Pharmacological Approaches in Neurofibromatosis Type 1-Associated Nervous System Tumors
Abstract
Simple Summary
Abstract
1. Introduction
2. Benign Nerve Sheath Tumors (Neurofibroma)
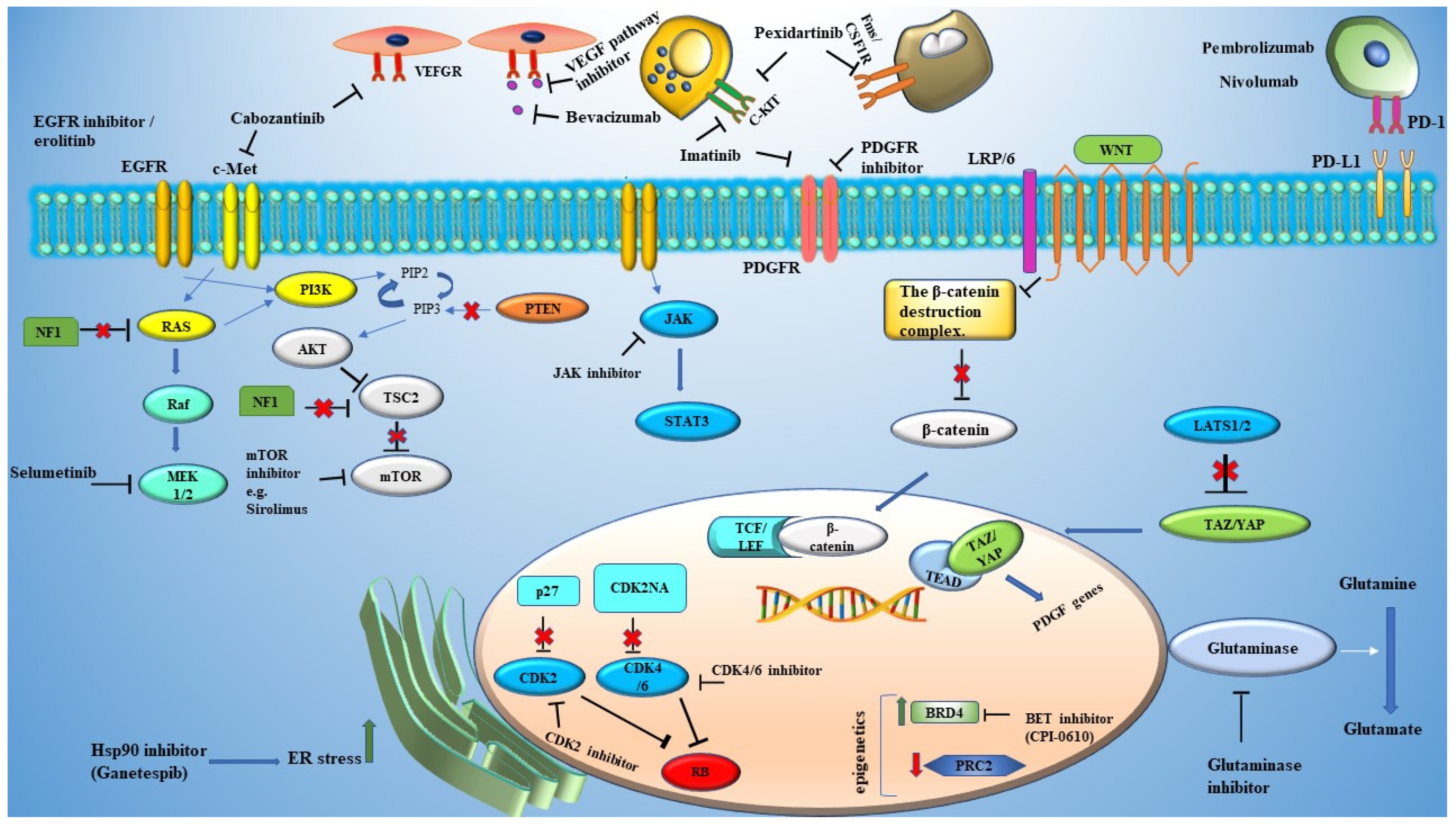
2.1. Targeting RAS Signaling
2.2. Targeting Mammalian Target of Rapamycin (mTOR)
2.3. Targeting Microenvironments
2.4. Runt-Related Transcription Factor (RUNX) Inhibition
3. Malignant Nerve Sheath Tumor
3.1. Developmental Genes in MPNSTs
3.2. Endoplasmic Reticulum (ER) Stress in MPNSTs
3.3. Targeting Cancer Metabolism
3.4. Epigenetic and MPNSTs
3.5. Harnessing the Immune System
4. Glioma
5. Non-Coding RNA in NF1 Tumors
6. “Hit the Bullseye”
7. Genetically Engineered Models for NF1
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kim, A.; Gillespie, A.; Dombi, E.; Goodwin, A.; Goodspeed, W.; Fox, E.; Balis, F.M.; Widemann, B.C. Characteristics of Children Enrolled in Treatment Trials for NF1-Related Plexiform Neurofibromas. Neurology 2009, 73, 1273–1279. [Google Scholar] [CrossRef]
- Easton, D.F.; Ponder, M.A.; Huson, S.M.; Ponder, B.A. An Analysis of Variation in Expression of Neurofibromatosis (NF) Type 1 (NF1): Evidence for Modifying Genes. Am. J. Hum. Genet. 1993, 53, 305–313. [Google Scholar]
- Evans, D.G.R.; O’Hara, C.; Wilding, A.; Ingham, S.L.; Howard, E.; Dawson, J.; Moran, A.; Scott-Kitching, V.; Holt, F.; Huson, S.M. Mortality in Neurofibromatosis 1: In North West England: An Assessment of Actuarial Survival in a Region of the UK since 1989. Eur. J. Hum. Genet. 2011, 19, 1187–1191. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, S.A.; Yang, Q.; Friedman, J.M. Mortality in Neurofibromatosis 1: An Analysis Using U.S. Death Certificates. Am. J. Hum. Genet. 2001, 68, 1110–1118. [Google Scholar] [CrossRef] [PubMed]
- Masocco, M.; Kodra, Y.; Vichi, M.; Conti, S.; Kanieff, M.; Pace, M.; Frova, L.; Taruscio, D. Mortality Associated with Neurofibromatosis Type 1: A Study Based on Italian Death Certificates (1995–2006). Orphanet J. Rare Dis. 2011, 6, 11. [Google Scholar] [CrossRef]
- Khosrotehrani, K.; Bastuji-Garin, S.; Zeller, J.; Revuz, J.; Wolkenstein, P. Clinical Risk Factors for Mortality in Patients with Neurofibromatosis 1: A Cohort Study of 378 Patients. Arch. Derm. 2003, 139, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Martin, G.A.; Viskoohil, D.; Bollag, G.; McCabe, P.C.; Crosier, W.J.; Haubruck, H.; Conroy, L.; Clark, R.; O’Connell, P.; Cawthon, R.M.; et al. The GAP-Related Domain of the Neurofibromatosis Type 1 Gene Product Interacts with Ras P21. Cell 1990, 63, 843–849. [Google Scholar] [CrossRef]
- Ballester, R.; Marchuk, D.; Boguski, M.; Saulino, A.; Letcher, R.; Wigler, M.; Collins, F. The NF1 Locus Encodes a Protein Functionally Related to Mammalian GAP and Yeast IRA Proteins. Cell 1990, 63, 851–859. [Google Scholar] [CrossRef]
- Corral, T.; Jiménez, M.; Hernández-Muñoz, I.; de Castro, I.P.; Pellicer, A. NF1 Modulates the Effects of Ras Oncogenes: Evidence of Other NF1 Function besides Its GAP Activity: NF1. J. Cell. Physiol. 2003, 197, 214–224. [Google Scholar] [CrossRef]
- D’angelo, I.; Welti, S.; Bonneau, F.; Scheffzek, K. A Novel Bipartite Phospholipid-binding Module in the Neurofibromatosis Type 1 Protein. EMBO Rep. 2006, 7, 174–179. [Google Scholar] [CrossRef]
- Scheffzek, K.; Shivalingaiah, G. Ras-Specific GTPase-Activating Proteins—Structures, Mechanisms, and Interactions. Cold Spring Harb. Perspect. Med. 2019, 9, a031500. [Google Scholar] [CrossRef] [PubMed]
- Kehrer-Sawatzki, H.; Mautner, V.-F.; Cooper, D.N. Emerging Genotype–Phenotype Relationships in Patients with Large NF1 Deletions. Hum. Genet. 2017, 136, 349–376. [Google Scholar] [CrossRef]
- Koczkowska, M.; Callens, T.; Chen, Y.; Gomes, A.; Hicks, A.D.; Sharp, A.; Johns, E.; Uhas, K.A.; Armstrong, L.; Bosanko, K.A.; et al. Clinical Spectrum of Individuals with Pathogenic N F1 Missense Variants Affecting p.Met1149, p.Arg1276, and p.Lys1423: Genotype–Phenotype Study in Neurofibromatosis Type 1. Hum. Mutat. 2020, 41, 299–315. [Google Scholar] [CrossRef] [PubMed]
- Koczkowska, M.; Chen, Y.; Callens, T.; Gomes, A.; Sharp, A.; Johnson, S.; Hsiao, M.-C.; Chen, Z.; Balasubramanian, M.; Barnett, C.P.; et al. Genotype-Phenotype Correlation in NF1: Evidence for a More Severe Phenotype Associated with Missense Mutations Affecting NF1 Codons 844–848. Am. J. Hum. Genet. 2018, 102, 69–87. [Google Scholar] [CrossRef] [PubMed]
- Hirbe, A.C.; Gutmann, D.H. Neurofibromatosis Type 1: A Multidisciplinary Approach to Care. Lancet Neurol. 2014, 13, 834–843. [Google Scholar] [CrossRef]
- Prada, C.E.; Rangwala, F.A.; Martin, L.J.; Lovell, A.M.; Saal, H.M.; Schorry, E.K.; Hopkin, R.J. Pediatric Plexiform Neurofibromas: Impact on Morbidity and Mortality in Neurofibromatosis Type 1. J. Pediatrics 2012, 160, 461–467. [Google Scholar] [CrossRef]
- Evans, D.G.R. Malignant Peripheral Nerve Sheath Tumours in Neurofibromatosis 1. J. Med. Genet. 2002, 39, 311–314. [Google Scholar] [CrossRef]
- Ortonne, N.; Wolkenstein, P.; Blakeley, J.O.; Korf, B.; Plotkin, S.R.; Riccardi, V.M.; Miller, D.C.; Huson, S.; Peltonen, J.; Rosenberg, A.; et al. Cutaneous Neurofibromas: Current Clinical and Pathologic Issues. Neurology 2018, 91, S5–S13. [Google Scholar] [CrossRef]
- Bollag, G.; Clapp, D.W.; Shih, S.; Adler, F.; Zhang, Y.Y.; Thompson, P.; Lange, B.J.; Freedman, M.H.; McCormick, F.; Jacks, T.; et al. Loss of NF1 Results in Activation of the Ras Signaling Pathway and Leads to Aberrant Growth in Haematopoietic Cells. Nat. Genet. 1996, 12, 144–148. [Google Scholar] [CrossRef]
- Hiatt, K.K.; Ingram, D.A.; Zhang, Y.; Bollag, G.; Clapp, D.W. Neurofibromin GTPase-Activating Protein-Related Domains Restore Normal Growth in Nf1−/− Cells. J. Biol. Chem. 2001, 276, 7240–7245. [Google Scholar] [CrossRef]
- Jessen, W.J.; Miller, S.J.; Jousma, E.; Wu, J.; Rizvi, T.A.; Brundage, M.E.; Eaves, D.; Widemann, B.; Kim, M.-O.; Dombi, E.; et al. MEK Inhibition Exhibits Efficacy in Human and Mouse Neurofibromatosis Tumors. J. Clin. Investig. 2013, 123, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Dombi, E.; Baldwin, A.; Marcus, L.J.; Fisher, M.J.; Weiss, B.; Kim, A.; Whitcomb, P.; Martin, S.; Aschbacher-Smith, L.E.; Rizvi, T.A.; et al. Activity of Selumetinib in Neurofibromatosis Type 1–Related Plexiform Neurofibromas. N. Engl. J. Med. 2016, 375, 2550–2560. [Google Scholar] [CrossRef]
- Gross, A.M.; Wolters, P.L.; Dombi, E.; Baldwin, A.; Whitcomb, P.; Fisher, M.J.; Weiss, B.; Kim, A.; Bornhorst, M.; Shah, A.C.; et al. Selumetinib in Children with Inoperable Plexiform Neurofibromas. N. Engl. J. Med. 2020, 382, 1430–1442. [Google Scholar] [CrossRef]
- Great Ormond Street Hospital for Children NHS Foundation Trust. A Paediatric Phase I/II Study of Intermittent Dosing of the Mek-1 Inhibitor Selumetinib in Children with Neurofibromatosis Type-1 And Inoperable Plexiform Neurofibroma And/Or Progressive Optic Pathway Glioma. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- National Cancer Institute (NCI). Phase II Trial of the MEK1/2 Inhibitor Selumetinib (AZD6244 Hydrogen Sulfate in Adults with Neurofibromatosis Type 1 (NF1) and Inoperable Plexiform Neurofibromas. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- National Cancer Institute (NCI). Pilot Study of the MEK1/2 Inhibitor Selumetinib (AZD6244 Hydrogen Sulfate) for Adults with Neurofibromatosis Type 1 (NF1) and Cutaneous Neurofibromas (CNF). Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Vaassen, P.; Dürr, N.; Röhrig, A.; Willing, R.; Rosenbaum, T. Trametinib Induces Neurofibroma Shrinkage and Enables Surgery. Neuropediatrics 2019, 50, 300–303. [Google Scholar] [CrossRef] [PubMed]
- Perreault, D. A Phase 2 Study of Trametinib for Patients with Pediatric Glioma or Plexiform Neurofibroma with Refractory Tumor and Activation of the MAPK/ERK Pathway. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Korf, B. A Phase II Study of Binimetinib in Children and Adults with NF1 Associated Plexiform Neurofibromas (PNOC010). Available online: clinicaltrials.gov (accessed on 25 May 2021).
- SpringWorks Therapeutics, Inc. A Phase 2b Trial of the MEK 1/2 Inhibitor (MEKi) PD-0325901 in Adult and Pediatric Patients with Neurofibromatosis Type 1 (NF1)-Associated Inoperable Plexiform Neurofibromas (PNs) That Are Causing Significant Morbidity. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Jousma, E.; Rizvi, T.A.; Wu, J.; Janhofer, D.; Dombi, E.; Dunn, R.S.; Kim, M.-O.; Masters, A.R.; Jones, D.R.; Cripe, T.P.; et al. Preclinical Assessments of the MEK Inhibitor PD-0325901 in a Mouse Model of Neurofibromatosis Type 1: MEK Inhibition in Neurofibroma. Pediatr. Blood Cancer 2015, 62, 1709–1716. [Google Scholar] [CrossRef]
- Cho, K.-N.; Lee, K.-I. Chemistry and Biology of Ras Farnesyltransferase. Arch. Pharm. Res. 2002, 25, 759–769. [Google Scholar] [CrossRef] [PubMed]
- Yan, N.; Ricca, C.; Fletcher, J.; Glover, T.; Seizinger, B.R.; Manne, V. Farnesyltransferase Inhibitors Block the Neurofibromatosis Type I (NF1) Malignant Phenotype. Cancer Res. 1995, 55, 3569–3575. [Google Scholar]
- Widemann, B.C.; Dombi, E.; Gillespie, A.; Wolters, P.L.; Belasco, J.; Goldman, S.; Korf, B.R.; Solomon, J.; Martin, S.; Salzer, W.; et al. Phase 2 Randomized, Flexible Crossover, Double-Blinded, Placebo-Controlled Trial of the Farnesyltransferase Inhibitor Tipifarnib in Children and Young Adults with Neurofibromatosis Type 1 and Progressive Plexiform Neurofibromas. Neuro-Oncol. 2014, 16, 707–718. [Google Scholar] [CrossRef]
- Saxton, R.A.; Sabatini, D.M. MTOR Signaling in Growth, Metabolism, and Disease. Cell 2017, 168, 960–976. [Google Scholar] [CrossRef]
- Johannessen, C.M.; Reczek, E.E.; James, M.F.; Brems, H.; Legius, E.; Cichowski, K. The NF1 Tumor Suppressor Critically Regulates TSC2 and MTOR. Proc. Natl. Acad. Sci. USA 2005, 102, 8573–8578. [Google Scholar] [CrossRef]
- Bhola, P.; Banerjee, S.; Mukherjee, J.; Balasubramanium, A.; Arun, V.; Karim, Z.; Burrell, K.; Croul, S.; Gutmann, D.H.; Guha, A. Preclinical In Vivo Evaluation of Rapamycin in Human Malignant Peripheral Nerve Sheath Explant Xenograft. Int. J. Cancer 2010, 126, 563–571. [Google Scholar] [CrossRef]
- Johannessen, C.M.; Johnson, B.W.; Williams, S.M.G.; Chan, A.W.; Reczek, E.E.; Lynch, R.C.; Rioth, M.J.; McClatchey, A.; Ryeom, S.; Cichowski, K. TORC1 Is Essential for NF1-Associated Malignancies. Curr. Biol. 2008, 18, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Johansson, G.; Mahller, Y.Y.; Collins, M.H.; Kim, M.-O.; Nobukuni, T.; Perentesis, J.; Cripe, T.P.; Lane, H.A.; Kozma, S.C.; Thomas, G.; et al. Effective In Vivo Targeting of the Mammalian Target of Rapamycin Pathway in Malignant Peripheral Nerve Sheath Tumors. Mol. Cancer 2008, 7, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Weiss, B.; Widemann, B.C.; Wolters, P.; Dombi, E.; Vinks, A.; Cantor, A.; Perentesis, J.; Schorry, E.; Ullrich, N.; Gutmann, D.H.; et al. Sirolimus for Progressive Neurofibromatosis Type 1-Associated Plexiform Neurofibromas: A Neurofibromatosis Clinical Trials Consortium Phase II Study. Neuro-Oncol. 2015, 17, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Slopis, J.M.; Arevalo, O.; Bell, C.S.; Hebert, A.A.; Northrup, H.; Riascos, R.F.; Samuels, J.A.; Smith, K.C.; Tate, P.; Koenig, M.K. Treatment of Disfiguring Cutaneous Lesions in Neurofibromatosis-1 with Everolimus: A Phase II, Open-Label, Single-Arm Trial. Drugs R D 2018, 18, 295–302. [Google Scholar] [CrossRef]
- Bissell, M.J.; Hines, W.C. Why Don’t We Get More Cancer? A Proposed Role of the Microenvironment in Restraining Cancer Progression. Nat. Med. 2011, 17, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Grivennikov, S.I.; Greten, F.R.; Karin, M. Immunity, Inflammation, and Cancer. Cell 2010, 140, 883–899. [Google Scholar] [CrossRef] [PubMed]
- Staser, K.; Yang, F.-C.; Clapp, D.W. Pathogenesis of Plexiform Neurofibroma: Tumor-Stromal/Hematopoietic Interactions in Tumor Progression. Annu. Rev. Pathol. Mech. Dis. 2012, 7, 469–495. [Google Scholar] [CrossRef]
- Le, L.Q.; Parada, L.F. Tumor Microenvironment and Neurofibromatosis Type I: Connecting the GAPs. Oncogene 2007, 26, 4609–4616. [Google Scholar] [CrossRef]
- Riccardi, V.M. Mast-Cell Stabilization to Decrease Neurofibroma Growth: Preliminary Experience with Ketotifen. Arch. Derm. 1987, 123, 1011. [Google Scholar] [CrossRef]
- Riccardi, V.M. A Controlled Multiphase Trial of Ketotifen to Minimize Neurofibroma-Associated Pain and Itching. Arch. Derm. 1993, 129, 577. [Google Scholar] [CrossRef] [PubMed]
- Riccardi, V.M. Ketotifen Suppression of NF1 Neurofibroma Growth over 30 Years. Am. J. Med. Genet. 2015, 167, 1570–1577. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.-C.; Ingram, D.A.; Chen, S.; Zhu, Y.; Yuan, J.; Li, X.; Yang, X.; Knowles, S.; Horn, W.; Li, Y.; et al. Nf1-Dependent Tumors Require a Microenvironment Containing Nf1+/− and c-Kit-Dependent Bone Marrow. Cell 2008, 135, 437–448. [Google Scholar] [CrossRef]
- Robertson, K.A.; Nalepa, G.; Yang, F.-C.; Bowers, D.C.; Ho, C.Y.; Hutchins, G.D.; Croop, J.M.; Vik, T.A.; Denne, S.C.; Parada, L.F.; et al. Imatinib Mesylate for Plexiform Neurofibromas in Patients with Neurofibromatosis Type 1: A Phase 2 Trial. Lancet Oncol. 2012, 13, 1218–1224. [Google Scholar] [CrossRef]
- Wei, J.; Freytag, M.; Schober, Y.; Nockher, W.A.; Mautner, V.F.; Friedrich, R.E.; Manley, P.W.; Kluwe, L.; Kurtz, A. Nilotinib Is More Potent than Imatinib for Treating Plexiform Neurofibroma In Vitro and In Vivo. PLoS ONE 2014, 9, e107760. [Google Scholar] [CrossRef] [PubMed]
- Indiana University Pilot Study of Tasigna®/Nilotinib (AMN107) in Neurofibromatosis (NF1) Patients with Plexiform Neurofibromas. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Noy, R.; Pollard, J.W. Tumor-Associated Macrophages: From Mechanisms to Therapy. Immunity 2014, 41, 49–61. [Google Scholar] [CrossRef]
- Fletcher, J.S.; Springer, M.G.; Choi, K.; Jousma, E.; Rizvi, T.A.; Dombi, E.; Kim, M.-O.; Wu, J.; Ratner, N. STAT3 Inhibition Reduces Macrophage Number and Tumor Growth in Neurofibroma. Oncogene 2019, 38, 2876–2884. [Google Scholar] [CrossRef]
- Prada, C.E.; Jousma, E.; Rizvi, T.A.; Wu, J.; Dunn, R.S.; Mayes, D.A.; Cancelas, J.A.; Dombi, E.; Kim, M.-O.; West, B.L.; et al. Neurofibroma-Associated Macrophages Play Roles in Tumor Growth and Response to Pharmacological Inhibition. Acta Neuropathol. 2013, 125, 159–168. [Google Scholar] [CrossRef]
- National Cancer Institute (NCI). Phase I/II Trial of PLX3397 in Children and Young Adults with Refractory Leukemias and Refractory Solid Tumors Including Neurofibromatosis Type 1 (NF1) Associated Plexiform Neurofibromas (PN). Clinical trial registration NCT02390752. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Yao, Z.; Zhang, J.; Zhang, B.; Liang, G.; Chen, X.; Yao, F.; Xu, X.; Wu, H.; He, Q.; Ding, L.; et al. Imatinib Prevents Lung Cancer Metastasis by Inhibiting M2-like Polarization of Macrophages. Pharmacol. Res. 2018, 133, 121–131. [Google Scholar] [CrossRef]
- Fletcher, J.S.; Wu, J.; Jessen, W.J.; Pundavela, J.; Miller, J.A.; Dombi, E.; Kim, M.-O.; Rizvi, T.A.; Chetal, K.; Salomonis, N.; et al. Cxcr3-Expressing Leukocytes Are Necessary for Neurofibroma Formation in Mice. JCI Insight 2019, 4, e98601. [Google Scholar] [CrossRef]
- Stetson, D.B.; Medzhitov, R. Type I Interferons in Host Defense. Immunity 2006, 25, 373–381. [Google Scholar] [CrossRef]
- López de Padilla, C.M.; Niewold, T.B. The Type I Interferons: Basic Concepts and Clinical Relevance in Immune-Mediated Inflammatory Diseases. Gene 2016, 576, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Musella, M.; Manic, G.; De Maria, R.; Vitale, I.; Sistigu, A. Type-I-Interferons in Infection and Cancer: Unanticipated Dynamics with Therapeutic Implications. OncoImmunology 2017, 6, e1314424. [Google Scholar] [CrossRef]
- Asmana Ningrum, R. Human Interferon Alpha-2b: A Therapeutic Protein for Cancer Treatment. Scientifica 2014, 2014, 970315. [Google Scholar] [CrossRef]
- Jakacki, R.I.; Dombi, E.; Steinberg, S.M.; Goldman, S.; Kieran, M.W.; Ullrich, N.J.; Pollack, I.F.; Goodwin, A.; Manley, P.E.; Fangusaro, J.; et al. Phase II Trial of Pegylated Interferon Alfa-2b in Young Patients with Neurofibromatosis Type 1 and Unresectable Plexiform Neurofibromas. Neuro-Oncol. 2017, 19, 289–297. [Google Scholar] [CrossRef]
- Kawasaki, T.; Kawai, T. Toll-Like Receptor Signaling Pathways. Front. Immunol. 2014, 5, 461. [Google Scholar] [CrossRef] [PubMed]
- Urosevic, M.; Dummer, R. Role of Imiquimod in Skin Cancer Treatment. Am. J. Clin. Dermatol. 2004, 5, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.K.; Riccardi, V.M.; Plotkin, S.R.; Weinberg, H.; Anderson, R.R.; Blakeley, J.O.; Jarnagin, K.; Lee, J. Considerations for Development of Therapies for Cutaneous Neurofibroma. Neurology 2018, 91, S21–S30. [Google Scholar] [CrossRef]
- Weis, S.M.; Cheresh, D.A. Tumor Angiogenesis: Molecular Pathways and Therapeutic Targets. Nat. Med. 2011, 17, 1359–1370. [Google Scholar] [CrossRef] [PubMed]
- Munchhof, A.M.; Li, F.; Mead, L.; White, H.; Fenoglio, A.; Yang, F.-C.; Ingram, D.A. Function of the NF1 Tumor Suppressor Gene in Angiogenesis. Blood 2005, 106, 3680. [Google Scholar] [CrossRef]
- Kim, A.; Dombi, E.; Tepas, K.; Fox, E.; Martin, S.; Wolters, P.; Balis, F.M.; Jayaprakash, N.; Turkbey, B.; Muradyan, N.; et al. Phase I Trial and Pharmacokinetic Study of Sorafenib in Children with Neurofibromatosis Type I and Plexiform Neurofibromas: Sorafenib in Plexiform Neurofibromas. Pediatr. Blood Cancer 2013, 60, 396–401. [Google Scholar] [CrossRef]
- Shih, C.-S. A Pilot Study of Sutent®/Sunitinib (SU11248), an Oral Multi-Targeted Tyrosine Kinase Inhibitor in Subjects with NF-1 Plexiform Neurofibromas. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Neurofibromatosis Clinical Trials Consortium; Fisher, M.J.; Shih, C.-S.; Rhodes, S.D.; Armstrong, A.E.; Wolters, P.L.; Dombi, E.; Zhang, C.; Angus, S.P.; Johnson, G.L.; et al. Cabozantinib for Neurofibromatosis Type 1–Related Plexiform Neurofibromas: A Phase 2 Trial. Nat. Med. 2021, 27, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Korf, B. A Phase II Study of Cabozantinib (XL184) for Plexiform Neurofibromas in Subjects with Neurofibromatosis Type 1 in Children and Adults. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Widemann, B.C.; Babovic-Vuksanovic, D.; Dombi, E.; Wolters, P.L.; Goldman, S.; Martin, S.; Goodwin, A.; Goodspeed, W.; Kieran, M.W.; Cohen, B.; et al. Phase II Trial of Pirfenidone in Children and Young Adults with Neurofibromatosis Type 1 and Progressive Plexiform Neurofibromas: Pirfenidone Phase II Trial in NF1 and PN. Pediatr. Blood Cancer 2014, 61, 1598–1602. [Google Scholar] [CrossRef] [PubMed]
- Mevel, R.; Draper, J.E.; Lie-a-Ling, M.; Kouskoff, V.; Lacaud, G. RUNX Transcription Factors: Orchestrators of Development. Development 2019, 146, dev148296. [Google Scholar] [CrossRef]
- Li, H.; Zhao, X.; Yan, X.; Jessen, W.J.; Kim, M.-O.; Dombi, E.; Liu, P.P.; Huang, G.; Wu, J. Runx1 Contributes to Neurofibromatosis Type 1 Neurofibroma Formation. Oncogene 2016, 35, 1468–1474. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.; Choi, K.; Liu, W.; Rose, J.; Zhao, C.; Yu, Y.; Na, Y.; Cai, Y.; Coover, R.A.; Lin, Y.; et al. RUNX Represses Pmp22 to Drive Neurofibromagenesis. Sci. Adv. 2019, 5, eaau8389. [Google Scholar] [CrossRef] [PubMed]
- Kolberg, M.; Holand, M.; Agesen, T.H.; Brekke, H.R.; Liestol, K.; Hall, K.S.; Mertens, F.; Picci, P.; Smeland, S.; Lothe, R.A. Survival Meta-Analyses for >1800 Malignant Peripheral Nerve Sheath Tumor Patients with and without Neurofibromatosis Type 1. Neuro-Oncol. 2013, 15, 135–147. [Google Scholar] [CrossRef]
- Brohl, A.S.; Kahen, E.; Yoder, S.J.; Teer, J.K.; Reed, D.R. The Genomic Landscape of Malignant Peripheral Nerve Sheath Tumors: Diverse Drivers of Ras Pathway Activation. Sci. Rep. 2017, 7, 14992. [Google Scholar] [CrossRef]
- Lee, W.; Teckie, S.; Wiesner, T.; Ran, L.; Prieto Granada, C.N.; Lin, M.; Zhu, S.; Cao, Z.; Liang, Y.; Sboner, A.; et al. PRC2 Is Recurrently Inactivated through EED or SUZ12 Loss in Malignant Peripheral Nerve Sheath Tumors. Nat. Genet. 2014, 46, 1227–1232. [Google Scholar] [CrossRef]
- Amirnasr, A.; Verdijk, R.M.; van Kuijk, P.F.; Taal, W.; Sleijfer, S.; Wiemer, E.A.C. Expression and Inhibition of BRD4, EZH2 and TOP2A in Neurofibromas and Malignant Peripheral Nerve Sheath Tumors. PLoS ONE 2017, 12, e0183155. [Google Scholar] [CrossRef]
- Torres, K.E.; Liu, J.; Young, E.; Huang, K.-L.; Ghadimi, M.; Lusby, K.; Lazar, A.J.; Lev, D. Expression of ‘Drugable’ Tyrosine Kinase Receptors in Malignant Peripheral Nerve Sheath Tumour: Potential Molecular Therapeutic Targets for a Chemoresistant Cancer: Correspondence. Histopathology 2011, 59, 156–159. [Google Scholar] [CrossRef][Green Version]
- DeClue, J.E.; Heffelfinger, S.; Benvenuto, G.; Ling, B.; Li, S.; Rui, W.; Vass, W.C.; Viskochil, D.; Ratner, N. Epidermal Growth Factor Receptor Expression in Neurofibromatosis Type 1–Related Tumors and NF1 Animal Models. J. Clin. Investig. 2000, 105, 1233–1241. [Google Scholar] [CrossRef]
- Holtkamp, N.; Malzer, E.; Zietsch, J.; Okuducu, A.F.; Mucha, J.; Mawrin, C.; Mautner, V.-F.; Schildhaus, H.-U.; von Deimling, A. EGFR and ErbB2 in Malignant Peripheral Nerve Sheath Tumors and Implications for Targeted Therapy. Neuro-Oncol. 2008, 10, 946–957. [Google Scholar] [CrossRef]
- Albritton, K.H.; Rankin, C.; Coffin, C.M.; Ratner, N.; Budd, G.T.; Schuetze, S.M.; Randall, R.L.; Declue, J.E.; Borden, E.C. Phase II Study of Erlotinib in Metastatic or Unresectable Malignant Peripheral Nerve Sheath Tumors (MPNST). JCO 2006, 24, 9518. [Google Scholar] [CrossRef]
- Endo, M.; Yamamoto, H.; Setsu, N.; Kohashi, K.; Takahashi, Y.; Ishii, T.; Iida, K.; Matsumoto, Y.; Hakozaki, M.; Aoki, M.; et al. Prognostic Significance of AKT/MTOR and MAPK Pathways and Antitumor Effect of MTOR Inhibitor in NF1-Related and Sporadic Malignant Peripheral Nerve Sheath Tumors. Clin. Cancer Res. 2013, 19, 450–461. [Google Scholar] [CrossRef] [PubMed]
- Watson, A.L.; Anderson, L.K.; Greeley, A.D.; Keng, V.W.; Rahrmann, E.P.; Halfond, A.L.; Powell, N.M.; Collins, M.H.; Rizvi, T.; Moertel, C.L.; et al. Co-Targeting the MAPK and PI3K/AKT/MTOR Pathways in Two Genetically Engineered Mouse Models of Schwann Cell Tumors Reduces Tumor Grade and Multiplicity. Oncotarget 2014, 5, 1502–1514. [Google Scholar] [CrossRef] [PubMed]
- Sarcoma Alliance for Research through Collaboration SARC031: A Phase 2 Trial of the MEK Inhibitor Selumetinib (AZD6244 Hydrogen Sulfate) in Combination with the MTOR Inhibitor Sirolimus for Patients with Unresectable or Metastatic Malignant Peripheral Nerve Sheath Tumors. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Widemann, B.C.; Lu, Y.; Reinke, D.; Okuno, S.H.; Meyer, C.F.; Cote, G.M.; Chugh, R.; Milhem, M.M.; Hirbe, A.C.; Kim, A.; et al. Targeting Sporadic and Neurofibromatosis Type 1 (NF1) Related Refractory Malignant Peripheral Nerve Sheath Tumors (MPNST) in a Phase II Study of Everolimus in Combination with Bevacizumab (SARC016). Sarcoma 2019, 2019, 7656747. [Google Scholar] [CrossRef]
- Varelas, X. The Hippo Pathway Effectors TAZ and YAP in Development, Homeostasis and Disease. Development 2014, 141, 1614–1626. [Google Scholar] [CrossRef] [PubMed]
- Harvey, K.F.; Zhang, X.; Thomas, D.M. The Hippo Pathway and Human Cancer. Nat. Rev. Cancer 2013, 13, 246–257. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.M.N.; Deng, Y.; Wang, J.; Zhao, C.; Wang, J.; Rao, R.; Xu, L.; Zhou, W.; Choi, K.; Rizvi, T.A.; et al. Programming of Schwann Cells by Lats1/2-TAZ/YAP Signaling Drives Malignant Peripheral Nerve Sheath Tumorigenesis. Cancer Cell 2018, 33, 292–308.e7. [Google Scholar] [CrossRef] [PubMed]
- Clevers, H.; Nusse, R. Wnt/β-Catenin Signaling and Disease. Cell 2012, 149, 1192–1205. [Google Scholar] [CrossRef]
- Watson, A.L.; Rahrmann, E.P.; Moriarity, B.S.; Choi, K.; Conboy, C.B.; Greeley, A.D.; Halfond, A.L.; Anderson, L.K.; Wahl, B.R.; Keng, V.W.; et al. Canonical Wnt/β-Catenin Signaling Drives Human Schwann Cell Transformation, Progression, and Tumor Maintenance. Cancer Discov. 2013, 3, 674–689. [Google Scholar] [CrossRef] [PubMed]
- Urra, H.; Dufey, E.; Avril, T.; Chevet, E.; Hetz, C. Endoplasmic Reticulum Stress and the Hallmarks of Cancer. Trends Cancer 2016, 2, 252–262. [Google Scholar] [CrossRef] [PubMed]
- De Raedt, T.; Walton, Z.; Yecies, J.L.; Li, D.; Chen, Y.; Malone, C.F.; Maertens, O.; Jeong, S.M.; Bronson, R.T.; Lebleu, V.; et al. Exploiting Cancer Cell Vulnerabilities to Develop a Combination Therapy for Ras-Driven Tumors. Cancer Cell 2011, 20, 400–413. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Lu, Y.; Okuno, S.H.; Reinke, D.; Maertens, O.; Perentesis, J.; Basu, M.; Wolters, P.L.; De Raedt, T.; Chawla, S.; et al. Targeting Refractory Sarcomas and Malignant Peripheral Nerve Sheath Tumors in a Phase I/II Study of Sirolimus in Combination with Ganetespib (SARC023). Sarcoma 2020, 2020, e5784876. [Google Scholar] [CrossRef] [PubMed]
- Pavlova, N.N.; Thompson, C.B. The Emerging Hallmarks of Cancer Metabolism. Cell Metab. 2016, 23, 27–47. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, T.N.; Patwardhan, P.P.; Cremers, S.; Schwartz, G.K. Targeted Inhibition of Glutaminase as a Potential New Approach for the Treatment of NF1 Associated Soft Tissue Malignancies. Oncotarget 2017, 8, 94054–94068. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute (NCI). A Phase II Basket Trial of Glutaminase Inhibitor (BeGIN) CB-839 HCl in Patients with NF1 Aberrations, NF1 Mutant Malignant Peripheral Nerve Sheath Tumors (MPNST), KEAP1/NRF2 and LKB1 Aberrant Tumors. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Cheng, Y.; He, C.; Wang, M.; Ma, X.; Mo, F.; Yang, S.; Han, J.; Wei, X. Targeting Epigenetic Regulators for Cancer Therapy: Mechanisms and Advances in Clinical Trials. Signal Transduct. Target. Ther. 2019, 4, 62. [Google Scholar] [CrossRef]
- Doroshow, D.B.; Eder, J.P.; LoRusso, P.M. BET Inhibitors: A Novel Epigenetic Approach. Ann. Oncol. 2017, 28, 1776–1787. [Google Scholar] [CrossRef]
- Patel, A.J.; Liao, C.-P.; Chen, Z.; Liu, C.; Wang, Y.; Le, L.Q. BET Bromodomain Inhibition Triggers Apoptosis of NF1-Associated Malignant Peripheral Nerve Sheath Tumors through Bim Induction. Cell Rep. 2014, 6, 81–92. [Google Scholar] [CrossRef]
- Cooper, J.M.; Patel, A.J.; Chen, Z.; Liao, C.-P.; Chen, K.; Mo, J.; Wang, Y.; Le, L.Q. Overcoming BET Inhibitor Resistance in Malignant Peripheral Nerve Sheath Tumors. Clin. Cancer Res. 2019, 25, 3404–3416. [Google Scholar] [CrossRef] [PubMed]
- De Raedt, T.; Beert, E.; Pasmant, E.; Luscan, A.; Brems, H.; Ortonne, N.; Helin, K.; Hornick, J.L.; Mautner, V.; Kehrer-Sawatzki, H.; et al. PRC2 Loss Amplifies Ras-Driven Transcription and Confers Sensitivity to BRD4-Based Therapies. Nature 2014, 514, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Wojcik, J.B.; Marchione, D.M.; Sidoli, S.; Djedid, A.; Lisby, A.; Majewski, J.; Garcia, B.A. Epigenomic Reordering Induced by Polycomb Loss Drives Oncogenesis but Leads to Therapeutic Vulnerabilities in Malignant Peripheral Nerve Sheath Tumors. Cancer Res. 2019, 79, 3205–3219. [Google Scholar] [CrossRef] [PubMed]
- University of Texas Southwestern Medical Center. A Phase 2 Study of CPI-0610, a Small Molecule Inhibitor of Bromodomain and Extra-Terminal (BET) Proteins, in Patients with Malignant Peripheral Nerve Sheath Tumors. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Esfahani, K.; Roudaia, L.; Buhlaiga, N.; Del Rincon, S.V.; Papneja, N.; Miller, W.H. A Review of Cancer Immunotherapy: From the Past to the Present, to the Future. Curr. Oncol. 2020, 27, 87–97. [Google Scholar] [CrossRef]
- Pediatric Brain Tumor Consortium. Phase I/II Trial of Dabrafenib, Trametinib, and Hydroxychloroquine (HCQ) for BRAF V600E-Mutant or Trametinib and HCQ for BRAF Fusion/Duplication Positive or NF1-Associated Recurrent or Progressive Gliomas in Children and Young Adults. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- National Cancer Institute (NCI). DART: Dual Anti-CTLA-4 and Anti-PD-1 Blockade in Rare Tumors. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Deyle, D.R.; Escobar, D.Z.; Peng, K.-W.; Babovic-Vuksanovic, D. Oncolytic Measles Virus as a Novel Therapy for Malignant Peripheral Nerve Sheath Tumors. Gene 2015, 565, 140–145. [Google Scholar] [CrossRef]
- Mayo Clinic. Phase I Trial of Intratumoral Administration of a NIS-Expressing Derivative Manufactured From a Genetically Engineered Strain of Measles Virus in Patients with Unresectable or Recurrent Malignant Peripheral Nerve Sheath Tumor. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Antoszczyk, S.; Spyra, M.; Mautner, V.F.; Kurtz, A.; Stemmer-Rachamimov, A.O.; Martuza, R.L.; Rabkin, S.D. Treatment of Orthotopic Malignant Peripheral Nerve Sheath Tumors with Oncolytic Herpes Simplex Virus. Neuro-Oncol. 2014, 16, 1057–1066. [Google Scholar] [CrossRef]
- Cripe, T. A Phase I Dose Escalation Study of Intratumoral or Intravenous Herpes Simplex Virus-1 Mutant HSV1716 in Patients with Refractory Non-Central Nervous System (Non-CNS) Solid Tumors. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Campian, J.; Gutmann, D.H. CNS Tumors in Neurofibromatosis. JCO 2017, 35, 2378–2385. [Google Scholar] [CrossRef]
- Cassina, M.; Frizziero, L.; Opocher, E.; Parrozzani, R.; Sorrentino, U.; Viscardi, E.; Miglionico, G.; Midena, E.; Clementi, M.; Trevisson, E. Optic Pathway Glioma in Type 1 Neurofibromatosis: Review of Its Pathogenesis, Diagnostic Assessment, and Treatment Recommendations. Cancers 2019, 11, 1790. [Google Scholar] [CrossRef]
- Kaul, A.; Toonen, J.A.; Cimino, P.J.; Gianino, S.M.; Gutmann, D.H. Akt- or MEK-Mediated MTOR Inhibition Suppresses Nf1 Optic Glioma Growth. Neuro-Oncol. 2015, 17, 843–853. [Google Scholar] [CrossRef]
- Hegedus, B.; Banerjee, D.; Yeh, T.-H.; Rothermich, S.; Perry, A.; Rubin, J.B.; Garbow, J.R.; Gutmann, D.H. Preclinical Cancer Therapy in a Mouse Model of Neurofibromatosis-1 Optic Glioma. Cancer Res. 2008, 68, 1520–1528. [Google Scholar] [CrossRef]
- Fangusaro, J.; Onar-Thomas, A.; Young Poussaint, T.; Wu, S.; Ligon, A.H.; Lindeman, N.; Banerjee, A.; Packer, R.J.; Kilburn, L.B.; Goldman, S.; et al. Selumetinib in Paediatric Patients with BRAF-Aberrant or Neurofibromatosis Type 1-Associated Recurrent, Refractory, or Progressive Low-Grade Glioma: A Multicentre, Phase 2 Trial. Lancet Oncol. 2019, 20, 1011–1022. [Google Scholar] [CrossRef]
- National Cancer Institute (NCI). A Phase 3 Randomized Study of Selumetinib Versus Carboplatin/Vincristine in Newly Diagnosed or Previously Untreated Neurofibromatosis Type 1 (NF1) Associated Low-Grade Glioma (LGG). Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Ullrich, N.J.; Prabhu, S.P.; Reddy, A.T.; Fisher, M.J.; Packer, R.; Goldman, S.; Robison, N.J.; Gutmann, D.H.; Viskochil, D.H.; Allen, J.C.; et al. A Phase II Study of Continuous Oral MTOR Inhibitor Everolimus for Recurrent, Radiographic-Progressive Neurofibromatosis Type 1–Associated Pediatric Low-Grade Glioma: A Neurofibromatosis Clinical Trials Consortium Study. Neuro-Oncol. 2020, 22, 1527–1535. [Google Scholar] [CrossRef] [PubMed]
- Bartels, U.; Hawkins, C.; Ma, J.; Ho, M.; Dirks, P.; Rutka, J.; Stephens, D.; Bouffet, E. Vascularity and Angiogenesis as Predictors of Growth in Optic Pathway/Hypothalamic Gliomas. J. Neurosurg. Pediatr. 2006, 104, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Bartels, U.K.; Hawkins, C.; Ma, J.; Ray, A.; Dirks, P.; Rutka, J.; Bouffet, E. Microvessel Density Predicts Behavior in Pediatric Optic Pathway/Hypothalamic Gliomas. JCO 2004, 22, 1556. [Google Scholar] [CrossRef]
- Avery, R.A.; Hwang, E.I.; Jakacki, R.I.; Packer, R.J. Marked Recovery of Vision in Children with Optic Pathway Gliomas Treated with Bevacizumab. JAMA Ophthalmol. 2014, 132, 111. [Google Scholar] [CrossRef] [PubMed]
- Zhukova, N.; Rajagopal, R.; Lam, A.; Coleman, L.; Shipman, P.; Walwyn, T.; Williams, M.; Sullivan, M.; Campbell, M.; Bhatia, K.; et al. Use of Bevacizumab as a Single Agent or in Adjunct with Traditional Chemotherapy Regimens in Children with Unresectable or Progressive Low-Grade Glioma. Cancer Med. 2019, 8, 40–50. [Google Scholar] [CrossRef]
- Hwang, E.I.; Jakacki, R.I.; Fisher, M.J.; Kilburn, L.B.; Horn, M.; Vezina, G.; Rood, B.R.; Packer, R.J. Long-Term Efficacy and Toxicity of Bevacizumab-Based Therapy in Children with Recurrent Low-Grade Gliomas: Bevacizumab-Based Treatment in Pediatric LGG Update. Pediatr. Blood Cancer 2013, 60, 776–782. [Google Scholar] [CrossRef]
- Terashima, K.; Kiyotani, C.; Kimura, Y.; Osumi, T.; Shioda, Y.; Kato, M.; Tomizawa, D.; Usami, K.; Ogiwara, H.; Aoki, H.; et al. LGG-41. Bevacizumab for Pediatric Low-Grade Glioma. Neuro-Oncol. 2018, 20, i113. [Google Scholar] [CrossRef][Green Version]
- Vanan, M.I.; Kazina, C.; Clark, I.; Krawitz, S.; Bunge, M. PDCT-02. Marked Recovery of Vision in a Child with Optic Pathway Glioma Treated Upfront with Bevacizumab and Vinblastine. Neuro-Oncol. 2016, 18, vi145. [Google Scholar] [CrossRef]
- Pong, W.W.; Higer, S.B.; Gianino, S.M.; Emnett, R.J.; Gutmann, D.H. Reduced Microglial CX3CR1 Expression Delays Neurofibromatosis-1 Glioma Formation: Microglia and Gliomagenesis. Ann. Neurol 2013, 73, 303–308. [Google Scholar] [CrossRef]
- Solga, A.C.; Pong, W.W.; Kim, K.-Y.; Cimino, P.J.; Toonen, J.A.; Walker, J.; Wylie, T.; Magrini, V.; Griffith, M.; Griffith, O.L.; et al. RNA Sequencing of Tumor-Associated Microglia Reveals Ccl5 as a Stromal Chemokine Critical for Neurofibromatosis-1 Glioma Growth. Neoplasia 2015, 17, 776–788. [Google Scholar] [CrossRef]
- Pan, Y.; Bush, E.C.; Toonen, J.A.; Ma, Y.; Solga, A.C.; Sims, P.A.; Gutmann, D.H. Whole Tumor RNA-Sequencing and Deconvolution Reveal a Clinically-Prognostic PTEN/PI3K-Regulated Glioma Transcriptional Signature. Oncotarget 2017, 8, 52474–52487. [Google Scholar] [CrossRef]
- Pan, Y.; Xiong, M.; Chen, R.; Ma, Y.; Corman, C.; Maricos, M.; Kindler, U.; Semtner, M.; Chen, Y.-H.; Dahiya, S.; et al. Athymic Mice Reveal a Requirement for T-Cell–Microglia Interactions in Establishing a Microenvironment Supportive of Nf1 Low-Grade Glioma Growth. Genes Dev. 2018, 32, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Pan, Y.; Gutmann, D.H. Genetic and Genomic Alterations Differentially Dictate Low-Grade Glioma Growth through Cancer Stem Cell–Specific Chemokine Recruitment of T Cells and Microglia. Neuro-Oncol. 2019, 21, 1250–1262. [Google Scholar] [CrossRef] [PubMed]
- D’Angelo, F.; Ceccarelli, M.; Tala; Garofano, L.; Zhang, J.; Frattini, V.; Caruso, F.P.; Lewis, G.; Alfaro, K.D.; Bauchet, L.; et al. The Molecular Landscape of Glioma in Patients with Neurofibromatosis 1. Nat. Med. 2019, 25, 176–187. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute (NCI). A Phase 1 and Phase II and Re-Treatment Study of AZD6244 for Recurrent or Refractory Pediatric Low Grade Glioma. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Davies, F.; Baz, R. Lenalidomide Mode of Action: Linking Bench and Clinical Findings. Blood Rev. 2010, 24, S13–S19. [Google Scholar] [CrossRef]
- National Cancer Institute (NCI). A Phase II Randomized Trial of Lenalidomide (NSC # 703813) in Pediatric Patients with Recurrent, Refractory or Progressive Juvenile Pilocytic Astrocytomas and Optic Pathway Gliomas. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Aguilera, D. A Phase II Study of Pegylated Interferon ALFA-2b in Children with Recurrent or Refractory and Radiographically or Clinically Progressive Juvenile Pilocytic Astrocytomas and Optic Pathway Gliomas. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Robison, N. Phase I Study of MEK162 for Children with Progressive or Recurrent Cancer and a Phase II Study for Children with Low-Grade Gliomas and Other Ras/Raf/MAP Pathway Activated Tumors. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Bouffet, E. A Phase II, Open-Labeled, Multi-Center, Randomized Controlled Trial of Vinblastine +/- Bevacizumab for the Treatment of Chemotherapy-Naïve Children with Unresectable or Progressive Low Grade Glioma (LGG). Available online: clinicaltrials.gov (accessed on 25 May 2021).
- National Cancer Institute (NCI). A Phase I Trial of Pomalidomide for Children with Recurrent, Progressive, or Refractory CNS Tumors. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Kyi, C.; Roudko, V.; Sabado, R.; Saenger, Y.; Loging, W.; Mandeli, J.; Thin, T.H.; Lehrer, D.; Donovan, M.; Posner, M.; et al. Therapeutic Immune Modulation against Solid Cancers with Intratumoral Poly-ICLC: A Pilot Trial. Clin. Cancer Res. 2018, 24, 4937–4948. [Google Scholar] [CrossRef]
- Korf, B. A Phase II Trial of Poly-ICLC for Progressive, Previously Treated Low-Grade Gliomas in Children and Young Adults with Neurofibromatosis Type 1. Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Novartis Pharmaceuticals. Phase II Open-Label Global Study to Evaluate the Effect of Dabrafenib in Combination with Trametinib in Children and Adolescent Patients with BRAF V600 Mutation Positive Low Grade Glioma (LGG) or Relapsed or Refractory High Grade Glioma (HGG). Available online: clinicaltrials.gov (accessed on 25 May 2021).
- Slack, F.J.; Chinnaiyan, A.M. The Role of Non-Coding RNAs in Oncology. Cell 2019, 179, 1033–1055. [Google Scholar] [CrossRef] [PubMed]
- Masliah-Planchon, J.; Pasmant, E.; Luscan, A.; Laurendeau, I.; Ortonne, N.; Hivelin, M.; Varin, J.; Valeyrie-Allanore, L.; Dumaine, V.; Lantieri, L.; et al. MicroRNAome Profiling in Benign and Malignant Neurofibromatosis Type 1-Associated Nerve Sheath Tumors: Evidences of PTEN Pathway Alterations in Early NF1 Tumorigenesis. BMC Genom. 2013, 14, 473. [Google Scholar] [CrossRef]
- Amirnasr, A.; Verdijk, R.M.; van Kuijk, P.F.; Kartal, P.; Vriends, A.L.M.; French, P.J.; van Royen, M.E.; Taal, W.; Sleijfer, S.; Wiemer, E.A.C. Deregulated MicroRNAs in Neurofibromatosis Type 1 Derived Malignant Peripheral Nerve Sheath Tumors. Sci. Rep. 2020, 10, 2927. [Google Scholar] [CrossRef]
- Chirshev, E.; Oberg, K.C.; Ioffe, Y.J.; Unternaehrer, J.J. Let-7 as Biomarker, Prognostic Indicator, and Therapy for Precision Medicine in Cancer. Clin. Transl. Med. 2019, 8, 24. [Google Scholar] [CrossRef]
- Wang, M.; Wang, Z.; Zhu, X.; Guan, S.; Liu, Z. NFKB1-MiR-612-FAIM2 Pathway Regulates Tumorigenesis in Neurofibromatosis Type 1. Vitr. Cell. Dev. Biol.-Anim. 2019, 55, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Chai, G.; Liu, N.; Ma, J.; Li, H.; Oblinger, J.L.; Prahalad, A.K.; Gong, M.; Chang, L.-S.; Wallace, M.; Muir, D.; et al. MicroRNA-10b Regulates Tumorigenesis in Neurofibromatosis Type 1. Cancer Sci. 2010, 101, 1997–2004. [Google Scholar] [CrossRef]
- Subramanian, S.; Thayanithy, V.; West, R.B.; Lee, C.-H.; Beck, A.H.; Zhu, S.; Downs-Kelly, E.; Montgomery, K.; Goldblum, J.R.; Hogendoorn, P.C.; et al. Genome-Wide Transcriptome Analyses Reveal P53 Inactivation Mediated Loss of MiR-34a Expression in Malignant Peripheral Nerve Sheath Tumours: Gene Expression Profiles of Peripheral Nerve Sheath Tumours. J. Pathol. 2010, 220, 58–70. [Google Scholar] [CrossRef] [PubMed]
- Dunbar, C.E.; High, K.A.; Joung, J.K.; Kohn, D.B.; Ozawa, K.; Sadelain, M. Gene Therapy Comes of Age. Science 2018, 359, eaan4672. [Google Scholar] [CrossRef]
- Bai, R.-Y.; Esposito, D.; Tam, A.J.; McCormick, F.; Riggins, G.J.; Wade Clapp, D.; Staedtke, V. Feasibility of Using NF1-GRD and AAV for Gene Replacement Therapy in NF1-Associated Tumors. Gene 2019, 26, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Gene Therapy Initiative. Gilbert Family Foundation. Available online: https://www.gilbertfamilyfoundation.org/gilbert-nf-research-alliance/gilbert-gene-therapy-initiative/ (accessed on 24 May 2021).
- Gutmann, D.H.; Giovannini, M. Mouse Models of Neurofibromatosis 1 and 2. Neoplasia 2002, 4, 279–290. [Google Scholar] [CrossRef]
- Hirbe, A.C.; Dahiya, S.; Friedmann-Morvinski, D.; Verma, I.M.; Clapp, D.W.; Gutmann, D.H. Spatially- and Temporally-Controlled Postnatal P53 Knockdown Cooperates with Embryonic Schwann Cell Precursor Nf1 Gene Loss to Promote Malignant Peripheral Nerve Sheath Tumor Formation. Oncotarget 2016, 7, 7403–7414. [Google Scholar] [CrossRef]
- Scherer, A.; Stephens, V.R.; McGivney, G.R.; Gutierrez, W.R.; Laverty, E.A.; Knepper-Adrian, V.; Dodd, R.D. Distinct Tumor Microenvironments Are a Defining Feature of Strain-Specific CRISPR/Cas9-Induced MPNSTs. Genes 2020, 11, 583. [Google Scholar] [CrossRef]
- Perrin, G.Q.; Li, H.; Fishbein, L.; Thomson, S.A.; Hwang, M.S.; Scarborough, M.T.; Yachnis, A.T.; Wallace, M.R.; Mareci, T.H.; Muir, D. An Orthotopic Xenograft Model of Intraneural NF1 MPNST Suggests a Potential Association between Steroid Hormones and Tumor Cell Proliferation. Lab. Investig. 2007, 87, 1092–1102. [Google Scholar] [CrossRef]
- Uthoff, J.; Larson, J.; Sato, T.S.; Hammond, E.; Schroeder, K.E.; Rohret, F.; Rogers, C.S.; Quelle, D.E.; Darbro, B.W.; Khanna, R.; et al. Longitudinal Phenotype Development in a Minipig Model of Neurofibromatosis Type 1. Sci. Rep. 2020, 10, 5046. [Google Scholar] [CrossRef] [PubMed]
- Isakson, S.H.; Rizzardi, A.E.; Coutts, A.W.; Carlson, D.F.; Kirstein, M.N.; Fisher, J.; Vitte, J.; Williams, K.B.; Pluhar, G.E.; Dahiya, S.; et al. Genetically Engineered Minipigs Model the Major Clinical Features of Human Neurofibromatosis Type 1. Commun. Biol. 2018, 1, 158. [Google Scholar] [CrossRef] [PubMed]
| Drug | Patients | Mechanism of Action | Clinical Trial |
|---|---|---|---|
| Selumetinib | Young patients with recurrent or refractory low-grade glioma | MEK inhibitor | NCT01089101 Phase I/II [134] |
| Lenalidomide | Low dose or high dose in younger patients with recurrent, refractory, or progressive pilocytic astrocytoma of optic pathway glioma | Anti-angiogenesis, tumor apoptosis, and immunomodulatory effect [135] | NCT01553149 Phase II [136] |
| Pegylated interferon alfa-2b (PEG-Intron) | Children with juvenile pilocytic astrocytoma and optic pathway gliomas | Activate (JAK/STAT) pathway | NCT02343224 Phase II [137] |
| Trametinib | Pediatric neuro-oncology patients with refractory tumors and activation of the MAPK/ERK pathway | MEK inhibitor | NCT03363217 Phase I/II [28] |
| MEK162 | Children with RAS/RAF pathway activated tumors | Selective inhibitor of MEK | NCT02285439 Phase I/II [138] |
| Vinblastine +/− Bevacizumab | Children with unresectable or progressive low-grade glioma (LGG) | Alkaloid chemotherapy and antiangiogenetic | NCT02840409 Phase II [139] |
| Pomalidomide | Younger patients with recurrent, progressive, or refractory central nervous system tumors | Anti-proliferative and immunomodulator | NCT02415153 Phase I [140] |
| Dabrafenib, Trametinib and Hydroxychloroquine | Patients with recurrent LGG or HGG with a BRAF aberration | BRAF inhibitor, MEK inhibitor, and autophagy inhibitor | NCT04201457 Phase I/II [108] |
| Poly-ICLC | Low-grade gliomas in pediatric patients with NF1 | Immune stimulant, Toll-like receptor-3 agonist [141] | NCT04544007 Phase II [142] |
| Dabrafenib and trametinib | Patients with BRAF V600 mutation positive low-grade glioma or relapsed or refractory high-grade glioma. | BRAF inhibitor and MEK inhibitor | NCT02684058 [143] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rabab’h, O.; Gharaibeh, A.; Al-Ramadan, A.; Ismail, M.; Shah, J. Pharmacological Approaches in Neurofibromatosis Type 1-Associated Nervous System Tumors. Cancers 2021, 13, 3880. https://doi.org/10.3390/cancers13153880
Rabab’h O, Gharaibeh A, Al-Ramadan A, Ismail M, Shah J. Pharmacological Approaches in Neurofibromatosis Type 1-Associated Nervous System Tumors. Cancers. 2021; 13(15):3880. https://doi.org/10.3390/cancers13153880
Chicago/Turabian StyleRabab’h, Omar, Abeer Gharaibeh, Ali Al-Ramadan, Manar Ismail, and Jawad Shah. 2021. "Pharmacological Approaches in Neurofibromatosis Type 1-Associated Nervous System Tumors" Cancers 13, no. 15: 3880. https://doi.org/10.3390/cancers13153880
APA StyleRabab’h, O., Gharaibeh, A., Al-Ramadan, A., Ismail, M., & Shah, J. (2021). Pharmacological Approaches in Neurofibromatosis Type 1-Associated Nervous System Tumors. Cancers, 13(15), 3880. https://doi.org/10.3390/cancers13153880






