Emerging Role of Fascin-1 in the Pathogenesis, Diagnosis, and Treatment of the Gastrointestinal Cancers
Abstract
Simple Summary
Abstract
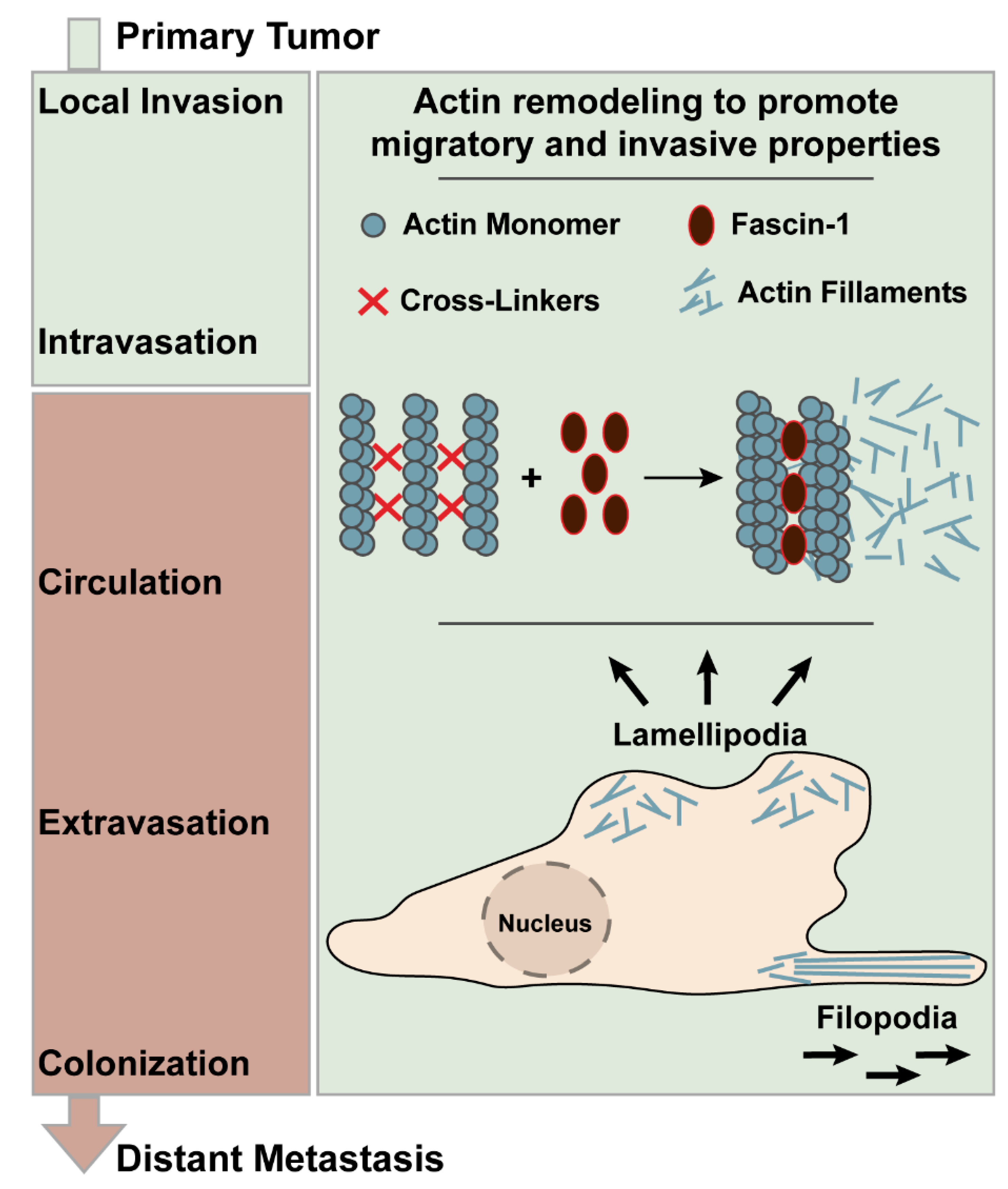
1. Introduction
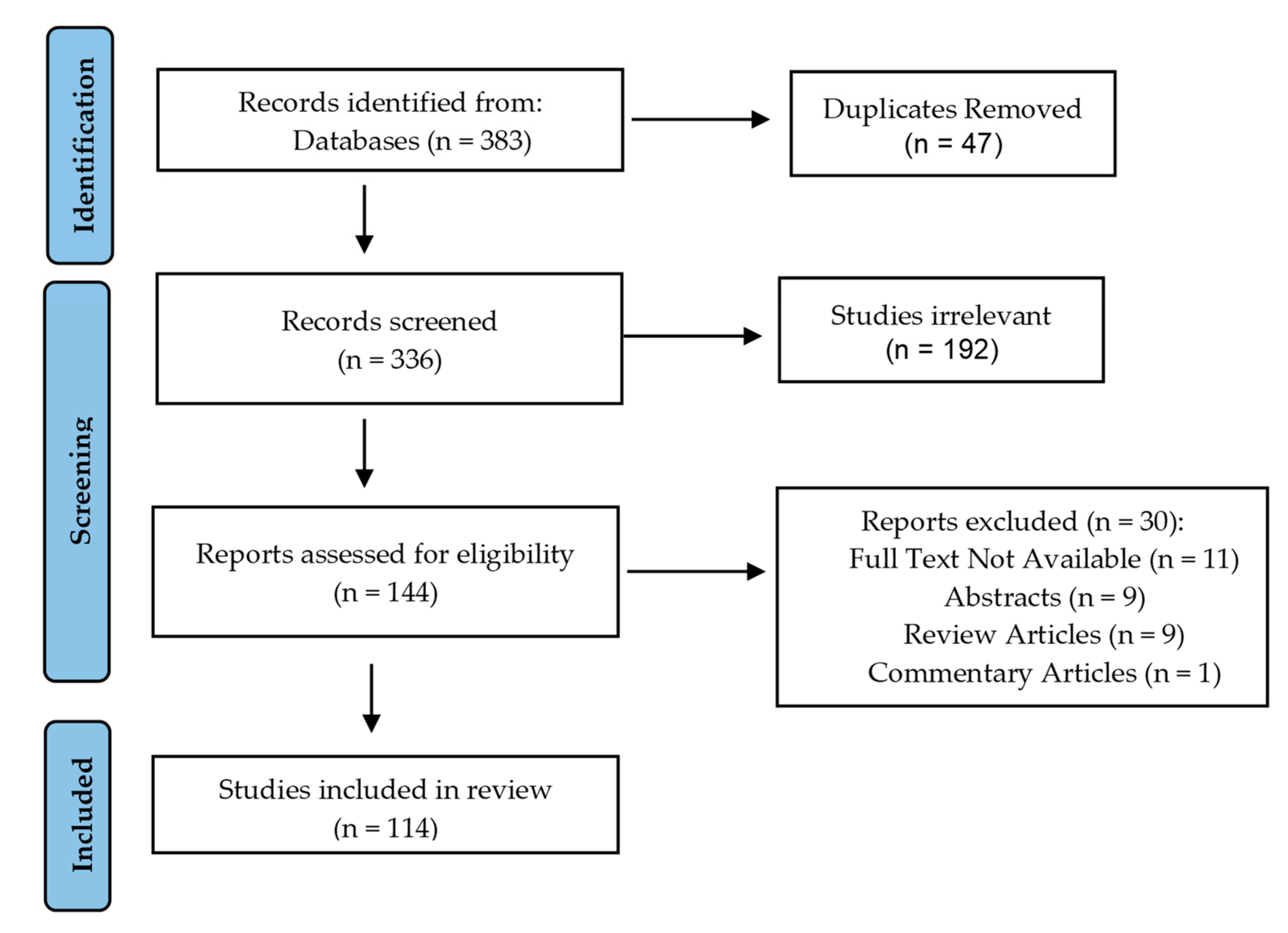
2. Methods
3. Fasin-1 and Esophageal Squamous Cell Carcinoma
3.1. Expression Pattern of Fascin-1 in ESCC and Its Potential as a Prognostic Marker
3.2. Outcome of Fascin-1 Overexpression in ESCC
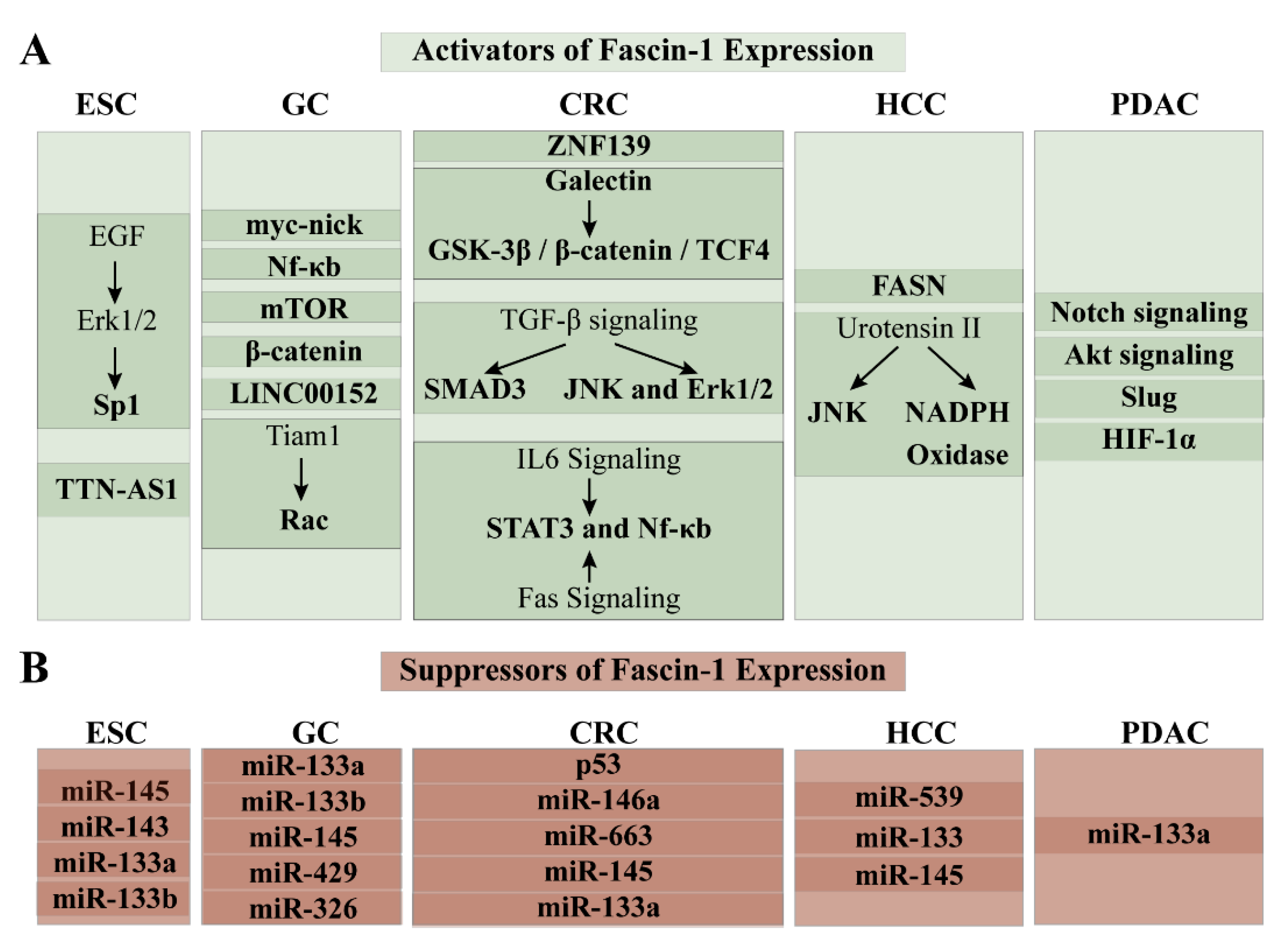
3.3. Regulation of Fascin-1 Expression in ESCC
4. Fascin-1 and Gastric Carcinoma
4.1. Expression Pattern of Fascin-1 in GC and Its Potential as a Prognostic Marker
4.2. Regulation of Fascin-1 Expression in GC
5. Fascin-1 and Colorectal Cancer (CRC)
5.1. Expression Pattern of Fascin-1 in CRC and Its Potential as a Prognostic Marker
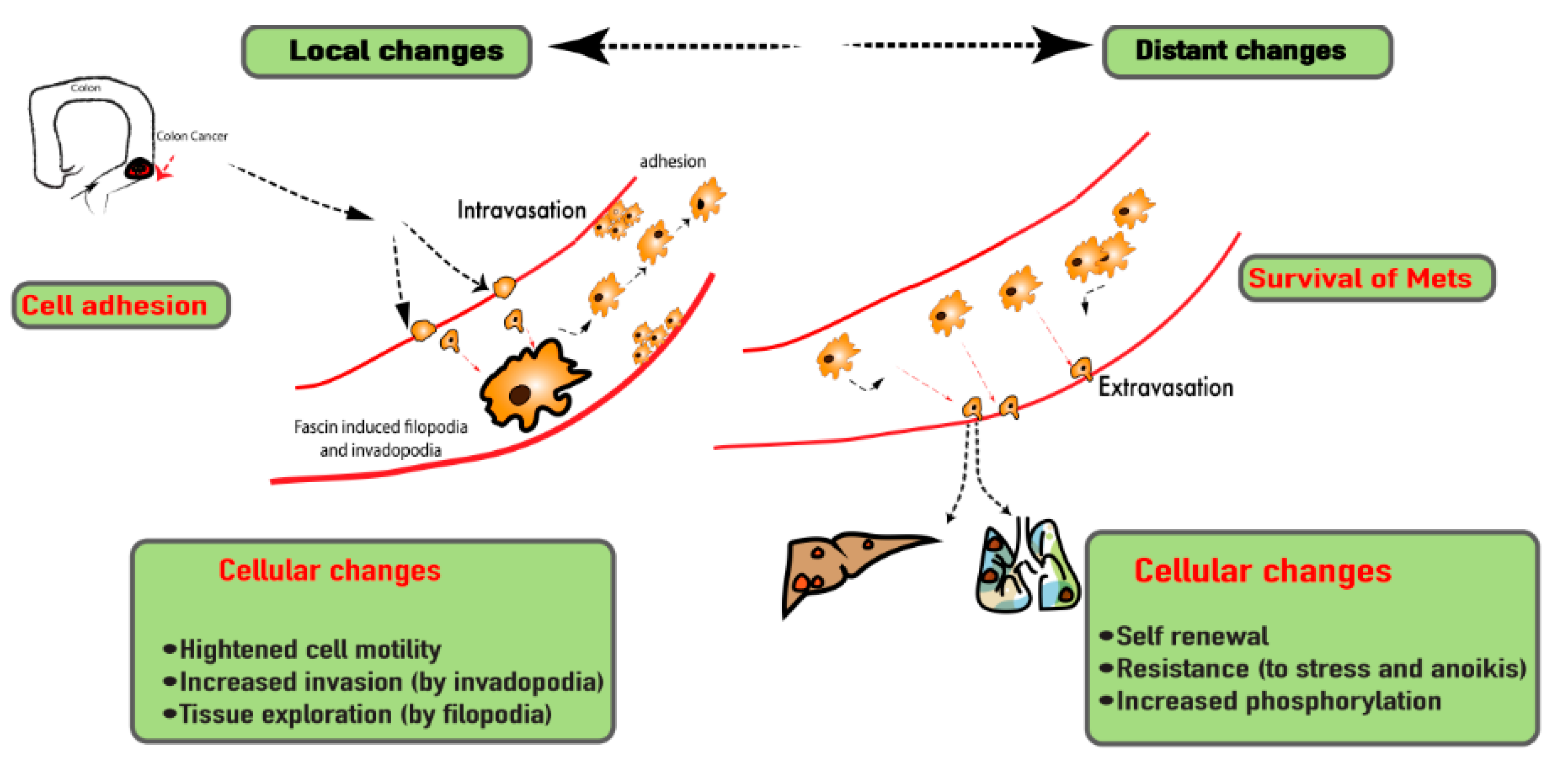
5.2. Outcome of Fascin-1 Overexpression and Suppression in CRC
5.3. Fascin-1 as a Therapeutic Target in CRC
5.4. Regulation of Regulation of Fascin-1 in CRC5
6. Fascin-1 and Hepatocellular Carcinoma
6.1. Expression Pattern of Fascin-1 in HCC and Its Potential as a Prognostic Marker
6.2. Outcome of Fascin-1 Overexpression in HCC
6.3. Fascin-1 as a Therapeutic Target in HCC
6.4. Regulation of Fascin-1 Expression in HCC
7. Fascin-1 and Pancreatic Cancer
7.1. Expression Pattern of Fascin-1 in Pancreatic Carcinomas
7.2. Fascin-1 as a Prognostic/Diagnostic Marker in PDAC
7.3. Outcome of Fascin-1 Overexpression and Suppression in PDAC
7.4. Fascin-1 as a Therapeutic Target in PDAC
7.5. Regulation of Fascin-1 in PDAC
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviation
| ABD | actin-binding domain |
| BMI1 | BMI1 proto-oncogene, polycomb ring finger |
| CTGF | Connective tissue growth factor |
| CRC | colorectal carcinoma |
| CYR61 | Cysteine-rich, angiogenic inducer 61 |
| EBV | Epstein-Barr virus |
| EGFR | epidermal growth factor receptor |
| EMMPRIN | extracellular matrix metalloproteinase inducer |
| EMT | epithelial-to-mesenchymal transition |
| ESCC | esophageal squamous cell carcinoma |
| EUS-FNA | endoscopic ultrasound-guided fine-needle aspiration |
| FASN | fatty acid synthase |
| GC | gastric carcinoma |
| GI tract | gastrointestinal tract |
| HCC | hepatocellular carcinoma |
| HIF-1α | hypoxia inducible factor 1 |
| IHC | immunohistochemistry |
| IPMN | intraductal papillary mucinous neoplasms |
| ITPN | intraductal tubulopapillary neoplasms |
| MMP-2 | matrix metalloproteinase-2 |
| MMP-9 | matrix metalloproteinase-9 |
| PanIN | pancreatic intraepithelial neoplasia |
| PDAC | pancreatic adenoracinoma |
| PKC | Protein Kinase C |
| rpNK-lysin | recombinant porcine natural killer lysin |
| TGF-β | transforming growth factor |
| Sp1 | specificity protein 1 |
| STAT3 | signal transducer and activator of transcription |
| Tiam1 | T lymphoma invasion and metastasis 1 |
| UII | urotensin II |
| ZNF139 | zinc finger protein 139 |
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2021. CA Cancer J. Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Riihimäki, M.; Hemminki, A.; Sundquist, J.; Hemminki, K. Patterns of metastasis in colon and rectal cancer. Sci Rep. 2016, 6, 29765. [Google Scholar] [CrossRef] [PubMed]
- Riihimäki, M.; Hemminki, A.; Sundquist, K.; Sundquist, J.; Hemminki, K. Metastatic spread in patients with gastric cancer. Oncotarget 2016, 7, 52307–52316. [Google Scholar] [CrossRef] [PubMed]
- Fares, J.; Fares, M.Y.; Khachfe, H.H.; Salhab, H.A.; Fares, Y. Molecular principles of metastasis: A hallmark of cancer revisited. Signal. Transduct. Target. Ther. 2020, 5, 28. [Google Scholar] [CrossRef]
- Svitkina, T. The Actin Cytoskeleton and Actin-Based Motility. Cold Spring Harb. Perspect. Biol. 2018, 10, a018267. [Google Scholar] [CrossRef]
- Innocenti, M. New insights into the formation and the function of lamellipodia and ruffles in mesenchymal cell migration. Cell Adh. Migr. 2018, 12, 401–416. [Google Scholar] [CrossRef] [PubMed]
- Izdebska, M.; Zielińska, W.; Grzanka, D.; Gagat, M. The Role of Actin Dynamics and Actin-Binding Proteins Expression in Epithelial-to-Mesenchymal Transition and Its Association with Cancer Progression and Evaluation of Possible Therapeutic Targets. Biomed. Res. Int. 2018, 2018, 4578373. [Google Scholar] [CrossRef]
- Jayo, A.; Parsons, M. Fascin: A key regulator of cytoskeletal dynamics. Int. J. Biochem. Cell Biol. 2010, 42, 1614–1617. [Google Scholar] [CrossRef] [PubMed]
- Kureishy, N.; Sapountzi, V.; Prag, S.; Anilkumar, N.; Adams, J.C. Fascins, and their roles in cell structure and function. Bioessays 2002, 24, 350–361. [Google Scholar] [CrossRef] [PubMed]
- Sedeh, R.S.; Fedorov, A.A.; Fedorov, E.V.; Ono, S.; Matsumura, F.; Almo, S.C.; Bathe, M. Structure, evolutionary conservation, and conformational dynamics of Homo sapiens fascin-1, an F-actin crosslinking protein. J. Mol. Biol. 2010, 400, 589–604. [Google Scholar] [CrossRef]
- Jayo, A.; Malboubi, M.; Antoku, S.; Chang, W.; Ortiz-Zapater, E.; Groen, C.; Pfisterer, K.; Tootle, T.; Charras, G.; Gundersen, G.G.; et al. Fascin Regulates Nuclear Movement and Deformation in Migrating Cells. Dev. Cell 2016, 38, 371–383. [Google Scholar] [CrossRef]
- Lin, S.; Huang, C.; Gunda, V.; Sun, J.; Chellappan, S.P.; Li, Z.; Izumi, V.; Fang, B.; Koomen, J.; Singh, P.K.; et al. Fascin Controls Metastatic Colonization and Mitochondrial Oxidative Phosphorylation by Remodeling Mitochondrial Actin Filaments. Cell Rep. 2019, 28, 2824–2836.e8. [Google Scholar] [CrossRef] [PubMed]
- Ai, D.; Zhu, H.; Ren, W.; Chen, Y.; Liu, Q.; Deng, J.; Ye, J.; Fan, J.; Zhao, K. Patterns of distant organ metastases in esophageal cancer: A population-based study. J. Thorac. Dis. 2017, 9, 3023–3030. [Google Scholar] [CrossRef]
- Hu, N.; Qian, L.; Hu, Y.; Shou, J.-Z.; Wang, C.; Giffen, C.; Wang, Q.-H.; Wang, Y.; Goldstein, A.M.; Emmert-Buck, M.; et al. Quantitative real-time RT-PCR validation of differential mRNA expression of SPARC, FADD, Fascin, COL7A1, CK4, TGM3, ECM1, PPL and EVPLin esophageal squamous cell carcinoma. BMC Cancer 2006, 6, 33. [Google Scholar] [CrossRef]
- Shen, T.Y.; Mei, L.L.; Qiu, Y.T.; Shi, Z.Z. Identification of candidate target genes of genomic aberrations in esophageal squamous cell carcinoma. Oncol. Lett. 2016, 12, 2956–2961. [Google Scholar] [CrossRef] [PubMed]
- Takikita, M.; Hu, N.; Shou, J.Z.; Giffen, C.; Wang, Q.H.; Wang, C.; Hewitt, S.M.; Taylor, P.R. Fascin and CK4 as biomarkers for esophageal squamous cell carcinoma. Anticancer Res. 2011, 31, 945–952. [Google Scholar]
- Zhang, H.; Xu, L.; Xiao, D.; Xie, J.; Zeng, H.; Cai, W.; Niu, Y.; Yang, Z.; Shen, Z.; Li, E. Fascin is a potential biomarker for early-stage oesophageal squamous cell carcinoma. J. Clin. Pathol. 2006, 59, 958–964. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Ito, T.; Inoue, H.; Okumura, T.; Tanaka, E.; Tsunoda, S.; Higashiyama, M.; Watanabe, G.; Imamura, M.; Shimada, Y. Prognostic significance of fascin overexpression in human esophageal squamous cell carcinoma. Clin. Cancer Res. 2005, 11, 2597–2605. [Google Scholar] [CrossRef]
- Hsu, K.F.; Lin, C.K.; Yu, C.P.; Tzao, C.; Lee, S.C.; Lee, Y.Y.; Tsai, W.C.; Jin, J.S. Cortactin, fascin, and survivin expression associated with clinicopathological parameters in esophageal squamous cell carcinoma. Dis. Esophagus 2009, 22, 402–408. [Google Scholar] [CrossRef]
- Cao, H.H.; Zheng, C.P.; Wang, S.H.; Wu, J.Y.; Shen, J.H.; Xu, X.E.; Fu, J.H.; Wu, Z.Y.; Li, E.M.; Xu, L.Y. A molecular prognostic model predicts esophageal squamous cell carcinoma prognosis. PLoS ONE 2014, 9, e106007. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Gu, Y.; Lu, W.; Liu, X.; Fu, H. Fascin1 promotes gastric cancer progression by facilitatingcell migrationand epithelial-mesenchymal transition. Pathol. Res. Pract. 2018, 214, 1362–1369. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zheng, H.; Hara, T.; Takahashi, H.; Masuda, S.; Wang, Z.; Yang, X.; Guan, Y.; Takano, Y. Aberrant expression of cortactin and fascin are effective markers for pathogenesis, invasion, metastasis and prognosis of gastric carcinomas. Int. J. Oncol. 2008, 33, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, Y.; Shimada, Y.; Kawamura, J.; Yamasaki, S.; Imamura, M. The prognostic relevance of fascin expression in human gastric carcinoma. Oncology 2004, 67, 262–270. [Google Scholar] [CrossRef]
- Tsai, W.C.; Jin, J.S.; Chang, W.K.; Chan, D.C.; Yeh, M.K.; Cherng, S.C.; Lin, L.F.; Sheu, L.F.; Chao, Y.C. Association of cortactin and fascin-1 expression in gastric adenocarcinoma: Correlation with clinicopathological parameters. J. Histochem. Cytochem. 2007, 55, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Tu, L.; Xu, J.; Wang, M.; Zhao, W.Y.; Zhang, Z.Z.; Zhu, C.C.; Tang, D.F.; Zhang, Y.Q.; Wang, D.H.; Zuo, J.; et al. Correlations of fascin-1 and cadherin-17 protein expression with clinicopathologic features and prognosis of patients with gastric cancer. Tumour Biol. 2016, 37, 8775–8782. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Kim, D.C.; Kim, M.C.; Jung, G.J.; Kim, K.H.; Jang, J.S.; Kwon, H.C.; Kim, Y.M.; Jeong, J.S. Fascin expression is related to poor survival in gastric cancer. Pathol. Int. 2012, 62, 777–784. [Google Scholar] [CrossRef]
- Puppa, G.; Maisonneuve, P.; Sonzogni, A.; Masullo, M.; Chiappa, A.; Valerio, M.; Zampino, M.G.; Franceschetti, I.; Capelli, P.; Chilosi, M.; et al. Independent prognostic value of fascin immunoreactivity in stage III-IV colonic adenocarcinoma. Br. J. Cancer 2007, 96, 1118–1126. [Google Scholar] [CrossRef][Green Version]
- Qualtrough, D.; Singh, K.; Banu, N.; Paraskeva, C.; Pignatelli, M. The actin-bundling protein fascin is overexpressed in colorectal adenomas and promotes motility in adenoma cells in vitro. Br. J. Cancer 2009, 101, 1124–1129. [Google Scholar] [CrossRef][Green Version]
- Vignjevic, D.; Schoumacher, M.; Gavert, N.; Janssen, K.P.; Jih, G.; Laé, M.; Louvard, D.; Ben-Ze’ev, A.; Robine, S. Fascin, a novel target of beta-catenin-TCF signaling, is expressed at the invasive front of human colon cancer. Cancer Res. 2007, 67, 6844–6853. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Skacel, M.; Lavery, I.C.; Mukherjee, A.L.; Casey, G.; Adams, J.C. Prognostic significance of fascin expression in advanced colorectal cancer: An immunohistochemical study of colorectal adenomas and adenocarcinomas. BMC Cancer 2006, 6, 241. [Google Scholar] [CrossRef]
- Jung, E.J.; Lee, J.H.; Min, B.W.; Kim, Y.S.; Choi, J.S. Clinicopathologic significance of fascin, extracellular matrix metalloproteinase inducer, and ezrin expressions in colorectal adenocarcinoma. Indian J. Pathol. Microbiol. 2011, 54, 32–36. [Google Scholar] [CrossRef]
- Tsai, W.C.; Chao, Y.C.; Sheu, L.F.; Chang, J.L.; Nieh, S.; Jin, J.S. Overexpression of fascin-1 in advanced colorectal adenocarcinoma: Tissue microarray analysis of immunostaining scores with clinicopathological parameters. Dis. Markers 2007, 23, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Ozerhan, I.H.; Ersoz, N.; Onguru, O.; Ozturk, M.; Kurt, B.; Cetiner, S. Fascin expression in colorectal carcinomas. Clinics (São Paulo) 2010, 65, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Piskor, B.M.; Pryczynicz, A.; Lubowicka, E.; Miniewska, K.; Zinczuk, J.; Zareba, K.; Guzinska-Ustymowicz, K. Immunohistochemical expression of Fascin-1 in colorectal cancer in relation to clinical and pathological parameters. Folia Histochem. Cytobiol. 2018, 56, 106–112. [Google Scholar] [CrossRef]
- Roseweir, A.K.; Kong, C.Y.; Park, J.H.; Bennett, L.; Powell, A.; Quinn, J.; van Wyk, H.C.; Horgan, P.G.; McMillan, D.C.; Edwards, J.; et al. A novel tumor-based epithelial-to-mesenchymal transition score that associates with prognosis and metastasis in patients with Stage II/III colorectal cancer. Int. J. Cancer 2019, 144, 150–159. [Google Scholar] [CrossRef]
- Tampakis, A.; Tampaki, E.C.; Nonni, A.; Kostakis, I.D.; Posabella, A.; Kontzoglou, K.; von Flüe, M.; Felekouras, E.; Kouraklis, G.; Nikiteas, N. High fascin-1 expression in colorectal cancer identifies patients at high risk for early disease recurrence and associated mortality. BMC Cancer 2021, 21, 153. [Google Scholar] [CrossRef]
- Oh, S.Y.; Kim, Y.B.; Suh, K.W.; Paek, O.J.; Moon, H.Y. Prognostic impact of fascin-1 expression is more significant in advanced colorectal cancer. J. Surg. Res. 2012, 172, 102–108. [Google Scholar] [CrossRef]
- Koçer, N.E.; Kayaselçuk, F. Is availability of anti-EGFR therapy for the colorectal adenocarcinomas showing fascin expression limited? Target. Oncol. 2014, 9, 171–175. [Google Scholar] [CrossRef]
- Huang, X.; Ji, J.; Xue, H.; Zhang, F.; Han, X.; Cai, Y.; Zhang, J.; Ji, G. Fascin and cortactin expression is correlated with a poor prognosis in hepatocellular carcinoma. Eur. J. Gastroenterol. Hepatol. 2012, 24, 633–639. [Google Scholar] [CrossRef]
- Iguchi, T.; Aishima, S.; Umeda, K.; Sanefuji, K.; Fujita, N.; Sugimachi, K.; Gion, T.; Taketomi, A.; Maehara, Y.; Tsuneyoshi, M. Fascin expression in progression and prognosis of hepatocellular carcinoma. J. Surg. Oncol. 2009, 100, 575–579. [Google Scholar] [CrossRef]
- Lin, C.K.; Jin, J.S.; Yu, C.P.; Tsai, W.C. Expression of LGR8 and related biomarkers in hepatocellular carcinoma: Correlation with clinicopathological parameters. Chin. J. Physiol. 2011, 54, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Misiura, M.; Zińczuk, J.; Zaręba, K.; Kamińska, D.; Guzińska-Ustymowicz, K.; Pryczynicz, A. Actin-Bundling Proteins (Actinin-4 and Fascin-1) are Involved in the Development of Pancreatic Intraepithelial Neoplasia (PanIN). Am. J. Med. Sci. 2020, 359, 147–155. [Google Scholar] [CrossRef]
- Tsai, W.C.; Chao, Y.C.; Sheu, L.F.; Lin, Y.F.; Nieh, S.; Chen, A.; Yu, C.P.; Jin, J.S. EMMPRIN and fascin overexpression associated with clinicopathologic parameters of pancreatobiliary adenocarcinoma in Chinese people. Apmis 2007, 115, 929–938. [Google Scholar] [CrossRef]
- Li, A.; Morton, J.P.; Ma, Y.; Karim, S.A.; Zhou, Y.; Faller, W.J.; Woodham, E.F.; Morris, H.T.; Stevenson, R.P.; Juin, A.; et al. Fascin is regulated by slug, promotes progression of pancreatic cancer in mice, and is associated with patient outcomes. Gastroenterology 2014, 146, 1386–1396.e17. [Google Scholar] [CrossRef]
- Maitra, A.; Adsay, N.V.; Argani, P.; Iacobuzio-Donahue, C.; De Marzo, A.; Cameron, J.L.; Yeo, C.J.; Hruban, R.H. Multicomponent analysis of the pancreatic adenocarcinoma progression model using a pancreatic intraepithelial neoplasia tissue microarray. Mod. Pathol. 2003, 16, 902–912. [Google Scholar] [CrossRef]
- Zhao, X.; Gao, S.; Ren, H.; Sun, W.; Zhang, H.; Sun, J.; Yang, S.; Hao, J. Hypoxia-inducible factor-1 promotes pancreatic ductal adenocarcinoma invasion and metastasis by activating transcription of the actin-bundling protein fascin. Cancer Res. 2014, 74, 2455–2464. [Google Scholar] [CrossRef]
- Tsai, W.C.; Lin, C.K.; Lee, H.S.; Gao, H.W.; Nieh, S.; Chan, D.C.; Jin, J.S. The correlation of cortactin and fascin-1 expression with clinicopathological parameters in pancreatic and ampulla of Vater adenocarcinoma. Apmis 2013, 121, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, B.; Ludwig, O.J.; Collins, B.T.; Cortese, C. Immunostaining as an adjunct to cytology for diagnosis of pancreatic adenocarcinoma. Clin. Gastroenterol. Hepatol. 2008, 6, 1425–1431. [Google Scholar] [CrossRef] [PubMed]
- Dim, D.C.; Jiang, F.; Qiu, Q.; Li, T.; Darwin, P.; Rodgers, W.H.; Peng, H.Q. The usefulness of S100P, mesothelin, fascin, prostate stem cell antigen, and 14-3-3 sigma in diagnosing pancreatic adenocarcinoma in cytological specimens obtained by endoscopic ultrasound guided fine-needle aspiration. Diagn. Cytopathol. 2014, 42, 193–199. [Google Scholar] [CrossRef]
- Tan, H.; Zhang, H.; Xie, J.; Chen, B.; Wen, C.; Guo, X.; Zhao, Q.; Wu, Z.; Shen, J.; Wu, J.; et al. A novel staging model to classify oesophageal squamous cell carcinoma patients in China. Br. J. Cancer 2014, 110, 2109–2115. [Google Scholar] [CrossRef]
- Wang, C.; Wang, J.; Chen, Z.; Gao, Y.; He, J. Immunohistochemical prognostic markers of esophageal squamous cell carcinoma: A systematic review. Chin. J. Cancer 2017, 36, 65. [Google Scholar] [CrossRef]
- Perisetti, A.; Bellamkonda, M.; Konda, M.; Edwards, S.; Ali Khan, S.; Bansal, P.; Hu, Z.D.; Goyal, H. Tumor-associated antigens and their antibodies in the screening, diagnosis, and monitoring of esophageal cancers. Eur. J. Gastroenterol. Hepatol. 2020, 32, 779–788. [Google Scholar] [CrossRef]
- Chen, W.X.; Hong, X.B.; Hong, C.Q.; Liu, M.; Li, L.; Huang, L.S.; Xu, L.Y.; Xu, Y.W.; Peng, Y.H.; Li, E.M. Tumor-associated autoantibodies against Fascin as a novel diagnostic biomarker for esophageal squamous cell carcinoma. Clin. Res. Hepatol. Gastroenterol. 2017, 41, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Du, Z.P.; Wu, B.L.; Xie, J.J.; Lin, X.H.; Qiu, X.Y.; Zhan, X.F.; Wang, S.H.; Shen, J.H.; Li, E.M.; Xu, L.Y. Network Analyses of Gene Expression following Fascin Knockdown in Esophageal Squamous Cell Carcinoma Cells. Asian Pac. J. Cancer Prev. 2015, 16, 5445–5451. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Xie, J.J.; Xu, L.Y.; Zhang, H.H.; Cai, W.J.; Mai, R.Q.; Xie, Y.M.; Yang, Z.M.; Niu, Y.D.; Shen, Z.Y.; Li, E.M. Role of fascin in the proliferation and invasiveness of esophageal carcinoma cells. Biochem. Biophys. Res. Commun. 2005, 337, 355–362. [Google Scholar] [CrossRef]
- Xie, J.J.; Xu, L.Y.; Wu, J.Y.; Shen, Z.Y.; Zhao, Q.; Du, Z.P.; Lv, Z.; Gu, W.; Pan, F.; Xu, X.E.; et al. Involvement of CYR61 and CTGF in the fascin-mediated proliferation and invasiveness of esophageal squamous cell carcinomas cells. Am. J. Pathol. 2010, 176, 939–951. [Google Scholar] [CrossRef]
- Ortiz, C.M.; Ito, T.; Hashimoto, Y.; Nagayama, S.; Iwai, A.; Tsunoda, S.; Sato, F.; Martorell, M.; Garcia, J.A.; Perez, A.; et al. Effects of small interfering RNAs targeting fascin on human esophageal squamous cell carcinoma cell lines. Diagn. Pathol. 2010, 5, 41. [Google Scholar] [CrossRef]
- Hou, J.; Guo, Z.Y.; Xie, J.J.; Li, E.M.; Xu, L.Y. Fascin overexpression is regulated by the transactivation of the promoter but not by its hypomethylation in esophageal squamous cell carcinoma. Mol. Med. Rep. 2009, 2, 843–849. [Google Scholar] [CrossRef]
- Lu, X.F.; Li, E.M.; Du, Z.P.; Xie, J.J.; Guo, Z.Y.; Gao, S.Y.; Liao, L.D.; Shen, Z.Y.; Xie, D.; Xu, L.Y. Specificity protein 1 regulates fascin expression in esophageal squamous cell carcinoma as the result of the epidermal growth factor/extracellular signal-regulated kinase signaling pathway activation. Cell. Mol. Life Sci. 2010, 67, 3313–3329. [Google Scholar] [CrossRef]
- Kano, M.; Seki, N.; Kikkawa, N.; Fujimura, L.; Hoshino, I.; Akutsu, Y.; Chiyomaru, T.; Enokida, H.; Nakagawa, M.; Matsubara, H. miR-145, miR-133a and miR-133b: Tumor-suppressive miRNAs target FSCN1 in esophageal squamous cell carcinoma. Int. J. Cancer 2010, 127, 2804–2814. [Google Scholar] [CrossRef]
- Akanuma, N.; Hoshino, I.; Akutsu, Y.; Murakami, K.; Isozaki, Y.; Maruyama, T.; Yusup, G.; Qin, W.; Toyozumi, T.; Takahashi, M.; et al. MicroRNA-133a regulates the mRNAs of two invadopodia-related proteins, FSCN1 and MMP14, in esophageal cancer. Br. J. Cancer 2014, 110, 189–198. [Google Scholar] [CrossRef]
- Liu, R.; Liao, J.; Yang, M.; Sheng, J.; Yang, H.; Wang, Y.; Pan, E.; Guo, W.; Pu, Y.; Kim, S.J.; et al. The cluster of miR-143 and miR-145 affects the risk for esophageal squamous cell carcinoma through co-regulating fascin homolog 1. PLoS ONE 2012, 7, e33987. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.N.; Li, K.; Liu, Y.; Yang, C.L.; He, C.Y.; Wang, H.R. Down-regulation of long noncoding RNA PVT1 inhibits esophageal carcinoma cell migration and invasion and promotes cell apoptosis via microRNA-145-mediated inhibition of FSCN1. Mol. Oncol. 2019, 13, 2554–2573. [Google Scholar] [CrossRef]
- Lin, C.; Zhang, S.; Wang, Y.; Wang, Y.; Nice, E.; Guo, C.; Zhang, E.; Yu, L.; Li, M.; Liu, C.; et al. Functional Role of a Novel Long Noncoding RNA TTN-AS1 in Esophageal Squamous Cell Carcinoma Progression and Metastasis. Clin. Cancer Res. 2018, 24, 486–498. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.-M.; Wang, X.-N.; Shi, H.-S.; Xie, J.-J.; Du, Z.-P.; Liao, L.-D.; Nie, P.-J.; Xu, L.-Y.; Li, E.-M. Fascin phosphorylation sites combine to regulate esophageal squamous cancer cell behavior. Amino Acids 2017, 49, 943–955. [Google Scholar] [CrossRef]
- Zhao, Q.; Shen, J.H.; Shen, Z.Y.; Wu, Z.Y.; Xu, X.E.; Xie, J.J.; Wu, J.Y.; Huang, Q.; Lu, X.F.; Li, E.M.; et al. Phosphorylation of fascin decreases the risk of poor survival in patients with esophageal squamous cell carcinoma. J. Histochem. Cytochem. 2010, 58, 979–988. [Google Scholar] [CrossRef]
- Zheng, H.C.; Zhao, S. The meta and bioinformatics analysis of fascin expression in gastric cancer: A potential marker for aggressiveness and worse prognosis. Oncotarget 2017, 8, 105574–105583. [Google Scholar] [CrossRef]
- Son, B.K.; Kim, D.H.; Min, K.W.; Kim, E.K.; Kwon, M.J. Smad4/Fascin index is highly prognostic in patients with diffuse type EBV-associated gastric cancer. Pathol. Res. Pract. 2018, 214, 475–481. [Google Scholar] [CrossRef]
- Hao, Y.J.; Li, Y.; Fan, L.Q.; Zhao, Q.; Tan, B.B.; Jiao, Z.K.; Zhao, X.F.; Zhang, Z.D.; Wang, D. Role of RNA-interference-induced zinc finger protein 139 suppression in gastric cancer cell sensitivity to chemotherapeutic agents. Oncol. Lett. 2015, 10, 1333–1338. [Google Scholar] [CrossRef][Green Version]
- Kim, S.J.; Choi, I.J.; Cheong, T.C.; Lee, S.J.; Lotan, R.; Park, S.H.; Chun, K.H. Galectin-3 increases gastric cancer cell motility by up-regulating fascin-1 expression. Gastroenterology 2010, 138, 1035–1045.e2. [Google Scholar] [CrossRef]
- Li, L.; Cao, F.; Liu, B.; Luo, X.; Ma, X.; Hu, Z. TGF-β induces fascin expression in gastric cancer via phosphorylation of smad3 linker area. Am. J. Cancer Res. 2015, 5, 1890–1896. [Google Scholar] [PubMed]
- Fu, H.; Hu, Z.; Wen, J.; Wang, K.; Liu, Y. TGF-beta promotes invasion and metastasis of gastric cancer cells by increasing fascin1 expression via ERK and JNK signal pathways. Acta Biochim. Biophys. Sin. 2009, 41, 648–656. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Qian, C.J.; Ye, B.; Zhao, Z.Q.; Wei, J.; Liang, Y.; Zhang, X. Signal transducer and activator of transcription 3 signaling upregulates fascin via nuclear factor-κB in gastric cancer: Implications in cell invasion and migration. Oncol. Lett. 2014, 7, 902–908. [Google Scholar] [CrossRef]
- Yang, Y.; Zhao, Q.; Cai, Z.; Cheng, G.; Chen, M.; Wang, J.; Zhong, H. Fas Signaling Promotes Gastric Cancer Metastasis through STAT3-Dependent Upregulation of Fascin. PLoS ONE 2015, 10, e0125132. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Bai, H.; Zou, D.; Hong, T.; Liu, J.; Huang, J.; He, P.; Zhou, Q.; He, J. The role of microRNA-133b and its target gene FSCN1 in gastric cancer. J. Exp. Clin. Cancer Res. 2014, 33, 99. [Google Scholar] [CrossRef]
- Lai, C.; Chen, Z.; Li, R. MicroRNA-133a inhibits proliferation and invasion, and induces apoptosis in gastric carcinoma cells via targeting fascin actin-bundling protein 1. Mol. Med. Rep. 2015, 12, 1473–1478. [Google Scholar] [CrossRef]
- Xue, M.; Zhao, L.; Yang, F.; Li, Z.; Li, G. MicroRNA-145 inhibits the malignant phenotypes of gastric carcinoma cells via downregulation of fascin 1 expression. Mol. Med. Rep. 2016, 13, 1033–1039. [Google Scholar] [CrossRef][Green Version]
- Zhang, M.; Dong, B.B.; Lu, M.; Zheng, M.J.; Chen, H.; Ding, J.Z.; Xu, A.M.; Xu, Y.H. miR-429 functions as a tumor suppressor by targeting FSCN1 in gastric cancer cells. OncoTargets Ther. 2016, 9, 1123–1133. [Google Scholar] [CrossRef]
- Li, Y.; Gao, Y.; Xu, Y.; Ma, H.; Yang, M. Down-regulation of miR-326 is associated with poor prognosis and promotes growth and metastasis by targeting FSCN1 in gastric cancer. Growth Factors 2015, 33, 267–274. [Google Scholar] [CrossRef]
- Wang, C.Q.; Wang, Y.; Huang, B.F.; Tang, C.H.; Du, Z.; Zeng, Y.; Wang, Q.; Shao, J.K.; Jin, L.L. High Expression of Both Resistin and Fascin-1 Predicts a Poor Prognosis in Patients with Colorectal Cancer. Biomed. Res. Int. 2020, 2020, 8753175. [Google Scholar] [CrossRef]
- Kanda, Y.; Kawaguchi, T.; Osaki, M.; Onuma, K.; Ochiya, T.; Kitagawa, T.; Okada, F. Fascin protein stabilization by miR-146a implicated in the process of a chronic inflammation-related colon carcinogenesis model. Inflamm. Res. 2018, 67, 839–846. [Google Scholar] [CrossRef] [PubMed]
- Al-Antary, N.; Farghaly, H.; Aboulkassim, T.; Yasmeen, A.; Akil, N.; Al Moustafa, A.E. Epstein-Barr virus and its association with Fascin expression in colorectal cancers in the Syrian population: A tissue microarray study. Hum. Vaccines Immunother. 2017, 13, 1573–1578. [Google Scholar] [CrossRef] [PubMed]
- Ghabreau, L.; Segal, E.; Yasmeen, A.; Kassab, A.; Akil, N.; Al Moustafa, A.-E. High-risk human papillomavirus infections in colorectal cancer in the Syrian population and their association with Fascin, Id-1 and P-cadherin expressions: A tissue microarray study. Clin. Cancer Investig. J. 2012, 1, 26–30. [Google Scholar] [CrossRef]
- Shi, S.; Zheng, H.C.; Zhang, Z.G. Roles of Fascin mRNA expression in colorectal cancer: Meta-analysis and bioinformatics analysis. Mol. Clin. Oncol. 2020, 13, 119–128. [Google Scholar] [CrossRef]
- Tan, V.Y.; Lewis, S.J.; Adams, J.C.; Martin, R.M. Association of fascin-1 with mortality, disease progression and metastasis in carcinomas: A systematic review and meta-analysis. BMC Med. 2013, 11, 52. [Google Scholar] [CrossRef] [PubMed]
- Alajez, N.M. Significance of BMI1 and FSCN1 expression in colorectal cancer. Saudi J. Gastroenterol. 2016, 22, 288–293. [Google Scholar] [CrossRef]
- Schoumacher, M.; El-Marjou, F.; Laé, M.; Kambou, N.; Louvard, D.; Robine, S.; Vignjevic, D.M. Conditional expression of fascin increases tumor progression in a mouse model of intestinal cancer. Eur. J. Cell Biol. 2014, 93, 388–395. [Google Scholar] [CrossRef]
- Jawhari, A.U.; Buda, A.; Jenkins, M.; Shehzad, K.; Sarraf, C.; Noda, M.; Farthing, M.J.; Pignatelli, M.; Adams, J.C. Fascin, an actin-bundling protein, modulates colonic epithelial cell invasiveness and differentiation in vitro. Am. J. Pathol. 2003, 162, 69–80. [Google Scholar] [CrossRef]
- Kanda, Y.; Kawaguchi, T.; Kuramitsu, Y.; Kitagawa, T.; Kobayashi, T.; Takahashi, N.; Tazawa, H.; Habelhah, H.; Hamada, J.; Kobayashi, M.; et al. Fascin regulates chronic inflammation-related human colon carcinogenesis by inhibiting cell anoikis. Proteomics 2014, 14, 1031–1041. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Parsons, M.; Adams, J.C. Dual actin-bundling and protein kinase C-binding activities of fascin regulate carcinoma cell migration downstream of Rac and contribute to metastasis. Mol. Biol. Cell 2007, 18, 4591–4602. [Google Scholar] [CrossRef]
- Chen, M.B.; Wei, M.X.; Han, J.Y.; Wu, X.Y.; Li, C.; Wang, J.; Shen, W.; Lu, P.H. MicroRNA-451 regulates AMPK/mTORC1 signaling and fascin1 expression in HT-29 colorectal cancer. Cell Signal. 2014, 26, 102–109. [Google Scholar] [CrossRef]
- Montoro-García, S.; Alburquerque-González, B.; Bernabé-García, Á.; Bernabé-García, M.; Rodrigues, P.C.; den-Haan, H.; Luque, I.; Nicolás, F.J.; Pérez-Sánchez, H.; Cayuela, M.L.; et al. Novel anti-invasive properties of a Fascin1 inhibitor on colorectal cancer cells. J. Mol. Med. 2020, 98, 383–394. [Google Scholar] [CrossRef]
- Huang, F.K.; Han, S.; Xing, B.; Huang, J.; Liu, B.; Bordeleau, F.; Reinhart-King, C.A.; Zhang, J.J.; Huang, X.Y. Targeted inhibition of fascin function blocks tumour invasion and metastatic colonization. Nat. Commun. 2015, 6, 7465. [Google Scholar] [CrossRef]
- Mahmoud, A.; Elkhalifa, D.; Alali, F.; Al Moustafa, A.E.; Khalil, A. Novel Polymethoxylated Chalcones as Potential Compounds Against KRAS-Mutant Colorectal Cancers. Curr. Pharm. Des. 2020, 26, 1622–1633. [Google Scholar] [CrossRef]
- Alburquerque-González, B.; Bernabé-García, M.; Montoro-García, S.; Bernabé-García, Á.; Rodrigues, P.C.; Ruiz-Sanz, J.; López-Calderón, F.F.; Luque, I.; Nicolas, F.J.; Cayuela, M.L.; et al. New role of the antidepressant imipramine as a Fascin1 inhibitor in colorectal cancer cells. Exp. Mol. Med. 2020, 52, 281–292. [Google Scholar] [CrossRef]
- Walker, A.J.; Card, T.; Bates, T.E.; Muir, K. Tricyclic antidepressants and the incidence of certain cancers: A study using the GPRD. Br. J. Cancer 2011, 104, 193–197. [Google Scholar] [CrossRef]
- Alburquerque-González, B.; Bernabé-García, Á.; Bernabé-García, M.; Ruiz-Sanz, J.; López-Calderón, F.F.; Gonnelli, L.; Banci, L.; Peña-García, J.; Luque, I.; Nicolás, F.J.; et al. The FDA-Approved Antiviral Raltegravir Inhibits Fascin1-Dependent Invasion of Colorectal Tumor Cells In Vitro and In Vivo. Cancers 2021, 13, 861. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Fan, K.; Sun, N.; Yin, W.; Sun, Y.; Sun, P.; Jahejo, A.R.; Li, H. Recombinant porcine NK-lysin inhibits the invasion of hepatocellular carcinoma cells in vitro. Int. J. Biol. Macromol. 2019, 140, 1249–1259. [Google Scholar] [CrossRef]
- Elewa, M.A.; Al-Gayyar, M.M.; Schaalan, M.F.; Abd El Galil, K.H.; Ebrahim, M.A.; El-Shishtawy, M.M. Hepatoprotective and anti-tumor effects of targeting MMP-9 in hepatocellular carcinoma and its relation to vascular invasion markers. Clin. Exp. Metastasis 2015, 32, 479–493. [Google Scholar] [CrossRef]
- Schenk, M.; Aykut, B.; Teske, C.; Giese, N.A.; Weitz, J.; Welsch, T. Salinomycin inhibits growth of pancreatic cancer and cancer cell migration by disruption of actin stress fiber integrity. Cancer Lett. 2015, 358, 161–169. [Google Scholar] [CrossRef]
- Anderson, S.; Poudel, K.R.; Roh-Johnson, M.; Brabletz, T.; Yu, M.; Borenstein-Auerbach, N.; Grady, W.N.; Bai, J.; Moens, C.B.; Eisenman, R.N.; et al. MYC-nick promotes cell migration by inducing fascin expression and Cdc42 activation. Proc. Natl. Acad. Sci. USA 2016, 113, E5481–E5490. [Google Scholar] [CrossRef]
- Conacci-Sorrell, M.; Ngouenet, C.; Anderson, S.; Brabletz, T.; Eisenman, R.N. Stress-induced cleavage of Myc promotes cancer cell survival. Genes Dev. 2014, 28, 689–707. [Google Scholar] [CrossRef] [PubMed]
- Sui, X.; Zhu, J.; Tang, H.; Wang, C.; Zhou, J.; Han, W.; Wang, X.; Fang, Y.; Xu, Y.; Li, D.; et al. p53 controls colorectal cancer cell invasion by inhibiting the NF-κB-mediated activation of Fascin. Oncotarget 2015, 6, 22869–22879. [Google Scholar] [CrossRef] [PubMed]
- Ou, C.; Sun, Z.; He, X.; Li, X.; Fan, S.; Zheng, X.; Peng, Q.; Li, G.; Li, X.; Ma, J. Targeting YAP1/LINC00152/FSCN1 Signaling Axis Prevents the Progression of Colorectal Cancer. Adv. Sci. 2020, 7, 1901380. [Google Scholar] [CrossRef]
- Yu, S.; Xie, H.; Zhang, J.; Wang, D.; Song, Y.; Zhang, S.; Zheng, S.; Wang, J. MicroRNA-663 suppresses the proliferation and invasion of colorectal cancer cells by directly targeting FSCN1. Mol. Med. Rep. 2017, 16, 9707–9714. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Feng, Y.; Zhu, J.; Ou, C.; Deng, Z.; Chen, M.; Huang, W.; Li, L. MicroRNA-145 inhibits tumour growth and metastasis in colorectal cancer by targeting fascin-1. Br. J. Cancer 2014, 110, 2300–2309. [Google Scholar] [CrossRef] [PubMed]
- Wan, T.M.; Lam, C.S.; Ng, L.; Chow, A.K.; Wong, S.K.; Li, H.S.; Man, J.H.; Lo, O.S.; Foo, D.; Cheung, A.; et al. The clinicopathological significance of miR-133a in colorectal cancer. Dis. Markers 2014, 2014, 919283. [Google Scholar] [CrossRef]
- Zheng, K.; Liu, W.; Liu, Y.; Jiang, C.; Qian, Q. MicroRNA-133a suppresses colorectal cancer cell invasion by targeting Fascin1. Oncol. Lett. 2015, 9, 869–874. [Google Scholar] [CrossRef]
- Parsons, M.; Adams, J.C. Rac regulates the interaction of fascin with protein kinase C in cell migration. J. Cell Sci. 2008, 121, 2805–2813. [Google Scholar] [CrossRef]
- Liu, L.; Zhao, L.; Zhang, Y.; Zhang, Q.; Ding, Y. Proteomic analysis of Tiam1-mediated metastasis in colorectal cancer. Cell Biol. Int. 2007, 31, 805–814. [Google Scholar] [CrossRef]
- Hayashi, Y.; Osanai, M.; Lee, G.H. Fascin-1 expression correlates with repression of E-cadherin expression in hepatocellular carcinoma cells and augments their invasiveness in combination with matrix metalloproteinases. Cancer Sci. 2011, 102, 1228–1235. [Google Scholar] [CrossRef]
- Radović, S.; Dorić, M.; Zujo, H.; Hukić, A.; Kuskunović, S.; Babić, M.; Tomić, I. Interdigitating dendritic cell sarcoma of the liver and lung: A case report with morphohological and immunohistochemical features of tumor. Bosn. J. Basic Med. Sci. 2012, 12, 203–206. [Google Scholar] [CrossRef]
- Zhang, Y.; Lu, Y.; Zhang, C.; Huang, D.; Wu, W.; Zhang, Y.; Shen, J.; Cai, Y.; Chen, W.; Yao, W. FSCN-1 increases doxorubicin resistance in hepatocellular carcinoma through promotion of epithelial-mesenchymal transition. Int. J. Oncol. 2018, 52, 1455–1464. [Google Scholar] [CrossRef]
- Kim, S.H.; Kim, Y.; Kim, M.; Kim, D.S.; Lee, S.C.; Chi, S.W.; Lee, D.H.; Park, S.G.; Park, B.C.; Bae, K.H.; et al. Comparative proteomic analysis of mouse melanoma cell line B16, a metastatic descendant B16F10, and B16 overexpressing the metastasis-associated tyrosine phosphatase PRL-3. Oncol. Res. 2009, 17, 601–612. [Google Scholar] [CrossRef]
- Huang, J.; Tang, Y.; Zou, X.; Lu, Y.; She, S.; Zhang, W.; Ren, H.; Yang, Y.; Hu, H. Identification of the fatty acid synthase interaction network via iTRAQ-based proteomics indicates the potential molecular mechanisms of liver cancer metastasis. Cancer Cell Int. 2020, 20, 332. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.Y.; Shi, Z.M.; Yu, X.T.; Feng, P.; Wang, X.J. The effects of urotensin II on migration and invasion are mediated by NADPH oxidase-derived reactive oxygen species through the c-Jun N-terminal kinase pathway in human hepatoma cells. Peptides 2017, 88, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Hong, W.; Zhou, C.; Jiang, Z.; Wang, G.; Wei, G.; Li, X. miR-539 inhibits FSCN1 expression and suppresses hepatocellular carcinoma migration and invasion. Oncol. Rep. 2017, 37, 2593–2602. [Google Scholar] [CrossRef]
- Wang, G.; Zhu, S.; Gu, Y.; Chen, Q.; Liu, X.; Fu, H. MicroRNA-145 and MicroRNA-133a Inhibited Proliferation, Migration, and Invasion, While Promoted Apoptosis in Hepatocellular Carcinoma Cells Via Targeting FSCN1. Dig. Dis. Sci. 2015, 60, 3044–3052. [Google Scholar] [CrossRef] [PubMed]
- Iacobuzio-Donahue, C.A.; Ashfaq, R.; Maitra, A.; Adsay, N.V.; Shen-Ong, G.L.; Berg, K.; Hollingsworth, M.A.; Cameron, J.L.; Yeo, C.J.; Kern, S.E.; et al. Highly expressed genes in pancreatic ductal adenocarcinomas: A comprehensive characterization and comparison of the transcription profiles obtained from three major technologies. Cancer Res. 2003, 63, 8614–8622. [Google Scholar] [PubMed]
- Date, K.; Okabayashi, T.; Shima, Y.; Iwata, J.; Sumiyoshi, T.; Kozuki, A.; Morita, S.; Hata, Y.; Noda, Y.; Nishioka, A.; et al. Clinicopathological features and surgical outcomes of intraductal tubulopapillary neoplasm of the pancreas: A systematic review. Langenbeck’s Arch. Surg. 2016, 401, 439–447. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Inoue, T.; Eguchi, T.; Miyasaka, Y.; Ohuchida, K.; Mizumoto, K.; Yamada, T.; Yamaguchi, K.; Tanaka, M.; Tsuneyoshi, M. Fascin overexpression in intraductal papillary mucinous neoplasms (adenomas, borderline neoplasms, and carcinomas) of the pancreas, correlated with increased histological grade. Mod. Pathol. 2007, 20, 552–561. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.; Maitra, A.; Saavedra, J.A.; Klimstra, D.S.; Adsay, N.V.; Hruban, R.H. Expression of novel markers of pancreatic ductal adenocarcinoma in pancreatic nonductal neoplasms: Additional evidence of different genetic pathways. Mod. Pathol. 2005, 18, 752–761. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.C.; Wu, C.C.; Ng, K.F.; Wu, R.C.; Chen, H.M.; Chen, T.C. Follicular dendritic cell sarcoma mimicking giant cell carcinoma of the pancreas. Pathol. Int. 2006, 56, 466–470. [Google Scholar] [CrossRef]
- Lu, Z.; Hu, L.; Evers, S.; Chen, J.; Shen, Y. Differential expression profiling of human pancreatic adenocarcinoma and healthy pancreatic tissue. Proteomics 2004, 4, 3975–3988. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.F.; Yu, S.N.; Lu, Z.H.; Liu, J.P.; Chen, J. Fascin promotes the motility and invasiveness of pancreatic cancer cells. World J. Gastroenterol. 2011, 17, 4470–4478. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Kuramitsu, Y.; Murakami, K.; Ryozawa, S.; Taba, K.; Kaino, S.; Zhang, X.; Sakaida, I.; Nakamura, K. Proteomic differential display analysis for TS-1-resistant and -sensitive pancreatic cancer cells using two-dimensional gel electrophoresis and mass spectrometry. Anticancer Res. 2011, 31, 2103–2108. [Google Scholar] [PubMed]
- Qian, C.J.; Chen, Y.Y.; Zhang, X.; Liu, F.Q.; Yue, T.T.; Ye, B.; Yao, J. Notch4 inhibition reduces migration and invasion and enhances sensitivity to docetaxel by inhibiting Akt/fascin in pancreatic cancer cells. Oncol. Lett. 2016, 12, 3499–3505. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Dang, X.; Li, W.; Ma, Q. miR-133a functions as a tumor suppressor and directly targets FSCN1 in pancreatic cancer. Oncol. Res. 2013, 21, 353–363. [Google Scholar] [CrossRef]
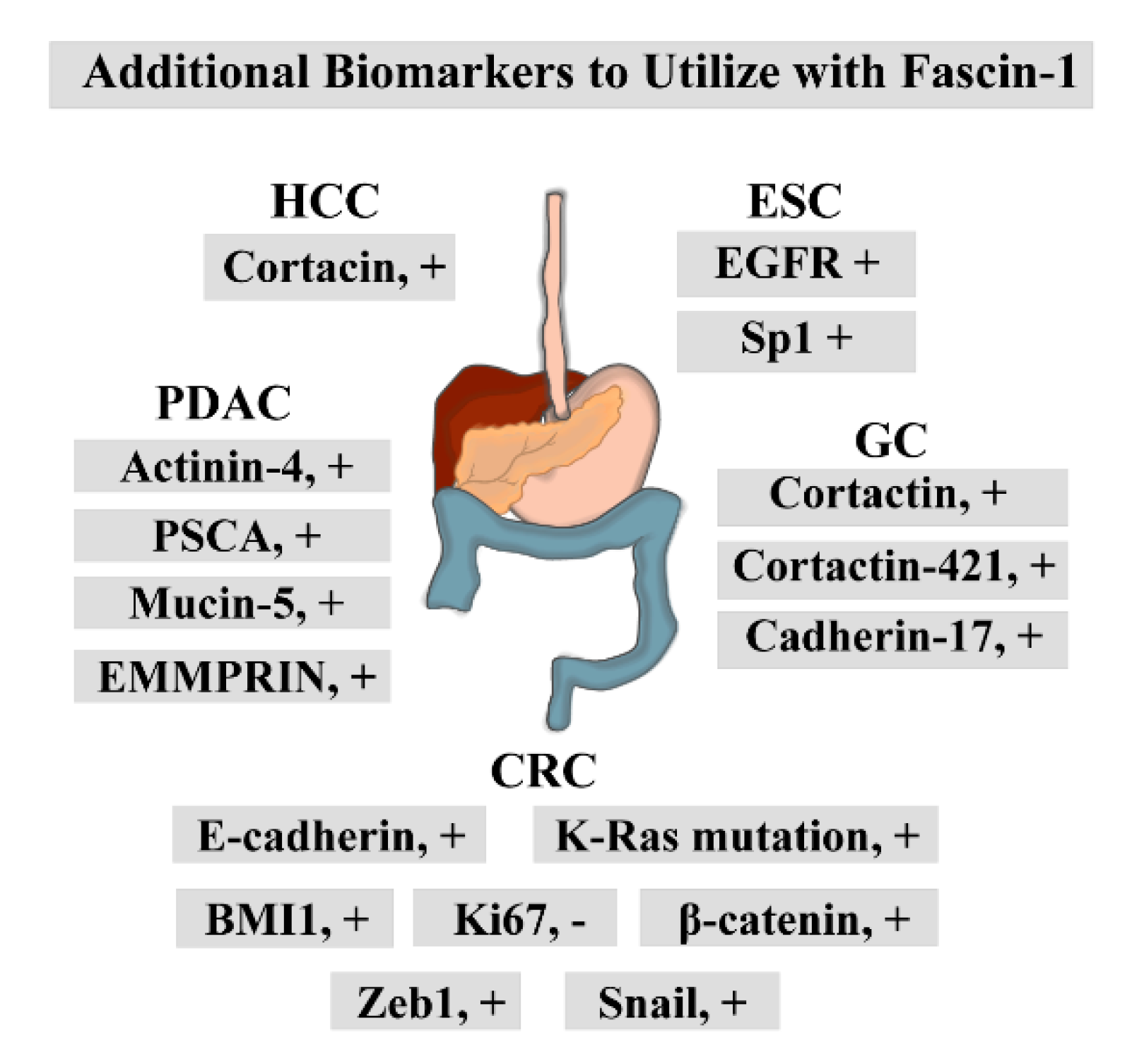
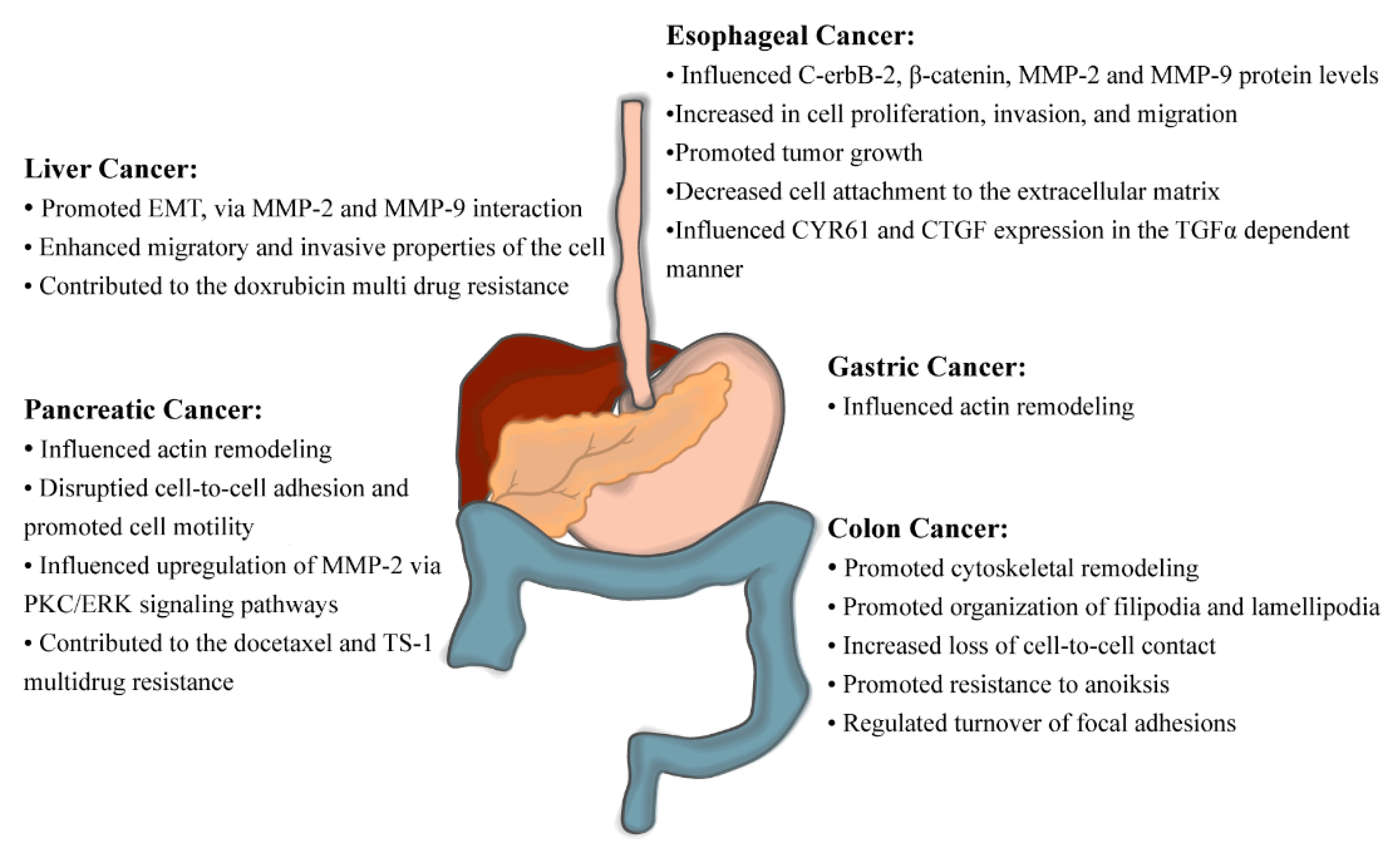
| Type of Cancer | Refs. | Methods | Correlation Between High Fascin-1 Expression and: | Independent Factor | |||
|---|---|---|---|---|---|---|---|
| Lymph Node Metastasis | Distant Metastasis | Reduced Survival | Other Clinicopathological Outcomes | ||||
| Esophageal Cancer | [18] | IHC, rt-PCR, WB | + | N/A | N/A | Stage-dependent progression of ESCC | N/A |
| Cell proliferation | |||||||
| [19] | IHC | + | N/A | + | Tumor stage (III and IV) | + | |
| [20] | IHC | + | + | N/A | Tumor differentiation | N/A | |
| Poor differentiation | |||||||
| T4 stage | |||||||
| [21] | IHC | + | + | + | Advanced tumor | + | |
| Gastric Adenocarcinoma | [22] | rt-qPCR | + | + | N/A | Tumor differentiation | N/A |
| Advanced tumor | |||||||
| [23] | IHC | + | N/A | + | Tumor size | N/A | |
| Depth of invasion | |||||||
| Lymphatic and venous invasion | |||||||
| UICC staging | |||||||
| [24] | IHC | + | - | + | Extent of primary tumor | - | |
| Age | |||||||
| Serosal invasion | |||||||
| Histopathological grading | |||||||
| TNM staging | |||||||
| Recurrence | |||||||
| [25] | IHC | N/A | N/A | + | TNM staging | N/A | |
| High-grade histopathological differentiation | |||||||
| [26] | IHC | N/A | N/A | + | Tumor size | + | |
| [27] | IHC | + | N/A | + | High clinical stage | + | |
| High T stage | |||||||
| Lymphovascular invasion | |||||||
| The intestinal type of Lauren classification | |||||||
| Colorectal Adenocarcinoma | [28] | IHC | + | + | + | Tumor grade and stage | + |
| Mucinous differentiation | |||||||
| Extranodal tumor extension | |||||||
| Increased recurrence rate | |||||||
| Cancer progression | |||||||
| [29] | IHC | N/A | N/A | N/A | Tumor size | N/A | |
| Histological type | |||||||
| Degree of dysplasia | |||||||
| [30] | IHC and qPCR | + | + | N/A | High expression in stage III/IV CRC | N/A | |
| [31] | IHC | N/A | N/A | + | Worse prognosis for stage III/IV patients | N/A | |
| [32] | IHC | N/A | N/A | + | Advanced tumor depth | + | |
| [33] | IHC | N/A | N/A | N/A | Advanced dysplasia | N/A | |
| High-grade histopathological differentiation | |||||||
| Advanced T stage | |||||||
| [34] | IHC | + | N/A | + | Invasive tumors and advanced cancer stage | N/A | |
| [35] | IHC | N/A | N/A | N/A | Adenocarcinoma type without mucosal component | N/A | |
| [36] | IHC | N/A | N/A | + | Increased tumor budding | N/A | |
| Systemic inflammation | |||||||
| Decreased memory T-cells | |||||||
| [37] | IHC | + | + | + | Progressive anatomic disease extent | + | |
| Higher T classification | |||||||
| High-grade tumors | |||||||
| Increased vascular invasion | |||||||
| [38] | IHC | N/A | + | + | Increased recurrence rate | + | |
| [39] | IHC | N/A | N/A | N/A | High expression in anti-EGFR resistant CRC | N/A | |
| Hepatocellular Carcinoma | [40] | IHC | + | + | + | Histological differentiation | N/A |
| Metastasis | |||||||
| [41] | IHC | + | + | + | Advanced Differentiation | N/A | |
| Tumor size | |||||||
| Regional and distant metastasis | |||||||
| [42] | IHC | N/A | N/A | N/A | No correlation with clinicopathological parameters | N/A | |
| Pancreatic Adenocarcinoma | [43] | IHC | N/A | N/A | N/A | Advanced PanIN, stage-dependent | N/A |
| [44] | IHC | + | + | + | Advanced tumor grade | N/A | |
| Advanced T stages | |||||||
| Histological grade and clinical stages | |||||||
| [45] | IHC | N/A | N/A | + | Increased recurrence rate | N/A | |
| Increased vascular invasion | |||||||
| [46] | IHC | N/A | N/A | N/A | Advanced PanIN, stage-dependent | N/A | |
| [47] | IHC | N/A | N/A | + | Advanced tumor grade | N/A | |
| [48] | IHC | N/A | N/A | + | Histological grade | N/A | |
| American Joint Committee on Cancer Stage | |||||||
| [49] | IHC | N/A | N/A | N/A | N/A: High background with anti-fascin-1 | N/A | |
| [50] | IHC | N/A | N/A | N/A | N/A: Antibody with high specificity but low sensitivity | N/A | |
| Compound | Cancer Type | Cell Lines | In Vivo Models | Clinical Trial Data | Ref. |
|---|---|---|---|---|---|
| Compound G2 | Colorectal Cancer | HCT-116, DLD-1 | Zebrafish model of invasion | No | [93] |
| Polymethoxylated Chalcones 3/14 | HCT-116, LoVo, HT-29, NCE-1 E6/E7 | Unknown | No | [95] | |
| Imipramine | SW-480, DLD-1, HCT-15, HCT-116, HT-29, LS174T, SW-620, LoVo | Zebrafish model of invasion | No | [96] | |
| Raltegravir | HCT-116, DLD-1 | Zebrafish model of invasion | No | [98] | |
| Natural Killer Lysine | Hepatocellular Carcinoma | SMMC-7721, 97-H, HepG2 | Unknown | No | [99] |
| Doxycycline | Unknown | Thiocetamide HCC animal model | No | [100] | |
| Salinomycin | Pancreatic Adenocarcinoma | AsPC-1, Colo357, MiaPaCa-2, PANC-1, Panc02 | Orthotopic injection of Panc02 in C57Bl/6 mice | No | [101] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ristic, B.; Kopel, J.; Sherazi, S.A.A.; Gupta, S.; Sachdeva, S.; Bansal, P.; Ali, A.; Perisetti, A.; Goyal, H. Emerging Role of Fascin-1 in the Pathogenesis, Diagnosis, and Treatment of the Gastrointestinal Cancers. Cancers 2021, 13, 2536. https://doi.org/10.3390/cancers13112536
Ristic B, Kopel J, Sherazi SAA, Gupta S, Sachdeva S, Bansal P, Ali A, Perisetti A, Goyal H. Emerging Role of Fascin-1 in the Pathogenesis, Diagnosis, and Treatment of the Gastrointestinal Cancers. Cancers. 2021; 13(11):2536. https://doi.org/10.3390/cancers13112536
Chicago/Turabian StyleRistic, Bojana, Jonathan Kopel, Syed A. A. Sherazi, Shweta Gupta, Sonali Sachdeva, Pardeep Bansal, Aman Ali, Abhilash Perisetti, and Hemant Goyal. 2021. "Emerging Role of Fascin-1 in the Pathogenesis, Diagnosis, and Treatment of the Gastrointestinal Cancers" Cancers 13, no. 11: 2536. https://doi.org/10.3390/cancers13112536
APA StyleRistic, B., Kopel, J., Sherazi, S. A. A., Gupta, S., Sachdeva, S., Bansal, P., Ali, A., Perisetti, A., & Goyal, H. (2021). Emerging Role of Fascin-1 in the Pathogenesis, Diagnosis, and Treatment of the Gastrointestinal Cancers. Cancers, 13(11), 2536. https://doi.org/10.3390/cancers13112536







