Detecting Progression of Melanocytic Choroidal Tumors by Sequential Imaging: Is Ultrasonography Necessary?
Abstract
1. Introduction
2. Methods
3. Results
4. Discussion
4.1. Main Finding
4.2. How Important Is Monitoring Tumor Thickness?
4.3. Value of Ultrasonographic Evaluation in Addition to Measurement of Tumor Thickness
4.4. Optimal Method for Measuring Tumor Thickness
4.5. Benefits of Omitting Ultrasonography from the Monitoring of Choroidal Nevi
4.6. Strengths and Weaknesses
4.7. Further Research
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Au, A.; Gupta, O. The economics of telemedicine for vitreoretinal diseases. Curr. Opin. Ophthalmol. 2011, 22, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.; Edwards, R.T. Diabetic retinopathy screening: A systematic review of the economic evidence. Diabet. Med. 2010, 27, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Kirkizlar, E.; Serban, N.; Sisson, J.A.; Swann, J.L.; Barnes, C.S.; Williams, M.D. Evaluation of telemedicine for screening of diabetic retinopathy in the Veterans Health Administration. Ophthalmology 2013, 120, 2604–2610. [Google Scholar] [CrossRef] [PubMed]
- Quinn, G.E.; Ying, G.S.; Daniel, E.; Hildebrand, P.L.; Ells, A.; Baumritter, A.; Kemper, A.R.; Schron, E.B.; Wade, K.; e-ROP Cooperative Group. Validity of a telemedicine system for the evaluation of acute-phase retinopathy of prematurity. JAMA Ophthalmol. 2014, 132, 1178–1184. [Google Scholar] [CrossRef]
- Thomas, S.M.; Jeyaraman, M.M.; Hodge, W.G.; Hutnik, C.; Costella, J.; Malvankar-Mehta, M.S. The effectiveness of teleglaucoma versus in-patient examination for glaucoma screening: A systematic review and meta-analysis. PLoS ONE 2014, 9, e113779. [Google Scholar] [CrossRef]
- Roelofs, K.; Weis, E. Tele-Oncology: A Validation Study of Choroidal and Iris Nevi. Ocul. Oncol. Pathol. 2019, 5, 298–302. [Google Scholar] [CrossRef]
- Lapere, S.; Weis, E. Tele-ophthalmology for the monitoring of choroidal and iris nevi: A pilot study. Can. J. Ophthalmol. 2018, 53, 471–473. [Google Scholar] [CrossRef]
- Balaskas, K.; Gray, J.; Blows, P.; Rajai, A.; Flaye, D.; Peto, T.; Sagoo, M.S. Management of choroidal naevomelanocytic lesions: Feasibility and safety of a virtual clinic model. Br. J. Ophthalmol. 2016, 100, 665–670. [Google Scholar] [CrossRef]
- Damato, B. Managing patients with choroidal melanoma in the COVID-19 era: A personal perspective. Br. J. Ophthalmol. 2020, 104, 885–886. [Google Scholar] [CrossRef]
- Sumich, P.; Mitchell, P.; Wang, J.J. Choroidal nevi in a white population: The Blue Mountains Eye Study. Arch. Ophthalmol. 1998, 116, 645–650. [Google Scholar] [CrossRef]
- Jonas, J.B.; You, Q.S.; Xu, L.; Wang, Y.X. Choroidal nevi in adult Chinese. Ophthalmology 2008, 115, 1102-1102.E1. [Google Scholar] [CrossRef]
- Greenstein, M.B.; Myers, C.E.; Meuer, S.M.; Klein, B.E.; Cotch, M.F.; Wong, T.Y.; Klein, R. Prevalence and characteristics of choroidal nevi: The multi-ethnic study of atherosclerosis. Ophthalmology 2011, 118, 2468–2473. [Google Scholar] [CrossRef]
- Qiu, M.; Shields, C.L. Choroidal Nevus in the United States Adult Population: Racial Disparities and Associated Factors in the National Health and Nutrition Examination Survey. Ophthalmology 2015, 122, 2071–2083. [Google Scholar] [CrossRef]
- Ng, C.H.; Wang, J.J.; Mitchell, P.; Amirul Islam, F.M.; Wong, T.Y. Prevalence and characteristics of choroidal nevi in an Asian vs. White population. Arch. Ophthalmol. 2009, 127, 314–319. [Google Scholar] [CrossRef]
- Nangia, V.; Jonas, J.B.; Agarwal, S.; Khare, A.; Lambat, S.; Panda-Jonas, S. Choroidal nevi in adult Indians: The Central India Eye and Medical Study. Br. J. Ophthalmol. 2012, 96, 1443–1444. [Google Scholar] [CrossRef]
- Singh, A.D.; Kalyani, P.; Topham, A. Estimating the risk of malignant transformation of a choroidal nevus. Ophthalmology 2005, 112, 1784–1789. [Google Scholar] [CrossRef]
- Kivelä, T.; Eskelin, S. Transformation of nevus to melanoma. Ophthalmology 2006, 113, 887–888. [Google Scholar] [CrossRef]
- Eskelin, S.; Pyrhönen, S.; Summanen, P.; Hahka-Kemppinen, M.; Kivelä, T. Tumor doubling times in metastatic malignant melanoma of the uvea: Tumor progression before and after treatment. Ophthalmology 2000, 107, 1443–1449. [Google Scholar] [CrossRef]
- Damato, B. Ocular treatment of choroidal melanoma in relation to the prevention of metastatic death—A personal view. Prog. Retin. Eye Res. 2018, 66, 187–199. [Google Scholar] [CrossRef]
- Wollweber, M.; Roth, B. Raman Sensing and Its Multimodal Combination with Optoacoustics and OCT for Applications in the Life Sciences. Sensors 2019, 19, 2387. [Google Scholar] [CrossRef]
- Drexler, W.; Liu, M.; Kumar, A.; Kamali, T.; Unterhuber, A.; Leitgeb, R.A. Optical coherence tomography today: Speed, contrast, and multimodality. J. Biomed. Opt. 2014, 19, 071412. [Google Scholar] [CrossRef]
- Callaway, N.F.; Mruthyunjaya, P. Widefield imaging of retinal and choroidal tumors. Int. J. Retin. Vitr. 2019, 5, 49. [Google Scholar] [CrossRef] [PubMed]
- Shields, C.L.; Shields, J.A.; Kiratli, H.; De Potter, P.; Cater, J.R. Risk factors for growth and metastasis of small choroidal melanocytic lesions. Ophthalmology 1995, 102, 1351–1361. [Google Scholar] [CrossRef]
- Shields, C.L.; Cater, J.; Shields, J.A.; Singh, A.D.; Santos, M.C.; Carvalho, C. Combination of clinical factors predictive of growth of small choroidal melanocytic tumors. Arch. Ophthalmol. 2000, 118, 360–364. [Google Scholar] [CrossRef]
- Shields, C.L.; Furuta, M.; Berman, E.L.; Zahler, J.D.; Hoberman, D.M.; Dinh, D.H.; Mashayekhi, A.; Shields, J.A. Choroidal nevus transformation into melanoma: Analysis of 2514 consecutive cases. Arch. Ophthalmol. 2009, 127, 981–987. [Google Scholar] [CrossRef] [PubMed]
- Collaborative Ocular Melanoma Study Group. Factors predictive of growth and treatment of small choroidal melanoma: COMS Report No. 5. The Collaborative Ocular Melanoma Study Group. Arch. Ophthalmol. 1997, 115, 1537–1544. [Google Scholar] [CrossRef]
- Shields, C.L.; Dalvin, L.A.; Ancona-Lezama, D.; Yu, M.D.; Di Nicola, M.; Williams, B.K., Jr.; Lucio-Alvarez, J.A.; Ang, S.M.; Maloney, S.; Welch, R.J.; et al. Choroidal nevus imaging features in 3,806 cases and risk factors for transformation into melanoma in 2,355 cases: The 2020 Taylor R. Smith and Victor T. Curtin Lecture. Retina 2019, 39, 1840–1851. [Google Scholar] [CrossRef]
- Al-Harby, L.; Sagoo, M.; O’Day, R.; Hay, G.; Arora, A.; Keane, P.A.; Cohen, M.L.; Damato, B. External Validation of the MOLES Scoring System for Estimating Risk of Malignancy in Melanocytic Choroidal Tumors. 2020. Submitted for publication. [Google Scholar]
- Roelofs, K.A.; O’Day, R.; Al Harby, L.; Arora, A.K.; Cohen, V.M.L.; Sagoo, M.S.; Damato, B. The MOLES system for planning management of melanocytic choroidal tumors: Is it safe? Cancers 2020, 12, 1311. [Google Scholar] [CrossRef]
- Kivelä, T.; Simpson, E.; Grossniklaus, H. Uveal Melanoma, 8th ed.; Springer: New York, NY, USA, 2017. [Google Scholar]
- Gass, J.D. Observation of suspected choroidal and ciliary body melanomas for evidence of growth prior to enucleation. Ophthalmology 1980, 87, 523–528. [Google Scholar] [CrossRef]
- Mims, J.L., 3rd; Shields, J.A. Follow-up studies of suspicious choroidal nevi. Ophthalmology 1978, 85, 929–943. [Google Scholar] [CrossRef]
- Shields, C.L.; Lim, L.S.; Dalvin, L.A.; Shields, J.A. Small choroidal melanoma: Detection with multimodal imaging and management with plaque radiotherapy or AU-011 nanoparticle therapy. Curr. Opin. Ophthalmol. 2019, 30, 206–214. [Google Scholar] [CrossRef]
- Dalvin, L.A.; Shields, C.L.; Ancona-Lezama, D.A.; Yu, M.D.; Di Nicola, M.; Williams, B.K., Jr.; Lucio-Alvarez, J.A.; Ang, S.M.; Maloney, S.M.; Welch, R.J.; et al. Combination of multimodal imaging features predictive of choroidal nevus transformation into melanoma. Br. J. Ophthalmol. 2019, 103, 1441–1447. [Google Scholar] [CrossRef]
- Doro, D.; Kotsafti, O.; Cimatti, P. Long-term echographic surveillance of elevated choroidal nevi. Am. J. Ophthalmol. 2013, 156, 438–443. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.U.; Kaliki, S.; Shields, C.L.; Ferenczy, S.R.; Harmon, S.A.; Shields, J.A. Enhanced depth imaging optical coherence tomography of choroidal nevus in 104 cases. Ophthalmology 2012, 119, 1066–1072. [Google Scholar] [CrossRef]
- Dolz-Marco, R.; Hasanreisoglu, M.; Shields, J.A.; Shields, C.L. Posterior scleral bowing with choroidal nevus on enhanced-depth imaging optical coherence tomography. JAMA Ophthalmol. 2015, 133, 1165–1170. [Google Scholar] [CrossRef]
- Jonna, G.; Daniels, A.B. Enhanced depth imaging OCT of ultrasonographically flat choroidal nevi demonstrates 5 distinct patterns. Ophthalmol. Retin. 2019, 3, 270–277. [Google Scholar] [CrossRef]
- Martins, M.F.; Kiefer, K.; Kanecadan, L.A.A.; Garcia, P.N.; Belfort, R.N.; Allemann, N. Comparisons of choroidal nevus measurements obtained using 10- and 20-MHz ultrasound and spectral domain optical coherence tomography. Arq. Bras. Oftalmol. 2017, 80, 78–83. [Google Scholar] [CrossRef]
- Barsam, A.S.; Gibbons, A.; McClellan, A.J.; Harbour, J.W.; Smiddy, W.E. Follow the nevus: The cost-utility of monitoring for growth of choroidal nevi. Int. J. Ophthalmol. 2019, 12, 1456–1464. [Google Scholar] [CrossRef]
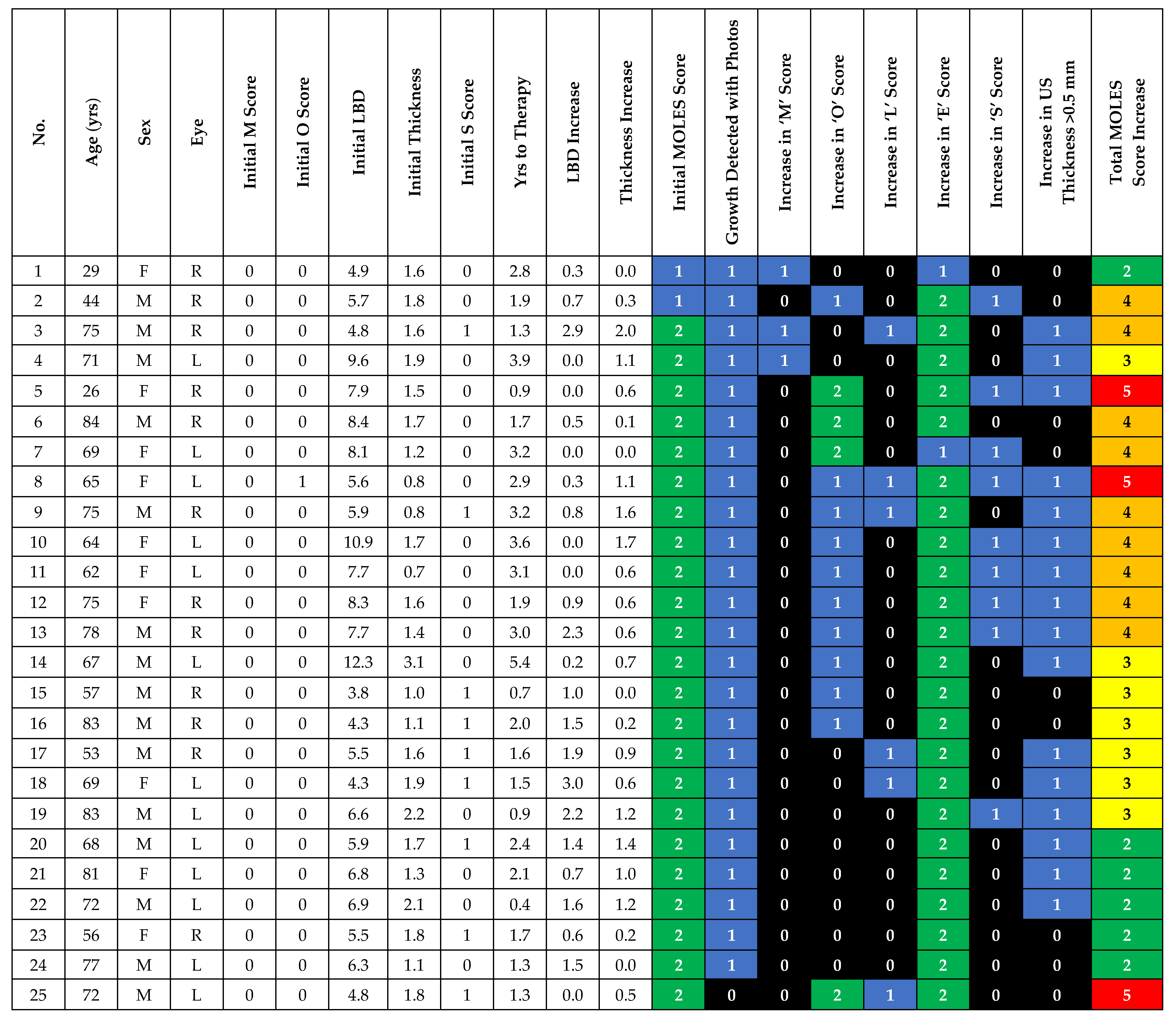
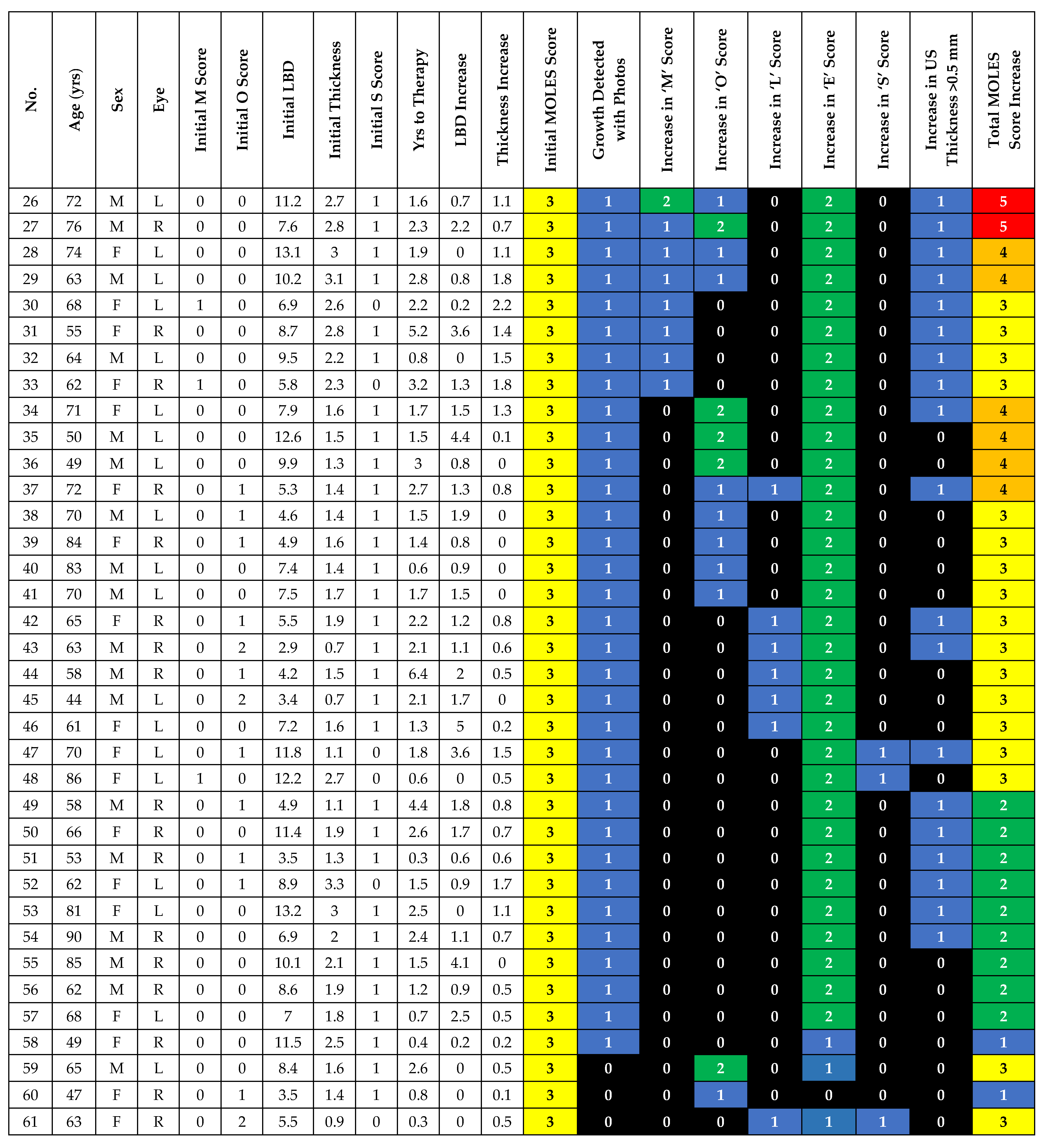
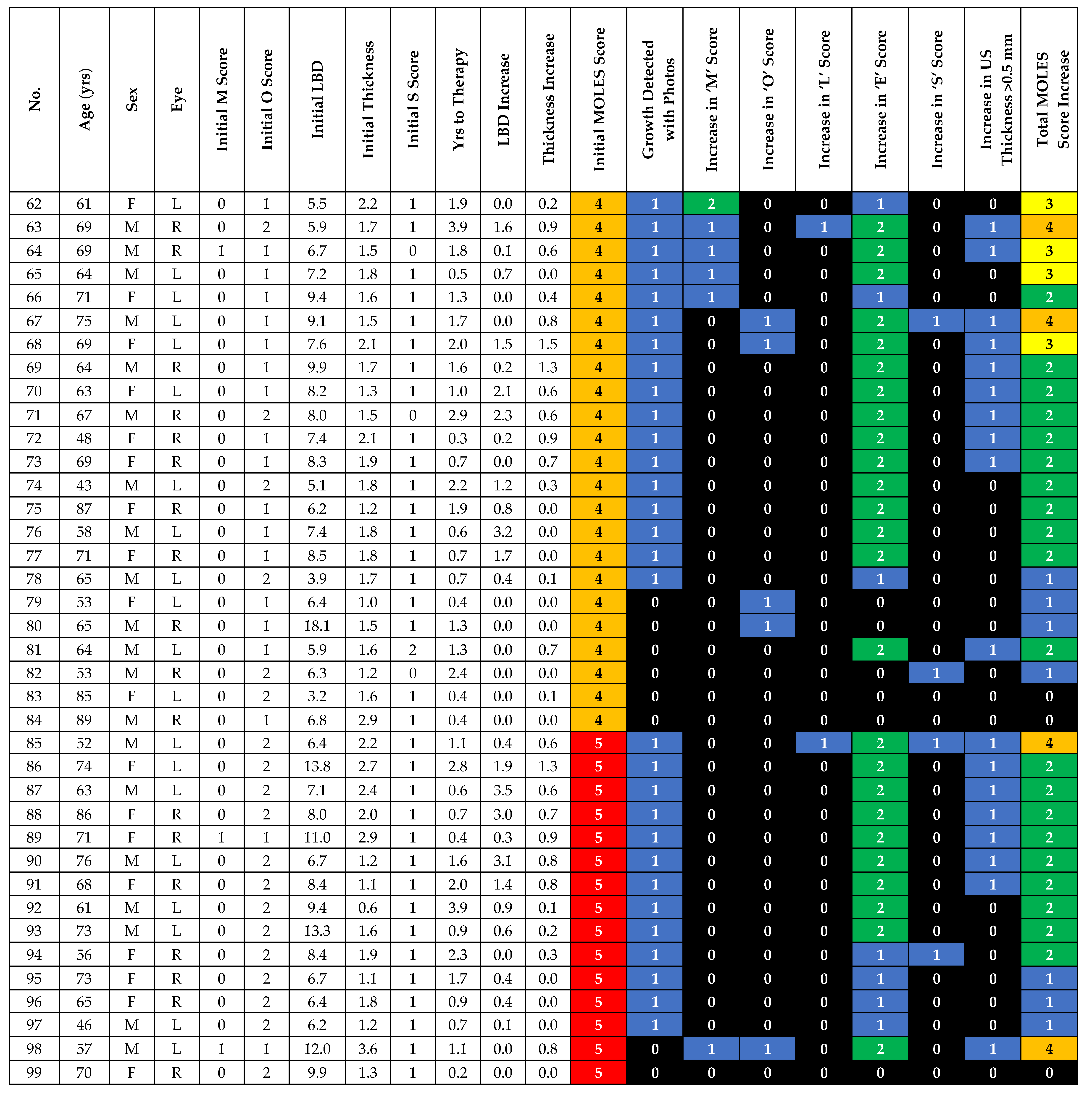

| Risk Factor | Severity | Score |
|---|---|---|
| Mushroom Shape | Absent | 0 |
| Unsure/Early growth through RPE | 1 | |
| Present | 2 | |
| Orange Pigment | Absent | 0 |
| Unsure/Trace (i.e., Dusting) | 1 | |
| Confluent clumps | 2 | |
| Large Size * | Thickness & Diameter | |
| Thickness < 1.0 mm (‘flat/minimal thickening’) and diameter < 3DD | 0 | |
| Thickness = 1.0–2.0 mm (‘subtle dome shape’) and/or diameter = 3–4DD | 1 | |
| Thickness > 2.0 mm (‘significant thickening’) and/or diameter > 4DD | 2 | |
| Enlargement | None (or lesion not documented or mentioned to patient previously) | 0 |
| Unsure (i.e. Poor image quality) | 1 | |
| Definite (confirmed with sequential imaging) | 2 | |
| Subretinal Fluid ** | Absent | 0 |
| Trace (if minimal and detected only with OCT) | 1 | |
| Definite (if seen without OCT) | 2 | |
| Total Score |
| MOLES Score | Suggested Management |
|---|---|
| 0 = Common Nevus | Monitoring in community with color photography every 1–2 years. |
| 1 = Low-Risk Nevus 2 = High-Risk Nevus | Non-urgent referral for specialist investigation comprising wide-field photography, autofluorescence imaging, optical coherence tomography and, in selected cases, ultrasonography. Subsequent surveillance to be undertaken at a specialist clinic or in the community according to risk of malignancy. |
| 3 = Probable Melanoma | Urgent referral to ophthalmologist with urgent onward referral to ocular oncologist if suspicion of malignancy is confirmed. |
| Patient Features | Sub-Category | n (%) | |
|---|---|---|---|
| Sex | Female | 46 (46.5) | |
| Male | 53 (53.5) | ||
| Age (Yrs) | <66 | 47 (47.5) | |
| >65 | 52 (52.5) | ||
| Eye | Left | 52 (52.5) | |
| Right | 47 (47.5) | ||
| Treatment | Laser | 2 (2.0) | |
| Plaque | 80 (80.8) | ||
| Proton beam | 17 (17.2) | ||
| Tumor Features | Initial (%) | Final (%) | |
| Mushroom Shape | Nil | 93 (93.9) | 80 (80.8) |
| Incipient | 6 (6.1) | 13 (13.1) | |
| Definite | 0 (0.0) | 6 (6.1) | |
| Orange Pigment | Nil | 47 (47.5) | 24 (24.2) |
| Dusting | 30 (30.3) | 34 (34.3) | |
| Confluent | 22 (22.2) | 41 (41.4) | |
| Largest Basal Tumor Diameter (mm) | <4.5 | 10 (10.1) | 5 (5.1) |
| 4.5–6.0 | 20 (20.2) | 10 (10.1) | |
| >6.0 | 69 (69.7) | 84 (84.8) | |
| Tumor Thickness (mm) | <1.0 | 0 (0.0) | 0 (0.0) |
| 1.0–2.0 | 74 (74.7) | 43 (43.4) | |
| >2.0 | 25 (25.3) | 56 (56.6) | |
| Tumor Size | Small | 2 (2.0) | 0 (0.0) |
| Borderline | 26 (26.3) | 12 (12.1) | |
| Large | 71 (71.7) | 87 (87.9) | |
| Enlargement | Nil | 99 (100) | 8 (8.1) |
| Possible | - | 14 (14.1) | |
| Definite | - | 77 (77.8) | |
| Subretinal Fluid | Nil | 25 (25.3) | 12 (12.1) |
| Minimal | 73 (73.7) | 83 (83.8) | |
| Significant | 1 (1.0) | 4 (4.0) | |
| LBD Increase | <0.6 mm | - | 40 (40.4) |
| >0.5 mm | - | 59 (59.6) | |
| Thickness Increase | <0.6 mm | - | 46 (46.5) |
| >0.5 mm | - | 53 (53.5) | |
| Duration of Monitoring (Yrs) | <1 | 28 (28.3) | |
| 1–2 | 37 (37.4) | ||
| >2 | 34 (34.3) | ||
| MOLES Score | 0 | 0 (0.0) | 0 (0.0) |
| 1 | 2 (2.0) | 0 (0.0) | |
| 2 | 23 (23.2) | 0 (0.0) | |
| 3 | 36 (36.4) | 2 (2.0) | |
| 4 | 23 (23.2) | 8 (8.1) | |
| 5 | 15 (15.2) | 24 (24.2) | |
| >5 | - | 65 (65.7) | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roelofs, K.A.; O’Day, R.; Al Harby, L.; Hay, G.; Arora, A.K.; Cohen, V.M.L.; Sagoo, M.S.; Damato, B.E. Detecting Progression of Melanocytic Choroidal Tumors by Sequential Imaging: Is Ultrasonography Necessary? Cancers 2020, 12, 1856. https://doi.org/10.3390/cancers12071856
Roelofs KA, O’Day R, Al Harby L, Hay G, Arora AK, Cohen VML, Sagoo MS, Damato BE. Detecting Progression of Melanocytic Choroidal Tumors by Sequential Imaging: Is Ultrasonography Necessary? Cancers. 2020; 12(7):1856. https://doi.org/10.3390/cancers12071856
Chicago/Turabian StyleRoelofs, Kelsey A., Roderick O’Day, Lamis Al Harby, Gordon Hay, Amit K. Arora, Victoria M. L. Cohen, Mandeep S. Sagoo, and Bertil E. Damato. 2020. "Detecting Progression of Melanocytic Choroidal Tumors by Sequential Imaging: Is Ultrasonography Necessary?" Cancers 12, no. 7: 1856. https://doi.org/10.3390/cancers12071856
APA StyleRoelofs, K. A., O’Day, R., Al Harby, L., Hay, G., Arora, A. K., Cohen, V. M. L., Sagoo, M. S., & Damato, B. E. (2020). Detecting Progression of Melanocytic Choroidal Tumors by Sequential Imaging: Is Ultrasonography Necessary? Cancers, 12(7), 1856. https://doi.org/10.3390/cancers12071856






