Is It Time to Call Time on Bone Marrow Biopsy for Staging Ewing Sarcoma (ES)?
Abstract
:Simple Summary
Abstract
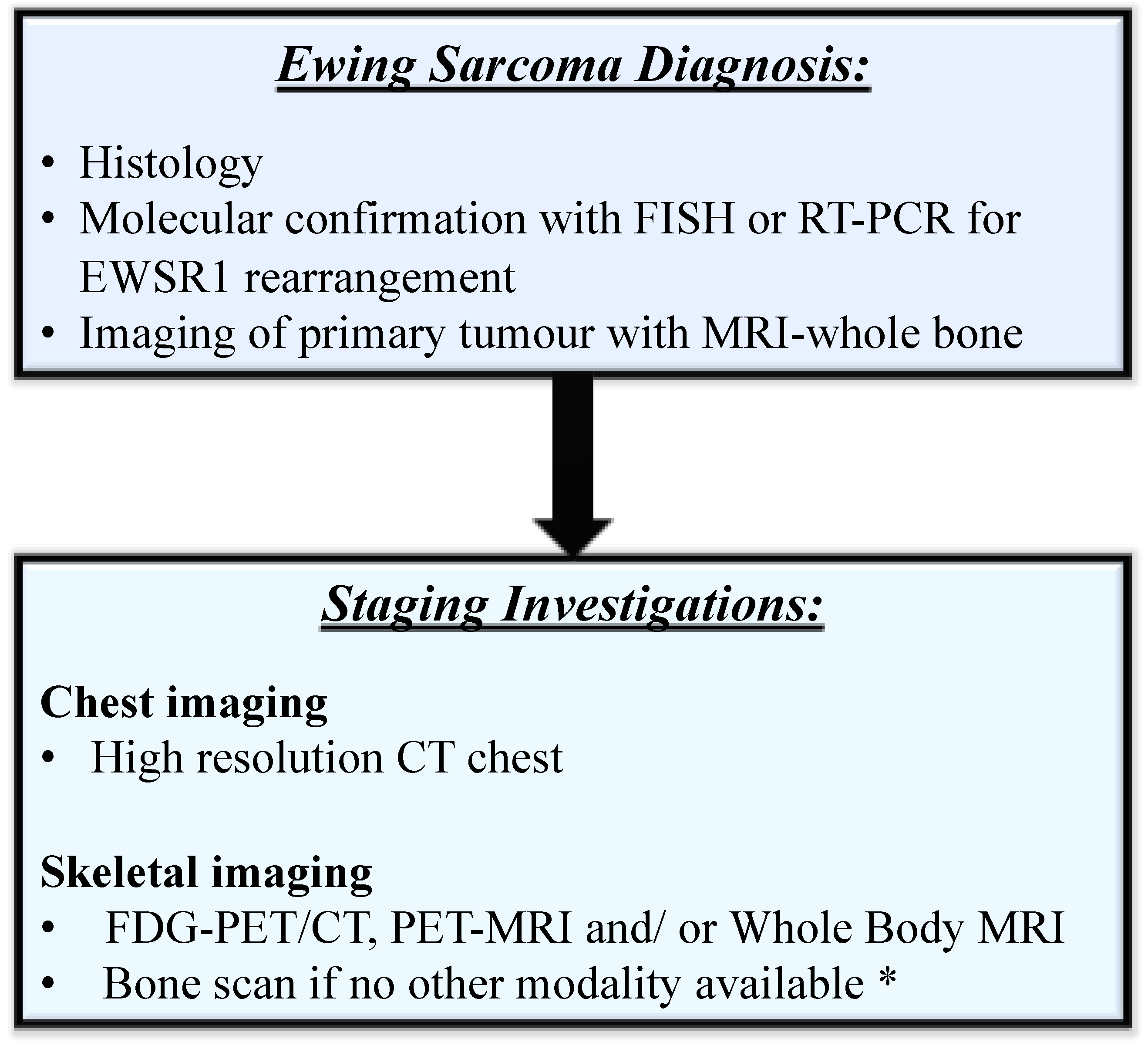
1. Introduction
2. Prognosis and Management of ES
3. FDG-PET/CT and WB-MRI Staging in ES
4. FDG-PET/CT Detection of BM Metastases
5. Current International Guidelines
6. Recommendations
7. Limitations
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ginsberg, J.W.; Johnson, S.Y.; Horowitz, M.E. Ewing’s sarcoma family of tumors: Ewing’s sarcoma of bone and soft tissue and the peripheral primitive neuroectodermal tumors. In Principles and Practice of Pediatric Oncology, 4th ed.; Pizzo, P.P., Ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2002; pp. 973–1016. [Google Scholar]
- Bain, B.J. Bone marrow biopsy morbidity and mortality. Br. J. Haematol. 2003, 121, 949–951. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaspar, N.; Hawkins, D.S.; Dirksen, U.; Lewis, I.J.; Ferrari, S.; Le Deley, M.C.; Kovar, H.; Grimer, R.; Whelan, J.; Claude, L.; et al. Ewing Sarcoma: Current Management and Future Approaches Through Collaboration. J. Clin. Oncol. 2015, 33, 3036–3046. [Google Scholar] [CrossRef]
- Oberlin, O.; Rey, A.; Desfachelles, A.S.; Philip, T.; Plantaz, D.; Schmitt, C.; Plouvier, E.; Lejars, O.; Rubie, H.; Terrier, P.; et al. Impact of high-dose busulfan plus melphalan as consolidation in metastatic Ewing tumors: A study by the Societe Francaise des Cancers de l’Enfant. J. Clin. Oncol. 2006, 24, 3997–4002. [Google Scholar] [CrossRef]
- Ladenstein, R.; Potschger, U.; Le Deley, M.C.; Whelan, J.; Paulussen, M.; Oberlin, O.; van den Berg, H.; Dirksen, U.; Hjorth, L.; Michon, J.; et al. Primary disseminated multifocal Ewing sarcoma: Results of the Euro-EWING 99 trial. J. Clin. Oncol. 2010, 28, 3284–3291. [Google Scholar] [CrossRef] [PubMed]
- Womer, R.B.; West, D.C.; Krailo, M.D.; Dickman, P.S.; Pawel, B.R.; Grier, H.E.; Marcus, K.; Sailer, S.; Healey, J.H.; Dormans, J.P.; et al. Randomized controlled trial of interval-compressed chemotherapy for the treatment of localized Ewing sarcoma: A report from the Children’s Oncology Group. J. Clin. Oncol. 2012, 30, 4148–4154. [Google Scholar] [CrossRef] [Green Version]
- Brennan, B.K.L.; Marec-Berard, P.; Martin-Broto, J.; Gelderblom, H.; Gaspar, N.; Strauss, S.; Urgelles, A.S.; Anderton, J.; Laurence, V.; Whelan, J.; et al. Comparison of two chemotherapy regimens in Ewing sarcoma (ES): Overall and subgroup results of the Euro Ewing 2012 randomized trial (EE2012). J. Clin. Oncol. 2020, 38, 11500. [Google Scholar] [CrossRef]
- Whelan, J.; Le Deley, M.C.; Dirksen, U.; Le Teuff, G.; Brennan, B.; Gaspar, N.; Hawkins, D.S.; Amler, S.; Bauer, S.; Bielack, S.; et al. High-Dose Chemotherapy and Blood Autologous Stem-Cell Rescue Compared with Standard Chemotherapy in Localized High-Risk Ewing Sarcoma: Results of Euro-E.W.I.N.G.99 and Ewing-2008. J. Clin. Oncol. 2018, JCO2018782516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dirksen, U.; Brennan, B.; Le Deley, M.C.; Cozic, N.; van den Berg, H.; Bhadri, V.; Brichard, B.; Claude, L.; Craft, A.; Amler, S.; et al. High-Dose Chemotherapy Compared With Standard Chemotherapy and Lung Radiation in Ewing Sarcoma With Pulmonary Metastases: Results of the European Ewing Tumour Working Initiative of National Groups, 99 Trial and EWING 2008. J. Clin. Oncol. 2019, 37, 3192–3202. [Google Scholar] [CrossRef] [PubMed]
- Falk, A.T.; Moureau-Zabotto, L.; Ouali, M.; Penel, N.; Italiano, A.; Bay, J.O.; Olivier, T.; Sunyach, M.P.; Boudou-Roquette, P.; Salas, S.; et al. Effect on survival of local ablative treatment of metastases from sarcomas: A study of the French sarcoma group. Clin. Oncol. (R Coll. Radiol.) 2015, 27, 48–55. [Google Scholar] [CrossRef]
- Bongiovanni, A.; Recine, F.; Foca, F.; Fausti, V.; Riva, N.; Fabbri, G.; Severi, S.; Liverani, C.; De Vita, A.; Spadazzi, C.; et al. Metastatic neuroendocrine neoplasia treatments in patients over 70 years of age. Endocr. Connect. 2018, 7, 1535–1541. [Google Scholar] [CrossRef] [Green Version]
- Hardie, C.M.; Allouni, A.; Edwards, S.; Ahmed, N.; Maraveyas, A.; Matteucci, P.L. PET-CT for staging pT4b melanomas prior to sentinel lymph node biopsy: A 5-year review. Melanoma Res. 2021. [Google Scholar] [CrossRef] [PubMed]
- Cacicedo, J.; Fernandez, I.; Del Hoyo, O.; Dolado, A.; Gomez-Suarez, J.; Hortelano, E.; Sancho, A.; Pijoan, J.I.; Alvarez, J.; Espinosa, J.M.; et al. Should PET/CT be implemented in the routine imaging work-up of locally advanced head and neck squamous cell carcinoma? A prospective analysis. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 1378–1389. [Google Scholar] [CrossRef]
- Scarsbrook, A.B.S. Evidence-based indications for the use of PET-CT in the United Kingdom 2016. In Royal College of Physicians of London, Royal College of Physicians and Surgeons of Glasgow, Royal College of Physicians of Edinburgh, The Royal College of Radiologists, British Nuclear Medicine Society, Administration of Radioactive Substances Advisory Committee; The Royal College of Radiologists: London, UK, 2016. [Google Scholar]
- Cavo, M.; Terpos, E.; Nanni, C.; Moreau, P.; Lentzsch, S.; Zweegman, S.; Hillengass, J.; Engelhardt, M.; Usmani, S.Z.; Vesole, D.H.; et al. Role of (18)F-FDG PET/CT in the diagnosis and management of multiple myeloma and other plasma cell disorders: A consensus statement by the International Myeloma Working Group. Lancet Oncol. 2017, 18, e206–e217. [Google Scholar] [CrossRef]
- Cheson, B.D.; Pfistner, B.; Juweid, M.E.; Gascoyne, R.D.; Specht, L.; Horning, S.J.; Coiffier, B.; Fisher, R.I.; Hagenbeek, A.; Zucca, E.; et al. Revised response criteria for malignant lymphoma. J. Clin. Oncol. 2007, 25, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Shim, S.S.; Lee, K.S.; Kim, B.T.; Chung, M.J.; Lee, E.J.; Han, J.; Choi, J.Y.; Kwon, O.J.; Shim, Y.M.; Kim, S. Non-small cell lung cancer: Prospective comparison of integrated FDG PET/CT and CT alone for preoperative staging. Radiology 2005, 236, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Park, J.R.; Bagatell, R.; Cohn, S.L.; Pearson, A.D.; Villablanca, J.G.; Berthold, F.; Burchill, S.; Boubaker, A.; McHugh, K.; Nuchtern, J.G.; et al. Revisions to the International Neuroblastoma Response Criteria: A Consensus Statement From the National Cancer Institute Clinical Trials Planning Meeting. J. Clin. Oncol. 2017, 35, 2580–2587. [Google Scholar] [CrossRef]
- El-Hennawy, G.; Moustafa, H.; Omar, W.; Elkinaai, N.; Kamel, A.; Zaki, I.; Farid, N.; El-Kholy, E. Different (18) F-FDG PET parameters for the prediction of histological response to neoadjuvant chemotherapy in pediatric Ewing sarcoma family of tumors. Pediatr. Blood Cancer 2020, 67, e28605. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, D.S.; Schuetze, S.M.; Butrynski, J.E.; Rajendran, J.G.; Vernon, C.B.; Conrad, E.U., 3rd; Eary, J.F. [18F]Fluorodeoxyglucose positron emission tomography predicts outcome for Ewing sarcoma family of tumors. J. Clin. Oncol. 2005, 23, 8828–8834. [Google Scholar] [CrossRef]
- Sobic Saranovic, D.P.; Nikitovic, M.; Saponjski, J.; Grozdic Milojevic, I.; Paripovic, L.; Saranovic, D.; Beatovic, S.; Artiko, V.M. Post-treatment FDG PET/CT predicts progression-free survival in young patients with small round blue cell tumors: Ewing sarcoma and PNET. Eur. J. Radiol. 2020, 129, 109076. [Google Scholar] [CrossRef]
- Palmerini, E.; Colangeli, M.; Nanni, C.; Fanti, S.; Marchesi, E.; Paioli, A.; Picci, P.; Cambioli, S.; Donati, D.; Cevolani, L.; et al. The role of FDG PET/CT in patients treated with neoadjuvant chemotherapy for localized bone sarcomas. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 215–223. [Google Scholar] [CrossRef] [Green Version]
- Daldrup-Link, H.E.; Franzius, C.; Link, T.M.; Laukamp, D.; Sciuk, J.; Jurgens, H.; Schober, O.; Rummeny, E.J. Whole-body MR imaging for detection of bone metastases in children and young adults: Comparison with skeletal scintigraphy and FDG PET. AJR Am. J. Roentgenol. 2001, 177, 229–236. [Google Scholar] [CrossRef]
- Treglia, G.; Salsano, M.; Stefanelli, A.; Mattoli, M.V.; Giordano, A.; Bonomo, L. Diagnostic accuracy of (1)(8)F-FDG-PET and PET/CT in patients with Ewing sarcoma family tumours: A systematic review and a meta-analysis. Skelet. Radiol. 2012, 41, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Newman, E.N.; Jones, R.L.; Hawkins, D.S. An evaluation of [F-18]-fluorodeoxy-D-glucose positron emission tomography, bone scan, and bone marrow aspiration/biopsy as staging investigations in Ewing sarcoma. Pediatr. Blood Cancer 2013, 60, 1113–1117. [Google Scholar] [CrossRef]
- London, K.; Stege, C.; Cross, S.; Onikul, E.; Graf, N.; Kaspers, G.; Dalla-Pozza, L.; Howman-Giles, R. 18F-FDG PET/CT compared to conventional imaging modalities in pediatric primary bone tumors. Pediatr. Radiol. 2012, 42, 418–430. [Google Scholar] [CrossRef]
- Volker, T.; Denecke, T.; Steffen, I.; Misch, D.; Schonberger, S.; Plotkin, M.; Ruf, J.; Furth, C.; Stover, B.; Hautzel, H.; et al. Positron emission tomography for staging of pediatric sarcoma patients: Results of a prospective multicenter trial. J. Clin. Oncol. 2007, 25, 5435–5441. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Li, F.; Yan, Z.; Ma, Y.; Xiong, F.; Cai, X.; Zhang, Q.; Liu, F.; Dong, J. Effectiveness of 18F-FDG PET/CT in the diagnosis, staging and recurrence monitoring of Ewing sarcoma family of tumors: A meta-analysis of 23 studies. Medicine (Baltimore) 2018, 97, e13457. [Google Scholar] [CrossRef] [PubMed]
- Guimaraes, M.D.; Noschang, J.; Teixeira, S.R.; Santos, M.K.; Lederman, H.M.; Tostes, V.; Kundra, V.; Oliveira, A.D.; Hochhegger, B.; Marchiori, E. Whole-body MRI in pediatric patients with cancer. Cancer Imaging 2017, 17, 6. [Google Scholar] [CrossRef] [Green Version]
- Frat, A.; Agildere, M.; Gencoglu, A.; Cakir, B.; Akin, O.; Akcali, Z.; Aktas, A. Value of whole-body turbo short tau inversion recovery magnetic resonance imaging with panoramic table for detecting bone metastases: Comparison with 99MTc-methylene diphosphonate scintigraphy. J. Comput. Assist. Tomogr. 2006, 30, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Walker, R.; Kessar, P.; Blanchard, R.; Dimasi, M.; Harper, K.; DeCarvalho, V.; Yucel, E.K.; Patriquin, L.; Eustace, S. Turbo STIR magnetic resonance imaging as a whole-body screening tool for metastases in patients with breast carcinoma: Preliminary clinical experience. J. Magn. Reson. Imaging 2000, 11, 343–350. [Google Scholar] [CrossRef]
- Taylor, S.A.; Mallett, S.; Ball, S.; Beare, S.; Bhatnagar, G.; Bhowmik, A.; Boavida, P.; Bridgewater, J.; Clarke, C.S.; Duggan, M.; et al. Diagnostic accuracy of whole-body MRI versus standard imaging pathways for metastatic disease in newly diagnosed non-small-cell lung cancer: The prospective Streamline L trial. Lancet Respir. Med. 2019, 7, 523–532. [Google Scholar] [CrossRef] [Green Version]
- Taylor, S.A.; Mallett, S.; Beare, S.; Bhatnagar, G.; Blunt, D.; Boavida, P.; Bridgewater, J.; Clarke, C.S.; Duggan, M.; Ellis, S.; et al. Diagnostic accuracy of whole-body MRI versus standard imaging pathways for metastatic disease in newly diagnosed colorectal cancer: The prospective Streamline C trial. Lancet Gastroenterol. Hepatol. 2019, 4, 529–537. [Google Scholar] [CrossRef] [Green Version]
- Taylor, S.A.; Mallett, S.; Miles, A.; Beare, S.; Bhatnagar, G.; Bridgewater, J.; Glynne-Jones, R.; Goh, V.; Groves, A.M.; Janes, S.M.; et al. Streamlining staging of lung and colorectal cancer with whole body MRI; study protocols for two multicentre, non-randomised, single-arm, prospective diagnostic accuracy studies (Streamline C and Streamline L). BMC Cancer 2017, 17, 299. [Google Scholar] [CrossRef] [Green Version]
- Smets, A.M.; Deurloo, E.E.; Slager, T.J.E.; Stoker, J.; Bipat, S. Whole-body magnetic resonance imaging for detection of skeletal metastases in children and young people with primary solid tumors—Systematic review. Pediatr. Radiol. 2018, 48, 241–252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, J.; Seith, A.; Kumar, A.; Sharma, R.; Bakhshi, S.; Kumar, R.; Agarwala, S. Whole-body MR imaging with the use of parallel imaging for detection of skeletal metastases in pediatric patients with small-cell neoplasms: Comparison with skeletal scintigraphy and FDG PET/CT. Pediatr. Radiol. 2008, 38, 953–962. [Google Scholar] [CrossRef]
- Aryal, A.; Kumar, V.S.; Shamim, S.A.; Gamanagatti, S.; Khan, S.A. What Is the Comparative Ability of 18F-FDG PET/CT, 99mTc-MDP Skeletal Scintigraphy, and Whole-body MRI as a Staging Investigation to Detect Skeletal Metastases in Patients with Osteosarcoma and Ewing Sarcoma? Clin. Orthop. Relat. Res. 2021. [Google Scholar] [CrossRef] [PubMed]
- Gyorke, T.; Zajic, T.; Lange, A.; Schafer, O.; Moser, E.; Mako, E.; Brink, I. Impact of FDG PET for staging of Ewing sarcomas and primitive neuroectodermal tumours. Nucl. Med. Commun. 2006, 27, 17–24. [Google Scholar] [CrossRef]
- Quartuccio, N.; Fox, J.; Kuk, D.; Wexler, L.H.; Baldari, S.; Cistaro, A.; Schoder, H. Pediatric bone sarcoma: Diagnostic performance of (1)(8)F-FDG PET/CT versus conventional imaging for initial staging and follow-up. AJR Am. J. Roentgenol. 2015, 204, 153–160. [Google Scholar] [CrossRef]
- Kalus, S.; Saifuddin, A. Whole-body MRI vs bone scintigraphy in the staging of Ewing sarcoma of bone: A 12-year single-institution review. Eur. Radiol. 2019, 29, 5700–5708. [Google Scholar] [CrossRef]
- Bosma, S.E.; Vriens, D.; Gelderblom, H.; van de Sande, M.A.J.; Dijkstra, P.D.S.; Bloem, J.L. (18)F-FDG PET-CT versus MRI for detection of skeletal metastasis in Ewing sarcoma. Skelet. Radiol. 2019, 48, 1735–1746. [Google Scholar] [CrossRef] [Green Version]
- Cesari, M.; Righi, A.; Colangeli, M.; Gambarotti, M.; Spinnato, P.; Ferraro, A.; Longhi, A.; Abate, M.E.; Palmerini, E.; Paioli, A.; et al. Bone marrow biopsy in the initial staging of Ewing sarcoma: Experience from a single institution. Pediatr. Blood Cancer 2019, 66, e27653. [Google Scholar] [CrossRef]
- Yagci-Kupeli, B.; Kocyigit-Deveci, E.; Adamhasan, F.; Kupeli, S. The Value of 18F-FDG PET/CT in Detecting Bone Marrow Involvement in Childhood Cancers. J. Pediatr. Hematol. Oncol. 2019, 41, 438–441. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, C.; Shimoi, T.; Sumiyoshi Okuma, H.; Kawachi, A.; Sudo, K.; Shimomura, A.; Noguchi, E.; Kodaira, M.; Yunokawa, M.; Yonemori, K.; et al. Bone marrow examination in patients with Ewing sarcoma/peripheral primitive neuroectodermal tumor without metastasis based on (18)F-fluorodeoxyglucose positron emission tomography/computed tomography. Med. Oncol. 2019, 36, 58. [Google Scholar] [CrossRef] [Green Version]
- Kasalak, O.; Glaudemans, A.; Overbosch, J.; Jutte, P.C.; Kwee, T.C. Can FDG-PET/CT replace blind bone marrow biopsy of the posterior iliac crest in Ewing sarcoma? Skelet. Radiol. 2018, 47, 363–367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zapata, C.P.; Cuglievan, B.; Zapata, C.M.; Olavarrieta, R.; Raskin, S.; Desai, K.; De Angulo, G. PET/CT versus bone marrow biopsy in the initial evaluation of bone marrow infiltration in various pediatric malignancies. Pediatr. Blood Cancer 2018, 65. [Google Scholar] [CrossRef] [PubMed]
- Kopp, L.M.; Hu, C.; Rozo, B.; White-Collins, A.; Huh, W.W.; Yarborough, A.; Herzog, C.E.; Hingorani, P. Utility of bone marrow aspiration and biopsy in initial staging of Ewing sarcoma. Pediatr. Blood Cancer 2015, 62, 12–15. [Google Scholar] [CrossRef] [PubMed]
- Oberlin, O.; Bayle, C.; Hartmann, O.; Terrier-Lacombe, M.J.; Lemerle, J. Incidence of bone marrow involvement in Ewing’s sarcoma: Value of extensive investigation of the bone marrow. Med. Pediatr. Oncol. 1995, 24, 343–346. [Google Scholar] [CrossRef]
- Campbell, K.M.; Shulman, D.S.; Grier, H.E.; DuBois, S.G. Role of bone marrow biopsy for staging new patients with Ewing sarcoma: A systematic review. Pediatr. Blood Cancer 2021, 68, e28807. [Google Scholar] [CrossRef]
- Breitegger, B.; Holzer, L.A.; Beham-Schmid, C.; Urban, C.; Liegl-Atzwanger, B.; Leithner, A. Bone marrow aspirations in Ewing sarcomas: Are they still necessary? A single-center retrospective analysis and review of the literature. J. Cancer Res. Ther. 2020, 16, 713–717. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.A.A. Bone marrow and not bone is the primary site for skeletal metastasis: Critical role of [18F] fluorodeoxyglucose positron emission tomography in this setting. Am. Soc. Clin. Oncol. 2006. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Torigian, D.; Alavi, A. Evolving concept of imaging bone marrow metastasis in the twenty-first century: Critical role of FDG-PET. Eur. J. Nucl. Med. Mol. Imaging 2008, 35, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Hoilund-Carlsen, P.F.; Hess, S.; Werner, T.J.; Alavi, A. Cancer metastasizes to the bone marrow and not to the bone: Time for a paradigm shift! Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 893–897. [Google Scholar] [CrossRef] [Green Version]
- Meyer, J.S.; Nadel, H.R.; Marina, N.; Womer, R.B.; Brown, K.L.; Eary, J.F.; Gorlick, R.; Grier, H.E.; Randall, R.L.; Lawlor, E.R.; et al. Imaging guidelines for children with Ewing sarcoma and osteosarcoma: A report from the Children’s Oncology Group Bone Tumor Committee. Pediatr. Blood Cancer 2008, 51, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Gerrand, C.; Athanasou, N.; Brennan, B.; Grimer, R.; Judson, I.; Morland, B.; Peake, D.; Seddon, B.; Whelan, J.; British Sarcoma, G. UK guidelines for the management of bone sarcomas. Clin. Sarcoma Res. 2016, 6, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biermann, J.S.; Chow, W.; Reed, D.R.; Lucas, D.; Adkins, D.R.; Agulnik, M.; Benjamin, R.S.; Brigman, B.; Budd, G.T.; Curry, W.T.; et al. NCCN Guidelines Insights: Bone Cancer, Version 2.2017. J. Natl. Compr. Cancer Netw. 2017, 15, 155–167. [Google Scholar] [CrossRef] [Green Version]
- Casali, P.G.; Bielack, S.; Abecassis, N.; Aro, H.T.; Bauer, S.; Biagini, R.; Bonvalot, S.; Boukovinas, I.; Bovee, J.; Brennan, B.; et al. Bone sarcomas: ESMO-PaedCan-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2018, 29, iv79–iv95. [Google Scholar] [CrossRef]
- Cheson, B.D.; Fisher, R.I.; Barrington, S.F.; Cavalli, F.; Schwartz, L.H.; Zucca, E.; Lister, T.A.; Alliance, A.L.; Lymphoma, G.; Eastern Cooperative Oncology Group; et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: The Lugano classification. J. Clin. Oncol. 2014, 32, 3059–3068. [Google Scholar] [CrossRef] [PubMed]
- Voltin, C.A.; Goergen, H.; Baues, C.; Fuchs, M.; Mettler, J.; Kreissl, S.; Oertl, J.; Klaeser, B.; Moccia, A.; Drzezga, A.; et al. Value of bone marrow biopsy in Hodgkin lymphoma patients staged by FDG PET: Results from the German Hodgkin Study Group trials HD16, HD17, and HD18. Ann. Oncol. 2018, 29, 1926–1931. [Google Scholar] [CrossRef] [PubMed]

| Ref. | Pt No. | Patient Group | Anatomic Imaging | Functional Imaging | BMB | Outcomes |
|---|---|---|---|---|---|---|
| Cesari [42] | 504 | ES of bone Median age 16 y (1–68 y) 1998–2017 | Chest CT (n = 504), bone scintigraphy (n = 366), WB MRI (n = 1), WB CT (n = 1) | FDG-PET/CT (n = 130) Bone scan and FDG-PET/CT (n = 6) | BMAB (n = 504, unilateral) |
|
| Yagci-Kupeli [43] | 94 | 94 solid tumours (ES n = 16) Median age 12 y (1–18 y) 2014–2017 | FDG-PET/CT | BMB |
| |
| Inagaki [44] | 26 | ES Median age 26 y (11–53 y) 2010–2016 | CT contrast (n = 25), MRI primary (n = 11) | FDG-PET/CT (n = 26) | BMAB (n = 26, unilateral) |
|
| Kasalak [45] | 20 | ES Mean age 15.9 y (5–57 y) 2009–2017 | FDG-PET/CT (n = 20) | BMB (18 bilateral, 2 unilateral) |
| |
| Zapata [46] | 69 | 69 mixed solid tumours ES (n = 7): mean age 10.7 y (3–16 y) 2009–2014 | FDG-PET/CT (n = 69)-reported on presence of bone marrow disease | BMAB |
| |
| Kopp [47] | 116 | ES Median age 13 y (1–38 y) 2000–2012 | Chest CT, MRI primary | Bone scans | BMAB (n = 111/116, bilateral) |
|
| Newman [25] | 91 | ES Median age 14.9 y (3.8–56.2 y) 2001–2011 | FDG-PET/CT (n = 80) Bone scan (n = 74) | BMA/B (n = 80 patients: 59 aspirates, 62 bilateral biopsies) |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ingley, K.M.; Wan, S.; Vöö, S.; Windsor, R.; Michelagnoli, M.; Saifuddin, A.; Strauss, S.J. Is It Time to Call Time on Bone Marrow Biopsy for Staging Ewing Sarcoma (ES)? Cancers 2021, 13, 3261. https://doi.org/10.3390/cancers13133261
Ingley KM, Wan S, Vöö S, Windsor R, Michelagnoli M, Saifuddin A, Strauss SJ. Is It Time to Call Time on Bone Marrow Biopsy for Staging Ewing Sarcoma (ES)? Cancers. 2021; 13(13):3261. https://doi.org/10.3390/cancers13133261
Chicago/Turabian StyleIngley, Katrina M., Simon Wan, Stefan Vöö, Rachael Windsor, Maria Michelagnoli, Asif Saifuddin, and Sandra J. Strauss. 2021. "Is It Time to Call Time on Bone Marrow Biopsy for Staging Ewing Sarcoma (ES)?" Cancers 13, no. 13: 3261. https://doi.org/10.3390/cancers13133261
APA StyleIngley, K. M., Wan, S., Vöö, S., Windsor, R., Michelagnoli, M., Saifuddin, A., & Strauss, S. J. (2021). Is It Time to Call Time on Bone Marrow Biopsy for Staging Ewing Sarcoma (ES)? Cancers, 13(13), 3261. https://doi.org/10.3390/cancers13133261






