Targeting Dopamine Receptor D2 by Imipridone Suppresses Uterine Serous Cancer Malignant Phenotype
Abstract
1. Introduction
2. Results
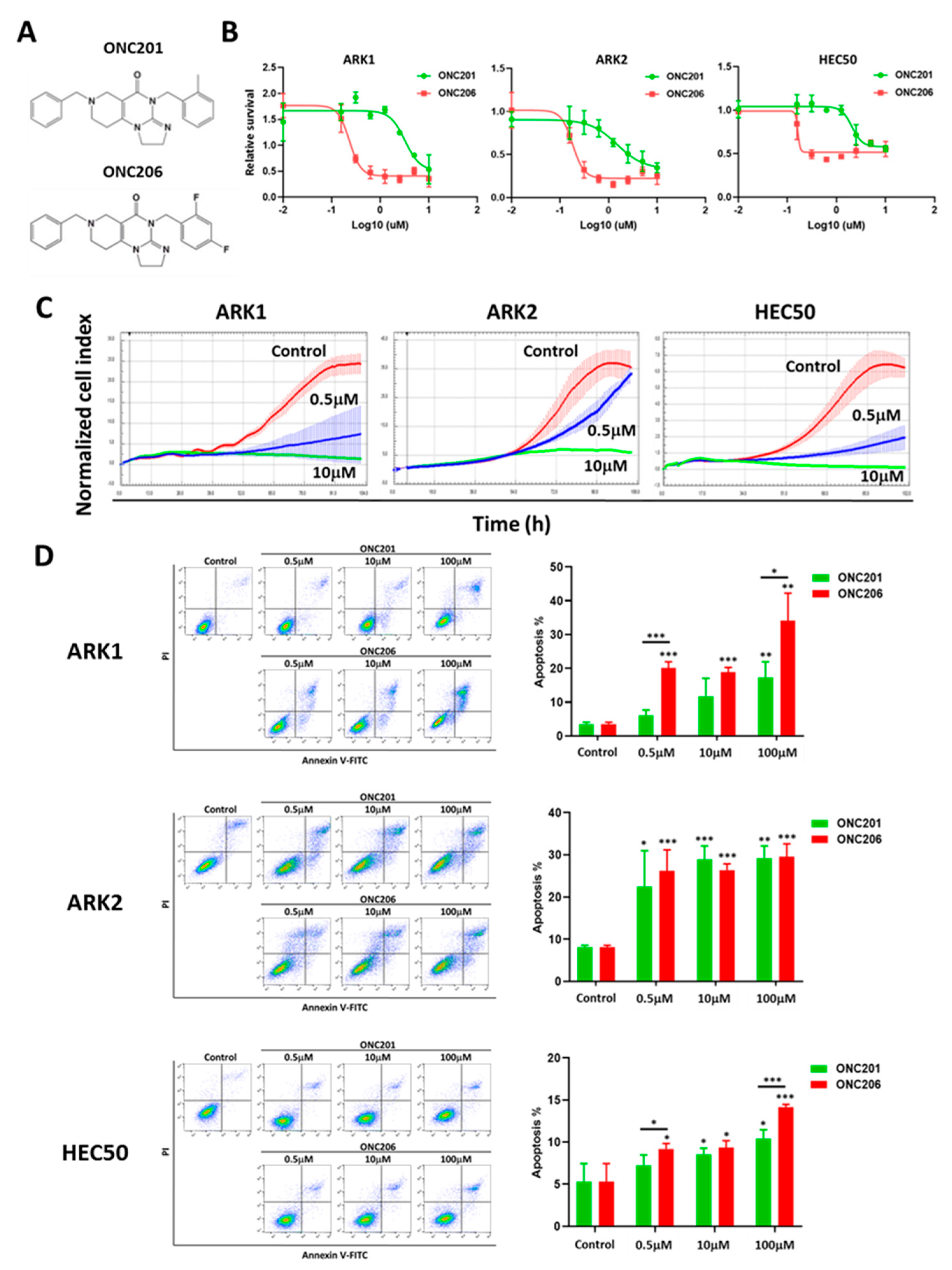
2.1. ONC206 Inhibits Cell Proliferation and Induces Apoptosis
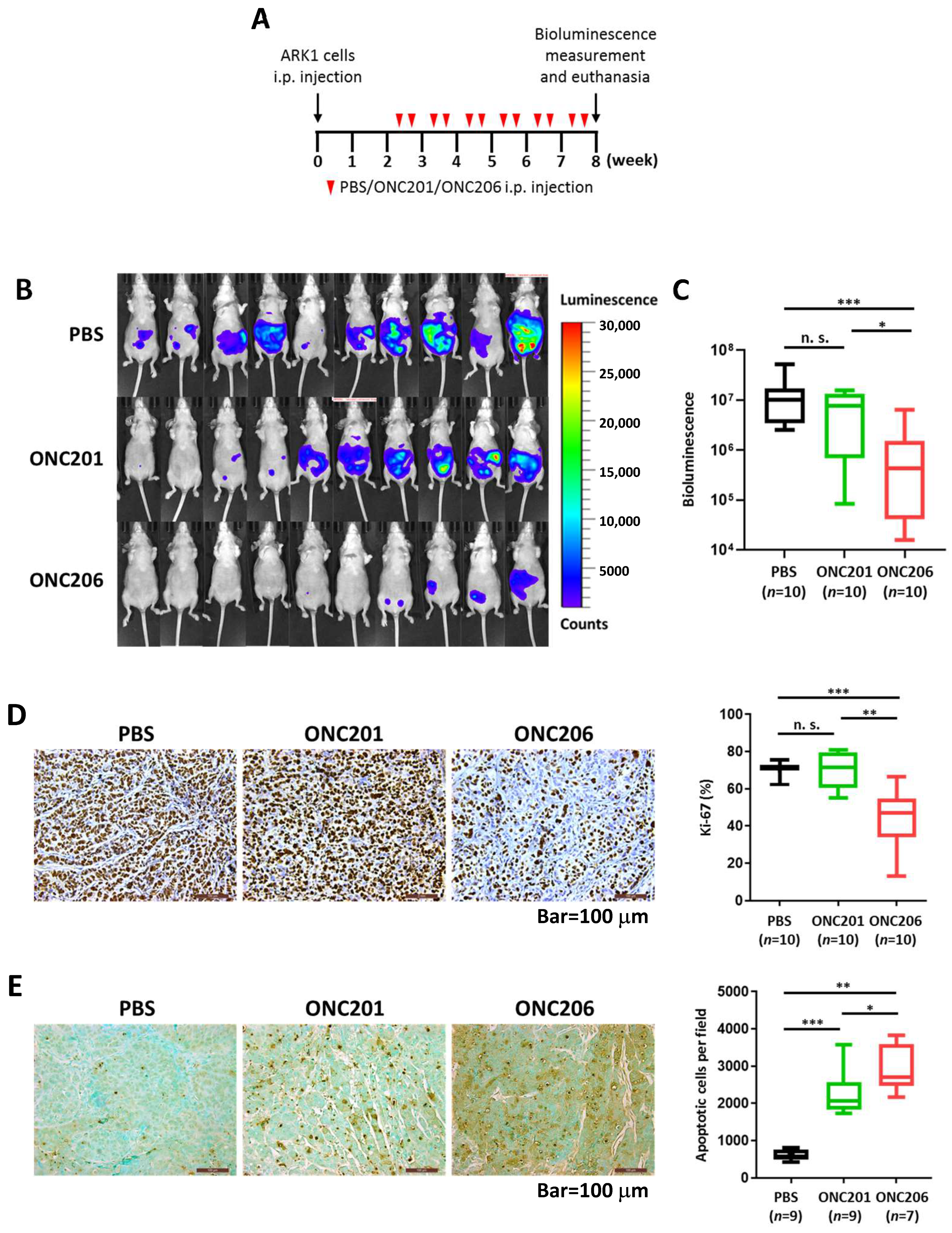
2.2. ONC206 Reduces Uterine Tumor Burden in Xenograft Mouse Model
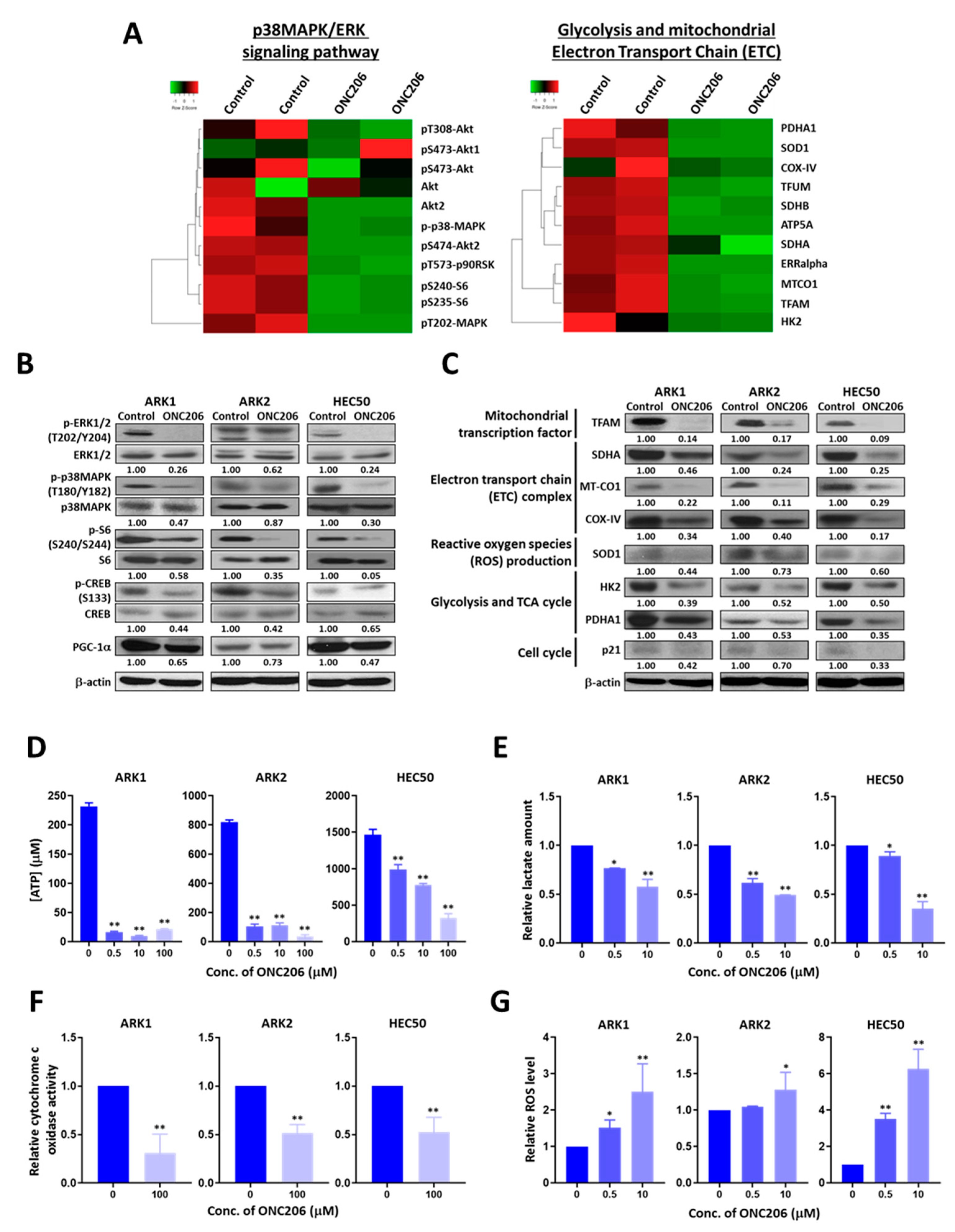
2.3. ONC206 Suppresses USC Progression through the p38MAPK/ERK Pathways and Metabolic Reprogramming
2.4. ONC206 Suppresses Glycolysis and Oxidative Phosphorylation (OXPHOS) in USC Cells
2.5. DRD2 Mediates the Tumor-Suppressive Effect of ONC206 in USC Cells
2.6. DRD2 Overexpression Is Associated with USC Progression and Patient Survival
2.7. ONC206 Synergizes with Paclitaxel for USC Cell Viability
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Cell Lines and Culture Conditions
4.3. MTT Assay
4.4. xCELLigence Real-Time Cell Analysis System
4.5. Apoptosis Assay
4.6. Tumor Xenografts
4.7. Immunohistochemistry
4.8. TUNEL Assay
4.9. Reverse Phase Protein Array (RPPA)
4.10. Western Blot Analysis
4.11. ATP Production Assay
4.12. Cytochrome c Oxidase Activity Assay
4.13. Lactate Production Assay
4.14. Reactive Oxygen Species (ROS) Detection Assay
4.15. DRD2 Knockout Using CRISPR-Cas9
4.16. Quantitative Reverse Transcription PCR Analysis
4.17. Analysis of DRD2 Expression in The Caner Genome Atlas (TCGA) Data
4.18. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, C.A.; Cheung, M.K.; Osann, K.; Chen, L.; Teng, N.N.; Longacre, T.A.; Powell, M.A.; Hendrickson, M.R.; Kapp, D.S.; Chan, J.K. Uterine papillary serous and clear cell carcinomas predict for poorer survival compared to grade 3 endometrioid corpus cancers. Br. J. Cancer 2006, 94, 642–646. [Google Scholar] [CrossRef]
- Fader, A.N.; Santin, A.D.; Gehrig, P.A. Early stage uterine serous carcinoma: Management updates and genomic advances. Gynecol. Oncol. 2013, 129, 244–250. [Google Scholar] [CrossRef]
- Berton-Rigaud, D.; Devouassoux-Shisheboran, M.; Ledermann, J.A.; Leitao, M.M.; Powell, M.A.; Poveda, A.; Beale, P.; Glasspool, R.; Creutzberg, C.L.; Harter, P.; et al. Gynecologic Cancer InterGroup (GCIG) consensus review for uterine and ovarian carcinosarcoma. Int. J. Gynecol. Cancer 2014, 24 (Suppl. 3), S55–S60. [Google Scholar] [CrossRef]
- Matei, D.; Filiaci, V.; Randall, M.E.; Mutch, D.; Steinhoff, M.M.; DiSilvestro, P.A.; Moxley, K.M.; Kim, Y.M.; Powell, M.A.; O’Malley, D.M.; et al. Adjuvant Chemotherapy plus Radiation for Locally Advanced Endometrial Cancer. N. Engl. J. Med. 2019, 380, 2317–2326. [Google Scholar] [CrossRef] [PubMed]
- Randall, M.E.; Filiaci, V.; McMeekin, D.S.; Von Gruenigen, V.; Huang, H.; Yashar, C.M.; Mannel, R.S.; Kim, J.-W.; Salani, R.; DiSilvestro, P.A.; et al. Phase III Trial: Adjuvant Pelvic Radiation Therapy Versus Vaginal Brachytherapy Plus Paclitaxel/Carboplatin in High-Intermediate and High-Risk Early Stage Endometrial Cancer. J. Clin. Oncol. 2019, 37, 1810–1818. [Google Scholar] [CrossRef]
- de Boer, S.M.; Powell, M.E.; Mileshkin, L.; Katsaros, D.; Bessette, P.; Haie-Meder, C.; Ottevanger, P.B.; Ledermann, J.A.; Khaw, P.; Colombo, A.; et al. Adjuvant chemoradiotherapy versus radiotherapy alone for women with high-risk endometrial cancer (PORTEC-3): Final results of an international, open-label, multicentre, randomised, phase 3 trial. Lancet Oncol. 2018, 19, 295–309. [Google Scholar] [CrossRef]
- Slomovitz, B.M.; Burke, T.W.; Eifel, P.J.; Ramondetta, L.M.; Silva, E.G.; Jhingran, A.; Oh, J.C.; Atkinson, E.N.; Broaddus, R.R.; Gershenson, D.M.; et al. Uterine papillary serous carcinoma (UPSC): A single institution review of 129 cases. Gynecol. Oncol. 2003, 91, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Boruta, D.M.; Gehrig, P.A.; Groben, P.A.; Bae-Jump, V.; Boggess, J.F.; Fowler, W.C.; van Le, M.D.L. Uterine serous and grade 3 endometrioid carcinomas: Is there a survival difference? Cancer 2004, 101, 2214–2221. [Google Scholar] [CrossRef]
- Mahdi, H.; Rizzo, A.; Rose, P.G. Outcome of recurrent uterine papillary serous carcinoma treated with platinum-based chemotherapy. Int. J. Gynecol. Cancer 2015, 25, 467–473. [Google Scholar] [CrossRef]
- Fader, A.N.; Drake, R.D.; O’Malley, D.M.; Gibbons, H.E.; Huh, W.K.; Havrilesky, L.J.; Gehrig, P.A.; Tuller, E.; Axtell, A.E.; Zanotti, K.M.; et al. Platinum/taxane-based chemotherapy with or without radiation therapy favorably impacts survival outcomes in stage I uterine papillary serous carcinoma. Cancer 2009, 115, 2119–2127. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.E.; Krigsfeld, G.; Mayes, P.A.; Patel, L.; Dicker, D.T.; Patel, A.S.; Dolloff, N.G.; Messaris, E.; Scata, K.A.; Wang, W.; et al. Dual inactivation of Akt and ERK by TIC10 signals Foxo3a nuclear translocation, TRAIL gene induction, and potent antitumor effects. Sci. Transl. Med. 2013, 5, 171ra17. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, J.M.; Espinoza, S.; Gainetdinov, R.R. Dopamine receptors—IUPHAR Review 13. Br. J. Pharm. 2015, 172, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, V.V.; Madhukar, N.S.; Gilvary, C.; Kline, C.L.B.; Oster, S.; El-Deiry, W.S.; Elemento, O.; Doherty, F.C.; VanEngelenburg, A.; Durrant, J.; et al. Dopamine Receptor D5 is a Modulator of Tumor Response to Dopamine Receptor D2 Antagonism. Clin. Cancer Res. 2019, 25, 2305–2313. [Google Scholar] [CrossRef]
- Allen, J.E.; Kline, C.L.B.; Prabhu, V.V.; Wagner, J.; Ishizawa, J.; Madhukar, N.; Lev, A.; Baumeister, M.; Zhou, L.; Lulla, A.; et al. Discovery and clinical introduction of first-in-class imipridone ONC201. Oncotarget 2016, 7, 74380–74392. [Google Scholar] [CrossRef]
- Bar-Shavit, R.; Maoz, M.; Kancharla, A.; Nag, J.K.; Agranovich, D.; Grisaru-Granovsky, S.; Uziely, B. G Protein-Coupled Receptors in Cancer. Int. J. Mol. Sci. 2016, 17, 1320. [Google Scholar] [CrossRef]
- Wagner, J.; Kline, C.L.; Ralff, M.D.; Lev, A.; Lulla, A.R.; Zhou, L.; Olson, G.L.; Nallaganchu, B.R.; Benes, C.H.; Allen, J.E.; et al. Preclinical evaluation of the imipridone family, analogs of clinical stage anti-cancer small molecule ONC201, reveals potent anti-cancer effects of ONC212. Cell Cycle 2017, 16, 1790–1799. [Google Scholar] [CrossRef]
- Ishida, C.T.; Zhang, Y.; Bianchetti, E.; Shu, C.; Nguyen, T.T.T.; Kleiner, G.; Sanchez-Quintero, M.J.; Quinzii, C.M.; Westhoff, M.-A.; Karpel-Massler, G.; et al. Metabolic Reprogramming by Dual AKT/ERK Inhibition through Imipridones Elicits Unique Vulnerabilities in Glioblastoma. Clin. Cancer Res. 2018, 24, 5392–5406. [Google Scholar] [CrossRef]
- Prabhu, V.V.; Kawakibi, A.R.; Madhukar, N.; Garnett, M.J.; McDermott, U.; Benes, C.H.; Anantharaman, L.; Charter, N.; Deacon, S.; Van Engelenburg, A.; et al. Abstract 3877: IND-enabling characterization of DRD2/3 imipridone antagonist ONC206 for oncology. Cancer Res. 2019, 79, 3877. [Google Scholar]
- Stein, M.N.; Malhotra, J.; Tarapore, R.S.; Malhotra, U.; Silk, A.W.; Chan, N.; Rodriguez, L.; Aisner, J.; Aiken, R.D.; Mayer, T.; et al. Safety and enhanced immunostimulatory activity of the DRD2 antagonist ONC201 in advanced solid tumor patients with weekly oral administration. J. Immunother. Cancer 2019, 7, 136. [Google Scholar] [CrossRef]
- Fang, Z.; Wang, J.; Clark, L.H.; Sun, W.; Yin, Y.; Kong, W.; Pierce, S.R.; West, L.; Sullivan, S.A.; Tran, A.-Q.; et al. ONC201 demonstrates anti-tumorigenic and anti-metastatic activity in uterine serous carcinoma in vitro. Am. J. Cancer Res. 2018, 8, 1551–1563. [Google Scholar] [PubMed]
- Stein, M.N.; Bertino, J.R.; Kaufman, H.L.; Mayer, T.; Moss, R.; Silk, A.; Chan, N.; Malhotra, J.; Rodriguez, L.; Aisner, J.; et al. First-in-Human Clinical Trial of Oral ONC201 in Patients with Refractory Solid Tumors. Clin. Cancer Res. 2017, 23, 4163–4169. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.E.; Krigsfeld, G.; Patel, L.; Mayes, P.A.; Dicker, D.T.; Wu, G.S.; EL-Deiry, W.S. Identification of TRAIL-inducing compounds highlights small molecule ONC201/TIC10 as a unique anti-cancer agent that activates the TRAIL pathway. Mol. Cancer 2015, 14, 99. [Google Scholar] [CrossRef] [PubMed]
- Klein, M.O.; Battagello, D.S.; Cardoso’, A.R.; Hauser, D.N.; Bittencourt, J.C.; Correa, R.G. Dopamine: Functions, Signaling, and Association with Neurological Diseases. Cell. Mol. Neurobiol. 2019, 39, 31–59. [Google Scholar] [CrossRef]
- Jandaghi, P.; Najafabadi, H.; Bauer, A.S.; Papadakis, A.I.; Fassan, M.; Hall, A.; Monast, A.; Doeberitz, M.V.K.; Neoptolemos, J.P.; Costello, E.; et al. Expression of DRD2 Is Increased in Human Pancreatic Ductal Adenocarcinoma and Inhibitors Slow Tumor Growth in Mice. Gastroenterology 2016, 151, 1218–1231. [Google Scholar] [CrossRef]
- Li, J.; Zhu, S.; Kozono, D.; Ng, K.; Futalan, D.; Shen, Y.; Akers, J.C.; Steed, T.; Kushwaha, D.; Schlabach, M.; et al. Genome-wide shRNA screen revealed integrated mitogenic signaling between dopamine receptor D2 (DRD2) and epidermal growth factor receptor (EGFR) in glioblastoma. Oncotarget 2014, 5, 882–893. [Google Scholar] [CrossRef]
- Bakadlag, R.; Jandaghi, P.; Hoheisel, J.D.; Riazalhosseini, Y. The potential of dopamine receptor D2 (DRD2) as a therapeutic target for tackling pancreatic cancer. Expert Opin. Targets 2019, 23, 365–367. [Google Scholar] [CrossRef]
- Ishizawa, J.; Zarabi, S.F.; Davis, R.E.; Halgas, O.; Nii, T.; Jitkova, Y.; Zhao, R.; St-Germain, J.; Heese, L.E.; Egan, G.; et al. Mitochondrial ClpP-Mediated Proteolysis Induces Selective Cancer Cell Lethality. Cancer Cell 2019, 35, 721–737.e9. [Google Scholar] [CrossRef]
- Graves, P.; Aponte-Collazo, L.J.; Fennell, E.M.J.; Graves, A.C.; Hale, A.E.; Dicheva, N.; Herring, L.E.; Gilbert, T.S.K.; East, M.P.; McDonald, I.M.; et al. Mitochondrial Protease ClpP is a Target for the Anticancer Compounds ONC201 and Related Analogues. ACS Chem. Biol. 2019, 14, 1020–1029. [Google Scholar] [CrossRef]
- Tan, Z.; Luo, X.; Xiao, L.; Tang, M.; Bode, A.M.; Dong, Z.; Cao, Y. The Role of PGC1alpha in Cancer Metabolism and its Therapeutic Implications. Mol. Cancer 2016, 15, 774–782. [Google Scholar] [CrossRef]
- Knutti, D.; Kressler, D.; Kralli, A. Regulation of the transcriptional coactivator PGC-1 via MAPK-sensitive interaction with a repressor. Proc. Natl. Acad. Sci. USA 2001, 98, 9713–9718. [Google Scholar] [CrossRef]
- Vazquez, F.; Lim, J.-H.; Chim, H.; Bhalla, K.; Girnun, G.; Pierce, K.; Clish, C.B.; Granter, S.R.; Widlund, H.R.; Spiegelman, B.M.; et al. PGC1alpha expression defines a subset of human melanoma tumors with increased mitochondrial capacity and resistance to oxidative stress. Cancer Cell 2013, 23, 287–301. [Google Scholar] [CrossRef] [PubMed]
- Fisher, R.P.; Clayton, D.A. Purification and characterization of human mitochondrial transcription factor 1. Mol. Cell Biol. 1988, 8, 3496–3509. [Google Scholar] [CrossRef] [PubMed]
- Parisi, M.A.; Clayton, D.A. Similarity of human mitochondrial transcription factor 1 to high mobility group proteins. Science 1991, 252, 965–969. [Google Scholar] [CrossRef] [PubMed]
- Ekstrand, M.I.; Falkenberg, M.; Rantanen, A.; Park, C.B.; Gaspari, M.; Hultenby, K.; Rustin, P.; Gustafsson, C.M.; Larsson, N.-G. Mitochondrial transcription factor A regulates mtDNA copy number in mammals. Hum. Mol. Genet. 2004, 13, 935–944. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.C. Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Res. 2010, 70, 440–446. [Google Scholar] [CrossRef]

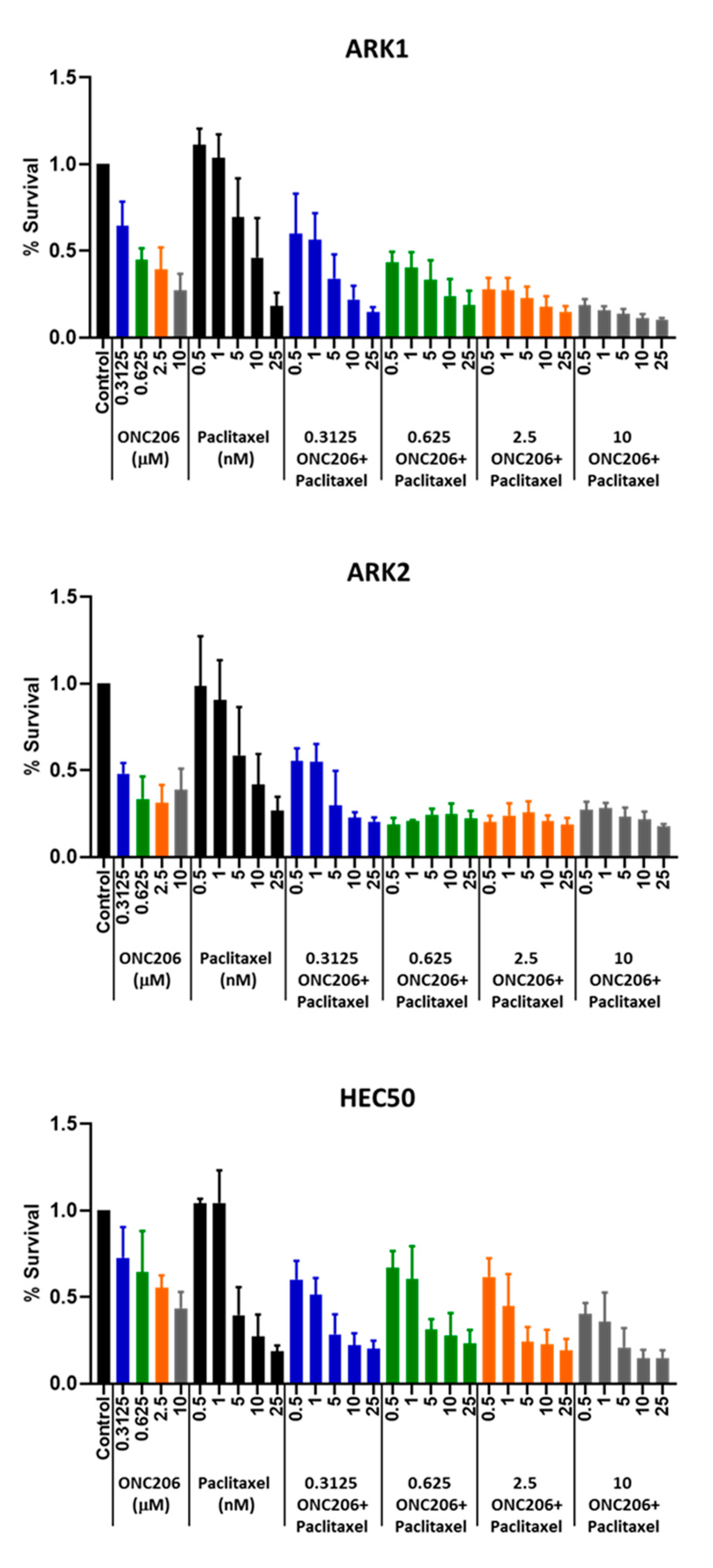
| ONC206 (μM) | Paclitaxel (nM) | Combination Index | ||
|---|---|---|---|---|
| ARK1 | ARK2 | HEC50 | ||
| 0.3125 | 0.5 | 1.180 | 0.179 | 0.613 |
| 1 | 0.889 | 0.267 | 0.527 | |
| 5 | 0.431 | 0.658 | 0.496 | |
| 10 | 0.488 | 0.480 | 0.582 | |
| 0.625 | 0.5 | 0.464 | 0.224 | 1.702 |
| 1 | 0.389 | 0.379 | 1.292 | |
| 5 | 0.484 | 0.164 | 0.619 | |
| 10 | 0.554 | 0.204 | 0.919 | |
| 2.5 | 0.5 | 0.321 | 0.441 | 2.637 |
| 1 | 0.335 | 0.301 | 0.592 | |
| 5 | 0.392 | 0.432 | 0.371 | |
| 10 | 0.463 | 0.177 | 0.627 | |
| 10 | 0.5 | 0.347 | 0.111 | 0.801 |
| 1 | 0.225 | 0.184 | 0.559 | |
| 5 | 0.281 | 0.117 | 0.304 | |
| 10 | 0.345 | 0.398 | 0.284 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, W.; Zhang, L.; Ferri-Borgogno, S.; Kwan, S.-Y.; Lewis, K.E.; Cun, H.T.; Yeung, T.-L.; Soliman, P.T.; Tarapore, R.S.; Allen, J.E.; et al. Targeting Dopamine Receptor D2 by Imipridone Suppresses Uterine Serous Cancer Malignant Phenotype. Cancers 2020, 12, 2436. https://doi.org/10.3390/cancers12092436
Hu W, Zhang L, Ferri-Borgogno S, Kwan S-Y, Lewis KE, Cun HT, Yeung T-L, Soliman PT, Tarapore RS, Allen JE, et al. Targeting Dopamine Receptor D2 by Imipridone Suppresses Uterine Serous Cancer Malignant Phenotype. Cancers. 2020; 12(9):2436. https://doi.org/10.3390/cancers12092436
Chicago/Turabian StyleHu, Wen, Li Zhang, Sammy Ferri-Borgogno, Suet-Ying Kwan, Kelsey E. Lewis, Han T. Cun, Tsz-Lun Yeung, Pamela T. Soliman, Rohinton S. Tarapore, Joshua E. Allen, and et al. 2020. "Targeting Dopamine Receptor D2 by Imipridone Suppresses Uterine Serous Cancer Malignant Phenotype" Cancers 12, no. 9: 2436. https://doi.org/10.3390/cancers12092436
APA StyleHu, W., Zhang, L., Ferri-Borgogno, S., Kwan, S.-Y., Lewis, K. E., Cun, H. T., Yeung, T.-L., Soliman, P. T., Tarapore, R. S., Allen, J. E., Guan, X., Lu, K. H., Mok, S. C., & Au-Yeung, C.-L. (2020). Targeting Dopamine Receptor D2 by Imipridone Suppresses Uterine Serous Cancer Malignant Phenotype. Cancers, 12(9), 2436. https://doi.org/10.3390/cancers12092436






