Comparison of Hodgkin’s Lymphoma in Children and Adolescents. A Twenty Year Experience with MH’96 and LH2004 AIEOP (Italian Association of Pediatric Hematology and Oncology) Protocols
Abstract
1. Introduction
- To evaluate the prognosis of adolescents in comparison with younger children when treated with the same protocol;
- to compare the results achieved in both age categories with the most recent Italian Association of Pediatric Hematology and Oncology (AIEOP) chemo-radiotherapeutic protocol (LH2004) and the previous one (MH’96); and
- to identify significant differences in clinical presentation of children and of adolescents, as well as an analysis of age as a prognostic factor that might impact the outcome of these categories of patients differently.
2. Materials and Methods
2.1. Response Criteria
2.2. Treatment
2.2.1. MH’96 Protocol
2.2.2. LH2004 Protocol
2.3. Statistical Methods
3. Results
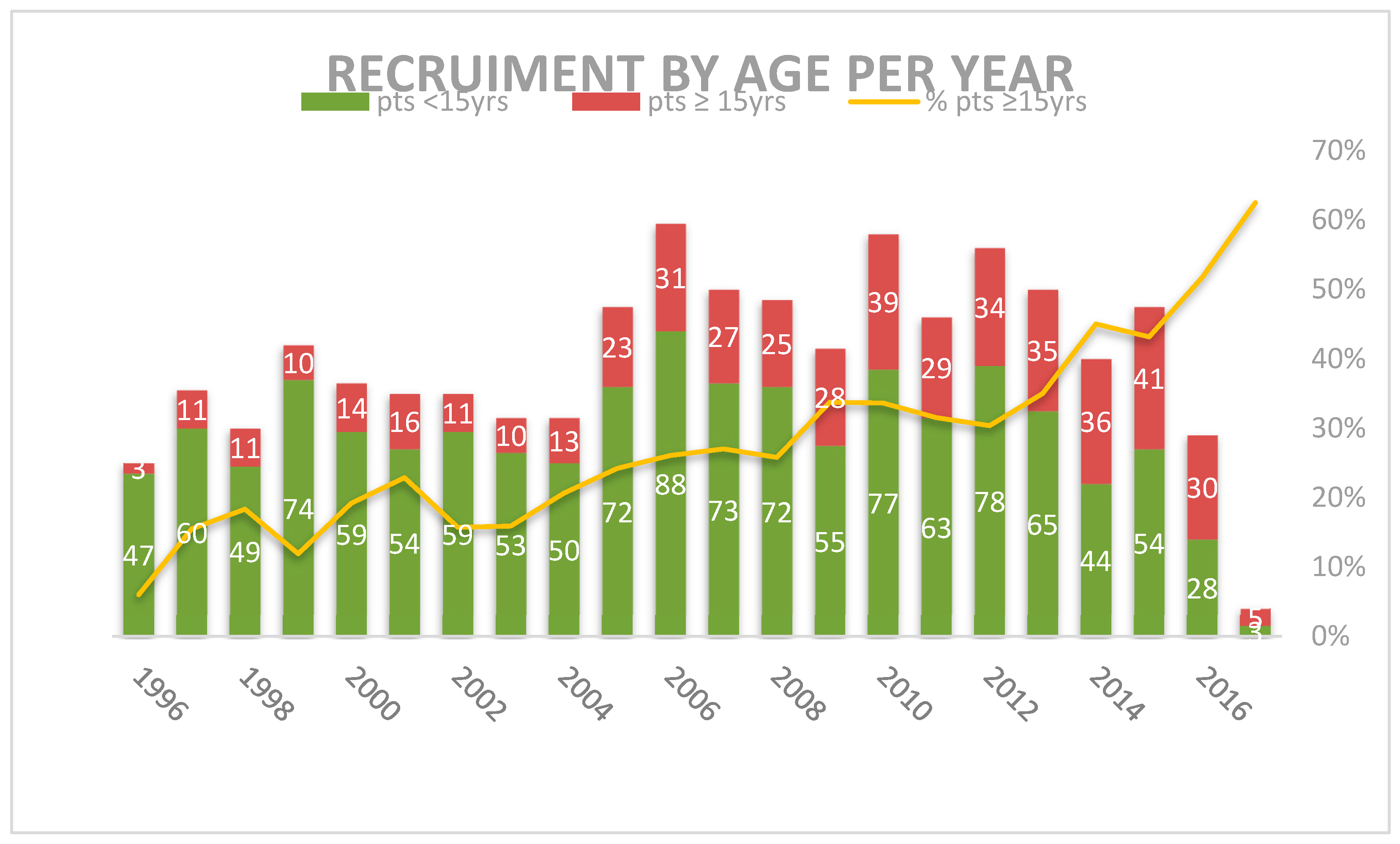
3.1. Patients Characteristics
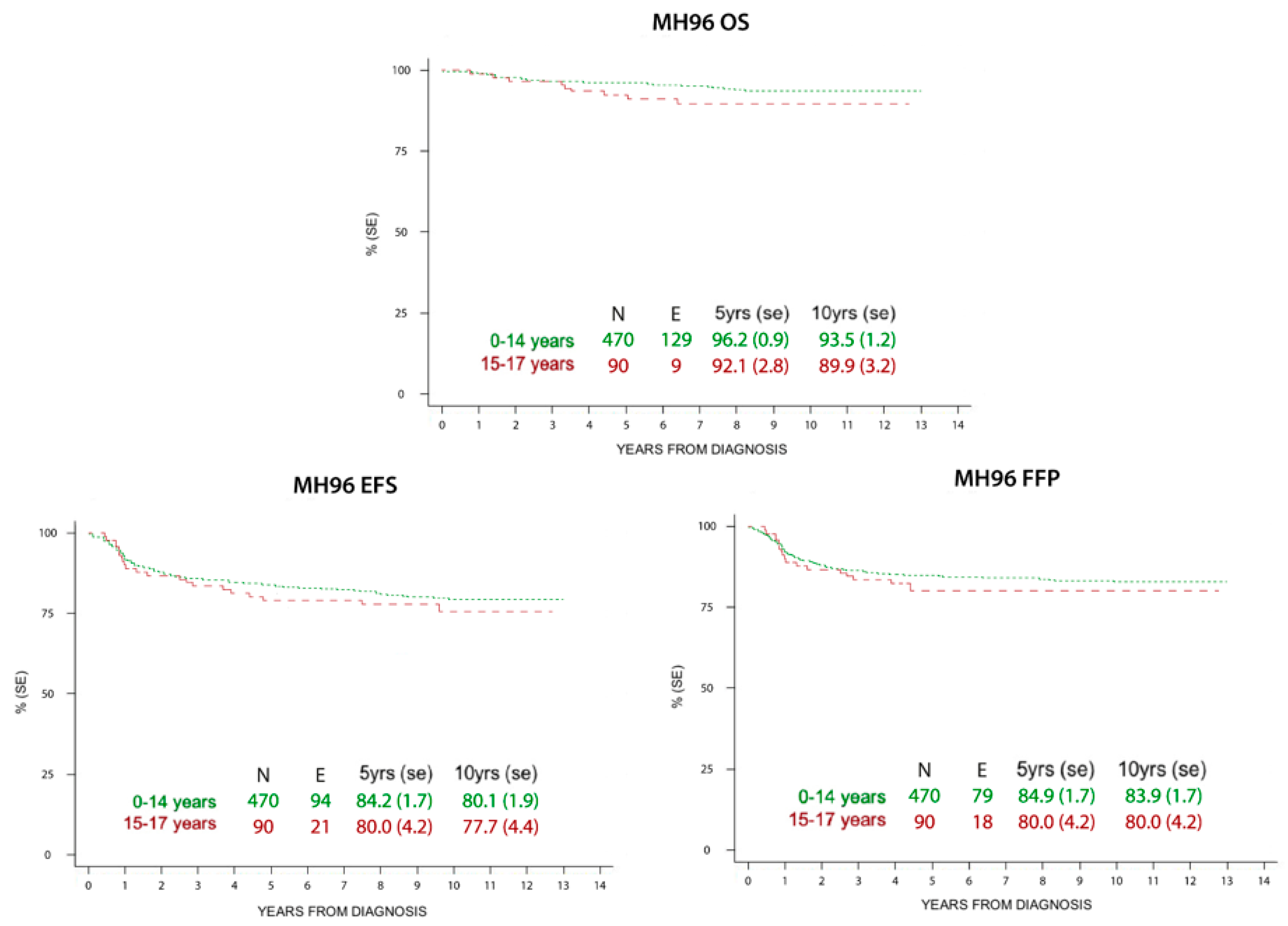
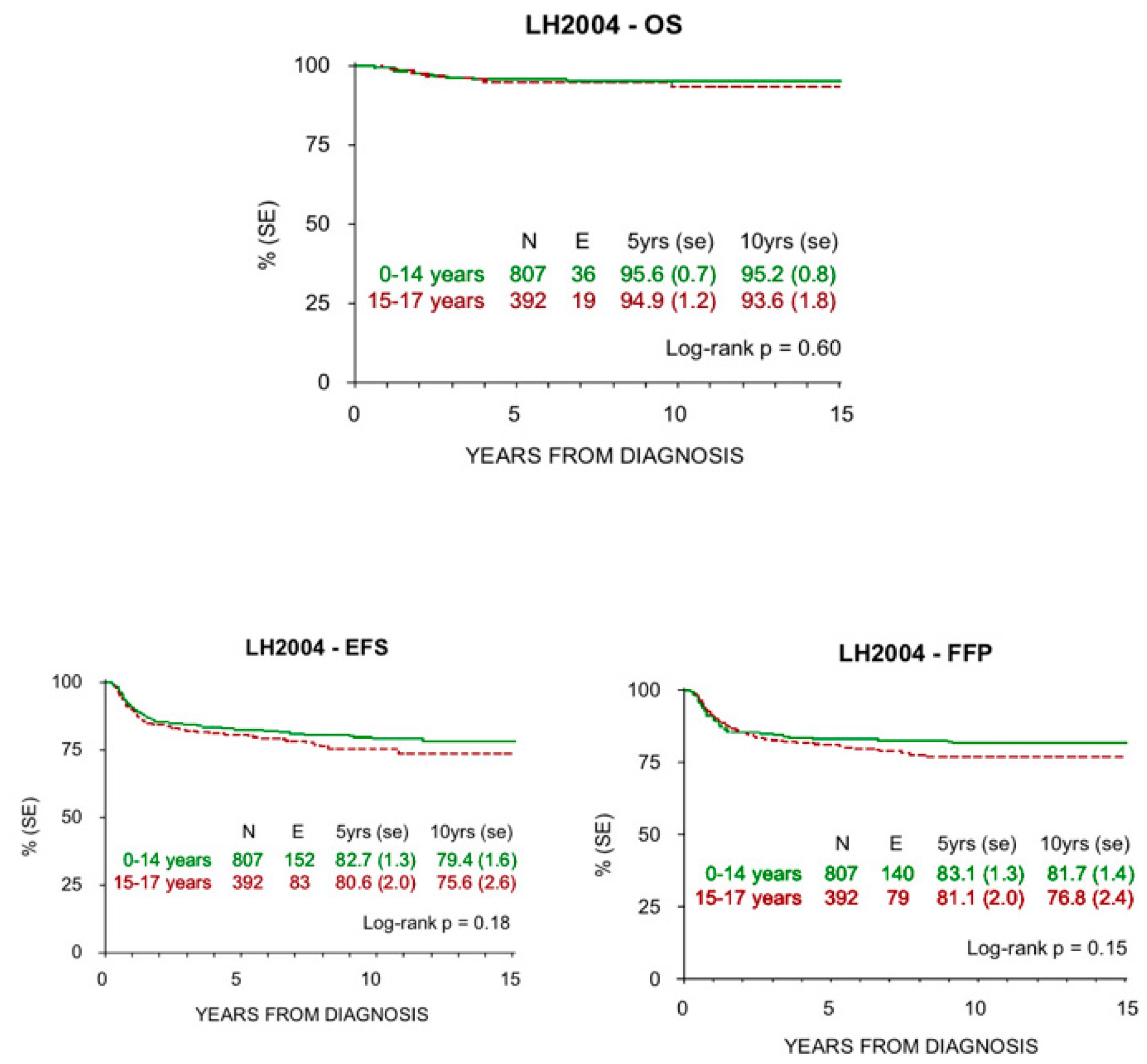
3.2. Survival
4. Discussion
5. Conclusions
6. Strengths and Limitations
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Birch, J.M.; Pang, D.; Alston, R.D.; Rowan, S.; Geraci, M.; Moran, A.; Eden, T.O.B. Survival from cancer in teenagers and young adults in England, 1979–2003. Br. J. Cancer 2008, 99, 830–835. [Google Scholar] [CrossRef] [PubMed]
- Crombie, J.L.; LaCasce, A.S. Current considerations in AYA Hodgkin lymphoma. Br. J. Haematol. 2019, 184, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Bleyer, A.; O’Leary, M.; Barr, R. Cancer Epidemiology in Older Adolescents and Young Adults 15 to 29 Years of Age, Including SEER Incidence and Survival: 1975–2000; NIH Pub. No. 06-5767; National Cancer Institute: Bethesda, MD, USA, 2006.
- Bleyer, A.; Budd, T.; Montello, M. Adolescents and young adults with cancer: The scope of the problem and criticality of clinical trials. Cancer 2006, 107, 1645–1655. [Google Scholar] [CrossRef]
- Barr, R.D.; Ferrari, A.; Ries, L.; Whelan, J.; Bleyer, W.A. Cancer in Adolescents and Young Adults: A Narrative Review of the Current Status and a View of the Future. JAMA Pediatr. 2016, 170, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Taddeo, D.; Egedy, M.; Frappier, J.-Y. Adherence to treatment in adolescents. Paediatr. Child Health 2008, 13, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Mobley, E.M.; Charlton, M.E.; Ward, M.M.; Lynch, C.F. Nonmetropolitan residence and other factors affecting clinical trial enrollment for adolescents and young adults with cancer in a US population-based study. Cancer 2019. [Google Scholar] [CrossRef] [PubMed]
- AIRTUM Working Group; CCM; AIEOP Working Group. Italian cancer figures, report 2012: Cancer in children and adolescents. Epidemiol. Prev. 2013, 37, 1–225. [Google Scholar]
- Stiller, C.A. International patterns of cancer incidence in adolescents. Cancer Treat. Rev. 2007, 33, 631–645. [Google Scholar] [CrossRef]
- Burnelli, R.; Rinieri, S.; Rondelli, R.; Todesco, A.; Bianchi, M.; Garaventa, A.; Zecca, M.; Indolfi, P.; Conter, V.; Santoro, N.; et al. Long-term results of the AIEOP MH’96 childhood Hodgkin’s lymphoma trial and focus on significance of response to chemotherapy and its implication in low risk patients to avoid radiotherapy. Leuk. Lymphoma 2018, 1–10. [Google Scholar] [CrossRef]
- Swerdlow, S.H.; Campo, E.; Pileri, S.A.; Harris, N.L.; Stein, H.; Siebert, R.; Advani, R.; Ghielmini, M.; Salles, G.A.; Zelenetz, A.D.; et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood 2016, 127, 2375–2390. [Google Scholar] [CrossRef]
- Kaplan, E.L.; Meier, P. Nonparametric Estimation from Incomplete Observations. J. Am. Stat. Assoc. 1958, 53, 457–481. [Google Scholar] [CrossRef]
- Kalbfleisch, J.D.; Prentice, R.L. The Statistical Analysis of Failure Time Data; John Wiley & Sons, Inc.: New York, NY, USA, 1980; 321p, ISBN 0-471-05519-0. [Google Scholar] [CrossRef]
- Ferrari, A.; Dama, E.; Pession, A.; Rondelli, R.; Pascucci, C.; Locatelli, F.; Ferrari, S.; Mascarin, M.; Merletti, F.; Masera, G.; et al. Adolescents with cancer in Italy: Entry into the national cooperative paediatric oncology group AIEOP trials. Eur. J. Cancer 2009, 45, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, A.; Rondelli, R.; Pession, A.; Mascarin, M.; Buzzoni, C.; Mosso, M.L.; Maule, M.; Barisone, E.; Bertolotti, M.; Clerici, C.A.; et al. Adolescents with Cancer in Italy: Improving Access to National Cooperative Pediatric Oncology Group (AIEOP) Centers. Pediatr. Blood Cancer 2016, 63, 1116–1119. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, A.; Quarello, P.; Mascarin, M.; Milano, G.M.; Tornesello, A.; Bertolotti, M.; Spinelli, M.; Ballotta, P.; Read Borghi, M.; Maule, M.; et al. Evolving Services for Adolescents with Cancer in Italy: Access to Pediatric Oncology Centers and Dedicated Projects. J. Adolesc. Young Adult Oncol. 2019. [Google Scholar] [CrossRef]
- Pieters, R.S.; Wagner, H.; Baker, S.; Morano, K.; Ulin, K.; Cicchetti, M.G.; Bishop-Jodoin, M.; FitzGerald, T.J. The impact of protocol assignment for older adolescents with hodgkin lymphoma. Front. Oncol. 2014, 4, 317. [Google Scholar] [CrossRef][Green Version]
- Reedijk, A.M.J.; Zijtregtop, E.A.M.; Coebergh, J.W.W.; Meyer-Wentrup, F.A.G.; Hebeda, K.M.; Zwaan, C.M.; Janssens, G.O.R.; Pieters, R.; Plattel, W.J.; Dinmohamed, A.G.; et al. Improved survival for adolescents and young adults with Hodgkin lymphoma and continued high survival for children in the Netherlands: A population-based study during 1990-2015. Br. J. Haematol. 2020. [Google Scholar] [CrossRef]
- Williams, L.A.; Richardson, M.; Marcotte, E.L.; Poynter, J.N.; Spector, L.G. Sex ratio among childhood cancers by single year of age. Pediatr. Blood Cancer 2019, e27620. [Google Scholar] [CrossRef]
- Landman-Parker, J.; Pacquement, H.; Leblanc, T.; Habrand, J.L.; Terrier-Lacombe, M.J.; Bertrand, Y.; Perel, Y.; Robert, A.; Coze, C.; Thuret, I.; et al. Localized Childhood Hodgkin’s Disease: Response-Adapted Chemotherapy with Etoposide, Bleomycin, Vinblastine, and Prednisone Before Low-Dose Radiation Therapy—Results of the French Society of Pediatric Oncology Study MDH90. J. Clin. Oncol. 2000, 18, 1500–1507. [Google Scholar] [CrossRef]
- Clavel, J.; Steliarova-Foucher, E.; Berger, C.; Danon, S.; Valerianova, Z. Hodgkin’s disease incidence and survival in European children and adolescents (1978-1997): Report from the Automated Cancer Information System project. Eur. J. Cancer Oxf. Engl. 1990 2006, 42, 2037–2049. [Google Scholar] [CrossRef]
- Englund, A.; Glimelius, I.; Rostgaard, K.; Smedby, K.E.; Eloranta, S.; Molin, D.; Kuusk, T.; de Brown, P.N.; Kamper, P.; Hjalgrim, H.; et al. Hodgkin lymphoma in children, adolescents and young adults—A comparative study of clinical presentation and treatment outcome. Acta Oncol. Stockh. Swed. 2017, 1–7. [Google Scholar] [CrossRef]
- Fernández, K.S.; Schwartz, C.L.; Chen, L.; Constine, L.S.; Chauvenet, A.; de Alarcón, P.A. Outcome of Adolescents and Young Adults Compared to Children with Hodgkin Lymphoma Treated with Response-Based Chemotherapy on Pediatric Protocols: A Children’s Oncology Group Report. Pediatr. Blood Cancer 2017, 64. [Google Scholar] [CrossRef] [PubMed]
- Marcos-Gragera, R.; Solans, M.; Galceran, J.; Fernández-Delgado, R.; Fernández-Teijeiro, A.; Mateos, A.; Quirós-Garcia, J.R.; Fuster-Camarena, N.; De Castro, V.; Sánchez, M.J.; et al. Childhood and adolescent lymphoma in Spain: Incidence and survival trends over 20 years. Clin. Transl. Oncol. Off. Publ. Fed. Span. Oncol. Soc. Natl. Cancer Inst. Mex. 2018. [Google Scholar] [CrossRef]
- Bigenwald, C.; Galimard, J.-E.; Quero, L.; Cabannes-Hamy, A.; Thieblemont, C.; Boissel, N.; Brice, P. Hodgkin lymphoma in adolescent and young adults: Insights from an adult tertiary single-center cohort of 349 patients. Oncotarget 2017, 8, 80073–80082. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pettit, T.; Sue, L.; Waugh, V.; Ballantine, K.; Gardner, K.; Bremer, L.; Pemberton, L.; Allison, L.; Adams, S.; Chou, E.; et al. An Age Stratified Analysis of the Access to Care Continuum Across Three Tumor Groups: Are There Delays for AYA? J. Adolesc. Young Adult Oncol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Glaser, S.L.; Clarke, C.A.; Keegan, T.H.M.; Chang, E.T.; Weisenburger, D.D. Time Trends in Rates of Hodgkin Lymphoma Histologic Subtypes: True Incidence Changes or Evolving Diagnostic Practice? Cancer Epidemiol. Prev. Biomark. 2015, 24, 1474–1488. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, C.L. The management of Hodgkin disease in the young child. Curr. Opin. Pediatr. 2003, 15, 10. [Google Scholar] [CrossRef]
- Young, L.S.; Murray, P.G. Epstein-Barr virus and oncogenesis: From latent genes to tumours. Oncogene 2003, 22, 5108–5121. [Google Scholar] [CrossRef]
- Jessica, H.; Waxman, I.M.; Kelly, K.M.; Erin, M.; Cairo, M.S. Adolescent non-Hodgkin lymphoma and Hodgkin lymphoma: State of the science. Br. J. Haematol. 2008, 144, 24–40. [Google Scholar] [CrossRef]
- Blum, K.A.; Keller, F.G.; Castellino, S.; Phan, A.; Flowers, C.R. Incidence and outcomes of lymphoid malignancies in adolescent and young adult patients in the United States. Br. J. Haematol. 2018, 183, 385–399. [Google Scholar] [CrossRef]
- Zaucha, J.M.; Chauvie, S.; Zaucha, R.; Biggii, A.; Gallamini, A. The role of PET/CT in the modern treatment of Hodgkin lymphoma. Cancer Treat. Rev. 2019, 77, 44–56. [Google Scholar] [CrossRef]
- Cistaro, A.; Cassalia, L.; Ferrara, C.; Quartuccio, N.; Evangelista, L.; Bianchi, M.; Fagioli, F.; Bisi, G.; Baldari, S.; Zanella, A.; et al. Italian Multicenter Study on Accuracy of 18F-FDG PET/CT in Assessing Bone Marrow Involvement in Pediatric Hodgkin Lymphoma. Clin. Lymphoma Myeloma Leuk. 2018, 18, e267–e273. [Google Scholar] [CrossRef] [PubMed]
- Lopci, E.; Mascarin, M.; Piccardo, A.; Castello, A.; Elia, C.; Guerra, L.; Borsatti, E.; Sala, A.; Todesco, A.; Zucchetta, P.; et al. FDG PET in response evaluation of bulky masses in paediatric Hodgkin’s lymphoma (HL) patients enrolled in the Italian AIEOP-LH2004 trial. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Satish, S.; Ambinder, R.F. Hodgkin lymphoma: A review and update on recent progress. CA Cancer J. Clin. 2018, 68, 116–132. [Google Scholar] [CrossRef]
- Yung, L.; Smith, P.; Hancock, B.W.; Hoskin, P.; Gilson, D.; Vernon, C.; Linch, D.C. Long Term Outcome in Adolescents with Hodgkin’s Lymphoma: Poor Results using Regimens Designed for Adults. Leuk. Lymphoma 2004, 45, 1579–1585. [Google Scholar] [CrossRef]
- Herbertson, R.A.; Evans, L.S.; Hutchinson, J.; Horsman, J.; Hancock, B.W. Poor outcome in adolescents with high-risk Hodgkin lymphoma. Int. J. Oncol. 2008, 33, 145–151. [Google Scholar] [CrossRef]
- Rodriguez, L.; Research Fellow, Division of Haematology/Oncology, Department of Paediatrics, SickKids Hospital and University of Toronto; Punnett, A.; Fellowship Program Training Director, Division of Haematology/Oncology, Department of Paediatrics, SickKids Hospital and University of Toronto. Canada Hodgkin’s Lymphoma in Adolescents and Young Adults. Oncol. Hematol. Rev. US 2012, 8, 116. [Google Scholar] [CrossRef][Green Version]
- Koumarianou, A.; Xiros, N.; Papageorgiou, E.; Pectasides, D.; Economopoulos, T. Survival improvement of young patients, aged 16-23, with Hodgkin lymphoma (HL) during the last three decades. Anticancer Res. 2007, 27, 1191–1197. [Google Scholar]
- Eichenauer, D.A.; Bredenfeld, H.; Haverkamp, H.; Müller, H.; Franklin, J.; Fuchs, M.; Borchmann, P.; Müller-Hermelink, H.-K.; Eich, H.T.; Müller, R.-P.; et al. Hodgkin’s lymphoma in adolescents treated with adult protocols: A report from the German Hodgkin study group. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2009, 27, 6079–6085. [Google Scholar] [CrossRef]
- Foltz, L.M.; Song, K.W.; Connors, J.M. Hodgkin’s lymphoma in adolescents. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2006, 24, 2520–2526. [Google Scholar] [CrossRef]
- Müller, J.; Illés, A.; Molnár, Z.; Rosta, A.; Váróczy, L.; Kovács, G. Adolescent hodgkin lymphoma: Are treatment results more favorable with pediatric than with adult regimens? J. Pediatr. Hematol. Oncol. 2011, 33, e60–e63. [Google Scholar] [CrossRef]
- Henderson, T.O.; Parsons, S.K.; Wroblewski, K.E.; Chen, L.; Hong, F.; Smith, S.M.; McNeer, J.L.; Advani, R.H.; Gascoyne, R.D.; Constine, L.S.; et al. Outcomes in adolescents and young adults with Hodgkin lymphoma treated on US cooperative group protocols: An adult intergroup (E2496) and Children’s Oncology Group (COG AHOD0031) comparative analysis. Cancer Cytopathol. 2018, 124, 136–144. [Google Scholar] [CrossRef] [PubMed]
- FitzGerald, T.J.; Bishop-Jodoin, M. Hodgkin Lymphoma: Differences in Treatment Between Europe and the United States/North America: Evolving Trends in Protocol Therapy. Clin. Med. Insights Oncol. 2018, 12, 1179554918754885. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Baxter, N.N.; Hodgson, D.; Punnett, A.; Sutradhar, R.; Pole, J.D.; Nagamuthu, C.; Lau, C.; Nathan, P.C. Treatment patterns and outcomes in adolescents and young adults with Hodgkin lymphoma in pediatric versus adult centers: An IMPACT Cohort Study. Cancer Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Cramer, P.; Andrieu, J.M. Hodgkin’s disease in childhood and adolescence: Results of chemotherapy-radiotherapy in clinical stages IA-IIB. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 1985, 3, 1495–1502. [Google Scholar] [CrossRef]
- Schellong, G.; Pötter, R.; Brämswig, J.; Wagner, W.; Prott, F.J.; Dörffel, W.; Körholz, D.; Mann, G.; Rath, B.; Reiter, A.; et al. High cure rates and reduced long-term toxicity in pediatric Hodgkin’s disease: The German-Austrian multicenter trial DAL-HD-90. The German-Austrian Pediatric Hodgkin’s Disease Study Group. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 1999, 17, 3736–3744. [Google Scholar] [CrossRef]
- Mauz-Körholz, C.; Hasenclever, D.; Dörffel, W.; Ruschke, K.; Pelz, T.; Voigt, A.; Stiefel, M.; Winkler, M.; Vilser, C.; Dieckmann, K.; et al. Procarbazine-free OEPA-COPDAC chemotherapy in boys and standard OPPA-COPP in girls have comparable effectiveness in pediatric Hodgkin’s lymphoma: The GPOH-HD-2002 study. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2010, 28, 3680–3686. [Google Scholar] [CrossRef]
- Raze, T.; Lacour, B.; Cowppli-Bony, A.; Delafosse, P.; Velten, M.; Trétarre, B.; Defossez, G.; Hammas, K.; Woronoff, A.-S.; Ganry, O.; et al. Cancer Among Adolescents and Young Adults Between 2000 and 2016 in France: Incidence and Improved Survival. J. Adolesc. Young Adult Oncol. 2020. [Google Scholar] [CrossRef]
- Dony, A.; Belhabri, A.; Bertrand, Y.; Sebban, C.; Cony-Makhoul, P.; Sobh, M.; Rogasik, M.; Salles, G.; Anglaret, B.; Freycon, C.; et al. Pattern of Care and Outcomes of Adolescent and Young Adults with Lymphoma Treated in the Rhône-Alpes Region. J. Adolesc. Young Adult Oncol. 2019, 8, 684–696. [Google Scholar] [CrossRef]
- Weiner, M.A.; Leventhal, B.; Brecher, M.L.; Marcus, R.B.; Cantor, A.; Gieser, P.W.; Ternberg, J.L.; Behm, F.G.; Wharam, M.D.; Chauvenet, A.R. Randomized study of intensive MOPP-ABVD with or without low-dose total-nodal radiation therapy in the treatment of stages IIB, IIIA2, IIIB, and IV Hodgkin’s disease in pediatric patients: A Pediatric Oncology Group study. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 1997, 15, 2769–2779. [Google Scholar] [CrossRef]
- Zawati, I.; Adouni, O.; Finetti, P.; Manai, M.; Manai, M.; Gamoudi, A.; Birnbaum, D.; Bertucci, F.; Mezlini, A. Adolescents and young adults with classical Hodgkin lymphoma in North Tunisia: Insights from an adult single-institutional study. Cancer Radiother. J. Soc. Francaise Radiother. Oncol. 2020. [Google Scholar] [CrossRef]
- Anderson, C.; Lund, J.L.; Weaver, M.A.; Wood, W.A.; Olshan, A.F.; Nichols, H.B. Noncancer mortality among adolescents and young adults with cancer. Cancer 2019. [Google Scholar] [CrossRef] [PubMed]
- Hahn, E.E.; Wu, Y.-L.; Munoz-Plaza, C.E.; Garcia Delgadillo, J.; Cooper, R.M.; Chao, C.R. Use of recommended posttreatment services for adolescent and young adult survivors of Hodgkin lymphoma. Cancer 2019, 125. [Google Scholar] [CrossRef] [PubMed]
- Cepelova, M.; Kruseova, J.; Luks, A.; Capek, V.; Cepela, P.; Potockova, J.; Kraml, P. Accelerated atherosclerosis, hyperlipoproteinemia and insulin resistance in long-term survivors of Hodgkin lymphoma during childhood and adolescence. Neoplasma 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Glimelius, I.; Englund, A.; Rostgaard, K.; Smedby, K.E.; Eloranta, S.; de Nully Brown, P.; Johansen, C.; Kamper, P.; Ljungman, G.; Hjalgrim, L.L.; et al. Distribution of hospital care among pediatric and young adult Hodgkin lymphoma survivors-A population-based cohort study from Sweden and Denmark. Cancer Med. 2019. [Google Scholar] [CrossRef]
- Jachimowicz, R.D.; Engert, A. The Challenging Aspects of Managing Adolescents and Young Adults with Hodgkin’s Lymphoma. Acta Haematol. 2014, 132, 274–278. [Google Scholar] [CrossRef] [PubMed]
- Aleman, B.M.P.; van den Belt-Dusebout, A.W.; Klokman, W.J.; Van’t Veer, M.B.; Bartelink, H.; van Leeuwen, F.E. Long-term cause-specific mortality of patients treated for Hodgkin’s disease. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2003, 21, 3431–3439. [Google Scholar] [CrossRef]
- Xavier, A.C.; Epperla, N.; Taub, J.W.; Costa, L.J. Excess mortality among 10-year survivors of classical Hodgkin lymphoma in adolescents and young adults. Am. J. Hematol. 2018, 93, 238–245. [Google Scholar] [CrossRef]
- Reulen, R.C.; Frobisher, C.; Winter, D.L.; Kelly, J.; Lancashire, E.R.; Stiller, C.A.; Pritchard-Jones, K.; Jenkinson, H.C.; Hawkins, M.M.; British Childhood Cancer Survivor Study Steering Group. Long-term risks of subsequent primary neoplasms among survivors of childhood cancer. JAMA 2011, 305, 2311–2319. [Google Scholar] [CrossRef]
- Van Leeuwen, F.E.; Ng, A.K. Long-term risk of second malignancy and cardiovascular disease after Hodgkin lymphoma treatment. Hematology 2016, 2016, 323–330. [Google Scholar] [CrossRef]
- O’Brien, M.M.; Donaldson, S.S.; Balise, R.R.; Whittemore, A.S.; Link, M.P. Second malignant neoplasms in survivors of pediatric Hodgkin’s lymphoma treated with low-dose radiation and chemotherapy. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2010, 28, 1232–1239. [Google Scholar] [CrossRef]
- Krishnan, B.; Morgan, G.J. Non-Hodgkin lymphoma secondary to cancer chemotherapy. Cancer Epidemiol. Biomark. Prev. Publ. Am. Assoc. Cancer Res. Cosponsored Am. Soc. Prev. Oncol. 2007, 16, 377–380. [Google Scholar] [CrossRef] [PubMed]
- Rueffer, U.; Josting, A.; Franklin, J.; May, M.; Sieber, M.; Breuer, K.; Engert, A.; Diehl, V.; German Hodgkin’s Lymphoma Study Group. Non-Hodgkin’s lymphoma after primary Hodgkin’s disease in the German Hodgkin’s Lymphoma Study Group: Incidence, treatment, and prognosis. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2001, 19, 2026–2032. [Google Scholar] [CrossRef] [PubMed]
- Keegan, T.H.M.; Ries, L.A.G.; Barr, R.D.; Geiger, A.M.; Dahlke, D.V.; Pollock, B.H.; Bleyer, W.A.; National Cancer Institute Next Steps for Adolescent and Young Adult Oncology Epidemiology Working Group. Comparison of cancer survival trends in the United States of adolescents and young adults with those in children and older adults. Cancer 2016, 122, 1009–1016. [Google Scholar] [CrossRef]
- Karim-Kos, H.E.; Hackl, M.; Mann, G.; Urban, C.; Woehrer, A.; Slavc, I.; Ladenstein, R. Trends in incidence, survival and mortality of childhood and adolescent cancer in Austria, 1994–2011. Cancer Epidemiol. 2016, 42, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Stark, D.; Bowen, D.; Dunwoodie, E.; Feltbower, R.; Johnson, R.; Moran, A.; Stiller, C.; O’Hara, C. Survival patterns in teenagers and young adults with cancer in the United Kingdom: Comparisons with younger and older age groups. Eur. J. Cancer Oxf. Engl. 2015, 51, 2643–2654. [Google Scholar] [CrossRef] [PubMed]
| Treatment Groups | MH’96 | LH2004 |
|---|---|---|
| 1 IA, IIA supradiaphragmatic no bulky no bulky, no pulmonary hilum, <4 lymphatic sites, or IA, IIA infradiaphragmatic < 4 lymphatic sites | 3× ABVD +: -CR + no initial mediastinal involvement: stop -others: RT -CR o PR ≥ 75%: 20 Gy IF -PR < 75%: 36 Gy IF | 3× ABVD +: -CR: stop -others: RT 25.2 Gy IF |
| 2 patients not included in groups 1 or 3 | 4× COPP/ABV + RT: -CR or PR ≥ 75%: 20 Gy IF -PR < 75%: 36 Gy IF | 4× COPP/ABV +: -CR: RT 14.4 Gy IF -PR: 2× IEP + RT -CR: 14.4 Gy IF -PR: 25.2 Gy IF |
| 3 IIIB, IV; M/T ≥ 0.33 all stages | 6× COPP/ABV + RT: -CR or PR ≥ 75%: 20 Gy IF -PR < 75%: 36 Gy IF | 4× COPP/ABV +: -CR: 2× COPP/ABV + RT 14.4 Gy IF -PR: 2× IEP + RT -CR: 14.4 Gy IF -PR: 25.2 Gy IF |
| If RP ≤ 50% after 2° Cycle: GR1: IEP/OPPA/COPP/IEP + RT; GR2 & 3: IEP/OPPA/IEP/OPPA/IEP + RT | ||
| ABVD Doxorubicin: 25 mg/m2 IV days 1 Bleomycin: 10 mg/m2 IV days 1 & 15 Vinblastine: 6 mg/m2 IV days 1 &.15 DTIC: 375 mg/m2 IV days 1 & 15 | COPP/ABV Cyclophosphamide: 600 mg/m2 IV day 1 Vincristine: 1.4 mg/m2 IV day 1 Prednisone: 40 mg/m2 orally days 1–14 Procarbazine: 100 mg/m2 orally days 1–7 Doxorubicin: 35 mg/m2 IV day 8 Bleomycin: 10 mg/m2 IV day 8 Vinblastine: 6 mg/m2 IV day 8 | IEP Ifosfamide: 2000 mg/m2 IV days 1–5 Etoposide: 120 mg/m2 IV days 1–5 Prednisone: 100 mg/m2 orally days 1–5 |
| OPPA Vincristine: 1.5 mg/m2 IV days 1& 8 & 15 Procarbazine: 100 mg/m2 orally days 1–14 Prednisone: 60 mg/m2 IV days 1–14 Doxorubicin: 40 mg/m2 IV days 1&15 | COPP Cyclophosphamide: 500 mg/m2 IV days 1 & 8 Vincristine: 1.5 mg/m2 IV days 1 & 8 Procarbazine: 100 mg/m2 orally days 1–15 Prednisone: 40 mg/m2 orally days 1–15 | |
| MH-‘96 | LH-2004 | |||||
|---|---|---|---|---|---|---|
| Children (<15 years) N (%) | Adolescents (15–18 years) N (%) | p Value | Children (<15 years) N (%) | Adolescents (15–18 years) N (%) | p Value | |
| Number | 470 (83.9) | 90 (16.1) | 807 (67.1) | 392 (32.9) | p = 0.000 1 | |
| Sex | p = 0.429 | p = 0.001 | ||||
| Males | 277 (59.0) | 49 (54.4) | 470 (58.2) | 189 (48.2) | ||
| Females | 193 (41.0) | 41 (45.6) | 337 (41.8) | 203 (51.8) | ||
| Histology | all vs. all (except n.c.): p = 0.024 | all vs. all (except n.c.): p = 0.006 | ||||
| Classic | ||||||
| Nodular Sclerosis | 304 (64.7) | 72 (80.0) | 589 (73) | 317 (80.9) | ||
| Mixed Cellularity | 106 (22.6) | 12 (13.3) | 79 (9.8) | 25 (6.4) | ||
| Lymphocyte-Rich | 2 (0.4) | 11 (1.4) | 3 (0.8) | |||
| Lymphocyte Depletion | 3 (0.6) | 8 (1.0) | 2 (0.5) | |||
| NOS | 26 (3.2) | 14 (3.6) | ||||
| Nodular Lymphocyte Predominance | 51 (10.9) | 3 (3.3) | 73 (9.0) | 16 (4.0) | ||
| n.c. | 4 (0.8) | 3 (3.3) | 21 (2.6) | 15 (3.8)) | ||
| Stage | p = 0.259 | p = 0.066 | ||||
| I | 62 (13.2) | 8 (9.0) | 45 (5.6) | 10 (2.5) | ||
| II | 222 (47.2) | 52 (57.7) | 405 (50.2) | 216 (55.1) | ||
| III | 99 (21.1) | 18 (20.0) | 186 (23) | 81 (20.7) | ||
| IV | 87 (18.5) | 12 (13.3) | 171 (21.2) | 85 (21.7) | ||
| Symptoms | p = 0.041 | p = 0.000 | ||||
| A | 309 (65.7) | 49 (54.4) | 503 (62.3) | 202 (51.5) | ||
| B | 161 (34.3) | 41 (45.6) | 304 (37.7) | 190 (48.5) | ||
| Mediastinum Bulky | p = 0.106 | p = 0.022 | ||||
| Yes | 202 (43.0) | 47 (52.2) | 351 (43.5) | 198 (50.5) | ||
| No | 268 (57.0) | 43 (47.8) | 456 (56.5) | 194 (49.5) | ||
| Extra-Nodal Involvement | p = 0.428 | p = 0.304 | ||||
| Yes | 101 (21.5) | 16 (17.8) | 208 (25.8) | 112 (28.6) | ||
| No | 369 (78.5) | 74 (82.2) | 599 (74.2) | 280 (71.4) | ||
| Treatment Group | p = 0.075 | p = 0.000 | ||||
| 1 | 143 (30.4) | 17 (19.0) | 146(18.1) | 35 (8.9) | ||
| 2 | 71 (15.1) | 14 (15.5) | 176 (21.8) | 97 (24.8) | ||
| 3 | 256 (54.5) | 59 (65.5) | 485 (60.1) | 260 (66.3) | ||
| RT in Group 1 | p = 0.048 2 | p = 0.247 | ||||
| Yes | 81 (56.6) | 14 (82.4) | 37 (25.3) | 13 (37.1) | ||
| No | 60 (42.0) | 3 (17.6) | 95 (65.1) | 21 (60) | ||
| Missing | 2 (1.4) | 14 (9.6) | 1 (2.9) | |||
| Time to PD/Relapses | p = 0.73 | p = 0.051 | ||||
| Number | 75 | 17 | 137 | 78 | ||
| Mean (Months) | 22.3 | 20.7 | 17.9 | 20.6 | ||
| [Range] | [2–128] | [6–57] | [2.5–110] | [2.6–99] | ||
| Median (Months) | 13.3 | 12.8 | 11.9 | 12.1 | ||
| Children (<15 years) | Adolescents (15–18 years) | |||||
|---|---|---|---|---|---|---|
| MH-’96 N (%) | LH-2004 N (%) | p Value | MH-’96 N (%) | LH-2004 N (%) | p Value | |
| Number | 470 (36.8) | 807 (63.2) | 90 (18.6) | 392 (81.4) | ||
| Sex | p = 0.808 | p = 0.286 | ||||
| Males | 277 (59.0) | 470 (58.2) | 49 (54.4) | 189 (48.2) | ||
| Females | 193 (41.0) | 337 (41.8) | 41 (45.6) | 203 (51.8) | ||
| Histology | All vs. all (except n.c.): p = 0.000 | All vs. all (except n.c.): p = 0.107 | ||||
| Classic | ||||||
| Nodular Sclerosis | 304 (64.7) | 589 (73) | 72 (80.0) | 317 (80.9) | ||
| Mixed Cellularity | 106 (22.6) | 79 (9.8) | 12 (13.3) | 25 (6.4) | ||
| Lymphocytes Rich | 2 (0.4) | 11 (1.4) | 3 (0.8) | |||
| Lymphocyte Depletion | 3 (0.6) | 8 (1.0) | 2 (0.5) | |||
| NOS | 26 (3.2) | 14 (3.6) | ||||
| Nodular Lymphocyte Predominance | 51 (10.9) | 73 (9.0) | 3 (3.3) | 16 (4.0) | ||
| n.c. | 4 (0.8) | 21 (2.6) | 3 (3.3) | 15 (3.8) | ||
| Stage | p = 0.000 | p = 0.015 | ||||
| I | 62 (13.2) | 45 (5.6) | 8 (9.0) | 10 (2.5) | ||
| II | 222 (47.2) | 405 (50.2) | 52 (57.7) | 216 (55.1) | ||
| III | 99 (21.1) | 186 (23) | 18 (20) | 81 (20.7) | ||
| IV | 87 (18.5) | 171 (21.2) | 12 (13.3) | 85 (21.7) | ||
| Symptoms | p = 0.221 | p = 0.618 | ||||
| A | 309 (65.7) | 503 (62.3) | 49 (54.4) | 202 (51.5) | ||
| B | 161 (34.3) | 304 (37.7) | 41 (45.6) | 190 (48.5) | ||
| Mediastinum Bulky | p = 0.858 | p = 0.770 | ||||
| Yes | 202 (43) | 351 (43.5) | 47 (52.2) | 198 (50.5) | ||
| No | 268 (57) | 456 (56.5) | 43 (47.8) | 194 (49.5) | ||
| Extra-nodal Involvement | p = 0.085 | p = 0.001 | ||||
| Yes | 101 (21.5) | 208 (25.8) | 16 (17.8) | 112 (28.6) | ||
| No | 369 (78.5) | 599 (74.2) | 74 (82.2) | 280 (71.4) | ||
| Therapeutic Group | p = 0.000 | p = 0.009 | ||||
| 1 | 143 (30.4) | 146 (18.1) | 17 (19.0) | 35 (8.9) | ||
| 2 | 71 (15.1) | 176 (21.8) | 14 (15.5) | 97 (24.8) | ||
| 3 | 256 (54.5) | 485 (60.1) | 59 (65.5) | 260 (66.3) | ||
| Radiotherapy in Group 1 | p = 0.000 | p = 0.003 * | ||||
| Yes | 81 (56.6) | 37 (25.3) | 14 (82.4) | 13 (37.1) | ||
| No | 60 (42) | 95 (65.1) | 3 (17.6) | 21 (60) | ||
| Missing | 2 (1.4) | 14 (9.6) | 1 (2.9) | |||
| Time to PD/Relapse | p = 0.012 | p = 0.90 | ||||
| Number | 75 | 137 | 17 | 78 | ||
| Mean (Months) | 22.3 | 17.9 | 20.7 | 20.6 | ||
| [Range] | [2–128] | [2.5–110] | [6–57] | [2.6–99] | ||
| Median (Months) | 13.3 | 11.9 | 12.8 | 12.1 | ||
| GROUP 1 | GROUP 2 | GROUP 3 | ||||
|---|---|---|---|---|---|---|
| at 5 years | at 10 years | at 5 years | at 10 years | at 5 years | at 10 years | |
| MH96 | ||||||
| <15 years | 98.6% | 97.8% | 98.6% | 98.0% | 94.1% | 89.7% |
| ≥15 years | 100% | 100% | 84.6% | 84.6% | 91.5% | 88.0% |
| LH2004 | ||||||
| <15 years | 100% | 100% | 98.2% | 97.0% | 93.3% | 93.0% |
| ≥15 years | 100% | 100% | 97.6% | 97.6% | 93.2% | 91.2% |
| Study | Country | Years | Patients N° | Treatment Protocol Pediatric/Adult, N | 5 y %OS | 10 y %OS | %EFS |
|---|---|---|---|---|---|---|---|
| Significant Worse Prognosis in Adolescents vs. Children or Adults or no Improvement over Time | |||||||
| Weiner, 1997 [51] | US | 1987–1992 | Children (0–13 y) 76 Adolescents (14–20 y) 103 | Pediatric POG 8725 | - | - | 5 y: 89 5 y: 72 |
| Clavel, 2006 [21] | Europe | 1978–1997 | Children (0–14 y) 3528 Adolescents (15–19 y) 1862 | - | 1978–82: 87, 1983–87: 91, 1988–92: 93, 1993–97: 93 1978–82: 80, 1983–87: 81, 1988–92: 90, 1993–97: 88 | - | - |
| Herbertson, 2008 [37] | UK | 1969–1998 | Adolescents (15–19 y) 63 Young Adults (20–25 y) 82 | Adult | 85.4 91.4 | 20 y: 76.3 20 y: 86.9 | 5 y: 59.9, 20 y: 56.1 5 y: 69.7, 20 y: 54.6 |
| Keegan, 2016 [65] | US | 2002–2006 | Children (0–14 y) 234 AYAs (15–39 y) 2651 | - | 96.2 94.6 | ||
| Karim-Kos, 2016 [66] | Austria | 1994–2008 | Children (0–14 y) 144 Adolescents (15–19 y) 244 | - | 1994–98: 95.9, 1999–03: 94.7, 2004–08:100 1994–98: 97, 1999–03: 94.6, 2004–08: 91.7 | - | - |
| Marcos-Gragera, 2018 [24] | Spain | 1983–2007 | Children (0–14 y) 327 Adolescents (15–19 y) 454 | - | 1991–95: 99, 1996–00: 86, 2001–05: 96 1991–95: 93, 1996–00: 91, 2001–05: 93 | - | - |
| Significant Better Prognosis in Adolescents Treated in Pediatric Centers vs. Adult Centers | |||||||
| Müller, 2011 [42] | Hungary | 1990–2004 | Adolescents (14–18 y) 155 | Adult, 48 Pediatric, 107 | 89.4 92.8 | 83.1 89.6 | 5 y: 69.6, 10 y: 59.1 5 y: 82.4, 10 y: 82.4 |
| Henderson, 2018 [43] | US | 1996–2009 | AYAs (17–21 y) 505 | Adult E2496, 114 Pediatric COG AHOD0031, 391 | 89 97 | - | - |
| No Statistical Difference in Survival between Adolescents vs. Children or Pediatric vs. Adult Treatment | |||||||
| Cramer&Andieu, 1985 [46] | France | 1972–1980 | Children (0–14 y) 32 Adolescents (15–19 y) 40 | Adult H 72 and H 77 | - | 12 y: 90.6 12 y: 92.4 | - |
| Schellong, 1999 [47] | Germany | 1990–1995 | <10 155 10–15 279 15–18 136 | Pediatric DAL-HD-90 | 97 98 97 | - | 92 91 92 |
| Landman-Parker, 2000 [20] | France | 1990–1996 | Children (0–18 y) 202 | Pediatric MDH90 | 97.5 | - | 5 y: 91.1 |
| Mauz-Körholz, 2010 [48] | Germany | 2002–2005 | Children (0–12 y) 169 Adolescents (13–18 y) 404 | Pediatric GPOH-HD-2002 | 98.2 97 | - | 5 y: 93.95 y: 86.9 |
| Stark, 2015 [67] | UK | 2001–2005 | Children (0–12 y) 226 AYAs (13–124 y) 1494 | - | 95 94 | - | - |
| Bigenwald, 2017 [25] | France | 1979–2013 | Adolescents (15–21 y) 176 Young Adults (21–25 y) 173 | Adult | >90 >90 | >90 >90 | - |
| Englund, 2017 [22] | Denmark and Sweden | 1990–2010 | Children (0–9 y) 55 Adolescents (10–17 y) 364 Young Adults (18–24 y) 653 | Pediatric, 315 Adult, 757 | - | D 93, S 95 D 95, S 95 | 10 y: D 79, S 88 10 y: D 85, S 88 |
| Fernández, 2017 [23] | US | 1996–2001 | Children (0–14 y) 268 Adolescents (15–21 y) 203 | Pediatric P9425 and P9426 | P9425: 95.4, P9426: 97.4 P9425: 92.9, P9426: 97.9 | - | 5 y: 87.1 5 y: 85.9 |
| Dony, 2019 [50] | Rhône-Alpes | 2000–2005 | AYAs (13–25 y) 198 | Pediatric, 49 Adult, 149 | 98 95 | - | - |
| Raze, 2020 [49] | France | 2000–2016 | Adolescents (15–19 y) 557 Young Adults (20–24 y) 703 | - | 2000–07: 97.6, 2008–15: 98 2000–07: 95.8, 2008–15: 95.5 | - | - |
| Zawati, 2020 [52] | Tunisia | 2000–2015 | Adolescents (15–25 y) 29 Young Adults (25–39 y) 37 | Adult | 84% 71% | - | 5 y: 48% 5 y: 54% |
| Gupta, 2020 [45] | Canada | 1992–2012 | AYAs (15–21 y) 954 | Pediatric, 243 Adult, 711 | >90 >90 | >90 >90 | 10 y: 83.8 10 y: 82.8 |
| Burnelli, 2020 | Italy | 1996–2004 | Children (0–14 y) 470 Adolescents (15–18) 90 | Pediatric AIEOP MH’96 | 96.2 92.1 | 93.5 89.9 | 5 y: 84.2, 10 y: 80.1 5 y: 80.0 10 y: 77.7 |
| Burnelli, 2020 | Italy | 2004–2017 | Children (0–14 y) 807 Adolescents (15–18) 392 | Pediatric AIEOP LH2004 | 95.6 94.9 | 95.2 93.6 | 5 y: 82.7 10 y: 79.4 5 y: 80.6 10 y: 75.6 |
| Significant Improvement of Adolescents’ Survival over Time | |||||||
| Koumarianou, 2007 [39] | Greece | 1978–2003 | AYAs (16–23 y) 55 | 1978–87 MOPP; 17 1988–93 MOPP/ABVD; 20 1994–03 ABVD or BEACOPP; 18 | 65 80 100 | - - - | 5 y: 53 5 y: 65 5 y: 88.5 |
| Reedijk, 2020 [18] | Netherlands | 1990–2015 | Children (0–14 y) 436 Adolescents (15–17 y) 490 Young Adults (18–24 y) 1693 | Pediatric and Adult | 1990–94:93 to 2010–15: 98 1990–94: 84 to 2010–15: 96 1990–94: 90 to 2010–15: 98 | 1990–94: 80 to 2005–09: 95 1990–94: 88 to 2005–09: 94 | |
| Other Studies Reporting Survival Data in Adolescents | |||||||
| Yung, 2004 [36] | UK | 1970–1997 | Adolescents (15–17 y) 209 | Adult | 81 | 76, 20 y: 68 | 5 y: 50, 10 y: 45, 20 y: 41 |
| Foltz, 2006 [41] | Canada | 1981–2004 | Adolescents (16–21 y) 259 Young Adults (22–45 y) 890 | Adult | 94 95 | 91, 20 y: 85 89, 20 y: 81 | - |
| Eichenauer, 2009 [40] | Germany | 1988–1998 | Adolescents (15–20 y) 557 Young Adults (21–45 y) 3228 | Adult GHSG HD4–9 | 6 y: 93.6 6 y: 90.9 | 12 y: 92.3 12 y: 87.1 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burnelli, R.; Fiumana, G.; Rondelli, R.; Pillon, M.; Sala, A.; Garaventa, A.; D’Amore, E.S.G.; Sabattini, E.; Buffardi, S.; Bianchi, M.; et al. Comparison of Hodgkin’s Lymphoma in Children and Adolescents. A Twenty Year Experience with MH’96 and LH2004 AIEOP (Italian Association of Pediatric Hematology and Oncology) Protocols. Cancers 2020, 12, 1620. https://doi.org/10.3390/cancers12061620
Burnelli R, Fiumana G, Rondelli R, Pillon M, Sala A, Garaventa A, D’Amore ESG, Sabattini E, Buffardi S, Bianchi M, et al. Comparison of Hodgkin’s Lymphoma in Children and Adolescents. A Twenty Year Experience with MH’96 and LH2004 AIEOP (Italian Association of Pediatric Hematology and Oncology) Protocols. Cancers. 2020; 12(6):1620. https://doi.org/10.3390/cancers12061620
Chicago/Turabian StyleBurnelli, Roberta, Giulia Fiumana, Roberto Rondelli, Marta Pillon, Alessandra Sala, Alberto Garaventa, Emanuele S.G. D’Amore, Elena Sabattini, Salvatore Buffardi, Maurizio Bianchi, and et al. 2020. "Comparison of Hodgkin’s Lymphoma in Children and Adolescents. A Twenty Year Experience with MH’96 and LH2004 AIEOP (Italian Association of Pediatric Hematology and Oncology) Protocols" Cancers 12, no. 6: 1620. https://doi.org/10.3390/cancers12061620
APA StyleBurnelli, R., Fiumana, G., Rondelli, R., Pillon, M., Sala, A., Garaventa, A., D’Amore, E. S. G., Sabattini, E., Buffardi, S., Bianchi, M., Vinti, L., Zecca, M., Muggeo, P., Provenzi, M., Farruggia, P., Rossi, F., D’Amico, S., Facchini, E., Bernasconi, S., ... Mascarin, M. (2020). Comparison of Hodgkin’s Lymphoma in Children and Adolescents. A Twenty Year Experience with MH’96 and LH2004 AIEOP (Italian Association of Pediatric Hematology and Oncology) Protocols. Cancers, 12(6), 1620. https://doi.org/10.3390/cancers12061620







