Hypofractionated Radiotherapy for Anaplastic Thyroid Cancer: Systematic Review and Pooled Analysis
Simple Summary
Abstract
1. Introduction
2. Results
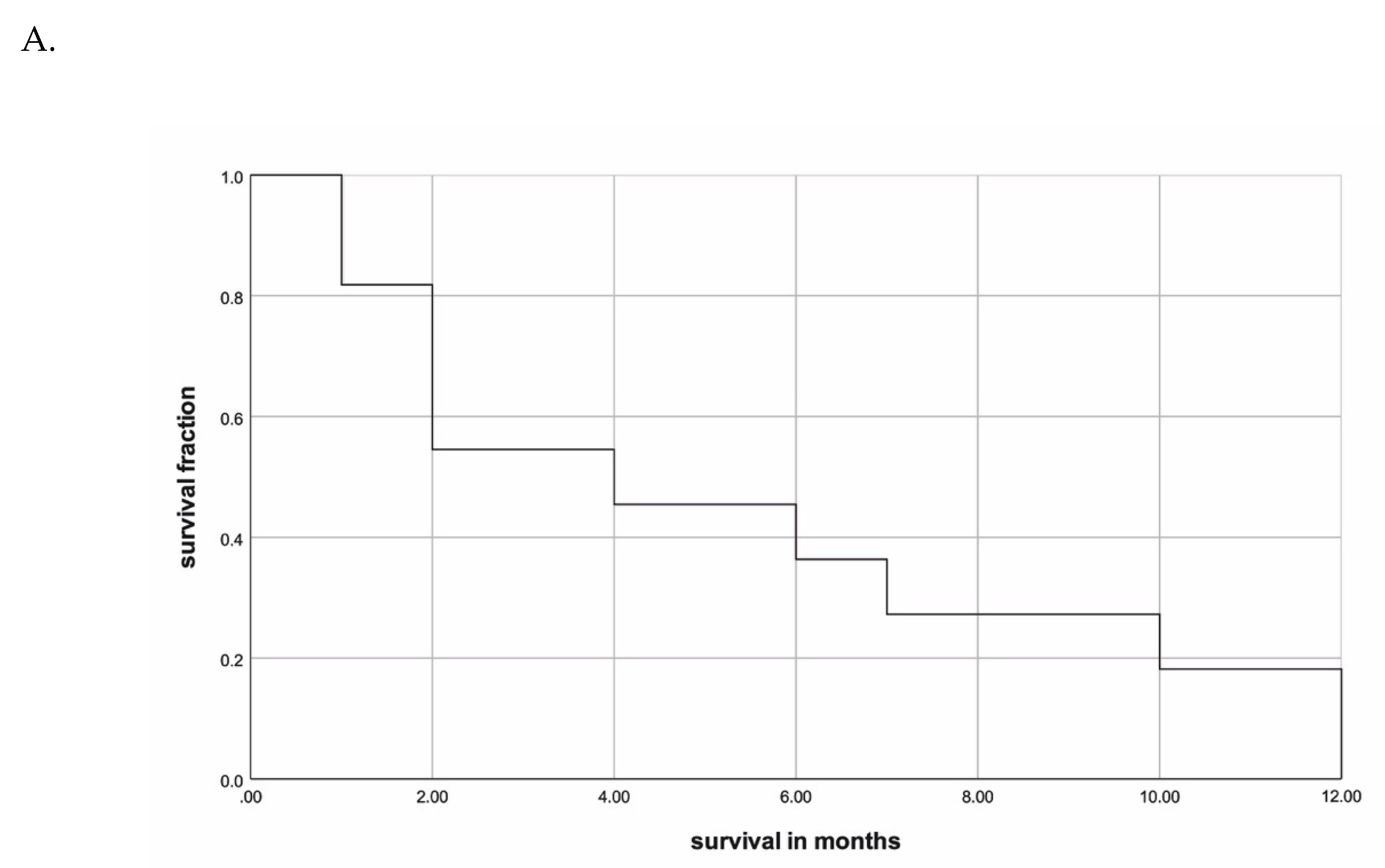
2.1. Single Center Evaluation
2.1.1. Treatment
2.1.2. Outcome
2.1.3. Treatment-Related Toxicity
2.2. Systematic Review
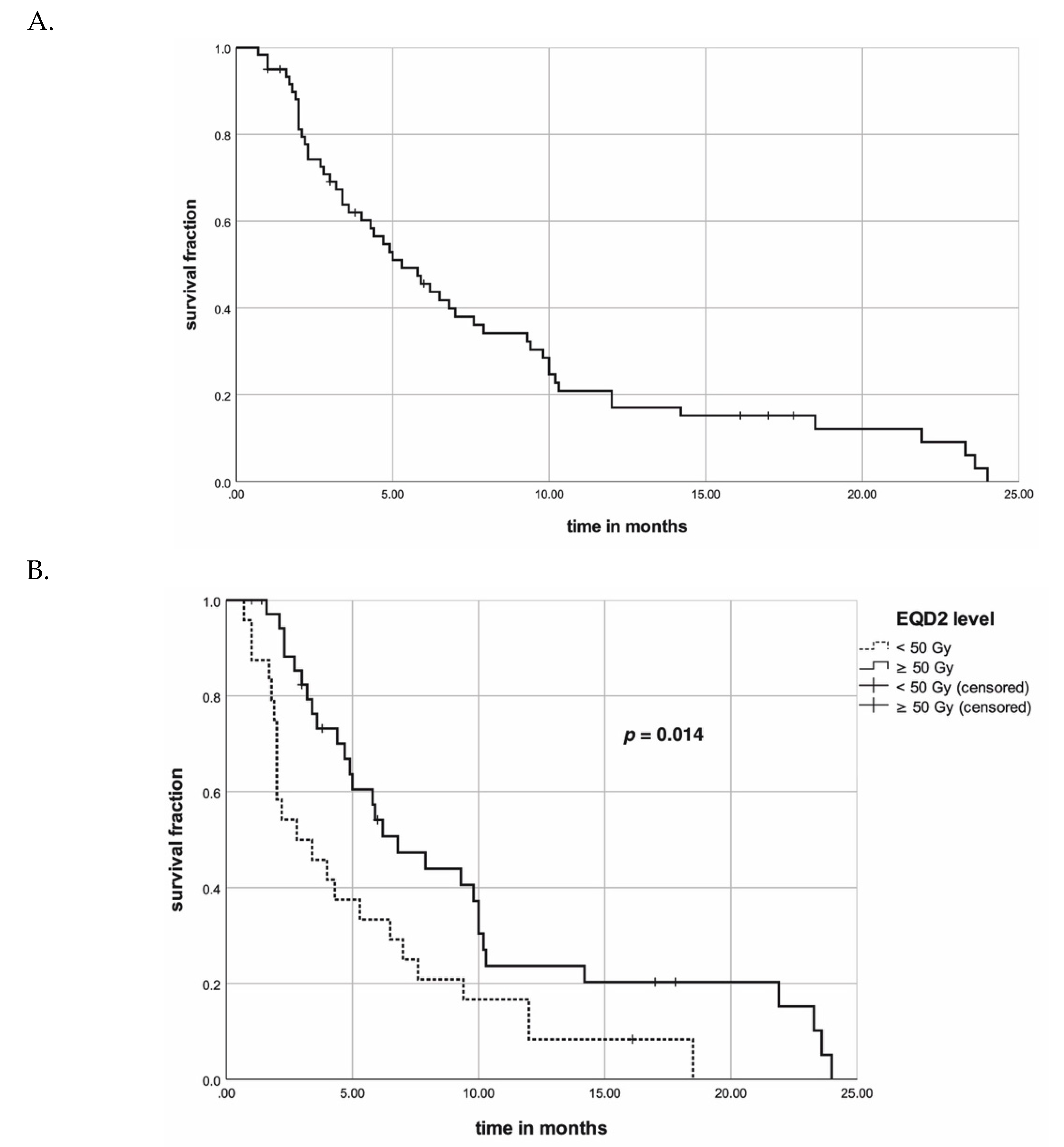
2.3. Pooled Data Evaluation
2.4. Propensity Score Matching (PSM)
3. Discussion
4. Patients and Methods
4.1. Single Center Evaluation
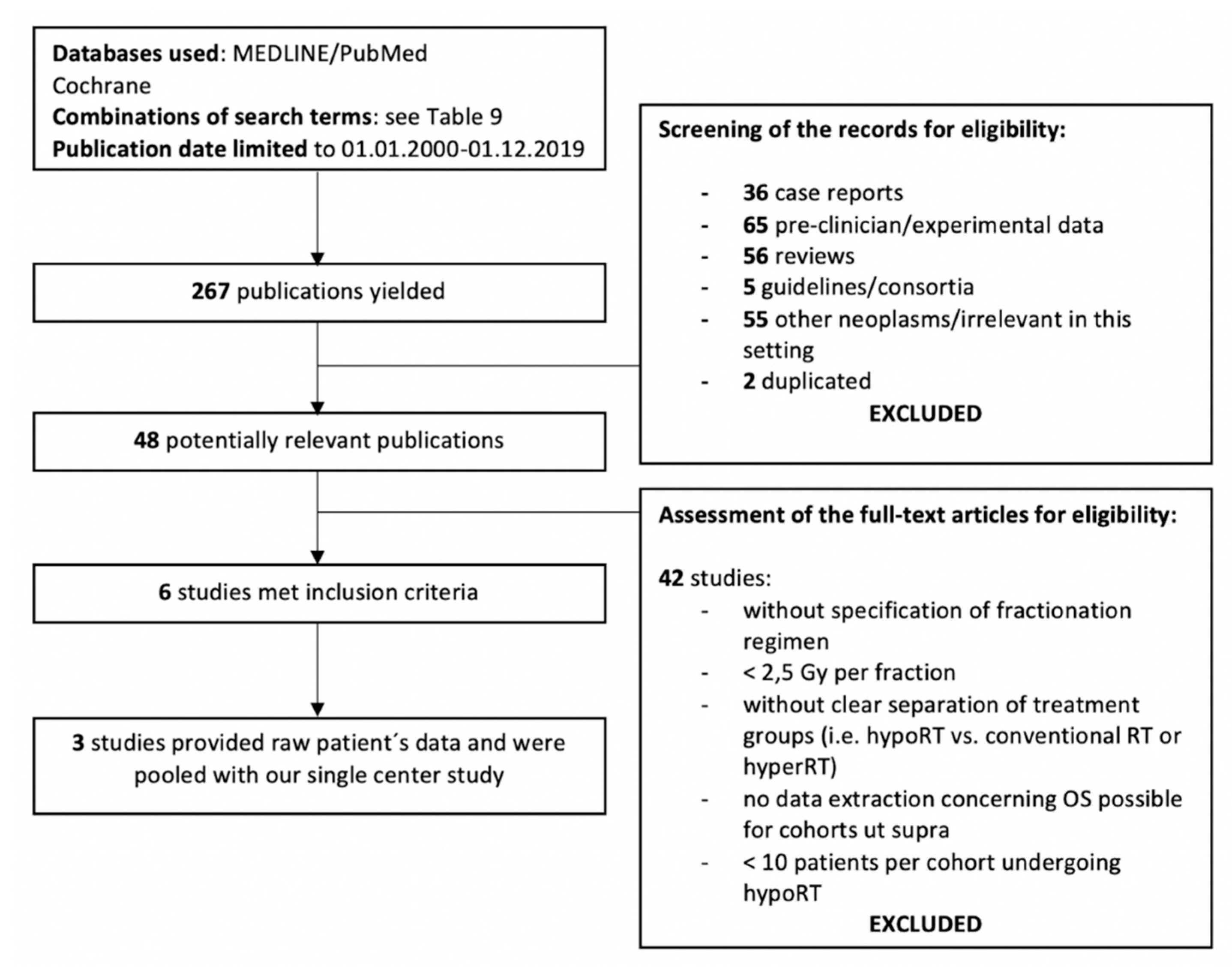
4.2. Systematic Review of Literature
4.3. Pooled Analysis and Data Management
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Dal Maso, L.; Tavilla, A.; Pacini, F.; Serraino, D.; van Dijk, B.A.C.; Chirlaque, M.D.; Capocaccia, R.; Larrañaga, N.; Colonna, M.; Agius, D.; et al. Survival of 86,690 patients with thyroid cancer: A population-based study in 29 European countries from EUROCARE-5. Eur. J. Cancer 2017, 77, 140–152. [Google Scholar] [CrossRef] [PubMed]
- Neff, R.L.; Farrar, W.B.; Kloos, R.T.; Burman, K.D. Anaplastic Thyroid Cancer. Endocrinol. Metab. Clin. N. Am. 2008, 37, 525–538. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Fuchs, T.L.; Dogan, S.; Landa, I.; Katabi, N.; Fagin, J.A.; Tuttle, R.M.; Sherman, E.J.; Gill, A.J.; Ghossein, R. Dissecting Anaplastic Thyroid Carcinoma (ATC): A Comprehensive Clinical, histologic, Immunophenotypic, and Molecular Study of 360 Cases. Thyroid 2020. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.-K.; Song, Y.S.; Lee, E.K.; Hwang, J.; Kim, H.H.; Jung, G.; Kim, Y.A.; Kim, S.; Cho, S.W.; Won, J.-K.; et al. Integrative analysis of genomic and transcriptomic characteristics associated with progression of aggressive thyroid cancer. Nat. Commun. 2019, 10, 2764. [Google Scholar] [CrossRef] [PubMed]
- Haddad, R.I.; Nasr, C.; Bischoff, L.; Busaidy, N.L.; Byrd, D.; Callender, G.; Dickson, P.; Duh, Q.-Y.; Ehya, H.; Goldner, W.; et al. NCCN Guidelines Insights: Thyroid Carcinoma, Version 2.2018. J. Natl. Compr. Cancer Netw. 2018, 16, 1429–1440. [Google Scholar] [CrossRef]
- Filetti, S.; Durante, C.; Hartl, D.; Leboulleux, S.; Locati, L.D.; Newbold, K.; Papotti, M.G.; Berruti, A. Thyroid cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2019, 30, 1856–1883. [Google Scholar] [CrossRef]
- Smallridge, R.C.; Ain, K.B.; Asa, S.L.; Bible, K.C.; Brierley, J.D.; Burman, K.D.; Kebebew, E.; Lee, N.Y.; Nikiforov, Y.E.; Rosenthal, M.S.; et al. American Thyroid Association Guidelines for Management of Patients with Anaplastic Thyroid Cancer. Thyroid 2012, 22, 1104–1139. [Google Scholar] [CrossRef]
- Vulpe, H.; Kwan, J.Y.Y.; McNiven, A.; Brierley, J.D.; Tsang, R.; Chan, B.; Goldstein, D.P.; Le, L.W.; Hope, A.; Giuliani, M. Patterns of failure in anaplastic and differentiated thyroid carcinoma treated with intensity-modulated radiotherapy. Curr. Oncol. 2017, 24, 226. [Google Scholar] [CrossRef]
- Caudell, J.J.; Ward, M.C.; Riaz, N.; Zakem, S.J.; Awan, M.J.; Dunlap, N.E.; Isrow, D.; Hassanzadeh, C.; Vargo, J.A.; Heron, D.E.; et al. Volume, Dose, and Fractionation Considerations for IMRT-based Reirradiation in Head and Neck Cancer: A Multi-institution Analysis. Int. J. Radiat. Oncol. 2018, 100, 606–617. [Google Scholar] [CrossRef]
- Dandekar, P.; Harmer, C.; Barbachano, Y.; Rhys-Evans, P.; Harrington, K.; Nutting, C.; Newbold, K. Hyperfractionated Accelerated Radiotherapy (HART) for Anaplastic Thyroid Carcinoma: Toxicity and Survival Analysis. Int. J. Radiat. Oncol. 2009, 74, 518–521. [Google Scholar] [CrossRef]
- Oweida, A.; Phan, A.; Vancourt, B.; Robin, T.; Hararah, M.K.; Bhatia, S.; Milner, D.; Lennon, S.; Pike, L.; Raben, D.; et al. Hypofractionated Radiotherapy Is Superior to Conventional Fractionation in an Orthotopic Model of Anaplastic Thyroid Cancer. Thyroid 2018, 28, 739–747. [Google Scholar] [CrossRef] [PubMed]
- Stavas, M.J.; Shinohara, E.T.; Attia, A.; Ning, M.S.; Friedman, J.M.; Cmelak, A.J. Short Course High Dose Radiotherapy in the Treatment of Anaplastic Thyroid Carcinoma. J. Thyroid Res. 2014, 2014, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N.; Matsushita, H.; Umezawa, R.; Yamamoto, T.; Ishikawa, Y.; Katagiri, Y.; Tasaka, S.; Takeda, K.; Fukui, K.; Kadoya, N.; et al. Hypofractionated Radiotherapy for Anaplastic Thyroid Carcinoma: 15 Years of Experience in a Single Institution. Eur. Thyroid J. 2019, 8, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Nachalon, Y.; Stern-Shavit, S.; Bachar, G.; Shvero, J.; Limon, D.; Popovtzer, A. Aggressive Palliation and Survival in Anaplastic Thyroid Carcinoma. JAMA Otolaryngol. Neck Surg. 2015, 141, 1128. [Google Scholar] [CrossRef] [PubMed]
- Goutsouliak, V.; Hay, J.H. Anaplastic thyroid cancer in British Columbia 1985–1999: A population-based study. Clin. Oncol. 2005, 17, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tsang, R.; Asa, S.; Dickson, B.; Arenovich, T.; Brierley, J. Clinical outcome of anaplastic thyroid carcinoma treated with radiotherapy of once- and twice-daily fractionation regimens. Cancer 2006, 107, 1786–1792. [Google Scholar] [CrossRef] [PubMed]
- So, K.; Smith, R.E.; Davis, S.R. Radiotherapy in anaplastic thyroid carcinoma: An Australian experience. J. Med. Imaging Radiat. Oncol. 2017, 61, 279–287. [Google Scholar] [CrossRef]
- Morrison, J.A.; Pike, L.A.; Lund, G.; Zhou, Q.; Kessler, B.E.; Bauerle, K.T.; Sams, S.B.; Haugen, B.R.; Schweppe, R.E. Characterization of Thyroid Cancer Cell Lines in Murine Orthotopic and Intracardiac Metastasis Models. Horm. Cancer 2015, 6, 87–99. [Google Scholar] [CrossRef]
- Fan, D.; Ma, J.; Bell, A.C.; Groen, A.H.; Olsen, K.S.; Lok, B.H.; Leeman, J.E.; Anderson, E.; Riaz, N.; McBride, S.; et al. Outcomes of multimodal therapy in a large series of patients with anaplastic thyroid cancer. Cancer 2020, 126, 444–452. [Google Scholar] [CrossRef]
- Glaser, S.M.; Mandish, S.F.; Gill, B.S.; Balasubramani, G.K.; Clump, D.A.; Beriwal, S. Anaplastic thyroid cancer: Prognostic factors, patterns of care, and overall survival: Anaplastic Thyroid Cancer. Head Neck 2016, 38, E2083–E2090. [Google Scholar] [CrossRef]
- Kim, T.Y.; Kim, K.W.; Jung, T.S.; Kim, J.M.; Kim, S.W.; Chung, K.-W.; Kim, E.Y.; Gong, G.; Oh, Y.L.; Cho, S.Y.; et al. Prognostic factors for Korean patients with anaplastic thyroid carcinoma. Head Neck 2007, 29, 765–772. [Google Scholar] [CrossRef]
- Lee, D.Y.; Won, J.-K.; Lee, S.-H.; Park, D.J.; Jung, K.C.; Sung, M.-W.; Wu, H.-G.; Kim, K.H.; Park, Y.J.; Hah, J.H. Changes of Clinicopathologic Characteristics and Survival Outcomes of Anaplastic and Poorly Differentiated Thyroid Carcinoma. Thyroid 2016, 26, 404–413. [Google Scholar] [CrossRef] [PubMed]
- Haigh, P.I.; Ituarte, P.H.G.; Wu, H.S.; Treseler, P.A.; Posner, M.D.; Quivey, J.M.; Duh, Q.Y.; Clark, O.H. Completely resected anaplastic thyroid carcinoma combined with adjuvant chemotherapy and irradiation is associated with prolonged survival. Cancer 2001, 91, 2335–2342. [Google Scholar] [CrossRef]
- Lee, D.Y.; Won, J.-K.; Choi, H.S.; Park, D.J.; Jung, K.C.; Sung, M.-W.; Kim, K.H.; Hah, J.H.; Park, Y.J. Recurrence and Survival After Gross Total Removal of Resectable Undifferentiated or Poorly Differentiated Thyroid Carcinoma. Thyroid 2016, 26, 1259–1268. [Google Scholar] [CrossRef] [PubMed]
- Are, C.; Shaha, A.R. Anaplastic Thyroid Carcinoma: Biology, Pathogenesis, Prognostic Factors, and Treatment Approaches. Ann. Surg. Oncol. 2006, 13, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Wendler, J.; Kroiss, M.; Gast, K.; Kreissl, M.C.; Allelein, S.; Lichtenauer, U.; Blaser, R.; Spitzweg, C.; Fassnacht, M.; Schott, M.; et al. Clinical presentation, treatment and outcome of anaplastic thyroid carcinoma: Results of a multicenter study in Germany. Eur. J. Endocrinol. 2016, 175, 521–529. [Google Scholar] [CrossRef]
- Pezzi, T.A.; Mohamed, A.S.R.; Sheu, T.; Blanchard, P.; Sandulache, V.C.; Lai, S.Y.; Cabanillas, M.E.; Williams, M.D.; Pezzi, C.M.; Lu, C.; et al. Radiation therapy dose is associated with improved survival for unresected anaplastic thyroid carcinoma: Outcomes from the National Cancer Data Base: Unresected Anaplastic Thyroid Carcinoma. Cancer 2017, 123, 1653–1661. [Google Scholar] [CrossRef]
- Sugitani, I.; Miyauchi, A.; Sugino, K.; Okamoto, T.; Yoshida, A.; Suzuki, S. Prognostic Factors and Treatment Outcomes for Anaplastic Thyroid Carcinoma: ATC Research Consortium of Japan Cohort Study of 677 Patients. World J. Surg. 2012, 36, 1247–1254. [Google Scholar] [CrossRef]
- Veness, M.J.; Porter, G.S.; Morgan, G.J. Anaplastic thyroid carcinoma: Dismal outcome despite current treatment approach. ANZ J. Surg. 2004, 74, 559–562. [Google Scholar] [CrossRef]
- De Ruysscher, D.; Niedermann, G.; Burnet, N.G.; Siva, S.; Lee, A.W.M.; Hegi-Johnson, F. Radiotherapy toxicity. Nat. Rev. Dis. Primer 2019, 5, 13. [Google Scholar] [CrossRef]
- Teckie, S.; Lok, B.H.; Rao, S.; Gutiontov, S.I.; Yamada, Y.; Berry, S.L.; Zelefsky, M.J.; Lee, N.Y. High-dose hypofractionated radiotherapy is effective and safe for tumors in the head-and-neck. Oral Oncol. 2016, 60, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Moulder, J.E.; Seymour, C. Radiation fractionation: The search for isoeffect relationships and mechanisms. Int. J. Radiat. Biol. 2018, 94, 743–751. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Choi, S.H.; Yoon, H.I.; Lee, J.; Kim, T.H.; Kim, J.W.; Lee, I.J. Treatment outcomes of radiotherapy for anaplastic thyroid cancer. Radiat. Oncol. J. 2018, 36, 103–113. [Google Scholar] [CrossRef] [PubMed]
| Parameter | n |
|---|---|
| Age, years | |
| <73 | 5 (46%) |
| ≥73 | 6 (55%) |
| Gender | |
| Male | 4 (36%) |
| Female | 7 (64%) |
| ECOG-PS | |
| 0 | 2 (18%) |
| 1 | 7 (64%) |
| 2 | 2 (18%) |
| T stage | |
| 3 | 1 (9%) |
| 4 | 10 (91%) |
| N stage | |
| 0 | 1 (9%) |
| 0 | 10 (91%) |
| M stage | |
| 0 | 3 (27%) |
| 1 | 8 (73%) |
| UICC stage | |
| IVB | 3 (27%) |
| IVC | 8 (73%) |
| Surgery | |
| No | 8 (73%) |
| Yes | 3 (27%) |
| Concurrent chemotherapy | |
| No | 4 (36%) |
| Yes | 7 (64%) |
| Treatment | |
| RT/CRT | 8 (73%) |
| S+CRT | 3 (27%) |
| EQD2 level | |
| <45 Gy | 5 (46%) |
| ≥45 Gy | 6 (55%) |
| RT technique | |
| 3D-CRT | 8 (73%) |
| IMRT | 3 (27%) |
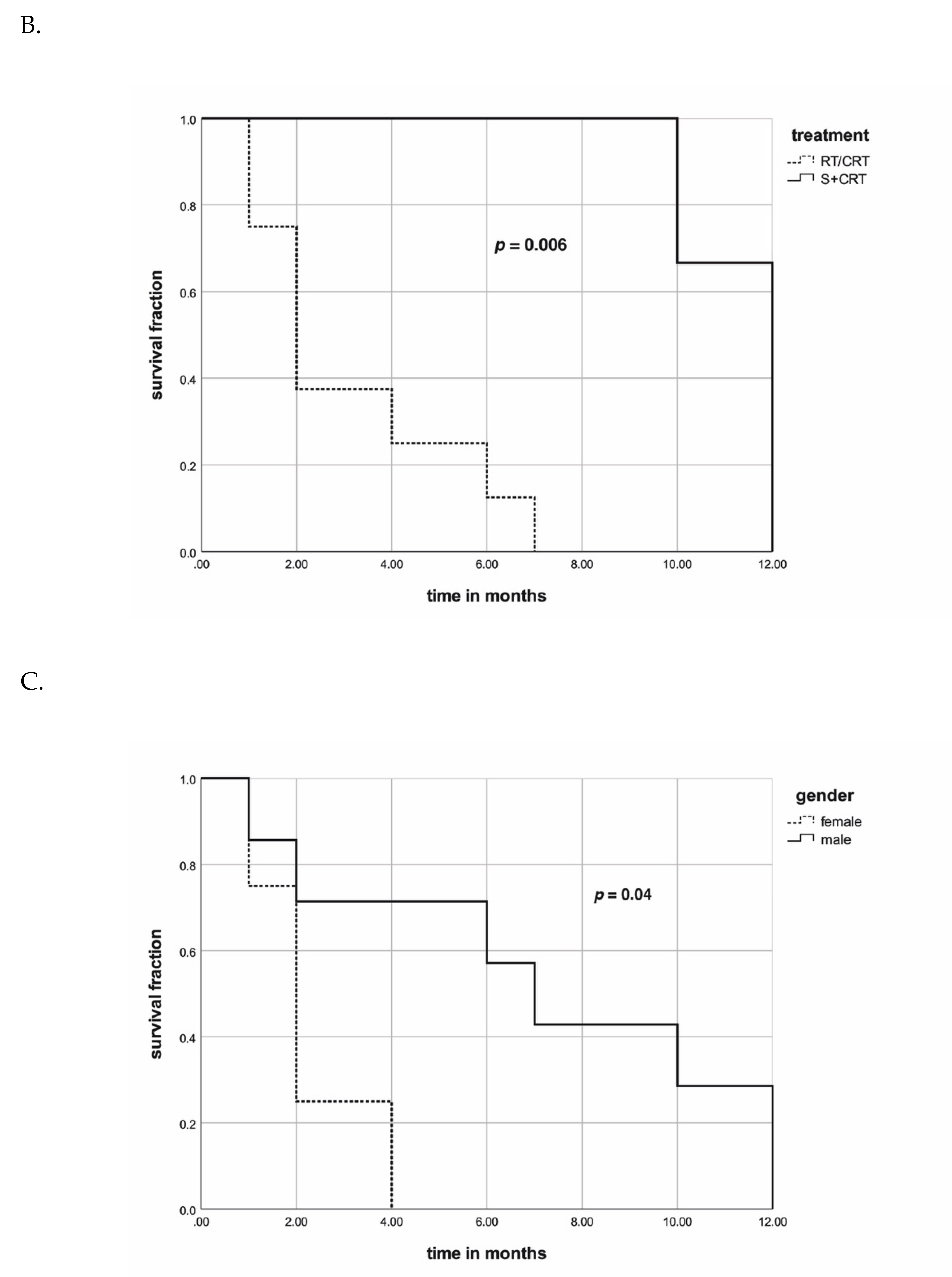
| Parameter | At 3 | At 6 | At 9 | p-Value | p-Value |
|---|---|---|---|---|---|
| Months | Months | Months | (Univariate Analysis) | (Multivariate Analysis) | |
| Age, years | |||||
| <73 | 60% | 40% | 40% | 0.106 | |
| ≥73 | 50% | 17% | 0% | ||
| Gender | |||||
| Male | 71% | 57% | 43% | 0.04 | 0.349 |
| Female | 25% | 0% | 0% | ||
| ECOG-PS | |||||
| 0 | 100% | 50% | 50% | ||
| 1 | 57% | 43% | 29% | 0.326 | |
| 2 | 0% | 0% | 0% | ||
| M stage | |||||
| 0 | 100% | 67% | 67% | 0.179 | |
| 1 | 38% | 25% | 13% | ||
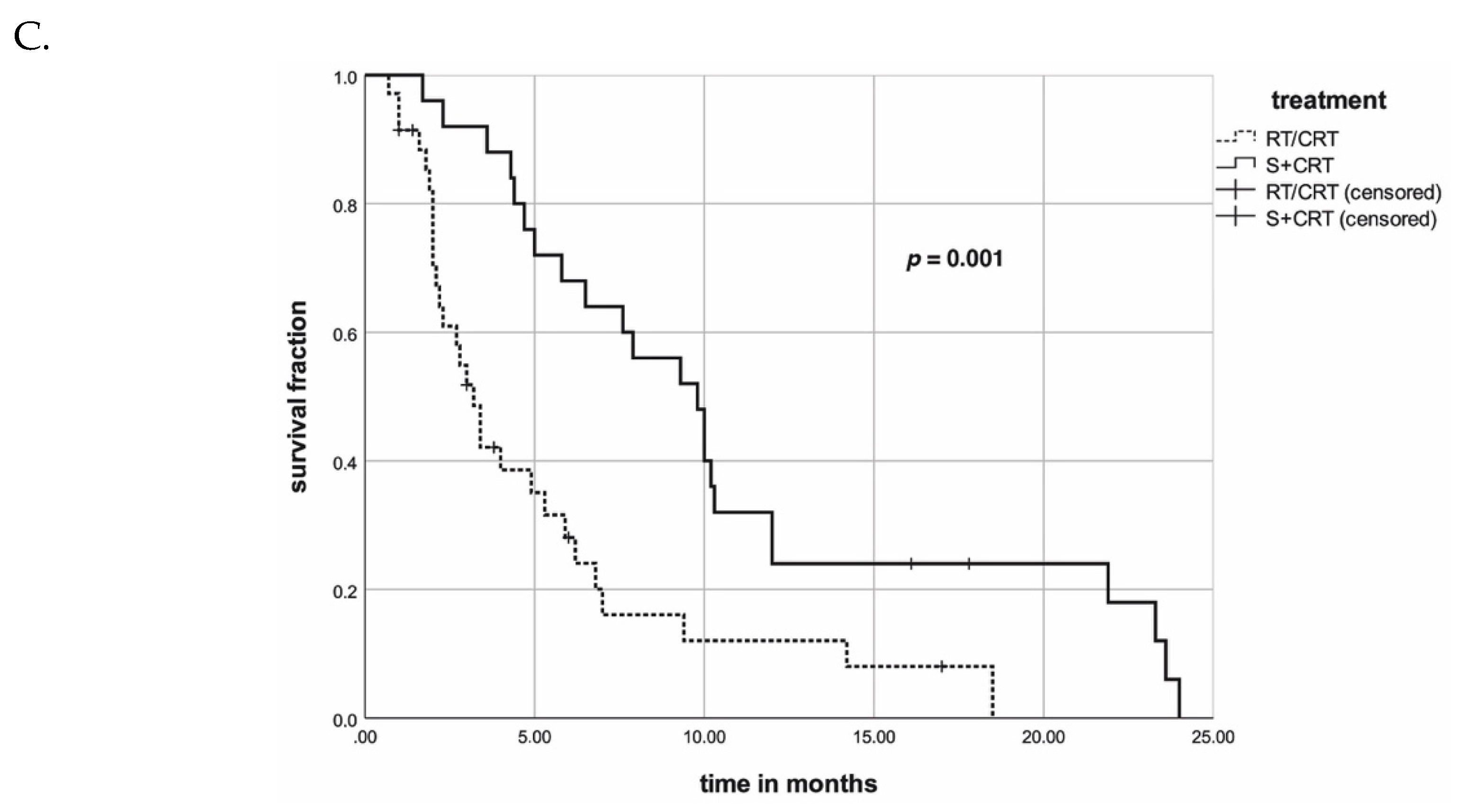
| Treatment | |||||
| RT/CRT | 38% | 13% | 0% | 0.006 | 0.941 |
| S+CRT | 100% | 100% | 100% | ||
| Concurrent chemotherapy | |||||
| No | 57 | 29 | 14 | 0.327 | |
| Yes | 50 | 50 | 50 | ||
| EQD2 level | |||||
| < 45 Gy | 50% | 33% | 17% | 0.241 | |
| ≥ 45 Gy | 60% | 40% | 40% | ||
| RT technique | |||||
| 3D-CRT | 50% | 38% | 38% | 0.701 | |
| IMRT | 67% | 33% | 0% |
| Author | Number of Patients (n) | Median Age (Years) | Presence of Metastases | Stage Distribution IVA/IVB/IVC (%) | hypoRT in (n) Patients | Surgery | ChT | Detailed RT Information |
|---|---|---|---|---|---|---|---|---|
| Goutsouliak et al. (2004) [16] | 62 referred cases: 57: received radiotherapy, 33 in palliative intention | 84 (n = 62) | 16% (n = 62) | NR/NR/16 (n = 62) | 49 | 21.0% (n = 62) | 14.5% (n = 62) | (1) n = 33: 30 Gy/3 Gy (2) n = 8: 50 Gy/2.5 Gy (3) n = 5: 40 Gy/2.67 Gy (4) n = 2: 45 Gy/2.5 Gy (5) n = 1: 45 Gy/3 Gy |
| Wang et al. (2006) [17] | 47: 24 in palliative intention | 70.5 (46.1–89.7) (n = 24) | 25% (IVC) | 8/67/25 (n = 24) | 24 | 37.5% | 16.7% | Median 20 Gy/4Gy (5–40Gy/4Gy) |
| Stavas et al. (2014) [12] | 17 | 70 (59–84) | 47% (n = 17) | 41/12/47 (n = 17) AJCC 7th edition | 17 | 82.4% | 88.2% | Median 54Gy/3Gy (40–62.5Gy/2.5–4Gy) |
| Nachalon et al. (2015) [14] | 26 patients: 12 treated in palliative intention | NR | 52% (n = 23) | NR/NR/52% (n = 12) | 23 | 21.7% | 47.8% | (1) resectable & ECOG<2: S+RCT 60–70Gy (n = 5) (2) non-resect. & ECOG<2: RCT 70Gy (n = 6) (3) M+ & ECOG<2: 50Gy (n = 9) (4) M+ & ECOG>2: 30Gy (n = 3) (5) no treatment (n = 3) |
| So et al. (2017) [15] | 30: 18 treated in palliative intention | 78 (63–92, n = 14) | 50% (n = 14) | 21/29/50 (n = 14) | 14 | 28.6% | 14.3% | Median 45 Gy/3 Gy (18–45Gy/2.5–6Gy) |
| Takahashi et al. (2018) [13] | 33 | 71 (49–87) (n = 25) | 40% (IVC) | 19/32/40 (other 1.2%) | 25 | 28.0% | 48.0% | Median 50Gy/5Gy (5–60Gy/3–6Gy) |
| Author | Median FU (95% CI) in Months | Survival after 3 Months | Survival after 6 Months | Survival after 12 Months | Median Survival (95% CI) in Months |
|---|---|---|---|---|---|
| Goutsouliak et al. (2004) [15] | 3 (0.6–20) | NR | NR | NR | 3 (0.6–20) NR for other hypofractionated treatment modalities |
| Wang et al. (2006) [16] | 4.7 (0.2–114) | 54.2% | 16.7% | 0% | 3.2 (0.2–NR, <9) |
| Stavas et al. (2014) [12] | 9.3 (4.6–14) | 94.1% | 70.6% | 41.2% | 9.3 (4.6–14) |
| Nachalon et al. (2015) [14] | 6 (2.1–9.8) | NR | NR | NR | 6 (2.1–9.8) |
| So et al. (2017) [17] | 3.4 (1.9–4.9) | 57.1% | 21.4% | 7.1% | 3.4 (1.9–4.9) |
| Takahashi et al. (2018) [13] | 3 (2.5–3.5) | 42.9% | 23.8% | 4.8% | 3 (2.5–3.5) |
| Author | CTCAE/RTOG ≥ Grade 3 Events | Local Recurrence | |||||
|---|---|---|---|---|---|---|---|
| Dysphagia | Dyspnea | Dysphonia | Mucositis | Dermatitis | Other Symptoms or Supportive Interventions | ||
| Goutsouliak et al. (2004) [15] | NR | NR | NR | NR | NR | NR | NR |
| Wang et al. (2006) [16] | NR | NR | NR | 0 | 0 | 4.2% esophagitis | 20.1% |
| Stavas et al. (2014) [12] | 24% | NR | NR | NR | 24% | 18% esophagitis; 23.5% PEG post-RT | 18% |
| Nachalon et al. (2015) [14] | 0 | 0 | 0 | 0 | 0 | 23%: PEG 35%: tracheostomy | NR |
| So et al. (2017) [17] | 0 | 0 | 0 | 0 | 0 | 0 | 29% |
| Takahashi et al. (2018) [13] | 26% | NR | NR | 5% | 5% | 10% tracheal necrosis & injury to carotid artery | 28% 5% died from LR |
| Parameter | n |
|---|---|
| Age, years | |
| <73 | 30 (50%) |
| ≥73 | 30 (50%) |
| Gender | |
| Male | 31 (52%) |
| Female | 29 (48%) |
| ECOG-PS | |
| 0–1 | 35 (58%) |
| 2–4 | 25 (42%) |
| UICC stage | |
| IVA | 6 (10%) |
| IVB | 22 (37%) |
| IVC | 30 (50%) |
| unknown | 2 (3%) |
| EQD2 level | |
| <50 Gy | 24 (40%) |
| ≥50 Gy | 36 (60%) |
| Single dose | |
| 2.5–3.5 Gy | 40 (67%) |
| 4–5 Gy | 20 (33%) |
| Concurrent chemotherapy | |
| No | 23 (38%) |
| Yes | 37 (62%) |
| Treatment | |
| RT | 24 (40%) |
| CRT | 11 (18%) |
| S + CRT | 25 (42%) |
| Parameter | At Three Months | At Six Months | At 12 Months | p-Value (Univariate Analysis) | p-Value (Multivariate Analysis) |
|---|---|---|---|---|---|
| Age, years | |||||
| <73 | 77% | 58% | 19% | 0.068 | |
| ≥73 | 62% | 35% | 15% | ||
| Gender | |||||
| Male | 76% | 50% | 8% | 0.743 | |
| Female | 62% | 41% | 24% | ||
| ECOG-PS | |||||
| 0–1 | 77% | 49% | 15% | 0.95 | |
| 2–4 | 59% | 41% | 30% | ||
| UICC stage | |||||
| IVA/B | 74% | 51% | 25% | 0.119 | |
| IVC | 67% | 42% | 11% | ||
| EQD2 level | |||||
| <50 Gy | 50% | 33% | 8% | 0.014 | 0.065 |
| 50 Gy | 82% | 54% | 24% | ||
| Single dose | |||||
| 2.5–3.5 Gy | 73% | 51% | 23% | 0.077 | |
| 4–5 Gy | 62% | 34% | 6% | ||
| Concurrent chemotherapy | |||||
| No | 73% | 44% | 11% | 0.286 | |
| Yes | 67% | 47% | 20% | ||
| Treatment | |||||
| RT/CRT | 52% | 24% | 12% | <0.001 | 0.003 |
| S+CRT | 92% | 68% | 32% |
| Parameter | Entire PSM-Cohort N (%) | Normofractionated Subgroup N (%) | Hypofractionated Subgroup N (%) | p-Value |
|---|---|---|---|---|
| Total | 54 (100) | 18 (33) | 36 (67) | |
| Age, years (range) | 70 (54–86) | 68 (55–83) | 71 (54–86) | 0.235 |
| Gender | ||||
| Male | 27 (50) | 9 (50) | 18 (50) | 0.999 |
| Female | 27 (50) | 9 (50) | 18 (50) | |
| ECOG | ||||
| 0–1 | 48 (89) | 16 (89) | 42 (89) | 0.999 |
| 2–4 | 6 (11) | 2 (11) | 4 (11) | |
| UICC stage | ||||
| IVA | 4 (10) | 2 (11) | 2 (6) | |
| IVB | 23 (37) | 13 (72) | 10 (28) | 0.002 |
| IVC | 26 (50) | 3 (17) | 23 (64) | |
| EQD2 level | ||||
| <50 Gy | 14 (26) | 2 (11) | 12 (33) | 0.082 |
| ≥50 Gy | 40 (74) | 16 (89) | 24 (67) | |
| Concurrent chemotherapy | ||||
| No | 18 (33) | 6 (33) | 12(33) | 0.999 |
| Yes | 36 (67) | 12 (67) | 24 (67) | |
| Treatment | ||||
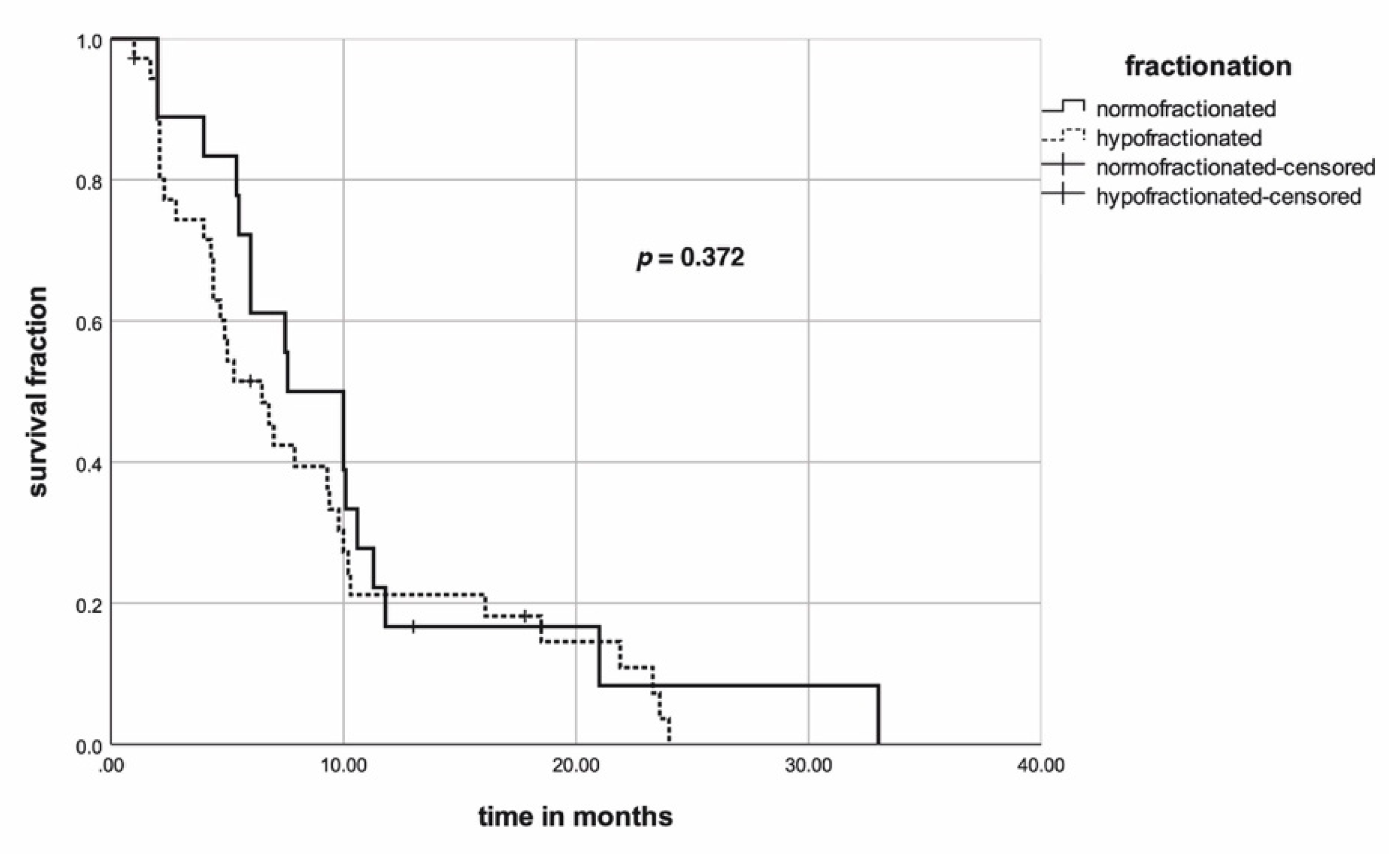
| RT/CRT | 26 (48) | 6 (33) | 20 (56) | 0.128 |
| S+CRT/RT | 28 (52) | 12 (67) | 16 (45) |
| Term | Studies Identified | |
|---|---|---|
| 1 | (Radiotherapy or radiotherap* or radio-therap* or irradiation or irradiat* or re-irradiat* or reirradiat*) | 530,114 |
| 2 | (“anaplastic thyroid cancer” or “anaplastic thyroid carcinoma” or ATC) | 4658 |
| 3 | 1 and 2 | 333 |
| 4 | “2000/01/01” [PDat]: “2019/12/01” [PDat] | 267 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliinyk, D.; Augustin, T.; Koehler, V.F.; Rauch, J.; Belka, C.; Spitzweg, C.; Käsmann, L. Hypofractionated Radiotherapy for Anaplastic Thyroid Cancer: Systematic Review and Pooled Analysis. Cancers 2020, 12, 2506. https://doi.org/10.3390/cancers12092506
Oliinyk D, Augustin T, Koehler VF, Rauch J, Belka C, Spitzweg C, Käsmann L. Hypofractionated Radiotherapy for Anaplastic Thyroid Cancer: Systematic Review and Pooled Analysis. Cancers. 2020; 12(9):2506. https://doi.org/10.3390/cancers12092506
Chicago/Turabian StyleOliinyk, Dmytro, Teresa Augustin, Viktoria Florentine Koehler, Josefine Rauch, Claus Belka, Christine Spitzweg, and Lukas Käsmann. 2020. "Hypofractionated Radiotherapy for Anaplastic Thyroid Cancer: Systematic Review and Pooled Analysis" Cancers 12, no. 9: 2506. https://doi.org/10.3390/cancers12092506
APA StyleOliinyk, D., Augustin, T., Koehler, V. F., Rauch, J., Belka, C., Spitzweg, C., & Käsmann, L. (2020). Hypofractionated Radiotherapy for Anaplastic Thyroid Cancer: Systematic Review and Pooled Analysis. Cancers, 12(9), 2506. https://doi.org/10.3390/cancers12092506






