1. Introduction
Gastrointestinal stromal tumors (GISTs) are the most common mesenchymal malignancies of the gastrointestinal tract arising from the specialized cells in the bowel wall, the interstitial cells of Cajal (ICCs), or their stem cell-like precursors. Since GISTs are frequently driven by the auto-activated, mutant KIT receptor tyrosine kinase gene or by the platelet-derived growth factor receptor alpha (PDGFR-α) [
1,
2,
3], most of the tumors have a durable response to receptor tyrosine kinase inhibitor imatinib mesylate (Gleevec), which is currently considered as an effective drug for first-line therapy for GIST patients [
4,
5]. Unfortunately, despite the impressive response rates, more than 50% of patients with advanced and metastatic forms of the disease develop secondary resistance to imatinib (IM)-based therapy within 2 years after initiation of treatment [
6,
7]. To overcome secondary resistance to IM, more potent KIT inhibitors have been developed that are currently used as second- and third-line therapies for these patients. These include sunitinib malate (Sutent) [
8] and regorafenib (Stivarga) [
9], respectively. As KIT mutations are known as the most common mechanisms of secondary resistance to IM in GISTs, the alternative molecular mechanisms are also described and include hemi- or homozygous deletion of the wild-type KIT allele [
10], overexpression of focal adhesion kinase (FAK) [
11], amplification of the insulin-like growth factor receptor I (IGF-1R) [
12], the BRAF V600E mutation (5% of GISTs) [
13], and RTK “switch” (loss of c-KIT and gain of MET/AXL) [
14]. We found recently that secondary resistance to IM in GISTs might also be due to the activation of the FGF-signaling pathway [
15], whereas the inhibition of this pathway in IM-resistant GISTs effectively restores their sensitivity to IM both in vivo and in vitro [
16]. This was in agreement with previous findings illustrating that IM-induced long-term KIT inhibition causes a feedback activation of FGFR-mediated signaling in GIST, thereby suggesting that FGFR inhibition may improve the long-term efficacy of IM in GIST patients [
17]. Moreover, FGF-2 has been shown as a potent factor capable of desensitizing GIST to IM treatment, thereby providing insights into the protective role of FGF-signaling in IM-treated GISTs and illustrating the novel molecular mechanism of IM resistance for GIST patients lacking secondary resistance mutations in KIT [
18]. Based on these findings, a phase Ib study was performed to evaluate the toxicity and dosing schedule of a combination of IM and BGJ398, a well-known selective FGFR-inhibitor. The stable disease for more than 32 weeks was observed in ~25% of heavily pretreated patients with IM-refractory GISTs (3/12). Unfortunately, toxicity was encountered with the combination therapy indicated above, and the trial was terminated before the dosing schedule of the combination therapy was determined [
19].
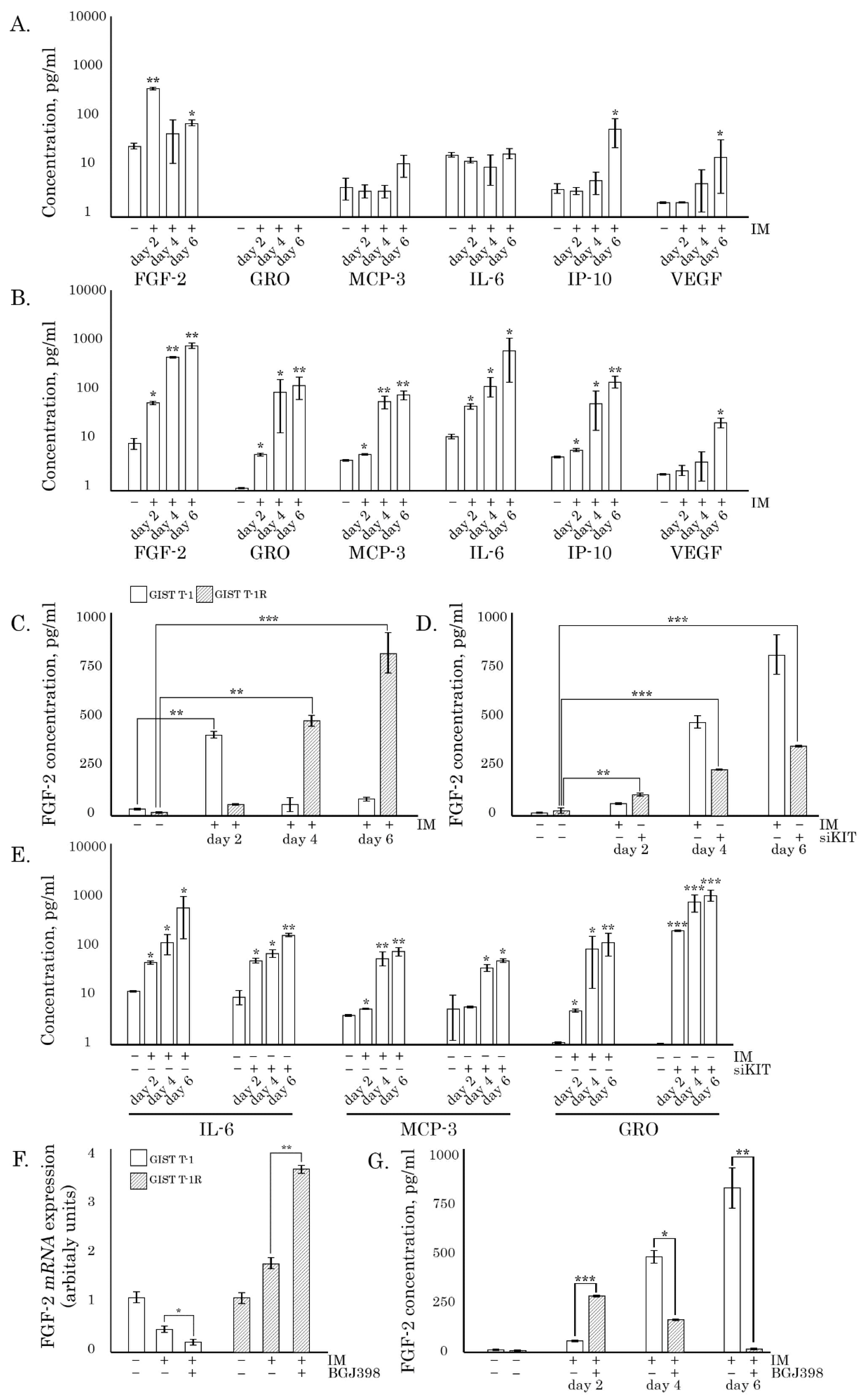
We show here for the first time that IM induces profound changes in the cellular secretome of IM-resistant GISTs, leading to the release of multiple chemokines, including FGF-2, GRO, and MCP 1-3. Most importantly, IM promotes the malignant behavior of IM-resistant GISTs by stimulating their migration, invasion, and colony formation of tumor cells via the activation of the FGF-2/FGFR autocrine loop. IM-induced activation of FGF-signaling in IM-resistant GISTs was revealed in vivo by utilizing xenograft models and tumor specimens from GIST patients who have progressed after initiation of IM-based therapy. Altogether, this data suggests that the continuation of IM-based therapy for GIST patients with secondary resistance to IM might have a negative impact on disease progression due to the activation of FGF-signaling, thereby providing a rationale to evaluate the effectiveness of FGFR-inhibitors to improve the therapeutic strategies for IM-resistant GISTs.
3. Discussion
Fibroblast growth factors (FGFs) and their receptors (FGFR1–4) are known to be involved in the regulation of a wide spectrum of physiologic cellular processes, such as differentiation, proliferation, migration, survival, and angiogenesis. Meanwhile, aberrant FGFR-signaling is also well-documented for the broad spectrum of human malignancies and plays a regulatory role in tumor development and progression. Most importantly, the activation of FGF-signaling in tumors might mediate their sensitivity to the current therapeutic regimens, thereby affecting the effectiveness of anticancer therapies and disease prognosis.
In addition to well-documented FGFR mutations and/or
FGFR amplifications leading to the activation of FGFR-signaling in multiple human malignancies, aberrant FGFR-signaling in cancer cells might be a result of the activation of autocrine/paracrine loops leading to increased secretion of FGFs by tumor or stromal cells and thereby promoting the survival and proliferation of cancer cells. The increased activities of autocrine and paracrine loops in human malignancies also play an important regulatory role in their pathogenesis due to the well-known abilities of FGFR ligands, especially FGF-2, to regulate angiogenesis and promote tumor growth, invasiveness, and disease progression. Moreover, in addition to the direct proangiogenic effects of FGFR-signaling, the activation of this pathway might have a strong impact on neoangiogenesis via the activation of VEGFR-signaling and thereby may synergize with VEGFR and platelet-derived growth factor receptor (PDGFR)-mediated pathways to promote tumor vascularization [
25,
26]. Activation of autocrine/paracrine FGF/FGFR-mediated loops was evidenced for multiple malignancies, including non–small cell lung carcinoma, hepatocellular carcinoma, and breast, prostate, and colorectal cancers [
27,
28,
29]. The increased activity of FGF-2/FGFR autocrine/paracrine loops in malignancies is also considered as a potent mechanism of tumor resistance to conventional therapies, including targeted therapies [
30,
31].
However, to date, a little is known about the role of autocrine/paracrine FGF-2/FGFR-mediated signaling in GIST biology and their sensitivity to targeted-based therapy. For example, Li F. and coauthors demonstrated an increased expression of FGF-2 and FGFR-1 in the majority of primary GIST samples, thus suggesting the activation of FGF-2-mediated autocrine and/or paracrine loops [
17]. Javidi-Sharifi N. and coauthors demonstrated that FGF-2 expression was increased in IM-resistant GIST cells, and KIT- and FGFR-3 inhibitors synergized to inhibit GISTs growth in vitro, thereby suggesting the crosstalk between KIT and FGFR3 in GISTs [
18]. Consistent with these findings, our present data illustrates the activation of the FGF2/FGFR2 autocrine pathway in IM-resistant GISTs induced by c-KIT inhibition (by imatinib and siRNA, as well). In particular, we show here for the first time that KIT inhibition in GISTs induced the profound changes in GIST secretomes, reflecting a massive secretion of the multiple chemokines (e.g., MCP-3, GRO, IP-10, FGF-2, and VEGF) known to be elevated in many types of human cancers, and promoting cancer cell invasion and migration. In particular, we observed that FGF-2, the FGFR1-4 ligand, was actively produced by IM- or
siKIT-treated GISTs (both of IM-naive and -resistant) in a time-dependent manner, thus suggesting the activation of the autocrine FGF-2/FGFR loop could serve as a compensatory mechanism of the c-KIT-signaling pathway inhibited in GISTs (
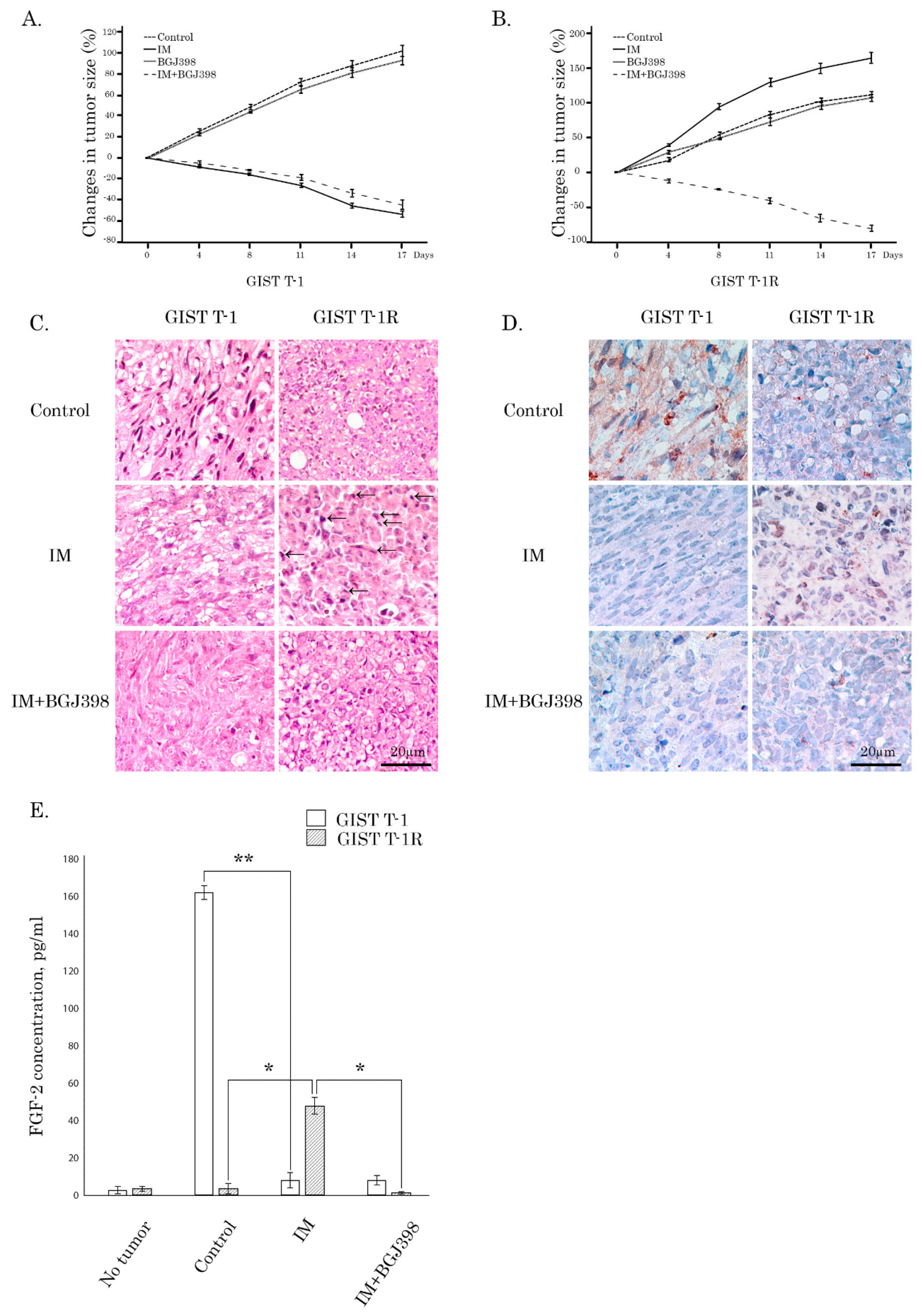
Figure 1D). Activation of the FGF2/FGFR autocrine loop in IM-treated GISTs was also revealed by in vivo studies illustrating nuclear localization of FGF-2 in GIST xenografts after IM treatment (
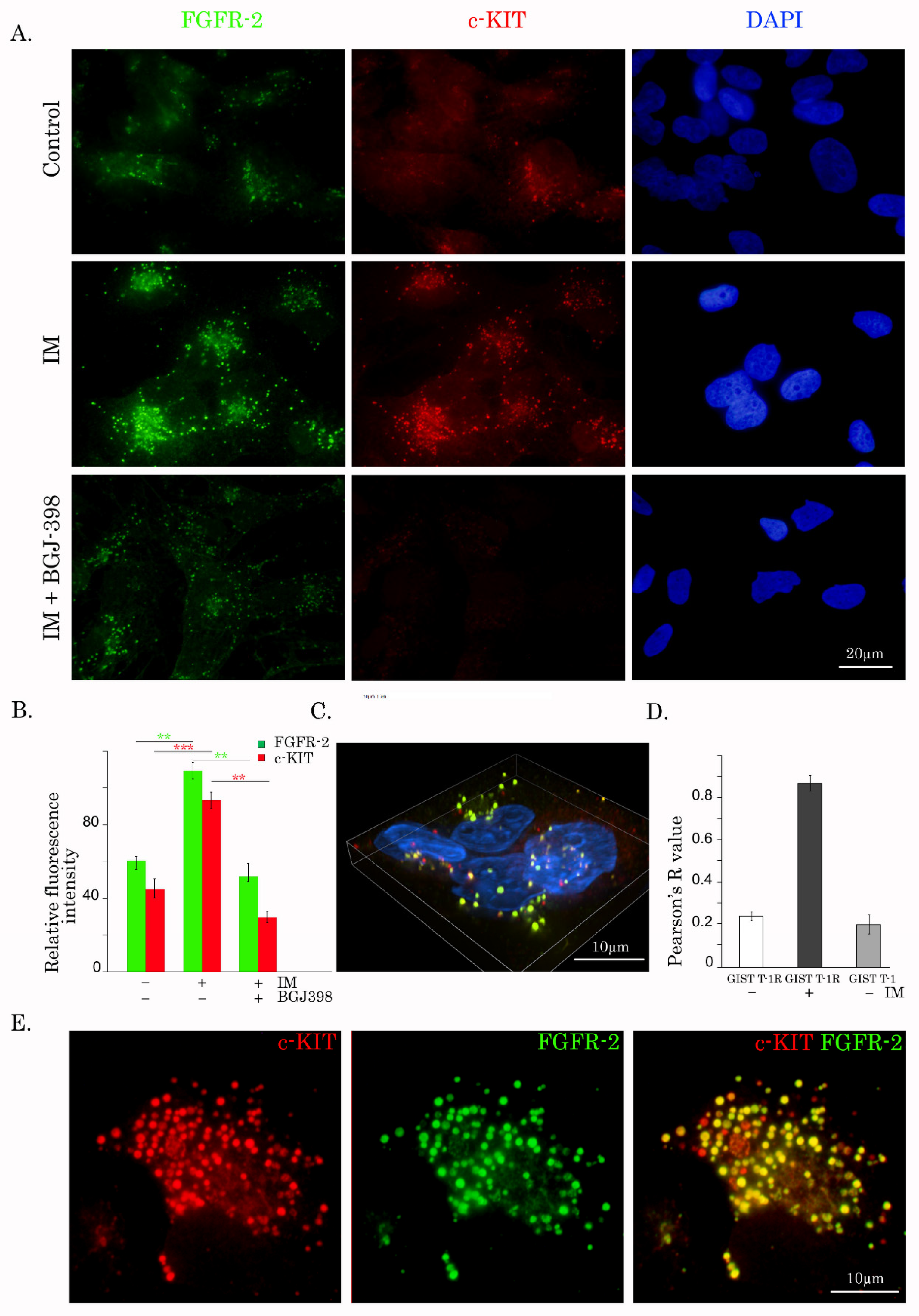
Figure 6D) and tumor specimens of GIST patients who received IM-based therapy (
Figure 7A). The molecular mechanisms involved in FGF-2 trafficking into the nucleus in IM-treated GISTs remain to be further elucidated and might include its association with the cytoplasmic microtubule-associated protein translokin, FGF-2-interacting factor (FIF). The first one is known to be important for low molecular weight (18-kDa) FGF-2, harboring a C-terminal nuclear localization sequence (NLS) [
32], whereas the second one regulates the nuclear trafficking of all the high molecular weight isoforms (22-, 22.5-, 24-, and 34-kDa) of FGF-2 containing N-terminal NLS [
33].
Besides active angiogenesis and increased growth kinetics in tumors exhibiting the activation of FGF-2/FGFR autocrine and/or paracrine loops, activated FGF-signaling was shown as a potent regulator of migration and invasion of cancer cells via diverse molecular mechanisms [
34,
35,
36,
37,
38]. Most of them reflect the paracrine loop-mediated mechanisms. For example, fibroblast cell surface-associated fibroblast growth factor (FGF)-2 was shown to induce the cell-contact-dependent migration and invasion of colorectal cancer cells via the activation of the FGF-2-FGFRs-SRC-αvβ5 integrin axis [
37], whereas the activation of FGF-2/FGFR-1 paracrine signaling in breast cancer cells induces the expression of the connective tissue growth factor (CTGF), thereby promoting the migration and invasion of tumor cells [
38].
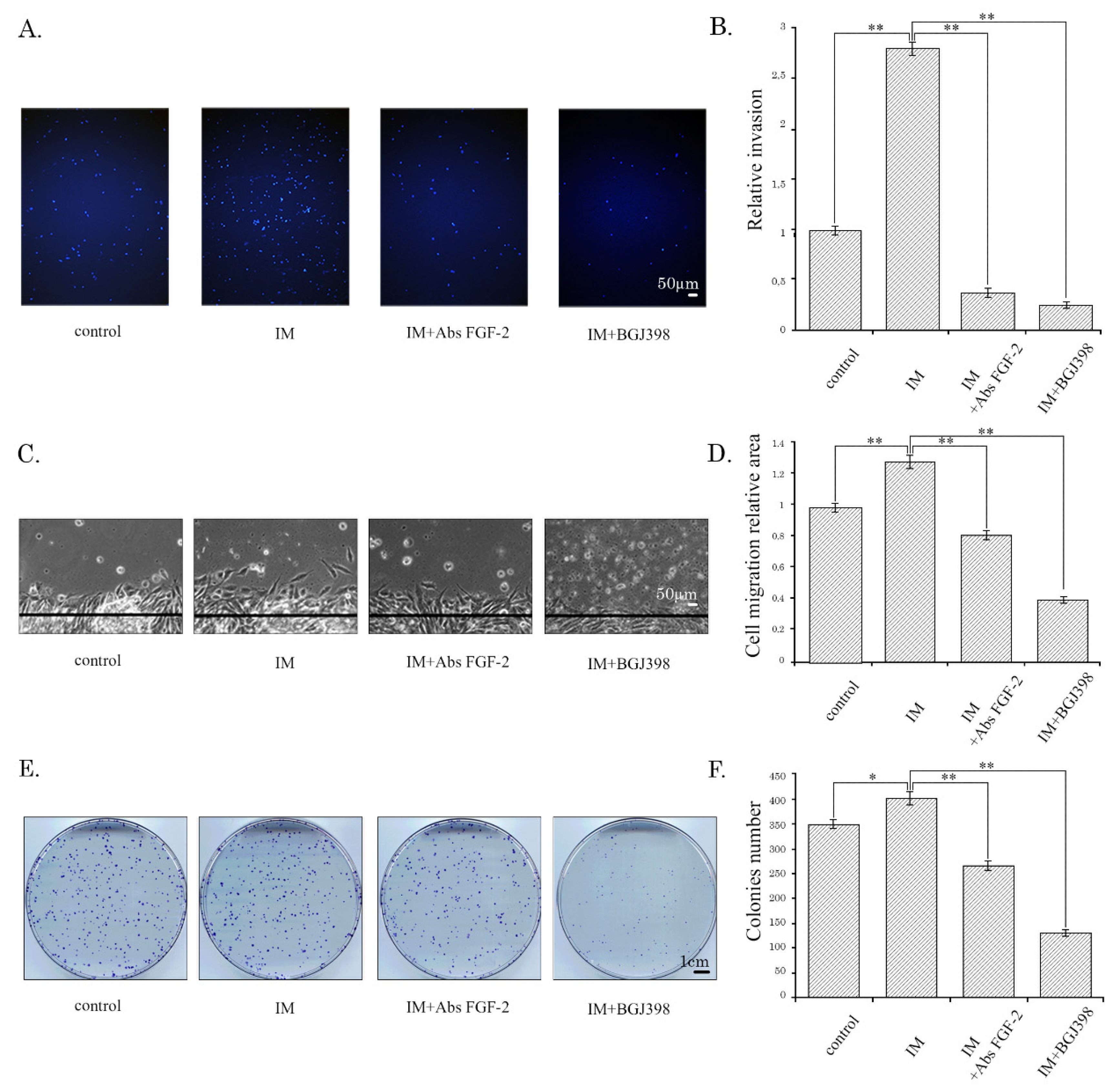
Consistent with these findings, we showed here for the first time that increased production of FGF-2 by IM-treated GIST cells reflecting the activation of the FGF-2/FGFR autocrine loop can serve as a mechanism maintaining the aggressive behavior of IM-resistant tumor cells through the facilitation of their migration and invasion. Interruption of the FGF-2/FGFR signaling via neutralizing anti-FGFR-2 Abs abolished this effect, thereby revealing that IM-induced activation of the FGF-2/FGFR autocrine loop is responsible for increased migration and invasion of IM-resistant GIST T-1R cells. Similarly, BGJ398, a selective FGFR inhibitor, abolished IM-induced increase of migration and invasion of IM-resistant GISTs (
Figure 2 and
Figure S3). Of note, we also observed the large-size spherical colonies in IM-treated GIST 430 cells (
Figure S3E,G), thereby suggesting that IM might potentiate the growth of the cells with stem-like cell (SC) properties. Again, this effect of IM was abolished by BGJ398 or anti-FGF-2 Abs (
Figure S3E–H), thereby illustrating the IM-induced activation of the FGF2/FGFR autocrine pathway is not restricted to one particular GIST cell line. To validate the SC-like phenotype in GIST 430 after IM treatment, further experiments are needed, including the cell culture in specific (e.g., non-adherent) culture conditions, by utilizing ultra-low attachment surfaces and examination of markers specific for the CSC/progenitor population, such as aldehyde dehydrogenase (
ALDH1A1) mRNA expression and ALDH activity and expression, as was shown before [
39].
Thus, IM activates the FGF-2/FGFR autocrine loop in various IM-resistant GIST cell lines and this mechanism might have a negative impact on the disease progression due to the activation of tumor cell migration, invasion, and enrichment of tumors with stem-like cell (SC) properties.
Of note, IM-induced activation of the FGF-2/FGFR loop in GIST might be a factor mediating tumor resistance to IM. Indeed, an exogenous FGF-2 rescued IM-naive GIST cells from IM-induced apoptotic cell death (
Figure 5E,F) and significantly (~4-fold) increased IC50 values of IM in IM-naïve GIST cells (
Figure 5B). The last one is consistent with previous findings illustrating a protective role of FGF-2 in IM-naive GIST cell lines (GIST T-1 and 882) treated with the low doses of IM [
18]. The anti-apoptotic activity of FGF-2 in GISTs might be due to the different molecular mechanisms. It was recently shown that the anti-apoptotic activity of FGF-1 and -2 is not solely due to the activation of FGFR-signaling (e.g., AKT- and MAPK-mediated pathways) and might be a result from the intracellular translocation of the ligands into the nucleus [
40]. Indeed, besides the activation of the tyrosine kinase receptors, secreted FGF-1 and FGF-2 can also translocate across the cellular membranes into the cytosol upon internalization via FGFR-dependent endocytosis, and this process is tightly regulated by several cytosolic proteins, including heat shock protein 90 (HSP90), PI3K- and p38 kinase [
36,
37,
38]. Nuclear import of FGFR ligands is more specific and depends on the interactions between FGF-1 and -2 with LRRC59 protein and translokin, respectively [
32,
41]. Despite the complexity of the mechanisms involved in FGF-1 and -2 internalization and intracellular trafficking, it is known that nuclear transportation of these ligands is frequently observed during various types of stress conditions, such as exposure to the toxic agents, oxidative stress, hyperosmolarity, and serum deprivation, thereby highlighting their protective role in the nucleus during the unfavorable conditions indicated above. Indeed, the nuclear translocation of FGF-1 was shown to promote cell survival via the downregulation of p53-dependent apoptosis [
40,
41,
42]. Similarly, the antiapoptotic activity of intracellular FGF-2 was shown to be mediated via its binding to apoptosis inhibitor 5 (API5) [
33]. Our data illustrates that FGF-2 is actively secreted by IM-treated and KIT-inhibited GISTs (
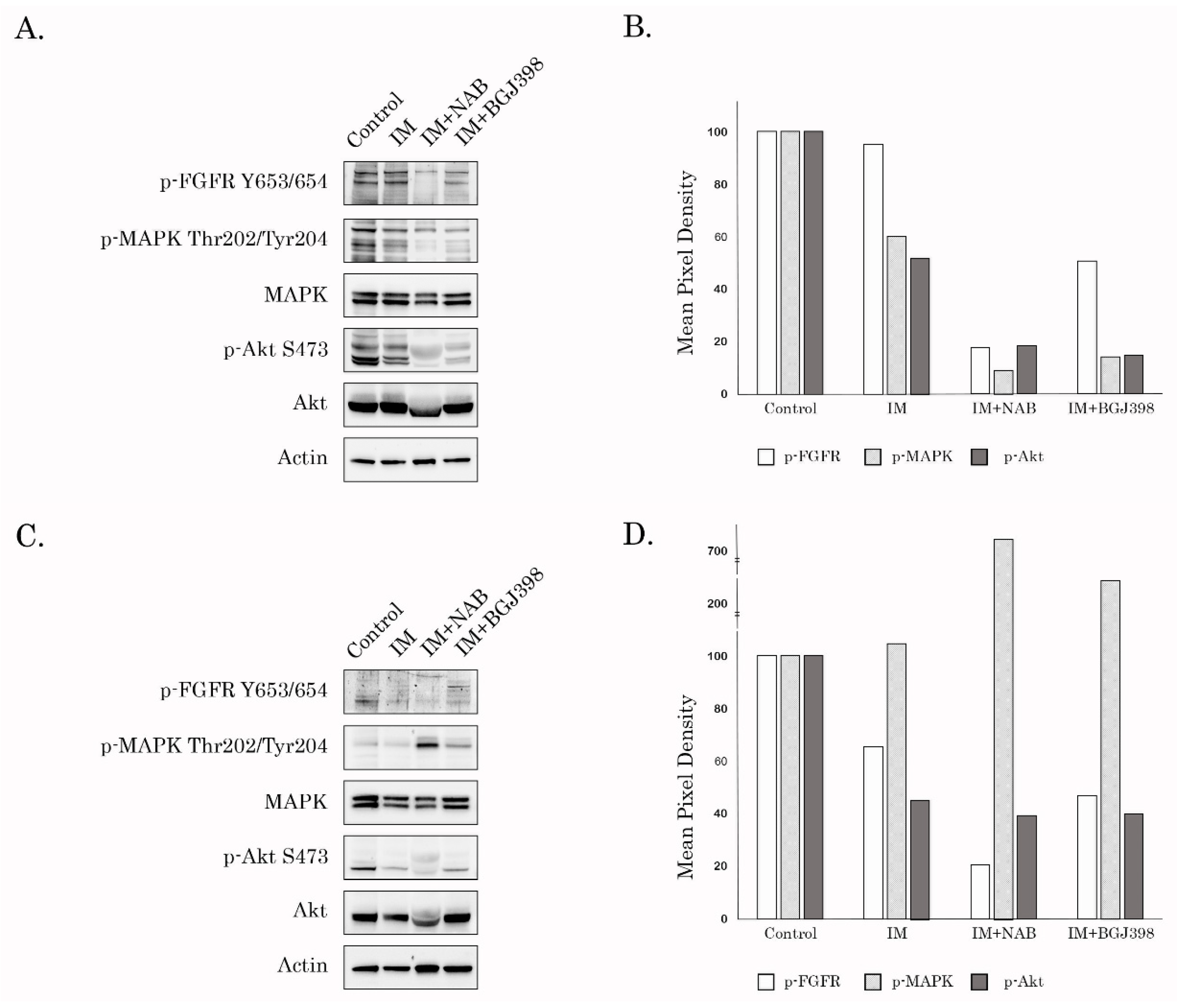
Figure 1D), activates FGF-signaling via phosphorylation of FGFR1 and -2 (
Figure 3) and translocates into the nucleus (
Figure 6D and
Figure 7A), thereby illustrating the variety of FGF-dependent mechanisms of GIST resistance to IM-induced apoptosis.
Taken together, IM-induced activation of FGF-signaling pathway in GISTs is in agreement with a paradigm indicating that FGFRs and their ligand, FGF-2, could work as a surrogate signaling pathway under the inhibition of RTK-signaling and mediate tumor resistance to RTK inhibitors (RTKi) and chemotherapeutic agents as well. For example, FGF-2-FGFR-1 activation through an autocrine loop was found as a mechanism of acquired resistance in non-small cell lung cancer (NSLC) to EGFR-tyrosine kinase inhibitor gefitinib [
30,
31]. Moreover, FGF-2 leaking out from gefitinib-treated NSLC cells was proposed as a survival factor for tumor cells that remained alive after the first exposure of EGFR-TKIs and further maintained their resistance to EGFR-TKIs [
42,
43]. FGF2-mediated tumor resistance to a broad spectrum of chemotherapeutic drugs with diverse structures and mechanisms of action is also well-documented. For example, Song S. et al. demonstrated that increased concentrations of bFGF in the conditioned medium of solid and metastatic tumors might mediate their resistance to certain chemotherapeutic agents—paclitaxel, doxorubicin, and 5-fluorouracil—whereas the inhibition of bFGF by monoclonal antibody abolished this effect [
44]. Similarly, the elevated intracellular level of bFGF in CLL was associated with their resistance to chemotherapy and correlated with an advanced stage of the disease [
45].
Most importantly, an increased FGF-2 level was found in plasma samples of patients affected by diverse malignancies, such as leukemia and lung and breast cancers, especially when metastases are present and therefore suggested increased FGF-2 levels in serum as a complement predictor of poor prognosis. For example, high serum levels of bFGF were found to be associated with poor outcomes in small cell lung cancer [
46] and non-Hodgkin’s lymphoma [
47]. In addition, high levels of FGF-2 in plasma were associated with large tumor size in head and neck cancers [
48] and with tumor growth kinetics in advanced colorectal cancer [
49]. Increased basic-FGF plasma concentrations are also correlated with high tumor grade and stage, metastatic spreading, and poor prognosis in kidney cancer patients, thereby illustrating that fibroblast growth factor receptor (FGFR) pathway is an attractive therapeutic target for patients with renal cell carcinoma [
50].
Consistent with this data, we observed the elevated FGF-2 levels in serum of IM-resistant GIST xenografts treated with IM when compared to non-treated mice bearing IM-resistant GISTs (
Figure 6E). Of note, FGF-2 levels in serum correlated with the xenograft volume and size, thereby suggesting a significant impact of autocrine FGF-2-mediated signaling for GIST behavior during IM treatment. Moreover, increased FGF-2 expression was also observed in IM-treated GIST xenografts, when compared to controls (
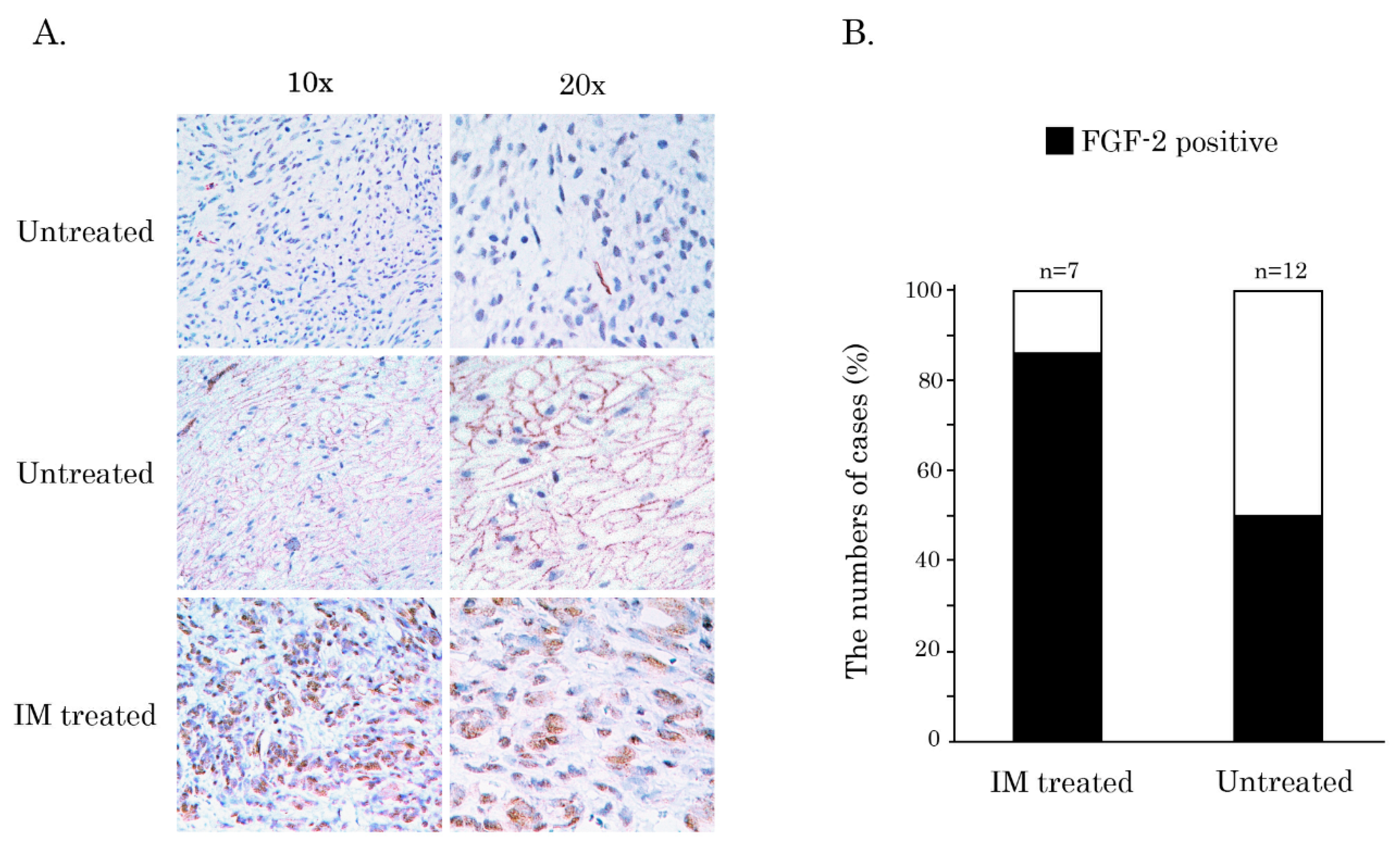
Figure 6D, middle panel). IHC-staining also revealed a substantial increase of FGF-2 expression in GIST human specimens after IM-based therapy when compared to non-treated patients with primary GISTs (
Figure 7A,B). Most importantly, this was also associated with the increased numbers of mitotic cells in GISTs after IM treatment (
Figure 6C, middle panel), thereby revealing IM’s ability to promote aggressive phenotypes in GISTs during long-term therapy.
Overall, our present data illustrate that continuation of IM-based therapy might have a negative impact on GISTs’ behavior after the tumors acquire resistance to IM. This is due to IM-induced activation of the autocrine FGF-2/FGFR loop, thereby facilitating disease progression via stimulation of GIST proliferation, migration, and invasion. This, in turn, provides a rationale to further evaluate the effectiveness of FGFR-inhibitors to improve the therapeutic strategies for GIST patients with an acquired (i.e., secondary) resistance to IM.
4. Materials and Methods
4.1. Chemical Compounds
Imatinib mesylate (IM) and BGJ398 were purchased from SelleckChem (Houston, TX, USA).
4.2. Antibodies
Primary antibodies used for immunoblotting and immunofluorescence were as follows: phospho-MAPK (Erk1/2) Thr202/Tyr204 (#4370S), MAPK (Erk1/2) (#4696S), phospho-AKT S473 (#4060P), AKT (#4691p), phospho-FGFR Y653/654 (#3476S), FGFR1 (#9740S), FGFR2 (#23328S) phospho-KIT Y719 (#3391S) (Cell Signaling, Danvers, MA, USA), c-KIT (#A4502, Dako, Carpinteria, CA, USA), beta-actin (A00730-200, GenScript, Piscataway, NJ, USA). HRP-conjugated secondary antibodies used for Western blotting were from Santa Cruz (Santa Cruz Biotechnology, Santa Cruz, CA, USA). Primary antibodies used for IHC-staining were as follows: c-KIT (#14-1172-82, Thermo Fisher Scientific, Waltham, MA, USA), DOG-1 (#244R, Cell Marque, Rocklin, CA, USA), a cleaved form of caspase-3 (#9662S, Cell Signaling, Danvers, MA, USA) and FGF-2 (sc-365106, Santa Cruz Biotechnology, Santa Cruz, CA, USA). Neutralizing antibody against bFGF (Anti-FGF2/basic FGF #05-117) and human recombinant FGF-2/basic (FGF2 #01-106) were obtained from Merck KGaA (Darmstadt, Germany).
4.3. Cell Lines and Culture Conditions
The IM-sensitive GIST T-1 cell line was established from human metastatic pleural tumors arising from stomach GIST and exhibited a heterozygous 57-base pair deletion (V570-Y578) in
KIT exon 11 [
20]. The IM-resistant GIST T-1R subline was generated in our laboratory, as shown elsewhere [
15]. IM-resistant GIST 430 cell line was established from human GIST that acquired clinical resistance to IM-based therapy. This GIST cell line exhibited a heterozygous primary
KIT exon 11 deletion (V560_L576del) and a secondary
KIT exon 13 point mutation (V654A) [
21]. GIST cell lines indicated above were cultured in a humidified atmosphere of 5% CO
2 at 37 °C (LamSystems, Russia), as previously described [
16].
4.4. TUNEL Assay
To evaluate apoptotic cell death in GIST cells, the terminal deoxynucleotidyl transferase nick end labeling (TUNEL) assay was performed by using a commercially available in situ apoptosis detection kit (DeadEnd™ Fluorometric TUNEL System, Madison, WI, USA). TUNEL-positive cells were counted by using a ×40 objective on an Olympus BX63 fluorescence microscope.
4.5. Cellular Survival MTS-Based Assay
IM-naïve and -resistant GIST cell lines indicated above were plated on 96-well flat-bottomed plates (Corning Inc., Corning, NY, USA) and allowed to attach and grow for at least 24 h before treatment with RTK inhibitors (e.g., IM and BGJ398) that were introduced into the cell culture for 48–72 h with indicated concentrations alone or in combination with each other. For specific experiments, cells were treated with RTKis in the presence or absence of FGF-2. To calculate the IC50 values of the RTKi, MTS reagent (Promega, Madison, WI, USA) was introduced into the cell culture for at least 1 h to assess the live cell numbers. The cellular viability was assayed at 492 nm on a MultiScan FC plate reader (Thermo Fisher Scientific, Waltham, MA, USA). Resulting IC50 values were defined as the compound concentration required to inhibit cellular growth by 50%. This data was normalized to the DMSO-treated (i.e., control) cells.
4.6. Real-Time Monitoring of Cell Proliferation
The growth curves of GIST T1 cells cultured in the presence of IM, FGF2 alone, or in combination were analyzed by using the iCELLigence system (ACEA Biosciences, San Diego, CA, USA). For this purpose, tumor cells were plated on the electronic microtiter plates (E-Plate; Roche Diagnostics, GmbH, Mannheim, Germany) for at least 24 h and further treated with IM (0.25 µM), FGF-2 (100 ng/mL) alone or in combination for 72 h. The cells treated with solvent (DMSO) were used as a negative control. The cell index (CI) was measured every 30 min until the end time-point of the experiment (72 h). Normalized cell index (NCI) values were also analyzed by using RTCA software (Roche Diagnostics, GmbH, Mannheim, Germany).
4.7. Western Blotting and Coimmunoprecipitation (Co-IP)
To examine the protein expression in GIST cells, whole-cell lysates (WCL) were prepared by scraping the cells growing as monolayer into RIPA buffer (25 mM Tris-HCl pH 7.6, 150 mM NaCl, 5 mM EDTA, 1% NP-40, 1% sodium deoxycholate, 0.1% SDS), supplemented with the cocktail of protease and phosphatase inhibitors. The cellular lysates were further incubated for 1 h at 4 °C and clarified by centrifugation for 30 min at 13,000 rpm at 4 °C. The protein concentrations in WCL were calculated by the Bradford assay. The protein samples (30 μg) were loaded on the 4–12% Bis-Tris or 3–8% Tris-acetate NuPAGE gels (Invitrogen, Carlsbad, CA, USA), transferred to a nitrocellulose membrane (Bio-Rad, Hercules, CA, USA), probed with a specific antibody, and visualized by enhanced chemiluminescence (Western Lightning Plus-ECL reagent, Perkin Elmer, Waltham, MA, USA). Densitometric analysis of Western blotting images was performed by using NIH ImageJ software (Bethesda, MD, USA).
For Co-IP, GISTs were lysed by TEB buffer (50 mM Tris-HCl pH 7.5, 150 mM NaCl, 1% NP-40, 10% glycerol) and supplemented with the cocktail of protease and phosphatase inhibitors. The lysates were clarified by centrifugation, as shown above, and cultured with the specific Abs overnight at 4 °C in a rotating device. The next day, the samples were treated with protein A/G Sepharose beads (Santa Cruz Biotechnology, Santa Cruz, CA, USA) for 1 h (rotating device at 4 °C). Finally, the beads were washed with TEB buffer and the 1X samples were subjected for Western blotting, as indicated above.
4.8. Immunofluorescence Staining
Cells were seeded on glass coverslips pre-coated with poly-L-lysine (Sigma-Aldrich, St. Louis, MO, USA) and allowed to attach for 48 h before treatment. For immunofluorescence staining, the cells were washed with PBS, fixed in 2% paraformaldehyde for 10 min at room temperature, washed with PBS again, and additionally fixed with ice-cold methanol for 10 min. Methanol was removed with PBS washing (5 times for 5 min). Alternatively, washed cells were fixed in 4% paraformaldehyde in PBS for 30 min at 4 °C and permeabilized with 0.5% Triton X-100 for 5 min. The cells were blocked for 30 min by using 10% normal goat serum and further incubated with primary antibodies for overnight at 4 °C. The next day the cells were washed with PBS, incubated with Alexa Fluor 488 or TexRed-conjugated secondary antibodies (Invitrogen, Carlsbad, CA, USA) for 30 min at room temperature in the dark. Finally, a DAPI stain (Sigma-Aldrich, St. Louis, MO, USA) was used for 1 min to outline the nuclei; the coverslips were washed with PBS twice and mounted on glass slides. The cells were visualized on an Olympus BX63 fluorescence microscope. Images were captured using a Spot advanced imaging system or a Nikon N-SIM confocal system.
4.9. D-IF Microscopy (Wide-Field with Deconvolution)
Glass coverslips with cells after IF staining were mounted on PVA (polyvinyl alcohol, Sigma-Aldrich, St. Louis, MO, USA). Samples were examined using N-SIM microscopy (Nikon) with an EMCCD camera (iXon 897, AndorT). Then, 488 and 561 nm laser excitation and 100x/1.49 NA oil immersion objectives were used. Exposure conditions were adjusted to get a typical yield of about 5000 max counts (16-bit raw image) for minimal bleaching. Images have been captured as serial optical sections of the same cell in z-axis with 0.12 μm z-steps. For serial images in wide-field mode, the AutoQuant blind deconvolution algorithm was used. Image analysis, visualization, archiving, and volume views were performed using NIS-Elements 4.2 microscope imaging software (Nikon).
4.10. Colocalization Analysis
Co-localization of FGFR2 and c-KIT was performed on thresholded super-resolution images to exclude false-positives of the background generated by SIM reconstruction algorithms. Co-localization was analyzed by the Coloc 2 plugin included in the FIJI package, run on ROIs containing prominent features. Costes’s algorithm was applied for automatic threshold detection [
51], and Pearson coefficients were calculated for the above-threshold pixels.
Co-localization analysis for FGFR1 and c-KIT was performed in FIJI Software (Laboratory for Optical and Computational Instrumentation, University of Wisconsin, Madison, WI, USA). The Parallel Spectral Deconvolution plugin was applied to restore the contrast of the images, and the degree of co-localization between FGFR1 and c-KIT was measured by using the Coloc2 plugin. Spearman’s rank correlation values of at least 40 cells per each experimental condition were calculated, and their means were compared by using the analysis of variance (ANOVA) with subsequent pairwise comparisons (
t-test with Bonferroni adjustment) in R software (R Foundation for Statistical Computing, Vienna, Austria; URL
https://www.R-project.org/).
4.11. Wound-Healing Assay (Scratch Assay)
Cells were pre-cultured with DMSO (control), and IM (1 µM) alone or in the presence of anti-FGF-2 mAbs (5 mg/mL) or BGJ398 (1 µM) for 72 h and further seeded in 6-well plates and the tip of a 200 μL micropipette was used to make a scratch on a confluent monolayer of cells to create a wound. The detached cells were rinsed with PBS and then DMEM-Hi medium was added. The wound area was photographed by an AxioVert 200 microscope (Carl Zeiss, Germany) at 10x objective magnification, an AxioCam MRc camera (Carl Zeiss, Germany) and AxioVision 4.6 software (Carl Zeiss, Germany). The wound closure areas were calculated at 12 h after incubation to show the closure of the wounds. Three independent experiments were photographed and quantified under a microscope. The data were normalized to the wound areas of the control (non-treated) cells.
4.12. Transwell Invasion Assay
The cells were pretreated with IM alone and in the presence of anti-FGF-2 or BGJ398 for 72 h, as shown for the wound-healing assay. The inserts containing 8-μm pores in 12-well Transwell plates (Greiner Bio-One, Austria) were coated with Matrigel (Corning Inc., Corning, NY, USA) and hardened for 24 h. Cells (5 × 105) were placed in the upper inserts coated with Matrigel. Culture medium supplemented with 15% FBS was placed into the lower wells, whereas upper wells did not contain the growth factors (FBS—0%). After 24 h, the cells that migrated through the pores of inserts were fixed by 70% ethanol, stained with DAPI (Sigma-Aldrich, St. Louis, MO, USA), and counted by fluorescence microscopy Axiovert 200 (Carl Zeiss, Germany). Cells on the upper side of the membrane were carefully removed with cotton swabs. The numbers of invaded cells per field of view treated with IM alone or in presence of BGJ398 or anti-FGF-2 Abs were calculated. At least 9 fields in each case were counted. The experiments were performed in triplicate.
4.13. Colony Formation Assay
The cells were pretreated with IM alone and in the presence of anti-FGF-2 or BGJ398 for 72 h, as shown before. The cells were trypsinized, washed with PBS twice, and seeded for colony formation (approximately 500 cells for p100 culture dish) in complete DMEM-Hi medium with 10% FBS (Thermo Fisher Scientific, Waltham, MA, USA). The colonies were fixed by 70% ethanol, stained with Giemsa stain, and counted using Colony V1.1 software (Fujifilm, Japan) after 10 days of incubation. The experiments were performed in triplicate.
4.14. KIT/FGFR-2 Silencing Using siRNA
For FGFR2 and KIT knockdown experiments, On-Target Plus Smartpool siRNAs (Dharmacon, Lafayette, CO, USA) targeting FGFR-2 (# L-003132-00-0005) or KIT (# L-003150-00-0005) were diluted in Opti-MEM (Thermo Fisher Scientific, Waltham, MA, USA) and complexed with RNAimax (Invitrogen, Carlsbad, CA, USA) to a final concentration of 40 nM oligonucleotides. The oligomer–RNAimax complex was added to the culture medium, and after 48–72 h, the knockdown was validated by immunoblotting.
4.15. RNA Extraction and Real-Time Quantitative PCR
Total RNA was extracted from GISTs and converted into cDNA, as previously described [
15]. Briefly, 1 µL template cDNA was utilized for a real-time qPCR reaction with 5× qPCRmix-HS SYBR (PB025, Evrogen, Moscow, Russia) and forward and reverse primers (10 mM of each) for experimental or control genes. Real-time qPCR was performed by using the CFX96 Real-Time detection system (Bio-Rad, Hercules, CA, USA) according to the manufacturer’s protocol. The levels of each mRNA were normalized relative to GAPDH.
4.16. Enzyme-Linked Immunosorbent Assay
The levels of FGF2 in the serum of mice bearing IM-naive and -resistant GIST xenografts were measured using a human FGF2 ELISA Kit (R&D Systems, Tustin, CA, USA), according to the manufacturer’s protocol. The plates were read at 405 nm on a MultiScan FC plate reader (Thermo Fisher Scientific, Waltham, MA, USA).
4.17. Multiplex Analysis of Cytokines
A 41-multiplex analysis of an array of granules (MILLIPLEX MAP human cytokine/chemokine magnetic bead panel, Merck, Kenilworth, NJ, USA) was used to examine the levels of cytokines/chemokines in GIST supernatants. A MAGPIX® reader (Bio-Rad, Hercules, CA, USA) was used with xPONENT software (Luminex Corp., Austin, Texas, USA). The Luminex MAGPIX® instrument was previously calibrated with the MAGPIX® Calibration Kit (EMD Millipore, Merck, Kenilworth, NJ, USA). The performance of the assay was verified with the MAGPIX® Performance Verification Kit (EMD Millipore, Merck, Kenilworth, NJ, USA). MILLIPLEX® analytes were used for counting the results and optimization of the performance of the MAGPIX reader.
4.18. Tumor Samples for KIT Mutational Analysis and Tissue Microarrays
A total of 30 patients diagnosed with GIST were enrolled in the present study, including 28 localized tumors from patients treated with primary complete surgery. The protocols for tumor sample collection and clinical record review were approved by the Ethics Committee of the Kazan State Medical University (IRB no. 98-0352B), and the patients provided informed written consent for the use of their tissues and clinical data in research. All personal information was removed. The slides were stained for hematoxylin/eosin and reviewed to confirm the diagnosis of GISTs; CD117 and DOG-1 expression were also evaluated by immunohistochemistry (IHC)-staining. In addition, the number of mitotic cells was calculated in 50 high-power fields (HPFs). Clinical risk score from primary GISTs was calculated according to the NIH GIST Consensus Criteria developed by Fletcher et al. [
52] and classification of the risk categories proposed by the Armed Forces Institute of Pathology (AFIP) [
53]. GIST specimens were examined for hot-spot mutation sites of
KIT (exons 9, 11, 13, and 17). For this purpose, DNA was extracted from formalin-fixed, paraffin-embedded (FFPE) histological sections that were previously verified by pathologists (S.P. and E.M.). Tumor tissues were cut from the section and incubated in the extraction buffer (50 mmol/L KCl, 10 mmol/L Tris (pH8.3), 2.5 mmol/L MgCl
2) supplemented with 60 μg/mL proteinase K for overnight at 55 °C. Proteinase K was inactivated by 10 min incubation at 95 °C. The lysates were centrifuged for 5 min and supernatants were used for PCR. Amplification of
c-KIT was performed by PCR using specific oligonucleotide primers for exons 9, 11, 13, and 17 (
Table S2) and a DNA Technology Termocycler, RF. PCR was carried out in 50 μL total reaction volume, including initial DNA denaturation at 94 °C for 15 min, 35 cycles each at 94 °C for 30 s, primer annealing at the temperature indicated (
Table S3) for 30 s, elongation at 72 °C for 15 s, followed by one cycle at 72 °C for 2 min. PCR products were visualized by electrophoresis in 2% agarose, cut from the gel and purified by Wizard
® SV Gel and PCR Clean-Up System (Promega, Madison, WI, USA). Direct sequencing was performed by Applied Biosystems DNA analyzer with the same primers shown in the
Table S2 and ABI PRISM
® BigDye™ Terminator v. 3.1 and an Applied Biosystems 3730 DNA Analyzer Sequencing ready reaction kit (Applied Biosystems, Foster City, CA, USA), according to the manufacturer’s protocol. DNA sequence alignment was performed with the NCBI Reference Sequence Database, employing online Alignment Search Tool BLAST [
54] and Chromas SeqMan NGen version 12.0/Chromas (Technelysium Pty Ltd., South Brisbane, Australia) for multiple sequence alignment.
Formalin-fixed, paraffin-embedded tissue samples of representative tumor regions were used for the tissue microarray analysis (TMA). For this, the tissue cylinders (1.0 mm) were punched out from representative areas of each tissue block and transferred into a recipient paraffin block by using a manual tissue arrayer. Paraffin-embedded tissue sections were deparaffinized and rehydrated. Heat-induced epitope retrieval was applied before the staining with a primary antibody recognizing FGF-2, c-KIT, and DOG-1. Next, tissue microarrays were followed by a specific secondary antibody using the DAB Map detection kit (Ventana Medical Systems, Tucson, AR, USA). Tissue sections were also counterstained with hematoxylin and analyzed by light microscopy (Leica, Wetzlar, Germany). As mentioned before, all stained specimens were analyzed by 2 independent pathologists, S.P. and E.M.
4.19. GIST Xenograft Models
Subcutaneous tumor xenografts were established by s.c. inoculation of GIST cells (100 μL of 1 × 107 cells/mL) into the flank areas of 5–8-week-old female nu/nu mice. The animal protocols were approved by the N.N. Blokhin National Medical Research Center of Oncology committee (des. 2019-1. from 15.01.2019) for Ethics of Animal Experimentation, and all the experiments were conducted in accordance with the Decision No. 81 of the Council of the Eurasian Economic Commission dated November 3, 2016 “On approval of Rules of good laboratory practice of the Eurasian Economic Union in the sphere of circulation of medicines” and Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes.
And all the experiments were conducted in accordance with the Guidelines for Animal Experiments in N.N. Blokhin National Medical Research Center of Oncology. GIST xenografts were allowed to reach an appropriate volume (~200 mm
3) before the animals were randomized into the treatment groups (
n = 4). Mice were orally administered either 50 μL of a vehicle (negative control), IM (50 mg/kg), BGJ398 (20 mg/kg), or a combination of the drugs indicated above. The changes in tumor volume, weight, and general health were recorded every second day. After the experiments reached to its end-point, the animals were sacrificed, tumors were excised and subjected to a histopathologic examination by 2 independent pathologists, S.P. and E.M. Formalin-fixed, paraffin-embedded (FFPE) tissues were sectioned at 4 μM for hematoxylin and eosin (H&E) stain and were also subjected to IHC-staining for FGF-2 expression. Histopathologic grading of the response to the treatment was also assessed by calculating the areas of necrosis, myxoid degeneration, or fibrosis, as previously described [
41]. The images were captured using ScanScope XT (Aperio technologies inc., Vista, CA, USA).
4.20. Statistics
All the experiments were repeated a minimum of 3 times. The results are presented as the mean ± standard deviation (SD) for each group. Difference was considered significant at
p < 0.05. The distribution of patients at risk of recurrence of GISTs was carried out according to the National Institute of Health (NIH) consensus criteria [
52] and the Armed Forces Institute of Pathology (AFIP) criteria [
53]. The relationship between FGF-2 expression and low/high risk of GIST patients was evaluated by drawing the receiver operating characteristic (AUROC) curve. The association between FGF-2 expression and patient’s groups was evaluated using the chi-square test or the Mann-Whitney U test.