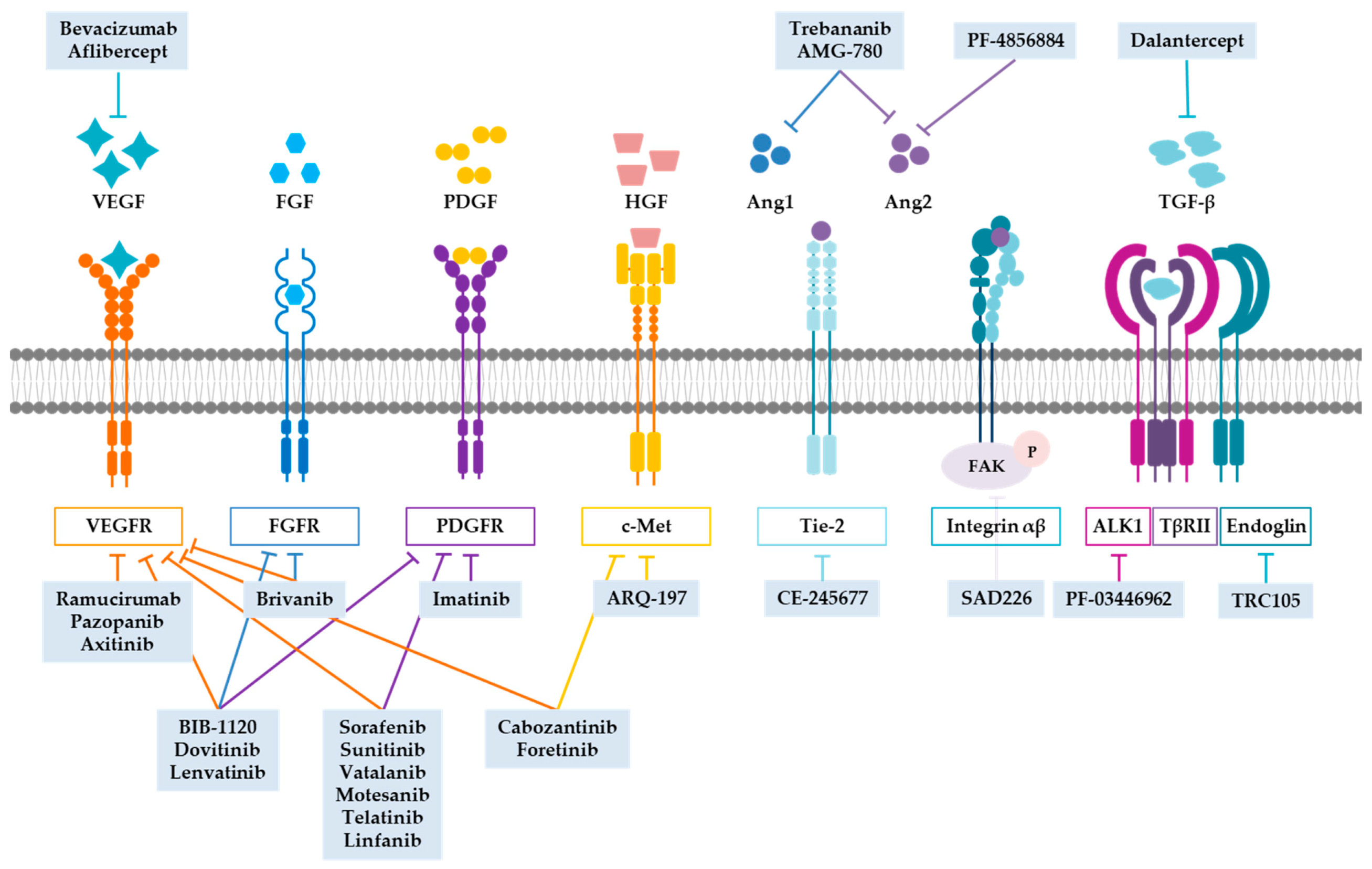
2. Tumor Angiogenesis
Main Targets of Antiangiogenic Therapy
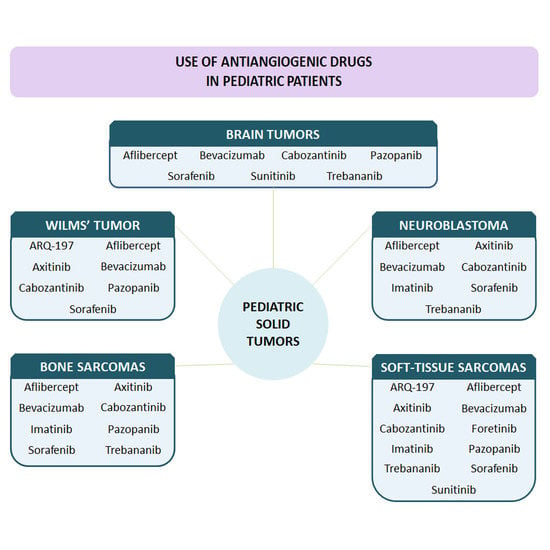
3. Antiangiogenics and Brain Tumors
3.1. Low-Grade Gliomas
3.2. Medulloblastomas
3.3. High-Grade Gliomas
3.4. Ependymomas
3.5. Other Brain Tumors
4. Antiangiogenics and Neuroblastoma
5. Antiangiogenics and Wilms’ Tumor
6. Antiangiogenics and Soft-Tissue Sarcomas
6.1. Rhabdomyosarcomas
6.2. Other Soft-Tissue Sarcomas
7. Antiangiogenics and Bone Sarcomas
7.1. Osteosarcomas
7.2. Ewing’s Sarcoma
8. Conclusions and Future Perspectives
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Cunningham, R.M.; Walton, M.A.; Carter, P.M. The Major Causes of Death in Children and Adolescents in the United States. N. Engl. J. Med. 2018, 379, 2468–2475. [Google Scholar] [CrossRef]
- Kattner, P.; Strobel, H.; Khoshnevis, N.; Grunert, M.; Bartholomae, S.; Pruss, M.; Fitzel, R.; Halatsch, M.-E.; Schilberg, K.; Siegelin, M.D.; et al. Compare and contrast: Pediatric cancer versus adult malignancies. Cancer Metastasis Rev. 2019, 38, 673–682. [Google Scholar] [CrossRef]
- Force, L.M.; Abdollahpour, I.; Advani, S.M.; Agius, D.; Ahmadian, E.; Alahdab, F.; Alam, T.; Alebel, A.; Alipour, V.; Allen, C.A.; et al. The global burden of childhood and adolescent cancer in 2017: An analysis of the Global Burden of Disease Study 2017. Lancet Oncol. 2019, 20, 1211–1225. [Google Scholar] [CrossRef]
- Barone, A.; Casey, D.; McKee, A.E.; Reaman, G. Cancer drugs approved for use in children: Impact of legislative initiatives and future opportunities. Pediatr. Blood Cancer 2019, 66, e27809. [Google Scholar] [CrossRef]
- de Jong, M.C.; Kors, W.A.; de Graaf, P.; Castelijns, J.A.; Kivelä, T.; Moll, A.C. Trilateral retinoblastoma: A systematic review and meta-analysis. Lancet Oncol. 2014, 15, 1157–1167. [Google Scholar] [CrossRef]
- Saletta, F.; Pozza, L.D.; Byrne, J.A. Genetic causes of cancer predisposition in children and adolescents. Transl. Pediatr. 2015, 4, 67–75. [Google Scholar]
- Brodeur, G.M.; Nichols, K.E.; Plon, S.E.; Schiffman, J.D.; Malkin, D. Pediatric Cancer Predisposition and Surveillance: An Overview, and a Tribute to Alfred G. Knudson Jr. Clin. Cancer Res. 2017, 23, e1–e5. [Google Scholar] [CrossRef]
- Choong, S.S.; Latiff, Z.A.; Mohamed, M.; Lim, L.L.W.; Chen, K.S.; Vengidasan, L.; Razali, H.; Rahman, E.J.A.; Ariffin, H.; Malaysian Society of Paediatric Haematology-Oncology. Childhood adrenocortical carcinoma as a sentinel cancer for detecting families with germlineTP53mutations. Clin. Genet. 2012, 82, 564–568. [Google Scholar] [CrossRef]
- Gröbner, S.N.; Worst, B.C.; Weischenfeldt, J.; Buchhalter, I.; Kleinheinz, K.; Rudneva, V.A.; Johann, P.D.; Balasubramanian, G.P.; Segura-Wang, M.; Brabetz, S.; et al. The landscape of genomic alterations across childhood cancers. Nature 2018, 555, 321–327. [Google Scholar] [CrossRef]
- Campbell, B.B.; Light, N.; Fabrizio, D.; Zatzman, M.; Fuligni, F.; de Borja, R.; Davidson, S.; Edwards, M.; Elvin, J.A.; Hodel, K.P.; et al. Comprehensive Analysis of Hypermutation in Human Cancer. Cell 2017, 171, 1042–1056.e10. [Google Scholar] [CrossRef]
- Núñez-Gómez, E.; Pericacho, M.; Ollauri-Ibáñez, C.; Bernabéu, C.; López-Novoa, J.M. The role of endoglin in post-ischemic revascularization. Angiogenesis 2016, 20, 1–24. [Google Scholar] [CrossRef]
- Ollauri-Ibáñez, C.; López-Novoa, J.M.; Pericacho, M. Endoglin-based biological therapy in the treatment of angiogenesis-dependent pathologies. Expert Opin. Biol. Ther. 2017, 17, 1053–1063. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.; Hurwitz, H.I.; Sandler, A.B.; Miles, D.; Coleman, R.L.; Deurloo, R.; Chinot, O.L. Bevacizumab (Avastin®) in cancer treatment: A review of 15 years of clinical experience and future outlook. Cancer Treat. Rev. 2020, 86, 102017. [Google Scholar] [CrossRef]
- Koch, A.E.; Distler, O. Vasculopathy and disordered angiogenesis in selected rheumatic diseases: Rheumatoid arthritis and systemic sclerosis. Arthritis Res. Ther. 2007, 9, S3. [Google Scholar] [CrossRef]
- Potente, M.; Carmeliet, P. The Link Between Angiogenesis and Endothelial Metabolism. Annu. Rev. Physiol. 2017, 79, 43–66. [Google Scholar] [CrossRef]
- Sie, M.; Dunnen, W.F.D.; Hoving, E.W.; de Bont, E.S. Anti-angiogenic therapy in pediatric brain tumors: An effective strategy? Crit. Rev. Oncol. 2014, 89, 418–432. [Google Scholar] [CrossRef]
- Sherwood, L.M.; Parris, E.E.; Folkman, J. Tumor Angiogenesis: Therapeutic Implications. N. Engl. J. Med. 1971, 285, 1182–1186. [Google Scholar] [CrossRef]
- Viallard, C.; Larrivée, B. Tumor angiogenesis and vascular normalization: Alternative therapeutic targets. Angiogenesis 2017, 20, 409–426. [Google Scholar] [CrossRef]
- Nagy, J.A.; Chang, S.-H.; Dvorak, A.M.; Dvorak, H.F. Why are tumour blood vessels abnormal and why is it important to know? Br. J. Cancer 2009, 100, 865–869. [Google Scholar] [CrossRef]
- Fukumura, D.; Duda, D.G.; Munn, L.L.; Jain, R.K. Tumor Microvasculature and Microenvironment: Novel Insights Through Intravital Imaging in Pre-Clinical Models. Microcirculation 2010, 17, 206–225. [Google Scholar] [CrossRef]
- Zirlik, K.; Duyster, J. Anti-Angiogenics: Current Situation and Future Perspectives. Oncol. Res. Treat. 2018, 41, 166–171. [Google Scholar] [CrossRef]
- Bergers, G.; Hanahan, D. Modes of resistance to anti-angiogenic therapy. Nat. Rev. Cancer 2008, 8, 592–603. [Google Scholar] [CrossRef]
- Zhang, Y.; Cedervall, J.; Hamidi, A.; Herre, M.; Viitaniemi, K.; D’Amico, G.; Miao, Z.; Unnithan, R.V.M.; Vaccaro, A.; van Hooren, L.; et al. Platelet-Specific PDGFB Ablation Impairs Tumor Vessel Integrity and Promotes Metastasis. Cancer Res. 2020, 80, 3345–3358. [Google Scholar] [CrossRef]
- Petrillo, M.; Scambia, G.; Ferrandina, G. Novel targets for VEGF-independent anti-angiogenic drugs. Expert Opin. Investig. Drugs 2012, 21, 451–472. [Google Scholar] [CrossRef]
- Pietras, K.; Sjöblom, T.; Rubin, K.; Heldin, C.-H.; Östman, A. PDGF receptors as cancer drug targets. Cancer Cell 2003, 3, 439–443. [Google Scholar] [CrossRef]
- Mccarty, M.F.; Somcio, R.J.; Stoeltzing, O.; Wey, J.; Fan, F.; Liu, W.; Bucana, C.; Ellis, L.M. Overexpression of PDGF-BB decreases colorectal and pancreatic cancer growth by increasing tumor pericyte content. J. Clin. Investig. 2007, 117, 2114–2122. [Google Scholar] [CrossRef]
- Iqbal, N.; Iqbal, N. Imatinib: A Breakthrough of Targeted Therapy in Cancer. Chemother. Res. Pract. 2014, 2014. [Google Scholar] [CrossRef]
- Papadopoulos, N.; Lennartsson, J. The PDGF/PDGFR pathway as a drug target. Mol. Asp. Med. 2018, 62, 75–88. [Google Scholar] [CrossRef]
- Czyz, M. Fibroblast Growth Factor Receptor Signaling in Skin Cancers. Cells 2019, 8, 540. [Google Scholar] [CrossRef]
- Babina, I.S.; Turner, N.C. Advances and challenges in targeting FGFR signalling in cancer. Nat. Rev. Cancer 2017, 17, 318. [Google Scholar] [CrossRef]
- Xue, W.-J.; Li, M.T.; Chen, L.; Sun, L.-P.; Li, Y. Recent developments and advances of FGFR as a potential target in cancer. Futur. Med. Chem. 2018, 10, 2109–2126. [Google Scholar] [CrossRef]
- Sanada, F.; Fujikawa, T.; Shibata, K.; Taniyama, Y.; Rakugi, H.; Morishita, R. Therapeutic Angiogenesis Using HGF Plasmid. Ann. Vasc. Dis. 2020, 13, 109–115. [Google Scholar] [CrossRef]
- Nicolini, G.; Forini, F.; Kusmic, C.; Iervasi, G.; Balzan, S. Angiopoietin 2 signal complexity in cardiovascular disease and cancer. Life Sci. 2019, 239, 117080. [Google Scholar] [CrossRef]
- Akwii, R.G.; Sajib, M.S.; Zahra, F.T.; Mikelis, C.M. Role of Angiopoietin-2 in Vascular Physiology and Pathophysiology. Cells 2019, 8, 471. [Google Scholar] [CrossRef]
- Yu, X.; Ye, F. Role of Angiopoietins in Development of Cancer and Neoplasia Associated with Viral Infection. Cells 2020, 9, 457. [Google Scholar] [CrossRef]
- Dowlati, A.; Vlahovic, G.; Natale, R.B.; Rasmussen, E.; Singh, I.; Hwang, Y.C.; Rossi, J.; Bass, M.B.; Friberg, G.; Pickett, C.A. A Phase I, First-in-Human Study of AMG 780, an Angiopoietin-1 and -2 Inhibitor, in Patients with Advanced Solid Tumors. Clin. Cancer Res. 2016, 22, 4574–4584. [Google Scholar] [CrossRef]
- Alday-Parejo, B.; Stupp, R.; Rüegg, C. Are Integrins Still Practicable Targets for Anti-Cancer Therapy? Cancers 2019, 11, 978. [Google Scholar] [CrossRef]
- Lv, P.-C.; Jiang, A.-Q.; Zhang, W.-M.; Zhu, H.-L. FAK inhibitors in Cancer, a patent review. Expert Opin. Ther. Patents 2018, 28, 139–145. [Google Scholar] [CrossRef]
- Nana, F.A.; Vanderputten, M.; Ocak, S. Role of Focal Adhesion Kinase in Small-Cell Lung Cancer and Its Potential as a Therapeutic Target. Cancers 2019, 11, 1683. [Google Scholar] [CrossRef]
- Ma, X.; Cui, Z.; Du, Z.; Lin, H. Transforming growth factor-β signaling, a potential mechanism associated with diabetes mellitus and pancreatic cancer? J. Cell. Physiol. 2020, 235, 5882–5892. [Google Scholar] [CrossRef]
- Bach, D.-H.; Park, H.J.; Lee, S.K. The Dual Role of Bone Morphogenetic Proteins in Cancer. Mol. Ther. Oncolytics 2018, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- De Vinuesa, A.G.; Bocci, M.; Pietras, K.; Dijke, P. Ten Targeting tumour vasculature by inhibiting activin receptor-like kinase (ALK)1 function. Biochem. Soc. Trans. 2016, 44, 1142–1149. [Google Scholar] [CrossRef] [PubMed]
- Cunha, S.I.; Pietras, K. ALK1 as an emerging target for antiangiogenic therapy of cancer. Blood 2011, 117, 6999–7006. [Google Scholar] [CrossRef]
- Hawinkels, L.J.; de Vinuesa, A.G.; Dijke, P.T. Activin receptor-like kinase 1 as a target for anti-angiogenesis therapy. Expert Opin. Investig. Drugs 2013, 22, 1371–1383. [Google Scholar] [CrossRef]
- Ollauri-Ibáñez, C.; Núñez-Gómez, E.; Egido-Turrión, C.; Silva-Sousa, L.; Díaz-Rodríguez, E.; Rodríguez-Barbero, A.; López-Novoa, J.M.; Pericacho, M. Continuous endoglin (CD105) overexpression disrupts angiogenesis and facilitates tumor cell metastasis. Angiogenesis 2020, 23, 231–247. [Google Scholar] [CrossRef]
- Paauwe, M.; Heijkants, R.C.; Oudt, C.H.; van Pelt, G.W.; Cui, C.; Theuer, C.P.; Hardwick, J.C.H.; Sier, C.F.M.; Hawinkels, L.J.A.C. Endoglin targeting inhibits tumor angiogenesis and metastatic spread in breast cancer. Oncogene 2016, 35, 4069–4079. [Google Scholar] [CrossRef]
- Uneda, S.; Toi, H.; Tsujie, T.; Tsujie, M.; Harada, N.; Tsai, H.; Seon, B.K. Anti-endoglin monoclonal antibodies are effective for suppressing metastasis and the primary tumors by targeting tumor vasculature. Int. J. Cancer 2009, 125, 1446–1453. [Google Scholar] [CrossRef]
- Gao, S.; Wang, S.; Fan, R.; Hu, J. Recent advances in the molecular mechanism of thalidomide teratogenicity. Biomed. Pharmacother. 2020, 127, 110114. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kim, Y.-M. Tumor endothelial cells as a potential target of metronomic chemotherapy. Arch. Pharmacal Res. 2019, 42, 1–13. [Google Scholar] [CrossRef]
- Cojocaru, E.; Mihăilă, D.; Leon-Constantin, M.M.; Cobzaru, R.G.; Rîpă, C.V.; Trandafirescu, M.F.; Trandafir, L.M. Update in pediatric primary brain tumors—From histology to genetically defined tumors. Rom. J. Morphol. Embryol. 2019, 60, 761–767. [Google Scholar]
- Pillay-Smiley, N.; Alden, T.; Hartsell, W.; Fangusaro, J. Severe Radiation Necrosis Successfully Treated with Bevacizumab in an Infant with Low-Grade Glioma and Tumor-Associated Intractable Trigeminal Neuralgia. Pediatr. Blood Cancer 2016, 63, 1671–1673. [Google Scholar] [CrossRef] [PubMed]
- Strenger, V.; Lackner, H.; Mayer, R.; Sminia, P.; Sovinz, P.; Mokry, M.; Pilhatsch, A.; Benesch, M.; Schwinger, W.; Seidel, M.; et al. Incidence and clinical course of radionecrosis in children with brain tumors: A 20-year longitudinal observational study. Strahlenther. Onkol. 2013, 189, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Plimpton, S.R.; Stence, N.; Hemenway, M.; Hankinson, T.C.; Foreman, N.; Liu, A.K. Cerebral Radiation Necrosis in Pediatric Patients. Pediatr. Hematol. Oncol. 2013, 32, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Barone, A.; Rubin, J.B. Opportunities and Challenges for Successful Use of Bevacizumab in Pediatrics. Front. Oncol. 2013, 3, 92. [Google Scholar] [CrossRef]
- Sie, M.; de Bont, E.S.J.M.; Scherpen, F.J.G.; Hoving, E.W.; Dunnen, W.F.A.D. Tumour vasculature and angiogenic profile of paediatric pilocytic astrocytoma; is it much different from glioblastoma? Neuropathol. Appl. Neurobiol. 2010, 36, 636–647. [Google Scholar] [CrossRef]
- Hsu, C.H.; Lober, R.M.; Li, M.D.; Partap, S.; Murphy, P.A.; Barnes, P.D.; Fisher, P.; Yeom, K.W. Decreased tumor apparent diffusion coefficient correlates with objective response of pediatric low-grade glioma to bevacizumab. J. Neuro-Oncol. 2015, 122, 491–496. [Google Scholar] [CrossRef]
- Hwang, E.I.; Jakacki, R.I.; Fisher, M.J.; Kilburn, L.; Horn, M.; Vezina, G.; Rood, B.R.; Packer, R.J. Long-term efficacy and toxicity of bevacizumab-based therapy in children with recurrent low-grade gliomas. Pediatr. Blood Cancer 2012, 60, 776–782. [Google Scholar] [CrossRef]
- Gorsi, H.S.; Khanna, P.; Tumblin, M.; Yeh-Nayre, L.; Milburn, M.; Elster, J.D.; Crawford, J.R. Single-agent bevacizumab in the treatment of recurrent or refractory pediatric low-grade glioma: A single institutional experience. Pediatr. Blood Cancer 2018, 65, e27234. [Google Scholar] [CrossRef]
- Zhukova, N.; Rajagopal, R.; Lam, A.; Coleman, L.; Shipman, P.; Walwyn, T.; Williams, M.; Sullivan, M.; Campbell, M.; Bhatia, K.; et al. Use of bevacizumab as a single agent or in adjunct with traditional chemotherapy regimens in children with unresectable or progressive low-grade glioma. Cancer Med. 2018, 8, 40–50. [Google Scholar] [CrossRef]
- Avery, R.A.; Hwang, E.I.; Jakacki, R.I.; Packer, R.J. Marked Recovery of Vision in Children with Optic Pathway Gliomas Treated with Bevacizumab. JAMA Ophthalmol. 2014, 132, 111–114. [Google Scholar] [CrossRef]
- Gururangan, S.; Fangusaro, J.; Poussaint, T.Y.; McLendon, R.E.; Onar-Thomas, A.; Wu, S.; Packer, R.J.; Banerjee, A.; Gilbertson, R.J.; Fahey, F.; et al. Efficacy of bevacizumab plus irinotecan in children with recurrent low-grade gliomas—A Pediatric Brain Tumor Consortium study. Neuro-Oncology 2014, 16, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Kalra, M.; Heath, J.A.; Kellie, S.J.; Pozza, L.D.; Stevens, M.M.; Swamy, S.; McCowage, G.B. Confirmation of Bevacizumab Activity, and Maintenance of Efficacy in Retreatment After Subsequent Relapse, in Pediatric Low-grade Glioma. J. Pediatr. Hematol. 2015, 37, e341–e346. [Google Scholar] [CrossRef] [PubMed]
- Okada, K.; Yamasaki, K.; Tanaka, C.; Fujisaki, H.; Osugi, Y.; Hara, J. Phase I Study of Bevacizumab Plus Irinotecan in Pediatric Patients with Recurrent/Refractory Solid Tumors. Jpn. J. Clin. Oncol. 2013, 43, 1073–1079. [Google Scholar] [CrossRef] [PubMed]
- Packer, R.J.; Jakacki, R.; Horn, M.; Rood, B.; Vezina, G.; Macdonald, T.; Fisher, M.J.; Cohen, B. Objective response of multiply recurrent low-grade gliomas to bevacizumab and irinotecan. Pediatr. Blood Cancer 2009, 52, 791–795. [Google Scholar] [CrossRef] [PubMed]
- Couec, M.-L.; André, N.; Thebaud, E.; Minckes, O.; Rialland, X.; Corradini, N.; Aerts, I.; Marec-Bérard, P.; Bourdeaut, F.; Leblond, P.; et al. Bevacizumab and irinotecan in children with recurrent or refractory brain tumors: Toxicity and efficacy trends. Pediatr. Blood Cancer 2012, 59, 34–38. [Google Scholar] [CrossRef]
- Pilotto, C.; Beshlawi, I.; Thomas, A.; Grundy, R.G. Vascular stenosis in a child with visual pathway glioma treated with bevacizumab: A case report and review of literature. Child’s Nerv. Syst. 2017, 34, 781–785. [Google Scholar] [CrossRef]
- Thomas, A.A.; Tucker, S.M.; Nelson, C.J.; Nickerson, J.P.; Durham, S.R.; Homans, A.C. Anaplastic pleomorphic xanthoastrocytoma with leptomeningeal dissemination responsive to BRAF inhibition and bevacizumab. Pediatr. Blood Cancer 2019, 66, e27465. [Google Scholar] [CrossRef]
- Metts, R.D.; Bartynski, W.; Welsh, C.T.; Kinsman, S.; Bredlau, A.-L. Bevacizumab Therapy for Pilomyxoid Astrocytoma. J. Pediatr. Hematol. 2017, 39, e219–e223. [Google Scholar] [CrossRef]
- Karajannis, M.; Legault, G.; Fisher, M.J.; Milla, S.S.; Cohen, K.J.; Wisoff, J.H.; Harter, D.H.; Goldberg, J.D.; Hochman, T.; Merkelson, A.; et al. Phase II study of sorafenib in children with recurrent or progressive low-grade astrocytomas. Neuro-Oncology 2014, 16, 1408–1416. [Google Scholar] [CrossRef]
- Legault, G.; Kieran, M.W.; Scott, R.M.; Chordas, C.; Milla, S.S.; Karajannis, M. Recurrent Ascites in a Patient with Low-grade Astrocytoma and Ventriculo-Peritoneal Shunt Treated with the Multikinase Inhibitor Sorafenib. J. Pediatr. Hematol. 2014, 36, e533–e535. [Google Scholar] [CrossRef]
- Thompson, E.M.; Keir, S.T.; Venkatraman, T.; Lascola, C.; Yeom, K.W.; Nixon, A.B.; Liu, Y.; Picard, D.; Remke, M.; Bigner, D.D.; et al. The role of angiogenesis in Group 3 medulloblastoma pathogenesis and survival. Neuro-Oncology 2017, 19, 1217–1227. [Google Scholar] [CrossRef] [PubMed]
- Peyrl, A.; Chocholous, M.; Kieran, M.W.; Azizi, A.A.; Prucker, C.; Czech, T.; Dieckmann, K.; Schmook, M.-T.; Haberler, C.; Leiss, U.; et al. Antiangiogenic metronomic therapy for children with recurrent embryonal brain tumors. Pediatr. Blood Cancer 2012, 59, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, D.; Mazewski, C.; Fangusaro, J.; Macdonald, T.J.; McNall-Knapp, R.Y.; Hayes, L.L.; Kim, S.; Castellino, R.C. Response to bevacizumab, irinotecan, and temozolomide in children with relapsed medulloblastoma: A multi-institutional experience. Child’s Nerv. Syst. 2013, 29, 589–596. [Google Scholar] [CrossRef]
- Piha-Paul, S.A.; Shin, S.J.; Vats, T.; Guha-Thakurta, N.; Aaron, J.; Rytting, M.; Kleinerman, E.; Kurzrock, R. Pediatric patients with refractory central nervous system tumors: Experiences of a clinical trial combining bevacizumab and temsirolimus. Anticancer. Res. 2014, 34, 1939–1945. [Google Scholar]
- Craveiro, R.B.; Ehrhardt, M.; Holst, M.; Pietsch, T.; Dilloo, D. In comparative analysis of multi-kinase inhibitors for targeted medulloblastoma therapy pazopanib exhibits promising in vitro and in vivo efficacy. Oncotarget 2014, 5, 7149. [Google Scholar] [CrossRef] [PubMed]
- Abouantoun, T.J.; Castellino, R.C.; Macdonald, T.J. Sunitinib induces PTEN expression and inhibits PDGFR signaling and migration of medulloblastoma cells. J. Neuro-Oncol. 2010, 101, 215–226. [Google Scholar] [CrossRef]
- Yang, F.; Jove, V.; Xin, H.; Hedvat, M.; van Meter, T.E.; Yu, H. Sunitinib Induces Apoptosis and Growth Arrest of Medulloblastoma Tumor Cells by Inhibiting STAT3 and AKT Signaling Pathways. Mol. Cancer Res. 2010, 8, 35–45. [Google Scholar] [CrossRef]
- André, N.; Verschuur, A.; Rössler, J.K.; Sterba, J. Anti-angiogenic therapies for children with cancer. Curr. Cancer Drug Targets 2010, 10, 879–889. [Google Scholar] [CrossRef]
- Becher, O.J.; Hambardzumyan, D.; Walker, T.R.; Helmy, K.; Nazarian, J.; Albrecht, S.; Hiner, R.L.; Gall, S.; Huse, J.T.; Jabado, N.; et al. Preclinical Evaluation of Radiation and Perifosine in a Genetically and Histologically Accurate Model of Brainstem Glioma. Cancer Res. 2010, 70, 2548–2557. [Google Scholar] [CrossRef]
- Parekh, C.; Jubran, R.; Erdreich-Epstein, A.; Panigrahy, A.; Bluml, S.; Finlay, J.; Dhall, G. Treatment of children with recurrent high grade gliomas with a bevacizumab containing regimen. J. Neuro-Oncol. 2010, 103, 673–680. [Google Scholar] [CrossRef]
- Narayana, A.; Kunnakkat, S.; Chacko-Mathew, J.; Gardner, S.; Karajannis, M.; Raza, S.; Wisoff, J.; Weiner, H.; Harter, D.; Allen, J. Bevacizumab in recurrent high-grade pediatric gliomas. Neuro-Oncology 2010, 12, 985–990. [Google Scholar] [CrossRef]
- Gururangan, S.; Chi, S.N.; Poussaint, T.Y.; Onar-Thomas, A.; Gilbertson, R.J.; Vajapeyam, S.; Friedman, H.S.; Packer, R.J.; Rood, B.N.; Boyett, J.M.; et al. Lack of Efficacy of Bevacizumab Plus Irinotecan in Children with Recurrent Malignant Glioma and Diffuse Brainstem Glioma: A Pediatric Brain Tumor Consortium Study. J. Clin. Oncol. 2010, 28, 3069–3075. [Google Scholar] [CrossRef]
- Umeda, K.; Shibata, H.; Saida, S.; Hiramatsu, H.; Arakawa, Y.; Mizowaki, T.; Nishiuchi, R.; Adachi, S.; Heike, T.; Watanabe, K.-I. Long-term efficacy of bevacizumab and irinotecan in recurrent pediatric glioblastoma. Pediatr. Int. 2015, 57, 169–171. [Google Scholar] [CrossRef]
- Aguilera, D.; Mazewski, C.; Hayes, L.; Jordan, C.; Esiashivilli, N.; Janns, A.; Macdonald, T.J. Prolonged Survival After Treatment of Diffuse Intrinsic Pontine Glioma with Radiation, Temozolamide, and Bevacizumab. J. Pediatr. Hematol. 2013, 35, e42–e46. [Google Scholar] [CrossRef]
- Hummel, T.R.; Salloum, R.; Drissi, R.; Kumar, S.; Sobo, M.; Goldman, S.; Pai, A.L.H.; Leach, J.; Lane, A.; Pruitt, D.; et al. A pilot study of bevacizumab-based therapy in patients with newly diagnosed high-grade gliomas and diffuse intrinsic pontine gliomas. J. Neuro-Oncol. 2015, 127, 53–61. [Google Scholar] [CrossRef]
- Grill, J.; Massimino, M.; Bouffet, E.; Azizi, A.A.; McCowage, G.; Cañete, A.; Saran, F.; le Deley, M.-C.; Varlet, P.; Morgan, P.S.; et al. Phase II, Open-Label, Randomized, Multicenter Trial (HERBY) of Bevacizumab in Pediatric Patients with Newly Diagnosed High-Grade Glioma. J. Clin. Oncol. 2018, 36, 951–958. [Google Scholar] [CrossRef]
- Su, J.M.; Murray, J.C.; McNall-Knapp, R.Y.; Bowers, D.C.; Shah, S.; Adesina, A.M.; Paulino, A.C.; Jo, E.; Mo, Q.; Baxter, P.A.; et al. A phase 2 study of valproic acid and radiation, followed by maintenance valproic acid and bevacizumab in children with newly diagnosed diffuse intrinsic pontine glioma or high-grade glioma. Pediatr. Blood Cancer 2020, 67, e28283. [Google Scholar] [CrossRef]
- Rokes, C.A.; Remke, M.; Guha-Thakurta, N.; Witt, O.; Korshunov, A.; Pfister, S.; Wolff, J.E. Sorafenib Plus Valproic Acid for Infant Spinal Glioblastoma. J. Pediatr. Hematol. 2010, 32, 511–514. [Google Scholar] [CrossRef]
- Wetmore, C.; Daryani, V.M.; Billups, C.A.; Boyett, J.M.; Leary, S.; Tanos, R.; Goldsmith, K.C.; Stewart, C.F.; Blaney, S.M.; Gajjar, A. Phase II evaluation of sunitinib in the treatment of recurrent or refractory high-grade glioma or ependymoma in children: A children’s Oncology Group Study ACNS1021. Cancer Med. 2016, 5, 1416–1424. [Google Scholar] [CrossRef]
- Truffaux, N.; Philippe, C.; Paulsson, J.; Andreiuolo, F.; Guerrini-Rousseau, L.; Cornilleau, G.; le Dret, L.; Richon, C.; Lacroix, L.; Puget, S.; et al. Preclinical evaluation of dasatinib alone and in combination with cabozantinib for the treatment of diffuse intrinsic pontine glioma. Neuro-Oncology 2014, 17, 953–964. [Google Scholar] [CrossRef]
- International Cancer Genome Consortium PedBrain Tumor Project; Bender, S.; Gronych, J.; Warnatz, H.-J.; Hutter, B.; Gröbner, S.; Ryzhova, M.; Pfaff, E.; Hovestadt, V.; Weinberg, F.; et al. Recurrent MET fusion genes represent a drug target in pediatric glioblastoma. Nat. Med. 2016, 22, 1314–1320. [Google Scholar] [CrossRef]
- Scholz, A.; Harter, P.N.; Cremer, S.; Yalcin, B.H.; Gurnik, S.; Yamaji, M.; di Tacchio, M.; Sommer, K.; Baumgarten, P.; Bähr, O.; et al. Endothelial cell-derived angiopoietin-2 is a therapeutic target in treatment-naive and bevacizumab-resistant glioblastoma. EMBO Mol. Med. 2016, 8, 39–57. [Google Scholar] [CrossRef] [PubMed]
- Leary, S.E.; Park, J.R.; Reid, J.M.; Ralya, A.T.; Baruchel, S.; Wu, B.; Roberts, T.P.L.; Liu, X.; Minard, C.G.; Fox, E.; et al. Pediatric Phase I Trial and Pharmacokinetic Study of Trebananib in Relapsed Solid Tumors, Including Primary Tumors of the Central Nervous System ADVL1115: A Children’s Oncology Group Phase I Consortium Report. Clin. Cancer Res. 2017, 23, 6062–6069. [Google Scholar] [CrossRef] [PubMed]
- Gururangan, S.; Fangusaro, J.; Poussaint, T.Y.; Onar-Thomas, A.; Gilbertson, R.J.; Vajapeyam, S.; Gajjar, A.; Goldman, S.; Friedman, H.S.; Packer, R.J.; et al. Lack of efficacy of bevacizumab + irinotecan in cases of pediatric recurrent ependymoma-a Pediatric Brain Tumor Consortium study. Neuro-Oncology 2012, 14, 1404–1412. [Google Scholar] [CrossRef]
- de Wire, M.; Fouladi, M.; Turner, D.C.; Wetmore, C.; Hawkins, C.; Jacobs, C.; Yuan, Y.; Liu, D.; Goldman, S.; Fisher, P.; et al. An open-label, two-stage, phase II study of bevacizumab and lapatinib in children with recurrent or refractory ependymoma: A collaborative ependymoma research network study (CERN). J. Neuro-Oncology 2015, 123, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Hosoya, T.; Kambe, A.; Nishimura, Y.; Sakamoto, M.; Maegaki, Y.; Kurosaki, M. Pediatric Case of Li-Fraumeni Syndrome Complicated with Supratentorial Anaplastic Ependymoma. World Neurosurg. 2018, 120, 125–128. [Google Scholar] [CrossRef]
- Bender, J.G.; Lee, A.; Reid, J.M.; Baruchel, S.; Roberts, T.; Voss, S.D.; Wu, B.; Ahern, C.H.; Ingle, A.M.; Harris, P.; et al. Phase I Pharmacokinetic and Pharmacodynamic Study of Pazopanib in Children with Soft Tissue Sarcoma and Other Refractory Solid Tumors: A Children’s Oncology Group Phase I Consortium Report. J. Clin. Oncol. 2013, 31, 3034–3043. [Google Scholar] [CrossRef]
- Chuk, M.; Widemann, B.C.; Minard, C.G.; Liu, X.; Kim, A.; Bernhardt, M.B.; Kudgus, R.A.; Reid, J.M.; Voss, S.D.; Blaney, S.; et al. A phase 1 study of cabozantinib in children and adolescents with recurrent or refractory solid tumors, including CNS tumors: Trial ADVL1211, a report from the Children’s Oncology Group. Pediatr. Blood Cancer 2018, 65, e27077. [Google Scholar] [CrossRef]
- Donson, A.M.; Amani, V.; Warner, E.A.; Griesinger, A.M.; Witt, D.A.; Levy, J.M.M.; Hoffman, L.; Hankinson, T.C.; Handler, M.H.; Vibhakar, R.; et al. Identification of FDA-Approved Oncology Drugs with Selective Potency in High-Risk Childhood Ependymoma. Mol. Cancer Ther. 2018, 17, 1984–1994. [Google Scholar] [CrossRef]
- Dutta, P.; Reddy, K.S.; Rai, A.; Madugundu, A.K.; Solanki, H.S.; Bhansali, A.; Radotra, B.D.; Kumar, N.; Collier, D.; Iacovazzo, D.; et al. Surgery, Octreotide, Temozolomide, Bevacizumab, Radiotherapy, and Pegvisomant Treatment of an AIP Mutation-Positive Child. J. Clin. Endocrinol. Metab. 2019, 104, 3539–3544. [Google Scholar] [CrossRef]
- Borghi, G.; Michaeli, O.; Salvador, H.; Alderete, D.; Ponce, N.F.; Zapotocky, M.; Hansford, J.R.; Malalasekera, V.S.; Toledano, H.; Maguire, B.; et al. Bevacizumab for NF2-associated vestibular schwannomas of childhood and adolescence. Pediatr. Blood Cancer 2020, 67, e28228. [Google Scholar] [CrossRef]
- de Pasquale, M.D.; de Ioris, M.A.; Gallo, A.; Mastronuzzi, A.; Crocoli, A.; Cozza, R.; Boldrini, R. Peripheral medulloepithelioma: A rare tumor with a potential target therapy. J. Transl. Med. 2014, 12, 49. [Google Scholar] [CrossRef] [PubMed]
- Cornelius, A.; Foley, J.; Bond, J.; Nagulapally, A.B.; Steinbrecher, J.; Hendricks, W.P.D.; Rich, M.; Yendrembam, S.; Bergendahl, G.; Trent, J.M.; et al. Molecular Guided Therapy Provides Sustained Clinical Response in Refractory Choroid Plexus Carcinoma. Front. Pharmacol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S. Targeting the Tumor Microenvironment in Neuroblastoma: Recent Advances and Future Directions. Cancers 2020, 12, 2057. [Google Scholar] [CrossRef]
- Ribatti, D. Anti-angiogenesis in neuroblastoma. Crit. Rev. Oncol. Hematol. 2013, 1028, 133–142. [Google Scholar] [CrossRef]
- Dungwa, J.V.; Uparkar, U.; May, M.T.; Ramani, P. Angiogenin up-regulation correlates with adverse clinicopathological and biological factors, increased microvascular density and poor patient outcome in neuroblastomas. Histopathology 2012, 60, 911–923. [Google Scholar] [CrossRef]
- Gheytanchi, E.; Mehrazma, M.; Madjd, Z. Expression of Ki-67, p53 and VEGF in Pediatric Neuroblastoma. Asian Pac. J. Cancer Prev. 2014, 15, 3065–3070. [Google Scholar] [CrossRef]
- Jakovljević, G.; Culić, S.; Stepan, J.; Kosuta, I.; Seiwerth, S. Relationship between tumor vascularity and vascular endothelial growth factor as prognostic factors for patients with neuroblastoma. Coll. Antropol. 2011, 35, 1071–1079. [Google Scholar]
- Zins, K.; Kovatchki, D.; Lucas, T.; Abraham, D. PlGF and VEGF-A Regulate Growth of High-Risk MYCN-Single Copy Neuroblastoma Xenografts via Different Mechanisms. Int. J. Mol. Sci. 2016, 17, 1613. [Google Scholar] [CrossRef]
- Nowicki, M.; Konwerska, A.; Ostalska-Nowicka, D.; Derwich, K.; Miśkowiak, B.; Kondraciuk, B.; Samulak, D.; Witt, M. Vascular endothelial growth factor (VEGF)-C a potent risk factor in children diagnosed with stadium 4 neuroblastoma. Folia Histochem. Cytobiol. 2009, 46, 493–499. [Google Scholar] [CrossRef][Green Version]
- Ćavar, S.; Jelašić, D.; Seiwerth, S.; Milošević, M.; Hutinec, Z.; Mišić, M. Endoglin (CD 105) as a potential prognostic factor in neuroblastoma. Pediatr. Blood Cancer 2015, 62, 770–775. [Google Scholar] [CrossRef] [PubMed]
- Segerström, L.; Fuchs, D.; Bäckman, U.; Holmquist, K.; Christofferson, R.; Azarbayjani, F. The Anti-VEGF Antibody Bevacizumab Potently Reduces the Growth Rate of High-Risk Neuroblastoma Xenografts. Pediatr. Res. 2006, 60, 576–581. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Modak, S.; Kushner, B.H.; Basu, E.; Roberts, S.S.; Cheung, N.-K.V. Combination of bevacizumab, irinotecan, and temozolomide for refractory or relapsed neuroblastoma: Results of a phase II study. Pediatr. Blood Cancer 2017, 64, e26448. [Google Scholar] [CrossRef] [PubMed]
- Dickson, P.V.; Hamner, J.B.; Sims, T.L.; Fraga, C.H.; Ng, C.Y.; Rajasekeran, S.; Hagedorn, N.L.; McCarville, M.B.; Stewart, C.F.; Davidoff, A.M. Bevacizumab-Induced Transient Remodeling of the Vasculature in Neuroblastoma Xenografts Results in Improved Delivery and Efficacy of Systemically Administered Chemotherapy. Clin. Cancer Res. 2007, 13, 3942–3950. [Google Scholar] [CrossRef] [PubMed]
- Sims, T.L.; Williams, R.F.; Ng, C.Y.; Rosati, S.F.; Spence, Y.; Davidoff, A.M. Bevacizumab suppresses neuroblastoma progression in the setting of minimal disease. Surgery 2008, 144, 269–275. [Google Scholar] [CrossRef][Green Version]
- Hartwich, J.; Orr, W.S.; Ng, C.Y.; Spence, Y.; Morton, C.; Davidoff, A.M. HIF-1α activation mediates resistance to anti-angiogenic therapy in neuroblastoma xenografts. J. Pediatr. Surg. 2013, 48, 39–46. [Google Scholar] [CrossRef]
- Patterson, D.M.; Gao, D.; Trahan, D.N.; Johnson, B.A.; Ludwig, A.; Barbieri, E.; Chen, Z.; Diaz-Miron, J.; Vassilev, L.; Shohet, J.M.; et al. Effect of MDM2 and vascular endothelial growth factor inhibition on tumor angiogenesis and metastasis in neuroblastoma. Angiogenesis 2011, 14, 255–266. [Google Scholar] [CrossRef]
- Iyer, R.; Evans, A.E.; Qi, X.; Ho, R.; Minturn, J.E.; Zhao, H.; Balamuth, N.; Maris, J.M.; Brodeur, G.M. Lestaurtinib Enhances the Antitumor Efficacy of Chemotherapy in Murine Xenograft Models of Neuroblastoma. Clin. Cancer Res. 2010, 16, 1478–1485. [Google Scholar] [CrossRef]
- Bocca, P.; di Carlo, E.; Caruana, I.; Emionite, L.; Cilli, M.; de Angelis, B.; Quintarelli, C.; Pezzolo, A.; Raffaghello, L.; Morandi, F.; et al. Bevacizumab-mediated tumor vasculature remodelling improves tumor infiltration and antitumor efficacy of GD2-CAR T cells in a human neuroblastoma preclinical model. OncoImmunology 2017, 7, e1378843. [Google Scholar] [CrossRef]
- Zhu, R.; Wang, Z.; Liang, P.; He, X.; Zhuang, X.; Huang, R.; Wang, M.; Wang, Q.; Qian, Y.; Wang, S. Efficient VEGF targeting delivery of DOX using Bevacizumab conjugated SiO2@LDH for anti-neuroblastoma therapy. Acta Biomater. 2017, 63, 163–180. [Google Scholar] [CrossRef]
- Glincher, R.; Price, A.P.; Laquaglia, M.P.; Kushner, B.H.; Modak, S. Bevacizumab-associated Bowel Microperforation in a Patient with Neuroblastoma. J. Pediatr. Hematol. 2018, 40, e394–e396. [Google Scholar] [CrossRef] [PubMed]
- Lowery, C.D.; Blosser, W.; Dowless, M.; Renschler, M.; Perez, L.V.; Stephens, J.; Pytowski, B.; Wasserstrom, H.; Stancato, L.F.; Falcon, B. Anti-VEGFR2 therapy delays growth of preclinical pediatric tumor models and enhances anti-tumor activity of chemotherapy. Oncotarget 2019, 10, 5523–5533. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Mokhtari, R.B.; Sheikh, R.; Wu, B.; Zhang, L.; Xu, P.; Man, S.; Oliveira, I.D.; Yeger, H.; Kerbel, R.S.; et al. Metronomic Oral Topotecan with Pazopanib Is an Active Antiangiogenic Regimen in Mouse Models of Aggressive Pediatric Solid Tumor. Clin. Cancer Res. 2011, 17, 5656–5667. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Mokhtari, R.B.; Oliveira, I.D.; Islam, S.; Toledo, S.R.C.; Yeger, H.; Baruchel, S. Tumor Dynamics in Response to Antiangiogenic Therapy with Oral Metronomic Topotecan and Pazopanib in Neuroblastoma Xenografts. Transl. Oncol. 2013, 6, 493–503. [Google Scholar] [CrossRef][Green Version]
- Rössler, J.K.; Monnet, Y.; Farace, F.; Opolon, P.; Daudigeos-Dubus, E.; Bourredjem, A.; Vassal, G.; Geoerger, B. The selective VEGFR1-3 inhibitor axitinib (AG-013736) shows antitumor activity in human neuroblastoma xenografts. Int. J. Cancer 2010, 128, 2748–2758. [Google Scholar] [CrossRef]
- Sidarovich, V.; de Mariano, M.; Aveic, S.; Pancher, M.; Adami, V.; Gatto, P.; Pizzini, S.; Pasini, L.; Croce, M.; Parodi, F.; et al. A High-Content Screening of Anticancer Compounds Suggests the Multiple Tyrosine Kinase Inhibitor Ponatinib for Repurposing in Neuroblastoma Therapy. Mol. Cancer Ther. 2018, 17, 1405–1415. [Google Scholar] [CrossRef]
- Maris, J.M.; Courtright, J.; Houghton, P.J.; Morton, C.L.; Kolb, E.A.; Lock, R.B.; Tajbakhsh, M.; Reynolds, C.P.; Keir, S.T.; Wu, J.; et al. Initial testing (stage 1) of sunitinib by the pediatric preclinical testing program. Pediatr. Blood Cancer 2008, 51, 42–48. [Google Scholar] [CrossRef]
- Yang, F.; Jove, V.; Buettner, R.; Xin, H.; Wu, J.; Wang, Y.; Nam, S.; Xu, Y.; Ara, T.; de Clerck, Y.A.; et al. Sorafenib inhibits endogenous and IL-6/S1P induced JAK2-STAT3 signaling in human neuroblastoma, associated with growth suppression and apoptosis. Cancer Biol. Ther. 2012, 13, 534–541. [Google Scholar] [CrossRef]
- Kakodkar, N.C.; Peddinti, R.R.; Tian, Y.; Bs, L.J.G.; Chlenski, A.; Yang, Q.; Bs, H.R.S.; Maitland, M.L.; Cohn, S.L. Sorafenib inhibits neuroblastoma cell proliferation and signaling, blocks angiogenesis, and impairs tumor growth. Pediatr. Blood Cancer 2012, 59, 642–647. [Google Scholar] [CrossRef]
- Chai, H.; Luo, A.Z.; Weerasinghe, P.; Brown, R.E. Sorafenib downregulates ERK/Akt and STAT3 survival pathways and induces apoptosis in a human neuroblastoma cell line. Int. J. Clin. Exp. Pathol. 2010, 3, 408–415. [Google Scholar]
- Calero, R.; Morchon, E.; Johnsen, J.I.; Serrano, R. Sunitinib Suppress Neuroblastoma Growth through Degradation of MYCN and Inhibition of Angiogenesis. PLoS ONE 2014, 9, e95628. [Google Scholar] [CrossRef]
- Nilsson, M.B.; Zage, P.E.; Zeng, L.; Xu, L.; Cascone, T.; Wu, H.K.; Saigal, B.; Zweidler-McKay, P.A.; Heymach, J.V. Multiple receptor tyrosine kinases regulate HIF-1α and HIF-2α in normoxia and hypoxia in neuroblastoma: Implications for antiangiogenic mechanisms of multikinase inhibitors. Oncogene 2010, 29, 2938–2949. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Smith, K.M.; Chong, A.L.; Stempak, D.; Yeger, H.; Marrano, P.; Thorner, P.S.; Irwin, M.S.; Kaplan, D.R.; Baruchel, S. In Vivo Antitumor and Antimetastatic Activity of Sunitinib in Preclinical Neuroblastoma Mouse Model. Neoplasia 2009, 11, 426–435. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Sun, J.D.; Zhang, L.; Mokhtari, R.B.; Wu, B.; Meng, F.; Liu, Q.; Bhupathi, D.; Wang, Y.; Yeger, H.; et al. Hypoxia-Targeting Drug Evofosfamide (TH-302) Enhances Sunitinib Activity in Neuroblastoma Xenograft Models. Transl. Oncol. 2018, 11, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Okada, K.; Nakano, Y.; Yamasaki, K.; Nitani, C.; Fujisaki, H.; Hara, J. Sorafenib treatment in children with relapsed and refractory neuroblastoma: An experience of four cases. Cancer Med. 2016, 5, 1947–1949. [Google Scholar] [CrossRef] [PubMed]
- Beppu, K.; Jaboine, J.; Merchant, M.S.; Mackall, C.L.; Thiele, C.J. Effect of Imatinib Mesylate on Neuroblastoma Tumorigenesis and Vascular Endothelial Growth Factor Expression. J. Natl. Cancer Inst. 2004, 96, 46–55. [Google Scholar] [CrossRef]
- Palmberg, E.; Johnsen, J.I.; Paulsson, J.; Gleissman, H.; Wickström, M.; Edgren, M.; Östman, A.; Kogmer, P.; Lindskog, M. Metronomic scheduling of imatinib abrogates clonogenicity of neuroblastoma cells and enhances their susceptibility to selected chemotherapeutic drugs in vitro and in vivo. Int. J. Cancer 2009, 124, 1227–1234. [Google Scholar] [CrossRef]
- Timeus, F.; Crescenzio, N.; Doria, A.; Foglia, L.; Pagliano, S.; Ricotti, E.; Fagioli, F.; Tovo, P.-A.; di Montezemolo, L.C. In vitro anti-neuroblastoma activity of saquinavir and its association with imatinib. Oncol. Rep. 2011, 27, 734–740. [Google Scholar] [CrossRef]
- Rössler, J.K.; Zambrzycka, I.; Lagodny, J.; Kontny, U.; Niemeyer, C.M. Effect of STI-571 (imatinib mesylate) in combination with retinoic acid and γ-irradiation on viability of neuroblastoma cells. Biochem. Biophys. Res. Commun. 2006, 342, 1405–1412. [Google Scholar] [CrossRef]
- Neumann, I.; Foell, J.L.; Bremer, M.; Volkmer, I.; Korholz, D.; Burdach, S.; Staege, M.S. Retinoic acid enhances sensitivity of neuroblastoma cells for imatinib mesylate. Pediatr. Blood Cancer 2010, 55, 464–470. [Google Scholar] [CrossRef]
- Bond, M.; Bernstein, M.L.; Pappo, A.; Schultz, K.R.; Krailo, M.; Blaney, S.M.; Adamson, P.C. A phase II study of imatinib mesylate in children with refractory or relapsed solid tumors: A Children’s Oncology Group study. Pediatr. Blood Cancer 2007, 50, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Calafiore, L.; Amoroso, L.; Alberighi, O.D.C.; Luksch, R.; Zanazzo, G.; Castellano, A.; Podda, M.; Dominici, C.; Haupt, R.; Corrias, M.V.; et al. Two-stage phase II study of imatinib mesylate in subjects with refractory or relapsing neuroblastoma. Ann. Oncol. 2013, 24, 1406–1413. [Google Scholar] [CrossRef] [PubMed]
- Morandi, F.; Amoroso, L.; Dondero, A.; Castriconi, R.; Parodi, S.; Luksch, R.; Casale, F.; Castellano, A.; Garaventa, A.; Moretta, A.; et al. Updated clinical and biological information from the two-stage phase II study of imatinib mesylate in subjects with relapsed/refractory neuroblastoma. OncoImmunology 2018, 7, e1468953. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Scorsone, K.A.; Woodfield, S.E.; Zage, P.E. Sensitivity of neuroblastoma to the novel kinase inhibitor cabozantinib is mediated by ERK inhibition. Cancer Chemother. Pharmacol. 2015, 76, 977–987. [Google Scholar] [CrossRef]
- Perisa, M.P.; Storey, M.; Streby, K.A.; Ranalli, M.A.; Skeens, M.; Shah, N. Cabozantinib for relapsed neuroblastoma: Single institution case series. Pediatr. Blood Cancer 2020, 67, e28317. [Google Scholar] [CrossRef]
- Wu, H.-W.; Sheard, M.A.; Malvar, J.; Fernandez, G.E.; Declerck, Y.A.; Blavier, L.; Shimada, H.; Theuer, C.P.; Sposto, R.; Seeger, R.C. Anti-CD105 Antibody Eliminates Tumor Microenvironment Cells and Enhances Anti-GD2 Antibody Immunotherapy of Neuroblastoma with Activated Natural Killer Cells. Clin. Cancer Res. 2019, 25, 4761–4774. [Google Scholar] [CrossRef]
- Leslie, S.W.; Sajjad, H.; Murphy, P.B. Wilms Tumor; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Yasemin, O.; Isin, K.; Güllüoğlu, M.G.; Inci, A.; Veli, U.; Özlük, Y.; Kilicaslan, I.; Ayan, I.; Uysal, V. The prognostic significance of angiogenesis and the effect of vascular endothelial growth factor on angiogenic process in Wilms’ tumour. Pathology 2006, 38, 408–414. [Google Scholar] [CrossRef]
- Abramson, L.P.; Grundy, P.E.; Rademaker, A.W.; Helenowski, I.; Cornwell, M.; Katzenstein, H.M.; Reynolds, M.; Arensman, R.M.; Crawford, S.E. Increased microvascular density predicts relapse in Wilms’ tumor. J. Pediatr. Surg. 2003, 38, 325–330. [Google Scholar] [CrossRef]
- Ghanem, M.A.; van Steenbrugge, G.J.; Sudaryo, M.K.; Mathoera, R.B.; Nijman, J.M.; van der Kwast, T.H. Expression and prognostic relevance of vascular endothelial growth factor (VEGF) and its receptor (FLT-1) in nephroblastoma. J. Clin. Pathol. 2003, 56, 107–113. [Google Scholar] [CrossRef]
- Nowicki, M.; Ostalska-Nowicka, D.; Kaczmarek, M.; Miskowiak, B.; Witt, M. The significance of VEGF-C/VEGFR-2 interaction in the neovascularization and prognosis of nephroblastoma (Wilms’ tumour). Histopathology 2007, 50, 358–364. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, D.; Chen, X.-R.; Fan, Y.; Wang, J. Expression of vascular endothelial growth factor (VEGF) and VEGF-C in serum and tissue of Wilms tumor. Chin. Med. J. 2011, 124, 3716–3720. [Google Scholar] [PubMed]
- Benesch, M.; Windelberg, M.; Sauseng, W.; Witt, V.; Fleischhack, G.; Lackner, H.; Gadner, H.; Bode, U.; Urban, C. Compassionate use of bevacizumab (Avastin®) in children and young adults with refractory or recurrent solid tumors. Ann. Oncol. 2007, 19, 807–813. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Burney, I.A.; Al-Moundhri, M.S. Near complete resolution of refractory, relapsed, metastatic Wilms’ tumour in an adolescent with bevacizumab. J. Coll. Physicians Surg. Pak. 2014, 2, 5. [Google Scholar]
- Schiavetti, A.; Varrasso, G.; Collini, P.; Clerico, A. Vincristine, Irinotecan, and Bevacizumab in Relapsed Wilms Tumor with Diffuse Anaplasia. J. Pediatr. Hematol. 2018, 40, 331–333. [Google Scholar] [CrossRef]
- Venkatramani, R.; Malogolowkin, M.H.; Mascarenhas, L. Treatment of multiply relapsed wilms tumor with vincristine, irinotecan, temozolomide and bevacizumab. Pediatr. Blood Cancer 2014, 61, 756–759. [Google Scholar] [CrossRef]
- Venkatramani, R.; Malogolowkin, M.; Davidson, T.B.; May, W.; Sposto, R.; Mascarenhas, L. A Phase I Study of Vincristine, Irinotecan, Temozolomide and Bevacizumab (Vitb) in Pediatric Patients with Relapsed Solid Tumors. PLoS ONE 2013, 8, e68416. [Google Scholar] [CrossRef]
- Interiano, R.B.; McCarville, M.B.; Wu, J.; Davidoff, A.M.; Sandoval, J.; Navid, F. Pneumothorax as a complication of combination antiangiogenic therapy in children and young adults with refractory/recurrent solid tumors. J. Pediatr. Surg. 2015, 50, 1484–1489. [Google Scholar] [CrossRef][Green Version]
- Kim, A.; Widemann, B.C.; Krailo, M.; Ms, N.J.; Fox, E.; Weigel, B.J.; Blaney, S.M. Phase 2 trial of sorafenib in children and young adults with refractory solid tumors: A report from the Children’s Oncology Group. Pediatr. Blood Cancer 2015, 62, 1562–1566. [Google Scholar] [CrossRef]
- Anderson, B.; Jasty-Rao, R.; Wu, Y.-M.; Paul, T.; Robinson, D.; Mody, R. Exceptional Response to Cabozantinib in a Patient with Multiply Relapsed Wilms Tumor. JCO Precis. Oncol. 2018, 2, 1–5. [Google Scholar] [CrossRef]
- Kirschner, K.M.; Sciesielski, L.K.; Scholz, H. Wilms’ tumour protein Wt1 stimulates transcription of the gene encoding vascular endothelial cadherin. Pflüg. Arch. Eur. J. Physiol. 2010, 460, 1051–1061. [Google Scholar] [CrossRef]
- Giordano, G.; Campanini, N.; Rocco, A.; Donofrio, V.; Bertolini, P.; Falleti, J.; Pettinato, G. C-kit protein expression in Wilms’ tumour: An immunohistochemical study. Eur. J. Surg. Oncol. 2009, 35, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Thompson, P.A.; Chintagumpala, M. Targeted Therapy in Bone and Soft Tissue Sarcoma in Children and Adolescents. Curr. Oncol. Rep. 2012, 14, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Lautz, T.B.; Hayes-Jordan, A.A. Recent progress in pediatric soft tissue sarcoma therapy. Semin. Pediatr. Surg. 2019, 28, 150862. [Google Scholar] [CrossRef] [PubMed]
- Dubois, S.; Demetri, G.D. Markers of angiogenesis and clinical features in patients with sarcoma. Cancer 2007, 109, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Potti, A.; Ganti, A.K.; Tendulkar, K.; Sholes, K.; Chitajallu, S.; Koch, M.; Kargas, S. Determination of vascular endothelial growth factor (VEGF) overexpression in soft tissue sarcomas and the role of overexpression in leiomyosarcoma. J. Cancer Res. Clin. Oncol. 2004, 130, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Wachtel, M.; Schäfer, B.W. Targets for cancer therapy in childhood sarcomas. Cancer Treat. Rev. 2010, 36, 318–327. [Google Scholar] [CrossRef]
- Chisholm, J.C.; Merks, J.H.M.; Casanova, M.; Bisogno, G.; Orbach, D.; Gentet, J.-C.; Thomassin-Defachelles, A.-S.; Chastagner, P.; Lowis, S.; Ronghe, M.; et al. Open-label, multicentre, randomised, phase II study of the EpSSG and the ITCC evaluating the addition of bevacizumab to chemotherapy in childhood and adolescent patients with metastatic soft tissue sarcoma (the BERNIE study). Eur. J. Cancer 2017, 83, 177–184. [Google Scholar] [CrossRef]
- Bender, J.G.; Blaney, S.M.; Borinstein, S.; Reid, J.M.; Baruchel, S.; Ahern, C.; Ingle, A.M.; Yamashiro, D.J.; Chen, A.; Weigel, B.; et al. A Phase I Trial and Pharmacokinetic Study of Aflibercept (VEGF Trap) in Children with Refractory Solid Tumors: A Children’s Oncology Group Phase I Consortium Report. Clin. Cancer Res. 2012, 18, 5081–5089. [Google Scholar] [CrossRef]
- Ferguson, M.; Hingorani, P.; Gupta, A.A. Emerging molecular-targeted therapies in early-phase clinical trials and preclinical models. Am. Soc. Clin. Oncol. Annu. Meet. 2013, 33, 420–424. [Google Scholar]
- Vo, K.T.; Matthay, K.K.; Dubois, S.G. Targeted antiangiogenic agents in combination with cytotoxic chemotherapy in preclinical and clinical studies in sarcoma. Clin. Sarcoma Res. 2016, 6, 1–11. [Google Scholar] [CrossRef]
- Russo, I.; di Paolo, V.; Crocoli, A.; Mastronuzzi, A.; Serra, A.; di Paolo, P.L.; di Giannatale, A.; Miele, E.; Milano, G.M. A Chart Review on the Feasibility and Safety of the Vincristine Irinotecan Pazopanib (VIPaz) Association in Children and Adolescents With Resistant or Relapsed Sarcomas. Front. Oncol. 2020, 10, 1228. [Google Scholar] [CrossRef]
- Paulino, A.C.; Pappo, A. Alveolar rhabdomyosarcoma of the extremity and nodal metastasis: Is the in-transit lymphatic system at risk? Pediatr. Blood Cancer 2009, 53, 1332–1333. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, E.; Nishijo, K.; McCleish, A.T.; Michalek, J.E.; Grayson, M.H.; Infante, A.J.; Abboud, H.E.; Legallo, R.D.; Qualman, S.J.; Rubin, B.P.; et al. PDGFR-A is a therapeutic target in alveolar rhabdomyosarcoma. Oncogene 2008, 27, 6550–6560. [Google Scholar] [CrossRef]
- McDowell, H.P.; Meco, D.; Riccardi, A.; Tanno, B.; Berardi, A.C.; Raschellà, G.; Riccardi, R.; Dominici, C. Imatinib mesylate potentiates topotecan antitumor activity in rhabdomyosarcoma preclinical models. Int. J. Cancer 2007, 120, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- Gruenwald, V.; Länger, F.; Raatschen, H.-J.; Beilken, A. Proof of principle for bevacizumab activity in desmoid-type fibromatosis. Clin. Sarcoma Res. 2016, 6, 5. [Google Scholar] [CrossRef]
- Azizi, A.A.; Haberler, C.; Czech, T.; Gupper, A.; Prayer, D.; Breitschopf, H.; Acker, T.; Slavc, I. Vascular-endothelial-growth-factor (VEGF) expression and possible response to angiogenesis inhibitor bevacizumab in metastatic alveolar soft part sarcoma. Lancet Oncol. 2006, 7, 521–523. [Google Scholar] [CrossRef]
- Conde, N.; Cruz, O.; Albert, A.; Mora, J. Antiangiogenic treatment as a pre-operative management of alveolar soft-part sarcoma. Pediatr. Blood Cancer 2011, 57, 1071–1073. [Google Scholar] [CrossRef] [PubMed]
- Hingorani, P.; Eshun, F.; White-Collins, A.; Watanabe, M. Gemcitabine, Docetaxel, and Bevacizumab in Relapsed and Refractory Pediatric Sarcomas. J. Pediatr. Hematol. 2012, 34, 524–527. [Google Scholar] [CrossRef] [PubMed]
- Jeng, M.; Fuh, B.; Blatt, J.; Gupta, A.; Merrow, A.C.; Hammill, A.; Adams, D. Malignant transformation of infantile hemangioma to angiosarcoma: Response to chemotherapy with bevacizumab. Pediatr. Blood Cancer 2014, 61, 2115–2117. [Google Scholar] [CrossRef] [PubMed]
- Federico, S.M.; Caldwell, K.J.; McCarville, M.B.; Daryani, V.M.; Stewart, C.F.; Mao, S.; Wu, J.; Davidoff, A.M.; Santana, V.M.; Furman, W.L.; et al. Phase I expansion cohort to evaluate the combination of bevacizumab, sorafenib and low-dose cyclophosphamide in children and young adults with refractory or recurrent solid tumours. Eur. J. Cancer 2020, 132, 35–42. [Google Scholar] [CrossRef]
- Casanova, M.; Basso, E.; Magni, C.; Bergamaschi, L.; Chiaravalli, S.; Carta, R.; Tirtei, E.; Massimino, M.; Fagioli, F.; Ferrari, A. Response to pazopanib in two pediatric patents with pretreated relapsing synovial sarcoma. Tumori 2017, 103, e1–e3. [Google Scholar] [CrossRef] [PubMed]
- Menegaz, B.A.; Cuglievan, B.; Benson, J.; Camacho, P.; Lamhamedi-Cherradi, S.; Leung, C.H.; Warneke, C.L.; Huh, W.; Subbiah, V.; Benjamin, R.S.; et al. Clinical Activity of Pazopanib in Patients with Advanced Desmoplastic Small Round Cell Tumor. Oncology 2018, 23, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Yanagisawa, R.; Noguchi, M.; Fujita, K.; Sakashita, K.; Sano, K.; Ogiso, Y.; Katsuyama, Y.; Kondo, Y.; Komori, K.; Fujihara, I.; et al. Preoperative Treatment with Pazopanib in a Case of Chemotherapy-Resistant Infantile Fibrosarcoma. Pediatr. Blood Cancer 2016, 63, 348–351. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Kurata, T.; Sano, K.; Suzuki, S.; Kaneko, T.; Motobayashi, M.; Shigemura, T.; Sumi, T.; Koike, K.; Nakazawa, Y. Dramatic Reduction in Tumor Size During 5 Months of Pazopanib Therapy in Combination with Ifosfamide, Carboplatin, and Etoposide in an Early Infant With Progressive Soft Tissue Sarcoma. J. Pediatr. Hematol. 2017, 39, 154–156. [Google Scholar] [CrossRef]
- Funakoshi, Y.; Okada, M.; Kawata, S.; Ito, N.; Abe, K.; Moriuchi, H. The Significant Effects of Pazopanib on Multiple Pulmonary Metastatic Lesions of Alveolar Soft Part Sarcoma: A Case Report. J. Pediatr. Hematol. Oncol. 2017, 39, 238–239. [Google Scholar] [CrossRef]
- Weiss, A.R.; Chen, Y.-L.; Scharschmidt, T.J.; Chi, Y.-Y.; Tian, J.; Black, J.O.; Davis, J.L.; Fanburg-Smith, J.C.; Zambrano, E.; Anderson, J.; et al. Pathological response in children and adults with large unresected intermediate-grade or high-grade soft tissue sarcoma receiving preoperative chemoradiotherapy with or without pazopanib (ARST1321): A multicentre, randomised, open-label, phase 2 trial. Lancet Oncol. 2020, 21, 1110–1122. [Google Scholar] [CrossRef]
- Geller, J.; Fox, E.; Turpin, B.K.; Goldstein, S.L.; Liu, X.; Minard, C.G.; Kudgus, R.A.; Reid, J.M.; Berg, S.L.; Weigel, B.J. A study of axitinib, a VEGF receptor tyrosine kinase inhibitor, in children and adolescents with recurrent or refractory solid tumors: A Children’s Oncology Group phase 1 and pilot consortium trial (ADVL1315). Cancer 2018, 124, 4548–4555. [Google Scholar] [CrossRef]
- Hilbert, M.; Mary, P.; Larroquet, M.; Serinet, M.O.; Helfre, S.; Brisse, H.; Coulomb, A.; Orbach, D. Alveolar soft part sarcoma in childhood: Is Sunitinib-Sutent® treatment an effective approach? Pediatr. Blood Cancer 2012, 58, 475–476. [Google Scholar] [CrossRef]
- Orbach, D.; Brennan, B.; Casanova, M.; Bergeron, C.; Mosseri, V.; Francotte, N.; van Noesel, M.; Rey, A.; Bisogno, G.; Pierron, G.; et al. Paediatric and adolescent alveolar soft part sarcoma: A joint series from European cooperative groups. Pediatr. Blood Cancer 2013, 60, 1826–1832. [Google Scholar] [CrossRef]
- Malouf, G.G.; Beinse, G.; Adam, J.; Mir, O.; Chamseddine, A.N.; Terrier, P.; Honoré, C.; Spano, J.; Italiano, A.; Kurtz, J.; et al. Brain Metastases and Place of Antiangiogenic Therapies in Alveolar Soft Part Sarcoma: A Retrospective Analysis of the French Sarcoma Group. Oncology 2019, 24, 980–988. [Google Scholar] [CrossRef]
- Flores, R.J.; Harrison, D.J.; Federman, N.C.; Furman, W.L.; Huh, W.W.; Broaddus, E.G.; Okcu, M.F.; Venkatramani, R. Alveolar soft part sarcoma in children and young adults: A report of 69 cases. Pediatr. Blood Cancer 2018, 65, e26953. [Google Scholar] [CrossRef] [PubMed]
- Reed, D.R.; Mascarenhas, L.; Manning, K.; Hale, G.A.; Goldberg, J.; Gill, J.; Sandler, E.; Isakoff, M.S.; Smith, T.; Caracciolo, J.; et al. Pediatric phase I trial of oral sorafenib and topotecan in refractory or recurrent pediatric solid malignancies. Cancer Med. 2015, 5, 294–303. [Google Scholar] [CrossRef] [PubMed]
- de Sanctis, R.; Bertuzzi, A.F.; Bisogno, G.; Carli, M.; Ferrari, A.; Comandone, A.; Santoro, A. Imatinib mesylate in desmoplastic small round cell tumors. Future Oncol. 2017, 13, 1233–1237. [Google Scholar] [CrossRef] [PubMed]
- Chugh, R.; Wathen, J.K.; Patel, S.R.; Maki, R.G.; Meyers, P.A.; Schuetze, S.M.; Priebat, D.A.; Thomas, D.; Jacobson, J.A.; Samuels, B.L.; et al. Efficacy of Imatinib in Aggressive Fibromatosis: Results of a Phase II Multicenter Sarcoma Alliance for Research through Collaboration (SARC) Trial. Clin. Cancer Res. 2010, 16, 4884–4891. [Google Scholar] [CrossRef] [PubMed]
- Posso-De Los Rios, C.J.; Lara-Corrales, I.; Ho, N. Dermatofibrosarcoma protuberans in pediatric patients: A report of 17 cases. J. Cutan. Med. Surg. 2014, 18, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Price, V.E.; Fletcher, J.A.; Zielenska, M.; Cole, W.; Viero, S.; Manson, D.E.; Stuart, M.; Pappo, A.S. Imatinib mesylate: An attractive alternative in young children with large, surgically challenging dermatofibrosarcoma protuberans. Pediatr. Blood Cancer 2005, 44, 511–515. [Google Scholar] [CrossRef]
- Gooskens, S.L.M.; Oranje, A.P.; van Adrichem, L.N.A.; de Waard-van der Spek, F.B.; den Hollander, J.C.; van de Ven, C.P.; van den Heuvel-Eibrink, M.M. Imatinib mesylate for children with dermatofibrosarcoma protuberans (DFSP). Pediatr. Blood Cancer 2010, 55, 369–373. [Google Scholar] [CrossRef]
- Goldberg, J.M.; Gavcovich, T.; Saigal, G.; Goldman, J.W.; Rosen, L.S. Extended Progression-Free Survival in Two Patients with Alveolar Soft Part Sarcoma Exposed to Tivantinib. J. Clin. Oncol. 2014, 32, e114–e116. [Google Scholar] [CrossRef]
- Danieau, G.; Morice, S.; Rédini, F.; Verrecchia, F.; Royer, B.B.-L.B.-L. New Insights about the Wnt/β-Catenin Signaling Pathway in Primary Bone Tumors and Their Microenvironment: A Promising Target to Develop Therapeutic Strategies? Int. J. Mol. Sci. 2019, 20, 3751. [Google Scholar] [CrossRef]
- Mikulić, D.; Ilić, I.; Cepulić, M.; Orlić, D.; Giljević, J.S.; Fattorini, I.; Seiwerth, S. Tumor angiogenesis and outcome in osteosarcoma. Pediatr. Hematol. Oncol. 2004, 21, 611–619. [Google Scholar] [CrossRef]
- Ren, T.; Qing, Y.; Dai, N.; Li, M.; Qian, C.; Yang, Y.; Cheng, Y.; Li, Z.; Zhang, S.; Zhong, Z.; et al. Apurinic/apyrimidinic endonuclease 1 induced upregulation of fibroblast growth factor 2 and its receptor 3 induces angiogenesis in human osteosarcoma cells. Cancer Sci. 2014, 105, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Mantadakis, E.; Kim, G.; Reisch, J.; McHard, K.; Maale, G.; Leavey, P.J.; Timmons, C. Lack of Prognostic Significance of Intratumoral Angiogenesis in Nonmetastatic Osteosarcoma. J. Pediatr. Hematol. 2001, 23, 286–289. [Google Scholar] [CrossRef] [PubMed]
- Kreuter, M.; Bieker, R.; Bielaek, S.S.; Auras, T.; Buerger, H.; Gosheger, G.; Jurgens, H.; Berdel, W.E.; Mesters, R.M. Prognostic relevance of increased angiogenesis in osteosarcoma. Clin. Cancer Res. 2004, 10, 8531–8537. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, F.; Zhang, Z.; Wang, D.; Cui, B.; Zeng, F.; Huang, L.; Zhang, Q.; Sun, Q. High expression levels of Cyr61 and VEGF are associated with poor prognosis in osteosarcoma. Pathol. Res. Pract. 2017, 213, 895–899. [Google Scholar] [CrossRef]
- Lammli, J.; Fan, M.; Rosenthal, H.G.; Patni, M.; Rinehart, E.; Vergara, G.; Ablah, E.; Wooley, P.H.; Lucas, G.; Yang, S.-Y. Expression of Vascular Endothelial Growth Factor correlates with the advance of clinical osteosarcoma. Int. Orthop. 2012, 36, 2307–2313. [Google Scholar] [CrossRef]
- Hoang, B.H.; Dyke, J.P.; Koutcher, J.A.; Huvos, A.G.; Mizobuchi, H.; Mazza, B.A.; Gorlick, R.; Healey, J.H. VEGF Expression in Osteosarcoma Correlates with Vascular Permeability by Dynamic MRI. Clin. Orthop. Relat. Res. 2004, 426, 32–38. [Google Scholar] [CrossRef]
- Tabone, M.D.; Brugières, L.; Piperno-Neumann, S.; Selva, M.A.; Marec-Bérard, P.; Pacquement, H.; Lervat, C.; Corradini, N.; Gentet, J.C.; Couderc, R.; et al. Prognostic impact of blood and urinary angiogenic factor levels at diagnosis and during treatment in patients with osteosarcoma: A prospective study. BMC Cancer 2017, 17, 1–10. [Google Scholar] [CrossRef]
- Kubo, T.; Piperdi, S.; Rosenblum, J.; Antonescu, C.R.; Chen, W.; Kim, H.-S.; Huvos, A.G.; Sowers, R.; Meyers, P.A.; Healey, J.H.; et al. Platelet-derived growth factor receptor as a prognostic marker and a therapeutic target for imatinib mesylate therapy in osteosarcoma. Cancer 2008, 112, 2119–2129. [Google Scholar] [CrossRef]
- Navid, F.; Santana, V.M.; Neel, M.; McCarville, M.B.; Shulkin, B.L.; Wu, J.; Billups, C.A.; Mao, S.; Daryani, V.M.; Stewart, C.F.; et al. A phase II trial evaluating the feasibility of adding bevacizumab to standard osteosarcoma therapy. Int. J. Cancer 2017, 141, 1469–1477. [Google Scholar] [CrossRef]
- Kuo, C.; Kent, P.M.; Logan, A.D.; Tamulonis, K.B.; Dalton, K.L.; Batus, M.; Fernandez, K.; McFall, R.E. Docetaxel, bevacizumab, and gemcitabine for very high risk sarcomas in adolescents and young adults: A single-center experience. Pediatr. Blood Cancer 2017, 64, e26265. [Google Scholar] [CrossRef]
- Subbiah, V.; Wagner, M.J.; McGuire, M.F.; Sarwari, N.M.; Devarajan, E.; Lewis, V.O.; Westin, S.; Kato, S.; Brown, R.E.; Anderson, P. Personalized comprehensive molecular profiling of high-risk osteosarcoma: Implications and limitations for precision medicine. Oncotarget 2015, 6, 40642–40654. [Google Scholar] [CrossRef] [PubMed]
- Penel-Page, M.; Ray-Coquard, I.; Larcade, J.; Girodet, M.; Bouclier, L.; Rogasik, M.; Corradini, N.; Entz-Werle, N.; Brugieres, L.; Domont, J.; et al. Off-label use of targeted therapies in osteosarcomas: Data from the French registry OUTC’S (Observatoire de l’Utilisation des Thérapies Ciblées dans les Sarcomes). BMC Cancer 2015, 15, 854. [Google Scholar] [CrossRef] [PubMed]
- Umeda, K.; Kato, I.; Saida, S.; Okamoto, T.; Adachi, S. Pazopanib for second recurrence of osteosarcoma in pediatric patients. Pediatr. Int. 2017, 59, 937–938. [Google Scholar] [CrossRef] [PubMed]
- Czarnecka, A.M.; Synoradzki, K.; Firlej, W.; Bartnik, E.; Sobczuk, P.; Fiedorowicz, M.; Grieb, P.; Rutkowski, P. Molecular Biology of Osteosarcoma. Cancers 2020, 12, 2130. [Google Scholar] [CrossRef] [PubMed]
- Raciborska, A.; Bilska, K. Sorafenib in patients with progressed and refractory bone tumors. Med Oncol. 2018, 35, 126. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, A.E.; Walterhouse, D.O.; Leavey, P.; Reichek, J.; Walz, A.L. Prolonged response to sorafenib in a patient with refractory metastatic osteosarcoma and a somatic PDGFRA D846V mutation. Pediatr. Blood Cancer 2019, 66, e27493. [Google Scholar] [CrossRef]
- Stewart, E.; Federico, S.; Karlstrom, A.; Shelat, A.; Sablauer, A.; Pappo, A.; Dyer, M.A. The Childhood Solid Tumor Network: A new resource for the developmental biology and oncology research communities. Dev. Biol. 2015, 411, 287–293. [Google Scholar] [CrossRef]
- du Bois, S.G.; Marina, N.; Glade-Bender, J. Angiogenesis and vascular targeting in Ewing sarcoma: A review of preclinical and clinical data. Cancer 2010, 116, 749–757. [Google Scholar] [CrossRef]
- Mikulić, D.; Ilić, I.; Ćepulić, M.; Giljević, J.S.; Orlić, D.; Župančić, B.; Fattorini, I.; Seiwerth, S. Angiogenesis and Ewing sarcoma—Relationship to pulmonary metastasis and survival. J. Pediatr. Surg. 2006, 41, 524–529. [Google Scholar] [CrossRef]
- Wagner, L.; Turpin, B.; Nagarajan, R.; Weiss, B.D.; Cripe, T.; Geller, J. Pilot study of vincristine, oral irinotecan, and temozolomide (VOIT regimen) combined with bevacizumab in pediatric patients with recurrent solid tumors or brain tumors. Pediatr. Blood Cancer 2013, 60, 1447–1451. [Google Scholar] [CrossRef]
- Tamura, A.; Yamamoto, N.; Nino, N.; Ichikawa, T.; Nakatani, N.; Nakamura, S.; Saito, A.; Kozaki, A.; Kishimoto, K.; Ishida, T.; et al. Pazopanib maintenance therapy after tandem high-dose chemotherapy for disseminated Ewing sarcoma. Int. Cancer Conf. J. 2019, 8, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Nan, X.; Wang, J.; Cheng, H.; Yin, Z.; Sheng, J.; Qiu, B.; Lau, C.C.; Yustein, J.T.; Zhao, H.; Wong, S.T.C. Imatinib revives the therapeutic potential of metformin on ewing sarcoma by attenuating tumor hypoxic response and inhibiting convergent signaling pathways. Cancer Lett. 2019, 469, 195–206. [Google Scholar] [CrossRef]
- Wang, Y.; Mandal, D.; Wang, S.; Kleinerman, E.S.; Pollock, R.E.; Lev, D.; Hayes-Jordan, A. Platelet-derived growth factor receptor β inhibition increases tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) sensitivity: Imatinib and TRAIL dual therapy. Cancer 2010, 116, 3892–3902. [Google Scholar] [CrossRef]
- Merchant, M.S.; Woo, C.W.; Mackall, C.L.; Thiele, C.J. Potential use of imatinib in Ewing’s sarcoma: Evidence for in vitro and in vivo activity. J. Natl. Cancer Inst. 2002, 94, 1673–1679. [Google Scholar] [CrossRef] [PubMed]
- Scotlandi, K.; Manara, M.C.; Strammiello, R.; Landuzzi, L.; Benini, S.; Perdichizzi, S.; Serra, M.; Astolfi, A.; Nicoletti, G.; Lollini, P.-L.; et al. c-kit Receptor Expression in Ewing’s Sarcoma: Lack of Prognostic Value but Therapeutic Targeting Opportunities in Appropriate Conditions. J. Clin. Oncol. 2003, 21, 1952–1960. [Google Scholar] [CrossRef] [PubMed]
- Italiano, A.; Mir, O.; Mathoulin-Pelissier, S.; Penel, N.; Piperno-Neumann, S.; Bompas, E.; Chevreau, C.; Duffaud, F.; Entz-Werlé, N.; Saada, E.; et al. Cabozantinib in patients with advanced Ewing sarcoma or osteosarcoma (CABONE): A multicentre, single-arm, phase 2 trial. Lancet Oncol. 2020, 21, 446–455. [Google Scholar] [CrossRef]
- Voss, S.D.; Bender, J.G.; Spunt, S.L.; Dubois, S.G.; Widemann, B.C.; Park, J.R.; Leary, S.E.; Nelson, M.D.; Adamson, P.C.; Blaney, S.M.; et al. Growth plate abnormalities in pediatric cancer patients undergoing phase 1 anti-angiogenic therapy: A report from the Children’s Oncology Group Phase I Consortium. Pediatr. Blood Cancer 2014, 62, 45–51. [Google Scholar] [CrossRef]
- Ramjiawan, R.R.; Griffioen, A.W.; Duda, D.G. Anti-angiogenesis for cancer revisited: Is there a role for combinations with immunotherapy? Angiogenesis 2017, 20, 185–204. [Google Scholar] [CrossRef]
- Jain, R.K. Normalization of Tumor Vasculature: An Emerging Concept in Antiangiogenic Therapy. Science 2005, 307, 58–62. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ollauri-Ibáñez, C.; Astigarraga, I. Use of Antiangiogenic Therapies in Pediatric Solid Tumors. Cancers 2021, 13, 253. https://doi.org/10.3390/cancers13020253
Ollauri-Ibáñez C, Astigarraga I. Use of Antiangiogenic Therapies in Pediatric Solid Tumors. Cancers. 2021; 13(2):253. https://doi.org/10.3390/cancers13020253
Chicago/Turabian StyleOllauri-Ibáñez, Claudia, and Itziar Astigarraga. 2021. "Use of Antiangiogenic Therapies in Pediatric Solid Tumors" Cancers 13, no. 2: 253. https://doi.org/10.3390/cancers13020253
APA StyleOllauri-Ibáñez, C., & Astigarraga, I. (2021). Use of Antiangiogenic Therapies in Pediatric Solid Tumors. Cancers, 13(2), 253. https://doi.org/10.3390/cancers13020253