Tumor-Associated Macrophages in Bladder Cancer: Biological Role, Impact on Therapeutic Response and Perspectives for Immunotherapy
Abstract
:Simple Summary
Abstract
1. Introduction
1.1. Tumor-Associated Macrophages
1.2. Bladder Cancer
2. Macrophages in Bladder Cancer
2.1. Association of Macrophages, Clinicopathological Features and Outcomes in Bladder Cancer
2.2. Bladder Cancer Recruits TAMs
2.3. Bladder Cancer Favors M2-like Polarization of TAM to Promote Tumor Progression
3. Macrophages Influence Bladder Cancer Treatments
3.1. Macrophages and NMIBC Treatments
3.1.1. Transurethral Resection of Bladder Tumor (TURBT)
3.1.2. Bacillus Calmette-Guérin (BCG)
3.2. Macrophages and MIBC Treatments
3.2.1. Radical cystectomy (RC)
3.2.2. Chemotherapy
3.2.3. Immune Checkpoint Inhibitors (ICIs)
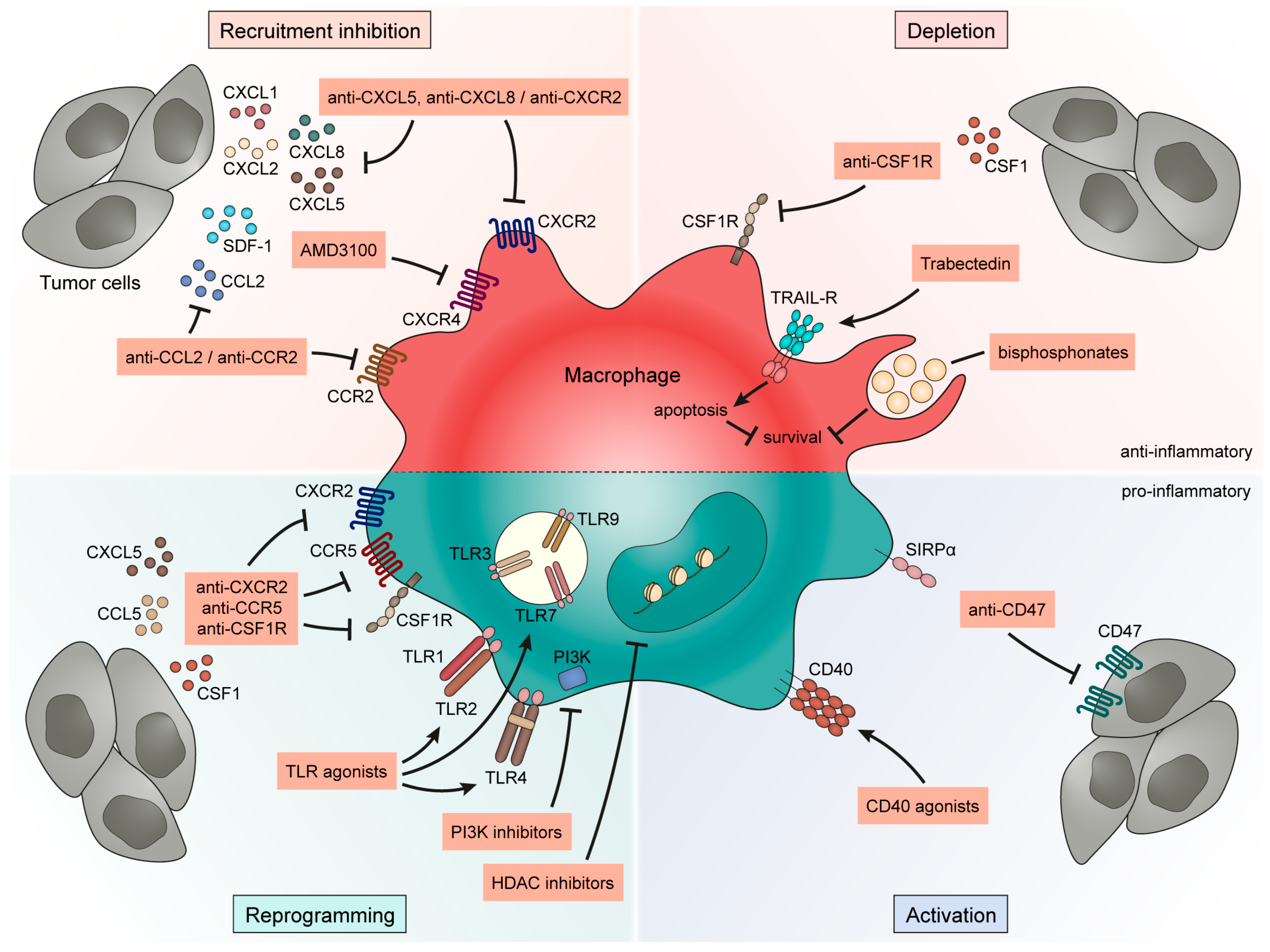
4. Targeting TAMs to Improve Bladder Cancer Outcome
4.1. Strategies to Inhibit Macrophage Recruitment in Bladder Cancer
4.1.1. CCL2-CCR2 Axis
4.1.2. Stromal Cell-Derived Factor 1 (SDF-1)-CXCR4 Axis
4.1.3. CXCL-CXCR2 Axis
4.2. Macrophage Depletion for Bladder Cancer Treatment
4.2.1. CSF1-CSF1 R Axis
4.2.2. Trabectedin
4.2.3. Bisphosphonates
4.3. Reprogramming of Tumor-Associated Macrophages in Bladder Cancer
4.3.1. Chemokines
4.3.2. Toll-like Receptor (TLR) Agonists
4.3.3. Histone Deacetylase (HDAC) Inhibitors
4.3.4. Phosphoinositide 3-Kinase (PI3 K) Inhibitors
4.4. Activation of Tumor-Associated Macrophages in Bladder Cancer
4.4.1. CD40-CD40 L Pathway
4.4.2. CD47-Signal Regulatory Protein-α (SIRPα) Axis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mariano, L.L.; Ingersoll, M.A. Bladder resident macrophages: Mucosal sentinels. Cell. Immunol. 2018, 330, 136–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engel, D.R.; Maurer, J.; Tittel, A.P.; Weisheit, C.; Cavlar, T.; Schumak, B.; Limmer, A.; Van Rooijen, N.; Trautwein, C.; Tacke, F.; et al. CCR2 Mediates Homeostatic and Inflammatory Release of Gr1highMonocytes from the Bone Marrow, but Is Dispensable for Bladder Infiltration in Bacterial Urinary Tract Infection. J. Immunol. 2008, 181, 5579–5586. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Waldhuber, A.; Puthia, M.; Wieser, A.; Cirl, C.; Dürr, S.; Neumann-Pfeifer, S.; Albrecht, S.; Römmler, F.; Müller, T.; Zheng, Y.; et al. Uropathogenic Escherichia coli strain CFT073 disrupts NLRP3 inflammasome activation. J. Clin. Investig. 2016, 126, 2425–2436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mora-Bau, G.; Platt, A.M.; Van Rooijen, N.; Randolph, G.J.; Albert, M.L.; Ingersoll, M.A. Macrophages Subvert Adaptive Immunity to Urinary Tract Infection. PLoS Pathog. 2015, 11, e1005044. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Marchesi, F.; Malesci, A.; Laghi, L. Europe PMC Funders Group Tumor-Associated Macrophages as Treatment Targets in Oncology. Nat. Rev. Clin. Oncol. 2018, 14, 399–416. [Google Scholar] [CrossRef] [PubMed]
- Guttman, O.; Lewis, E.C. M2-like Macrophages and Tumor-Associated Macrophages: Overlapping and Distinguishing Properties En Route to a Safe Therapeutic Potential. Integr. Cancer Sci. Ther. 2016, 3, 554–561. [Google Scholar]
- Bottazzi, B.; Walter, S.; Govoni, D.; Colotta, F.; Mantovani, A. Monocyte chemotactic cytokine gene transfer modulates macrophage infiltration, growth and susceptibility to IL-2 therapy of a murine melanoma. Cytokine 1991, 3, 519–1285. [Google Scholar] [CrossRef]
- Sinha, P.; Clements, V.K.; Ostrand-Rosenberg, S. Reduction of Myeloid-Derived Suppressor Cells and Induction of M1 Macrophages Facilitate the Rejection of Established Metastatic Disease. J. Immunol. 2005, 174, 636–645. [Google Scholar] [CrossRef] [Green Version]
- Takao, S.; Smith, E.H.; Wang, D.; Chan, C.K.; Bulkley, G.B.; Klein, A.S. Role of reactive oxygen metabolites in murine peritoneal macrophage phagocytosis and phagocytic killing. Am. J. Physiol. Physiol. 1996, 271, C1278–C1284. [Google Scholar] [CrossRef]
- Wynn, T.A.; Chawla, A.; Pollard, J.W. Macrophage biology in development, homeostasis and disease. Nature 2013, 496, 445–455. [Google Scholar] [CrossRef]
- Noy, R.; Pollard, J.W. Tumor-Associated Macrophages: From Mechanisms to Therapy. Immunity 2014, 41, 49–61. [Google Scholar] [CrossRef] [Green Version]
- Mantovani, A.; Sica, A.; Sozzani, S.; Allavena, P.; Vecchi, A.; Locati, M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 2004, 25, 677–686. [Google Scholar] [CrossRef]
- Jayasingam, S.D.; Citartan, M.; Thang, T.H.; Zin, A.A.M.; Ang, K.C.; Ch’Ng, E.S. Evaluating the Polarization of Tumor-Associated Macrophages Into M1 and M2 Phenotypes in Human Cancer Tissue: Technicalities and Challenges in Routine Clinical Practice. Front. Oncol. 2020, 9, 1512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ge, Z.; Ding, S. The Crosstalk Between Tumor-Associated Macrophages (TAMs) and Tumor Cells and the Corresponding Targeted Therapy. Front. Oncol. 2020, 10, 590941. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Pamer, E.G. Monocyte recruitment during infection and inflammation. Nat. Rev. Immunol. 2011, 11, 762–774. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qian, B.-Z.; Pollard, J.W. Macrophage Diversity Enhances Tumor Progression and Metastasis. Cell 2010, 141, 39–51. [Google Scholar] [CrossRef] [Green Version]
- Gabrilovich, D.I.; Ostrand-Rosenberg, S.; Bronte, V. Coordinated regulation of myeloid cells by tumours. Nat. Rev. Immunol. 2012, 12, 253–268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rath, M.; Muller, I.; Kropf, P.; Closs, E.I.; Munder, M. Metabolism via Arginase or Nitric Oxide Synthase: Two Competing Arginine Pathways in Macrophages. Front. Immunol. 2014, 5, 532. [Google Scholar] [CrossRef] [Green Version]
- Sica, A.; Mantovani, A. Macrophage plasticity and polarization: In vivo veritas. J. Clin. Investig. 2012, 122, 787–795. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamat, A.M.; Hahn, N.M.; Efstathiou, J.A.; Lerner, S.P.; Malmström, P.-U.; Choi, W.; Guo, C.C.; Lotan, Y.; Kassouf, W. Bladder Cancer. Lancet 2016, 6736, 1–15. [Google Scholar] [CrossRef]
- Sanli, O.; Dobruch, J.; Knowles, M.A.; Burger, M.; Alemozaffar, M.; Nielsen, M.E.; Lotan, Y. Bladder Cancer. Nat. Rev. Dis. Prim. 2017, 3, 17022. [Google Scholar] [CrossRef] [PubMed]
- Knowles, M.A.; Hurst, C.D. Molecular biology of bladder cancer: New insights into pathogenesis and clinical diversity. Nat. Rev. Cancer 2015, 15, 25–41. [Google Scholar] [CrossRef] [PubMed]
- Fu, H.; Zhu, Y.; Wang, Y.; Liu, Z.; Zhang, J.; Xie, H.; Fu, Q.; Dai, B.; Ye, D.; Xu, J. Identification and Validation of Stromal Immunotype Predict Survival and Benefit from Adjuvant Chemotherapy in Patients with Muscle-Invasive Bladder Cancer. Clin. Cancer Res. 2018, 24, 3069–3078. [Google Scholar] [CrossRef] [Green Version]
- Wang, B.; Liu, H.; Dong, X.; Wu, S.; Zeng, H.; Liu, Z.; Wan, D.; Dong, W.; He, W.; Chen, X.; et al. High CD204+ tumor-infiltrating macrophage density predicts a poor prognosis in patients with urothelial cell carcinoma of the bladder. Oncotarget 2015, 6, 20204–20214. [Google Scholar] [CrossRef]
- Lima, L.; Oliveira, D.; Tavares, A.; Amaro, T.; Cruz, R.; Oliveira, M.J.; Ferreira, J.A.; Santos, L.L. The predominance of M2-polarized macrophages in the stroma of low-hypoxic bladder tumors is associated with BCG immunotherapy failure. Urol. Oncol. Semin. Orig. Investig. 2014, 32, 449–457. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Ye, Y.-L.; Li, M.-X.; Ye, S.-B.; Huang, W.-R.; Cai, T.-T.; He, J.; Peng, J.-Y.; Duan, T.-H.; Cui, J.; et al. CXCL2/MIF-CXCR2 signaling promotes the recruitment of myeloid-derived suppressor cells and is correlated with prognosis in bladder cancer. Oncogene 2016, 36, 2095–2104. [Google Scholar] [CrossRef] [PubMed]
- Saint, F.; Patard, J.; Muscatelli, B.G.; Belda, M.L.; Medina, S.G.D.D.; Abbou, C.; Chopin, D. Evaluation of cellular tumour rejection mechanisms in the peritumoral bladder wall after bacillus Calmette-Guérin treatment. BJU Int. 2001, 88, 602–610. [Google Scholar] [CrossRef] [Green Version]
- Hanada, T.; Nakagawa, M.; Emoto, A.; Nomura, T.; Nasu, N.; Nomura, Y. Prognostic value of tumor-associated macrophage count in human bladder cancer. Int. J. Urol. 2000, 7, 263–269. [Google Scholar] [CrossRef]
- Ayari, C.; LaRue, H.; Hovington, H.; Decobert, M.; Harel, F.; Bergeron, A.; Têtu, B.; Lacombe, L.; Fradet, Y. Bladder Tumor Infiltrating Mature Dendritic Cells and Macrophages as Predictors of Response to Bacillus Calmette-Guérin Immunotherapy. Eur. Urol. 2009, 55, 1386–1396. [Google Scholar] [CrossRef]
- Boström, M.M.; Irjala, H.; Mirtti, T.; Taimen, P.; Kauko, T.; Ålgars, A.; Jalkanen, S.; Boström, P.J. Tumor-Associated Macrophages Provide Significant Prognostic Information in Urothelial Bladder Cancer. PLoS ONE 2015, 10, e0133552. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Shen, W.; Zhang, Y.; Liu, M.; Zhang, L.; Liu, Q.; Lu, H.H.; Bo, J. Accumulation of myeloid-derived suppressor cells (MDSCs) induced by low levels of IL-6 correlates with poor prognosis in bladder cancer. Oncotarget 2017, 8, 38378–38388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, C.-P.; Liu, L.-X.; Shyr, C.-R. Tumor-associated Macrophages Facilitate Bladder Cancer Progression by Increasing Cell Growth, Migration, Invasion and Cytokine Expression. Anticancer. Res. 2020, 40, 2715–2724. [Google Scholar] [CrossRef] [PubMed]
- Miyake, M.; Tatsumi, Y.; Gotoh, D.; Ohnishi, S.; Owari, T.; Iida, K.; Ohnishi, K.; Hori, S.; Morizawa, Y.; Itami, Y.; et al. Regulatory T Cells and Tumor-Associated Macrophages in the Tumor Microenvironment in Non-Muscle Invasive Bladder Cancer Treated with Intravesical Bacille Calmette-Guérin: A Long-Term Follow-Up Study of a Japanese Cohort. Int. J. Mol. Sci. 2017, 18, 2186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Z.; Wang, L.; Tian, J.; Man, H.; Li, P.; Shan, B. High expression of B7-H3 and CD163 in cancer tissues indicates malignant clinicopathological status and poor prognosis of patients with urothelial cell carcinoma of the bladder. Oncol. Lett. 2018, 15, 6519–6526. [Google Scholar] [CrossRef] [Green Version]
- Takeuchi, H.; Tanaka, M.; Tanaka, A.; Tsunemi, A.; Yamamoto, H. Predominance of M2-polarized macrophages in bladder cancer affects angiogenesis, tumor grade and invasiveness. Oncol. Lett. 2016, 11, 3403–3408. [Google Scholar] [CrossRef] [Green Version]
- Martinez, V.M.G.; Rubio, C.; Martínez-Fernández, M.; Segovia, C.; López-Calderón, F.; I Garin, M.; Teijeira, A.; Munera-Maravilla, E.; Varas, A.; Sacedón, R.; et al. BMP4 Induces M2 Macrophage Polarization and Favors Tumor Progression in Bladder Cancer. Clin. Cancer Res. 2017, 23, 7388–7399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xue, Y.; Tong, L.; Liu, F.L.; Liu, A.; Zeng, S.; Xiong, Q.; Yang, Z.; He, X.; Sun, Y.; Xu, C. Tumor-infiltrating M2 macrophages driven by specific genomic alterations are associated with prognosis in bladder cancer. Oncol. Rep. 2019, 42, 581–594. [Google Scholar] [CrossRef]
- Leblond, M.M.; Tillé, L.; Nassiri, S.; Gilfillan, C.B.; Imbratta, C.; Schmittnaegel, M.; Ries, C.H.; Speiser, D.E.; Verdeil, G. CD40 Agonist Restores the Antitumor Efficacy of Anti-PD1 Therapy in Muscle-Invasive Bladder Cancer in an IFN I/II-Mediated Manner. Cancer Immunol. Res. 2020, 8, 1180–1192. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.-K.; Zhao, X.-K.; Xia, Y.-C.; Zhu, X.; Xiao, P. Increased Circulating Immunosuppressive CD14+HLA-DR−/Low Cells Correlate with Clinical Cancer Stage and Pathological Grade in Patients with Bladder Carcinoma. J. Int. Med. Res. 2011, 39, 1381–1391. [Google Scholar] [CrossRef] [PubMed]
- Chevalier, M.; Trabanelli, S.; Racle, J.; Salomé, B.; Cesson, V.; Gharbi, D.; Bohner, P.; Domingos-Pereira, S.; Dartiguenave, F.; Fritschi, A.-S.; et al. ILC2-modulated T cell-to-MDSC balance is associated with bladder cancer recurrence. J. Clin. Investig. 2017, 127, 2916–2929. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miyake, M.; Hori, S.; Morizawa, Y.; Tatsumi, Y.; Nakai, Y.; Anai, S.; Torimoto, K.; Aoki, K.; Tanaka, N.; Shimada, K.; et al. CXCL1-Mediated Interaction of Cancer Cells with Tumor-Associated Macrophages and Cancer-Associated Fibroblasts Promotes Tumor Progression in Human Bladder Cancer. Neoplasia 2016, 18, 636–646. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, G.; Zhang, L.; Liu, M.; Liu, Q.; Duan, X.; Bo, J. CD163+ macrophages predict a poor prognosis in patients with primary T1 high-grade urothelial carcinoma of the bladder. World J. Urol. 2019, 37, 2721–2726. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Hao, S.; Ye, Y.; Wei, J.; Tang, Y.; Tan, L.; Liao, Z.; Zhang, M.; Li, J.; Gui, C.; et al. Identification of an Immune-Related Risk Signature Correlates With Immunophenotype and Predicts Anti-PD-L1 Efficacy of Urothelial Cancer. Front. Cell Dev. Biol. 2021, 9, 646982. [Google Scholar] [CrossRef] [PubMed]
- Sjödahl, G.; Lövgren, K.; Lauss, M.; Chebil, G.; Patschan, O.; Gudjonsson, S.; Månsson, W.; Fernö, M.; Leandersson, K.; Lindgren, D.; et al. Infiltration of CD3+ and CD68+ cells in bladder cancer is subtype specific and affects the outcome of patients with muscle-invasive tumors11Grant support: The Swedish Cancer Society, the Swedish research council, the Nilsson Cancer foundation, the BioCARE Strategic Cancer Research program, the Lund Medical Faculty, and FoU Landstinget Kronoberg and Södra Regionvårdnämnden. Urol. Oncol. Semin. Orig. Investig. 2014, 32, 791–797. [Google Scholar] [CrossRef] [Green Version]
- Hu, B.; Wang, Z.; Zeng, H.; Qi, Y.; Chen, Y.; Wang, T.; Wang, J.; Chang, Y.; Bai, Q.; Xia, Y.; et al. Blockade of DC-SIGN+ tumor-associated macrophages reactivates anti-tumor immunity and improves immunotherapy in muscle-invasive bladder cancer. Cancer Res. 2020, 80, 1707–1719. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, L.-R.; Zhang, N.; Chen, S.-T.; He, J.; Liu, Y.-H.; Han, Y.-Q.; Shi, X.-Q.; Yang, J.-J.; Mu, D.-Y.; Fu, G.-H.; et al. PD-1-Positive Tumor-Associated Macrophages Define Poor Clinical Outcomes in Patients With Muscle Invasive Bladder Cancer Through Potential CD68/PD-1 Complex Interactions. Front. Oncol. 2021, 11, 679928. [Google Scholar] [CrossRef]
- Asano, T.; Ohnishi, K.; Shiota, T.; Motoshima, T.; Sugiyama, Y.; Yatsuda, J.; Kamba, T.; Ishizaka, K.; Komohara, Y. CD 169-positive sinus macrophages in the lymph nodes determine bladder cancer prognosis. Cancer Sci. 2018, 109, 1723–1730. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Sfakianos, J.P.; Beaumont, K.G.; Akturk, G.; Horowitz, A.; Sebra, R.P.; Farkas, A.M.; Gnjatic, S.; Hake, A.; Izadmehr, S.; et al. Myeloid Cell–associated Resistance to PD-1/PD-L1 Blockade in Urothelial Cancer Revealed Through Bulk and Single-cell RNA Sequencing. Clin. Cancer Res. 2021, 1–14. [Google Scholar] [CrossRef]
- Chai, C.-Y.; Chen, W.-T.; Hung, W.-C.; Kang, W.-Y.; Huang, Y.-C.; Su, Y.-C.; Yang, C.-H. Hypoxia-inducible factor-1 expression correlates with focal macrophage infiltration, angiogenesis and unfavourable prognosis in urothelial carcinoma. J. Clin. Pathol. 2008, 61, 658–664. [Google Scholar] [CrossRef]
- Chen, C.; He, W.; Huang, J.; Wang, B.; Li, H.; Cai, Q.; Su, F.; Bi, J.; Liu, H.; Zhang, B.; et al. LNMAT1 promotes lymphatic metastasis of bladder cancer via CCL2 dependent macrophage recruitment. Nat. Commun. 2018, 9, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Cheah, M.T.; Chen, J.Y.; Sahoo, D.; Contreras-Trujillo, H.; Volkmer, A.K.; Scheeren, F.; Volkmer, J.-P.; Weissman, I.L. CD14-expressing cancer cells establish the inflammatory and proliferative tumor microenvironment in bladder cancer. Proc. Natl. Acad. Sci. USA 2015, 112, 4725–4730. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Onita, T.; Ji, P.G.; Xuan, J.W.; Sakai, H.; Kanetake, H.; Maxwell, P.H.; Fong, G.-H.; Gabril, M.Y.; Moussa, M.; Chin, J.L. Hypoxia-induced, perinecrotic expression of endothelial Per-ARNT-Sim domain protein-1/hypoxia-inducible factor-2alpha correlates with tumor progression, vascularization, and focal macrophage infiltration in bladder cancer. Clin. Cancer Res. 2002, 8, 471–480. [Google Scholar]
- Koga, F.; Kageyama, Y.; Kawakami, S.; Fujii, Y.; Hyochi, N.; Ando, N.; Takizawa, T.; Saito, K.; Iwai, A.; Masuda, H.; et al. Prognostic Significance of Endothelial Per-Arnt-Sim Domain Protein 1/Hypoxia-Inducible Factor-2α Expression in a Subset of Tumor Associated Macrophages in Invasive Bladder Cancer. J. Urol. 2004, 171, 1080–1084. [Google Scholar] [CrossRef] [PubMed]
- Prima, V.; Kaliberova, L.N.; Kaliberov, S.; Curiel, D.T.; Kusmartsev, S. COX2/MPGES1/PGE2 Pathway Regulates PD-L1 Expression in Tumor-Associated Macrophages and Myeloid-Derived Suppressor Cells. Proc. Natl. Acad. Sci. USA 2017, 114, 1117–1122. [Google Scholar] [CrossRef] [Green Version]
- Wu, A.T.H.; Srivastava, P.; Yadav, V.K.; Tzeng, D.T.W.; Iamsaard, S.; Su, E.C.Y.; Hsiao, M.; Liu, M.C. Ovatodiolide, Isolated from Anisomeles Indica, Suppresses Bladder Carcinogenesis through Suppression of MTOR/β-Catenin/CDK6 and Exosomal MiR-21 Derived from M2 Tumor-Associated Macrophages. Toxicol. Appl. Pharmacol. 2020, 401, 115109. [Google Scholar] [CrossRef]
- Wang, X.; Ni, S.; Chen, Q.; Ma, L.; Jiao, Z.; Wang, C.; Jia, G. Bladder cancer cells induce immunosuppression of T cells by supporting PD-L1 expression in tumour macrophages partially through interleukin 10. Cell Biol. Int. 2017, 41, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wang, D.; Xu, T.; Liu, P.; Cao, Y.; Wang, Y.; Yang, X.; Xu, X.; Wang, X.; Niu, H. Bladder cancer cells re-educate TAMs through lactate shuttling in the microfluidic cancer microenvironment. Oncotarget 2015, 6, 39196–39210. [Google Scholar] [CrossRef] [Green Version]
- Lin, F.; Yin, H.; Li, X.; Zhu, G.; He, W.; Gou, X. Bladder cancer cell-secreted exosomal miR-21 activates the PI3K/AKT pathway in macrophages to promote cancer progression. Int. J. Oncol. 2019, 56, 151–164. [Google Scholar] [CrossRef] [Green Version]
- Leblond, M.M.; Gérault, A.; Corroyer-Dulmont, A.; MacKenzie, E.T.; Petit, E.; Bernaudin, M.; Valable, S. Hypoxia induces macrophage polarization and re-education toward an M2 phenotype in U87 and U251 glioblastoma models. OncoImmunology 2015, 5, e1056442. [Google Scholar] [CrossRef] [Green Version]
- Takeda, N.; O’Dea, E.L.; Doedens, A.; Kim, J.; Weidemann, A.; Stockmann, C.; Asagiri, M.; Simon, M.C.; Hoffmann, A.; Johnson, R.S. Differential Activation and Antagonistic Function of HIF-α Isoforms in Macrophages Are Essential for NO Homeostasis. Genes Dev. 2010, 24, 491–501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colegio, O.R.; Chu, N.-Q.; Szabo, A.L.; Chu, T.; Rhebergen, A.M.; Jairam, V.; Cyrus, N.; Brokowski, C.E.; Eisenbarth, S.C.; Phillips, G.M.; et al. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature 2014, 513, 559–563. [Google Scholar] [CrossRef]
- Dufresne, M.; Dumas, G.; Asselin, É.; Carrier, C.; Pouliot, M.; Reyes-Moreno, C. Pro-inflammatory type-1 and anti-inflammatory type-2 macrophages differentially modulate cell survival and invasion of human bladder carcinoma T24 cells. Mol. Immunol. 2011, 48, 1556–1567. [Google Scholar] [CrossRef] [PubMed]
- Reusser, N.M.; Dalton, H.J.; Pradeep, S.; Gonzalez-Villasana, V.; Jennings, N.B.; Vasquez, H.G.; Wen, Y.; Rupaimoole, R.; Nagaraja, A.S.; Gharpure, K.; et al. Clodronate inhibits tumor angiogenesis in mouse models of ovarian cancer. Cancer Biol. Ther. 2014, 15, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Tervahartiala, M.; Taimen, P.; Mirtti, T.; Koskinen, I.; Ecke, T.; Jalkanen, S.; Boström, P.J. Immunological tumor status may predict response to neoadjuvant chemotherapy and outcome after radical cystectomy in bladder cancer. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Yang, H.; Kim, C.; Kim, M.-J.; A Schwendener, R.; Alitalo, K.; Heston, W.; Kim, I.; Kim, W.-J.; Koh, G.Y. Soluble vascular endothelial growth factor receptor-3 suppresses lymphangiogenesis and lymphatic metastasis in bladder cancer. Mol. Cancer 2011, 10, 36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Q.; Mao, Z.; Sun, J. NF-ΚB Inhibitor, BAY11-7082, Suppresses M2 Tumor-Associated Macrophage Induced EMT Potential via MiR-30a/NF-ΚB/Snail Signaling in Bladder Cancer Cells. Gene 2019, 710, 91–97. [Google Scholar] [CrossRef]
- Wu, H.; Zhang, X.; Han, D.; Cao, J.; Tian, J. Tumour-associated macrophages mediate the invasion and metastasis of bladder cancer cells through CXCL8. PeerJ 2020, 8, e8721. [Google Scholar] [CrossRef]
- Ahmed, M.; Sottnik, J.L.; Dancik, G.M.; Sahu, D.; Hansel, D.E.; Theodorescu, D.; Schwartz, M.A. An Osteopontin/CD44 Axis in RhoGDI2-Mediated Metastasis Suppression. Cancer Cell 2016, 30, 432–443. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiu, S.; Deng, L.; Liao, X.; Nie, L.; Qi, F.; Jin, K.; Tu, X.; Zheng, X.; Li, J.; Liu, L.; et al. Tumor-associated macrophages promote bladder tumor growth through PI3K/AKT signal induced by collagen. Cancer Sci. 2019, 110, 2110–2118. [Google Scholar] [CrossRef] [PubMed]
- Eruslanov, E.; Stoffs, T.L.; Kim, W.-J.; Daurkin, I.; Gilbert, S.M.; Su, L.-M.; Vieweg, J.; Daaka, Y.; Kusmartsev, S. Expansion of CCR8+ Inflammatory Myeloid Cells in Cancer Patients with Urothelial and Renal Carcinomas. Clin. Cancer Res. 2013, 19, 1670–1680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dominguez-Gutierrez, P.R.; Kwenda, E.P.; Donelan, W.; O’Malley, P.; Crispen, P.L.; Kusmartsev, S. Hyal2 Expression in Tumor-Associated Myeloid Cells Mediates Cancer-Related Inflammation in Bladder Cancer. Cancer Res. 2021, 81, 648–657. [Google Scholar] [CrossRef]
- Qi, Y.; Chang, Y.; Wang, Z.; Chen, L.; Kong, Y.; Zhang, P.; Liu, Z.; Zhou, Q.; Chen, Y.; Wang, J.; et al. Tumor-associated macrophages expressing galectin-9 identify immunoevasive subtype muscle-invasive bladder cancer with poor prognosis but favorable adjuvant chemotherapeutic response. Cancer Immunol. Immunother. 2019, 68, 2067–2080. [Google Scholar] [CrossRef]
- Takeyama, Y.; Kato, M.; Tamada, S.; Azuma, Y.; Shimizu, Y.; Iguchi, T.; Yamasaki, T.; Gi, M.; Wanibuchi, H.; Nakatani, T. Myeloid-derived suppressor cells are essential partners for immune checkpoint inhibitors in the treatment of cisplatin-resistant bladder cancer. Cancer Lett. 2020, 479, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Beltran, A.; Cimadamore, A.; Blanca, A.; Massari, F.; Vau, N.; Scarpelli, M.; Cheng, L.; Montironi, R. Immune Checkpoint Inhibitors for the Treatment of Bladder Cancer. Cancers 2021, 13, 131. [Google Scholar] [CrossRef] [PubMed]
- De Palma, M.; Lewis, C.E. Macrophage Regulation of Tumor Responses to Anticancer Therapies. Cancer Cell 2013, 23, 277–286. [Google Scholar] [CrossRef] [Green Version]
- Fallah, J.; Diaz-Montero, C.M.; Rayman, P.; Wei, W.; Finke, J.H.; Kim, J.S.; Pavicic, P.G.; Lamenza, M.; Dann, P.; Company, D.; et al. Myeloid-Derived Suppressor Cells in Nonmetastatic Urothelial Carcinoma of Bladder Is Associated With Pathologic Complete Response and Overall Survival. Clin. Genitourin. Cancer 2020, 18, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, H.; Torigoe, T.; Honma, I.; Sato, E.; Asanuma, H.; Hirohashi, Y.; Sato, N.; Tsukamoto, T. Effect of Human Leukocyte Antigen Class I Expression of Tumor Cells on Outcome of Intravesical Instillation of Bacillus Calmette-Guerin Immunotherapy for Bladder Cancer. Clin. Cancer Res. 2006, 12, 4641–4644. [Google Scholar] [CrossRef] [Green Version]
- Takayama, H.; Nishimura, K.; Tsujimura, A.; Nakai, Y.; Nakayama, M.; Aozasa, K.; Okuyama, A.; Nonomura, N. Increased Infiltration of Tumor Associated Macrophages is Associated With Poor Prognosis of Bladder Carcinoma In Situ After Intravesical Bacillus Calmette-Guerin Instillation. J. Urol. 2009, 181, 1894–1900. [Google Scholar] [CrossRef]
- Ajili, F.; Kourda, N.; Darouiche, A.; Chebil, M.; Boubaker, S. Prognostic Value of Tumor-associated Macrophages Count in Human Non-muscle-invasive Bladder Cancer Treated by BCG Immunotherapy. Ultrastruct. Pathol. 2013, 37, 56–61. [Google Scholar] [CrossRef]
- Parizi, M.K.; Shariat, S.F.; Margulis, V.; Mori, K.; Lotan, Y. Value of tumour-infiltrating immune cells in predicting response to intravesical BCG in patients with non-muscle-invasive bladder cancer: A systematic review and meta-analysis. BJU Int. 2021, 127, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Suriano, F.; Santini, D.; Perrone, G.; Amato, M.; Vincenzi, B.; Tonini, G.; Muda, A.O.; Boggia, S.; Buscarini, M.; Pantano, F. Tumor associated macrophages polarization dictates the efficacy of BCG instillation in non-muscle invasive urothelial bladder cancer. J. Exp. Clin. Cancer Res. 2013, 32, 87. [Google Scholar] [CrossRef] [Green Version]
- Pichler, R.; Fritz, J.; Zavadil, C.; Schäfer, G.; Culig, Z.; Brunner, A. Tumor-infiltrating immune cell subpopulations influence the oncologic outcome after intravesical Bacillus Calmette-Guérin therapy in bladder cancer. Oncotarget 2016, 7, 39916–39930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taubert, H.; Eckstein, M.; Epple, E.; Jung, R.; Weigelt, K.; Lieb, V.; Sikic, D.; Stöhr, R.; Geppert, C.; Weyerer, V.; et al. Immune Cell-Associated Protein Expression Helps to Predict Survival in Muscle-Invasive Urothelial Bladder Cancer Patients after Radical Cystectomy and Optional Adjuvant Chemotherapy. Cells 2021, 10, 159. [Google Scholar] [CrossRef] [PubMed]
- Ornstein, M.; Diaz-Montero, C.M.; Rayman, P.; Elson, P.; Haywood, S.; Finke, J.H.; Kim, J.S.; Pavicic, P.G.; Lamenza, M.; Devonshire, S.; et al. Myeloid-derived suppressors cells (MDSC) correlate with clinicopathologic factors and pathologic complete response (PCR) in patients with urothelial carcinoma (UC) undergoing cystectomy. Urol. Oncol. Semin. Orig. Investig. 2018, 36, 405–412. [Google Scholar] [CrossRef]
- Mariathasan, S.; Turley, S.J.; Nickles, D.; Castiglioni, A.; Yuen, K.; Wang, Y.; Kadel, E.E., III; Koeppen, H.; Astarita, J.L.; Cubas, R.; et al. TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature 2018, 554, 544–548. [Google Scholar] [CrossRef]
- Powles, T.; Eder, J.P.; Fine, G.D.; Braiteh, F.S.; Loriot, Y.; Cruz, C.; Bellmunt, J.; Burris, H.A.; Petrylak, D.P.; Teng, S.-L.; et al. MPDL3280A (anti-PD-L1) treatment leads to clinical activity in metastatic bladder cancer. Nature 2014, 515, 558–562. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, J.E.; Hoffman-Censits, J.; Powles, T.; van der Heijden, M.S.; Balar, A.; Necchi, A.; Dawson, N.; O’Donnell, P.H.; Balmanoukian, A.; Loriot, Y.; et al. Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: A single-arm, multicentre, phase 2 trial. Lancet 2016, 387, 1909–1920. [Google Scholar] [CrossRef] [Green Version]
- Powles, T.; Durán, I.; Van Der Heijden, M.S.; Loriot, Y.; Vogelzang, N.J.; De Giorgi, U.; Oudard, S.; Retz, M.M.; Castellano, D.; Bamias, A.; et al. Atezolizumab versus chemotherapy in patients with platinum-treated locally advanced or metastatic urothelial carcinoma (IMvigor211): A multicentre, open-label, phase 3 randomised controlled trial. Lancet 2018, 391, 748–757. [Google Scholar] [CrossRef]
- Bellmunt, J.; De Wit, R.; Vaughn, D.J.; Fradet, Y.; Lee, J.-L.; Fong, L.; Vogelzang, N.J.; Climent, M.A.; Petrylak, D.P.; Choueiri, T.K.; et al. Pembrolizumab as Second-Line Therapy for Advanced Urothelial Carcinoma. N. Engl. J. Med. 2017, 376, 1015–1026. [Google Scholar] [CrossRef] [Green Version]
- Sharma, P.; Siefker-Radtke, A.; De Braud, F.; Basso, U.; Calvo, E.; Bono, P.; Morse, M.A.; Ascierto, P.A.; Lopez-Martin, J.; Brossart, P.; et al. Nivolumab Alone and With Ipilimumab in Previously Treated Metastatic Urothelial Carcinoma: CheckMate 032 Nivolumab 1 mg/kg Plus Ipilimumab 3 mg/kg Expansion Cohort Results. J. Clin. Oncol. 2019, 37, 1608–1616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeng, D.; Ye, Z.; Wu, J.; Zhou, R.; Fan, X.; Wang, G.; Huang, Y.; Wu, J.; Sun, H.; Wang, M.; et al. Macrophage correlates with immunophenotype and predicts anti-PD-L1 response of urothelial cancer. Theranostics 2020, 10, 7002–7014. [Google Scholar] [CrossRef] [PubMed]
- Anfray, C.; Ummarino, A.; Andón, F.T.; Allavena, P. Current Strategies to Target Tumor-Associated-Macrophages to Improve Anti-Tumor Immune Responses. Cells 2019, 9, 46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Redelman-Sidi, G.; Glickman, M.S.; Bochner, B. The mechanism of action of BCG therapy for bladder cancer—A current perspective. Nat. Rev. Urol. 2014, 11, 153–162. [Google Scholar] [CrossRef]
- Miyake, M.; Hori, S.; Ohnishi, S.; Owari, T.; Iida, K.; Ohnishi, K.; Morizawa, Y.; Gotoh, D.; Itami, Y.; Nakai, Y.; et al. Clinical Impact of the Increase in Immunosuppressive Cell-Related Gene Expression in Urine Sediment during Intravesical Bacillus Calmette-Guérin. Diseases 2019, 7, 44. [Google Scholar] [CrossRef] [Green Version]
- De Boer, E.C.; De Jong, W.H.; Van Der Meijden, A.P.M.; Steerenberg, P.A.; Witjes, F.; Vegt, P.D.J.; Debruyne, F.M.J.; Ruitenberg, E.J. Leukocytes in the urine after intravesical BCG treatment for superficial bladder cancer. Urol. Res. 1991, 19, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.M.; Alexandroff, A.B.; Kelly, R.W.; Skibinska, A.; Esuvaranathan, K.; Prescott, S.; Chisholm, G.D.; James, K. Changes in urinary cytokines and soluble intercellular adhesion molecule-1 (ICAM-1) in bladder cancer patients after Bacillus Calmette-Guérin (BCG) immunotherapy. Clin. Exp. Immunol. 2008, 99, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Reale, R.I.M.; Reale, M.; Intorno, R.; Tenaglia, R.; Feliciani, C.; Barbacane, R.C.; Santoni, A.; Conti, P. Production of MCP-1 and RANTES in bladder cancer patients after bacillus Calmette-Guerin immunotherapy. Cancer Immunol. Immunother. 2002, 51, 91–98. [Google Scholar] [CrossRef]
- Luo, Y.; Chen, X.; O’Donnell, M.A. Mycobacterium bovis bacillus Calmette-Guérin (BCG) induces human CC- and CXC-chemokines in vitro and in vivo. Clin. Exp. Immunol. 2006, 147, 370–378. [Google Scholar] [CrossRef] [PubMed]
- Tan, Q.L.; Zhou, C.Y.; Cheng, L.; Luo, M.; Liu, C.P.; Xu, W.X.; Zhang, X.; Zeng, X. Immunotherapy of Bacillus Calmette-Guérin by Targeting Macrophages against Bladder Cancer in a NOD/Scid IL2Rg-/- Mouse Model. Mol. Med. Rep. 2020, 22, 362–370. [Google Scholar]
- De Boer, E.C.; De Jong, W.H.; Steerenberg, P.A.; Aarden, L.A.; Tetteroo, E.; De Groot, E.R.; Van Der Meijden, A.P.M.; Vegt, P.D.J.; Debruyne, F.M.J.; Ruitenberg, E.J. Induction of urinary interleukin-1 (IL-1), IL-2, IL-6, and tumour necrosis factor during intravesical immunotherapy with bacillus Calmette-Guérin in superficial bladder cancer. Cancer Immunol. Immunother. 1992, 34, 306–312. [Google Scholar] [CrossRef]
- Pryor, K.; Goddard, J.H.R.; Goldstein, D.M.; Stricker, P.D.; Russell, P.; Golovsky, D.; Penny, R. Bacillus Calmette-Guerin (BCG) enhances monocyte- and lymphocyte-mediated bladder tumour cell killing. Br. J. Cancer 1995, 71, 801–807. [Google Scholar] [CrossRef] [Green Version]
- Yamada, H.; Matsumoto, S.; Matsumoto, T.; Yamada, T.; Yamashita, U. MURINE IL-2 Secreting Recombinant Bacillus Calmette-Guérin Augments Macrophage-Mediated Cytotoxicity Against Murine Bladder Cancer MBT-2. J. Urol. 2000, 164, 526–531. [Google Scholar] [CrossRef]
- Luo, Y.; Yamada, H.; Evanoff, D.P.; Chen, X. Role of Th1-Stimulating Cytokines in Bacillus Calmette-Guérin (BCG)-Induced Macrophage Cytotoxicity against Mouse Bladder Cancer MBT-2 Cells. Clin. Exp. Immunol. 2006, 146, 181–188. [Google Scholar] [CrossRef]
- Luo, Y.; Han, R.; Evanoff, D.P.; Chen, X. Interleukin-10 inhibits Mycobacterium bovis bacillus Calmette-Guérin (BCG)-induced macrophage cytotoxicity against bladder cancer cells. Clin. Exp. Immunol. 2010, 160, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Copland, A.; Sparrow, A.; Hart, P.; Diogo, G.R.; Paul, M.; Azuma, M.; Reljic, R. Bacillus Calmette-Guérin Induces PD-L1 Expression on Antigen-Presenting Cells via Autocrine and Paracrine Interleukin-STAT3 Circuits. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Lodillinsky, C.; Langle, Y.; Guionet, A.; Gongora, A.; Baldi, A.; Sandes, E.O.; Casabé, A.; Eiján, A.M. Bacillus Calmette Guerin Induces Fibroblast Activation Both Directly and through Macrophages in a Mouse Bladder Cancer Model. PLoS ONE 2010, 5, e13571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sahai, E.; Astsaturov, I.; Cukierman, E.; DeNardo, D.G.; Egeblad, M.; Evans, R.M.; Fearon, D.; Greten, F.R.; Hingorani, S.R.; Hunter, T.; et al. A framework for advancing our understanding of cancer-associated fibroblasts. Nat. Rev. Cancer 2020, 20, 174–186. [Google Scholar] [CrossRef] [Green Version]
- Inman, B.A.; Sebo, T.J.; Frigola, X.; Dong, H.; Bergstralh, E.J.; Fradet, Y.; Lacombe, L.; Kwon, E.D. 879: PD-L1 (B7-H1) Expression by Urothelial Carcinoma of the Bladder and BCG-Induced Granulomata: Associations with Localized Stage Progression. J. Urol. 2007, 177, 292. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, S.-D.; McCrudden, C.; Chan, K.-W.; Lin, Y.; Kwok, H.-F. The prognostic significance of PD-L1 in bladder cancer. Oncol. Rep. 2015, 33, 3075–3084. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, B.; Pan, W.; Yang, M.; Yang, W.; He, W.; Chen, X.; Bi, J.; Jiang, N.; Huang, J.; Lin, T. Programmed death ligand-1 is associated with tumor infiltrating lymphocytes and poorer survival in urothelial cell carcinoma of the bladder. Cancer Sci. 2019, 110, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Xylinas, E.; Robinson, B.; Kluth, L.; Volkmer, B.; Hautmann, R.; Küfer, R.; Zerbib, M.; Kwon, E.; Thompson, R.; Boorjian, S.; et al. Association of T-cell co-regulatory protein expression with clinical outcomes following radical cystectomy for urothelial carcinoma of the bladder. Eur. J. Surg. Oncol. 2014, 40, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, J.; Wada, Y.; Matsumoto, K.; Azuma, M.; Kikuchi, K.; Ueda, S. Overexpression of B7-H1 (PD-L1) significantly associates with tumor grade and postoperative prognosis in human urothelial cancers. Cancer Immunol. Immunother. 2007, 56, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- Boorjian, S.A.; Sheinin, Y.; Crispen, P.L.; Farmer, S.A.; Lohse, C.M.; Kuntz, S.M.; Leibovich, B.C.; Kwon, E.D.; Frank, I. T-Cell Coregulatory Molecule Expression in Urothelial Cell Carcinoma: Clinicopathologic Correlations and Association with Survival. Clin. Cancer Res. 2008, 14, 4800–4808. [Google Scholar] [CrossRef] [Green Version]
- Bellmunt, J.; Mullane, S.A.; Werner, L.; Fay, A.P.; Callea, M.; Leow, J.; Taplin, M.E.; Choueiri, T.K.; Hodi, F.S.; Freeman, G.J.; et al. Association of PD-L1 expression on tumor-infiltrating mononuclear cells and overall survival in patients with urothelial carcinoma. Ann. Oncol. 2015, 26, 812–817. [Google Scholar] [CrossRef] [PubMed]
- Pichler, R.; Heidegger, I.; Fritz, J.; Danzl, M.; Sprung, S.; Zelger, B.; Brunner, A.; Pircher, A. PD-L1 expression in bladder cancer and metastasis and its influence on oncologic outcome after cystectomy. Oncotarget 2017, 8, 66849–66864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, D.H.; Jeong, J.Y.; Song, W. Prognostic Value of Programmed Death Ligand-1 Expression on Tumor-Infiltrating Immune Cells in Patients Treated with Cisplatin-Based Combination Adjuvant Chemotherapy Following Radical Cystectomy for Muscle-Invasive Bladder Cancer: A Retrospective Cohort Study. OncoTargets Ther. 2021, ume 14, 845–855. [Google Scholar] [CrossRef]
- Wankowicz, S.; Werner, L.; Orsola, A.; Novak, J.; Bowden, M.; Choueiri, T.K.; De Torres, I.; Morote, J.; Freeman, G.J.; Signoretti, S.; et al. Differential Expression of PD-L1 in High Grade T1 vs Muscle Invasive Bladder Carcinoma and its Prognostic Implications. J. Urol. 2017, 198, 817–823. [Google Scholar] [CrossRef]
- Breyer, J.; Wirtz, R.M.; Otto, W.; Erben, P.; Worst, T.; Stoehr, R.; Eckstein, M.; Denzinger, S.; Burger, M.; Hartmann, A. High PDL1 mRNA expression predicts better survival of stage pT1 non-muscle-invasive bladder cancer (NMIBC) patients. Cancer Immunol. Immunother. 2017, 67, 403–412. [Google Scholar] [CrossRef]
- Alexandrov, L.B.; Nik-Zainal, S.; Wedge, D.C.; Aparicio, S.A.J.R.; Behjati, S.; Biankin, A.V.; Bignell, G.R.; Bolli, N.; Borg, A.; Børresen-Dale, A.L.; et al. Signatures of Mutational Processes in Human Cancer. Nature 2013, 500, 415–421. [Google Scholar] [CrossRef] [Green Version]
- Sonpavde, G. PD-1 and PD-L1 Inhibitors as Salvage Therapy for Urothelial Carcinoma. N. Engl. J. Med. 2017, 376, 1073–1074. [Google Scholar] [CrossRef]
- Peranzoni, E.; Lemoine, J.; Vimeux, L.; Feuillet, V.; Barrin, S.; Kantari-Mimoun, C.; Bercovici, N.; Guérin, M.; Biton, J.; Ouakrim, H.; et al. Macrophages impede CD8 T cells from reaching tumor cells and limit the efficacy of anti-PD-1 treatment. Proc. Natl. Acad. Sci. USA 2018, 115, E4041–E4050. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- House, I.G.; Savas, P.; Lai, J.; Chen, A.X.Y.; Oliver, A.J.; Teo, Z.L.; Todd, K.L.; Henderson, M.A.; Giuffrida, L.; Petley, E.V.; et al. Macrophage-Derived CXCL9 and CXCL10 Are Required for Antitumor Immune Responses Following Immune Checkpoint Blockade. Clin. Cancer Res. 2020, 26, 487–504. [Google Scholar] [CrossRef] [Green Version]
- Guerriero, J.L.; Sotayo, A.; Ponichtera, H.E.; Castrillon, J.A.; Pourzia, A.L.; Schad, S.; Johnson, S.F.; Carrasco, R.D.; Lazo, S.; Bronson, R.T.; et al. Class IIa HDAC inhibition reduces breast tumours and metastases through anti-tumour macrophages. Nat. Cell Biol. 2017, 543, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Rodell, C.B.; Arlauckas, S.P.; Cuccarese, M.F.; Garris, C.S.; Li, R.; Ahmed, M.S.; Kohler, R.H.; Pittet, M.J.; Weissleder, R. TLR7/8-Agonist-Loaded Nanoparticles Promote the Polarization of Tumour-Associated Macrophages to Enhance Cancer Immunotherapy. Nat. Biomed. Eng. 2018, 2, 578–588. [Google Scholar] [CrossRef]
- Zhao, R.; Wan, Q.; Wang, Y.; Wu, Y.; Xiao, S.; Li, Q.; Shen, X.; Zhuang, W.; Zhou, Y.; Xia, L.; et al. M1-like TAMs are required for the efficacy of PD-L1/PD-1 blockades in gastric cancer. OncoImmunology 2021, 10, 1862520. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.A.; Patel, V.G. The role of PD-L1 expression as a predictive biomarker: An analysis of all US Food and Drug Administration (FDA) approvals of immune checkpoint inhibitors. J. Immunother. Cancer 2019, 7, 1–8. [Google Scholar] [CrossRef]
- Arwert, E.; Harney, A.S.; Entenberg, D.; Wang, Y.; Sahai, E.; Pollard, J.W.; Condeelis, J.S. A Unidirectional Transition from Migratory to Perivascular Macrophage Is Required for Tumor Cell Intravasation. Cell Rep. 2018, 23, 1239–1248. [Google Scholar] [CrossRef] [Green Version]
- Qian, B.-Z.; Li, J.; Zhang, H.; Kitamura, T.; Zhang, J.; Campion, L.R.; Kaiser, E.A.; Snyder, L.A.; Pollard, J.W. CCL2 recruits inflammatory monocytes to facilitate breast-tumour metastasis. Nature 2011, 475, 222–225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Yao, W.; Yuan, Y.; Chen, P.; Li, B.; Li, J.; Chu, R.; Song, H.; Xie, D.; Jiang, X.; et al. Targeting of tumour-infiltrating macrophages via CCL2/CCR2 signalling as a therapeutic strategy against hepatocellular carcinoma. Gut 2015, 66, 157–167. [Google Scholar] [CrossRef]
- Zhu, X.; Fujita, M.; Snyder, L.A.; Okada, H. Systemic delivery of neutralizing antibody targeting CCL2 for glioma therapy. J. Neuro-Oncol. 2011, 104, 83–92. [Google Scholar] [CrossRef] [Green Version]
- Connolly, K.A.; Belt, B.A.; Figueroa, N.M.; Murthy, A.; Patel, A.; Kim, M.; Lord, E.M.; Linehan, D.C.; Gerber, S.A. Increasing the efficacy of radiotherapy by modulating the CCR2/CCR5 chemokine axes. Oncotarget 2016, 7, 86522–86535. [Google Scholar] [CrossRef] [Green Version]
- Wu, X.; Singh, R.; Hsu, D.K.; Zhou, Y.; Yu, S.; Han, D.; Shi, Z.; Huynh, M.; Campbell, J.J.; Hwang, S.T. A Small Molecule CCR2 Antagonist Depletes Tumor Macrophages and Synergizes with Anti–PD-1 in a Murine Model of Cutaneous T-Cell Lymphoma (CTCL). J. Investig. Dermatol. 2020, 140, 1390–1400.e4. [Google Scholar] [CrossRef] [PubMed]
- Bonapace, L.; Coissieux, M.-M.; Wyckoff, J.; Mertz, K.D.; Varga, Z.; Junt, T.; Bentires-Alj, M. Cessation of CCL2 Inhibition Accelerates Breast Cancer Metastasis by Promoting Angiogenesis. Nature 2014, 515, 130–133. [Google Scholar] [CrossRef]
- Amann, B.; Perabo, F.G.; Wirger, A.; Hugenschmidt, H.; Schultze-Seemann, W. Urinary levels of monocyte chemo-attractant protein-1 correlate with tumour stage and grade in patients with bladder cancer. Br. J. Urol. 1998, 82, 118–121. [Google Scholar] [CrossRef]
- Eckstein, M.; Epple, E.; Jung, R.; Weigelt, K.; Lieb, V.; Sikic, D.; Stöhr, R.; Geppert, C.; Weyerer, V.; Bertz, S.; et al. CCL2 Expression in Tumor Cells and Tumor-Infiltrating Immune Cells Shows Divergent Prognostic Potential for Bladder Cancer Patients Depending on Lymph Node Stage. Cancers 2020, 12, 1253. [Google Scholar] [CrossRef]
- Eruslanov, E.; Neuberger, M.; Daurkin, I.; Perrin, G.Q.; Algood, C.; Dahm, P.; Rosser, C.; Vieweg, J.; Gilbert, S.M.; Kusmartsev, S. Circulating and tumor-infiltrating myeloid cell subsets in patients with bladder cancer. Int. J. Cancer 2012, 130, 1109–1119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mu, X.-Y.; Wang, R.-J.; Yao, Z.-X.; Zheng, Z.; Jiang, J.-T.; Tan, M.-Y.; Sun, F.; Fan, J.; Wang, X.; Zheng, J.-H.; et al. RS 504393 inhibits M-MDSCs recruiting in immune microenvironment of bladder cancer after gemcitabine treatment. Mol. Immunol. 2019, 109, 140–148. [Google Scholar] [CrossRef]
- Kioi, M.; Vogel, O.H.; Schultz, G.; Hoffman, R.M.; Harsh, G.R.; Brown, J.M. Inhibition of vasculogenesis, but not angiogenesis, prevents the recurrence of glioblastoma after irradiation in mice. J. Clin. Investig. 2010, 120, 694–705. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.M.; Thomas, R.; Nagpal, S.; Recht, L. Macrophage exclusion after radiation therapy (MERT): A new and effective way to increase the therapeutic ratio of radiotherapy. Radiother. Oncol. 2020, 144, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Hughes, R.; Qian, B.-Z.; Rowan, C.; Muthana, M.; Keklikoglou, I.; Olson, O.; Tazzyman, S.; Danson, S.; Addison, C.; Clemons, M.; et al. Perivascular M2 Macrophages Stimulate Tumor Relapse after Chemotherapy. Cancer Res. 2015, 75, 3479–3491. [Google Scholar] [CrossRef] [Green Version]
- Seo, Y.D.; Jiang, X.; Sullivan, K.M.; Jalikis, F.G.; Smythe, K.S.; Abbasi, A.; Vignali, M.; Park, J.O.; Daniel, S.K.; Pollack, S.M.; et al. Mobilization of CD8+ T Cells via CXCR4 Blockade Facilitates PD-1 Checkpoint Therapy in Human Pancreatic Cancer. Clin. Cancer Res. 2019, 25, 3934–3945. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, D.; Xin, M.; Wang, J.; Xu, H.; Huo, Q.; Tang, Z.; Wang, H. Chemokine receptor CXCR4 and its ligand CXCL12 expressions and clinical significance in bladder cancer. Genet. Mol. Res. 2015, 14, 17699–17707. [Google Scholar] [CrossRef] [PubMed]
- Batsi, O.; Giannopoulou, I.; Nesseris, I.; Valavanis, C.; Gakiopoulou, H.; Patsouris, E.S.; Arapandoni-Dadioti, P.; Lazaris, A.C. Immunohistochemical evaluation of CXCL12-CXCR4 axis and VEGFR3 expression in primary urothelial cancer and its recurrence. Anticancer. Res. 2014, 34, 3537–3542. [Google Scholar] [PubMed]
- Li, Y.; Chen, M.; Yuan, J.; Zhou, X.; He, W.; Zu, X.; Qi, L. CXCR4 Expression in Bladder Transitional Cell Carcinoma and Its Relationship with Clinicopathological Features. Urol. Int. 2014, 92, 157–163. [Google Scholar] [CrossRef]
- Liu, Y.; Wu, Y.; Zhang, P.; Xu, C.; Liu, Z.; He, C.; Liu, Y.; Kang, Z. CXCL12 and CD3E as Indicators for Tumor Microenvironment Modulation in Bladder Cancer and Their Correlations With Immune Infiltration and Molecular Subtypes. Front. Oncol. 2021, 11, 636870. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ou, D.-H.; Zhuang, D.-W.; Zheng, Z.-F.; Lin, M.-E. In silico analysis of the immune microenvironment in bladder cancer. BMC Cancer 2020, 20, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Che, J.; Song, R.; Chen, B.; Dong, X. Targeting CXCR1/2: The Medicinal Potential as Cancer Immunotherapy Agents, Antagonists Research Highlights and Challenges Ahead. Eur. J. Med. Chem. 2019, 6, 111853. [Google Scholar] [CrossRef]
- Wang, G.; Lu, X.; Dey, P.; Deng, P.; Wu, C.C.; Jiang, S.; Fang, Z.; Zhao, K.; Konaparthi, R.; Hua, S.; et al. Targeting YAP-Dependent MDSC Infiltration Impairs Tumor Progression. Cancer Discov. 2016, 6, 80–95. [Google Scholar] [CrossRef] [Green Version]
- Burnier, A.; Shimizu, Y.; Dai, Y.; Nakashima, M.; Matsui, Y.; Ogawa, O.; Rosser, C.J.; Furuya, H. CXCL1 is elevated in the urine of bladder cancer patients. SpringerPlus 2015, 4, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Nakashima, M.; Matsui, Y.; Kobayashi, T.; Saito, R.; Hatahira, S.; Kawakami, K.; Nakamura, E.; Nishiyama, H.; Ogawa, O. Urine CXCL1 as a biomarker for tumor detection and outcome prediction in bladder cancer. Cancer Biomarkers 2015, 15, 357–364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawanishi, H.; Matsui, Y.; Ito, M.; Watanabe, J.; Takahashi, T.; Nishizawa, K.; Nishiyama, H.; Kamoto, T.; Mikami, Y.; Tanaka, Y.; et al. Secreted CXCL1 Is a Potential Mediator and Marker of the Tumor Invasion of Bladder Cancer. Clin. Cancer Res. 2008, 14, 2579–2587. [Google Scholar] [CrossRef] [Green Version]
- Miyake, M.; Lawton, A.; Goodison, S.; Urquidi, V.; Gomes-Giacoia, E.; Zhang, G.; Ross, S.; Kim, J.; Rosser, C.J. Chemokine (C-X-C) ligand 1 (CXCL1) protein expression is increased in aggressive bladder cancers. BMC Cancer 2013, 13, 322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, Y.; Guan, Z.; Chen, J.; Xie, H.; Yang, Z.; Fan, J.; Wang, X.; Li, L. CXCL5/CXCR2 axis promotes bladder cancer cell migration and invasion by activating PI3K/AKT-induced upregulation of MMP2/MMP9. Int. J. Oncol. 2015, 47, 690–700. [Google Scholar] [CrossRef] [Green Version]
- Zhu, X.; Qiao, Y.; Liu, W.; Wang, W.; Shen, H.; Lu, Y.; Hao, G.; Zheng, J.; Tian, Y. CXCL5 is a potential diagnostic and prognostic marker for bladder cancer patients. Tumor Biol. 2016, 37, 4569–4577. [Google Scholar] [CrossRef]
- Urquidi, V.; Chang, M.; Dai, Y.; Kim, J.; Wolfson, E.D.; Goodison, S.; Rosser, C.J. IL-8 as a urinary biomarker for the detection of bladder cancer. BMC Urol. 2012, 12, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mian, B.M.; Dinney, C.P.N.; Bermejo, C.E.; Sweeney, P.; Tellez, C.; Yang, X.D.; Gudas, J.M.; McConkey, D.J.; Bar-Eli, M. Fully Human Anti-Interleukin 8 Antibody Inhibits Tumor Growth in Orthotopic Bladder Cancer Xenografts via down-Regulation of Matrix Metalloproteases and Nuclear Factor-ΚB. Clin. Cancer Res. 2003, 9, 3167–3175. [Google Scholar] [PubMed]
- Strachan, D.C.; Ruffell, B.; Oei, Y.; Bissell, M.J.; Coussens, L.M.; Pryer, N.; Daniel, D. CSF1R inhibition delays cervical and mammary tumor growth in murine models by attenuating the turnover of tumor-associated macrophages and enhancing infiltration by CD8+T cells. OncoImmunology 2013, 2, e26968. [Google Scholar] [CrossRef] [Green Version]
- Ries, C.H.; Cannarile, M.A.; Hoves, S.; Benz, J.; Wartha, K.; Runza, V.; Rey-Giraud, F.; Pradel, L.P.; Feuerhake, F.; Klaman, I.; et al. Targeting Tumor-Associated Macrophages with Anti-CSF-1R Antibody Reveals a Strategy for Cancer Therapy. Cancer Cell 2014, 25, 846–859. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Candido, J.B.; Morton, J.; Bailey, P.; Campbell, A.D.; Karim, S.A.; Jamieson, T.; Lapienyte, L.; Gopinathan, A.; Clark, W.; McGhee, E.J.; et al. CSF1R+ Macrophages Sustain Pancreatic Tumor Growth through T Cell Suppression and Maintenance of Key Gene Programs that Define the Squamous Subtype. Cell Rep. 2018, 23, 1448–1460. [Google Scholar] [CrossRef] [Green Version]
- Neubert, N.J.; Schmittnaegel, M.; Bordry, N.; Nassiri, S.; Wald, N.; Martignier, C.; Tillé, L.; Homicsko, K.; Damsky, W.; Hajjami, H.M.-E.; et al. T cell–induced CSF1 promotes melanoma resistance to PD1 blockade. Sci. Transl. Med. 2018, 10, eaan3311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Champelovier, P.; Boucard, N.; Levacher, G.; Simon, A.; Seigneurin, D.; Praloran, V. Plasminogen- and colony-stimulating factor-1-associated markers in bladder carcinoma: Diagnostic value of urokinase plasminogen activator receptor and plasminogen activator inhibitor type-2 using immunocytochemical analysis. Urol. Res. 2002, 30, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Lee, M.M.; De Silva, D.M.; Roy, A.; Wright, C.E.; Wong, T.K.; Costello, R.; Olaku, O.; Grubb, R.L.; Agarwal, P.K.; et al. Autocrine signaling by receptor tyrosine kinases in urothelial carcinoma of the bladder. PLoS ONE 2021, 16, 0241766. [Google Scholar] [CrossRef]
- Morizawa, Y.; Miyake, M.; Shimada, K.; Hori, S.; Tatsumi, Y.; Nakai, Y.; Tanaka, N.; Fujii, T.; Fujimoto, K. Colony-stimulating factors detected in tumor cells and voided urine are potential prognostic markers for patients with muscle-invasive bladder cancer undergoing radical cystectomy. Res. Rep. Urol. 2018, 10, 103–111. [Google Scholar] [CrossRef] [Green Version]
- Germano, G.; Frapolli, R.; Belgiovine, C.; Anselmo, A.; Pesce, S.; Liguori, M.; Erba, E.; Uboldi, S.; Zucchetti, M.; Pasqualini, F.; et al. Role of Macrophage Targeting in the Antitumor Activity of Trabectedin. Cancer Cell 2013, 23, 249–262. [Google Scholar] [CrossRef] [Green Version]
- Banerjee, P.; Zhang, R.; Ivan, C.; Galletti, G.; Clise-Dwyer, K.; Barbaglio, F.; Scarfo’, L.; Aracil, M.; Klein, C.; Wierda, W.; et al. Trabectedin Reveals a Strategy of Immunomodulation in Chronic Lymphocytic Leukemia. Cancer Immunol. Res. 2019, 7, 2036–2051. [Google Scholar] [CrossRef] [PubMed]
- Carminati, L.; Pinessi, D.; Borsotti, P.; Minoli, L.; Giavazzi, R.; D’Incalci, M.; Belotti, D.; Taraboletti, G. Antimetastatic and antiangiogenic activity of trabectedin in cutaneous melanoma. Carcinogenesis 2019, 40, 303–312. [Google Scholar] [CrossRef]
- Jones, J.; Sinder, B.; Paige, D.; Soki, F.; Koh, A.; Thiele, S.; Shiozawa, Y.; Hofbauer, L.; Daignault, S.; Roca, H.; et al. Trabectedin Reduces Skeletal Prostate Cancer Tumor Size in Association with Effects on M2 Macrophages and Efferocytosis. Neoplasia 2019, 21, 172–184. [Google Scholar] [CrossRef]
- Casagrande, N.; Borghese, C.; Favero, A.; Vicenzetto, C.; Aldinucci, D. Trabectedin overcomes doxorubicin-resistance, counteracts tumor-immunosuppressive reprogramming of monocytes and decreases xenograft growth in Hodgkin lymphoma. Cancer Lett. 2021, 500, 182–193. [Google Scholar] [CrossRef]
- Rogers, T.L.; Holen, I. Tumour macrophages as potential targets of bisphosphonates. J. Transl. Med. 2011, 9, 177. [Google Scholar] [CrossRef] [Green Version]
- Hiraoka, K.; Zenmyo, M.; Watari, K.; Iguchi, H.; Fotovati, A.; Kimura, Y.N.; Hosoi, F.; Shoda, T.; Nagata, K.; Osada, H.; et al. Inhibition of bone and muscle metastases of lung cancer cells by a decrease in the number of monocytes/macrophages. Cancer Sci. 2008, 99, 1595–1602. [Google Scholar] [CrossRef] [PubMed]
- Fritz, J.; Tennis, M.; Orlicky, D.J.; Lin, H.; Eju, C.; Redente, E.F.; Choo, K.S.; Staab, T.A.; Bouchard, R.J.; Merrick, D.T.; et al. Depletion of Tumor-Associated Macrophages Slows the Growth of Chemically Induced Mouse Lung Adenocarcinomas. Front. Immunol. 2014, 5, 587. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, W.; Zhu, X.-D.; Sun, H.-C.; Xiong, Y.-Q.; Zhuang, P.-Y.; Xu, H.-X.; Kong, L.-Q.; Wang, L.; Wu, W.-Z.; Tang, Z.-Y. Depletion of Tumor-Associated Macrophages Enhances the Effect of Sorafenib in Metastatic Liver Cancer Models by Antimetastatic and Antiangiogenic Effects. Clin. Cancer Res. 2010, 16, 3420–3430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeisberger, S.M.; Odermatt, B.; Marty, C.; Zehnder-Fjällman, A.H.M.; Ballmer-Hofer, K.; A Schwendener, R. Clodronate-liposome-mediated depletion of tumour-associated macrophages: A new and highly effective antiangiogenic therapy approach. Br. J. Cancer 2006, 95, 272–281. [Google Scholar] [CrossRef]
- Klug, F.; Prakash, H.; Huber, P.E.; Seibel, T.; Bender, N.; Halama, N.; Pfirschke, C.; Voss, R.H.; Timke, C.; Umansky, L.; et al. Low-Dose Irradiation Programs Macrophage Differentiation to an iNOS+/M1 Phenotype that Orchestrates Effective T Cell Immunotherapy. Cancer Cell 2013, 24, 589–602. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pyonteck, S.M.; Akkari, L.; Schuhmacher, A.J.; Bowman, R.L.; Sevenich, L.; Quail, D.F.; Olson, O.C.; Quick, M.L.; Huse, J.T.; Teijeiro, V.; et al. CSF-1R inhibition alters macrophage polarization and blocks glioma progression. Nat. Med. 2013, 19, 1264–1272. [Google Scholar] [CrossRef] [Green Version]
- Akkari, L.; Bowman, R.L.; Tessier, J.; Klemm, F.; Handgraaf, S.M.; de Groot, M.; Quail, D.F.; Tillard, L.; Gadiot, J.; Huse, J.T.; et al. Dynamic changes in glioma macrophage populations after radiotherapy reveal CSF-1R inhibition as a strategy to overcome resistance. Sci. Transl. Med. 2020, 12, eaaw7843. [Google Scholar] [CrossRef]
- Zhu, Y.; Knolhoff, B.L.; Meyer, M.; Nywening, T.M.; West, B.; Luo, J.; Wang-Gillam, A.; Goedegebuure, S.P.; Linehan, D.C.; DeNardo, D.G. CSF1/CSF1R Blockade Reprograms Tumor-Infiltrating Macrophages and Improves Response to T-cell Checkpoint Immunotherapy in Pancreatic Cancer Models. Cancer Res. 2014, 74, 5057–5069. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Mitri, D.; Mirenda, M.; Vasilevska, J.; Calcinotto, A.; Delaleu, N.; Revandkar, A.; Gil, V.; Boysen, G.; Losa, M.; Mosole, S.; et al. Re-Education of Tumor-Associated Macrophages by CXCR2 Blockade Drives Senescence and Tumor Inhibition in Advanced Prostate Cancer. Cell Rep. 2019, 28, 2156–2168.e5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pahlavanneshan, S.; Sayadmanesh, A.; Ebrahimiyan, H.; Basiri, M. Toll-Like Receptor-Based Strategies for Cancer Immunotherapy. J. Immunol. Res. 2021, 2021, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Akira, S.; Takeda, K. Toll-like Receptor Signalling. Nat. Rev. Immunol. 2004, 4, 499–511. [Google Scholar] [CrossRef]
- Huang, Z.; Yang, Y.; Jiang, Y.; Shao, J.; Sun, X.; Chen, J.; Dong, L.; Zhang, J. Anti-tumor immune responses of tumor-associated macrophages via toll-like receptor 4 triggered by cationic polymers. Biomaterial 2013, 34, 746–755. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.; Tufa, D.M.; Chatterjee, D.; Mühlradt, P.F.; Schmidt, R.E.; Jacobs, R. The TLR-2/TLR-6 agonist macrophage-activating lipopeptide-2 augments human NK cell cytotoxicity when PGE2 production by monocytes is inhibited by a COX-2 blocker. Cancer Immunol. Immunother. 2015, 64, 1175–1184. [Google Scholar] [CrossRef] [PubMed]
- Vidyarthi, A.; Khan, N.; Agnihotri, T.; Negi, S.; Das, D.K.; Aqdas, M.; Chatterjee, D.; Colegio, O.; Tewari, M.K.; Agrewala, J.N. TLR-3 Stimulation Skews M2 Macrophages to M1 Through IFN-αβ Signaling and Restricts Tumor Progression. Front. Immunol. 2018, 9, 9. [Google Scholar] [CrossRef]
- Zhao, J.; Zhang, Z.; Xue, Y.; Wang, G.; Cheng, Y.; Pan, Y.; Zhao, S.; Hou, Y. Anti-tumor macrophages activated by ferumoxytol combined or surface-functionalized with the TLR3 agonist poly(I:C) promote melanoma regression. Theranostics 2018, 8, 6307–6321. [Google Scholar] [CrossRef]
- Maeda, A.; Digifico, E.; Andón, F.T.; Mantovani, A.; Allavena, P. Poly(I:C) stimulation is superior than Imiquimod to induce the antitumoral functional profile of tumor-conditioned macrophages. Eur. J. Immunol. 2019, 49, 801–811. [Google Scholar] [CrossRef]
- Liu, Z.; Xie, Y.; Xiong, Y.; Liu, S.; Qiu, C.; Zhu, Z.; Mao, H.; Yu, M.; Wang, X. TLR 7/8 Agonist Reverses Oxaliplatin Resistance in Colorectal Cancer via Directing the Myeloid-Derived Suppressor Cells to Tumoricidal M1-Macrophages. Cancer Lett. 2020, 469, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.B.; Schwartz, M.; Kawamoto, H.; You, X.; Hwang, D.; Liu, H.; Scherr, D.S. Antitumor Effects of Imidazoquinolines in Urothelial Cell Carcinoma of the Bladder. J. Urol. 2007, 177, 2347–2351. [Google Scholar] [CrossRef]
- Hayashi, T.; Crain, B.; Corr, M.; Chan, M.; Cottam, H.B.; Maj, R.; Barberis, A.; Leoni, L.; Carson, D.A. Intravesical Toll-like receptor 7 agonist R-837: Optimization of its formulation in an orthotopic mouse model of bladder cancer. Int. J. Urol. 2010, 17, 483–490. [Google Scholar] [CrossRef] [PubMed]
- De Camargo, J.A.; Passos, G.R.; Ferrari, K.L.; Billis, A.; Saad, M.J.; Reis, L.O. Intravesical Immunomodulatory Imiquimod Enhances Bacillus Calmette-Guérin Downregulation of Nonmuscle-invasive Bladder Cancer. Clin. Genitourin. Cancer 2018, 16, e587–e593. [Google Scholar] [CrossRef]
- Donin, N.M.; Chamie, K.; Lenis, A.T.; Pantuck, A.J.; Reddy, M.; Kivlin, D.; Holldack, J.; Pozzi, R.; Hakim, G.; Karsh, L.I.; et al. A phase 2 study of TMX-101, intravesical imiquimod, for the treatment of carcinoma in situ bladder cancer. Urol. Oncol. Semin. Orig. Investig. 2017, 35, 39.e1–39.e7. [Google Scholar] [CrossRef]
- Ayari, C.; Besançon, M.; Bergeron, A.; LaRue, H.; Bussières, V.; Fradet, Y. Poly(I:C) Potentiates Bacillus Calmette–Guérin Immunotherapy for Bladder Cancer. Cancer Immunol. Immunother. 2016, 65, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Plote, D.; Choi, W.; Mokkapati, S.; Sundi, D.; Ferguson, J.E.; Duplisea, J.; Parker, N.R.; Yla-Herttuala, S.; SUO CTC Bladder Committee; McConkey, D.; et al. Inhibition of urothelial carcinoma through targeted type I interferon-mediated immune activation. OncoImmunology 2019, 8, e1577125. [Google Scholar] [CrossRef] [PubMed]
- Karagiannis, D.; Rampias, T. HDAC Inhibitors: Dissecting Mechanisms of Action to Counter Tumor Heterogeneity. Cancers 2021, 13, 3575. [Google Scholar] [CrossRef] [PubMed]
- Lobera, M.; Madauss, K.P.; Pohlhaus, D.T.; Wright, Q.; Trocha, M.; Schmidt, D.R.; Baloglu, E.; Trump, R.P.; Head, M.S.; A Hofmann, G.; et al. Selective class IIa histone deacetylase inhibition via a nonchelating zinc-binding group. Nat. Chem. Biol. 2013, 9, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-D.; Park, S.-M.; Ha, H.C.; Lee, A.R.; Won, H.; Cha, H.; Cho, S.; Cho, J.M. HDAC Inhibitor, CG-745, Enhances the Anti-Cancer Effect of Anti-PD-1 Immune Checkpoint Inhibitor by Modulation of the Immune Microenvironment. J. Cancer 2020, 11, 4059–4072. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Su, X.; Liu, R.; Pan, Y.; Fang, J.; Cao, L.; Feng, C.; Shang, Q.; Chen, Y.; Shao, C.; et al. HDAC inhibition potentiates anti-tumor activity of macrophages and enhances anti-PD-L1-mediated tumor suppression. Oncogene 2021, 40, 1836–1850. [Google Scholar] [CrossRef]
- Lu, Z.; Zou, J.; Li, S.; Topper, M.J.; Tao, Y.; Zhang, H.; Jiao, X.; Xie, W.; Kong, X.; Vaz, M.; et al. Epigenetic therapy inhibits metastases by disrupting premetastatic niches. Nature 2020, 579, 284–290. [Google Scholar] [CrossRef]
- Briere, D.; Sudhakar, N.; Woods, D.M.; Hallin, J.; Engstrom, L.D.; Aranda, R.; Chiang, H.; Sodré, A.L.; Olson, P.; Weber, J.S.; et al. The class I/IV HDAC inhibitor mocetinostat increases tumor antigen presentation, decreases immune suppressive cell types and augments checkpoint inhibitor therapy. Cancer Immunol. Immunother. 2018, 67, 381–392. [Google Scholar] [CrossRef]
- Toriyama, S.; Horinaka, M.; Yasuda, S.; Taniguchi, T.; Aono, Y.; Takamura, T.; Morioka, Y.; Miki, T.; Ukimura, O.; Sakai, T. A Histone Deacetylase Inhibitor, OBP-801, and Celecoxib Synergistically Inhibit the Cell Growth with Apoptosis via a DR5-Dependent Pathway in Bladder Cancer Cells. Mol. Cancer Ther. 2016, 15, 2066–2075. [Google Scholar] [CrossRef] [Green Version]
- Kaletsch, A.; Pinkerneil, M.; Hoffmann, M.J.; Vasudevan, A.A.J.; Wang, C.; Hansen, F.K.; Wiek, C.; Hanenberg, H.; Gertzen, C.; Gohlke, H.; et al. Effects of novel HDAC inhibitors on urothelial carcinoma cells. Clin. Epigenetics 2018, 10, 100. [Google Scholar] [CrossRef] [Green Version]
- Hsu, F.-S.; Wu, J.-T.; Lin, J.-Y.; Yang, S.-P.; Kuo, K.-L.; Lin, W.-C.; Shi, C.-S.; Chow, P.-M.; Liao, S.-M.; Pan, C.-I.; et al. Histone Deacetylase Inhibitor, Trichostatin A, Synergistically Enhances Paclitaxel-Induced Cytotoxicity in Urothelial Carcinoma Cells by Suppressing the ERK Pathway. Int. J. Mol. Sci. 2019, 20, 1162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuroki, H.; Anraku, T.; Kazama, A.; Shirono, Y.; Bilim, V.; Tomita, Y. Histone deacetylase 6 inhibition in urothelial cancer as a potential new strategy for cancer treatment. Oncol. Lett. 2020, 21, 1. [Google Scholar] [CrossRef]
- Burke, B.; Eden, C.; Perez, C.; Belshoff, A.; Hart, S.; Plaza-Rojas, L.; Delos Reyes, M.; Prajapati, K.; Voelkel-Johnson, C.; Henry, E.; et al. Inhibition of Histone Deacetylase (HDAC) Enhances Checkpoint Blockade Efficacy by Rendering Blad-der Cancer Cells Visible for T Cell-Mediated Destruction. Front. Oncol. 2020, 10, 1–13. [Google Scholar] [CrossRef]
- Grivas, P.; Mortazavi, A.; Picus, J.; Hahn, N.M.; Milowsky, M.I.; Hart, L.L.; Alva, A.; Bellmunt, J.; Pal, S.K.; Bambury, R.M.; et al. Mocetinostat for Patients with Previously Treated, Locally Advanced/Metastatic Urothelial Carcinoma and Inac-tivating Alterations of Acetyltransferase Genes. Cancer 2019, 125, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Quinn, D.I.; Tsao-Wei, D.D.; Twardowski, P.; Aparicio, A.M.; Frankel, P.; Chatta, G.; Wright, J.J.; Groshen, S.G.; Khoo, S.; Lenz, H.J.; et al. Phase II Study of the Histone Deacetylase Inhibitor Vorinostat (Suberoylanilide Hydroxamic Acid; SA-HA) in Recurrent or Metastatic Transitional Cell Carcinoma of the Urothelium—An NCI-CTEP Sponsored: California Cancer Consortium Trial, NCI 6879. Invest. New Drugs 2021, 39, 812–820. [Google Scholar] [CrossRef]
- Sotsios, Y.; Ward, S.G. Phosphoinositide 3-kinase: A key biochemical signal for cell migration in response to chemokines. Immunol. Rev. 2000, 177, 217–235. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, E.; Katanaev, V.L.; Garlanda, C.; Azzolino, O.; Pirola, L.; Silengo, L.; Sozzani, S.; Mantovani, A.; Altruda, F.; Wymann, M.P. Central Role for G Protein-Coupled Phosphoinositide 3-Kinase γ in Inflammation. Science 2000, 287, 1049–1052. [Google Scholar] [CrossRef] [PubMed]
- Kaneda, M.M.; Messer, K.S.; Ralainirina, N.; Li, H.; Leem, C.J.; Gorjestani, S.; Woo, G.; Nguyen, A.V.; Figueiredo, C.C.; Foubert, P.; et al. PI3Kγ is a molecular switch that controls immune suppression. Nature 2016, 539, 437–442. [Google Scholar] [CrossRef] [Green Version]
- Kaneda, M.M.; Cappello, P.; Nguyen, A.V.; Ralainirina, N.; Hardamon, C.R.; Foubert, P.; Schmid, M.C.; Sun, P.; Mose, E.; Bouvet, M.; et al. Macrophage PI3Kγ Drives Pancreatic Ductal Adenocarcinoma Progression. Cancer Discov. 2016, 6, 870–885. [Google Scholar] [CrossRef] [Green Version]
- Foubert, P.; Kaneda, M.M.; Varner, J.A. PI3Kγ Activates Integrin α4 and Promotes Immune Suppressive Myeloid Cell Polarization during Tumor Progression. Cancer Immunol. Res. 2017, 5, 957–968. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, K.; Quispe, C.; Javed, Z.; Iqbal, M.J.; Sadia, H.; Raza, S.; Irshad, A.; Salehi, B.; Reiner, Ž.; Sharifi-Rad, J. Resve-ratrol, Curcumin, Paclitaxel and MiRNAs Mediated Regulation of PI3K/Akt/MTOR Pathway: Go Four Better to Treat Blad-der Cancer. Cancer Cell Int. 2020, 20, 1–19. [Google Scholar] [CrossRef]
- Van Kooten, C.; Banchereau, J. CD40-CD40 Ligand. J. Leukoc. Biol. 2000, 67, 2–17. [Google Scholar] [CrossRef] [PubMed]
- Beatty, G.; Chiorean, E.G.; Fishman, M.P.; Saboury, B.; Teitelbaum, U.R.; Sun, W.; Huhn, R.D.; Song, W.; Li, D.; Sharp, L.L.; et al. CD40 Agonists Alter Tumor Stroma and Show Efficacy Against Pancreatic Carcinoma in Mice and Humans. Science 2011, 331, 1612–1616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wiehagen, K.R.; Girgis, N.M.; Yamada, D.H.; Smith, A.A.; Chan, S.R.; Grewal, I.S.; Quigley, M.; Verona, R.I. Combi-nation of CD40 Agonism and CSF-1R Blockade Reconditions Tumor-Associated Macrophages and Drives Potent Antitumor Immunity. Cancer Immunol. Res. 2017, 5, 1109–1121. [Google Scholar] [CrossRef] [Green Version]
- Hoves, S.; Ooi, C.-H.; Wolter, C.; Sade, H.; Bissinger, S.; Schmittnaegel, M.; Ast, O.; Giusti, A.M.; Wartha, K.; Runza, V.; et al. Rapid activation of tumor-associated macrophages boosts preexisting tumor immunity. J. Exp. Med. 2018, 215, 859–876. [Google Scholar] [CrossRef] [PubMed]
- Kashyap, A.S.; Schmittnaegel, M.; Rigamonti, N.; Pais-Ferreira, D.; Mueller, P.; Buchi, M.; Ooi, C.-H.; Kreuzaler, M.; Hirschmann, P.; Guichard, A.; et al. Optimized Antiangiogenic Reprogramming of the Tumor Microenvironment Poten-tiates CD40 Immunotherapy. Proc. Natl. Acad. Sci. USA 2020, 117, 541–551. [Google Scholar] [CrossRef] [Green Version]
- Long, K.B.; Gladney, W.L.; Tooker, G.M.; Graham, K.; Fraietta, J.A.; Beatty, G.L. IFNγ and CCL2 Cooperate to Redi-rect Tumor-Infiltrating Monocytes to Degrade Fibrosis and Enhance Chemotherapy Efficacy in Pancreatic Carcinoma. Cancer Discov. 2016, 6, 400–413. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.Q.; Zeng, S.; Vitiello, G.A.; Seifert, A.M.; Medina, B.D.; Beckman, M.J.; Loo, J.; Santamaria-Barria, J.; Malt-baek, J.H.; Param, N.J.; et al. Macrophages and CD8+ T Cells Mediate the Anti-Tumor Efficacy of Combined CD40 Ligation and Imatinib Therapy in Gastrointestinal Stromal Tumors. Cancer Immunol. Res. 2018, 6, 434–447. [Google Scholar] [CrossRef] [Green Version]
- Winograd, R.; Byrne, K.; Evans, R.A.; Odorizzi, P.M.; Meyer, A.R.L.; Bajor, D.L.; Clendenin, C.; Stanger, B.Z.; Furth, E.E.; Wherry, E.J.; et al. Induction of T-cell Immunity Overcomes Complete Resistance to PD-1 and CTLA-4 Blockade and Improves Survival in Pancreatic Carcinoma. Cancer Immunol. Res. 2015, 3, 399–411. [Google Scholar] [CrossRef] [Green Version]
- Zippelius, A.; Schreiner, J.; Herzig, P.; Muller, P. Induced PD-L1 Expression Mediates Acquired Resistance to Agonis-tic Anti-CD40 Treatment. Cancer Immunol. Res. 2015, 3, 236–244. [Google Scholar] [CrossRef] [Green Version]
- Garris, C.S.; Wong, J.L.; Ravetch, J.V.; Knorr, D.A. Dendritic Cell Targeting with Fc-Enhanced CD40 Antibody Ago-nists Induces Durable Antitumor Immunity in Humanized Mouse Models of Bladder Cancer. Sci. Transl. Med. 2021, 13, eabd1346. [Google Scholar] [CrossRef] [PubMed]
- Mangsbo, S.M.; Broos, S.; Fletcher, E.; Veitonmäki, N.; Furebring, C.; Dahlén, E.; Norlén, P.; Lindstedt, M.; Tötter-man, T.H.; Ellmark, P. The Human Agonistic CD40 Antibody ADC-1013 Eradicates Bladder Tumors and Generates T-Cell-Dependent Tumor Immunity. Clin. Cancer Res. 2015, 21, 1115–1126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandin, L.C.; Orlova, A.; Gustafsson, E.; Ellmark, P.; Tolmachev, V.; Tötterman, T.H.; Mangsbo, S.M. Locally Deli-vered CD40 Agonist Antibody Accumulates in Secondary Lymphoid Organs and Eradicates Experimental Disseminated Bladder Cancer. Cancer Immunol. Res. 2014, 2, 80–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murata, Y.; Kotani, T.; Ohnishi, H.; Matozaki, T. The CD47-SIRPα Signalling System: Its Physiological Roles and Therapeutic Application. J. Biochem. 2014, 155, 335–344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Edris, B.; Weiskopf, K.; Volkmer, A.K.; Volkmer, J.P.; Willingham, S.B.; Contreras-Trujillo, H.; Liu, J.; Majeti, R.; West, R.B.; Fletcher, J.A.; et al. Antibody Therapy Targeting the CD47 Protein Is Effective in a Model of Aggressive Metasta-tic Leiomyosarcoma. Proc. Natl. Acad. Sci. USA 2012, 109, 6656–6661. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weiskopf, K.; Jahchan, N.S.; Schnorr, P.; Cristea, S.; Ring, A.; Maute, R.L.; Volkmer, A.K.; Volkmer, J.-P.; Liu, J.; Lim, J.S.; et al. CD47-blocking immunotherapies stimulate macrophage-mediated destruction of small-cell lung cancer. J. Clin. Investig. 2016, 126, 2610–2620. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Wei, H.; Gao, P.; Yu, H.; Wang, K.; Fu, Z.; Ju, B.; Zhao, M.; Dong, S.; Li, Z.; et al. CD47 promotes ovarian cancer progression by inhibiting macrophage phagocytosis. Oncotarget 2017, 8, 39021–39032. [Google Scholar] [CrossRef] [Green Version]
- Gu, S.; Ni, T.; Wang, J.; Liu, Y.; Fan, Q.; Wang, Y.; Huang, T.; Chu, Y.; Sun, X.; Wang, Y. CD47 Blockade Inhibits Tumor Progression through Promoting Phagocytosis of Tumor Cells by M2 Polarized Macrophages in Endometrial Cancer. J. Immunol. Res. 2018, 2018, 6156757. [Google Scholar] [CrossRef]
- Willingham, S.B.; Volkmer, J.-P.; Gentles, A.J.; Sahoo, D.; Dalerba, P.; Mitra, S.S.; Wang, J.; Contreras-Trujillo, H.; Martin, R.; Cohen, J.D.; et al. The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumors. Proc. Natl. Acad. Sci. USA 2012, 109, 6662–6667. [Google Scholar] [CrossRef] [Green Version]
- Chan, K.S.; Espinosa, I.; Chao, M.; Wong, D.; Ailles, L.; Diehn, M.; Gill, H.; Presti, J.; Chang, H.Y.; Van De Rijn, M.; et al. Identification, Molecular Characterization, Clinical Prognosis, and Therapeutic Targeting of Human Bladder Tu-mor-Initiating Cells. Proc. Natl. Acad. Sci. USA 2009, 106, 14016–14021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kiss, B.; Berg, N.S.V.D.; Ertsey, R.; McKenna, K.; Mach, K.E.; Zhang, C.A.; Volkmer, J.-P.; Weissman, I.L.; Rosenthal, E.L.; Liao, J.C. CD47-Targeted Near-Infrared Photoimmunotherapy for Human Bladder Cancer. Clin. Cancer Res. 2019, 25, 3561–3571. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Cell Types and Markers | Bladder Cancer Cohorts | Types of Sample | Findings | References |
|---|---|---|---|---|
| CD68+ (TAM) | 40 pTa-pT1 23 ≥ pT2 | FFPE tissue | High TAM count was associated with poor 5-year survival | Hanada et al. [29] |
| CD68+ (TAM) | 81 pTa-pT1 11 pT2 | FFPE tissue | High number of TAMs was significantly associated with risk of progression | Bostrom et al. [31] |
| CD68+ (TAM) | 112 pTa 89 pT1 93 MIBC | FFPE tissue | High CD68/CD3 ratio was associated with poor OS | Sjödahl et al. [45] |
| CD68+ (TAM) | 10 low grades 34 high grades | FFPE tissue | High TAM count was associated with poor DFS but not OS | Chai et al. [50] |
| CD163+ (TAM) | 115 MIBC | FFPE tissue | High TAM count was associated with poor PFS and OS | Xu et al. [35] |
| CD163+ (TAM) | 94 high grade pT1 | FFPE tissue | TAMs were associated with tumor recurrence and progression | Yang et al. [43] |
| CD204+round cells (TAM) | 155 NMIBC | FFPE tissue | High number of TAMs correlated with a high risk of recurrence | Miyake et al. [42] |
| TAM | MIBC (no metastatic disease) | TCGA database | A signature with low T cells, low NK cells, high Treg and high TAMs is associated with poor DFS and OS | Fu et al. [24] |
| TAM | 406 MIBC | TCGA database | Patients in the high-risk group had a signature with low CD8+ T cells, CD4+ T cells and high abundance of M0 macrophages | Li et al. [44] |
| CD204+CD68+ (CD204+ macrophages) | 212 pTa-pT1 90 pT2-pT4 | FFPE tissue | High number of CD204+ macrophages in tumor stroma was associated with poor OS | Wang et al. [25] |
| DC-SIGN+CD68+ (DC-SIGN+TAM) | 257 MIBC | FFPE tissue | DC-SIGN+ TAMs may contribute to progression and poor prognosis | Hu et al. [46] |
| M2-like TAM | 429 MIBC | TCGA database | High M2-like TAM signature was associated with poor OS and DFS | Xue et al. [38] |
| M2-like TAM | 402 MIBC | TCGA database | M2-like TAM signature was associated with significantly worse 5-year OS and DFS outcomes | Jiang et al. [47] |
| CD169+CD68+ (CD169+macrophages) | 44 MIBC | FFPE tissue | CD169+ macrophages in tumor-draining lymph nodes were positively correlated with a favorable prognosis | Asano et al. [48] |
| CD33+ (MDSC) | 70 NMIBC 27 MIBC | FFPE tissue | The number of tumor-infiltrating CD33+ MDSCs was significantly inversely correlated with patient OS | Zhang et al. [27] |
| CD11b+CD33lowHLA-DR− (MDSC) | 71 pTa-pT1 42 pT2-pT4 | PBMC | High numbers of circulating CD11b+ CD33lowHLA-DR− cells were correlated with poor OS | Yang et al. [32] |
| Cell Types and Markers. | Bladder Cancer Cohorts | Treatments | Findings | References |
|---|---|---|---|---|
| CD68+ (TAM) | 40 pTa-pT1 23 ≥ pT2 | TURBT or RC | Patients with a high TAM count showed higher rates of cystectomy than those with a low TAM count | Hanada et al. [29] |
| CD68+ (TAM) and MAC387+ CD68+ (MAC387+ TAM) | 81 pTa-pT1 11 pT2 | TURBT or RC |
| Bostrom et al. [31] |
| CD68+ (macrophage) and CD204+ CD68+ (CD204+macrophages) | 212 pTa-pT1 90 pT2-pT4 | TURBT or RC | Total macrophages and CD204+ macrophages in the stroma were associated with poor OS after surgery | Wang et al. [25] |
| CD68+ (TAM) and HIF-2α+ CD68+ (HIF-2α+ TAM) | 22 pT1 20 pT2 23 pT3 4 pT4 | RC |
| Koga et al. [54] |
| CD33+ HLA-DR− (MDSC) | 65 pTa-pT1 44 ≥ pT2 | RC | The percentage of total MDSC in PBMC before RC was significantly lower in patients who experienced pathological complete response | Fallah et al. [77] |
| CD68+ (TAM) | 3 pTa 9 pT1 18 pTis | BCG | Higher CD68+ cells in tumor after BCG are correlated with better RFS | Kitamura et al. [78] |
| CD68+ (TAM) | 53 NMIBC | BCG | High TAM is associated with poor RFS in high-risk NMIBC after BCG | Ayari et al. [30] |
| CD68+ (TAM) | 41 CIS | BCG | Low TAM count is associated with good RFS after BCG | Takayama et al. [79] |
| CD68+ (TAM) | 12 pTa 15 pT1 | BCG | High TAM count is associated with shorter RFS after BCG treatment | Aliji et al. [80] |
| CD68+ (TAM) | 304 NMIBC | BCG | Pre-BCG treatment TAMs are associated with worse RFS in patients with NMIBC | Kardoust et al. [81] |
| CD204+ (TAM) | 68 pTa 73 pT1 13 pTis | BCG | High counts of TAM showed association with short PFS after BCG | Miyake et al. [34] |
| CD163+ CD68+ (CD163+ macrophages) | 40 pTa 59 pT1 | BCG | High density of CD163+ macrophage counts in the stroma but not in the tumor was related with BCG failures | Lima et al. [26] |
| iNOS+ CD68+ (iNOS+ TAM) and CD163+ CD68+ (CD163+ TAM) | 40 NMIBC | BCG |
| Suriano et al. [82] |
| CD68+ (TAM) and CD163+ CD68+ (CD163+ TAM) | 9 pTa 21 pT1 10 pTis | BCG |
| Pichler et al. [83] |
| Lin−CD14+ CD33+ HLA-DR− (M-MDSC) | 4 pTa 20 pT1 3 pTis 1 pT2 | BCG | Low T cell/M-MDSC ratio after BCG treatment correlates with poor RFS & PFS | Chevalier et al. [41] |
| CD68+ (TAM) | 49 pT2 69 ≥ pT3 | platinum-based chemotherapy | An immunotype containing low T cells, low NK cells, high Treg and high TAM is associated with increase OS and DFS after chemotherapy in pT3-T4 patients | Fu et al. [24] |
| DC-SIGN+ CD68+ (DC-SIGN+ TAM) | 137 pT2 | cisplatin-based chemotherapy | High DC-SIGN+ TAM infiltration was strongly associated with unresponsiveness to adjuvant chemotherapy in MIBC | Hu et al. [46] |
| Galectin-9+ CD68+ (Gal9+ TAM) | 141 ≥ vpT2 | platinum-based chemotherapy | Survival benefits after postoperative adjuvant chemotherapy among patients with high Gal9+ TAM, whereas patients with low Gal9+ TAM showed no benefit to chemotherapy | Qi et al. [73] |
| CD68+ (TAM) and CD163+CD68+ (CD163+TAM) | 44 pT2 85 pT3 39 pT4 | Adjuvant chemotherapy |
| Taubert et al. [84] |
| CD33+ HLA-DR− (MDSC) | 49 < pT2 36 ≥ pT2 | Neoadjuvant chemotherapy | Circulating MDSCs were negatively associated with pathologic complete response in patients treated with neoadjuvant therapy | Ornstein et al. [85] |
| PD-L1+ tumor-infiltrating immune cells | Metastatic urothelial carcinoma (IMvigor210) | Atezolizumab | PD-L1 expression on immune cells was significantly associated with response to Atezolizumab | Mariathasan et al. [86] |
| PD-L1+ tumor-infiltrating immune cells | Metastatic urothelial bladder cancer (IMvigor211) | Atezolizumab | Tumors expressing PD-L1+ tumor-infiltrating immune cells had particularly high response rates | Powles et al. [87] |
| PD-L1+ tumor-infiltrating immune cells | Metastatic urothelial carcinoma | Atezolizumab | Higher levels of PD-L1 immunohistochemistry expression on immune cells were associated with a higher response rate to Atezolizumab and longer OS | Rosenberg et al. [88] |
| PD-L1+ tumor-infiltrating immune cells | Metastatic urothelial bladder cancer (IMvigor211) | Atezolizumab | Overexpression of PD-L1 resulted in a more favorable outcome with both chemotherapy and Atezolizumab | Powles et al. [89] |
| PD-L1+ tumor-infiltrating immune cells | Advanced urothelial cancer (KEYNOTE-045) | Pembrolizumab | The benefit of Pembrolizumab appeared to be independent of PD-L1 expression on infiltrating immune cells | Bellmunt et al. [90] |
| PD-L1+ cells | Unresectable locally advanced or metastatic urothelial carcinoma (CheckMate 032) | Nivolumab + Ipilimumab | Responses were observed regardless of PD-L1 expression levels | Sharma et al. [91] |
| M1-like TAM | Metastatic urothelial carcinoma (IMvigor210) | Atezolizumab | M1 frequency is a robust biomarker for predicting the prognosis and response to immune checkpoint blockades | Zeng et al. [92] |
| Pro-tumorigenic inflammation signature | Metastatic urothelial carcinoma (IMvigor210) | Atezolizumab | Pro-tumorigenic inflammation in individual tumor microenvironments is associated with PD-1 and PD-L1 resistance | Wang et al. [93] |
| Metastatic urothelial carcinoma (CheckMate 275) | Nivolumab |
| Targeted Pathways | Agent Names | Combinations | Tumor Types | Clinical Phases | Trial Numbers |
|---|---|---|---|---|---|
| TLR | BDB001 | Atezolizumab + Radiotherapy | Advanced solid tumors * | II | NCT03915678 |
| Imiquimod | Carcinoma in situ bladder cancer | II | NCT01731652 | ||
| Imiquimod | TRK-950 | Advanced solid tumors * | I | NCT03872947 | |
| Poly(I:C) | PGV001 + Atezolizumab | Urothelial/bladder cancer | I | NCT03359239 | |
| Poly(I:C) | Durvalumab +/− Tremelimumab | Advanced solid tumors * | I/II | NCT02643303 | |
| HDAC | Abexinostat | Pembrolizumab | Advanced solid tumor * | I | NCT03590054 |
| Belinostat | Bladder cancer | I/II | NCT00421889 | ||
| Chidamide | Tislelizumab | Bladder cancer stage IV | II | NCT04562311 | |
| Domatinostat | Nivolumab +/− Ipilimumab | Urothelial carcinoma | I | NCT04871594 | |
| Entinostat | Pembrolizumab | MIBC | II | NCT03978624 | |
| FR901228 | Advanced urothelial carcinoma | II | NCT00087295 | ||
| Mocetinostat | Urothelial carcinoma | II | NCT02236195 | ||
| Romidepsin | Solid tumors * | I | NCT01638533 | ||
| Vorinostat | Locally recurrent or metastatic urothelial carcinoma | II | NCT00363883 | ||
| Vorinostat | Docetaxel | Advanced and relapsed solid tumors * | I | NCT00565227 | |
| Vorinostat | Pembrolizumab | Advanced urothelial cell carcinoma | I | NCT02619253 | |
| PI3K | Buparlisib | Metastatic urothelial carcinoma | II | NCT01551030 | |
| Copanlisib Copanlisib Hydrochloride GSK2636771 Taselisib | Advanced solid tumors * | II | NCT02465060 | ||
| Eganelisib | Nivolumab | Advanced urothelial carcinoma | II | NCT03980041 | |
| CD40 | APX005M | Urothelial carcinoma | I | NCT02482168 | |
| CDX-1140 | +/− CDX-301 +/− Pembrolizumab +/− Chemotherapy | Advanced solid tumors * | I | NCT03329950 | |
| CD47 | Hu5F9-G4 | Atezolizumab | Cisplatin-ineligible MIBC and locally advanced or metastatic urothelial carcinoma | I/II | NCT03869190 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leblond, M.M.; Zdimerova, H.; Desponds, E.; Verdeil, G. Tumor-Associated Macrophages in Bladder Cancer: Biological Role, Impact on Therapeutic Response and Perspectives for Immunotherapy. Cancers 2021, 13, 4712. https://doi.org/10.3390/cancers13184712
Leblond MM, Zdimerova H, Desponds E, Verdeil G. Tumor-Associated Macrophages in Bladder Cancer: Biological Role, Impact on Therapeutic Response and Perspectives for Immunotherapy. Cancers. 2021; 13(18):4712. https://doi.org/10.3390/cancers13184712
Chicago/Turabian StyleLeblond, Marine M., Hana Zdimerova, Emma Desponds, and Grégory Verdeil. 2021. "Tumor-Associated Macrophages in Bladder Cancer: Biological Role, Impact on Therapeutic Response and Perspectives for Immunotherapy" Cancers 13, no. 18: 4712. https://doi.org/10.3390/cancers13184712
APA StyleLeblond, M. M., Zdimerova, H., Desponds, E., & Verdeil, G. (2021). Tumor-Associated Macrophages in Bladder Cancer: Biological Role, Impact on Therapeutic Response and Perspectives for Immunotherapy. Cancers, 13(18), 4712. https://doi.org/10.3390/cancers13184712





