HER2/neu-Based Peptide Vaccination-Pulsed with B-Cell Epitope Induced Efficient Prophylactic and Therapeutic Antitumor Activities in TUBO Breast Cancer Mice Model
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Culture
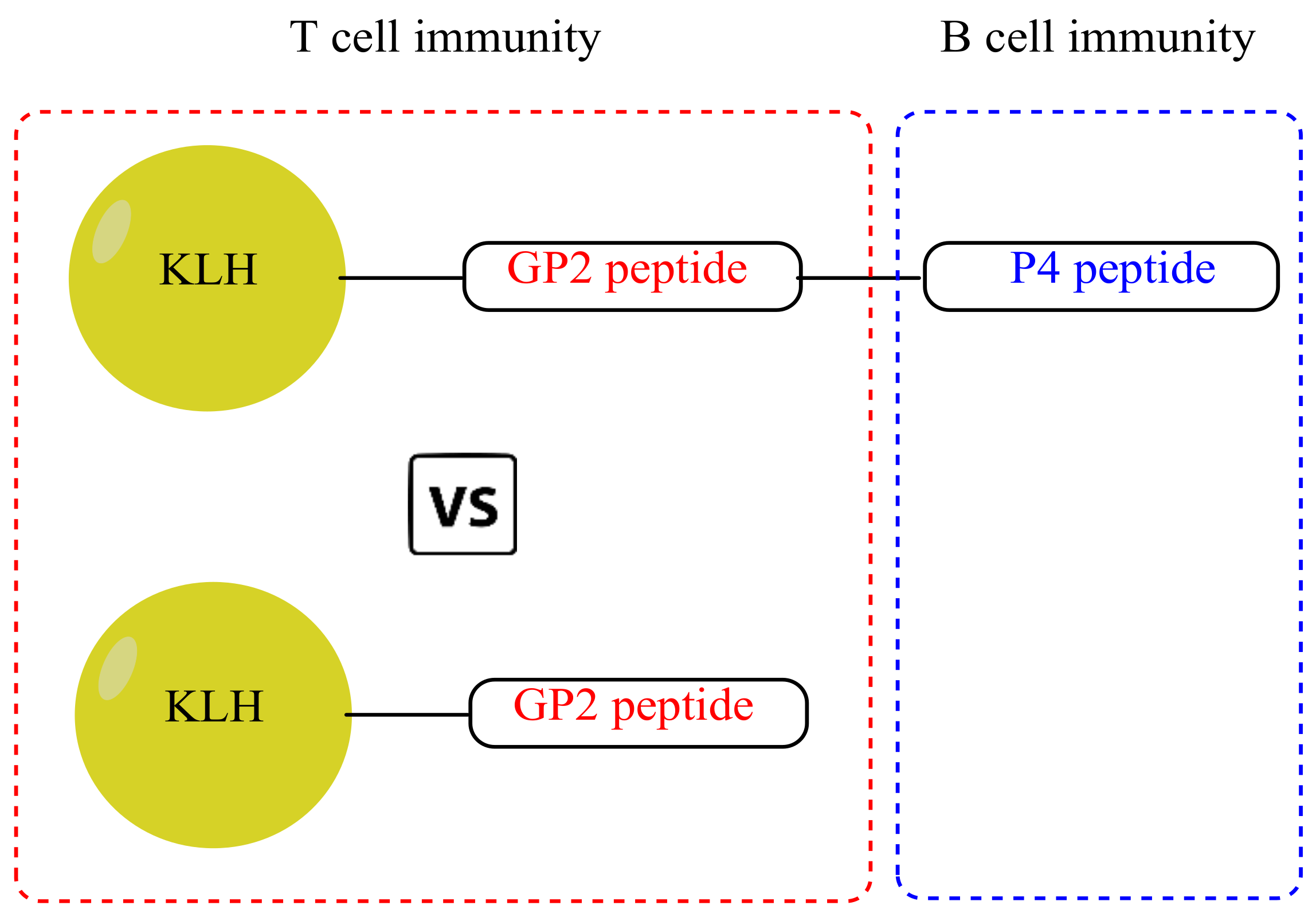
2.3. Protein–Peptide Conjugates
2.4. Experimental Animal
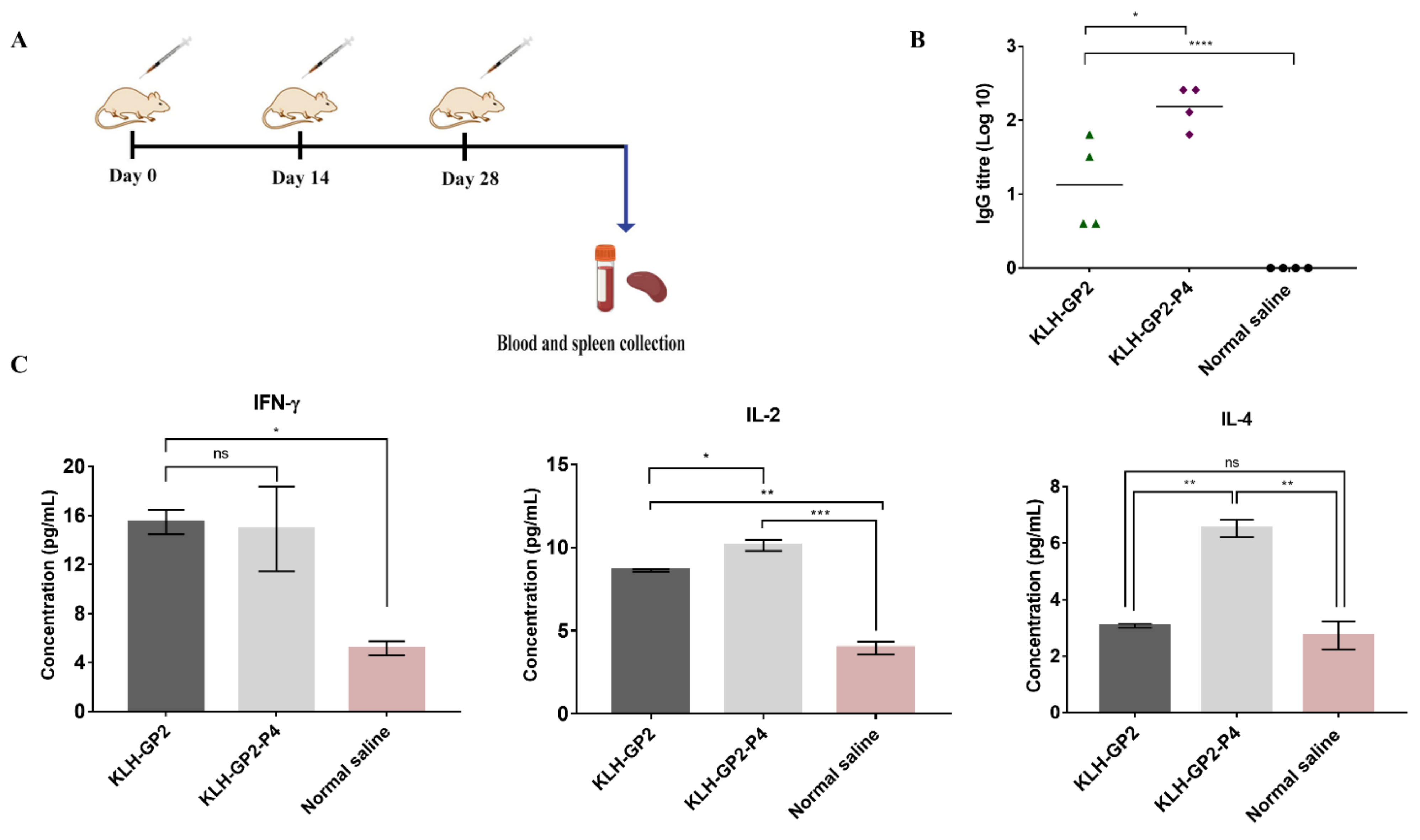
2.5. Mice Immunization for Immunogenicity Evaluations
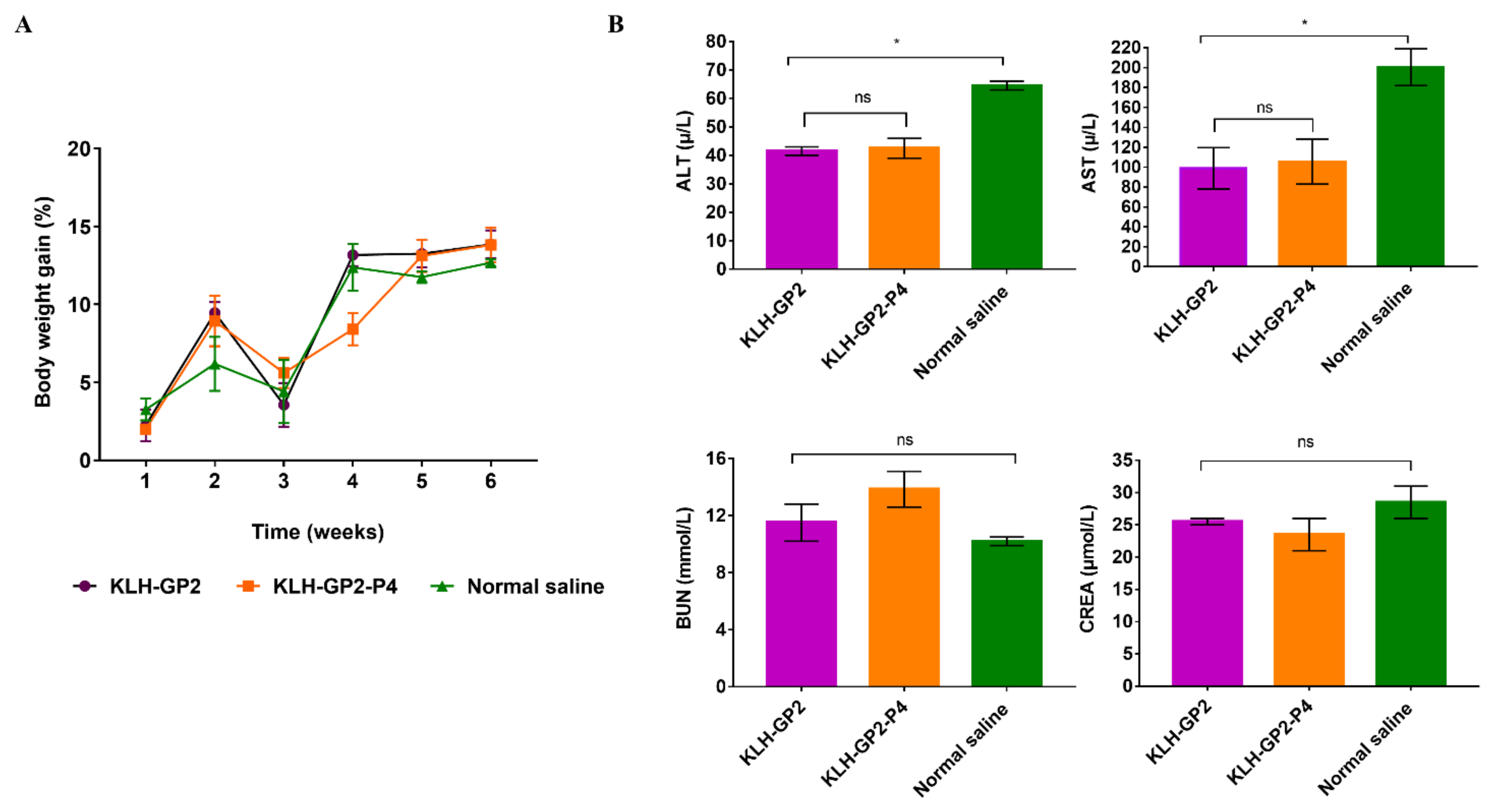
2.6. Reactogenicity and Safety Assessment of the Vaccine Candidates
2.7. Serum Biochemical Analyses
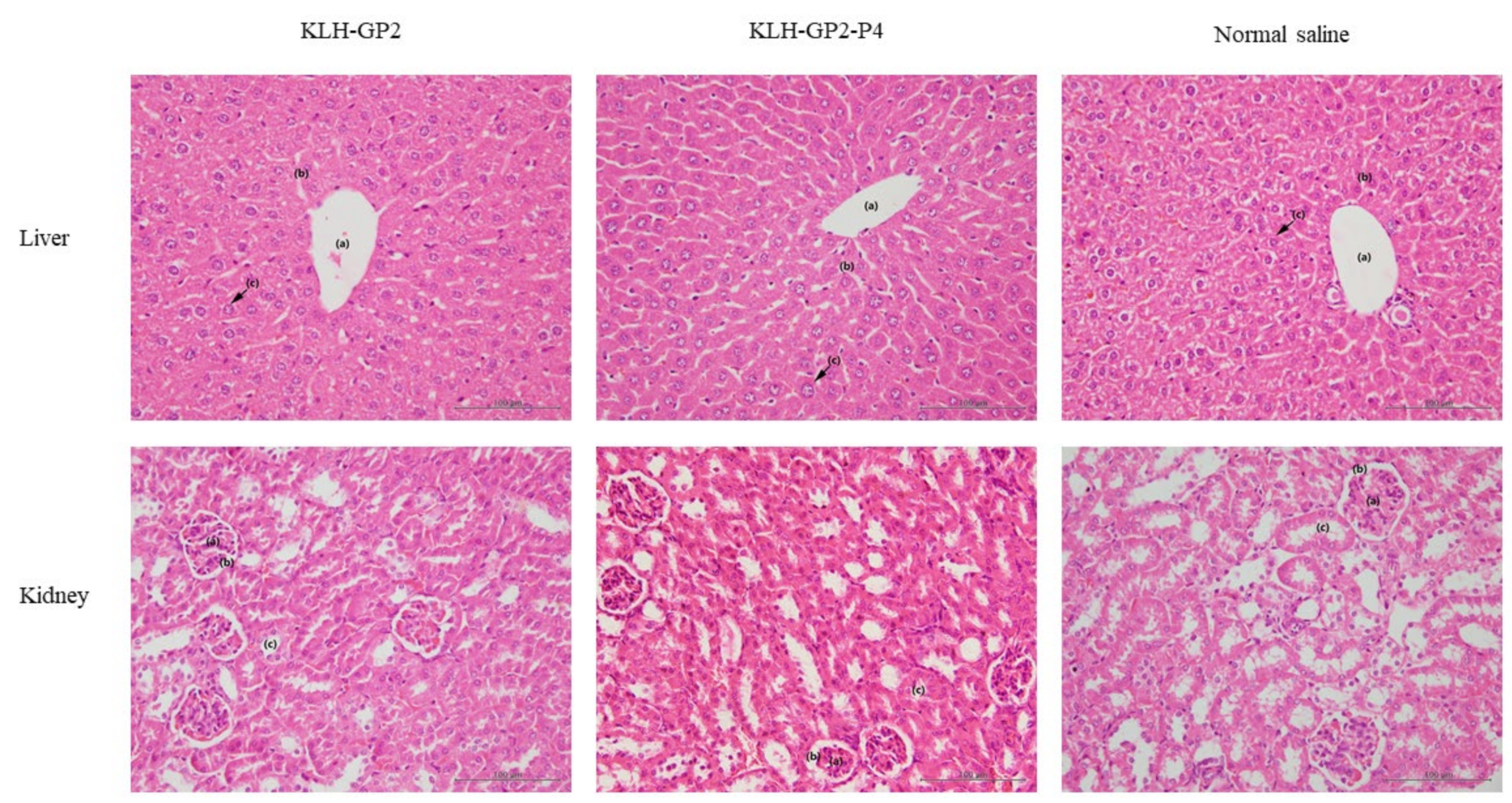
2.8. Histopathological Examination
2.9. Cytokines Quantification
2.10. HER2/neu-Specific Antibody Titers Evaluation
2.11. Indirect Serum Neutralization Assay
2.12. Establishment of BALB/c Mice Model of TUBO Breast Tumor
2.13. Prophylactic TUBO Challenge Experiment
2.14. Therapeutic TUBO Challenge Experiment
2.15. Statistical Analysis
3. Results
3.1. Antibodies and Cytokine Responses to Vaccination with Peptide Vaccine Candidates
3.2. Indirect Serum Neutralization Assay
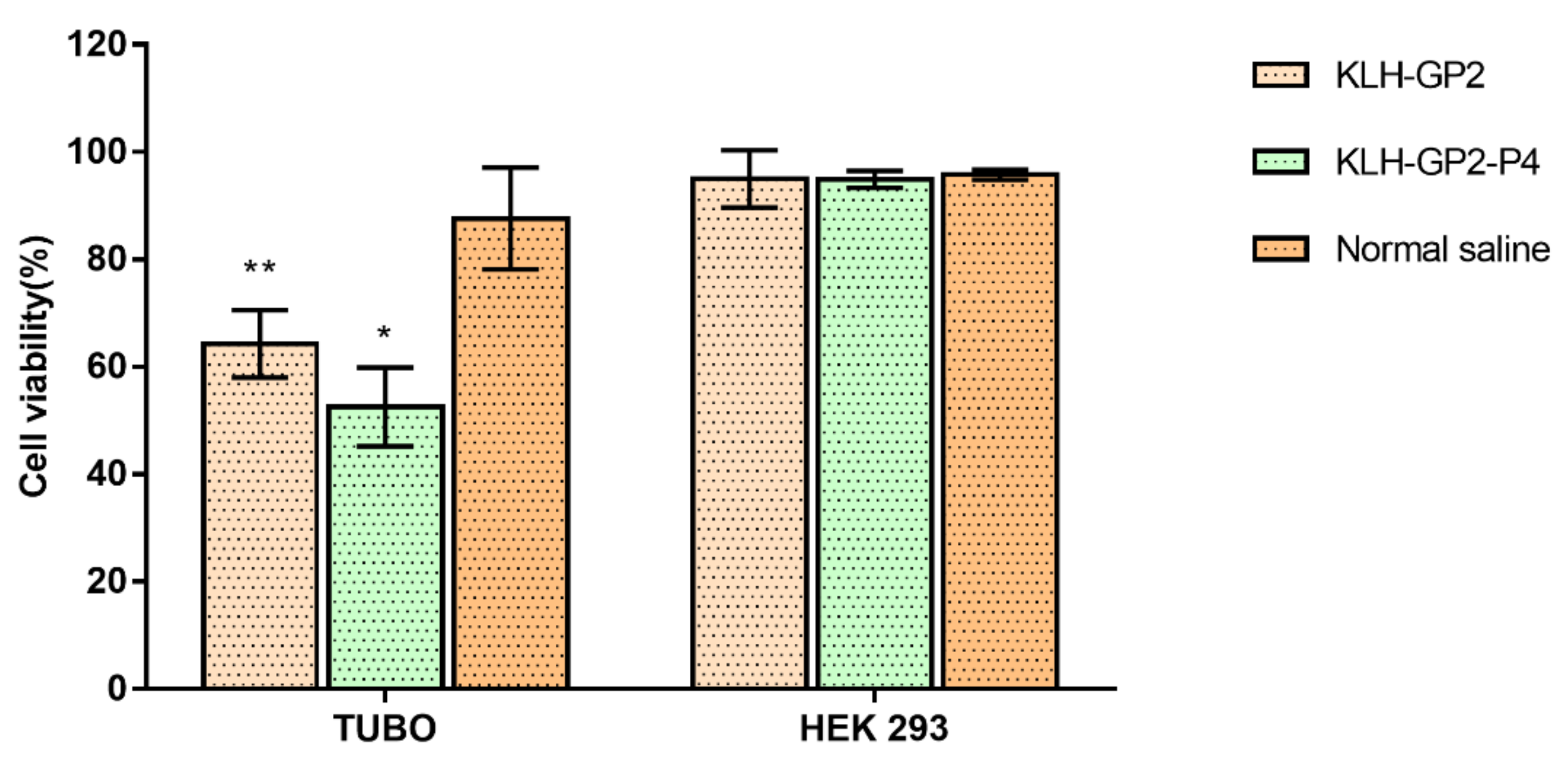
3.3. Reactogenicity and Safety Evaluations of Peptide-Based Vaccine Candidates in Mice
3.4. Establishment and Validation of TUBO-Derived Mouse Model of HER2/neu Positive Breast Cancer
3.5. Antitumor Efficacy of Prophylactic and Therapeutic Peptide Vaccination
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Aziz, M.H.; Ahmad, A. Epigenetic basis of cancer drug resistance. Cancer Drug Resist. 2020, 3, 113–116. [Google Scholar] [CrossRef] [Green Version]
- de la Peña, F.A.; Andrés, R.; Garcia-Sáenz, J.A.; Manso, L.; Margelí, M.; Dalmau, E.; Pernas, S.; Prat, A.; Servitja, S.; Ciruelos, E. SEOM clinical guidelines in early stage breast cancer (2018). Clin. Transl. Oncol. 2019, 21, 18–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamaguchi, Y.; Yamaue, H.; Okusaka, T.; Okuno, K.; Suzuki, H.; Fujioka, T.; Otsu, A.; Ohashi, Y.; Shimazawa, R.; Nishio, K.; et al. Guidance for peptide vaccines for the treatment of cancer. Guid. Pept. Vaccines Treat. Cancer. Cancer Sci. 2014, 105, 924–931. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shemesh, C.S.; Hsu, J.C.; Hosseini, I.; Shen, B.Q.; Rotte, A.; Twomey, P.; Girish, S.; Wu, B. Personalized Cancer Vaccines: Clinical Landscape, Challenges, and Opportunities. Mol. Ther. 2020, 27, 555–570. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, P.M.; Clifton, G.T.; Vreeland, T.J.; Adams, A.M.; O’Shea, A.E. AE37: A HER2-targeted vaccine for the prevention of breast cancer recurrence. Expert Opin. Investig. Drugs 2020, 30, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.L.B.; Soliman, H.; Czerniecki, B.J. The clinical development of vaccines for HER2+ breast cancer: Current landscape and future perspectives. Cancer Treat. Rev. 2017, 61, 107–115. [Google Scholar] [CrossRef]
- Farzad, N.; Barati, N.; Momtazi-Borojeni, A.A.; Yazdani, M.; Arab, A.; Razazan, A.; Shariat, S.; Mansourian, M.; Abbasi, A.; Saberi, Z.; et al. P435 HER2/neu-derived peptide conjugated to liposomes containing DOPE as an effective prophylactic vaccine formulation for breast cancer. Artif. Cells Nanomed. Biotechnol. 2019, 47, 665–673. [Google Scholar] [CrossRef]
- Skwarczynski, M.; Toth, I. Peptide-based synthetic vaccines. Chem. Sci. 2016, 7, 842–854. [Google Scholar] [CrossRef] [Green Version]
- Nevagi, R.J.; Toth, I.; Skwarczynski, M. Peptide-based vaccines. In Peptide Applications in Biomedicine 2018, Biotechnology and Bioengineering; Elsevier: Amsterdam, The Netherlands, 2018; pp. 327–358. [Google Scholar]
- Schneble, E.; Clifton, G.T.; Hale, D.F.; Peoples, G.E.J.V.D. Peptide-based cancer vaccine strategies and clinical results. Vaccine Des. 2016, 1403, 797–817. [Google Scholar]
- Pallerla, S.; Comeau, J.; Jois, S. Treatment of the Future: With Emphasis on HER2-Positive Breast Cancer. Int. J. Mol. Sci. 2021, 22, 779. [Google Scholar] [CrossRef] [PubMed]
- Brossart, P.; Wirths, S.; Stuhler, G.; Reichardt, V.L.; Kanz, L.; Brugger, W.J.B. Induction of cytotoxic T-lymphocyte responses in vivo after vaccinations with peptide-pulsed dendritic cells. J. Am. Soc. Hematol. 2000, 96, 3102–3108. [Google Scholar]
- Bezu, L.; Kepp, O.; Cerrato, G.; Pol, J.; Fucikova, J.; Spisek, R.; Galluzzi, L.J.O. Trial watch: Peptide-based vaccines in anticancer therapy. Oncoimmunology 2018, 7, e1511506. [Google Scholar] [CrossRef] [PubMed]
- de Paula Peres, L.; da Luz, F.A.C.; dos Anjos Pultz, B.; Brígido, P.C.; de Araújo, R.A.; Goulart, L.R.; Silva, M.J.B.J.B. Peptide vaccines in breast cancer: The immunological basis for clinical response. Biotechnol. Adv. 2015, 33, 1868–1877. [Google Scholar] [CrossRef] [PubMed]
- Clifton, G.T.; Mittendorf, E.A.; Peoples, G.E.J.I. Adjuvant HER2/neu peptide cancer vaccines in breast cancer. Immunotherapy 2015, 7, 1159–1168. [Google Scholar] [CrossRef]
- Azmi, F.; Ahmad Fuaad, A.A.H.; Skwarczynski, M.; Toth, I. Recent progress in adjuvant discovery for peptide-based subunit vaccines. Hum. Vaccines Immunother. 2014, 10, 778–796. [Google Scholar] [CrossRef] [Green Version]
- Swaminathan, A.; Lucas, R.M.; Dear, K.; McMichael, A.J. Keyhole limpet haemocyanin–a model antigen for human immunotoxicological studies. Br. J. Clin. Pharmacol. 2014, 78, 1135–1142. [Google Scholar] [CrossRef]
- Gandhi, R.T.; O’Neill, D.; Bosch, R.J.; Chan, E.S.; Bucy, R.P.; Shopis, J. AIDS Clinical Trials Group A5130 team. A randomized therapeutic vaccine trial of canarypox-HIV-pulsed dendritic cells vs. canarypox-HIV alone in HIV-1-infected patients on antiretroviral therapy. Vaccine 2009, 27, 6088–6094. [Google Scholar] [CrossRef] [Green Version]
- Tavakolpour, S.; Darvishi, M. The Roles of CD4+ T-Cells in Tumor Immunity. In Cancer Immunology; Springer: Cham, Switerland, 2020; pp. 63–90. [Google Scholar]
- Sarvaria, A.; Madrigal, J.A.; Saudemont, A.J.C. B cell regulation in cancer and anti-tumor immunity. Cell. Mol. Immunol. 2017, 14, 662–674. [Google Scholar] [CrossRef] [Green Version]
- Kaumaya, P.T. A paradigm shift: Cancer therapy with peptide-based B-cell epitopes and peptide immunotherapeutics targeting multiple solid tumor types: Emerging concepts and validation of combination immunotherapy. Hum. Vaccines Immunother. 2015, 11, 1368–1386. [Google Scholar] [CrossRef] [Green Version]
- Wiedermann, U.; Wiltschke, C.; Jasinska, J.; Kundi, M.; Zurbriggen, R.; Garner-Spitzer, E. A virosomal formulated Her-2/neu multi-peptide vaccine induces Her-2/neu-specific immune responses in patients with metastatic breast cancer: A phase I study. Breast Cancer Res. Treat. 2010, 119, 673–683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dakappagari, N.K.; Douglas, D.B.; Triozzi, P.L.; Stevens, V.C.; Kaumaya, P.T.J.C. Prevention of mammary tumors with a chimeric HER-2 B-cell epitope peptide vaccine. Cancer Res. 2000, 60, 3782–3789. [Google Scholar] [PubMed]
- Tobias, J.; Jasinska, J.; Baier, K.; Kundi, M.; Ede, N.; Zielinski, C.; Wiedermann, U.J.B. Enhanced and long term immunogenicity of a Her-2/neu multi-epitope vaccine conjugated to the carrier CRM197 in conjunction with the adjuvant Montanide. BMC Cancer 2017, 17, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lambraño, J.; Curtidor, H.; Avendaño, C.; Díaz-Arévalo, D.; Roa, L.; Vanegas, M.; Patarroyo, M.A. Preliminary Evaluation of the Safety and Immunogenicity of an Antimalarial Vaccine Candidate Modified Peptide (IMPIPS) Mixture in a Murine Model. J. Immunol. Res. 2019, 2019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azmi, F.; Fuaad, A.A.H.A.; Giddam, A.K.; Batzloff, M.R.; Good, M.F.; Skwarczynski, M.; Toth, I. Self-adjuvanting vaccine against group A streptococcus: Application of fibrillized peptide and immunostimulatory lipid as adjuvant. Bioorganic Med. Chem. 2014, 22, 6401–6408. [Google Scholar] [CrossRef] [Green Version]
- Sicca, F.; Martinuzzi, D.; Montomoli, E.; Huckriede, A. Comparison of influenza-specific neutralizing antibody titers determined using different assay readouts and hemagglutination inhibition titers: Good correlation but poor agreement. Vaccine 2020, 38, 2527–2541. [Google Scholar] [CrossRef]
- Zhang, F.; Hao, C.; Zhang, S.; Li, A.; Zhang, Q.; Wu, W.; Li, D. Oral immunization with recombinant enterovirus 71 VP1 formulated with chitosan protects mice against lethal challenge. Virol. J. 2014, 11, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Mohamad, N.E.; Keong Yeap, S.; Beh, B.K.; Romli, M.F.; Yusof, H.M.; Kristeen-Teo, Y.W.; Alitheen, N.B. Comparison of in vivo toxicity, antioxidant and immunomodulatory activities of coconut, nipah and pineapple juice vinegars. J. Sci. Food Agric. 2018, 98, 534–540. [Google Scholar] [CrossRef]
- Al-Awadhi, A.; Lee Murray, J.; Ibrahim, N.K. D eveloping anti-HER 2 vaccines: B reast cancer experience. Int. J. Cancer 2018, 143, 2126–2132. [Google Scholar] [CrossRef] [Green Version]
- Knutson, K.L.; Clynes, R.; Shreeder, B.; Yeramian, P.; Kemp, K.P.; Ballman, K.; Perez, E.A. Improved survival of HER2+ breast cancer patients treated with trastuzumab and chemotherapy is associated with host antibody immunity against the HER2 intracellular domain. Cancer Res. 2016, 76, 3702–3710. [Google Scholar] [CrossRef] [Green Version]
- Schubert, B.; Kohlbacher, O. Designing string-of-beads vaccines with optimal spacers. Genome Med. 2016, 8, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Geyer, H.; Wuhrer, M.; Resemann, A.; Geyer, R. Identification and characterization of keyhole limpet hemocyanin N-glycans mediating cross-reactivity with Schistosoma mansoni. J. Biol. Chem. 2005, 280, 40731–40748. [Google Scholar] [CrossRef] [Green Version]
- May, R.J.; Beenhouwer, D.O.; Scharff, M.D. Antibodies to keyhole limpet hemocyanin cross-react with an epitope on the polysaccharide capsule of Cryptococcus neoformans and other carbohydrates: Implications for vaccine development. J. Immunol. 2003, 171, 4905–4912. [Google Scholar] [CrossRef] [Green Version]
- Church, S.E.; Jensen, S.M.; Antony, P.A.; Restifo, N.P.; Fox, B.A. Tumor-specific CD 4+ T cells maintain effector and memory tumor-specific CD 8+ T cells. Eur. J. Immunol. 2014, 44, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Ni, L.; Lu, J. Interferon gamma in cancer immunotherapy. Cancer Med. 2018, 7, 4509–4516. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Liu, K.; Xu, J.; Zhan, T.; Liu, M.; Li, L.; Wang, X. Synthetic and immunological studies on the OCT4 immunodominant motif antigen-based anti-cancer vaccine. Cancer Biol. Med. 2020, 17, 132. [Google Scholar] [CrossRef] [PubMed]
- Zamani, P.; Teymouri, M.; Nikpoor, A.R.; Navashenaq, J.G.; Gholizadeh, Z.; Darban, S.A.; Jaafari, M.R.J.E.J. Nanoliposomal vaccine containing long multi-epitope peptide E75-AE36 pulsed PADRE-induced effective immune response in mice TUBO model of breast cancer. Eur. J. Cancer 2020, 129, 80–96. [Google Scholar] [CrossRef] [PubMed]
- Barati, N.; Razazan, A.; Nicastro, J.; Slavcev, R.; Arab, A.; Mosaffa, F.; Behravan, J.J.C. Immunogenicity and antitumor activity of the superlytic λF7 phage nanoparticles displaying a HER2/neu-derived peptide AE37 in a tumor model of BALB/c mice. Cancer Lett. 2018, 424, 109–116. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L. Multi-epitope vaccines: A promising strategy against tumors and viral infections. Cell. Mol. Immunol. 2018, 15, 182–184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, Y.; Hu, Q.; Zhang, R.; Zhou, B.; Xie, D.; Wang, Y.; Yang, L. Rational design of innate defense regulator peptides as tumor vaccine adjuvants. NPJ Vaccines 2021, 6, 1–13. [Google Scholar] [CrossRef]
- Xie, Y.; Pan, H.; Sun, H.; Li, D. A promising balanced Th1 and Th2 directing immunological adjuvant, saponins from the root of Platycodon grandiflorum. Vaccine 2008, 26, 3937–3945. [Google Scholar] [CrossRef] [PubMed]
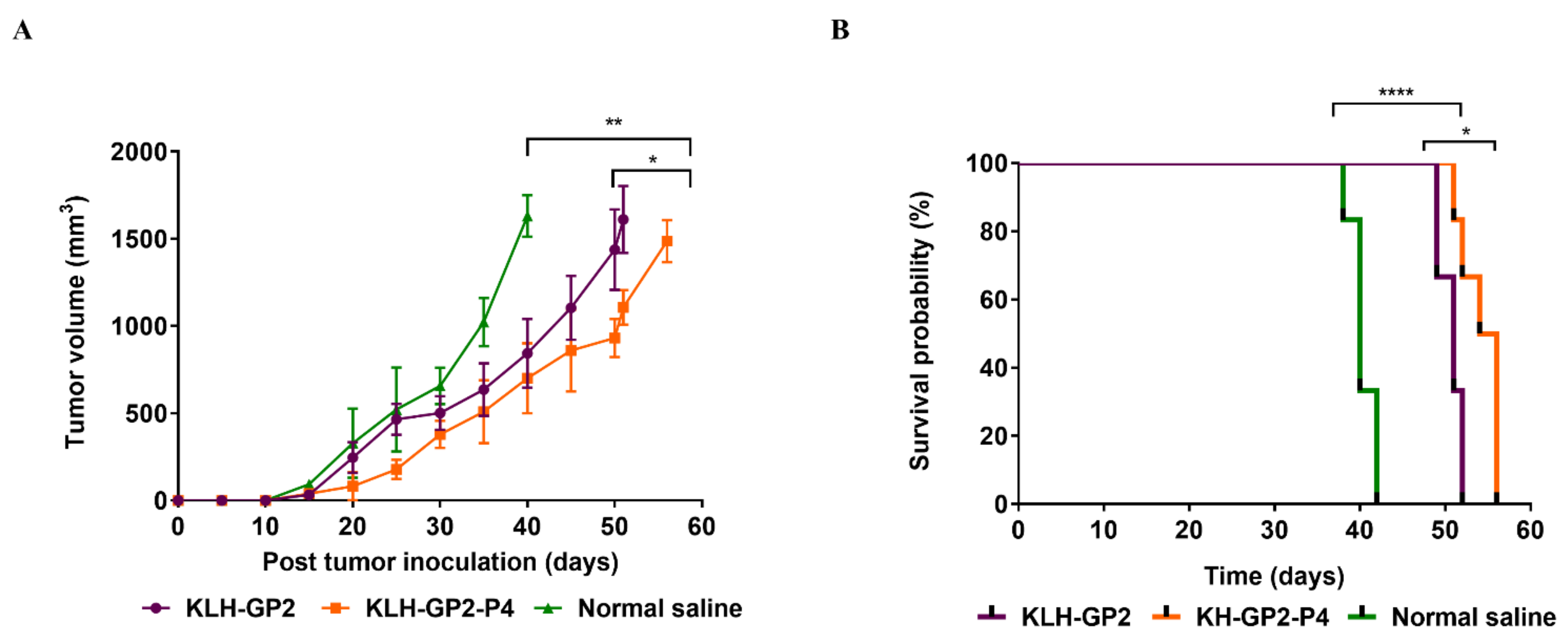
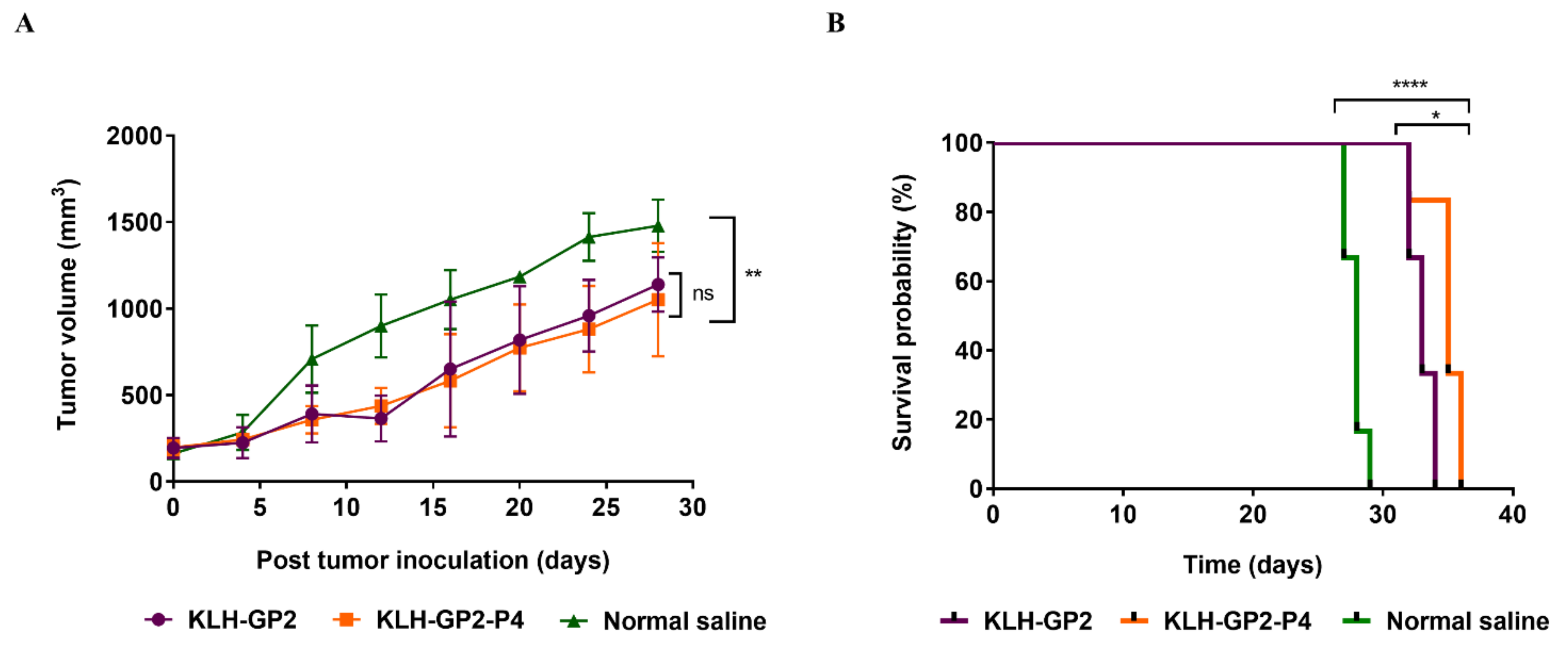
| Vaccine Candidates | MST a | TTE b | TGD (%) c | ILS (%) d |
|---|---|---|---|---|
| KLH–GP2 | 50 | 51 ± 2.12 ** | 27.5 ** | 25 ** |
| KLH–GP2–P4 | 56 | 56 ± 3.27 ** | 33 *** | 40 *** |
| Normal saline | 40 | 40 ± 1.51 | 0 | 0 |
| Vaccine Candidate | MST a | TTE b | TGD (%) c | ILS (%) d |
|---|---|---|---|---|
| KLH–GP2 | 33 | 34 ± 1.22 * | 17 * | 21 * |
| KLH–GP2–P4 | 35 | 36 ± 1.51 * | 24 ** | 25 ** |
| Normal saline | 28 | 29 ± 0.75 | 0 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nordin, M.L.; Mohamad Norpi, A.S.; Ng, P.Y.; Yusoff, K.; Abu, N.; Lim, K.P.; Azmi, F. HER2/neu-Based Peptide Vaccination-Pulsed with B-Cell Epitope Induced Efficient Prophylactic and Therapeutic Antitumor Activities in TUBO Breast Cancer Mice Model. Cancers 2021, 13, 4958. https://doi.org/10.3390/cancers13194958
Nordin ML, Mohamad Norpi AS, Ng PY, Yusoff K, Abu N, Lim KP, Azmi F. HER2/neu-Based Peptide Vaccination-Pulsed with B-Cell Epitope Induced Efficient Prophylactic and Therapeutic Antitumor Activities in TUBO Breast Cancer Mice Model. Cancers. 2021; 13(19):4958. https://doi.org/10.3390/cancers13194958
Chicago/Turabian StyleNordin, Muhammad Luqman, Abdin Shakirin Mohamad Norpi, Pei Yuen Ng, Khatijah Yusoff, Nadiah Abu, Kue Peng Lim, and Fazren Azmi. 2021. "HER2/neu-Based Peptide Vaccination-Pulsed with B-Cell Epitope Induced Efficient Prophylactic and Therapeutic Antitumor Activities in TUBO Breast Cancer Mice Model" Cancers 13, no. 19: 4958. https://doi.org/10.3390/cancers13194958
APA StyleNordin, M. L., Mohamad Norpi, A. S., Ng, P. Y., Yusoff, K., Abu, N., Lim, K. P., & Azmi, F. (2021). HER2/neu-Based Peptide Vaccination-Pulsed with B-Cell Epitope Induced Efficient Prophylactic and Therapeutic Antitumor Activities in TUBO Breast Cancer Mice Model. Cancers, 13(19), 4958. https://doi.org/10.3390/cancers13194958





