The Efficacy and Adverse Events in Patients with Head and Neck Cancer Following Radiotherapy Combined with S-1 Therapy: A Meta-Analysis
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
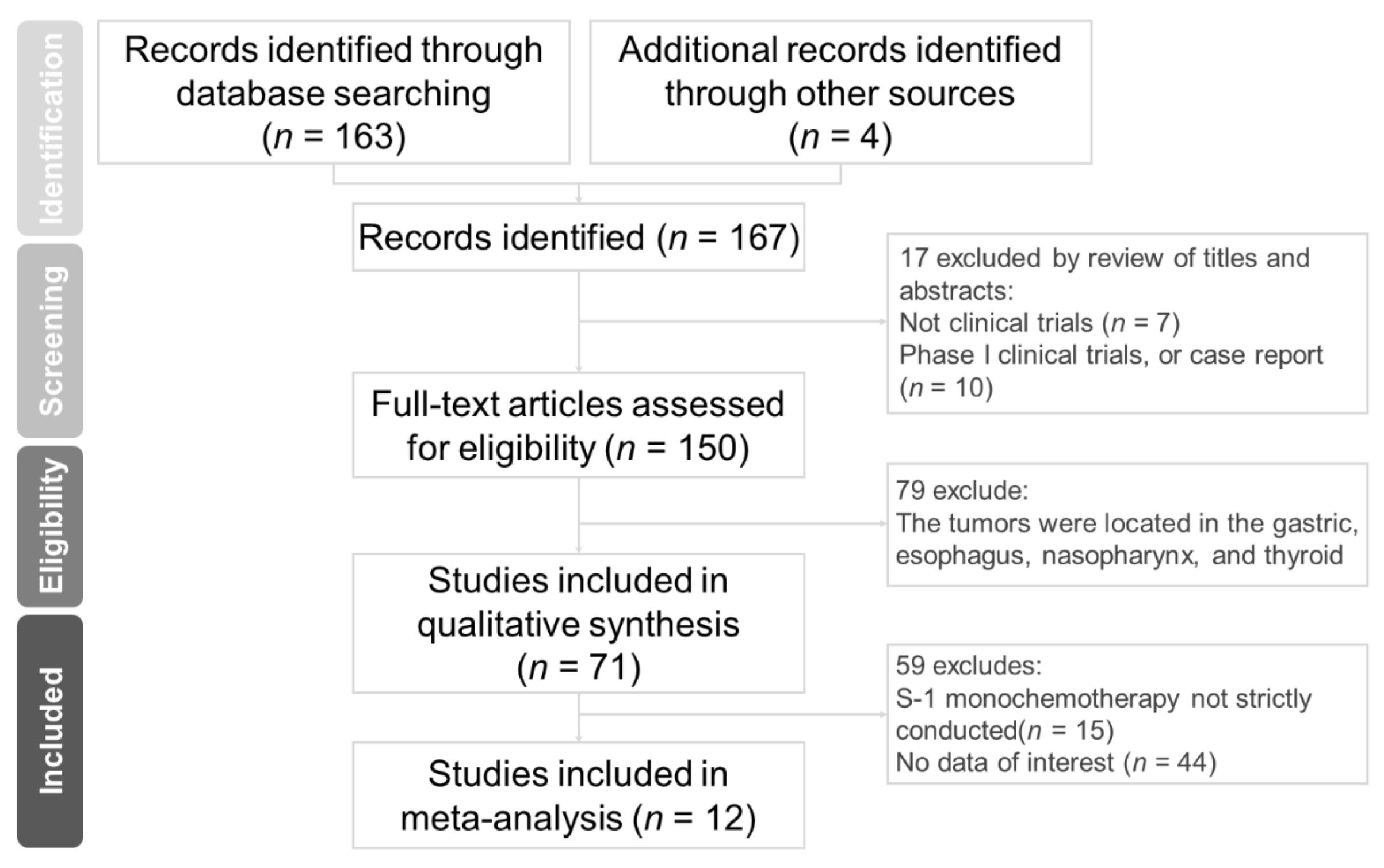
2.2. Study Selection
2.3. Data Extraction and Outcome Measures
2.4. Data Synthesis and Analysis
2.5. Bias Assessment and Quality Assessment
3. Results
3.1. Search Results and Study Characteristics
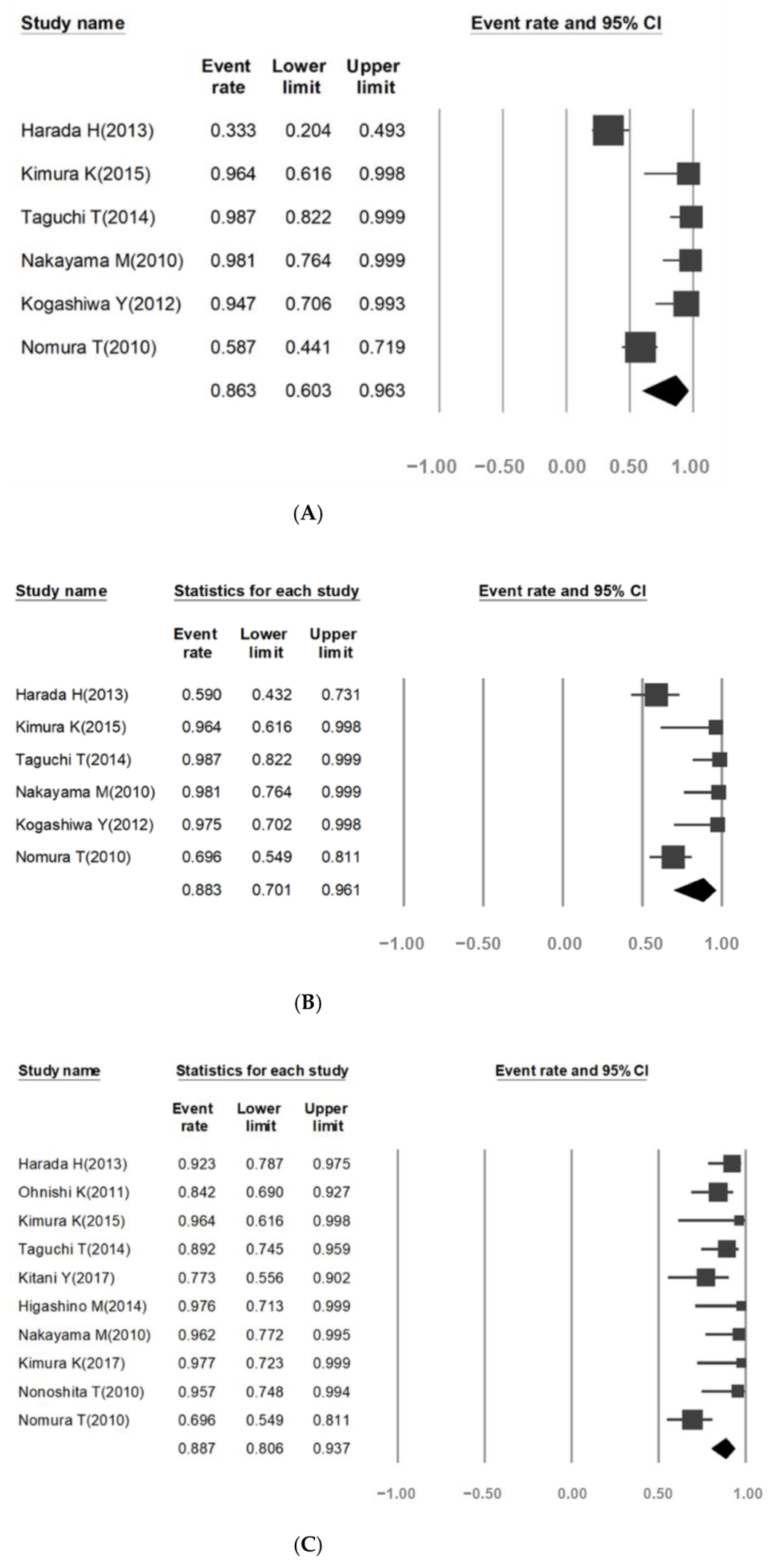
3.2. Effect of Tumor Response Rate
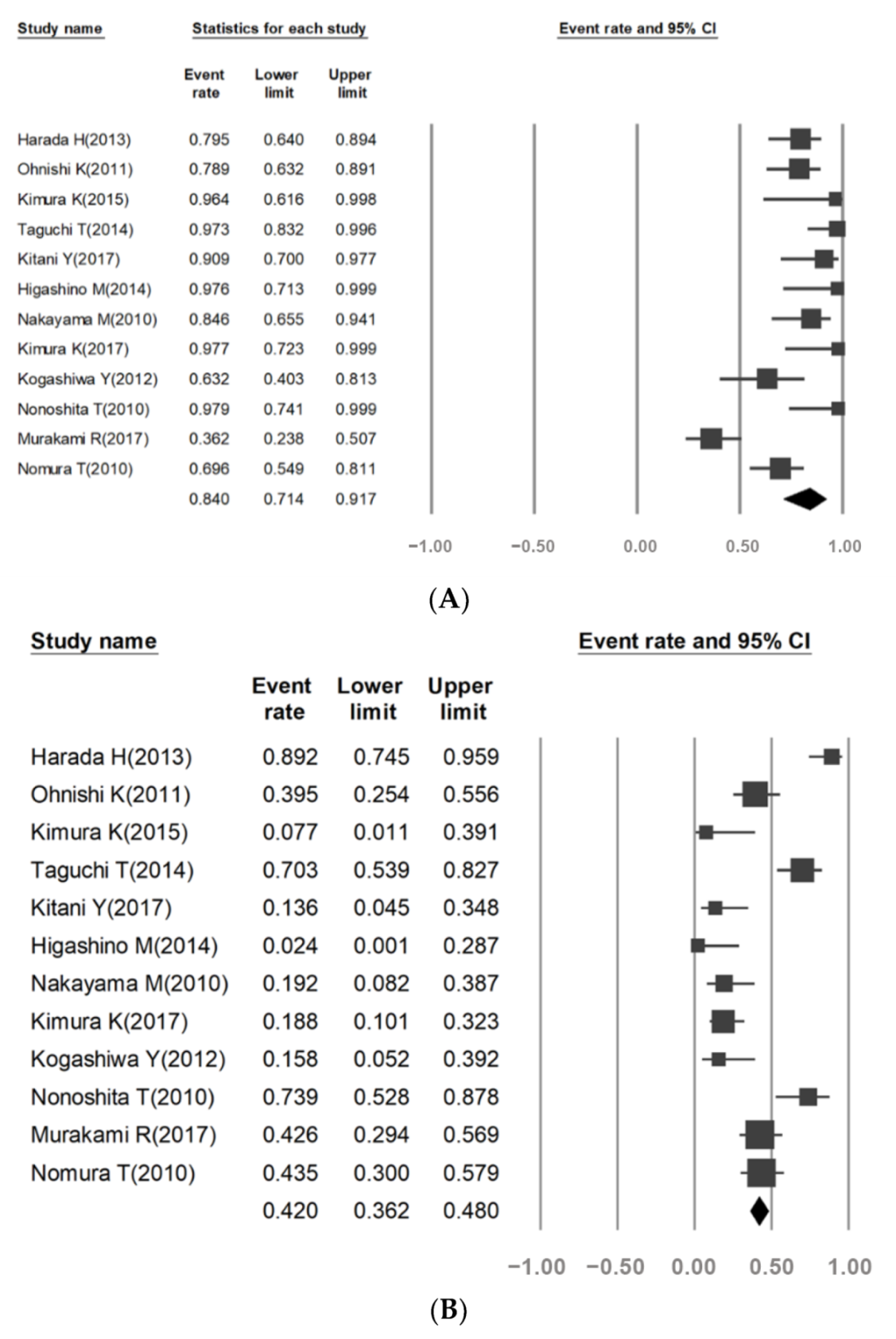
3.3. Survival Rate
3.4. Grade 3/4 Adverse Events (AEs) Rate
3.5. Sensitivity Analysis
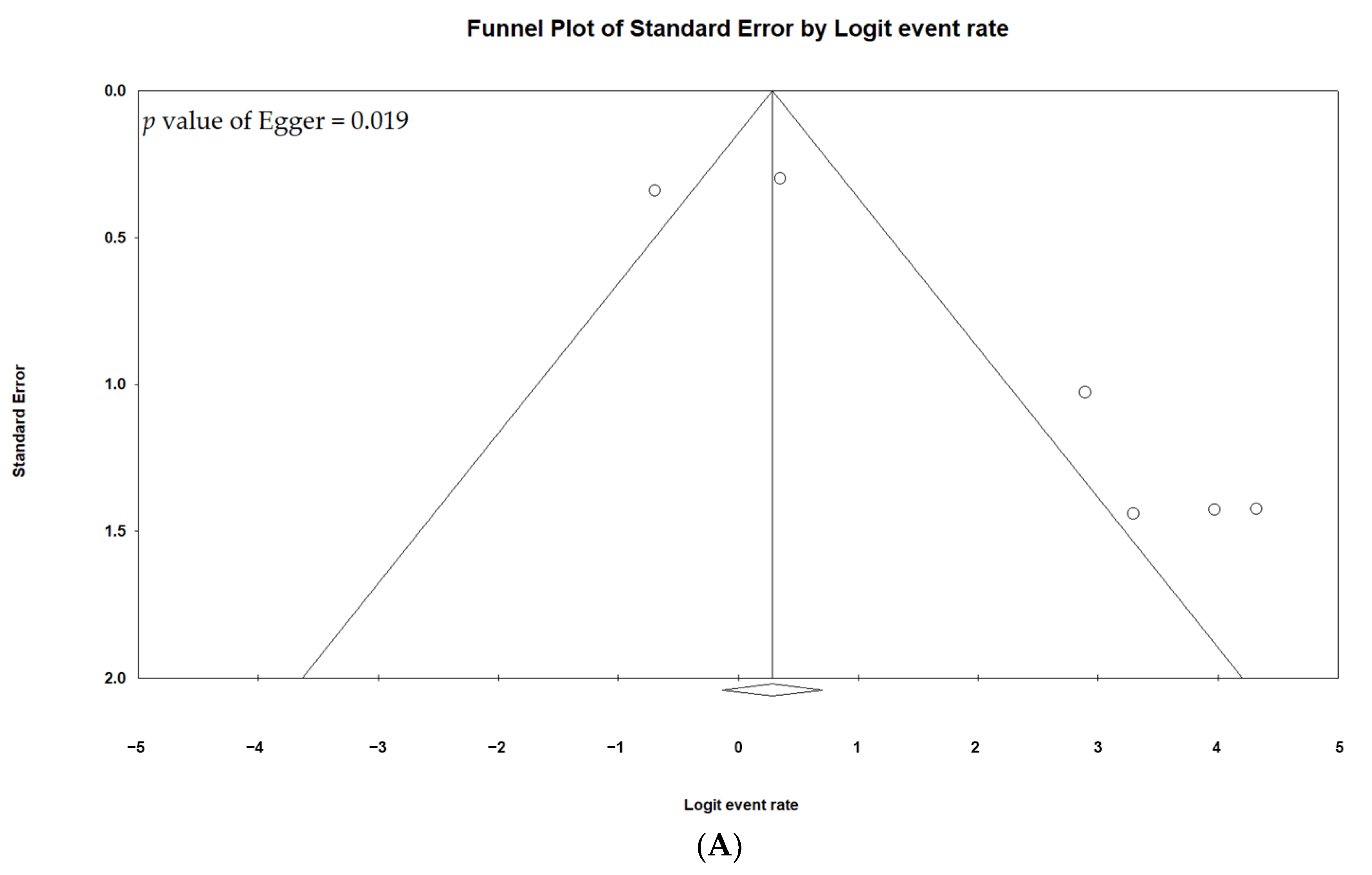
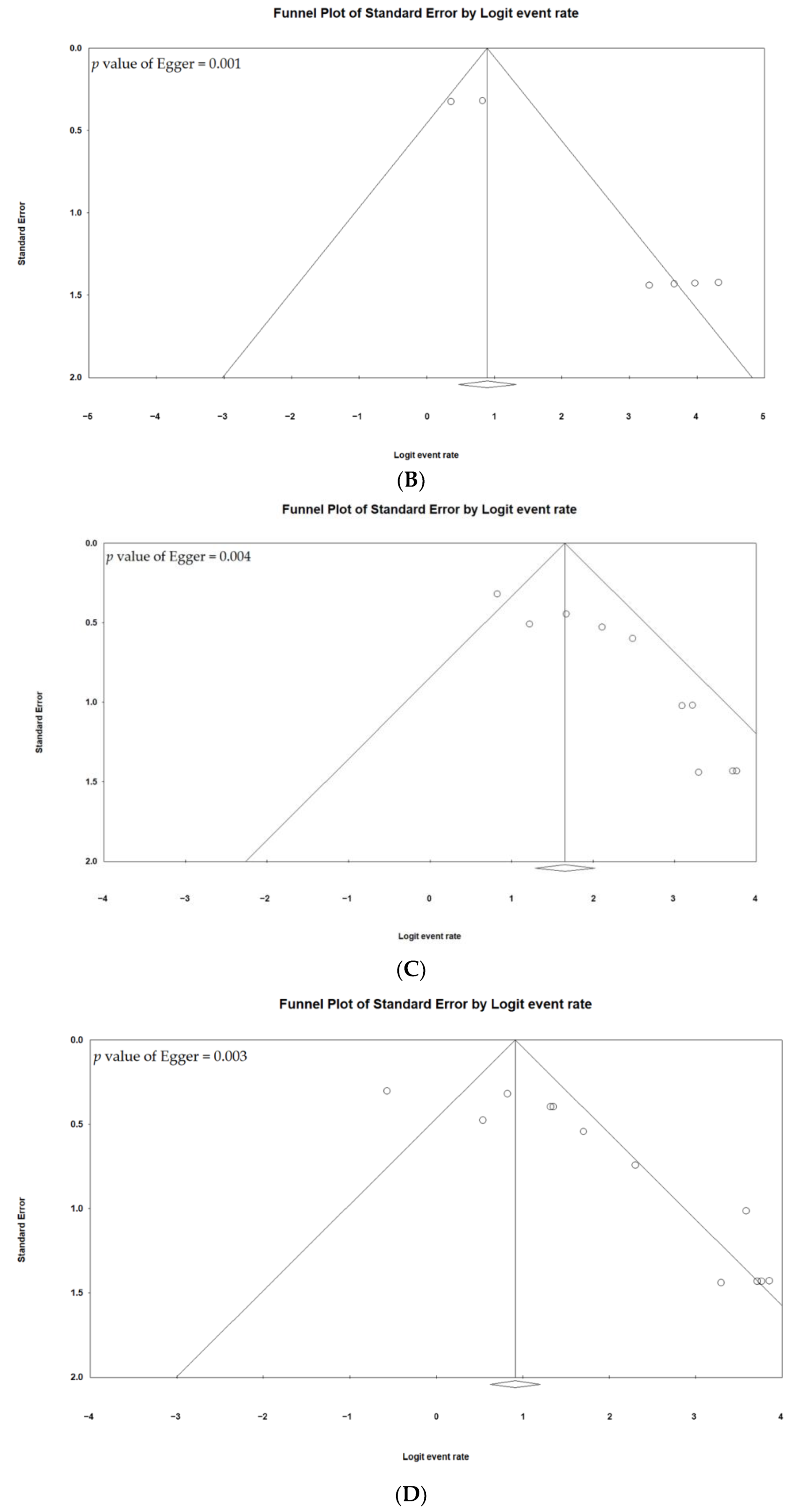
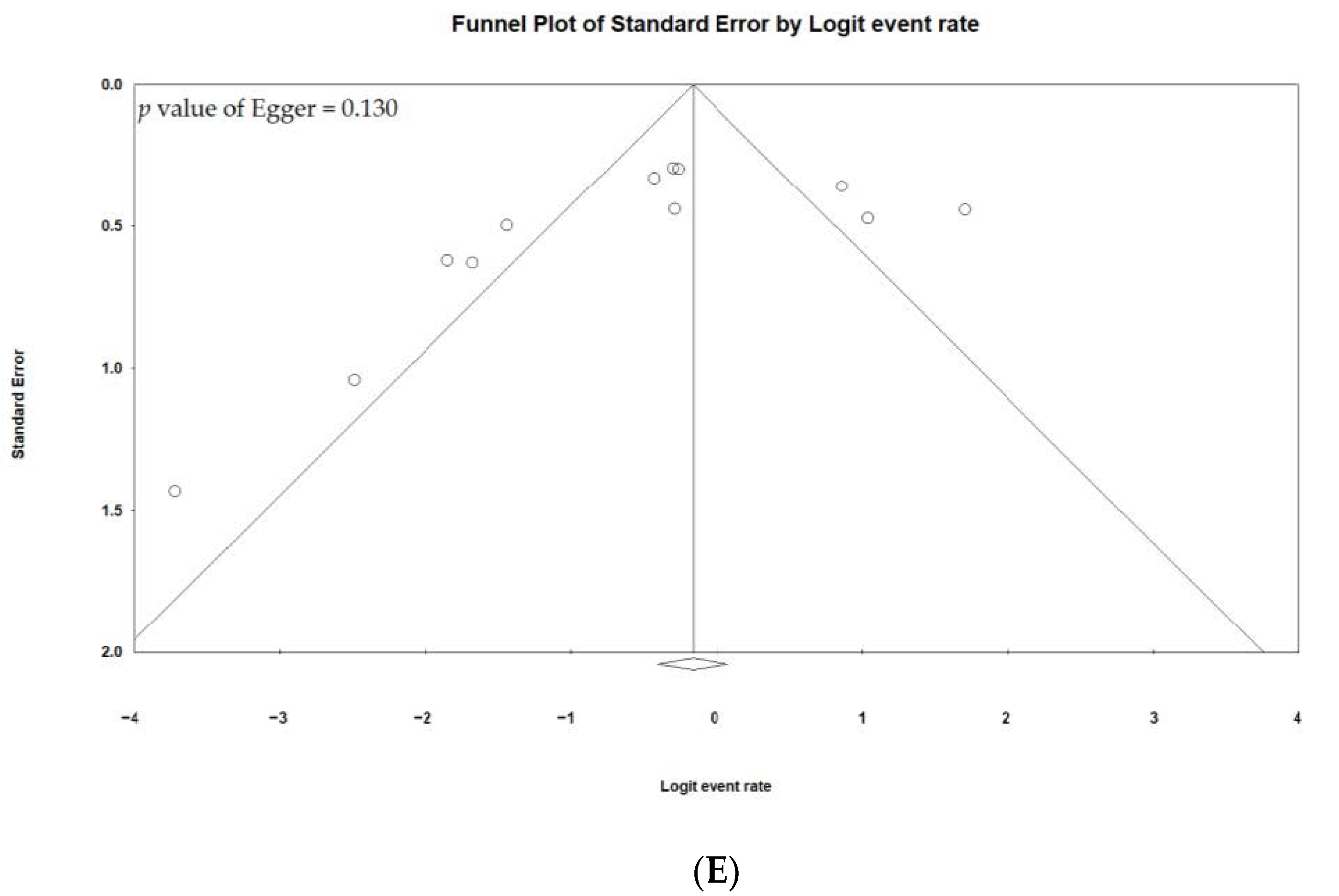
3.6. Publication Bias
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rush, J. Cancer statistics. JAMA 2013, 310, 982. [Google Scholar] [CrossRef]
- Cooper, J.S.; Pajak, T.F.; Forastiere, A.A.; Jacobs, J.; Campbell, B.H.; Saxman, S.B.; Kish, J.A.; Kim, H.E.; Cmelak, A.J.; Rotman, M.; et al. Postoperative concurrent radiotherapy and chemotherapy for high-risk squamous-cell carcinoma of the head and neck. New Engl. J. Med. 2004, 350, 1937–1944. [Google Scholar] [CrossRef] [PubMed]
- Montagna, E.; Cancello, G.; Dellapasqua, S.; Munzone, E.; Colleoni, M. Metronomic therapy and breast cancer: A systematic review. Cancer Treat. Rev. 2014, 40, 942–950. [Google Scholar] [CrossRef]
- Salem, A.D.; Gado, N.M.; Abdelaziz, N.N.; Essa, E.A.; Abdelhafeez, Z.M.; Kamel, T.H. Phase II trial of metronomic chemotherapy as salvage therapy for patients with metastatic breast cancer. J. Egypt. Natl. Cancer Inst. 2008, 20, 134–140. [Google Scholar]
- Nokihara, H.; Lu, S.; Mok, T.; Nakagawa, K.; Yamamoto, N.; Shi, Y.; Zhang, L.; Soo, R.; Yang, J.C.-H.; Sugawara, S.; et al. Randomized controlled trial of S-1 versus docetaxel in patients with non-small-cell lung cancer previously treated with platinum-based chemotherapy (East Asia S-1 Trial in Lung Cancer). Ann. Oncol. 2017, 28, 2698–2706. [Google Scholar] [CrossRef] [PubMed]
- Sakuramoto, S.; Sasako, M.; Yamaguchi, T.; Kinoshita, T.; Fujii, M.; Nashimoto, A.; Furukawa, H.; Nakajima, T.; Ohashi, Y.; Imamura, H.; et al. Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. New Engl. J. Med. 2007, 357, 1810–1820. [Google Scholar] [CrossRef]
- Tatsumi, K.; Fukushima, M.; Shirasaka, T.; Fujii, S. Inhibitory effects of pyrimidine, barbituric acid and pyridine derivatives on 5-fluorouracil degradation in rat liver extracts. Jpn. J. Cancer Res. 1987, 78, 748–755. [Google Scholar]
- Harada, K.; Ferdous, T.; Ueyama, Y. Gimeracil exerts radiosensitizing effects on oral squamous cell carcinoma cells in vitro and in vivo. Anticancer Res. 2016, 36, 5923–5930. [Google Scholar] [CrossRef]
- Diasio, R.B. Clinical implications of dihydropyrimidine dehydrogenase inhibition. Oncology 1999, 13, 17–21. [Google Scholar] [PubMed]
- Shirasaka, T.; Shimamato, Y.; Ohshimo, H.; Yamaguchi, M.; Kato, T.; Yonekura, K.; Fukushima, M. Development of a novel form of an oral 5-fluorouracil derivative (S-1) directed to the potentiation of the tumor selective cytotoxicity of 5-fluorouracil by two biochemical modulators. Anti-Cancer Drugs 1996, 7, 548–557. [Google Scholar] [CrossRef]
- Inuyama, Y.; Kida, A.; Tsukuda, M.; Kohno, N.; Satake, B. Late phase II study of S-1 in patients with advanced head and neck cancer. Gan Kagaku Ryoho. Cancer Chemother. 2001, 28, 1381–1390. [Google Scholar]
- Inuyama, Y.; Kida, A.; Tsukuda, M.; Kohno, N.; Satake, B. Early phase II study of S-1 in patients with advanced head and neck cancer. S-1 cooperative study group (head and neck working group). Gan Kagaku Ryoho. Cancer Chemother. 1998, 25, 1151–1158. [Google Scholar]
- Hutton, B.; Salanti, G.; Caldwell, D.M.; Chaimani, A.; Schmid, C.; Cameron, C.; Ioannidis, J.P.; E Straus, S.; Thorlund, K.; Jansen, J.P.; et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: Checklist and explanations. Ann. Intern. Med. 2015, 162, 777–784. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions, 2nd ed.; John Wiley & Sons: Chichester, UK, 2019. [Google Scholar]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Egger, M.; Smith, G.D.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629. [Google Scholar] [CrossRef] [PubMed]
- Begg, C.B.; Mazumdar, M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994, 50, 1088. [Google Scholar] [CrossRef] [PubMed]
- Furlan, A.D.; Pennick, V.; Bombardier, C.; Van Tulder, M. 2009 Updated method guidelines for systematic reviews in the Cochrane back review group. Spine 2009, 34, 1929–1941. [Google Scholar] [CrossRef] [PubMed]
- Roberts, C.; Torgerson, D. Understanding controlled trials: Baseline imbalance in randomised controlled trials. BMJ 1999, 319, 185. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Brozek, J.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; Debeer, H. GRADE guidelines: 1. Introduction—GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 2011, 64, 383–394. [Google Scholar] [CrossRef]
- Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 2010, 25, 603–605. [Google Scholar] [CrossRef]
- Harada, H.; Omura, K.; Tomioka, H.; Nakayama, H.; Hiraki, A.; Shinohara, M.; Yoshihama, Y.; Shintani, S. Multicenter phase II trial of preoperative chemoradiotherapy with S-1 for locally advanced oral squamous cell carcinoma. Cancer Chemother. Pharmacol. 2013, 71, 1059–1064. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ohnishi, K.; Shioyama, Y.; Nakamura, K.; Nakashima, T.; Ohga, S.; Nonoshita, T.; Yoshitake, T.; Terashima, K.; Komune, S.; Honda, H. Concurrent chemoradiotherapy with S-1 as first-line treatment for patients with oropharyngeal cancer. J. Radiat. Res. 2011, 52, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Kimura, K.; Itoh, Y.; Okada, T.; Nakahara, R.; Kawamura, M.; Kubota, S.; Itoh, J.; Hiramatsu, M.; Fujimoto, Y.; Shibata, T.; et al. Critical evaluation of a prospective study of concurrent chemoradiotherapy with S-1 for early glottic carcinoma. Anticancer Res. 2015, 35, 2385–2390. [Google Scholar]
- Taguchi, T.; Takahashi, M.; Nishimura, G.; Shiono, O.; Komatsu, M.; Sano, D.; Sakuma, Y.; Tanigaki, Y.; Kubota, A.; Taguri, M.; et al. Phase II study of concurrent chemoradiotherapy with S-1 in Patients with Stage II (T2N0M0) squamous cell carcinoma of the pharynx or larynx. Jpn. J. Clin. Oncol. 2014, 44, 1158–1163. [Google Scholar] [CrossRef] [PubMed]
- Kitani, Y.; Kubota, A.; Furukawa, M.; Hori, Y.; Nakayama, Y.; Nonaka, T.; Mizoguchi, N.; Kitani, Y.; Hatakeyama, H.; Oridate, N. Impact of combined modality treatment with radiotherapy and S-1 on T2N0 laryngeal cancer: Possible improvement in survival through the prevention of second primary cancer and distant metastasis. Oral Oncol. 2017, 71, 54–59. [Google Scholar] [CrossRef]
- Higashino, M.; Kawata, R.; Lee, K.; Nishikawa, S.; Ichihara, S.; Uesugi, Y. Radiotherapy concurrent with S-1 and radiotherapy alone for T2N0 glottic carcinoma: A retrospective comparative study. Auris Nasus Larynx 2014, 41, 364–368. [Google Scholar] [CrossRef]
- Nakayama, M.; Hayakawa, K.; Okamoto, M.; Niibe, Y.; Ishiyama, H.; Kotani, S. Phase I/II Trial of concurrent use of S-1 and radiation therapy for T2 glottic cancer. Jpn. J. Clin. Oncol. 2010, 40, 921–926. [Google Scholar] [CrossRef]
- Kimura, K.; Itoh, Y.; Okada, T.; Kubota, S.; Kawamura, M.; Nakahara, R.; Oie, Y.; Kozai, Y.; Takase, Y.; Tsuzuki, H.; et al. Optimized treatment strategy of radiotherapy for early glottic squamous cell carcinomas: An initial analysis. Nagoya J. Med. Sci. 2017, 79, 331–338. [Google Scholar] [PubMed]
- Kogashiwa, Y.; Nagafuji, H.; Kohno, N. Feasibility of concurrent chemoradiotherapy with S-1 administered on alternate days for elderly patients with head and neck cancer. Anticancer Res. 2012, 32, 4035–4040. [Google Scholar]
- Nonoshita, T.; Shioyama, Y.; Nakamura, K.; Nakashima, T.; Ohga, S.; Yoshitake, T.; Ohnishi, K.; Terashima, K.; Asai, K.; Honda, H. Concurrent chemoradiotherapy with S-1 for T2N0 glottic squamous cell carcinoma. J. Radiat. Res. 2010, 51, 481–484. [Google Scholar] [CrossRef] [PubMed]
- Murakami, R.; Semba, A.; Kawahara, K.; Matsuyama, K.; Hiraki, A.; Nagata, M.; Toya, R.; Yamashita, Y.; Oya, N.; Nakayama, H. Concurrent chemoradiotherapy with S-1 in patients with stage III–IV oral squamous cell carcinoma: A retrospective analysis of nodal classification based on the neck node level. Mol. Clin. Oncol. 2017, 7, 140–144. [Google Scholar] [CrossRef]
- Nomura, T.; Murakami, R.; Toya, R.; Teshima, K.; Nakahara, A.; Hirai, T.; Hiraki, A.; Nakayama, H.; Yoshitake, Y.; Ota, K.; et al. Phase II study of preoperative concurrent chemoradiation therapy with S-1 in patients with T4 oral squamous cell carcinoma. Int. J. Radiat. Oncol. 2010, 76, 1347–1352. [Google Scholar] [CrossRef] [PubMed]
- Pignon, J.-P.; le Maître, A.; Maillard, E.; Bourhis, J. Meta-analysis of chemotherapy in head and neck cancer (MACH-NC): An update on 93 randomised trials and 17,346 patients. Radiother. Oncol. 2009, 92, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Budach, W.; Hehr, T.; Budach, V.; Belka, C.; Dietz, K. A meta-analysis of hyperfractionated and accelerated radiotherapy and combined chemotherapy and radiotherapy regimens in unresected locally advanced squamous cell carcinoma of the head and neck. BMC Cancer 2006, 6, 28. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Hamaguchi, T.; Goto, M.; Muro, K.; Matsumura, Y.; Shimada, Y.; Shirao, K.; Nagayama, S. Plasma concentrations of 5-fluorouracil and F-β-alanine following oral administration of S-1, a dihydropyrimidine dehydrogenase inhibitory fluoropyrimidine, as compared with protracted venous infusion of 5-fluorouracil. Br. J. Cancer 2003, 89, 816–820. [Google Scholar] [CrossRef] [PubMed]
- Hirata, K.; Horikoshi, N.; Aiba, K.; Okazaki, M.; Denno, R.; Sasaki, K.; Nakano, Y.; Ishizuka, H.; Yamada, Y.; Uno, S.; et al. Pharmacokinetic study of S-1, a novel oral fluorouracil antitumor drug. Clin. Cancer Res. 1999, 5, 2000–2005. [Google Scholar] [PubMed]
- Byfield, J.E.; Calabro-Jones, P.; Klisak, I.; Kulhanian, F. Pharmacologic requirements for obtaining sensitization of human tumor cells in vitro to combined 5-fluorouracil or ftorafur and X rays. Int. J. Radiat. Oncol. 1982, 8, 1923–1933. [Google Scholar] [CrossRef]
- Smalley, S.R.; Kimler, B.F.; Evans, R.G. 5-Fluorouracil modulation of radiosensitivity in cultured human carcinoma cells. Int. J. Radiat. Oncol. 1991, 20, 207–211. [Google Scholar] [CrossRef]
- Nakata, K.; Sakata, K.-I.; Someya, M.; Miura, K.; Hayashi, J.; Hori, M.; Takagi, M.; Himi, T.; Kondo, A.; Hareyama, M. Phase I study of oral S-1 and concurrent radiotherapy in patients with head and neck cancer. J. Radiat. Res. 2013, 54, 679–683. [Google Scholar] [CrossRef]
- Fujimoto, Y.; Kato, S.; Itoh, Y.; Naganawa, S.; Nakashima, T. A phase I study of concurrent chemoradiotherapy using oral S-1 for head and neck cancer. Anticancer Res. 2014, 34, 209–213. [Google Scholar]
- Inoue, T.; Inoue, T.; Ikeda, H.; Teshima, T.; Murayama, S. Prognostic factor of telecobalt therapy for early glottic carcinoma. Cancer 1992, 70, 2797–2801. [Google Scholar] [CrossRef]
- Mendenhall, W.M.; Werning, J.W.; Hinerman, R.W.; Amdur, R.J.; Villaret, D.B. Management of T1-T2 glottic carcinomas. Cancer 2004, 100, 1786–1792. [Google Scholar] [CrossRef]
- Inoue, T.; Matayoshi, Y.; Inoue, T.; Ikeda, H.; Teshima, T.; Murayama, S. Prognostic factors in telecobalt therapy for early supraglottic carcinoma. Cancer 1993, 72, 57–61. [Google Scholar] [CrossRef]
- Wong, C.S.; Ang, K.K.; Fletcher, G.H.; Thames, H.D.; Peters, L.J.; Byers, R.M.; Oswald, M.J. Definitive radiotherapy for squamous cell carcinoma of the tonsillar fossa. Int. J. Radiat. Oncol. 1989, 16, 657–662. [Google Scholar] [CrossRef]
- Mendenhall, W.M.; Parsons, J.T.; Stringer, S.P.; Cassisi, N.J.; Million, R.R. Radiotherapy alone or combined with neck dissection for T1−T2 carcinoma of the pyriform sinus: An alternative to conservation surgery. Int. J. Radiat. Oncol. 1993, 27, 1017–1027. [Google Scholar] [CrossRef]
- Selek, U.; Garden, A.S.; Morrison, W.H.; El-Naggar, A.K.; Rosenthal, D.; Ang, K. Radiation therapy for early-stage carcinoma of the oropharynx. Int. J. Radiat. Oncol. 2004, 59, 743–751. [Google Scholar] [CrossRef]
- Mello, F.W.; Melo, G.; Pasetto, J.J.; Silva, C.A.B.; Warnakulasuriya, S.; Rivero, E.R.C. The synergistic effect of tobacco and alcohol consumption on oral squamous cell carcinoma: A systematic review and meta-analysis. Clin. Oral Investig. 2019, 23, 2849–2859. [Google Scholar] [CrossRef]
- Hama, T.; Tokumaru, Y.; Fujii, M.; Yane, K.; Okami, K.; Kato, K.; Masuda, M.; Mineta, H.; Nakashima, T.; Sugasawa, M.; et al. Prevalence of human papillomavirus in oropharyngeal cancer: A multicenter study in Japan. Oncology 2014, 87, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Pfister, D.G.; Spencer, S.; Adelstein, D.; Adkins, D.; Anzai, Y.; Brizel, D.M.; Bruce, J.Y.; Busse, P.M.; Caudell, J.J.; Cmelak, A.J.; et al. Head and neck cancers, version 2.2020, NCCN clinical practice guidelines in oncology. J. Natl. Compr. Cancer Netw. 2020, 18, 873–898. [Google Scholar] [CrossRef] [PubMed]
- Ajani, J.A.; Rodriguez, W.; Bodoky, G.; Moiseyenko, V.; Lichinitser, M.; Gorbunova, V.; Vynnychenko, I.; Garin, A.; Lang, I.; Falcon, S. Multicenter phase III comparison of cisplatin/S-1 With cisplatin/infusional fluorouracil in advanced gastric or gastroesophageal adenocarcinoma study: The FLAGS trial. J. Clin. Oncol. 2010, 28, 1547–1553. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.; Lu, H.; Li, J.; Shen, L.; Chen, Z.; Shi, Y.; Song, S.; Qin, S.; Liu, J.; Ouyang, X. Ramdomized 3-armed phase III study of S-1 monotherapy versus S-1/CDDP (SP) versus 5-FU/CDDP (FP) in patients (pts) with advanced gastric cancer (AGC): SC-101 study. J. Clin. Oncol. 2008, 26, 4533. [Google Scholar] [CrossRef]
| Author, Year | Country | Trial Design | Schedule | Age (Years) Median (Range) | Patients Evaluated | Tumor Response | LCR-3 | OS-3 | Grade 3/4 AEs | Evaluation Criteria | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| OR | CB | ||||||||||
| Harada, 2013 [22] | Japan | Single-arm phase II | CRT with S-1 | 56.5 (21–75) | 39 | 13 | 23 | 91.5 | 78.9 | 33 | RECIST/NCICTC |
| Ohnishi, 2011 [23] | Japan | Single-arm Retrospective | CRT with S-1 | 61 (37–82) | 38 | NA | NA | 83 | 79 | 15 | NA/CTCAE |
| Kimura, 2015 [24] | Japan | Single-arm phase I/II | CRT with S-1 | 67 (59–75) | 13 | 13 | 13 | 100 | 100 | 1 | RECIST/NCICTC |
| Taguchi, 2014 [25] | Japan | Single-arm phase II | CRT with S-1 | 68 (49–79) | 37 | 37 | 37 | 89 | 97.2 | 26 | RECIST/CTCAE |
| Kitani, 2017 [26] | Japan | Retrospective comparative | CRT with S-1 | 69 (48–91) | 22 | NA | NA | 75 | 90 | 3 | NA/CTCAE |
| Higashino, 2014 [27] | Japan | Retrospective comparative | CRT with S-1 | 66 (53–81) | 20 | NA | NA | 100 | 100 | 0 | NA/NCITCTC |
| Nakayama, 2010 [28] | Japan | Single-arm phase I/II | CRT with S-1 | 66 (53–80) | 26 | 26 | 26 | 94.7 | 85.4 | 5 | RECIST/NCICTC |
| Kimura, 2017 [29] | Japan | Retrospective comparative | CRT with S-1 | 68.5 (44–94) | 21 | NA | NA | 100 | 100 | 9 | NA/CTCAE |
| Kogashiwa, 2012 [30] | Japan | Single-arm Retrospective | CRT with S-1 | 84 (75–98) | 19 | 18 | 19 | NA | 61.2 | 3 | RECIST/NCICTC |
| Nonoshita, 2010 [31] | Japan | Single-arm Retrospective | CRT with S-1 | 64 (52–78) | 23 | NA | NA | 95.4 | 100 | 17 | NA/CTCAE |
| Murakami, 2017 [32] | Japan | Single-arm Retrospective | CRT with S-1 | 79 (45–91) | 47 | NA | NA | NA | 37.3 | 20 | NA/ NCICTC |
| Nomura, 2010 [33] | Japan | Single-arm phase II | CRT with S-1 | 76 (50–88) | 46 | 27 | 32 | 69 | 69 | 20 | RECIST/NCICTC |
| Endpoints and Adverse Events | Oral Cancer | Laryngeal Cancer | p Value | ||
|---|---|---|---|---|---|
| No. of Trials | Rate % (95%CI) | No. of Trials | Rate % (95%CI) | ||
| Objective response | 2 | 46.0 (23.5–70.4) | 3 | 97.0 (90.2–99.1) | 0.002 |
| Clinical benefit | 2 | 64.0 (53.8–74.0) | 3 | 97.8 (82.2–99.9) | <0.001 |
| 3-year local control rate * | 2 | 82.9 (49–96.1) | 7 | 91.2 (83.5–95.5) | <0.001 |
| 3-year overall survival * | 3 | 60.6 (35.5–81.1) | 7 | 89.9 (78.4–95.6) | <0.001 |
| Grade 3/4 adverse events | 3 | 58.2 (31.8–80.6) | 6 | 23.8 (8.8–50.5) | 0.381 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shih, H.-S.; Jhou, H.-J.; Ou, Y.-H.; Liu, Y.-T.; Kor, C.-T.; Chen, A.W.-G.; Chen, M.-K. The Efficacy and Adverse Events in Patients with Head and Neck Cancer Following Radiotherapy Combined with S-1 Therapy: A Meta-Analysis. Cancers 2021, 13, 2971. https://doi.org/10.3390/cancers13122971
Shih H-S, Jhou H-J, Ou Y-H, Liu Y-T, Kor C-T, Chen AW-G, Chen M-K. The Efficacy and Adverse Events in Patients with Head and Neck Cancer Following Radiotherapy Combined with S-1 Therapy: A Meta-Analysis. Cancers. 2021; 13(12):2971. https://doi.org/10.3390/cancers13122971
Chicago/Turabian StyleShih, Hung-Sheng, Hong-Jie Jhou, Yang-Hao Ou, Yen-Tze Liu, Chew-Teng Kor, Andy Wei-Ge Chen, and Mu-Kuan Chen. 2021. "The Efficacy and Adverse Events in Patients with Head and Neck Cancer Following Radiotherapy Combined with S-1 Therapy: A Meta-Analysis" Cancers 13, no. 12: 2971. https://doi.org/10.3390/cancers13122971
APA StyleShih, H.-S., Jhou, H.-J., Ou, Y.-H., Liu, Y.-T., Kor, C.-T., Chen, A. W.-G., & Chen, M.-K. (2021). The Efficacy and Adverse Events in Patients with Head and Neck Cancer Following Radiotherapy Combined with S-1 Therapy: A Meta-Analysis. Cancers, 13(12), 2971. https://doi.org/10.3390/cancers13122971






