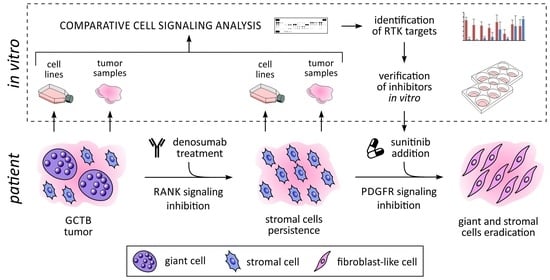
New Target for Precision Medicine Treatment of Giant-Cell Tumor of Bone: Sunitinib Is Effective in the Treatment of Neoplastic Stromal Cells with Activated PDGFRβ Signaling
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Tumor Samples
2.2. Cell Lines and Cell Culture
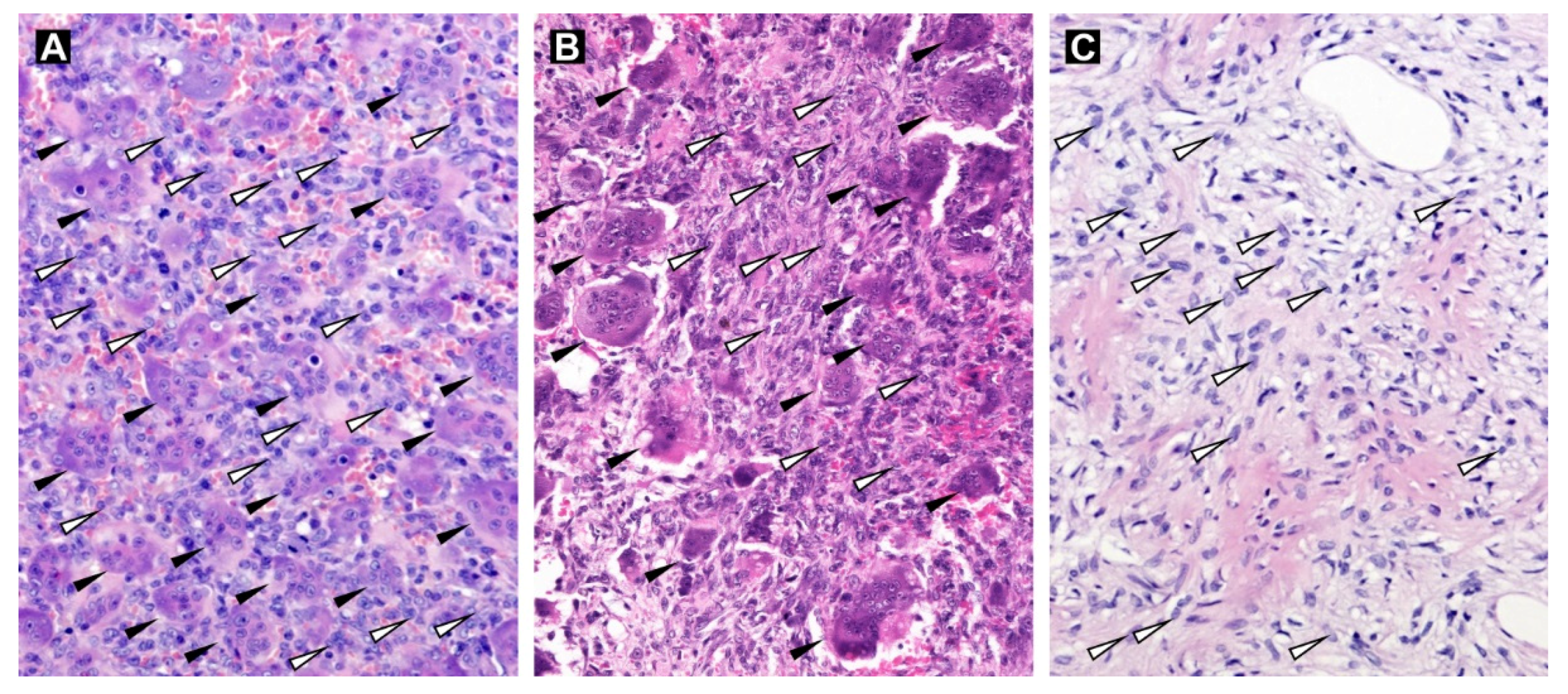
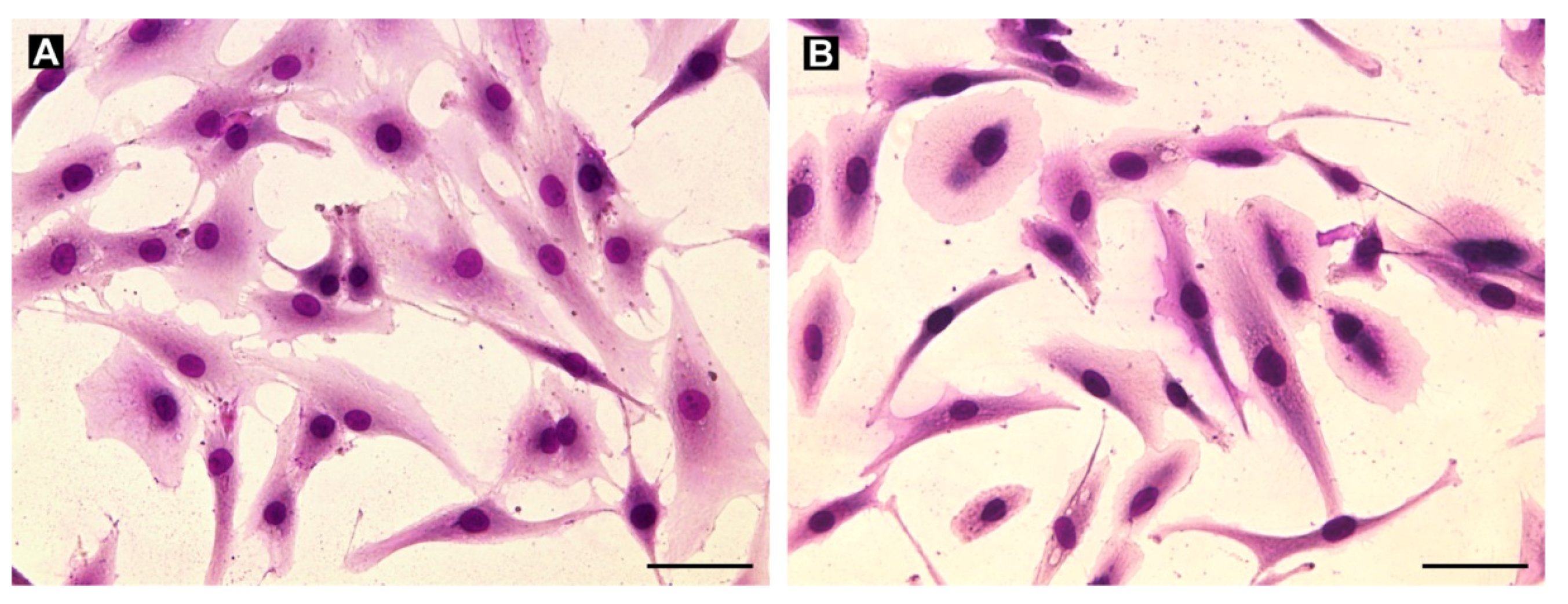
2.3. Evaluation of Tissue and Cell Morphology
2.4. Immunoblotting
2.5. Phosphoprotein Array Analysis
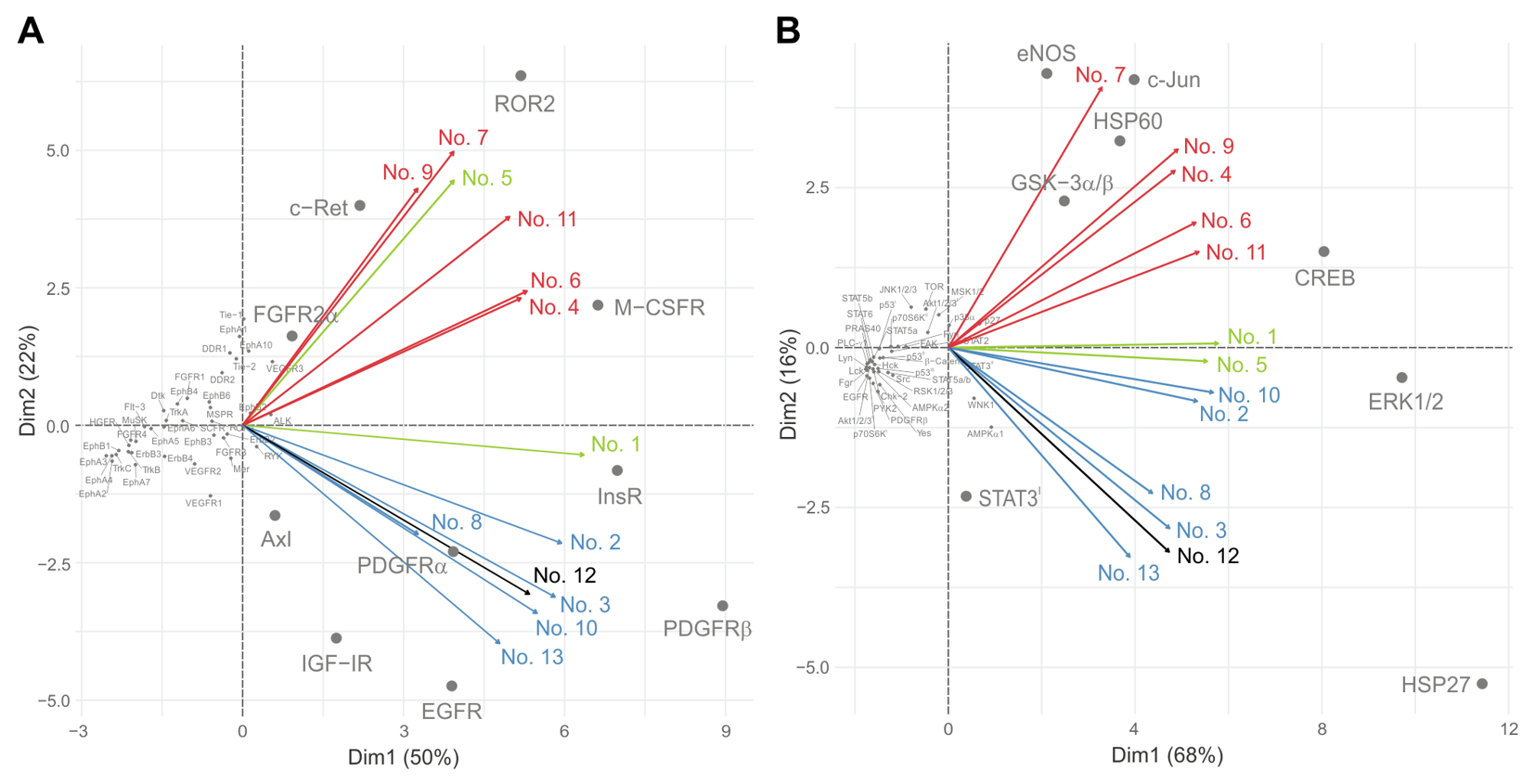
2.6. Principal Component Analysis
2.7. MTT Assay
2.8. Statistical Analysis
3. Results
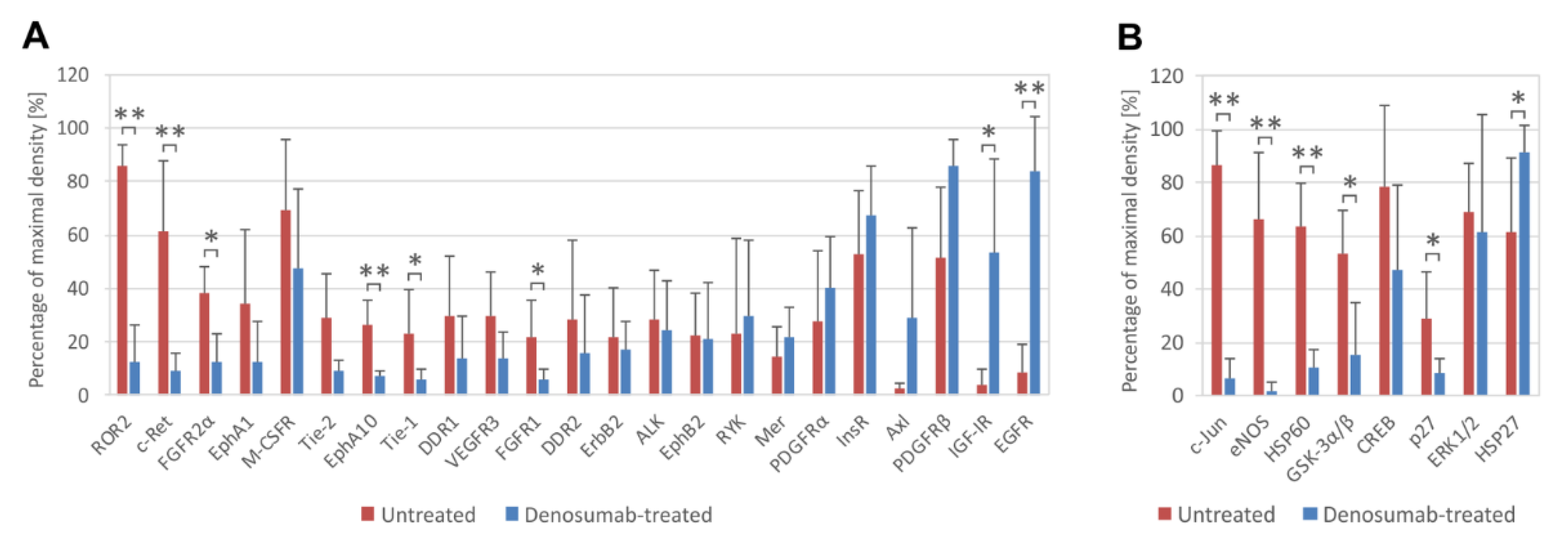
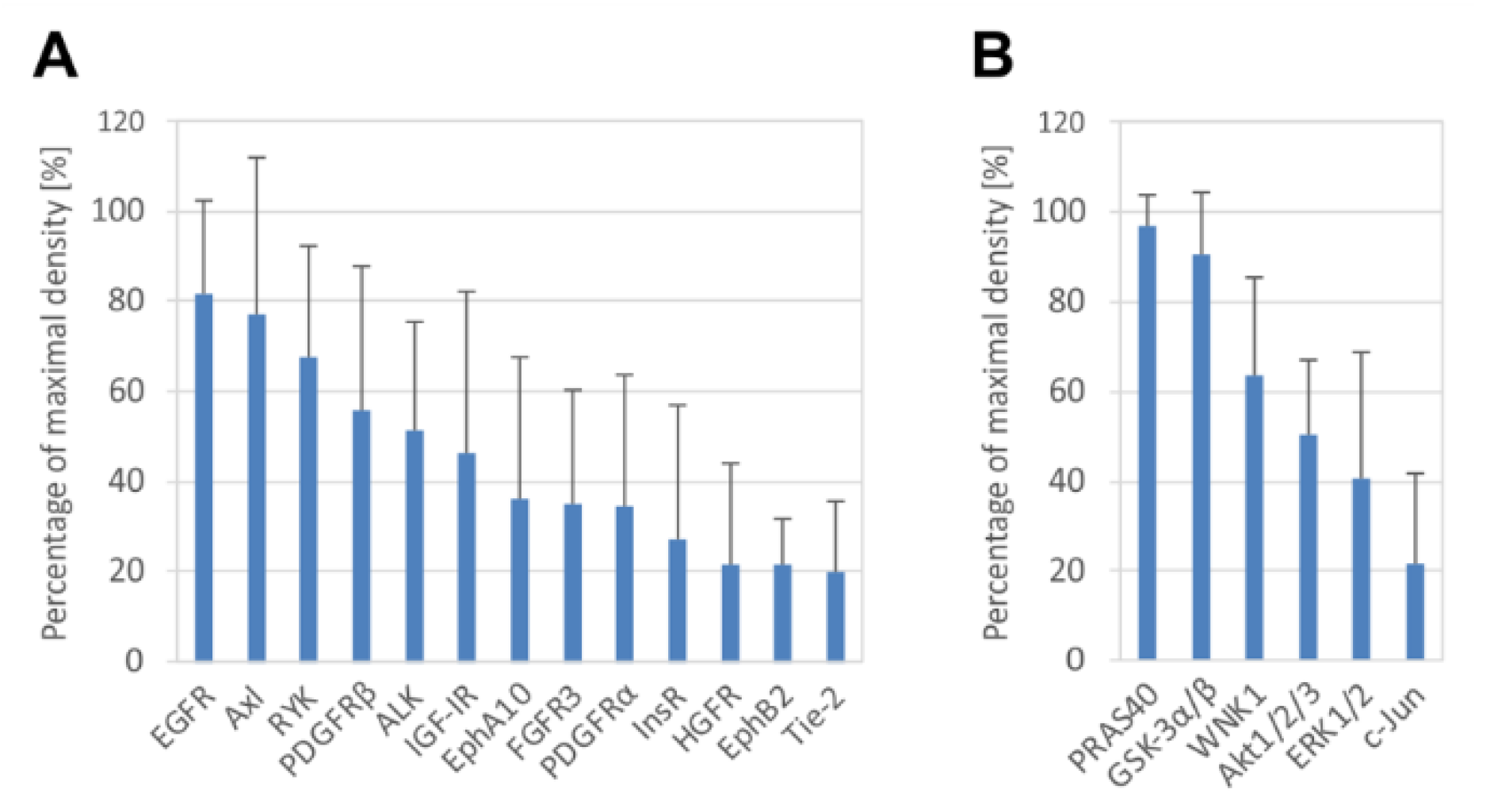
3.1. Denosumab-Treated Tumor Samples Exhibit Specific Profiles of Phosphorylated Kinases
3.2. GCTB-Derived Cell Lines Resemble Denosumab-Treated Cancer Tissues Regarding RTK Phosphorylation Profiles
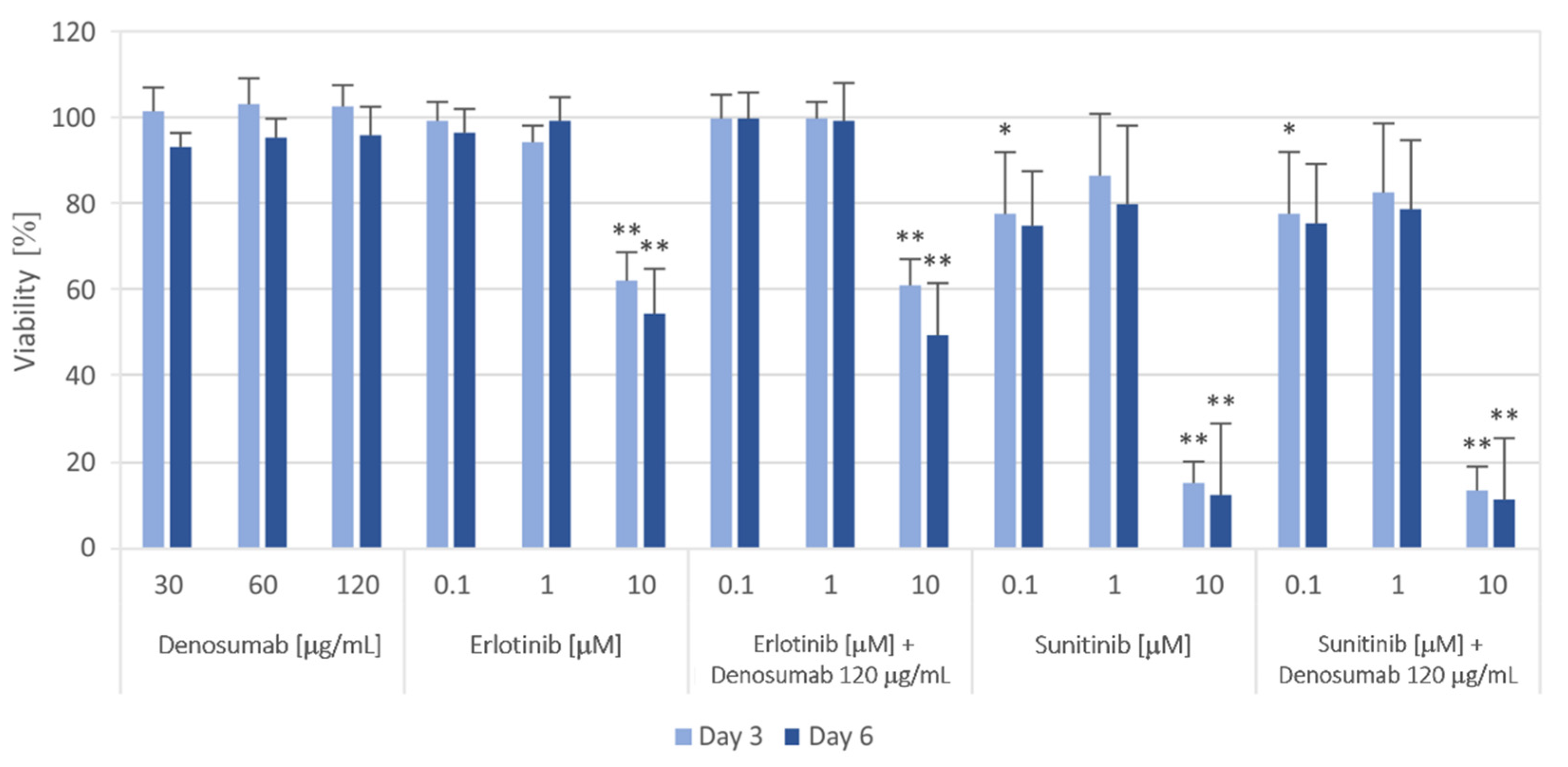
3.3. GCTB-Derived Cell Lines Are Sensitive to Tyrosine Kinase Inhibitors but Not to RANKL Inhibitor
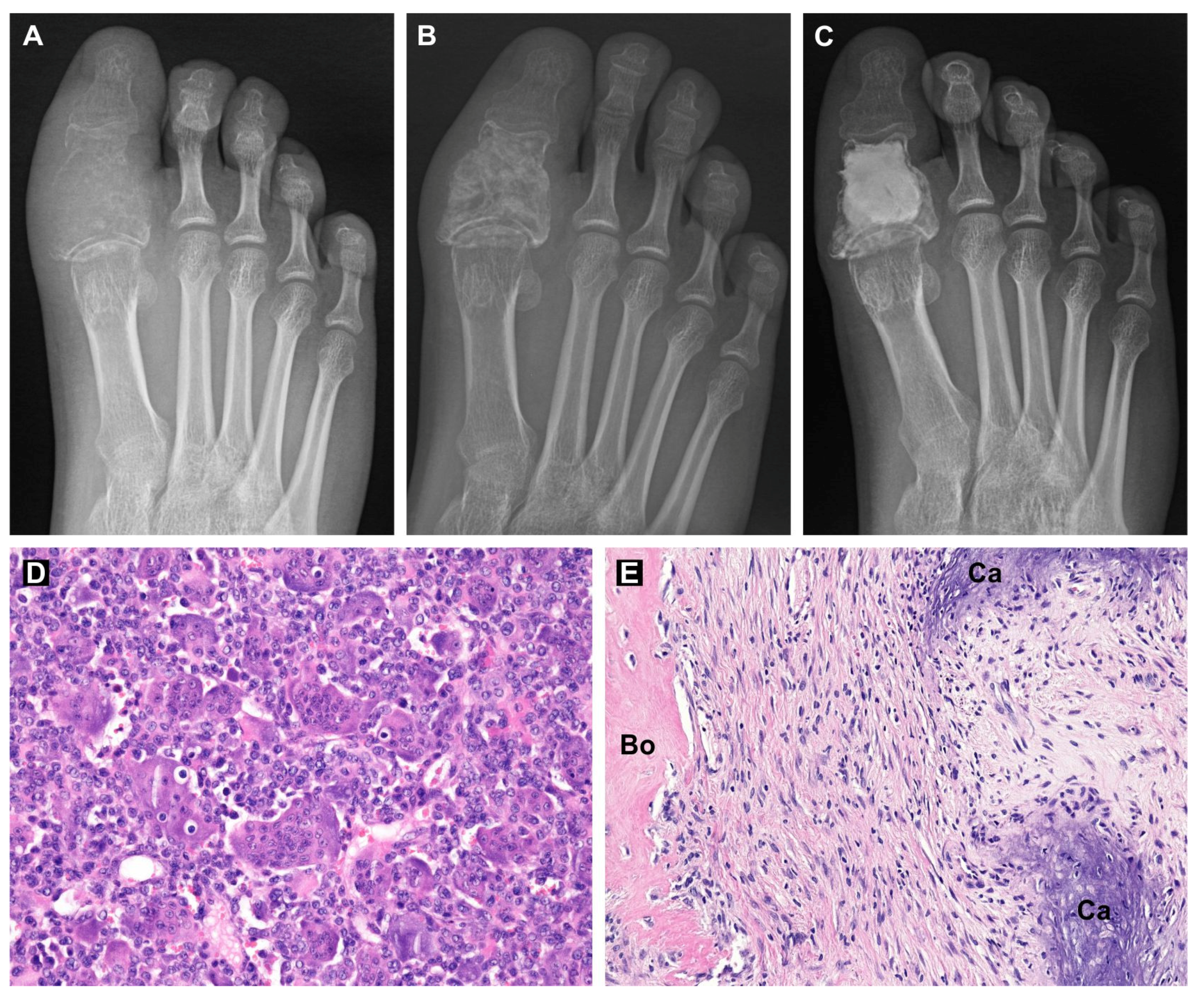
3.4. From Bench to Bedside: The Use of Sunitinib in GCTB Treatment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Campanacci, M.; Baldini, N.; Boriani, S.; Sudanese, A. Giant-Cell Tumor of Bone. J. Bone Joint Surg. Am. 1987, 69, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Athanasou, N.; Bansal, M.; Forsyth, R.; Reid, R.; Sapi, Z. Giant cell tumor of bone. In WHO Classification of Tumours of Soft Tissue and Bone; Fletcher, C.D.M., Bridge, J., Hogendoor, P., Mertens, F., Eds.; IARC Press: Lyon, France, 2013; pp. 321–324. ISBN 978-92-832-2434-1. [Google Scholar]
- Turcotte, R.E. Giant Cell Tumor of Bone. Orthop. Clin. North Am. 2006, 37, 35–51. [Google Scholar] [CrossRef] [PubMed]
- Unni, K.K.; Inwards, C.Y. Dahlin’s Bone Tumors: General Aspects and Data on 10,165 Cases, 6th ed.; LWW: Philadelphia, PA, USA, 2009; ISBN 978-0-7817-6242-7. [Google Scholar]
- Ruggieri, P.; Mavrogenis, A.F.; Ussia, G.; Angelini, A.; Papagelopoulos, P.J.; Mercuri, M. Recurrence After and Complications Associated With Adjuvant Treatments for Sacral Giant Cell Tumor. Clin. Orthop. Relat. Res. 2010, 468, 2954–2961. [Google Scholar] [CrossRef]
- Balke, M.; Schremper, L.; Gebert, C.; Ahrens, H.; Streitbuerger, A.; Koehler, G.; Hardes, J.; Gosheger, G. Giant cell tumor of bone: Treatment and outcome of 214 cases. J. Cancer Res. Clin. Oncol. 2008, 134, 969–978. [Google Scholar] [CrossRef]
- Malek, F.A.A.; Krueger, P.M.; Hatmi, Z.N.; Malayeri, A.; Faezipour, H.; Donnell, R.J.O. Local control of long bone giant cell tumour using curettage, burring and bone grafting without adjuvant therapy. Int. Orthop. 2006, 30, 495–498. [Google Scholar] [CrossRef]
- Saiz, P.; Virkus, W.; Piasecki, P.; Templeton, A.; Shott, S.; Gitelis, S. Results of Giant Cell Tumor of Bone Treated With Intralesional Excision. Clin. Orthop. Relat. Res. 2004, 424, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Jeys, L.M.; Suneja, R.; Chami, G.; Grimer, R.J.; Carter, S.R.; Tillman, R.M. Impending fractures in giant cell tumours of the distal femur: Incidence and outcome. Int. Orthop. 2006, 30, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.-F.; Tang, J.-Y.; Li, K.-H. RANK pathway in giant cell tumor of bone: Pathogenesis and therapeutic aspects. Tumor Biol. 2015, 36, 495–501. [Google Scholar] [CrossRef]
- Thomas, D.M. RANKL, denosumab, and giant cell tumor of bone. Curr. Opin. Oncol. 2012, 24, 397–403. [Google Scholar] [CrossRef]
- Chawla, S.; Henshaw, R.; Seeger, L.; Choy, E.; Blay, J.-Y.; Ferrari, S.; Kroep, J.; Grimer, R.; Reichardt, P.; Rutkowski, P.; et al. Safety and efficacy of denosumab for adults and skeletally mature adolescents with giant cell tumour of bone: Interim analysis of an open-label, parallel-group, phase 2 study. Lancet Oncol. 2013, 14, 901–908. [Google Scholar] [CrossRef]
- Arbeitsgemeinschaft Knochentumoren; Becker, W.T.; Dohle, J.; Bernd, L.; Braun, A.; Cserhati, M.; Enderle, A.; Hovy, L.; Matejovsky, Z.; Szendroi, M.; et al. Local Recurrence of Giant Cell Tumor of Bone after Intralesional Treatment with and without Adjuvant Therapy. J. Bone Jt. Surg. Am. Vol. 2008, 90, 1060–1067. [Google Scholar] [CrossRef] [PubMed]
- Rutkowski, P.; Gaston, L.; Borkowska, A.; Stacchiotti, S.; Gelderblom, H.; Baldi, G.G.; Palmerini, E.; Casali, P.; Gronchi, A.; Parry, M.; et al. Denosumab treatment of inoperable or locally advanced giant cell tumor of bone—Multicenter analysis outside clinical trial. Eur. J. Surg. Oncol. EJSO 2018, 44, 1384–1390. [Google Scholar] [CrossRef] [PubMed]
- Palmerini, E.; Chawla, N.; Ferrari, S.; Sudan, M.; Picci, P.; Marchesi, E.; Leopardi, M.P.; Syed, I.; Sankhala, K.; Parthasarathy, P.; et al. Denosumab in advanced/unresectable giant-cell tumour of bone (GCTB): For how long? Eur. J. Cancer 2017, 76, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Ulas, A.; Akinci, M.B.; Silay, K.; Sendur, M.A.N.; DeDe, D.S.; Yalcin, B. Denosumab: Excellent response of metastatic giant cell tumor of the bone. J. BUON Off. J. Balk. Union Oncol. 2015, 20, 666–667. [Google Scholar]
- Mak, I.W.; Evaniew, N.; Popovic, S.; Tozer, R.; Ghert, M. A Translational Study of the Neoplastic Cells of Giant Cell Tumor of Bone Following Neoadjuvant Denosumab. J. Bone Jt. Surg. Am. Vol. 2014, 96, e127. [Google Scholar] [CrossRef] [PubMed]
- Balla, P.; Moskovszky, L.; Sapi, Z.; Forsyth, R.; Knowles, H.; Athanasou, N.A.; Szendroi, M.; Kopper, L.; Rajnai, H.; Pinter, F.; et al. Epidermal growth factor receptor signalling contributes to osteoblastic stromal cell proliferation, osteoclastogenesis and disease progression in giant cell tumour of bone. Histopathology 2011, 59, 376–389. [Google Scholar] [CrossRef]
- Guenther, R.; Krenn, V.; Morawietz, L.; Dankof, A.; Melcher, I.; Schaser, K.-D.; Kasper, H.-U.; Kuban, R.-J.; Ungethüm, U.; Sers, C. Giant cell tumors of the bone: Molecular profiling and expression analysis of Ephrin A1 receptor, Claudin 7, CD52, FGFR3 and AMFR. Pathol. Res. Pr. 2005, 201, 649–663. [Google Scholar] [CrossRef]
- Liu, L.; Aleksandrowicz, E.; Fan, P.; Schonsiegel, F.; Zhang, Y.; Sahr, H.; Gladkich, J.; Mattern, J.; Depeweg, D.; Lehner, B.; et al. Enrichment of c-Met+ tumorigenic stromal cells of giant cell tumor of bone and targeting by cabozantinib. Cell Death Dis. 2014, 5, e1471. [Google Scholar] [CrossRef]
- Zhou, Z.; Li, Y.; Wang, X.; Hu, J.; Kuang, M.; Wang, Z.; Li, S.; Xu, W.; Xiao, J. ALCAM+ stromal cells: Role in giant cell tumor of bone progression. Cell Death Dis. 2018, 9, 1–13. [Google Scholar] [CrossRef]
- Maros, M.E.; Schnaidt, S.; Balla, P.; Kelemen, Z.; Sapi, Z.; Szendroi, M.; Laszlo, T.; Forsyth, R.; Picci, P.; Krenacs, T. In situ cell cycle analysis in giant cell tumor of bone reveals patients with elevated risk of reduced progression-free survival. Bone 2019, 127, 188–198. [Google Scholar] [CrossRef]
- Neradil, J.; Kyr, M.; Polaskova, K.; Kren, L.; Macigova, P.; Skoda, J.; Sterba, J.; Veselska, R. Phospho-Protein Arrays as Effective Tools for Screening Possible Targets for Kinase Inhibitors and Their Use in Precision Pediatric Oncology. Front. Oncol. 2019, 9, 930. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Meth. 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Kassambara, A.; Mundt, F. Factoextra: Extract and Visualize the Results of Multivariate Data Analyses. R package Version 1.0.7. 2020. Available online: https://CRAN.R-project.org/package=factoextra (accessed on 2 May 2021).
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, 2018. Available online: https://www.R-project.org (accessed on 2 May 2021).
- Sramek, M.; Neradil, J.; Macigova, P.; Mudry, P.; Polaskova, K.; Slaby, O.; Noskova, H.; Sterba, J.; Veselska, R. Effects of Sunitinib and Other Kinase Inhibitors on Cells Harboring a PDGFRB Mutation Associated with Infantile Myofibromatosis. Int. J. Mol. Sci. 2018, 19, 2599. [Google Scholar] [CrossRef]
- Mukaihara, K.; Suehara, Y.; Kohsaka, S.; Akaike, K.; Tanabe, Y.; Kubota, D.; Ishii, M.; Fujimura, T.; Kazuno, S.; Okubo, T.; et al. Protein Expression Profiling of Giant Cell Tumors of Bone Treated with Denosumab. PLoS ONE 2016, 11, e0148401. [Google Scholar] [CrossRef][Green Version]
- Roskoski, R. The role of small molecule platelet-derived growth factor receptor (PDGFR) inhibitors in the treatment of neoplastic disorders. Pharmacol. Res. 2018, 129, 65–83. [Google Scholar] [CrossRef]
- Van der Heijden, L.; Dijkstra, P.S.; Blay, J.-Y.; Gelderblom, H. Giant cell tumour of bone in the denosumab era. Eur. J. Cancer 2017, 77, 75–83. [Google Scholar] [CrossRef]
- Klenke, F.M.; Wenger, D.E.; Inwards, C.Y.; Rose, P.S.; Sim, F.H. Giant Cell Tumor of Bone: Risk Factors for Recurrence. Clin. Orthop. Relat. Res. 2011, 469, 591–599. [Google Scholar] [CrossRef]
- Van Langevelde, K.; McCarthy, C.L. Radiological findings of denosumab treatment for giant cell tumours of bone. Skelet. Radiol. 2020, 49, 1345–1358. [Google Scholar] [CrossRef]
- Thomas, D.M.; Skubitz, K.M. Giant cell tumour of bone. Curr. Opin. Oncol. 2009, 21, 338–344. [Google Scholar] [CrossRef]
- Baud’Huin, M.; Renault, R.; Charrier, C.; Riet, A.; Moreau, A.; Brion, R.; Gouin, F.; Duplomb, L.; Heymann, D. Interleukin-34 is expressed by giant cell tumours of bone and plays a key role in RANKL-induced osteoclastogenesis. J. Pathol. 2010, 221, 77–86. [Google Scholar] [CrossRef]
- Oreffo, R.O.; Marshall, G.J.; Kirchen, M.; Garcia, C.; Gallwitz, W.E.; Chavez, J.; Mundy, G.R.; Bonewald, L.F. Characterization of a Cell Line Derived from a Human Giant Cell Tumor That Stimulates Osteoclastic Bone Resorption. Clin. Orthop. Relat. Res. 1993, 296, 229–241. [Google Scholar] [CrossRef]
- Hadjidakis, D.J.; Androulakis, I.I. Bone Remodeling. Ann. N. Y. Acad. Sci. 2006, 1092, 385–396. [Google Scholar] [CrossRef]
- Uehara, S.; Udagawa, N.; Kobayashi, Y. Regulation of osteoclast function via Rho-Pkn3-c-Src pathways. J. Oral Biosci. 2019, 61, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Brun, J.; Andreasen, C.M.; Ejersted, C.; Andersen, T.L.; Caverzasio, J.; Thouverey, C. PDGF Receptor Signaling in Osteoblast Lineage Cells Controls Bone Resorption Through Upregulation of Csf1 Expression. J. Bone Miner. Res. 2020, 35, 2458–2469. [Google Scholar] [CrossRef]
- David, J.-P.; Sabapathy, K.; Hoffmann, O.; Idarraga, M.H.; Wagner, E.F. JNK1 modulates osteoclastogenesis through both c-Jun phosphorylation-dependent and -independent mechanisms. J. Cell Sci. 2002, 115, 4317–4325. [Google Scholar] [CrossRef]
- Boyle, W.J.; Simonet, W.S.; Lacey, D.L. Osteoclast differentiation and activation. Nat. Cell Biol. 2003, 423, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Baloul, S.S. Osteoclastogenesis and Osteogenesis during Tooth Movement. Front Oral Biol 2016, 18, 75–79. [Google Scholar] [CrossRef]
- Wang, L.; Han, L.; Xue, P.; Hu, X.; Wong, S.-W.; Deng, M.; Tseng, H.C.; Huang, B.-W.; Ko, C.-C. Dopamine suppresses osteoclast differentiation via cAMP/PKA/CREB pathway. Cell. Signal. 2021, 78, 109847. [Google Scholar] [CrossRef]
- Roccisana, J.; Kawanabe, N.; Kajiya, H.; Koide, M.; Roodman, G.D.; Reddy, S.V. Functional Role for Heat Shock Factors in the Transcriptional Regulation of Human RANK Ligand Gene Expression in Stromal/Osteoblast Cells. J. Biol. Chem. 2004, 279, 10500–10507. [Google Scholar] [CrossRef]
- Koh, J.-M.; Lee, Y.-S.; Kim, Y.S.; Park, S.-H.; Lee, S.H.; Kim, H.-H.; Lee, M.-S.; Lee, K.-U.; Kim, G.S. Heat shock protein 60 causes osteoclastic bone resorption via toll-like receptor-2 in estrogen deficiency. Bone 2009, 45, 650–660. [Google Scholar] [CrossRef]
- Tariq, M.U.; Umer, M.; Khan, Z.; Saeed, J.; Siddiqui, M.A.; Din, N.U. Spectrum of histological features of Denosumab treated Giant Cell Tumor of Bone; potential pitfalls and diagnostic challenges for pathologists. Ann. Diagn. Pathol. 2020, 45, 151479. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Jiang, S.; Li, Z.; Dong, Y. Denosumab and Sunitinib in the treatment of giant-cell tumor of bone with pulmonary and bone metastases in an adolescent. Medicine 2019, 98, e17778. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, S.; Naot, D.; Callon, K.; Porteous, F.; Horne, A.; Wattie, D.; Watson, M.; Cornish, J.; Browett, P.; Grey, A. Imatinib Promotes Osteoblast Differentiation by Inhibiting PDGFR Signaling and Inhibits Osteoclastogenesis by Both Direct and Stromal Cell-Dependent Mechanisms. J. Bone Miner. Res. 2007, 22, 1679–1689. [Google Scholar] [CrossRef]
- O’Sullivan, S.; Tay, M.L.; Lin, J.; Bava, U.; Callon, K.; Cornish, J.; Naot, D.; Grey, A. Tyrosine Kinase Inhibitors Regulate OPG through Inhibition of PDGFRβ. PLoS ONE 2016, 11, e0164727. [Google Scholar] [CrossRef] [PubMed]
- Greenhalgh, J.; Dwan, K.; Boland, A.; Bates, V.; Vecchio, F.; Dundar, Y.; Jain, P.; Green, J.A. First-line treatment of advanced epidermal growth factor receptor (EGFR) mutation positive non-squamous non-small cell lung cancer. Cochrane Database Syst. Rev. 2016, 5, CD010383. [Google Scholar] [CrossRef]
- Kelley, R.K.; Ko, A. Erlotinib in the treatment of advanced pancreatic cancer. Biol. Targets Ther. 2008, 2, 83–95. [Google Scholar] [CrossRef]
- Motzer, R.J.; Ravaud, A.; Patard, J.-J.; Pandha, H.S.; George, D.J.; Patel, A.; Chang, Y.-H.; Escudier, B.; Donskov, F.; Magheli, A.; et al. Adjuvant Sunitinib for High-risk Renal Cell Carcinoma After Nephrectomy: Subgroup Analyses and Updated Overall Survival Results. Eur. Urol. 2018, 73, 62–68. [Google Scholar] [CrossRef]
- George, S.; Merriam, P.; Maki, R.G.; Abbeele, A.D.V.D.; Yap, J.; Akhurst, T.; Harmon, D.C.; Bhuchar, G.; O’Mara, M.M.; D’Adamo, D.R.; et al. Multicenter Phase II Trial of Sunitinib in the Treatment of Nongastrointestinal Stromal Tumor Sarcomas. J. Clin. Oncol. 2009, 27, 3154–3160. [Google Scholar] [CrossRef]
- Noguchi, R.; Yoshimatsu, Y.; Ono, T.; Sei, A.; Hirabayashi, K.; Ozawa, I.; Kikuta, K.; Kondo, T. Establishment and characterization of NCC-PLPS1-C1, a novel patient-derived cell line of pleomorphic liposarcoma. Hum. Cell 2021, 34, 688–697. [Google Scholar] [CrossRef]
- Basu Mallick, A.; Chawla, S.P. Giant Cell Tumor of Bone: An Update. Curr. Oncol. Rep. 2021, 23, 1–6. [Google Scholar] [CrossRef]
- Palmerini, E.; Staals, E.L.; Jones, L.B.; Donati, D.M.; Longhi, A.; Randall, R.L. Role of (Neo)adjuvant Denosumab for Giant Cell Tumor of Bone. Curr. Treat. Options Oncol. 2020, 21, 68. [Google Scholar] [CrossRef] [PubMed]
| Sample Number | Sex | Age at Diagnosis | Localization | Campanacci Grade | Denosumab Treatment | Cell Line |
|---|---|---|---|---|---|---|
| 1 | M | 33 | Proximal tibia | 3 | Yes † | GCTB1 |
| 2 | M | 46 | Proximal ulna | 3 | Yes | Not derived |
| 3 | M | 27 | Distal radius | 3 | Yes | GCTB3 |
| 4 | M | 37 | Distal radius | 3 | No | GCTB4 |
| 5 | F | 46 | Distal femur | 2 | Yes * | GCTB5 |
| 6 | M | 25 | Proximal fibula | 3 | No | GCTB6 |
| 7 | M | 41 | Distal femur | 2 | No | GCTB7 |
| 8 | M | 28 | Cuboid bone | 3 | Yes | GCTB8 |
| 9 | F | 45 | Proximal tibia | 2 | No | GCTB9 |
| 10 | F | 16 | Proximal fibula | 3 | Yes | GCTB10 |
| 11 | F | 51 | Proximal tibia | 2 | No | Derived but not used |
| 12 | M | 17 | Proximal phalanx of big toe | 3 | Yes # | Not derived |
| 13 | M | 40 | Distal radius | 2 | Yes | Derived but not used |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahdal, M.; Neradil, J.; Mudry, P.; Paukovcekova, S.; Staniczkova Zambo, I.; Urban, J.; Macsek, P.; Pazourek, L.; Tomas, T.; Veselska, R. New Target for Precision Medicine Treatment of Giant-Cell Tumor of Bone: Sunitinib Is Effective in the Treatment of Neoplastic Stromal Cells with Activated PDGFRβ Signaling. Cancers 2021, 13, 3543. https://doi.org/10.3390/cancers13143543
Mahdal M, Neradil J, Mudry P, Paukovcekova S, Staniczkova Zambo I, Urban J, Macsek P, Pazourek L, Tomas T, Veselska R. New Target for Precision Medicine Treatment of Giant-Cell Tumor of Bone: Sunitinib Is Effective in the Treatment of Neoplastic Stromal Cells with Activated PDGFRβ Signaling. Cancers. 2021; 13(14):3543. https://doi.org/10.3390/cancers13143543
Chicago/Turabian StyleMahdal, Michal, Jakub Neradil, Peter Mudry, Silvia Paukovcekova, Iva Staniczkova Zambo, Jiri Urban, Peter Macsek, Lukas Pazourek, Tomas Tomas, and Renata Veselska. 2021. "New Target for Precision Medicine Treatment of Giant-Cell Tumor of Bone: Sunitinib Is Effective in the Treatment of Neoplastic Stromal Cells with Activated PDGFRβ Signaling" Cancers 13, no. 14: 3543. https://doi.org/10.3390/cancers13143543
APA StyleMahdal, M., Neradil, J., Mudry, P., Paukovcekova, S., Staniczkova Zambo, I., Urban, J., Macsek, P., Pazourek, L., Tomas, T., & Veselska, R. (2021). New Target for Precision Medicine Treatment of Giant-Cell Tumor of Bone: Sunitinib Is Effective in the Treatment of Neoplastic Stromal Cells with Activated PDGFRβ Signaling. Cancers, 13(14), 3543. https://doi.org/10.3390/cancers13143543