Requirements for Designing an Effective Metallic Nanoparticle (NP)-Boosted Radiation Therapy (RT)
Abstract
:Simple Summary
Abstract
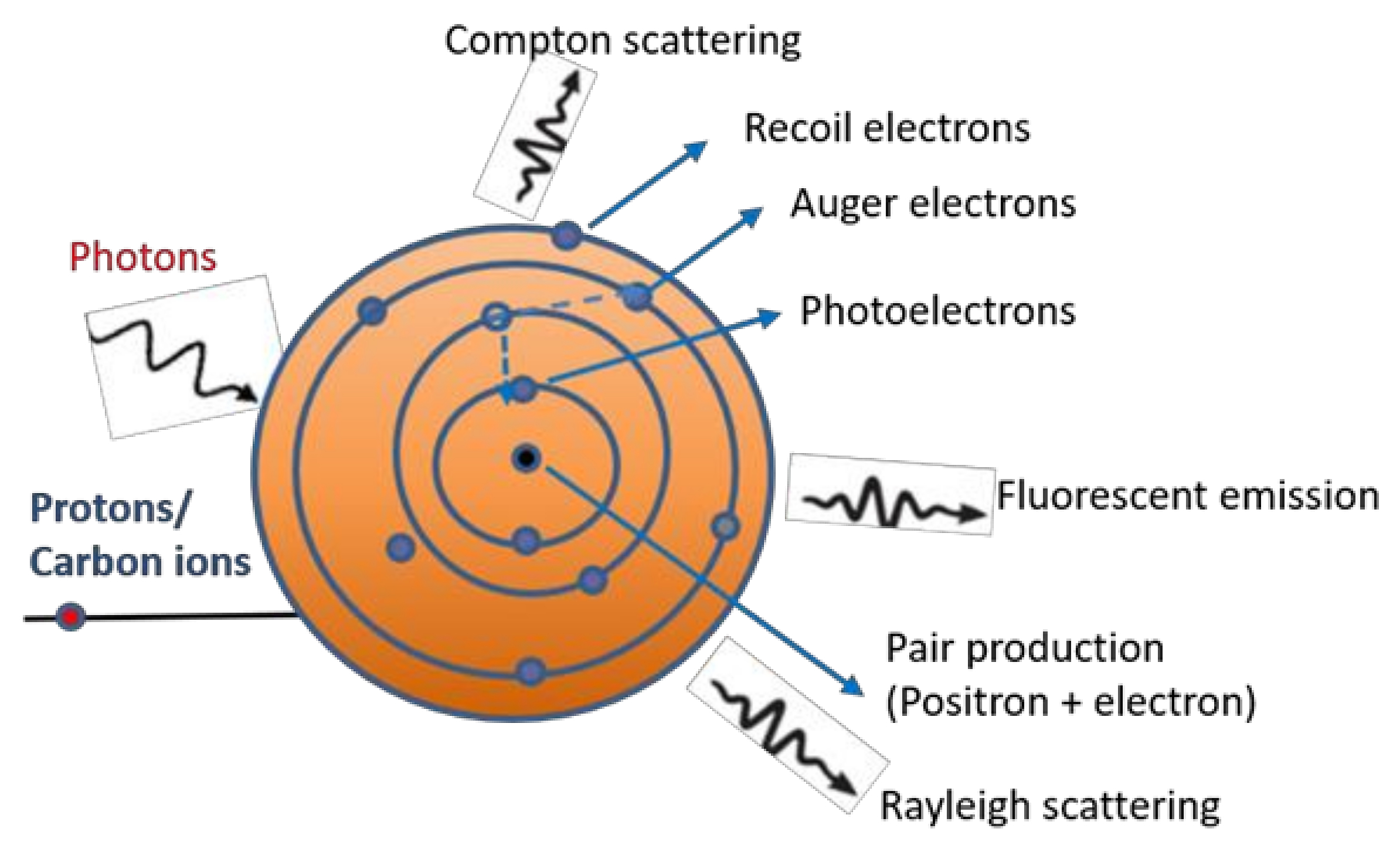
1. Introduction
1.1. Most Promising Metallic NPs Proposed for RT
1.1.1. Gold (Au, Z = 79)
1.1.2. Platinum (Pt, Z = 78)
1.1.3. Hafnium (Hf, Z = 72)
1.1.4. Gadolinium (Gd, Z = 64)
1.1.5. Silver (Ag, Z = 47)
1.1.6. Iron (Fe, Z = 26)
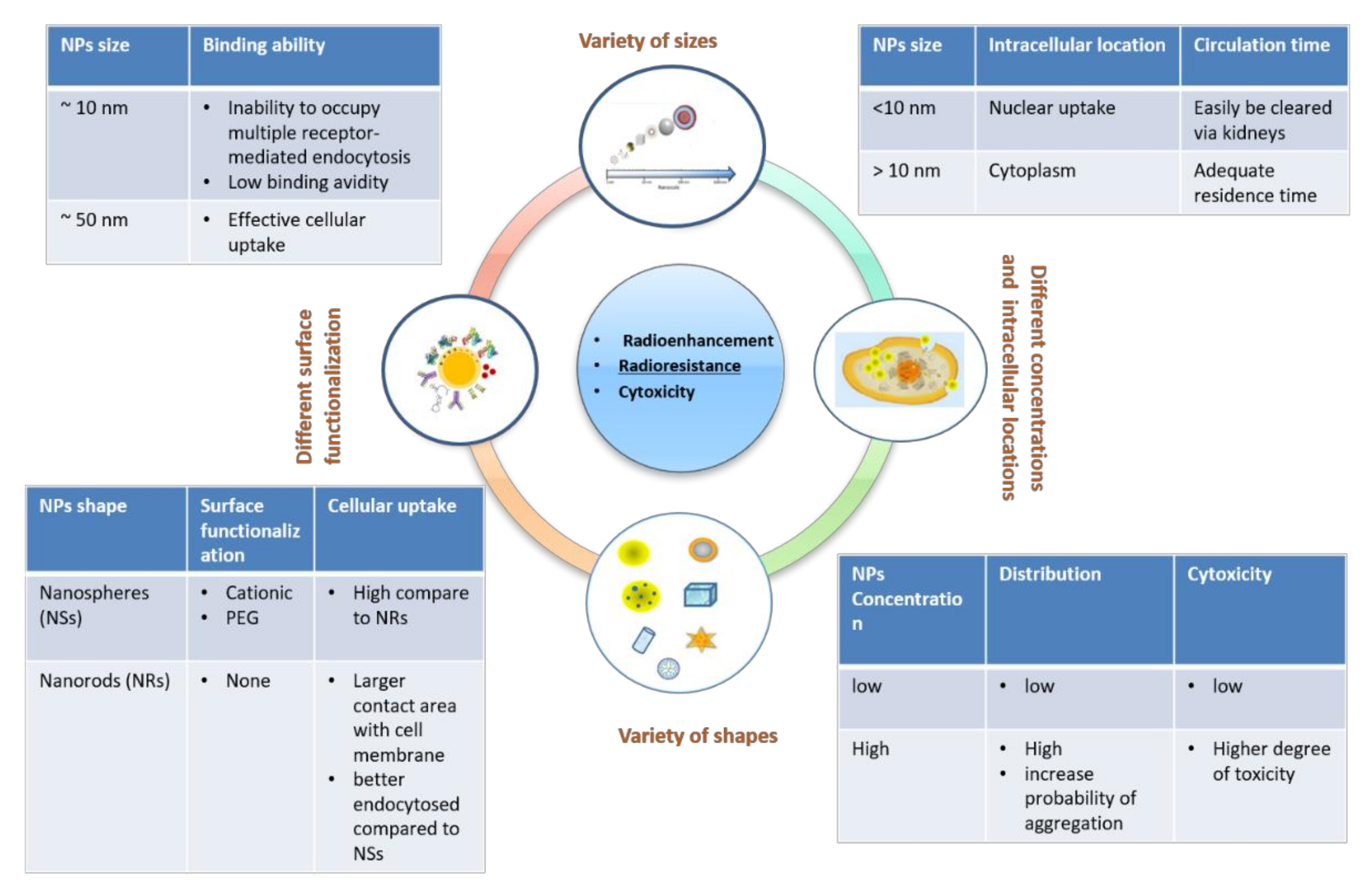
2. Effects of Size, Shapes, and Surface Treatments
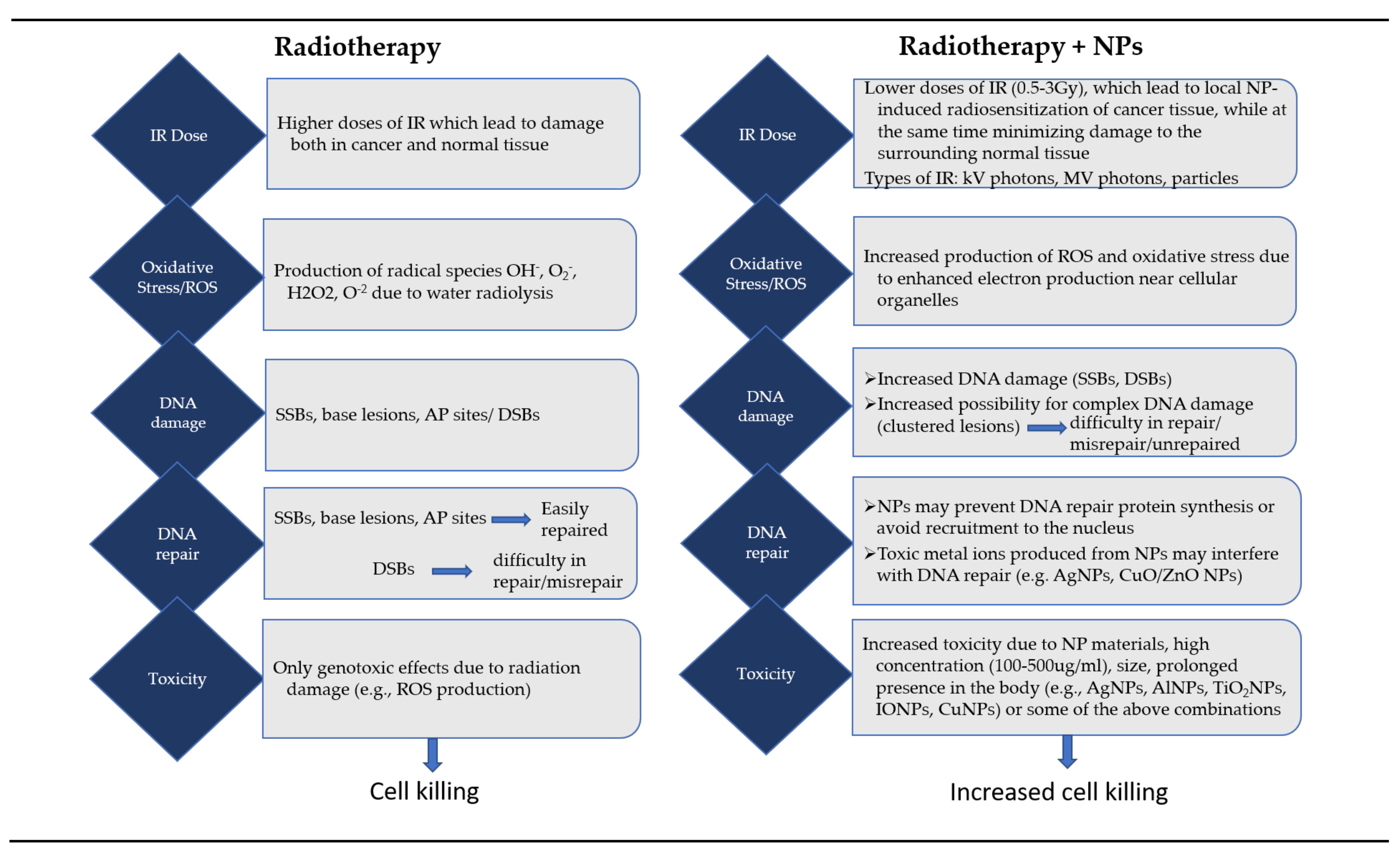
3. Biological/Biochemical Effects
3.1. Nanotoxicity
3.2. Cell Uptake and Localization
3.3. Oxidative Stress
3.4. Effect on Cell Cycle
3.5. Effect on DNA Damage
3.6. Effect on Apoptosis, Autophagy and Senescence
4. Exp. Procedures to Study the Biological Effects of NPs’ Radiosensitization
- Transmission electron microscopy (TEM): TEM can be used to monitor the cellular uptake of NPs and it can be used to detect DNA damage after IR in the presence and absence of NPs. Sample preparations for TEM analysis consists of several stages. After IR, the basic steps for cell or tissue preparation are the following: fixation in aldehyde buffer solution, embedding in gelatin, post fixation in osmium tetroxide, dehydration in ethanol, and embedding in resin (epoxy or acrylic). The choice of fixative and resin depends on the aim of the study (ultrastructural or Immunocyto-histochemical). The chemicals used for these stages such as the aldehyde solution for fixation are adequate to simultaneously preserve the ultrastructure morphology of cell organelles and the antigenicity. However, all chemical preparation stages, if not applied correctly, may lead to image artifacts. For a more detailed information, the reader can resort to a recent technical note about all the technicalities regarding TEM [162]. There exist several other techniques to study DNA damage, e.g., Immunocytochemistry and Gel Electrophoresis, which are considerably more timesaving, require fewer demanding skills and protocols, and are less expensive. However, for detection of type and location of the DNA damage, TEM is unique since the details of the damage cannot be detected in such magnification and resolution (nm scale) with any other method. One disadvantage with TEM is the difficulty in quantifying the damage. Usually, only 10–50 cells are analyzed each time, so the method provides better quality than other methods but cannot accurately quantify the results. However, the use of TEM was performed for the detection of both single and double staining to detect clustered DNA damage and especially DSBs after IR [163,164]. TEM uses normally the immunogold-labelling technique to characterize the DNA damage. Primary antibodies target specific repair proteins are localized by secondary Au-conjugated antibodies, similar to immunocytochemistry methodology. TEM was not used to study DNA damage induced NP radiosensitization until now, but it was used to monitor cellular uptake and distribution of NPs [77,105,165]. Moreover, recently, the technical use of TEM was thoroughly described as a means of studying NP-induced radiosensitization in vitro [162].
- Flow cytometry (FC): Flow cytometry can be used for DNA damage detection and cell cycle analysis. Propidium iodide is the most commonly used dye to quantitatively assess DNA content, and it is a very useful technique to study different checkpoints throughout the cell cycle [119]. Though flow cytometry is broadly used in radiation experiments, cell cycle analysis related to DNA damage-induced NPs radiosensitization was not performed until now. For instance, G2/M checkpoint prevents cells with damaged DNA from entering Mitosis. Phosphorylation at Ser10 of histone H3 is tightly correlated with chromosome condensation during mitosis [166]. An antibody that specifically recognizes the phosphorylated form of histone H3 (p-histone H3 Ser10) is used to identify mitotic cells. Cells are co-stained with anti-p-histone H3 Ser 10 antibody and propidium iodine to distinguish mitotic cells from G2 cells [167]. Milner et al., used flow cytometry on CHO cells to access DNA damage after irradiation with 60Co γ-source [168]. Nucleotides were extracted from cells, then they were stained with the fluorescent dye ethidium bromide and then exposed to laser light within FC. Flow cytometry can be also used to monitor NP uptake. For example, Shapero et al. [169] used flow cytometry to investigate cellular uptake and final localization of silica NPs of different sizes (50, 100, and 300 nm) inside A549 cells as a function of time. They showed that the uptake rate of silica NPs decreases with size; however, due to fluorescent intensity of NPs they suggested that results might be misleading if are not normalized.
- Immunofluorescence: Immunofluorescence uses primary antibodies, labelled with fluorescent secondary antibodies for visualization, specific to targeted DNA repair enzymes. The most common target for DNA damage detection, such as DSBs, is the phosphorylated histone γ-H2AX (phosphorylation at serine 139). Foci represent the DSBs in a 1:1 manner and are used as a DNA damage biomarker. Other DNA repair markers include RAD51, 53BP1 (p53-binding protein 1), phospho-p53, and PARP1 [170]. However, the specificity whether such DSB markers recognize only DSBs is a controversial issue. γH2AX also appears in SSB sites [171]. Beside repair enzymes, primary antibodies can be used to label the damage itself. In this case, the antibodies bind to specific DNA lesions such as 8-oxo-dG, which is used to detect base lesions [172]. Recently electrophoresis-based DNA fractionation methods were used to quantify DNA damage. Detection and quantification of γ-H2AX and 53BP1 is very often employed to assess metallic NP-induced radiosensitization [77,173].
- Agarose Gel Electrophoresis (AGE): DNA lesions can be identified through gel electrophoresis. This is a fast method that quantifies the average density of breaks and a variety of DNA lesions in nanogram quantities. AGE can be divided into two main groups: 1) Alkaline Gel Electrophoresis and 2) Glyoxal gel electrophoresis. Agarose gels can separate a mixture of molecules such as DNA fragments between 50 bp (3% agarose) and 500.000 bp (0.1% agarose) in an agarose matrix, with suitable electrophoresis buffers [174]. The gels are stained with ethidium bromide and the image acquisition of the DNA migration is performed with UV light. Alkaline agarose gels are mostly used for single stranded DNA but it is also used for others alkali-labile lesions as well such as 8-oxoguanine base lesions [175]. Glyoxal agarose gel electrophoresis fractionizes DNA the same way, but it also keeps alkali-labile sites intact.
- Pulsed-field gel electrophoresis (PFGE) and Comet Assay (CA): Methods such as the comet assay and pulsed-field gel electrophoresis (PFGE) [176,177] are based on the detection of DNA fragments by electrophoresis. In comet assay, the cells are embedded in agarose gel on a glass slide for microscopy and the DNA fragments are fractionated by electrophoresis. Because of the tail-like images, this method is called comet assay. In PFGE, cells are embedded in agarose gel, called plugs, and then the cells are lysed in these agarose plugs. Only the DNA fragments such as DSBs migrate in the gel, while the undamaged DNA remain. The CA does not need a large number of cells for the analysis, and it is not expensive, but it cannot distinguish the lengths of the DNA fragments. PFGE is there for better to be used for quantitative purposes. Gel electrophoresis was already used in several studies concerning metallic NP (especially AuNPs) radiosensitization [178,179].
- Clonogenic Survival Assay (CSA): One gold standard technique to access cell death and survival rate in radiobiology is CSA. After a stress induced situation, such as IR, survival assay determines the ability of cells to proliferate and form colonies after a few days’ incubation. Cells are seeded into petri dishes, treated with NPs, irradiated and then replated in low seeding densities and left to grow colonies for a few days (depending on the cell line). After these days, the colonies are stained with crystal violet in 80–100% methanol [180]. The individual colonies are counted and then a survival curve can be defined as a relationship between the radiation dose and the fraction of cells that were able to replicate and form colonies. Clonogenic assay is almost always used to compare cell radiosensitization in the presence and absence of metallic NPs [26,179,181].
- TUNEL Assay: TUNEL (terminal deoxynucleotidyl transferase dUTP nick end labeling) staining, also called the TUNEL Assay, detects the DNA breaks formed when DNA fragmentation occurs in the last phase of apoptosis [182]. TdT can label blunt ends of double-stranded DNA breaks as it attaches to deoxynucleotides to the 3′-hydroxyl terminus of DNA breaks. The nucleotides attached by TdT are stained with a fluorescent dye. Tunnel assay is an alternative assay to agarose gel electrophoreses to analyze the formation of DNA fragments during apoptosis. Teraoka et al., used this assay to access apoptotic cells by counting TUNEL-positive cells [156].
- Immunoblotting/Western blotting: Immunoblotting, or western blotting, is used to identify changes in protein expression following treatment. In this assay, protein expression is sampled at different time-points following X-ray irradiation to determine how pretreatment with NPs enhances the radiation sensitivity. This is performed both in the presence and absence of NPs [123]. This assay uses protein expression levels in different time points after radiation. Primary antibodies can be selected according to selected interest such as apoptosis, DNA damage and repair and oxidative stress.
- Immunocyto/histochemistry for Cellular Senescence detection: There is a number of senescence biomarkers. The most common markers are SA-β-Gal, senescence-associated secretory phenotype (SASP) [183], cell cycle inhibitors p16Ink4a and p21Cip1 [184], and lipofuscin [185]. Oxidative stress is one of the possible reasons leading to senescence. Irradiation alone can lead to increased senescent phenotype [186]. Since ROS production can be responsible for NP-induced radiosensitization, it is essential to also study whether or not irradiated cells incubated with NPs lead to increased senescence compared to the irradiated alone. Until now there are only few groups who address senescence [173,187].
5. Clinicals Trials
5.1. The Use of NPs in Radiotherapy
5.2. Limitations and Challenges in Using Nanoparticles in Clinic
6. Discussion
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Castillo, M.H.; Button, T.M.; Doerr, R.; Homs, M.I.; Pruett, C.W.; Pearce, J.I. Effects of radiotherapy on mandibular reconstruction plates. Am. J. Surg. 1988, 156, 261–263. [Google Scholar] [CrossRef]
- Allal, A.S.; Richter, M.; Russo, M.; Rouzaud, M.; Dulguerov, P.; Kurtz, J.M. Dose variation at bone/titanium interfaces using titanium hollow screw osseointegrating reconstruction plates. Int. J. Radiat. Oncol. 1998, 40, 215–219. [Google Scholar] [CrossRef]
- Mirkin, C.A.; Meade, T.J.; Petrosko, S.H.; Stegh, A.H. Nanotechnology-Based Precision Tools for the Detection and Treatment of Cancer; Springer International Publishing: Cham, Switzerland, 2020; Volume 166. [Google Scholar]
- Reft, C.; Alecu, R.; Das, I.J.; Gerbi, B.J.; Keall, P.; Lief, E.; Mijnheer, B.J.; Papanikolaou, N.; Sibata, C.; Van Dyk, J. Dosimetric considerations for patients with HIP prostheses undergoing pelvic irradiation. Report of the AAPM Radiation Therapy Committee Task Group 63. Med. Phys. 2003, 30, 1162–1182. [Google Scholar] [CrossRef]
- Rousselle, A.; Amelot, A.; Thariat, J.; Jacob, J.; Mercy, G.; De Marzi, L.; Feuvret, L. Metallic implants and CT artefacts in the CTV area: Where are we in 2020? Cancer Radiother. 2020, 24, 658–666. [Google Scholar] [CrossRef]
- Ceresa, C.; Bravin, A.; Cavaletti, G.; Pellei, M.; Santini, C. The Combined Therapeutical Effect of Metal-based Drugs and Radiation Therapy: The Present Status of Research. Curr. Med. Chem. 2014, 21, 2237–2265. [Google Scholar] [CrossRef]
- Amato, E.; Italiano, A.; Leotta, S.; Pergolizzi, S.; Torrisi, L. Monte Carlo study of the dose enhancement effect of gold nanoparticles during X-ray therapies and evaluation of the anti-angiogenic effect on tumour capillary vessels. J. X-ray Sci. Technol. 2013, 21, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, T. Radiotherapy Improvements by Using Au Nanoparticles. Recent Patents Nanotechnol. 2015, 9, 114–125. [Google Scholar] [CrossRef]
- Choi, J.; Kim, G.; Cho, S.B.; Im, H.-J. Radiosensitizing high-Z metal nanoparticles for enhanced radiotherapy of glioblastoma multiforme. J. Nanobiotechnol. 2020, 18, 122. [Google Scholar] [CrossRef]
- Kuncic, Z.; Lacombe, S. Nanoparticle radio-enhancement: Principles, progress and application to cancer treatment. Phys. Med. Biol. 2018, 63, 02tr01. [Google Scholar] [CrossRef]
- Tamanoi, F.; Matsumoto, K.; Doan, T.L.H.; Shiro, A.; Saitoh, H. Studies on the Exposure of Gadolinium Containing Nanoparticles with Monochromatic X-rays Drive Advances in Radiation Therapy. Nanomaterials (Basel) 2020, 10, 1341. [Google Scholar] [CrossRef] [PubMed]
- Dalzon, B.; Guidetti, M.; Testemale, D.; Reymond, S.; Proux, O.; Vollaire, J.; Collin-Faure, V.; Testard, I.; Fenel, D.; Schoehn, G.; et al. Utility of macrophages in an antitumor strategy based on the vectorization of iron oxide nanoparticles. Nanoscale 2019, 11, 9341–9352. [Google Scholar] [CrossRef] [Green Version]
- Matsumoto, K.; Saitoh, H.; Doan, T.L.H.; Shiro, A.; Nakai, K.; Komatsu, A.; Tsujimoto, M.; Yasuda, R.; Kawachi, T.; Tajima, T.; et al. Destruction of tumor mass by gadolinium-loaded nanoparticles irradiated with monochromatic X-rays: Implications for the Auger therapy. Sci. Rep. 2019, 9, 13275. [Google Scholar] [CrossRef] [PubMed]
- Bulin, A.-L.; Broekgaarden, M.; Chaput, F.; Baisamy, V.; Garrevoet, J.; Busser, B.; Brueckner, D.; Youssef, A.; Ravanat, J.-L.; Dujardin, C.; et al. Radiation Dose-Enhancement Is a Potent Radiotherapeutic Effect of Rare-Earth Composite Nanoscintillators in Preclinical Models of Glioblastoma. Adv. Sci. 2020, 7, 2001675. [Google Scholar] [CrossRef] [PubMed]
- Gagliardi, F.M.; Franich, R.D.; Geso, M. Nanoparticle dose enhancement of synchrotron radiation in PRESAGE dosimeters. J. Synchrotron Radiat. 2020, 27, 1590–1600. [Google Scholar] [CrossRef]
- Schuemann, J.; Berbeco, R.; Chithrani, D.B.; Cho, S.H.; Kumar, R.; McMahon, S.J.; Sridhar, S.; Krishnan, S. Roadmap to Clinical Use of Gold Nanoparticles for Radiation Sensitization. Int. J. Radiat. Oncol. 2016, 94, 189–205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Detappe, A.; Kunjachan, S.; Drané, P.; Kotb, S.; Myronakis, M.; Biancur, D.E.; Ireland, T.; Wagar, M.; Lux, F.; Tillement, O.; et al. Key clinical beam parameters for nanoparticle-mediated radiation dose amplification. Scientific Reports 2016, 6, 34040. [Google Scholar] [CrossRef] [PubMed]
- Tsiamas, P.; Liu, B.; Cifter, F.; Ngwa, W.F.; Berbeco, R.I.; Kappas, C.; Theodorou, K.; Marcus, K.; Makrigiorgos, M.G.; Sajo, E.; et al. Impact of beam quality on megavoltage radiotherapy treatment techniques utilizing gold nanoparticles for dose enhancement. Phys. Med. Biol. 2013, 58, 451–464. [Google Scholar] [CrossRef] [PubMed]
- Lechtman, E.; Mashouf, S.; Chattopadhyay, N.; Keller, B.M.; Lai, P.; Cai, Z.; Reilly, R.M.; Pignol, J.P. A Monte Carlo-based model of gold nanoparticle radiosensitization accounting for increased radiobiological effectiveness. Phys. Med. Biol. 2013, 58, 3075–3087. [Google Scholar] [CrossRef] [PubMed]
- McMahon, S.J.; Hyland, W.B.; Muir, M.F.; Coulter, J.A.; Jain, S.; Butterworth, K.T.; Schettino, G.; Dickson, G.R.; Hounsell, A.R.; O’Sullivan, J.M.; et al. Biological consequences of nanoscale energy deposition near irradiated heavy atom nanoparticles. Sci. Rep. 2011, 1, 18. [Google Scholar] [CrossRef] [Green Version]
- Chithrani, D.B.; Jelveh, S.; Jalali, F.; Van Prooijen, M.; Allen, C.; Bristow, R.G.; Hill, R.P.; Jaffray, D.A. Gold nanoparticles as radiation sensitizers in cancer therapy. Radiat. Res. 2010, 173, 719–728. [Google Scholar] [CrossRef]
- Rudek, B.; McNamara, A.; Ramos-Méndez, J.; Byrne, H.; Kuncic, Z.; Schuemann, J. Radio-enhancement by gold nanoparticles and their impact on water radiolysis for x-ray, proton and carbon-ion beams. Phys. Med. Biol. 2019, 64, 175005. [Google Scholar] [CrossRef]
- Seo, S.J.; Han, S.M.; Cho, J.H.; Hyodo, K.; Zaboronok, A.; You, H.; Peach, K.; Hill, M.A.; Kim, J.K. Enhanced production of reactive oxygen species by gadolinium oxide nanoparticles under core-inner-shell excitation by proton or monochromatic X-ray irradiation: Implication of the contribution from the interatomic de-excitation-mediated nanoradiator effect to dose enhancement. Radiat. Environ. Biophys. 2015, 54, 423–431. [Google Scholar] [CrossRef]
- Averback, R.S.; De La Rubia, T.D. Displacement Damage in Irradiated Metals and Semiconductors. In Solid State Physics; Ehrenreich, H., Spaepen, F., Eds.; Academic Press: Cambridge, MA, USA, 1998; Volume 51, pp. 281–402. [Google Scholar]
- Toulemonde, M.; Assmann, W.; Trautmann, C.; Grüner, F.; Mieskes, H.D.; Kucal, H.; Wang, Z.G. Electronic sputtering of metals and insulators by swift heavy ions. Nucl. Instrum. Methods Physi. Res. Sect. B Beam Interact. Mater. Atoms 2003, 212, 346–357. [Google Scholar] [CrossRef]
- Paro, A.D.; Shanmugam, I.; van de Ven, A.L. Nanoparticle-Mediated X-Ray Radiation Enhancement for Cancer Therapy. Methods Mol. Biol. 2017, 1530, 391–401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hashemi, S.; Aghamiri, M.R.; Kahani, M.; Jaberi, R. Investigation of gold nanoparticle effects in brachytherapy by an electron emitter ophthalmic plaque. Int. J. Nanomed. 2019, 14, 4157–4165. [Google Scholar] [CrossRef] [Green Version]
- Ngwa, W.; Boateng, F.; Kumar, R.; Irvine, D.J.; Formenti, S.; Ngoma, T.; Herskind, C.; Veldwijk, M.R.; Hildenbrand, G.L.; Hausmann, M.; et al. Smart Radiation Therapy Biomaterials. Int. J. Radiat. Oncol. 2017, 97, 624–637. [Google Scholar] [CrossRef] [Green Version]
- Kwatra, D.; Venugopal, A.; Anant, S. Nanoparticles in radiation therapy: A summary of various approaches to enhance radiosensitization in cancer. Transl. Cancer Res. 2013, 2, 330–342. [Google Scholar]
- Altundal, Y.; Cifter, G.; Detappe, A.; Sajo, E.; Tsiamas, P.; Zygmanski, P.; Berbeco, R.; Cormack, R.A.; Makrigiorgos, M.; Ngwa, W. New potential for enhancing concomitant chemoradiotherapy with FDA approved concentrations of cisplatin via the photoelectric effect. Phys. Med. Eur. J. Med. Phys. 2015, 31, 25–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cifter, G.; Altundal, Y.; Detappe, A.; Sajo, E.; Berbeco, R.; Makrigiorgos, M.; Ngwa, W. Dose enhancement during concomitant chemoradiotherapy using FDA approved concentrations of carboplatin and oxaliplatin nanoparticles. In Proceedings of the World Congress on Medical Physics and Biomedical Engineering, Toronto, ON, Canada, 7–12 June 2015; pp. 1723–1726. [Google Scholar]
- Danhier, F.; Feron, O.; Préat, V. To exploit the tumor microenvironment: Passive and active tumor targeting of nanocarriers for anti-cancer drug delivery. J. Control. Release 2010, 148, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Bamrungsap, S.; Zhao, Z.; Chen, T.; Wang, L.; Li, C.; Fu, T.; Tan, W. Nanotechnology in therapeutics: A focus on nanoparticles as a drug delivery system. Nanomedicine 2012, 7, 1253–1271. [Google Scholar] [CrossRef] [PubMed]
- Cappellano, G.; Comi, C.; Chiocchetti, A.; Dianzani, U. Exploiting PLGA-Based Biocompatible Nanoparticles for Next-Generation Tolerogenic Vaccines against Autoimmune Disease. Int. J. Mol. Sci. 2019, 20, 204. [Google Scholar] [CrossRef] [Green Version]
- Taratula, O.; Garbuzenko, O.B.; Chen, A.M.; Minko, T. Innovative strategy for treatment of lung cancer: Targeted nanotechnology-based inhalation co-delivery of anticancer drugs and siRNA. J. Drug Target. 2011, 19, 900–914. [Google Scholar] [CrossRef]
- Hao, Y.; Altundal, Y.; Moreau, M.; Sajo, E.; Kumar, R.; Ngwa, W. Potential for enhancing external beam radiotherapy for lung cancer using high-Z nanoparticles administered via inhalation. Phys. Med. Biol. 2015, 60, 7035–7043. [Google Scholar] [CrossRef] [Green Version]
- Lao, L.L.; Peppas, N.A.; Boey, F.Y.C.; Venkatraman, S.S. Modeling of drug release from bulk-degrading polymers. Int. J. Pharm. 2011, 418, 28–41. [Google Scholar] [CrossRef]
- Wolinsky, J.B.; Colson, Y.L.; Grinstaff, M.W. Local drug delivery strategies for cancer treatment: Gels, nanoparticles, polymeric films, rods, and wafers. J. Control. Release 2012, 159, 14–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- A matter of scale. Nat. Nanotechnol. 2016, 11, 733. [CrossRef] [PubMed] [Green Version]
- Meng, X.; Li, X. Size Limit and Energy Analysis of Nanoparticles during Wrapping Process by Membrane. Nanomaterials (Basel) 2018, 8, 899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coradeghini, R.; Gioria, S.; García, C.P.; Nativo, P.; Franchini, F.; Gilliland, D.; Ponti, J.; Rossi, F. Size-dependent toxicity and cell interaction mechanisms of gold nanoparticles on mouse fibroblasts. Toxicol. Lett. 2013, 217, 205–216. [Google Scholar] [CrossRef]
- Bahadar, H.; Maqbool, F.; Niaz, K.; Abdollahi, M. Toxicity of Nanoparticles and an Overview of Current Experimental Models. Iran Biomed. J. 2016, 20, 1–11. [Google Scholar] [CrossRef]
- Frank, D.; Tyagi, C.; Tomar, L.; Choonara, Y.E.; du Toit, L.C.; Kumar, P.; Penny, C.; Pillay, V. Overview of the role of nanotechnological innovations in the detection and treatment of solid tumors. Int. J. Nanomed. 2014, 9, 589–613. [Google Scholar] [CrossRef] [Green Version]
- Sanità, G.; Carrese, B.; Lamberti, A. Nanoparticle Surface Functionalization: How to Improve Biocompatibility and Cellular Internalization. Front. Mol. Biosci. 2020, 7. [Google Scholar] [CrossRef]
- Cui, L.; Tse, K.; Zahedi, P.; Harding, S.M.; Zafarana, G.; Jaffray, D.A.; Bristow, R.G.; Allen, C. Hypoxia and cellular localization influence the radiosensitizing effect of gold nanoparticles (AuNPs) in breast cancer cells. Radiat. Res. 2014, 182, 475–488. [Google Scholar] [CrossRef] [PubMed]
- McNamara, A.L.; Kam, W.W.Y.; Scales, N.; McMahon, S.J.; Bennett, J.W.; Byrne, H.L.; Schuemann, J.; Paganetti, H.; Banati, R.; Kuncic, Z. Dose enhancement effects to the nucleus and mitochondria from gold nanoparticles in the cytosol. Phys. Med. Biol. 2016, 61, 5993–6010. [Google Scholar] [CrossRef] [Green Version]
- Dykman, L.A.; Khlebtsov, N.G. Uptake of Engineered Gold Nanoparticles into Mammalian Cells. Chem. Rev. 2014, 114, 1258–1288. [Google Scholar] [CrossRef]
- Su, X.Y.; Liu, P.D.; Wu, H.; Gu, N. Enhancement of radiosensitization by metal-based nanoparticles in cancer radiation therapy. Cancer Biol. Med. 2014, 11, 86–91. [Google Scholar] [CrossRef]
- Hossain, M.; Su, M. Nanoparticle location and material dependent dose enhancement in X-ray radiation therapy. J. Phys. Chem. C Nanomater. Interfaces 2012, 116, 23047–23052. [Google Scholar] [CrossRef] [Green Version]
- Schürmann, R.; Vogel, S.; Ebel, K.; Bald, I. The Physico-Chemical Basis of DNA Radiosensitization: Implications for Cancer Radiation Therapy. Chemistry 2018, 24, 10271–10279. [Google Scholar] [CrossRef]
- Chithrani, B.D.; Ghazani, A.A.; Chan, W.C.W. Determining the Size and Shape Dependence of Gold Nanoparticle Uptake into Mammalian Cells. Nano Lett. 2006, 6, 662–668. [Google Scholar] [CrossRef]
- Li, F.; Li, Z.; Jin, X.; Liu, Y.; Li, P.; Shen, Z.; Wu, A.; Zheng, X.; Chen, W.; Li, Q. Radiosensitizing Effect of Gadolinium Oxide Nanocrystals in NSCLC Cells Under Carbon Ion Irradiation. Nanoscale Res. Lett. 2019, 14, 328. [Google Scholar] [CrossRef] [Green Version]
- Sah, B.; Antosh, M.P. Effect of size on gold nanoparticles in radiation therapy: Uptake and survival effects. J. Nanomed. 2019, 2, 1013. [Google Scholar]
- Torrisi, L.; Davidkova, M.; Havranek, V.; Cutroneo, M.; Torrisi, A. Physical study of proton therapy at CANAM laboratory on medulloblastoma cell lines DAOY. Radiat. Eff. Defects Solids 2020, 175, 863–878. [Google Scholar] [CrossRef]
- Johnstone, T.C.; Suntharalingam, K.; Lippard, S.J. The Next Generation of Platinum Drugs: Targeted Pt(II) Agents, Nanoparticle Delivery, and Pt(IV) Prodrugs. Chem. Rev. 2016, 116, 3436–3486. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Chen, L.; Huang, C.; Huang, Y.; Jia, N. Albumin-mediated platinum nanocrystals for in vivo enhanced computed tomography imaging. J. Mater. Chem. B 2017, 5, 3498–3510. [Google Scholar] [CrossRef] [PubMed]
- Gatto, F.; Moglianetti, M.; Pompa, P.P.; Bardi, G. Platinum Nanoparticles Decrease Reactive Oxygen Species and Modulate Gene Expression without Alteration of Immune Responses in THP-1 Monocytes. Nanomaterials 2018, 8, 392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Porcel, E.; Liehn, S.; Remita, H.; Usami, N.; Kobayashi, K.; Furusawa, Y.; Le Sech, C.; Lacombe, S. Platinum nanoparticles: A promising material for future cancer therapy? Nanotechnology 2010, 21, 85103. [Google Scholar] [CrossRef]
- Li, S.; Porcel, E.; Remita, H.; Marco, S.; Réfrégiers, M.; Dutertre, M.; Confalonieri, F.; Lacombe, S. Platinum nanoparticles: An exquisite tool to overcome radioresistance. Cancer Nanotechnol. 2017, 8, 4. [Google Scholar] [CrossRef]
- Buchtelova, H.; Dostalova, S.; Michalek, P.; Krizkova, S.; Strmiska, V.; Kopel, P.; Hynek, D.; Richtera, L.; Ridoskova, A.; Adam, P.; et al. Size-related cytotoxicological aspects of polyvinylpyrrolidone-capped platinum nanoparticles. Food Chem. Toxicol. 2017, 105, 337–346. [Google Scholar] [CrossRef]
- Li, Y.; Yun, K.H.; Lee, H.; Goh, S.H.; Suh, Y.G.; Choi, Y. Porous platinum nanoparticles as a high-Z and oxygen generating nanozyme for enhanced radiotherapy in vivo. Biomaterials 2019, 197, 12–19. [Google Scholar] [CrossRef]
- Muhammad Afiq, K.A.; Rashid, R.A.; Lazim, R.M.; Dollah, N.; Razak, K.A.; Rahman, W.N. Evaluation of radiosensitization effects by platinum nanodendrites for 6 MV photon beam radiotherapy. Radiat. Phys. Chem. 2018, 150, 40–45. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, P.; Li, F.; Jin, X.; Li, J.; Chen, W.; Li, Q. Metal-based NanoEnhancers for Future Radiotherapy: Radiosensitizing and Synergistic Effects on Tumor Cells. Theranostics 2018, 8, 1824–1849. [Google Scholar] [CrossRef]
- Maggiorella, L.; Barouch, G.; Devaux, C.; Pottier, A.; Deutsch, E.; Bourhis, J.; Borghi, E.; Levy, L. Nanoscale radiotherapy with hafnium oxide nanoparticles. Futur. Oncol. 2012, 8, 1167–1181. [Google Scholar] [CrossRef] [PubMed]
- Nanobiotix. Available online: https://www.nanobiotix.com/ (accessed on 28 April 2021).
- Bonvalot, S.; Rutkowski, P.L.; Thariat, J.; Carrère, S.; Ducassou, A.; Sunyach, M.P.; Agoston, P.; Hong, A.; Mervoyer, A.; Rastrelli, M.; et al. NBTXR3, a first-in-class radioenhancer hafnium oxide nanoparticle, plus radiotherapy versus radiotherapy alone in patients with locally advanced soft-tissue sarcoma (Act.In.Sarc): A multicentre, phase 2–3, randomised, controlled trial. Lancet Oncol. 2019, 20, 1148–1159. [Google Scholar] [CrossRef]
- Pottier, A.; Borghi, E.; Levy, L. Metals as radio-enhancers in oncology: The industry perspective. Biochem. Biophys. Res. Commun. 2015, 468, 471–475. [Google Scholar] [CrossRef]
- Verry, C.; Sancey, L.; Dufort, S.; Le Duc, G.; Mendoza, C.; Lux, F.; Grand, S.; Arnaud, J.; Quesada, J.L.; Villa, J.; et al. Treatment of multiple brain metastases using gadolinium nanoparticles and radiotherapy: NANO-RAD, a phase I study protocol. BMJ Open 2019, 9, e023591. [Google Scholar] [CrossRef]
- Coulter, J.A.; Hyland, W.B.; Nicol, J.; Currell, F.J. Radiosensitising Nanoparticles as Novel Cancer Therapeutics—Pipe Dream or Realistic Prospect? Clin. Oncol. 2013, 25, 593–603. [Google Scholar] [CrossRef] [Green Version]
- Xu, L.; Wang, Y.-Y.; Huang, J.; Chen, C.-Y.; Wang, Z.-X.; Xie, H. Silver nanoparticles: Synthesis, medical applications and biosafety. Theranostics 2020, 10, 8996–9031. [Google Scholar] [CrossRef]
- Sun, C.; Lee, J.S.; Zhang, M. Magnetic nanoparticles in MR imaging and drug delivery. Adv. Drug Deliv. Rev. 2008, 60, 1252–1265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Datta, N.R.; Krishnan, S.; Speiser, D.E.; Neufeld, E.; Kuster, N.; Bodis, S.; Hofmann, H. Magnetic nanoparticle-induced hyperthermia with appropriate payloads: Paul Ehrlich’s “magic (nano)bullet” for cancer theranostics? Cancer Treat. Rev. 2016, 50, 217–227. [Google Scholar] [CrossRef]
- Bergs, J.W.; Wacker, M.G.; Hehlgans, S.; Piiper, A.; Multhoff, G.; Rödel, C.; Rödel, F. The role of recent nanotechnology in enhancing the efficacy of radiation therapy. Biochim. Biophys. Acta 2015, 1856, 130–143. [Google Scholar] [CrossRef]
- Huo, S.; Jin, S.; Ma, X.; Xue, X.; Yang, K.; Kumar, A.; Wang, P.C.; Zhang, J.; Hu, Z.; Liang, X.-J. Ultrasmall gold nanoparticles as carriers for nucleus-based gene therapy due to size-dependent nuclear entry. ACS Nano 2014, 8, 5852–5862. [Google Scholar] [CrossRef]
- Jiang, W.; Kim, B.Y.S.; Rutka, J.T.; Chan, W.C.W. Nanoparticle-mediated cellular response is size-dependent. Nat. Nanotechnol. 2008, 3, 145–150. [Google Scholar] [CrossRef]
- Chithrani, D.B.; Dunne, M.; Stewart, J.; Allen, C.; Jaffray, D.A. Cellular uptake and transport of gold nanoparticles incorporated in a liposomal carrier. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Pagáčová, E.; Štefančíková, L.; Schmidt-Kaler, F.; Hildenbrand, G.; Vičar, T.; Depeš, D.; Lee, J.-H.; Bestvater, F.; Lacombe, S.; Porcel, E.; et al. Challenges and Contradictions of Metal Nano-Particle Applications for Radio-Sensitivity Enhancement in Cancer Therapy. Inter. J. Mol. Sci. 2019, 20, 588. [Google Scholar] [CrossRef] [Green Version]
- Chithrani, B.D.; Chan, W.C.W. Elucidating the Mechanism of Cellular Uptake and Removal of Protein-Coated Gold Nanoparticles of Different Sizes and Shapes. Nano Lett. 2007, 7, 1542–1550. [Google Scholar] [CrossRef]
- Parakhonskiy, B.; Zyuzin, M.V.; Yashchenok, A.; Carregal-Romero, S.; Rejman, J.; Möhwald, H.; Parak, W.J.; Skirtach, A.G. The influence of the size and aspect ratio of anisotropic, porous CaCO3 particles on their uptake by cells. J. Nanobiotechnol. 2015, 13, 53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Kröger, M.; Liu, W.K. Shape effect in cellular uptake of PEGylated nanoparticles: Comparison between sphere, rod, cube and disk. Nanoscale 2015, 7, 16631–16646. [Google Scholar] [CrossRef]
- Herd, H.; Daum, N.; Jones, A.T.; Huwer, H.; Ghandehari, H.; Lehr, C.-M. Nanoparticle Geometry and Surface Orientation Influence Mode of Cellular Uptake. ACS Nano 2013, 7, 1961–1973. [Google Scholar] [CrossRef] [Green Version]
- Behzadi, S.; Serpooshan, V.; Tao, W.; Hamaly, M.A.; Alkawareek, M.Y.; Dreaden, E.C.; Brown, D.; Alkilany, A.M.; Farokhzad, O.C.; Mahmoudi, M. Cellular uptake of nanoparticles: Journey inside the cell. Chem. Soc. Rev. 2017, 46, 4218–4244. [Google Scholar] [CrossRef]
- Lin, J.; Zhang, H.; Chen, Z.; Zheng, Y. Penetration of Lipid Membranes by Gold Nanoparticles: Insights into Cellular Uptake, Cytotoxicity, and Their Relationship. ACS Nano 2010, 4, 5421–5429. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.C.; Xie, J.; Wurm, P.A.; Xia, Y. Understanding the Role of Surface Charges in Cellular Adsorption versus Internalization by Selectively Removing Gold Nanoparticles on the Cell Surface with a I2/KI Etchant. Nano Lett. 2009, 9, 1080–1084. [Google Scholar] [CrossRef]
- Pan, L.; Liu, J.; Shi, J. Cancer cell nucleus-targeting nanocomposites for advanced tumor therapeutics. Chem. Soc. Rev. 2018, 47, 6930–6946. [Google Scholar] [CrossRef] [PubMed]
- Kang, B.; Mackey, M.A.; El-Sayed, M.A. Nuclear Targeting of Gold Nanoparticles in Cancer Cells Induces DNA Damage, Causing Cytokinesis Arrest and Apoptosis. J. Am. Chem. Soc. 2010, 132, 1517–1519. [Google Scholar] [CrossRef]
- Celina, J.Y.; Devika, B.C. Nuclear Targeting of Gold Nanoparticles for Improved Therapeutics. Curr. Top. Med. Chem. 2016, 16, 271–280. [Google Scholar] [CrossRef]
- Zhang, D.; Neumann, O.; Wang, H.; Yuwono, V.M.; Barhoumi, A.; Perham, M.; Hartgerink, J.D.; Wittung-Stafshede, P.; Halas, N.J. Gold Nanoparticles Can Induce the Formation of Protein-based Aggregates at Physiological pH. Nano Lett. 2009, 9, 666–671. [Google Scholar] [CrossRef]
- Lechtman, E.; Pignol, J.-P. Interplay between the gold nanoparticle sub-cellular localization, size, and the photon energy for radiosensitization. Scientific Reports 2017, 7, 13268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Butterworth, K.T.; McMahon, S.J.; Taggart, L.E.; Prise, K.M. Radiosensitization by gold nanoparticles: Effective at megavoltage energies and potential role of oxidative stress. Transl. Cancer Res. 2013, 2, 269–279. [Google Scholar]
- Sun, H.; Wang, X.; Zhai, S. The Rational Design and Biological Mechanisms of Nanoradiosensitizers. Nanomaterials 2020, 10, 504. [Google Scholar] [CrossRef] [Green Version]
- Rosa, S.; Connolly, C.; Schettino, G.; Butterworth, K.T.; Prise, K.M. Biological mechanisms of gold nanoparticle radiosensitization. Cancer Nanotechnol. 2017, 8, 2. [Google Scholar] [CrossRef] [Green Version]
- Yamagishi, Y.; Watari, A.; Hayata, Y.; Li, X.; Kondoh, M.; Yoshioka, Y.; Tsutsumi, Y.; Yagi, K. Acute and chronic nephrotoxicity of platinum nanoparticles in mice. Nanoscale Res. Lett. 2013, 8, 395. [Google Scholar] [CrossRef] [Green Version]
- Pelka, J.; Gehrke, H.; Esselen, M.; Türk, M.; Crone, M.; Bräse, S.; Muller, T.; Blank, H.; Send, W.; Zibat, V.; et al. Cellular uptake of platinum nanoparticles in human colon carcinoma cells and their impact on cellular redox systems and DNA integrity. Chem. Res. Toxicol. 2009, 22, 649–659. [Google Scholar] [CrossRef]
- Zhang, X.-D.; Guo, M.-L.; Wu, H.-Y.; Sun, Y.-M.; Ding, Y.-Q.; Feng, X.; Zhang, L.-A. Irradiation stability and cytotoxicity of gold nanoparticles for radiotherapy. Int. J. Nanomed. 2009, 4, 165–173. [Google Scholar] [CrossRef] [Green Version]
- Connor, E.E.; Mwamuka, J.; Gole, A.; Murphy, C.J.; Wyatt, M.D. Gold Nanoparticles Are Taken Up by Human Cells but Do Not Cause Acute Cytotoxicity. Small 2005, 1, 325–327. [Google Scholar] [CrossRef]
- Ying, E.; Hwang, H.M. In vitro evaluation of the cytotoxicity of iron oxide nanoparticles with different coatings and different sizes in A3 human T lymphocytes. Sci. Total Environ. 2010, 408, 4475–4481. [Google Scholar] [CrossRef]
- Nguyen, K.C.; Seligy, V.L.; Massarsky, A.; Moon, T.W.; Rippstein, P.; Tan, J.; Tayabali, A.F. Comparison of toxicity of uncoated and coated silver nanoparticles. J. Phys. Conf. Ser. 2013, 429, 012025. [Google Scholar] [CrossRef] [Green Version]
- Cho, W.-S.; Cho, M.; Jeong, J.; Choi, M.; Cho, H.-Y.; Han, B.S.; Kim, S.H.; Kim, H.O.; Lim, Y.T.; Chung, B.H.; et al. Acute toxicity and pharmacokinetics of 13 nm-sized PEG-coated gold nanoparticles. Toxicol. Appl. Pharmacol. 2009, 236, 16–24. [Google Scholar] [CrossRef]
- Feng, Z.V.; Gunsolus, I.L.; Qiu, T.A.; Hurley, K.R.; Nyberg, L.H.; Frew, H.; Johnson, K.P.; Vartanian, A.M.; Jacob, L.M.; Lohse, S.E.; et al. Impacts of gold nanoparticle charge and ligand type on surface binding and toxicity to Gram-negative and Gram-positive bacteria. Chem. Sci. 2015, 6, 5186–5196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hillaireau, H.; Couvreur, P. Nanocarriers’ entry into the cell: Relevance to drug delivery. Cell. Mol. Life Sci. 2009, 66, 2873–2896. [Google Scholar] [CrossRef] [PubMed]
- Mao, Z.; Zhou, X.; Gao, C. Influence of structure and properties of colloidal biomaterials on cellular uptake and cell functions. Biomater. Sci. 2013, 1, 896–911. [Google Scholar] [CrossRef]
- Doherty, G.J.; McMahon, H.T. Mechanisms of Endocytosis. Annu. Rev. Biochem. 2009, 78, 857–902. [Google Scholar] [CrossRef] [Green Version]
- Ma, X.; Wu, Y.; Jin, S.; Tian, Y.; Zhang, X.; Zhao, Y.; Yu, L.; Liang, X.-J. Gold Nanoparticles Induce Autophagosome Accumulation through Size-Dependent Nanoparticle Uptake and Lysosome Impairment. ACS Nano 2011, 5, 8629–8639. [Google Scholar] [CrossRef]
- Xie, X.; Liao, J.; Shao, X.; Li, Q.; Lin, Y. The Effect of shape on Cellular Uptake of Gold Nanoparticles in the forms of Stars, Rods, and Triangles. Sci. Rep. 2017, 7, 3827. [Google Scholar] [CrossRef]
- Liu, C.-G.; Han, Y.-H.; Kankala, R.K.; Wang, S.-B.; Chen, A.-Z. Subcellular Performance of Nanoparticles in Cancer Therapy. Int. J. Nanomedicine 2020, 15, 675–704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, S.; Coulter, J.A.; Hounsell, A.R.; Butterworth, K.T.; McMahon, S.J.; Hyland, W.B.; Muir, M.F.; Dickson, G.R.; Prise, K.M.; Currell, F.J.; et al. Cell-Specific Radiosensitization by Gold Nanoparticles at Megavoltage Radiation Energies. Int. J. Radiat. Oncol. Biol. Phys. 2011, 79, 531–539. [Google Scholar] [CrossRef] [Green Version]
- Peckys, D.B.; De Jonge, N. Visualizing Gold Nanoparticle Uptake in Live Cells with Liquid Scanning Transmission Electron Microscopy. Nano Lett. 2011, 11, 1733–1738. [Google Scholar] [CrossRef]
- Tremi, I.; Anagnostopoulos, D.; Spyratou, E.; Gkeka, P.; Georgakilas, A.G.; Chatgilialoglu, C.; Cournia, Z. Effect of 5-trans Isomer of Arachidonic Acid on Model Liposomal Membranes Studied by a Combined Simulation and Experimental Approach. J. Membr. Biol. 2018, 251, 475–489. [Google Scholar] [CrossRef]
- Howard, D.; Sebastian, S.; Le, Q.V.-C.; Thierry, B.; Kempson, I. Chemical Mechanisms of Nanoparticle Radiosensitization and Radioprotection: A Review of Structure-Function Relationships Influencing Reactive Oxygen Species. Int. J. Mol. Sci. 2020, 21, 579. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vilotte, F.; Jumeau, R.; Bourhis, J. High Z nanoparticles and radiotherapy: A critical view. Lancet Oncol. 2019, 20, e557. [Google Scholar] [CrossRef] [Green Version]
- Baldacchino, G.; Brun, E.; Denden, I.; Bouhadoun, S.; Roux, R.; Khodja, H.; Sicard-Roselli, C. Importance of radiolytic reactions during high-LET irradiation modalities: LET effect, role of O2 and radiosensitization by nanoparticles. Cancer Nanotechnol. 2019, 10, 3. [Google Scholar] [CrossRef]
- Sakhrani, N.M.; Padh, H. Organelle targeting: Third level of drug targeting. Drug Des. Dev. Ther. 2013, 7, 585–599. [Google Scholar] [CrossRef] [Green Version]
- Geng, F.; Song, K.; Xing, J.Z.; Yuan, C.; Yan, S.; Yang, Q.; Chen, J.; Kong, B. Thio-glucose bound gold nanoparticles enhance radio-cytotoxic targeting of ovarian cancer. Nanotechnology 2011, 22, 285101. [Google Scholar] [CrossRef]
- Taggart, L.E.; McMahon, S.J.; Currell, F.J.; Prise, K.M.; Butterworth, K.T. The role of mitochondrial function in gold nanoparticle mediated radiosensitisation. Cancer Nanotechnol. 2014, 5, 5. [Google Scholar] [CrossRef] [Green Version]
- Cheng, K.; Sano, M.; Jenkins, C.H.; Zhang, G.; Vernekohl, D.; Zhao, W.; Wei, C.; Zhang, Y.; Zhang, Z.; Liu, Y.; et al. Synergistically Enhancing the Therapeutic Effect of Radiation Therapy with Radiation Activatable and Reactive Oxygen Species-Releasing Nanostructures. ACS Nano 2018, 12, 4946–4958. [Google Scholar] [CrossRef] [PubMed]
- Swanner, J.; Mims, J.; Carroll, D.L.; Akman, S.A.; Furdui, C.M.; Torti, S.V.; Singh, R.N. Differential cytotoxic and radiosensitizing effects of silver nanoparticles on triple-negative breast cancer and non-triple-negative breast cells. Int. J. Nanomed. 2015, 10, 3937–3953. [Google Scholar] [CrossRef] [Green Version]
- Bouras, A.; Kaluzova, M.; Hadjipanayis, C.G. Radiosensitivity enhancement of radioresistant glioblastoma by epidermal growth factor receptor antibody-conjugated iron-oxide nanoparticles. J. Neurooncol. 2015, 124, 13–22. [Google Scholar] [CrossRef] [Green Version]
- Wang, B. Analyzing Cell Cycle Checkpoints in Response to Ionizing Radiation in Mammalian Cells. In Cell Cycle Control: Mechanisms and Protocols; Noguchi, E., Gadaleta, M.C., Eds.; Springer: New York, NY, USA, 2014; pp. 313–320. [Google Scholar] [CrossRef]
- Fernet, M.; Mégnin-Chanet, F.; Hall, J.; Favaudon, V. Control of the G2/M checkpoints after exposure to low doses of ionising radiation: Implications for hyper-radiosensitivity. DNA Repair 2010, 9, 48–57. [Google Scholar] [CrossRef]
- Pawlik, T.M.; Keyomarsi, K. Role of cell cycle in mediating sensitivity to radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2004, 59, 928–942. [Google Scholar] [CrossRef]
- Xu, W.; Luo, T.; Li, P.; Zhou, C.; Cui, D.; Pang, B.; Ren, Q.; Fu, S. RGD-conjugated gold nanorods induce radiosensitization in melanoma cancer cells by downregulating α(v)β₃ expression. Int. J. Nanomed. 2012, 7, 915–924. [Google Scholar] [CrossRef] [Green Version]
- Roa, W.; Zhang, X.; Guo, L.; Shaw, A.; Hu, X.; Xiong, Y.; Gulavita, S.; Patel, S.; Sun, X.; Chen, J.; et al. Gold nanoparticle sensitize radiotherapy of prostate cancer cells by regulation of the cell cycle. Nanotechnology 2009, 20, 375101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, E.H.; Kim, M.-S.; Song, H.S.; Yoo, S.H.; Sai, S.; Chung, K.; Sung, J.; Jeong, Y.K.; Jo, Y.; Yoon, M. Gold nanoparticles as a potent radiosensitizer in neutron therapy. Oncotarget 2017, 8, 112390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, C.; Jiang, Y.; Li, X.; Hu, L. Thioglucose-bound gold nanoparticles increase the radiosensitivity of a triple-negative breast cancer cell line (MDA-MB-231). Breast Cancer 2015, 22, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Shetake, N.G.; Kumar, A.; Pandey, B.N. Iron-oxide nanoparticles target intracellular HSP90 to induce tumor radio-sensitization. Biochim. Biophys. Acta (BBA) Gen. Subj. 2019, 1863, 857–869. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Gong, S.; Wang, J.; Yu, L.; Chen, Y.; Li, N.; Tang, B. A nuclear-targeted titanium dioxide radiosensitizer for cell cycle regulation and enhanced radiotherapy. Chem. Commun. 2019, 55, 8182–8185. [Google Scholar] [CrossRef] [PubMed]
- Fournier, C.; Taucher-Scholz, G. Radiation induced cell cycle arrest: An overview of specific effects following high-LET exposure. Radiother. Oncol. 2004, 73, S119–S122. [Google Scholar] [CrossRef]
- Lücke-Huhle, C. Alpha-Irradiation-Induced G2 Delay: A Period of Cell Recovery. Radiat. Res. 1982, 89, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Blakely, E.A.; Kronenberg, A. Heavy-Ion Radiobiology: New Approaches to Delineate Mechanisms Underlying Enhanced Biological Effectiveness. Radiat. Res. 1998, 150, S126–S145. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Kong, Y.; Yang, Z.; Liu, Y.; Liu, R.; Geng, Y.; Luo, H.; Zhang, H.; Li, H.; Feng, S.; et al. Preliminary study on radiosensitivity to carbon ions in human breast cancer. J. Radiat. Res. 2020, 61, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Tsuboi, Y.; Tsuchida, T.; Nose, K.; Ando, K. Cytotoxic effect of accelerated carbon beams on glioblastoma cell lines with p53 mutation: Clonogenic survival and cell-cycle analysis. Int. J. Radiat. Biol. 1998, 74, 71–79. [Google Scholar] [CrossRef]
- Meijer, A.E.; Ekedahl, J.; Joseph, B.; Castro, J.; Harms-Ringdahl, M.; Zhivotovsky, B.; Lewensohn, R. High-LET radiation induces apoptosis in lymphoblastoid cell lines derived from ataxia-telangiectasia patients. Int. J. Radiat. Biol. 2001, 77, 309–317. [Google Scholar] [CrossRef]
- Goto, S.; Watanabe, M.; Yatagai, F. Delayed Cell Cycle Progression in Human Lymphoblastoid Cells after Exposure to High-LET Radiation Correlates with Extremely Localized DNA Damage. Radiat. Res. 2002, 158, 678–686. [Google Scholar] [CrossRef]
- Gadbois, D.M.; Crissman, H.A.; Nastasi, A.; Habbersett, R.; Wang, S.-K.; Chen, D.; Lehnert, B.E. Alterations in the Progression of Cells through the Cell Cycle after Exposure to Alpha Particles or Gamma Rays. Radiat. Res. 1996, 146, 414–424. [Google Scholar] [CrossRef]
- Antoccia, A.; Sgura, A.; Berardinelli, F.; Cavinato, M.; Cherubini, R.; Gerardi, S.; Tanzarella, C. Cell Cycle Perturbations and Genotoxic Effects in Human Primary Fibroblasts Induced by Low-energy Protons and X/γ-rays. J. Radiat. Res. 2009, 50, 457–468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Truong, K.; Bradley, S.; Baginski, B.; Wilson, J.R.; Medlin, D.; Zheng, L.; Wilson, R.K.; Rusin, M.; Takacs, E.; Dean, D. The effect of well-characterized, very low-dose x-ray radiation on fibroblasts. PLoS ONE 2018, 13, e0190330. [Google Scholar] [CrossRef] [Green Version]
- Zhao, H.; Zhuang, Y.; Li, R.; Liu, Y.; Mei, Z.; He, Z.; Zhou, F.; Zhou, Y. Effects of different doses of X-ray irradiation on cell apoptosis, cell cycle, DNA damage repair and glycolysis in HeLa cells. Oncol. Lett. 2019, 17, 42–54. [Google Scholar] [CrossRef] [PubMed]
- McQuaid, H.N.; Muir, M.F.; Taggart, L.E.; McMahon, S.J.; Coulter, J.A.; Hyland, W.B.; Jain, S.; Butterworth, K.T.; Schettino, G.; Prise, K.M.; et al. Imaging and radiation effects of gold nanoparticles in tumour cells. Sci. Rep. 2016, 6, 19442. [Google Scholar] [CrossRef] [PubMed]
- Butterworth, K.T.; Wyer, J.A.; Brennan-Fournet, M.; Latimer, C.J.; Shah, M.B.; Currell, F.J.; Hirst, D.G. Variation of Strand Break Yield for Plasmid DNA Irradiated with High-Z Metal Nanoparticles. Radiat. Res. 2008, 170, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Ghita, M.; McMahon, S.J.; Taggart, L.E.; Butterworth, K.T.; Schettino, G.; Prise, K.M. A mechanistic study of gold nanoparticle radiosensitisation using targeted microbeam irradiation. Sci. Rep. 2017, 7, 44752. [Google Scholar] [CrossRef] [Green Version]
- Alphandéry, E. Nano-Therapies for Glioblastoma Treatment. Cancers 2020, 12, 242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, N.; Yang, W.; Bao, Y.; Xu, H.; Qin, S.; Tu, Y. BSA capped Au nanoparticle as an efficient sensitizer for glioblastoma tumor radiation therapy. RSC Adv. 2015, 5, 40514–40520. [Google Scholar] [CrossRef]
- Štefančíková, L.; Lacombe, S.; Salado, D.; Porcel, E.; Pagáčová, E.; Tillement, O.; Lux, F.; Depeš, D.; Kozubek, S.; Falk, M. Effect of gadolinium-based nanoparticles on nuclear DNA damage and repair in glioblastoma tumor cells. J. Nanobiotechnol. 2016, 14, 63. [Google Scholar] [CrossRef] [Green Version]
- Sun, L.; Joh, D.Y.; Al-Zaki, A.; Stangl, M.; Murty, S.; Davis, J.J.; Baumann, B.C.; Alonso-Basanta, M.; Kao, G.D.; Tsourkas, A.; et al. Theranostic Application of Mixed Gold and Superparamagnetic Iron Oxide Nanoparticle Micelles in Glioblastoma Multiforme. J. Biomed. Nanotechnol. 2016, 12, 347–356. [Google Scholar] [CrossRef] [Green Version]
- Fathy, M.M.; Mohamed, F.S.; Elbialy, N.; Elshemey, W.M. Multifunctional Chitosan-Capped Gold Nanoparticles for enhanced cancer chemo-radiotherapy: An invitro study. Phys. Med. Eur. J. Med. Phys. 2018, 48, 76–83. [Google Scholar] [CrossRef]
- Zhou, H.; Zhang, Y.; Su, G.; Zhai, S.; Yan, B. Enhanced cancer cell killing by a targeting gold nanoconstruct with doxorubicin payload under X-ray irradiation. RSC Adv. 2013, 3, 21596–21603. [Google Scholar] [CrossRef]
- Setua, S.; Ouberai, M.; Piccirillo, S.G.; Watts, C.; Welland, M. Cisplatin-tethered gold nanospheres for multimodal chemo-radiotherapy of glioblastoma. Nanoscale 2014, 6, 10865–10873. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zangeneh, M.; Nedaei, H.A.; Mozdarani, H.; Mahmoudzadeh, A.; Kharrazi, S.; Salimi, M. The role and mechanisms of zinc oxide nanoparticles in the improvement of the radiosensitivity of lung cancer cells in clinically relevant megavoltage radiation energies in-vitro. Nanomed. J. 2019, 6, 276–290. [Google Scholar] [CrossRef]
- Xia, Q.; Li, H.; Liu, Y.; Zhang, S.; Feng, Q.; Xiao, K. The effect of particle size on the genotoxicity of gold nanoparticles. J. Biomed. Mater. Res. Part A 2017, 105, 710–719. [Google Scholar] [CrossRef]
- Chatterjee, N.; Yang, J.; Kim, S.; Joo, S.W.; Choi, J. Diameter size and aspect ratio as critical determinants of uptake, stress response, global metabolomics and epigenetic alterations in multi-wall carbon nanotubes. Carbon 2016, 108, 529–540. [Google Scholar] [CrossRef]
- May, S.; Hirsch, C.; Rippl, A.; Bohmer, N.; Kaiser, J.-P.; Diener, L.; Wichser, A.; Bürkle, A.; Wick, P. Transient DNA damage following exposure to gold nanoparticles. Nanoscale 2018, 10, 15723–15735. [Google Scholar] [CrossRef] [Green Version]
- Szegezdi, E.; Logue, S.E.; Gorman, A.M.; Samali, A. Mediators of endoplasmic reticulum stress-induced apoptosis. EMBO Rep. 2006, 7, 880–885. [Google Scholar] [CrossRef] [Green Version]
- Bialik, S.; Dasari, S.K.; Kimchi, A. Autophagy-dependent cell death—Where, how and why a cell eats itself to death. J. Cell Sci. 2018, 131, jcs215152. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.; Lin, J.; Liu, P.; Huang, Z.; Zhao, P.; Jin, H.; Wang, C.; Wen, L.; Gu, N. Is the autophagy a friend or foe in the silver nanoparticles associated radiotherapy for glioma? Biomaterials 2015, 62, 47–57. [Google Scholar] [CrossRef]
- Teraoka, S.; Kakei, Y.; Akashi, M.; Iwata, E.; Hasegawa, T.; Miyawaki, D.; Sasaki, R.; Komori, T. Gold nanoparticles enhance X-ray irradiation-induced apoptosis in head and neck squamous cell carcinoma in vitro. Biomed. Rep. 2018, 9, 415–420. [Google Scholar] [CrossRef] [Green Version]
- Verano-Braga, T.; Miethling-Graff, R.; Wojdyla, K.; Rogowska-Wrzesinska, A.; Brewer, J.R.; Erdmann, H.; Kjeldsen, F. Insights into the Cellular Response Triggered by Silver Nanoparticles Using Quantitative Proteomics. ACS Nano 2014, 8, 2161–2175. [Google Scholar] [CrossRef]
- Li, Y.; Sun, L.; Jin, M.; Du, Z.; Liu, X.; Guo, C.; Li, Y.; Huang, P.; Sun, Z. Size-dependent cytotoxicity of amorphous silica nanoparticles in human hepatoma HepG2 cells. Toxicol. Vitr. 2011, 25, 1343–1352. [Google Scholar] [CrossRef]
- Liu, P.; Jin, H.; Guo, Z.; Ma, J.; Zhao, J.; Li, D.; Wu, H.; Gu, N. Silver nanoparticles outperform gold nanoparticles in radiosensitizing U251 cells in vitro and in an intracranial mouse model of glioma. Int. J. Nanomed. 2016, 11, 5003–5014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mikuła-Pietrasik, J.; Niklas, A.; Uruski, P.; Tykarski, A.; Książek, K. Mechanisms and significance of therapy-induced and spontaneous senescence of cancer cells. Cell. Mol. Life Sci. 2020, 77, 213–229. [Google Scholar] [CrossRef] [Green Version]
- Nelson, G.; Kucheryavenko, O.; Wordsworth, J.; von Zglinicki, T. The senescent bystander effect is caused by ROS-activated NF-κB signalling. Mech. Ageing Dev. 2018, 170, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Tremi, I.; Havaki, S.; Georgitsopoulou, S.; Lagopati, N.; Georgakilas, V.; Gorgoulis, V.G.; Georgakilas, A.G. A Guide for Using Transmission Electron Microscopy for Studying the Radiosensitizing Effects of Gold Nanoparticles In Vitro. Nanomaterials 2021, 11, 859. [Google Scholar] [CrossRef] [PubMed]
- Lorat, Y.; Brunner, C.U.; Schanz, S.; Jakob, B.; Taucher-Scholz, G.; Rübe, C.E. Nanoscale analysis of clustered DNA damage after high-LET irradiation by quantitative electron microscopy—The heavy burden to repair. DNA Repair 2015, 28, 93–106. [Google Scholar] [CrossRef]
- Lorat, Y.; Fleckenstein, J.; Görlinger, P.; Rübe, C.; Rübe, C.E. Assessment of DNA damage by 53PB1 and pKu70 detection in peripheral blood lymphocytes by immunofluorescence and high-resolution transmission electron microscopy. Strahlenther. Onkol. 2020, 196, 821–833. [Google Scholar] [CrossRef] [Green Version]
- Reifarth, M.; Hoeppener, S.; Schubert, U.S. Uptake and Intracellular Fate of Engineered Nanoparticles in Mammalian Cells: Capabilities and Limitations of Transmission Electron Microscopy—Polymer-Based Nanoparticles. Adv. Mater. 2018, 30, 1703704. [Google Scholar] [CrossRef] [PubMed]
- Goto, H.; Tomono, Y.; Ajiro, K.; Kosako, H.; Fujita, M.; Sakurai, M.; Okawa, K.; Iwamatsu, A.; Okigaki, T.; Takahashi, T.; et al. Identification of a Novel Phosphorylation Site on Histone H3 Coupled with Mitotic Chromosome Condensation. J. Biol. Chem. 1999, 274, 25543–25549. [Google Scholar] [CrossRef] [Green Version]
- Xu, B.; Kim, S.; Kastan, M.B. Involvement of Brca1 in S-phase and G(2)-phase checkpoints after ionizing irradiation. Mol. Cell. Biol. 2001, 21, 3445–3450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Milner, A.E.; Vaughan, A.T.M.; Clark, I.P. Measurement of DNA Damage in Mammalian Cells Using Flow Cytometry. Radiat. Res. 1987, 110, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Shapero, K.; Fenaroli, F.; Lynch, I.; Cottell, D.C.; Salvati, A.; Dawson, K.A. Time and space resolved uptake study of silica nanoparticles by human cells. Mol. BioSyst. 2011, 7, 371–378. [Google Scholar] [CrossRef] [Green Version]
- Maliszewska-Olejniczak, K.; Dróżdż, A.; Waluś, M.; Dorosz, M.; Gryziński, M.A. Immunofluorescence Imaging of DNA Damage and Repair Foci in Human Colon Cancer Cells. J. Vis. Exp. 2020. [Google Scholar] [CrossRef] [PubMed]
- Vrouwe, M.G.; Pines, A.; Overmeer, R.M.; Hanada, K.; Mullenders, L.H.F. UV-induced photolesions elicit ATR-kinase-dependent signaling in non-cycling cells through nucleotide excision repair-dependent and -independent pathways. J. Cell Sci. 2011, 124, 435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soultanakis, R.P.; Melamede, R.J.; Bespalov, I.A.; Wallace, S.S.; Beckman, K.B.; Ames, B.N.; Taatjes, D.J.; Janssen-Heininger, Y.M.W. Fluorescence detection of 8-oxoguanine in nuclear and mitochondrial DNA of cultured cells using a recombinant Fab and confocal scanning laser microscopy. Free Radic. Biol. Med. 2000, 28, 987–998. [Google Scholar] [CrossRef]
- Bromma, K.; Cicon, L.; Beckham, W.; Chithrani, D.B. Gold nanoparticle mediated radiation response among key cell components of the tumour microenvironment for the advancement of cancer nanotechnology. Sci. Rep. 2020, 10, 12096. [Google Scholar] [CrossRef]
- Drouin, R.; Gao, S.; Holmquist, G.P. Agarose Gel Electrophoresis for DNA Damage Analysis. In Technologies for Detection of DNA Damage and Mutations; Pfeifer, G.P., Ed.; Springer: Boston, MA, USA, 1996; pp. 37–43. [Google Scholar] [CrossRef]
- Sutherland, B.M.; Bennett, P.V.; Sutherland, J.C. DNA Damage Quantitation by Alkaline Gel Electrophoresis. In DNA Repair Protocols: Mammalian Systems; Henderson, D.S., Ed.; Humana Press: Totowa, NJ, USA, 2006; pp. 251–273. [Google Scholar] [CrossRef] [Green Version]
- Sarkaria, J.N.; Bush, C.; Eady, J.J.; Peacock, J.H.; Steel, G.G.; Yarnold, J.R. Comparison between Pulsed-Field Gel Electrophoresis and the Comet Assay as Predictive Assays for Radiosensitivity in Fibroblasts. Radiat. Res. 1998, 150, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Olive, P.L.; Banáth, J.P. The comet assay: A method to measure DNA damage in individual cells. Nat. Protoc. 2006, 1, 23–29. [Google Scholar] [CrossRef]
- Falk, M.; Wolinsky, M.; Veldwijk, M.R.; Hildenbrand, G.; Hausmann, M. Gold nanoparticle enhanced radiosensitivity of cells: Considerations and contradictions from model systems and basic investigations of cell damaging for radiation therapy. In Nanoparticle Enhanced Radiation Therapy; IOP Publishing: Bristol, UK, 2020; pp. 1011–1025. [Google Scholar] [CrossRef]
- Pandey, A.; Vighetto, V.; Di Marzio, N.; Ferraro, F.; Hirsch, M.; Ferrante, N.; Mitra, S.; Grattoni, A.; Filgueira, C.S. Gold Nanoparticles Radio-Sensitize and Reduce Cell Survival in Lewis Lung Carcinoma. Nanomaterials (Basel) 2020, 10, 1717. [Google Scholar] [CrossRef]
- Franken, N.A.P.; Rodermond, H.M.; Stap, J.; Haveman, J.; van Bree, C. Clonogenic assay of cells in vitro. Nat. Protoc. 2006, 1, 2315–2319. [Google Scholar] [CrossRef] [PubMed]
- Kazmi, F.; Vallis, K.A.; Vellayappan, B.A.; Bandla, A.; Yukun, D.; Carlisle, R. Megavoltage Radiosensitization of Gold Nanoparticles on a Glioblastoma Cancer Cell Line Using a Clinical Platform. Int. J. Mol. Sci. 2020, 21, 429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kyrylkova, K.; Kyryachenko, S.; Leid, M.; Kioussi, C. Detection of Apoptosis by TUNEL Assay. In Odontogenesis: Methods and Protocols; Kioussi, C., Ed.; Humana Press: Totowa, NJ, USA, 2012; pp. 41–47. [Google Scholar] [CrossRef]
- Coppé, J.-P.; Desprez, P.-Y.; Krtolica, A.; Campisi, J. The senescence-associated secretory phenotype: The dark side of tumor suppression. Annu. Rev. Pathol. 2010, 5, 99–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caliò, A.; Zamò, A.; Ponzoni, M.; Zanolin, M.E.; Ferreri, A.J.M.; Pedron, S.; Montagna, L.; Parolini, C.; Fraifeld, V.E.; Wolfson, M.; et al. Cellular Senescence Markers p16INK4a and p21CIP1/WAF Are Predictors of Hodgkin Lymphoma Outcome. Clin. Cancer Res. 2015, 21, 5164. [Google Scholar] [CrossRef] [Green Version]
- Evangelou, K.; Lougiakis, N.; Rizou, S.V.; Kotsinas, A.; Kletsas, D.; Muñoz-Espín, D.; Kastrinakis, N.G.; Pouli, N.; Marakos, P.; Townsend, P.; et al. Robust, universal biomarker assay to detect senescent cells in biological specimens. Aging Cell 2017, 16, 192–197. [Google Scholar] [CrossRef]
- Li, M.; You, L.; Xue, J.; Lu, Y. Ionizing Radiation-Induced Cellular Senescence in Normal, Non-transformed Cells and the Involved DNA Damage Response: A Mini Review. Front. Pharmacol. 2018, 9, 522. [Google Scholar] [CrossRef]
- Hellevik, T.; Pettersen, I.; Berg, V.; Winberg, J.O.; Moe, B.T.; Bartnes, K.; Paulssen, R.H.; Busund, L.-T.; Bremnes, R.; Chalmers, A.; et al. Cancer-associated fibroblasts from human NSCLC survive ablative doses of radiation but their invasive capacity is reduced. Radiat. Oncol. 2012, 7, 59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McGinnity, T.L.; Dominguez, O.; Curtis, T.E.; Nallathamby, P.D.; Hoffman, A.J.; Roeder, R.K. Hafnia (HfO2) nanoparticles as an X-ray contrast agent and mid-infrared biosensor. Nanoscale 2016, 8, 13627–13637. [Google Scholar] [CrossRef]
- Mendoza, J.G.; Aguilar Frutis, M.A.; Flores, G.A.; Hipólito, M.G.; Maciel Cerda, A.; Nieto, J.A.; Montalvo, T.R.; Falcony, C. Synthesis and characterization of hafnium oxide films for thermo and photoluminescence applications. Appl. Radiat. Isot. 2010, 68, 696–699. [Google Scholar] [CrossRef]
- Pottier, A.; Borghi, E.; Levy, L. New Use of Metals as Nanosized Radioenhancers. Anticancer Res. 2014, 34, 443. [Google Scholar] [PubMed]
- Bonvalot, S.; Pechoux, C.L.; Baere, T.D.; Buy, X.; Italiano, A.; Stockle, E.; Terrier, P.; Lassau, N.; Cesne, A.L.; Sargos, P.; et al. Phase I study of NBTXR3 nanoparticles, in patients with advanced soft tissue sarcoma (STS). J. Clin. Oncol. 2014, 32, 10563. [Google Scholar] [CrossRef]
- Kratz, F.; Elsadek, B. Clinical impact of serum proteins on drug delivery. J. Control. Release 2012, 161, 429–445. [Google Scholar] [CrossRef]
- Shreffler, J.W.; Pullan, J.E.; Dailey, K.M.; Mallik, S.; Brooks, A.E. Overcoming Hurdles in Nanoparticle Clinical Translation: The Influence of Experimental Design and Surface Modification. Int. J. Mol. Sci. 2019, 20, 6056. [Google Scholar] [CrossRef] [Green Version]
- Thomas, O.S.; Weber, W. Overcoming Physiological Barriers to Nanoparticle Delivery—Are We There Yet? Front. Bioeng. Biotechnol. 2019, 7. [Google Scholar] [CrossRef]
- Davis, M.E.; Zuckerman, J.E.; Choi, C.H.J.; Seligson, D.; Tolcher, A.; Alabi, C.A.; Yen, Y.; Heidel, J.D.; Ribas, A. Evidence of RNAi in humans from systemically administered siRNA via targeted nanoparticles. Nature 2010, 464, 1067–1070. [Google Scholar] [CrossRef]
- Phillips, E.; Penate-Medina, O.; Zanzonico, P.B.; Carvajal, R.D.; Mohan, P.; Ye, Y.; Humm, J.; Gönen, M.; Kalaigian, H.; Schöder, H.; et al. Clinical translation of an ultrasmall inorganic optical-PET imaging nanoparticle probe. Sci. Transl. Med. 2014, 6, 260ra149. [Google Scholar] [CrossRef] [Green Version]
- Torrisi, L.; Restuccia, N.; Cuzzocrea, S.; Paterniti, I.; Ielo, I.; Pergolizzi, S.; Cutroneo, M.; Kovacik, L. Laser-produced Au nanoparticles as X-ray contrast agents for diagnostic imaging. Gold Bull. 2017, 50, 51–60. [Google Scholar] [CrossRef]
- Zhang, Y.-N.; Poon, W.; Tavares, A.J.; McGilvray, I.D.; Chan, W.C.W. Nanoparticle–liver interactions: Cellular uptake and hepatobiliary elimination. J. Control. Release 2016, 240, 332–348. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.S.; Liu, W.; Misra, P.; Tanaka, E.; Zimmer, J.P.; Itty Ipe, B.; Bawendi, M.G.; Frangioni, J.V. Renal clearance of quantum dots. Nat. Biotechnol. 2007, 25, 1165–1170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Albanese, A.; Tang, P.S.; Chan, W.C.W. The Effect of Nanoparticle Size, Shape, and Surface Chemistry on Biological Systems. Annu. Rev. Biomed. Eng. 2012, 14, 1–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sancey, L.; Lux, F.; Kotb, S.; Roux, S.; Dufort, S.; Bianchi, A.; Crémillieux, Y.; Fries, P.; Coll, J.-L.; Rodriguez-Lafrasse, C.; et al. The use of theranostic gadolinium-based nanoprobes to improve radiotherapy efficacy. Br. J. Radiol. 2014, 87, 20140134. [Google Scholar] [CrossRef]
- Poller, W.C.; Pieber, M.; Boehm-Sturm, P.; Ramberger, E.; Karampelas, V.; Möller, K.; Schleicher, M.; Wiekhorst, F.; Löwa, N.; Wagner, S.; et al. Very small superparamagnetic iron oxide nanoparticles: Long-term fate and metabolic processing in atherosclerotic mice. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 2575–2586. [Google Scholar] [CrossRef] [PubMed]
- Pouliquen, D.; Le Jeune, J.J.; Perdrisot, R.; Ermias, A.; Jallet, P. Iron oxide nanoparticles for use as an MRI contrast agent: Pharmacokinetics and metabolism. Magn. Reson. Imaging 1991, 9, 275–283. [Google Scholar] [CrossRef]
- Sadauskas, E.; Wallin, H.; Stoltenberg, M.; Vogel, U.; Doering, P.; Larsen, A.; Danscher, G. Kupffer cells are central in the removal of nanoparticles from the organism. Part. Fibre Toxicol. 2007, 4, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sadauskas, E.; Danscher, G.; Stoltenberg, M.; Vogel, U.; Larsen, A.; Wallin, H. Protracted elimination of gold nanoparticles from mouse liver. Nanomed. Nanotechnol. Biol. Med. 2009, 5, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Lammers, T.; Peschke, P.; Kühnlein, R.; Subr, V.; Ulbrich, K.; Huber, P.; Hennink, W.; Storm, G. Effect of Intratumoral Injection on the Biodistribution, the Therapeutic Potential of HPMA Copolymer-Based Drug Delivery Systems. Neoplasia 2006, 8, 788–795. [Google Scholar] [CrossRef] [Green Version]
- Hara, D.; Tao, W.; Totiger, T.; Pourmand, A.; Dogan, N.; Ford, J.C.; Shi, J.; Pollack, A. Prostate Cancer Targeted X-ray Fluorescence Imaging via Gold Nanoparticles Functionalized with Prostate-Specific Membrane Antigen (PSMA). Int. J. Radiat. Oncol. Biol. Phys. 2021. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Kang, P.M. Systemic Review of Biodegradable Nanomaterials in Nanomedicine. Nanomaterials 2020, 10, 656. [Google Scholar] [CrossRef] [Green Version]
- Wolfe, T.; Chatterjee, D.; Lee, J.; Grant, J.D.; Bhattarai, S.; Tailor, R.; Goodrich, G.; Nicolucci, P.; Krishnan, S. Targeted gold nanoparticles enhance sensitization of prostate tumors to megavoltage radiation therapy in vivo. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 1277–1283. [Google Scholar] [CrossRef] [Green Version]
- Dilmanian, F.A.; Krishnan, S.; McLaughlin, W.E.; Lukaniec, B.; Baker, J.T.; Ailawadi, S.; Hirsch, K.N.; Cattell, R.F.; Roy, R.; Helfer, J.; et al. Merging Orthovoltage X-Ray Minibeams spare the proximal tissues while producing a solid beam at the target. Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, P.; Marshall, T.I.; Currell, F.J.; Kacperek, A.; Schettino, G.; Prise, K.M. Variations in the Processing of DNA Double-Strand Breaks Along 60-MeV Therapeutic Proton Beams. Int. J. Radiat. Oncol. Biol. Phys. 2016, 95, 86–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Held, K.D.; Kawamura, H.; Kaminuma, T.; Paz, A.E.S.; Yoshida, Y.; Liu, Q.; Willers, H.; Takahashi, A. Effects of Charged Particles on Human Tumor Cells. Front. Oncol. 2016, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vozenin, M.-C.; De Fornel, P.; Petersson, K.; Favaudon, V.; Jaccard, M.; Germond, J.-F.; Petit, B.; Burki, M.; Ferrand, G.; Patin, D.; et al. The Advantage of FLASH Radiotherapy Confirmed in Mini-pig and Cat-cancer Patients. Clin. Cancer Res. 2019, 25, 35–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Griffin, R.J.; Limoli, C.L.; Simone, C.B., II. Radiation Research Special Issue: New Beam Delivery Modalities are Shaping the Future of Radiotherapy. Radiat. Res. 2020, 194, 567–570. [Google Scholar] [CrossRef]
| Name (Sponsor) | Particle Type/Drug | Application/Indication | Patients | Type of IR/Dose | ClinicalTrials.gov Identifier (Phase) | Status |
|---|---|---|---|---|---|---|
| AGuIX (University Hospital, Grenoble) | Gadolinium-based nanoparticles | Target tissue: Multiple brain metastases Application: Sequentially rising dose levels of AGuIX NPs were injected in combination with whole brain radiation therapy (Ph I). Based on adapted dose, AGuIX NPs are injected in combination with whole brain radiation therapy (Ph II). | 15 100 | X-rays/30Gy (3Gy/session) | NCT02820454 (Ph I) [68] NCT03818386 (Ph II) | Completed recruiting |
| AGuIX (University Hospital, Grenoble) | Gadolinium-based nanoparticles | Target tissue: Pancreatic and lung cancer Application: AGuIX NPs are injected in combination with MR-guided stereotactic body radiation therapy (SBRT) | 100 | X-rays/- | NCT04789486 (Ph I/II) | recruiting |
| AGulX (Centre Francois Baclesse) | Polysiloxane and Gadolinium-based nanoparticles | Target tissue: Relapsing tumors of the cephalo-spino-iliosacral axis particularly base of the skull, pharyngeal wall, parapharyngeal, and retropharyngeal lymph nodes, etc. Application: AGuIX NPs are injected in combination with photon therapy | 46 | Photon therapy/- | NCT04784221 (Ph II) | Not yet recruiting |
| NBTXR3 PEP503 (Nanobiotix) | Hafnium oxide-based nanoparticles | Target tissue: locally advanced soft-tissue sarcoma of the Extremity and Trunk Wall Application: NPs were stimulated with external radiation to enhance tumor cell death via electron production | 180 | External beam radiotherapy/ 50Gy(2Gy/fraction) | NCT02379845 (Ph II/III) [66] | completed |
| NBTXR3 PEP503 (Nanobiotix) | Hafnium oxide-based nanoparticles | Target tissue: locally advanced squamous cell carcinoma of the oral Application: NPs are stimulated with external radiation to enhance tumor cell death via electron production | 48 | Intensity-Modulated Radiation Therapy /70Gy (2Gy/fraction) | NCT01946867 (Ph I) | recruiting |
| NBTXR3 PEP503 (Nanobiotix) | Hafnium oxide- based nanoparticles/PD-I inhibitor | Target tissue: advanced cancer types Application: NPs are stimulated with external radiation to enhance tumor cell death via electron production in combination with immunotherapy | 60 | Radiotherapy/- | NCT03589339 (Ph I) | recruiting |
| NBTXR3 PEP503 (Nanobiotix) | Hafnium oxide-based nanoparticles | Target tissue: liver cancers Application: NPs are stimulated with external radiation to enhance tumor cell death via electron production NCT02721056 | 23 (initial nu. 200) | Stereotactic Body Radiation Therapy/45Gy (15Gy/fraction) or 50Gy (5Gy/fraction) | NCT02721056 (Ph I), Ph II will be designed independently | terminated |
| NBTXR3 PEP503 (Nanobiotix) | Hafnium oxide-based nanoparticles | Target tissue: Prostate Adenocarcinoma Application: NPs are stimulated with external radiation with and without brachytherapy to enhance tumor cell death via electron production. | 5(initial nu. 96) | External Beam Radiation Therapy/45Gy (1.8Gy/ fraction) or Brachytherapy Boost/15Gy + EBRT 45Gy (1.8/fraction) | NCT02805894 (PhI/II) | terminated |
| SPION | Iron oxide Nanoparticles/ ferumoxytol | Target tissue: Primary and metastatic hepatic cancers Application: MR-Linac imaging with SPION in combination with radiotherapy to increase detection and efficacy. | 25 | Stereotactic Body Radiotherapy (LINAC)/- | NCT04682847 | recruiting |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tremi, I.; Spyratou, E.; Souli, M.; Efstathopoulos, E.P.; Makropoulou, M.; Georgakilas, A.G.; Sihver, L. Requirements for Designing an Effective Metallic Nanoparticle (NP)-Boosted Radiation Therapy (RT). Cancers 2021, 13, 3185. https://doi.org/10.3390/cancers13133185
Tremi I, Spyratou E, Souli M, Efstathopoulos EP, Makropoulou M, Georgakilas AG, Sihver L. Requirements for Designing an Effective Metallic Nanoparticle (NP)-Boosted Radiation Therapy (RT). Cancers. 2021; 13(13):3185. https://doi.org/10.3390/cancers13133185
Chicago/Turabian StyleTremi, Ioanna, Ellas Spyratou, Maria Souli, Efstathios P. Efstathopoulos, Mersini Makropoulou, Alexandros G. Georgakilas, and Lembit Sihver. 2021. "Requirements for Designing an Effective Metallic Nanoparticle (NP)-Boosted Radiation Therapy (RT)" Cancers 13, no. 13: 3185. https://doi.org/10.3390/cancers13133185
APA StyleTremi, I., Spyratou, E., Souli, M., Efstathopoulos, E. P., Makropoulou, M., Georgakilas, A. G., & Sihver, L. (2021). Requirements for Designing an Effective Metallic Nanoparticle (NP)-Boosted Radiation Therapy (RT). Cancers, 13(13), 3185. https://doi.org/10.3390/cancers13133185










