The Prognostic Effect of KRAS Mutations in Non-Small Cell Lung Carcinoma Revisited: A Norwegian Multicentre Study
Abstract
:Simple Summary
Abstract
1. Introduction
2. Material and Methods
2.1. Ethics
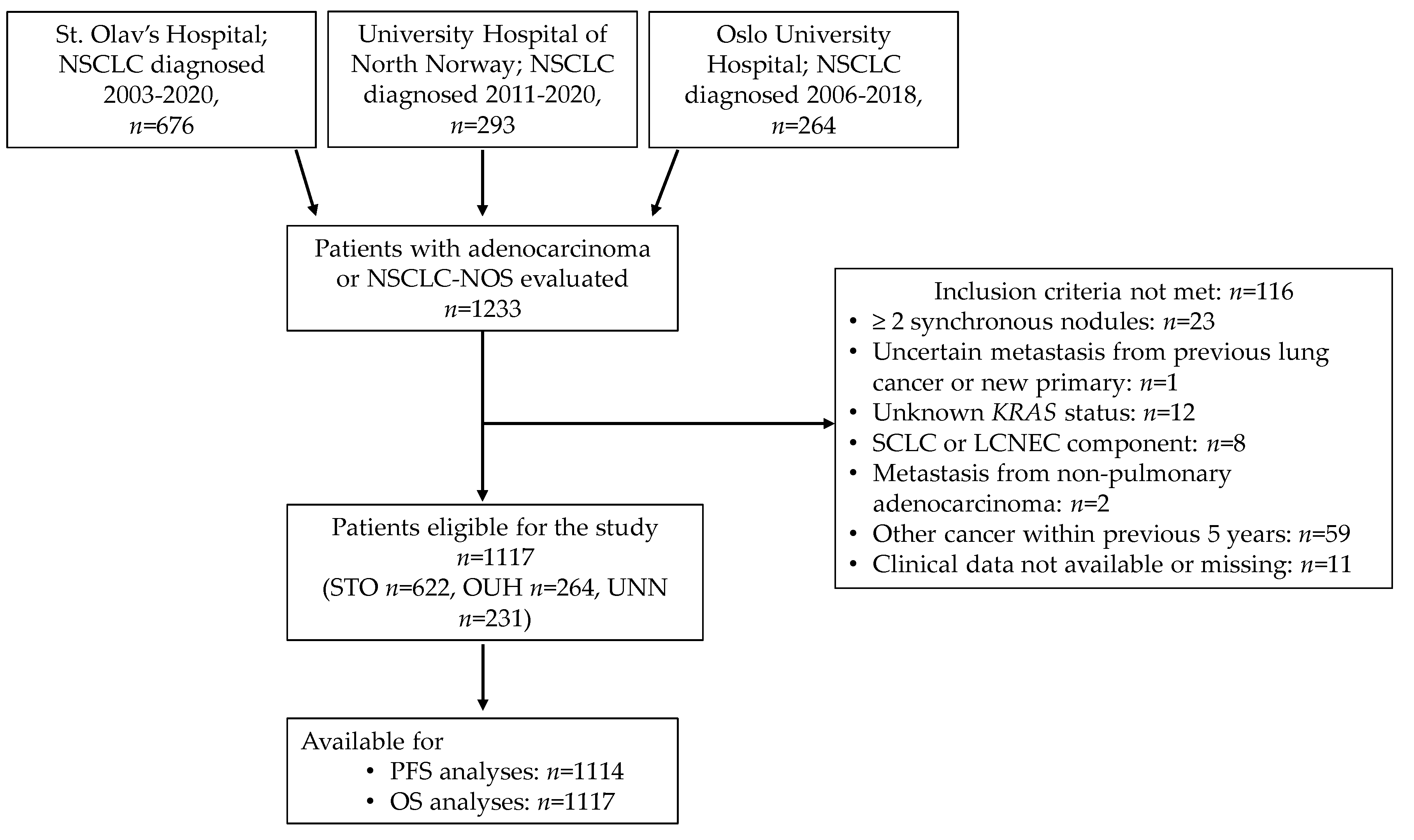
2.2. Patient Inclusion and Tumour Specimens
2.3. Statistics
3. Results
3.1. Patient Characteristics
3.2. KRAS Mutation Status and Correlations with Clinical Characteristics
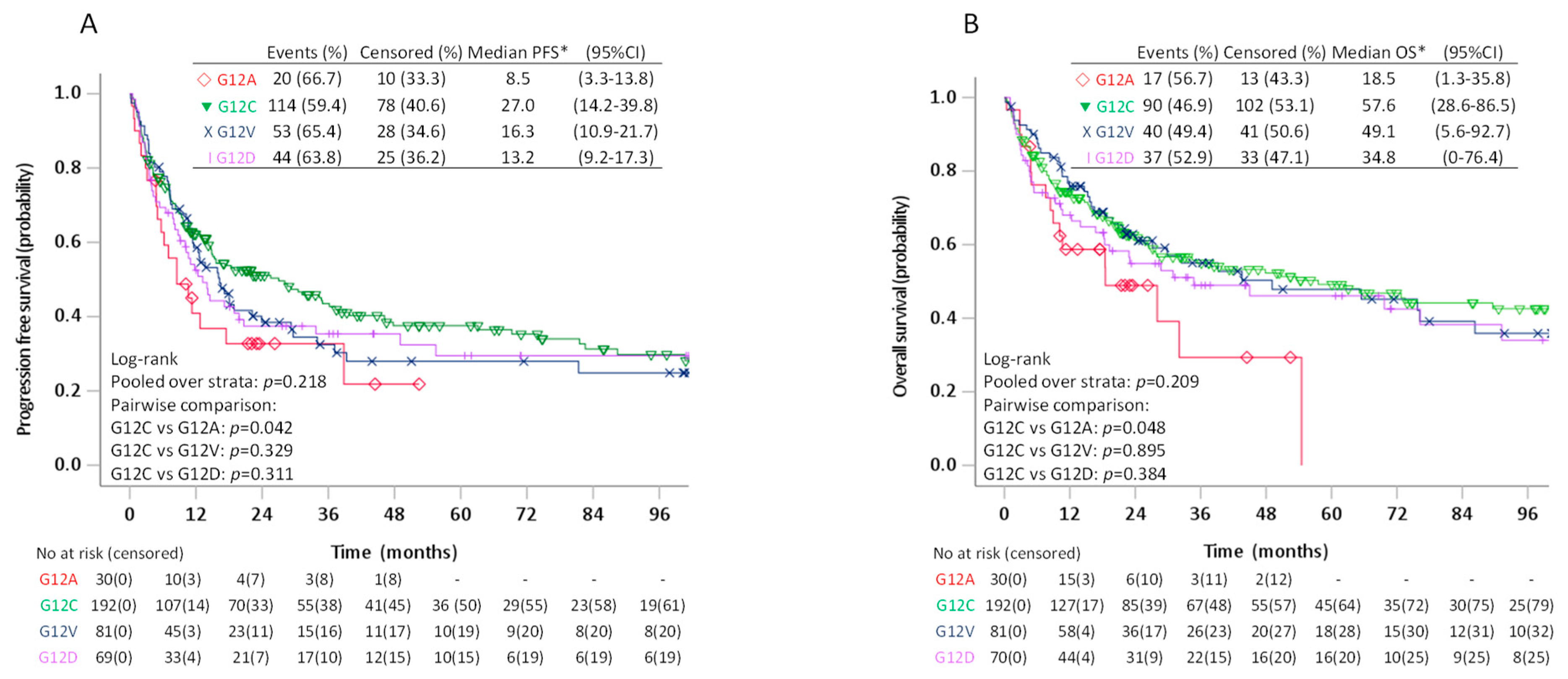
3.3. Mutation Status and Survival
3.3.1. Whole Cohort (Stage I–IV)
3.3.2. Curative Surgery
3.3.3. Non-Curative Treatment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dogan, S.; Shen, R.; Ang, D.C.; Johnson, M.L.; D’Angelo, S.P.; Paik, P.K.; Brzostowski, E.B.; Riely, G.J.; Kris, M.G.; Zakowski, M.F.; et al. Molecular epidemiology of EGFR and KRAS mutations in 3,026 lung adenocarcinomas: Higher susceptibility of women to smoking-related KRAS-mutant cancers. Clin. Cancer Res. 2012, 18, 6169–6177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El Osta, B.; Behera, M.; Kim, S.; Berry, L.D.; Sica, G.; Pillai, R.N.; Owonikoko, T.K.; Kris, M.G.; Johnson, B.E.; Kwiatkowski, D.J.; et al. Characteristics and Outcomes of Patients With Metastatic KRAS-Mutant Lung Adenocarcinomas: The Lung Cancer Mutation Consortium Experience. J. Thorac. Oncol. 2019, 14, 876–889. [Google Scholar] [CrossRef]
- Kohno, T.; Nakaoku, T.; Tsuta, K.; Tsuchihara, K.; Matsumoto, S.; Yoh, K.; Goto, K. Beyond ALK-RET, ROS1 and other oncogene fusions in lung cancer. Transl. Lung Cancer Res. 2015, 4, 156–164. [Google Scholar] [PubMed]
- Finn, S.P.; Addeo, A.; Dafni, U.; Thunnissen, E.; Bubendorf, L.; Madsen, L.B.; Biernat, W.; Verbeken, E.; Hernandez-Losa, J.; Marchetti, A.; et al. Prognostic Impact of KRAS G12C Mutation in Patients With NSCLC: Results From the European Thoracic Oncology Platform Lungscape Project. J. Thorac. Oncol. 2021, 16, 990–1002. [Google Scholar] [CrossRef]
- D’Angelo, S.P.; Janjigian, Y.Y.; Ahye, N.; Riely, G.J.; Chaft, J.E.; Sima, C.S.; Shen, R.; Zheng, J.; Dycoco, J.; Kris, M.G.; et al. Distinct Clinical Course of EGFR-Mutant Resected Lung Cancers: Results of Testing of 1118 Surgical Specimens and Effects of Adjuvant Gefitinib and Erlotinib. J. Thorac. Oncol. 2012, 7, 1815–1822. [Google Scholar] [CrossRef] [Green Version]
- Shepherd, F.A.; Domerg, C.; Hainaut, P.; Jänne, P.A.; Pignon, J.-P.; Graziano, S.; Douillard, J.-Y.; Brambilla, E.; Le Chevalier, T.; Seymour, L.; et al. Pooled analysis of the prognostic and predictive effects of KRAS mutation status and KRAS mutation subtype in early-stage resected non-small-cell lung cancer in four trials of adjuvant chemotherapy. J. Clin. Oncol. 2013, 31, 2173–2181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rulli, E.; Marabese, M.; Torri, V.; Farina, G.; Veronese, S.; Bettini, A.; Longo, F.; Moscetti, L.; Ganzinelli, M.; Lauricella, C.; et al. Value of KRAS as prognostic or predictive marker in NSCLC: Results from the TAILOR trial. Ann. Oncol. 2015, 26, 2079–2084. [Google Scholar] [CrossRef] [PubMed]
- Zer, A.; Ding, K.; Lee, S.M.; Goss, G.D.; Seymour, L.; Ellis, P.M.; Hackshaw, A.; Bradbury, P.A.; Han, L.; O’Callaghan, C.J.; et al. Pooled Analysis of the Prognostic and Predictive Value of KRAS Mutation Status and Mutation Subtype in Patients with Non Small Cell Lung Cancer Treated with Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitors. J. Thorac. Oncol. 2016, 11, 312–323. [Google Scholar] [CrossRef] [Green Version]
- Shepherd, F.A.; Lacas, B.; Le Teuff, G.; Hainaut, P.; Jänne, P.A.; Pignon, J.-P.; Le Chevalier, T.; Seymour, L.; Douillard, J.-Y.; Graziano, S.; et al. Pooled Analysis of the Prognostic and Predictive Effects of TP53 Comutation Status Combined With KRAS or EGFR Mutation in Early-Stage Resected Non-Small-Cell Lung Cancer in Four Trials of Adjuvant Chemotherapy. J. Clin. Oncol. 2017, 35, 2018–2027. [Google Scholar] [CrossRef]
- Izar, B.; Zhou, H.; Heist, R.S.; Azzoli, C.G.; Muzikansky, A.; Scribner, E.E.F.; Bernardo, L.A.; Dias-Santagata, D.; Iafrate, A.J.; Lanuti, M. The Prognostic Impact of KRAS, Its Codon and Amino Acid Specific Mutations, on Survival in Resected Stage I Lung Adenocarcinoma. J. Thorac. Oncol. 2014, 9, 1363–1369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kadota, K.; Sima, C.S.; Arcila, M.E.; Hedvat, C.; Kris, M.G.; Jones, D.R.; Adusumilli, P.S.; Travis, W.D. KRAS Mutation Is a Significant Prognostic Factor in Early-stage Lung Adenocarcinoma. Am. J. Surg. Pathol. 2016, 40, 1579–1590. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arbour, K.C.; Jordan, E.; Kim, H.R.; Dienstag, J.; Yu, H.A.; Sanchez-Vega, F.; Lito, P.; Berger, M.; Solit, D.B.; Hellmann, M.; et al. Effects of Co-occurring Genomic Alterations on Outcomes in Patients with KRAS-Mutant Non–Small Cell Lung Cancer. Clin. Cancer Res. 2018, 24, 334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aredo, J.V.; Padda, S.K.; Kunder, C.A.; Han, S.S.; Neal, J.W.; Shrager, J.B.; Wakelee, H.A. Impact of KRAS mutation subtype and concurrent pathogenic mutations on non-small cell lung cancer outcomes. Lung Cancer 2019, 133, 144–150. [Google Scholar] [CrossRef]
- Vetter, I.R.; Wittinghofer, A. The Guanine Nucleotide-Binding Switch in Three Dimensions. Science 2001, 294, 1299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tate, J.G.; Bamford, S.; Jubb, H.C.; Sondka, Z.; Beare, D.M.; Bindal, N.; Boutselakis, H.; Cole, C.G.; Creatore, C.; Dawson, E.; et al. COSMIC: The Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 2018, 47, D941–D947. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sebastian, M.; Eberhardt, W.E.E.; Hoffknecht, P.; Metzenmacher, M.; Wehler, T.; Kokowski, K.; Alt, J.; Schütte, W.; Büttner, R.; Heukamp, L.C.; et al. KRAS G12C-mutated advanced non-small cell lung cancer: A real-world cohort from the German prospective, observational, nation-wide CRISP Registry (AIO-TRK-0315). Lung Cancer 2021, 154, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Scheffzek, K.; Ahmadian, M.R.; Kabsch, W.; Wiesmüller, L.; Lautwein, A.; Schmitz, F.; Wittinghofer, A. The Ras-RasGAP complex: Structural basis for GTPase activation and its loss in oncogenic Ras mutants. Science 1997, 277, 333–338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hunter, J.C.; Manandhar, A.; Carrasco, M.A.; Gurbani, D.; Gondi, S.; Westover, K.D. Biochemical and Structural Analysis of Common Cancer-Associated KRAS Mutations. Mol. Cancer Res. 2015, 13, 1325. [Google Scholar] [CrossRef] [Green Version]
- Céspedes, M.V.; Sancho, F.J.; Guerrero, S.; Parreño, M.; Casanova, I.; Pavón, M.A.; Marcuello, E.; Trias, M.; Cascante, M.; Capellà, G.; et al. K-ras Asp12 mutant neither interacts with Raf, nor signals through Erk and is less tumorigenic than K-ras Val12. Carcinogenesis 2006, 27, 2190–2200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ihle, N.T.; Byers, L.A.; Kim, E.S.; Saintigny, P.; Lee, J.J.; Blumenschein, G.R.; Tsao, A.; Liu, S.; Larsen, J.E.; Wang, J.; et al. Effect of KRAS Oncogene Substitutions on Protein Behavior: Implications for Signaling and Clinical Outcome. JNCI J. Natl. Cancer Inst. 2012, 104, 228–239. [Google Scholar] [CrossRef]
- Travis, W.D.; Brambilla, E.; Burke, A.P.; Marx, A.; Nicholson, A.G. WHO Classification of Tumours of the Lung, Pleura, Thymus and Heart. WHO Classification of Tumours, 4th ed.; International Agency for Research on Cancer: Lyon, France, 2015; Volume 7. [Google Scholar]
- Detterbeck, F.C.; Boffa, D.J.; Kim, A.W.; Tanoue, L.T. The Eighth Edition Lung Cancer Stage Classification. Chest 2017, 151, 193–203. [Google Scholar] [CrossRef] [PubMed]
- La Fleur, L.; Falk-Sörqvist, E.; Smeds, P.; Berglund, A.; Sundström, M.; Mattsson, J.S.M.; Brandén, E.; Koyi, H.; Isaksson, J.; Brunnström, H.; et al. Mutation patterns in a population-based non-small cell lung cancer cohort and prognostic impact of concomitant mutations in KRAS and TP53 or STK11. Lung Cancer 2019, 130, 50–58. [Google Scholar] [CrossRef] [Green Version]
- Villaruz, L.C.; Socinski, M.A.; Cunningham, D.E.; Chiosea, S.I.; Burns, T.F.; Siegfried, J.M.; Dacic, S. The prognostic and predictive value of KRAS oncogene substitutions in lung adenocarcinoma. Cancer 2013, 119, 2268–2274. [Google Scholar] [CrossRef] [Green Version]
- Yu, H.A.; Sima, C.S.; Shen, R.; Kass, S.; Gainor, J.; Shaw, A.; Hames, M.; Iams, W.; Aston, J.; Lovly, C.M.; et al. Prognostic Impact of KRAS Mutation Subtypes in 677 Patients with Metastatic Lung Adenocarcinomas. J. Thorac. Oncol. 2015, 10, 431–437. [Google Scholar] [CrossRef] [Green Version]
- Cui, W.; Franchini, F.; Alexander, M.; Officer, A.; Wong, H.-L.; Ijzerman, M.; Desai, J.; Solomon, B.J. Real world outcomes in KRAS G12C mutation positive non-small cell lung cancer. Lung Cancer 2020, 146, 310–317. [Google Scholar] [CrossRef]
- Ruppert, A.-M.; Beau-Faller, M.; Debieuvre, D.; Ouafik, L.H.; Westeel, V.; Rouquette, I.; Mazières, J.; Bringuier, P.-P.; Monnet, I.; Escande, F.; et al. Outcomes of advanced Non-Small Cell Lung Cancer patients From the IFCT Biomarkers France study by KRAS Mutation Subtypes. JTO Clin. Res. Rep. 2020, 100052. [Google Scholar] [CrossRef]
- Nadal, E.; Chen, G.; Prensner, J.R.; Shiratsuchi, H.; Sam, C.; Zhao, L.; Kalemkerian, G.P.; Brenner, D.; Lin, J.; Reddy, R.M.; et al. KRAS-G12C Mutation Is Associated with Poor Outcome in Surgically Resected Lung Adenocarcinoma. J. Thorac. Oncol. 2014, 9, 1513–1522. [Google Scholar] [CrossRef] [Green Version]
- Stolze, B.; Reinhart, S.; Bulllinger, L.; Fröhling, S.; Scholl, C. Comparative analysis of KRAS codon 12, 13, 18, 61, and 117 mutations using human MCF10A isogenic cell lines. Sci. Rep. 2015, 5, 8535. [Google Scholar] [CrossRef] [PubMed]
- Skoulidis, F.; Byers, L.A.; Diao, L.; Papadimitrakopoulou, V.A.; Tong, P.; Izzo, J.; Behrens, C.; Kadara, H.; Parra, E.R.; Canales, J.R.; et al. Co-occurring Genomic Alterations Define Major Subsets of KRAS Mutant Lung Adenocarcinoma with Distinct Biology, Immune Profiles, and Therapeutic Vulnerabilities. Cancer Discov. 2015, 5, 860. [Google Scholar] [CrossRef] [Green Version]
- Koyama, S.; Akbay, E.A.; Li, Y.Y.; Aref, A.R.; Skoulidis, F.; Herter-Sprie, G.S.; Buczkowski, K.A.; Liu, Y.; Awad, M.M.; Denning, W.L.; et al. STK11/LKB1 Deficiency Promotes Neutrophil Recruitment and Proinflammatory Cytokine Production to Suppress T-cell Activity in the Lung Tumor Microenvironment. Cancer Res. 2016, 76, 999. [Google Scholar] [CrossRef] [Green Version]
- Skoulidis, F.; Goldberg, M.E.; Greenawalt, D.M.; Hellmann, M.D.; Awad, M.M.; Gainor, J.F.; Schrock, A.B.; Hartmaier, R.J.; Trabucco, S.E.; Gay, L.; et al. STK11/LKB1 Mutations and PD-1 Inhibitor Resistance in KRAS-Mutant Lung Adenocarcinoma. Cancer Discov. 2018, 8, 822–835. [Google Scholar] [CrossRef] [Green Version]
- Scheffler, M.; Ihle, M.A.; Hein, R.; Merkelbach-Bruse, S.; Scheel, A.H.; Siemanowski, J.; Brägelmann, J.; Kron, A.; Abedpour, N.; Ueckeroth, F.; et al. K-ras Mutation Subtypes in NSCLC and Associated Co-occuring Mutations in Other Oncogenic Pathways. J. Thorac. Oncol. 2019, 14, 606–616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, Z.Y.; Zhong, W.Z.; Zhang, X.C.; Su, J.; Xie, Z.; Liu, S.Y.; Tu, H.Y.; Chen, H.J.; Sun, Y.L.; Zhou, Q.; et al. Potential Predictive Value of TP53 and KRAS Mutation Status for Response to PD-1 Blockade Immunotherapy in Lung Adenocarcinoma. Clin. Cancer Res. 2017, 23, 3012–3024. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagy, Á.; Pongor, L.S.; Szabó, A.; Santarpia, M.; Győrffy, B. KRAS driven expression signature has prognostic power superior to mutation status in non-small cell lung cancer. Int. J. Cancer 2017, 140, 930–937. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Henry, J.T.; Coker, O.; Chowdhury, S.; Shen, J.P.; Morris, V.K.; Dasari, A.; Raghav, K.; Nusrat, M.; Kee, B.; Parseghian, C.; et al. Comprehensive Clinical and Molecular Characterization of KRASG12C-Mutant Colorectal Cancer. JCO Precis. Oncol. 2021, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Santos, E.; Martin-Zanca, D.; Reddy, E.P.; Pierotti, M.A.; Della Porta, G.; Barbacid, M. Malignant activation of a K-ras oncogene in lung carcinoma but not in normal tissue of the same patient. Science 1984, 223, 661. [Google Scholar] [CrossRef] [PubMed]
- Hong, D.S.; Fakih, M.G.; Strickler, J.H.; Desai, J.; Durm, G.A.; Shapiro, G.I.; Falchook, G.S.; Price, T.J.; Sacher, A.; Denlinger, C.S.; et al. KRASG12C Inhibition with Sotorasib in Advanced Solid Tumors. N. Engl. J. Med. 2020, 383, 1207–1217. [Google Scholar] [CrossRef]
- Skoulidis, F.; Li, B.T.; Dy, G.K.; Price, T.J.; Falchook, G.S.; Wolf, J.; Italiano, A.; Schuler, M.; Borghaei, H.; Barlesi, F.; et al. Sotorasib for Lung Cancers with KRAS p.G12C Mutation. N. Engl. J. Med. 2021, 384, 2371–2381. [Google Scholar] [CrossRef]
- Xue, J.Y.; Zhao, Y.; Aronowitz, J.; Mai, T.T.; Vides, A.; Qeriqi, B.; Kim, D.; Li, C.; de Stanchina, E.; Mazutis, L.; et al. Rapid non-uniform adaptation to conformation-specific KRAS(G12C) inhibition. Nature 2020, 577, 421–425. [Google Scholar] [CrossRef] [PubMed]
- Awad, M.M.; Liu, S.; Rybkin, I.I.; Arbour, K.C.; Dilly, J.; Zhu, V.W.; Johnson, M.L.; Heist, R.S.; Patil, T.; Riely, G.J.; et al. Acquired Resistance to KRASG12C Inhibition in Cancer. N. Engl. J. Med. 2021, 384, 2382–2393. [Google Scholar] [CrossRef]



| Characteristic | Total | KRAS wt n = 697 | KRAS mut n = 420 | |||
|---|---|---|---|---|---|---|
| Age (years) | ||||||
| <50 | 46 | (4.1) | 29 | (4.2) | 17 | (4.0) |
| 50–60 | 169 | (15.1) | 104 | (14.9) | 65 | (15.5) |
| >60 | 902 | (80.8) | 564 | (80.9) | 338 | (80.5) |
| Hospital | ||||||
| STO | 622 | (55.7) | 391 | (56.1) | 231 | (55.0) |
| UNN | 231 | (20.7) | 144 | (20.7) | 87 | (20.7) |
| OUH | 264 | (23.6) | 162 | (23.2) | 102 | (24.3) |
| Sex | ||||||
| Female | 592 | (53.0) | 353 | (50.6) | 239 | (56.9) |
| Male | 525 | (47.0) | 344 | (49.4) | 181 | (43.1) |
| Smoking history | ||||||
| Never smoker | 126 | (11.3) | 113 | (16.2) | 13 | (3.1) |
| Former/current smoker | 991 | (88.7) | 584 | (83.8) | 407 | (96.9) |
| Ethnicity | ||||||
| Scandinavian/European | 1106 | (99.0) | 686 | (98.4) | 420 | (100.0) |
| African | 4 | (0.4) | 4 | (0.6) | 0 | (0.0) |
| Asian | 7 | (0.6) | 7 | (1.0) | 0 | (0.0) |
| ECOG PS | ||||||
| 0–1 | 1037 | (92.8) | 650 | (93.3) | 387 | (92.1) |
| 2 | 59 | (5.3) | 35 | (5.0) | 24 | (5.7) |
| 3–4 | 21 | (1.9) | 12 | (1.7) | 9 | (2.1) |
| Histology | ||||||
| Adenocarcinoma | 1063 | (95.2) | 650 | (93.3) | 413 | (98.3) |
| Adenosquamous carcinoma | 6 | (0.5) | 6 | (0.9) | 0 | (0.0) |
| Large cell carcinoma | 3 | (0.3) | 3 | (0.4) | 0 | (0.0) |
| NSCLC-NOS | 36 | (3.2) | 31 | (4.4) | 5 | (1.2) |
| MIA | 2 | (0.2) | 2 | (0.3) | 0 | (0.0) |
| Other non-squamous NSCLC | 7 | (0.6) | 5 | (0.7) | 2 | (0.5) |
| EGFR status | ||||||
| No | 948 | (84.9) | 528 | (75.8) | 420 | (100.0) |
| Yes | 142 | (12.7) | 142 | (20.4) | 0 | (0.0) |
| Not assessed | 27 | (2.4) | 27 | (3.9) | 0 | (0.0) |
| ALK rearrangement | ||||||
| No | 1011 | (90.5) | 620 | (89.0) | 391 | (93.1) |
| Yes | 12 | (1.1) | 12 | (1.7) | 0 | (0.0) |
| Not assessed | 94 | (8.4) | 65 | (9.3) | 29 | (6.9) |
| ROS1 rearrangement | ||||||
| No | 552 | (49.4) | 342 | (49.1) | 210 | (50.0) |
| Yes | 3 | (.3) | 3 | (0.4) | 0 | (0.0) |
| Not assessed | 562 | (50.3) | 352 | (50.5) | 210 | (50.0) |
| Disease stage | ||||||
| IA | 227 | (20.3) | 141 | (20.2) | 86 | (20.5) |
| IB | 132 | (11.8) | 77 | (11.0) | 55 | (13.1) |
| IIA | 40 | (3.6) | 19 | (2.7) | 21 | (5.0) |
| IIB | 108 | (9.7) | 75 | (10.8) | 33 | (7.9) |
| IIIA | 138 | (12.4) | 83 | (11.9) | 55 | (13.1) |
| IIIB | 67 | (6.0) | 42 | (6.0) | 25 | (6.0) |
| IIIC | 25 | (2.2) | 16 | (2.3) | 9 | (2.1) |
| IVA | 206 | (18.4) | 140 | (20.1) | 66 | (15.7) |
| IVB | 174 | (15.6) | 104 | (14.9) | 70 | (16.7) |
| Treatment intention | ||||||
| Curative | 671 | (60.1) | 414 | (59.4) | 257 | (61.2) |
| Palliative | 400 | (35.8) | 251 | (36.0) | 149 | (35.5) |
| No treatment | 46 | (4.1) | 32 | (4.6) | 14 | (3.3) |
| Variable | PFS | OS | ||||||
|---|---|---|---|---|---|---|---|---|
| n | HR | 95% CI | p | n | HR | 95% CI | p | |
| Age (years) | ||||||||
| <50 | 46 | 1 (ref) | 46 | 1 (ref) | ||||
| 50–60 | 166 | 1.07 | 0.71–1.60) | 0.762 | 169 | 1.49 | (0.90–2.45) | 0.118 |
| >60 | 902 | 1.04 | (0.72–1.51) | 0.818 | 902 | 1.68 | (1.06–2.67) | 0.027 |
| Sex | ||||||||
| Women | 590 | 1 (ref) | 592 | 1 (ref) | ||||
| Men | 524 | 1.10 | (0.94–1.27) | 0.233 | 525 | 1.13 | (0.96–1.34) | 0.137 |
| Smoking history | ||||||||
| Never | 126 | 1 (ref) | 126 | 1 (ref) | ||||
| Former/current | 988 | 1.48 | (1.15–1.91) | 0.002 | 991 | 1.59 | (1.19–2.12) | 0.002 |
| ECOG PS | ||||||||
| 0–1 | 1034 | 1 (ref) | 1037 | 1 (ref) | ||||
| 2 | 59 | 4.30 | (3.24–5.71) | <0.001 | 59 | 6.41 | (4.80–8.56) | <0.001 |
| 3–4 | 21 | 4.55 | (2.76–7.48) | <0.001 | 21 | 6.91 | (4.18–11.41) | <0.001 |
| Stage | ||||||||
| I | 359 | 1 (ref) | 359 | 1 (ref) | ||||
| II | 148 | 2.15 | (1.61–2.86) | <0.001 | 148 | 2.11 | (1.51–2.95) | <0.001 |
| III | 230 | 4.37 | (3.43–5.57) | <0.001 | 230 | 3.89 | (2.93–5.15) | <0.001 |
| IV | 377 | 12.74 | (10.01–16.09) | <0.001 | 380 | 12.46 | (9.61–16.14) | <0.001 |
| Surgery | ||||||||
| No | 534 | 1 (ref) | 534 | 1 (ref) | ||||
| Yes | 580 | 0.15 | (0.12–0.17) | <0.001 | 583 | 0.13 | (0.11–0.16) | <0.001 |
| Curative RT +/− CT first line | ||||||||
| No | 1023 | 1 (ref) | 1023 | 1 (ref) | ||||
| Yes | 91 | 1.28 | (0.99–1.64) | 0.060 | 91 | 1.05 | (0.78–1.43) | 0.740 |
| Palliative CT and/or RT first line | ||||||||
| No | 843 | 1 (ref) | 846 | 1 (ref) | ||||
| Yes | 271 | 5.75 | (4.85–6.81) | <0.001 | 271 | 5.54 | ((4.64–6.63)) | <0.001 |
| History of TKI (any line) | ||||||||
| No | 1025 | 1 (ref) | 1028 | 1 (ref) | ||||
| Yes | 89 | 1.56 | (1.22–1.99) | <0.001 | 89 | 1.32 | (0.86–2.03) | 0.198 |
| History of ICI (any line) | ||||||||
| No | 939 | 1 (ref) | 941 | 1 (ref) | ||||
| Yes | 175 | 2.19 | (1.81–2.64) | <0.001 | 176 | 1.39 | ((1.11–1.74)) | 0.006 |
| KRAS status | ||||||||
| Wild type | 695 | (1 (ref) | 697 | 1 (ref) | ||||
| Mutated | 419 | 1.04 | (0.89–1.21) | 0.664 | 420 | 1.01 | (0.85–1.19) | 0.940 |
| KRAS G12C status | ||||||||
| Wild type | 695 | 1 (ref) | 697 | 1 (ref) | ||||
| G12C | 192 | 0.91 | (0.74–1.12) | 0.379 | 193 | 0.91 | (0.72–1.14) | 0.414 |
| KRAS non-G12C | 227 | 1.15 | (0.96–1.39) | 0.132 | 227 | 1.10 | (0.89–1.35) | 0.383 |
| Raf vs. PI3K preference | ||||||||
| Raf | 245 | 1 (ref) | 245 | 1 (ref) | ||||
| PI3K | 69 | 1.11 | (0.79–1.55) | 0.554 | 70 | 1.09 | (0.75–1.58) | 0.649 |
| Ral A/B vs. PI3K preference | ||||||||
| RalA/B | 192 | 1 (ref) | 192 | 1 (ref) | ||||
| PI3K | 69 | 1.20 | (0.85–1.70) | 0.312 | 70 | 1.19 | (0.81–1.74) | 0.385 |
| Type KRAS mutation | ||||||||
| G12A | 30 | 1 (ref) | 30 | 1 (ref) | ||||
| G12C | 192 | 0.62 | (0.38–1.00) | 0.050 | 192 | 0.59 | (0.35–0.99) | 0.044 |
| G12V | 81 | 0.72 | (0.43–1.21) | 0.220 | 81 | 0.60 | (0.34–1.06) | 0.080 |
| G12D | 69 | 0.74 | (0.44–1.26) | 0.274 | 70 | 0.69 | (0.39–1.24) | 0.216 |
| Variable | PFS | OS | PFS | OS | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HR | 95%CI | p | HR | 95%CI | p | HR | 95%CI | p | HR | 95%CI | p | |
| Age (years) | 1.01 | (1.00–1.01) | 0.207 | 1.02 | (1.01–1.03) | <0.001 | 1.01 | (1.00–1.01) | 0.214 | 1.02 | (1.01–1.03) | <0.001 |
| Sex | ||||||||||||
| Women | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| Men | 1.09 | (0.93–1.27) | 0.280 | 1.05 | (0.88–1.24) | 0.603 | 1.09 | (0.93–1.27) | 0.283 | 1.05 | (0.89–1.24) | 0.596 |
| Smoking history | ||||||||||||
| Never | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| Former/current | 1.56 | (1.19–2.05) | <0.001 | 1.71 | (1.24–2.33) | 0.001 | 1.56 | (1.19–2.05) | 0.001 | 1.70 | (1.25–2.33) | 0.001 |
| ECOG PS | ||||||||||||
| 0–1 | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| 2 | 2.02 | (1.51–2.71) | <0.001 | 2.90 | (2.15–3.92) | <0.001 | 2.01 | (1.50–2.70) | <0.001 | 2.91 | (2.16–3.94) | <0.001 |
| 3–4 | 4.27 | (2.56–7.14) | <0.001 | 6.51 | (3.83–11.05) | <0.001 | 4.25 | (2.54–7.11) | <0.001 | 6.56 | (3.86–11.14) | <0.001 |
| Stage | ||||||||||||
| I | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| II | 2.11 | (1.58–2.81) | <0.001 | 2.09 | (1.49–2.92) | <0.001 | 2.11 | (1.58–2.82) | <0.001 | 2.08 | (1.49–2.92) | <0.001 |
| III | 3.02 | (2.28–3.99) | <0.001 | 2.67 | (1.92–3.71) | <0.001 | 3.02 | (2.28–3.99) | <0.001 | 2.67 | (1.92–3.72) | <0.001 |
| IV | 4.61 | (3.18–6.67) | <0.001 | 4.34 | (2.91–6.47) | <0.001 | 4.61 | (3.19–6.68) | <0.001 | 4.34 | (2.91–6.47) | <0.001 |
| Surgery | ||||||||||||
| No | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| Yes | 0.36 | (0.25–0.52) | <0.001 | 0.25 | (0.11–0.37) | <0.001 | 0.36 | (0.25–0.52) | <0.001 | 0.25 | (0.17–0.37) | <0.001 |
| Curative RT +/− CT first line | ||||||||||||
| No | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| Yes | 0.74 | (0.51–1.09) | 0.127 | 0.47 | (0.31–0.73) | <0.001 | 0.74 | (0.51–1.09) | 0.130 | 0.47 | (0.31–0.73) | 0.001 |
| Palliative CT and/or RT first line | ||||||||||||
| No | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| Yes | 1.49 | (1.19–1.88) | <0.001 | 1.16 | (0.90–1.48) | 0.250 | 1.49 | (1.18–1.87) | 0.001 | 1.16 | (0.91–1.48) | 0.236 |
| History of TKI (any line) | ||||||||||||
| No | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| Yes | 0.78 | (0.58–1.03) | 0.081 | 0.65 | (0.47–0.90) | 0.009 | 0.78 | (0.58–1.03) | 0.081 | 0.65 | (0.47–0.90) | 0.009 |
| KRAS status | ||||||||||||
| Wild type | 1 (ref) | 1 (ref) | - | - | - | - | - | - | ||||
| Mutated | 0.98 | (0.83–1.15) | 0.801 | 0.96 | (0.80–1.15) | 0.678 | - | - | - | - | - | - |
| KRAS G12C status | ||||||||||||
| Wild type | - | - | - | - | - | - | 1 (ref) | 1 (ref) | ||||
| G12C | - | - | - | - | - | - | 0.96 | (0.77–1.19) | 0.691 | 1.00 | (0.79–1.28) | 0.972 |
| KRAS non-G12C | - | - | - | - | - | - | 1.00 | (0.82–1.21) | 0.977 | 0.93 | (0.75–1.16) | 0.530 |
| Variable | PFS | OS | ||||
|---|---|---|---|---|---|---|
| HR | 95%CI | p | HR | 95%CI | p | |
| Age at time of diagnosis | 1.00 | (0.99–1.02) | 0.820 | 1.01 | (0.99–1.03) | 0.142 |
| Sex | ||||||
| Women | 1 (ref) | 1/ref) | ||||
| Men | 1.16 | (0.89–1.52) | 0.283 | 1.01 | (0.75–1.37) | 0.940 |
| Smoking history | ||||||
| Never smoker | 1 (ref) | 1 (ref) | ||||
| Former/current smoker | 0.93 | (0.40–2.14) | 0.858 | 0.77 | (0.31–1.94) | 0.582 |
| ECOG PS | ||||||
| 0–1 | 1 (ref) | 1 (ref) | ||||
| 2 | 1.99 | (1.16–3.40) | 0.012 | 2.66 | (1.53–4.61) | 0.001 |
| 3–4 | 3.82 | (1.64–8.88) | 0.002 | 6.76 | (2.86–16.00) | 0.000 |
| Stage | ||||||
| I | 1 (ref) | 1 (ref) | ||||
| II | 1.66 | (1.01–2.74) | 0.047 | 1.45 | (0.80–2.60) | 0.218 |
| III | 2.69 | (1.66–4.35) | 0.000 | 2.02 | (1.13–3.60) | 0.018 |
| IV | 2.49 | (1.29–4.82) | 0.007 | 2.06 | (0.99–4.29) | 0.054 |
| Surgery | ||||||
| No | 1 (ref) | 1 (ref) | ||||
| Yes | 0.24 | (0.13–0.46) | 0.000 | 0.17 | (0.08–0.34) | 0.000 |
| Curative RT +/− CT first line | ||||||
| No | 1 (ref) | 1 (ref) | ||||
| Yes | 0.53 | (0.27–1.03) | 0.062 | 0.26 | (0.11–0.59) | 0.001 |
| Palliative CT and/or RT first line | ||||||
| No | 1 (ref) | 1 (ref) | ||||
| Yes | 1.53 | (1.01–2.33) | 0.046 | 1.26 | (0.80–1.99) | 0.314 |
| KRAS mutation | ||||||
| G12A | 1 (ref) | 1 (ref) | ||||
| G12C | 0.76 | (0.45–1.26) | 0.281 | 0.97 | (0.54–1.73) | 0.907 |
| G12V | 0.79 | (0.46–1.37) | 0.399 | 0.83 | (0.44–1.56) | 0.561 |
| G12D | 0.76 | (0.44–1.33) | 0.333 | 0.87 | (0.46–1.64) | 0.659 |
| Variable | PFS | OS | PFS | OS | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HR | 95%CI | p | HR | 95%CI | p | HR | 95%CI | p | HR | 95%CI | p | |
| Age (years) | 1.02 | (1.00–1.03) | 0.070 | 1.02 | (1.00–1.04) | 0.027 | 1.02 | (1.00–1.04) | 0.085 | 1.01 | (0.99–1.03) | 0.200 |
| Sex | ||||||||||||
| Women | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| Men | 1.00 | (0.75–1.35) | 0.981 | 0.88 | (0.63–1.22) | 0.431 | 0.90 | (0.64–1.25) | 0.529 | 0.69 | (0.47–1.01) | 0.054 |
| Smoking history | ||||||||||||
| Never | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| Former/current | 1.02 | (0.36–2.88) | 0.975 | 0.93 | (0.28–3.09) | 0.911 | 1.31 | (0.40–4.33) | 0.653 | 1.34 | (0.31–5.67) | 0.695 |
| ECOG PS | ||||||||||||
| 0–1 | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| 2 | 2.23 | (1.32–3.76) | 0.003 | 3.03 | (1.79–5.14) | <0.001 | 2.77 | (1.46–5.23) | 0.002 | 4.89 | (2.49–9.58) | <0.001 |
| 3–4 | 6.68 | (2.89–15.5) | <0.001 | 9.21 | (3.85–22.02) | <0.001 | 6.02 | (2.41–14.99) | <0.001 | 10.95 | (4.18–28.68) | <0.001 |
| Stage | ||||||||||||
| I | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| II | 1.55 | (0.88–2.71) | 0.128 | 1.52 | (0.79–2.94) | 0.212 | 1.35 | (0.71–2.58) | 0.363 | 1.03 | (0.47–2.26) | 0.946 |
| III | 2.61 | (1.49–4.58) | 0.001 | 2.71 | (1.45–5.06) | 0.002 | 3.21 | (1.77–5.85) | <0.001 | 2.61 | (1.30–5.25) | 0.007 |
| IV | 2.74 | (1.36–5.52) | 0.005 | 3.08 | (1.50–6.32) | 0.002 | 3.24 | (1.46–7.19) | 0.004 | 1.94 | (0.80–4.71) | 0.143 |
| Surgery | ||||||||||||
| No | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| Yes | 0.234 | (0.11–0.48) | <0.001 | 0.22 | (0.1–0.48) | <0.001 | 0.28 | (0.13–0.62) | 0.002 | 0.16 | (0.07–0.39) | <0.001 |
| Curative RT +/− CT first line | ||||||||||||
| No | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| Yes | 0.617 | (0.30–1.27) | 0.189 | 0.35 | (0.14–0.83) | 0.017 | 0.57 | (0.26–1.28) | 0.173 | 0.22 | (0.08–0.58) | 0.002 |
| Palliative CT and/or RT first line | ||||||||||||
| No | 1 (ref) | 1 (ref) | 1 (ref) | 1 (ref) | ||||||||
| Yes | 1.56 | (1.00–2.44) | 0.052 | 1.27 | (0.77–2.09) | 0.350 | 1.15 | (0.94–2.48) | 0.089 | 1.32 | (0.78–2.24) | 0.302 |
| PI3K vs. Raf | ||||||||||||
| Raf | 1 (ref) | 1 (ref) | - | - | - | - | - | - | ||||
| PI3K | 0.903 | (0.64–1.28) | 0.570 | 0.89 | (0.60–1.30) | 0.530 | - | - | - | - | - | - |
| PI3K vs. RalA/B | ||||||||||||
| RalA/B | - | - | - | - | - | - | 1 (ref) | 1 (ref) | ||||
| PI3K | - | - | - | - | - | - | 0.94 | (0.65–1.35) | 0.720 | 0.86 | (0.58–1.29) | 0.474 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wahl, S.G.F.; Dai, H.Y.; Emdal, E.F.; Berg, T.; Halvorsen, T.O.; Ottestad, A.L.; Lund-Iversen, M.; Brustugun, O.T.; Førde, D.; Paulsen, E.-E.; et al. The Prognostic Effect of KRAS Mutations in Non-Small Cell Lung Carcinoma Revisited: A Norwegian Multicentre Study. Cancers 2021, 13, 4294. https://doi.org/10.3390/cancers13174294
Wahl SGF, Dai HY, Emdal EF, Berg T, Halvorsen TO, Ottestad AL, Lund-Iversen M, Brustugun OT, Førde D, Paulsen E-E, et al. The Prognostic Effect of KRAS Mutations in Non-Small Cell Lung Carcinoma Revisited: A Norwegian Multicentre Study. Cancers. 2021; 13(17):4294. https://doi.org/10.3390/cancers13174294
Chicago/Turabian StyleWahl, Sissel Gyrid Freim, Hong Yan Dai, Elisabeth Fritzke Emdal, Thomas Berg, Tarje Onsøien Halvorsen, Anine Larsen Ottestad, Marius Lund-Iversen, Odd Terje Brustugun, Dagny Førde, Erna-Elise Paulsen, and et al. 2021. "The Prognostic Effect of KRAS Mutations in Non-Small Cell Lung Carcinoma Revisited: A Norwegian Multicentre Study" Cancers 13, no. 17: 4294. https://doi.org/10.3390/cancers13174294
APA StyleWahl, S. G. F., Dai, H. Y., Emdal, E. F., Berg, T., Halvorsen, T. O., Ottestad, A. L., Lund-Iversen, M., Brustugun, O. T., Førde, D., Paulsen, E.-E., Donnem, T., Andersen, S., Grønberg, B. H., & Richardsen, E. (2021). The Prognostic Effect of KRAS Mutations in Non-Small Cell Lung Carcinoma Revisited: A Norwegian Multicentre Study. Cancers, 13(17), 4294. https://doi.org/10.3390/cancers13174294






