Genetic and Proteinic Linkage of MAO and COMT with Oral Potentially Malignant Disorders and Cancers of the Oral Cavity and Pharynx
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.2. Genetic Polymorphisms Analysis of MAO and COMT
2.3. Single Nucleotide Polymorphism Assay
2.4. Cell Cultures and Cytotoxicity Assay
2.5. Real-Time qRT-PCR Analysis
2.6. Protein Extraction and Western Blotting
2.7. Immunohistochemistry
2.8. Statistical Analysis
3. Results
3.1. Characterization of Study Population
3.2. SNPs of MAO/COMT and Substance Use (BQ, Cigarette, and Alcohol) on Risk Assessment of Oral and Pharyngeal Cancers and OPMD
3.3. Gene–Gene Joint Effects of MAO/COMT SNPs on Risk Assessment
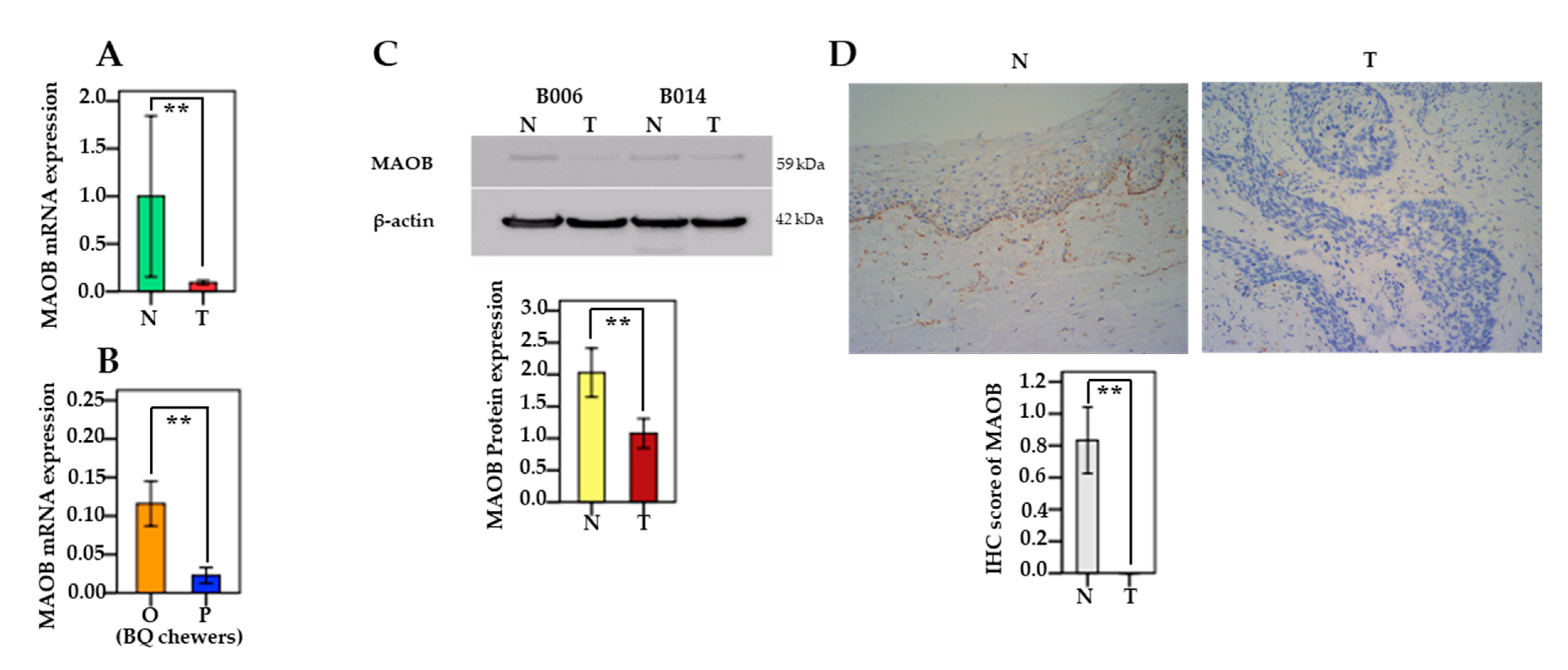
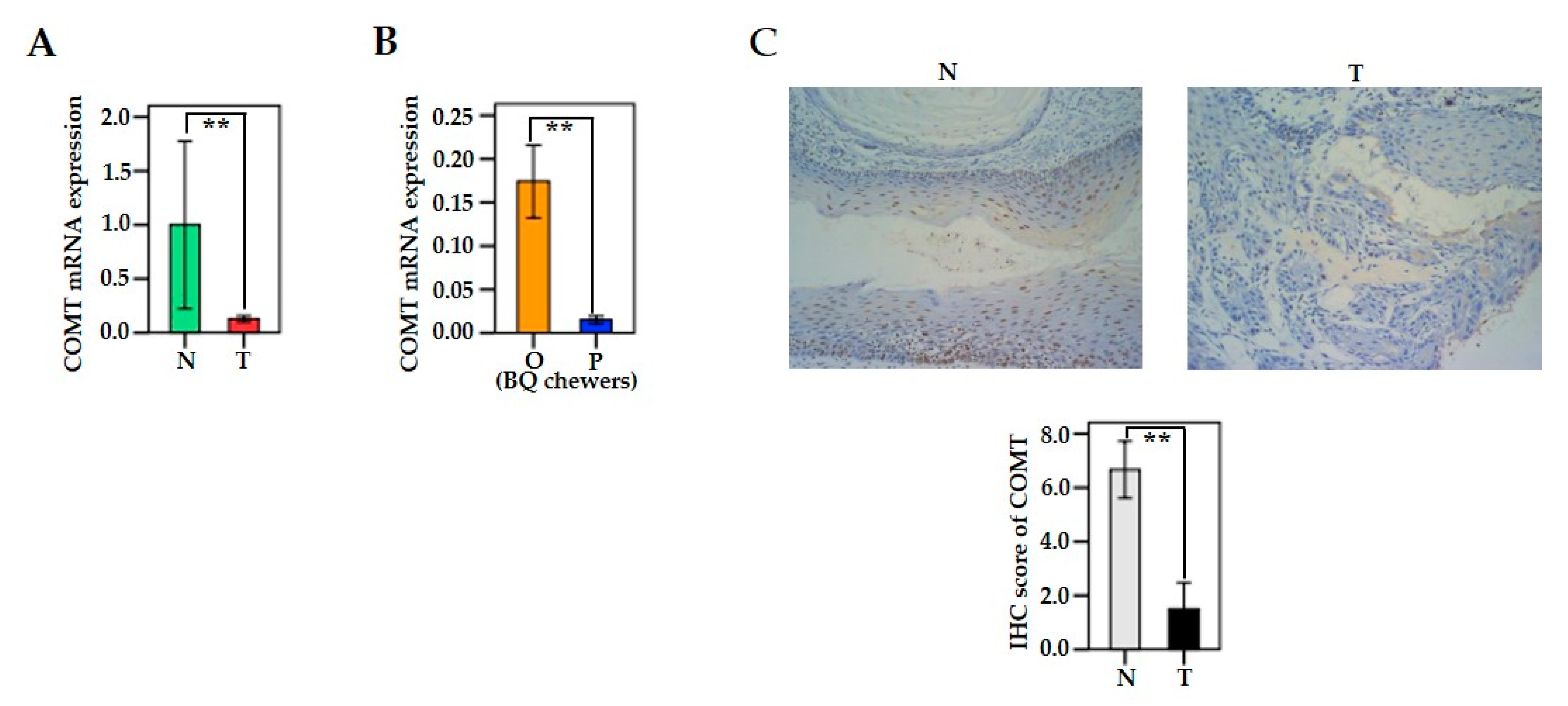
3.4. The mRNA and Protein Levels of MAO/COMT in Oral and Pharyngeal Cancerous Tissues and Non-Cancerous Tissues
3.5. Associations Between MAO/COMT mRNAs and Genetic Polymorphisms
3.6. Diagnostic Performance of MAO/COMT Biomarkers
3.7. Associations between MAO/COMT mRNA and BQ Use
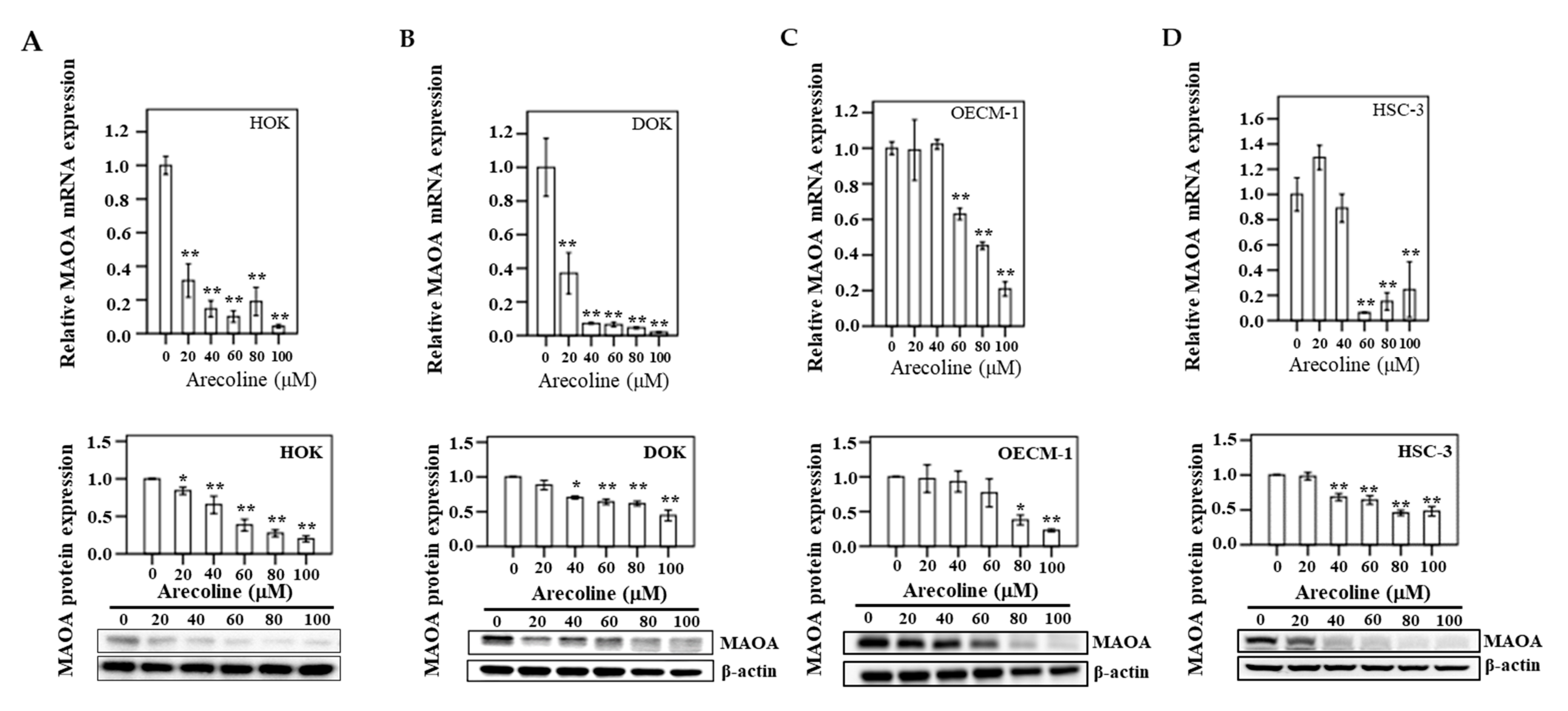
3.8. MAOA Expression in Human Oral Epithelial Cells
3.9. MAOB Expression in Human Oral Epithelial Cells
3.10. COMT Expression in Human Oral Epithelial Cells
4. Discussion
Study Limitation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- International Agency for Research on Cancer. Global Cancer Observatory. Cancer Today. 2019. Available online: https://gco.iarc.fr/. (accessed on 16 September 2019).
- Chen, P.H.; Mahmood, Q.; Mariottini, G.L.; Chiang, T.A.; Lee, K.W. Adverse health effects of betel quid and the risk of oral and pharyngeal cancers. Biomed. Res. Int. 2017, 2017, 3904098. [Google Scholar] [CrossRef] [Green Version]
- Cancer Registration System Annual Report; Ministry of Health and Welfare: Taipei City, Taiwan, 2018.
- Gupta, P.C.; Ray, C.S. Epidemiology of betel quid usage. Ann. Acad. Med. Singap. 2004, 33, 31–36. [Google Scholar]
- Ko, Y.C.; Chiang, T.A.; Chang, S.J.; Hsieh, S.F. Prevalence of betel quid chewing habit in Taiwan and related sociodemographic factors. J. Oral Pathol. Med. 1992, 21, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Ko, A.M.; Warnakulasuriya, S.; Yin, B.L.; Sunarjo, Z.R.B.; Ibrahim, S.O.; Liu, Z.W.; Li, W.H.; Zhang, S.S.; Ibrahim, S.O.; et al. Intercountry prevalences and practices of betel-quid use in south, southeast and eastern Asia regions and associated oral preneoplastic disorders: An international collaborative study by Asian betel-quid consortium of south and east Asia. Int. J. Cancer 2011, 129, 1741–1751. [Google Scholar] [CrossRef] [Green Version]
- Edmondson, D.E. Hydrogen peroxide produced by mitochondrial monoamine oxidase catalysis: Biological implications. Curr. Pharm. Des. 2014, 20, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Bach, A.W.; Lan, N.C.; Johnson, D.L.; Abell, C.W.; Bembenek, M.E.; Kwan, S.W.; Seeburg, P.H.; Shih, J.C. cDNA cloning of human liver monoamine oxidase A and B: Molecular basis of differences in enzymatic properties. Proc. Natl. Acad. Sci. USA 1988, 85, 4934–4938. [Google Scholar] [CrossRef] [Green Version]
- Berry, M.D.; Juorio, A.V.; Paterson, I.A. The functional role of monoamine oxidases A and B in the mammalian central nervous system. Prog. Neurobiol. 1994, 42, 375–391. [Google Scholar] [CrossRef]
- van Amsterdam, J.; Talhout, R.; Vleeming, W.; Opperhuizen, A. Contribution of monoamine oxidase (MAO) inhibition to tobacco and alcohol addiction. Life Sci. 2006, 79, 1969–1973. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Breakefield, X.O. Human monoamine oxidase A gene determines levels of enzyme activity. Am. J. Hum. Genet. 1991, 49, 383–392. [Google Scholar]
- Shih, J.C. Monoamine oxidase isoenzymes: Genes, functions and targets for behavior and cancer therapy. J. Neural Transm. 2018, 125, 1553–1566. [Google Scholar] [CrossRef]
- Rybaczyk, L.A.; Bashaw, M.J.; Pathak, D.R.; Huang, K. An indicator of cancer: Downregulation of monoamine oxidase-A in multiple organs and species. BMC Genom. 2008, 9, 134. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Yang, X.M.; Wang, Y.H.; Feng, M.X.; Liu, X.J.; Zhang, Y.L.; Huang, S.; Wu, Z.; Xue, F.; Qin, W.X.; et al. Monoamine oxidase A suppresses hepatocellular carcinoma metastasis by inhibiting the adrenergic system and its transactivation of EGFR signaling. J. Hepatol. 2014, 60, 1225–1234. [Google Scholar] [CrossRef] [Green Version]
- Grossman, M.H.; Emanuel, B.S.; Budarf, M.L. Chromosomal mapping of the human catechol-O-methyltransferase gene to 22q11.1----q11.2. Genomics 1992, 12, 822–825. [Google Scholar] [CrossRef]
- Cavalieri, E.L.; Stack, D.E.; Devanesan, P.D.; Todorovic, R.; Dwivedy, I.; Higginbotham, S.; Johansson, S.L.; Patil, K.D.; Gross, M.L.; Gooden, J.K.; et al. Molecular origin of cancer: Catechol estrogen-3,4-quinones as endogenous tumor initiators. Proc. Natl. Acad. Sci. USA 1997, 94, 10937–10942. [Google Scholar] [CrossRef] [Green Version]
- Bastos, P.; Gomes, T.; Ribeiro, L. Catechol-O-methyltransferase (COMT): An update on its role in cancer, neurological and cardiovascular diseases. Rev. Physiol. Biochem. Pharm. 2017, 173, 1–39. [Google Scholar]
- Chen, P.H.; Tu, H.P.; Wang, S.J.; Ko, A.M.; Lee, C.P.; Chiang, T.A.; Tsai, Y.S.; Lee, C.H.; Shieh, T.Y.; Ko, C.H.; et al. Monoamine oxidase A variants are associated with heavy betel quid use. Addict. Biol. 2012, 17, 786–797. [Google Scholar] [CrossRef]
- Ko, A.M.; Lee, C.H.; Ko, A.M.; Ko, Y.C. Betel quid dependence mechanism and potential cessation therapy. Prog. Neuropsychopharmacol. Biol. Psychiatry 2020, 103, 109982. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.Y.; Wang, W.C.; Su, C.W.; Hsu, C.W.; Yuan, S.S.; Chen, Y.K. Overexpression of sprouty 1 protein in human oral squamous cell carcinogenesis. J. Dent. Sci. 2021, 16, 21–28. [Google Scholar] [CrossRef]
- Chen, P.H.; Chung, C.M.; Wang, Y.Y.; Huang, H.W.; Huang, B.; Lee, K.W.; Yuan, S.S.; Wu, C.W.; Lin, L.S.; Chan, L.P. CYP26A1 is a novel biomarker for betel quid-related oral and pharyngeal cancers. Diagnostics 2020, 10, 982. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.H.; Lee, K.W.; Chen, C.H.; Shieh, T.Y.; Ho, P.S.; Wang, S.J.; Lee, C.H.; Yang, S.F.; Chen, M.K.; Chiang, S.L.; et al. CYP26B1 is a novel candidate gene for betel quid-related oral squamous cell carcinoma. Oral Oncol. 2011, 47, 594–600. [Google Scholar] [CrossRef]
- Wu, S.J.; Chen, Y.J.; Shieh, T.Y.; Chen, C.M.; Wang, Y.Y.; Lee, K.T.; Lin, Y.M.; Chien, P.H.; Chen, P.H. Association study between novel CYP26 polymorphisms and the risk of betel quid-related malignant oral disorders. Sci. World J. 2015, 2015, 160185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Franzblau, A. A Primer of Statistics for Non-Statisticians; Harcourt Brace & World: New York, NY, USA, 1958. [Google Scholar]
- Singh, J.A.; Yang, S.; Strand, V.; Simon, L.; Forsythe, A.; Hamburger, S.; Chen, L. Validation of pain and patient global scales in chronic gout: Data from two randomised controlled trials. Ann. Rheum. Dis. 2011, 70, 1277–1281. [Google Scholar] [CrossRef] [Green Version]
- Winstock, A.R.; Trivedy, C.R.; Warnakulasuriya, K.A.A.S.; Peters, T.J. A dependency syndrome related to areca nut use: Some medical and psychological aspects among areca nut users in the Gujarat community in the UK. Addict. Biol 2000, 5, 173–179. [Google Scholar] [CrossRef]
- IARC Betel-quid and areca-nut chewing and some areca-nut-derived nitrosamines. IARC Monogr Eval. Carcinog. Risks Hum. 2004, 85, 1–334.
- Lee, C.H.; Ko, Y.C.; Huang, H.L.; Chao, Y.Y.; Tsai, C.C.; Shieh, T.Y.; Lin, L.M. The precancer risk of betel quid chewing, tobacco use and alcohol consumption in oral leukoplakia and oral submucous fibrosis in southern Taiwan. Br. J. Cancer 2003, 88, 366–372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sundqvist, K.; Liu, Y.; Nair, J.; Bartsch, H.; Arvidson, K.; Grafstrom, R.C. Cytotoxic and genotoxic effects of areca nut-related compounds in cultured human buccal epithelial cells. Cancer Res. 1989, 49, 5294–5298. [Google Scholar] [PubMed]
- Jeng, J.H.; Lan, W.H.; Hahn, L.J.; Hsieh, C.C.; Kuo, M.Y. Inhibition of the migration, attachment, spreading, growth and collagen synthesis of human gingival fibroblasts by arecoline, a major areca alkaloid, in vitro. J. Oral Pathol. Med. 1996, 25, 371–375. [Google Scholar] [CrossRef] [PubMed]
- Jeng, J.H.; Kuo, M.L.; Hahn, L.J.; Kuo, M.Y. Genotoxic and non-genotoxic effects of betel quid ingredients on oral mucosal fibroblasts in vitro. J. Dent. Res. 1994, 73, 1043–1049. [Google Scholar] [CrossRef] [Green Version]
- Tseng, S.K.; Chang, M.C.; Su, C.Y.; Chi, L.Y.; Chang, J.Z.; Tseng, W.Y.; Yeung, S.Y.; Hsu, M.L.; Jeng, J.H. Arecoline induced cell cycle arrest, apoptosis, and cytotoxicity to human endothelial cells. Clin. Oral Investig. 2012, 16, 1267–1273. [Google Scholar] [CrossRef]
- Chang, M.C.; Ho, Y.S.; Lee, P.H.; Chan, C.P.; Lee, J.J.; Hahn, L.J.; Wang, Y.J.; Jeng, J.H. Areca nut extract and arecoline induced the cell cycle arrest but not apoptosis of cultured oral KB epithelial cells: Association of glutathione, reactive oxygen species and mitochondrial membrane potential. Carcinogenesis 2001, 22, 1527–1535. [Google Scholar] [CrossRef] [Green Version]
- Sjoberg, R.L.; Wu, W.Y.; Dahlin, A.M.; Tsavachidis, S.; Gliogene, G.; Bondy, M.L.; Melin, B. Role of monoamine-oxidase-A-gene variation in the development of glioblastoma in males: A case control study. J. Neurooncol. 2019, 145, 287–294. [Google Scholar] [CrossRef] [Green Version]
- Petersson, F. EBV-associated non-keratinizing nasopharyngeal carcinoma with prominent spindled cell and whorling patterns: A previously unreported histological variant in a patient presenting with dermatomyositis. Head Neck Pathol. 2020, 14, 203–207. [Google Scholar] [CrossRef]
- Yang, B.; Jiang, J.; Du, H.; Geng, G.; Jiang, Z.; Yao, C.; Zhang, Q.; Jin, L. Decreased monoamine oxidase (MAO) activity and MAO-A expression as diagnostic indicators of human esophageal cancers. Biomarkers 2009, 14, 624–629. [Google Scholar] [CrossRef] [PubMed]
- Bharti, R.; Dey, G.; Das, A.K.; Mandal, M. Differential expression of IL-6/IL-6R and MAO-A regulates invasion/angiogenesis in breast cancer. Br. J. Cancer 2018, 118, 1442–1452. [Google Scholar] [CrossRef] [PubMed]
- Ojetti, V.; Persiani, R.; Cananzi, F.C.; Sensi, C.; Piscaglia, A.C.; Saulnier, N.; Biondi, A.; Gasbarrini, A.; D’Ugo, D. cDNA-microarray analysis as a new tool to predict lymph node metastasis in gastric cancer. World J. Surg. 2014, 38, 2058–2064. [Google Scholar] [CrossRef] [PubMed]
- Alpini, G.; Invernizzi, P.; Gaudio, E.; Venter, J.; Kopriva, S.; Bernuzzi, F.; Onori, P.; Franchitto, A.; Coufal, M.; Frampton, G.; et al. Serotonin metabolism is dysregulated in cholangiocarcinoma, which has implications for tumor growth. Cancer Res. 2008, 68, 9184–9193. [Google Scholar] [CrossRef] [Green Version]
- Huang, L.; Frampton, G.; Rao, A.; Zhang, K.S.; Chen, W.; Lai, J.M.; Yin, X.Y.; Walker, K.; Culbreath, B.; Leyva-Illades, D.; et al. Monoamine oxidase A expression is suppressed in human cholangiocarcinoma via coordinated epigenetic and IL-6-driven events. Lab. Investig. 2012, 92, 1451–1460. [Google Scholar] [CrossRef]
- Grouzmann, E.; Matter, M.; Bilz, S.; Herren, A.; Triponez, F.; Henzen, C.; Kim, K.S.; Zulewski, H.; Buclin, T.; Brakch, N.; et al. Monoamine oxidase A down-regulation contributes to high metanephrine concentration in pheochromocytoma. J. Clin. Endocrinol. Metab. 2012, 97, 2773–2781. [Google Scholar] [CrossRef] [Green Version]
- Mikula, M.; Rubel, T.; Karczmarski, J.; Goryca, K.; Dadlez, M.; Ostrowski, J. Integrating proteomic and transcriptomic high-throughput surveys for search of new biomarkers of colon tumors. Funct. Integr. Genom. 2011, 11, 215–224. [Google Scholar] [CrossRef]
- Wu, J.B.; Shao, C.; Li, X.; Li, Q.; Hu, P.; Shi, C.; Li, Y.; Chen, Y.T.; Yin, F.; Liao, C.P.; et al. Monoamine oxidase A mediates prostate tumorigenesis and cancer metastasis. J. Clin. Investig. 2014, 124, 2891–2908. [Google Scholar] [CrossRef] [Green Version]
- True, L.; Coleman, I.; Hawley, S.; Huang, C.Y.; Gifford, D.; Coleman, R.; Beer, T.M.; Gelmann, E.; Datta, M.; Mostaghel, E.; et al. A molecular correlate to the Gleason grading system for prostate adenocarcinoma. Proc. Natl. Acad Sci. USA 2006, 103, 10991–10996. [Google Scholar] [CrossRef] [Green Version]
- Peehl, D.M.; Coram, M.; Khine, H.; Reese, S.; Nolley, R.; Zhao, H. The significance of monoamine oxidase-A expression in high grade prostate cancer. J. Urol. 2008, 180, 2206–2211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flamand, V.; Zhao, H.; Peehl, D.M. Targeting monoamine oxidase A in advanced prostate cancer. J. Cancer Res. Clin. Oncol. 2010, 136, 1761–1771. [Google Scholar] [CrossRef] [Green Version]
- Hodorova, I.; Rybarova, S.; Vecanova, J.; Solar, P.; Domorakova, I.; Adamkov, M.; Mihalik, J. Comparison of expression pattern of monoamine oxidase A with histopathologic subtypes and tumour grade of renal cell carcinoma. Med. Sci. Monit. 2012, 18, 482–486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, F.; Hu, L.; Ma, Y.; Huang, B.; Xiu, Z.; Zhang, P.; Zhou, K.; Tang, X. Increased expression of monoamine oxidase A is associated with epithelial to mesenchymal transition and clinicopathological features in non-small cell lung cancer. Oncol. Lett. 2018, 15, 3245–3251. [Google Scholar] [CrossRef]
- Li, P.C.; Siddiqi, I.N.; Mottok, A.; Loo, E.Y.; Wu, C.H.; Cozen, W.; Steidl, C.; Shih, J.C. Monoamine oxidase A is highly expressed in classical Hodgkin lymphoma. J. Pathol. 2017, 243, 220–229. [Google Scholar] [CrossRef]
- Bhat, A.A.; Syed, N.; Therachiyil, L.; Nisar, S.; Hashem, S.; Macha, M.A.; Yadav, S.K.; Krishnankutty, R.; Muralitharan, S.; Al-Naemi, H.; et al. Claudin-1, A Double-Edged Sword in Cancer. Int. J. Mol. Sci. 2020, 21, 569. [Google Scholar] [CrossRef] [Green Version]
- Tabata, Y.; Shidoji, Y. Hepatic monoamine oxidase B is involved in endogenous geranylgeranoic acid synthesis in mammalian liver cells. J. Lipid. Res. 2020, 61, 778–789. [Google Scholar] [CrossRef] [Green Version]
- Oh, S.Y.; Kang, S.M.; Kang, S.H.; Lee, H.J.; Kwon, T.G.; Kim, J.W.; Lee, S.T.; Choi, S.Y.; Hong, S.H. Potential salivary mRNA biomarkers for early detection of oral cancer. J. Clin. Med. 2020, 9, 243. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.C.; Han, Y.Y.; Zhang, X.M.; Xiao, N.; Jiang, T.; Zhu, S.; Wang, E.P.; Chen, C.B. miR-522 facilitates the prosperities of endometrial carcinoma cells by directly binding to monoamine oxidase B. Kaohsiung J. Med. Sci. 2019, 35, 598–606. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Chen, H.; Zhao, X. The prognostic significance of immune-related metabolic enzyme MTHFD2 in head and neck squamous cell carcinoma. Diagnostics 2020, 10, 689. [Google Scholar] [CrossRef]
- Sharpe, M.A.; Baskin, D.S. Monoamine oxidase B levels are highly expressed in human gliomas and are correlated with the expression of HiF-1alpha and with transcription factors Sp1 and Sp3. OncoTargets Ther. 2016, 7, 3379–3393. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.C.; Chien, M.H.; Lai, T.C.; Su, C.Y.; Jan, Y.H.; Hsiao, M.; Chen, C.L. Monoamine oxidase B expression correlates with a poor prognosis in colorectal cancer patients and is significantly associated with epithelial-to-mesenchymal transition-related gene signatures. Int. J. Mol. Sci. 2020, 21, 2813. [Google Scholar] [CrossRef]
- Huang, C.G.; Iv, G.D.; Liu, T.; Liu, Q.; Feng, J.G.; Lu, X.M. Polymorphisms of COMT and XPD and risk of esophageal squamous cell carcinoma in a population of Yili Prefecture, in Xinjiang, China. Biomarkers 2011, 16, 37–41. [Google Scholar] [CrossRef]
- Pan, J.L.; Gao, J.; Hou, J.H.; Hu, D.Z.; Li, L. Interaction between environmental risk factors and catechol-O-methyltransferase (COMT) and X-ray repair cross-complementing protein 1 (XRCC1) gene polymorphisms in risk of lung cancer among non-smoking chinese women: A case-control study. Med. Sci. Monit. 2018, 24, 5689–5697. [Google Scholar] [CrossRef]
- Zhou, Q.; Wang, Y.; Chen, A.; Tao, Y.; Song, H.; Li, W.; Tao, J.; Zuo, M. Association between the COMT Val158Met polymorphism and risk of cancer: Evidence from 99 case-control studies. OncoTargets Ther. 2015, 8, 2791–2803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thompson, P.A.; Shields, P.G.; Freudenheim, J.L.; Stone, A.; Vena, J.E.; Marshall, J.R.; Graham, S.; Laughlin, R.; Nemoto, T.; Kadlubar, F.F.; et al. Genetic polymorphisms in catechol-O-methyltransferase, menopausal status, and breast cancer risk. Cancer Res. 1998, 58, 2107–2110. [Google Scholar]
- Huang, C.S.; Chern, H.D.; Chang, K.J.; Cheng, C.W.; Hsu, S.M.; Shen, C.Y. Breast cancer risk associated with genotype polymorphism of the estrogen-metabolizing genes CYP17, CYP1A1, and COMT: A multigenic study on cancer susceptibility. Cancer Res. 1999, 59, 4870–4875. [Google Scholar] [PubMed]
- Lajin, B.; Hamzeh, A.R.; Ghabreau, L.; Mohamed, A.; Al Moustafa, A.E.; Alachkar, A. Catechol-O-methyltransferase Val 108/158 Met polymorphism and breast cancer risk: A case control study in Syria. Breast Cancer-Tokyo 2013, 20, 62–66. [Google Scholar] [CrossRef]
- Nock, N.L.; Cicek, M.S.; Li, L.; Liu, X.; Rybicki, B.A.; Moreira, A.; Plummer, S.J.; Casey, G.; Witte, J.S. Polymorphisms in estrogen bioactivation, detoxification and oxidative DNA base excision repair genes and prostate cancer risk. Carcinogenesis 2006, 27, 1842–1848. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolpert, B.J.; Amr, S.; Saleh, D.A.; Ezzat, S.; Gouda, I.; Loay, I.; Hifnawy, T.; Abdel-Hamid, M.; Mikhail, N.N.; Zhan, M.; et al. Associations differ by sex for catechol-O-methyltransferase genotypes and bladder cancer risk in South Egypt. Urol. Oncol. Semin. Orig. Investig. 2012, 30, 841–847. [Google Scholar] [CrossRef] [Green Version]
- Yager, J.D. Catechol-O-methyltransferase: Characteristics, polymorphisms and role in breast cancer. Drug Discov. Today Dis. Mech. 2012, 9, e41–e46. [Google Scholar] [CrossRef] [Green Version]
- Sasaki, M.; Kaneuchi, M.; Sakuragi, N.; Dahiya, R. Multiple promoters of catechol-O-methyltransferase gene are selectively inactivated by CpG hypermethylation in endometrial cancer. Cancer Res. 2003, 63, 3101–3106. [Google Scholar] [PubMed]
- Tolba, M.F.; Omar, H.A.; Hersi, F.; Nunes, A.C.F.; Noreddin, A.M. The impact of catechol-O-methyl transferase knockdown on the cell proliferation of hormone-responsive cancers. Mol. Cell. Endocrinol. 2019, 488, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Wu, Q.; Hong, X.; Zhou, L.; Zhang, J.; You, L.; Wang, W.; Wu, H.; Dai, H.; Zhao, Y. Catechol-O-methyltransferase, a new target for pancreatic cancer therapy. Cancer Sci. 2015, 106, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Wu, Q.; Hong, X.; Xiong, G.; Xiao, Y.; Zhou, J.; Wang, W.; Wu, H.; Zhou, L.; Song, W.; et al. Catechol-O-methyltransferase inhibits colorectal cancer cell proliferation and invasion. Arch. Med. Res. 2015, 46, 17–23. [Google Scholar] [CrossRef]
- Zhao, Y.-N.; Zhang, W.; Chen, Y.-C.; Fang, F.; Liu, X.-Q. Relative imbalances in the expression of catechol-O-methyltransferase and cytochrome P450 in breast cancer tissue and their association with breast carcinoma. Maturitas 2012, 72, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Zhang, J.; Zhou, L.; You, L.; Zhao, Y.; Li, J. Increased COMT expression in pancreatic cancer and correlation with clinicopathologic parameters. Sci. China Life Sci. 2012, 55, 747–752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.C.; Cheng, A.J.; Lee, L.Y.; Huang, Y.C.; Chang, J.T. Multifaceted mechanisms of areca nuts in oral carcinogenesis: The molecular pathology from precancerous condition to malignant transformation. J. Cancer 2019, 10, 4054–4062. [Google Scholar] [CrossRef] [Green Version]
- Ji, W.T.; Yang, S.R.; Chen, J.Y.; Cheng, Y.P.; Lee, Y.R.; Chiang, M.K.; Chen, H.R. Arecoline downregulates levels of p21 and p27 through the reactive oxygen species/mTOR complex 1 pathway and may contribute to oral squamous cell carcinoma. Cancer Sci. 2012, 103, 1221–1229. [Google Scholar] [CrossRef]
- Ji, W.T.; Lee, C.I.; Chen, J.Y.; Cheng, Y.P.; Yang, S.R.; Chen, J.H.; Chen, H.R. Areca nut extract induces pyknotic necrosis in serum-starved oral cells via increasing reactive oxygen species and inhibiting GSK3beta: An implication for cytopathic effects in betel quid chewers. PLoS ONE 2013, 8, e63295. [Google Scholar] [CrossRef] [PubMed]
- Katakwar, P.; Metgud, R.; Naik, S.; Mittal, R. Oxidative stress marker in oral cancer: A review. J. Canc. Res. 2016, 12, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Dawling, S.; Roodi, N.; Mernaugh, R.L.; Wang, X.; Parl, F.F. Catechol-O-methyltransferase (COMT)-mediated metabolism of catechol estrogens: Comparison of wild-type and variant COMT isoforms. Cancer Res. 2001, 61, 6716–6722. [Google Scholar] [PubMed]
- Mishra, R. Biomarkers of oral premalignant epithelial lesions for clinical application. Oral Oncol. 2012, 48, 578–584. [Google Scholar] [CrossRef]
- Wang, C.P.; Chen, T.C.; Chen, H.H.; Hsu, W.L.; Chang, Y.L. Prevalence of current oral HPV infection among 100 betel nut chewers or cigarette smokers in Northern Taiwan. J. Med. Assoc. 2019, 118, 203–208. [Google Scholar] [CrossRef]
- Anand, R.; Dhingra, C.; Prasad, S.; Menon, I. Betel nut chewing and its deleterious effects on oral cavity. J. Cancer Res. 2014, 10, 499–505. [Google Scholar]






| Oral and Pharyngeal Cancers (n = 297) | OPMD (n = 40) | Control (n = 193) | p-Value | ||||
|---|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | |||||
| Demography and Substance use Factors | n | (%) a | n | (%) | n | (%) | |
| Age, years (mean ± SD) | 53.97 ± 10.32 † | 50.83 ± 11.57 | 46.08 ± 12.94 | <0.001 *b | |||
| Age group ≤ 50 (years) | 117 | (39.4) | 22 | (55.0) | 127 | (65.8) | <0.001 *c |
| Age group > 50 (years) | 180 | (60.6) | 18 | (45.0) | 66 | (34.2) | |
| BMI (kg/m2) | 24.58 ± 4.07 | 26.52 ± 4.14 | 26.27 ± 27.74 | 0.668 b | |||
| Ethnicity | |||||||
| Minnan | 220 | (74.1) | 25 | (62.5) | 158 | (81.9) | 0.016 *c |
| Non-Minnan | 77 | (25.9) | 15 | (37.5) | 35 | (18.1) | |
| Marital status | |||||||
| Unmarried | 41 | (13.8) | 7 | (17.5) | 13 | (6.7) | 0.027 *c |
| Married | 256 | (86.2) | 33 | (82.5) | 180 | (93.3) | |
| Education level | |||||||
| Low (≤9 years) | 197 | (66.3) | 25 | (62.5) | 84 | (43.5) | <0.001 *c |
| High (>9 years) | 100 | (33.7) | 15 | (37.5) | 109 | (56.5) | |
| Alcohol drinking status | |||||||
| Never | 71 | (23.9) | 13 | (32.5) | 96 | (49.7) | <0.001 *c |
| Current or former | 226 | (76.1) | 27 | (67.5) | 97 | (50.3) | |
| Age at starting drinking (years) | 22.61 ± 6.61 | 20.25 ± 3.95 | 20.68 ± 7.72 | 0.047 *b | |||
| Years of drinking | 27.91 ± 10.81† | 27.22 ± 12.91 | 22.12 ± 11.35 | <0.001 b | |||
| Former | |||||||
| Age at quitting drinking (years) | 48.58 ± 11.55 † | 39.00 ± 12.61 | 38.47 ± 11.73 | <0.001 b | |||
| Years of quitting alcohol | 8.86 ± 7.90 | 12.57 ± 14.49 | 7.44 ± 6.97 | 0.249 b | |||
| BQ chewing status | |||||||
| Never | 40 | (13.5) | 7 | (17.5) | 130 | (67.4) | <0.001 *c |
| Current or former | 257 | (86.5) | 33 | (82.5) | 63 | (32.6) | |
| Age at starting chewing (years) | 22.44 ± 6.39† | 22.66 ± 8.26‡ | 19.00 ± 4.86 | <0.001 *b | |||
| Years of chewing | 24.61 ± 10.48† | 21.72 ± 9.64 | 19.97 ± 10.74 | 0.007 *b | |||
| Average amount of chewing (quid) | 28.96 ± 27.12 | 40.43 ± 36.91‡ | 20.18 ± 14.31 | 0.006 *b | |||
| Cumulative lifetime BQ use (pack-years) | 76.10 ± 89.07† | 86.24 ± 83.87 | 41.56 ± 37.10 | 0.016 *b | |||
| Type of BQ material | |||||||
| BQ with inflorescence of Piper betel Linn. | 27 | (10.5) | 3 | (9.1) | 13 | (20.6) | 0.093c |
| BQ with Piper betel leaf | 186 | (72.4) | 23 | (69.7) | 35 | (55.6) | |
| Mixed | 44 | (17.1) | 7 | (21.2) | 15 | (23.8) | |
| BQ juice swallowing | |||||||
| Swallowed | 109 | (42.4) | 15 | (45.5) | 9 | (14.3) | <0.001 *c |
| Never swallowed | 148 | (57.6) | 18 | (54.6) | 54 | (85.7) | |
| Former | |||||||
| Age at quitting chewing (years) | 45.45 ± 11.49 † | 42.00 ± 8.82 | 36.84 ± 9.76 | <0.001 *b | |||
| Years of quitting BQ | 10.15 ± 7.55 | 11.33 ± 9.31 | 9.60 ± 8.76 | 0.792 b | |||
| Cigarette smoking status | |||||||
| Never | 36 | (12.1) | 5 | (12.5) | 43 | (22.3) | 0.009 *c |
| Current or former | 261 | (87.9) | 35 | (87.5) | 150 | (77.7) | |
| Age at starting smoking (years) | 19.53 ± 4.02 † | 18.31 ± 3.81 | 18.29 ± 5.15 | 0.026 *b | |||
| Years of smoking | 29.81 ± 11.42 † | 28.16 ± 11.56 | 24.21 ± 10.57 | <0.001 *b | |||
| Average amount of smoking | 23.88 ± 13.73 † | 29.41 ± 16.74 ‡ | 15.51 ± 10.67 | <0.001 *b | |||
| Cumulative lifetime cigarette use (pack-years) | 37.04 ± 25.83 | 41.52 ± 27.06 | 27.33 ± 19.49 | 0.066 b | |||
| Former | |||||||
| Age at quitting smoking (years) | 46.96 ± 11.87 † | 43.33 ± 13.50 | 38.74 ± 14.34 | 0.009 *b | |||
| Years of quitting cigarette | 10.42 ± 9.69 | 17.83 ± 13.42 | 14.78 ± 13.03 | 0.076 b | |||
| Oral and Pharyngeal Cancers (n = 297) | OPMD (n = 40) | Control (n = 193) | Oral and Pharyngeal Cancers vs. Control | OPMD vs. Control | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Assessment | Risk Assessment | ||||||||||||
| SNPs | BQ | n | (%) a | n | (%) | n | (%) | AOR | (95% CI) b | p | AOR | (95% CI) b | p |
| MAOA | |||||||||||||
| rs6323 | |||||||||||||
| T | 98 | (33.0) | 10 | (25.0) | 89 | (46.1) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| G | 199 | (67.0) | 30 | (75.0) | 104 | (53.9) | 1.98 | (1.23–3.20) * | 0.005 | 2.89 | (1.27–6.58) * | 0.012 | |
| T | (−) | 14 | (4.7) | 5 | (12.5) | 57 | (29.5) | 1.00 | 1.00 | (Reference) | |||
| G | (−) | 26 | (8.8) | 2 | (5.0) | 73 | (37.8) | 1.72 | (0.77–3.87) | 0.187 | 0.37 | (0.07–2.01) | 0.247 |
| T | (+) | 84 | (28.3) | 5 | (12.5) | 32 | (16.6) | 12.67 | (5.44–29.53) * | <0.001 | 1.95 | (0.45–8.54) | 0.374 |
| G | (+) | 173 | (58.3) | 28 | (70.0) | 31 | (16.1) | 31.15 | (13.43–72.27) * | <0.001 | 12.77 | (3.61–45.20) * | <0.001 |
| rs1137070 | |||||||||||||
| C | 97 | (32.7) | 10 | (25.0) | 103 | (53.4) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| T | 200 | (67.3) | 30 | (75.0) | 90 | (46.6) | 2.37 | (1.47–3.81) * | <0.001 | 3.25 | (1.43–7.38) * | 0.005 | |
| C | (−) | 14 | (4.7) | 5 | (12.5) | 70 | (36.3) | 1.00 | (Reference) | 1.00 | (Reference) | ||
| T | (−) | 26 | (8.8) | 2 | (5.0) | 60 | (31.1) | 2.30 | (1.03–5.13) * | 0.043 | 0.52 | (0.10–2.86) | 0.455 |
| C | (+) | 83 | (28.0) | 5 | (12.5) | 33 | (17.1) | 13.21 | (5.90–29.57) * | <0.001 | 2.32 | (0.56–9.67) | 0.249 |
| T | (+) | 174 | (58.6) | 28 | (70.0) | 30 | (15.5) | 36.22 | (16.24–80.78) * | <0.001 | 15.47 | (4.67–51.26) * | <0.001 |
| rs5906957 | |||||||||||||
| G | 104 | (35.0) | 10 | (25.0) | 79 | (40.9) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| A | 193 | (65.0) | 30 | (75.0) | 114 | (59.1) | 1.67 | (1.03–2.71) * | 0.036 | 2.55 | (1.11–5.85) * | 0.027 | |
| G | (−) | 15 | (5.1) | 5 | (12.5) | 48 | (24.9) | 1.00 | (Reference) | 1.00 | (Reference) | ||
| A | (−) | 25 | (8.4) | 2 | (5.0) | 82 | (42.5) | 1.24 | (0.55–2.80) | 0.598 | 0.27 | (0.05–1.50) | 0.136 |
| G | (+) | 89 | (30.0) | 5 | (12.5) | 31 | (16.1) | 10.70 | (4.57–25.03) * | <0.001 | 1.58 | (0.36–7.01) | 0.547 |
| A | (+) | 168 | (56.6) | 28 | (70.0) | 32 | (16.6) | 24.15 | (10.44–55.89) * | <0.001 | 10.04 | (2.83–35.56) * | <0.001 |
| MAOB | |||||||||||||
| rs6324 | |||||||||||||
| A | 74 | (24.9) | 4 | (10.0) | 157 | (81.4) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| G | 223 | (75.1) | 36 | (90.0) | 36 | (18.7) | 13.00 | (7.35–22.98) * | <0.001 | 37.45 | (11.94–117.46) * | <0.001 | |
| A | (−) | 5 | (1.7) | 1 | (2.5) | 108 | (56.0) | 1.00 | (Reference) | 1.00 | (Reference) | ||
| G | (−) | 35 | (11.8) | 6 | (15.0) | 22 | (11.4) | 30.24 | (10.19–89.73) * | <0.001 | 29.73 | (3.33–265.50) * | 0.002 |
| A | (+) | 69 | (23.2) | 3 | (7.5) | 49 | (25.4) | 35.34 | (12.14–102.86) * | <0.001 | 8.12 | (0.76–86.96) | 0.084 |
| G | (+) | 188 | (63.3) | 30 | (75.0) | 14 | (7.3) | 307.03 | (97.43–967.54) * | <0.001 | 246.94 | (28.08–NA) * | <0.001 |
| rs1799836 | |||||||||||||
| C | 50 | (16.8) | 5 | (12.5) | 34 | (17.6) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| T | 247 | (83.2) | 35 | (87.5) | 159 | (82.4) | 1.36 | (0.75–2.47) | 0.309 | 1.97 | (0.68–5.73) | 0.213 | |
| C | (−) | 5 | (1.7) | 2 | (5.0) | 19 | (9.8) | 1.00 | (Reference) | 1.00 | |||
| T | (−) | 35 | (11.8) | 5 | (12.5) | 111 | (57.5) | 1.46 | (0.47–4.59) | 0.513 | 0.44 | (0.08–2.53) | 0.354 |
| C | (+) | 45 | (15.2) | 3 | (7.5) | 15 | (7.8) | 15.67 | (4.40–55.86) * | <0.001 | 1.93 | (0.25–14.87) | 0.530 |
| T | (+) | 212 | (71.4) | 30 | (75.0) | 48 | (24.9) | 22.60 | (7.13–71.62) * | <0.001 | 6.32 | (1.21–33.08) * | 0.029 |
| rs3027452 | |||||||||||||
| A | 41 | (13.8) | 5 | (12.5) | 27 | (14.0) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| G | 256 | (86.2) | 35 | (87.5) | 166 | (86.0) | 1.38 | (0.73–2.62) | 0.329 | 1.57 | (0.53–4.64) | 0.417 | |
| A | (−) | 4 | (1.4) | 2 | (5.0) | 13 | (6.7) | 1.00 | (Reference) | 1.00 | (Reference) | ||
| G | (−) | 36 | (12.1) | 5 | (12.5) | 117 | (60.6) | 1.14 | (0.32–4.13) | 0.841 | 0.26 | (0.04–1.59) | 0.145 |
| A | (+) | 37 | (12.5) | 3 | (7.5) | 14 | (7.3) | 11.51 | (2.78–47.73) * | <0.001 | 1.38 | (0.17–11.12) | 0.765 |
| G | (+) | 220 | (74.1) | 30 | (75.0) | 49 | (25.4) | 18.57 | (5.06–68.18) * | <0.001 | 4.00 | (0.73–22.03) | 0.112 |
| COMT | |||||||||||||
| rs4633 | |||||||||||||
| T/T | 15 | (5.1) | 4 | (10.0) | 17 | (8.8) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| C/T | 91 | (30.6) | 7 | (17.5) | 81 | (42.0) | 1.05 | (0.40–2.76) | 0.920 | 0.30 | (0.07–1.26) | 0.100 | |
| C/C | 191 | (64.3) | 29 | (72.5) | 95 | (49.2) | 1.86 | (0.74–4.71) | 0.189 | 1.06 | (0.29–3.81) | 0.931 | |
| T/T+C/T | 106 | (35.7) | 11 | (27.5) | 98 | (50.8) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| C/C | 191 | (64.3) | 29 | (72.5) | 95 | (49.2) | 1.78 | (1.11–2.85) * | 0.016 | 2.65 | (1.19–5.88) * | 0.017 | |
| T/T+C/T | (−) | 13 | (4.4) | 2 | (5.0) | 68 | (35.2) | 1.00 | (Reference) | 1.00 | (Reference) | ||
| C/C | (−) | 27 | (9.1) | 5 | (12.5) | 62 | (32.1) | 1.82 | (0.81–4.07) | 0.145 | 2.55 | (0.47–13.92) | 0.280 |
| T/T+C/T | (+) | 93 | (31.3) | 9 | (22.5) | 30 | (15.5) | 15.38 | (6.81–34.72) * | <0.001 | 10.12 | (1.84–55.53) * | 0.008 |
| C/C | (+) | 164 | (55.2) | 24 | (60.0) | 33 | (17.1) | 27.14 | (12.16–60.56) * | <0.001 | 26.86 | (5.24–137.65) * | <0.001 |
| rs9605030 | |||||||||||||
| C/C | 177 | (59.6) | 16 | (40.0) | 115 | (59.6) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| C/T | 103 | (34.7) | 20 | (50.0) | 69 | (35.8) | 1.06 | (0.65–1.73) | 0.829 | 2.38 | (1.09–5.21) * | 0.030 | |
| T/T | 17 | (5.7) | 4 | (10.0) | 9 | (4.7) | 1.07 | (0.38–3.00) | 0.904 | 3.29 | (0.81–13.36) | 0.096 | |
| C/C+C/T | 280 | (94.3) | 36 | (90.0) | 184 | (95.3) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| T/T | 17 | (5.7) | 4 | (10.0) | 9 | (4.7) | 1.04 | (0.38–2.90) | 0.935 | 2.23 | (0.58–8.51) | 0.242 | |
| C/C+C/T | (−) | 40 | (13.5) | 5 | (12.5) | 123 | (63.7) | 1.00 | (Reference) | 1.00 | (Reference) | ||
| T/T | (−) | 0 | (0.0) | 2 | (5.0) | 7 | (3.6) | NA | 0.983 | 4.18 | (0.61–28.75) | 0.147 | |
| C/C+C/T | (+) | 240 | (80.8) | 31 | (77.5) | 61 | (31.6) | 13.77 | (7.72–24.54) * | <0.001 | 12.58 | (3.98–39.83) * | <0.001 |
| T/T | (+) | 17 | (5.7) | 2 | (5.0) | 2 | (1.0) | 31.57 | (6.10–163.34) * | <0.001 | 32.29 | (3.28–317.97) * | 0.003 |
| rs9606186 | |||||||||||||
| C/C | 22 | (7.4) | 3 | (7.5) | 23 | (11.9) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| G/C | 125 | (42.1) | 12 | (30.0) | 84 | (43.5) | 2.28 | (1.03–5.02) * | 0.042 | 1.60 | (0.38–6.65) | 0.521 | |
| G/G | 150 | (50.5) | 25 | (62.5) | 86 | (44.6) | 2.72 | (1.25–5.94) * | 0.012 | 3.29 | (0.84–12.88) | 0.087 | |
| C/C+G/C | 147 | (49.5) | 15 | (37.5) | 107 | (55.4) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| G/G | 150 | (50.5) | 25 | (62.5) | 86 | (44.6) | 1.41 | (0.89–2.24) | 0.144 | 2.29 | (1.08–4.84) * | 0.031 | |
| C/C+G/C | (−) | 19 | (6.4) | 3 | (7.5) | 70 | (36.3) | 1.00 | (Reference) | 1.00 | (Reference) | ||
| G/G | (−) | 21 | (7.1) | 4 | (10.0) | 60 | (31.1) | 1.44 | (0.66–3.13) | 0.364 | 1.53 | (0.32–7.27) | 0.594 |
| C/C+G/C | (+) | 128 | (43.1) | 12 | (30.0) | 37 | (19.2) | 15.38 | (7.44–31.82) * | <0.001 | 8.13 | (1.93–34.27) * | 0.004 |
| G/G | (+) | 129 | (43.4) | 21 | (52.5) | 26 | (13.5) | 22.13 | (10.23–47.84) * | <0.001 | 20.73 | (4.97–86.52) * | <0.001 |
| Oral and Pharyngeal Cancers (n = 297) | OPMD (n = 40) | Control (n = 193) | Oral and Pharyngeal Cancers vs. Control | OPMD vs. Control | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Assessment | Risk Assessment | |||||||||||||
| SNPs | n | (%) a | n | (%) | n | (%) | AOR | (95% CI) b | p | AOR | (95% CI) b | p | ||
| MAOA | MAOB | |||||||||||||
| rs6323 | rs6324 | |||||||||||||
| T | A | 19 | (6.4) | 2 | (5.0) | 74 | (38.3) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| G | A | 55 | (18.5) | 2 | (5.0) | 83 | (43.0) | 3.16 | (1.49–6.71) * | 0.003 | 1.05 | (0.14–7.98) | 0.962 | |
| T | G | 79 | (26.6) | 8 | (20.0) | 15 | (7.8) | 22.53 | (8.96–56.70) * | <0.001 | 20.18 | (3.63–112.27) * | 0.001 | |
| G | G | 144 | (48.5) | 28 | (70.0) | 21 | (10.9) | 30.32 | (13.07–70.35) * | <0.001 | 53.21 | (10.91–259.62) * | <0.001 | |
| MAOA | MAOB | |||||||||||||
| rs1137070 | rs6324 | |||||||||||||
| C | A | 19 | (6.4) | 2 | (5.0) | 81 | (42.0) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| T | A | 55 | (18.5) | 2 | (5.0) | 76 | (39.4) | 3.28 | (1.53–7.01) * | 0.002 | 0.90 | (0.12–6.84) | 0.919 | |
| C | G | 78 | (26.3) | 8 | (20.0) | 22 | (11.4) | 15.53 | (6.55–36.83) * | <0.001 | 12.37 | (2.31–66.20) * | 0.003 | |
| T | G | 145 | (48.8) | 28 | (70.0) | 14 | (7.3) | 45.56 | (18.51–112.17) * | <0.001 | 70.59 | (14.17–351.78) * | <0.001 | |
| MAOA | COMT | |||||||||||||
| rs6323 | rs4633 | |||||||||||||
| T | T/T+C/T | 44 | (14.8) | 5 | (12.5) | 42 | (21.8) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| T | C/C | 54 | (18.2) | 5 | (12.5) | 47 | (24.4) | 1.18 | (0.57–2.46) | 0.659 | 1.09 | (0.27–4.31) | 0.906 | |
| G | T/T+C/T | 62 | (20.9) | 6 | (15.0) | 56 | (29.0) | 1.34 | (0.65–2.77) | 0.422 | 1.25 | (0.33–4.69) | 0.745 | |
| G | C/C | 137 | (46.1) | 24 | (60.0) | 48 | (24.9) | 2.94 | (1.50–5.76) * | 0.002 | 4.74 | (1.54–14.57) * | 0.007 | |
| MAOA | COMT | |||||||||||||
| rs1137070 | rs4633 | |||||||||||||
| C | T/T+C/T | 44 | (14.8) | 5 | (12.5) | 54 | (28.0) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| C | C/C | 53 | (17.9) | 5 | (12.5) | 49 | (25.4) | 1.47 | (0.72–2.99) | 0.293 | 1.26 | (0.32–4.90) | 0.744 | |
| T | T/T+C/T | 62 | (20.9) | 6 | (15.0) | 44 | (22.8) | 2.02 | (0.99–4.14) | 0.053 | 1.63 | (0.44–6.10) | 0.465 | |
| T | C/C | 138 | (46.5) | 24 | (60.0) | 46 | (23.8) | 3.57 | (1.86–6.82) * | <0.001 | 5.31 | (1.76–16.00) * | 0.003 | |
| MAOB | COMT | |||||||||||||
| rs6324 | rs4633 | |||||||||||||
| A | T/T+C/T | 18 | (6.1) | 0 | (0.0) | 77 | (39.9) | 1.00 | (Reference) | 1.00 | (Reference) | |||
| A | C/C | 56 | (18.9) | 4 | (10.0) | 80 | (41.5) | 2.63 | (1.22–5.67) * | 0.013 | NA | |||
| G | T/T+C/T | 88 | (29.6) | 11 | (27.5) | 21 | (10.9) | 15.19 | (6.30–36.60) * | <0.001 | NA | |||
| G | C/C | 135 | (45.5) | 25 | (62.5) | 15 | (7.8) | 39.27 | (15.66–98.47) * | <0.001 | NA | |||
| MAOA | MAOB | COMT | ||||||||||||
| rs6323 | rs6324 | rs4633 | ||||||||||||
| T | A | T/T+C/T | 8 | (2.7) | 0 | (0.0) | 34 | (17.6) | 1.00 | (Reference) | 1.00 | (Reference) | ||
| T | A | C/C | 11 | (3.7) | 2 | (5.0) | 40 | (20.7) | 1.23 | (0.35–4.27) | 0.746 | NA | ||
| G | A | T/T+C/T | 10 | (3.4) | 0 | (0.0) | 43 | (22.3) | 1.43 | (0.39–5.18) | 0.591 | NA | ||
| G | A | C/C | 45 | (15.2) | 2 | (5.0) | 40 | (20.7) | 5.24 | (1.72–15.98) * | 0.004 | NA | ||
| T | G | T/T+C/T | 36 | (12.1) | 5 | (12.5) | 8 | (4.2) | 17.76 | (4.69–67.25) * | <0.001 | NA | ||
| G | G | T/T+C/T | 52 | (17.5) | 6 | (15.0) | 13 | (6.7) | 19.31 | (5.53–67.46) * | <0.001 | NA | ||
| T | G | C/C | 43 | (14.5) | 3 | (7.5) | 7 | (3.6) | 36.43 | (9.08–146.12) * | <0.001 | NA | ||
| G | G | C/C | 92 | (31.0) | 22 | (55.0) | 8 | (4.2) | 56.99 | (15.82–205.31) * | <0.001 | NA | ||
| MAOA | MAOB | COMT | ||||||||||||
| rs1137070 | rs6324 | rs4633 | ||||||||||||
| C | A | T/T+C/T | 8 | (2.7) | 0 | (0.0) | 41 | (21.2) | 1.00 | (Reference) | 1.00 | (Reference) | ||
| C | A | C/C | 11 | (3.7) | 2 | (5.0) | 40 | (20.7) | 1.44 | (0.41–5.03) | 0.570 | NA | ||
| T | A | T/T+C/T | 10 | (3.4) | 0 | (0.0) | 36 | (18.7) | 1.74 | (0.48–6.33) | 0.404 | NA | ||
| T | A | C/C | 45 | (15.2) | 2 | (5.0) | 40 | (20.7) | 5.68 | (1.85–17.42) * | 0.002 | NA | ||
| C | G | T/T+C/T | 36 | (12.1) | 5 | (12.5) | 13 | (6.7) | 11.21 | (3.23–38.89) * | 0.001 | NA | ||
| C | G | C/C | 42 | (14.1) | 3 | (7.5) | 9 | (4.7) | 35.29 | (9.09–136.95) * | <0.001 | NA | ||
| T | G | T/T+C/T | 52 | (17.5) | 6 | (15.0) | 8 | (4.2) | 40.69 | (10.47–158.05) * | <0.001 | NA | ||
| T | G | C/C | 93 | (31.3) | 22 | (55.0) | 6 | (3.1) | 78.62 | (20.37–303.45) * | <0.001 | NA | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, P.-H.; Wang, Y.-Y.; Lan, T.-H.; Chan, L.-P.; Yuan, S.-S. Genetic and Proteinic Linkage of MAO and COMT with Oral Potentially Malignant Disorders and Cancers of the Oral Cavity and Pharynx. Cancers 2021, 13, 3268. https://doi.org/10.3390/cancers13133268
Chen P-H, Wang Y-Y, Lan T-H, Chan L-P, Yuan S-S. Genetic and Proteinic Linkage of MAO and COMT with Oral Potentially Malignant Disorders and Cancers of the Oral Cavity and Pharynx. Cancers. 2021; 13(13):3268. https://doi.org/10.3390/cancers13133268
Chicago/Turabian StyleChen, Ping-Ho, Yen-Yun Wang, Ting-Hsun Lan, Leong-Perng Chan, and Shyng-Shiou Yuan. 2021. "Genetic and Proteinic Linkage of MAO and COMT with Oral Potentially Malignant Disorders and Cancers of the Oral Cavity and Pharynx" Cancers 13, no. 13: 3268. https://doi.org/10.3390/cancers13133268
APA StyleChen, P.-H., Wang, Y.-Y., Lan, T.-H., Chan, L.-P., & Yuan, S.-S. (2021). Genetic and Proteinic Linkage of MAO and COMT with Oral Potentially Malignant Disorders and Cancers of the Oral Cavity and Pharynx. Cancers, 13(13), 3268. https://doi.org/10.3390/cancers13133268







