Radiation Pneumonitis in Thoracic Cancer Patients: Multi-Center Voxel-Based Analysis
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.1.1. 3D Conformal Radiation Therapy
2.1.2. Intensity-Modulated Radiation Therapy
2.1.3. Stereotactic Body Radiation Therapy
2.1.4. Passive-Scattering Proton Therapy
2.2. Spatial Normalization
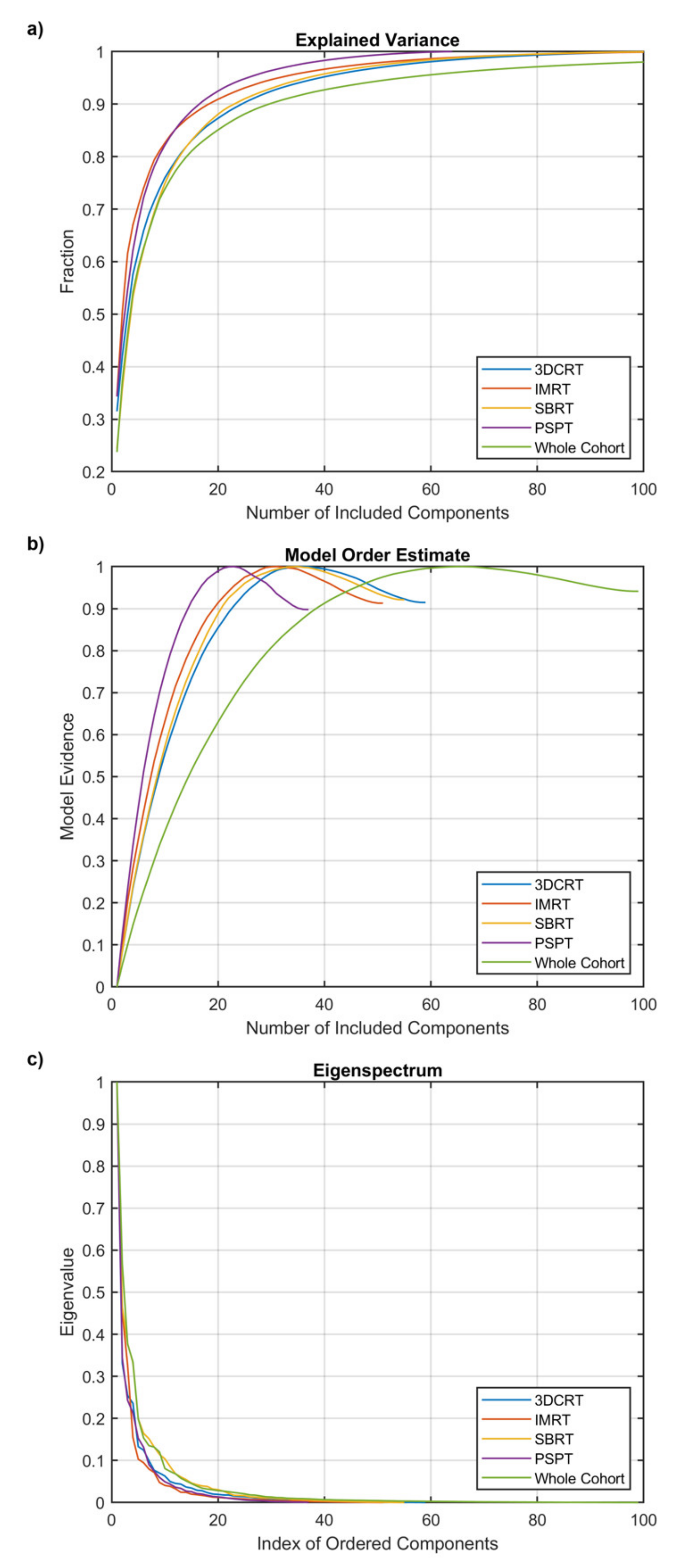
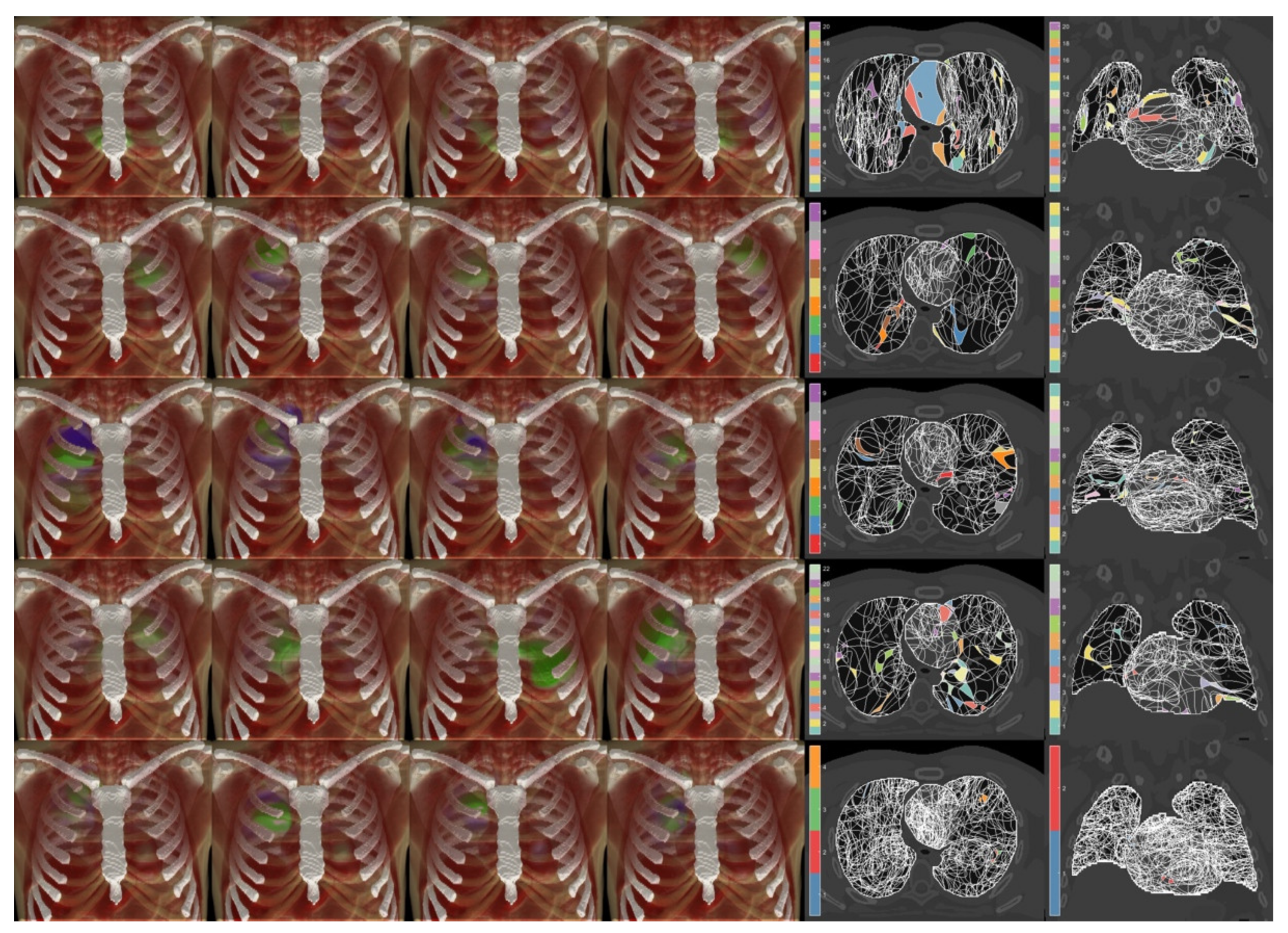
2.3. Characterization of Spatial Properties of Dosimetric Data
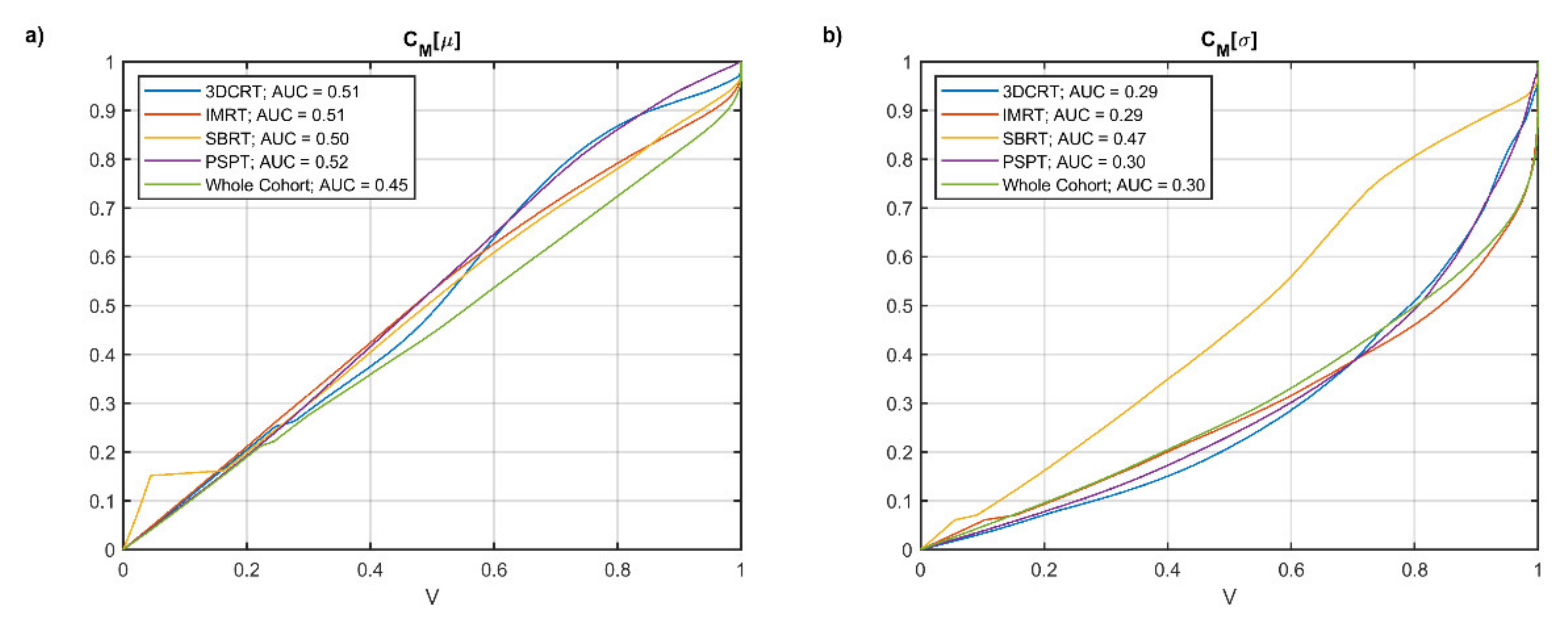
2.4. Voxel-Based Statistical Analysis
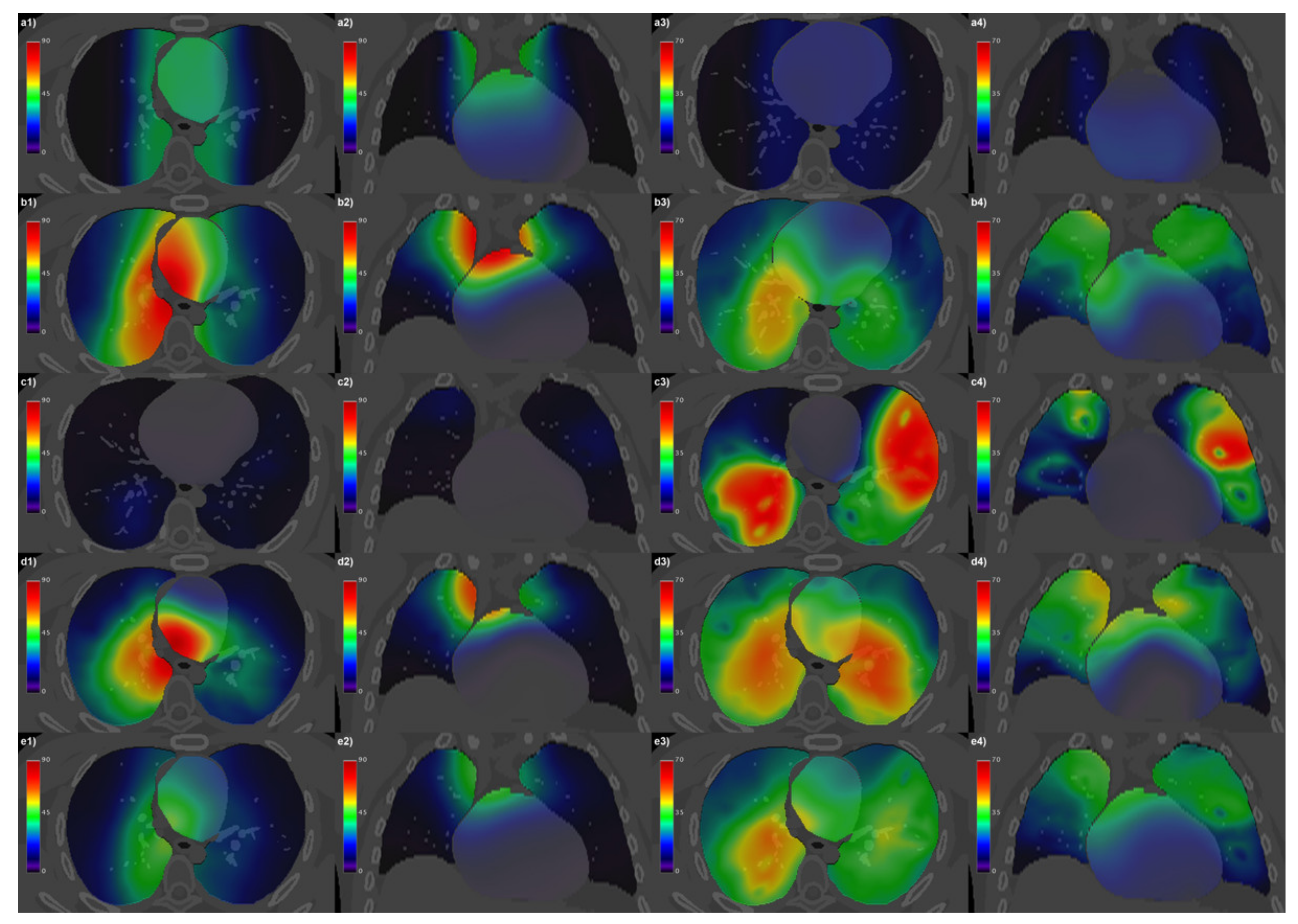
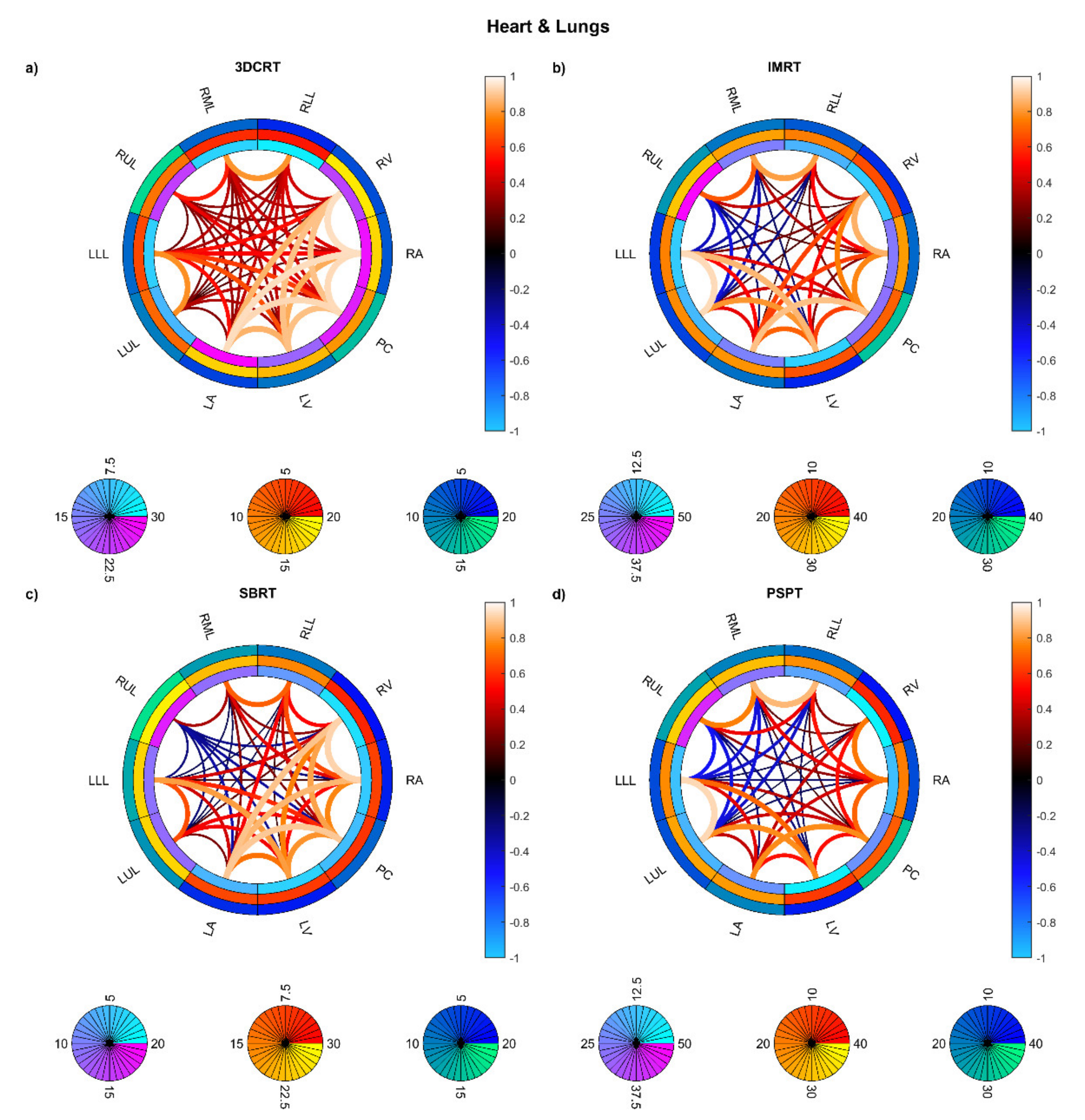
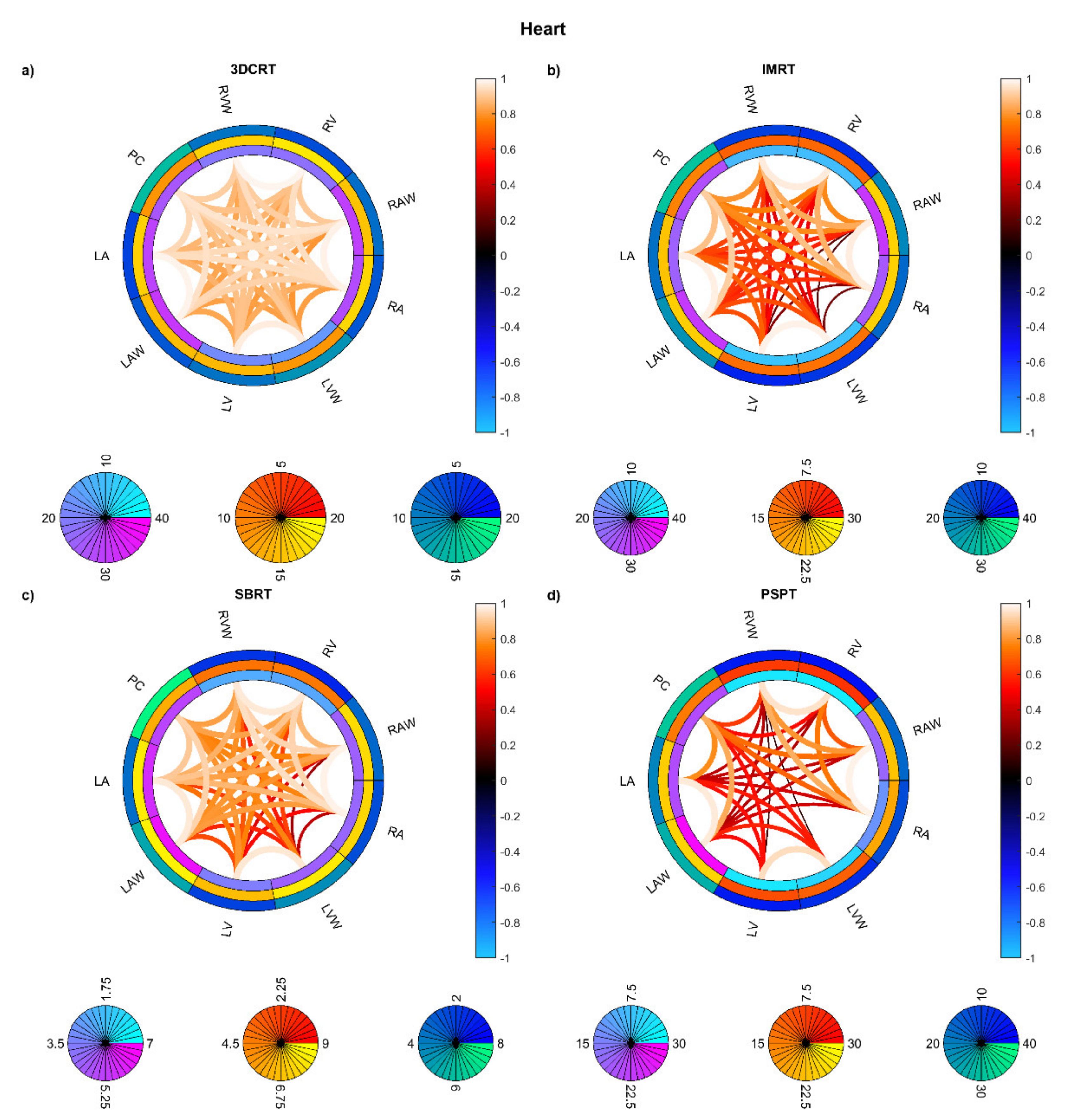
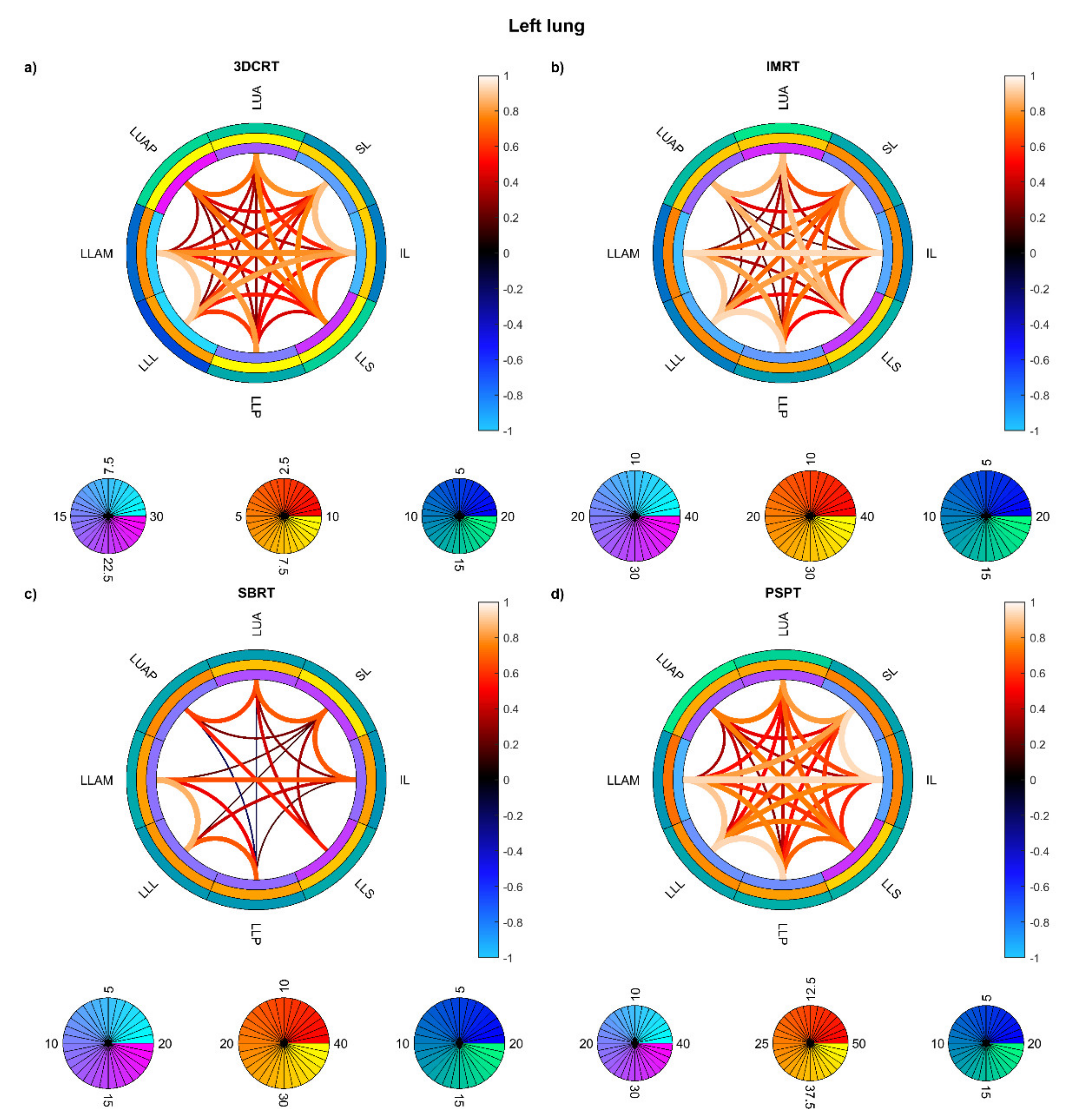
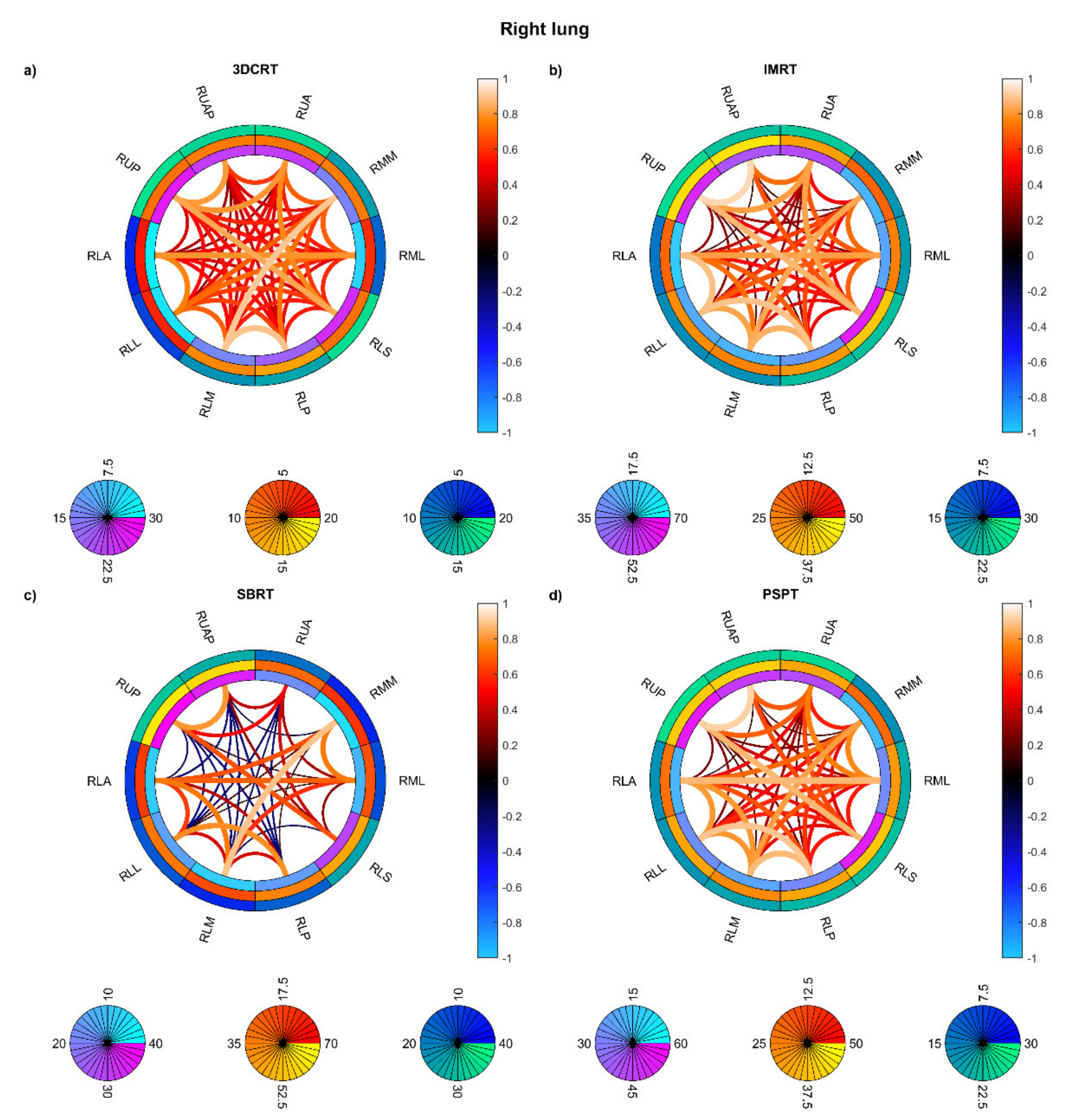
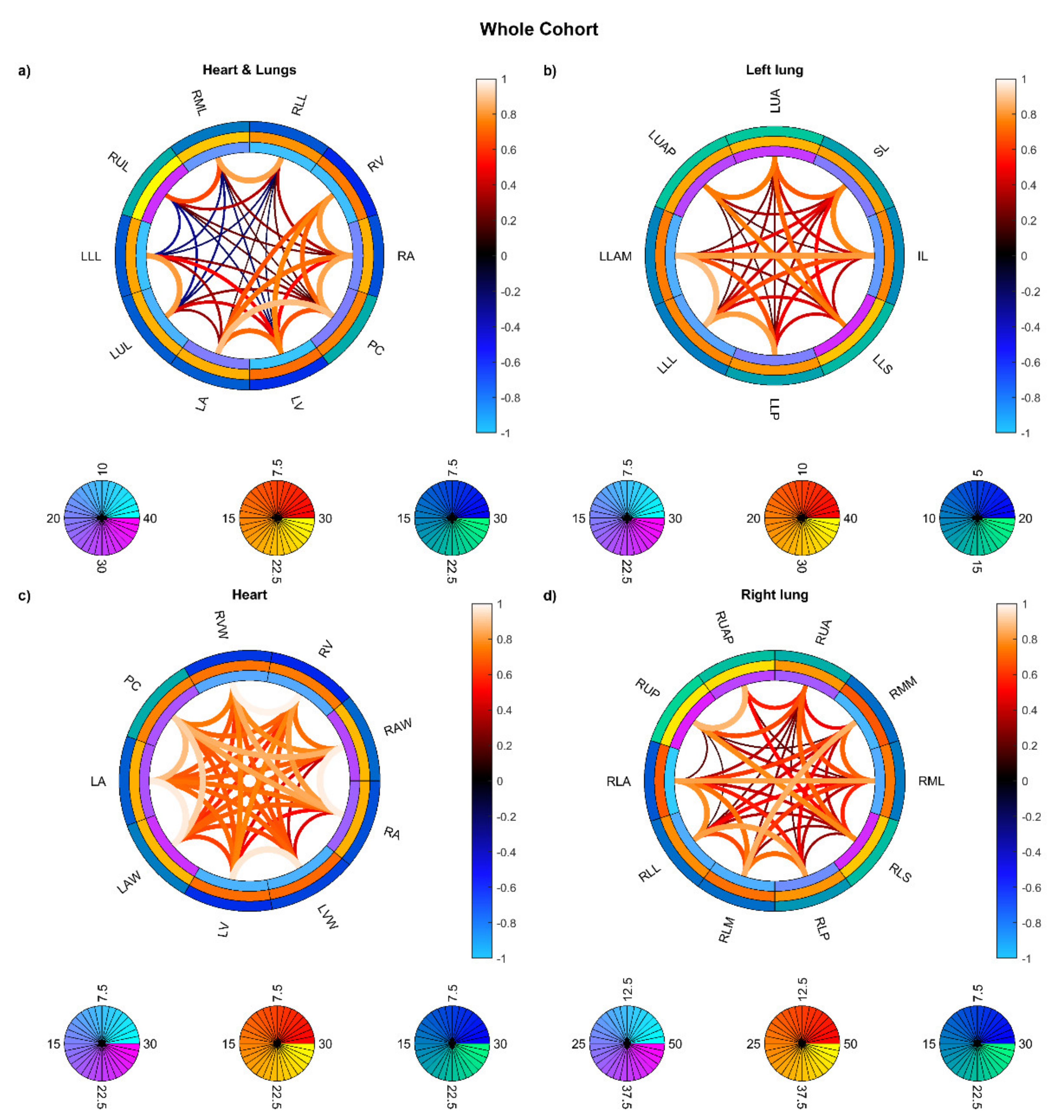
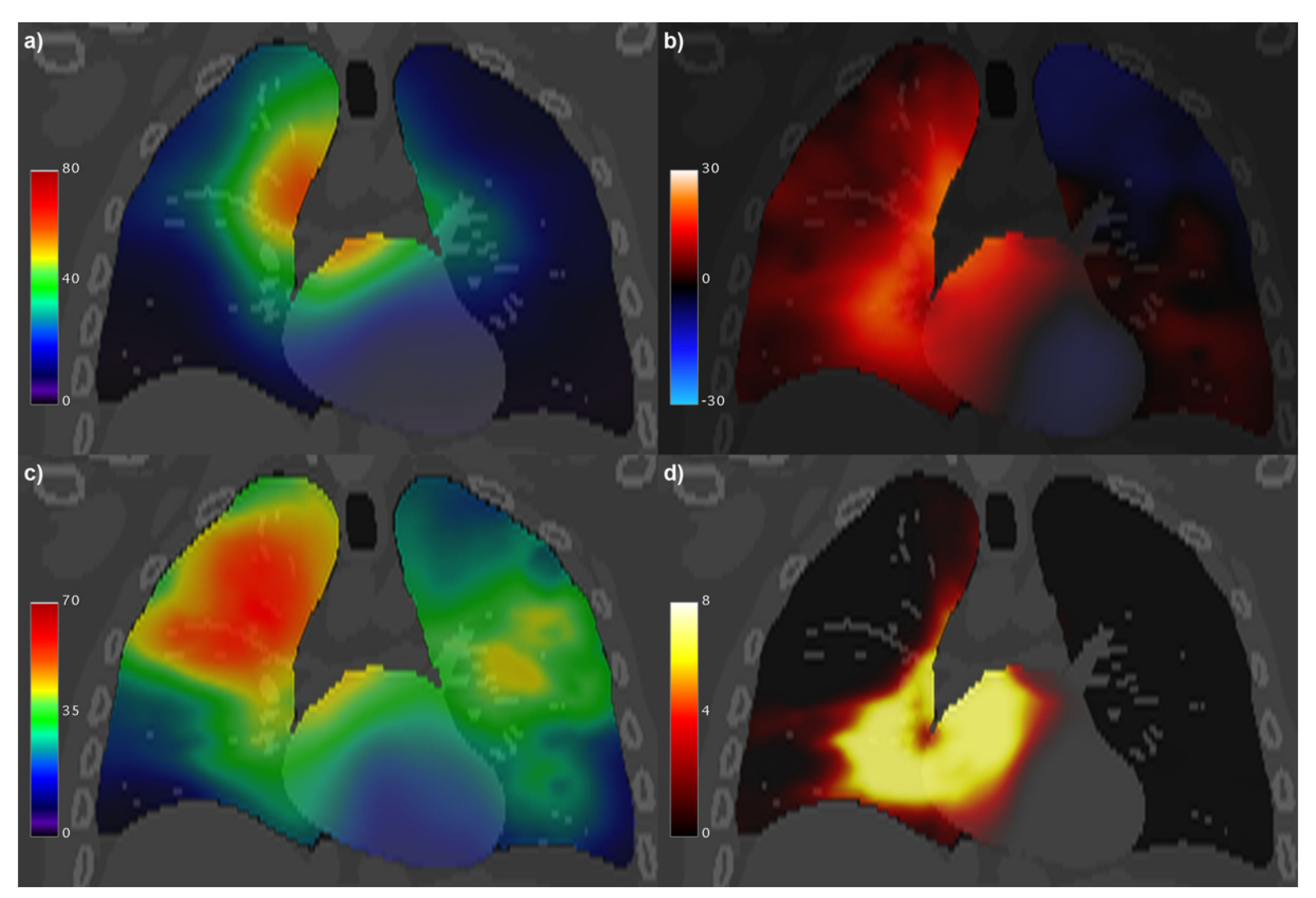
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jain, V.; Berman, A.T. Radiation Pneumonitis: Old Problem, New Tricks. Cancers 2018, 10, 222. [Google Scholar] [CrossRef] [Green Version]
- Cella, L.; Liuzzi, R.; D’Avino, V.; Conson, M.; Di Biase, A.; Picardi, M.; Pugliese, N.; Solla, R.; Salvatore, M.; Pacelli, R. Pulmonary damage in Hodgkin’s lymphoma patients treated with sequential chemo-radiotherapy: Predictors of radiation-induced lung injury. Acta Oncol. 2014, 53, 613–619. [Google Scholar] [CrossRef] [Green Version]
- Palma, G.; Monti, S.; Buonanno, A.; Pacelli, R.; Cella, L. PACE: A Probabilistic Atlas for Normal Tissue Complication Estimation in Radiation Oncology. Front. Oncol. 2019, 9, 130. [Google Scholar] [CrossRef] [Green Version]
- Wiegman, E.M.; Meertens, H.; Konings, A.W.; Kampinga, H.H.; Coppes, R.P. Loco-regional differences in pulmonary function and density after partial rat lung irradiation. Radiother. Oncol. 2003, 69, 11–19. [Google Scholar] [CrossRef]
- Defraene, G.; van Elmpt, W.; Crijns, W.; De Ruysscher, D. Regional variability in radiation-induced lung damage can be predicted by baseline CT numbers. Radiother. Oncol. 2017, 122, 300–306. [Google Scholar] [CrossRef]
- Madani, I.; De Ruyck, K.; Goeminne, H.; De Neve, W.; Thierens, H.; Van Meerbeeck, J. Predicting risk of radiation-induced lung injury. J. Thorac. Oncol. 2007, 2, 864–874. [Google Scholar] [CrossRef] [Green Version]
- Seppenwoolde, Y.; De Jaeger, K.; Boersma, L.J.; Belderbos, J.S.; Lebesque, J.V. Regional differences in lung radiosensitivity after radiotherapy for non-small-cell lung cancer. Int. J. Radiat. Oncol. Biol. Phys. 2004, 60, 748–758. [Google Scholar] [CrossRef] [PubMed]
- Cella, L.; Lomax, A.; Miralbell, R. New techniques in hadrontherapy: Intensity modulated proton beams. Phys. Med. 2001, 17 (Suppl. 1), 100–102. [Google Scholar]
- Mirri, M.A.; Arcangeli, G.; Benassi, M.; d’Angelo, A.; Pinzi, V.; Caterino, M.; Rinaldi, M.; Ceribelli, A.; Strigari, L. Hypofractionated Conformal Radiotherapy (HCRT) for primary and metastatic lung cancers with small dimension: Efficacy and toxicity. Strahlenther. Onkol. 2009, 185, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Tommasino, F.; Durante, M.; D’Avino, V.; Liuzzi, R.; Conson, M.; Farace, P.; Palma, G.; Schwarz, M.; Cella, L.; Pacelli, R. Model-based approach for quantitative estimates of skin, heart, and lung toxicity risk for left-side photon and proton irradiation after breast-conserving surgery. Acta Oncol. 2017, 56, 730–736. [Google Scholar] [CrossRef] [Green Version]
- Palma, G.; Monti, S.; Conson, M.; Pacelli, R.; Cella, L. Normal tissue complication probability (NTCP) models for modern radiation therapy. Semin. Oncol. 2019, 46, 210–218. [Google Scholar] [CrossRef]
- Monti, S.; Palma, G.; D’Avino, V.; Gerardi, M.; Marvaso, G.; Ciardo, D.; Pacelli, R.; Jereczek-Fossa, B.A.; Alterio, D.; Cella, L. Voxel-based analysis unveils regional dose differences associated with radiation-induced morbidity in head and neck cancer patients. Sci. Rep. 2017, 7, 7220. [Google Scholar] [CrossRef] [PubMed]
- Palma, G.; Monti, S.; D’Avino, V.; Conson, M.; Liuzzi, R.; Pressello, M.C.; Donato, V.; Deasy, J.O.; Quarantelli, M.; Pacelli, R.; et al. A Voxel-Based Approach to Explore Local Dose Differences Associated With Radiation-Induced Lung Damage. Int. J. Radiat. Oncol. Biol. Phys. 2016, 96, 127–133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cella, L.; Monti, S.; Xu, T.; Liuzzi, R.; Stanzione, A.; Durante, M.; Mohan, R.; Liao, Z.; Palma, G. Probing thoracic dose patterns associated to pericardial effusion and mortality in patients treated with photons and protons for locally advanced non-small-cell lung cancer. Radiother. Oncol. 2021. [Google Scholar] [CrossRef]
- Cella, L.; Oh, J.H.; Deasy, J.O.; Palma, G.; Liuzzi, R.; D’Avino, V.; Conson, M.; Picardi, M.; Salvatore, M.; Pacelli, R. Predicting radiation-induced valvular heart damage. Acta Oncol. 2015, 54, 1796–1804. [Google Scholar] [CrossRef]
- Cella, L.; Palma, G.; Deasy, J.O.; Oh, J.H.; Liuzzi, R.; D’Avino, V.; Conson, M.; Pugliese, N.; Picardi, M.; Salvatore, M.; et al. Complication probability models for radiation-induced heart valvular dysfunction: Do heart-lung interactions play a role? PLoS ONE 2014, 9, e111753. [Google Scholar] [CrossRef]
- Liao, Z.; Lee, J.J.; Komaki, R.; Gomez, D.R.; O’Reilly, M.S.; Fossella, F.V.; Blumenschein, G.R., Jr.; Heymach, J.V.; Vaporciyan, A.A.; Swisher, S.G.; et al. Bayesian Adaptive Randomization Trial of Passive Scattering Proton Therapy and Intensity-Modulated Photon Radiotherapy for Locally Advanced Non-Small-Cell Lung Cancer. J. Clin. Oncol. 2018, 36, 1813–1822. [Google Scholar] [CrossRef]
- Palma, G.; Monti, S.; Conson, M.; Xu, T.; Hahn, S.; Durante, M.; Mohan, R.; Liao, Z.; Cella, L. NTCP Models for Severe Radiation Induced Dermatitis After IMRT or Proton Therapy for Thoracic Cancer Patients. Front. Oncol. 2020, 10, 344. [Google Scholar] [CrossRef] [PubMed]
- Palma, G.; Monti, S.; Thor, M.; Rimner, A.; Deasy, J.O.; Cella, L. Spatial signature of dose patterns associated with acute radiation-induced lung damage in lung cancer patients treated with stereotactic body radiation therapy. Phys. Med. Biol. 2019, 64, 155006. [Google Scholar] [CrossRef] [PubMed]
- Segars, W.P.; Sturgeon, G.; Mendonca, S.; Grimes, J.; Tsui, B.M. 4D XCAT phantom for multimodality imaging research. Med. Phys. 2010, 37, 4902–4915. [Google Scholar] [CrossRef]
- Conson, M.; Cella, L.; Pacelli, R.; Comerci, M.; Liuzzi, R.; Salvatore, M.; Quarantelli, M. Automated delineation of brain structures in patients undergoing radiotherapy for primary brain tumors: From atlas to dose-volume histograms. Radiother. Oncol. 2014, 112, 326–331. [Google Scholar] [CrossRef] [Green Version]
- Klein, S.; Staring, M.; Murphy, K.; Viergever, M.A.; Pluim, J.P. elastix: A toolbox for intensity-based medical image registration. IEEE Trans. Med. Imaging 2010, 29, 196–205. [Google Scholar] [CrossRef]
- Palma, G.; Monti, S.; Cella, L. Voxel-based analysis in radiation oncology: A methodological cookbook. Phys. Med. 2020, 69, 192–204. [Google Scholar] [CrossRef] [PubMed]
- Beckmann, C.F.; Smith, S.M. Probabilistic independent component analysis for functional magnetic resonance imaging. IEEE Trans. Med. Imaging 2004, 23, 137–152. [Google Scholar] [CrossRef]
- Irimia, A.; Chambers, M.C.; Torgerson, C.M.; Van Horn, J.D. Circular representation of human cortical networks for subject and population-level connectomic visualization. Neuroimage 2012, 60, 1340–1351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, C.; Witte, M.; Heemsbergen, W.; van Herk, M. Multiple comparisons permutation test for image based data mining in radiotherapy. Radiat. Oncol. 2013, 8, 293. [Google Scholar] [CrossRef] [PubMed]
- Palma, G.; Taffelli, A.; Fellin, F.; D’Avino, V.; Scartoni, D.; Tommasino, F.; Scifoni, E.; Durante, M.; Amichetti, M.; Schwarz, M.; et al. Modelling the risk of radiation induced alopecia in brain tumor patients treated with scanned proton beams. Radiother. Oncol. 2020, 144, 127–134. [Google Scholar] [CrossRef]
- Mylona, E.; Acosta, O.; Lizee, T.; Lafond, C.; Crehange, G.; Magne, N.; Chiavassa, S.; Supiot, S.; Ospina Arango, J.D.; Campillo-Gimenez, B.; et al. Voxel-Based Analysis for Identification of Urethrovesical Subregions Predicting Urinary Toxicity After Prostate Cancer Radiation Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2019, 104, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Drean, G.; Acosta, O.; Lafond, C.; Simon, A.; de Crevoisier, R.; Haigron, P. Interindividual registration and dose mapping for voxelwise population analysis of rectal toxicity in prostate cancer radiotherapy. Med. Phys. 2016, 43, 2721–2730. [Google Scholar] [CrossRef] [PubMed]
- Abravan, A.; Faivre-Finn, C.; Kennedy, J.; McWilliam, A.; van Herk, M. Radiotherapy-Related Lymphopenia Affects Overall Survival in Patients With Lung Cancer. J. Thorac. Oncol. 2020, 15, 1624–1635. [Google Scholar] [CrossRef]
- Beasley, W.; Thor, M.; McWilliam, A.; Green, A.; Mackay, R.; Slevin, N.; Olsson, C.; Pettersson, N.; Finizia, C.; Estilo, C.; et al. Image-based Data Mining to Probe Dosimetric Correlates of Radiation-induced Trismus. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, 1330–1338. [Google Scholar] [CrossRef]
- Yahya, N.; Ebert, M.A.; House, M.J.; Kennedy, A.; Matthews, J.; Joseph, D.J.; Denham, J.W. Modeling Urinary Dysfunction After External Beam Radiation Therapy of the Prostate Using Bladder Dose-Surface Maps: Evidence of Spatially Variable Response of the Bladder Surface. Int. J. Radiat. Oncol. Biol. Phys. 2017, 97, 420–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coda, A.R.; Anzilotti, S.; Boscia, F.; Greco, A.; Panico, M.; Gargiulo, S.; Gramanzini, M.; Zannetti, A.; Albanese, S.; Pignataro, G.; et al. In vivo imaging of CNS microglial activation/macrophage infiltration with combined [(18)F] DPA-714-PET and SPIO-MRI in a mouse model of relapsing remitting experimental autoimmune encephalomyelitis. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 40–52. [Google Scholar] [CrossRef] [PubMed]
- Pontillo, G.; Petracca, M.; Monti, S.; Quarantelli, M.; Criscuolo, C.; Lanzillo, R.; Tedeschi, E.; Elefante, A.; Brescia Morra, V.; Brunetti, A.; et al. Unraveling Deep Gray Matter Atrophy and Iron and Myelin Changes in Multiple Sclerosis. AJNR Am. J. Neuroradiol. 2021. [Google Scholar] [CrossRef]
- Shortall, J.; Palma, G.; Mistry, H.; Vasquez Osorio, E.; McWilliam, A.; Choudhury, A.; Aznar, M.; van Herk, M.; Green, A. Flogging a Dead Salmon? Reduced Dose Posterior to Prostate Correlates With Increased PSA Progression in Voxel-Based Analysis of 3 Randomized Phase 3 Trials. Int. J. Radiat. Oncol. Biol. Phys. 2021, 110, 696–699. [Google Scholar] [CrossRef] [PubMed]
- Voshart, D.C.; Wiedemann, J.; van Luijk, P.; Barazzuol, L. Regional Responses in Radiation-Induced Normal Tissue Damage. Cancers 2021, 13, 367. [Google Scholar] [CrossRef]
- Cella, L.; Gagliardi, G.; Hedman, M.; Palma, G. Injuries From Asymptomatic COVID-19 Disease: New Hidden Toxicity Risk Factors in Thoracic Radiation Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2020, 108, 394–396. [Google Scholar] [CrossRef] [PubMed]
- Ghobadi, G.; van der Veen, S.; Bartelds, B.; de Boer, R.A.; Dickinson, M.G.; de Jong, J.R.; Faber, H.; Niemantsverdriet, M.; Brandenburg, S.; Berger, R.M.; et al. Physiological interaction of heart and lung in thoracic irradiation. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, e639–e646. [Google Scholar] [CrossRef] [Green Version]
- Monti, S.; Pacelli, R.; Cella, L.; Palma, G. Inter-patient image registration algorithms to disentangle regional dose bioeffects. Sci. Rep. 2018, 8, 4915. [Google Scholar] [CrossRef]
- Tomita, N.; Okuda, K.; Ogawa, Y.; Iida, M.; Eguchi, Y.; Kitagawa, Y.; Uchiyama, K.; Takaoka, T.; Nakanishi, R.; Shibamoto, Y. Relationship between radiation doses to heart substructures and radiation pneumonitis in patients with thymic epithelial tumors. Sci. Rep. 2020, 10, 11191. [Google Scholar] [CrossRef]
- Palma, G.; Monti, S.; Xu, T.; Scifoni, E.; Yang, P.; Hahn, S.M.; Durante, M.; Mohan, R.; Liao, Z.; Cella, L. Spatial Dose Patterns Associated With Radiation Pneumonitis in a Randomized Trial Comparing Intensity-Modulated Photon Therapy With Passive Scattering Proton Therapy for Locally Advanced Non-Small Cell Lung Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2019, 104, 1124–1132. [Google Scholar] [CrossRef] [PubMed]
- Huang, E.X.; Hope, A.J.; Lindsay, P.E.; Trovo, M.; El Naqa, I.; Deasy, J.O.; Bradley, J.D. Heart irradiation as a risk factor for radiation pneumonitis. Acta Oncol. 2011, 50, 51–60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cella, L.; D’Avino, V.; Palma, G.; Conson, M.; Liuzzi, R.; Picardi, M.; Pressello, M.C.; Boboc, G.I.; Battistini, R.; Donato, V.; et al. Modeling the risk of radiation-induced lung fibrosis: Irradiated heart tissue is as important as irradiated lung. Radiother. Oncol. 2015, 117, 36–43. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palma, G.; Monti, S.; Pacelli, R.; Liao, Z.; Deasy, J.O.; Mohan, R.; Cella, L. Radiation Pneumonitis in Thoracic Cancer Patients: Multi-Center Voxel-Based Analysis. Cancers 2021, 13, 3553. https://doi.org/10.3390/cancers13143553
Palma G, Monti S, Pacelli R, Liao Z, Deasy JO, Mohan R, Cella L. Radiation Pneumonitis in Thoracic Cancer Patients: Multi-Center Voxel-Based Analysis. Cancers. 2021; 13(14):3553. https://doi.org/10.3390/cancers13143553
Chicago/Turabian StylePalma, Giuseppe, Serena Monti, Roberto Pacelli, Zhongxing Liao, Joseph O. Deasy, Radhe Mohan, and Laura Cella. 2021. "Radiation Pneumonitis in Thoracic Cancer Patients: Multi-Center Voxel-Based Analysis" Cancers 13, no. 14: 3553. https://doi.org/10.3390/cancers13143553
APA StylePalma, G., Monti, S., Pacelli, R., Liao, Z., Deasy, J. O., Mohan, R., & Cella, L. (2021). Radiation Pneumonitis in Thoracic Cancer Patients: Multi-Center Voxel-Based Analysis. Cancers, 13(14), 3553. https://doi.org/10.3390/cancers13143553







