Predicting Response to Neoadjuvant Therapy in Colorectal Cancer Patients the Role of Messenger-and Micro-RNA Profiling
Abstract
1. Introduction
2. Methods
2.1. Protocol and Registration
2.2. Literature Search
2.3. Inclusion and Exclusion Criteria
2.4. Identification and Selection of Studies
2.5. Data Extraction and Synthesis
3. Results
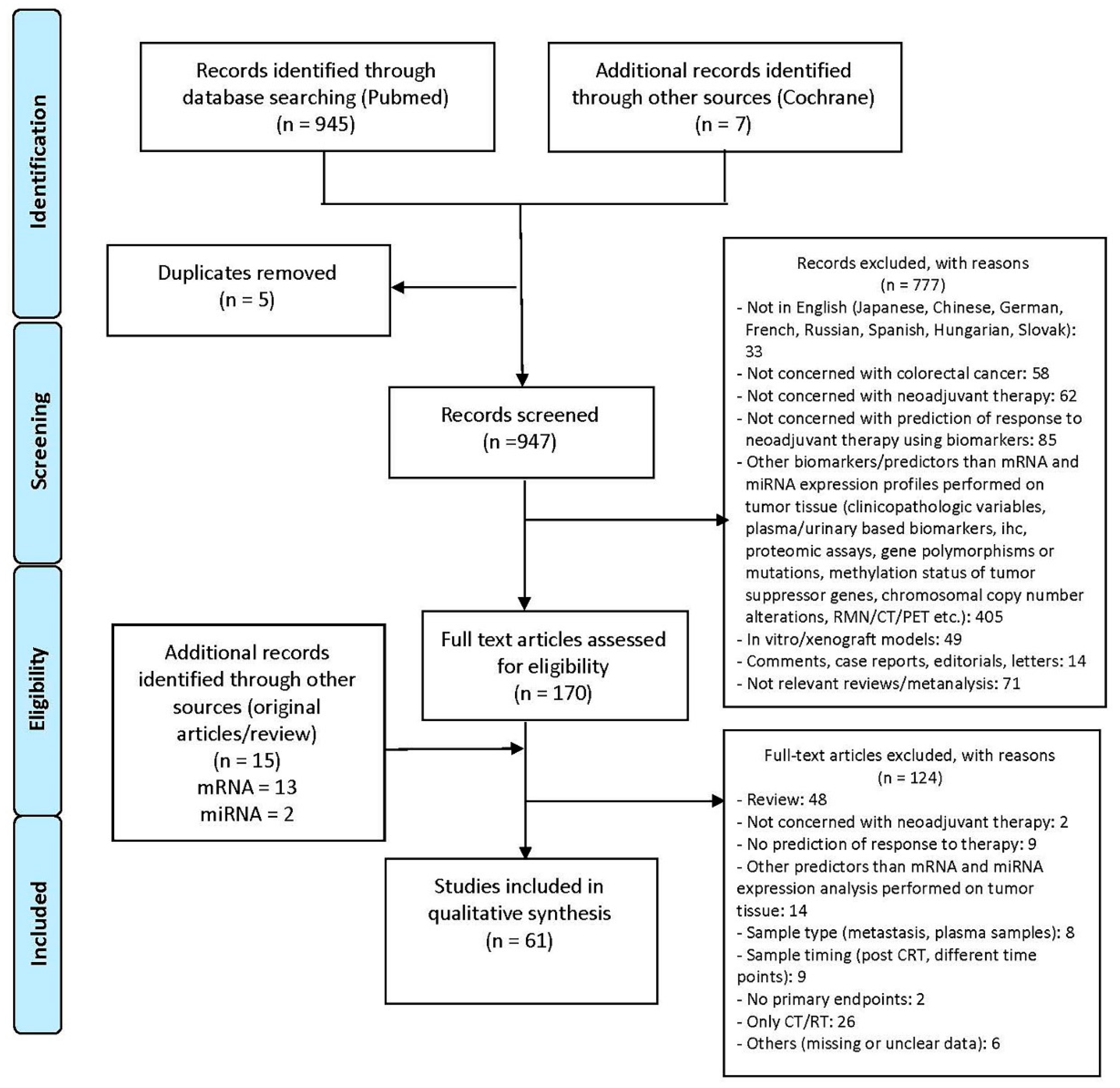
3.1. Literature Search
3.2. Analysis of mRNA Expression Data
3.2.1. Genes Modified in Two or More Studies
- (1)
- (2)
- (3)
- (4)
- (ITPK1 encodes the protein Inositol-Tetrakiphosphate 1-Kinase, an enzyme regulating the synthesis of inositol tetraphosphate and downstream products. Inositol metabolism plays a role in the development of the neural tube and maintenance of histone gene-suppression function. Its differentially relative expression (R versus NR) was analysed in three studies. In two of them, it was increased in nCRT responders [23,24]. In one study, it was decreased, but only when associated with downsizing [25].
- (5)
- (6)
- LGR5 encodes Leucine Rich Repeat Containing G Protein-Coupled Receptor 5, a receptor involved in the Wnt signalling pathway; it also plays a role in the formation and maintenance of adult intestinal stem cells during postembryonic development. Associated diseases include colon adenoma. Its differentially relative expression (R versus NR) was analysed in two studies. In both, it was decreased in nCRT responders [22,27].
- (7)
- MT-ND4 encodes Mitochondrially Encoded NADH Dehydrogenase 4 protein, involved in many pathways, such as respiratory electron transport, ATP synthesis, heat production by uncoupling proteins and GABAergic synapse. Its differentially relative expression (R versus NR) was analysed in two studies. In both, it was increased in nCRT responders [23,24].
- (8)
- (9)
- (10)
- (11)
- RRM1 encodes the protein Ribonucleotide Reductase Catalytic Subunit M1, subunit of ribonucleotide reductase, an enzyme essential for the conversion of ribonucleotides into deoxyribonucleotides, which are important for DNA replication and repair. Its differentially relative expression (R versus NR) was analysed in two studies. In both, it was increased in nCRT responders [20,29].
- (12)
- (13)
- TYMP encodes the protein Pyrimidine Metabolism Enzyme Thymidine Phosphorylase, an angiogenic factor that promotes angiogenesis in vivo and stimulates the in vitro growth of endothelial cells. Its differentially relative expression (R versus NR) was analysed in five studies. In four of them, it was increased in nCRT responders [30,31,32,33]. In one study, it was decreased, but only when associated with no distant recurrence [34].
- (14)
- TYMS encodes thymidylate synthase, an enzyme responsible for DNA methylation, playing a pivotal role in DNA replication and repair. Its differentially relative expression (R versus NR) was analysed in eight studies and was decreased in nCRT responders [14,32,34,35,36,37,38]. In particular, nCRT response was reported always in terms of tumor regression grade (according to various classification) in all eight studies. Moreover, nCRT response was also reported as downstaging in two studies [14,35] and as disease-free survival in another two studies [34,35]. Another studies [39] reported nCRT response in terms of disease specific survival and recurrence free survival; therefore, it was excluded in the systematic analysis of all genes differentially expressed.
- (15)
3.2.2. Main Biological Function of other Genes Identified as nCRT Predictors
3.3. Analysis of miRNA Expression Data
3.3.1. Upregulated miRNA in Responders
3.3.2. Downregulated miRNA in Responders
4. Discussion
4.1. miRNAs Differential Expression
4.2. Identification of Targeted Proteins
4.3. Correlation of miRNA with Suppressed Oncogenic Mutation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Globocan. Available online: https://gco.iarc.fr/today/home (accessed on 27 May 2020).
- American Cancer Society. Colorectal Cancer Facts & Figures. Available online: https://www.cancer.org (accessed on 27 May 2020).
- Stefano Tardivo, W.M.; Torri, E.; Poli, A. Rectal Cancer: Epidemiology and Burden of Disease; Springer: Berlin/Heidelber, Germany, 2005. [Google Scholar]
- Gaertner, W.B.; Kwaan, M.R.; Madoff, R.D.; Melton, G.B. Rectal cancer: An evidence-based update for primary care providers. World J. Gastroenterol. 2015, 21, 7659–7671. [Google Scholar] [CrossRef] [PubMed]
- Beddy, D.; Hyland, J.M.; Winter, D.C.; Lim, C.; White, A.; Moriarty, M.; Armstrong, J.; Fennelly, D.; Gibbons, D.; Sheahan, K. A simplified tumor regression grade correlates with survival in locally advanced rectal carcinoma treated with neoadjuvant chemoradiotherapy. Ann. Surg. Oncol. 2008, 15, 3471–3477. [Google Scholar] [CrossRef] [PubMed]
- Topova, L.; Hellmich, G.; Puffer, E.; Schubert, C.; Christen, N.; Boldt, T.; Wiedemann, B.; Witzigmann, H.; Stelzner, S. Prognostic value of tumor response to neoadjuvant therapy in rectal carcinoma. Dis. Colon. Rectum. 2011, 54, 401–411. [Google Scholar] [CrossRef]
- Williamson, J.S.; Harris, D.A.; Beynon, J.; Jenkins, G.J. Review of the development of DNA methylation as a marker of response to neoadjuvant therapy and outcomes in rectal cancer. Clin. Epigenetics. 2015, 7, 70. [Google Scholar] [CrossRef]
- Izzotti, A. Molecular medicine and the development of cancer chemopreventive agents. Ann. N. Y. Acad. Sci. 2012, 1259, 26–32. [Google Scholar] [CrossRef]
- Calin, G.A.; Croce, C.M. MicroRNA signatures in human cancers. Nat. Rev. Cancer 2006, 6, 857–866. [Google Scholar] [CrossRef]
- Alvarez-Garcia, I.; Miska, E.A. MicroRNA functions in animal development and human disease. Development 2005, 132, 4653–4662. [Google Scholar] [CrossRef] [PubMed]
- Jansson, M.D.; Lund, A.H. MicroRNA and cancer. Mol. Oncol. 2012, 6, 590–610. [Google Scholar] [CrossRef] [PubMed]
- Izzotti, A.; Carozzo, S.; Pulliero, A.; Zhabayeva, D.; Ravetti, J.L.; Bersimbaev, R. Extracellular MicroRNA in liquid biopsy: Applicability in cancer diagnosis and prevention. Am. J. Cancer Res. 2016, 6, 1461–1493. [Google Scholar]
- Prospero Database. Available online: https://www.crd.york.ac.uk/PROSPERO/ (accessed on 27 May 2020).
- Cosmis catalogue, C. Available online: https://cancer.sanger.ac.uk/cosmic (accessed on 27 May 2020).
- Human Gene Database—GENE CARDS. Available online: https://genecards.org (accessed on 27 May 2020).
- Rodel, F.; Frey, B.; Leitmann, W.; Capalbo, G.; Weiss, C.; Rodel, C. Survivin antisense oligonucleotides effectively radiosensitize colorectal cancer cells in both tissue culture and murine xenograft models. Int. J. Radiat. Oncol. Biol. Phys. 2008, 71, 247–255. [Google Scholar] [CrossRef]
- Huh, J.W.; Lee, J.H.; Kim, H.R. Pretreatment expression of 13 molecular markers as a predictor of tumor responses after neoadjuvant chemoradiation in rectal cancer. Ann. Surg. 2014, 259, 508–515. [Google Scholar] [CrossRef] [PubMed]
- Horisberger, K.; Erben, P.; Strobel, P.; Bohn, B.; Hahn, M.; Kahler, G.; Wenz, F.; Hochhaus, A.; Post, S.; Willeke, F.; et al. Annexin and survivin in locally advanced rectal cancer: Indicators of resistance to preoperative chemoradiotherapy? Onkologie 2010, 33, 439–444. [Google Scholar] [CrossRef]
- Rimkus, C.; Friederichs, J.; Boulesteix, A.L.; Theisen, J.; Mages, J.; Becker, K.; Nekarda, H.; Rosenberg, R.; Janssen, K.P.; Siewert, J.R. Microarray-based prediction of tumor response to neoadjuvant radiochemotherapy of patients with locally advanced rectal cancer. Clin. Gastroenterol. Hepatol. 2008, 6, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Palma, P.; Cano, C.; Conde-Muino, R.; Comino, A.; Bueno, P.; Ferron, J.A.; Cuadros, M. Expression profiling of rectal tumors defines response to neoadjuvant treatment related genes. PLoS ONE 2014, 9, e112189. [Google Scholar] [CrossRef] [PubMed]
- Toiyama, Y.; Inoue, Y.; Saigusa, S.; Okugawa, Y.; Yokoe, T.; Tanaka, K.; Miki, C.; Kusunoki, M. Gene expression profiles of epidermal growth factor receptor, vascular endothelial growth factor and hypoxia-inducible factor-1 with special reference to local responsiveness to neoadjuvant chemoradiotherapy and disease recurrence after rectal cancer surgery. Clin. Oncol. R. Coll. Radiol. 2010, 22, 272–280. [Google Scholar]
- Saigusa, S.; Tanaka, K.; Toiyama, Y.; Matsushita, K.; Kawamura, M.; Okugawa, Y.; Hiro, J.; Inoue, Y.; Uchida, K.; Mohri, Y.; et al. Gene expression profiles of tumor regression grade in locally advanced rectal cancer after neoadjuvant chemoradiotherapy. Oncol. Rep. 2012, 28, 855–861. [Google Scholar] [CrossRef]
- Lopes-Ramos, C.; Koyama, F.C.; Habr-Gama, A.; Salim, A.C.; Bettoni, F.; Asprino, P.F.; Franca, G.S.; Gama-Rodrigues, J.; Parmigiani, R.B.; Perez, R.O.; et al. Comprehensive evaluation of the effectiveness of gene expression signatures to predict complete response to neoadjuvant chemoradiotherapy and guide surgical intervention in rectal cancer. Cancer Genet. 2015, 208, 319–326. [Google Scholar] [CrossRef]
- Koyama, F.C.; Lopes Ramos, C.M.; Ledesma, F.; Alves, V.A.F.; Fernandes, J.M.; Vailati, B.B.; Sao Juliao, G.P.; Habr-Gama, A.; Gama-Rodrigues, J.; Perez, R.O.; et al. Effect of Akt activation and experimental pharmacological inhibition on responses to neoadjuvant chemoradiotherapy in rectal cancer. Br. J. Surg. 2018, 105, e192–e203. [Google Scholar] [CrossRef]
- Ghadimi, B.M.; Grade, M.; Difilippantonio, M.J.; Varma, S.; Simon, R.; Montagna, C.; Fuzesi, L.; Langer, C.; Becker, H.; Liersch, T.; et al. Effectiveness of gene expression profiling for response prediction of rectal adenocarcinomas to preoperative chemoradiotherapy. J. Clin. Oncol. 2005, 23, 1826–1838. [Google Scholar] [CrossRef]
- Lee, Y.Y.; Wei, Y.C.; Tian, Y.F.; Sun, D.P.; Sheu, M.J.; Yang, C.C.; Lin, L.C.; Lin, C.Y.; Hsing, C.H.; Li, W.S.; et al. Overexpression of Transcobalamin 1 is an Independent Negative Prognosticator in Rectal Cancers Receiving Concurrent Chemoradiotherapy. J. Cancer 2017, 8, 1330–1337. [Google Scholar] [CrossRef]
- Saigusa, S.; Inoue, Y.; Tanaka, K.; Toiyama, Y.; Kawamura, M.; Okugawa, Y.; Okigami, M.; Hiro, J.; Uchida, K.; Mohri, Y.; et al. Significant correlation between LKB1 and LGR5 gene expression and the association with poor recurrence-free survival in rectal cancer after preoperative chemoradiotherapy. J. Cancer Res. Clin. Oncol. 2013, 139, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Conde-Muino, R.; Cano, C.; Sanchez-Martin, V.; Herrera, A.; Comino, A.; Medina, P.P.; Palma, P.; Cuadros, M. Preoperative chemoradiotherapy for rectal cancer: The sensitizer role of the association between miR-375 and c-Myc. Oncotarget 2017, 8, 82294–82302. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nishioka, M.; Shimada, M.; Kurita, N.; Iwata, T.; Morimoto, S.; Yoshikawa, K.; Higashijima, J.; Miyatani, T. Gene expression profile can predict pathological response to preoperative chemoradiotherapy in rectal cancer. Cancer Genom. Proteom. 2011, 8, 87–92. [Google Scholar]
- Kocakova, I.; Svoboda, M.; Kubosova, K.; Chrenko, V.; Roubalova, E.; Krejci, E.; Sefr, R.; Slampa, P.; Frgala, T.; Zaloudik, J. Preoperative radiotherapy and concomitant capecitabine treatment induce thymidylate synthase and thymidine phosphorylase mRNAs in rectal carcinoma. Neoplasma 2007, 54, 447–453. [Google Scholar] [PubMed]
- Chiorean, E.G.; Sanghani, S.; Schiel, M.A.; Yu, M.; Burns, M.; Tong, Y.; Hinkle, D.T.; Coleman, N.; Robb, B.; LeBlanc, J.; et al. Phase II and gene expression analysis trial of neoadjuvant capecitabine plus irinotecan followed by capecitabine-based chemoradiotherapy for locally advanced rectal cancer: Hoosier Oncology Group GI03–53. Cancer Chemother. Pharmacol. 2012, 70, 25–32. [Google Scholar] [CrossRef]
- Sakai, K.; Kazama, S.; Nagai, Y.; Murono, K.; Tanaka, T.; Ishihara, S.; Sunami, E.; Tomida, S.; Nishio, K.; Watanabe, T. Chemoradiation provides a physiological selective pressure that increases the expansion of aberrant TP53 tumor variants in residual rectal cancerous regions. Oncotarget 2014, 5, 9641–9649. [Google Scholar] [CrossRef]
- Sadahiro, S.; Suzuki, T.; Tanaka, A.; Okada, K.; Saito, G.; Kamijo, A.; Nagase, H. Increase in Gene Expression of TYMP, DPYD and HIF1A Are Associated with Response to Preoperative Chemoradiotherapy Including S-1 or UFT for Rectal Cancer. Anticancer Res. 2016, 36, 2433–2440. [Google Scholar]
- Tanaka, K.; Saigusa, S.; Toiyama, Y.; Koike, Y.; Okugawa, Y.; Yokoe, T.; Inoue, Y.; Kobayashi, M.; Miki, C.; Kusunoki, M. TS and DPD mRNA levels on formalin-fixed paraffin-embedded specimens as predictors for distant recurrence of rectal cancer treated with preoperative chemoradiotherapy. J. Surg. Oncol. 2012, 105, 529–534. [Google Scholar] [CrossRef]
- Jakob, C.; Aust, D.E.; Meyer, W.; Baretton, G.B.; Schwabe, W.; Hausler, P.; Becker, H.; Liersch, T. Thymidylate synthase, thymidine phosphorylase, dihydropyrimidine dehydrogenase expression, and histological tumour regression after 5-FU-based neo-adjuvant chemoradiotherapy in rectal cancer. J. Pathol. 2004, 204, 562–568. [Google Scholar] [CrossRef]
- Kim, I.J.; Lim, S.B.; Kang, H.C.; Chang, H.J.; Ahn, S.A.; Park, H.W.; Jang, S.G.; Park, J.H.; Kim, D.Y.; Jung, K.H.; et al. Microarray gene expression profiling for predicting complete response to preoperative chemoradiotherapy in patients with advanced rectal cancer. Dis. Colon. Rectum. 2007, 50, 1342–1353. [Google Scholar] [CrossRef]
- Jakob, C.; Liersch, T.; Meyer, W.; Becker, H.; Baretton, G.B.; Aust, D.E. Predictive value of Ki67 and p53 in locally advanced rectal cancer: Correlation with thymidylate synthase and histopathological tumor regression after neoadjuvant 5-FU-based chemoradiotherapy. World J. Gastroenterol. 2008, 14, 1060–1066. [Google Scholar] [CrossRef] [PubMed]
- Casado, E.; Garcia, V.M.; Sanchez, J.J.; Blanco, M.; Maurel, J.; Feliu, J.; Fernandez-Martos, C.; de Castro, J.; Castelo, B.; Belda-Iniesta, C.; et al. A combined strategy of SAGE and quantitative PCR Provides a 13-gene signature that predicts preoperative chemoradiotherapy response and outcome in rectal cancer. Clin. Cancer Res. 2011, 17, 4145–4154. [Google Scholar] [CrossRef] [PubMed]
- Kornmann, M.; Schwabe, W.; Sander, S.; Kron, M.; Strater, J.; Polat, S.; Kettner, E.; Weiser, H.F.; Baumann, W.; Schramm, H.; et al. Thymidylate synthase and dihydropyrimidine dehydrogenase mRNA expression levels: Predictors for survival in colorectal cancer patients receiving adjuvant 5-fluorouracil. Clin. Cancer Res. 2003, 9, 4116–4124. [Google Scholar] [PubMed]
- Grimminger, P.P.; Danenberg, P.; Dellas, K.; Arnold, D.; Rodel, C.; Machiels, J.P.; Haustermans, K.; Debucquoy, A.; Velenik, V.; Sempoux, C.; et al. Biomarkers for cetuximab-based neoadjuvant radiochemotherapy in locally advanced rectal cancer. Clin. Cancer Res. 2011, 17, 3469–3477. [Google Scholar] [CrossRef] [PubMed]
- Empuku, S.; Nakajima, K.; Akagi, T.; Kaneko, K.; Hijiya, N.; Etoh, T.; Shiraishi, N.; Moriyama, M.; Inomata, M. An 80-gene set to predict response to preoperative chemoradiotherapy for rectal cancer by principle component analysis. Mol. Clin. Oncol. 2016, 4, 733–739. [Google Scholar] [CrossRef]
- Gim, J.; Cho, Y.B.; Hong, H.K.; Kim, H.C.; Yun, S.H.; Wu, H.G.; Jeong, S.Y.; Joung, J.G.; Park, T.; Park, W.Y.; et al. Predicting multi-class responses to preoperative chemoradiotherapy in rectal cancer patients. Radiat. Oncol. 2016, 11, 50. [Google Scholar] [CrossRef]
- Liersch, T.; Grade, M.; Gaedcke, J.; Varma, S.; Difilippantonio, M.J.; Langer, C.; Hess, C.F.; Becker, H.; Ried, T.; Ghadimi, B.M. Preoperative chemoradiotherapy in locally advanced rectal cancer: Correlation of a gene expression-based response signature with recurrence. Cancer Genet. Cytogenet. 2009, 190, 57–65. [Google Scholar] [CrossRef]
- Hur, H.; Tulina, I.; Cho, M.S.; Min, B.S.; Koom, W.S.; Lim, J.S.; Ahn, J.B.; Kim, N.K. Biomarker-Based Scoring System for Prediction of Tumor Response After Preoperative Chemoradiotherapy in Rectal Cancer by Reverse Transcriptase Polymerase Chain Reaction Analysis. Dis. Colon. Rectum. 2016, 59, 1174–1182. [Google Scholar] [CrossRef]
- Nakao, T.; Iwata, T.; Hotchi, M.; Yoshikawa, K.; Higashijima, J.; Nishi, M.; Takasu, C.; Eto, S.; Teraoku, H.; Shimada, M. Prediction of response to preoperative chemoradiotherapy and establishment of individualized therapy in advanced rectal cancer. Oncol. Rep. 2015, 34, 1961–1967. [Google Scholar] [CrossRef][Green Version]
- Carames, C.; Cristobal, I.; Moreno, V.; Marin, J.P.; Gonzalez-Alonso, P.; Torrejon, B.; Minguez, P.; Leon, A.; Martin, J.I.; Hernandez, R.; et al. MicroRNA-31 Emerges as a Predictive Biomarker of Pathological Response and Outcome in Locally Advanced Rectal Cancer. Int. J. Mol. Sci. 2016, 17, 878. [Google Scholar] [CrossRef]
- Hotchi, M.; Shimada, M.; Kurita, N.; Iwata, T.; Sato, H.; Morimoto, S.; Yoshikawa, K.; Higashijima, J.; Miyatani, T. microRNA expression is able to predict response to chemoradiotherapy in rectal cancer. Mol. Clin. Oncol. 2013, 1, 137–142. [Google Scholar] [CrossRef]
- Della Vittoria Scarpati, G.; Falcetta, F.; Carlomagno, C.; Ubezio, P.; Marchini, S.; De Stefano, A.; Singh, V.K.; D’Incalci, M.; De Placido, S.; Pepe, S. A specific miRNA signature correlates with complete pathological response to neoadjuvant chemoradiotherapy in locally advanced rectal cancer. Int. J. Radiat. Oncol. Biol. Phys. 2012, 83, 1113–1119. [Google Scholar] [CrossRef]
- Ma, W.; Yu, J.; Qi, X.; Liang, L.; Zhang, Y.; Ding, Y.; Lin, X.; Li, G. Radiation-induced microRNA-622 causes radioresistance in colorectal cancer cells by down-regulating Rb. Oncotarget 2015, 6, 15984–15994. [Google Scholar] [CrossRef]
- Svoboda, M.; Sana, J.; Fabian, P.; Kocakova, I.; Gombosova, J.; Nekvindova, J.; Radova, L.; Vyzula, R.; Slaby, O. MicroRNA expression profile associated with response to neoadjuvant chemoradiotherapy in locally advanced rectal cancer patients. Radiat. Oncol. 2012, 7, 195. [Google Scholar] [CrossRef]
- Eriksen, A.H.M.; Sorensen, F.B.; Andersen, R.F.; Jakobsen, A.; Hansen, T.F. Association between the expression of microRNAs and the response of patients with locally advanced rectal cancer to preoperative chemoradiotherapy. Oncol. Lett. 2017, 14, 201–209. [Google Scholar] [CrossRef][Green Version]
- Findlay, V.J.; Wang, C.; Nogueira, L.M.; Hurst, K.; Quirk, D.; Ethier, S.P.; Staveley O’Carroll, K.F.; Watson, D.K.; Camp, E.R. SNAI2 modulates colorectal cancer 5-fluorouracil sensitivity through miR145 repression. Mol. Cancer Ther. 2014, 13, 2713–2726. [Google Scholar] [CrossRef]
- Drebber, U.; Lay, M.; Wedemeyer, I.; Vallbohmer, D.; Bollschweiler, E.; Brabender, J.; Monig, S.P.; Holscher, A.H.; Dienes, H.P.; Odenthal, M. Altered levels of the onco-microRNA 21 and the tumor-supressor microRNAs 143 and 145 in advanced rectal cancer indicate successful neoadjuvant chemoradiotherapy. Int. J. Oncol. 2011, 39, 409–415. [Google Scholar] [CrossRef]
- D’Angelo, E.; Zanon, C.; Sensi, F.; Digito, M.; Rugge, M.; Fassan, M.; Scarpa, M.; Pucciarelli, S.; Nitti, D.; Agostini, M. miR-194 as predictive biomarker of responsiveness to neoadjuvant chemoradiotherapy in patients with locally advanced rectal adenocarcinoma. J. Clin. Pathol. 2018, 71, 344–350. [Google Scholar] [CrossRef]
- D’Angelo, E.; Fassan, M.; Maretto, I.; Pucciarelli, S.; Zanon, C.; Digito, M.; Rugge, M.; Nitti, D.; Agostini, M. Serum miR-125b is a non-invasive predictive biomarker of the pre-operative chemoradiotherapy responsiveness in patients with rectal adenocarcinoma. Oncotarget 2016, 7, 28647–28657. [Google Scholar] [CrossRef]
- Yu, J.; Li, N.; Wang, X.; Ren, H.; Wang, W.; Wang, S.; Song, Y.; Liu, Y.; Li, Y.; Zhou, X.; et al. Circulating serum microRNA-345 correlates with unfavorable pathological response to preoperative chemoradiotherapy in locally advanced rectal cancer. Oncotarget 2016, 7, 64233–64243. [Google Scholar] [CrossRef]
- Carames, C.; Cristobal, I.; Moreno, V.; del Puerto, L.; Moreno, I.; Rodriguez, M.; Marin, J.P.; Correa, A.V.; Hernandez, R.; Zenzola, V.; et al. MicroRNA-21 predicts response to preoperative chemoradiotherapy in locally advanced rectal cancer. Int. J. Colorectal. Dis. 2015, 30, 899–906. [Google Scholar] [CrossRef] [PubMed]
- Kheirelseid, E.A.; Miller, N.; Chang, K.H.; Curran, C.; Hennessey, E.; Sheehan, M.; Newell, J.; Lemetre, C.; Balls, G.; Kerin, M.J. miRNA expressions in rectal cancer as predictors of response to neoadjuvant chemoradiation therapy. Int. J. Colorectal. Dis. 2013, 28, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Chu, Q.; Xie, G.; Han, H.; Chen, Z.; Xu, B.; Yue, Z. Circadian gene expression predicts patient response to neoadjuvant chemoradiation therapy for rectal cancer. Int. J. Clin. Exp. Pathol. 2015, 8, 10985–10994. [Google Scholar] [PubMed]
- Vignard, J.; Mirey, G.; Salles, B. Ionizing-radiation induced DNA double-strand breaks: A direct and indirect lighting up. Radiother. Oncol. 2013, 108, 362–369. [Google Scholar] [CrossRef]
- Multhoff, G.; Radons, J. Radiation, inflammation, and immune responses in cancer. Front. Oncol. 2012, 2, 58. [Google Scholar] [CrossRef]
- Di Maggio, F.M.; Minafra, L.; Forte, G.I.; Cammarata, F.P.; Lio, D.; Messa, C.; Gilardi, M.C.; Bravata, V. Portrait of inflammatory response to ionizing radiation treatment. J. Inflamm. 2015, 12, 14. [Google Scholar] [CrossRef]
- Schaue, D.; Micewicz, E.D.; Ratikan, J.A.; Xie, M.W.; Cheng, G.; McBride, W.H. Radiation and inflammation. Semin. Radiat. Oncol. 2015, 25, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Kaur, P.; Asea, A. Radiation-induced effects and the immune system in cancer. Front. Oncol. 2012, 2, 191. [Google Scholar] [CrossRef]
- Chakraborty, M.; Abrams, S.I.; Coleman, C.N.; Camphausen, K.; Schlom, J.; Hodge, J.W. External beam radiation of tumors alters phenotype of tumor cells to render them susceptible to vaccine-mediated T-cell killing. Cancer Res. 2004, 64, 4328–4337. [Google Scholar] [CrossRef]
- Ganss, R.; Ryschich, E.; Klar, E.; Arnold, B.; Hammerling, G.J. Combination of T-cell therapy and trigger of inflammation induces remodeling of the vasculature and tumor eradication. Cancer Res. 2002, 62, 1462–1470. [Google Scholar]
- Surace, L.; Guckenberger, M.; van den Broek, M. Radiation holidays stimulate tumor immunity. Oncotarget 2015, 6, 15716–15717. [Google Scholar] [CrossRef] [PubMed]
- Surace, L.; Scheifinger, N.A.; Gupta, A.; van den Broek, M. Radiotherapy supports tumor-specific immunity by acute inflammation. Oncoimmunology 2016, 5, e1060391. [Google Scholar] [CrossRef] [PubMed]
- Erreni, M.; Mantovani, A.; Allavena, P. Tumor-associated Macrophages (TAM) and Inflammation in Colorectal Cancer. Cancer Microenviron. 2011, 4, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Takano, Y.; Masuda, T.; Iinuma, H.; Yamaguchi, R.; Sato, K.; Tobo, T.; Hirata, H.; Kuroda, Y.; Nambara, S.; Hayashi, N.; et al. Circulating exosomal microRNA-203 is associated with metastasis possibly via inducing tumor-associated macrophages in colorectal cancer. Oncotarget 2017, 8, 78598–78613. [Google Scholar] [CrossRef]
- Fong, M.Y.; Zhou, W.; Liu, L.; Alontaga, A.Y.; Chandra, M.; Ashby, J.; Chow, A.; O’Connor, S.T.; Li, S.; Chin, A.R.; et al. Breast-cancer-secreted miR-122 reprograms glucose metabolism in premetastatic niche to promote metastasis. Nat. Cell. Biol. 2015, 17, 183–194. [Google Scholar] [CrossRef]
- Costa-Silva, B.; Aiello, N.M.; Ocean, A.J.; Singh, S.; Zhang, H.; Thakur, B.K.; Becker, A.; Hoshino, A.; Mark, M.T.; Molina, H.; et al. Pancreatic cancer exosomes initiate pre-metastatic niche formation in the liver. Nat. Cell. Biol. 2015, 17, 816–826. [Google Scholar] [CrossRef]
- Yin, Y.; Yao, S.; Hu, Y.; Feng, Y.; Li, M.; Bian, Z.; Zhang, J.; Qin, Y.; Qi, X.; Zhou, L.; et al. The Immune-microenvironment Confers Chemoresistance of Colorectal Cancer through Macrophage-Derived IL6. Clin. Cancer Res. 2017, 23, 7375–7387. [Google Scholar] [CrossRef]
- Metzger, B.; Chambeau, L.; Begon, D.Y.; Faber, C.; Kayser, J.; Berchem, G.; Pauly, M.; Boniver, J.; Delvenne, P.; Dicato, M.; et al. The human epidermal growth factor receptor (EGFR) gene in European patients with advanced colorectal cancer harbors infrequent mutations in its tyrosine kinase domain. BMC Med. Genet. 2011, 12, 144. [Google Scholar] [CrossRef]
- Oh, B.Y.; Lee, R.A.; Chung, S.S.; Kim, K.H. Epidermal growth factor receptor mutations in colorectal cancer patients. J. Korean. Soc. Coloproctol. 2011, 27, 127–132. [Google Scholar] [CrossRef]
- Moroni, M.; Veronese, S.; Benvenuti, S.; Marrapese, G.; Sartore-Bianchi, A.; Di Nicolantonio, F.; Gambacorta, M.; Siena, S.; Bardelli, A. Gene copy number for epidermal growth factor receptor (EGFR) and clinical response to antiEGFR treatment in colorectal cancer: A cohort study. Lancet Oncol. 2005, 6, 279–286. [Google Scholar] [CrossRef]
- Sartore-Bianchi, A.; Moroni, M.; Veronese, S.; Carnaghi, C.; Bajetta, E.; Luppi, G.; Sobrero, A.; Barone, C.; Cascinu, S.; Colucci, G.; et al. Epidermal growth factor receptor gene copy number and clinical outcome of metastatic colorectal cancer treated with panitumumab. J. Clin. Oncol. 2007, 25, 3238–3245. [Google Scholar] [CrossRef]
- Graham, A.M.; Presnell, J.S. Hypoxia Inducible Factor (HIF) transcription factor family expansion, diversification, divergence and selection in eukaryotes. PLoS ONE 2017, 12, e0179545. [Google Scholar] [CrossRef]
- Talks, K.L.; Turley, H.; Gatter, K.C.; Maxwell, P.H.; Pugh, C.W.; Ratcliffe, P.J.; Harris, A.L. The expression and distribution of the hypoxia-inducible factors HIF-1alpha and HIF-2alpha in normal human tissues, cancers, and tumor-associated macrophages. Am. J. Pathol. 2000, 157, 411–421. [Google Scholar] [CrossRef]
- Liu, R.; Truax, A.D.; Chen, L.; Hu, P.; Li, Z.; Chen, J.; Song, C.; Ting, J.P. Expression profile of innate immune receptors, NLRs and AIM2, in human colorectal cancer: Correlation with cancer stages and inflammasome components. Oncotarget 2015, 6, 33456–33469. [Google Scholar] [CrossRef]
- Zhang, W.; Shi, X.; Peng, Y.; Wu, M.; Zhang, P.; Xie, R.; Wu, Y.; Yan, Q.; Liu, S.; Wang, J. HIF-1alpha Promotes Epithelial-Mesenchymal Transition and Metastasis through Direct Regulation of ZEB1 in Colorectal Cancer. PLoS ONE 2015, 10, e0129603. [Google Scholar]
- Jing, S.W.; Wang, Y.D.; Kuroda, M.; Su, J.W.; Sun, G.G.; Liu, Q.; Cheng, Y.J.; Yang, C.R. HIF-1alpha contributes to hypoxia-induced invasion and metastasis of esophageal carcinoma via inhibiting E-cadherin and promoting MMP-2 expression. Acta. Med. Okayama 2012, 66, 399–407. [Google Scholar]
- TargetScan. Available online: http://www.targetscan.org/vert_71/ (accessed on 27 May 2020).
- Nakajima, G.; Hayashi, K.; Xi, Y.; Kudo, K.; Uchida, K.; Takasaki, K.; Yamamoto, M.; Ju, J. Non-coding MicroRNAs hsa-let-7g and hsa-miR-181b are Associated with Chemoresponse to S-1 in Colon Cancer. Cancer Genom. Proteom. 2006, 3, 317–324. [Google Scholar]
- Knechtel, G.; Szkandera, J.; Stotz, M.; Hofmann, G.; Langsenlehner, U.; Krippl, P.; Samonigg, H.; Renner, W.; Langner, C.; Dehchamani, D.; et al. Single nucleotide polymorphisms in the hypoxia-inducible factor-1 gene and colorectal cancer risk. Mol. Carcinog. 2010, 49, 805–809. [Google Scholar] [CrossRef]
- Kuwai, T.; Kitadai, Y.; Tanaka, S.; Hiyama, T.; Tanimoto, K.; Chayama, K. Mutation of the von Hippel-Lindau (VHL) gene in human colorectal carcinoma: Association with cytoplasmic accumulation of hypoxia-inducible factor (HIF)-1alpha. Cancer Sci. 2004, 95, 149–153. [Google Scholar] [CrossRef]
- NCBI VEGFA Vascular Endothelial Growth Factor A [Homo Sapiens (Human)]. Available online: https://www.ncbi.nlm.nih.gov/gene/7422 (accessed on 27 May 2020).
- Vlajnic, T.; Andreozzi, M.C.; Schneider, S.; Tornillo, L.; Karamitopoulou, E.; Lugli, A.; Ruiz, C.; Zlobec, I.; Terracciano, L. VEGFA gene locus (6p12) amplification identifies a small but highly aggressive subgroup of colorectal cancer [corrected] patients. Mod. Pathol. 2011, 24, 1404–1412. [Google Scholar] [CrossRef]
- Burmeister, K.; Quagliata, L.; Andreozzi, M.; Eppenberger-Castori, S.; Matter, M.S.; Perrina, V.; Grobholz, R.; Jochum, W.; Horber, D.; Moosmann, P.; et al. Vascular endothelial growth factor A amplification in colorectal cancer is associated with reduced M1 and M2 macrophages and diminished PD-1-expressing lymphocytes. PLoS ONE 2017, 12, e0175563. [Google Scholar] [CrossRef]
- Overman, M.J.; McDermott, R.; Leach, J.L.; Lonardi, S.; Lenz, H.J.; Morse, M.A.; Desai, J.; Hill, A.; Axelson, M.; Moss, R.A.; et al. Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): An open-label, multicentre, phase 2 study. Lancet Oncol. 2017, 18, 1182–1191. [Google Scholar] [CrossRef]
- Cao, D.; Hou, M.; Guan, Y.S.; Jiang, M.; Yang, Y.; Gou, H.F. Expression of HIF-1alpha and VEGF in colorectal cancer: Association with clinical outcomes and prognostic implications. BMC Cancer 2009, 9, 432. [Google Scholar] [CrossRef] [PubMed]
- Riabov, V.; Gudima, A.; Wang, N.; Mickley, A.; Orekhov, A.; Kzhyshkowska, J. Role of tumor associated macrophages in tumor angiogenesis and lymphangiogenesis. Front. Physiol. 2014, 5, 75. [Google Scholar] [CrossRef] [PubMed]
- Linde, N.; Lederle, W.; Depner, S.; van Rooijen, N.; Gutschalk, C.M.; Mueller, M.M. Vascular endothelial growth factor-induced skin carcinogenesis depends on recruitment and alternative activation of macrophages. J. Pathol. 2012, 227, 17–28. [Google Scholar] [CrossRef]
- Geretto, M.; Pulliero, A.; Rosano, C.; Zhabayeva, D.; Bersimbaev, R.; Izzotti, A. Resistance to cancer chemotherapeutic drugs is determined by pivotal microRNA regulators. Am. J. Cancer Res. 2017, 7, 1350–1371. [Google Scholar]
- Xia, L.; Zhang, D.; Du, R.; Pan, Y.; Zhao, L.; Sun, S.; Hong, L.; Liu, J.; Fan, D. miR-15b and miR-16 modulate multidrug resistance by targeting BCL2 in human gastric cancer cells. Int. J. Cancer 2008, 123, 372–379. [Google Scholar] [CrossRef]
- Blower, P.E.; Chung, J.H.; Verducci, J.S.; Lin, S.; Park, J.K.; Dai, Z.; Liu, C.G.; Schmittgen, T.D.; Reinhold, W.C.; Croce, C.M.; et al. MicroRNAs modulate the chemosensitivity of tumor cells. Mol. Cancer Ther. 2008, 7, 1–9. [Google Scholar] [CrossRef]
- Cittelly, D.M.; Das, P.M.; Salvo, V.A.; Fonseca, J.P.; Burow, M.E.; Jones, F.E. Oncogenic HER2{Delta}16 suppresses miR-15a/16 and deregulates BCL-2 to promote endocrine resistance of breast tumors. Carcinogenesis 2010, 31, 2049–2057. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Wu, H.; Liu, X.; Evans, B.R.; Medina, D.J.; Liu, C.G.; Yang, J.M. Role of MicroRNA miR-27a and miR-451 in the regulation of MDR1/P-glycoprotein expression in human cancer cells. Biochem. Pharmacol. 2008, 76, 582–588. [Google Scholar] [CrossRef]
- Yao, J.; Li, Z.; Wang, X.; Xu, P.; Zhao, L.; Qian, J. MiR-125a regulates chemo-sensitivity to gemcitabine in human pancreatic cancer cells through targeting A20. Acta. Biochim. Biophys. Sin. 2016, 48, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.J.; Zhi, X.; Wang, Y.; Zhang, Z.; Hao, Z.; Ye, R.; Tang, Z.; Qian, F.; Wang, Q.; Zhu, J. Comprehensive Proteomic Characterization of the Human Colorectal Carcinoma Reveals Signature Proteins and Perturbed Pathways. Sci. Rep. 2017, 7, 42436. [Google Scholar] [CrossRef] [PubMed]
- Agostini, M.; Janssen, K.P.; Kim, I.J.; D’Angelo, E.; Pizzini, S.; Zangrando, A.; Zanon, C.; Pastrello, C.; Maretto, I.; Digito, M.; et al. An integrative approach for the identification of prognostic and predictive biomarkers in rectal cancer. Oncotarget 2015, 6, 32561–32574. [Google Scholar] [CrossRef] [PubMed]
- Yeoh, L.C.; Loh, C.K.; Gooi, B.H.; Singh, M.; Gam, L.H. Hydrophobic protein in colorectal cancer in relation to tumor stages and grades. World J. Gastroenterol. 2010, 16, 2754–2763. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Wang, H.; Li, J.; Liu, Y.; Ding, Y. Overexpression of Rho GDP-dissociation inhibitor alpha is associated with tumor progression and poor prognosis of colorectal cancer. J. Proteome. Res. 2008, 7, 3994–4003. [Google Scholar] [CrossRef] [PubMed]
- Bowden, D.L.; Sutton, P.A.; Wall, M.A.; Jithesh, P.V.; Jenkins, R.E.; Palmer, D.H.; Goldring, C.E.; Parsons, J.L.; Park, B.K.; Kitteringham, N.R.; et al. Proteomic profiling of rectal cancer reveals acid ceramidase is implicated in radiation response. J. Proteom. 2018, 179, 53–60. [Google Scholar] [CrossRef]
- Chauvin, A.; Wang, C.S.; Geha, S.; Garde-Granger, P.; Mathieu, A.A.; Lacasse, V.; Boisvert, F.M. The response to neoadjuvant chemoradiotherapy with 5-fluorouracil in locally advanced rectal cancer patients: A predictive proteomic signature. Clin. Proteomics 2018, 15, 16. [Google Scholar] [CrossRef]
- Repetto, O.; De Re, V.; De Paoli, A.; Belluco, C.; Alessandrini, L.; Canzonieri, V.; Cannizzaro, R. Identification of protein clusters predictive of tumor response in rectal cancer patients receiving neoadjuvant chemo-radiotherapy. Oncotarget 2017, 8, 28328–28341. [Google Scholar] [CrossRef]
- Raskov, H.; Pommergaard, H.C.; Burcharth, J.; Rosenberg, J. Colorectal carcinogenesis—Update and perspectives. World J. Gastroenterol. 2014, 20, 18151–18164. [Google Scholar] [CrossRef]
- Armaghany, T.; Wilson, J.D.; Chu, Q.; Mills, G. Genetic alterations in colorectal cancer. Gastrointest. Cancer Res. 2012, 5, 19–27. [Google Scholar]
- Huang, H.; Liu, N.; Guo, H.; Liao, S.; Li, X.; Yang, C.; Liu, S.; Song, W.; Liu, C.; Guan, L. L-carnitine is an endogenous HDAC inhibitor selectively inhibiting cancer cell growth in vivo and in vitro. PLoS ONE 2012, 7, e49062. [Google Scholar] [CrossRef] [PubMed]
- Abulí, A.; Fernández-Rozadilla, C.; Giráldez, M.D.; Muñoz, J.; Gonzalo, V.; Bessa, X.; Bujanda, L.; Reñé, J.M.; Lanas, A.; García, A.M.; et al. A two-phase case-control study for colorectal cancer genetic susceptibility: Candidate genes from chromosomal regions 9q22 and 3q22. Br. J. Cancer. 2011, 105, 870–875. [Google Scholar] [CrossRef] [PubMed]
- Van Gisbergen, M.W.; Voets, A.M.; Starmans, M.H.; de Coo, I.F.M.; Yadak, R.; Hoffman, R.F.; Boutros, P.C.; Smeets, H.J.M.; Dubois, L.; Lambin, P. How do changes in the mtDNA and mitochondrial dysfunction influence cancer and cancer therapy? Challenges, opportunities and models. Mutat. Res. Rev. Mutat. Res. 2015, 764, 16–30. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Huang, J.; Peng, J.; Wu, X.; Zhang, Y.; Zhu, W.; Guo, L. Upregulation of the inwardly rectifying potassium channel Kir2.1 (KCNJ2) modulates multidrug resistance of small-cell lung cancer under the regulation of miR-7 and the Ras/MAPK pathway. Mol. Cancer. 2015, 14, 59. [Google Scholar] [CrossRef] [PubMed]
- Burton, A.; Azevedo, C.; Andreassi, C.; Riccio, A.S.; Sairadi, A. Epigenetic control by inositol pyrophosphates. Proc. Natl. Acad. Sci. USA 2013, 110, 18970–18975. [Google Scholar] [CrossRef] [PubMed]
- Resta, N.; Simone, C.; Mareni, C.; Montera, M.; Gentile, M.; Susca, F.; Gristina, R.; Pozzi, S.; Bertario, L.; Bufo, P.; et al. STK11 mutations in Peutz-Jeghers syndrome and sporadic colon cancer. Cancer Res. 1998, 58, 4799–4801. [Google Scholar] [PubMed]
- Takahashi, H.; Ishii, H.; Nishida, N.; Ichiro, T.; Tsunekazu, M.; Masataka, I.; Takehiko, Y.; Kosi, M.; Hirofumi, Y.; Mitsugu, S.; et al. Significance of Lgr5(+ve) cancer stem cells in the colon and rectum. Ann. Surg. Oncol. 2011, 18, 1166–1174. [Google Scholar] [CrossRef]
- Liu, Y.F.; Yang, A.; Liu, W.; Wang, C.; Wang, M.; Zhang, L.; Wang, D.; Dong, J.F.; Li, M. NME2 reduces proliferation, migration and invasion of gastric cancer cells to limit metastasis. PLoS ONE 2015, 10, e0115968. [Google Scholar] [CrossRef]
- Zhou, Y.; Zheng, X.; Xu, B.; Chen, L.; Wang, Q.; Deng, H.; Jiang, J. Circular RNA hsa_circ_0004015 regulates the proliferation, invasion, and TKI drug resistance of non-small cell lung cancer by miR-1183/PDPK1 signaling pathway. Biochem. Biophys. Res. Commun. 2019, 508, 527–535. [Google Scholar] [CrossRef]
- Jordheim, L.P.; Sève, P.; Trédan, O.; Dumontet, C. The ribonucleotide reductase large subunit (RRM1) as a predictive factor in patients with cancer. Lancet Oncol. 2011, 12, 693–702. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, J.Y.; Lu, L.J.; Wang, C.H.; Wang, L.H. MiR-630 promotes epithelial ovarian cancer proliferation and invasion via targeting KLF6. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 4542–4547. [Google Scholar] [PubMed]
- Eoh, K.J.; Lee, S.H.; Kim, H.J.; Jung-Yun, L.; Sunghoon, K.; Sang Wun, K.; Young, T.K.; Eun, J.N. MicroRNA-630 inhibitor sensitizes chemoresistant ovarian cancer to chemotherapy by enhancing apoptosis. Biochem. Biophys. Res. Commun. 2018, 497, 513–520. [Google Scholar] [CrossRef] [PubMed]
| Top miRNAs | Target mRNAs | Gene Name | Total Context Score |
|---|---|---|---|
| miR-145 | TRIAP1 | TP53 regulated inhibitor of apoptosis 1 | 0.17 |
| EGFR | epidermal growth factor receptor | 0.30 | |
| BAK1 | BCL2-antagonist/killer 1 | 0.20 | |
| miR-223 | KRAS | Kirsten rat sarcoma viral oncogene homolog | 0.09 |
| miR-622 | TP53 | tumor protein p53 | 0.33 |
| KRAS | Kirsten rat sarcoma viral oncogene homolog | 0.24 | |
| HIF1A | Hypoxia-inducible factor 1, alpha subunit | 0.17 | |
| VEGFA | vascular endothelial growth factor A | 0.16 | |
| EGFR | epidermal growth factor receptor | 0.07 | |
| MKI67 | antigen identified by monoclonal antibody Ki-67 | 0.10 | |
| HIF1A | hypoxia-inducible factor 1, alpha subunit | 0.20 | |
| miR-1246 | KRAS | Kirsten rat sarcoma viral oncogene homolog | 0.18 |
| Protein | mRNA | miRNA |
|---|---|---|
| ABLIM1 | miR-153; miR-335 | |
| ALDH3A1 | miR-145 | |
| AMDHD2 | miR-483; miR-1224 | |
| ARID1B | let-7a; miR-92a; miR-144; miR-363 | |
| ASAH1 | miR-92a; miR-335 | |
| B3GALT5 | miR-622 | |
| CBX1 | miR-17; miR-20a; miR-20b; miR-106a; miR-223; mir-590 | |
| CDK | CDK5R1 | miR-92a; miR-196b; miR-363 |
| CDK | CDKN1(A) | let-7a; let-7g; let-7e; mir-16; miR-17; miR-20a; miR-106a; miR-125a; miR-145; miR-335; miR-363; miR-450a; miR-486; miR-542; miR-1909 |
| CD177 | miR-335 | |
| CNNM4 | miR-92a; miR-363; miR-450b; miR-765; miR-1224 | |
| DPYD | miR-494 | |
| ENSA | let-7e; miR-1224 | |
| ERBB2 | miR-21; miR-125a; miR-193a; miR-205; miR-486 | |
| FASTKD2 | miR-16 | |
| FTO | miR-450a | |
| glucose-6-phosphate 1-dehydrogenase | miR-335 | |
| GTF2E1 | mir-31; mir-92a; mir-363; miR-561 | |
| IFIT1 | miR-126; miR-335 | |
| LDH-A | miR-34b; miR-190b; miR-450b | |
| LSM12 | miR-561 | |
| MCMBP | miR-31; miR-154 | |
| PIP4K2B | miR-16; miR-215 | |
| PVR | miR-16; miR-17; miR-20a; miR-20b; miR-106a; miR-142; miR-519c | |
| PVRL1 | miR-765 | |
| RB1 | miR-17; miR-20a; miR-20b; miR-21; miR-99a; miR-106a; miR-99a; miR-144; miR-215; miR-335; miR-450b; miR-494; miR-519c; miR-590; miR-622 | |
| SLC25A33 | miR-17; miR-20a; miR-20b; miR-92a; miR-106a | |
| SLC5A6 | miR-379; let-7a; let-7e; let-7g | |
| TRMT5 | miR-20a; miR-153; miR-205 | |
| TSPAN6 | miR-16; miR-17; miR-20a; miR-20b; miR-106a; miR-142; miR-144; miR-145 | |
| TYMP | miR-92a |
| Ontology | mRNA | miRNA | Reference |
|---|---|---|---|
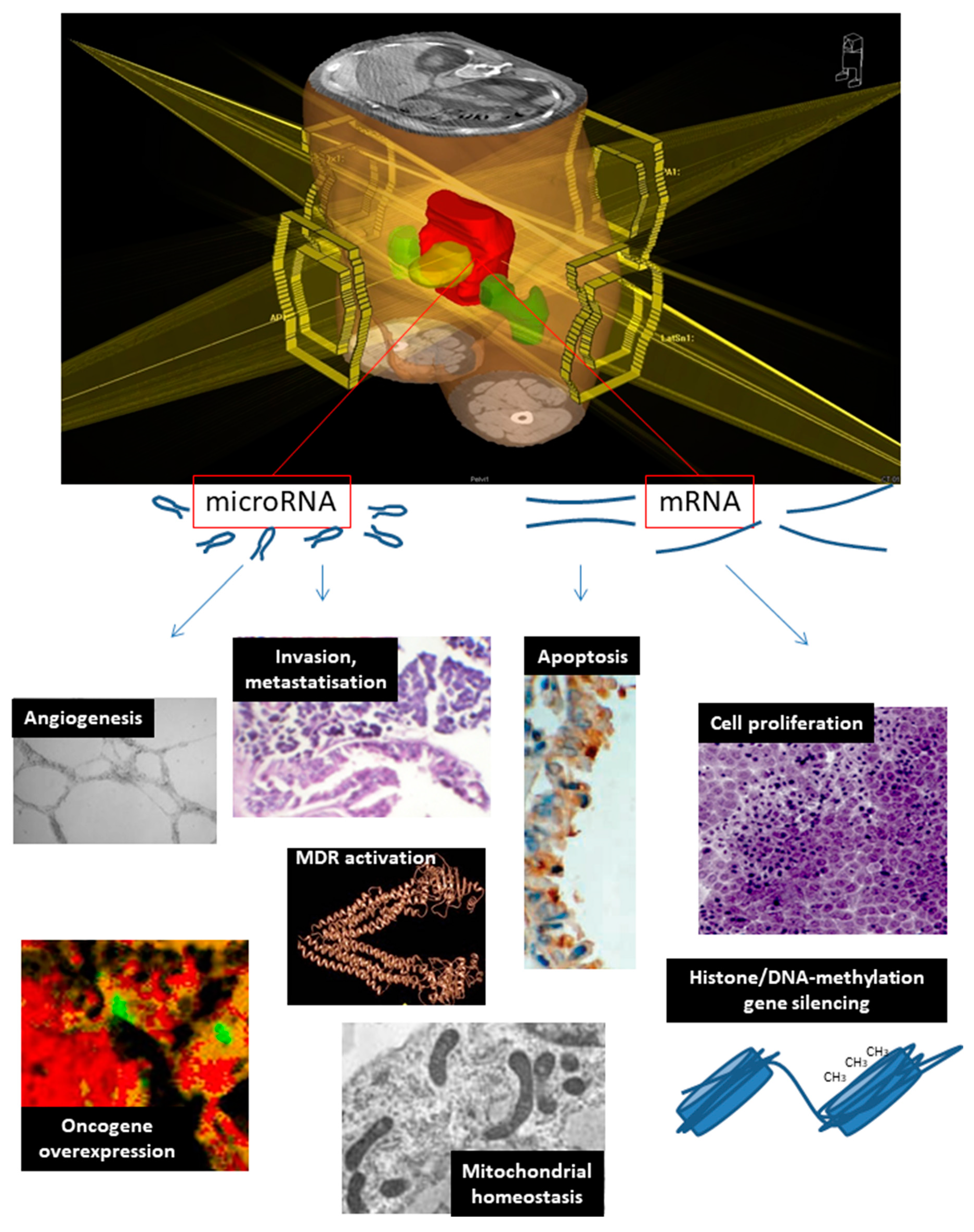
| Mitochondrial function, CRC genetic susceptibility | MT-ND4, MT-ND6, CDV3 | [109,110,111,112] | |
| Histone gene silencing, signal transduction | ITPK1, STK11 | [113,114] | |
| Oncogene overexpression | LGR5 (c-myc), MYC | let-7f (K-RAS), miR-21 (PTEN), miR-622 (k-RAS) | [115] |
| Invasion and metastatization | NME2, RRM1 | miR-1183 | [116,117,118] |
| Cell proliferation | EGFR, RRM1, STK11 | let-7f, miR-21, miR-622, miR-630, miR-1183 | [21,22,116,117,119,120] |
| Multidrug resistance | KCNJ2, RRM1 | miR-630, miR-1183 | [117,119,120] |
| Angiogenesis | TYMP, VEGFA | let-7f, miR-145 | [30,31,32,33,34,40] |
| DNA methylation | TYMS | [35,36,37,38,39] | |
| Apoptosis inhibition | BIRC5 | miR-630 | [16,17,18,120] |
| Gene | % of Cases Affected | Gain of Function (%) | Loss of Function (%) | Survival in Mutation Carriers vs. Wild Type |
|---|---|---|---|---|
| EGFR | 14 | 8 | 1 | = |
| LGR5 | 9 | 1 | 5 | Increased |
| MYC | 8 | 23 | 0 | Decreased |
| VEGFA | 7 | 9 | 0 | = |
| STK11 | 7 | 2 | 14 | = |
| TYMS | 4 | 7 | 2 | Increased |
| ITPK1 | 4 | 1 | 6 | Decreased |
| BIRC5 | 4 | 9 | 0 | = |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Izzotti, A.; Ceccaroli, C.; Geretto, M.; Ruggieri, F.G.; Schenone, S.; Di Maria, E. Predicting Response to Neoadjuvant Therapy in Colorectal Cancer Patients the Role of Messenger-and Micro-RNA Profiling. Cancers 2020, 12, 1652. https://doi.org/10.3390/cancers12061652
Izzotti A, Ceccaroli C, Geretto M, Ruggieri FG, Schenone S, Di Maria E. Predicting Response to Neoadjuvant Therapy in Colorectal Cancer Patients the Role of Messenger-and Micro-RNA Profiling. Cancers. 2020; 12(6):1652. https://doi.org/10.3390/cancers12061652
Chicago/Turabian StyleIzzotti, Alberto, Chiara Ceccaroli, Marta Geretto, Filippo Grillo Ruggieri, Sara Schenone, and Emilio Di Maria. 2020. "Predicting Response to Neoadjuvant Therapy in Colorectal Cancer Patients the Role of Messenger-and Micro-RNA Profiling" Cancers 12, no. 6: 1652. https://doi.org/10.3390/cancers12061652
APA StyleIzzotti, A., Ceccaroli, C., Geretto, M., Ruggieri, F. G., Schenone, S., & Di Maria, E. (2020). Predicting Response to Neoadjuvant Therapy in Colorectal Cancer Patients the Role of Messenger-and Micro-RNA Profiling. Cancers, 12(6), 1652. https://doi.org/10.3390/cancers12061652








