Ectopic Expression of Ankrd2 Affects Proliferation, Motility and Clonogenic Potential of Human Osteosarcoma Cells
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Cultures, Transfection and Cellular Treatments
2.2. Plasmids
2.3. Stable Silencing
2.4. RT-PCR and qPCR
2.5. Cell Proliferation/Viability
2.6. Cell Motility Assays
- Wound healing assay: Cells were seeded at 70% of confluence. Once confluence was reached, three wounds (“scratches”) were applied to the monolayer using a 200 µL pipette tip. The wounds were observed and captured in triplicate at the time of wounding (T0) and after 7, 24 and 48 h, under light microscopy using a microscope (Carl Zeiss, Munich, Germany) equipped with an AxioVision digital camera. The width of the scratches was measured using the AxioVision Rel 4.7 Software and the percentage (%) of the wound closure calculated according the formula: ((Tn-T0)/T0) × 100, where T0 is the width of the scratch at the beginning and Tn is the width at 7, 24 and 48 h after the scratch.
- Transwell migration assay: Sub-confluent flasks of Ankrd2-expressing OS-derived cells and their respective controls were starved overnight in IMDM GlutaMAX without FCS. The next day, 2.5 × 105 cells were added to the upper side of the Boyden chamber (Cell Biolabs, Inc., San Diego, CA USA) in triplicate in serum-free medium. FBS-conditioned medium was added to the lower chambers. After 24 h of incubation at 37 °C, non-migrated cells were carefully removed from the upper side of the chamber. Cells that had migrated to the bottom side of the chamber were fixed, and stained with crystal violet and destained. The OD560 was measured on a Tecan Infinite M200 Pro spectrophotometer (Tecan, Männedorf, Switzerland).
2.7. Clonogenic Assay
2.8. Anchorage-Independent Growth Assay
2.9. Protein Extracts and Immunoblot
2.10. Immunofluorescence
2.11. Gelatin Zymography
2.12. Image Processing and Statistical Analysis
3. Results
3.1. Ankrd2 is Expressed in Human Osteosarcoma Cell Lines
3.2. Characterization of Clones of OS Cell Lines Stably Expressing wt and S99A Ankrd2
3.3. Effect of Ankrd2 Overexpression on the Proliferation of OS Cells
3.4. Effect of Ankrd2 Overexpression on OS Cellular Motility
3.4.1. Wound Healing
3.4.2. Chemotactic Migration
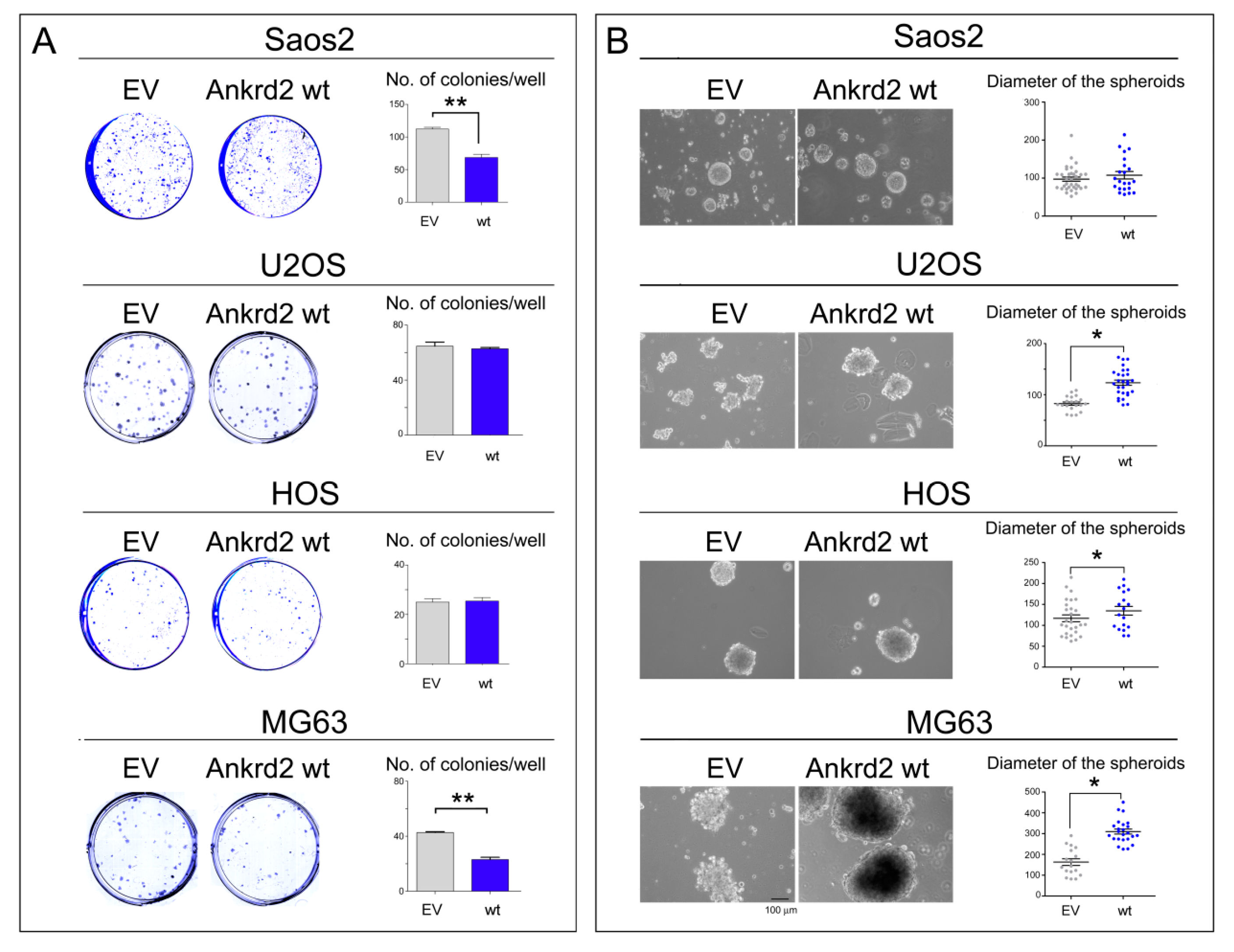
3.4.3. Clonogenic Potential
3.4.4. Anchorage Independent Growth
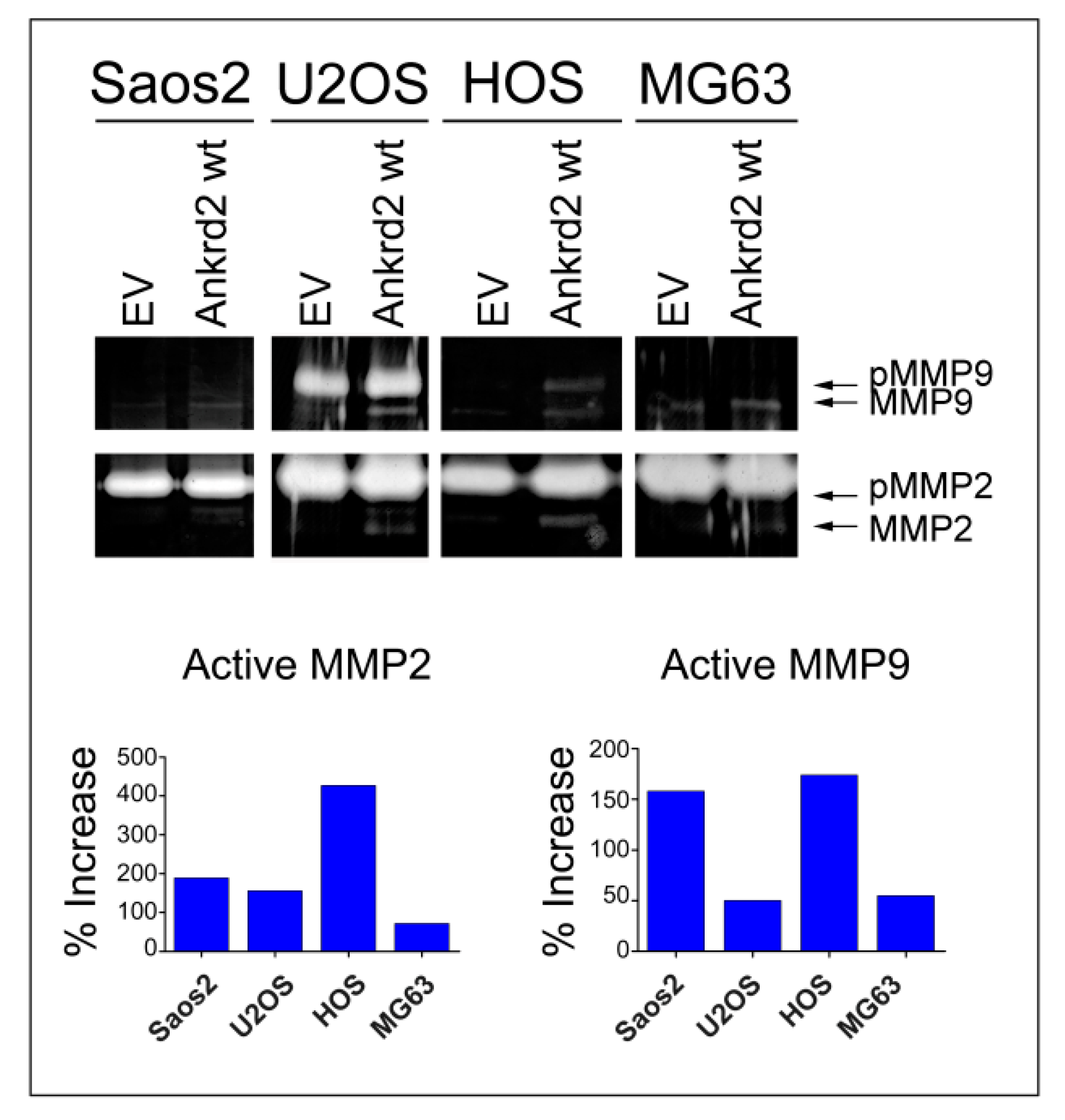
3.5. Effect of Ankrd2 Overexpression on Degradation of the Extracellular Matrix
3.6. Effect of Ankrd2 Silencing in OS Cells
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Miller, M.K.; Bang, M.-L.; Witt, C.C.; Labeit, D.; Trombitas, C.; Watanabe, K.; Granzier, H.; McElhinny, A.S.; Gregorio, C.C.; Labeit, S. The Muscle Ankyrin Repeat Proteins: CARP, ankrd2/Arpp and DARP as a Family of Titin Filament-based Stress Response Molecules. J. Mol. Biol. 2003, 333, 951–964. [Google Scholar] [CrossRef] [PubMed]
- Moriyama, M.; Tsukamotoa, Y.; Fujiwarabc, M.; Kondoad, G.; Nakadaa, C.; Babaa, T.; Ishiguroa, N.; Miyazakia, A.; Nakamuraa, K.; Horia, N.; et al. Identification of a Novel Human Ankyrin-Repeated Protein Homologous to CARP. Biochem. Biophys. Res. Commun. 2001, 285, 715–723. [Google Scholar] [CrossRef] [PubMed]
- Ishiguro, N.; Baba, T.; Ishida, T.; Takeuchi, K.; Osaki, M.; Araki, N.; Okada, E.; Takahashi, S.; Saito, M.; Watanabe, M.; et al. Carp, a Cardiac Ankyrin-Repeated Protein, and Its New Homologue, Arpp, Are Differentially Expressed in Heart, Skeletal Muscle, and Rhabdomyosarcomas. Am. J. Pathol. 2002, 160, 1767–1778. [Google Scholar] [CrossRef]
- Pallavicini, A.; Kojić, S.; Bean, C.; Vainzof, M.; Salamon, M.; Ievolella, C.; Bortoletto, G.; Pacchioni, B.; Zatz, M.; Lanfranchi, G.; et al. Characterization of Human Skeletal Muscle Ankrd2. Biochem. Biophys. Res. Commun. 2001, 285, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Jasnic-Savovic, J.; Krause, S.; Savic, S.; Kojic, A.; Kovcic, V.; Boskovic, S.; Nestorovic, A.; Rakicevic, L.; Schreiber-Katz, O.; Vogel, J.G.; et al. Differential expression and localization of Ankrd2 isoforms in human skeletal and cardiac muscles. Histochem. Cell Biol. 2016, 146, 569–584. [Google Scholar] [CrossRef] [PubMed]
- Cenni, V.; Kojic, S.; Capanni, C.; Faulkner, G.; Lattanzi, G. Ankrd2 in Mechanotransduction and Oxidative Stress Response in Skeletal Muscle: New Cues for the Pathogenesis of Muscular Laminopathies. Oxidative Med. Cell. Longev. 2019, 2019, 7318796. [Google Scholar] [CrossRef]
- Belgrano, A.; Rakicevic, L.; Mittempergher, L.; Campanaro, S.; Martinelli, V.C.; Mouly, V.; Valle, G.; Kojic, S.; Faulkner, G. Multi-Tasking Role of the Mechanosensing Protein Ankrd2 in the Signaling Network of Striated Muscle. PLoS ONE 2011, 6, e25519. [Google Scholar] [CrossRef]
- Tsukamoto, Y.; Hijiya, N.; Yano, S.; Yokoyama, S.; Nakada, C.; Uchida, T.; Matsuura, K.; Moriyama, M. Arpp/Ankrd2, a member of the muscle ankyrin repeat proteins (MARPs), translocates from the I-band to the nucleus after muscle injury. Histochem. Cell Biol. 2007, 129, 55–64. [Google Scholar] [CrossRef]
- Bean, C.; Facchinello, N.; Faulkner, G.; Lanfranchi, G. The effects of Ankrd2 alteration indicate its involvement in cell cycle regulation during muscle differentiation. Biochim. Biophys. Acta 2008, 1783, 1023–1035. [Google Scholar] [CrossRef]
- Cenni, V.; Bavelloni, A.; Beretti, F.; Tagliavini, F.; Manzoli, L.; Lattanzi, G.; Maraldi, N.M.; Cocco, L.; Marmiroli, S. Ankrd2/ARPP is a novel Akt2 specific substrate and regulates myogenic differentiation upon cellular exposure to H2O2. Mol. Biol. Cell 2011, 22, 2946–2956. [Google Scholar] [CrossRef]
- Bean, C.; Verma, N.K.; Yamamoto, D.L.; Chemello, F.; Cenni, V.; Filomena, M.C.; Chen, J.; Bang, M.L.; Lanfranchi, G. Ankrd2 is a modulator of NF-κB-mediated inflammatory responses during muscle differentiation. Cell Death Dis. 2014, 5, e1002. [Google Scholar] [CrossRef] [PubMed]
- Saraf, A.J.; Fenger, J.M.; Roberts, R.D. Osteosarcoma: Accelerating Progress Makes for a Hopeful Future. Front. Oncol. 2018, 8, 4. [Google Scholar] [CrossRef] [PubMed]
- Scotlandi, K.; Picci, P.; Kovar, H. Targeted therapies in bone sarcomas. Curr. Cancer Drug Targets 2009, 9, 843–853. [Google Scholar] [CrossRef] [PubMed]
- Hattinger, C.M.; Serra, M. Role of pharmacogenetics of drug-metabolizing enzymes in treating osteosarcoma. Expert Opin. Drug Metab. Toxicol. 2015, 11, 1449–1463. [Google Scholar] [CrossRef]
- Harrison, D.J.; Geller, D.S.; Gill, J.; Lewis, V.O.; Gorlick, R. Current and future therapeutic approaches for osteosarcoma. Expert Rev. Anticancer Ther. 2017, 18, 39–50. [Google Scholar] [CrossRef]
- Ishiguro, N.; Motoi, T.; Osaki, M.; Araki, N.; Minamizaki, T.; Moriyama, M.; Ito, H.; Yoshida, H. Immunohistochemical analysis of a muscle ankyrin-repeat protein, Arpp, in paraffin-embedded tumors: Evaluation of Arpp as a tumor marker for rhabdomyosarcoma. Hum. Pathol. 2005, 36, 620–625. [Google Scholar] [CrossRef]
- Piazzi, M.; Bavelloni, A.; Greco, S.; Focaccia, E.; Orsini, A.; Benini, S.; Gambarotti, M.; Faenza, I.; Blalock, W. Expression of the double-stranded RNA-dependent kinase PKR influences osteosarcoma attachment independent growth, migration, and invasion. J. Cell. Physiol. 2020, 235, 1103–1119. [Google Scholar] [CrossRef]
- Angori, S.; Capanni, C.; Faulkner, G.; Bean, C.; Boriani, G.; Lattanzi, G.; Cenni, V. Emery-Dreifuss Muscular Dystrophy-Associated Mutant Forms of Lamin A Recruit the Stress Responsive Protein Ankrd2 into the Nucleus, Affecting the Cellular Response to Oxidative Stress. Cell. Physiol. Biochem. 2017, 42, 169–184. [Google Scholar] [CrossRef]
- Evangelisti, C.; Paganelli, F.; Giuntini, G.; Mattioli, E.; Cappellini, A.; Ramazzotti, G.; Faenza, I.; Maltarello, M.C.; Martelli, A.M.; Scotlandi, K.; et al. Lamin A and Prelamin A Counteract Migration of Osteosarcoma Cells. Cells 2020, 9, 774. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhou, J.; Ji, Y.; Yu, B. Elevated expression of AKT2 correlates with disease severity and poor prognosis in human osteosarcoma. Mol. Med. Rep. 2014, 10, 737–742. [Google Scholar] [CrossRef][Green Version]
- Bjørnland, K.; Flatmark, K.; Pettersen, S.; Aaasen, A.O.; Fodstad, Ø.; Mælandsmo, G.M. Matrix Metalloproteinases Participate in Osteosarcoma Invasion. J. Surg. Res. 2005, 127, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Shomori, K.; Nagashima, Y.; Kuroda, N.; Honjo, A.; Tsukamoto, Y.; Tokuyasu, N.; Maeta, N.; Matsuura, K.; Hijiya, N.; Yano, S.; et al. ARPP protein is selectively expressed in renal oncocytoma, but rarely in renal cell carcinomas. Mod. Pathol. 2007, 20, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.; Henderson, B.R.; Emmanuel, C.; Harnett, P.; DeFazio, A. Inhibition of ANKRD1 sensitizes human ovarian cancer cells to endoplasmic reticulum stress-induced apoptosis. Oncogene 2014, 34, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, M.; Yamashita, K.; Waraya, M.; Minatani, N.; Ushiku, H.; Kojo, K.; Ema, A.; Kosaka, Y.; Katoh, H.; Sengoku, N.; et al. Epigenetic regulation of ZEB1-RAB25/ESRP1 axis plays a critical role in phenylbutyrate treatment-resistant breast cancer. Oncotarget 2015, 7, 1741–1753. [Google Scholar] [CrossRef]
- Hui, B.; Ji, H.; Xu, Y.; Wang, J.; Ma, Z.; Zhang, C.; Wang, K.-M.; Zhou, Y. RREB1-induced upregulation of the lncRNA AGAP2-AS1 regulates the proliferation and migration of pancreatic cancer partly through suppressing ANKRD1 and ANGPTL4. Cell Death Dis. 2019, 10, 207. [Google Scholar] [CrossRef]
- Takahashi, A.; Seike, M.; Chiba, M.; Takahashi, S.; Nakamichi, S.; Matsumoto, M.; Takeuchi, S.; Minegishi, Y.; Noro, R.; Kunugi, S.; et al. Ankyrin Repeat Domain 1 Overexpression is Associated with Common Resistance to Afatinib and Osimertinib in EGFR-mutant Lung Cancer. Sci. Rep. 2018, 8, 14896. [Google Scholar] [CrossRef]
- Zhang, L.M.; Su, L.X.; Hu, J.Z.; Wang, D.M.; Ju, H.Y.; Li, X.; Han, Y.F.; Xia, W.Y.; Guo, W.; Ren, G.X.; et al. Epigenetic regulation of VENTXP1 suppresses tumor proliferation via miR-205-5p/ANKRD2/NF-kB signaling in head and neck squamous cell carcinoma. Cell Death Dis. 2020, 11, 838. [Google Scholar] [CrossRef]
- Zadra, G.; Batista, J.L.; Loda, M. Dissecting the Dual Role of AMPK in Cancer: From Experimental to Human Studies. Mol. Cancer Res. 2015, 13, 1059–1072. [Google Scholar] [CrossRef]
- Fazio, C.; Ricciardiello, L. Inflammation and Notch signaling: A crosstalk with opposite effects on tumorigenesis. Cell Death Dis. 2016, 7, e2515. [Google Scholar] [CrossRef]
- Colaprico, A.; Olsen, C.; Bailey, M.H.; Odom, G.J.; Terkelsen, T.; Silva, T.C.; Olsen, A.V.; Cantini, L.; Zinovyev, A.; Barillot, E.; et al. Interpreting pathways to discover cancer driver genes with Moonlight. Nat. Commun. 2020, 11, 69. [Google Scholar] [CrossRef]
- Wagner, F.; Holzapfel, B.M.; Thibaudeau, L.; Straub, M.; Ling, M.-T.; Grifka, J.; Loessner, D.; Lévesque, J.-P.; Hutmacher, D.W. A Validated Preclinical Animal Model for Primary Bone Tumor Research. J. Bone Jt. Surg. 2016, 98, 916–925. [Google Scholar] [CrossRef] [PubMed]
- Wagner, F.; Holzapfel, B.M.; Martine, L.C.; McGovern, J.; Lahr, C.A.; Boxberg, M.; Prodinger, P.M.; Grässel, S.; Loessner, D.; Hutmacher, D.W. A humanized bone microenvironment uncovers HIF2 alpha as a latent marker for osteosarcoma. Acta Biomater. 2019, 89, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Wagner, F.; Holzapfel, B.M.; McGovern, J.A.; Shafiee, A.; Baldwin, J.G.; Martine, L.C.; Lahr, C.A.; Wunner, F.M.; Friis, T.; Bas, O.; et al. Humanization of bone and bone marrow in an orthotopic site reveals new potential therapeutic targets in osteosarcoma. Biomaterials 2018, 171, 230–246. [Google Scholar] [CrossRef] [PubMed]
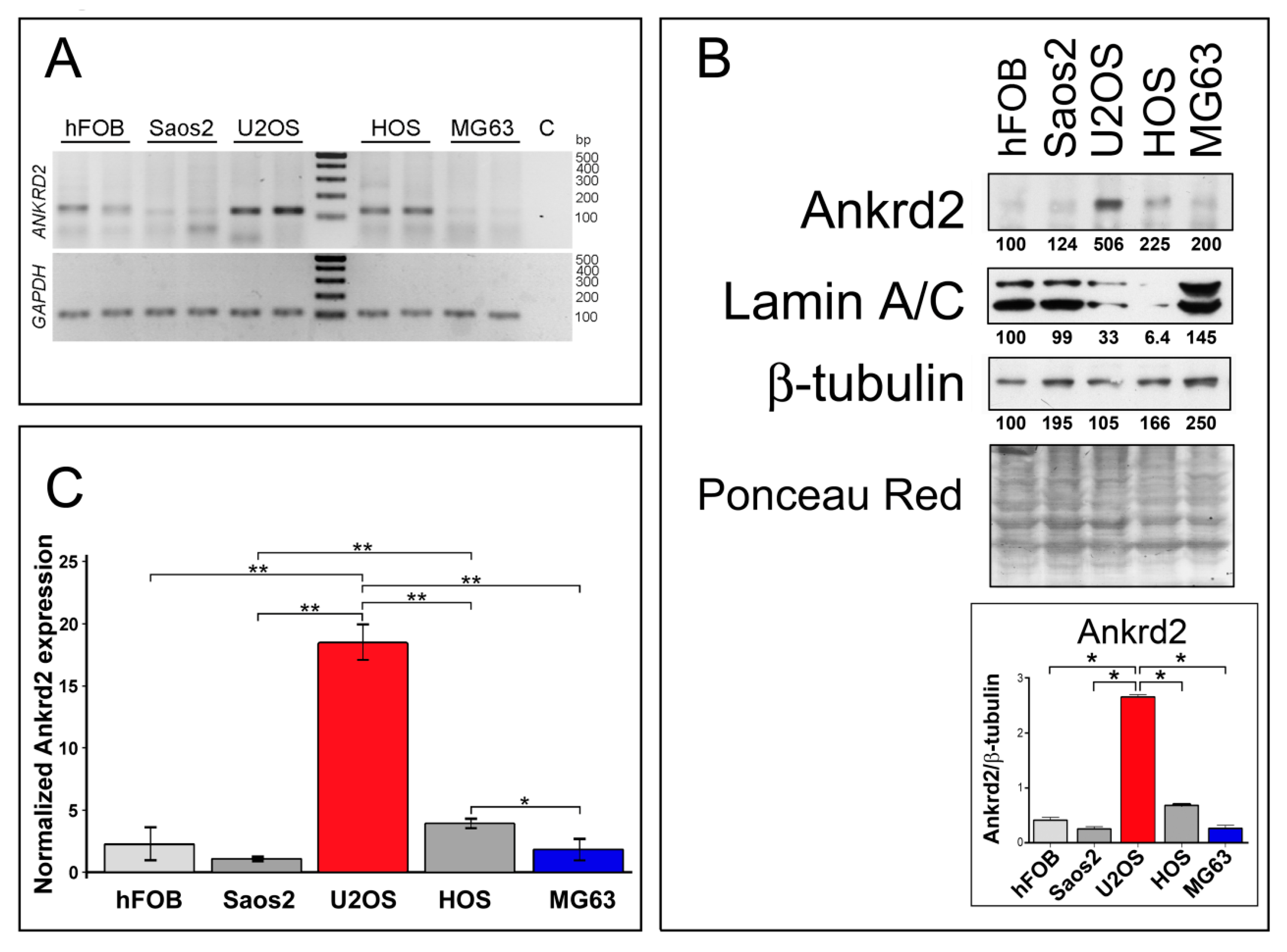
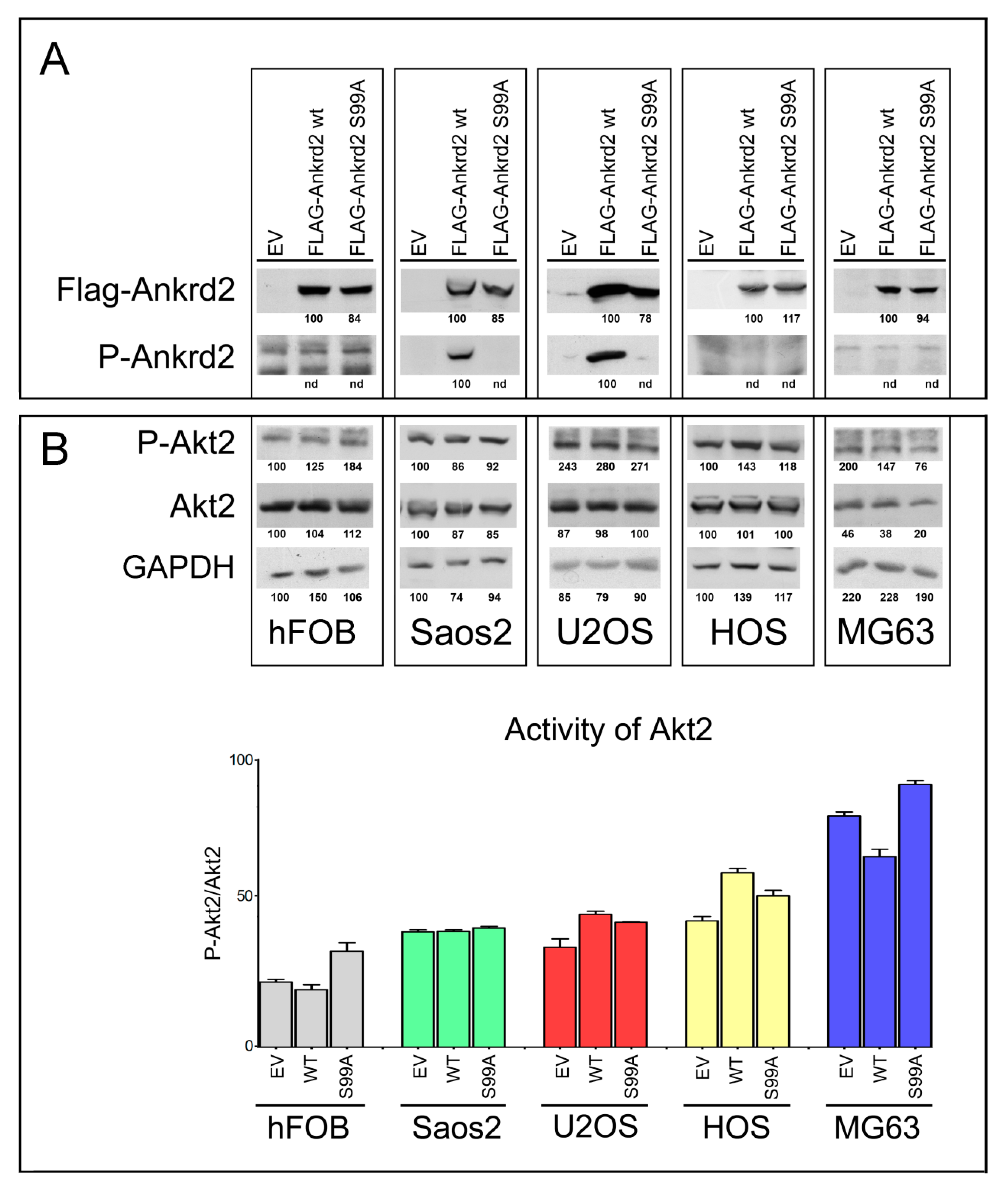
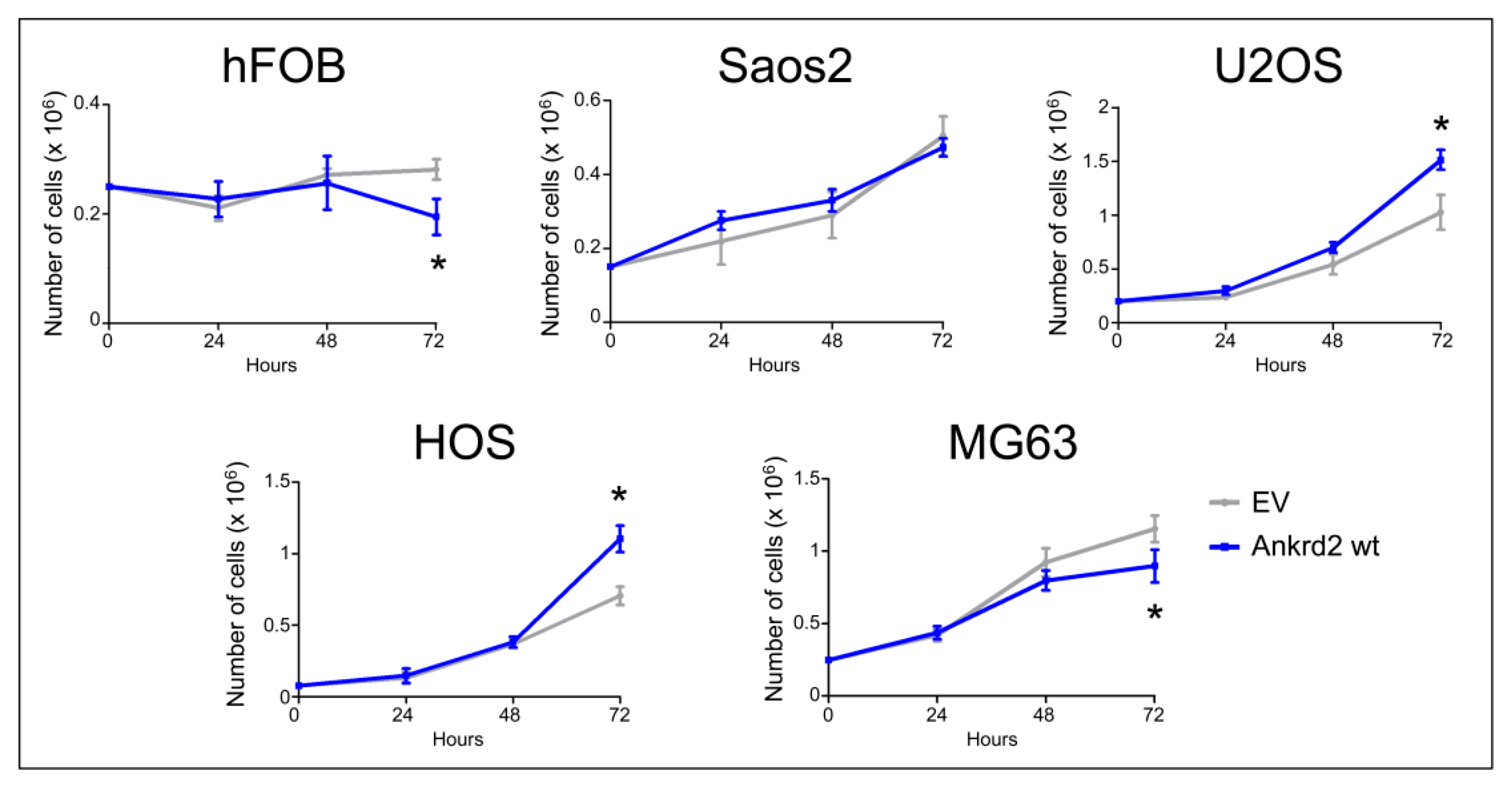
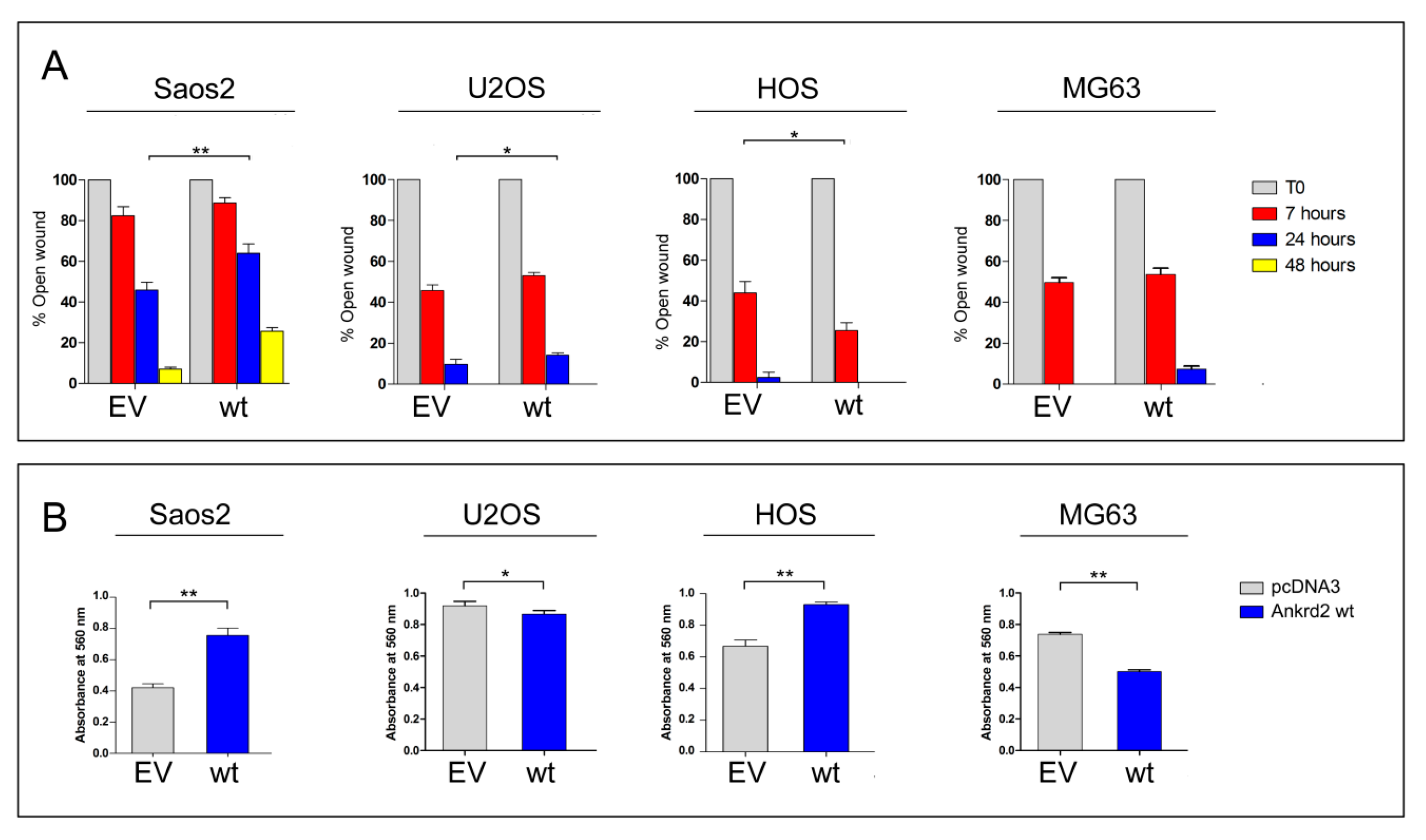

| Osteosarcoma-Derived Cell Line | Effect on Proliferation | Effect on Migration (24 h from Wounding) | Effect on Transwell Migration | Effect on Clonogenicity (Anchorage Dependent) | Effect on Clonogenicity (Anchorage Independent) | Effect on MMP2/9 Activity |
|---|---|---|---|---|---|---|
| Saos2 | = | - | + | - | = | + |
| U2OS | + | - | - | = | + | + |
| HOS | + | + | + | = | + | + |
| MG63 | - | - | - | - | + | + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piazzi, M.; Kojic, S.; Capanni, C.; Stamenkovic, N.; Bavelloni, A.; Marin, O.; Lattanzi, G.; Blalock, W.; Cenni, V. Ectopic Expression of Ankrd2 Affects Proliferation, Motility and Clonogenic Potential of Human Osteosarcoma Cells. Cancers 2021, 13, 174. https://doi.org/10.3390/cancers13020174
Piazzi M, Kojic S, Capanni C, Stamenkovic N, Bavelloni A, Marin O, Lattanzi G, Blalock W, Cenni V. Ectopic Expression of Ankrd2 Affects Proliferation, Motility and Clonogenic Potential of Human Osteosarcoma Cells. Cancers. 2021; 13(2):174. https://doi.org/10.3390/cancers13020174
Chicago/Turabian StylePiazzi, Manuela, Snezana Kojic, Cristina Capanni, Nemanja Stamenkovic, Alberto Bavelloni, Oriano Marin, Giovanna Lattanzi, William Blalock, and Vittoria Cenni. 2021. "Ectopic Expression of Ankrd2 Affects Proliferation, Motility and Clonogenic Potential of Human Osteosarcoma Cells" Cancers 13, no. 2: 174. https://doi.org/10.3390/cancers13020174
APA StylePiazzi, M., Kojic, S., Capanni, C., Stamenkovic, N., Bavelloni, A., Marin, O., Lattanzi, G., Blalock, W., & Cenni, V. (2021). Ectopic Expression of Ankrd2 Affects Proliferation, Motility and Clonogenic Potential of Human Osteosarcoma Cells. Cancers, 13(2), 174. https://doi.org/10.3390/cancers13020174







