Prognostic Utility of Platelet–Lymphocyte Ratio, Neutrophil–Lymphocyte Ratio and Monocyte–Lymphocyte Ratio in Head and Neck Cancers: A Detailed PRISMA Compliant Systematic Review and Meta-Analysis
Abstract
Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
2.2. Study Selection
2.3. Inclusion Criteria
- The studies must discuss the survival outcome of HNC cancer patients based on PLR, NLR and MLR levels.
- The survival outcome must be presented in the form of HR (hazard ratios) and 95% CI (confidence intervals).
- The survival outcome must be presented in the form of Kaplan–Meier curves, along with patient cohort information, for each treatment arm represented in the KM Curves. (This is required only if the HR and 95% CI values have not been presented in the manuscript, as the above information is required to extract approximated HR values.)
2.4. Exclusion Criteria
- Conference abstracts, reviews, letters to the editor and other non-clinical literature will not be considered for either systematic review or meta-analysis.
- Included studies must be clinical studies or involve patient samples. (In vitro, in silico and animal studies will be excluded.)
- Unpublished or non-peer-reviewed literature will be excluded.
- Studies that do not focus on survival outcomes and prognosis aspects of PLR, NLR and MLR in cancer patients will not be considered.
- If the sample size of each individual study is of low power (sample size < 10), they will be excluded.
2.5. Data Extraction and Recording
- Author names;
- Year of publication;
- Marker studied (PLR, NLR or MLR);
- Size of patient cohort;
- Diagnostic methods;
- Follow-up period;
- Gender split of cohort;
- TNM staging split of cohort;
- Survival endpoint of each study (overall survival, disease-free survival, disease-specific survival);
- General features of each study (will be presented as short qualitative opinions/observations of reviewers, for each study being included).
2.6. Quality Assessment
2.7. Meta-Analysis
2.8. Assessment of Heterogeneity
2.9. Subgroup Analysis
2.10. Publication Bias
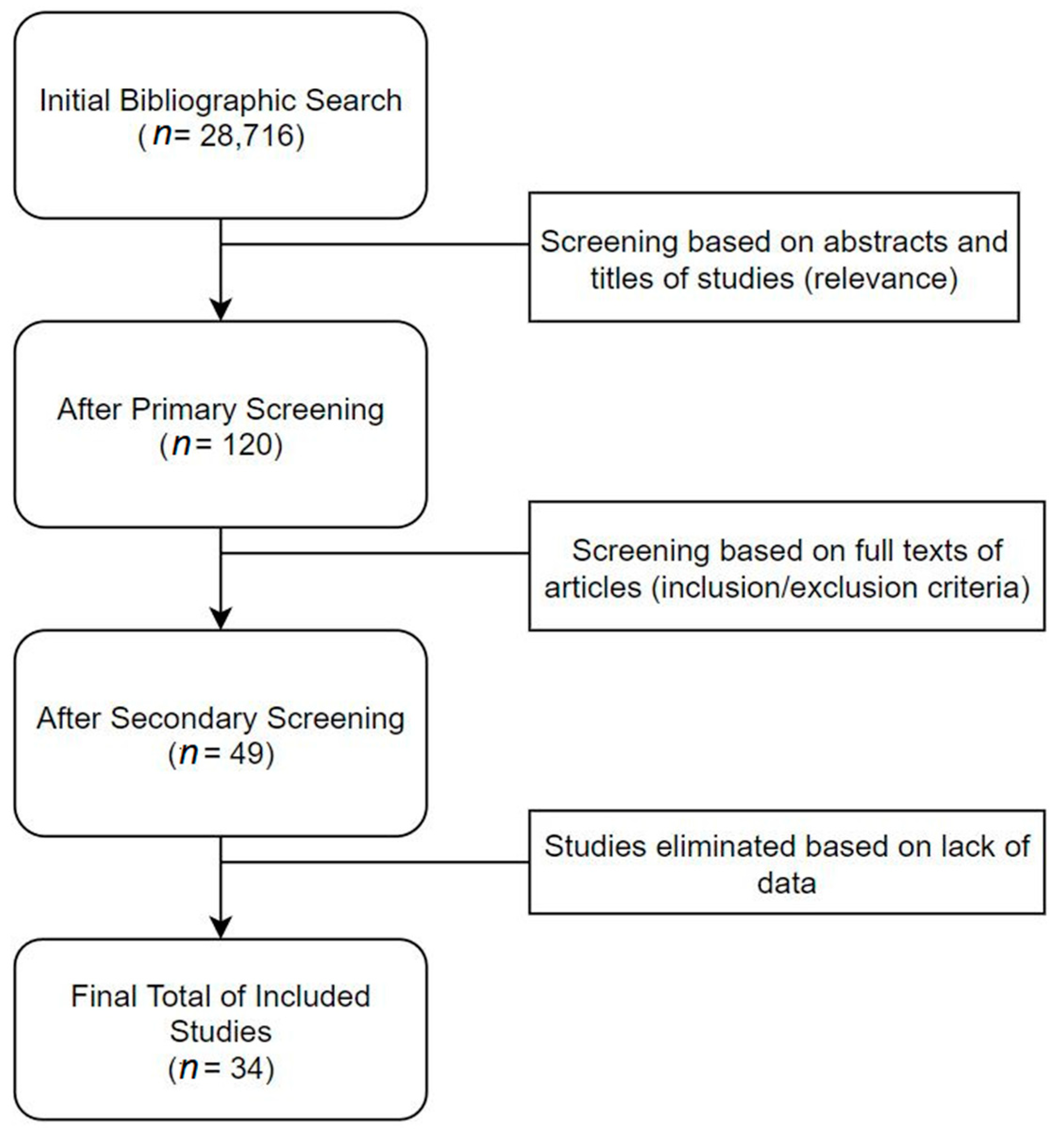
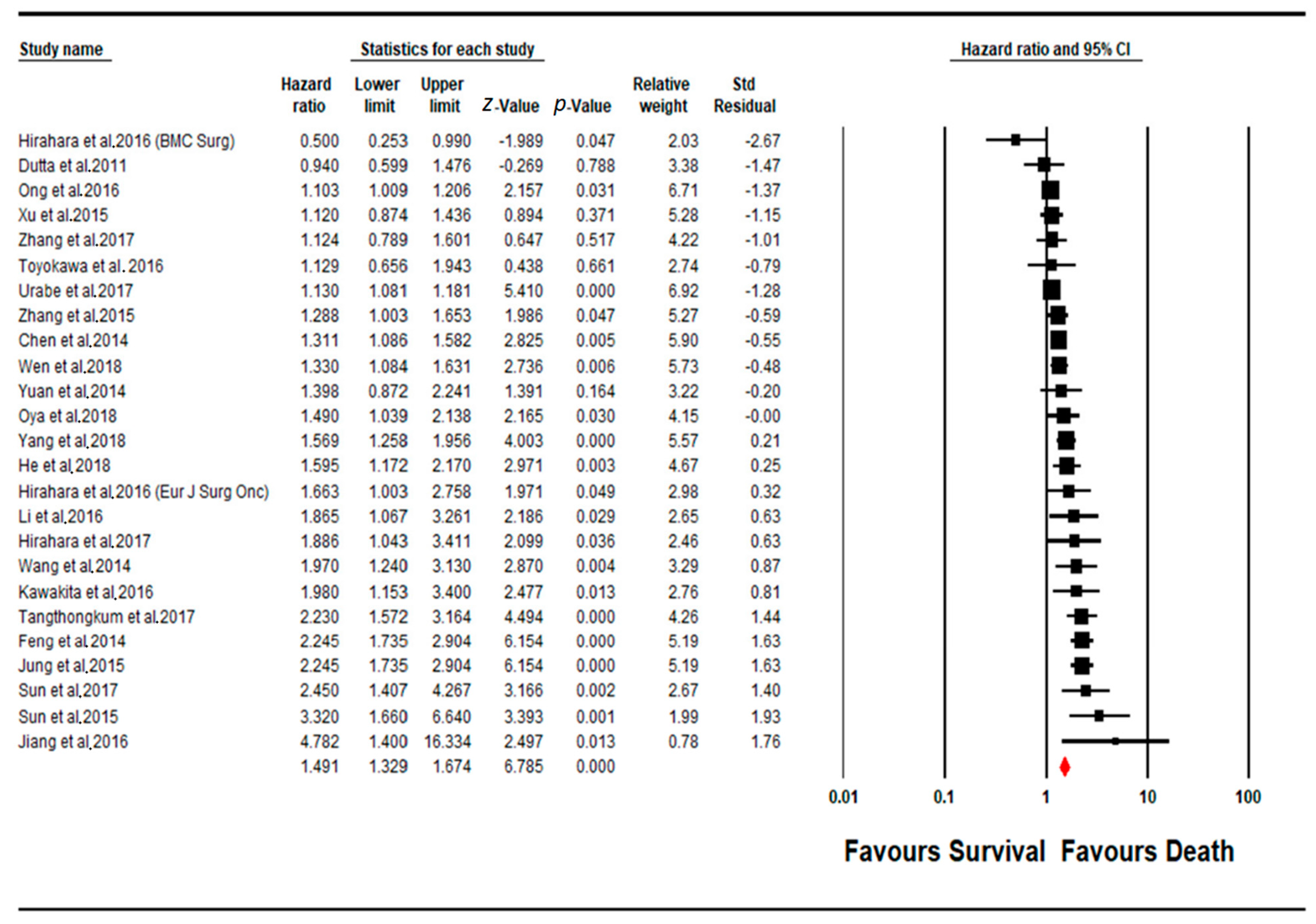
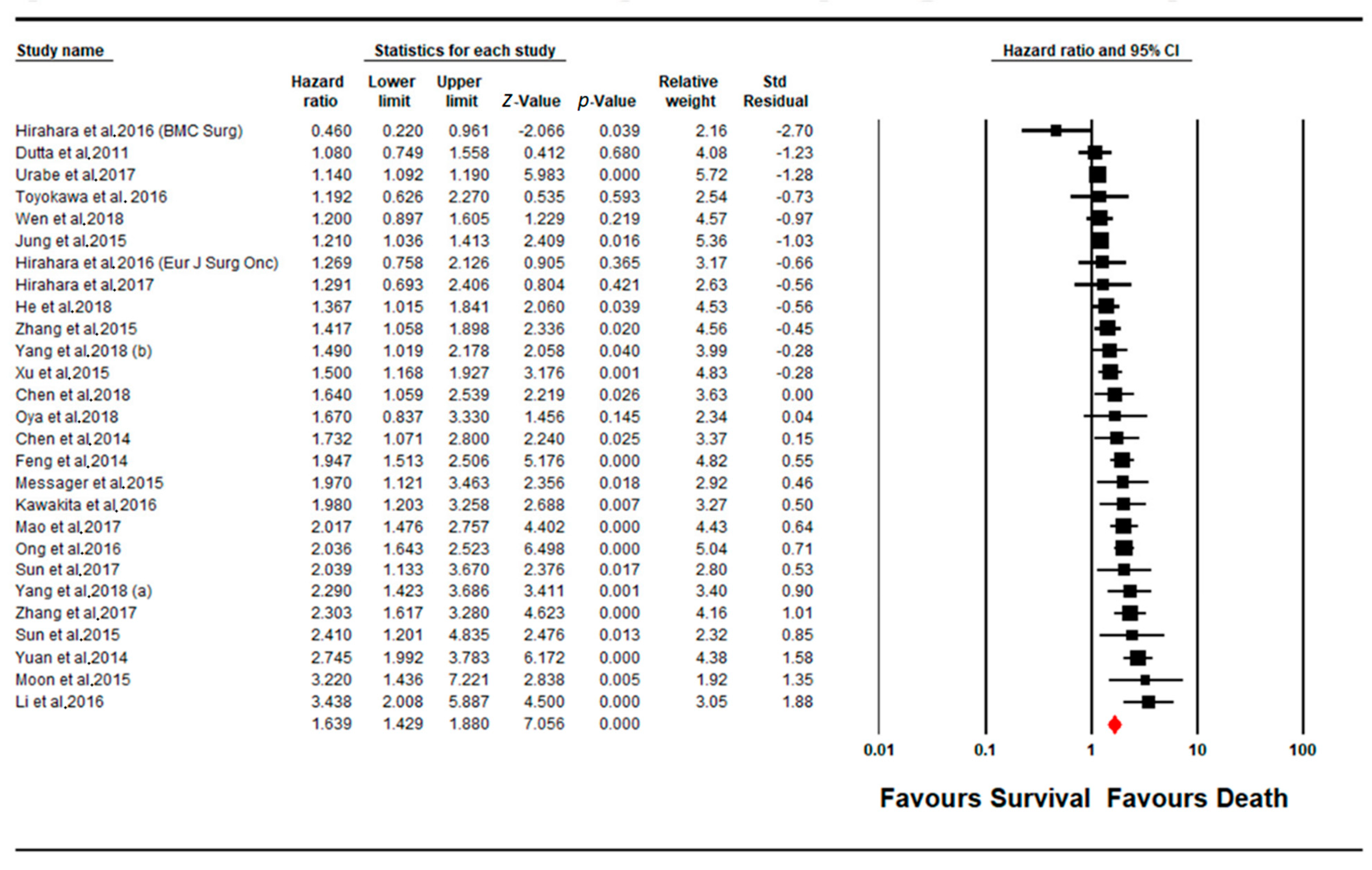
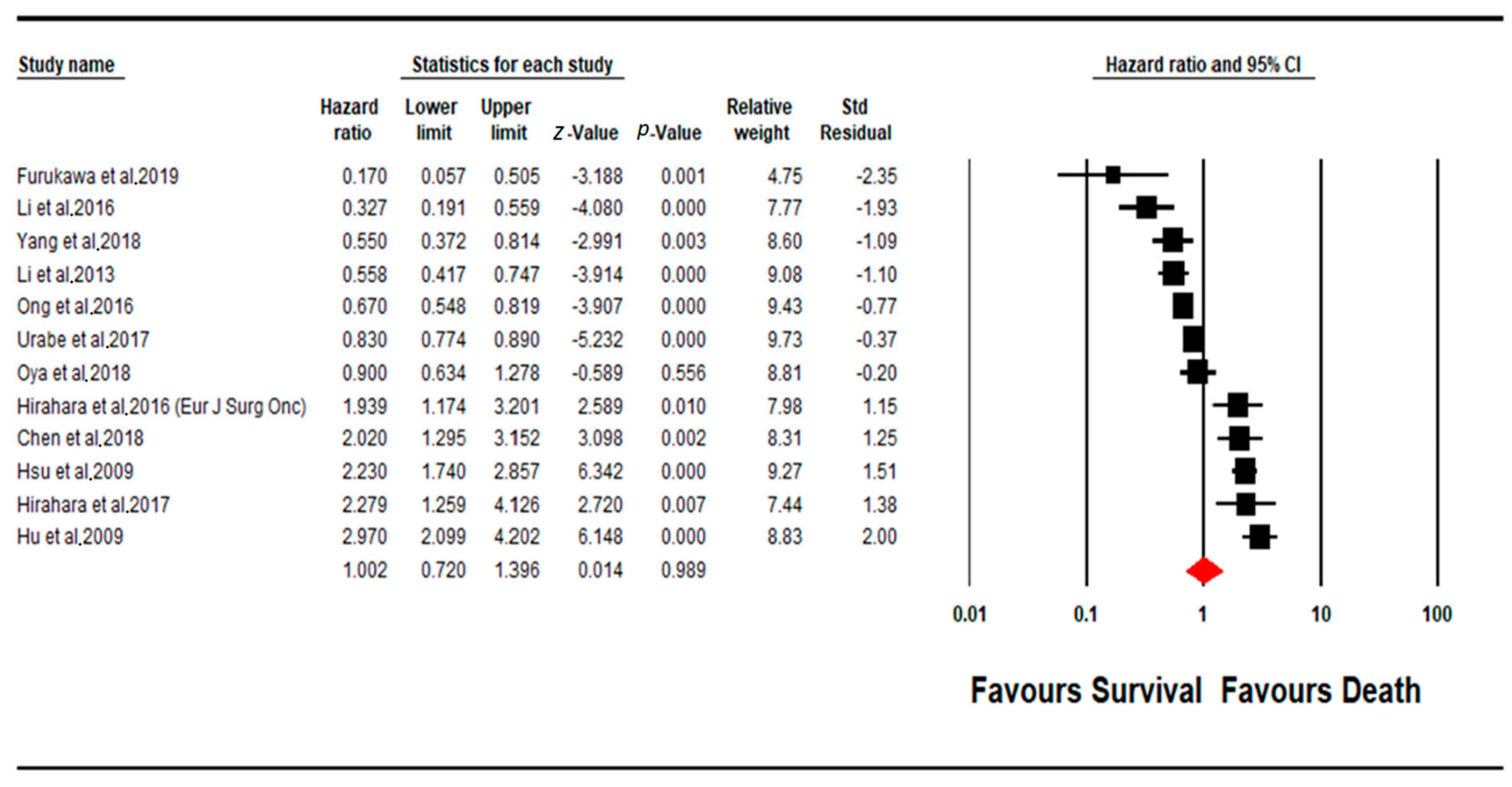
3. Results
4. Meta-Analysis
4.1. Meta-Analysis PLR Subgroup
4.2. Meta-Analysis NLR Subgroup
4.3. Meta-Analysis MLR Subgroup
4.4. Publication Bias
4.5. Quality Assessment
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, M.; Zhao, J.; Zhang, L.; Wei, F.; Lian, Y.; Wu, Y.; Gong, Z.; Zhang, S.; Zhou, J.; Cao, K. Role of tumor microenvironment in tumorigenesis. J. Cancer 2017, 8, 761. [Google Scholar] [CrossRef]
- Colotta, F.; Allavena, P.; Sica, A.; Garlanda, C.; Mantovani, A. Cancer-related inflammation, the seventh hallmark of cancer: Links to genetic instability. Carcinogenesis 2009, 30, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Wang, W.J.; Zhi, Q.; Shen, M.; Jiang, M.; Bian, X.; Gong, F.R.; Zhou, C.; Lian, L.; Wu, M.Y.; et al. Neutrophil/lymphocyte ratio is a more sensitive systemic inflammatory response biomarker than platelet/lymphocyte ratio in the prognosis evaluation of unresectable pancreatic cancer. Oncotarget 2017, 8, 88835–88844. [Google Scholar] [CrossRef] [PubMed]
- Akinci Ozyurek, B.; Sahin Ozdemirel, T.; Buyukyaylaci Ozden, S.; Erdogan, Y.; Kaplan, B.; Kaplan, T. Prognostic Value of the Neutrophil to Lymphocyte Ratio (NLR) in Lung Cancer Cases. Asian Pac. J. Cancer Prev. 2017, 18, 1417–1421. [Google Scholar] [CrossRef] [PubMed]
- Krenn-Pilko, S.; Langsenlehner, U.; Thurner, E.M.; Stojakovic, T.; Pichler, M.; Gerger, A.; Kapp, K.S.; Langsenlehner, T. The elevated preoperative platelet-to-lymphocyte ratio predicts poor prognosis in breast cancer patients. Br. J. Cancer 2014, 110, 2524. [Google Scholar] [CrossRef]
- Yan, M.; Jurasz, P. The role of platelets in the tumor microenvironment: From solid tumors to leukemia. Biochim. Biophys. Acta BBA Mol. Cell Res. 2016, 1863, 392–400. [Google Scholar] [CrossRef]
- Shi, C.; Pamer, E.G. Monocyte recruitment during infection and inflammation. Nat. Reviews. Immunol. 2011, 11, 762–774. [Google Scholar] [CrossRef]
- McMillan, D.C. An inflammation-based prognostic score and its role in the nutrition-based management of patients with cancer: Nutrition Society and BAPEN Medical Symposium on ‘Nutrition support in cancer therapy’. Proc. Nutr. Soc. 2008, 67, 257–262. [Google Scholar] [CrossRef]
- Fearon, K.C.H. Cancer cachexia: Developing multimodal therapy for a multidimensional problem. Eur. J. Cancer 2008, 44, 1124–1132. [Google Scholar] [CrossRef]
- Pardoll, D.M.; Topalian, S.L. The role of CD4+ T cell responses in antitumor immunity. Curr. Opin. Immunol. 1998, 10, 588–594. [Google Scholar] [CrossRef]
- Hung, K.; Hayashi, R.; Lafond-Walker, A.; Lowenstein, C.; Pardoll, D.; Levitsky, H. The central role of CD4+ T cells in the antitumor immune response. J. Exp. Med. 1998, 188, 2357–2368. [Google Scholar] [CrossRef]
- Chanmee, T.; Ontong, P.; Konno, K.; Itano, N. Tumor-associated macrophages as major players in the tumor microenvironment. Cancers 2014, 6, 1670–1690. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Qin, X.; Wang, H.; Xia, Y.; Li, Y.; Chen, X.; Shang, L.; Tai, Y.T.; Feng, X.; Acharya, P.; et al. Elevated neutrophil-to-lymphocyte ratio and monocyte-to-lymphocyte ratio and decreased platelet-to-lymphocyte ratio are associated with poor prognosis in multiple myeloma. Oncotarget 2017, 8, 18792–18801. [Google Scholar] [CrossRef]
- Sun, X.; Cui, F.; Yin, H.; Wu, D.; Wang, N.; Yuan, M.; Fei, Y.; Wang, Q. Association between EGFR mutation and expression of BRCA1 and RAP80 in non-small cell lung cancer. Oncol. Lett. 2018, 16, 2201–2206. [Google Scholar] [CrossRef]
- Rosen, E.M.; Fan, S.; Pestell, R.G.; Goldberg, I.D. BRCA1 gene in breast cancer. J. Cell. Physiol. 2003, 196, 19–41. [Google Scholar] [CrossRef] [PubMed]
- Brennan, D.J.; Gallagher, W.M. Prognostic ability of a panel of immunohistochemistry markers—retailoring of an ‘old solution’. Breast Cancer Res. BCR 2008, 10, 102. [Google Scholar] [CrossRef] [PubMed]
- Mocellin, S.; Rossi, C.R.; Pilati, P.; Nitti, D.; Marincola, F.M. Quantitative real-time PCR: A powerful ally in cancer research. Trends Mol. Med. 2003, 9, 189–195. [Google Scholar] [CrossRef]
- Ying, H.-Q.; Deng, Q.-W.; He, B.-S.; Pan, Y.-Q.; Wang, F.; Sun, H.-L.; Chen, J.; Liu, X.; Wang, S.-K. The prognostic value of preoperative NLR, d-NLR, PLR and LMR for predicting clinical outcome in surgical colorectal cancer patients. Med Oncol. 2014, 31, 305. [Google Scholar] [CrossRef]
- Chen, L.; Zeng, H.; Yang, J.; Lu, Y.; Zhang, D.; Wang, J.; Kuang, C.; Zhu, S.; Wang, M.; Ma, X. Survival and prognostic analysis of preoperative inflammatory markers in patients undergoing surgical resection for laryngeal squamous cell carcinoma. BMC Cancer 2018, 18, 816. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, H.Y.; Li, J.; Shao, X.Y.; Zhang, C.X. The elevated NLR, PLR and PLT may predict the prognosis of patients with colorectal cancer: A systematic review and meta-analysis. Oncotarget 2017, 8, 68837–68846. [Google Scholar] [CrossRef]
- Solmaz, S.; Uzun, O.; Acar, C.; Sevindik, O.G.; Piskin, O.; Ozsan, H.G.; Demirkan, F.; Undar, B.; Alacacioglu, A.; Ozcan, M.A. Is the platelet-to-lymphocyte ratio a new prognostic marker in multiple myeloma? J. Lab. Physicians 2018, 10, 363. [Google Scholar] [CrossRef]
- Mellor, K.L.; Powell, A.G.M.T.; Lewis, W.G. Systematic Review and Meta-Analysis of the Prognostic Significance of Neutrophil-Lymphocyte Ratio (NLR) After R0 Gastrectomy for Cancer. J. Gastrointest. Cancer 2018, 49, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Zhou, S.; Liu, Y.; Zhai, L.; Sun, X. Prognostic value of systemic inflammatory markers in ovarian Cancer: A PRISMA-compliant meta-analysis and systematic review. BMC Cancer 2018, 18, 443. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef]
- Kumarasamy, C.; Sabarimurugan, S.; Madurantakam, R.M.; Lakhotiya, K.; Samiappan, S.; Baxi, S.; Nachimuthu, R.; Gothandam, K.M.; Jayaraj, R. Prognostic significance of blood inflammatory biomarkers NLR, PLR, and LMR in cancer—A protocol for systematic review and meta-analysis. Medicine 2019, 98, e14834. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.A.; Shea, B.; O’connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses; Ottawa Hospital Research Institute: Ottawa, ON, Canada, 2009. [Google Scholar]
- Jayaraj, R.; Kumarasamy, C.; Madhav, M.R.; Pandey, V.; Sabarimurugan, S.; Ramesh, N.; Gothandam, K.M.; Baxi, S. Comment on “Systematic Review and Meta-Analysis of Diagnostic Accuracy of miRNAs in Patients with Pancreatic Cancer”. Dis. Markers 2018, 2018, 2. [Google Scholar] [CrossRef]
- Jayaraj, R.; Kumarasamy, C. Comment on ‘Prognostic biomarkers for oral tongue squamous cell carcinoma: A systematic review and meta-analysis’. Br. J. Cancer 2018, 118, e11. [Google Scholar] [CrossRef] [PubMed]
- Jayaraj, R.; Kumarasamy, C.J.O.O. Comment on, “Survival for HPV-positive oropharyngeal squamous cell carcinoma with surgical versus non-surgical treatment approach: A systematic review and meta-analysis”. Oral. Oncol. 2018, 90, 137–138. [Google Scholar] [CrossRef]
- Jayaraj, R.; Kumarasamy, C.; Sabarimurugan, S.; Baxi, S.J.F.i.P. Commentary: Blood-Derived microRNAs for Pancreatic Cancer Diagnosis: A Narrative Review and Meta-Analysis. Front. Physiol. 2018, 9, 1896. [Google Scholar] [CrossRef]
- Jayaraj, R.; Kumarasamy, C.J.O.O. Conceptual interpretation of analysing and reporting of results on systematic review and meta-analysis of optimal extent of lateral neck dissection for well-differentiated thyroid carcinoma with metastatic lateral neck lymph nodes. Oral. Oncol. 2019, 89, 153–154. [Google Scholar] [CrossRef]
- Borenstein, M.; Rothstein, H. Comprehensive Meta-Analysis; Biostat, 1999; Available online: https://www.meta-analysis.com/downloads/Meta-Analysis%20Manual%20V3.pdf (accessed on 17 August 2021).
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.T.; Rothstein, H.R. A basic introduction to fixed-effect and random-effects models for meta-analysis. Res. Synth. Methods 2010, 1, 97–111. [Google Scholar] [CrossRef]
- Jayaraj, R.; Kumarasamy, C.; Gothandam, K.M. Letter to the editor “Prognostic value of microRNAs in colorectal cancer: A meta-analysis”. Cancer Manag. Res. 2018, 10, 3501–3503. [Google Scholar] [CrossRef] [PubMed]
- Jayaraj, R.; Kumarasamy, C.J.O.o. Letter to the Editor about the Article: “Performance of different imaging techniques in the diagnosis of head and neck cancer mandibular invasion: A systematic review and meta-analysis”. Oral. Oncol. 2018, 89, 159–160. [Google Scholar] [CrossRef]
- Jayaraj, R.; Kumarasamy, C.; Sabarimurugan, S.; Baxi, S. Letter to the Editor in response to the article,” The epidemiology of oral human papillomavirus infection in healthy populations: A systematic review and meta-analysis”. Oral. Oncol. 2018, 84, 121–122. [Google Scholar] [CrossRef] [PubMed]
- Jayaraj, R.; Kumarasamy, C.; Samiappan, S.; Swaminathan, P. Letter to the Editor regarding, “The prognostic role of PD-L1 expression for survival in head and neck squamous cell carcinoma: A systematic review and meta-analysis”. Oral. Oncol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Jayaraj, R.; Kumarasamy, C.; Madurantakam Royam, M.; Devi, A.; Baxi, S. Letter to the editor: Is HIF-1α a viable prognostic indicator in OSCC? A critical review of a meta-analysis study. World J. Surg. Oncol. 2018, 16, 111. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ Br. Med. J. 2003, 327, 557. [Google Scholar] [CrossRef]
- von Hippel, P.T. The heterogeneity statistic I (2) can be biased in small meta-analyses. BMC Med. Res. Methodol. 2015, 15, 35. [Google Scholar] [CrossRef] [PubMed]
- Huedo-Medina, T.B.; Sánchez-Meca, J.; Marín-Martínez, F.; Botella, J. Assessing heterogeneity in meta-analysis: Q statistic or I2 index? Psychol. Methods 2006, 11, 193. [Google Scholar] [CrossRef] [PubMed]
- Deeks, J.J.; Higgins, J.P.T.; Altman, D.G. Analysing data and undertaking meta-analyses. In Cochrane Handbook for Systematic Reviews of Interventions; Cochrane Book Series; Wiley: Hoboken, NJ, USA, 2008; pp. 243–296. [Google Scholar]
- Egger, M.; Smith, G.D.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Orwin, R.G. A fail-safe N for effect size in meta-analysis. J. Educ. Stat. 1983, 8, 157–159. [Google Scholar] [CrossRef]
- Begg, C.B.; Mazumdar, M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994, 50, 1088–1101. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Bedford, M.; Begum, R.; Mitchell, H.; Hodson, J.; Whiting, J.; Griffiths, E. The value of inflammation based prognostic scores in patients undergoing surgical resection for oesophageal and gastric carcinoma. J. Surg. Oncol. 2018, 117, 1697–1707. [Google Scholar] [CrossRef] [PubMed]
- Kawakita, D.; Tada, Y.; Imanishi, Y.; Beppu, S.; Tsukahara, K.; Kano, S.; Ozawa, H.; Okami, K.; Sato, Y.; Shimizu, A. Impact of hematological inflammatory markers on clinical outcome in patients with salivary duct carcinoma: A multi-institutional study in Japan. Oncotarget 2017, 8, 1083. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Sun, P.; Dai, Q.-s.; Weng, H.-w.; Li, H.-p.; Ye, S. The Glasgow Prognostic Score predicts poor survival in cisplatin-based treated patients with metastatic nasopharyngeal carcinoma. PLoS ONE 2014, 9, e112581. [Google Scholar] [CrossRef]
- Zhang, F.; Chen, Z.; Wang, P.; Hu, X.; Gao, Y.; He, J. Combination of platelet count and mean platelet volume (COP-MPV) predicts postoperative prognosis in both resectable early and advanced stage esophageal squamous cell cancer patients. Tumor Biol. 2016, 37, 9323–9331. [Google Scholar] [CrossRef]
- Jiang, K.; Lei, J.; Li, C.; Shu, K.; Li, W.; Zhang, Y.; Li, Z.; Gong, R.; Zhu, J. Comparison of the prognostic values of selected inflammation based scores in patients with medullary thyroid carcinoma: A pilot study. J. Surg. Oncol. 2017, 116, 281–287. [Google Scholar] [CrossRef]
- Li, X.H.; Chang, H.; Xu, B.Q.; Tao, Y.L.; Gao, J.; Chen, C.; Qu, C.; Zhou, S.; Liu, S.R.; Wang, X.H. An inflammatory biomarker-based nomogram to predict prognosis of patients with nasopharyngeal carcinoma: An analysis of a prospective study. Cancer Med. 2017, 6, 310–319. [Google Scholar] [CrossRef]
- Feng, J.-F.; Huang, Y.; Chen, Q.-X. Preoperative platelet lymphocyte ratio (PLR) is superior to neutrophil lymphocyte ratio (NLR) as a predictive factor in patients with esophageal squamous cell carcinoma. World J. Surg. Oncol. 2014, 12, 1–6. [Google Scholar] [CrossRef]
- Jiang, R.; Zou, X.; Hu, W.; Fan, Y.-Y.; Yan, Y.; Zhang, M.-X.; You, R.; Sun, R.; Luo, D.-H.; Chen, Q.-Y. The elevated pretreatment platelet-to-lymphocyte ratio predicts poor outcome in nasopharyngeal carcinoma patients. Tumor Biol. 2015, 36, 7775–7787. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Park, S.Y.; Park, S.-j.; Park, J. Prognostic value of the neutrophil-to-lymphocyte ratio for overall and disease-free survival in patients with surgically treated esophageal squamous cell carcinoma. Tumor Biol. 2016, 37, 7149–7154. [Google Scholar] [CrossRef] [PubMed]
- He, Y.F.; Luo, H.Q.; Wang, W.; Chen, J.; Yao, Y.W.; Yan, Y.; Wu, S.S.; Hu, X.X.; Ke, L.H.; Niu, J.Y. Preoperative NLR and PLR in the middle or lower ESCC patients with radical operation. Eur. J. Cancer Care 2017, 26, e12445. [Google Scholar] [CrossRef] [PubMed]
- Jiang, K.; Lei, J.; Chen, W.; Gong, Y.; Luo, H.; Li, Z.; Gong, R.; Zhu, J. Association of the preoperative neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios with lymph node metastasis and recurrence in patients with medullary thyroid carcinoma. Medicine 2016, 95, e5079. [Google Scholar] [CrossRef] [PubMed]
- Hirahara, N.; Tajima, Y.; Fujii, Y.; Yamamoto, T.; Hyakudomi, R.; Hirayama, T.; Taniura, T.; Ishitobi, K.; Kidani, A.; Kawabata, Y. A novel prognostic scoring system using inflammatory response biomarkers for esophageal squamous cell carcinoma. World J. Surg. 2018, 42, 172–184. [Google Scholar] [CrossRef]
- Ong, H.S.; Gokavarapu, S.; Wang, L.Z.; Tian, Z.; Zhang, C.P. Low pretreatment lymphocyte-monocyte ratio and high platelet-lymphocyte ratio indicate poor cancer outcome in early tongue cancer. Int. J. Oral Maxillofac. Surg. 2017, 75, 1762–1774. [Google Scholar] [CrossRef]
- Mao, Y.; Fu, Y.; Gao, Y.; Yang, A.; Zhang, Q. Platelet-to-lymphocyte ratio predicts long-term survival in laryngeal cancer. Eur. Arch. Oto-Rhino-Laryngol. 2018, 275, 553–559. [Google Scholar] [CrossRef]
- Dutta, S.; Crumley, A.B.C.; Fullarton, G.M.; Horgan, P.G.; McMillan, D.C. Comparison of the prognostic value of tumour and patient related factors in patients undergoing potentially curative resection of gastric cancer. Am. J. Surg. 2012, 204, 294–299. [Google Scholar] [CrossRef]
- Li, J.-P.; Chen, S.-L.; Liu, X.-M.; He, X.; Xing, S.; Liu, Y.-J.; Lin, Y.-H.; Liu, W.-L. A novel inflammation-based stage (I Stage) predicts overall survival of patients with nasopharyngeal carcinoma. Int. J. Mol. Sci. 2016, 17, 1900. [Google Scholar] [CrossRef]
- Messager, M.; Neofytou, K.; Chaudry, M.A.; Allum, W.H. Prognostic impact of preoperative platelets to lymphocytes ratio (PLR) on survival for oesophageal and junctional carcinoma treated with neoadjuvant chemotherapy: A retrospective monocentric study on 153 patients. Eur. J. Surg. Oncol. 2015, 41, 1316–1323. [Google Scholar] [CrossRef]
- Hirahara, N.; Matsubara, T.; Kawahara, D.; Nakada, S.; Ishibashi, S.; Tajima, Y. Prognostic significance of preoperative inflammatory response biomarkers in patients undergoing curative thoracoscopic esophagectomy for esophageal squamous cell carcinoma. Eur. J. Surg. Oncol. 2017, 43, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.; Roh, J.-L.; Lee, S.-w.; Kim, S.-B.; Choi, S.-H.; Nam, S.Y.; Kim, S.Y. Prognostic value of nutritional and hematologic markers in head and neck squamous cell carcinoma treated by chemoradiotherapy. Radiother. Oncol. 2016, 118, 330–334. [Google Scholar] [CrossRef]
- Hirahara, N.; Matsubara, T.; Mizota, Y.; Ishibashi, S.; Tajima, Y. Prognostic value of preoperative inflammatory response biomarkers in patients with esophageal cancer who undergo a curative thoracoscopic esophagectomy. BMC Surg. 2016, 16, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Turri–Zanoni, M.; Salzano, G.; Lambertoni, A.; Giovannardi, M.; Karligkiotis, A.; Battaglia, P.; Castelnuovo, P. Prognostic value of pretreatment peripheral blood markers in paranasal sinus cancer: Neutrophil-to-lymphocyte and platelet-to-lymphocyte ratio. Head Neck 2017, 39, 730–736. [Google Scholar] [CrossRef]
- Xie, X.; Luo, K.J.; Hu, Y.; Wang, J.Y.; Chen, J. Prognostic value of preoperative platelet–lymphocyte and neutrophil–lymphocyte ratio in patients undergoing surgery for esophageal squamous cell cancer. Dis. Esophagus 2016, 29, 79–85. [Google Scholar] [CrossRef]
- Bojaxhiu, B.; Templeton, A.J.; Elicin, O.; Shelan, M.; Zaugg, K.; Walser, M.; Giger, R.; Aebersold, D.M.; Dal Pra, A. Relation of baseline neutrophil-to-lymphocyte ratio to survival and toxicity in head and neck cancer patients treated with (chemo-) radiation. Radiat. Oncol. 2018, 13, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; Chen, C.; Xia, Y.; Bi, X.; Liu, P.; Zhang, F.; Yang, H.; An, X.; Jiang, W.; Wang, F. The ratio of C-reactive protein/albumin is a novel inflammatory predictor of overall survival in cisplatin-based treated patients with metastatic nasopharyngeal carcinoma. Dis. Markers 2017, 2017, 6570808. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Zhang, L.; Luo, M.; Hu, G.; Mei, Q.; Liu, D.; Long, G.; Hu, G. Pretreatment hematologic markers as prognostic factors in patients with nasopharyngeal carcinoma: Neutrophil-lymphocyte ratio and platelet-lymphocyte ratio. Head Neck 2016, 38, E1332–E1340. [Google Scholar] [CrossRef]
- Tangthongkum, M.; Tiyanuchit, S.; Kirtsreesakul, V.; Supanimitjaroenporn, P.; Sinkitjaroenchai, W. Platelet to lymphocyte ratio and red cell distribution width as prognostic factors for survival and recurrence in patients with oral cancer. Eur. Arch. Oto-Rhino-Laryngol. 2017, 274, 3985–3992. [Google Scholar] [CrossRef]
- Ozturk, K.; Akyildiz, N.S.; Uslu, M.; Gode, S.; Uluoz, U. The effect of preoperative neutrophil, platelet and lymphocyte counts on local recurrence and survival in early-stage tongue cancer. Eur. Arch. Oto-Rhino-Laryngol. 2016, 273, 4425–4429. [Google Scholar] [CrossRef]
- Toyokawa, T.; Kubo, N.; Tamura, T.; Sakurai, K.; Amano, R.; Tanaka, H.; Muguruma, K.; Yashiro, M.; Hirakawa, K.; Ohira, M. The pretreatment Controlling Nutritional Status (CONUT) score is an independent prognostic factor in patients with resectable thoracic esophageal squamous cell carcinoma: Results from a retrospective study. BMC Cancer 2016, 16, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Urabe, M.; Yamashita, H.; Watanabe, T.; Seto, Y. Comparison of prognostic abilities among preoperative laboratory data indices in patients with resectable gastric and esophagogastric junction adenocarcinoma. World J. Surg. 2018, 42, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.-f.; Chang, B.-y.; Chen, X.-q.; Liu, P.-p.; Wuxiao, Z.-j.; Wang, Z.-h.; Li, S.; Jiang, W.-q.; Xia, Z.-j. A prognostic model based on pretreatment platelet lymphocyte ratio for stage IE/IIE upper aerodigestive tract extranodal NK/T cell lymphoma, nasal type. Med. Oncol. 2014, 31, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.-l.; Wang, F.-h.; Zhang, D.-s.; Qiu, M.-z.; Ren, C.; Jin, Y.; Zhou, Y.-x.; Wang, D.-s.; He, M.-m.; Bai, L. A novel inflammation-based prognostic score in esophageal squamous cell carcinoma: The C-reactive protein/albumin ratio. BMC Cancer 2015, 15, 1–11. [Google Scholar] [CrossRef]
- Xu, X.-L.; Yu, H.-Q.; Hu, W.; Song, Q.; Mao, W.-M. A novel inflammation-based prognostic score, the C-reactive protein/albumin ratio predicts the prognosis of patients with operable esophageal squamous cell carcinoma. PLoS ONE 2015, 10, e0138657. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, H.; Zhou, L.; Deng, T.; Ning, T.; Liu, R.; Zhang, L.; Wang, X.; Ge, S.; Li, H. Platelet to lymphocyte ratio is a predictive marker of prognosis and therapeutic effect of postoperative chemotherapy in non-metastatic esophageal squamous cell carcinoma. Clin. Chim. Acta 2018, 479, 160–165. [Google Scholar] [CrossRef]
- Ye, L.; Oei, R.W.; Kong, F.; Xu, T.; Shen, C.; Wang, X.; He, X.; Kong, L.; Hu, C.; Ying, H. Prognostic values of hematological biomarkers in nasopharyngeal carcinoma patients treated with intensity-modulated radiotherapy. Eur. Arch. Oto-Rhino-Laryngol. 2018, 275, 1309–1317. [Google Scholar] [CrossRef]
- Yuan, D.; Zhu, K.; Li, K.; Yan, R.; Jia, Y.; Dang, C. The preoperative neutrophil–lymphocyte ratio predicts recurrence and survival among patients undergoing R0 resections of adenocarcinomas of the esophagogastric junction. J. Surg. Oncol. 2014, 110, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Su, Y.; Chen, Z.; Wei, Z.; Han, W.; Xu, A. The prognostic value of preoperative inflammation-based prognostic scores and nutritional status for overall survival in resected patients with nonmetastatic Siewert type II/III adenocarcinoma of esophagogastric junction. Medicine 2017, 96, e7647. [Google Scholar] [CrossRef] [PubMed]
- Hsu, W.H.; Hsu, P.K.; Hsieh, C.C.; Huang, C.S.; Wu, Y.C. The metastatic lymph node number and ratio are independent prognostic factors in esophageal cancer. J. Gastrointest. Surg. 2009, 13, 1913–1920. [Google Scholar] [CrossRef]
- Chien, H.-C.; Chen, H.-S.; Wu, S.-C.; Hsu, P.-K.; Liu, C.-Y.; Wang, B.-Y.; Shih, C.-H.; Liu, C.-C. The prognostic value of metastatic lymph node number and ratio in oesophageal squamous cell carcinoma patients with or without neoadjuvant chemoradiation. Eur. J. Cardiothorac. Surg. 2016, 50, 337–343. [Google Scholar] [CrossRef]
- Furukawa, K.; Kawasaki, G.; Naruse, T.; Umeda, M. Prognostic Significance of Pretreatment Lymphocyte–to–Monocyte Ratio in Patients with Tongue Cancer. Anticancer Res. 2019, 39, 405. [Google Scholar] [CrossRef] [PubMed]
- Hsueh, C.; Tao, L.; Zhang, M.; Cao, W.; Gong, H.; Zhou, J.; Zhou, L. The prognostic value of preoperative neutrophils, platelets, lymphocytes, monocytes and calculated ratios in patients with laryngeal squamous cell cancer. Oncotarget 2017, 8, 60514–60527. [Google Scholar] [CrossRef]
- Huang, Y.; Feng, J.-F. Low preoperative lymphocyte to monocyte ratio predicts poor cancer-specific survival in patients with esophageal squamous cell carcinoma. OncoTargets Ther. 2015, 8, 137–145. [Google Scholar]
- Kano, S.; Homma, A.; Hatakeyama, H.; Mizumachi, T.; Sakashita, T.; Kakizaki, T.; Fukuda, S. Pretreatment lymphocyte-to-monocyte ratio as an independent prognostic factor for head and neck cancer. Head Neck 2017, 39, 247–253. [Google Scholar] [CrossRef]
- Li, J.; Jiang, R.; Liu, W.-S.; Liu, Q.; Xu, M.; Feng, Q.-S.; Chen, L.-Z.; Bei, J.-X.; Chen, M.-Y.; Zeng, Y.-X. A Large Cohort Study Reveals the Association of Elevated Peripheral Blood Lymphocyte-to-Monocyte Ratio with Favorable Prognosis in Nasopharyngeal Carcinoma. PLoS ONE 2013, 8, e83069. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.-S.; Huang, Y.; Yang, X.; Feng, J.-F. A nomogram to predict prognostic values of various inflammatory biomarkers in patients with esophageal squamous cell carcinoma. Am. J. Cancer Res. 2015, 5, 2180–2189. [Google Scholar]
- Oya, R.; Takenaka, Y.; Aoki, K.; Hamaguchi, H.; Takemura, K.; Nozawa, M.; Kitamura, T.; Yamamoto, Y.; Uno, A. Prognostic Significance of Hematologic Markers in Patients with Head and Neck Squamous Cell Carcinomas. Int. J. Otorhinolaryngol. Head Neck Surg. 2018, 7, 55–65. [Google Scholar] [CrossRef][Green Version]
- Yang, J.; Hsueh, C.-Y.; Cao, W.; Zhou, L. Pretreatment lymphocyte-to-monocyte ratio as an independent prognostic factor for hypopharyngeal squamous cell carcinoma. Acta Otolaryngol. 2018, 138, 734–740. [Google Scholar] [CrossRef] [PubMed]
- Tham, T.; Olson, C.; Khaymovich, J.; Herman, S.W.; Costantino, P.D. The lymphocyte-to-monocyte ratio as a prognostic indicator in head and neck cancer: A systematic review and meta-analysis. Eur. Arch. Oto-Rhino-Laryngol. 2018, 275, 1663–1670. [Google Scholar] [CrossRef]
| Author Name | Year of Publication | Prognostic Parameter (PLR/NLR/MLR) | Cohort Size | Anatomic Location of Cancer | Country of Study | Type of Study | Gender | Stage of Cancer | Metastasis | Risk Factors | Age |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Wen et al. [47] | 2018 | PLR NLR | 723 | Esophageal (65%) Gastric (35%) | UK | retrospective study | male (75.5%) female (24.5%) | T0 (4.6%) T1 (14.4%) T2 (21.0%) T3 (51.9%) T4 (8.2%) | M1 2.5% | NA | 66.1 ± 10.5 |
| Kawakita et al. [48] | 2016 | PLR NLR | 140 | Parotid gland (78%) Submandibular gland (20%) Others (2%) | Japan | retrospective study | male (86%) female (14%) | T1 (9%) T2 (26%) T3 (20%) 4a (44%) 4b (1%) | M0 (93%) M1 (7%) | NA | 64 (26–84) |
| Chen et al. [49] | 2014 | NLR PLR | 211 | Nasopharynx (100%) | China | retrospective study | male (85.8%) female (14.2%) | NA | NA | NA | 46 (14–72) |
| Zhang et al. [50] | 2015 | NLR PLR | 468 | Esophagus (100%) | China | retrospective study | male (80.3%) female (19.7%) | stage I (9.8 %) stage II (42.6 %) stage III (47.6 %) | NA | Smoking Alcohol consumption | 59.5 ± 9.0 |
| Jiang et al. [51] | 2017 | PLR NLR LMR | 78 | Thyroid (100%) | China | retrospective study | male (43.6%) female (56.4%) | stage I (23.1%) stage II (20.5%) stage III (20.5%) stage IV (35.9%) | M1 (2.6%) | NA | 47.3 ± 13.8 |
| Li et al. [52] | 2016 | PLR NLR LMR | 388 | Nasopharynx (100%) | China | retrospective study | NA | NA | M0 (100%) | NA | |
| Feng et al. [53] | 2014 | NLR PLR | 483 | Esophagus (100%) | China | retrospective study | male (85.1%) female (14.9%) | T1 (18.0%) T2 (16.6%) T3 (54.9%) T4 (10.5%) | M0 (56.7%) M1 (43.3%) | NA | 59.1 ± 8.0 |
| Jiang et al. [54] | 2015 | PLR | 1261 | Nasopharynx (100%) | China | retrospective study | male (72.9%) female (27.1%) | Clinical stage I (3.25) II (14.8%) III (52.7%) IV (29.4%) Tumor stage T1 (8.6%) T2 (23.6%) T3 (46.2%) T4 (21.5%) Node stage N0 (16.8%) N1 (36.6%) N2 (37.7%) N3 (9.0%) | M1 (12.4) M0 (87.6) | Smoking Chronic HBV infection Cardiovascular disease Diabetes mellitus Family history of NPC | 46 (39–55) |
| Jung et al. [55] | 2015 | NLR PLR | 119 | Esophagus (100%) | Korea | retrospective study | male (94.1%) female (5.9%) | Pathological stage I (31.1%) II 33 (27.7%) III 49 (41.2) | M0 (100%) | NA | 63.64 ± 8.42 |
| He et al. [56] | 2016 | NLR PLR | 317 | Esophagus (100%) | China | retrospective study | male (84.5%) female (15.5%) | TNM I–II (68.5%) III–IV (31.5%) | M0 (53.9%) M1 (46.1%) | Smoking Alcohol consumption | 60 (37–77) |
| Jiang et al. [57] | 2016 | NLR PLR | 70 | Thyroid (100%) | China | retrospective study | male (40%) female (60%) | Stage III or IV (50.0%) | M1 (2.9%) | NA | 47.7 ± 13.9 |
| Hirahara et al. [58] | 2017 | LMR NLR PLR | 147 | Esophagus (100%) | Japan | retrospective study | male (89.8%) female (10.2% | Pathological stage Ia–1b (40.1%) 2a–2b (22.4%) 3a–3c (37.4%) | NA | NA | NA |
| Ong et al. [59] | 2016 | LMR NLR PLR | 133 | Tongue (100%) | China | retrospective study | male (53.4%) female (46.6%) | pT classification, n (%) T1 (39.1%) T2 (60.9%) | M0 (96.2%) M1 (3.8%) | NA | 51.92 (24–74) |
| Mao et al. [60] | 2017 | PLR | 899 | Larynx (100%) | China | retrospective study | male (97.1%) female (2.9%) | T 1 (22.9%) 2 (28.9%) 3 (29.1%) 4 (19.0%) N 0 (81.2%) 1 (9.7%) 2 (8.6%) 3 (0.6%) | M0 (100%) | Smoking Alcohol consumption | 60 (22–87) |
| Dutta et al. [61] | 2011 | NLR PLR | 112 | Esophagus (100%) | UK | retrospective study | male (75.9%) female (24.1%) | TNM stage I 17.9% II 34.8% III 46.4% IV 0.9% | NA | NA | <65 (60.7%) 65–74 (33.9%) ≥75 (5.4%) |
| Li et al. [62] | 2016 | NLR PLR | 409 | Nasopharynx (100%) | China | retrospective study | male (70.4%) female (29.6%) | (I–II) (18.8%) (III–IV) (81.2%) | M0 (84.4%) M1 (15.6%) | NA | 45 (18–77) |
| Messager et al. [63] | 2015 | PLR | 153 | Esophagus (100%) | UK | live patient samples | male (83.7%) female (16.3%) | ||||
| Hirahara et al. [64] | 2016 | LMR NLR PLR | 147 | Esophagus (100%) | Japan | retrospective study | male (89.8%) female (10.2%) | Pathological stage Ia–Ib 40.1% IIa–Iib 22.5% IIIa–IIIc 37.4% | NA | NA | NA |
| Moon et al. [65] | 2015 | NLR PLR | 153 | Oropharynx 33.3% Nasopharynx 31.4% Larynx 18.3% Hypopharynx 17.0% | Korea | live patient samples | male (84.3%) female (15.7%) | Clinical TNM stage T1/T2 17.6%/36.6% T3/T4 14.4%/31.4% N0/N1 21.6%/20.9% N2/N3 49.7%/7.8% Overall I/II 4.6%/17.0% Overall III/IV 13.7%/64.75% | M0 (100%) | Smoking Alcohol consumption | 57 (16–78) |
| Hirahara et al. [66] | 2016 | LMR NLR PLR | 147 | Esophagus (100%) | Japan | retrospective study | male (89.8%) female (10.2%) | pathological stage 1a–1b (40.2%) 2a–2b (22.4%) 3a–3c (37.4%) | NA | NA | |
| Turri–Zanoni et al. [67] | 2016 | NLR PLR | 215 | Paranasal sinus (100%) | Italy | retrospective study | male (34%) female (66%) | pT classification pT1 19% pT2 18% pT3 22% pT4a 16% pT4b 25% | M0 (100%) | NA | 65 (8–87) |
| Xie et al. [68] | 2014 | NLR PLR | 317 | Esophagus (100%) | China | retrospective study | male (77%) female (23%) | Tumor stage Stage I 88.4 Stage II 69.7 Stage III 42.4 | M0 (100%) | NA | 58.1 ± 8.9 |
| Bojaxhiu et al. [69] | 2018 | NLR PLR | 186 | Oral cavity (28%) Oropharynx (45%) Hypopharynx (15%) Larynx (13%) | Switzerland | retrospective study | male (79%) female (22%) | UICC stage, N (%) I (3%) II (6%) III (24%) IV (68%) | M0 (100%) | Smoking Alcohol consumption | 61 (41–88) |
| Sun et al. [70] | 2017 | NLR PLR | 148 | Nasopharynx (100%) | China | retrospective study | male (83.8%) female (16.2%) | M0 (100%) | Smoking | 45 (24–72) | |
| Sun et al. [71] | 2015 | NLR PLR | 251 | Nasopharynx (100%) | China | retrospective study | male (71.7%) female (28.3%) | UICC/AJCC stage I 2.4% II 15.9% III 47.4% IV 34.3% | M0 (100%) | NA | 46 (15–76) |
| Tangthongkum et al. [72] | 2017 | PLR | 274 | Oral (100%) | Thailand | retrospective study | males (64.4%) females (35.6%) | NA | NA | NA | 60 (21–92) |
| Ozturk et al. [73] | 2016 | NLR PLR | 57 | Tongue (100%) | Turkey | retrospective study | male (38.6%) female (61.4%) | stage I (64.9 %) stage II (35.1 %) | M0 (100%) | NA | 57.8 (23–88) |
| Toyokawa et al. [74] | 2016 | NLR PLR | 185 | Esophagus (100%) | Japan | retrospective study | male (82.2 %) female (17.8%) | Clinical TNM stage I 36.2% II 42.2% III/IV 21.6% | M0 (100%) | NA | <65 51.4% ≥65 48.6% |
| Urabe et al. [75] | 2017 | LMR NLR PLR | 1363 | Resectable Gastric and Esophagogastric Junction | Japan | retrospective study | male (71.5%) female (28.5%) | T stage T1 58% T2 11.8% T3 17.6% T4 12.6% | M0 (100%) | NA | NA |
| Wang et al. [76] | 2014 | PLR | 252 | Upper aerodigestive tract | China | retrospective study | male (68.7%) female (31.3%) | Ann Arbor stage IE 61.5% IIE 38.5 % | NA | NA | 41 (9–80) |
| Wei et al. [77] | 2015 | NLR PLR | 423 | Esophagus (100%) | China | retrospective study | male (80.6%) female (19.4) | TNM stage (AJCC, 7th) I (12.8%) II (39.7%) III (33.6%) IV (13.9%) | M0 (86.1) M1 (13.9) | NA | 58 (24–88) |
| Xu et al. [78] | 2015 | NLR PLR | 468 | Esophagus (100%) | China | retrospective study | male (88.9%) female (11.1%) | Clinical stage I II IIIA IIIB + IIIC | NA | Smoking Alcohol consumption | 58 |
| Yang et al. [79] | 2018 | NLR PLR | 515 | Esophagus (100%) | China | retrospective study | male (81.2%) female (18.8%) | TNM Stage I II III | M0 (100%) | NA | 61(33–92) |
| Ye et al. [80] | 2018 | NLR PLR | 427 | Nasopharynx (100%) | China | retrospective study | male (71.9%) female (28.1%) | TNM stage I 2.1 II 18.7% III 48.7% IV 30.5% | yes | NA | 48 (17–82) |
| Yuan et al. [81] | 2014 | NLR PLR | 327 | Esophagogastric junction (100%) | China | retrospective study | male (86.2%) female(13.8%) | pTNM stage I and II (45.9%) III and IV (54.1%) | yes | NA | 63.1± 9.7 (39–77) |
| Zhang et al. [82] | 2017 | NLR PLR | 355 | Esophagogastric junction (100%) | China | retrospective study | male (79.2%) female (20.8%) | TNM stage I, II (43.4%) III, IV (56.6%) | NA | NA | 64 (34–82) |
| Chen et al. [20] | 2018 | NLR PLR MLR | 361 | Larynx (100%) | China | retrospective study | male (97.8%) female (2.2%) | TNM stage I 31.6% II 32.7% III 19.7% IV 16.0% | yes | NA | 60 (35–87) |
| Hsu et al. [83] | 2009 | MLR | 1069 | Esophagus (100%) | Taiwan | retrospective study | male (94.5%) female (5.5%) | stage I 53 (10.9%) II 197 (40.4%) III 138 (28.3%) IV 100 (20.5%) | yes | NA | 63.8 (34–88) |
| Chien et al. [84] | 2016 | MLR | 2025 | Esophagus (100%) | Taiwan | retrospective study | male (94.1%) female (5.9%) | cStage T1–2N0 T3–4N0 T1–2N (+) T3–4N (+) Unknown | yes | NA | 55.2 ± 9.8 |
| Furukawa et al. [85] | 2019 | LMR | 103 | Tongue (100%) | Japan | retrospective study | male (53.8%) female (46.2%) | stage I, II 84.5% III, IV 15.5% | yes | Smoking Alcohol consumption | 63 (26–92) |
| Hsueh et al. [86] | 2017 | NLR PLR LMR | 979 | Larynx (100%) | China | retrospective study | male (97.5%) female (2.5%) | stage I 23.7% II 36.4% III 26.8% IV 13.1% | yes | NA | 60.81 ± 9.68 |
| Huang et al. [87] | 2015 | LMR | 348 | Esophagus (100%) | China | retrospective study | male (87.1%) female (12.9%) | NA | yes | NA | 59.2 ±7.8 |
| Kano et al. [88] | 2016 | NLR PLR LMR | 285 | Larynx (23.5%) Oropharynx (40.7%) Hypopharynx (35.8%) | Japan | retrospective study | male (88.4%) female (11.6%) | Clinical stage I, II (22.1%) III, IV (77.9%) | no | NA | 61 (37–80) |
| Li et al. [89] | 2013 | LMR | 1547 | Nasopharynx (100%) | China | retrospective study | male 72.7% female 27.3% | Overall stage I-II (21.6%) III-IV (78.4%) | yes | NA | 51 (6–87) |
| Li et al. [52] | 2017 | NLR PLR LMR | 249 | Nasopharynx (100%) | China | prospective study | male (73.9%) female (26.1%) | Clinical stage I-II 26.1 III-IV 73.9 | yes | NA | ≤50 (65.9%) >50 (34.1%) |
| Liu et al. [90] | 2015 | NLR PLR LMR | 326 | Esophagus (100%) | China | retrospective study | male (86.8%) female (13.2%) | T stage T1 (18.1%) T2 (18.4%) T3 (53.7%) T4 (9.8%) | NA | NA | 59.2 ± 7.9 (38–80) |
| Oya et al. [91] | 2018 | NLR PLR LMR | 441 | Oral cavity 44% Larynx 28% Oropharynx 10% Hypopharynx 13 % Other 5% | Japan | retrospective study | male (73%) female (27%) | Stage I 32% II 18% III 15% IV 35% | no | NA | 68 (27–92) |
| Yang et al. [92] | 2018 | NLR LMR | 197 | Hypopharyngeal (100%) | China | retrospective study | male (99.0%) female (0.01%) | Clinical stage I (2.0%) II (13.2%) III (27.5%) IV (57.4%) | yes | Smoking Drinking | <59 (50.8%) ≥59 (49.2%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumarasamy, C.; Tiwary, V.; Sunil, K.; Suresh, D.; Shetty, S.; Muthukaliannan, G.K.; Baxi, S.; Jayaraj, R. Prognostic Utility of Platelet–Lymphocyte Ratio, Neutrophil–Lymphocyte Ratio and Monocyte–Lymphocyte Ratio in Head and Neck Cancers: A Detailed PRISMA Compliant Systematic Review and Meta-Analysis. Cancers 2021, 13, 4166. https://doi.org/10.3390/cancers13164166
Kumarasamy C, Tiwary V, Sunil K, Suresh D, Shetty S, Muthukaliannan GK, Baxi S, Jayaraj R. Prognostic Utility of Platelet–Lymphocyte Ratio, Neutrophil–Lymphocyte Ratio and Monocyte–Lymphocyte Ratio in Head and Neck Cancers: A Detailed PRISMA Compliant Systematic Review and Meta-Analysis. Cancers. 2021; 13(16):4166. https://doi.org/10.3390/cancers13164166
Chicago/Turabian StyleKumarasamy, Chellan, Vaibhav Tiwary, Krishnan Sunil, Deepa Suresh, Sameep Shetty, Gothandam Kodiveri Muthukaliannan, Siddhartha Baxi, and Rama Jayaraj. 2021. "Prognostic Utility of Platelet–Lymphocyte Ratio, Neutrophil–Lymphocyte Ratio and Monocyte–Lymphocyte Ratio in Head and Neck Cancers: A Detailed PRISMA Compliant Systematic Review and Meta-Analysis" Cancers 13, no. 16: 4166. https://doi.org/10.3390/cancers13164166
APA StyleKumarasamy, C., Tiwary, V., Sunil, K., Suresh, D., Shetty, S., Muthukaliannan, G. K., Baxi, S., & Jayaraj, R. (2021). Prognostic Utility of Platelet–Lymphocyte Ratio, Neutrophil–Lymphocyte Ratio and Monocyte–Lymphocyte Ratio in Head and Neck Cancers: A Detailed PRISMA Compliant Systematic Review and Meta-Analysis. Cancers, 13(16), 4166. https://doi.org/10.3390/cancers13164166










