Combination of a 15-SNP Polygenic Risk Score and Classical Risk Factors for the Prediction of Breast Cancer Risk in Cypriot Women
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. SNP Selection and Genotyping
2.3. Statistical Analysis
3. Results
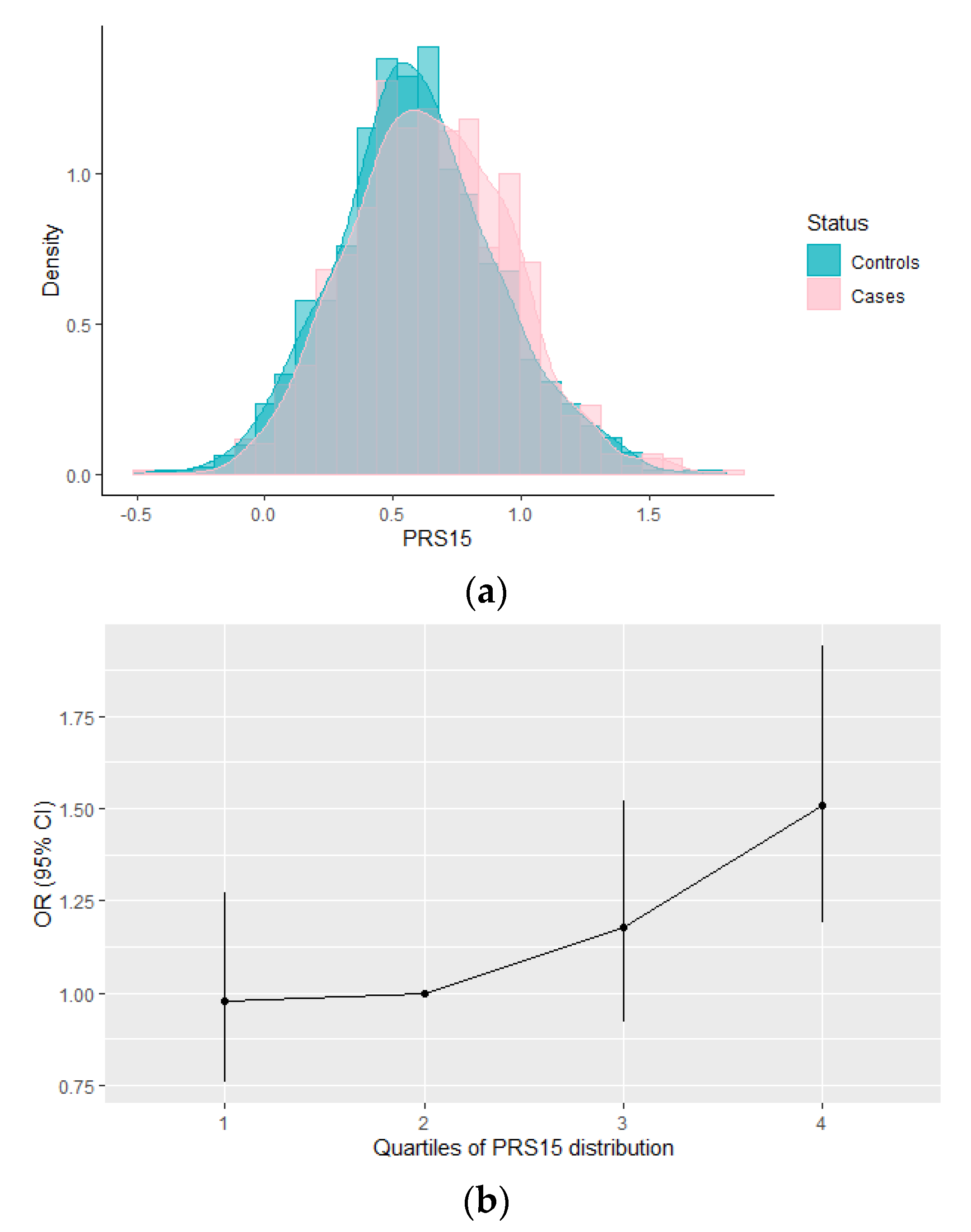
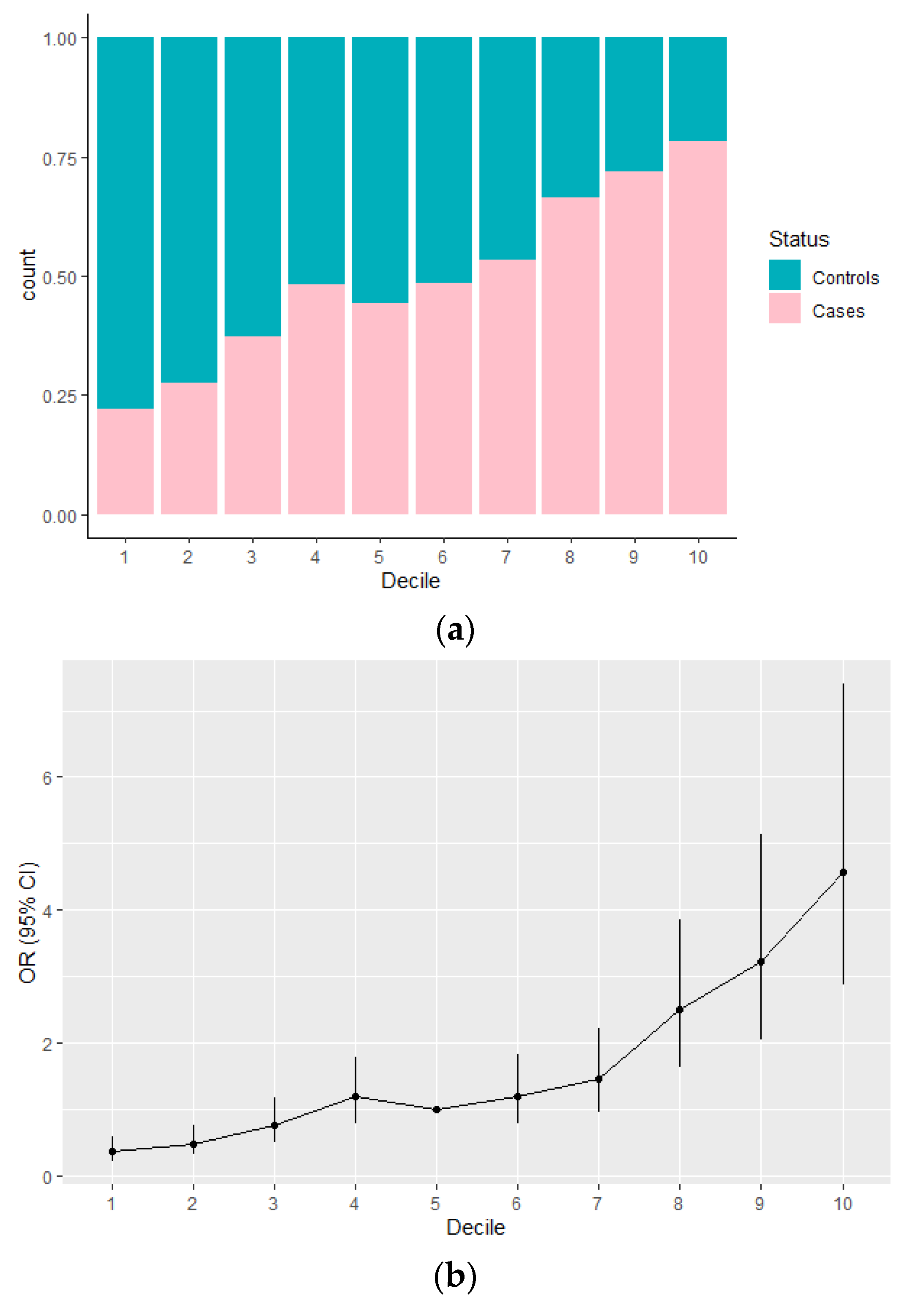
3.1. Evaluation of the PRS15 and Its Association with Breast Cancer Risk in Greek-Cypriot Women
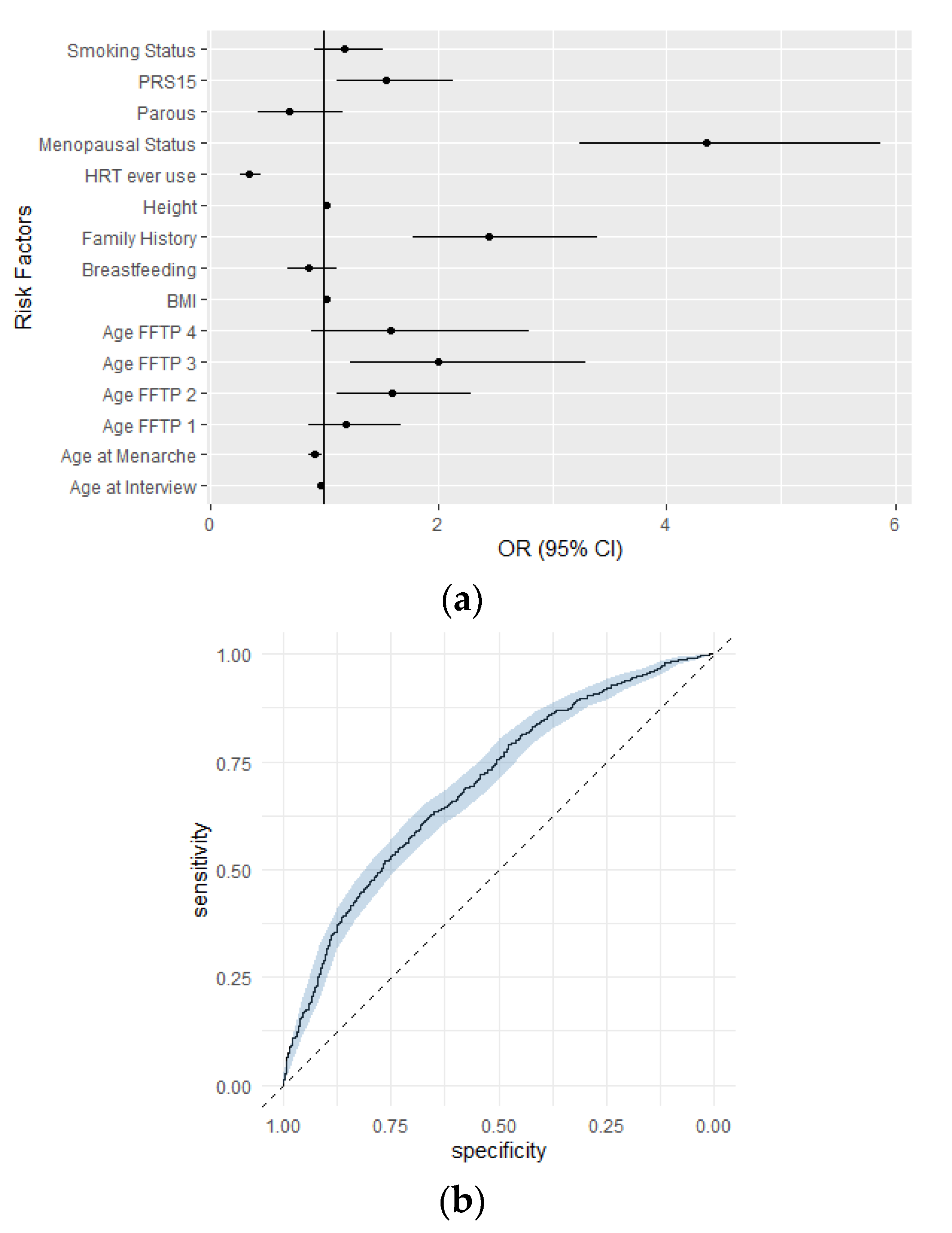
3.2. Association between the Integrated Risk Model Consisting of PRS15 and Classical Risk Factors with Breast Cancer Risk in Greek-Cypriot Women
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Green, J.; Cairns, B.J.; Casabonne, D.; Wright, F.L.; Reeves, G.; Beral, V. Height and cancer incidence in the Million Women Study: Prospective cohort, and meta-analysis of prospective studies of height and total cancer risk. Lancet Oncol. 2011, 12, 785–794. [Google Scholar] [CrossRef] [Green Version]
- Hamajima, N.; Hirose, K.; Tajima, K.; Rohan, T.; Calle, E.E.; Heath, C.W.; Coates, R.J.; Liff, J.M.; Talamini, R.; Chantarakul, N.; et al. Alcohol, tobacco and breast cancer-Collaborative reanalysis of individual data from 53 epidemiological studies, including 58 515 women with breast cancer and 95 067 women without the disease. Br. J. Cancer 2002, 87, 1234–1245. [Google Scholar] [CrossRef]
- Hamajima, N.; Hirose, K.; Tajima, K.; Rohan, T.; Friedenreich, C.M.; Calle, E.E.; Gapstur, S.M.; Patel, A.V.; Coates, R.J.; Liff, J.M.; et al. Menarche, menopause, and breast cancer risk: Individual participant meta-analysis, including 118 964 women with breast cancer from 117 epidemiological studies. Lancet Oncol. 2012, 13, 1141–1151. [Google Scholar] [CrossRef]
- Morimoto, L.M.; White, E.; Chen, Z.; Chlebowski, R.T.; Hays, J.; Kuller, L.; Lopez, A.M.; Manson, J.A.; Margolis, K.L.; Muti, P.C.; et al. Obesity, body size, and risk of postmenopausal breast cancer: The women’s health initiative (United States). Cancer Causes Control 2002, 13, 741–751. [Google Scholar] [CrossRef] [PubMed]
- Collaborative Group on Hormonal Factors in Breast Cancer. Breast cancer and hormone replacement therapy: Collaborative reanalysis of data from 51 epidemiological studies of 52,705 women with breast cancer and 108,411 women without breast cancer. Collaborative Group on Hormonal Factors in Breast Cancer. Lancet 1997, 350, 1047–1059. [Google Scholar] [CrossRef]
- Michailidou, K.; Beesley, J.; Lindstrom, S.; Canisius, S.; Dennis, J.; Lush, M.J.; Maranian, M.J.; Bolla, M.K.; Wang, Q.; Shah, M.; et al. Genome-wide association analysis of more than 120,000 individuals identifies 15 new susceptibility loci for breast cancer. Nat. Genet. 2015, 47, 373–380. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Michailidou, K.; Hall, P.; Gonzalez-Neira, A.; Ghoussaini, M.; Dennis, J.; Milne, R.L.; Schmidt, M.K.; Chang-Claude, J.; Bojesen, S.E.; Bolla, M.K.; et al. Large-scale genotyping identifies 41 new loci associated with breast cancer risk. Nat. Genet. 2013, 45, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Michailidou, K.; Lindström, S.; Dennis, J.; Beesley, J.; Hui, S.; Kar, S.; Lemaçon, A.; Soucy, P.; Glubb, D.; Rostamianfar, A.; et al. Association analysis identifies 65 new breast cancer risk loci. Nature 2017, 551, 92–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, H.; Ahearn, T.U.; Lecarpentier, J.; Barnes, D.; Beesley, J.; Qi, G.; Jiang, X.; O’Mara, T.A.; Zhao, N.; Bolla, M.K.; et al. Genome-wide association study identifies 32 novel breast cancer susceptibility loci from overall and subtype-specific analyses. Nat. Genet. 2020, 52, 572–581. [Google Scholar] [CrossRef]
- Pashayan, N.; Morris, S.; Gilbert, F.J.; Pharoah, P.D.P. Cost-effectiveness and Benefit-to-Harm Ratio of Risk-Stratified Screening for Breast Cancer A Life-Table Model. JAMA Oncol. 2018, 4, 1504–1510. [Google Scholar] [CrossRef] [Green Version]
- Burton, H.; Chowdhury, S.; Dent, T.; Hall, A.; Pashayan, N.; Pharoah, P. Public health implications from COGS and potential for risk stratification and screening. Nat. Genet. 2013, 45, 349–351. [Google Scholar] [CrossRef]
- Khera, A.V.; Chaffin, M.; Aragam, K.G.; Haas, M.E.; Roselli, C.; Choi, S.H.; Natarajan, P.; Lander, E.S.; Lubitz, S.A.; Ellinor, P.T.; et al. Genome-wide polygenic scores for common diseases identify individuals with risk equivalent to monogenic mutations. Nat. Genet. 2018, 50, 1219–1224. [Google Scholar] [CrossRef]
- Mavaddat, N.; Michailidou, K.; Dennis, J.; Lush, M.; Fachal, L.; Lee, A.; Tyrer, J.P.; Chen, T.H.; Wang, Q.; Bolla, M.K.; et al. Polygenic Risk Scores for Prediction of Breast Cancer and Breast Cancer Subtypes. Am. J. Hum. Genet. 2019, 104, 21–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mavaddat, N.; P Pharoah, P.D.; Michailidou, K.; Tyrer, J.; Brook, M.N.; Bolla, M.K.; Wang, Q.; Dennis, J.; Dunning, A.M.; Shah, M.; et al. Prediction of Breast Cancer Risk Based on Profiling With Common Genetic Variants. JNCI J. Natl. Cancer Inst. 2015, 107, 36. [Google Scholar] [CrossRef] [PubMed]
- Torkamani, A.; Wineinger, N.E.; Topol, E.J. The personal and clinical utility of polygenic risk scores. Nat. Rev. Genet. 2018, 19, 581–590. [Google Scholar] [CrossRef]
- Zhang, Y.D.; Hurson, A.N.; Zhang, H.; Choudhury, P.P.; Easton, D.F.; Milne, R.L.; Simard, J.; Hall, P.; Michailidou, K.; Dennis, J.; et al. Assessment of polygenic architecture and risk prediction based on common variants across fourteen cancers. Nat. Commun. 2020, 11, 3353. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, N.; Shi, J.; García-Closas, M. Developing and evaluating polygenic risk prediction models for stratified disease prevention. Nat. Rev. Genet. 2016, 17, 392–406. [Google Scholar] [CrossRef] [PubMed]
- Hurson, A.N.; Pal Choudhury, P.; Gao, C.; Hüsing, A.; Eriksson, M.; Shi, M.; Jones, M.E.; Evans, D.G.R.; Milne, R.L.; Gaudet, M.M.; et al. Prospective evaluation of a breast-cancer risk model integrating classical risk factors and polygenic risk in 15 cohorts from six countries. Int. J. Epidemiol. 2021. [Google Scholar] [CrossRef]
- Maas, P.; Barrdahl, M.; Joshi, A.D.; Auer, P.L.; Gaudet, M.M.; Milne, R.L.; Schumacher, F.R.; Anderson, W.F.; Check, D.; Chattopadhyay, S.; et al. Breast Cancer Risk from Modifiable and Nonmodifiable Risk Factors among White Women in the United States. JAMA Oncol. 2016, 2, 1295–1302. [Google Scholar] [CrossRef] [PubMed]
- Wand, H.; Lambert, S.A.; Tamburro, C.; Iacocca, M.A.; O’Sullivan, J.W.; Sillari, C.; Kullo, I.J.; Rowley, R.; Dron, J.S.; Brockman, D.; et al. Improving reporting standards for polygenic scores in risk prediction studies. Nature 2021, 591, 211–219. [Google Scholar] [CrossRef]
- Triviño, J.C.; Ceba, A.; Rubio-Solsona, E.; Serra, D.; Sanchez-Guiu, I.; Ribas, G.; Rosa, R.; Cabo, M.; Bernad, L.; Pita, G.; et al. Combination of phenotype and polygenic risk score in breast cancer risk evaluation in the Spanish population: A case –control study. BMC Cancer 2020, 20, 1079. [Google Scholar] [CrossRef]
- Garcia-Closas, M.; Gunsoy, N.B.; Chatterjee, N. Combined associations of genetic and environmental risk factors: Implications for prevention of breast cancer. J. Natl. Cancer Inst. 2014, 106, dju305. [Google Scholar] [CrossRef]
- Kapoor, P.M.; Mavaddat, N.; Choudhury, P.P.; Wilcox, A.N.; Lindström, S.; Behrens, S.; Michailidou, K.; Dennis, J.; Bolla, M.K.; Wang, Q.; et al. Combined Associations of a Polygenic Risk Score and Classical Risk Factors With Breast Cancer Risk. J. Natl. Cancer Inst. 2021, 113, 329–337. [Google Scholar] [CrossRef]
- Lee, A.; Mavaddat, N.; Wilcox, A.N.; Cunningham, A.P.; Carver, T.; Hartley, S.; Babb de Villiers, C.; Izquierdo, A.; Simard, J.; Schmidt, M.K.; et al. BOADICEA: A comprehensive breast cancer risk prediction model incorporating genetic and nongenetic risk factors. Genet. Med. 2019, 21, 1708–1718. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Veen, E.M.; Brentnall, A.R.; Byers, H.; Harkness, E.F.; Astley, S.M.; Sampson, S.; Howell, A.; Newman, W.G.; Cuzick, J.; Evans, G.R. Use of single-nucleotide polymorphisms and mammographic density plus classic risk factors for breast cancer risk prediction. JAMA Oncol. 2018, 4, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, P.P.; Wilcox, A.N.; Brook, M.N.; Zhang, Y.; Ahearn, T.; Orr, N.; Coulson, P.; Schoemaker, M.J.; Jones, M.E.; Gail, M.H.; et al. Comparative validation of breast cancer risk prediction models and projections for future risk stratification. J. Natl. Cancer Inst. 2021, 112, 278–285. [Google Scholar] [CrossRef]
- Lakeman, I.M.M.; Rodríguez-Girondo, M.; Lee, A.; Ruiter, R.; Stricker, B.H.; Wijnant, S.R.A.; Kavousi, M.; Antoniou, A.C.; Schmidt, M.K.; Uitterlinden, A.G.; et al. Validation of the BOADICEA model and a 313-variant polygenic risk score for breast cancer risk prediction in a Dutch prospective cohort. Genet. Med. Off. J. Am. Coll. Med. Genet. 2020, 22, 1803–1811. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Rice, M.; Tworoger, S.S.; Rosner, B.A.; Eliassen, A.H.; Tamimi, R.M.; Joshi, A.D.; Lindstrom, S.; Qian, J.; Colditz, G.A.; et al. Addition of a polygenic risk score, mammographic density, and endogenous hormones to existing breast cancer risk prediction models: A nested case-control study. PLoS Med. 2018, 15, e1002644. [Google Scholar] [CrossRef] [PubMed]
- Yanes, T.; Young, M.A.; Meiser, B.; James, P.A. Clinical applications of polygenic breast cancer risk: A critical review and perspectives of an emerging field. Breast Cancer Res. 2020, 22, 21. [Google Scholar] [CrossRef] [Green Version]
- Pavlou, P.; Demetriou, A. Cyprus Cancer Registry (CyCR). In Progress Report August 2016; Ministry of Health: Nicosia, Cyprus, 2016; p. 86. [Google Scholar]
- Loizidou, M.A.; Hadjisavvas, A.; Ioannidis, J.P.A.; Kyriacou, K. Replication of genome-wide discovered breast cancer risk loci in the Cypriot population. Breast Cancer Res. Treat. 2011, 128, 267–272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hadjisavvas, A.; Loizidou, M.A.; Middleton, N.; Michael, T.; Papachristoforou, R.; Kakouri, E.; Daniel, M.; Papadopoulos, P.; Malas, S.; Marcou, Y.; et al. An investigation of breast cancer risk factors in Cyprus: A case control study. BMC Cancer 2010, 10, 447. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, S.; Thomas, G.; Ghoussaini, M.; Healey, C.S.; Humphreys, M.K.; Platte, R.; Morrison, J.; Maranian, M.; Pooley, K.A.; Luben, R.; et al. Newly discovered breast cancer susceptibility loci on 3p24 and 17q23.2. Nat. Genet. 2009, 41, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Easton, D.F.; Pooley, K.A.; Dunning, A.M.; Pharoah, P.D.P.; Thompson, D.; Ballinger, D.G.; Struewing, J.P.; Morrison, J.; Field, H.; Luben, R.; et al. Genome-wide association study identifies novel breast cancer susceptibility loci. Nature 2007, 447, 1087–1093. [Google Scholar] [CrossRef] [Green Version]
- Ghoussaini, M.; Fletcher, O.; Michailidou, K.; Turnbull, C.; Schmidt, M.K.; Dicks, E.; Dennis, J.; Wang, Q.; Humphreys, M.K.; Luccarini, C.; et al. Genome-wide association analysis identifies three new breast cancer susceptibility loci. Nat. Genet. 2012, 44, 312–318. [Google Scholar] [CrossRef]
- Stacey, S.N.; Manolescu, A.; Sulem, P.; Rafnar, T.; Gudmundsson, J.; Gudjonsson, S.A.; Masson, G.; Jakobsdottir, M.; Thorlacius, S.; Helgason, A.; et al. Common variants on chromosomes 2q35 and 16q12 confer susceptibility to estrogen receptor-positive breast cancer. Nat. Genet. 2007, 39, 865–869. [Google Scholar] [CrossRef]
- Thomas, G.; Jacobs, K.B.; Kraft, P.; Yeager, M.; Wacholder, S.; Cox, D.G.; Hankinson, S.E.; Hutchinson, A.; Wang, Z.; Yu, K.; et al. A multistage genome-wide association study in breast cancer identifies two new risk alleles at 1p11.2 and 14q24.1 (RAD51L1). Nat. Genet. 2009, 41, 579–584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turnbull, C.; Ahmed, S.; Morrison, J.; Pernet, D.; Renwick, A.; Maranian, M.; Seal, S.; Ghoussaini, M.; Hines, S.; Healey, C.S.; et al. Genome-wide association study identifies five new breast cancer susceptibility loci. Nat. Genet. 2010, 42, 504–507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, W.; Long, J.; Gao, Y.T.; Li, C.; Zheng, Y.; Xiang, Y.B.; Wen, W.; Levy, S.; Deming, S.L.; Haines, J.L.; et al. Genome-wide association study identifies a new breast cancer susceptibility locus at 6q25.1. Nat. Genet. 2009, 41, 324–328. [Google Scholar] [CrossRef] [Green Version]
- Team, R.C. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Pharoah, P.D.; Antoniou, A.; Bobrow, M.; Zimmern, R.L.; Easton, D.F.; Ponder, B.A. Polygenic susceptibility to breast cancer and implications for prevention. Nat. Genet. 2002, 31, 33–36. [Google Scholar] [CrossRef] [PubMed]
- Robin, X.; Turck, N.; Hainard, A.; Tiberti, N.; Lisacek, F.; Sanchez, J.C.; Müller, M. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 2011, 12, 77. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Closas, M.; Chatterjee, N. Assessment of breast cancer risk: Which tools to use? Lancet Oncol. 2019, 20, 463–464. [Google Scholar] [CrossRef]
- Lambert, S.A.; Gil, L.; Jupp, S.; Ritchie, S.C.; Xu, Y.; Buniello, A.; McMahon, A.; Abraham, G.; Chapman, M.; Parkinson, H.; et al. The Polygenic Score Catalog as an open database for reproducibility and systematic evaluation. Nat. Genet. 2021, 53, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, A.; Song, M.; Brook, M.N.; Milne, R.L.; Mavaddat, N.; Michailidou, K.; Bolla, M.K.; Wang, Q.; Dennis, J.; Wilcox, A.N.; et al. Joint associations of a polygenic risk score and environmental risk factors for breast cancer in the Breast Cancer Association Consortium. Int. J. Epidemiol. 2018, 47, 526–536. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| CHR 1 | SNP | Position 2 | Alleles 3 | MAF 4 | iCOGS OR 5 | iCOGS p-Value 6 | MASTOS MAF 7 | MASTOS OR 8 | MASTOS p-Value | |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | rs11249433 | 121280613 | A/G | 0.4 | 1.09 (1.07–1.12) | 0.46 | 1.00 (0.89–1.12) | 0.98 | ||
| 2 | rs13387042 | 217905832 | A/G | 0.49 | 0.88 (0.86–0.9) | 0.45 | 0.85 (0.75–0.95) | 0.005 | ||
| 3 | rs4973768 | 27416013 | C/T | 0.47 | 1.1 (1.08–1.12) | 0.45 | 0.89 (0.78–1.00) | 0.055 | ||
| 5 | rs889312 | 56031884 | A/C | 0.28 | 1.12 (1.1–1.15) | 0.29 | 1.18 (1.04–1.34) | 0.01 | ||
| 6 | rs2046210 | 151948366 | G/A | 0.34 | 1.08 (1.06–1.1) | 0.41 | 1.13 (1.00–1.27) | 0.047 | ||
| 8 | rs13281615 | 128355618 | A/G | 0.4 | 1.1 (1.08–1.12) | 0.48 | 1.07 (0.95–1.20) | 0.26 | ||
| 9 | rs1011970 | 22062134 | G/T | 0.17 | 1.06 (1.03–1.08) | 0.19 | 1.15 (0.99–1.33) | 0.07 | ||
| 10 | rs2981582 | 123352317 | G/A | 0.38 | 1.26 (1.24–1.28) | 0.44 | 1.16 (1.03–1.31) | 0.01 | ||
| 10 | rs10995190 | 64278682 | G/A | 0.16 | 0.86 (0.83–0.88) | 0.14 | 0.97 (0.82–1.15) | 0.7 | ||
| 10 | rs704010 | 80841148 | C/T | 0.38 | 1.08 (1.06–1.1) | 0.37 | 1.01 (0.90–1.14) | 0.83 | ||
| 11 | rs3817198 | 1909006 | T/C | 0.31 | 1.07 (1.05–1.09) | 0.31 | 0.97 (0.85–1.09) | 0.59 | ||
| 11 | rs614367 | 69328764 | C/T | 0.15 | 1.21 (1.18–1.24) | 0.11 | 1.09 (0.91–1.31) | 0.36 | ||
| 16 | rs3803662 | 52586341 | G/A | 0.26 | 1.24 (1.21–1.26) | 0.33 | 1.01 (0.89–1.14) | 0.86 | ||
| 17 | rs6504950 | 53056471 | G/A | 0.28 | 0.94 (0.92–0.96) | 0.26 | 0.94 (0.82–1.07) | 0.34 | ||
| 21 | rs2823093 | 16520832 | G/A | 0.27 | 0.93 (0.91–0.95) | 0.73 | 1.07 (0.94–1.23) | 0.28 |
| Decile. | Controls (%) | Cases (%) | OR (95% CI) | p-Value |
|---|---|---|---|---|
| 1 | 139 (15.4) | 39 (4.4) | 0.36 (0.22–0.57) | 1.55 × 10−5 |
| 2 | 135 (15) | 51 (5.8) | 0.48 (0.31–0.75) | 0.001 |
| 3 | 108 (12) | 64 (7.3) | 0.75 (0.49–1.16) | 0.2 |
| 4 | 98 (10.9) | 91 (10.3) | 1.18 (0.78–1.79) | 0.44 |
| 5 | 94 (10.4) | 74 (8.4) | 1 | - |
| 6 | 92 (10.2) | 86 (9.8) | 1.19 (0.78–1.82) | 0.43 |
| 7 | 89 (9.9) | 102 (11.6) | 1.46 (0.96–2.21) | 0.08 |
| 8 | 62 (6.9) | 122 (13.9) | 2.5 (1.63–3.86) | 3.17 × 10−5 |
| 9 | 45 (5) | 114 (13) | 3.22 (2.04–5.13) | 6.46 × 10−7 |
| 10 | 38 (4.2) | 137 (15.6) | 4.58 (2.88–7.4) | 2.44 × 10−10 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yiangou, K.; Kyriacou, K.; Kakouri, E.; Marcou, Y.; Panayiotidis, M.I.; Loizidou, M.A.; Hadjisavvas, A.; Michailidou, K. Combination of a 15-SNP Polygenic Risk Score and Classical Risk Factors for the Prediction of Breast Cancer Risk in Cypriot Women. Cancers 2021, 13, 4568. https://doi.org/10.3390/cancers13184568
Yiangou K, Kyriacou K, Kakouri E, Marcou Y, Panayiotidis MI, Loizidou MA, Hadjisavvas A, Michailidou K. Combination of a 15-SNP Polygenic Risk Score and Classical Risk Factors for the Prediction of Breast Cancer Risk in Cypriot Women. Cancers. 2021; 13(18):4568. https://doi.org/10.3390/cancers13184568
Chicago/Turabian StyleYiangou, Kristia, Kyriacos Kyriacou, Eleni Kakouri, Yiola Marcou, Mihalis I. Panayiotidis, Maria A. Loizidou, Andreas Hadjisavvas, and Kyriaki Michailidou. 2021. "Combination of a 15-SNP Polygenic Risk Score and Classical Risk Factors for the Prediction of Breast Cancer Risk in Cypriot Women" Cancers 13, no. 18: 4568. https://doi.org/10.3390/cancers13184568
APA StyleYiangou, K., Kyriacou, K., Kakouri, E., Marcou, Y., Panayiotidis, M. I., Loizidou, M. A., Hadjisavvas, A., & Michailidou, K. (2021). Combination of a 15-SNP Polygenic Risk Score and Classical Risk Factors for the Prediction of Breast Cancer Risk in Cypriot Women. Cancers, 13(18), 4568. https://doi.org/10.3390/cancers13184568








