Occupational Exposure to Carcinogens and Occupational Epidemiological Cancer Studies in Iran: A Review
Abstract
Simple Summary
Abstract
1. Introduction
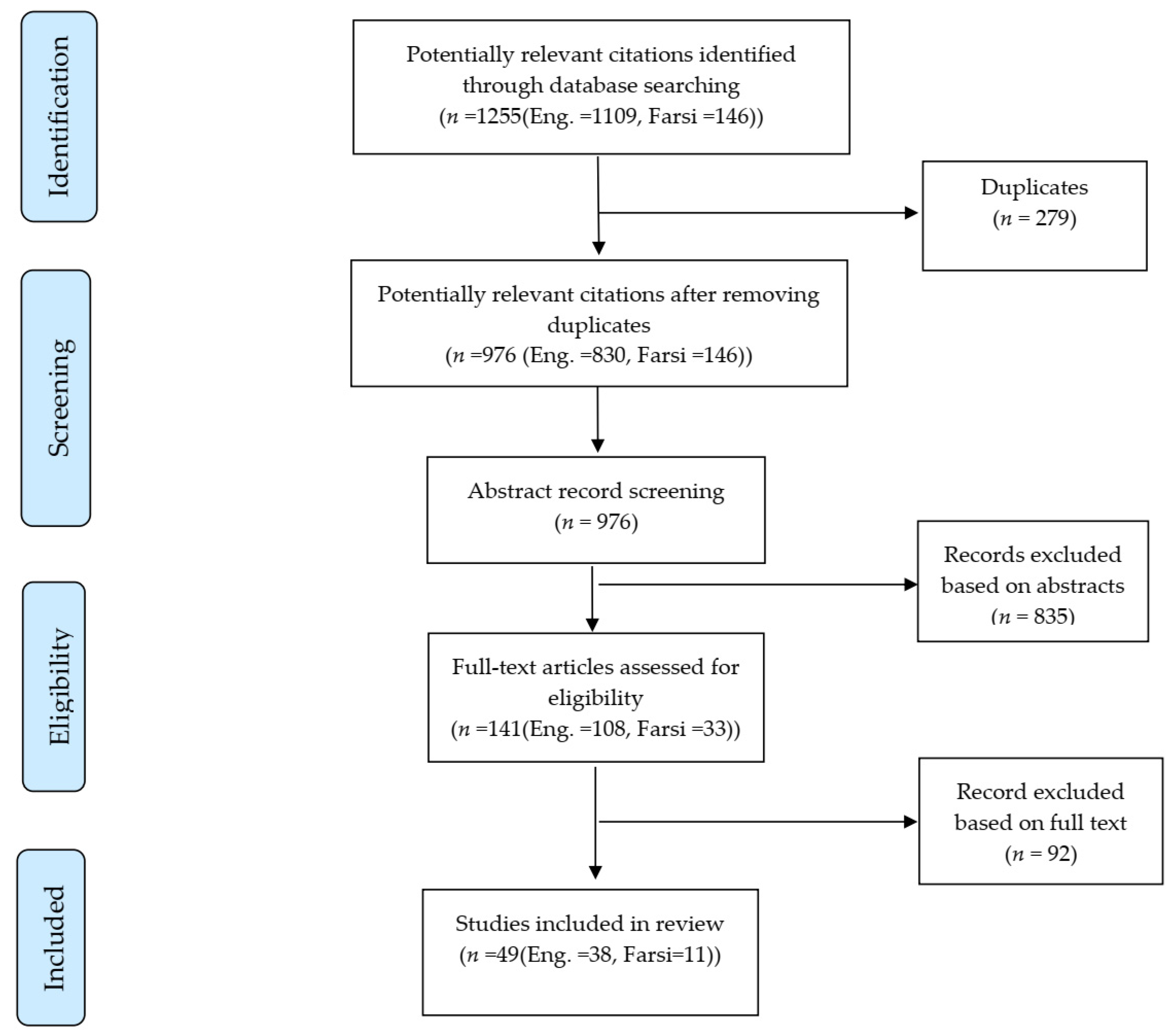
2. Materials and Methods
2.1. Literature Search and Inclusion/Exclusion Criteria
2.2. Data Extraction
3. Results
3.1. Epidemiological Studies
3.2. Exposure Monitoring Studies Including Cancer Risk Predictions
| Reference/Location/Study Year | Industry | Population/Sample Size/Description | Exposure Agents Assessed | Type of Assessment/Frequency of Measurement | Outcome/Results |
|---|---|---|---|---|---|
| Heibati/North of Iran/2016 [27] | Petroleum | 50 workers selected randomly (tanker loading workers, tank-gauging workers, drivers, firefighters, and office workers) | BTEXs 1 (benzene, toluene, ethylbenzene, and xylene) | Personal air sampling; over one 8-h work shift | Elevated ELCR 2 in tanker loading and tank-gauging workers attributable to tasks, e.g., dispensing, loading, and unloading of petrol |
| Shanh/Iran/2013–2014 [23] | Petroleum | 338 samples from 169 workers in 21 petrochemical complexes including mechanics, security, samplers, site men, technicians, laboratory staff, and office workers | VOCs 3 (benzene, epiclorohydrine, trichloroethylene, styrene, ethyl benzene, and 1,3-butadiene) | Personal air sampling; two samples taken over a work shift for each subject; each sample duration 3.5–4 h | Elevated LCR 4 for all workers attributed to VOC carcinogenic components; the highest LCR was attributable to benzene exposure |
| Sadeghi-Yarandi/Isfahan, Iran/2018 [24] | Petroleum | 150 samples from 50 workers including fire-fighters, polybutadiene latex (PBL), dryer, and coagulation workers, mechanics, electricians, packing, and laboratory workers | 1,3-butadiene | Personal air sampling; 90 min for each sample (beginning, middle, and end of work shift) | Elevated LCR for all workers, highest LCR attributable to 1,3-butadiene observed in safety and fire-fighters of petroleum complexes |
| Barkhordari/Iran/2013 [21] | Petroleum | 30 welders were assigned to different groups by exposure to Ni and Cr (VI) and also health situation (healthy and asthmatic welders) | Nickel and hexavalent chromium from welding fumes | Personal air measurement data; parameters (e.g., intensity and duration of exposure) were used from previous studies; information on daily work hours, daily tasks collected by questionnaires | Elevated ILCR 5 in exposed group in both asthmatic and healthy welders |
| Golbabaie/Iran/study year is unknown [22] | Petrochemical | Not applicable | Benzene | Exposure data used from previous prospective study in rubber industry workers; exposure index (EI) determined by experts using physical characteristics of exposure agent (e.g., the olfactory threshold value, inhalable fraction and vapor), and preventive measures including personal and environmental protection. | Elevated risk of leukemia attributable to benzene exposure in all workers |
| Harati/Iran/2016 [35] | Petrochemical | 123 samples from 60 workers | VOCs (benzene, toluene, xylene, pentane, hexane, heptane, octane, and nonane), and hydrogen sulfide (H2S) | Personal air sampling; two samples from each worker (one to measure VOCs, another to measure H2S), and 3 control samples; details on sampling duration were not provided | Elevated ELCR in workers attributable to benzene exposure in all workers |
| Hazrati/Ardabil, Iran/study year is unknown [39] | Gasoline and CNG 6 refueling stations | Samples from 24 refueling stations including 15 petrol and 9 CNG stations; the population sample size was not clarified | BTEXs | Personal air sampling over a full work shift | Elevated long-term exposure cancer risk related to BTEX compound exposure |
| Javadi/Isfahan, Iran/2016 [34] | Gasoline and CNG refueling stations | 24 samples from 12 refueling stations (12 workers, 12 stationary air sampling) | BTEXs | Personal air sampling and stationary air sampling over a full work shift | Elevated risk of cancer attributable to benzene exposure |
| Omidi/Tehran, Iran/2018 [26] | Poultry slaughterhouse | 200 samples and 40 blank samples from all workers | VOCs (benzene, toluene, ethylbenzene), and CS2 (Carbon disulphide) | Personal air sampling; 4 samples in a different hours of a full work shift; sampling repeated in 5 consecutive weeks for each worker; each sampling duration was between 50 and 90 min | Elevated LCR attributable to benzene exposure |
| Mohammadyan/Neyshabur, Iran/2017–2018 [50] | Electronic manufacturing, plastic compress unit | 141 samples from 59 workers including primary granule warehouse, plastic injection workers, shift managers, miscellaneous (forklift drivers, quality control engineers, and crew) | Styrene | Personal air sampling, details on sampling duration were not provided | Elevated LCR attributable to styrene exposure in plastic injection operators and shift supervisors |
| Mohammadyan/Neyshabur, Iran/2017–2018 [28] | Electronics manufacturing | 40 samples from 40 female soldering workers (cutting electrical wires and coating, initial soldering, voltage testing and secondary soldering workers, and shift supervisors) | Lead | Personal air sampling over a full work shift | ELCR not increased in high-risk exposed group |
| Tayfeh Rahimian/Tehran, Iran/study year is unknown [49] | Manufacturing of plastics products | 100 workers from mixing, winder, coating, finishing, rewinder, warehouse used to manufacture two plastic products | Chloride vinyl | Personal air sampling over a full work shift | Elevated ELCR attributable to duration of chloride vinyl exposure |
| Mazinani/Tehran, Iran/study year is unknown [48] | Manufacturing of plastics products | 54 samples from 4 workshops in different units including cutting, stoning, pressing, and packing | Formaldehyde | Stationary air sampling over a full work shift. Indoor air temperature, pressure, and relative humidity used in analyses | Elevated risk of cancer attributable to formaldehyde exposure in all units; the highest level of exposure was in stoning and pressing units |
| Azari/Iran/study year is unknown [25] | Manufacturing of shoes | Sample from 48 workers in 12 workshops including cutting, modelling, fitting, and finishing tasks | Benzene, toluene | Personal air sampling, once a month during 3 consecutive months, details on sampling duration were not provided | Elevated risk of leukemia attributable to benzene and toluene exposure in shoemakers; the level of exposure was higher than the threshold limit value (TLV) |
| Sanjari/Iran/study year is unknown [51] | Aluminum rolling | 103 samples from different units including slabbing, production, painting, and washing workshops | Chemical exposure including aluminum products, silica, rock wool, iron oxide, manganese, sulfuric acid, benzene, ethylbenzene, toluene and xylene | Stationary air sampling; details on sampling duration were not provided; indoor air temperature, pressure, and humidity used in analyses | Elevated ELCR attributable to benzene exposure; level of exposure to sulfuric acid in washing lines and manganese in the manufacturing unit were higher than in other units |
| Zarei/Tehran, Iran/2017 [40] | Foundry | Sampling of breathing zone of 55 workers from different units including machine operator, painting, furnace, and cleaning; information on number of samples was not provided | Crystalline silica | Stationary air sampling, 4 h during a work shift | Elevated lung ELCR attributable to crystalline silica exposure; All workers exposed to a higher level than the definite acceptable limit recommended by OSHA 7 |
| Omidianidost/Tehran, Iran/2011 [52] | Foundry | 80 samples of 80 workers from workshops used for 29 small foundries including 10 iron cast, 3 brass, and an aluminum foundry | Crystalline silica | Personal air sampling over a full work shift | Elevated risk of lung cancer mortality; 50 percent of workers exposed to greater than the acceptable threshold limit value by OSHA |
| Omidi/Iran/study year is unknown [36] | Steel manufacturing | 50 samples from the breathing zone of 372 workers in energy and biochemistry units, benzol refinement and experimental furnace units | BTEXs | Stationary air sampling over a full work shift | Elevated cancer risk attributable to benzene exposure; exposure to BTEX components in the benzol refinement unit was higher than in other units |
| Normohammadi/Tehran, Iran/2010–2011 [42] | Construction | 60 demolition workers from 4 demolition sites; 15 samples from each site | Crystalline silica | Personal air sampling over a full work shift, meteorological parameters including air temperature and wind speed were used in the analysis | Elevated ELCR of lung cancer attributable to crystalline silica exposure in workers |
| Tavakol/Tehran, Iran/study year is unknown [43] | Construction | 85 samples from 85 construction workers including supervisors, stonemason, batching and concrete workers, and labors | Crystalline silica | Personal air sampling over a full work shift | Elevated ELCR of lung cancer attributable to crystalline silica exposure in construction work; batching and concrete workers had the highest average exposure that was greater than the threshold limit recommended by OSHA |
| Moghadam/Neyshabour, Iran/2015 [53] | Concrete manufacturing | Sampling of breathing zone of 72 workers from autoclave units, wing tube and cutter line, mixing, packing, and quality control units; information on number of samples was not provided | Crystalline silica | Stationary air sampling; information on sampling duration was not provided | Elevated ELCR for lung cancer attributable to crystalline silica exposure |
| Azari/Tehran, Iran/study year is unknown [44] | Stone cutting and milling, foundry work, glass manufacturing, asphalt, construction, sand and gravel mining, sand blast, ceramics, bricks and cement | 200 workers from 50 workplaces (4 workers in each) including stone cutting and milling, foundry, glass manufacturing, asphalt preparation, ceramic, brick and concrete manufacturing, and construction | Crystalline silica | Personal air sampling over a full work shift | Elevated lung ELCR attributable to cumulative exposure of crystalline silica (direct correlation between the level of exposure and lung cancer risk) |
| Mohammadi/Markazi, Iran/2015 [41] | Ceramic pin insulator manufacturing | 60 samples from 5 units including pressing, production, coating, furnace, and packing (12 samples in each unit) | Crystalline silica | Stationary air sampling; information on sampling duration was not provided | The highest mortality risk of lung cancer attributable to crystalline silica exposure was estimated for furnace workers |
| Yahyaei/Rasht, Iran/2018 [47] | Hospital | 65 employees in pathology labs including pathologist, lab technician, office worker, service workers | Formaldehyde | Personal air sampling during direct exposure to formaldehyde (8:00 and 12:00 a.m.); 25 min for each task; stationary air sampling during a full work shift | Elevated individual lifetime cancer risk 100–1000 times higher than the acceptable cancer risk in all exposed laboratory staff; exposure level in all staff members was higher than the acceptable level by OSHA |
| Pourtaghi/Tehran, Iran/2018 [46] | Hospital | 68 samples from the breathing zone of 72 hospital staff | Formaldehyde | Stationary air sampling during one full work shift | Elevated LCR attributable to intensity of formaldehyde exposure greater than the recommended acceptable limit by OSHA |
| Jalali/Iran/2019 [45] | Hospital | Sampling of breathing zone of 60 pathology laboratory staff members; information on number of samples was not provided | Formaldehyde | Stationary air sampling during one full work shift | Highest LCR attributable to formaldehyde in lab technicians |
| Zarei/Tehran/Iran/2010 [29] | Brake shoe and clutch disk manufacturing | 61 workers including weighing, mixing, pressing, and finishing occupations | Asbestos | Personal air sampling; 4 h over a full work shift | Elevated risk of lung cancer ELCR attributable to cumulative exposure of asbestos; exposure levels for all workers far greater than the occupational exposure limits recommended by OSHA |
| Jafari/Isfahan, Iran/study year is unknown [54] | Asbestos-cement products manufacturing | 97 workers from 4 units including milling, cutting, and cutting | Asbestos | Personal air sampling; samples collected from different units of the factory over a full work shift | Elevated risk of lung, mesothelioma, and gastrointestinal cancer mortality after 20 years of exposure; greater risk in the dry cutting unit than the wet cutting unit |
| Harati/Iran/2015 [37] | Automobile manufacturing | 46 samples from 20 workers | BTEXs, Silica | Personal air sampling (2 times for each worker over a full work shift), and 6 stationary air samples over a full work shift | Elevated hematological cancer risk corresponding to cumulative exposure to benzene and crystalline silica |
| Dehghani/Iran/2016 [38] | Automobile manufacturing | 34 samples from breathing zones of workers: cabin maker, pre-paint, and painting units | BTEXs | Stationary air sampling over a full work shift | Elevated risk of cancer in painting unit workers attributable to benzene and ethyl benzene with 30 years of exposure |
| Baghani/Ardabil, Iran/2017 [31] | Beauty salons | 50 beauty salons across the Ardabil Province | BTEXs | Stationary air sampling; indoor air temperature, pressure, and relative humidity were used in the analysis, samples taken during the afternoon (14:00 to 19:00) | Elevated LTCR 8 attributable to BTEX components |
| Hadei/Tehran, Iran/2016–2017 [30] | Beauty salons | 360 samples from 20 beauty salons, 180 samples each for indoor and outdoor; (3 samples for each component including BTEXs, formaldehyde, and acetaldehyde) | BTEXs, formaldehyde, and acetaldehyde | Stationary air sampling; over a full work shift during 3 consecutive months; air temperature, pressure, and relative humidity were used in the analysis | Elevated risk of cancer attributable to benzene, formaldehyde, and acetaldehyde exposure; exposure to different components affected by tasks: benzene and toluene (hair dying), formaldehyde (hair style and nail treatments), and xylene (hair styling) |
| Hazrati/Ardabil, Iran/study year is unknown [32] | Waterpipe café | 87 samples from 81 waterpipe cafés | BTEXs | Stationary air sampling; each sample duration lased for 50 min; sampling from 14:00 to 19:00; 6 additional samples taken directly from the smoke mainstream of the waterpipe (4 from fruit flavored tobacco and 2 from regular tobacco). | Elevated long-term exposure cancer risk attributable to benzene exposure |
| Rostami/Ardabil, Iran/2018 [33] | Waterpipe café | 51 samples from 51 waterpipe cafés | PAHs 9 | Stationary air sampling; from breathing zone of smokers and employees; air temperature, pressure, and relative humidity were used in the analysis | Elevated risk of cancer attributable to PAH component inhalation |
3.3. Burden of Cancer Studies
3.4. Publications over Time and Location
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Disclaimer
References
- Torbat, A.E. Industrialization and dependency: The case of Iran. ECO Econ. J. 2010, 2, 3. [Google Scholar]
- Anttila, S.L.; Boffetta, P. Occupational Cancers; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Ge, C.B.; Friesen, M.C.; Kromhout, H.; Peters, S.; Rothman, N.; Lan, Q.; Vermeulen, R. Use and Reliability of Exposure Assessment Methods in Occupational Case–Control Studies in the General Population: Past, Present, and Future. Ann. Work. Expo. Health 2018, 62, 1047–1063. [Google Scholar] [CrossRef] [PubMed]
- Fritschi, L. OccIDEAS—Occupational exposure assessment in community-based studies. Occup. Med. 2019, 69, 156–157. [Google Scholar] [CrossRef] [PubMed]
- Emami, H.; Ilbeigi, A.; Khodadad, K. An Overview of Asbestos and Malignant Pleural Mesothelioma: An Iranian Perspective. Asian Pac. J. Cancer Prev. 2017, 18, 2619–2624. [Google Scholar] [CrossRef]
- Olsson, A.; Kromhout, H. Occupational cancer burden: The contribution of exposure to process-generated substances at the workplace. Mol. Oncol. 2021, 15, 753–763. [Google Scholar] [CrossRef]
- Arroyave, W.D.; Mehta, S.S.; Guha, N.; Schwingl, P.; Taylor, K.W.; Glenn, B.; Radke, E.G.; Vilahur, N.; Carreón, T.; Nachman, R.M. Challenges and recommendations on the conduct of systematic reviews of observational epidemiologic studies in environmental and occupational health. J. Expo. Sci. Environ. Epidemiol. 2021, 31, 21–30. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; PRISMA-P Group. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef]
- Zafarghandi, M.R.; Soroush, M.R.; Mahmoodi, M.; Naieni, K.H.; Ardalan, A.; Dolatyari, A.; Falahati, F.; Mirmohammadkhani, M.; Mousavi, B.; Ghanei, M. Incidence of cancer in Iranian sulfur mustard exposed veterans: A long-term follow-up cohort study. Cancer Causes Control. 2012, 24, 99–105. [Google Scholar] [CrossRef]
- Khoubi, J.; Pourabdian, S.; Mohebbi, I.; Tajvidi, M.; Zaroorian, O.; Giahi, O. Association between the high risk occupations and bladder cancer in Iran: A case-control study. Int. J. Occup. Med. Environ. Health 2013, 26, 205–213. [Google Scholar] [CrossRef]
- Akbari, H.; Akbari, H.; Aminian, O.; Chavoshi, F.; Mohseni, H.; Saburi, A. Occupational risk of bladder cancer among Iranian male workers. Urol. Ann. 2014, 6, 135–138. [Google Scholar] [CrossRef]
- Farzaneh, F.; Mehrparvar, A.H.; Lotfi, M.H. Occupations and the Risk of Bladder Cancer in Yazd Province: A Case-Control Study. Int. J. Occup. Environ. Med. 2017, 8, 191–198. [Google Scholar] [CrossRef][Green Version]
- Ghadimi, T.; Gheitasi, B.; Nili, S.; Karimi, M.A.; Ghaderi, E. Occupation, smoking, opium, and bladder cancer: A case–control study. South Asian J. Cancer 2015, 4, 111–114. [Google Scholar] [CrossRef]
- Tajvidi, M.; Najafi, S.; Molana, S.H.; Gharib, S.; Hemati, S. The Relationship of kidney cancer and occupation in Isfahan province, Iran; a case-control study. J. Isfahan Med Sch. 2013, 31, 305–313. [Google Scholar]
- Aminian, O.; Abedi, A.; Chavoshi, F.; Ghasemi, M.; Najarkolaei, F.R. Evaluation of occupational risk factors in non-Hodgkin lymphoma and Hodgkin’s disease in Iranian men. Iran. J. Cancer Prev. 2012, 5, 189–193. [Google Scholar]
- Zakerinia, M.; Namdari, M.; Amirghofran, S. The Relationship between Exposure to Pesticides and the Occurrence of Lymphoid Neoplasm. Iran. Red Crescent Med. J. 2012, 14, 337–344. [Google Scholar]
- Amizadeh, M.; Safari-Kamalabadi, M.; Askari-Saryazdi, G.; Amizadeh, M.; Reihani-Kermani, H. Pesticide Exposure and Head and Neck Cancers: A Case-Control Study in an Agricultural Region. Iran. J. Otorhinolaryngol. 2017, 29, 275–285. [Google Scholar] [PubMed]
- Hosseini, M.; Naghan, P.A.; Karimi, S.; Seyed-Alinaghi, S.; Bahadori, M.; Khodadad, K.; Mohammadi, F.; Keynama, K.; Masjedi, M. Environmental risk factors for lung cancer in Iran: A case-control study. Int. J. Epidemiol. 2009, 38, 989–996. [Google Scholar] [CrossRef]
- Aghilinejad, M.; Kabir-Mokamelkhah, E.; Imanizade, Z.; Danesh, H. Occupational Class Groups as a Risk Factor for Gastrointestinal Cancer: A Case-Control Study. Int. J. Occup. Environ. Med. 2017, 8, 21–31. [Google Scholar] [CrossRef][Green Version]
- Rafeemanesh, E.; Taghizadekermani, A.; Khajedaluee, M.; Ahmadi, F. Evaluation of Breast Cancer Risk in Relation to Occupation. Middle East J. Cancer 2018, 9, 186–194. [Google Scholar]
- Barkhordari, A.; Sakhvidi, M.J.Z.; Sakhvidi, F.Z.; Halvani, G.; Firoozichahak, A.; Shirali, G. Cancer Risk Assessment in Welder’s Under Different Exposure Scenarios. Iran. J. Public Health 2014, 43, 666–673. [Google Scholar]
- Golbabaie, F.E.; Azari, M.; Jahangiri, M.; Rahimi, R.; Shahtaheri, J. Health risk assessment of chemical pollutants in a petrochemical complex. Iran Occup. Health 2012, 9, 11–21. [Google Scholar]
- Bahrami, A.; Ghorbani-Shahna, F.; Rahimnejad, S.; Farhadian, M. Risk Assessment of Workers’ Exposure to Volatile Organic Compounds in the Air of a Petrochemical Complex in Iran. Indian J. Occup. Environ. Med. 2017, 21, 121–127. [Google Scholar] [CrossRef]
- Sadeghi-Yarandi, M.; Karimi, A.; Ahmadi, V.; Sajedian, A.A.; Soltanzadeh, A.; Golbabaei, F. Cancer and non-cancer health risk assessment of occupational exposure to 1,3-butadiene in a petrochemical plant in Iran. Toxicol. Ind. Health 2020, 36, 960–970. [Google Scholar] [CrossRef] [PubMed]
- Azari, M.R.; Hosseini, V.; Jafari, M.J.; Soori, H.; Asadi, P.; Mousavion, S.M.A. Evaluation of Occupational Exposure of Shoe Makers to Benzene and Toluene Compounds in Shoe Manufacturing Workshops in East Tehran. Tanaffos 2012, 11, 43–49. [Google Scholar]
- Omidi, F.; Dehghani, F.; Fallahzadeh, R.A.; Miri, M.; Taghavi, M.; Eynipour, A. Probabilistic risk assessment of occupational exposure to volatile organic compounds in the rendering plant of a poultry slaughterhouse. Ecotoxicol. Environ. Saf. 2019, 176, 132–136. [Google Scholar] [CrossRef]
- Heibati, B.; Pollitt, K.J.G.; Karimi, A.; Yazdani-Charati, J.; Ducatman, A.; Shokrzadeh, M.; Mohammadyan, M. BTEX exposure assessment and quantitative risk assessment among petroleum product distributors. Ecotoxicol. Environ. Saf. 2017, 144, 445–449. [Google Scholar] [CrossRef]
- Mohammadyan, M.; Moosazadeh, M.; Khanjani, N.; Moghadam, S.R. Quantitative and semi-quantitative risk assessment of occupational exposure to lead among electrical solderers in Neyshabur, Iran. Environ. Sci. Pollut. Res. 2019, 26, 31207–31214. [Google Scholar] [CrossRef] [PubMed]
- Azari, M.R.; Nasermoaddeli, A.; Movahadi, M.; Mehrabi, Y.; Hatami, H.; Soori, H.; Moshfegh, E.; Ramazni, B. Risk assessment of lung cancer and asbestosis in workers exposed to asbestos fibers in brake shoe factory in Iran. Ind. Health 2010, 48, 38–42. [Google Scholar] [CrossRef]
- Hadei, M.; Hopke, P.K.; Shahsavani, A.; Moradi, M.; Yarahmadi, M.; Emam, B.; Rastkari, N. Indoor concentrations of VOCs in beauty salons; association with cosmetic practices and health risk assessment. J. Occup. Med. Toxicol. 2018, 13, 1–9. [Google Scholar] [CrossRef]
- Baghani, A.N.; Rostami, R.; Arfaeinia, H.; Hazrati, S.; Fazlzadeh, M.; Delikhoon, M. BTEX in indoor air of beauty salons: Risk assessment, levels and factors influencing their concentrations. Ecotoxicol. Environ. Saf. 2018, 159, 102–108. [Google Scholar] [CrossRef]
- Hazrati, S.; Rostami, R.; Fazlzadeh, M. BTEX in indoor air of waterpipe cafés: Levels and factors influencing their concentrations. Sci. Total. Environ. 2015, 524-525, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Rostami, R.; Zarei, A.; Saranjam, B.; Ghaffari, H.R.; Hazrati, S.; Poureshgh, Y.; Fazlzadeh, M. Exposure and risk assessment of PAHs in indoor air of waterpipe cafés in Ardebil, Iran. Build. Environ. 2019, 155, 47–57. [Google Scholar] [CrossRef]
- Javadi, I.M.; Heybati, B.; Elyasi, S. Occupational exposure of shahindej county refueling stations workers to BTEX compounds, in 2016. J. Res. Environ. Health 2017, 3, 74–83. [Google Scholar]
- Harati, B.; Shahtaheri, S.J.; Yousefi, H.A.; Harati, A.; Askari, A.; Abdolmohamadi, N. Cancer Risk Assessment for Workers Exposed to Pollution Source, a Petrochemical Company, Iran. Iran. J. Public Health 2020, 49, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Omidi, F.; Fallahzadeh, R.A.; Dehghani, F.; Harati, B.; Chamgordani, S.B.; Gharibi, V. Carcinogenic and non-carcinogenic risk assessment of exposure to volatile organic compounds (BTEX) using Monte-Carlo simulation technique in a steel industry. J. Health Saf. Work. 2018, 8, 299. [Google Scholar]
- Harati, B.; Shahtaheri, S.J.; Karimi, A.; Azam, K.; Ahmadi, A.; Rad, M.A.; Harati, A. Risk assessment of chemical pollutants in an automobile manufacturing. J. Health Saf. Work. 2017, 7, 121. [Google Scholar]
- Dehghani, F.; Golbabaei, F.; Zakerian, S.A.; Omidi, F.; Mansournia, M.A. Health risk assessment of exposure to volatile organic compounds (BTEX) in a painting unit of an automotive industry. J. Health Saf. Work. 2018, 8, 55. [Google Scholar]
- Hazrati, S.; Rostami, R.; Fazlzadeh, M.; Pourfarzi, F. Benzene, toluene, ethylbenzene and xylene concentrations in atmospheric ambient air of gasoline and CNG refueling stations. Air Qual. Atmos. Health 2016, 9, 403–409. [Google Scholar] [CrossRef]
- Zarei, F.; Azari, M.R.; Salehpour, S.; Khodakarim, S.; Kalantary, S.; Tavakol, E. Exposure assessment of core making workers to respirable crystalline silica dust. J. Health Saf. Work. 2017, 7, 1. [Google Scholar]
- Mohammadi, H.; Golbabaei, F.; Dehghan, S.F.; Normohammadi, M. Occupational exposure assessment to crystalline silica in an insulator industry: Determination the risk of mortality from silicosis and lung cancer. J. Health Saf. Work. 2017, 7, 45. [Google Scholar]
- Nourmohammadi, M.; Kakooei, H.; Omidi, L.; Yari, S.; Alimi, R. Risk Assessment of Exposure to Silica Dust in Building Demolition Sites. Saf. Health Work. 2016, 7, 251–255. [Google Scholar] [CrossRef]
- Tavakol, E.R.A.; Salehpour, S.; Khodakarim, S. Determination of Construction Workers’ Exposure to Respirable Crystalline Silica and Respirable Dust. J. Saf. Promot. Inj. Prev. 2016, 3, 263–270. [Google Scholar]
- Azari, M.R.; Rokni, M.; Salehpour, S.; Mehrabi, Y.; Jafari, M.J.; Moaddeli, A.N.; Movahedi, M.; Ramezankhani, A.; Hatami, H.; Mosavion, M.A.; et al. Risk assessment of workers exposed to crystalline silica aerosols in the east zone of Tehran. Tanaffos 2009, 8, 43–50. [Google Scholar]
- Jalali, M.; Moghadam, S.R.; Baziar, M.; Hesam, G.; Moradpour, Z.; Zakeri, H.R. Occupational exposure to formaldehyde, lifetime cancer probability, and hazard quotient in pathology lab employees in Iran: A quantitative risk assessment. Environ. Sci. Pollut. Res. 2021, 28, 1878–1888. [Google Scholar] [CrossRef]
- Pourtaghi, G.; Bahrami, A.; Shaban, I.; Taheri, E.; PirMohammadi, Z. Exposure Risk Assessment of Formaldehyde in Four Military Hospitals of Tehran, Iran. J. Occup. Hyg. Eng. 2020, 7, 21–30. [Google Scholar]
- Yahyaei, E.; Majlesi, B.; Joubani, M.N.; Pourbakhshi, Y.; Ghiyasi, S.; Rastani, M.J.; Heidari, M. Occupational Exposure and Risk Assessment of Formaldehyde in the Pathology Departments of Hospitals. Asian Pac. J. Cancer Prev. 2020, 21, 1303–1309. [Google Scholar] [CrossRef] [PubMed]
- Mazinani, M.V.S.; Khodakarim, S.; Kheiry, H.; Mosavi Mehraban, A.A.; Zendehdel, R. Evaluation of occupational exposure to formaldehyde from selected manufactures of dinnerware melamine production in Tehran. J. Saf. Promot. Inj. Prev. 2015, 3, 111–116. [Google Scholar]
- Tayfeh Rahimian, R.R.A.; Jafari, M.J.; Soori, H.; Saranjam, B.; Tavakol, E.; Karimi, M. Evaluation of Occupational Exposure with Vinyl Chloride Monomer in the Plastic Production Industry in Tehran. J. Saf. Promot. Inj. Prev. 2014, 1. [Google Scholar]
- Mohammadyan, M.; Moosazadeh, M.; Borji, A.; Khanjani, N.; Moghadam, S.R.; Moghadam, A.M.B. Health risk assessment of occupational exposure to styrene in Neyshabur electronic industries. Environ. Sci. Pollut. Res. 2019, 26, 11920–11927. [Google Scholar] [CrossRef] [PubMed]
- Sanjari, A.; Saeedi, R.; Khaloo, S.S. Semi-quantitative health risk assessment of exposure to chemicals in an aluminum rolling mill. Int. J. Occup. Saf. Ergon. 2021, 27, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Omidianidost, A.; Ghasemkhani, M.; Kakooei, H.; Shahtaheri, S.J.; Ghanbari, M. Risk Assessment of Occupational Exposure to Crystalline Silica in Small Foundries in Pakdasht, Iran. Iran. J. Public Health 2016, 45, 70–75. [Google Scholar]
- Moghadam, S.R.; Khanjani, N.; Mohamadyan, M.; Emkani, M.; Yari, S.; Tizabi, M.N.L.; Ganjali, A. Changes in Spirometry Indices and Lung Cancer Mortality Risk Estimation in Concrete Workers Exposed io Crystalline Silica. Asian Pac. J. Cancer Prev. 2020, 21, 2811–2817. [Google Scholar] [CrossRef]
- Jafari, M.J.; Karimi, A.; Mohammad Bardshahi, A. Prediction of cancer mortality by evaluation of asbestos fibers concentrations in an asbestos-cement products factory. Iran. J. Environ. Health Sci. Eng. 2010, 7, 165–172. [Google Scholar]
- Mosavi-Jarrahi, A.; Mohagheghi, M.; Kalaghchi, B.; Mousavi-Jarrahi, Y.; Noori, M.K. Estimating the incidence of lung cancer attributable to occupational exposure in Iran. Popul. Health Metrics 2009, 7, 7. [Google Scholar] [CrossRef] [PubMed]
- Mosavi-Jarrahi, A.; Mohagheghi, M.A.; Kalaghchi, B.; Mousavi-Jarrahi, Y.; Kolahi, A.-A.; Noori, M.K. Estimating the incidence of leukemia attributable to occupational exposure in Iran. Asian Pac. J. Cancer Prev. 2009, 10, 67–70. [Google Scholar] [PubMed]
- Abtahi, M.; Koolivand, A.; Dobaradaran, S.; Yaghmaeian, K.; Khaloo, S.S.; Jorfi, S.; Keshmiri, S.; Nafez, A.H.; Saeedi, R. National and subnational mortality and disability-adjusted life years (DALYs) attributable to 17 occupational risk factors in Iran, 1990–2015. Environ. Res. 2018, 165, 158–175. [Google Scholar] [CrossRef] [PubMed]
- Kauppinen, T.; Toikkanen, J.; Pedersen, D.; Young, R.; Ahrens, W.; Boffetta, P.; Hansen, J.; Kromhout, H.; Blasco, J.M.; Mirabelli, D.; et al. Occupational exposure to carcinogens in the European Union. Occup. Environ. Med. 2000, 57, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Kassebaum, N.J.; Arora, M.; Barber, R.M.; Bhutta, Z.; Brown, J.; Carter, A.; Casey, D.C.; Charlson, F.J.; Coates, M.; Coggeshall, M.; et al. Global, regional, and national disability-adjusted life-years (DALYs) for 315 diseases and injuries and healthy life expectancy (HALE), 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1603–1658. [Google Scholar] [CrossRef]
- GBD 2013 Risk Factors Collaborators; Forouzanfar, M.H.; Alexander, L.; Anderson, H.R.; Bachman, V.F.; Biryukov, S.; Brauer, M.; Burnett, R.; Casey, D.; Coates, M.; et al. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks in 188 countries, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 386, 2287–2323. [Google Scholar] [CrossRef]
- Maddahinasab, M.; Momenirad, A.; Tajarlou, R.; Razavi, M. Managing confidential information on petroleum projects in the case of third parties. Energy Policy Stud. 2019, 2, 49–61. [Google Scholar]
- Baan, R.; Grosse, Y.; Straif, K.; Lauby-Secretan, B.; El Ghissassi, F.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Freeman, C.; Galichet, L.; et al. A review of human carcinogens—Part F: Chemical agents and related occupations. Lancet Oncol. 2009, 10, 1143–1144. [Google Scholar] [CrossRef]
- Kromhout, H.; Oostendorp, Y.; Heederik, D.; Boleij, J.S.M. Agreement between qualitative exposure estimates and quantitative exposure measurements. Am. J. Ind. Med. 1987, 12, 551–562. [Google Scholar] [CrossRef] [PubMed]
- Kromhout, H. Design of measurement strategies for workplace exposures. Occup. Environ. Med. 2002, 59, 349–354. [Google Scholar] [CrossRef]
- Vainio, H.; Zendehdel, K. Occupational/Environmental Cancer at the International Congress on Cancer prevention & Early Detection: A Workshop Report. Basic Clin. Cancer Res. 2017, 9, 40–44. [Google Scholar]
- Aubrun, J.; Binet, S.; Bozec, C.; Brochard, P.; Dimerman, S.; Fontaine, B.; Guénel, P.; Luce, D.; Martinet, Y.; Moulin, J. Occupational cancer in France: Epidemiology, toxicology, prevention, and compensation. Environ. Health Perspect. 1999, 107, 245–252. [Google Scholar]
- Poustchi, H.; Eghtesad, S.; Kamangar, F.; Etemadi, A.; Keshtkar, A.-A.; Hekmatdoost, A.; Mohammad, H.S.; Mahmoudi, Z.; Shayanrad, A.; Roozafzai, F.; et al. Prospective Epidemiological Research Studies in Iran (the PERSIAN Cohort Study): Rationale, Objectives, and Design. Am. J. Epidemiol. 2018, 187, 647–655. [Google Scholar] [CrossRef]
- Hadji, M.; Marzban, M.; Gholipour, M.; Rashidian, H.; Naghibzadeh-Tahami, A.; Haghdoost, A.; Rezaianzadeh, A.; Rahimi-Movaghar, A.; Moradi, A.; Seyyedsalehi, M.; et al. National Study of Opium and Cancer in Iran (IROPICAN): Study Protocol and Results of the Pilot Phase. J. Glob. Oncol. 2018, 4, 192. [Google Scholar] [CrossRef]
- Blair, A.; Marrett, L.; Freeman, L.B. Occupational cancer in developed countries. Environ. Health 2011, 10, S9. [Google Scholar] [CrossRef]
| Study Characteristics | Cohort n = 1 (%) | Case-Control n = 11 (%) | Exposure Monitoring n = 34 (%) | Cancer Burden Studies n = 3 (%) |
|---|---|---|---|---|
| Direct exposure assessments | - | 0 (0.0) | 32 (94.1) | - |
| Personal Air Sampling | - | - | 16 (47.0) | - |
| Stationary Air Monitoring | - | - | 13(38.2) | - |
| Stationary/Personal Air Sampling | - | - | 3(8.8) | - |
| Indirect Exposure Assessment Methods: | 1 (100) | 11 (100.0) | 2 (5.8) | 3 (100) |
| Self-report of job or exposure history | - | 11 (100.0) | - | - |
| Available databases | - | - | 1 (2.9) | 3 (100) |
| Expert Assessment | - | - | 1 (2.9) | - |
| Not applicable | 1 (100) | - | - | - |
| Cancer Outcomes | ||||
| Cancer, general 1 | 1 (100) | - | 24 (70.5) | 1 (33.3) |
| Lung cancer | - | 1 (9.0) | 8 (24.3) | - |
| Bladder and urinary tract cancer | - | 5 (45.4) | - | 1 (33.3) |
| Hematological cancer | - | 2 (18.1) | 2 (8.1) | 1 (33.3) |
| Head and neck cancer | - | 1 (9.0) | - | - |
| Breast cancer | - | 1 (9.0) | - | - |
| Gastrointestinal cancer | - | 1 (9.0) | - | - |
| Reference/Location/Study Year | Cancer Sites | Population Size/Description | Source of Exposure Assessment | Exposure Duration Measure | Covariates Controlled for in Modelling | Outcome (Risk of Cancer) |
|---|---|---|---|---|---|---|
| Prospective cohort | ||||||
| Zafarghandi/Iran/1984–2010 [9] | Cancer | 7570; Male veterans, Exposed to sulfur mustard during the Iran–Iraq war (1984–1987); 7592; Male veterans, unexposed to sulfur mustard (instead, e.g., physical injuries). | Documented medical records on at least one acute exposure while the participant presented in the battlefield between 1984 and 1987 | Single exposure to sulfur mustard during the war | Stratified by age, smoking status, educational level, marital status | Yes |
| Case-control | ||||||
| Khoubi/Isfahan, Iran/2004–2009 [10] | Bladder | Cases: 300; bladder cancer patient recruited from registry Controls: 500; population-based controls | Questionnaire; Phone/face-to-face interview; Collected data on industry title, duration, hours of work per day, self-report of exposures | Lifetime occupation history: Jobs held > 6 months | Age, sex, smoking status (never/current/former smoker, duration of smoking (year), average number of cigarettes per day) | Yes |
| Aminian/Tehran, Iran/2007–2009 [11] | Bladder | Cases: 160; Male- Histologically confirmed Controls: 160 Male without occupational exposure to the chemicals (source of controls recruitment is unclear) | Study specific questionnaire; Face-to-face interview; Collected data on job title, duration, exposure to the specific chemicals in each job, history of cancer occurrence in coworkers | Current and former jobs | Sex | Yes |
| Farzaneh/Yazd, Iran/2009–2013 [12] | Bladder | Cases: 200; Histologically confirmed Controls: 200 healthy neighbors | Questionnaire; Face-to-face interview; Collected data on job titles | Lifetime occupation history: jobs held > 1 year | Age, sex, family history of bladder cancer, chronic urinary tract infections (times per year), kidney and bladder stones, hair dyeing, and educational level | Yes |
| Ghadimi/Kurdestan, Iran/2012–2015 [13] | Bladder | Cases: 152; Histologically confirmed recruited from cancer registries Controls: 152, Hospital based | Semi-structured questionnaire; Interview; Collected data on job titles, and tasks | Last 20 years jobs | Age, sex, and place of residence, univariable analysis | Yes |
| Tajvidi/Isfahan, Iran/2001–2010 [14] | Kidney | Cases: 200; kidney cancer cases recruited from cancer registry Controls: 400; healthy population based | Semi-structured questionnaire; Collected data on job title, and self-report of exposures | Not clear | Age, sex | Yes |
| Aminian/Tehran, Iran/2011–2015 [15] | Non- Hodgkin/Hodgkin’s lymphoma | Cases: 150; male; Histologically confirmed Controls: 150; relative controls (case’s brother or close relative) | Semi-structured questionnaire; Interview; Collected data on job title, duration, exposure to the specific chemicals in each job, history of cancer occurrence in coworkers | Current and former jobs: Jobs held > 1 year | Age, sex | Yes |
| Zakerinia/Fars, Iran/2007–2008 [16] | Malignant lymphoma | Cases: 200; Histologically confirmed Controls: 200; Hospital based | Semi-structured questionnaire; Face-to-face interview; Collected data on job history, specific exposures for each job, a question on extra jobs in farming including pesticides exposure (herbicides, fungicides, insecticides), reason for exposure, duration | Lifetime occupation history: Jobs held > 1 year | Age, sex, center | Non-Hodgkin lymphoma: Yes Hodgkin lymphoma: No |
| Amizadeh/Not clear [17] | Head and neck | Cases: 31; Histologically confirmed Controls: 32; Hospital based | Structured questionnaire; Face-to-face interview; Collected data on type of crops, tasks, duration, farming surface size, name of pesticide, frequency of apply per year, and methods, personal protective equipment; biomonitoring (residual pesticide was extracted from adipose tissue) | At least 1 year of agriculture | Age, sex, smoking status | Yes |
| Hosseini/Tehran, Iran/2002–2005 [18] | Lung | Cases: 242; primary cases; Histologically confirmed Controls: 484; Hospital based (242; Healthy visitors and 242; patients except oncology ward patients) | Structured questionnaire; Face-to-face interview; Collected data on workplace conditions, exposure to suspected occupational lung carcinogens | Not clear | Age, sex, and-place of resident | Yes |
| Aghilinejad/Tehran, Iran/2014–2015 [19] | Gastrointestinal | Cases: 243; Male; Histologically confirmed Controls: 243; Male; Hospital based (cancer patient other than gastrointestinal cancer) | Questionnaire; Interview; In the method the author stated, “occupational history” but no details provided on the collected data | Childhood until 5 years before cancer diagnosis | Age | Yes |
| Rafeemanesh/Khorasan-Razavi, Iran/2010–2014 [20] | Breast | Cases: 104; Histologically confirmed (employed women) Controls: 112; Women; Healthy controls (who referred to the health care centers to receive routine examination); Employed women | Questionnaire; Face-to-face Interview; Collected data on recent job title; In the methods “occupational exposures” are stated, with no details provided | Not clear | Not clear | Yes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hosseini, B.; Hall, A.L.; Zendehdel, K.; Kromhout, H.; Onyije, F.M.; Moradzadeh, R.; Zamanian, M.; Schüz, J.; Olsson, A. Occupational Exposure to Carcinogens and Occupational Epidemiological Cancer Studies in Iran: A Review. Cancers 2021, 13, 3581. https://doi.org/10.3390/cancers13143581
Hosseini B, Hall AL, Zendehdel K, Kromhout H, Onyije FM, Moradzadeh R, Zamanian M, Schüz J, Olsson A. Occupational Exposure to Carcinogens and Occupational Epidemiological Cancer Studies in Iran: A Review. Cancers. 2021; 13(14):3581. https://doi.org/10.3390/cancers13143581
Chicago/Turabian StyleHosseini, Bayan, Amy L. Hall, Kazem Zendehdel, Hans Kromhout, Felix M. Onyije, Rahmatollah Moradzadeh, Maryam Zamanian, Joachim Schüz, and Ann Olsson. 2021. "Occupational Exposure to Carcinogens and Occupational Epidemiological Cancer Studies in Iran: A Review" Cancers 13, no. 14: 3581. https://doi.org/10.3390/cancers13143581
APA StyleHosseini, B., Hall, A. L., Zendehdel, K., Kromhout, H., Onyije, F. M., Moradzadeh, R., Zamanian, M., Schüz, J., & Olsson, A. (2021). Occupational Exposure to Carcinogens and Occupational Epidemiological Cancer Studies in Iran: A Review. Cancers, 13(14), 3581. https://doi.org/10.3390/cancers13143581








