New Pathological and Clinical Insights in Endometrial Cancer in View of the Updated ESGO/ESTRO/ESP Guidelines
Abstract
Simple Summary
Abstract
1. Introduction
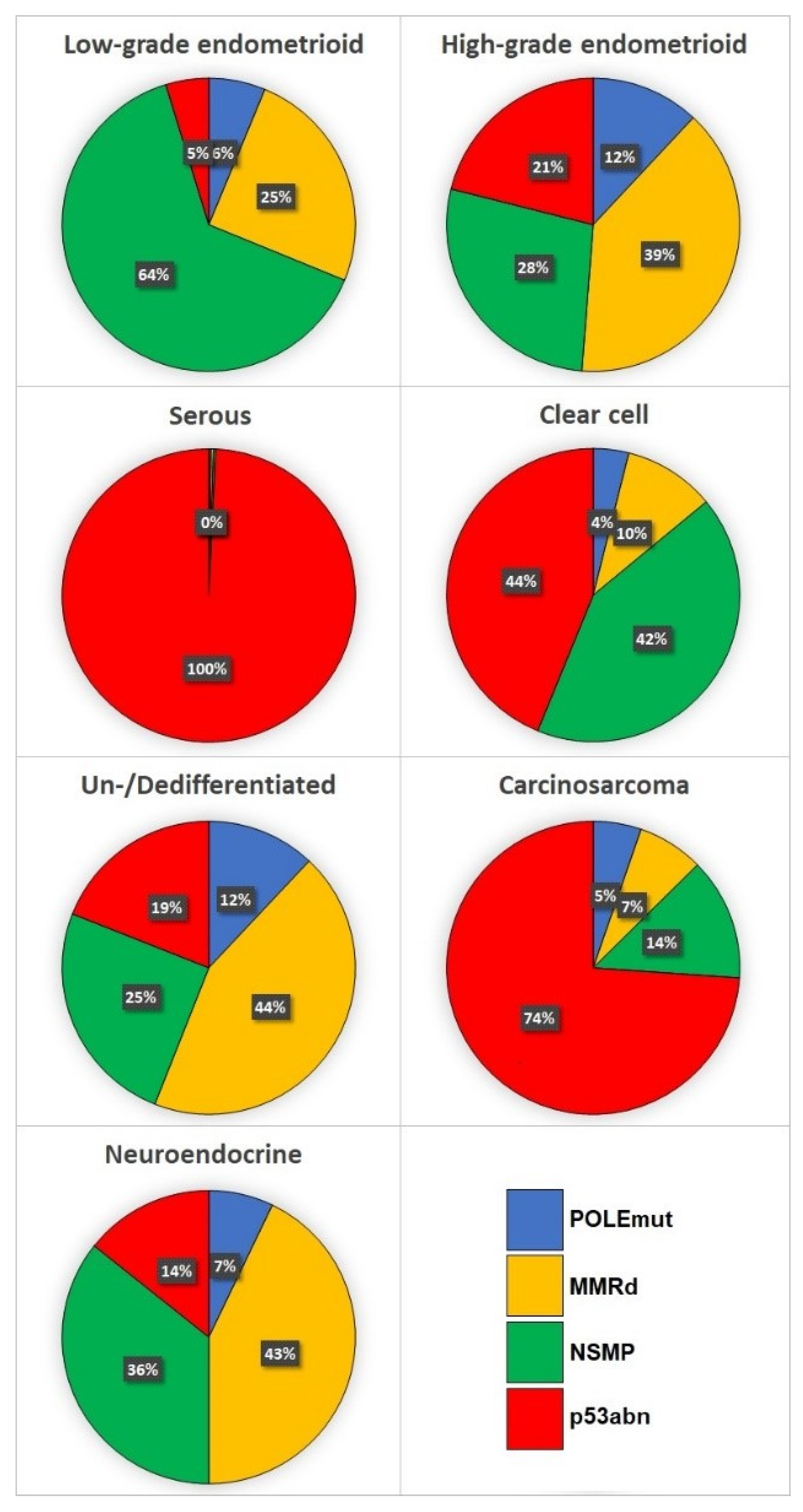
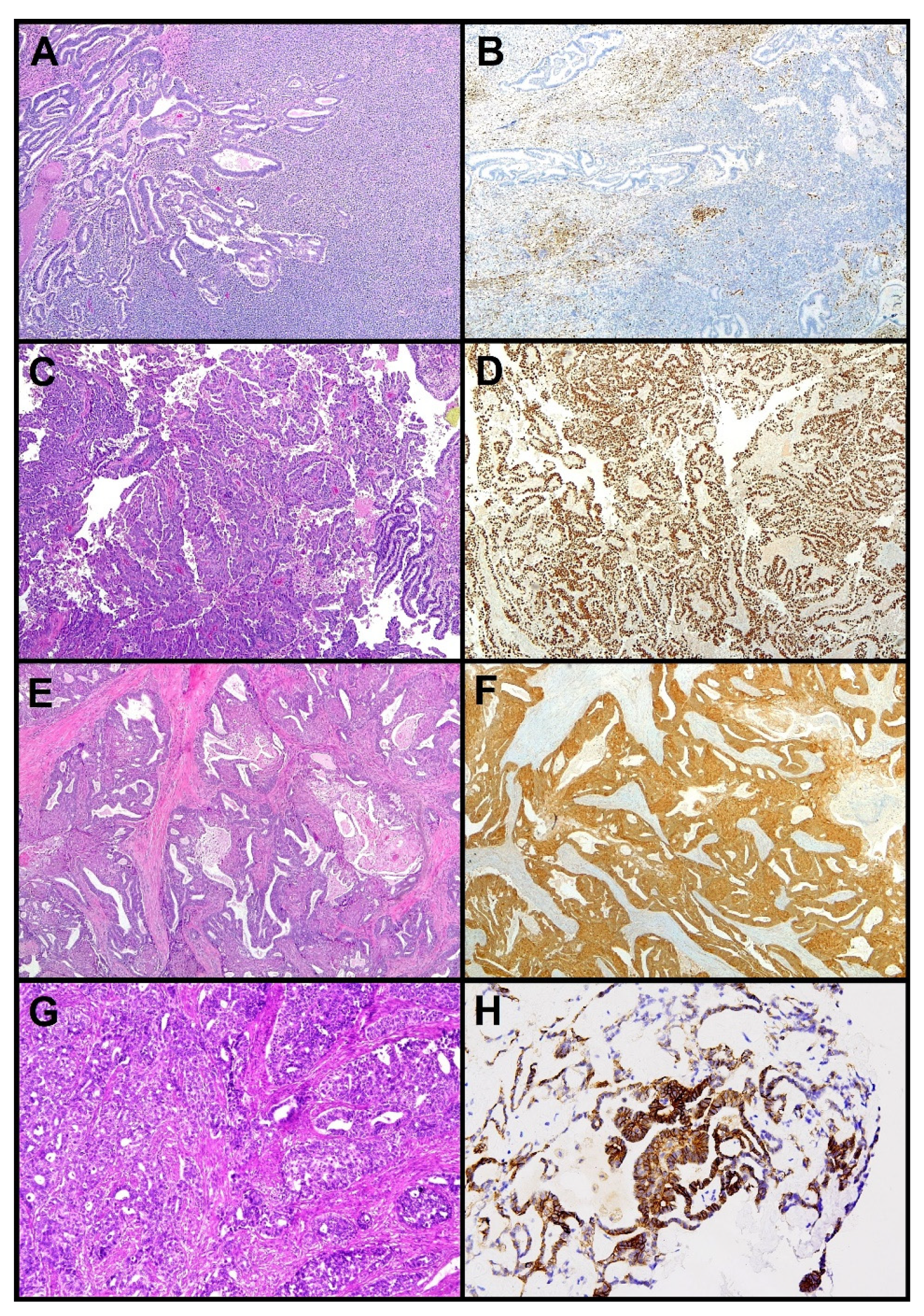
2. Endometrioid Carcinoma (EEC)
3. Serous Carcinoma (SEC)
4. Clear Cell Carcinoma (CCEC)
5. Mixed Carcinoma
6. Undifferentiated/Dedifferentiated Carcinoma (UEC/DEC)
7. Uterine Carcinosarcoma (UCS)
8. Neuroendocrine Carcinoma (NEEC)
9. Mesonephric-Like Carcinoma (MLEC)
10. Gastric-Type Carcinoma (GTEC)
11. Discussion
11.1. Prognostic Consistency of Molecular Groups
11.2. Interpretation Issues and Multiple Classifiers
- -
- Geographical loss of MLH1 and PMS2 due to heterogeneous hypermethylation within the tumor.
- -
- Geographical loss of MSH6 and/or MSH2 due to a secondary (non-germline) mutation in an MSH6 coding mononucleotide tract or a mutation in POLE.
- -
- Weak focal/patchy immunoreactivity for MSH6 can be seen with MSH2 loss of expression/germline mutations.
- -
- Subclonal loss of MMR protein expression.
- -
- MSS/MSI-Low with loss of MMR protein expression due to MLH1 promoter hypermethylation or somatic MMR variants.
- -
- MSI with retained/proficient MMR protein expression due to POLE variants.
11.3. Specific Treatment Options
11.4. CTNNB1-Mutant: The Fifth Molecular Group?
11.5. Unsolved Problems: Preoperative Setting and Interlaboratory Reproducibility
12. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| TCGA | The Cancer Genome Atlas |
| POLE | DNA polymerase epsilon |
| MSI | microsatellite instability |
| ESGO/ESTRO/ESP | European Society of Gynaecological Oncology, European SocieTy for Radiotherapy and Oncology, European Society of Pathology |
| USA | United States of America |
| EEC | endometrial endometrioid carcinoma |
| SEC | serous endometrial carcinoma |
| CCEC | clear cell endometrial carcinoma |
| UEC/DEC | Undifferentiated/dedifferentiated endometrial carcinoma |
| UCS | uterine carcinosarcoma |
| NEEC | neuroendocrine endometrial carcinoma |
| MLEC | mesonephric-like endometrial carcinoma |
| GTEC | gastric/gastrointestinal-type endometrial carcinoma |
| LVSI | lymphovascular space invasion |
| FIGO | International Federation of Gynecology and Obstetrics |
| DNA | Deoxyribonucleic Acid |
| SCNA | somatic copy-number alteration |
| MMR | mismatch repair |
| MMRd | MMR-deficient |
| p53abn | p53-abnormal |
| NSMP | no specific molecular profile |
| AEH/EIN | atypical endometrial hyperplasia/endometrioid intraepithelial neoplasia |
| MELF | microcystic, elongated, and fragmented |
| NCCN | National Comprehensive Cancer Network |
| TCs | Tumor cells |
| ICs | immune cells |
| CPS | combined positive score |
References
- Lortet-Tieulent, J.; Ferlay, J.; Bray, F.; Jemal, A. International Patterns and Trends in Endometrial Cancer Incidence, 1978–2013. J. Natl. Cancer Inst. 2017, 110, 354–361. [Google Scholar] [CrossRef]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Dyba, T.; Randi, G.; Bettio, M.; Gavin, A.; Visser, O.; Bray, F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018. Eur. J. Cancer 2018, 103, 356–387. [Google Scholar] [CrossRef]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef]
- Bokhman, J.V. Two pathogenetic types of endometrial carcinoma. Gynecol. Oncol. 1983, 15, 10–17. [Google Scholar] [CrossRef]
- Murali, R.; Soslow, R.A.; Weigelt, B. Classification of endometrial carcinoma: More than two types. Lancet Oncol. 2014, 15, e268–e278. [Google Scholar] [CrossRef]
- Zannoni, G.F.; Vellone, V.G.; Arena, V.; Prisco, M.G.; Scambia, G.; Carbone, A.; Gallo, D. Does high-grade endometrioid carcinoma (grade 3 FIGO) belong to type I or type II endometrial cancer? A clinical–pathological and immunohistochemical study. Virchows Arch. 2010, 457, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Bae, H.S.; Kim, H.; Kwon, S.Y.; Kim, K.-R.; Song, J.Y.; Kim, I. Should Endometrial Clear Cell Carcinoma be Classified as Type II Endometrial Carcinoma? Int. J. Gynecol. Pathol. 2015, 34, 74–84. [Google Scholar] [CrossRef]
- Colombo, N.; Creutzberg, C.; Amant, F.; Bosse, T.; González-Martín, A.; Ledermann, J.; Marth, C.; Nout, R.; Querleu, D.; Mirza, M.R.; et al. ESMO-ESGO-ESTRO Consensus Conference on Endometrial Cancer: Diagnosis, treatment and follow-up. Ann. Oncol. 2016, 27, 16–41. [Google Scholar] [CrossRef] [PubMed]
- WHO Classification of Tumours Editorial Board. Female Genital Tumours. In WHO Classification of Tumours Series, 5th ed.; International Agency for Research on Cancer: Lyon, France, 2020; Volume 4. [Google Scholar]
- Murali, R.; Delair, D.F.; Bean, S.M.; Abu-Rustum, N.R.; Soslow, R.A. Evolving Roles of Histologic Evaluation and Molecular/Genomic Profiling in the Management of Endometrial Cancer. J. Natl. Compr. Cancer Netw. 2018, 16, 201–209. [Google Scholar] [CrossRef]
- Santoro, A.; Piermattei, A.; Inzani, F.; Angelico, G.; Valente, M.; Arciuolo, D.; Spadola, S.; Martini, M.; Fanfani, F.; Fagotti, A.; et al. Frozen section accurately allows pathological characterization of endometrial cancer in patients with a preoperative ambiguous or inconclusive diagnoses: Our experience. BMC Cancer 2019, 19, 1096. [Google Scholar] [CrossRef] [PubMed]
- Cancer Genome Atlas Research Network; Kandoth, C.; Schultz, N.; Cherniack, A.D.; Akbani, R.; Liu, Y.; Shen, H.; Robertson, A.G.; Pashtan, I.; Shen, R.; et al. Integrated genomic characterization of endometrial carcinoma. Nature 2013, 497, 67–73. [Google Scholar] [CrossRef]
- Raffone, A.; Travaglino, A.; Mascolo, M.; Carbone, L.; Guida, M.; Insabato, L.; Zullo, F. TCGA molecular groups of endometrial cancer: Pooled data about prognosis. Gynecol. Oncol. 2019, 155, 374–383. [Google Scholar] [CrossRef]
- Talhouk, A.; McConechy, M.K.; Leung, S.; Li-Chang, H.H.; Kwon, J.S.; Melnyk, N.; Yang, W.; Senz, J.; Boyd, N.; Karnezis, A.N.; et al. A clinically applicable molecular-based classification for endometrial cancers. Br. J. Cancer 2015, 14, 299–310. [Google Scholar] [CrossRef]
- Talhouk, A.; McConechy, M.K.; Leung, S.; Yang, W.; Lum, A.; Senz, J.; Boyd, N.; Pike, J.; Anglesio, M.; Kwon, J.S.; et al. Confirmation of ProMisE: A simple, genomics-based clinical classifier for endometrial cancer. Cancer 2017, 123, 802–813. [Google Scholar] [CrossRef] [PubMed]
- Kommoss, S.; McConechy, M.; Leung, S.; Bunz, A.; Magrill, J.; Britton, H.; Grevenkamp, F.; Karnezis, A.; Yang, W.; Lum, A.; et al. Final validation of the ProMisE molecular classifier for endometrial carcinoma in a large population-based case series. Ann. Oncol. 2018, 29, 1180–1188. [Google Scholar] [CrossRef] [PubMed]
- Stelloo, E.; Nout, R.A.; Osse, E.M.; Juergenliemk-Schulz, I.J.; Jobsen, J.J.; Lutgens, L.C.; Van Der Steen-Banasik, E.M.; Nijman, H.W.; Putter, H.; Bosse, T.; et al. Improved Risk Assessment by Integrating Molecular and Clinicopathological Factors in Early-stage Endometrial Cancer—Combined Analysis of the PORTEC Cohorts. Clin. Cancer Res. 2016, 22, 4215–4224. [Google Scholar] [CrossRef]
- Concin, N.; Matias-Guiu, X.; Vergote, I.; Cibula, D.; Mirza, M.R.; Marnitz, S.; Ledermann, J.; Bosse, T.; Chargari, C.; Fagotti, A.; et al. ESGO/ESTRO/ESP Guidelines for the management of patients with endometrial carcinoma. Virchows Arch. 2021, 478, 153–190. [Google Scholar] [CrossRef] [PubMed]
- Travaglino, A.; Raffone, A.; Stradella, C.; Esposito, R.; Moretta, P.; Gallo, C.; Orlandi, G.; Insabato, L.; Zullo, F. Impact of endometrial carcinoma histotype on the prognostic value of the TCGA molecular subgroups. Arch. Gynecol. Obstet. 2020, 301, 1355–1363. [Google Scholar] [CrossRef]
- Murali, R.; Davidson, B.; Fadare, O.; Carlson, J.A.; Crum, C.P.; Gilks, C.B.; Irving, J.A.; Malpica, A.; Matias-Guiu, X.; McCluggage, W.G.; et al. High-grade Endometrial Carcinomas: Morphologic and Immunohistochemical Features, Diagnostic Challenges and Recommendations. Int. J. Gynecol. Pathol. 2019, 38 (Suppl. 1), S40–S63. [Google Scholar] [CrossRef]
- Travaglino, A.; Raffone, A.; Mollo, A.; Borrelli, G.; Alfano, P.; Zannoni, G.F.; Insabato, L.; Zullo, F. TCGA molecular subgroups and FIGO grade in endometrial endometrioid carcinoma. Arch. Gynecol. Obstet. 2020, 301, 1117–1125. [Google Scholar] [CrossRef]
- Raffone, A.; Travaglino, A.; Mascolo, M.; Carotenuto, C.; Guida, M.; Mollo, A.; Insabato, L.; Zullo, F. Histopathological characterization of ProMisE molecular groups of endometrial cancer. Gynecol. Oncol. 2020, 157, 252–259. [Google Scholar] [CrossRef]
- Bosse, T.; Nout, R.A.; McAlpine, J.N.; McConechy, M.K.; Britton, H.; Hussein, Y.R.; Gonzalez, C.; Ganesan, R.; Steele, J.C.; Harrison, B.T.; et al. Molecular Classification of Grade 3 Endometrioid Endometrial Cancers Identifies Distinct Prognostic Subgroups. Am. J. Surg. Pathol. 2018, 42, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Brett, M.A.; Atenafu, E.G.; Singh, N.; Ghatage, P.; Clarke, B.A.; Nelson, G.S.; Bernardini, M.Q.; Köbel, M. Equivalent Survival of p53 Mutated Endometrial Endometrioid Carcinoma Grade 3 and Endometrial Serous Carcinoma. Int. J. Gynecol. Pathol. 2021, 40, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, A.; Gupta, P.; Gupta, N.; Rajwanshi, A.; Rai, B.; Gainder, S. Role of Immunohistochemistry to Distinguish Grade 3 Endometrioid Carcinoma and Uterine Serous Carcinoma. Appl. Immunohistochem. Mol. Morphol. 2020, 28, 42–48. [Google Scholar] [CrossRef]
- Tortorella, L.; Restaino, S.; Zannoni, G.F.; Vizzielli, G.; Chiantera, V.; Cappuccio, S.; Gioè, A.; La Fera, E.; Dinoi, G.; Angelico, G.; et al. Substantial lymph-vascular space invasion (LVSI) as predictor of distant relapse and poor prognosis in low-risk early-stage endometrial cancer. J. Gynecol. Oncol. 2021, 32, e11. [Google Scholar] [CrossRef]
- Pasanen, A.; Loukovaara, M.; Bützow, R. Clinicopathological significance of deficient DNA mismatch repair and MLH1 promoter methylation in endometrioid endometrial carcinoma. Mod. Pathol. 2020, 33, 1443–1452. [Google Scholar] [CrossRef] [PubMed]
- Van Den Heerik, A.S.V.; Horeweg, N.; Nout, R.A.; Lutgens, L.C.; Van Der Steen-Banasik, E.M.; Westerveld, G.H.; Van Den Berg, H.A.; Slot, A.; Koppe, F.L.; Kommoss, S.; et al. PORTEC-4a: International randomized trial of molecular profile-based adjuvant treatment for women with high-intermediate risk endometrial cancer. Int. J. Gynecol. Cancer 2020, 30, 2002–2007. [Google Scholar] [CrossRef] [PubMed]
- Rau, T.T.; Bettschen, E.; Büchi, C.; Christe, L.; Rohner, A.; Müller, M.D.; Carlson, J.W.; Imboden, S.; Zlobec, I. Prognostic impact of tumor budding in endometrial carcinoma within distinct molecular subgroups. Mod. Pathol. 2021, 34, 222–232. [Google Scholar] [CrossRef]
- Santoro, A.; Angelico, G.; Inzani, F.; Spadola, S.; Arciuolo, D.; Valente, M.; Musarra, T.; Capelli, G.; Fanfani, F.; Gallotta, V.; et al. Pathological features, immunoprofile and mismatch repair protein expression status in uterine endometrioid carcinoma: Focus on MELF pattern of myoinvasion. Eur. J. Surg. Oncol. 2021, 47, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Angelico, G.; Santoro, A.; Straccia, P.; Inzani, F.; Cianfrini, F.; Spadola, S.; Arciuolo, D.; Valente, M.; D’Alessandris, N.; Mulè, A.; et al. Diagnostic and Prognostic Role of WT1 Immunohistochemical Expression in Uterine Carcinoma: A Systematic Review and Meta-Analysis across All Endometrial Carcinoma Histotypes. Diagnostics 2020, 10, 637. [Google Scholar] [CrossRef]
- Zheng, W.; Xiang, L.; Fadare, O.; Kong, B. A Proposed Model for Endometrial Serous Carcinogenesis. Am. J. Surg. Pathol. 2011, 35, e1–e14. [Google Scholar] [CrossRef]
- Stelloo, E.; Bosse, T.; Nout, R.A.; Mackay, H.J.; Church, D.N.; Nijman, H.W.; Leary, A.; Edmondson, R.J.; Powell, M.E.; Crosbie, E.J.; et al. Refining prognosis and identifying targetable pathways for high-risk endometrial cancer; a TransPORTEC initiative. Mod. Pathol. 2015, 28, 836–844. [Google Scholar] [CrossRef]
- Joehlin-Price, A.; Van Ziffle, J.; Hills, N.K.; Ladwig, N.; Rabban, J.T.; Garg, K. Molecularly Classified Uterine FIGO Grade 3 Endometrioid Carcinomas Show Distinctive Clinical Outcomes But Overlapping Morphologic Features. Am. J. Surg. Pathol. 2021, 45, 421–429. [Google Scholar] [CrossRef]
- Conlon, N.; Da Cruz Paula, A.; Ashley, C.W.; Segura, S.; De Brot, L.; da Silva, E.M.; Soslow, R.A.; Weigelt, B.; DeLair, D.F. Endometrial Carcinomas with a “Serous” Component in Young Women Are Enriched for DNA Mismatch Repair Deficiency, Lynch Syndrome, and POLE Exonuclease Domain Mutations. Am. J. Surg. Pathol. 2020, 44, 641–648. [Google Scholar] [CrossRef]
- Lax, S.F. Molecular genetic pathways in various types of endometrial carcinoma: From a phenotypical to a molecular-based classification. Virchows Arch. 2004, 444, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Fadare, O.; Liang, S.X.; Ulukus, E.C.; Chambers, S.K.; Zheng, W. Precursors of Endometrial Clear Cell Carcinoma. Am. J. Surg. Pathol. 2006, 30, 1519–1530. [Google Scholar] [CrossRef]
- Pors, J.; Segura, S.; Cheng, A.; Ji, J.X.; Tessier-Cloutier, B.; Cochrane, D.; Fix, D.J.; Park, K.; Gilks, B.; Hoang, L. Napsin-A and AMACR are Superior to HNF-1β in Distinguishing Between Mesonephric Carcinomas and Clear Cell Carcinomas of the Gynecologic Tract. Appl. Immunohistochem. Mol. Morphol. 2020, 28, 593–601. [Google Scholar] [CrossRef]
- Hoang, L.N.; McConechy, M.K.; Meng, B.; McIntyre, J.B.; Ewanowich, C.; Gilks, C.B.; Huntsman, D.G.; Köbel, M.; Lee, C.-H. Targeted mutation analysis of endometrial clear cell carcinoma. Histopathology 2014, 66, 664–674. [Google Scholar] [CrossRef] [PubMed]
- Travaglino, A.; Raffone, A.; Mascolo, M.; Guida, M.; Insabato, L.; Zannoni, G.F.; Zullo, F. Clear cell endometrial carcinoma and the TCGA classification. Histopathology 2020, 76, 336–338. [Google Scholar] [CrossRef]
- Zannoni, G.F.; Santoro, A.; Angelico, G.; Spadola, S.; Arciuolo, D.; Valente, M.; Inzani, F.; Pettinato, A.; Vatrano, S.; Fanfani, F.; et al. Clear cell carcinoma of the endometrium: An immunohistochemical and molecular analysis of 45 cases. Hum. Pathol. 2019, 92, 10–17. [Google Scholar] [CrossRef]
- Köbel, M.; Tessier-Cloutier, B.; Leo, J.; Hoang, L.N.; Gilks, C.B.; Soslow, R.A.; Delair, D.; Stewart, C.J.; Lee, C.-H. Frequent Mismatch Repair Protein Deficiency in Mixed Endometrioid and Clear Cell Carcinoma of the Endometrium. Int. J. Gynecol. Pathol. 2017, 36, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.R.; Cloutier, B.T.; Leung, S.; Cochrane, D.; Britton, H.; Pina, A.; Storness-Bliss, C.; Farnell, D.; Huang, L.; Shum, K.; et al. Molecular subtypes of clear cell carcinoma of the endometrium: Opportunities for prognostic and predictive stratification. Gynecol. Oncol. 2020, 158, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Delair, D.F.; Burke, K.A.; Selenica, P.; Lim, R.S.; Scott, S.N.; Middha, S.; Mohanty, A.S.; Cheng, D.T.; Berger, M.F.; Soslow, R.A.; et al. The genetic landscape of endometrial clear cell carcinomas. J. Pathol. 2017, 243, 230–241. [Google Scholar] [CrossRef] [PubMed]
- Ida, N.; Nakamura, K.; Saijo, M.; Nasu, A.; Yoshino, T.; Masuyama, H.; Yanai, H. DNA mismatch repair deficiency and p53 abnormality are age-related events in mixed endometrial carcinoma with a clear cell component. Pathol. Res. Pract. 2021, 220, 153383. [Google Scholar] [CrossRef]
- Hagemann, I.S.; Deng, W.; Zaino, R.J.; Powell, M.A.; Gunderson, C.; Cosgrove, C.; Mathews, C.; Pearl, M.L.; Waggoner, S.; Ghebre, R.; et al. The presence of an endometrioid component does not alter the clinicopathologic profile or survival of patients with uterine serous cancer: A gynecologic oncology group (GOG/NRG) study of 934 women. Gynecol. Oncol. 2021, 160, 660–668. [Google Scholar] [CrossRef]
- Matrai, C.; Motanagh, S.; Mirabelli, S.; Ma, L.; He, B.; Chapman-Davis, E.; Kurtis, B.; Elemento, O.; Mosquera, J.M.; Ellenson, L.H. Molecular Profiles of Mixed Endometrial Carcinoma. Am. J. Surg. Pathol. 2020, 44, 1104–1111. [Google Scholar] [CrossRef]
- Van Esterik, M.; Van Gool, I.C.; de Kroon, C.D.; Nout, R.A.; Creutzberg, C.L.; Smit, V.T.; Bosse, T.; Stelloo, E. Limited impact of intratumour heterogeneity on molecular risk assignment in endometrial cancer. Oncotarget 2017, 8, 25542–25551. [Google Scholar] [CrossRef]
- Kihara, A.; Amano, Y.; Matsubara, D.; Fukushima, N.; Fujiwara, H.; Niki, T. BRG1, INI1, and ARID1B Deficiency in Endometrial Carcinoma: A Clinicopathologic and Immunohistochemical Analysis of a Large Series from a Single Institution. Am. J. Surg. Pathol. 2020, 44, 1712–1724. [Google Scholar] [CrossRef]
- Busca, A.; Parra-Herran, C.; Nofech-Mozes, S.; Djordjevic, B.; Ismiil, N.; Cesari, M.; Nucci, M.R.; Mirkovic, J. Undifferentiated endometrial carcinoma arising in the background of high-grade endometrial carcinoma—Expanding the definition of dedifferentiated endometrial carcinoma. Histopathology 2020, 77, 769–780. [Google Scholar] [CrossRef]
- Tafe, L.J.; Garg, K.; Chew, I.; Tornos, C.; Soslow, R.A. Endometrial and ovarian carcinomas with undifferentiated components: Clinically aggressive and frequently underrecognized neoplasms. Mod. Pathol. 2010, 23, 781–789. [Google Scholar] [CrossRef] [PubMed]
- Santoro, A.; Angelico, G.; Travaglino, A.; Raffone, A.; Arciuolo, D.; D’Alessandris, N.; Inzani, F.; Zannoni, G.F. Clinico-pathological significance of TCGA classification and SWI/SNF proteins expression in undifferentiated/dedifferentiated endometrial carcinoma: A possible prognostic risk stratification. Gynecol. Oncol. 2021, 161, 629–635. [Google Scholar] [CrossRef]
- Travaglino, A.; Raffone, A.; Mascolo, M.; Guida, M.; Insabato, L.; Zannoni, G.F.; Zullo, F. TCGA Molecular Subgroups in Endometrial Undifferentiated/Dedifferentiated Carcinoma. Pathol. Oncol. Res. 2019, 26, 1411–1416. [Google Scholar] [CrossRef]
- Abu-Rustum, N.R.; Yashar, C.M.; Bradley, K. Clinical Practice Guidelines in Oncology (NCCN Guidelines®®)—Uterine Neoplasms; Version 2.2021—05/07/2021; NCCN: Plymouth Meeting, PA, USA, 2021. [Google Scholar]
- Segura, S.E.; Nobre, S.P.; Hussein, Y.R.; Abu-Rustum, N.R.; Weigelt, B.; Soslow, R.A.; Delair, D.F. DNA Mismatch Repair–deficient Endometrial Carcinosarcomas Portend Distinct Clinical, Morphologic, and Molecular Features Compared With Traditional Carcinosarcomas. Am. J. Surg. Pathol. 2020, 44, 1573–1579. [Google Scholar] [CrossRef]
- Kurman, R.; Carcangiu, M.; Herrington, C.; Young, R. World Health Organization Classification of Tumors of Female Reproductive Organs, 4th ed.; International Agency for Research on Cancer (IARC) Press: Lyon, France, 2014. [Google Scholar]
- Nam, J.-H.; Park, J.-Y. Update on treatment of uterine sarcoma. Curr. Opin. Obstet. Gynecol. 2010, 22, 36–42. [Google Scholar] [CrossRef]
- Matsuzaki, S.; Klar, M.; Matsuzaki, S.; Roman, L.D.; Sood, A.K.; Matsuo, K. Uterine carcinosarcoma: Contemporary clinical summary, molecular updates, and future research opportunity. Gynecol. Oncol. 2021, 160, 586–601. [Google Scholar] [CrossRef]
- Travaglino, A.; Raffone, A.; Gencarelli, A.; Mollo, A.; Guida, M.; Insabato, L.; Santoro, A.; Zannoni, G.F.; Zullo, F. TCGA Classification of Endometrial Cancer: The Place of Carcinosarcoma. Pathol. Oncol. Res. 2020, 26, 2067–2073. [Google Scholar] [CrossRef]
- Taskin, O.Ç.; Onder, S.; Topuz, S.; Sozen, H.; Sen, F.; Ilhan, R.; Yavuz, E. A Selected Immunohistochemical Panel Aids in Differential Diagnosis and Prognostic Stratification of Subtypes of High-grade Endometrial Carcinoma: A Clinicopathologic and Immunohistochemical Study at a Single Institution. Appl. Immunohistochem. Mol. Morphol. 2017, 25, 696–702. [Google Scholar] [CrossRef]
- Zhang, C.; Hu, W.; Jia, N.; Li, Q.; Hua, K.; Tao, X.; Wang, L.; Feng, W. Uterine Carcinosarcoma and High-Risk Endometrial Carcinomas: A Clinicopathological Comparison. Int. J. Gynecol. Cancer 2015, 25, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Prueksaritanond, N.; Chantape, W. Comparative Survival Outcomes of Uterine Papillary Serous Carcinoma, Clear Cell Carcinoma, Grade 3 Endometrioid Adenocarcinoma, and Carcinosarcoma of Endometrial Cancer in Rajavithi Hospital. J. Med. Assoc. Thail. Chotmaihet Thangphaet 2016, 99, 75–83. [Google Scholar]
- McConechy, M.K.; Hoang, L.N.; Chui, M.H.; Senz, J.; Yang, W.; Rozenberg, N.; MacKenzie, R.; McAlpine, J.N.; Huntsman, D.G.; Clarke, B.A.; et al. In-depth molecular profiling of the biphasic components of uterine carcinosarcomas. J. Pathol. Clin. Res. 2015, 1, 173–185. [Google Scholar] [CrossRef]
- Cherniack, A.D.; Shen, H.; Walter, V.; Stewart, C.; Murray, B.A.; Bowlby, R.; Hu, X.; Ling, S.; Soslow, R.A.; Broaddus, R.R.; et al. Integrated Molecular Characterization of Uterine Carcinosarcoma. Cancer Cell 2017, 31, 411–423. [Google Scholar] [CrossRef]
- Jones, T.E.; Pradhan, D.; Dabbs, D.J.; Bhargava, R.; Onisko, A.; Jones, M.W. Immunohistochemical Markers With Potential Diagnostic, Prognostic, and Therapeutic Significance in Uterine Carcinosarcoma: A Clinicopathologic Study of 43 Cases. Int. J. Gynecol. Pathol. 2021, 40, 84–93. [Google Scholar] [CrossRef]
- Howitt, B.E.; Dong, F.; Vivero, M.; Shah, V.; Lindeman, N.; Schoolmeester, J.K.; Baltay, M.; MacConaill, L.; Sholl, L.M.; Nucci, M.R.; et al. Molecular Characterization of Neuroendocrine Carcinomas of the Endometrium: Representation in All 4 TCGA Groups. Am. J. Surg. Pathol. 2020, 44, 1541–1548. [Google Scholar] [CrossRef]
- Inzani, F.; Santoro, A.; Angelico, G.; Feraco, A.; Spadola, S.; Arciuolo, D.; Valente, M.; Carlino, A.; Piermattei, A.; Scaglione, G.; et al. Neuroendocrine Carcinoma of the Uterine Cervix: A Clinicopathologic and Immunohistochemical Study with Focus on Novel Markers (Sst2–Sst5). Cancers 2020, 12, 1211. [Google Scholar] [CrossRef]
- Pocrnich, C.E.; Ramalingam, P.; Euscher, E.D.; Malpica, A. Neuroendocrine Carcinoma of the Endometrium: A Clinicopathologic Study of 25 Cases. Am. J. Surg. Pathol. 2016, 40, 577–586. [Google Scholar] [CrossRef]
- George, J.; Lim, J.S.; Jang, S.J.; Cun, Y.; Ozretić, L.; Kong, G.; Leenders, F.; Lu, X.; Fernández-Cuesta, L.; Bosco, G.; et al. Comprehensive genomic profiles of small cell lung cancer. Nat. Cell Biol. 2015, 524, 47–53. [Google Scholar] [CrossRef]
- Pors, J.; Cheng, A.; Leo, J.M.; Kinloch, M.A.; Gilks, B.; Hoang, L. A Comparison of GATA3, TTF1, CD10, and Calretinin in Identifying Mesonephric and Mesonephric-like Carcinomas of the Gynecologic Tract. Am. J. Surg. Pathol. 2018, 42, 1596–1606. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, E.M.; Fix, D.J.; Sebastiao, A.P.M.; Selenica, P.; Ferrando, L.; Kim, S.H.; Stylianou, A.; Paula, A.D.C.; Pareja, F.; Smith, E.S.; et al. Mesonephric and mesonephric-like carcinomas of the female genital tract: Molecular characterization including cases with mixed histology and matched metastases. Mod. Pathol. 2021, 1–18. [Google Scholar] [CrossRef]
- Horn, L.-C.; Höhn, A.K.; Krücken, I.; Stiller, M.; Obeck, U.; Brambs, C.E. Mesonephric-like adenocarcinomas of the uterine corpus: Report of a case series and review of the literature indicating poor prognosis for this subtype of endometrial adenocarcinoma. J. Cancer Res. Clin. Oncol. 2020, 146, 971–983. [Google Scholar] [CrossRef] [PubMed]
- Pors, J.; Segura, S.; Chiu, D.S.; Almadani, N.; Ren, H.; Fix, D.J.; Howitt, B.E.; Kolin, D.; McCluggage, W.G.; Mirkovic, J.; et al. Clinicopathologic Characteristics of Mesonephric Adenocarcinomas and Mesonephric-like Adenocarcinomas in the Gynecologic Tract: A Multi-institutional Study. Am. J. Surg. Pathol. 2021, 45, 498–506. [Google Scholar] [CrossRef]
- Wong, R.W.; Ralte, A.; Grondin, K.; Talia, K.L.; McCluggage, W.G. Endometrial Gastric (Gastrointestinal)-type Mucinous Lesions: Report of a Series Illustrating the Spectrum of Benign and Malignant Lesions. Am. J. Surg. Pathol. 2020, 44, 406–419. [Google Scholar] [CrossRef] [PubMed]
- Travaglino, A.; Raffone, A.; Gencarelli, A.; Mollo, A.; Zullo, F.; Insabato, L. Endometrial Gastric-type Carcinoma: An Aggressive and Morphologically Heterogenous New Histotype Arising From Gastric Metaplasia of the Endometrium. Am. J. Surg. Pathol. 2020, 44, 1002–1004. [Google Scholar] [CrossRef]
- Wong, R.W.; Talia, K.L.; McCluggage, W.G. Endometrial Gastric-type Carcinoma: An Aggressive and Morphologically Heterogenous New Histotype Arising from Gastric Metaplasia of the Endometrium. Am. J. Surg. Pathol. 2020, 44, 1736–1737. [Google Scholar] [CrossRef] [PubMed]
- Selenica, P.; Alemar, B.; Matrai, C.; Talia, K.L.; Veras, E.; Hussein, Y.; Oliva, E.; Beets-Tan, R.G.H.; Mikami, Y.; McCluggage, W.G.; et al. Massively parallel sequencing analysis of 68 gastric-type cervical adenocarcinomas reveals mutations in cell cycle-related genes and potentially targetable mutations. Mod. Pathol. 2021, 34, 1213–1225. [Google Scholar] [CrossRef]
- Ardighieri, L.; Palicelli, A.; Ferrari, F.; Bugatti, M.; Drera, E.; Sartori, E.; Odicino, F. Endometrial Carcinomas with Intestinal-Type Metaplasia/Differentiation: Does Mismatch Repair System Defects Matter? Case Report and Systematic Review of the Literature. J. Clin. Med. 2020, 9, 2552. [Google Scholar] [CrossRef]
- McAlpine, J.; Leon-Castillo, A.; Bosse, T. The rise of a novel classification system for endometrial carcinoma; integration of molecular subclasses. J. Pathol. 2018, 244, 538–549. [Google Scholar] [CrossRef] [PubMed]
- Raffone, A.; Travaglino, A.; Gabrielli, O.; Micheli, M.; Zuccalà, V.; Bitonti, G.; Camastra, C.; Gargiulo, V.; Insabato, L.; Zullo, F. Clinical features of ProMisE groups identify different phenotypes of patients with endometrial cancer. Arch. Gynecol. Obstet. 2021, 303, 1393–1400. [Google Scholar] [CrossRef]
- Moroney, M.R.; Davies, K.D.; Wilberger, A.C.; Sheeder, J.; Post, M.D.; Berning, A.A.; Fisher, C.; Lefkowits, C.; Guntupalli, S.R.; Behbakht, K.; et al. Molecular markers in recurrent stage I, grade 1 endometrioid endometrial cancers. Gynecol. Oncol. 2019, 153, 517–520. [Google Scholar] [CrossRef] [PubMed]
- Travaglino, A.; Raffone, A.; Gencarelli, A.; Saracinelli, S.; Riccardi, C.; Mollo, A.; Zullo, F.; Insabato, L. Clinico-pathological features associated with mismatch repair deficiency in endometrial undifferentiated/dedifferentiated carcinoma: A systematic review and meta-analysis. Gynecol. Oncol. 2021, 160, 579–585. [Google Scholar] [CrossRef]
- León-Castillo, A.; Britton, H.; McConechy, M.K.; McAlpine, J.N.; Nout, R.; Kommoss, S.; Brucker, S.Y.; Carlson, J.W.; Epstein, E.; Rau, T.T.; et al. Interpretation of somatic POLE mutations in endometrial carcinoma. J. Pathol. 2020, 250, 323–335. [Google Scholar] [CrossRef]
- Raffone, A.; Travaglino, A.; Cerbone, M.; De Luca, C.; Russo, D.; Di Maio, A.; De Marco, M.; Turco, M.C.; Insabato, L.; Zullo, F. Diagnostic accuracy of p53 immunohistochemistry as surrogate of TP53 sequencing in endometrial cancer. Pathol. Res. Pract. 2020, 216, 153025. [Google Scholar] [CrossRef]
- Köbel, M.; Ronnett, B.M.; Singh, N.; Soslow, R.A.; Gilks, C.B.; McCluggage, W.G. Interpretation of P53 Immunohistochemistry in Endometrial Carcinomas: Toward Increased Reproducibility. Int. J. Gynecol. Pathol. 2019, 38 (Suppl. 1), S123–S131. [Google Scholar] [CrossRef]
- Singh, N.; Wong, R.; Tchrakian, N.; Allen, S.G.; Clarke, B.; Gilks, B. The British Association of Gynaecological Pathologists—Interpretation and Reporting Terminology for Mismatch Repair Protein Immunohistochemistry in Endometrial Cancer. In BAGP Guidance Document: MMR Immunohistochemistry Interpretation and Terminology; Version 1.1; Histopathology Department University Hospitals Derby and Burton NHS Trust: Derby, UK, 2020. [Google Scholar]
- Raffone, A.; Travaglino, A.; Cerbone, M.; Gencarelli, A.; Mollo, A.; Insabato, L.; Zullo, F. Diagnostic Accuracy of Immunohistochemistry for Mismatch Repair Proteins as Surrogate of Microsatellite Instability Molecular Testing in Endometrial Cancer. Pathol. Oncol. Res. 2020, 26, 1417–1427. [Google Scholar] [CrossRef] [PubMed]
- Stelloo, E.; Jansen, A.M.L.; Osse, E.M.; Nout, R.A.; Creutzberg, C.L.; Ruano, D.; Church, D.N.; Morreau, H.; Smit, V.T.H.B.M.; van Wezel, T.; et al. Practical guidance for mismatch repair-deficiency testing in endometrial cancer. Ann. Oncol. 2017, 28, 96–102. [Google Scholar] [CrossRef]
- León-Castillo, A.; Gilvazquez, E.; Nout, R.; Smit, V.T.; McAlpine, J.N.; McConechy, M.; Kommoss, S.; Brucker, S.Y.; Carlson, J.W.; Epstein, E. Clinicopathological and molecular characterisation of ‘multiple-classifier’ endometrial carcinomas. J. Pathol. 2020, 250, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Marabelle, A.; Fakih, M.; Lopez, J.; Shah, M.; Shapira-Frommer, R.; Nakagawa, K.; Chung, H.C.; Kindler, H.L.; A Lopez-Martin, J.; Miller, W.H.; et al. Association of tumour mutational burden with outcomes in patients with advanced solid tumours treated with pembrolizumab: Prospective biomarker analysis of the multicohort, open-label, phase 2 KEYNOTE-158 study. Lancet Oncol. 2020, 21, 1353–1365. [Google Scholar] [CrossRef]
- Pasanen, A.; Ahvenainen, T.; Pellinen, T.; Vahteristo, P.; Loukovaara, M.; Bützow, R. PD-L1 Expression in Endometrial Carcinoma Cells and Intratumoral Immune Cells: Differences Across Histologic and TCGA-based Molecular Subgroups. Am. J. Surg. Pathol. 2020, 44, 174–181. [Google Scholar] [CrossRef]
- Banet, N.; Shahi, M.; Batista, D.; Yonescu, R.; Tanner, E.J.; Fader, A.N.; Cimino-Mathews, A. HER-2 Amplification in Uterine Serous Carcinoma and Serous Endometrial Intraepithelial Carcinoma. Am. J. Surg. Pathol. 2021, 45, 708–715. [Google Scholar] [CrossRef]
- Fader, A.N.; Roque, D.M.; Siegel, E.; Buza, N.; Hui, P.; Abdelghany, O.; Chambers, S.K.; Secord, A.A.; Havrilesky, L.; O’Malley, D.M.; et al. Randomized Phase II Trial of Carboplatin-Paclitaxel Versus Carboplatin-Paclitaxel-Trastuzumab in Uterine Serous Carcinomas That Overexpress Human Epidermal Growth Factor Receptor 2/neu. J. Clin. Oncol. 2018, 36, 2044–2051. [Google Scholar] [CrossRef] [PubMed]
- Rottmann, D.; Snir, O.L.; Wu, X.; Wong, S.; Hui, P.; Santin, A.D.; Buza, N. HER2 testing of gynecologic carcinosarcomas: Tumor stratification for potential targeted therapy. Mod. Pathol. 2020, 33, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Cagaanan, A.; Stelter, B.; Vu, N.; Rhode, E.N.; Stewart, T.; Hui, P.; Buza, N.; Al-Niaimi, A.; Flynn, C.; Weisman, P.S.; et al. HER2 Expression in Endometrial Cancers Diagnosed as Clear Cell Carcinoma. Int. J. Gynecol. Pathol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Auguste, A.; Genestie, C.; De Bruyn, M.; Adam, J.; Le Formal, A.; Drusch, F.; Pautier, P.; Crosbie, E.J.; Mackay, H.; Kitchener, H.C.; et al. Refinement of high-risk endometrial cancer classification using DNA damage response biomarkers: A TransPORTEC initiative. Mod. Pathol. 2018, 31, 1851–1861. [Google Scholar] [CrossRef] [PubMed]
- Kurnit, K.; Kim, G.N.; Fellman, B.M.; Urbauer, D.L.; Mills, G.B.; Zhang, W.; Broaddus, R.R. CTNNB1 (beta-catenin) mutation identifies low grade, early stage endometrial cancer patients at increased risk of recurrence. Mod. Pathol. 2017, 30, 1032–1041. [Google Scholar] [CrossRef]
- Zannoni, G.F.; Angelico, G.; Santoro, A. Aberrant non-canonical WNT pathway as key-driver of high-grade serous ovarian cancer development. Virchows Arch. 2020, 477, 321–322. [Google Scholar] [CrossRef] [PubMed]
- Travaglino, A.; Raffone, A.; Saccone, G.; De Luca, C.; Mollo, A.; Mascolo, M.; De Placido, G.; Insabato, L.; Zullo, F. Immunohistochemical Nuclear Expression of β-Catenin as a Surrogate of CTNNB1 Exon 3 Mutation in Endometrial Cancer. Am. J. Clin. Pathol. 2019, 151, 529–538. [Google Scholar] [CrossRef]
- Kim, G.; Kurnit, K.; Djordjevic, B.; Singh, C.; Munsell, M.F.; Wang, W.-L.; Lazar, A.J.; Zhang, W.; Broaddus, R. Nuclear β-catenin localization and mutation of the CTNNB1 gene: A context-dependent association. Mod. Pathol. 2018, 31, 1553–1559. [Google Scholar] [CrossRef]
- Meljen, V.T.; Mittenzwei, R.; Wong, J.; Puechl, A.; Whitaker, R.; Broadwater, G.; Hall, A.H.; Bean, S.M.; Bentley, R.C.; Elvin, J.A.; et al. Endometrial Adenocarcinomas With No Specific Molecular Profile. Int. J. Gynecol. Pathol. 2021. [Google Scholar] [CrossRef]
- Costigan, D.C.; Dong, F.; Nucci, M.R.; Howitt, B.E. Clinicopathologic and Immunohistochemical Correlates of CTNNB1 Mutated Endometrial Endometrioid Carcinoma. Int. J. Gynecol. Pathol. 2020, 39, 119–127. [Google Scholar] [CrossRef]
- Travaglino, A.; Raffone, A.; Gencarelli, A.; Raimondo, D.; Moretta, P.; Pignatiello, S.; Granata, M.; Seracchioli, R.; Zullo, F.; Insabato, L. Relationship between morular metaplasia and squamous differentiation in endometrial carcinoma. Pathol. Res. Pr. 2021, 217, 153307. [Google Scholar] [CrossRef]
- Abdulfatah, E.; Wakeling, E.; Sakr, S.; Al-Obaidy, K.; Bandyopadhyay, S.; Morris, R.; Feldman, G.; Ali-Fehmi, R. Molecular classification of endometrial carcinoma applied to endometrial biopsy specimens: Towards early personalized patient management. Gynecol. Oncol. 2019, 154, 467–474. [Google Scholar] [CrossRef]
- Plotkin, A.; Kuzeljevic, B.; De Villa, V.; Thompson, E.F.; Gilks, C.B.; Clarke, B.A.; Köbel, M.; McAlpine, J.N. Interlaboratory Concordance of ProMisE Molecular Classification of Endometrial Carcinoma Based on Endometrial Biopsy Specimens. Int. J. Gynecol. Pathol. 2019, 39. [Google Scholar] [CrossRef] [PubMed]
| LOW RISK | Stage I-II POLEmut Stage IA MMRd/NSMP low-grade EEC with no or focal LVSI |
| INTERMEDIATE RISK | Stage IB MMRd/NSMP low-grade EEC with no or focal LVSI Stage IA MMRd/NSMP high-grade EEC with no or focal LVSI Stage IA p53abn EEC with no myometrial invasion Stage IA non-endometrioid with no myometrial invasion |
| HIGH-INTERMEDIATE RISK | Stage I MMRd/NSMP EEC with LVSI Sage IB MMRd/NSMP high-grade EEC Stage II MMRd/NSMP EEC |
| HIGH RISK | Stage III–IVa EEC with no residual disease Stage I–IVa p53abn EEC with myometrial invasion and no residual disease Stage I–IVa non-endometrioid * with myometrial invasion and no residual disease |
| ADVANCED/METASTATIC | Stage III–Iva with residual disease Stage IVb |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santoro, A.; Angelico, G.; Travaglino, A.; Inzani, F.; Arciuolo, D.; Valente, M.; D’Alessandris, N.; Scaglione, G.; Fiorentino, V.; Raffone, A.; et al. New Pathological and Clinical Insights in Endometrial Cancer in View of the Updated ESGO/ESTRO/ESP Guidelines. Cancers 2021, 13, 2623. https://doi.org/10.3390/cancers13112623
Santoro A, Angelico G, Travaglino A, Inzani F, Arciuolo D, Valente M, D’Alessandris N, Scaglione G, Fiorentino V, Raffone A, et al. New Pathological and Clinical Insights in Endometrial Cancer in View of the Updated ESGO/ESTRO/ESP Guidelines. Cancers. 2021; 13(11):2623. https://doi.org/10.3390/cancers13112623
Chicago/Turabian StyleSantoro, Angela, Giuseppe Angelico, Antonio Travaglino, Frediano Inzani, Damiano Arciuolo, Michele Valente, Nicoletta D’Alessandris, Giulia Scaglione, Vincenzo Fiorentino, Antonio Raffone, and et al. 2021. "New Pathological and Clinical Insights in Endometrial Cancer in View of the Updated ESGO/ESTRO/ESP Guidelines" Cancers 13, no. 11: 2623. https://doi.org/10.3390/cancers13112623
APA StyleSantoro, A., Angelico, G., Travaglino, A., Inzani, F., Arciuolo, D., Valente, M., D’Alessandris, N., Scaglione, G., Fiorentino, V., Raffone, A., & Zannoni, G. F. (2021). New Pathological and Clinical Insights in Endometrial Cancer in View of the Updated ESGO/ESTRO/ESP Guidelines. Cancers, 13(11), 2623. https://doi.org/10.3390/cancers13112623









