The Immune Microenvironment of Chordomas: An Immunohistochemical Analysis
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Suzuki, K.; Kachala, S.S.; Kadota, K.; Shen, R.; Mo, Q.; Beer, D.G.; Rusch, V.W.; Travis, W.D.; Adusumilli, P.S. Prognostic Immune Markers in Non–Small Cell Lung Cancer. Clin. Cancer Res. 2011, 17, 5247–5256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karpathiou, G.; Casteillo, F.; Giroult, J.-B.; Forest, F.; Fournel, P.; Monaya, A.; Froudarakis, M.; Dumollard, J.M.; Prades, J.M.; Peoc’h, M. Prognostic impact of immune microenvironment in laryngeal and pharyngeal squamous cell carcinoma: Immune cell subtypes, immuno-suppressive pathways and clinicopathologic characteristics. Oncotarget 2017, 8, 19310–19322. [Google Scholar] [CrossRef] [Green Version]
- Camy, F.; Karpathiou, G.; Dumollard, J.M.; Magne, N.; Perrot, J.L.; Vassal, F.; Picot, T.; Mobarki, M.; Forest, F.; Casteillo, F.; et al. Brain metastasis PD-L1 and CD8 expression is dependent on primary tumor type and its PD-L1 and CD8 status. J. Immunother. Cancer 2020, 8, e000597. [Google Scholar] [CrossRef] [PubMed]
- Petitprez, F.; de Reyniès, A.; Keung, E.Z.; Chen, T.W.-W.; Sun, C.-M.; Calderaro, J.; Jeng, Y.-M.; Hsiao, L.-P.; Lacroix, L.; Bougoüin, A.; et al. B cells are associated with survival and immunotherapy response in sarcoma. Nature 2020, 577, 556–560. [Google Scholar] [CrossRef]
- Walcott, B.P.; Nahed, B.V.; Mohyeldin, A.; Coumans, J.-V.; Kahle, K.T.; Ferreira, M.J. Chordoma: Current concepts, management, and future directions. Lancet Oncol. 2012, 13, e69–e76. [Google Scholar] [CrossRef]
- Klemen, N.D.; Kelly, C.M.; Bartlett, E.K. The emerging role of immunotherapy for the treatment of sarcoma. J. Surg. Oncol. 2021, 123, 730–738. [Google Scholar] [CrossRef]
- Migliorini, D.; Mach, N.; Aguiar, D.; Vernet, R.; Landis, B.N.; Becker, M.; McKee, T.; Dutoit, V.; Dietrich, P.-Y. First report of clinical responses to immunotherapy in 3 relapsing cases of chordoma after failure of standard therapies. Oncoimmunology 2017, 6, e1338235. [Google Scholar] [CrossRef] [Green Version]
- Mathios, D.; Ruzevick, J.; Jackson, C.M.; Xu, H.; Shah, S.; Taube, J.M.; Burger, P.C.; McCarthy, E.F.; Quinones-Hinojosa, A.; Pardoll, D.M.; et al. PD-1, PD-L1, PD-L2 expression in the chordoma microenvironment. J. Neurooncol. 2015, 121, 251–259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feng, Y.; Shen, J.; Gao, Y.; Liao, Y.; Cote, G.; Choy, E.; Chebib, I.; Mankin, H.; Hornicek, F.; Duan, Z. Expression of programmed cell death ligand 1 (PD-L1) and prevalence of tumor-infiltrating lymphocytes (TILs) in chordoma. Oncotarget 2015, 6, 11139–11149. [Google Scholar] [CrossRef] [Green Version]
- Zou, M.-X.; Guo, K.-M.; Lv, G.-H.; Huang, W.; Li, J.; Wang, X.-B.; Jiang, Y.; She, X.-L. Clinicopathologic implications of CD8+/Foxp3+ ratio and miR-574-3p/PD-L1 axis in spinal chordoma patients. Cancer Immunol. Immunother. 2018, 67, 209–224. [Google Scholar] [CrossRef] [PubMed]
- Zou, M.-X.; Lv, G.-H.; Wang, X.-B.; Huang, W.; Li, J.; Jiang, Y.; She, X.-L. Clinical Impact of the Immune Microenvironment in Spinal Chordoma: Immunoscore as an Independent Favorable Prognostic Factor. Neurosurgery 2019, 84, E318–E333. [Google Scholar] [CrossRef] [PubMed]
- Zou, M.; Pan, Y.; Huang, W.; Zhang, T.; Escobar, D.; Wang, X.; Jiang, Y.; She, X.; Lv, G.; Li, J. A four-factor immune risk score signature predicts the clinical outcome of patients with spinal chordoma. Clin. Transl. Med. 2020, 10, 224–237. [Google Scholar] [CrossRef] [PubMed]
- Zou, M.-X.; Peng, A.-B.; Lv, G.-H.; Wang, X.-B.; Li, J.; She, X.-L.; Jiang, Y. Expression of programmed death-1 ligand (PD-L1) in tumor-infiltrating lymphocytes is associated with favorable spinal chordoma prognosis. Am. J. Transl. Res. 2016, 8, 3274–3287. [Google Scholar]
- Zou, M.-X.; Zheng, B.-W.; Liu, F.-S.; Wang, X.-B.; Hu, J.-R.; Huang, W.; Dai, Z.-H.; Zhang, Q.-S.; Liu, F.-B.; Zhong, H.; et al. The Relationship Between Tumor-Stroma Ratio, the Immune Microenvironment, and Survival in Patients With Spinal Chordoma. Neurosurgery 2019, 85, E1095–E1110. [Google Scholar] [CrossRef] [PubMed]
- Ruscetti, M.; Morris, J.P.; Mezzadra, R.; Russell, J.; Leibold, J.; Romesser, P.B.; Simon, J.; Kulick, A.; Ho, Y.; Fennell, M.; et al. Senescence-Induced Vascular Remodeling Creates Therapeutic Vulnerabilities in Pancreas Cancer. Cell 2020, 181, 424–441. [Google Scholar] [CrossRef]
- Torlakovic, E.; Lim, H.J.; Adam, J.; Barnes, P.; Bigras, G.; Chan, A.W.H.; Cheung, C.C.; Chung, J.-H.; Couture, C.; Fiset, P.O.; et al. “Interchangeability” of PD-L1 immunohistochemistry assays: A meta-analysis of diagnostic accuracy. Mod. Pathol. 2020, 33, 4–17. [Google Scholar] [CrossRef]
- Boothman, A.-M.; Scott, M.; Ratcliffe, M.; Whiteley, J.; Dennis, P.A.; Wadsworth, C.; Sharpe, A.; Rizvi, N.A.; Garassino, M.C.; Walker, J. Impact of Patient Characteristics, Prior Therapy, and Sample Type on Tumor Cell Programmed Cell Death Ligand 1 Expression in Patients with Advanced NSCLC Screened for the ATLANTIC Study. J. Thorac. Oncol. 2019, 14, 1390–1399. [Google Scholar] [CrossRef] [PubMed]
- Paver, E.C.; Cooper, W.A.; Colebatch, A.J.; Ferguson, P.M.; Hill, S.K.; Lum, T.; Shin, J.-S.; O’Toole, S.; Anderson, L.; Scolyer, R.A.; et al. Programmed death ligand-1 (PD-L1) as a predictive marker for immunotherapy in solid tumours: A guide to immunohistochemistry implementation and interpretation. Pathology 2021, 53, 141–156. [Google Scholar] [CrossRef]
- Emancipator, K.; Huang, L.; Aurora-Garg, D.; Bal, T.; Cohen, E.E.W.; Harrington, K.; Soulières, D.; Le Tourneau, C.; Licitra, L.; Burtness, B.; et al. Comparing programmed death ligand 1 scores for predicting pembrolizumab efficacy in head and neck cancer. Mod. Pathol. 2021, 34, 532–541. [Google Scholar] [CrossRef] [PubMed]
- Machado, I.; López-Guerrero, J.A.; Scotlandi, K.; Picci, P.; Llombart-Bosch, A. Immunohistochemical analysis and prognostic significance of PD-L1, PD-1, and CD8+ tumor-infiltrating lymphocytes in Ewing’s sarcoma family of tumors (ESFT). Virchows Arch. 2018, 472, 815–824. [Google Scholar] [CrossRef]
- Zhang, C.; Zheng, J.-H.; Lin, Z.-H.; Lv, H.-Y.; Ye, Z.-M.; Chen, Y.-P.; Zhang, X.-Y. Profiles of immune cell infiltration and immune-related genes in the tumor microenvironment of osteosarcoma. Aging 2020, 12, 3486–3501. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Brouchet, A.; Illac, C.; Gilhodes, J.; Bouvier, C.; Aubert, S.; Guinebretiere, J.-M.; Marie, B.; Larousserie, F.; Entz-Werlé, N.; de Pinieux, G.; et al. CD163-positive tumor-associated macrophages and CD8-positive cytotoxic lymphocytes are powerful diagnostic markers for the therapeutic stratification of osteosarcoma patients: An immunohistochemical analysis of the biopsies fromthe French OS2006 phase 3 t. Oncoimmunology 2017, 6, e1331193. [Google Scholar] [CrossRef] [PubMed]
- Fusco, N.; Vaira, V.; Righi, I.; Sajjadi, E.; Venetis, K.; Lopez, G.; Cattaneo, M.; Castellani, M.; Rosso, L.; Nosotti, M.; et al. Characterization of the immune microenvironment in malignant pleural mesothelioma reveals prognostic subgroups of patients. Lung Cancer 2020, 150, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Jia, Q.; Wei, H.; Yang, X.; Liu, T.; Zhao, J.; Ling, Y.; Wang, C.; Yu, H.; Li, Z.; et al. Apatinib in patients with advanced chordoma: A single-arm, single-centre, phase 2 study. Lancet Oncol. 2020, 21, 1244–1252. [Google Scholar] [CrossRef]
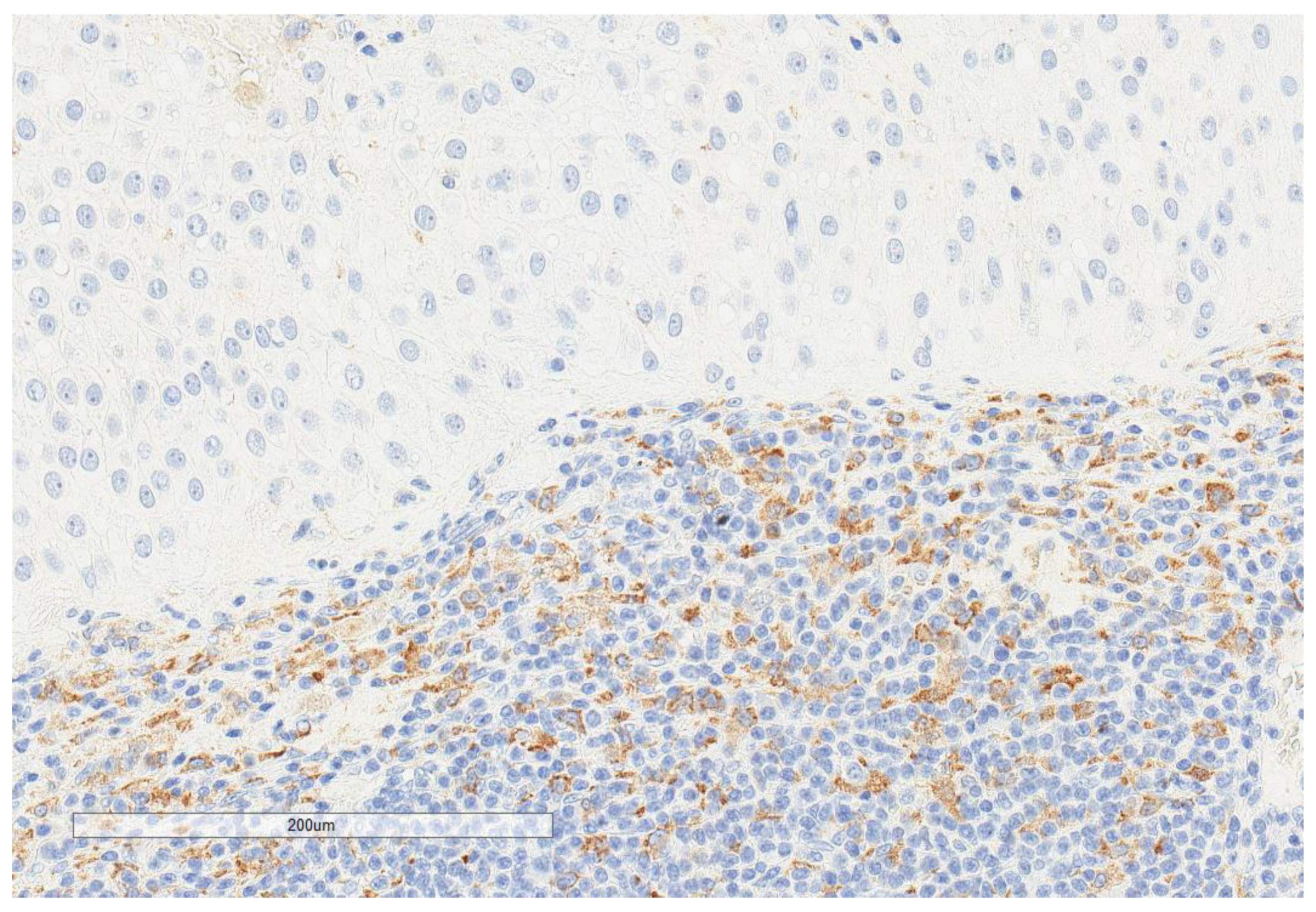
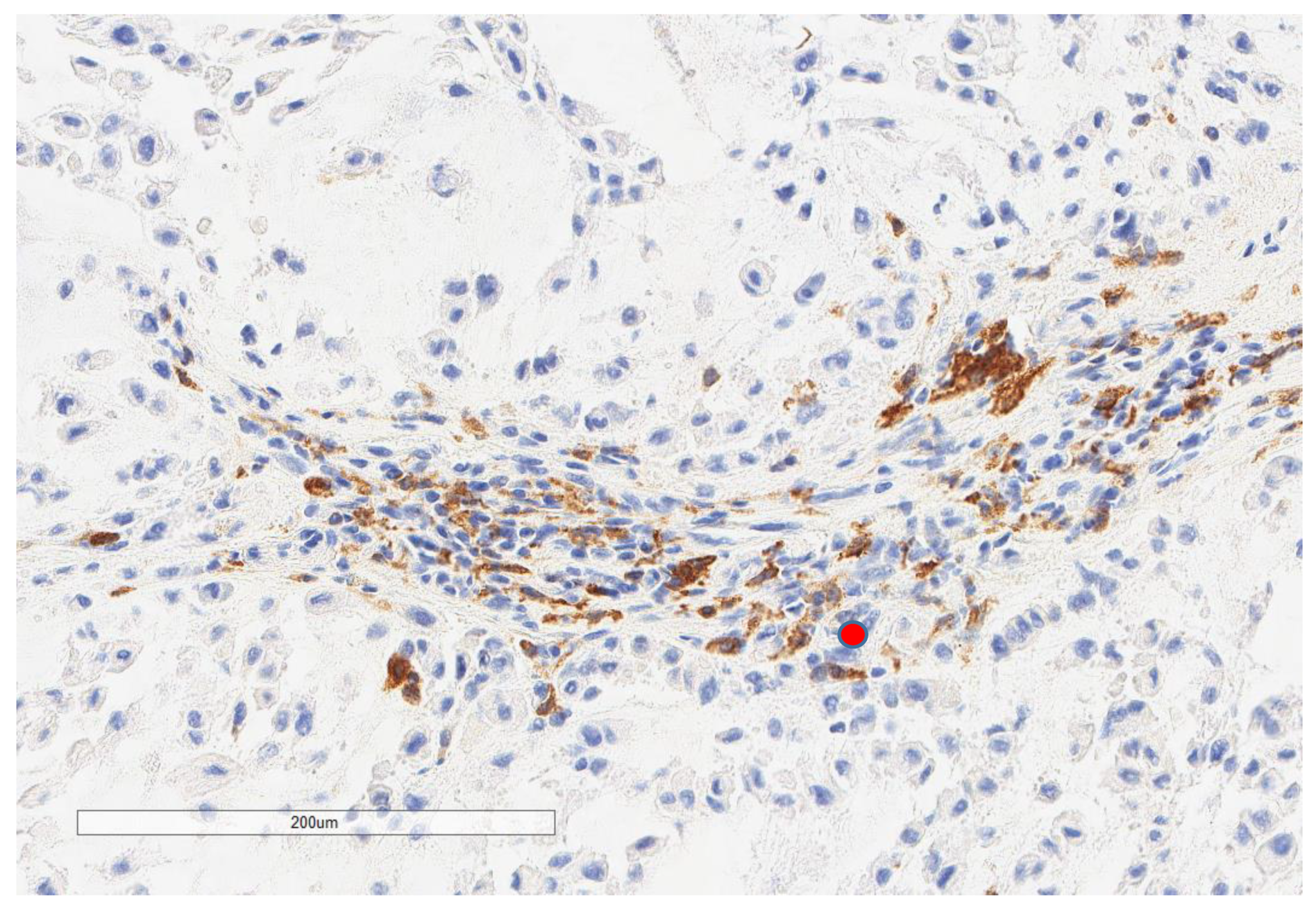

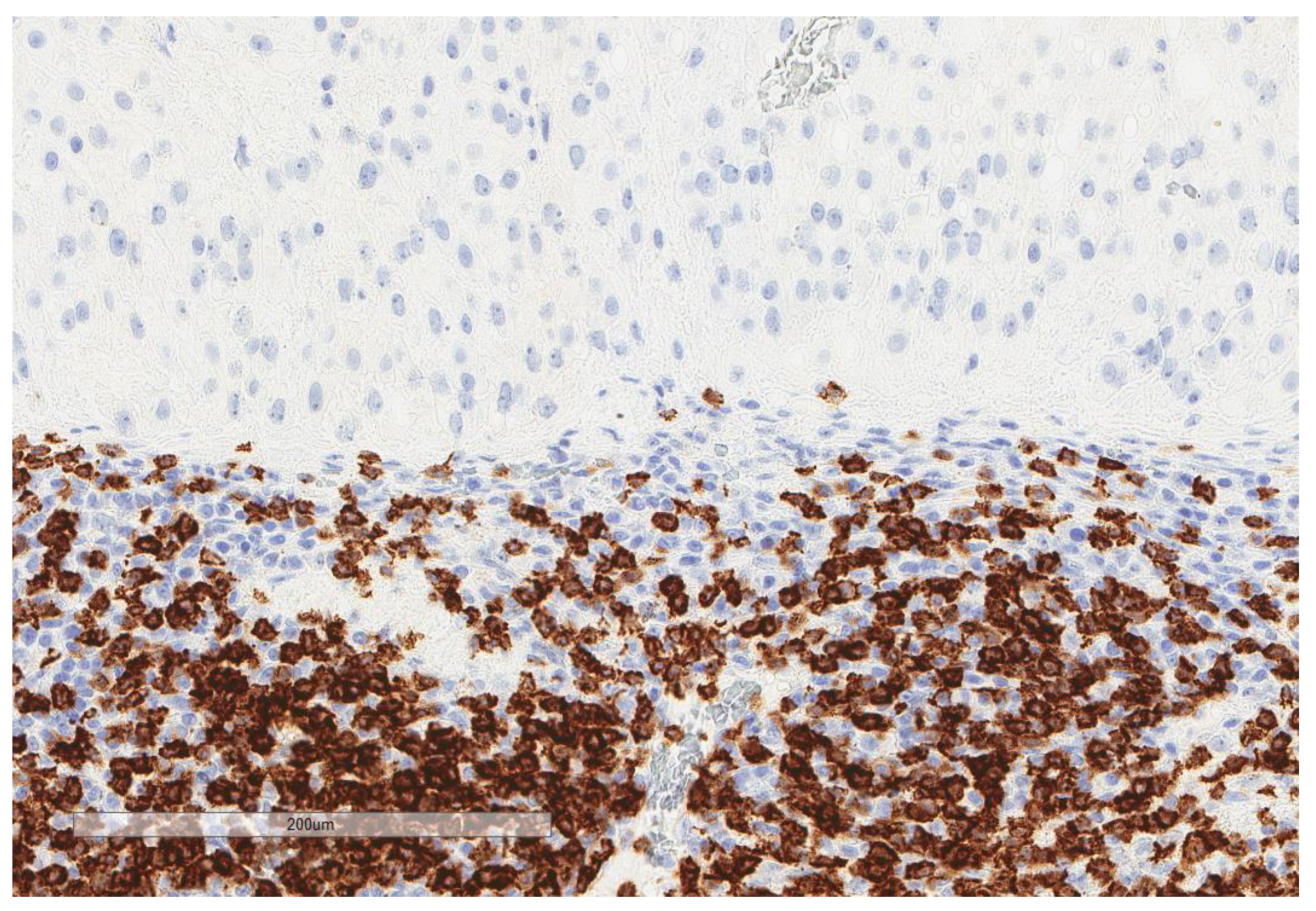

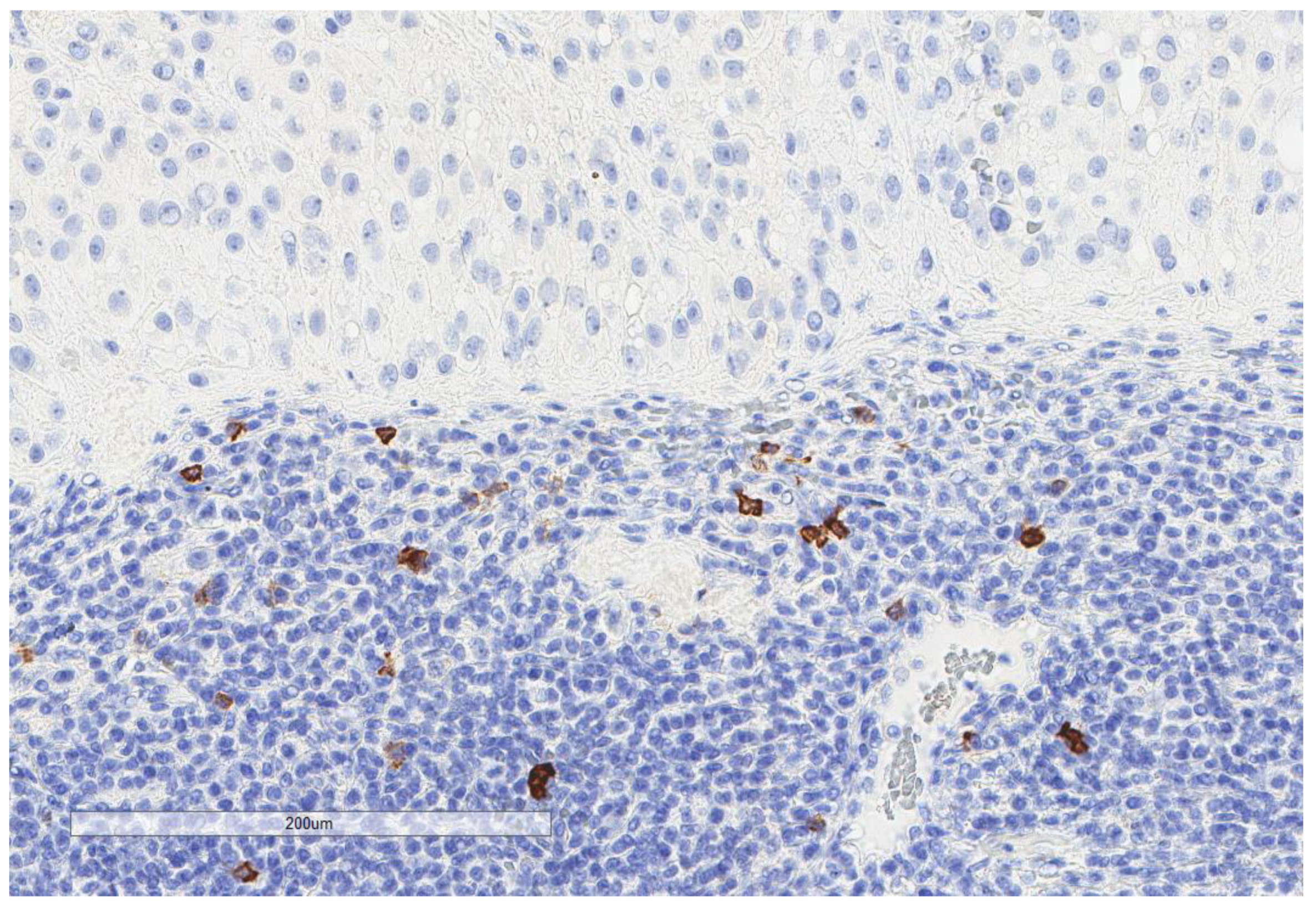
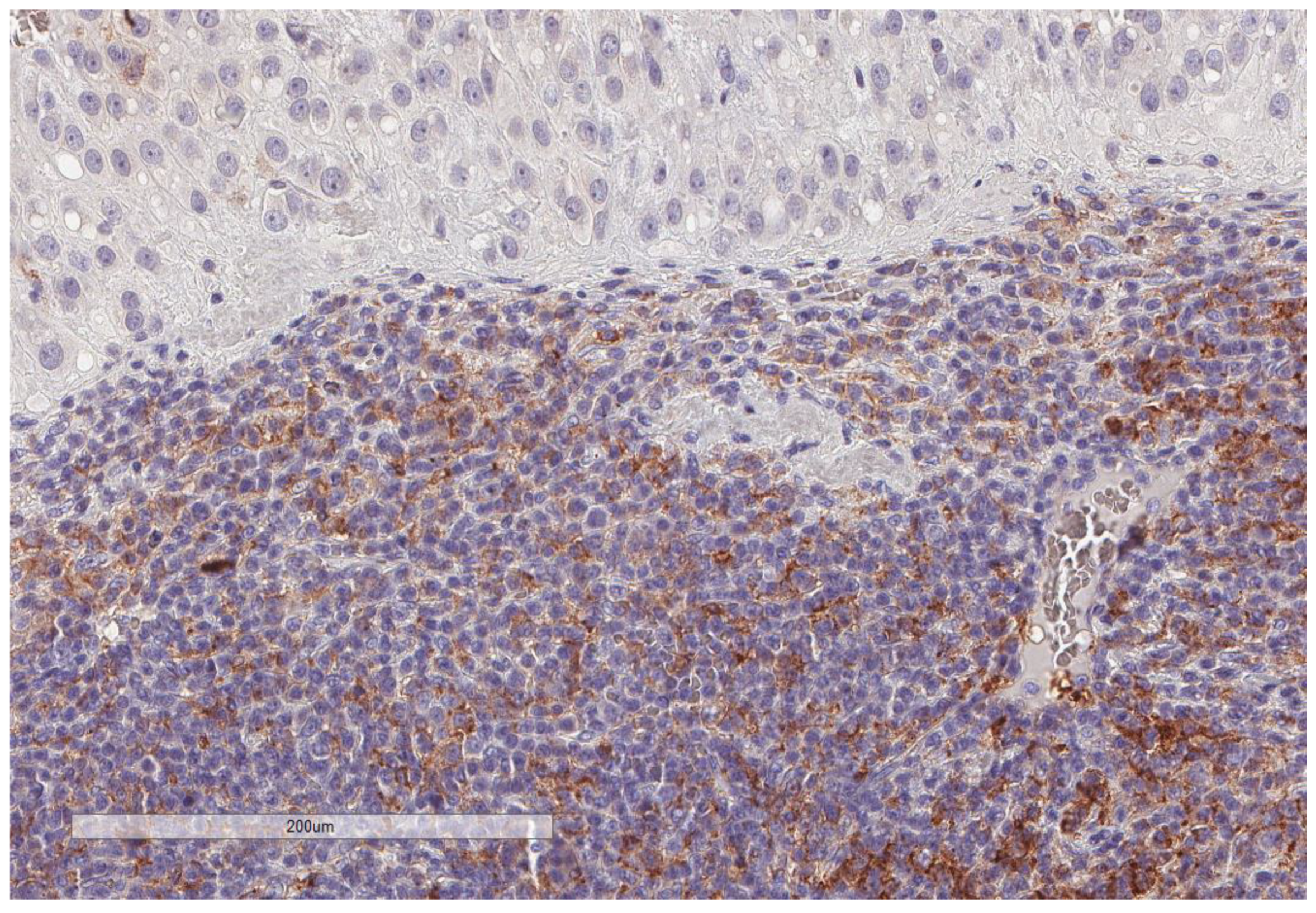

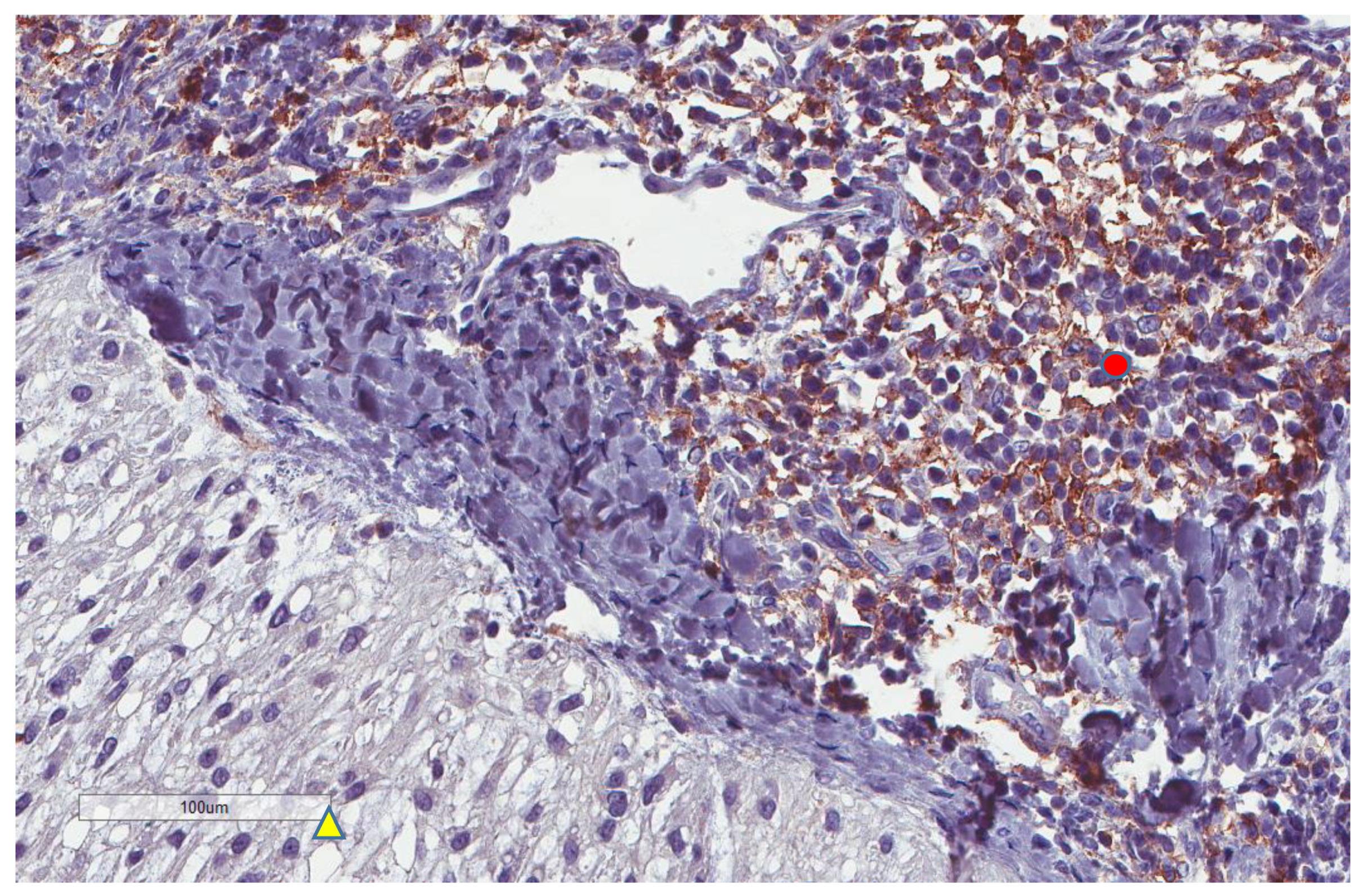

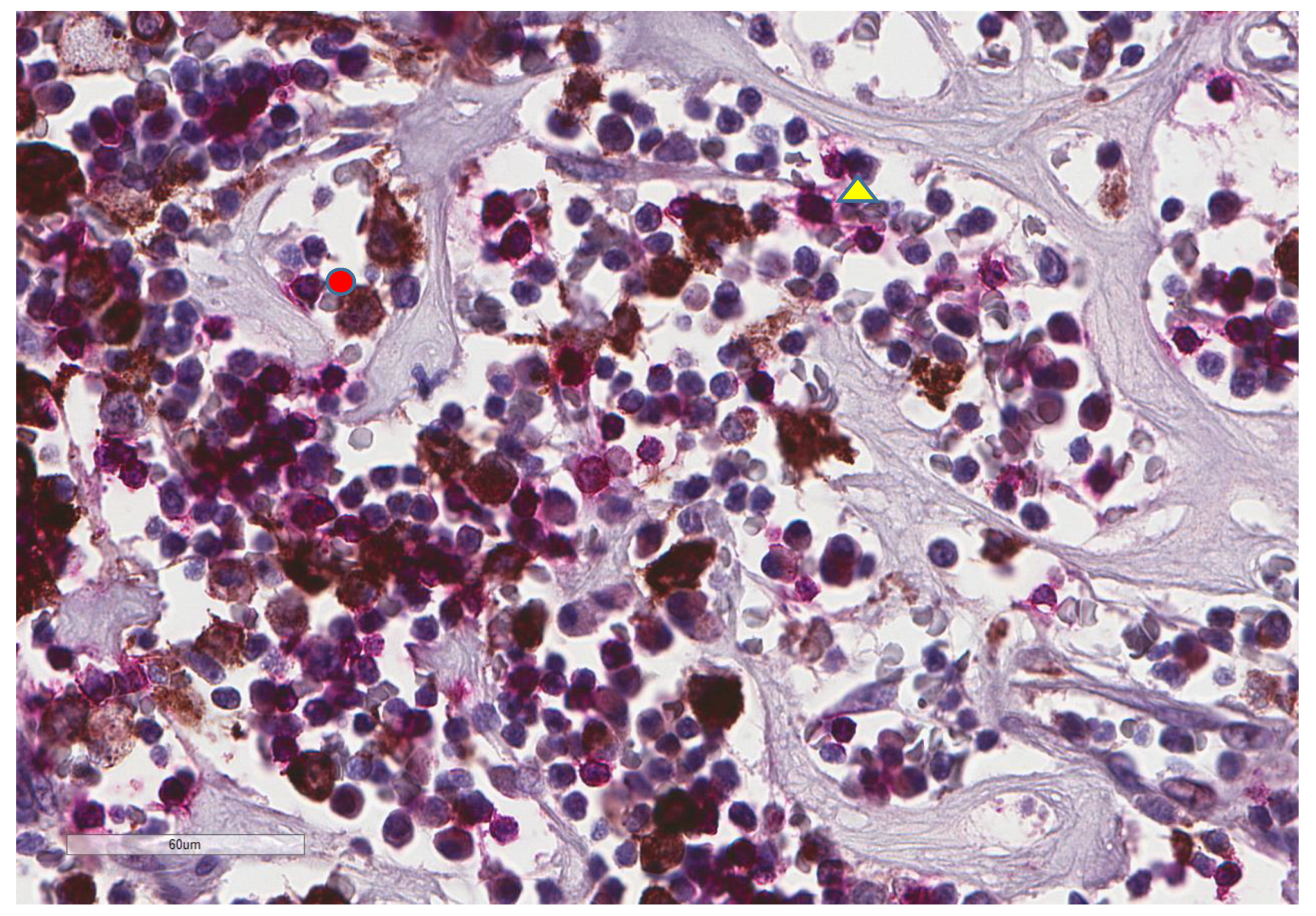

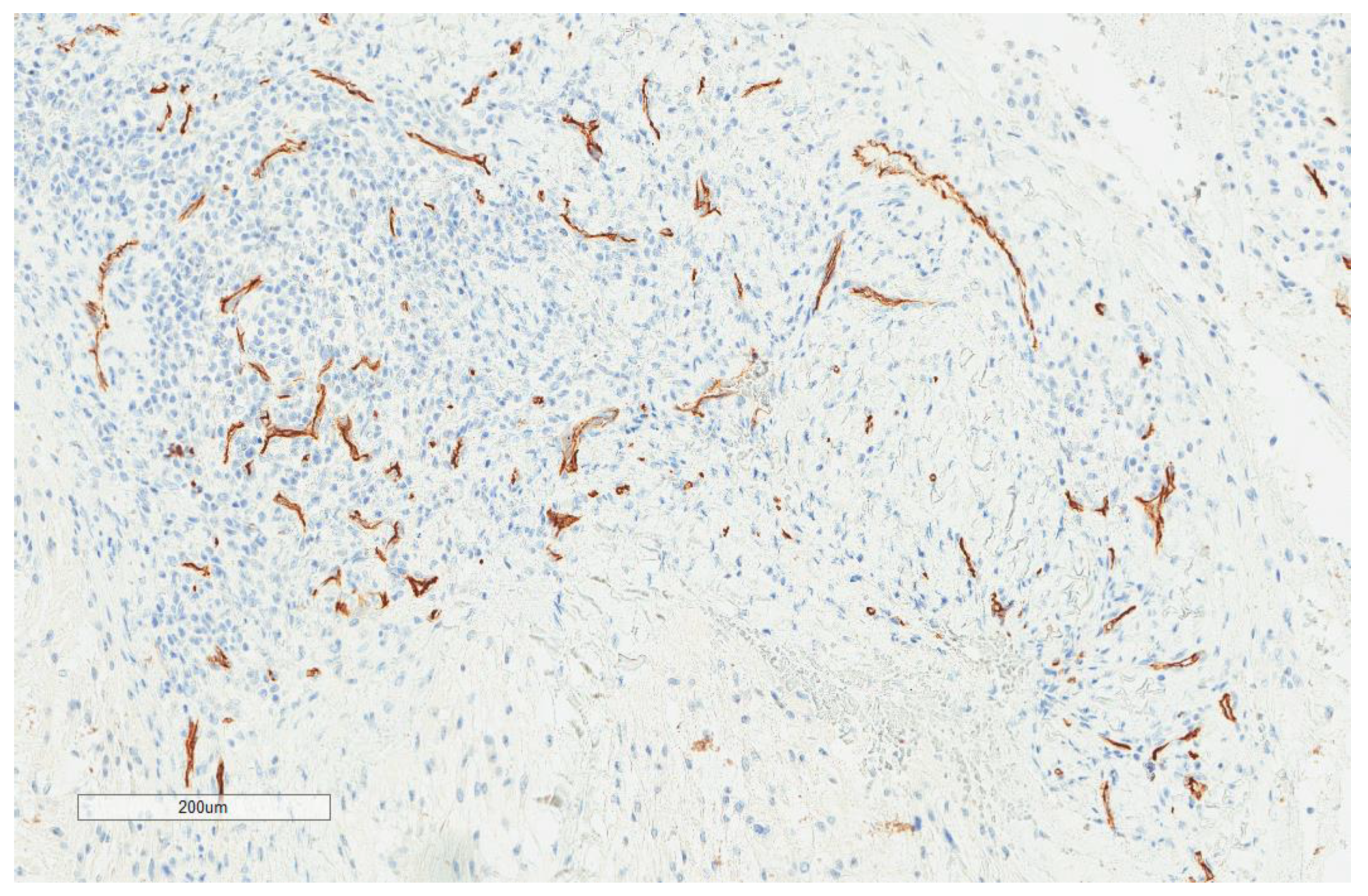
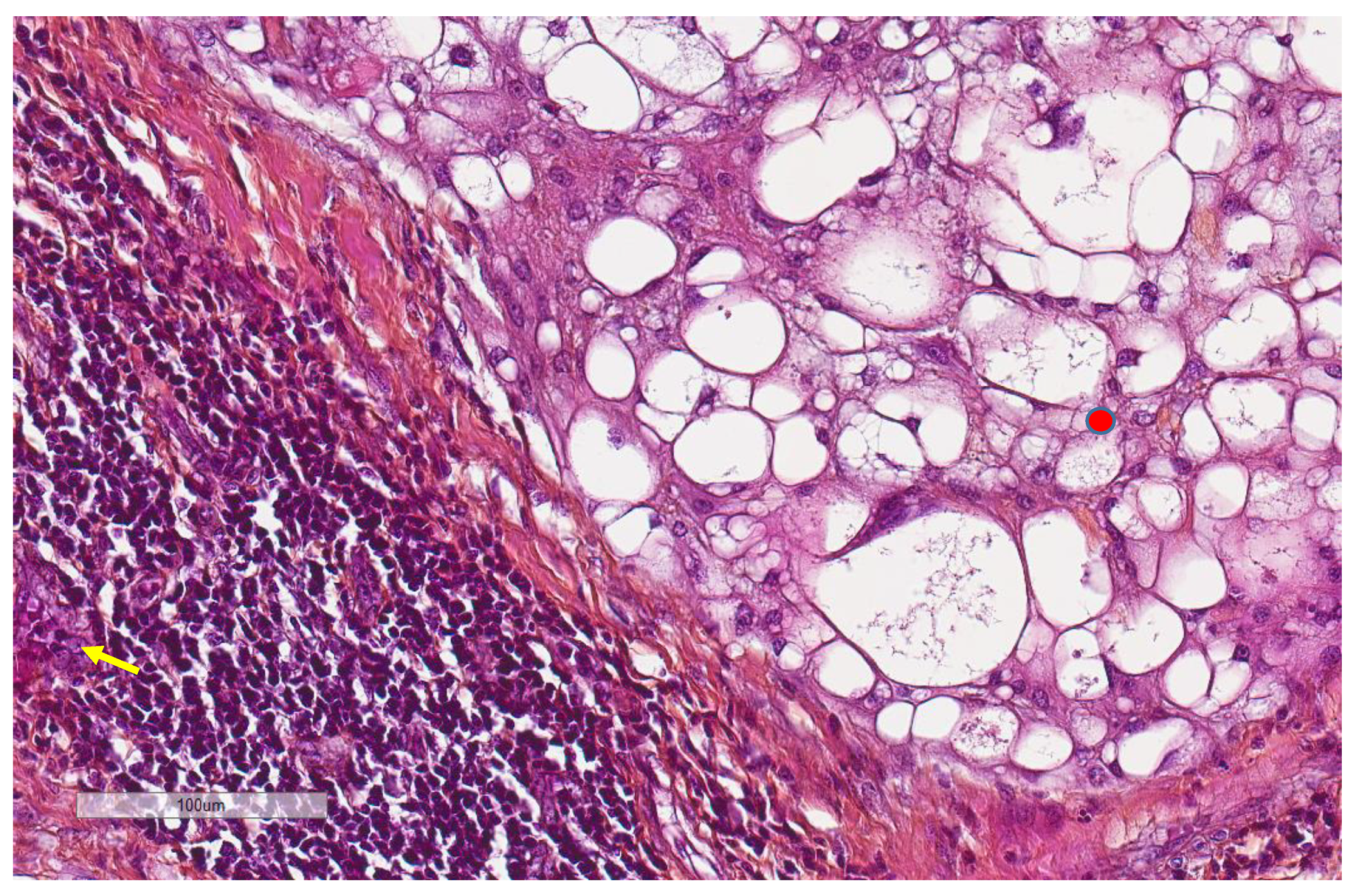
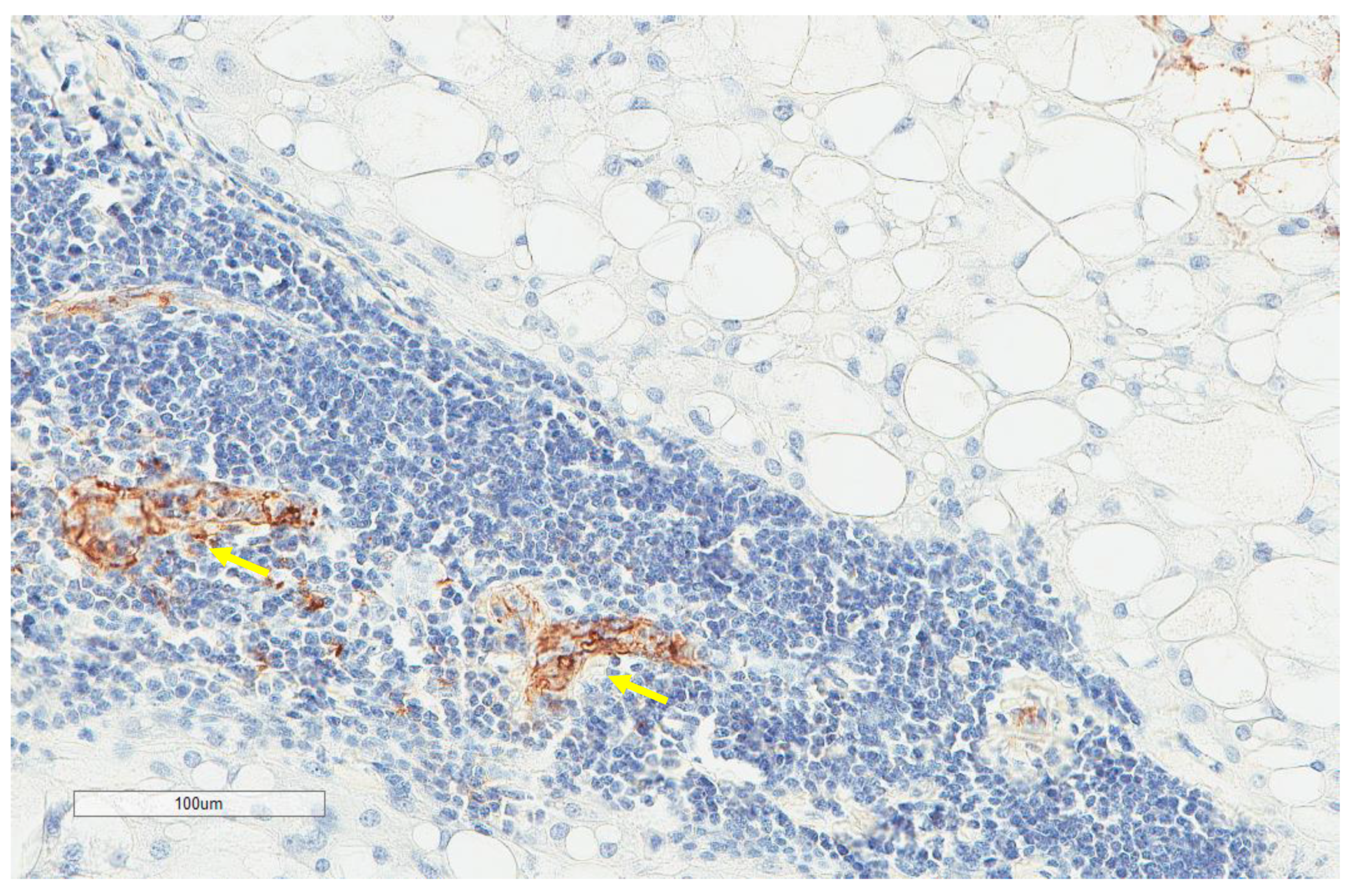
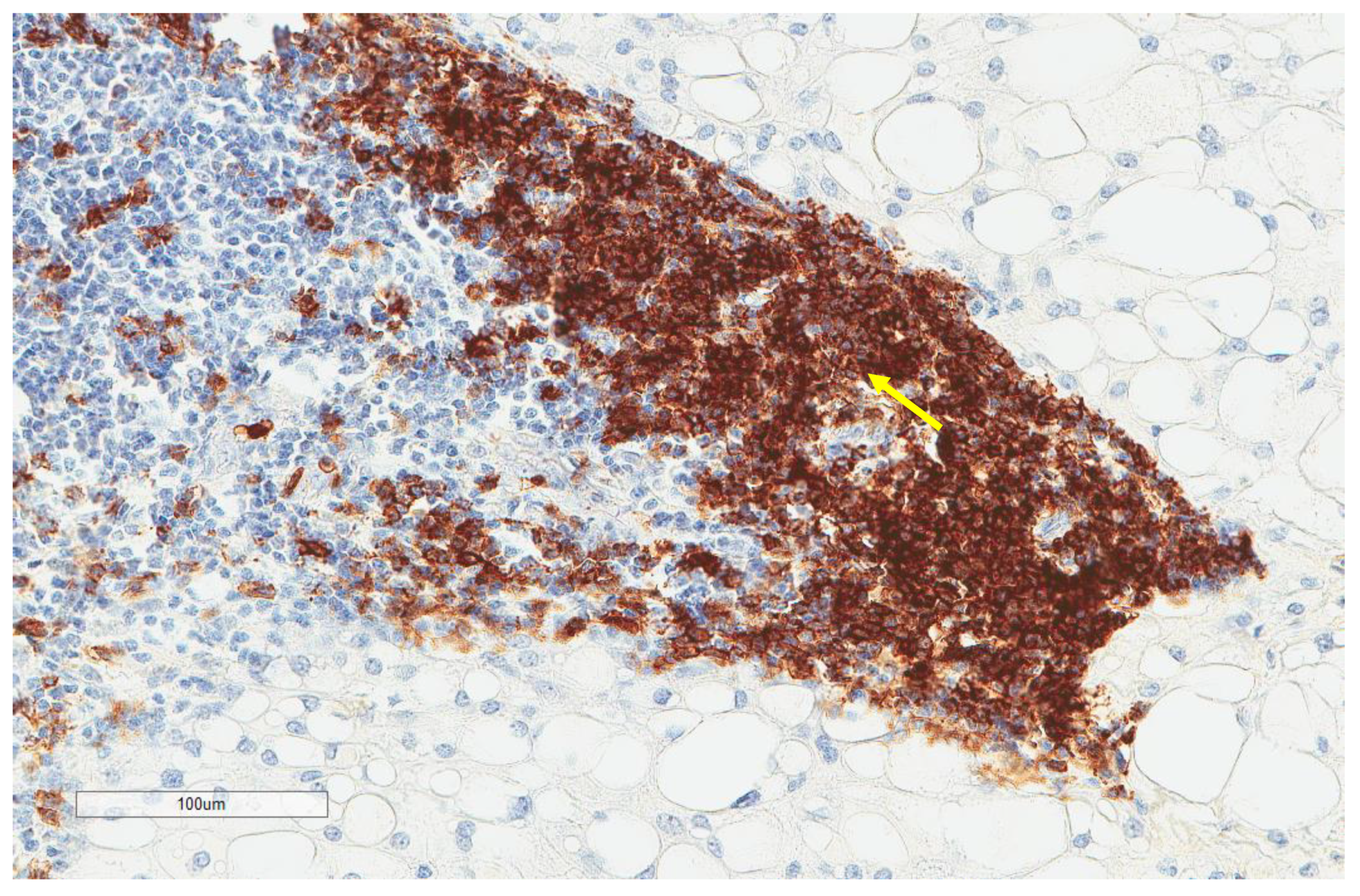
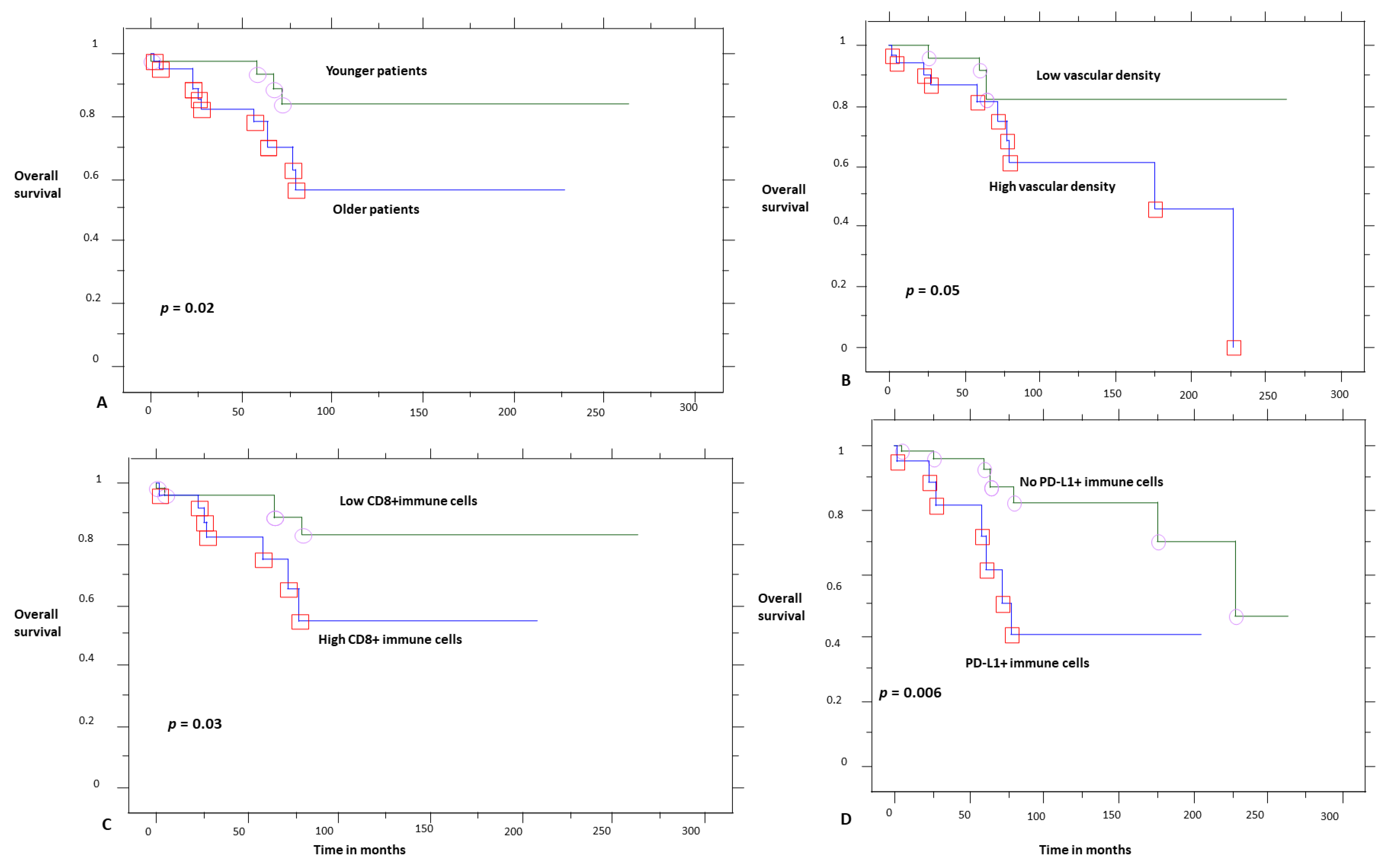
| Parameter | n, % |
|---|---|
| Age (n = 81) | |
| Range | 12–82 |
| Median | 64 |
| Mean ± SD | 58 ± 17.4 |
| Sex (n = 81) | |
| Female | 34, 42% |
| Male | 47, 58% |
| Localization (n = 81) | |
| Skull | 30, 37% |
| Sacrum | 27, 33.3% |
| Mobile spine | 24, 29.7% |
| Recurrence (n = 81) | |
| Yes | 52, 64.2% |
| No | 29, 35.8% |
| Multiple recurrences (n = 80) | |
| Yes | 24, 30% |
| No | 56, 70% |
| Tumor size (in mm **) | |
| Range | 19–144 |
| Median | 43 |
| Mean ± SD | 50.6 ± 27.5 |
| Principal treatment at diagnosis (n = 85) | |
| Surgical | 72, 84.7% |
| Radiotherapy | 10, 11.8% |
| Palliative | 3, 3.5% |
| Adjuvant therapy (n = 40) | |
| Radiotherapy | 36, 90% |
| Surgical | 3, 7.5% |
| immunotherapy | 1, 2.5% |
| Follow-up (n = 81) | |
| Range | 2–264 |
| Median | 60 |
| Mean ± SD | 69.5 ± 59.5 |
| Patient status (n = 81) | |
| Alive | 66, 81.5% |
| Dead * | 15, 18.5% |
| Overall survival (n = 81) | |
| Range | 2–264 |
| Median (log-rank) | Not reached |
| Parameter | n, % |
|---|---|
| CD20 (n = 73) | |
| Low (score 0–1) | 65, 89% |
| High (score 2–3) | 8, 11% |
| CD8 (n = 81) | |
| Low (score 0–1) | 55, 67.9% |
| High (score 2–3) | 26, 32.1% |
| CD4 (n = 41) | |
| Low (score 0–1) | 25, 61% |
| High (score 2–3) | 16, 39% |
| CD163 (n = 74) | |
| Low (score 0–1) | 45, 60.8% |
| High (score 2–3) | 29, 39.2% |
| PD-L1 inflammatory cells (n = 81) | |
| No (score 0) | 60, 74% |
| Yes (score 1–3) | 21, 26% |
| CD34 vascular density (n = 74) | |
| Range | 1–22 |
| Median (High and Low, n,%) | 3.5 (37, 50% and 37, 50%) |
| Mean ± SD | 5.8 ± 5.7 |
| MECA-79 vessels (n = 59) | |
| High | 5, 8.5% |
| Low | 54, 91.5% |
| Parameter | CD8 | CD163 | MECA-79 | CD34 | PD-L1 Inflammatory Cells | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| High | Low | p | High | Low | p | High | Low | p | High | Low | p | Yes | No | p | |
| CD20 | |||||||||||||||
| High | 6 (8.3%) | 2 (2.7%) | 0.01 * | 3 (4.1%) | 5 (6.9%) | 0.8 | 2 (3.4%) | 6 (10.2%) | 0.07 | 4 (5.5%) | 4 (5.5%) | 0.9 | 2 (2.7%) | 6 (8.2%) | 0.9 |
| Low | 20 (27.4%) | 45 (61.6%) | 26 (35.6%) | 39 (53.4%) | 3 (5.1%) | 48 (81.3%) | 33 (45.2%) | 32 (43.8%) | 17 (23.3%) | 48 (65.8%) | |||||
| CD8 | |||||||||||||||
| High | 14 (18.9%) | 12 (16.2%) | 0.05 | 2 (3.4%) | 24 (40.7%) | 0.8 | 22 (29.7%) | 4 (5.4%) | <0.0001 § | 13 (16.1%) | 13 (16.1%) | 0.0007 £ | |||
| Low | 15 (20.3%) | 33 (44.6%) | 3 (5.1%) | 30 (50.8%) | 15 (20.3%) | 33 (44.6%) | 8 (9.9%) | 47 (58%) | |||||||
| CD163 | |||||||||||||||
| High | 2 (3.4%) | 26 (44.1%) | 0.7 | 19 (25.7%) | 10 (13.5%) | 0.03 & | 14 (18.9%) | 15 (20.2%) | 0.0004 ° | ||||||
| Low | 3 (5.1%) | 28 (47.4%) | 18 (24.3%) | 27 (36.5%) | 5 (6.7%) | 40 (54%) | |||||||||
| MECA-79 | |||||||||||||||
| High | 3 (5.1%) | 2 (3.4%) | 0.8 | 2 (3.4%) | 3 (5.1%) | 0.6 | |||||||||
| Low | 30 (50.8%) | 24 (40.7%) | 16 (27.1%) | 38 (64.4%) | |||||||||||
| CD34 | |||||||||||||||
| High | 16 (21.6%) | 21 (28.4%) | 0.0005 + | ||||||||||||
| Low | 3 (4%) | 34 (46%) | |||||||||||||
| Parameter | Ten-Year Overall Survival Probability | p |
|---|---|---|
| CD20 | ||
| Low | 70% | 0.7 |
| High | 81% | |
| CD8 | ||
| Low | 81% | 0.03 |
| High | 51% | |
| CD4 | ||
| Low | 71% | 0.9 |
| High | 81% | |
| CD163 | ||
| Low | 71% | 0.9 |
| High | 71% | |
| PD-L1 inflammatory cells | ||
| No | 81% | 0.02 |
| Yes | 46% | |
| MECA-79 | ||
| High | 76% | 0.8 |
| Low | 61% | |
| CD34 | ||
| Low | 81% | 0.05 |
| High | 60% | |
| Tumor size | ||
| Low | 100% | 0.2 |
| High | 60% | |
| Patient age | ||
| Low | 82% | 0.02 |
| High | 58% | |
| Tissue age | ||
| Low | 71% | 0.9 |
| High | 71% |
| Variable | HR | CI | p |
|---|---|---|---|
| Age | 1.031 | 0.988–1.076 | 0.1646 |
| CD8 (absence) | 0.427 | 0.106–1.726 | 0.2325 |
| PD-L1 immune cells (absence) | 0.188 | 0.044–0.815 | 0.0255 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dridi, M.; Krebs-Drouot, L.; Meyronet, D.; Dumollard, J.M.; Vassal, F.; Jouanneau, E.; Jacquesson, T.; Barrey, C.; Grange, S.; Boutonnat, J.; et al. The Immune Microenvironment of Chordomas: An Immunohistochemical Analysis. Cancers 2021, 13, 3335. https://doi.org/10.3390/cancers13133335
Dridi M, Krebs-Drouot L, Meyronet D, Dumollard JM, Vassal F, Jouanneau E, Jacquesson T, Barrey C, Grange S, Boutonnat J, et al. The Immune Microenvironment of Chordomas: An Immunohistochemical Analysis. Cancers. 2021; 13(13):3335. https://doi.org/10.3390/cancers13133335
Chicago/Turabian StyleDridi, Maroa, Lila Krebs-Drouot, David Meyronet, Jean Marc Dumollard, François Vassal, Emmanuel Jouanneau, Timothée Jacquesson, Cédric Barrey, Sylvain Grange, Jean Boutonnat, and et al. 2021. "The Immune Microenvironment of Chordomas: An Immunohistochemical Analysis" Cancers 13, no. 13: 3335. https://doi.org/10.3390/cancers13133335
APA StyleDridi, M., Krebs-Drouot, L., Meyronet, D., Dumollard, J. M., Vassal, F., Jouanneau, E., Jacquesson, T., Barrey, C., Grange, S., Boutonnat, J., Péoc’h, M., & Karpathiou, G. (2021). The Immune Microenvironment of Chordomas: An Immunohistochemical Analysis. Cancers, 13(13), 3335. https://doi.org/10.3390/cancers13133335






