DNA FISH Diagnostic Assay on Cytological Samples of Thyroid Follicular Neoplasms †
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
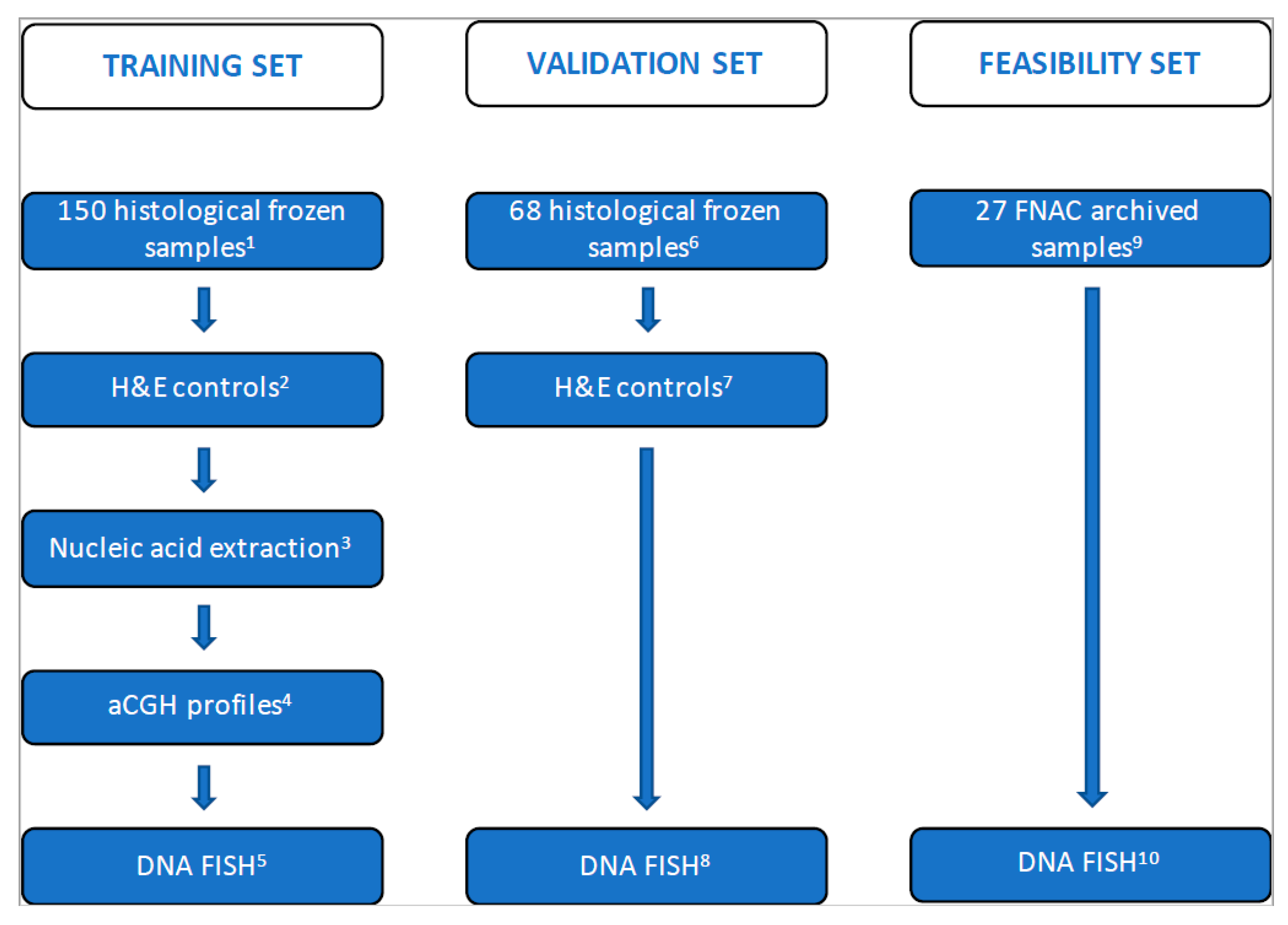
2.1. Study Design, Patients and Tissue Sample Characterization
2.2. DNA Ploidy Analysis
2.3. Nucleic Acid Extraction
2.4. Oligonucleotide aCGH Assay, Data Processing and Analysis
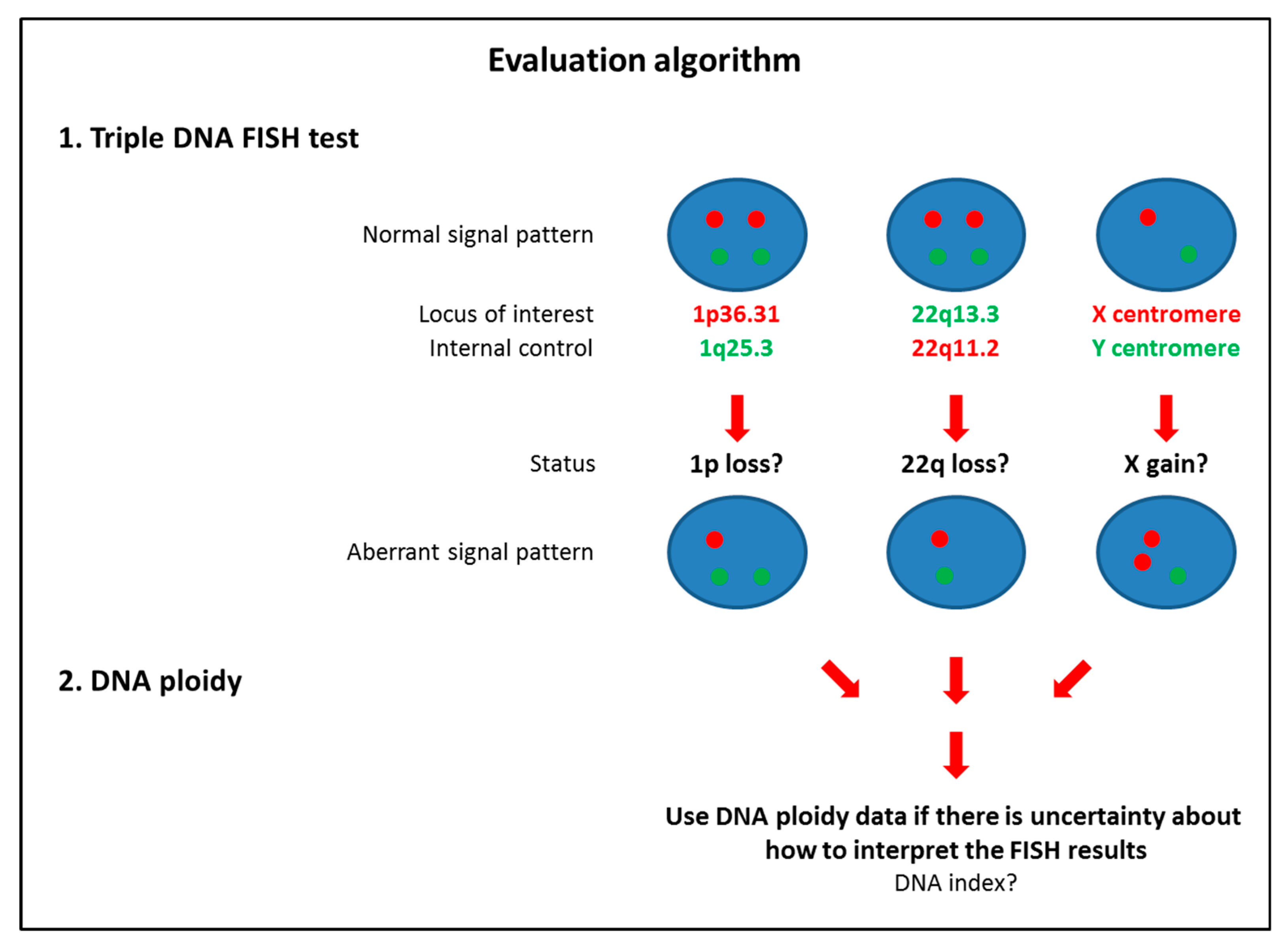
2.5. Triple DNA FISH Assay
2.6. Gene Mutation Analyses
2.7. Translocation Analyses
3. Results
3.1. Clinical, Histopathological and Biological Data
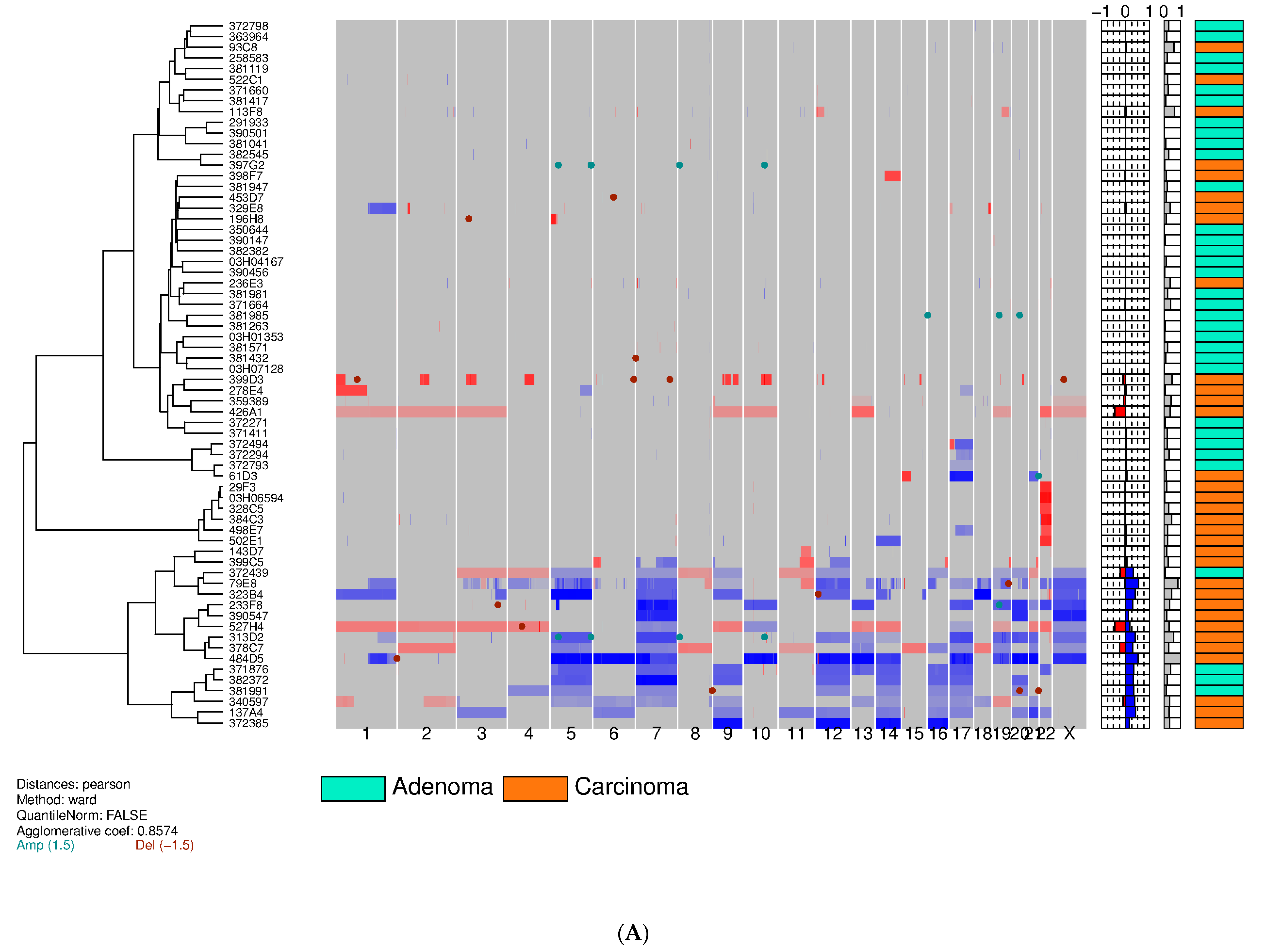
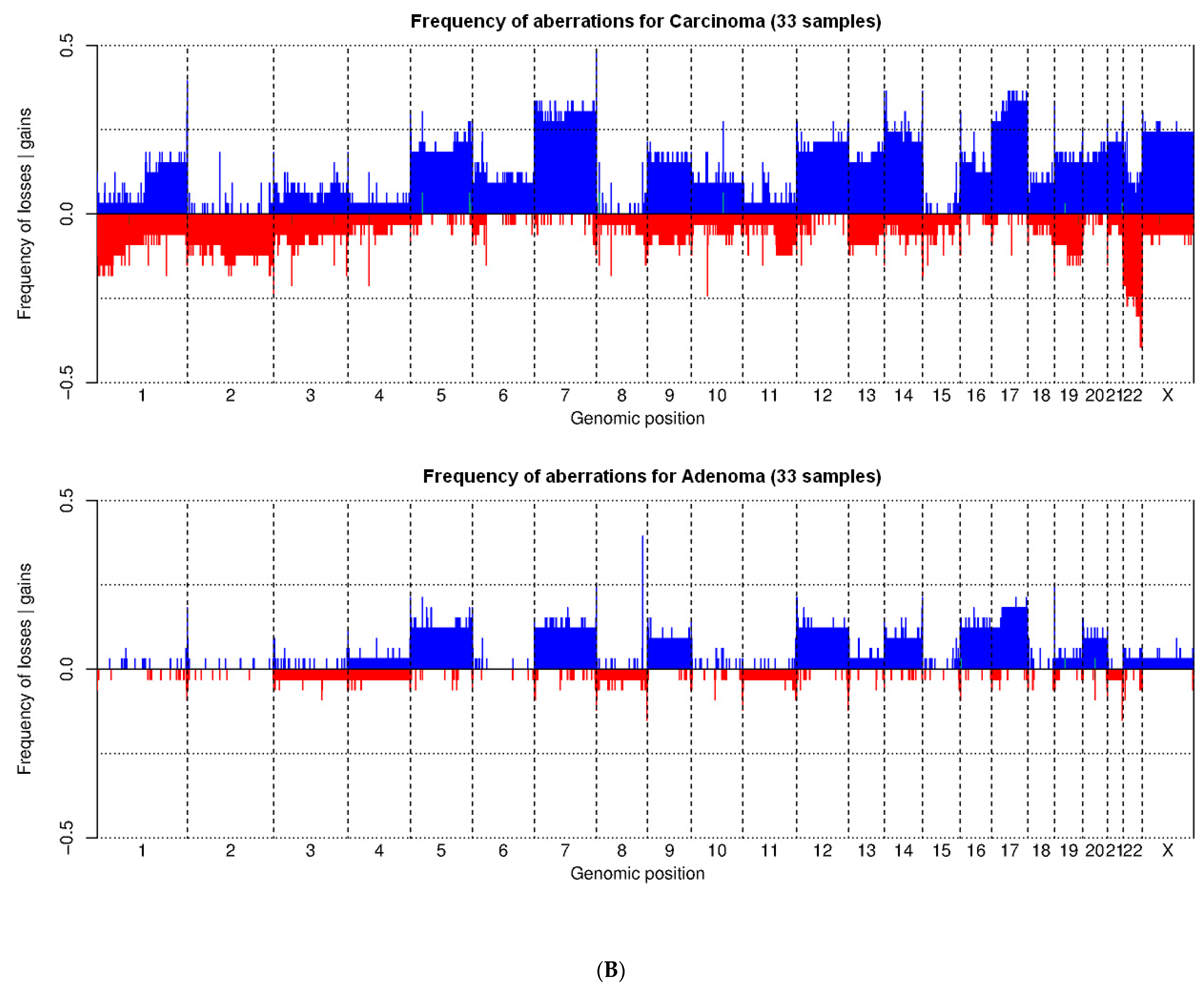
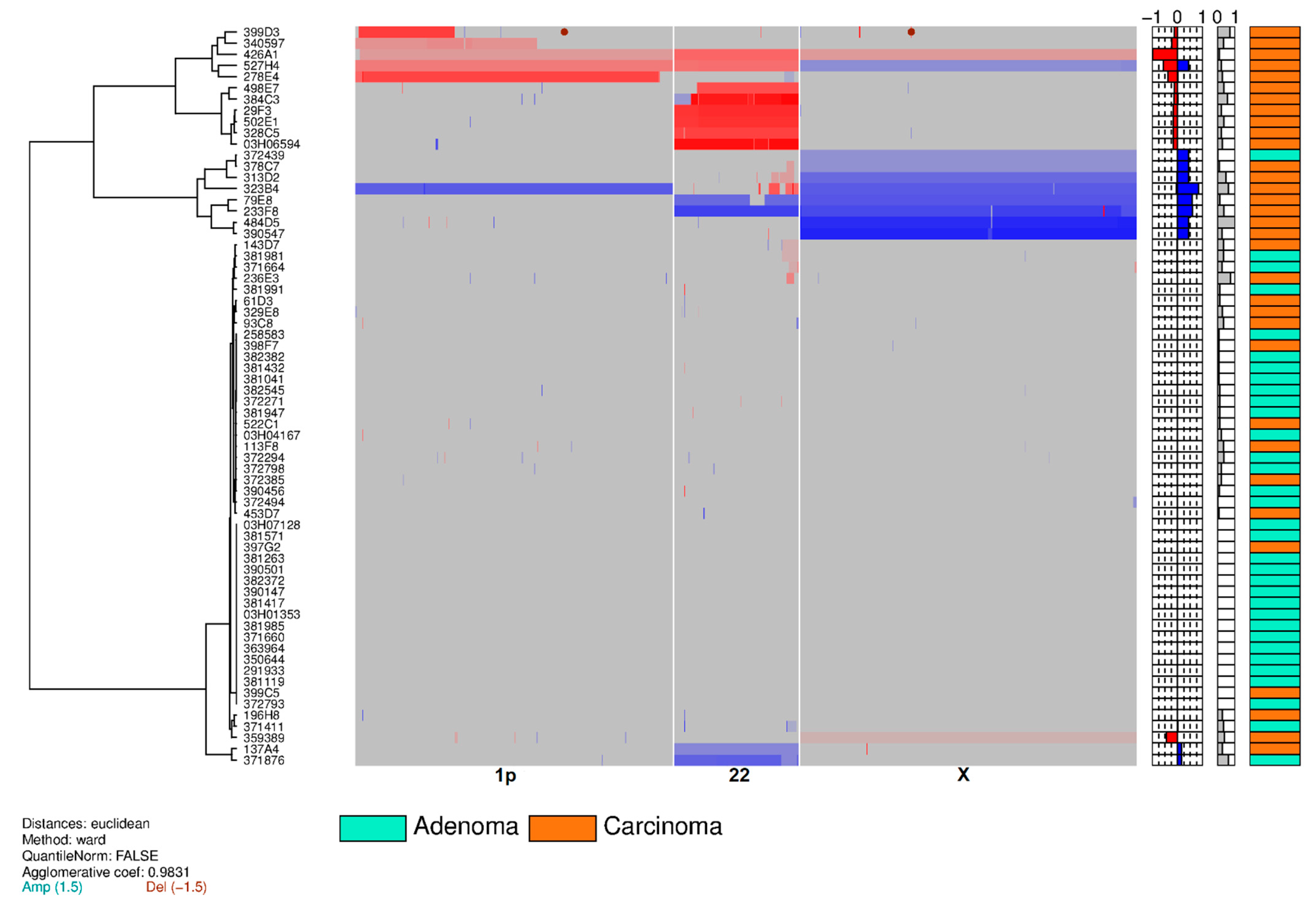
3.2. Training Set on Frozen Tissue
3.3. Validation Set on Touch Preparations from Frozen Tissue
3.4. Feasibility Set on Archived Already Stained Cytological Samples
3.5. FISH Accuracy
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American thyroid association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef]
- Gharib, H.; Papini, E.; Garber, J.R.; Duick, D.S.; Harrell, R.M.; Hegedus, L.; Paschke, R.; Valcavi, R.; Vitti, P. American association of clinical endocrinologists, American college of endocrinology, and associazione medici endocrinologi medical guidelines for clinical practice for the diagnosis and management of thyroid nodules—2016 update. Endocr. Pract. 2016, 22, 622–639. [Google Scholar] [CrossRef]
- Paschke, R.; Cantara, S.; Crescenzi, A.; Jarzab, B.; Musholt, T.J.; Simoes, M.S. European thyroid association guidelines regarding thyroid nodule molecular fine-needle aspiration cytology diagnostics. Eur. Thyroid J. 2017, 6, 115–129. [Google Scholar] [CrossRef]
- Schlumberger, M.J. Papillary and follicular thyroid carcinoma. N. Engl. J. Med. 1998, 338, 297–306. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef]
- Mazzaferri, E.L. Management of a solitary thyroid nodule. N. Engl. J. Med. 1993, 328, 553–559. [Google Scholar] [PubMed]
- Hegedüs, L. Clinical practice. The thyroid nodule. N. Engl. J. Med. 2004, 351, 1764–1771. [Google Scholar] [CrossRef] [PubMed]
- Burman, K.D.; Wartofsky, L. Clinical Practice. Thyroid nodules. N. Engl. J. Med. 2015, 373, 2347–2356. [Google Scholar] [CrossRef] [PubMed]
- Durante, C.; Grani, G.; Lamartina, L.; Filetti, S.; Mandel, S.J.; Cooper, D.S. The diagnosis and management of thyroid nodules: A review. JAMA 2018, 319, 914–924. [Google Scholar] [CrossRef]
- Haddad, R.I.; Nasr, C.; Bischoff, L.; Busaidy, N.L.; Byrd, D.; Callender, G.; Dickson, P.; Duh, Q.Y.; Ehya, H.; Goldner, W.; et al. NCCN guidelines insights: Thyroid carcinoma, version 2.2018. J. Natl. Compr. Cancer Netw. 2018, 16, 1429–1440. [Google Scholar] [CrossRef] [PubMed]
- Cibas, E.S.; Ali, S.Z. The Bethesda system for reporting thyroid cytopathology. Am. J. Clin. Pathol. 2009, 132, 658–665. [Google Scholar] [CrossRef]
- Cibas, E.S.; Ali, S.Z. The 2017 Bethesda system for reporting thyroid cytopathology. Thyroid 2017, 27, 1341–1346. [Google Scholar] [CrossRef] [PubMed]
- Bongiovanni, M.; Spitale, A.; Faquin, W.C.; Mazzucchelli, L.; Baloch, Z.W. The Bethesda system for reporting thyroid cytopathology: A meta-analysis. Acta Cytol. 2012, 56, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Nikiforov, Y.E.; Seethala, R.R.; Tallini, G.; Baloch, Z.W.; Basolo, F.; Thompson, L.D.R.; Barletta, J.A.; Wenig, B.M.; Al Ghuzlan, A.; Kakudo, K.; et al. Nomenclature revision for encapsulated follicular variant of papillary thyroid carcinoma: A paradigm shift to reduce overtreatment of indolent tumors. JAMA Oncol. 2016, 2, 1023–1029. [Google Scholar] [CrossRef] [PubMed]
- Nikiforov, Y.E.; Baloch, Z.W.; Hodak, S.P.; Giordano, T.J.; Lloyd, R.V.; Seethala, R.R.; Wenig, B.M. Change in diagnostic criteria for noninvasive follicular thyroid neoplasm with papillarylike nuclear features. JAMA Oncol. 2018, 4, 1125–1126. [Google Scholar] [CrossRef]
- World Health Organisation. Pathology and Genetics of Tumours of Endocrine Organs. In WHO Classification of Tumours of the Thyroid Gland, 3rd ed.; DeLellis, R.A., Llyod, R.V., Heitz, P.U., Eds.; International Agency for Research on Cancer (IARC) Press: Lyon, France, 2004; Volume 7. [Google Scholar]
- Aschebrook-Kilfoy, B.; Schechter, R.B.; Shih, Y.C.; Kaplan, E.L.; Chiu, B.C.; Angelos, P.; Grogan, R.H. The clinical and economic burden of a sustained increase in thyroid cancer incidence. Cancer Epidemiol. Biomark. Prev. 2013, 22, 1252–1259. [Google Scholar] [CrossRef] [PubMed]
- Nikiforov, Y.E. Role of molecular markers in thyroid nodule management: Then and now. Endocr. Pract. 2017, 23, 979–988. [Google Scholar] [CrossRef]
- Yeh, M.W. The rise and fall of surgical aggressiveness for known or suspected differentiated thyroid cancer. Cancer Cytopathol. 2020. [Google Scholar] [CrossRef]
- Cancer Genome Atlas Research Network. Integrated genomic characterization of papillary thyroid carcinoma. Cell 2014, 159, 676–690. [Google Scholar] [CrossRef]
- Giordano, T.J. Follicular cell thyroid neoplasia: Insights from genomics and the cancer genome atlas research network. Curr. Opin. Oncol. 2016, 28, 1–4. [Google Scholar] [CrossRef]
- Yoo, S.K.; Lee, S.; Kim, S.J.; Jee, H.G.; Kim, B.A.; Cho, H.; Song, Y.S.; Cho, S.W.; Won, J.K.; Shin, J.Y.; et al. Comprehensive analysis of the transcriptional and mutational landscape of follicular and papillary thyroid cancers. PLoS Genet. 2016, 12, e1006239. [Google Scholar] [CrossRef]
- Acquaviva, G.; Visani, M.; Repaci, A.; Rhoden, K.J.; De Biase, D.; Pession, A.; Giovanni, T. Molecular pathology of thyroid tumours of follicular cells: A review of genetic alterations and their clinicopathological relevance. Histopathology 2018, 72, 6–31. [Google Scholar] [CrossRef] [PubMed]
- Yakushina, V.D.; Lerner, L.V.; Lavrov, A.V. Gene fusions in thyroid cancer. Thyroid 2018, 28, 158–167. [Google Scholar] [CrossRef]
- Nikiforov, Y.E.; Ohori, N.P. Papillary Carcinoma. In Diagnostic Pathology and Molecular Genetics of the Thyroid: A Comprehensive Guide for Practicing Thyroid Pathology, 3rd ed.; Nikiforov, Y.E., Biddinger, P.W., Thompson, L.D.R., Eds.; Wolters Kluwer: Philadelphia, PA, USA, 2020; pp. 229–300. [Google Scholar]
- Efanov, A.A.; Brenner, A.V.; Bogdanova, T.I.; Kelly, L.M.; Liu, P.; Little, M.P.; Wald, A.I.; Hatch, M.; Zurnadzy, L.Y.; Nikiforova, M.N.; et al. Investigation of the relationship between radiation dose and gene mutations and fusions in post-chernobyl thyroid cancer. J. Natl. Cancer Inst. 2018, 110, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Nikiforov, Y.E. Thyroid Tumors: Classifications, and General Considerations. In Diagnostic Pathology and Molecular Genetics of the Thyroid: A Comprehensive Guide for Practicing Thyroid Pathology, 3rd ed.; Nikiforov, Y.E., Biddinger, P.W., Thompson, L.D.R., Eds.; Wolters Kluwer: Philadelphia, PA, USA, 2020; pp. 123–136. [Google Scholar]
- Nikiforov, Y.E.; Biddinger, P.W.; Thompson, L.D.R. Diagnostic Pathology and Molecular Genetics of the Thyroid: A Comprehensive Guide for Practicing Thyroid Pathology, 3rd ed.; Wolters Kluwer: Philadelphia, PA, USA, 2020. [Google Scholar]
- Gopal, R.K.; Kubler, K.; Calvo, S.E.; Polak, P.; Livitz, D.; Rosebrock, D.; Sadow, P.M.; Campbell, B.; Donovan, S.E.; Amin, S.; et al. Widespread chromosomal losses and mitochondrial DNA alterations as genetic drivers in Hurthle cell carcinoma. Cancer Cell 2018, 34, 242–255.e5. [Google Scholar] [CrossRef] [PubMed]
- Ganly, I.; Makarov, V.; Deraje, S.; Dong, Y.; Reznik, E.; Seshan, V.; Nanjangud, G.; Eng, S.; Bose, P.; Kuo, F.; et al. Integrated genomic analysis of Hürthle cell cancer reveals oncogenic drivers, recurrent mitochondrial mutations, and unique chromosomal landscapes. Cancer Cell 2018, 34, 256–270.e5. [Google Scholar] [CrossRef]
- Ganly, I.; McFadden, D.G. Short review: Genomic alterations in Hürthle cell carcinoma. Thyroid 2019, 29, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Grani, G.; Lamartina, L.; Durante, C.; Filetti, S.; Cooper, D.S. Follicular thyroid cancer and Hurthle cell carcinoma: Challenges in diagnosis, treatment, and clinical management. Lancet Diabetes Endocrinol. 2018, 6, 500–514. [Google Scholar] [CrossRef]
- Gonzalez, J.L.; Wang, H.H.; Ducatman, B.S. Fine-needle aspiration of Hürthle cell lesions. A cytomorphologic approach to diagnosis. Am. J. Clin. Pathol. 1993, 100, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Renshaw, A.A. Hürthle cell carcinoma is a better gold standard than Hürthle cell neoplasm for fine-needle aspiration of the thyroid: Defining more consistent and specific cytologic criteria. Cancer 2002, 96, 261–266. [Google Scholar] [CrossRef]
- Rossi, E.D.; Martini, M.; Straccia, P.; Raffaelli, M.; Pennacchia, I.; Marrucci, E.; Lombardi, C.P.; Pontecorvi, A.; Fadda, G. The cytologic category of oncocytic (Hurthle) cell neoplasm mostly includes low-risk lesions at histology: An institutional experience. Eur. J. Endocrinol. 2013, 169, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Auger, M. Hürthle cells in fine-needle aspirates of the thyroid: A review of their diagnostic criteria and significance. Cancer Cytopathol. 2014, 122, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Sobrinho-Simões, M.; Eloy, C.; Magalhães, J.; Lobo, C.; Amaro, T. Follicular thyroid carcinoma. Mod. Pathol. 2011, 24, S10–S18. [Google Scholar] [CrossRef] [PubMed]
- Rosai, J.; DeLellis, R.A.; Carcangiu, M.L.; Frable, W.J.; Tallini, G. Tumors of the Thyroid and Parathyroid Glands, AFIP Atlas of Tumor Pathology; American Registry of Pathology: Silver Spring, MD, USA, 2015. [Google Scholar]
- Ghossein, R. Update to the college of American pathologists reporting on thyroid carcinomas. Head Neck Pathol. 2009, 3, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Ghossein, R.A. Crucial parameters in thyroid carcinoma reporting—Challenges, controversies and clinical implications. Histopathology 2018, 72, 32–39. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. Follicular Thyroid Carcinoma. In WHO Classification of Tumors of Endocrine Organs, 4th ed.; Lloyd, R.V., Osamura, R.Y., Klöppel, G., Rosai, J., Eds.; International Agency for Research on Cancer (IARC): Lyon, France, 2017; pp. 92–95. [Google Scholar]
- Hiddemann, W.; Schumann, J.; Andreef, M.; Barlogie, B.; Herman, C.J.; Leif, R.C.; Mayall, B.H.; Murphy, R.F.; Sandberg, A.A. Convention on nomenclature for DNA cytometry. Committee on nomenclature, society for analytical cytology. Cancer Genet. Cytogenet. 1984, 13, 181–183. [Google Scholar] [CrossRef]
- Olshen, A.B.; Venkatraman, E.S.; Lucito, R.; Wigler, M. Circular binary segmentation for the analysis of array-based DNA copy number data. Biostatistics 2004, 5, 557–572. [Google Scholar] [CrossRef]
- Boichard, A.; Croux, L.; Al Ghuzlan, A.; Broutin, S.; Dupuy, C.; Leboulleux, S.; Schlumberger, M.; Bidart, J.M.; Lacroix, L. Somatic RAS mutations occur in a large proportion of sporadic RET-negative medullary thyroid carcinomas and extend to a previously unidentified exon. J. Clin. Endocrinol. Metab. 2012, 97, E2031–E2035. [Google Scholar] [CrossRef]
- Lacroix, L.; Mian, C.; Barrier, T.; Talbot, M.; Caillou, B.; Schlumberger, M.; Bidart, J.M. PAX8 and peroxisome proliferator-activated receptor gamma 1 gene expression status in benign and malignant thyroid tissues. Eur. J. Endocrinol. 2004, 151, 367–374. [Google Scholar] [CrossRef]
- Smida, J.; Salassidis, K.; Hieber, L.; Zitzelsberger, H.; Kellerer, A.M.; Demidchik, E.P.; Negele, T.; Spelsberg, F.; Lengfelder, E.; Werner, M.; et al. Distinct frequency of ret rearrangements in papillary thyroid carcinomas of children and adults from Belarus. Int. J. Cancer 1999, 80, 32–38. [Google Scholar] [CrossRef]
- Castro, P.; Eknaes, M.; Teixeira, M.R.; Danielsen, H.E.; Soares, P.; Lothe, R.A.; Sobrinho-Simoes, M. Adenomas and follicular carcinomas of the thyroid display two major patterns of chromosomal changes. J. Pathol. 2005, 206, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Hemmer, S.; Wasenius, V.M.; Knuutila, S.; Joensuu, H.; Franssila, K. Comparison of benign and malignant follicular thyroid tumours by comparative genomic hybridization. Br. J. Cancer 1998, 78, 1012–1017. [Google Scholar] [CrossRef] [PubMed]
- Hemmer, S.; Wasenius, V.M.; Knuutila, S.; Franssila, K.; Joensuu, H. DNA copy number changes in thyroid carcinoma. Am. J. Pathol. 1999, 154, 1539–1547. [Google Scholar] [CrossRef]
- Roque, L.; Rodrigues, R.; Pinto, A.; Moura-Nunes, V.; Soares, J. Chromosome imbalances in thyroid follicular neoplasms: A comparison between follicular adenomas and carcinomas. Genes Chromosom. Cancer 2003, 36, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Cope, L.; Sun, W.; Wang, Y.; Prasad, N.; Sangenario, L.; Talbot, K.; Somervell, H.; Westra, W.; Bishop, J.; et al. DNA copy number variations characterize benign and malignant thyroid tumors. J. Clin. Endocrinol. Metab. 2013, 98, E558–E566. [Google Scholar] [CrossRef]
- Singh, B.; Lim, D.; Cigudosa, J.C.; Ghossein, R.; Shaha, A.R.; Poluri, A.; Wreesmann, V.B.; Tuttle, M.; Shah, J.P.; Rao, P.H. Screening for genetic aberrations in papillary thyroid cancer by using comparative genomic hybridization. Surgery 2000, 128, 888–893. [Google Scholar] [CrossRef]
- Smit, J.W.; Van Zelderen-Bhola, S.; Merx, R.; De Leeuw, W.; Wessels, H.; Vink, R.; Morreau, H. A novel chromosomal translocation t(3;5)(q12;p15.3) and loss of heterozygosity on chromosome 22 in a multifocal follicular variant of papillary thyroid carcinoma presenting with skin metastases. Clin. Endocrinol. (Oxf.) 2001, 55, 543–548. [Google Scholar] [CrossRef]
- Swierniak, M.; Pfeifer, A.; Stokowy, T.; Rusinek, D.; Chekan, M.; Lange, D.; Krajewska, J.; Oczko-Wojciechowska, M.; Czarniecka, A.; Jarzab, M.; et al. Somatic mutation profiling of follicular thyroid cancer by next generation sequencing. Mol. Cell Endocrinol. 2016, 433, 130–137. [Google Scholar] [CrossRef]
- Nikiforov, Y.E.; Nikiforova, M.N. Molecular genetics and diagnosis of thyroid cancer. Nat. Rev. Endocrinol. 2011, 7, 569–580. [Google Scholar] [CrossRef]
- Pstrag, N.; Ziemnicka, K.; Bluyssen, H.; Wesoly, J. Thyroid cancers of follicular origin in a genomic light: In-depth overview of common and unique molecular marker candidates. Mol. Cancer 2018, 17, 116. [Google Scholar] [CrossRef]
- Song, Y.S.; Park, Y.J. Genomic characterization of differentiated thyroid carcinoma. Endocrinol. Metab. 2019, 34, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bakhoum, S.F.; Landau, D.A. Chromosomal instability as a driver of tumor heterogeneity and evolution. Cold Spring Harb. Perspect. Med. 2017, 7, a029611. [Google Scholar] [CrossRef] [PubMed]
- McGranahan, N.; Swanton, C. Clonal heterogeneity and tumor evolution: Past, present, and the future. Cell 2017, 168, 613–628. [Google Scholar] [CrossRef] [PubMed]
- Bakhoum, S.F.; Ngo, B.; Laughney, A.M.; Cavallo, J.A.; Murphy, C.J.; Ly, P.; Shah, P.; Sriram, R.K.; Watkins, T.B.K.; Taunk, N.K.; et al. Chromosomal instability drives metastasis through a cytosolic DNA response. Nature 2018, 553, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Sansregret, L.; Swanton, C. The role of aneuploidy in cancer evolution. Cold Spring Harb. Perspect. Med. 2017, 7, a028373. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.M.; Shih, J.; Ha, G.; Gao, G.F.; Zhang, X.; Berger, A.C.; Schumacher, S.E.; Wang, C.; Hu, H.; Liu, J.; et al. Genomic and functional approaches to understanding cancer aneuploidy. Cancer Cell 2018, 33, 676–689.e3. [Google Scholar] [CrossRef] [PubMed]
- Ward, L.S.; Brenta, G.; Medvedovic, M.; Fagin, J.A. Studies of allelic loss in thyroid tumors reveal major differences in chromosomal instability between papillary and follicular carcinomas. J. Clin. Endocrinol. Metab. 1998, 83, 525–530. [Google Scholar] [CrossRef]
- Roque, L.; Clode, A.; Belge, G.; Pinto, A.; Bartnitzke, S.; Santos, J.R.; Thode, B.; Bullerdiek, J.; Castedo, S.; Soares, J. Follicular thyroid carcinoma: Chromosome analysis of 19 cases. Genes Chromosom. Cancer 1998, 21, 250–255. [Google Scholar] [CrossRef]
- Greenebaum, E.; Koss, L.G.; Elequin, F.; Silver, C.E. The diagnostic value of flow cytometric DNA measurements in follicular tumors of the thyroid gland. Cancer 1985, 56, 2011–2018. [Google Scholar] [CrossRef]
- Schelfhout, L.J.; Cornelisse, C.J.; Goslings, B.M.; Hamming, J.F.; Kuipers-Dijkshoorn, N.J.; Van de Velde, C.J.; Fleuren, G.J. Frequency and degree of aneuploidy in benign and malignant thyroid neoplasms. Int. J. Cancer 1990, 45, 16–20. [Google Scholar] [CrossRef]
- Jung, S.H.; Kim, M.S.; Jung, C.K.; Park, H.C.; Kim, S.Y.; Liu, J.; Bae, J.S.; Lee, S.H.; Kim, T.M.; Lee, S.H.; et al. Mutational burdens and evolutionary ages of thyroid follicular adenoma are comparable to those of follicular carcinoma. Oncotarget 2016, 7, 69638–69648. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liu, H.; Min, S.; Shen, Y.; Li, W.; Chen, Y.; Wang, X. CDK16 overexpressed in non-small cell lung cancer and regulates cancer cell growth and apoptosis via a p27-dependent mechanism. Biomed. Pharmacother. 2018, 103, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, M.A.; Hay, I.D.; Bartelt, D.H., Jr.; Ritland, S.R.; Dahl, R.J.; Grant, C.S.; Jenkins, R.B. Cytogenetic and molecular genetic studies of follicular and papillary thyroid cancers. J. Clin. Investig. 1991, 88, 1596–1604. [Google Scholar] [CrossRef] [PubMed]
- Tung, W.S.; Shevlin, D.W.; Kaleem, Z.; Tribune, D.J.; Wells, S.A., Jr.; Goodfellow, P.J. Allelotype of follicular thyroid carcinomas reveals genetic instability consistent with frequent nondisjunctional chromosomal loss. Genes Chromosom. Cancer 1997, 19, 43–51. [Google Scholar] [CrossRef]
- Segev, D.L.; Saji, M.; Phillips, G.S.; Westra, W.H.; Takiyama, Y.; Piantadosi, S.; Smallridge, R.C.; Nishiyama, R.H.; Udelsman, R.; Zeiger, M.A. Polymerase chain reaction-based microsatellite polymorphism analysis of follicular and Hürthle cell neoplasms of the thyroid. J. Clin. Endocrinol. Metab. 1998, 83, 2036–2042. [Google Scholar] [CrossRef]
- Kitamura, Y.; Shimizu, K.; Ito, K.; Tanaka, S.; Emi, M. Allelotyping of follicular thyroid carcinoma: Frequent allelic losses in chromosome arms 7q, 11p, and 22q. J. Clin. Endocrinol. Metab. 2001, 86, 4268–4272. [Google Scholar] [CrossRef]
- Grebe, S.K.; McIver, B.; Hay, I.D.; Wu, P.S.; Maciel, L.M.; Drabkin, H.A.; Goellner, J.R.; Grant, C.S.; Jenkins, R.B.; Eberhardt, N.L. Frequent loss of heterozygosity on chromosomes 3p and 17p without VHL or p53 mutations suggests involvement of unidentified tumor suppressor genes in follicular thyroid carcinoma. J. Clin. Endocrinol. Metab. 1997, 82, 3684–3691. [Google Scholar] [CrossRef]
- Wu, G.; Mambo, E.; Guo, Z.; Hu, S.; Huang, X.; Gollin, S.M.; Trink, B.; Ladenson, P.W.; Sidransky, D.; Xing, M. Uncommon mutation, but common amplifications, of the PIK3CA gene in thyroid tumors. J. Clin. Endocrinol. Metab. 2005, 90, 4688–4693. [Google Scholar] [CrossRef]
- Storchova, Z.; Pellman, D. From polyploidy to aneuploidy, genome instability and cancer. Nat. Rev. Mol. Cell Biol. 2004, 5, 45–54. [Google Scholar] [CrossRef]
- Fox, D.T.; Duronio, R.J. Endoreplication and polyploidy: Insights into development and disease. Development 2013, 140, 3–12. [Google Scholar] [CrossRef]
- Shu, Z.; Row, S.; Deng, W.M. Endoreplication: The good, the bad, and the ugly. Trends Cell Biol. 2018, 28, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Lopez, S.; Lim, E.L.; Horswell, S.; Haase, K.; Huebner, A.; Dietzen, M.; Mourikis, T.P.; Watkins, T.B.K.; Rowan, A.; Dewhurst, S.M.; et al. Interplay between whole-genome doubling and the accumulation of deleterious alterations in cancer evolution. Nat. Genet. 2020, 52, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Vasudevan, A.; Baruah, P.S.; Smith, J.C.; Wang, Z.; Sayles, N.M.; Andrews, P.; Kendall, J.; Leu, J.; Chunduri, N.K.; Levy, D.; et al. Single-chromosomal gains can function as metastasis suppressors and promoters in colon cancer. Dev. Cell 2020, 52, 413–428.e6. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Prasad, M.; Lemon, W.J.; Hampel, H.; Wright, F.A.; Kornacker, K.; LiVolsi, V.; Frankel, W.; Kloos, R.T.; Eng, C.; et al. Gene expression in papillary thyroid carcinoma reveals highly consistent profiles. Proc. Natl. Acad. Sci. USA 2001, 98, 15044–15049. [Google Scholar] [CrossRef] [PubMed]
- Chevillard, S.; Ugolin, N.; Vielh, P.; Ory, K.; Levalois, C.; Elliott, D.; Clayman, G.L.; El-Naggar, A.K. Gene expression profiling of differentiated thyroid neoplasms: Diagnostic and clinical implications. Clin. Cancer Res. 2004, 10, 6586–6597. [Google Scholar] [CrossRef]
- Frattini, M.; Ferrario, C.; Bressan, P.; Balestra, D.; De Cecco, L.; Mondellini, P.; Bongarzone, I.; Collini, P.; Gariboldi, M.; Pilotti, S.; et al. Alternative mutations of BRAF, RET and NTRK1 are associated with similar but distinct gene expression patterns in papillary thyroid cancer. Oncogene 2004, 23, 7436–7440. [Google Scholar] [CrossRef]
- Giordano, T.J.; Kuick, R.; Thomas, D.G.; Misek, D.E.; Vinco, M.; Sanders, D.; Zhu, Z.; Ciampi, R.; Roh, M.; Shedden, K.; et al. Molecular classification of papillary thyroid carcinoma: Distinct BRAF, RAS, and RET/PTC mutation-specific gene expression profiles discovered by DNA microarray analysis. Oncogene 2005, 24, 6646–6656. [Google Scholar] [CrossRef]
- Vasko, V.; Espinosa, A.V.; Scouten, W.; He, H.; Auer, H.; Liyanarachchi, S.; Larin, A.; Savchenko, V.; Francis, G.L.; De la Chapelle, A.; et al. Gene expression and functional evidence of epithelial-to-mesenchymal transition in papillary thyroid carcinoma invasion. Proc. Natl. Acad. Sci. USA 2007, 104, 2803–2808. [Google Scholar] [CrossRef]
- Knauf, J.A.; Sartor, M.A.; Medvedovic, M.; Lundsmith, E.; Ryder, M.; Salzano, M.; Nikiforov, Y.E.; Giordano, T.J.; Ghossein, R.A.; Fagin, J.A. Progression of BRAF-induced thyroid cancer is associated with epithelial-mesenchymal transition requiring concomitant MAP kinase and TGFβ signaling. Oncogene 2011, 30, 3153–3162. [Google Scholar] [CrossRef]
- He, H.; Jazdzewski, K.; Li, W.; Liyanarachchi, S.; Nagy, R.; Volinia, S.; Calin, G.A.; Liu, C.G.; Franssila, K.; Suster, S.; et al. The role of microRNA genes in papillary thyroid carcinoma. Proc. Natl. Acad. Sci. USA 2005, 102, 19075–19080. [Google Scholar] [CrossRef]
- Pallante, P.; Visone, R.; Ferracin, M.; Ferraro, A.; Berlingieri, M.T.; Troncone, G.; Chiappetta, G.; Liu, C.G.; Santoro, M.; Negrini, M.; et al. MicroRNA deregulation in human thyroid papillary carcinomas. Endocr. Relat. Cancer 2006, 13, 497–508. [Google Scholar] [CrossRef] [PubMed]
- Nikiforova, M.N.; Tseng, G.C.; Steward, D.; Diorio, D.; Nikiforov, Y.E. MicroRNA expression profiling of thyroid tumors: Biological significance and diagnostic utility. J. Clin. Endocrinol. Metab. 2008, 93, 1600–1608. [Google Scholar] [CrossRef]
- Finn, S.; Smyth, P.; O’Regan, E.; Cahill, S.; Toner, M.; Timon, C.; Flavin, R.; O’Leary, J.; Sheils, O. Low-level genomic instability is a feature of papillary thyroid carcinoma: An array comparative genomic hybridization study of laser capture microdissected papillary thyroid carcinoma tumors and clonal cell lines. Arch. Pathol. Lab. Med. 2007, 131, 65–73. [Google Scholar] [PubMed]
- Roque, L.; Castedo, S.; Gomes, P.; Soares, P.; Clode, A.; Soares, J. Cytogenetic findings in 18 follicular thyroid adenomas. Cancer Genet. Cytogenet. 1993, 67, 1–6. [Google Scholar] [CrossRef]
- Belge, G.; Roque, L.; Soares, J.; Bruckmann, S.; Thode, B.; Fonseca, E.; Clode, A.; Bartnitzke, S.; Castedo, S.; Bullerdiek, J. Cytogenetic investigations of 340 thyroid hyperplasias and adenomas revealing correlations between cytogenetic findings and histology. Cancer Genet. Cytogenet. 1998, 101, 42–48. [Google Scholar] [CrossRef]
- Frisk, T.; Kytola, S.; Wallin, G.; Zedenius, J.; Larsson, C. Low frequency of numerical chromosomal aberrations in follicular thyroid tumors detected by comparative genomic hybridization. Genes Chromosom. Cancer 1999, 25, 349–353. [Google Scholar] [CrossRef]
- Da Silva, L.; James, D.; Simpson, P.T.; Walker, D.; Vargas, A.C.; Jayanthan, J.; Lakhani, S.R.; McNicol, A.M. Tumor heterogeneity in a follicular carcinoma of thyroid: A study by comparative genomic hybridization. Endocr. Pathol. 2011, 22, 103–107. [Google Scholar] [CrossRef]
- Kjellman, P.; Lagercrantz, S.; Höög, A.; Wallin, G.; Larsson, C.; Zedenius, J. Gain of 1q and loss of 9q21.3-q32 are associated with a less favorable prognosis in papillary thyroid carcinoma. Genes Chromosom. Cancer 2001, 32, 43–49. [Google Scholar] [CrossRef]
- Wreesmann, V.B.; Sieczka, E.M.; Socci, N.D.; Hezel, M.; Belbin, T.J.; Childs, G.; Patel, S.G.; Patel, K.N.; Tallini, G.; Prystowsky, M.; et al. Genome-wide profiling of papillary thyroid cancer identifies MUC1 as an independent prognostic marker. Cancer Res. 2004, 64, 3780–3789. [Google Scholar] [CrossRef]
- Rodrigues, R.; Roque, L.; Espadinha, C.; Pinto, A.; Domingues, R.; Dinis, J.; Catarino, A.; Pereira, T.; Leite, V. Comparative genomic hybridization, BRAF, RAS, RET, and oligo-array analysis in aneuploid papillary thyroid carcinomas. Oncol. Rep. 2007, 18, 917–926. [Google Scholar] [CrossRef]
- Baldan, F.; Mio, C.; Allegri, L.; Passon, N.; Lepore, S.M.; Russo, D.; Damante, G. Evaluation of somatic genomic imbalances in thyroid carcinomas of follicular origin by CGH-based approaches. Minerva Endocrinol. 2018, 43, 285–293. [Google Scholar] [PubMed]
- Nicolson, N.G.; Murtha, T.D.; Dong, W.; Paulsson, J.O.; Choi, J.; Barbieri, A.L.; Brown, T.C.; Kunstman, J.W.; Larsson, C.; Prasad, M.L.; et al. Comprehensive genetic analysis of follicular thyroid carcinoma predicts prognosis independent of histology. J. Clin. Endocrinol. Metab. 2018, 103, 2640–2650. [Google Scholar] [CrossRef] [PubMed]
- Martincorena, I.; Campbell, P.J. Somatic mutation in cancer and normal cells. Science 2015, 349, 1483–1489. [Google Scholar] [CrossRef] [PubMed]
- Giordano, T.J. Genomic hallmarks of thyroid neoplasia. Annu. Rev. Pathol. 2018, 13, 141–162. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Knauf, J.A.; Gonsky, R.; Wang, M.; Lai, E.H.; Chissoe, S.; Fagin, J.A.; Korenberg, J.R. From amplification to gene in thyroid cancer: A high-resolution mapped bacterial-artificial-chromosome resource for cancer chromosome aberrations guides gene discovery after comparative genome hybridization. Am. J. Hum. Genet. 1998, 63, 625–637. [Google Scholar] [CrossRef]
- Qureshi, A.A.; Collins, V.P.; Jani, P. Genomic differences in benign and malignant follicular thyroid tumours using 1-Mb array-comparative genomic hybridisation. Eur. Arch. Otorhinolaryngol. 2013, 270, 325–335. [Google Scholar] [CrossRef]
- Rossi, E.D.; Pantanowitz, L.; Faquin, W.C. The role of molecular testing for the indeterminate thyroid FNA. Genes 2019, 10, 736. [Google Scholar] [CrossRef]
- Eszlinger, M.; Lau, L.; Ghaznavi, S.; Symonds, C.; Chandarana, S.P.; Khalil, M.; Paschke, R. Molecular profiling of thyroid nodule fine-needle aspiration cytology. Nat. Rev. Endocrinol. 2017, 13, 415–424. [Google Scholar] [CrossRef]
- Zhang, M.; Lin, O. Molecular testing of thyroid nodules: A review of current available tests for fine-needle aspiration specimens. Arch. Pathol. Lab. Med. 2016, 140, 1338–1344. [Google Scholar] [CrossRef]
- Nishino, M.; Nikiforova, M. Update on molecular testing for cytologically indeterminate thyroid nodules. Arch. Pathol. Lab. Med. 2018, 142, 446–457. [Google Scholar] [CrossRef]
- Nikiforova, M.N.; Mercurio, S.; Wald, A.I.; Barbi de Moura, M.; Callenberg, K.; Santana-Santos, L.; Gooding, W.E.; Yip, L.; Ferris, R.L.; Nikiforov, Y.E. Analytical performance of the ThyroSeq v3 genomic classifier for cancer diagnosis in thyroid nodules. Cancer 2018, 124, 1682–1690. [Google Scholar] [CrossRef] [PubMed]
- Steward, D.L.; Carty, S.E.; Sippel, R.S.; Yang, S.P.; Sosa, J.A.; Sipos, J.A.; Figge, J.J.; Mandel, S.; Haugen, B.R.; Burman, K.D.; et al. Performance of a multigene genomic classifier in thyroid nodules with indeterminate cytology: A prospective blinded multicenter study. JAMA Oncol. 2019, 5, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Alexander, E.K.; Kennedy, G.C.; Baloch, Z.W.; Cibas, E.S.; Chudova, D.; Diggans, J.; Friedman, L.; Kloos, R.T.; LiVolsi, V.A.; Mandel, S.J.; et al. Preoperative diagnosis of benign thyroid nodules with indeterminate cytology. N. Engl. J. Med. 2012, 367, 705–715. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.N.; Angell, T.E.; Babiarz, J.; Barth, N.M.; Blevins, T.; Duh, Q.Y.; Ghossein, R.A.; Harrell, R.M.; Huang, J.; Kennedy, G.C.; et al. Performance of a genomic sequencing classifier for the preoperative diagnosis of cytologically indeterminate thyroid nodules. JAMA Surg. 2018, 153, 817–824. [Google Scholar] [CrossRef]
- Labourier, E.; Shifrin, A.; Busseniers, A.E.; Lupo, M.A.; Manganelli, M.L.; Andruss, B.; Wylie, D.; Beaudenon-Huibregtse, S. Molecular testing for miRNA, mRNA, and DNA on fine-needle aspiration improves the preoperative diagnosis of thyroid nodules with indeterminate cytology. J. Clin. Endocrinol. Metab. 2015, 100, 2743–2750. [Google Scholar] [CrossRef]
- Benjamin, H.; Schnitzer-Perlman, T.; Shtabsky, A.; VandenBussche, C.J.; Ali, S.Z.; Kolar, Z.; Pagni, F.; Bar, D.; Meiri, E. Analytical validity of a microRNA-based assay for diagnosing indeterminate thyroid FNA smears from routinely prepared cytology slides. Cancer Cytopathol. 2016, 124, 711–721. [Google Scholar] [CrossRef]
- Vargas-Salas, S.; Martinez, J.R.; Urra, S.; Dominguez, J.M.; Mena, N.; Uslar, T.; Lagos, M.; Henriquez, M.; Gonzalez, H.E. Genetic testing for indeterminate thyroid cytology: Review and meta-analysis. Endocr. Relat. Cancer 2018, 25, R163–R177. [Google Scholar] [CrossRef]
- Balentine, C.J.; Vanness, D.J.; Schneider, D.F. Cost-effectiveness of lobectomy versus genetic testing (Afirma®) for indeterminate thyroid nodules: Considering the costs of surveillance. Surgery 2018, 163, 88–96. [Google Scholar] [CrossRef]
- Nicholson, K.J.; Roberts, M.S.; McCoy, K.L.; Carty, S.E.; Yip, L. Molecular testing versus diagnostic lobectomy in Bethesda III/IV thyroid nodules: A cost-effectiveness analysis. Thyroid 2019, 29, 1237–1243. [Google Scholar] [CrossRef]
- Sciacchitano, S.; Lavra, L.; Ulivieri, A.; Magi, F.; De Francesco, G.P.; Bellotti, C.; Salehi, L.B.; Trovato, M.; Drago, C.; Bartolazzi, A. Comparative analysis of diagnostic performance, feasibility and cost of different test-methods for thyroid nodules with indeterminate cytology. Oncotarget 2017, 8, 49421–49442. [Google Scholar] [CrossRef]
- Renshaw, A.A.; Wang, E.; Wilbur, D.; Hughes, J.H.; Haja, J.; Henry, M.R. Interobserver agreement on microfollicles in thyroid fine-needle aspirates. Arch. Pathol. Lab. Med. 2006, 130, 148–152. [Google Scholar] [PubMed]
- Gerhard, R.; Da Cunha Santos, G. Inter- and intraobserver reproducibility of thyroid fine needle aspiration cytology: An analysis of discrepant cases. Cytopathology 2007, 18, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Ohori, N.P.; Schoedel, K.E. Variability in the atypia of undetermined significance/follicular lesion of undetermined significance diagnosis in the Bethesda system for reporting thyroid cytopathology: Sources and recommendations. Acta Cytol. 2011, 55, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Kholová, I.; Ludvíková, M. Thyroid atypia of undetermined significance or follicular lesion of undetermined significance: An indispensable Bethesda 2010 diagnostic category or waste garbage? Acta Cytol. 2014, 58, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Renshaw, A.A. Subclassification of atypical cells of undetermined significance in direct smears of fine-needle aspirations of the thyroid: Distinct patterns and associated risk of malignancy. Cancer Cytopathol. 2011, 119, 322–327. [Google Scholar] [CrossRef]
- Padmanabhan, V.; Marshall, C.B.; Akdas Barkan, G.; Ghofrani, M.; Laser, A.; Tolgay Ocal, I.; David Sturgis, C.; Souers, R.; Kurtycz, D.F. Reproducibility of atypia of undetermined significance/follicular lesion of undetermined significance category using the Bethesda system for reporting thyroid cytology when reviewing slides from different institutions: A study of interobserver variability among cytopathologists. Diagn. Cytopathol. 2017, 45, 399–405. [Google Scholar]
- Rossi, E.D.; Bizzarro, T.; Martini, M.; Larocca, L.M.; Schmitt, F.; Vielh, P. Cytopathology of follicular cell nodules. Adv. Anat. Pathol. 2017, 24, 45–55. [Google Scholar] [CrossRef]
- Bellevicine, C.; Sgariglia, R.; Migliatico, I.; Vigliar, E.; D’Anna, M.; Nacchio, M.A.; Serra, N.; Malapelle, U.; Bongiovanni, M.; Troncone, G. Different qualifiers of AUS/FLUS thyroid FNA have distinct BRAF, RAS, RET/PTC, and PAX8/PPARg alterations. Cancer Cytopathol. 2018, 126, 317–325. [Google Scholar] [CrossRef]
- Fazeli, R.; VandenBussche, C.J.; Bishop, J.A.; Ali, S.Z. Cytological diagnosis of follicular variant of papillary thyroid carcinoma before and after the Bethesda system for reporting thyroid cytopathology. Acta Cytol. 2016, 60, 14–18. [Google Scholar] [CrossRef]
- Grant, E.G.; Tessler, F.N.; Hoang, J.K.; Langer, J.E.; Beland, M.D.; Berland, L.L.; Cronan, J.J.; Desser, T.S.; Frates, M.C.; Hamper, U.M.; et al. Thyroid ultrasound reporting lexicon: White paper of the ACR thyroid imaging, reporting and data system (TIRADS) committee. J. Am. Coll. Radiol. 2015, 12, 1272–1279. [Google Scholar] [CrossRef]
- Russ, G.; Bonnema, S.J.; Erdogan, M.F.; Durante, C.; Ngu, R.; Leenhardt, L. European thyroid association guidelines for ultrasound malignancy risk stratification of thyroid nodules in adults: The EU-TIRADS. Eur. Thyroid J. 2017, 6, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Tessler, F.N.; Middleton, W.D.; Grant, E.G. Thyroid imaging reporting and data system (TI-RADS): A user’s guide. Radiology 2018, 287, 1082. [Google Scholar] [CrossRef] [PubMed]
- Chaigneau, E.; Russ, G.; Royer, B.; Bigorgne, C.; Bienvenu-Perrard, M.; Rouxel, A.; Leenhardt, L.; Belin, L.; Buffet, C. TIRADS score is of limited clinical value for risk stratification of indeterminate cytological results. Eur. J. Endocrinol. 2018, 179, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Castellana, M.; Castellana, C.; Treglia, G.; Giorgino, F.; Giovanella, L.; Russ, G.; Trimboli, P. Performance of five ultrasound risk stratification systems in selecting thyroid nodules for FNA. J. Clin. Endocrinol. Metab. 2020, 105, dgz170. [Google Scholar] [CrossRef] [PubMed]
- Persichetti, A.; Di Stasio, E.; Coccaro, C.; Graziano, F.; Bianchini, A.; Di Donna, V.; Corsello, S.; Valle, D.; Bizzarri, G.; Frasoldati, A.; et al. Inter- and intraobserver agreement in the assessment of thyroid nodule ultrasound features and classification systems: A blinded multicenter study. Thyroid 2020, 30, 237–242. [Google Scholar] [CrossRef]
- Modi, L.; Sun, W.; Shafizadeh, N.; Negron, R.; Yee-Chang, M.; Zhou, F.; Simsir, A.; Sheth, S.; Brandler, T.C. Does a higher American college of radiology thyroid imaging reporting and data system (ACR TI-RADS) score forecast an increased risk of malignancy? A correlation study of ACR TI-RADS with FNA cytology in the evaluation of thyroid nodules. Cancer Cytopathol. 2020. [Google Scholar] [CrossRef]
- Castellana, M.; Piccardo, A.; Virili, C.; Scappaticcio, L.; Grani, G.; Durante, C.; Giovanella, L.; Trimboli, P. Can ultrasound systems for risk stratification of thyroid nodules identify follicular carcinoma? Cancer Cytopathol. 2020, 128, 250–259. [Google Scholar] [CrossRef]
- Alexander, A.A. US-based risk stratification “guidelines” for thyroid nodules: Quō Vādis? J. Clin. Ultrasound 2020, 48, 127–133. [Google Scholar] [CrossRef]
- Li, X.; Zhang, S.; Zhang, Q.; Wei, X.; Pan, Y.; Zhao, J.; Xin, X.; Qin, C.; Wang, X.; Li, J.; et al. Diagnosis of thyroid cancer using deep convolutional neural network models applied to sonographic images: A retrospective, multicohort, diagnostic study. Lancet Oncol. 2019, 20, 193–201. [Google Scholar] [CrossRef]
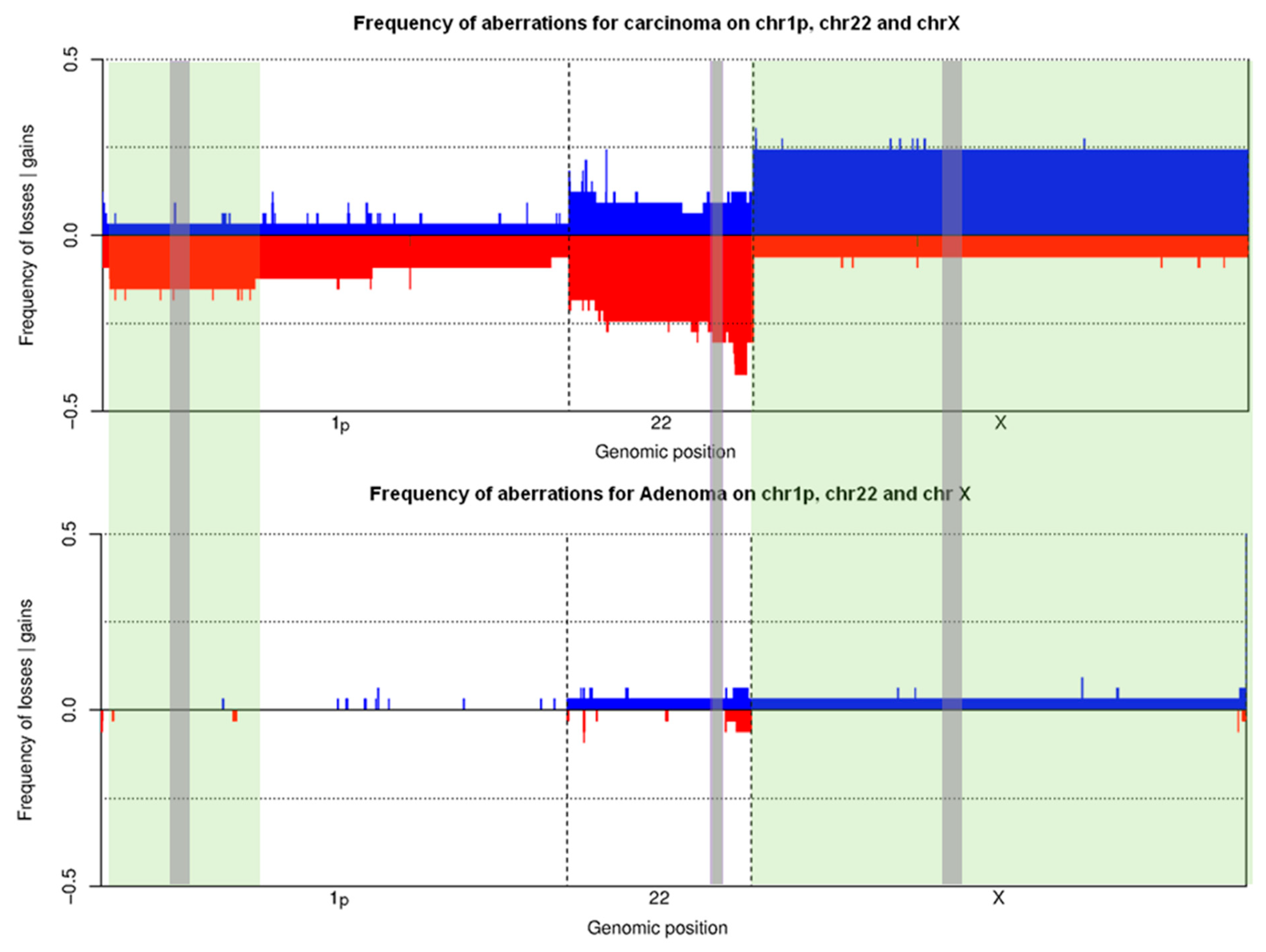


| Characteristics | Unit | Training Set | Validation Set | Total | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Adenoma | Carcinoma | Total | Adenoma | Carcinoma | Total | Adenoma | Carcinoma | Total | ||
| Patients | Number | 33 (50%) | 33 (50%) | 66 (100%) | 26 (43%) | 34 (57%) | 60 (100%) | 59 (47%) | 67 (53%) | 126 (100%) |
| Gender: | Number | |||||||||
| Female | 21 (64%) | 16 (48%) | 37 (56%) | 20 (77%) | 20 (59%) | 40 (67%) | 41 (69%) | 36 (54%) | 77 (61%) | |
| Male | 12 (36%) | 17 (52%) | 29 (44%) | 6 (23%) | 14 (41%) | 20 (33%) | 18 (31%) | 31 (46%) | 49 (39%) | |
| Age med. (range) | Years | 52 (21–70) | 53 (25–78) | 52 (21–78) | 47 (27–75) | 54 (21–82) | 51 (21–82) | 50 (21–75) | 53 (21–82) | 52 (21–82) |
| T size med. (range) | cm | 3 (1–6.4) | 4.5 (1–15) | 3.8 (1–15) | 2.8 (1.5–9) | 4.4 (1.5–11) | 3.7 (1.5–11) | 2.9 (1–9) | 4.5 (1–15) | 3.7 (1–15) |
| Follow-up med. (range) | Months | 35 (3–180) | 47 (0–140) | 41 (0–180) | 12 (1–48) | 42 (2–120) | 29 (1–120) | 25 (1–180) | 44 (0–140) | 35 (0–180) |
| Clinical status | Number, alive | 32 (100%) | 30 (91%) | 62 (95%) | 26 (100%) | 30 (88%) | 56 (93%) | 58 (100%) | 60 (90%) | 118 (94%) |
| Mutations: | ||||||||||
| KRAS/NA | 0 (0%)/1 | 0 (0%)/1 | – | 1 (5%)/4 | 0 (0%)/5 | – | 1 (2%)/5 | 0 (0%)/6 | – | |
| HRAS/NA | 1 (3%)/1 | 5 (16%)/2 | – | 1 (4%)/2 | 6 (20%)/4 | – | 2 (4%)/3 | 11 (18%)/6 | – | |
| NRAS/NA | 2 (6%)/1 | 6 (20%)/3 | – | 4 (17%)/2 | 8 (27%)/4 | – | 6 (11%)/3 | 14 (23%)/7 | – | |
| BRAF/NA | 0 (0%)/1 | 1 (3%)/1 | – | 0 (0%)/2 | 0 (0%)/4 | – | 0 (0%)/3 | 1 (2%)/6 | – | |
| CTNNB1/NA | 0 (0%)/1 | 0 (0%)/1 | – | – | – | – | 0 (0%)/1 | 0 (0%)/1 | – | |
| PIK3CA/NA | 0 (0%)/1 | 0 (0%)/3 | – | – | – | – | 0 (0%)/1 | 0 (0%)/3 | – | |
| Translocations: | ||||||||||
| PAX8/PPARγ1/NA | 0 (0%)/1 | 3 (12%)/7 | – | 0 (0%)/2 | 2 (6%)/1 | – | 0 (0%)/3 | 5 (8%)/8 | – | |
| RET/PTC1/NA | 0 (0%)/0 | 0 (0%)/7 | – | 0 (0%)/0 | 0(0%)/0 | – | 0 (0%)/0 | 0 (0%)/7 | – | |
| Chromosome | Training Set | Validation Set | Feasibility Set | Total | ||||
|---|---|---|---|---|---|---|---|---|
| Adenoma aCGH (%)/FISH (%) | Carcinoma aCGH (%)/FISH (%) | Adenoma aCGH (%)/FISH (%) | Carcinoma aCGH (%)/FISH (%) | Adenoma aCGH (%)/FISH (%) | Carcinoma aCGH (%)/FISH (%) | Adenoma aCGH (%)/FISH (%) | Carcinoma aCGH (%)/FISH (%) | |
| 1p36.31 loss | 0 (0%)/0 (0%) | 5 (15%)/5 (15%) | –/0 (0%) | –/3 (9%)# | –/0 (0%) | –/2 (14%) | 0 (0%)/0 (0%) | 5 (6%)/10 (12%) |
| 22q13.3 loss | 0 (0%)/1 (3%) | 8 (24%)/8 (24%) | –/0 (0%) | –/12 (35%)# | –/0 (0%) | –/2 (14%) | 0 (0%)/1 (1.4%) | 8 (9.9%)/22 (27%) |
| X gain | 1 (3%)/0 (0%) * | 8 (24%)/8 (24%) ** | –/0 (0%) | –/4 (12%)# | –/0 (0%) | –/2 (14%) | 1 (1.4%)/0 (0%) * | 8 (9.9%)/14 (17.3%) ** |
| Total | 2 alterations (1 aCGH, 1 FISH) in 2 patients (6%) | 22 alterations (21 aCGH, 21 FISH) ** in 18 patients *** (55%) | 0 alteration (FISH) (0%) | 19 alterations (FISH) in 16 patients (47%) | 0 alteration (FISH) (0%) | 6 alterations (FISH) in 6 patients (43%) | 2 alterations (1 aCGH, 1 FISH) in 2 patients (2.8%) | 47 alterations (21 aCGH, 46 FISH) ** in 40 patients (49.4%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vielh, P.; Balogh, Z.; Suciu, V.; Richon, C.; Job, B.; Meurice, G.; Valent, A.; Lacroix, L.; Marty, V.; Motte, N.; et al. DNA FISH Diagnostic Assay on Cytological Samples of Thyroid Follicular Neoplasms. Cancers 2020, 12, 2529. https://doi.org/10.3390/cancers12092529
Vielh P, Balogh Z, Suciu V, Richon C, Job B, Meurice G, Valent A, Lacroix L, Marty V, Motte N, et al. DNA FISH Diagnostic Assay on Cytological Samples of Thyroid Follicular Neoplasms. Cancers. 2020; 12(9):2529. https://doi.org/10.3390/cancers12092529
Chicago/Turabian StyleVielh, Philippe, Zsofia Balogh, Voichita Suciu, Catherine Richon, Bastien Job, Guillaume Meurice, Alexander Valent, Ludovic Lacroix, Virginie Marty, Nelly Motte, and et al. 2020. "DNA FISH Diagnostic Assay on Cytological Samples of Thyroid Follicular Neoplasms" Cancers 12, no. 9: 2529. https://doi.org/10.3390/cancers12092529
APA StyleVielh, P., Balogh, Z., Suciu, V., Richon, C., Job, B., Meurice, G., Valent, A., Lacroix, L., Marty, V., Motte, N., Dessen, P., Caillou, B., Ghuzlan, A. A., Bidart, J.-M., Lazar, V., Hofman, P., Scoazec, J.-Y., El-Naggar, A. K., & Schlumberger, M. (2020). DNA FISH Diagnostic Assay on Cytological Samples of Thyroid Follicular Neoplasms. Cancers, 12(9), 2529. https://doi.org/10.3390/cancers12092529







