The Expression Profile and Textural Characteristics of C595-Reactive MUC1 in Pancreatic Ductal Adenocarcinoma for Targeted Radionuclide Therapy
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
2.1. Surface Expression of C595-Reactive MUC1 on Pancreatic Cancer Cells
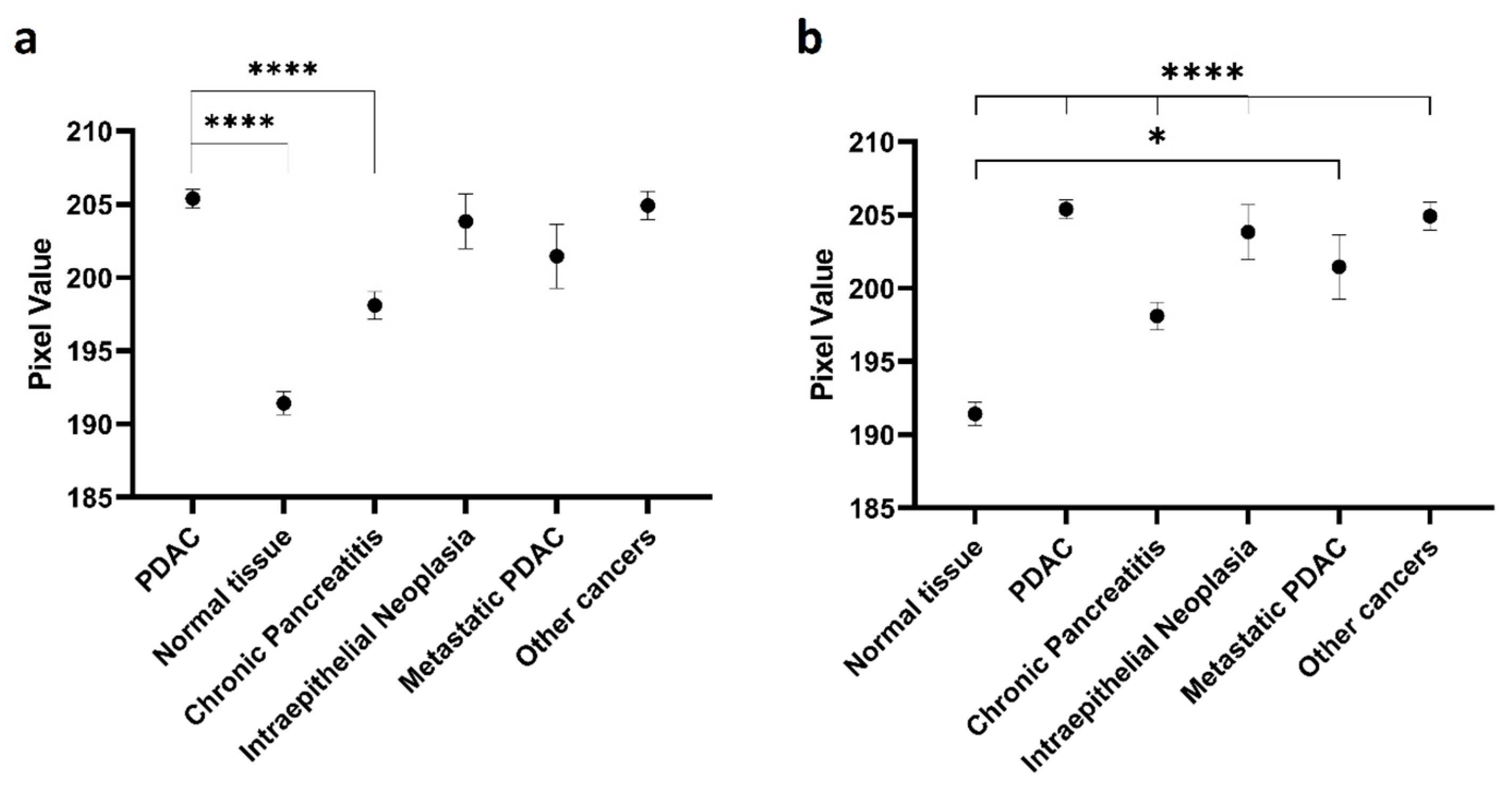
2.2. Expression of C595-Positive MUC1 on Pancreatic Tissues
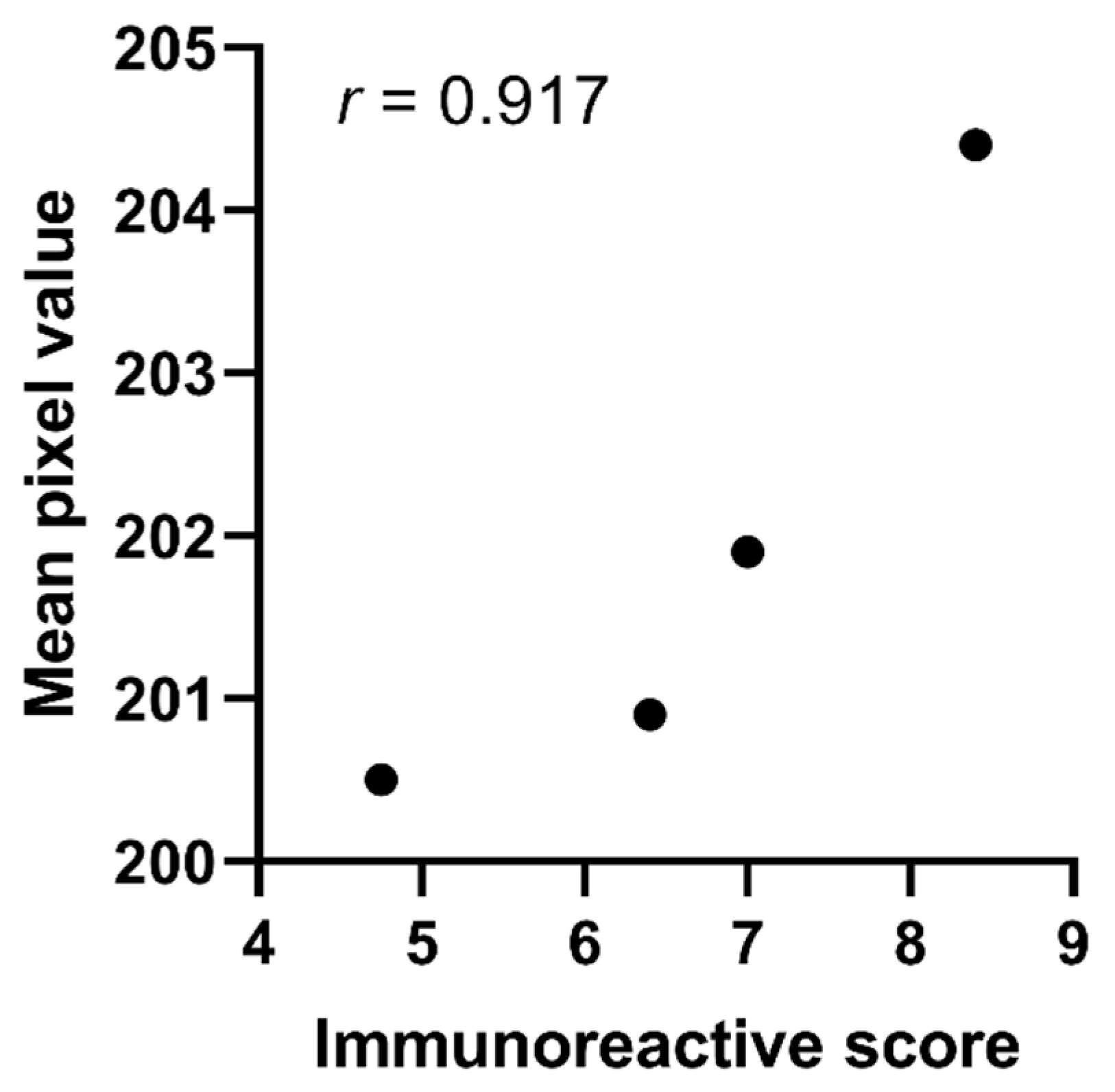
2.3. Validation of Pixel Analysis
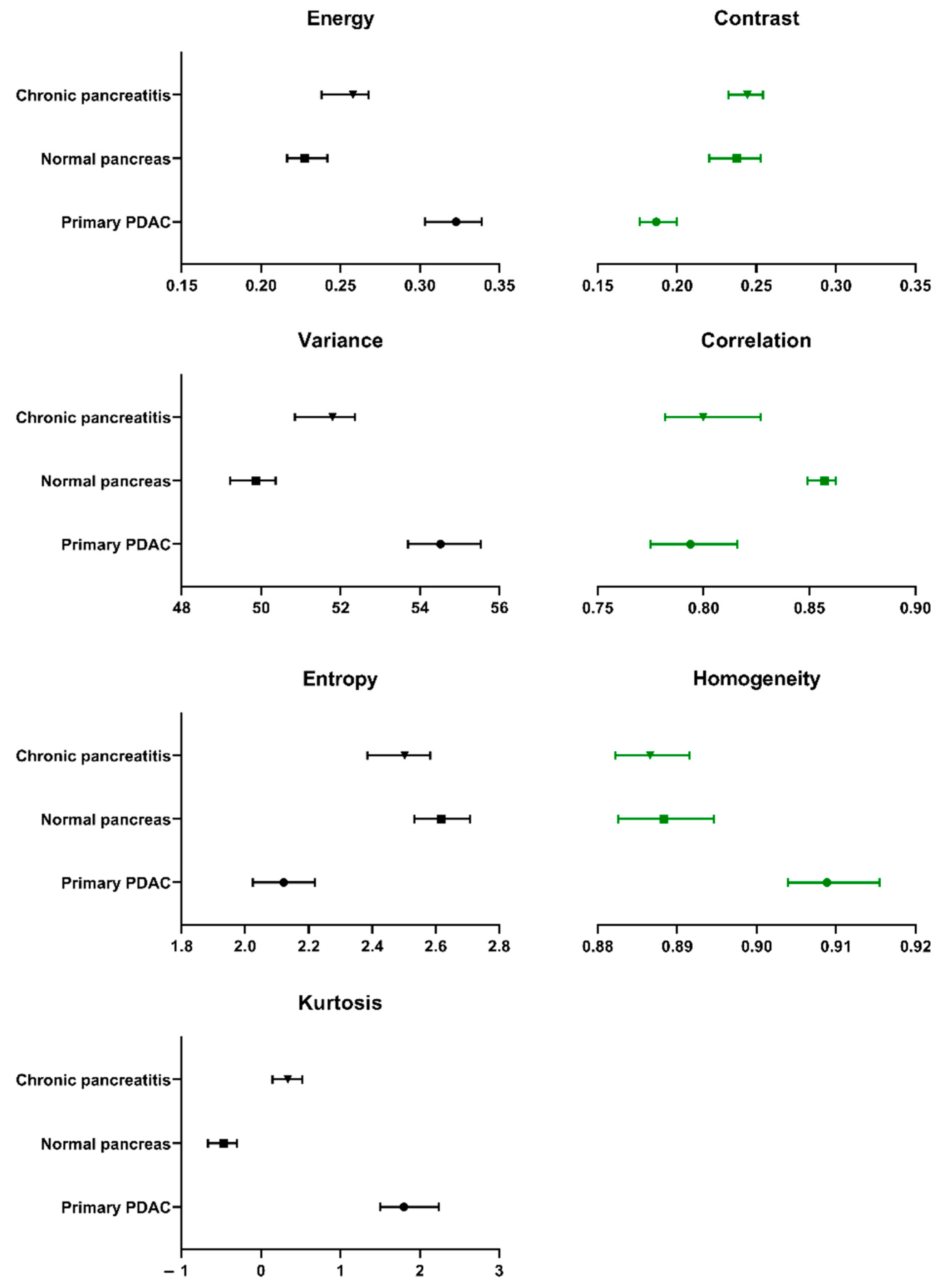
2.4. Textural Analysis of Pancreatic Tissues
3. Discussion
4. Materials and Methods
4.1. Antibodies
4.2. Cell Cultures
4.3. Flow Cytometry
4.4. Immunocytochemistry
4.5. Human Pancreatic Tissues
4.6. Immunohistochemical Staining
4.7. Analysis of Immunohistochemical Staining
4.7.1. Pixel Analysis
4.7.2. Validation of Pixel Analysis
4.7.3. Textural Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Australian Institute of Health and Welfare. Cancer in Australia: 2019; Government of Australia: Canberra, Australia, 2019.
- Lekka, K.; Tzitzi, E.; Giakoustidis, A.; Papadopoulos, V.; Giakoustidis, D. Contemporary management of borderline resectable pancreatic ductal adenocarcinoma. Ann. Hepato Biliary Pancreat. Surg. 2019, 23, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Adamska, A.; Domenichini, A.; Falasca, M. Pancreatic ductal adenocarcinoma: Current and evolving therapies. Int. J. Mol. Sci. 2017, 18, 1338. [Google Scholar] [CrossRef] [PubMed]
- Gudkov, S.V.; Shilyagina, N.Y.; Vodeneev, V.A.; Zvyagin, A.V. Targeted radionuclide therapy of human tumors. Int. J. Mol. Sci. 2015, 17, 33. [Google Scholar] [CrossRef] [PubMed]
- Al-Ejeh, F.; Pajic, M.; Shi, W.; Kalimutho, M.; Miranda, M.; Nagrial, A.M.; Chou, A.; Biankin, A.V.; Grimmond, S.M.; Brown, M.P.; et al. Gemcitabine and chk1 inhibition potentiate egfr-directed radioimmunotherapy against pancreatic ductal adenocarcinoma. Clin. Cancer Res. 2014, 20, 3187–3197. [Google Scholar] [CrossRef] [PubMed]
- Pasternack, J.B.; Domogauer, J.D.; Khullar, A.; Akudugu, J.M.; Howell, R.W. The advantage of antibody cocktails for targeted alpha therapy depends on specific activity. J. Nucl. Med. 2014, 55, 2012–2019. [Google Scholar] [CrossRef] [PubMed]
- Eilaghi, A.; Baig, S.; Zhang, Y.; Zhang, J.; Karanicolas, P.; Gallinger, S.; Khalvati, F.; Haider, M.A. Ct texture features are associated with overall survival in pancreatic ductal adenocarcinoma—a quantitative analysis. BMC Med Imaging 2017, 17, 38. [Google Scholar] [CrossRef]
- Rizzo, S.; Botta, F.; Raimondi, S.; Origgi, D.; Fanciullo, C.; Morganti, A.G.; Bellomi, M. Radiomics: The facts and the challenges of image analysis. Eur. Radiol. Exp. 2018, 2, 36. [Google Scholar] [CrossRef]
- Löfstedt, T.; Brynolfsson, P.; Asklund, T.; Nyholm, T.; Garpebring, A. Gray-level invariant haralick texture features. PLoS ONE 2019, 14, e0212110. [Google Scholar] [CrossRef]
- Constantinou, P.E.; Danysh, B.P.; Dharmaraj, N.; Carson, D.D. Transmembrane mucins as novel therapeutic targets. Expert Rev. Endocrinol. Metab. 2011, 6, 835–848. [Google Scholar] [CrossRef]
- Van Putten, J.P.M.; Strijbis, K. Transmembrane mucins: Signaling receptors at the intersection of inflammation and cancer. J. Innate Immun. 2017, 9, 281–299. [Google Scholar] [CrossRef]
- Suh, H.; Pillai, K.; Morris, D.L. Mucins in pancreatic cancer: Biological role, implications in carcinogenesis and applications in diagnosis and therapy. Am. J. Cancer Res. 2017, 7, 1372–1383. [Google Scholar] [PubMed]
- Hattrup, C.L.; Gendler, S.J. Structure and function of the cell surface (tethered) mucins. Annu. Rev. Physiol. 2008, 70, 431–457. [Google Scholar] [CrossRef] [PubMed]
- Nath, S.; Mukherjee, P. Muc1: A multifaceted oncoprotein with a key role in cancer progression. Trends Mol. Med. 2014, 20, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Dhar, P.; McAuley, J. The role of the cell surface mucin muc1 as a barrier to infection and regulator of inflammation. Front. Cell. Infect. Microbiol. 2019, 9, 117. [Google Scholar] [CrossRef]
- Gendler, S.J. Muc1, the renaissance molecule. J. Mammary Gland Biol. Neoplasia 2001, 6, 339–353. [Google Scholar] [CrossRef]
- Tinder, T.L.; Subramani, D.B.; Basu, G.D.; Bradley, J.M.; Schettini, J.; Million, A.; Skaar, T.; Mukherjee, P. Muc1 enhances tumor progression and contributes toward immunosuppression in a mouse model of spontaneous pancreatic adenocarcinoma. J. Immunol. 2008, 181, 3116–3125. [Google Scholar] [CrossRef]
- Roulois, D.; Grégoire, M.; Fonteneau, J.-F. Muc1-specific cytotoxic t lymphocytes in cancer therapy: Induction and challenge. Biomed. Res. Int. 2013, 2013, 871936. [Google Scholar] [CrossRef]
- Gendler, S.; Taylor-Papadimitriou, J.; Duhig, T.; Rothbard, J.; Burchell, J. A highly immunogenic region of a human polymorphic epithelial mucin expressed by carcinomas is made up of tandem repeats. J. Biol. Chem. 1988, 263, 12820–12823. [Google Scholar]
- Price, M.R.; Pugh, J.A.; Hudecz, F.; Griffiths, W.; Jacobs, E.; Symonds, I.M.; Clarke, A.J.; Chan, W.C.; Baldwin, R.W. C595—A monoclonal antibody against the protein core of human urinary epithelial mucin commonly expressed in breast carcinomas. Br. J. Cancer 1990, 61, 681–686. [Google Scholar] [CrossRef]
- Qu, C.; Li, Y.; Song, Y.; Rizvi, S.; Raja, C.; Zhang, D.; Samra, J.; Smith, R.; Perkins, A.C.; Apostolidis, C.; et al. Muc1 expression in primary and metastatic pancreatic cancer cells for in vitro treatment by 213bi-c595 radioimmunoconjugate. Br. J. Cancer 2004, 91, 2086–2093. [Google Scholar] [CrossRef]
- Wang, L.; Ma, J.; Liu, F.; Yu, Q.; Chu, G.; Perkins, A.C.; Li, Y. Expression of muc1 in primary and metastatic human epithelial ovarian cancer and its therapeutic significance. Gynecol. Oncol. 2007, 105, 695–702. [Google Scholar] [CrossRef]
- Croce, M.V.; Isla-Larrain, M.T.; Demichelis, S.O.; Segal-Eiras, A.; Gori, J.R.; Price, M.R. Tissue and serum muc1 mucin detection in breast cancer patients. Breast Cancer Res. Treat. 2003, 81, 195–207. [Google Scholar] [CrossRef]
- Stegmeier, F.; Warmuth, M.; Sellers, W.R.; Dorsch, M. Targeted cancer therapies in the twenty-first century: Lessons from imatinib. Clin. Pharmacol. Ther. 2010, 87, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Ocean, A.J.; Pennington, K.L.; Guarino, M.J.; Sheikh, A.; Bekaii-Saab, T.; Serafini, A.N.; Lee, D.; Sung, M.W.; Gulec, S.A.; Goldsmith, S.J.; et al. Fractionated radioimmunotherapy with 90y-clivatuzumab tetraxetan and low-dose gemcitabine is active in advanced pancreatic cancer: A phase 1 trial. Cancer 2012, 118, 5497–5506. [Google Scholar] [CrossRef] [PubMed]
- Picozzi, V.J.; Ramanathan, R.K.; Lowery, M.A.; Ocean, A.J.; Mitchel, E.P.; O’Neil, B.H.; Guarino, M.J.; Conkling, P.R.; Cohen, S.J.; Bahary, N.; et al. 90y-clivatuzumab tetraxetan with or without low-dose gemcitabine: A phase ib study in patients with metastatic pancreatic cancer after two or more prior therapies. Eur. J. Cancer 2015, 51, 1857–1864. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.Gov. Phase 3 Trial of 90y-Clivatuzumab Tetraxetan & Gemcitabine vs Placebo & Gemcitabine in Metastatic Pancreatic Cancer (Pancrit®-1) (nct01956812). Available online: https://clinicaltrials.gov/ct2/show/NCT01956812 (accessed on 29 August 2019).
- Wu, S.-t.; Williams, C.D.; Grover, P.A.; Moore, L.J.; Mukherjee, P. Early detection of pancreatic cancer in mouse models using a novel antibody, tab004. PLoS ONE 2018, 13, e0193260. [Google Scholar] [CrossRef]
- Park, J.Y.; Hiroshima, Y.; Lee, J.Y.; Maawy, A.A.; Hoffman, R.M.; Bouvet, M. Muc1 selectively targets human pancreatic cancer in orthotopic nude mouse models. PLoS ONE 2015, 10, e0122100. [Google Scholar] [CrossRef]
- Chhieng, D.C.; Benson, E.; Eltoum, I.; Eloubeidi, M.A.; Jhala, N.; Jhala, D.; Siegal, G.P.; Grizzle, W.E.; Manne, U. Muc1 and muc2 expression in pancreatic ductal carcinoma obtained by fine-needle aspiration. Cancer 2003, 99, 365–371. [Google Scholar] [CrossRef]
- Fung, K.; Vivier, D.; Keinänen, O.; Sarbisheh, E.K.; Price, E.W.; Zeglis, B.M. 89zr-labeled ar20.5: A muc1-targeting immunopet probe. Molecules 2020, 25, 2315. [Google Scholar] [CrossRef]
- Gold, D.V.; Karanjawala, Z.; Modrak, D.E.; Goldenberg, D.M.; Hruban, R.H. Pam4-reactive muc1 is a biomarker for early pancreatic adenocarcinoma. Clin. Cancer Res. 2007, 13, 7380–7387. [Google Scholar] [CrossRef]
- Boustani, J.; Grapin, M.; Laurent, P.-A.; Apetoh, L.; Mirjolet, C. The 6th r of radiobiology: Reactivation of anti-tumor immune response. Cancers 2019, 11, 860. [Google Scholar] [CrossRef] [PubMed]
- Marcu, L.; Bezak, E.; Allen, B.J. Global comparison of targeted alpha vs targeted beta therapy for cancer: In vitro, in vivo and clinical trials. Crit. Rev. Oncol. Hematol. 2018, 123, 7–20. [Google Scholar] [CrossRef] [PubMed]
- Silver, D.A.; Pellicer, I.; Fair, W.R.; Heston, W.D.; Cordon-Cardo, C. Prostate-specific membrane antigen expression in normal and malignant human tissues. Clin. Cancer Res. 1997, 82, 52–539. [Google Scholar]
- Ghosh, A.; Heston, W.D. Tumor target prostate specific membrane antigen (PSMA) and its regulation in prostate cancer. J. Cell. Biochem. 2004, 91, 528–539. [Google Scholar] [CrossRef] [PubMed]
- Rahbar, K.; Ahmadzadehfar, H.; Kratochwil, C.; Haberkorn, U.; Schäfers, M.; Essler, M.; Baum, R.P.; Kulkarni, H.R.; Schmidt, M.; Drzezga, A.; et al. German multicenter study investigating 177lu-psma-617 radioligand therapy in advanced prostate cancer patients. J. Nucl. Med. 2017, 58, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Kratochwil, C.; Bruchertseifer, F.; Rathke, H.; Hohenfellner, M.; Giesel, F.L.; Haberkorn, U.; Morgenstern, A. Targeted α-therapy of metastatic castration-resistant prostate cancer with (225)ac-psma-617: Swimmer-plot analysis suggests efficacy regarding duration of tumor control. J. Nucl. Med. 2018, 59, 795–802. [Google Scholar] [CrossRef]
- Sandrasegaran, K.; Lin, Y.; Asare-Sawiri, M.; Taiyini, T.; Tann, M. Ct texture analysis of pancreatic cancer. Eur. Radiol. 2019, 29, 1067–1073. [Google Scholar] [CrossRef]
- Kim, H.S.; Kim, Y.J.; Kim, K.G.; Park, J.S. Preoperative ct texture features predict prognosis after curative resection in pancreatic cancer. Sci. Rep. 2019, 9, 17389. [Google Scholar] [CrossRef]
- Scalco, E.; Rizzo, G. Texture analysis of medical images for radiotherapy applications. Br. J. Radiol. 2017, 90, 20160642. [Google Scholar] [CrossRef]
- Nasief, H.; Zheng, C.; Schott, D.; Hall, W.; Tsai, S.; Erickson, B.; Allen Li, X. A machine learning based delta-radiomics process for early prediction of treatment response of pancreatic cancer. NPJ Precis. Oncol. 2019, 3, 25. [Google Scholar] [CrossRef]
- Monzel, R. Haralicktexturefeatures. Available online: https://au.mathworks.com/matlabcentral/fileexchange/58769-haralicktexturefeatures (accessed on 20 March 2020).
- Haralick, R.M.; Shanmugam, K.; Dinstein, I. Textural features for image classification. IEEE Trans. Syst. Man Cybern. 1973, SMC-3, 610–621. [Google Scholar] [CrossRef]
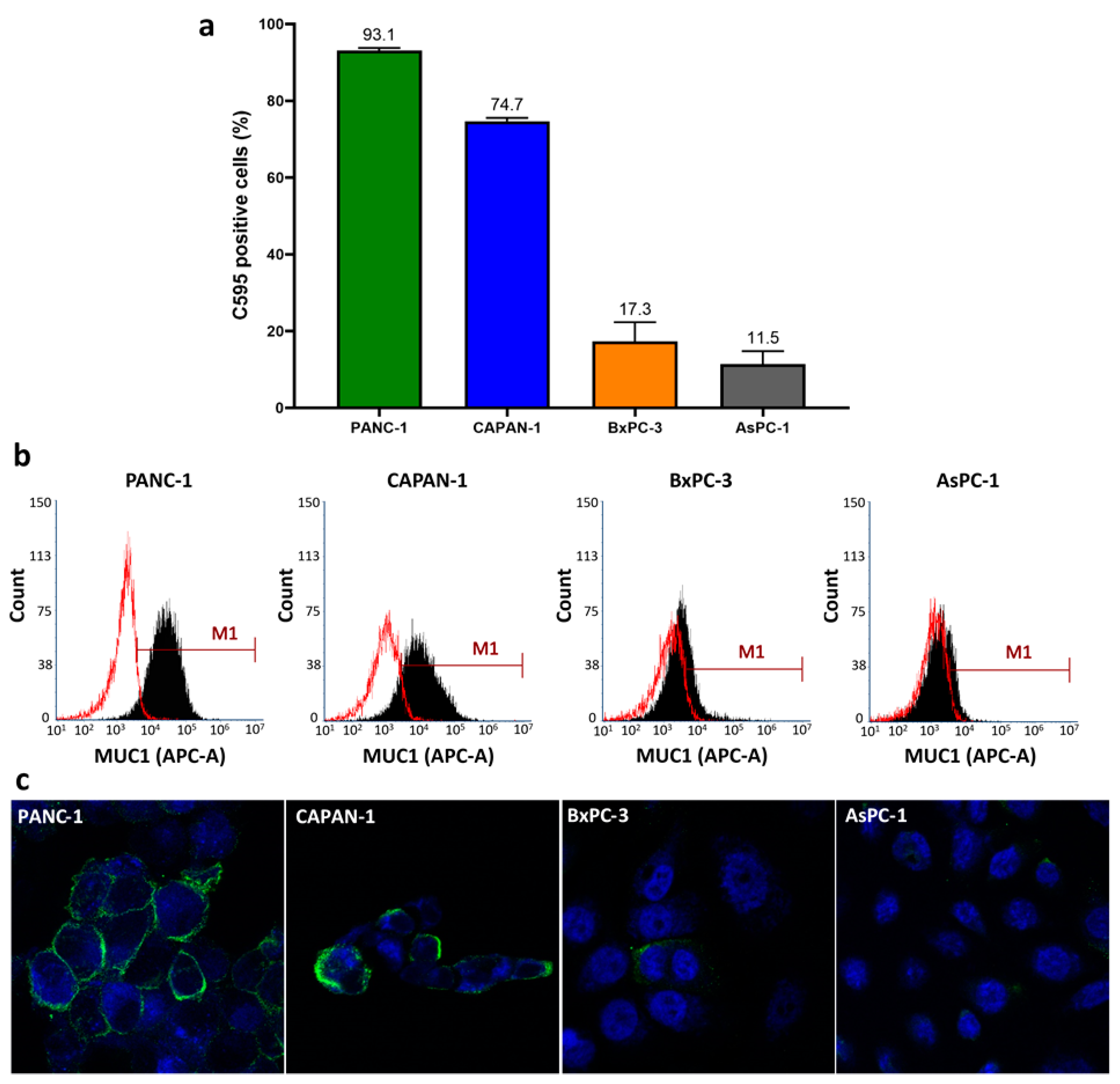
| Cell Line | Staining Intensity 1 | Staining Pattern |
|---|---|---|
| PANC-1 | 3 | Diffuse |
| CAPAN-1 | 2 | Focal |
| BxPC-3 | 1 | Diffuse |
| AsPC-1 | 1 | Focal |
| Disease Category | Sample Size (n) | Median Age (Range) |
|---|---|---|
| All samples | 357 | 54 (1083) |
| Normal pancreatic tissue | 97 | 54 (21–76) |
| Pancreatic ductal adenocarcinoma | 106 | 58 (23–83) |
| Metastatic pancreatic ductal adenocarcinoma | 10 | 59 (51–66) |
| Acute pancreatitis | 7 | 60 (47–68) |
| Chronic pancreatitis | 77 | 55 (10–76) |
| Pancreatic intraepithelial neoplasia | 22 | 52 (36–67) |
| Other cancer | 38 | 48 (17–77) |
| Disease Category | p-Values Compared to Control Tissue | |
|---|---|---|
| Test Tissues | PDAC | Normal Pancreatic Tissue |
| Pancreatic ductal adenocarcinoma | - | <0.0001 * |
| Normal tissue | <0.0001 * | - |
| Chronic pancreatitis | <0.0001 * | <0.0001 * |
| Pancreatic intraepithelial neoplasia | >0.9999 | <0.0001 * |
| Metastatic PDAC | 0.8882 | 0.0110 * |
| Other pancreatic cancers | >0.9999 | <0.0001 * |
| Disease Category | Average Score Assigned by Histopathologist | Average Immunoreactive Score 3 | Mean Pixel Value | |
|---|---|---|---|---|
| Percentage of Stained Cells 1 | Intensity of Staining 2 | |||
| Primary PDAC | 3.4 | 2.4 | 8.4 | 204.4 |
| Metastatic PDAC | 3.5 | 2.3 | 8.0 | 200.9 |
| Chronic Pancreatitis | 3.2 | 2.0 | 7.4 | 201.9 |
| Normal Pancreas | 2.8 | 1.6 | 4.8 | 200.5 |
| Textural Descriptor | PDAC | Normal Pancreas | Metastatic PDAC | Chronic Pancreatitis | PanIN | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Median | 95% CI | Median | 95% CI | Median | 95% CI | Median | 95% CI | Median | 95% CI | ||||||
| L | U | L | U | L | U | L | U | L | U | ||||||
| Sample size (n) | 106 | 97 | 10 | 77 | 22 | ||||||||||
| Energy | 0.3228 | 0.3031 | 0.3390 | 0.2273 | 0.2165 | 0.2418 | 0.2523 | 0.2308 | 0.3159 | 0.2580 | 0.2380 | 0.2676 | 0.2935 | 0.2579 | 0.3270 |
| Contrast | 0.1870 | 0.1766 | 0.199 | 0.2377 | 0.2204 | 0.2527 | 0.1988 | 0.1578 | 0.2155 | 0.2443 | 0.2324 | 0.2542 | 0.2241 | 0.1988 | 0.2348 |
| Correlation | 0.7939 | 0.7751 | 0.8159 | 0.8572 | 0.8492 | 0.8624 | 0.8446 | 0.7453 | 0.8741 | 0.7999 | 0.7818 | 0.8270 | 0.7853 | 0.7323 | 0.8189 |
| Variance | 54.52 | 53.70 | 55.55 | 49.87 | 49.23 | 50.37 | 51.32 | 50.68 | 54.98 | 51.80 | 50.85 | 52.37 | 53.31 | 51.89 | 55.27 |
| Homogeneity | 0.9089 | 0.9039 | 0.9155 | 0.8884 | 0.8826 | 0.8946 | 0.9035 | 0.8972 | 0.9224 | 0.8866 | 0.8823 | 0.8916 | 0.8948 | 0.8894 | 0.9061 |
| Sum Average | 14.73 | 14.60 | 14.88 | 14.02 | 13.95 | 14.09 | 14.27 | 14.14 | 14.82 | 14.35 | 14.21 | 14.42 | 14.57 | 14.37 | 14.85 |
| Sum Variance | 180.8 | 178.2 | 187.0 | 156.5 | 154.1 | 159.1 | 164.5 | 158.7 | 184.7 | 166.3 | 162.5 | 169.6 | 174.7 | 168.0 | 186.0 |
| Sum Entropy | 1.337 | 1.272 | 1.380 | 1.639 | 1.601 | 1.684 | 1.510 | 1.268 | 1.662 | 1.530 | 1.481 | 1.587 | 1.383 | 1.272 | 1.516 |
| Entropy | 2.122 | 2.025 | 2.219 | 2.617 | 2.533 | 2.709 | 2.371 | 2.060 | 2.633 | 2.502 | 2.385 | 2.583 | 2.212 | 2.038 | 2.420 |
| Difference Variance | 0.1580 | 0.1497 | 0.1669 | 0.1970 | 0.1835 | 0.2087 | 0.1659 | 0.1345 | 0.1793 | 0.2032 | 0.1927 | 0.2092 | 0.1853 | 0.1661 | 0.1970 |
| Difference Entropy | 0.4828 | 0.4678 | 0.5027 | 0.5526 | 0.5312 | 0.5721 | 0.5010 | 0.4370 | 0.5248 | 0.5612 | 0.5469 | 0.5742 | 0.5362 | 0.5013 | 0.5464 |
| Information Measure of Correlation I | −0.4427 | −0.4737 | −0.4253 | −0.4996 | −0.5174 | −0.4787 | −0.5072 | −0.5671 | −0.3923 | −0.4452 | −0.4738 | −0.4378 | −0.4367 | −0.4979 | −0.3747 |
| Information Measure of Correlation II | 0.9979 | 0.9973 | 0.9983 | 0.9996 | 0.9995 | 0.9996 | 0.9992 | 0.9971 | 0.9996 | 0.9991 | 0.9989 | 0.9994 | 0.9983 | 0.9970 | 0.9991 |
| Maximal Correlation Coefficient | 0.8169 | 0.7951 | 0.8287 | 0.8795 | 0.8694 | 0.8902 | 0.8605 | 0.7599 | 0.8930 | 0.8387 | 0.8196 | 0.8616 | 0.8144 | 0.7437 | 0.8709 |
| Kurtosis | 1.802 | 1.498 | 2.244 | −0.4732 | −0.666 | −0.3023 | 0.4964 | −0.6286 | 1.192 | 0.3385 | 0.1463 | 0.5188 | 0.7882 | −0.06413 | 1.767 |
| Textural Descriptor | Normal Tissue | Chronic Pancreatitis |
|---|---|---|
| p-Values Compared to PDAC Tissues (* p < 0.05) | ||
| Energy | <0.0001 * | <0.0001 * |
| Contrast | <0.0001 * | <0.0001 * |
| Correlation | <0.0001 * | 0.3614 |
| Variance | <0.0001 * | <0.0001 * |
| Homogeneity | <0.0001 * | <0.0001 * |
| Sum Average | <0.0001 * | <0.0001 * |
| Sum Variance | <0.0001 * | <0.0001 * |
| Sum Entropy | <0.0001 * | <0.0001 * |
| Entropy | <0.0001 * | <0.0001 * |
| Difference Variance | <0.0001 * | <0.0001 * |
| Difference Entropy | <0.0001 * | <0.0001 * |
| Information Measure of Correlation I | 0.0006 * | 0.7915 |
| Information Measure of Correlation II | <0.0001 * | <0.0001 * |
| Maximal Correlation Coefficient | <0.0001 * | 0.0230 * |
| Kurtosis | <0.0001 * | <0.0001 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hull, A.; Li, Y.; Bartholomeusz, D.; Hsieh, W.; Escarbe, S.; Ruszkiewicz, A.; Bezak, E. The Expression Profile and Textural Characteristics of C595-Reactive MUC1 in Pancreatic Ductal Adenocarcinoma for Targeted Radionuclide Therapy. Cancers 2021, 13, 61. https://doi.org/10.3390/cancers13010061
Hull A, Li Y, Bartholomeusz D, Hsieh W, Escarbe S, Ruszkiewicz A, Bezak E. The Expression Profile and Textural Characteristics of C595-Reactive MUC1 in Pancreatic Ductal Adenocarcinoma for Targeted Radionuclide Therapy. Cancers. 2021; 13(1):61. https://doi.org/10.3390/cancers13010061
Chicago/Turabian StyleHull, Ashleigh, Yanrui Li, Dylan Bartholomeusz, William Hsieh, Samantha Escarbe, Andrew Ruszkiewicz, and Eva Bezak. 2021. "The Expression Profile and Textural Characteristics of C595-Reactive MUC1 in Pancreatic Ductal Adenocarcinoma for Targeted Radionuclide Therapy" Cancers 13, no. 1: 61. https://doi.org/10.3390/cancers13010061
APA StyleHull, A., Li, Y., Bartholomeusz, D., Hsieh, W., Escarbe, S., Ruszkiewicz, A., & Bezak, E. (2021). The Expression Profile and Textural Characteristics of C595-Reactive MUC1 in Pancreatic Ductal Adenocarcinoma for Targeted Radionuclide Therapy. Cancers, 13(1), 61. https://doi.org/10.3390/cancers13010061





