Endocrine Adverse Events of Nivolumab in Non-Small Cell Lung Cancer Patients—Literature Review
Abstract
1. Introduction
- Activating molecules (CD28, CD27, OX40, CD137 and GITR (glucocorticoid-induced tumor necrosis factor receptor)), whose stimulation increases the proliferation, differentiation and activation of lymphocytes, positively stimulating the immune response;
- Inhibitory molecules (CTLA-4 (cytotoxic T-limphocyte-associated protein 4), PD-1 (programmed cell death-1), ICOS (inducible T-cell co-stimulator) and LAG-3 (lymphocyte activation gene-3)), the stimulation of which causes the functional depletion of lymphocytes, thus limiting T lymphocyte activity and causing immunosuppression [1].
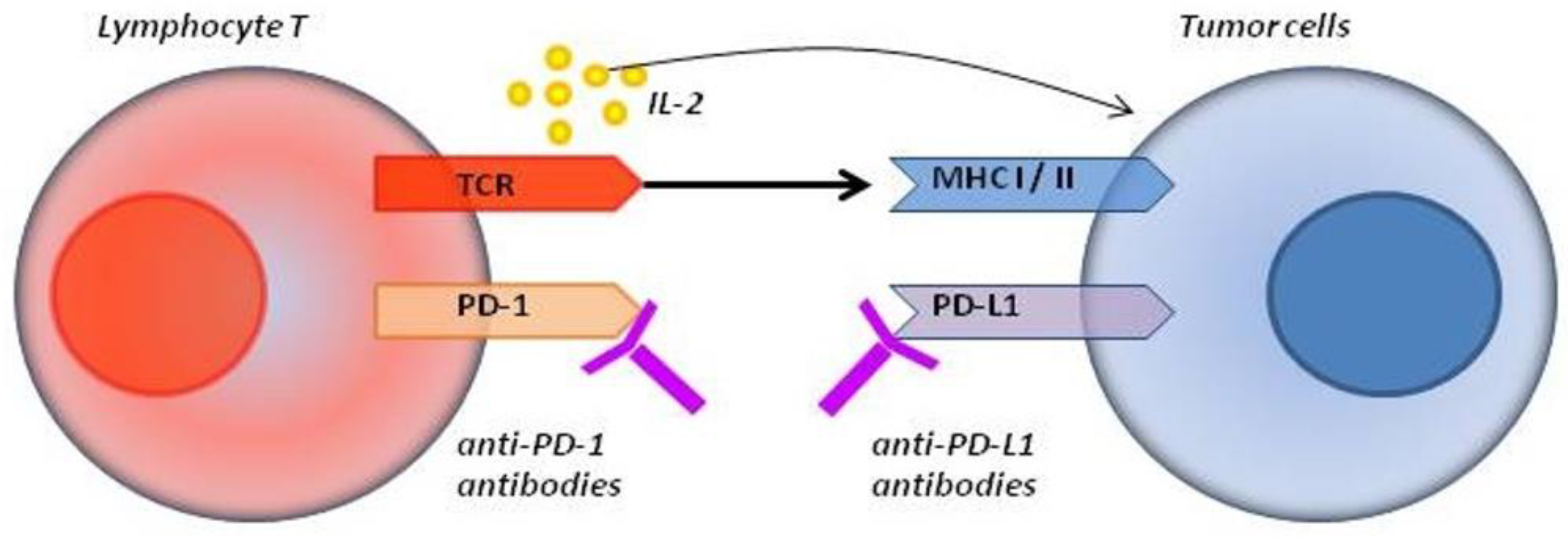
2. Inhibitory Molecules and Their Use in Oncology
3. Adverse Events Associated with Anti-PD-1 and Anti-PD-L1 Immunotherapy
4. Endocrine Adverse Effects Associated with Nivolumab Treatment
5. Pituitary Gland Disorders
5.1. Epidemiology
5.2. Mechanisms
5.3. Clinical Manifestations
5.4. Diagnostics
5.5. Treatment, Prognosis
6. Thyroid Gland Disorders
6.1. Epidemiology
6.2. Clinical Manifestations
6.3. Mechanisms
6.4. Diagnostics
6.5. Treatment, Prognosis
7. Disorders of Adrenal Glands
7.1. Epidemiology
7.2. Clinical Manifestations
7.3. Diagnostics
7.4. Treatment, Prognosis
8. Diabetes
8.1. Epidemiology
8.2. Clinical Manifestations
8.3. Mechanisms
8.4. Diagnostics
8.5. Treatment, Prognosis
9. Management of Adverse Events during Nivolumab Treatment
10. IrAE and Efficacy of Immunotherapy
11. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wojas-Krawczyk, K.; Szczyrek, M.; Krawczyk, P. Immunologia i immunoterapia niedrobnokomórkowego raka płuca. Nowotw. Płuca Klatki Piersiowej 2017, 1, 37–45. [Google Scholar]
- Ntali, G.; Kassi, E.; Alevizaki, M. Endocrine sequelae of immune checkpoint inhibitors. Hormones 2017, 16, 341–350. [Google Scholar] [PubMed]
- Zamani, M.R.; Aslani, S.; Salmaninejad, A.; Javan, M.R.; Rezaei, N. PD-1/PD-L1 and autoimmunity: A growing relationship. Cell. Immunol. 2016, 310, 27–41. [Google Scholar] [CrossRef]
- Guaraldi, F.; La Selva, R.; Samà, M.T.; D’Angelo, V.; Gori, D.; Fava, P.; Fierro, M.T.; Savoia, P.; Arvat, E. Characterization and implications of thyroid dysfunction induced by immune checkpoint inhibitors in real-life clinical practice: A long-term prospective study from a referral institution. J. Endocrinol. Investig. 2018, 41, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Riella, L.V.; Paterson, A.M.; Sharpe, A.H.; Chandraker, A. Role of the PD-1 pathway in the immune response. Am. J. Transpl. 2012, 12, 2575–2587. [Google Scholar] [CrossRef]
- Zak, K.M.; Kitel, R.; Przetocka, S. Structure of the complex of human programmed death 1, PD-1, and its Ligand PD-L1. Structure 2015, 23, 2341–2348. [Google Scholar] [CrossRef]
- Li, L.; Masood, A.; Bari, S.; Yavuz, S.; Grosbach, A.B. Autoimmune diabetes and thyroiditis complicating treatment with nivolumab. Case Rep. Oncol. 2017, 10, 230–234. [Google Scholar] [CrossRef]
- Herzberg, B.; Campo, M.J.; Gainor, J.F. Immune checkpoint inhibitors in non-small cell lung cancer. Oncologist 2017, 22, 81–88. [Google Scholar] [CrossRef]
- Gridelli, C.; Rossi, E.; De Chiara, G.; Ciardiello, F.; Sgambato, A. Thyroid-induced toxicity of check-point inhibitors immunotherapy in the treatment of advanced non-small cell lung cancer. J. Endocrinol. Diabetes 2016, 3, 1–10. [Google Scholar] [CrossRef]
- The Nobel Prize in Physiology or Medicine 2018. Available online: https://www.nobelprize.org/prizes/medicine/2018/press-release/ (accessed on 20 January 2020).
- Ishida, Y.; Agata, Y.; Shibahara, K.; Honjo, T. Induced expression of PD-1, a novel member of the immunoglobulin gene superfamily, upon programmed cell death. EMBO J. 1992, 11, 3887–3895. [Google Scholar] [CrossRef]
- Leach, D.R.; Krummel, M.F.; Allison, J.P. Enhancement of antitumor immunity by CTLA-4 blockade. Science 1996, 22, 1734–1736. [Google Scholar] [CrossRef] [PubMed]
- OPDIVO (Nivolumab). Highlights of Prescribing Information. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/125554s067lbl.pdf (accessed on 12 January 2020).
- Opdivo: EPAR- Medicine Overview. Available online: https://www.ema.europa.eu/en/documents/overview/opdivo-epar-medicine-overview_en.pdf (accessed on 20 January 2020).
- Pardol, D.M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef] [PubMed]
- Ott, P.A.; Hodi, F.S.; Robert, C. CTLA-4 and PD-1/PD-L1 blockade: New immunotherapeutic modalities with durable clinical benefit in melanoma patients. Clin. Cancer Res. 2013, 19, 5300–5309. [Google Scholar] [CrossRef] [PubMed]
- National Health Fund- Reimbursed Drugs List. Available online: NFZ/programylekowe:https://www.gov.pl/web/zdrowie/obwieszczenie-ministra-zdrowia-z-dnia-18-lutego-2020-r-w-sprawie-wykazu-refundowanych-lekow-srodkow-spozywczychspecjalnego-przeznaczenia-zywieniowego-oraz-wyrobow-medycznych-na-1-marca-2020-r (accessed on 27 February 2020).
- Weber, J.S.; Keahler, K.C.; Hauschild, A. Management of immune-related adverse events and kinetics of response with ipilimumab. J. Clin. Oncol. 2012, 30, 2691–2697. [Google Scholar] [CrossRef] [PubMed]
- Delivanis, D.A.; Gustafson, M.P.; Bornschlegl, S.; Merten, M.M.; Kottschade, L.; Withers, S.; Dietz, A.B.; Ryder, M. Pembrolizumab-induced thyroiditis: Comprehensive clinical review and insights into underlying involved mechanisms. J. Clin. Endocrinol. Metab. 2017, 102, 2770–2780. [Google Scholar] [CrossRef] [PubMed]
- Topalian, S.L.; Drake, C.G.; Pardoll, D.M. Targeting the PD-1/B7-H1 (PD-L1) pathway to activate anti-tumor immunity. Curr. Opin. Immunol. 2012, 24, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Toi, Y.; Sugawara, S.; Kawashima, Y.; Aiba, T.; Kawana, S.; Saito, R.; Tsurumi, K.; Suzuki, K.; Shimizu, H.; Sugisaka, J.; et al. Association of immune-related adverse events with clinical benefit in patients with advanced non-small-cell lung cancer treated with nivolumab. Oncologist 2018, 23, 1358–1365. [Google Scholar] [CrossRef]
- Borghaei, H.; Paz-Ares, L.; Horn, L.; Spigel, D.R.; Steins, M.; Ready, N.E.; Chow, L.Q.; Vokes, E.E.; Felip, E.; Holgado, E.; et al. Nivolumab versus docetaxel in advanced nonsquamous nonsmall-cell lung cancer. N. Engl. J. Med. 2015, 373, 1627–1639. [Google Scholar] [CrossRef]
- Herbst, R.S.; Baas, P.; Kim, D.W.; Felip, E.; Perez-Gracia, J.L.; Han, J.-Y.; Molina, J.; Kim, J.-H.; Arvis, C.D.; Ahn, M.-J.; et al. Pembrolizumab versus docetaxel for previously treated, PD-L1positive, advanced non-small-cell lung cancer (KEYNOTE-010): A randomised controlled trial. Lancet 2016, 387, 1540–1550. [Google Scholar] [CrossRef]
- Stelmachowska-Banaś, M.; Zgliczyński, W. Postępowanie w przypadku wystąpienia endokrynologicznych działań niepożądanych przy stosowaniu leku niwolumab. Onkol. Prakt. Klin. Edu. 2017, 3, 324–330. [Google Scholar]
- Herbst, R.S.; Soria, J.C.; Kowanetz, M.; Fine, G.D.; Hamid, O.; Gordon, M.S.; Sosman, J.A.; McDermott, D.F.; Powderly, J.D.; Gettinger, S.N.; et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 2014, 515, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Brahmer, J.; Reckamp, K.L.; Baas, P.; Crinò, L.; Eberhardt, W.E.E.; Poddubskaya, E.; Antonia, S.; Pluzanski, A.; Vokes, E.E.; Holgado, E.; et al. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N. Engl. J. Med. 2015, 373, 123–135. [Google Scholar] [CrossRef]
- Godwin, J.L.; Jaggi, S.; Sirisena, I.; Sharda, P.; Rao, A.D.; Mehra, R.; Veloski, C. Nivolumab-induced autoimmune diabetes mellitus presenting as diabetic ketoacidosis in a patient with metastatic lung cancer. J. Immunother. Cancer 2017, 5, 40. [Google Scholar] [CrossRef] [PubMed]
- Bellesoeur, A.; Ollier, E.; Allard, M.; Hirsch, L.; Boudou-Rouquette, P.; Arrondeau, J.; Thomas-Schoemann, A.; Tiako, M.; Khoudour, N.; Chapron, J.; et al. Is there an Exposure-Response Relationship for Nivolumab in Real-World NSCLC Patients? Cancers 2019, 11, 1784. [Google Scholar] [CrossRef] [PubMed]
- Judd, J.; Zibelman, M.; Handorf, E.; O’Neill, J.; Ramamurthy, C.; Bentota, S.; Doyle, J.; Uzzo, R.G.; Bauman, J.; Borghaei, H.; et al. Immune-related adverse events as a biomarker in non-melanoma patients treated with programmed cell death 1 inhibitors. Oncologist 2017, 22, 1232–1237. [Google Scholar] [CrossRef]
- Opdivo: Immune-Mediated Adverse Reactions Management Guide. Bristol-Myers Squibb. Available online: http://www.opdivohcp.com/servlet/servlet.FileDownload?file=00Pi000000onjAXEAY (accessed on 4 January 2020).
- Topalian, S.L.; Hodi, F.S.; Brahmer, J.R.; Gettinger, S.N.; Smith, D.C.; McDermott, D.F.; Powderly, J.D.; Carvajal, R.D.; Sosman, J.A.; Atkins, M.B.; et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N. Engl. J. Med. 2012, 366, 2443–2454. [Google Scholar] [CrossRef]
- Robert, C.; Long, G.V.; Brady, B.; Dutriaux, C.; Maio, M.; Mortier, L.; Hassel, J.C.; Rutkowski, P.; McNeil, C.; Kalinka-Warzocha, E.; et al. Nivolumab in previously untreated melanoma without BRAF mutation. N. Engl. J. Med. 2015, 372, 320–330. [Google Scholar] [CrossRef]
- Topalian, S.L.; Sznol, M.; McDermott, D.F.; Kluger, H.M.; Carvajal, R.C.; Sharfman, W.H.; Brahmer, J.R.; Lawrence, D.P.; Atkins, M.B.; Powderly, J.D.; et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J. Clin. Oncol. 2014, 32, 1020–1030. [Google Scholar] [CrossRef]
- Larkin, J.; Chiarion-Sileni, V.; Gonzalez, R.; Grob, J.J.; Cowey, C.L.; Lao, C.D.; Schadendorf, D.; Dummer, R.; Smylie, M.; Rutkowski, P.; et al. Combined Nivolumab and Ipilimumab or Monotherapy in Untreated Melanoma. N. Engl. J. Med. 2015, 373, 23–34. [Google Scholar] [CrossRef]
- Gettinger, S.; Rizvi, N.A.; Chow, L.Q.; Borghaei, H.; Brahmer, J.; Ready, N.; Gerber, D.E.; Shepherd, F.A.; Antonia, S.; Goldman, J.W.; et al. Nivolumab Monotherapy for First-Line Treatment of Advanced Non-Small-Cell Lung Cancer. J. Clin. Oncol. 2016, 34, 2980–2987. [Google Scholar] [CrossRef]
- Rizvi, N.A.; Mazieres, J.; Planchard, D.; Stinchcombe, T.E.; Dy, G.K.; Antonia, S.J.; Horn, L.; Lena, H.; Minenza, E.; Mennecier, B.; et al. Activity and safety of nivolumab, an anti-PD-1 immune checkpoint inhibitor, for patients with advanced, refractory squamous non-small-cell lung cancer (CheckMate 063): A phase 2, single-arm trial. Lancet Oncol. 2015, 16, 257–265. [Google Scholar] [CrossRef]
- Motzer, R.J.; Escudier, B.; McDermott, D.F.; George, S.; Hammers, H.J.; Srinivas, S.; Tykodi, S.S.; Sosman, J.A.; Procopio, G.; Plimack, E.R.; et al. Nivolumab versus Everolimus in Advanced Renal-Cell Carcinoma. N. Engl. J. Med. 2015, 373, 1803–1813. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, K.; Shono-Saito, T.; Yamate, T. A case of fulminant type 1 diabetes mellitus, with a precipitous decrease in pancreatic volume, induced by nivolumab for malignant melanoma: Analysis of HLA and CTLA-4 polymorphisms. Eur. J. Dermatol. 2017, 27, 184–185. [Google Scholar] [CrossRef]
- Sakurai, K.; Niitsuma, S.; Sato, R.; Takahashi, K.; Arihara, Z. Painless Thyroiditis and Fulminant Type 1 Diabetes Mellitus in a Patient Treated with an Immune Checkpoint Inhibitor, Nivolumab. Tohoku J. Exp. Med. 2018, 244, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, H.; Iwasaki, H.; Yamashita, T.; Yoshida, T.; Suganuma, N.; Yamanaka, T.; Masudo, K.; Nakayama, H.; Kohagura, K.; Rino, Y.; et al. Potential Risk Factors for Nivolumab-induced Thyroid Dysfunction. In Vivo 2017, 31, 1225–1228. [Google Scholar] [PubMed]
- Yamauchi, I.; Sakane, Y.; Fukuda, Y.; Fujii, T.; Taura, D.; Hirata, M.; Hirota, K.; Ueda, Y.; Kanai, Y.; Yamashita, Y.; et al. Clinical Features of Nivolumab-Induced Thyroiditis: A Case Series Study. Thyroid 2017, 27, 894–901. [Google Scholar] [CrossRef]
- Weber, J.; Gibney, G.; Kudchadkar, R.; Yu, B.; Cheng, P.; Martinez, A.J.; Kroeger, J.; Richards, A.; McCormick, L.; Moberg, V.; et al. Phase I/II Study of Metastatic Melanoma Patients Treated with Nivolumab Who Had Progressed after Ipilimumab. Cancer Immunol. Res. 2016, 4, 345–353. [Google Scholar] [CrossRef]
- Barroso-Sousa, R.; Barry, W.T.; Garrido-Castro, A.C.; Hodi, F.S.; Min, L.; Krop, I.E.; Tolaney, S.M. Incidence of Endocrine Dysfunction Following the Use of Different Immune Checkpoint Inhibitor Regimens: A Systematic Review and Meta-analysis. JAMA Oncol. 2018, 4, 173–182. [Google Scholar] [CrossRef]
- Tahir, S.A.; Gao, J.; Miura, Y.; Blando, J.; Tidwell, R.S.S.; Zhao, H.; Subudhi, S.K.; Tawbi, H.; Keung, E.; Wargo, J.; et al. Autoimmune antibodies correlate with immune checkpoint therapy- induced toxicities. Proc. Natl. Acad. Sci. USA 2019, 116, 22246–22251. [Google Scholar] [CrossRef]
- Byun, D.J.; Wolchok, J.D.; Rosenberg, L.M.; Girotra, M. Cancer immunotherapy-immune checkpoint blockade and associated endcrinopathies. Nat. Rev. Endocrinol. 2017, 13, 195–207. [Google Scholar] [CrossRef]
- Ryder, M.; Callahan, M.; Postow, M.A.; Wolchok, J.; Fagin, J.A. Endocrine-related adverse events following ipilimumab in patients with advanced melanoma: A comprehensive retrospective review from a single instytution. Endocr. Relat. Cancer 2014, 21, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Faje, A. Immunotherapy and hypophysitis: Clinical presentation, treatment, and biologic insights. Pituitary 2016, 19, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Shang, Y.H.; Zhang, Y.; Li, J.H.; Li, P.; Zhang, X. Risk of endocrine adverse events in cancer patients treated with PD-1 inhibitors: A systematic review and meta-analysis. Immunotherapy 2017, 9, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Iwama, S.; De Remigis, A.; Callahan, M.K.; Slovin, S.F.; Wolchok, J.D.; Caturegli, P. Pituitary expression of CTLA-4 mediates hypophysitis secondary to administration of CTLA-4 blocking antibody. Sci. Transl. Med. 2014, 6, 230ra45. [Google Scholar] [CrossRef] [PubMed]
- Ohara, N.; Kobayashi, M.; Ohashi, K.; Ito, R.; Ikeda, Y.; Kawaguchi, G.; Yoneoka, Y.; Hasegawa, G.; Takada, T. Isolated adrenocorticotropic hormone deficiency and thyroiditis associated with nivolumab therapy in a patient with advanced lung adenocarcinoma: A case report and review of the literature. J. Med. Case Rep. 2019, 13, 88. [Google Scholar] [CrossRef] [PubMed]
- Haanen, J.B.A.G.; Carbonnel, F.; Robert, C.; Kerr, K.M.; Peters, S.; Larkin, J.; Jordan, K. ESMO Guidelines Committee. Management of toxicities from immunotherapy: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2017, 28, iv119–iv142. [Google Scholar] [CrossRef]
- Corsello, S.M.; Barnabei, A.; Marchetti, P.; De Vecchis, L.; Salvatori, R.; Torino, F. Endocrine side effects induced by immune checkpoint inhibitors. J. Clin. Endocrinol. Metab. 2013, 98, 1361–1375. [Google Scholar] [CrossRef]
- Vancieri, G.; Bellia, A.; Lauro, D. Late-onset panhypopituitarism in a 72-year-old male patient treated with ipilimumab for metastatic melanoma: A case report. J. Endocrinol. Investig. 2016, 39, 805–806. [Google Scholar] [CrossRef]
- Machado, C.M.; Santos, L.A.; Barroso, A.; Oliveira, M.J. Nivolumab-induced Hypothyroidism Followed by Isolated ACTH Deficiency. BMJ Case Rep. 2019, 12, e231236. [Google Scholar] [CrossRef]
- Chang, J.; Tran, J.; Kamel, D.; Basu, A. Nivolumab-induce hypophysitis leading to hypopituitarism and secondary empty sella syndrome in a patient with non-small cell lung cancer. MBJ Case Rep. 2019, 12, e228135. [Google Scholar] [CrossRef]
- Sekizaki, T.; Kameda, H.; Oba, C.; Cho, K.Y.; Nakamura, A.; Miyoshi, H.; Osawa, T.; Shinohara, N.; Atsumi, T. Nivolumab-induced hypophysitis causing secondary adrenal insufficiency after transient ACTH elevation. Endocr. J. 2019, 66, 937–941. [Google Scholar] [CrossRef] [PubMed]
- Mishima, Y.; Fukaishi, T.; Inase, N.; Isogai, S. Nivolumab-induced Hypophysitis, Secondary Adrenal Insufficiency and Destructive Thyroiditis in a Patient with Lung Adenocarcinoma. Intern. Med. 2019, 58, 693–697. [Google Scholar] [CrossRef] [PubMed]
- Thapi, S.; Leiter, A.; Galsky, M.; Gallagher, E.J. Recovery from secondary adrenal insufficiency in a patient with immune checkpoint inhibitor therapy induced hypophysitis. J. Immunother. Cancer 2019, 7, 248. [Google Scholar] [CrossRef] [PubMed]
- Seki, T.; Yasuda, A.; Oki, M.; Kitajima, N.; Takagi, A.; Nakajima, N.; Miyajima, A.; Fukagawa, M. Secondary Adrenal Insufficiency Following Nivolumab Therapy in a Patient with Metastatic Renal Cell Carcinoma. Tokai J. Exp. Clin. Med. 2017, 42, 115–120. [Google Scholar] [PubMed]
- Kitajima, K.; Ashida, K.; Wada, N.; Suetsugu, R.; Takeichi, Y.; Sakamoto, S.; Uchi, H.; Matsushima, T.; Shiratsuchi, M.; Ohnaka, K.; et al. Isolated ACTH deficiency probably induced by autoimmune-related mechanism evoked with nivolumab. Jpn. J. Clin. Oncol. 2017, 47, 463–466. [Google Scholar] [CrossRef] [PubMed]
- Cukier, P.; Santini, F.C.; Scaranti, M.; Hoff, A.O. Endocrine side effects of cancer immunotherapy. Endocr. Relat. Cancer 2017, 24, T331–T347. [Google Scholar] [CrossRef]
- Takaya, K.; Sonoda, M.; Fuchigami, A.; Hiyoshi, T. Isolated adrenocorticotropic hormone deficiency caused by nivolumab in a patient with metastatic lung cancer. Intern. Med. 2017, 56, 2463–2469. [Google Scholar] [CrossRef]
- Akahori, H. Reversible impairment of the processing of proopiomelanocortin into ACTH in pituitary enlargement suspected of lymphocytic hypophysitis. Endocr. J. 2012, 59, 1121–1129. [Google Scholar] [CrossRef]
- Weber, J.S.; Yang, J.C.; Atkins, M.B.; Disis, M.L. Toxicities of immunotherapy for the practitioner. J. Clin. Oncol. 2015, 33, 2092–2099. [Google Scholar] [CrossRef]
- Joshi, M.N.; Whitelaw, B.C.; Palomar, M.T.; Wu, Y.; Carroll, P.V. Immune checkpoint inhibitor-related hypophysitis and endocrine dysfunction: Clinical review. Clin. Endocrinol. 2016, 85, 331–339. [Google Scholar] [CrossRef]
- Puzanov, I.; Diab, A.; Abdallah, K.; Bingham, C.O., 3rd; Brogdon, C.; Dadu, R.; Hamad, L.; Kim, S.; Lacouture, M.E.; LeBoeuf, N.R.; et al. Managing toxicities associated with immune checkpoint inhibitors: Consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J. Immunother. Cancer 2017, 5, 95. [Google Scholar] [CrossRef] [PubMed]
- Brahmer, J.R.; Lacchetti, C.; Schneider, B.J.; Atkins, M.B.; Brassil, K.J.; Caterino, J.M.; Chau, I.; Ernstoff, M.S.; Gardner, J.M.; Ginex, P.; et al. Management of Immune-Related Adverse Events in Patients Treated with Immune Checkpoint Inhibitor Therapy: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 2018, 36, 1714–1768. [Google Scholar] [CrossRef] [PubMed]
- Common Terminology Criteria for Adverse Events (CTCAE); Version 5; National Cancer Institute: Bethesda, MD, USA, 2003. Available online: https://ctep.cancer.gov/protocolDevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_8.5×11.pdf (accessed on 9 January 2020).
- Abdel-Rahman, O.; El Halawani, H.; Fouad, M. Risk of endocrine complications in cancer patients treated with immune check point inhibitors: A meta-analysis. Future Oncol. 2016, 12, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Osorio, J.C.; Ni, A.; Chaft, J.E.; Pollina, R.; Kasler, M.K.; Stephens, D.; Rodriguez, C.; Cambridge, L.; Rizvi, H.; Wolchok, J.D.; et al. Antibody-mediated thyroid dysfunction during T-cell checkpoint blockade in patients with non-small cell lung cancer. Ann. Oncol. 2017, 28, 583–589. [Google Scholar] [CrossRef] [PubMed]
- De Filette, J.; Jansen, Y.; Schreuer, M.; Everaert, H.; Velkeniers, B.; Neyns, B.; Bravenboer, B. Incidence of thyroid-related adverse events in melanoma patients treated with pembrolizumab. J. Clin. Endocrinol. Metab. 2016, 101, 4431–4439. [Google Scholar] [CrossRef] [PubMed]
- Peiró, I.; Palmero, R.; Iglesias, P.; Díez, J.J.; Simó-Servat, A.; Marín, J.A.; Jiménez, L.; Domingo-Domenech, E.; Mancho-Fora, N.; Nadal, E.; et al. Thyroid dysfunction induced by nivolumab: Searching for disease patterns and outcomes. Endocrine 2019, 64, 605–613. [Google Scholar] [CrossRef]
- Ramos-Levi, A.M.; Rogado, J.; Sanchez-Torres, J.M.; Colomer, R.; Marazuela, M. Nivolumab-induced thyroid dysfunction in patients with lung cancer. Endocrinol. Diabetes Nutr. 2019, 66, 26–34. [Google Scholar] [CrossRef]
- Brancatella, A.; Viola, N.; Brogioni, S.; Montanelli, L.; Sardella, C.; Vitti, P.; Marcocci, C.; Lupi, I.; Latrofa, F. Graves’ Disease Induced by Immune Checkpoint Inhibitors: A Case Report and Review of the Literature. Eur. Thyroid. J. 2019, 8, 192–195. [Google Scholar] [CrossRef]
- Narita, T.; Oiso, N.; Taketomo, Y.; Okahashi, K.; Yamauchi, K.; Sato, M.; Uchida, S.; Matsuda, H.; Kawada, A. Serological aggravation of autoimmune thyroid disease in two cases receiving nivolumab. J. Dermatol. 2016, 43, 210–214. [Google Scholar] [CrossRef]
- Gutzmer, R.; Koop, A.; Meier, F.; Hassel, J.C.; Terheyden, P.; Zimmer, L.; Heinzerling, L.; Ugurel, S.; Pföhler, C.; Gesierich, A.; et al. Programmed cell death protein-1 (PD-1) inhibitor therapy in patients with advanced melanoma and preexisting autoimmunity or ipilimumab-triigered autoimmunity. Eur. J. Cancer 2017, 75, 24–32. [Google Scholar] [CrossRef]
- Garon-Czmil, J.; Petitpain, N.; Rouby, F.; Sassier, M.; Babai, S.; Yelehe-Okouma, M.; Weryha, G.; Klein, M.; Gillet, P. Thyroiditis and immune check point inhibitors: The post-marketing experience using the French National Pharmacovigilance database. Fundam. Clin. Pharmacol. 2019, 33, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Campredon, P.; Imbert, P.; Mouly, C.; Grunenwald, S.; Mazières, J.; Caron, P. Severe Inflammatory Ophthalmopathy in a Euthyroid Patient during Nivolumab Treatment. Eur. Thyroid. J. 2018, 7, 84–87. [Google Scholar] [CrossRef] [PubMed]
- van Kooten, M.J.; van den Berg, G.; Glaudemans, A.W.J.M.; Hiltermann, T.J.N.; Groen, H.J.M.; Rutgers, A.; Links, T.P. Transient thyrotoxicosis during nivolumab treatment. Neth. J. Med. 2017, 75, 204–207. [Google Scholar] [PubMed]
- Elia, G.; Ferrari, S.M.; Galdiero, M.R.; Ragusa, F.; Paparo, S.R.; Ruffilli, I.; Varricchi, G.; Fallahi, P.; Antonelli, A. New insight in endocrine-related adverse events associated to immune checkpoint blockade. Best Pract. Res. Clin. Endocrinol. Metab. 2020, 34, 101370. [Google Scholar] [CrossRef]
- Kobayashi, T.; Iwama, S.; Yasuda, Y.; Okada, N.; Tsunekawa, T.; Onoue, T.; Takagi, H.; Hagiwara, D.; Ito, Y.; Morishita, Y.; et al. Patients with antithyroid antibodies are prone to develop destructive thyroiditis by nivolumab: A prospective study. J. Endocr. Soc. 2018, 2, 241–251. [Google Scholar] [CrossRef]
- Presotto, E.M.; Rastrelli, G.; Desideri, I.; V Scotti, V.; Gunnella, S.; Pimpinelli, N.; Vaccher, E.; Bearz, A.; Di Costanzo, F.; Bruggia, M.; et al. Endocrine toxicity in cancer patients treated with nivolumab or pembrolizumab: Results of a large multicentre study. J. Endocrinol. Investig. 2020, 43, 337–345. [Google Scholar] [CrossRef]
- Yamauchi, I.; Yasoda, A.; Matsumoto, S.; Sakamori, Y.; Kim, Y.H.; Nomura, M.; Otsuka, A.; Yamasaki, T.; Saito, R.; Kitamura, M.; et al. Incidence, features, and prognosis of immune-related adverse events involving the thyroid gland induced by nivolumab. PLoS ONE 2019, 14, e0216954. [Google Scholar] [CrossRef]
- Stagnaro-Green, A.; Schwartz, A.; Gismondi, R.; Tinelli, A.; Mangieri, T.; Negro, R. High rate of persistent hypothyroidism in a large-scale prospective study of postpartum thyroiditis in southern Italy. J. Clin. Endocrinol. Metab. 2011, 96, 652–657. [Google Scholar] [CrossRef]
- Wolchok, J.D.; Kluger, H.; Callahan, M.K.; Postow, M.A.; Rizvi, N.A.; Lesokhin, A.M.; Segal, N.H.; Ariyan, C.E.; Gordon, R.A.; Reed, K.; et al. Nivolumab plus ipilimumab in advanced melanoma. N. Engl. J. Med. 2013, 369, 122–133. [Google Scholar] [CrossRef]
- Trainer, H.; Hulse, P.; Higham, C.E.; Trainer, P.; Lorigan, P. Hyponatraemia secondary to nivolumab-induced primary adrenal failure. Endocrinol. Diabetes Metab. Case Rep. 2016, 2016, 16–0108. [Google Scholar] [CrossRef]
- Clinical Manifestations of Adrenal Insufficiency in Adults. Available online: https://www.uptodate.com/contents/clinical-manifestations-of-adrenal-insufficiency-inadults (accessed on 29 July 2020).
- Kotwal, A.; Haddox, C.; Block, M.; Kudva, Y.C. Immune checkpoint inhibitors: An emerging cause of insulin-dependent diabetes. BMJ Open Diabetes Res. Care 2019, 7, e000591. [Google Scholar] [CrossRef] [PubMed]
- de Filette, J.M.K.; Pen, J.J.; Decoster, L.; Vissers, T.; Bravenboer, B.; Van der Auwera, B.J.; Gorus, F.K.; Roep, B.O.; Aspeslagh, S.; Neyns, B.; et al. Immune checkpoint inhibitors and type 1 diabetes mellitus: A case report and systematic review. Eur. J. Endocrinol. 2019, 181, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.; Vudattu, N.; Sznol, M.; Gettinger, S.; Kluger, H.; Lupsa, B.; Herold, K.C. Precipitation of autoimmune diabetes with anti-PD-1 immunotherapy. Diabetes Care 2015, 38, e55–e57. [Google Scholar] [PubMed]
- Gauci, M.L.; Laly, P.; Vidal-Trecan, T.; Baroudjian, B.; Gottlieb, J.; Madjlessi-Ezra, N.; Da Meda, L.; Madelaine-Chambrin, I.; Bagot, M.; Basset-Seguin, N.; et al. Autoimmune diabetes induced by PD-1 inhibitor-retrospective analysis and pathogenesis: A case report and literature review. Cancer Immunol. Immunother. 2017, 66, 1399–1410. [Google Scholar] [CrossRef] [PubMed]
- Martin-Liberal, J.; Furness, A.J.; Joshi, K.; Peggs, K.S.; Quezada, S.A.; Larkin, J. Anti-programmed cell death-1 therapy and insulin-dependent diabetes: A case report. Cancer Immunol. Immunother. 2015, 64, 765–767. [Google Scholar] [CrossRef] [PubMed]
- Lowe, J.R.; Perry, D.J.; Salama, A.K.S.; Mathews, C.E.; Moss, L.G.; Hanks, B.A. Genetic risk analysis of a patient with fulminant autoimmune type 1 diabetes mellitus secondary to combination ipilimumab and nivolumab immunotherapy. J. Immunother. Cancer 2016, 4, 89. [Google Scholar] [CrossRef]
- Araujo, M.; Ligeiro, D.; Costa, L.; Marques, F.; Trindade, H.; Correia, J.M.; Fonseca, C. A case of fulminant Type 1 diabetes following anti-PD1 immunotherapy in a genetically susceptible patient. Immunotherapy 2017, 9, 531–535. [Google Scholar] [CrossRef]
- American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2020. Diabetes Care 2020, 43, S14–S31. [Google Scholar] [CrossRef]
- Sato, K.; Akamatsu, H.; Murakami, E.; Sasaki, S.; Kanai, K.; Hayata, A.; Tokudome, N.; Akamatsu, K.; Koh, Y.; Ueda, H.; et al. Correlation between immune-related adverse events and eficacy in nonsmall cell lung cancer treated with nivolumab. Lung Cancer 2018, 115, 71–74. [Google Scholar] [CrossRef]
- Weber, J.S.; Hodi, F.S.; Wolchok, J.D.; Topalian, S.L.; Schadendorf, D.; Larkin, J.; Sznol, M.; Long, G.V.; Li, H.; Waxman, I.M.; et al. Safety Profile of Nivolumab Monotherapy: A Pooled Analysis of Patients With Advanced Melanoma. J. Clin. Oncol. 2017, 35, 785–792. [Google Scholar] [CrossRef]
- Freeman-Keller, M.; Kim, Y.; Cronin, H.; Richards, A.; Gibney, G.; Weber, J.S. Nivolumab in resected and unresectable metastatic melanoma: Characteristics of immune-related adverse events and association with outcomes. Clin. Cancer Res. 2016, 22, 886–894. [Google Scholar] [CrossRef] [PubMed]
- Sanlorenzo, M.; Vujic, I.; Daud, A.; Algazi, A.; Gubens, M.; Luna, S.A.; Lin, K.; Quaglino, P.; Rappersberger, K.; Ortiz-Urda, S. Pembrolizumab Cutaneous Adverse Events and Their Association with Disease Progression. JAMA Dermatol. 2015, 151, 1206–1212. [Google Scholar] [CrossRef]
- Haratani, K.; Hayashi, H.; Chiba, Y.; Kudo, K.; Yonesaka, K.; Kato, R.; Kaneda, H.; Hasegawa, Y.; Tanaka, K.; Takeda, M.; et al. Association of Immune-Related Adverse Events with Nivolumab Efficacy in Non-Small-Cell Lung Cancer. JAMA Oncol. 2018, 4, 374–378. [Google Scholar] [CrossRef] [PubMed]
- Teraoka, S.; Fujimoto, D.; Morimoto, T.; Kawachi, H.; Ito, M.; Sato, Y.; Nagata, K.; Nakagawa, A.; Otsuka, K.; Uehara, K.; et al. Early Immune-Related Adverse Events and Association with Outcome in Advanced Non–Small Cell Lung Cancer Patients Treated with Nivolumab: A Prospective Cohort Study. J. Thorac. Oncol. 2017, 12, 1798–1805. [Google Scholar] [CrossRef] [PubMed]
- Hasan Ali, O.; Diem, S.; Markert, E.; Jochum, W.; Kerl, K.; French, L.E.; Speiser, D.E.; Früh, M.; Flatz, L. Characterization of nivolumab-associated skin reactions in patients with metastatic non-small cell lung cancer. Oncoimmunology 2016, 5, e1231292. [Google Scholar] [CrossRef] [PubMed]
- Campredon, P.; Mouly, C.; Lusque, A.; Bigay-Game, L.; Bousquet, E.; Mazieres, J.; Caron, P. Incidence of thyroid dysfunctions during treatment with nivolumab for non-small cell lung cancer: Retrospective study of 105 patients. La Presse Médicale 2019, 48, e199–e207. [Google Scholar] [CrossRef]
- Zhou, X.; Yao, Z.; Yang, H.; Liang, N.; Zhang, X.; Zhang, F. Are immune-related adverse events associated with the efficacy of immune checkpoint inhibitors in patients with cancer? A systematic review and meta-analysis. BMC Med. 2020, 18, 87. [Google Scholar] [CrossRef]
| Adverse Event | Occurrence Rate (%) [30] | Median Time of Occurrence of Adverse Event [30] (Months) | Frequency of Occurrence Observed in Other Studies |
|---|---|---|---|
| Hypophysitis | 0.6 | 4.9 (range 1.4 to 11 months) | 0.5–0.9 [31,32,33,34] |
| Adrenal Insufficiency | 1 | 4.3 (range 15 days to 21 months) | 0.85–1.9 [35,36,37] |
| Type 1 Diabetes Mellitus | 0.9 1 | 4.4 (range 15 days to 22 months) | 0.1–1.5 [32,37,38,39] |
| Hypothyroidism/Thyroiditis | 9 | 2.9 (range 1 day to 16.6 months) | 2.6–10.1 [33,34,35,36,40,41,42] |
| Hyperthyroidism | 2.7 | 1.5 (range 1 day to 14.2 months) | 0.8–15.3 [34,35,37,40,41,42] |
| Adverse Events | Intensity [14,68] | Recommended Nivolumab Dosage Modification | Monitoring/Treatment |
|---|---|---|---|
| Hypophysitis | Grade 1—asymptomatic or mild symptoms, e.g., fatigue, weakness, loss of appetite of mild intensity, no headache | Continued treatment | -Hormonal evaluation -Pituitary MRI -Clinical and hormonal evaluation every 1–3 weeks -Repeat MRI after one month if the symptoms remain and the hormonal tests and/or MRI are normal -Hormonal substitution if necessary (e.g., hydrocortisone 10 to 20 mg orally in the morning, 5 to 10 mg orally in early afternoon; levothyroxine by weight, testosterone or estrogen therapy as needed in those without contraindications. Always start corticosteroids several days before thyroid hormone to prevent precipitating adrenal crisis. Follow FT4 for thyroid hormone replacement titration (TSH is not accurate). ≥Grade 2: 1 mg/kg/day prednisone/prednisone equivalents -Substitution treatment if necessary -Analgesic treatment in case of headaches -In case of clinical improvement (with or without substitution treatment continuation), taper steroids over at least 1 month before resuming treatment -Return to treatment in case of return of AE to level 0–1 |
| Grade 2—moderate symptoms, e.g., headaches without vision disturbances, fatigue, worse well-being, hemodynamically stable, no electrolyte abnormalities | Withhold dose, temporary drug interruption | ||
| Grade 3—serious mass-effect symptoms, e.g., headaches, vision disturbances or severe symptoms of adrenal insufficiency | Withhold dose, temporary drug interruption | ||
| Grade 4 | Permanently discontinue | ||
| Adrenal insufficiency | Grade 1 Asymptomatic; | Clinical or diagnostic observations only; no intervention in terms of nivolumab treatment | -Clinical and hormonal evaluation repeated every 1–3 weeks -Glucocorticoids administered in the substitutional doses (hydrocortisone: 10 to 20 mg orally in the morning, 5 to 10 mg orally in early afternoon or prednisone: 5 to 10 mg daily, fludrocortisone (0.1 mg/d) for mineralocorticoid replacement in primary adrenal insufficiency -Grades 3–4: 1 to 2 mg/kg/day prednisone equivalents followed by corticosteroid taper over at least 1 month -Return to treatment in case of return of AE to level 0–1 |
| Grade 2 Moderate symptoms; | Consider withholding dose | ||
| Grade 3 Severe symptoms; | Discontinuation of treatment/hospitalization | ||
| Grade 4 Life-threatening symptoms | |||
| Type 1 diabetes | Grade 1 Asymptomatic or mild symptoms; fasting glucose value >ULN-160 mg/dl (>ULN-8.9 mmol/L), no evidence of ketosis | Clinical or diagnostic observations only; no intervention in terms of nivolumab treatment | -Monitoring and treatment of hyperglycemia -In case of AE returning to level 0–1, return to treatment |
| Grade 2 Moderate symptoms, fasting glucose value >160–250 mg/dl (>8.9–13.9 mmol/L) | |||
| Grade 3 Fasting glucose value >250–500 mg/dl (>13.9–27.8 mmol/l) | Withhold dose, hospitalization indicated | ||
| Grade 4 Fasting glucose value >500 mg/dl (>27.8 mmol/L) | Discontinuation of treatment, hospitalization indicated | ||
| Thyroid disorders-Hypothyroidism | Grade 1 Asymptomatic or few symptoms TSH elevated (<10 mUL/L), FT3, FT4-normal aTPO and anti-TG usually high | There are no recommended modifications of dosage of nivolumab. In severe cases, (Grade ≥ 3) consider withholding ICI | -Monitor thyroid function prior to and periodically during treatment (before each cycle) -Levothyroxine in case of hypothyroidism -Grade ≥ 3: Hospitalization Supportive therapy for severe cardio-respiratory symptoms Return to Grade 2—consider return to immunotherapy |
| Grade 2 Mild symptoms (fatigue, weight gain, constipations, dry skin, eyelid edema, puffy face) Low FT3 and/or FT4, TSH > 10 mUI/L aTPO/antiTg usually high | |||
| Grade 3–4 Moderate–severe symptoms (bradycardia, hypotension, pericardial effusion, depression, hypoventilation, stupor, lethargy to myxedema coma) -Very low FT4, FT3 -TSH very high -aTPO and antiTG usually high | |||
| Thyroid disorders Hyperthyroidism | Grade 1 Asymptomatic -FT3, FT4 normal -TSH suppressed (<0.1 mUI/L), antiTPO/aTG normal or high | There are no recommended modifications of dosage of nivolumab. In severe cases (Grade ≥ 3), consider withholding ICI | -TSH, FT4, FT3 before each cycle -Close monitoring of thyroid function to catch transition to hypothyroidism in patients with thyroiditis and hyperthyroidism -Overt hyperthyroidism: introduce beta-blocker (propranolol/atenolol/metoprolol) -Consider glucocorticoid therapy—prednisone 1 to 2 mg/kg/d or equivalent tapered over 1 to 2 weeks (not routinely) -Anti-thyroid drug (thiamazol/PTU) in case of Graves’ disease -Grade ≥ 3: Hospitalization -In case of destructive thyroiditis, consider treatment with oral glucocorticoid (prednisone equivalent 0.5–1 mg/kg/day followed by dose reduction) -In case of withholding ICI treatment, consider restarting when symptoms controlled |
| Grade 2 Mild symptoms: weight loss, increased appetite, anxiety and irritability, muscle weakness -TSH suppressed (<0.1 mUI/L) -FT4, FT4 high -aTPO/aTG normal or high -TSAbs high in case of Graves’ disease | |||
| Grade 3–4 Moderate to severe symptoms: arrhythmia, atrial fibrillation, tremor -TSH suppressed (<0.1 mUI/L) -FT4, FT4 high -aTPO/aTG normal or high -TSAbs high in case of Graves’ disease |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dudzińska, M.; Szczyrek, M.; Wojas-Krawczyk, K.; Świrska, J.; Chmielewska, I.; Zwolak, A. Endocrine Adverse Events of Nivolumab in Non-Small Cell Lung Cancer Patients—Literature Review. Cancers 2020, 12, 2314. https://doi.org/10.3390/cancers12082314
Dudzińska M, Szczyrek M, Wojas-Krawczyk K, Świrska J, Chmielewska I, Zwolak A. Endocrine Adverse Events of Nivolumab in Non-Small Cell Lung Cancer Patients—Literature Review. Cancers. 2020; 12(8):2314. https://doi.org/10.3390/cancers12082314
Chicago/Turabian StyleDudzińska, Marta, Michał Szczyrek, Kamila Wojas-Krawczyk, Joanna Świrska, Izabela Chmielewska, and Agnieszka Zwolak. 2020. "Endocrine Adverse Events of Nivolumab in Non-Small Cell Lung Cancer Patients—Literature Review" Cancers 12, no. 8: 2314. https://doi.org/10.3390/cancers12082314
APA StyleDudzińska, M., Szczyrek, M., Wojas-Krawczyk, K., Świrska, J., Chmielewska, I., & Zwolak, A. (2020). Endocrine Adverse Events of Nivolumab in Non-Small Cell Lung Cancer Patients—Literature Review. Cancers, 12(8), 2314. https://doi.org/10.3390/cancers12082314






