Potential Circulating Biomarkers of Recurrence after Hepatic Resection or Liver Transplantation in Hepatocellular Carcinoma Patients
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
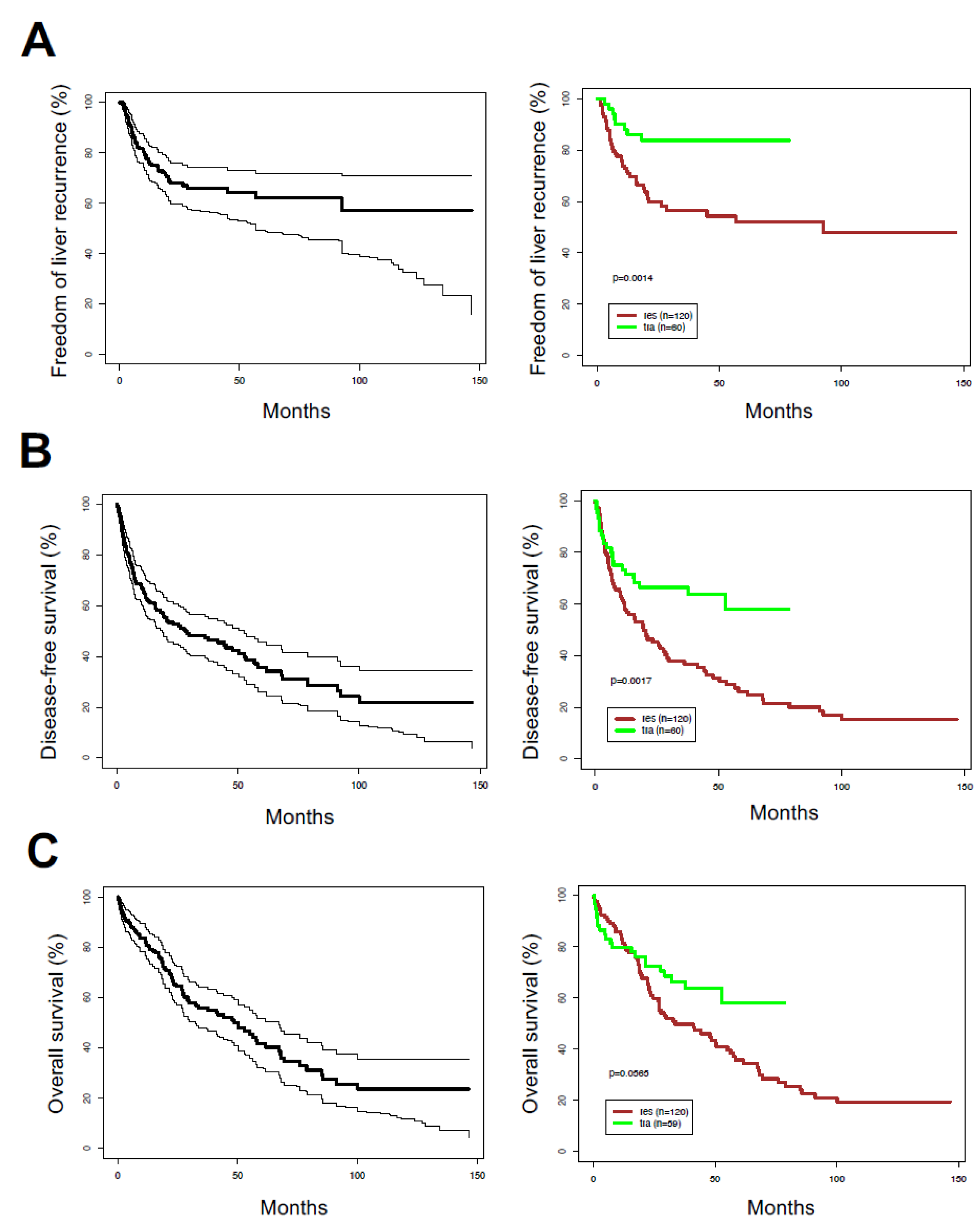
2.2. Post-Surgical Outcomes and Treatment
2.3. Levels of Circulating Inflammatory and Angiogenic Biomarkers in Surgical HCC Patients
2.4. Correlation between Clinical Variables with Recurrence and Survival after Surgical Treatments
2.5. Correlation between Circulating Inflammatory and Angiogenic Factors with Recurrence and Survival after Surgical Treatments
2.6. Elevated Gene Expression Levels of IL10 Downstream Mediators IL10RA and JAK1 Are Associated with Worse OS in HCC
2.7. Correlation between Circulating Biomarkers and Outcomes after Stratifying by Surgical Intervention and Milan Criteria
2.8. Analysis of Clinical Outcomes in a Proportional Hazards Model Including AFP and Plasma VEGF or VEGF-C after Adjusting for Milan Criteria Score
3. Discussion
4. Materials and Methods
4.1. Patients, Diagnosis and Therapeutic Strategy
4.2. Study Design
4.3. Measurement of Pro-Inflammatory and Angiogenic Biomarkers
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Global Burden of Disease Liver Cancer. The Burden of Primary Liver Cancer and Underlying Etiologies From 1990 to 2015 at the Global, Regional, and National Level: Results from the Global Burden of Disease Study 2015. JAMA Oncol. 2017, 3, 1683–1691. [Google Scholar] [CrossRef]
- Tabrizian, P.; Jibara, G.; Shrager, B.; Schwartz, M.; Roayaie, S. Recurrence of hepatocellular cancer after resection: Patterns, treatments, and prognosis. Ann. Surg. 2015, 261, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Fuster, J.; Bruix, J. The Barcelona approach: Diagnosis, staging, and treatment of hepatocellular carcinoma. Liver Transplant. 2004, 10, S115–S120. [Google Scholar] [CrossRef] [PubMed]
- Cucchetti, A.; Ercolani, G.; Vivarelli, M.; Cescon, M.; Ravaioli, M.; La Barba, G.; Zanello, M.; Grazi, G.L.; Pinna, A.D. Impact of model for end-stage liver disease (MELD) score on prognosis after hepatectomy for hepatocellular carcinoma on cirrhosis. Liver Transplant. 2006, 12, 966–971. [Google Scholar] [CrossRef] [PubMed]
- Citterio, D.; Facciorusso, A.; Sposito, C.; Rota, R.; Bhoori, S.; Mazzaferro, V. Hierarchic Interaction of Factors Associated With Liver Decompensation After Resection for Hepatocellular Carcinoma. JAMA Surg. 2016, 151, 846–853. [Google Scholar] [CrossRef]
- Mazzaferro, V.; Regalia, E.; Doci, R.; Andreola, S.; Pulvirenti, A.; Bozzetti, F.; Montalto, F.; Ammatuna, M.; Morabito, A.; Gennari, L. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N. Engl. J. Med. 1996, 334, 693–699. [Google Scholar] [CrossRef]
- Llovet, J.M.; Pavel, M.; Rimola, J.; Diaz, M.A.; Colmenero, J.; Saavedra-Perez, D.; Fondevila, C.; Ayuso, C.; Fuster, J.; Gines, P.; et al. Pilot study of living donor liver transplantation for patients with hepatocellular carcinoma exceeding Milan Criteria (Barcelona Clinic Liver Cancer extended criteria). Liver Transplant. 2018, 24, 369–379. [Google Scholar] [CrossRef]
- Toso, C.; Kneteman, N.M.; James Shapiro, A.M.; Bigam, D.L. The estimated number of patients with hepatocellular carcinoma selected for liver transplantation using expanded selection criteria. Transplant. Int. 2009, 22, 869–875. [Google Scholar] [CrossRef]
- Agopian, V.G.; Harlander-Locke, M.; Zarrinpar, A.; Kaldas, F.M.; Farmer, D.G.; Yersiz, H.; Finn, R.S.; Tong, M.; Hiatt, J.R.; Busuttil, R.W. A novel prognostic nomogram accurately predicts hepatocellular carcinoma recurrence after liver transplantation: Analysis of 865 consecutive liver transplant recipients. J. Am. Coll. Surg. 2015, 220, 416–427. [Google Scholar] [CrossRef]
- Halazun, K.J.; Najjar, M.; Abdelmessih, R.M.; Samstein, B.; Griesemer, A.D.; Guarrera, J.V.; Kato, T.; Verna, E.C.; Emond, J.C.; Brown, R.S., Jr. Recurrence After Liver Transplantation for Hepatocellular Carcinoma: A New MORAL to the Story. Ann. Surg. 2017, 265, 557–564. [Google Scholar] [CrossRef]
- Zhang, W.; Kim, R.; Quintini, C.; Hashimoto, K.; Fujiki, M.; Diago, T.; Eghtesad, B.; Miller, C.; Fung, J.; Tan, A.; et al. Prognostic role of plasma vascular endothelial growth factor in patients with hepatocellular carcinoma undergoing liver transplantation. Liver Transplant. 2015, 21, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Qin, S.; Merle, P.; Granito, A.; Huang, Y.H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Breder, V.; et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017, 389, 56–66. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.X.; Kang, Y.K.; Yen, C.J.; Finn, R.S.; Galle, P.R.; Llovet, J.M.; Assenat, E.; Brandi, G.; Pracht, M.; Lim, H.Y.; et al. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased alpha-fetoprotein concentrations (REACH-2): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019, 20, 282–296. [Google Scholar] [CrossRef]
- Abou-Alfa, G.K.; Meyer, T.; Cheng, A.L.; El-Khoueiry, A.B.; Rimassa, L.; Ryoo, B.Y.; Cicin, I.; Merle, P.; Chen, Y.; Park, J.W.; et al. Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma. N. Engl J. Med. 2018, 379, 54–63. [Google Scholar] [CrossRef]
- Tammela, T.; Zarkada, G.; Wallgard, E.; Murtomaki, A.; Suchting, S.; Wirzenius, M.; Waltari, M.; Hellstrom, M.; Schomber, T.; Peltonen, R.; et al. Blocking VEGFR-3 suppresses angiogenic sprouting and vascular network formation. Nature 2008, 454, 656–660. [Google Scholar] [CrossRef]
- Padera, T.P.; Jain, R.K. VEGFR3: A new target for antiangiogenesis therapy? Dev. Cell 2008, 15, 178–179. [Google Scholar] [CrossRef]
- Yamaguchi, R.; Yano, H.; Nakashima, O.; Akiba, J.; Nishida, N.; Kurogi, M.; Kojiro, M. Expression of vascular endothelial growth factor-C in human hepatocellular carcinoma. J. Gastroenterol. Hepatol. 2006, 21, 152–160. [Google Scholar] [CrossRef]
- Xu, M.; Doyle, M.M.; Banan, B.; Vachharajani, N.; Wang, X.; Saad, N.; Fowler, K.; Brunt, E.M.; Lin, Y.; Chapman, W.C. Neoadjuvant Locoregional Therapy and Recurrent Hepatocellular Carcinoma after Liver Transplantation. J. Am. Coll. Surg. 2017, 225, 28–40. [Google Scholar] [CrossRef]
- Jaroszewicz, J.; Januszkiewicz, M.; Flisiak, R.; Rogalska, M.; Kalinowska, A.; Wierzbicka, I. Circulating vascular endothelial growth factor and its soluble receptors in patients with liver cirrhosis: Possible association with hepatic function impairment. Cytokine 2008, 44, 14–17. [Google Scholar] [CrossRef]
- Duda, D.G.; Willett, C.G.; Ancukiewicz, M.; di Tomaso, E.; Shah, M.; Czito, B.G.; Bentley, R.; Poleski, M.; Lauwers, G.Y.; Carroll, M.; et al. Plasma soluble VEGFR-1 is a potential dual biomarker of response and toxicity for bevacizumab with chemoradiation in locally advanced rectal cancer. Oncologist 2010, 15, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.X.; Duda, D.G.; Sahani, D.V.; Jain, R.K. HCC and angiogenesis: Possible targets and future directions. Nat. Rev. Clin. Oncol. 2011, 8, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.X.; Sahani, D.V.; Duda, D.G.; di Tomaso, E.; Ancukiewicz, M.; Catalano, O.A.; Sindhwani, V.; Blaszkowsky, L.S.; Yoon, S.S.; Lahdenranta, J.; et al. Efficacy, safety, and potential biomarkers of sunitinib monotherapy in advanced hepatocellular carcinoma: A phase II study. J. Clin. Oncol. 2009, 27, 3027–3035. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Gea, V.; Toffanin, S.; Friedman, S.L.; Llovet, J.M. Role of the microenvironment in the pathogenesis and treatment of hepatocellular carcinoma. Gastroenterology 2013, 144, 512–527. [Google Scholar] [CrossRef] [PubMed]
- Hoshida, Y.; Villanueva, A.; Kobayashi, M.; Peix, J.; Chiang, D.Y.; Camargo, A.; Gupta, S.; Moore, J.; Wrobel, M.J.; Lerner, J.; et al. Gene expression in fixed tissues and outcome in hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 1995–2004. [Google Scholar] [CrossRef]
- Naugler, W.E.; Karin, M. The wolf in sheep’s clothing: The role of interleukin-6 in immunity, inflammation and cancer. Trends Mol. Med. 2008, 14, 109–119. [Google Scholar] [CrossRef]
- Zhu, A.X.; Duda, D.G.; Ancukiewicz, M.; di Tomaso, E.; Clark, J.W.; Miksad, R.; Fuchs, C.S.; Ryan, D.P.; Jain, R.K. Exploratory analysis of early toxicity of sunitinib in advanced hepatocellular carcinoma patients: Kinetics and potential biomarker value. Clin. Cancer Res. 2011, 17, 918–927. [Google Scholar] [CrossRef]
- Ringelhan, M.; Pfister, D.; O’Connor, T.; Pikarsky, E.; Heikenwalder, M. The immunology of hepatocellular carcinoma. Nat. Immunol. 2018, 19, 222–232. [Google Scholar] [CrossRef]
- Yang, J.D.; Nakamura, I.; Roberts, L.R. The tumor microenvironment in hepatocellular carcinoma: Current status and therapeutic targets. Semin. Cancer Biol. 2011, 21, 35–43. [Google Scholar] [CrossRef]
- Mazzaferro, V.; Sposito, C.; Zhou, J.; Pinna, A.D.; De Carlis, L.; Fan, J.; Cescon, M.; Di Sandro, S.; Yi-Feng, H.; Lauterio, A.; et al. Metroticket 2.0 Model for Analysis of Competing Risks of Death After Liver Transplantation for Hepatocellular Carcinoma. Gastroenterology 2018, 154, 128–139. [Google Scholar] [CrossRef]
- Bodzin, A.S.; Lunsford, K.E.; Markovic, D.; Harlander-Locke, M.P.; Busuttil, R.W.; Agopian, V.G. Predicting Mortality in Patients Developing Recurrent Hepatocellular Carcinoma After Liver Transplantation: Impact of Treatment Modality and Recurrence Characteristics. Ann. Surg. 2017, 266, 118–125. [Google Scholar] [CrossRef]
- Roayaie, S.; Obeidat, K.; Sposito, C.; Mariani, L.; Bhoori, S.; Pellegrinelli, A.; Labow, D.; Llovet, J.; Schwartz, M.; Mazzaferro, V. Resection of hepatocellular cancer </= 2 cm: Results from two western centers. Hepatology 2012. [Google Scholar] [CrossRef]
- Amarapurkar, A.D.; Vibhav; Kim, V. Angiogenesis in liver cirrhosis and hepatocellular carcinoma. Indian J. Pathol. Microbiol. 2008, 51, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Clavien, P.A.; Lesurtel, M.; Bossuyt, P.M.; Gores, G.J.; Langer, B.; Perrier, A. Recommendations for liver transplantation for hepatocellular carcinoma: An international consensus conference report. Lancet Oncol. 2012, 13, e11–e22. [Google Scholar] [CrossRef]
- Duvoux, C.; Roudot-Thoraval, F.; Decaens, T.; Pessione, F.; Badran, H.; Piardi, T.; Francoz, C.; Compagnon, P.; Vanlemmens, C.; Dumortier, J.; et al. Liver transplantation for hepatocellular carcinoma: A model including alpha-fetoprotein improves the performance of Milan criteria. Gastroenterology 2012, 143, 986–994. [Google Scholar] [CrossRef] [PubMed]
- Dumitra, T.C.; Dumitra, S.; Metrakos, P.P.; Barkun, J.S.; Chaudhury, P.; Deschenes, M.; Paraskevas, S.; Hassanain, M.; Tchervenkov, J.I. Pretransplantation alpha-fetoprotein slope and milan criteria: Strong predictors of hepatocellular carcinoma recurrence after transplantation. Transplantation 2013, 95, 228–233. [Google Scholar] [CrossRef]
- Chaiteerakij, R.; Zhang, X.; Addissie, B.D.; Mohamed, E.A.; Harmsen, W.S.; Theobald, P.J.; Peters, B.E.; Balsanek, J.G.; Ward, M.M.; Giama, N.H.; et al. Combinations of biomarkers and Milan criteria for predicting hepatocellular carcinoma recurrence after liver transplantation. Liver Transplant. 2015, 21, 599–606. [Google Scholar] [CrossRef]
- Song, P.P.; Xia, J.F.; Inagaki, Y.; Hasegawa, K.; Sakamoto, Y.; Kokudo, N.; Tang, W. Controversies regarding and perspectives on clinical utility of biomarkers in hepatocellular carcinoma. World J. Gastroenterol. 2016, 22, 262–274. [Google Scholar] [CrossRef]
- Bando, H.; Weich, H.A.; Brokelmann, M.; Horiguchi, S.; Funata, N.; Ogawa, T.; Toi, M. Association between intratumoral free and total VEGF, soluble VEGFR-1, VEGFR-2 and prognosis in breast cancer. Br. J. Cancer 2005, 92, 553–561. [Google Scholar] [CrossRef]
- Chao, Y.; Li, C.P.; Chau, G.Y.; Chen, C.P.; King, K.L.; Lui, W.Y.; Yen, S.H.; Chang, F.Y.; Chan, W.K.; Lee, S.D. Prognostic significance of vascular endothelial growth factor, basic fibroblast growth factor, and angiogenin in patients with resectable hepatocellular carcinoma after surgery. Ann. Surg. Oncol. 2003, 10, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Ren, X.; Chen, Y.; Liu, P.; Wei, X.; Li, H.; Ying, G.; Chen, K.; Winkler, H.; Hao, X. Dysfunctional activation of neurotensin/IL-8 pathway in hepatocellular carcinoma is associated with increased inflammatory response in microenvironment, more epithelial mesenchymal transition in cancer and worse prognosis in patients. PLoS ONE 2013, 8, e56069. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chau, G.Y.; Wu, C.W.; Lui, W.Y.; Chang, T.J.; Kao, H.L.; Wu, L.H.; King, K.L.; Loong, C.C.; Hsia, C.Y.; Chi, C.W. Serum interleukin-10 but not interleukin-6 is related to clinical outcome in patients with resectable hepatocellular carcinoma. Ann. Surg. 2000, 231, 552–558. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. European Association for the Study of the, L. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef] [PubMed]
| Characteristics/ Surgical Intervention | Liver Resection | Liver Transplantation | p-Value | |
|---|---|---|---|---|
| Age, years (n) | 64 (16, 79) (120) | 57 (30, 68) (60) | <0.0001 | |
| Milan criteria (n within, %) | 45/119 (37.8%) | 33/51 (64.7%) | 0.0015 | |
| Edmondson Steiner grade (n, %) | 0.044 | |||
| I/II | 10/116 (8.6%) | 10/44 (22.7%) | ||
| II/III | 96/116 (82.8%) | 31/44 (70.5%) | ||
| III/IV | 10/116 (8.6%) | 3/44 (6.8%) | ||
| Tumor size (cm ± SD, n) | 7.0 ± 3.6 (118) | 4.0 ± 1.7 (53) | <0.0001 | |
| Nodules (n, %) | 0.0009 | |||
| 1 | 100/120 (83.3%) | 35/57 (61.4%) | ||
| 2 | 14/120 (11.7%) | 11/57 (19.3%) | ||
| 3 | 3/120 (2.5%) | 8/57 (14.0%) | ||
| 4+ | 3/120 (2.5%) | 3/57 (3.7%) | ||
| Nodules (n ± SD, n) | 1.2 ± 0.6 (120) | 1.8 ± 1.4 (57) | 0.0007 | |
| INR (range, n) | 1.1 (1.0, 1.2) (41) | 1.4 (1.2, 1.5) (54) | <0.0001 | |
| Pre-operative HBV (n, %) | 49/119 (41.2%) | 42/60 (70.0%) | 0.0003 | |
| Pre-operative HCV (n, %) | 47/119 (39.5%) | 15/60 (25.0%) | 0.067 | |
| Pre-operative HDV (n, %) | 7/119 (5.9%) | 26/60 (43.3%) | <0.0001 | |
| Pre-operative cirrhosis (n, %) | 80/119 (67.2%) | 59/60 (98.3%) | <0.0001 | |
| CTP score (n, %) | 0 | 5/120 (29.2%) | 0/59 (0.0%) | <0.0001 |
| A | 78/120 (65.0%) | 17/59 (28.8%) | ||
| B | 7/120 (5.8%) | 42/59 (71.2%) | ||
| MELD score (median, range) | 8 (6–11) | 13 (7–28) | <0.0001 | |
| Surgery type (n, %) | <0.0001 | |||
| Liver resection | 108/120 (90.0%) | 0/60 (0.0%) | ||
| Liver resection + RFA/sorafenib/TACE/PCT | 12/120 (10.0%) | 0/60 (0.0%) | ||
| Liver transplantation | 0/120 (0.0%) | 59/60 (98.3%) | ||
| Liver transplantation + RFA/TACE | 0/120 (0.0%) | 1/60 (1.7%) | ||
| Biomarker | LR Patients (n) | LT Patients (n) | p Value |
|---|---|---|---|
| IFN-γ (pg/mL) | 7.5 (5.6, 13.3) (44) | 7.8 (4.9, 13.4) (58) | 0.91 |
| IL-6 (pg/mL) | 1.72 (1.60, 4.59) (44) | 4.8 (2.0, 30.7) (58) | 0.0001 |
| IL-8 (pg/mL) | 13.2 (7.4, 18.7) (44) | 22.6 (11.3, 51.3) (58) | 0.0017 |
| IL-10 (pg/mL) | 0.80 (0.57, 2.07) (45) | 1.23 (0.59, 4.49) (58) | 0.11 |
| TNF-α (pg/mL) | 3.5 (2.8, 4.1) (44) | 3.4 (2.6, 5.0) (58) | 0.84 |
| bFGF (pg/mL) | 4.3 (2.5, 8.6) (44) | 12.1 (6.2, 32.6) (58) | <0.0001 |
| PlGF (pg/mL) | 34.6 (28.8, 43.0) (44) | 27.2 (18.9, 35.1) (58) | 0.0007 |
| sVEGFR1 (pg/mL) | 116 (87, 230) (44) | 297 (134, 1166) (58) | 0.0002 |
| sTIE2 (pg/mL) | 5334 (4610, 7429) (44) | 6316 (5293, 8255) (58) | 0.046 |
| VEGF (pg/mL) | 82 (42, 152) (44) | 85 (39, 243) (58) | 0.97 |
| VEGF-C (pg/mL) | 102 (89, 193) (44) | 131 (89, 179) (58) | 0.26 |
| VEGF-D (pg/mL) | 697 (549, 987) (44) | 840 (681, 1162) (58) | 0.012 |
| AFP (ng/mL) | 12.8 (6.3, 20.0) (31) | 7.1 (3.3, 20.0) (53) | 0.13 |
| Clinical Variables/ Outcome | FLR | DFS | OS | |||
|---|---|---|---|---|---|---|
| HR [95% CI] (n) | p-Value | HR [95% CI] (n) | p-Value | HR [95% CI] (n) | p-Value | |
| All patients | ||||||
| Milan criteria | 0.58 [0.33,1.03] (173) | 0.065 | 0.64 [0.43, 0.97] (173) | 0.034 | 0.53 [0.34, 0.81] (173) | 0.0037 |
| Edmondson–Steiner grade | 4.45 [0.97,20.40] (160) | 0.054 | 1.75 [0.82, 3.71] (160) | 0.15 | 1.41 [0.63, 3.16] (160) | 0.40 |
| Tumor size | 1.03 [0.96, 1.11] (171) | 0.38 | 1.06 [1.00, 1.12] (171) | 0.050 | 1.06 [1.00, 1.12] (170) | 0.042 |
| HBV | 2.08 [1.20, 3.63] (179) | 0.0097 | 1.29 [0.87, 1.91] (179) | 0.21 | 1.06 [0.70, 1.61] (178) | 0.77 |
| HCV | 0.73 [0.41, 1.30] (179) | 0.28 | 0.83 [0.55, 1.24] (179) | 0.36 | 0.90 [0.59, 1.38] (178) | 0.63 |
| HDV | 1.48 [0.63, 3.48] (179) | 0.37 | 1.03 [0.54, 1.98] (179) | 0.92 | 0.85 [0.44, 1.65] (178) | 0.64 |
| LT | ||||||
| Milan criteria | 0.38 [0.09, 1.51] (54) | 0.17 | 0.48 [0.20, 1.16] (54) | 0.10 | 0.53 [0.21,1.31] (54) | 0.17 |
| Edmondson–Steiner grade | NA [NA, NA] (44) | N/A | 1.48 [0.27, 8.11] (44) | 0.65 | 1.58 [0.29, 8.66] (44) | 0.60 |
| Tumor size | 1.54 [0.97, 2.45] (53) | 0.067 | 1.27 [0.96, 1.66] (53) | 0.067 | 1.26 [0.95, 1.67] (52) | 0.11 |
| HBV | 1.69 [0.34, 8.39] (60) | 0.52 | 1.33 [0.52, 3.41] (60) | 0.55 | 1.45 [0.53, 3.98] (59) | 0.47 |
| HCV | 0.78 [0.16, 3.89] (60) | 0.77 | 0.56 [0.19, 1.65] (60) | 0.29 | 0.45 [0.13, 1.52] (59) | 0.20 |
| HDV | 0.73 [0.17, 3.05] (60) | 0.66 | 0.69 [0.29, 1.65] (60) | 0.41 | 0.71 [0.29, 1.71] (59) | 0.45 |
| LR | ||||||
| CTP Score | 2.72 [1.05, 7.05] (120) | 0.039 | 2.71 [1.41, 5.23] (120) | 0.0029 | 3.26 [1.66, 6.42] (120) | 0.0006 |
| Milan criteria | 0.64 [0.34, 1.18] (119) | 0.15 | 0.70 [0.44, 1.09] (119) | 0.11 | 0.53 [0.32, 0.86] (119) | 0.011 |
| Edmondson–Steiner grade | 3.79 [0.83, 17.31] (116) | 0.086 | 1.82 [0.76, 4.35] (116) | 0.18 | 1.39 [0.55, 3.52] (116) | 0.49 |
| Tumor size | 1.02 [0.95, 1.10] (118) | 0.067 | 1.05 [0.99, 1.11] (118) | 0.10 | 1.05 [0.99, 1.12] (118) | 0.083 |
| HBV | 2.14 [1.19, 3.86] (119) | 0.011 | 1.28 [0.83, 1.98] (119) | 0.27 | 0.00 [0.62, 1.57] (119) | 0.97 |
| HCV | 0.72 [0.39, 1.33] (119) | 0.30 | 0.89 [0.57, 1.39] (119) | 0.61 | 1.03 [0.64, 1.64] (119) | 0.91 |
| HDV | 2.13 [0.84, 5.42] (119) | 0.11 | 1.66 [0.72, 3.84] (119) | 0.24 | 1.07 [0.43, 2.65] (119) | 0.89 |
| Biomarkers/ Outcome | FLR | DFS | OS | |||
|---|---|---|---|---|---|---|
| HR (95% CI) (n) | p-Value | HR (95% CI) (n) | p-Value | HR (95% CI) (n) | p-Value | |
| All patients | ||||||
| sVEGFR1 | 0.88 (0.65,1.18) (102) | 0.40 | 0.94 (0.78,1.13) (102) | 0.50 | 0.90 (0.72,1.13) (101) | 0.38 |
| VEGF | 1.48 (1.09, 2.01) (102) | 0.012 | 1.25 (1.04, 1.51) (102) | 0.019 | 1.13 (0.92, 1.39) (101) | 0.25 |
| VEGF-C | 1.31 (0.74, 2.32) (102) | 0.35 | 1.49 (0.97, 2.29) (102) | 0.071 | 1.67 (0.98, 2.85) (101) | 0.058 |
| IFN-γ | 0.78 (0.51, 1.19) (102) | 0.25 | 0.74 (0.55, 1.01) (102) | 0.056 | 0.77 (0.55, 1.08) (101) | 0.13 |
| IL-6 | 0.99 (0.80, 1.24) (102) | 0.95 | 1.02 (0.89, 1.17) (102) | 0.72 | 1.01 (0.87, 1.17) (101) | 0.87 |
| IL-8 | 1.18 (0.89, 1.57) (102) | 0.25 | 1.12 (0.93, 1.34) (102) | 0.24 | 1.04 (0.85, 1.27) (101) | 0.70 |
| IL-10 | 0.88 (0.67, 1.17) (102) | 0.39 | 1.01 (0.87, 1.17) (102) | 0.93 | 1.01 (0.86, 1.17) (101) | 0.95 |
| AFP | 1.04 (0.69, 1.56) (84) | 0.85 | 0.96 (0.75, 1.24) (84) | 0.77 | 0.97 (0.74, 1.28) (83) | 0.84 |
| LT | ||||||
| sVEGFR1 | 0.69 (0.41, 1.14) (58) | 0.15 | 0.78 (0.60, 1.02) (58) | 0.069 | 0.74 (0.55, 0.99) (57) | 0.045 |
| VEGF | 1.51 (0.96, 2.38) (58) | 0.073 | 1.38 (1.07, 1.77) (58) | 0.012 | 1.31 (1.02, 1.69) (57) | 0.037 |
| VEGF-C | 2.19 (0.51, 9.51) (58) | 0.29 | 2.47 (1.08, 5.64) (58) | 0.033 | 2.49 (1.05, 5.93) (57) | 0.039 |
| IFN-γ | 0.68 (0.32, 1.43) (58) | 0.31 | 0.66 (0.42, 1.04) (58) | 0.071 | 0.61 (0.38, 0.99) (57) | 0.043 |
| IL-6 | 0.93 (0.67, 1.27) (58) | 0.63 | 0.97 (0.82, 1.15) (58) | 0.71 | 0.92 (0.74, 1.21) (57) | 0.39 |
| IL-8 | 0.92 (0.61, 1.38) (58) | 0.68 | 0.98 (0.78, 1.23) (58) | 0.84 | 0.95 (0.61, 1.38) (57) | 0.65 |
| IL-10 | 0.88 (0.61, 1.25) (58) | 0.47 | 0.92 (0.76, 1.11) (58) | 0.37 | 0.86 (0.69, 1.07) (57) | 0.18 |
| AFP | 0.89 (0.48, 1.65) (53) | 0.71 | 0.89 (0.63, 1.25) (53) | 0.50 | 0.93 (0.65, 1.33) (52) | 0.69 |
| LR | ||||||
| sVEGFR1 | 1.03 (0.75, 1.42) (44) | 0.84 | 1.15 (0.92, 1.42) (44) | 0.22 | 1.38 (0.99, 1.92) (44) | 0.061 |
| VEGF | 1.44 (0.96, 2.20) (44) | 0.078 | 1.08 (0.81, 1.44) (44) | 0.61 | 0.76 (0.53, 1.09) (44) | 0.14 |
| VEGF-C | 1.18 (0.63, 2.23) (44) | 0.60 | 1.21 (0.72, 2.04) (44) | 0.47 | 1.30 (0.66, 2.56) (44) | 0.45 |
| IFN-γ | 0.84 (0.50, 1.41) (44) | 0.51 | 0.83 (0.55, 1.27) (44) | 0.39 | 1.06 (0.63, 1.80) (44) | 0.81 |
| IL-6 | 1.08 (0.78, 1.51) (44) | 0.63 | 1.17 (0.92, 1.50) (44) | 0.20 | 1.34 (1.03, 1.74) (44) | 0.032 |
| IL-8 | 1.63 (1.09, 2.45) (44) | 0.018 | 1.54 (1.11, 2.12) (44) | 0.0093 | 1.35 (0.93, 1.96) (44) | 0.12 |
| IL-10 | 0.89 (0.56, 1.42) (44) | 0.63 | 1.35 (1.01, 1.79) (44) | 0.040 | 1.78 (1.28, 2.49) (44) | 0.0007 |
| AFP | 1.17 (0.67, 2.05) (31) | 0.59 | 1.06 (0.72, 1.58) (31) | 0.50 | 1.04 (0.66, 1.63) (31) | 0.87 |
| Biomarkers/ Outcome | FLR | DFS | OS | |||
|---|---|---|---|---|---|---|
| HR (95% CI) (n) | p-Value | HR (95% CI) (n) | p-Value | HR (95% CI) (n) | p-Value | |
| LT | ||||||
| sVEGFR1 | 0.49 (0.20, 1.17) (35) | 0.11 | 0.64 (0.41, 0.99) (35) | 0.044 | 0.63 (0.40, 0.98) (35) | 0.042 |
| VEGF | 1.45 (0.83, 2.51) (35) | 0.19 | 1.45 (1.04, 2.02) (35) | 0.027 | 1.44 (1.03, 2.01) (35) | 0.031 |
| VEGF-C | 2.12 (0.28, 16.23) (35) | 0.47 | 1.85 (0.56, 6.06) (35) | 0.31 | 2.02 (0.60, 6.79) (35) | 0.26 |
| IFN-γ | 0.84 (0.33, 2.14) (35) | 0.72 | 0.71 (0.38, 1.33) (35) | 0.28 | 0.71 (0.38, 1.34) (35) | 0.29 |
| IL-6 | 0.85 (0.52, 1.38) (35) | 0.50 | 0.89 (0.68, 1.17) (35) | 0.41 | 0.89 (0.68, 1.17) (35) | 0.40 |
| IL-8 | 0.83 (0.46, 1.50) (35) | 0.54 | 0.94 (0.68, 1.30) (35) | 0.73 | 0.95 (0.69, 1.31) (35) | 0.73 |
| IL-10 | 0.79 (0.43, 1.45) (35) | 0.45 | 0.91 (0.69, 1.20) (35) | 0.51 | 0.91 (0.68, 1.20) (35) | 0.49 |
| AFP | 0.97 (0.44, 2.15) (34) | 0.94 | 1.00 (0.62, 1.61) (34) | 0.99 | 0.98 (0.61, 1.58) (34) | 0.95 |
| LR | ||||||
| sVEGFR1 | 3.08 (1.18, 8.08) (19) | 0.022 | 2.40 (1.10, 5.26) (19) | 0.028 | 1.52 (0.60, 3.85) (19) | 0.38 |
| VEGF | 1.65 (0.92, 2.97) (19) | 0.095 | 1.38 (0.83, 2.28) (19) | 0.21 | 1.15 (0.47, 2.83) (19) | 0.76 |
| VEGF-C | 1.75 (0.48, 6.41) (19) | 0.40 | 2.19 (0.65, 7.35) (19) | 0.20 | 2.45 (0.43, 13.90) (19) | 0.31 |
| IFN-γ | 0.63 (0.22, 1.75) (19) | 0.37 | 0.48 (0.20, 1.16) (19) | 0.10 | 0.20 (0.02, 1.89) (19) | 0.16 |
| IL-6 | 0.58 (0.18, 1.86) (19) | 0.36 | 1.05 (0.63, 1.75) (19) | 0.85 | 1.67 (0.82, 3.39) (19) | 0.15 |
| IL-8 | 1.57 (0.84, 2.95) (19) | 0.16 | 1.45 (0.88, 2.39) (19) | 0.14 | 1.50 (0.79, 2.83) (19) | 0.22 |
| IL-10 | 0.49 (0.15, 1.57) (19) | 0.23 | 1.16 (0.63, 2.14) (19) | 0.63 | 1.94 (0.88, 4.29) (19) | 0.10 |
| AFP | 1.39 (0.64, 3.00) (16) | 0.40 | 1.07 (0.65, 1.77) (16) | 0.78 | 0.83 (0.42, 1.62) (16) | 0.58 |
| Biomarkers/ Outcome | FLR | DFS | OS | |||
|---|---|---|---|---|---|---|
| HR (95% CI) (n) | p-Value | HR (95% CI) (n) | p-Value | HR (95% CI) (n) | p-Value | |
| LT | ||||||
| sVEGFR1 | 1.00 (0.54, 1.83) (17) | 0.99 | 1.00 (0.68, 1.47) (17) | 0.99 | 0.89 (0.57, 1.41) (17) | 0.62 |
| VEGF | 2.00 (0.86, 4.65) (17) | 0.11 | 1.53 (0.94, 2.49) (17) | 0.085 | 1.34 (0.81, 2.23) (17) | 0.25 |
| VEGF-C | 127.32 (0.20, 80,654) (17) | 0.14 | 6.15 (1.60, 23.62) (17) | 0.0082 | 5.41 (1.42, 20.64) (17) | 0.014 |
| IFN-γ | 0.48 (0.15, 1.55) (17) | 0.22 | 0.75 (0.37, 1.50) (17) | 0.42 | 0.72 (0.34, 1.54) (17) | 0.40 |
| IL-6 | 1.07 (0.73, 1.59) (17) | 0.72 | 1.14 (0.91, 1.44) (17) | 0.26 | 1.06 (0.82,1.38) (17) | 0.65 |
| IL-8 | 1.31 (0.53, 3.24) (17) | 0.57 | 1.36 (0.79, 2.34) (17) | 0.26 | 1.30 (0.73, 2.29) (17) | 0.37 |
| IL-10 | 0.98 (0.61, 1.58) (17) | 0.93 | 1.02 (0.77, 1.34) (17) | 0.91 | 0.91 (0.64, 1.28) (17) | 0.58 |
| AFP | 0.71 (0.21, 2.36) (13) | 0.57 | 1.18 (0.57, 2.45) (13) | 0.65 | 1.60 (0.67, 3.83) (13) | 0.29 |
| LR | ||||||
| sVEGFR1 | 0.80 (0.45, 1.42) (25) | 0.44 | 1.06 (0.80, 1.39) (25) | 0.69 | 1.43 (0.97, 2.10) (25) | 0.073 |
| VEGF | 1.26 (0.74, 2.14) (25) | 0.39 | 0.97 (0.71, 1.32) (25) | 0.84 | 0.78 (0.52, 1.16) (25) | 0.22 |
| VEGF-C | 1.03 (0.47, 2.27) (25) | 0.94 | 1.03 (0.56, 1.90) (25) | 0.92 | 1.19 (0.60, 2.35) (25) | 0.61 |
| IFN-γ | 0.93 (0.52, 1.65) (25) | 0.81 | 0.99 (0.61, 1.59) (25) | 0.96 | 1.26 (0.73, 2.16) (25) | 0.40 |
| IL-6 | 1.33 (0.91, 1.94) (25) | 0.15 | 1.23 (0.91, 1.66) (25) | 0.17 | 1.13 (0.80, 1.58) (25) | 0.49 |
| IL-8 | 1.59 (0.93, 2.71) (25) | 0.093 | 1.53 (0.99, 2.36) (25) | 0.054 | 1.35 (0.78, 2.34) (25) | 0.28 |
| IL-10 | 1.09 (0.67, 1.78) (25) | 0.79 | 1.38 (0.99, 1.92) (25) | 0.060 | 1.56 (1.07, 2.27) (25) | 0.021 |
| AFP | 0.82 (0.37, 1.82) (15) | 0.63 | 1.05 (0.54, 2.06) (15) | 0.88 | 1.24 (0.66, 2.32) (15) | 0.50 |
| Biomarkers/ Outcome | Plasma VEGF | Plasma VEGF-C | ||||
|---|---|---|---|---|---|---|
| DFS | C-statistic | χ2 | p-value | C-statistic | χ2 | p-value |
| Milan as stratum | 0.690 (0.578, 0.802) | 8.22 | 0.0042 | 0.620 (0.453, 0.788) | 6.18 | 0.013 |
| Milan in regression | 0.729 (0.624, 0.835) | 8.44 | 0.0037 | 0.692 (0.555, 0.830) | 5.95 | 0.015 |
| OS | C-statistic | χ2 | p-value | C-statistic | χ2 | p-value |
| Milan as stratum | 0.674 (0.562, 0.785) | 6.12 | 0.013 | 0.629 (0.461, 0.797) | 5.86 | 0.016 |
| Milan in regression | 0.700 (0.585, 0.815) | 5.98 | 0.015 | 0.686 (0.543, 0.830) | 6.54 | 0.011 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duda, D.G.; Dima, S.O.; Cucu, D.; Sorop, A.; Klein, S.; Ancukiewicz, M.; Kitahara, S.; Iacob, S.; Bacalbasa, N.; Tomescu, D.; et al. Potential Circulating Biomarkers of Recurrence after Hepatic Resection or Liver Transplantation in Hepatocellular Carcinoma Patients. Cancers 2020, 12, 1275. https://doi.org/10.3390/cancers12051275
Duda DG, Dima SO, Cucu D, Sorop A, Klein S, Ancukiewicz M, Kitahara S, Iacob S, Bacalbasa N, Tomescu D, et al. Potential Circulating Biomarkers of Recurrence after Hepatic Resection or Liver Transplantation in Hepatocellular Carcinoma Patients. Cancers. 2020; 12(5):1275. https://doi.org/10.3390/cancers12051275
Chicago/Turabian StyleDuda, Dan G., Simona O. Dima, Dana Cucu, Andrei Sorop, Sebastian Klein, Marek Ancukiewicz, Shuji Kitahara, Speranta Iacob, Nicolae Bacalbasa, Dana Tomescu, and et al. 2020. "Potential Circulating Biomarkers of Recurrence after Hepatic Resection or Liver Transplantation in Hepatocellular Carcinoma Patients" Cancers 12, no. 5: 1275. https://doi.org/10.3390/cancers12051275
APA StyleDuda, D. G., Dima, S. O., Cucu, D., Sorop, A., Klein, S., Ancukiewicz, M., Kitahara, S., Iacob, S., Bacalbasa, N., Tomescu, D., Herlea, V., Tanase, C., Croitoru, A., & Popescu, I. (2020). Potential Circulating Biomarkers of Recurrence after Hepatic Resection or Liver Transplantation in Hepatocellular Carcinoma Patients. Cancers, 12(5), 1275. https://doi.org/10.3390/cancers12051275








