Improving Risk Stratification of Early Oral Tongue Cancer with TNM-Immune (TNM-I) Staging System
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
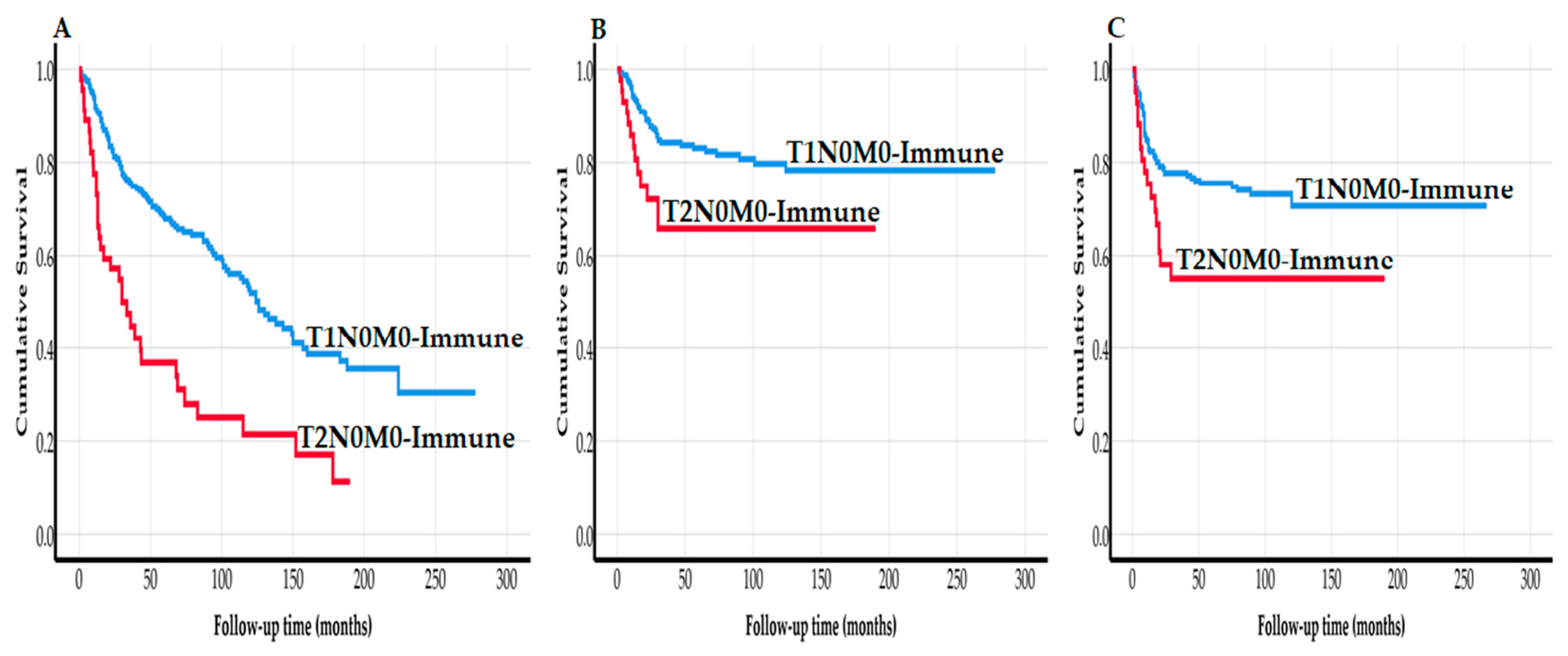
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Amin, M.B.; Edge, S.; Greene, F. AJCC Cancer Staging Manual, 8th ed.; Springer: New York, NY, USA, 2017. [Google Scholar]
- Almangush, A.; Pirinen, M.; Youssef, O.; Mäkitie, A.A.; Leivo, I. Risk stratification in oral squamous cell carcinoma using staging of the eighth American Joint Committee on Cancer: Systematic review and meta-analysis. Head Neck 2020, 42, 3002–3017. [Google Scholar] [CrossRef] [PubMed]
- Richards, C.H.; Roxburgh, C.S.; Powell, A.; Foulis, A.K.; Horgan, P.G.; McMillan, D. The clinical utility of the local inflammatory response in colorectal cancer. Eur. J. Cancer 2014, 50, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Zhou, Y.; Lu, X.; Bian, Z.; Chen, Y.; Zhou, J.; Zhang, L.; Hao, Z.; Zhang, M.; Liang, C. Immune response drives outcomes in prostate cancer: Implications for immunotherapy. Mol. Oncol. 2021, 15, 1358–1375. [Google Scholar] [CrossRef]
- Galon, J.; Bruni, D. Tumor Immunology and Tumor Evolution: Intertwined Histories. Immunity 2020, 52, 55–81. [Google Scholar] [CrossRef]
- Swisher, S.K.; Wu, Y.; Castaneda, C.A.; Lyons, G.R.; Yang, F.; Tapia, C.; Wang, X.; Casavilca, S.A.A.; Bassett, R.; Castillo, M.; et al. Interobserver Agreement Between Pathologists Assessing Tumor-Infiltrating Lymphocytes (TILs) in Breast Cancer Using Methodology Proposed by the International TILs Working Group. Ann. Surg. Oncol. 2016, 23, 2242–2248. [Google Scholar] [CrossRef]
- Heikkinen, I.; Bello, I.O.; Wahab, A.; Hagström, J.; Haglund, C.; Coletta, R.D.; Nieminen, P.; Mäkitie, A.A.; Salo, T.; Leivo, I.; et al. Assessment of Tumor-infiltrating Lymphocytes Predicts the Behavior of Early-stage Oral Tongue Cancer. Am. J. Surg. Pathol. 2019, 43, 1392–1396. [Google Scholar] [CrossRef]
- Almangush, A.; Leivo, I.; Mäkitie, A.A. Overall assessment of tumor-infiltrating lymphocytes in head and neck squamous cell carcinoma: time to take notice. Acta Oto-Laryngol. 2020, 140, 246–248. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kim, C.H.; Lee, Y.; Lee, J.H.; Chae, Y.-S. Tumour infiltrating lymphocytes are predictors of lymph node metastasis in early gastric cancers. Pathology 2017, 49, 589–595. [Google Scholar] [CrossRef]
- Taube, J.M. Emerging Immunologic Biomarkers: Setting the (TNM-Immune) Stage. Clin. Cancer Res. 2014, 20, 2023–2025. [Google Scholar] [CrossRef] [Green Version]
- Takada, K.; Kashiwagi, S.; Asano, Y.; Goto, W.; Kouhashi, R.; Yabumoto, A.; Morisaki, T.; Shibutani, M.; Takashima, T.; Fujita, H.; et al. Prediction of lymph node metastasis by tumor-infiltrating lymphocytes in T1 breast cancer. BMC Cancer 2020, 20, 1–13. [Google Scholar] [CrossRef]
- Corredor, G.; Wang, X.; Zhou, Y.; Lu, C.; Fu, P.; Syrigos, K.N.; Rimm, D.L.; Yang, M.; Romero, E.; Schalper, K.A.; et al. Spatial Architecture and Arrangement of Tumor-Infiltrating Lymphocytes for Predicting Likelihood of Recurrence in Early-Stage Non–Small Cell Lung Cancer. Clin. Cancer Res. 2019, 25, 1526–1534. [Google Scholar] [CrossRef] [Green Version]
- Hendry, S.; Salgado, R.; Gevaert, T.; Russell, P.A.; John, T.; Thapa, B.; Christie, M.; van de Vijver, K.; Estrada, M.V.; Gonzalez-Ericsson, P.I.; et al. Assessing Tumor-Infiltrating Lymphocytes in Solid Tumors: A Practical Review for Pathologists and Proposal for a Standardized Method from the International Immuno-Oncology Biomarkers Working Group: Part 2: TILs in Melanoma, Gastrointestinal Tract Carcinomas, Non–Small Cell Lung Carcinoma and Mesothelioma, Endometrial and Ovarian Carcinomas, Squamous Cell Carcinoma of the Head and Neck, Genitourinary Carcinomas, and Primary Brain Tumors. Adv. Anat. Pathol. 2017, 24, 311–335. [Google Scholar] [CrossRef] [PubMed]
- Badalamenti, G.; Fanale, D.; Incorvaia, L.; Barraco, N.; Listì, A.; Maragliano, R.; Vincenzi, B.; Calò, V.; Iovanna, J.L.; Bazan, V.; et al. Role of tumor-infiltrating lymphocytes in patients with solid tumors: Can a drop dig a stone? Cell. Immunol. 2019, 343, 103753. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [Green Version]
- Galon, J.; Mlecnik, B.; Bindea, G.; Angell, H.K.; Berger, A.; Lagorce, C.; Lugli, A.; Zlobec, I.; Hartmann, A.; Bifulco, C.; et al. Towards the introduction of the ‘Immunoscore’ in the classification of malignant tumours. J. Pathol. 2014, 232, 199–209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Donnem, T.; Kilvaer, T.K.; Andersen, S.; Richardsen, E.; Paulsen, E.E.; Hald, S.M.; Al-Saad, S.; Brustugun, O.T.; Helland, Å.; Lund-Iversen, M.; et al. Strategies for clinical implementation of TNM-Immunoscore in resected nonsmall-cell lung cancer. Ann. Oncol. 2016, 27, 225–232. [Google Scholar] [CrossRef]
- Spector, M.E.; Bellile, E.; Amlani, L.; Zarins, K.; Smith, J.; Brenner, J.C.; Rozek, L.; Nguyen, A.; Thomas, D.; McHugh, J.B.; et al. Prognostic Value of Tumor-Infiltrating Lymphocytes in Head and Neck Squamous Cell Carcinoma. JAMA Otolaryngol. Neck Surg. 2019, 145, 1012–1019. [Google Scholar] [CrossRef] [PubMed]
- Borsetto, D.; Tomasoni, M.; Payne, K.; Polesel, J.; Deganello, A.; Bossi, P.; Tysome, J.; Masterson, L.; Tirelli, G.; Tofanelli, M.; et al. Prognostic Significance of CD4+ and CD8+ Tumor-Infiltrating Lymphocytes in Head and Neck Squamous Cell Carcinoma: A Meta-Analysis. Cancers 2021, 13, 781. [Google Scholar] [CrossRef]
- Iseki, Y.; Shibutani, M.; Maeda, K.; Nagahara, H.; Fukuoka, T.; Matsutani, S.; Kashiwagi, S.; Tanaka, H.; Hirakawa, K.; Ohira, M. A new method for evaluating tumor-infiltrating lymphocytes (TILs) in colorectal cancer using hematoxylin and eosin (H-E)-stained tumor sections. PLoS ONE 2018, 13, e0192744. [Google Scholar] [CrossRef]
- Mangia, A.; Saponaro, C.; Vagheggini, A.; Opinto, G.; Centonze, M.; Vicenti, C.; Popescu, O.; Pastena, M.; Giotta, F.; Silvestris, N. Should Tumor Infiltrating Lymphocytes, Androgen Receptor, and FOXA1 Expression Predict the Clinical Outcome in Triple Negative Breast Cancer Patients? Cancers 2019, 11, 1393. [Google Scholar] [CrossRef] [Green Version]
- Shaban, M.; Khurram, S.A.; Fraz, M.M.; Alsubaie, N.; Masood, I.; Mushtaq, S.; Hassan, M.; Loya, A.; Rajpoot, N.M. A Novel Digital Score for Abundance of Tumour Infiltrating Lymphocytes Predicts Disease Free Survival in Oral Squamous Cell Carcinoma. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Parameter | Total (%) | T1N0M0-Immune | T2N0M0-Immune | p Value |
|---|---|---|---|---|
| Number (%) 243 (83.8%) | Number (%) 47 (16.2%) | |||
| Age | 0.844 | |||
| ≤60 | 121 (41.7%) | 102 (84.3%) | 19 (15.7%) | |
| >60 | 169 (58.3%) | 141 (83.4%) | 28 (16.6%) | |
| Gender | 0.164 | |||
| Men | 152 (52.4%) | 123 (80.9%) | 29 (19.1%) | |
| Women | 138 (47.6%) | 120 (87.0%) | 18 (13.0%) | |
| WHO Grade | 0.102 | |||
| Well-differentiated | 95 (32.8%) | 83 (87.4%) | 12 (12.6%) | |
| Moderately-differentiated | 125 (43.1%) | 107 (85.6%) | 18 (14.4%) | |
| Poorly-differentiated | 70 (24.1%) | 53 (75.7%) | 17 (24.3%) | |
| Perineural invasion | 0.013 | |||
| No | 254 (87.6%) | 218 (85.8%) | 36 (14.2%) | |
| Yes | 36 (12.4%) | 25 (69.4%) | 11 (30.6%) | |
| Pattern of invasion | 0.037 | |||
| Cohesive | 72 (24.8%) | 66 (91.7%) | 6 (8.3%) | |
| Infiltrative | 218 (75.2%) | 177 (81.2%) | 41 (18.8%) | |
| TNM AJCC 8 | 0.140 | |||
| T1N0M0 | 88 (30.3%) | 78 (88.6%) | 10 (11.4%) | |
| T2N0M0 | 202 (69.7%) | 165 (81.7%) | 37 (18.3%) |
| Parameter | Overall Survival | Disease-Specific Survival | Disease-Free Survival | |||
|---|---|---|---|---|---|---|
| Univariable Analysis HR (95% CI) p Value | Multivariable Analysis HR (95% CI) p Value | Univariable Analysis HR (95% CI) p Value | Multivariable Analysis HR (95% CI) p Value | Univariable Analysis HR (95% CI) p Value | Multivariable Analysis HR (95% CI) p Value | |
| Age | p < 0.001 | p < 0.001 | p = 0.012 | p = 0.010 | p = 0.015 | p = 0.014 |
| ≤60 | Reference | Reference | Reference | Reference | Reference | Reference |
| >60 | 2.32 (1.63–3.31) | 2.49 (1.73–3.61) | 2.12 (1.18–3.79) | 2.19 (1.20–4.01) | 1.81 (1.12–2.93) | 1.86 (1.13–3.05) |
| Gender | p = 0.164 | p = 0.022 | p = 0.339 | p = 0.433 | p = 0.571 | p = 0.888 |
| Men | Reference | Reference | Reference | Reference | Reference | Reference |
| Women | 0.79 (0.57–1.10) | 0.67 (0.47–0.94) | 1.29 (0.76–2.20) | 1.25 (0.72–2.17) | 1.14 (0.73–1.78) | 0.97 (0.61–1.54) |
| WHO Grade | p = 0.233 | p = 0.219 | p = 0.316 | p = 0.157 | p = 0.798 | p = 0.497 |
| Well | Reference | Reference | Reference | Reference | Reference | Reference |
| Moderate | 1.34 (0.92–1.96) | 1.39 (0.96–2.05) | 1.64 (0.87–3.12) | 1.85 (0.97–3.53) | 1.09 (0.65–1.86) | 1.15 (0.68–1.96) |
| Poor | 1.02 (0.65–1.61) | 1.18 (0.74–1.88) | 1.41 (0.66–2.99) | 1.78 (0.82–3.85) | 1.23 (0.68–2.22) | 1.44 (0.79–2.64) |
| Perineural invasion | p = 0.086 | p = 0.233 | p = 0.478 | p = 0.743 | p = 0.224 | p = 0.13 |
| No | Reference | Reference | Reference | Reference | Reference | Reference |
| Yes | 1.47 (0.95–2.28) | 1.32 (0.84–2.09) | 1.31 (0.62–2.78) | 1.14 (0.53–2.46) | 1.47 (0.79–2.71) | 1.65 (0.87–3.16) |
| TNM AJCC 8 | p = 0.473 | p = 0.697 | p = 0.339 | p = 0.239 | p = 0.108 | p = 0.075 |
| T1N0M0 | Reference | Reference | Reference | Reference | Reference | Reference |
| T2N0M0 | 1.15 (0.79–1.68) | 1.08 (0.73–1.61) | 1.36 (0.73–2.53) | 1.48 (0.77–2.83) | 0.69 (0.43–1.09) | 0.64 (0.39–1.05) |
| TNM-Immune | p < 0.001 | p < 0.001 | p = 0.012 | p = 0.008 | p = 0.015 | p = 0.017 |
| T1N0M0-Immune | Reference | Reference | Reference | Reference | Reference | Reference |
| T2N0M0-Immune | 2.52 (1.71–3.71) | 2.87 (1.92–4.28) | 2.22 (1.19–4.15) | 2.41 (1.26–4.60) | 1.96 (1.14–3.37) | 1.97 (1.13–3.43) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Almangush, A.; Bello, I.O.; Heikkinen, I.; Hagström, J.; Haglund, C.; Kowalski, L.P.; Coletta, R.D.; Mäkitie, A.A.; Salo, T.; Leivo, I. Improving Risk Stratification of Early Oral Tongue Cancer with TNM-Immune (TNM-I) Staging System. Cancers 2021, 13, 3235. https://doi.org/10.3390/cancers13133235
Almangush A, Bello IO, Heikkinen I, Hagström J, Haglund C, Kowalski LP, Coletta RD, Mäkitie AA, Salo T, Leivo I. Improving Risk Stratification of Early Oral Tongue Cancer with TNM-Immune (TNM-I) Staging System. Cancers. 2021; 13(13):3235. https://doi.org/10.3390/cancers13133235
Chicago/Turabian StyleAlmangush, Alhadi, Ibrahim O. Bello, Ilkka Heikkinen, Jaana Hagström, Caj Haglund, Luiz Paulo Kowalski, Ricardo D. Coletta, Antti A. Mäkitie, Tuula Salo, and Ilmo Leivo. 2021. "Improving Risk Stratification of Early Oral Tongue Cancer with TNM-Immune (TNM-I) Staging System" Cancers 13, no. 13: 3235. https://doi.org/10.3390/cancers13133235
APA StyleAlmangush, A., Bello, I. O., Heikkinen, I., Hagström, J., Haglund, C., Kowalski, L. P., Coletta, R. D., Mäkitie, A. A., Salo, T., & Leivo, I. (2021). Improving Risk Stratification of Early Oral Tongue Cancer with TNM-Immune (TNM-I) Staging System. Cancers, 13(13), 3235. https://doi.org/10.3390/cancers13133235








