Recent Developments of Systemic Chemotherapy for Gastric Cancer
Abstract
1. Introduction
2. Epidemiology
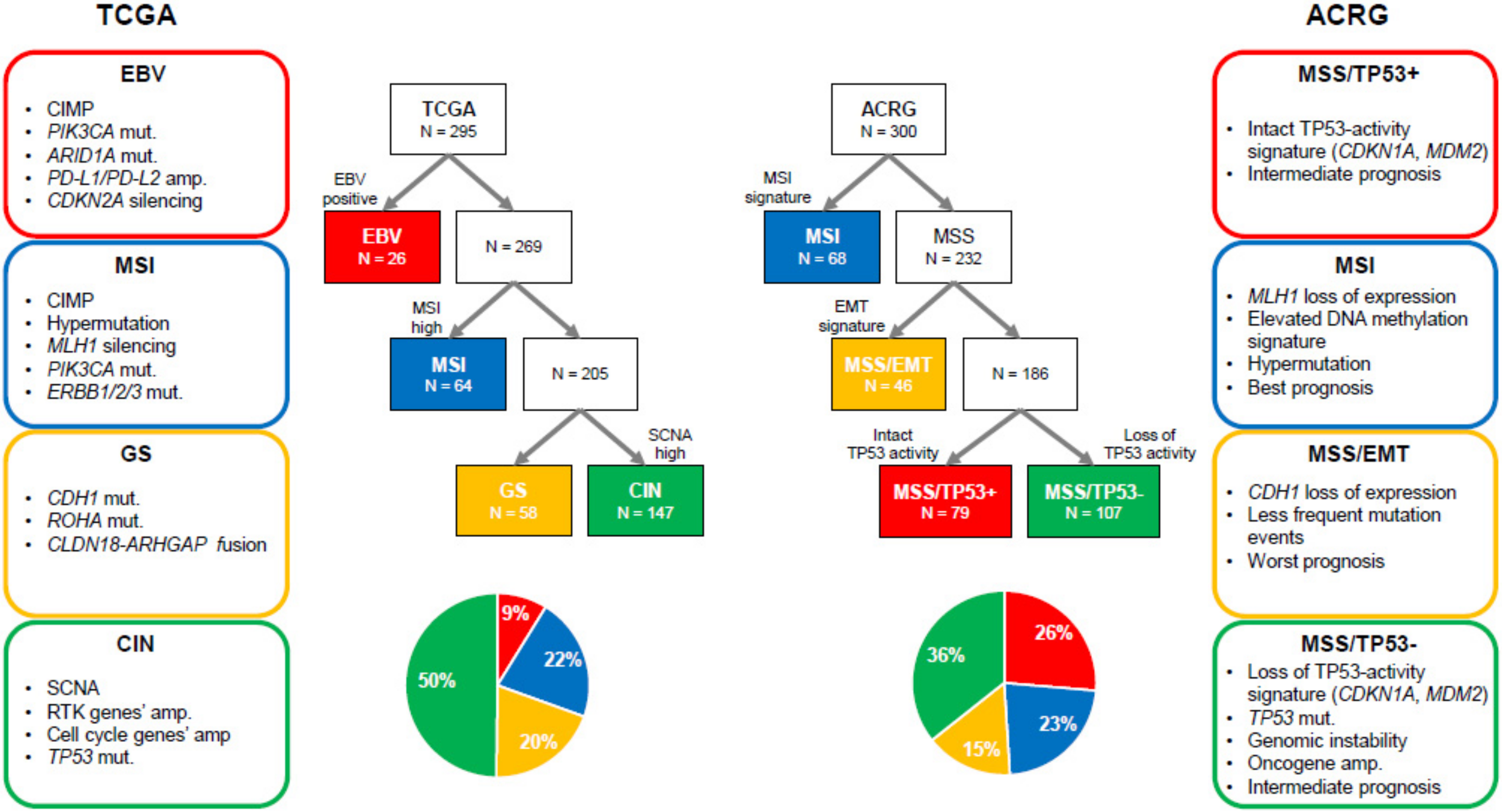
3. Molecular Findings in GC
4. Differences in Surgical Outcomes between Eastern and Western Countries
5. Development of Adjuvant Chemotherapy for Resectable GC
5.1. Western Standard Treatment
5.2. The Standard Treatment in the East Asia
5.3. Targeted Therapy for Resectable GC
5.4. Immunotherapy for Resectable GC
6. Development of Chemotherapy for Unresectable GC
6.1. Cytotoxic Agents for Advanced GC
6.1.1. Triplet Regimen in 1L Treatment
6.1.2. Trifluridine/Tipiracil (FTD/TPI, TAS-102)
6.2. Targeted Agents for Advanced GC
6.2.1. HER2-Targeted Therapies
6.2.2. VEGF/VEGFR-Targeted Therapies
6.2.3. Other Targeted Therapies
6.3. ICIs for Advanced GC
6.3.1. PD-1/PD-L1 Inhibitors as Monotherapies or Combination Therapies with Cytotoxic Agents
6.3.2. PD-1/PD-L1 Inhibitor Combination Therapy with Other ICIs
6.3.3. PD-1/PD-L1 Inhibitor Combination Therapy with Targeted Drugs
7. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- International Agency for Research on Cancer. GLOBOCAN 2018: Cancer Incidence and Mortality Worldwide. Available online: http://gco.iarc.fr/today/data/factsheets/cancers/39-All-cancers-fact-sheet.pdf (accessed on 24 March 2020).
- Colquhoun, A.; Arnold, M.; Ferlay, J.; Goodman, K.J.; Forman, D.; Soerjomataram, I. Global patterns of cardia and non-cardia gastric cancer incidence in 2012. Gut 2015, 64, 1881–1888. [Google Scholar] [CrossRef] [PubMed]
- de Martel, C.; Forman, D.; Plummer, M. Gastric cancer: Epidemiology and risk factors. Gastroenterol. Clin. North. Am. 2013, 42, 219–240. [Google Scholar] [CrossRef] [PubMed]
- Ajani, J.A.; Lee, J.; Sano, T.; Janjigian, Y.Y.; Fan, D.; Song, S. Gastric adenocarcinoma. Nat. Rev. Dis. Primers 2017, 3, 17036. [Google Scholar] [CrossRef] [PubMed]
- Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature 2014, 513, 202–209. [Google Scholar] [CrossRef]
- Cristescu, R.; Lee, J.; Nebozhyn, M.; Kim, K.M.; Ting, J.C.; Wong, S.S.; Liu, J.; Yue, Y.G.; Wang, J.; Yu, K.; et al. Molecular analysis of gastric cancer identifies subtypes associated with distinct clinical outcomes. Nat. Med. 2015, 21, 449–456. [Google Scholar] [CrossRef]
- Setia, N.; Agoston, A.T.; Han, H.S.; Mullen, J.T.; Duda, D.G.; Clark, J.W.; Deshpande, V.; Mino-Kenudson, M.; Srivastava, A.; Lennerz, J.K.; et al. A protein and mRNA expression-based classification of gastric cancer. Mod. Pathol. 2016, 29, 772–784. [Google Scholar] [CrossRef]
- Ahn, S.; Lee, S.J.; Kim, Y.; Kim, A.; Shin, N.; Choi, K.U.; Lee, C.H.; Huh, G.Y.; Kim, K.M.; Setia, N.; et al. High-throughput Protein and mRNA Expression-based Classification of Gastric Cancers Can Identify Clinically Distinct Subtypes, Concordant With Recent Molecular Classifications. Am. J. Surg. Pathol. 2017, 41, 106–115. [Google Scholar] [CrossRef]
- Chan, W.L.; Lam, K.O.; Lee, V.H.F.; Davidson, M.; So, T.H.; Li, J.S.; Chau, I.; Kwong, D.L.W. Gastric Cancer—From Aetiology to Management: Differences Between the East and the West. Clin. Oncol. 2019, 31, 570–577. [Google Scholar] [CrossRef]
- Ohtsu, A.; Yoshida, S.; Saijo, N. Disparities in Gastric Cancer Chemotherapy Between the East and West. J. Clin. Oncol. 2006, 24, 2188–2196. [Google Scholar] [CrossRef]
- Eusebi, L.H.; Telese, A.; Marasco, G.; Bazzoli, F.; Zagari, R.M. Gastric cancer prevention strategies: A global perspective. J. Gastroenterol. Hepatol. 2020. [Google Scholar] [CrossRef]
- Leung, W.K.; Wu, M.S.; Kakugawa, Y.; Kim, J.J.; Yeoh, K.G.; Goh, K.L.; Wu, K.C.; Wu, D.C.; Sollano, J.; Kachintorn, U.; et al. Screening for gastric cancer in Asia: Current evidence and practice. Lancet Oncol. 2008, 9, 279–287. [Google Scholar] [CrossRef]
- Hamashima, C.; Shabana, M.; Okada, K.; Okamoto, M.; Osaki, Y. Mortality reduction from gastric cancer by endoscopic and radiographic screening. Cancer Sci. 2015, 106, 1744–1749. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.; Kang, M.H.; Choi, K.S.; Suh, M.; Jun, J.K.; Park, E.C. Cost-effectiveness outcomes of the national gastric cancer screening program in South Korea. Asian Pac. J. Cancer Prev. 2013, 14, 2533–2540. [Google Scholar] [CrossRef] [PubMed]
- Sumiyama, K. Past and current trends in endoscopic diagnosis for early stage gastric cancer in Japan. Gastric Cancer 2017, 20, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Cancer Statistics in Japan 2016. Available online: https://ganjoho.jp/data/reg_stat/statistics/brochure/2016/cancer_statistics_2016_fig_J.pdf (accessed on 24 March 2020).
- Jim, M.A.; Pinheiro, P.S.; Carreira, H.; Espey, D.K.; Wiggins, C.L.; Weir, H.K. Stomach cancer survival in the United States by race and stage (2001–2009): Findings from the CONCORD-2 study. Cancer 2017, 123, 4994–5013. [Google Scholar] [CrossRef]
- Noguchi, Y.; Yoshikawa, T.; Tsuburaya, A.; Motohashi, H.; Karpeh, M.S.; Brennan, M.F. Is gastric carcinoma different between Japan and the United States? Cancer 2000, 89, 2237–2246. [Google Scholar] [CrossRef]
- Strong, V.E.; Song, K.Y.; Park, C.H.; Jacks, L.M.; Gonen, M.; Shah, M.; Coit, D.G.; Brennan, M.F. Comparison of gastric cancer survival following R0 resection in the United States and Korea using an internationally validated nomogram. Ann. Surg. 2010, 251, 640–646. [Google Scholar] [CrossRef]
- Talamonti, M.S.; Kim, S.P.; Yao, K.A.; Wayne, J.D.; Feinglass, J.; Bennett, C.L.; Rao, S. Surgical outcomes of patients with gastric carcinoma: The importance of primary tumor location and microvessel invasion. Surgery 2003, 134, 720–727. [Google Scholar] [CrossRef]
- Forman, D.; Newell, D.G.; Fullerton, F.; Yarnell, J.W.; Stacey, A.R.; Wald, N.; Sitas, F. Association between infection with Helicobacter pylori and risk of gastric cancer: Evidence from a prospective investigation. Br. Med. J. 1991, 302, 1302–1305. [Google Scholar] [CrossRef]
- Hooi, J.K.Y.; Lai, W.Y.; Ng, W.K.; Suen, M.M.Y.; Underwood, F.E.; Tanyingoh, D.; Malfertheiner, P.; Graham, D.Y.; Wong, V.W.S.; Wu, J.C.Y.; et al. Global Prevalence of Helicobacter pylori Infection: Systematic Review and Meta-Analysis. Gastroenterology 2017, 153, 420–429. [Google Scholar] [CrossRef] [PubMed]
- De Vries, A.C.; Kuipers, E.J. Review article: Helicobacter pylori eradication for the prevention of gastric cancer. Aliment. Pharmacol. Ther. 2007, 26, 25–35. [Google Scholar] [CrossRef]
- Wang, J.; Xu, L.; Shi, R.; Huang, X.; Li, S.W.; Huang, Z.; Zhang, G. Gastric atrophy and intestinal metaplasia before and after Helicobacter pylori eradication: A meta-analysis. Digestion 2011, 83, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Fuccio, L.; Zagari, R.M.; Eusebi, L.H.; Laterza, L.; Cennamo, V.; Ceroni, L.; Grilli, D.; Bazzoli, F. Meta-analysis: Can Helicobacter pylori eradication treatment reduce the risk for gastric cancer? Ann. Intern. Med. 2009, 151, 121–128. [Google Scholar] [CrossRef]
- Pasechnikov, V.; Chukov, S.; Fedorov, E.; Kikuste, I.; Leja, M. Gastric cancer: Prevention, screening and early diagnosis. World J. Gastroenterol. 2014, 20, 13842–13862. [Google Scholar] [CrossRef]
- Lordick, F.; Janjigian, Y.Y. Clinical impact of tumour biology in the management of gastroesophageal cancer. Nat. Rev. Clin. Oncol. 2016, 13, 348–360. [Google Scholar] [CrossRef] [PubMed]
- Gullo, I.; Carneiro, F.; Oliveira, C.; Almeida, G.M. Heterogeneity in Gastric Cancer: From Pure Morphology to Molecular Classifications. Pathobiology 2018, 85, 50–63. [Google Scholar] [CrossRef]
- Lauren, P. The Two Histological Main Types of Gastric Carcinoma: Diffuse and So-Called Intestinal-Type Carcinoma. An Attempt at a Histo-Clinical Classification. Acta Pathol. Microbiol. Scand. 1965, 64, 31–49. [Google Scholar] [CrossRef]
- Sasaki, S.; Nishikawa, J.; Sakai, K.; Iizasa, H.; Yoshiyama, H.; Yanagihara, M.; Shuto, T.; Shimokuri, K.; Kanda, T.; Suehiro, Y.; et al. EBV-associated gastric cancer evades T-cell immunity by PD-1/PD-L1 interactions. Gastric Cancer 2019, 22, 486–496. [Google Scholar] [CrossRef] [PubMed]
- Serra, O.; Galan, M.; Ginesta, M.M.; Calvo, M.; Sala, N.; Salazar, R. Comparison and applicability of molecular classifications for gastric cancer. Cancer Treat. Rev. 2019, 77, 29–34. [Google Scholar] [CrossRef]
- Sanjeevaiah, A.; Cheedella, N.; Hester, C.; Porembka, M.R. Gastric Cancer: Recent Molecular Classification Advances, Racial Disparity, and Management Implications. J. Oncol. Pract. 2018, 14, 217–224. [Google Scholar] [CrossRef]
- Lin, S.J.; Gagnon-Bartsch, J.A.; Tan, I.B.; Earle, S.; Ruff, L.; Pettinger, K.; Ylstra, B.; van Grieken, N.; Rha, S.Y.; Chung, H.C.; et al. Signatures of tumour immunity distinguish Asian and non-Asian gastric adenocarcinomas. Gut 2015, 64, 1721–1731. [Google Scholar] [CrossRef] [PubMed]
- Griniatsos, J.; Trafalis, D. Differences in gastric cancer surgery outcome between East and West: Differences in surgery or different diseases? J. Buon 2018, 23, 1210–1215. [Google Scholar] [PubMed]
- Hartgrink, H.H.; van de Velde, C.J.; Putter, H.; Bonenkamp, J.J.; Klein Kranenbarg, E.; Songun, I.; Welvaart, K.; van Krieken, J.H.; Meijer, S.; Plukker, J.T.; et al. Extended lymph node dissection for gastric cancer: Who may benefit? Final results of the randomized Dutch gastric cancer group trial. J. Clin. Oncol. 2004, 22, 2069–2077. [Google Scholar] [CrossRef] [PubMed]
- Cuschieri, A.; Weeden, S.; Fielding, J.; Bancewicz, J.; Craven, J.; Joypaul, V.; Sydes, M.; Fayers, P. Patient survival after D1 and D2 resections for gastric cancer: Long-term results of the MRC randomized surgical trial. Br. J. Cancer 1999, 79, 1522–1530. [Google Scholar] [CrossRef]
- Degiuli, M.; Sasako, M.; Ponti, A.; Vendrame, A.; Tomatis, M.; Mazza, C.; Borasi, A.; Capussotti, L.; Fronda, G.; Morino, M.; et al. Randomized clinical trial comparing survival after D1 or D2 gastrectomy for gastric cancer. Br. J. Surg. 2014, 101, 23–31. [Google Scholar] [CrossRef]
- Sasako, M.; Sano, T.; Yamamoto, S.; Kurokawa, Y.; Nashimoto, A.; Kurita, A.; Hiratsuka, M.; Tsujinaka, T.; Kinoshita, T.; Arai, K.; et al. D2 lymphadenectomy alone or with para-aortic nodal dissection for gastric cancer. N. Engl. J. Med. 2008, 359, 453–462. [Google Scholar] [CrossRef]
- Smyth, E.C.; Verheij, M.; Allum, W.; Cunningham, D.; Cervantes, A.; Arnold, D.; Committee, E.G. Gastric cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2016, 27, v38–v49. [Google Scholar] [CrossRef]
- NCCN Clinical Practice Guidelines in Oncology: Gastric Cancer. Version 1. 2020. Available online: https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf (accessed on 24 March 2020).
- Macdonald, J.S.; Smalley, S.R.; Benedetti, J.; Hundahl, S.A.; Estes, N.C.; Stemmermann, G.N.; Haller, D.G.; Ajani, J.A.; Gunderson, L.L.; Jessup, J.M.; et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N. Engl. J. Med. 2001, 345, 725–730. [Google Scholar] [CrossRef]
- Cunningham, D.; Allum, W.H.; Stenning, S.P.; Thompson, J.N.; Van de Velde, C.J.; Nicolson, M.; Scarffe, J.H.; Lofts, F.J.; Falk, S.J.; Iveson, T.J.; et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N. Engl. J. Med. 2006, 355, 11–20. [Google Scholar] [CrossRef]
- Al-Batran, S.E.; Homann, N.; Pauligk, C.; Goetze, T.O.; Meiler, J.; Kasper, S.; Kopp, H.G.; Mayer, F.; Haag, G.M.; Luley, K.; et al. Perioperative chemotherapy with fluorouracil plus leucovorin, oxaliplatin, and docetaxel versus fluorouracil or capecitabine plus cisplatin and epirubicin for locally advanced, resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4): A randomised, phase 2/3 trial. Lancet 2019, 393, 1948–1957. [Google Scholar] [CrossRef]
- Sakuramoto, S.; Sasako, M.; Yamaguchi, T.; Kinoshita, T.; Fujii, M.; Nashimoto, A.; Furukawa, H.; Nakajima, T.; Ohashi, Y.; Imamura, H.; et al. Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. N. Engl. J. Med. 2007, 357, 1810–1820. [Google Scholar] [CrossRef]
- Yoshida, K.; Kodera, Y.; Kochi, M.; Ichikawa, W.; Kakeji, Y.; Sano, T.; Nagao, N.; Takahashi, M.; Takagane, A.; Watanabe, T.; et al. Addition of Docetaxel to Oral Fluoropyrimidine Improves Efficacy in Patients With Stage III Gastric Cancer: Interim Analysis of JACCRO GC-07, a Randomized Controlled Trial. J. Clin. Oncol. 2019, 37, 1296–1304. [Google Scholar] [CrossRef] [PubMed]
- Bang, Y.J.; Kim, Y.W.; Yang, H.K.; Chung, H.C.; Park, Y.K.; Lee, K.H.; Lee, K.W.; Kim, Y.H.; Noh, S.I.; Cho, J.Y.; et al. Adjuvant capecitabine and oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): A phase 3 open-label, randomised controlled trial. Lancet 2012, 379, 315–321. [Google Scholar] [CrossRef]
- Lee, J.; Lim, D.H.; Kim, S.; Park, S.H.; Park, J.O.; Park, Y.S.; Lim, H.Y.; Choi, M.G.; Sohn, T.S.; Noh, J.H.; et al. Phase III trial comparing capecitabine plus cisplatin versus capecitabine plus cisplatin with concurrent capecitabine radiotherapy in completely resected gastric cancer with D2 lymph node dissection: The ARTIST trial. J. Clin. Oncol. 2012, 30, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Zang, D.Y.; Han, B.; Ji, J.H.; Kim, T.G.; Oh, S.Y.; Hwang, I.G.; Kim, J.H.; Shin, D.; Lim, D.H.; et al. ARTIST 2: Interim results of a phase III trial involving adjuvant chemotherapy and/or chemoradiotherapy after D2-gastrectomy in stage II/III gastric cancer (GC). J. Clin. Oncol. 2019, 37, 4001. [Google Scholar] [CrossRef]
- Kang, Y.K.; Yook, J.H.; Park, Y.K.; Kim, Y.W.; Kim, J.; Ryu, M.H.; Rha, S.Y.; Chung, I.J.; Kim, I.H.; Oh, S.C.; et al. Phase III randomized study of neoadjuvant chemotherapy (CT) with docetaxel(D), oxaliplatin(O) and S-1(S) (DOS) followed by surgery and adjuvant S-1, vs surgery and adjuvant S-1, for resectable advanced gastric cancer (GC) (PRODIGY). Ann. Oncol. 2019, 30, v876–v877. [Google Scholar] [CrossRef]
- Ji, J.; Shen, L.; Li, Z.; Zhang, X.; Liang, H.; Xue, Y.; Wang, Y.; Zhou, Z.; Yu, J.; Chen, L.; et al. Perioperative chemotherapy of oxaliplatin combined with S-1 (SOX) versus postoperative chemotherapy of SOX or oxaliplatin with capecitabine (XELOX) in locally advanced gastric adenocarcinoma with D2 gastrectomy: A randomized phase III trial (RESOLVE trial). Ann. Oncol. 2019, 30, v877. [Google Scholar] [CrossRef]
- Sasako, M.; Sakuramoto, S.; Katai, H.; Kinoshita, T.; Furukawa, H.; Yamaguchi, T.; Nashimoto, A.; Fujii, M.; Nakajima, T.; Ohashi, Y. Five-year outcomes of a randomized phase III trial comparing adjuvant chemotherapy with S-1 versus surgery alone in stage II or III gastric cancer. J. Clin. Oncol. 2011, 29, 4387–4393. [Google Scholar] [CrossRef]
- Noh, S.H.; Park, S.R.; Yang, H.K.; Chung, H.C.; Chung, I.J.; Kim, S.W.; Kim, H.H.; Choi, J.H.; Kim, H.K.; Yu, W.; et al. Adjuvant capecitabine plus oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): 5-year follow-up of an open-label, randomised phase 3 trial. Lancet Oncol. 2014, 15, 1389–1396. [Google Scholar] [CrossRef]
- Al-Batran, S.E.; Pauligk, C.; Homann, N.; Hartmann, J.T.; Moehler, M.; Probst, S.; Rethwisch, V.; Stoehlmacher-Williams, J.; Prasnikar, N.; Hollerbach, S.; et al. The feasibility of triple-drug chemotherapy combination in older adult patients with oesophagogastric cancer: A randomised trial of the Arbeitsgemeinschaft Internistische Onkologie (FLOT65+). Eur. J. Cancer 2013, 49, 835–842. [Google Scholar] [CrossRef]
- Cats, A.; Jansen, E.P.M.; van Grieken, N.C.T.; Sikorska, K.; Lind, P.; Nordsmark, M.; Meershoek-Klein Kranenbarg, E.; Boot, H.; Trip, A.K.; Swellengrebel, H.A.M.; et al. Chemotherapy versus chemoradiotherapy after surgery and preoperative chemotherapy for resectable gastric cancer (CRITICS): An international, open-label, randomised phase 3 trial. Lancet Oncol. 2018, 19, 616–628. [Google Scholar] [CrossRef]
- Cunningham, D.; Stenning, S.P.; Smyth, E.C.; Okines, A.F.; Allum, W.H.; Rowley, S.; Stevenson, L.; Grabsch, H.I.; Alderson, D.; Crosby, T.; et al. Peri-operative chemotherapy with or without bevacizumab in operable oesophagogastric adenocarcinoma (UK Medical Research Council ST03): Primary analysis results of a multicentre, open-label, randomised phase 2–3 trial. Lancet Oncol. 2017, 18, 357–370. [Google Scholar] [CrossRef]
- Bang, Y.J.; Van Cutsem, E.; Feyereislova, A.; Chung, H.C.; Shen, L.; Sawaki, A.; Lordick, F.; Ohtsu, A.; Omuro, Y.; Satoh, T.; et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): A phase 3, open-label, randomised controlled trial. Lancet 2010, 376, 687–697. [Google Scholar] [CrossRef]
- Hargadon, K.M.; Johnson, C.E.; Williams, C.J. Immune checkpoint blockade therapy for cancer: An overview of FDA-approved immune checkpoint inhibitors. Int. Immunopharmacol. 2018, 62, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.; Mandala, M.; Del Vecchio, M.; Gogas, H.J.; Arance, A.M.; Cowey, C.L.; Dalle, S.; Schenker, M.; Chiarion-Sileni, V.; Marquez-Rodas, I.; et al. Adjuvant Nivolumab versus Ipilimumab in Resected Stage III or IV Melanoma. N. Engl. J. Med. 2017, 377, 1824–1835. [Google Scholar] [CrossRef] [PubMed]
- Eggermont, A.M.M.; Chiarion-Sileni, V.; Grob, J.J.; Dummer, R.; Wolchok, J.D.; Schmidt, H.; Hamid, O.; Robert, C.; Ascierto, P.A.; Richards, J.M.; et al. Adjuvant ipilimumab versus placebo after complete resection of high-risk stage III melanoma (EORTC 18071): A randomised, double-blind, phase 3 trial. Lancet Oncol. 2015, 16, 522–530. [Google Scholar] [CrossRef]
- Muro, K.; Van Cutsem, E.; Narita, Y.; Pentheroudakis, G.; Baba, E.; Li, J.; Ryu, M.H.; Zamaniah, W.I.W.; Yong, W.P.; Yeh, K.H.; et al. Pan-Asian adapted ESMO Clinical Practice Guidelines for the management of patients with metastatic gastric cancer: A JSMO-ESMO initiative endorsed by CSCO, KSMO, MOS, SSO and TOS. Ann. Oncol. 2019, 30, 19–33. [Google Scholar] [CrossRef]
- Ford, H.E.R.; Marshall, A.; Bridgewater, J.A.; Janowitz, T.; Coxon, F.Y.; Wadsley, J.; Mansoor, W.; Fyfe, D.; Madhusudan, S.; Middleton, G.W.; et al. Docetaxel versus active symptom control for refractory oesophagogastric adenocarcinoma (COUGAR-02): An open-label, phase 3 randomised controlled trial. Lancet Oncol. 2014, 15, 78–86. [Google Scholar] [CrossRef]
- Thuss-Patience, P.C.; Kretzschmar, A.; Bichev, D.; Deist, T.; Hinke, A.; Breithaupt, K.; Dogan, Y.; Gebauer, B.; Schumacher, G.; Reichardt, P. Survival advantage for irinotecan versus best supportive care as second-line chemotherapy in gastric cancer—A randomised phase III study of the Arbeitsgemeinschaft Internistische Onkologie (AIO). Eur. J. Cancer 2011, 47, 2306–2314. [Google Scholar] [CrossRef]
- Kang, J.H.; Lee, S.I.; Lim, D.H.; Park, K.W.; Oh, S.Y.; Kwon, H.C.; Hwang, I.G.; Lee, S.C.; Nam, E.; Shin, D.B.; et al. Salvage chemotherapy for pretreated gastric cancer: A randomized phase III trial comparing chemotherapy plus best supportive care with best supportive care alone. J. Clin. Oncol. 2012, 30, 1513–1518. [Google Scholar] [CrossRef]
- Fuchs, C.S.; Tomasek, J.; Yong, C.J.; Dumitru, F.; Passalacqua, R.; Goswami, C.; Safran, H.; dos Santos, L.V.; Aprile, G.; Ferry, D.R.; et al. Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): An international, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet 2014, 383, 31–39. [Google Scholar] [CrossRef]
- Shitara, K.; Doi, T.; Dvorkin, M.; Mansoor, W.; Arkenau, H.T.; Prokharau, A.; Alsina, M.; Ghidini, M.; Faustino, C.; Gorbunova, V.; et al. Trifluridine/tipiracil versus placebo in patients with heavily pretreated metastatic gastric cancer (TAGS): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2018, 19, 1437–1448. [Google Scholar] [CrossRef]
- Kang, Y.K.; Boku, N.; Satoh, T.; Ryu, M.H.; Chao, Y.; Kato, K.; Chung, H.C.; Chen, J.S.; Muro, K.; Kang, W.K.; et al. Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017, 390, 2461–2471. [Google Scholar] [CrossRef]
- Fuchs, C.S.; Doi, T.; Jang, R.W.; Muro, K.; Satoh, T.; Machado, M.; Sun, W.; Jalal, S.I.; Shah, M.A.; Metges, J.P.; et al. Safety and Efficacy of Pembrolizumab Monotherapy in Patients With Previously Treated Advanced Gastric and Gastroesophageal Junction Cancer: Phase 2 Clinical KEYNOTE-059 Trial. JAMA Oncol. 2018, 4, e180013. [Google Scholar] [CrossRef]
- Takashima, A.; Iizumi, S.; Boku, N. Survival after failure of first-line chemotherapy in advanced gastric cancer patients: Differences between Japan and the rest of the world. Jpn. J. Clin. Oncol. 2017, 47, 583–589. [Google Scholar] [CrossRef]
- Iizumi, S.; Takashima, A.; Sakamaki, K.; Morita, S.; Boku, N. Survival impact of post-progression chemotherapy in advanced gastric cancer: Systematic review and meta-analysis. Cancer Chemother. Pharmacol. 2018, 81, 981–989. [Google Scholar] [CrossRef]
- Koo, D.H.; Ryu, M.H.; Ryoo, B.Y.; Seo, J.; Lee, M.Y.; Chang, H.M.; Lee, J.L.; Lee, S.S.; Kim, T.W.; Kang, Y.K. Improving trends in survival of patients who receive chemotherapy for metastatic or recurrent gastric cancer: 12 years of experience at a single institution. Gastric Cancer 2015, 18, 346–353. [Google Scholar] [CrossRef]
- Choi, I.S.; Choi, M.; Lee, J.H.; Kim, J.H.; Suh, K.J.; Lee, J.Y.; Kang, B.; Kim, J.W.; Kim, S.H.; Kim, J.W.; et al. Treatment patterns and outcomes in patients with metastatic gastric cancer receiving third-line chemotherapy: A population-based outcomes study. PLoS ONE 2018, 13, e0198544. [Google Scholar] [CrossRef]
- Chan, W.L.; Yuen, K.K.; Siu, S.W.; Lam, K.O.; Kwong, D.L. Third-line systemic treatment versus best supportive care for advanced/metastatic gastric cancer: A systematic review and meta-analysis. Crit. Rev. Oncol. Hematol. 2017, 116, 68–81. [Google Scholar] [CrossRef]
- Elimova, E.; Janjigian, Y.Y.; Mulcahy, M.; Catenacci, D.V.; Blum, M.A.; Almhanna, K.; Hecht, J.R.; Ajani, J.A. It Is Time to Stop Using Epirubicin to Treat Any Patient with Gastroesophageal Adenocarcinoma. J. Clin. Oncol. 2017, 35, 475–477. [Google Scholar] [CrossRef]
- Van Cutsem, E.; Moiseyenko, V.M.; Tjulandin, S.; Majlis, A.; Constenla, M.; Boni, C.; Rodrigues, A.; Fodor, M.; Chao, Y.; Voznyi, E.; et al. Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: A report of the V325 Study Group. J. Clin. Oncol. 2006, 24, 4991–4997. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.A.; Janjigian, Y.Y.; Stoller, R.; Shibata, S.; Kemeny, M.; Krishnamurthi, S.; Su, Y.B.; Ocean, A.; Capanu, M.; Mehrotra, B.; et al. Randomized Multicenter Phase II Study of Modified Docetaxel, Cisplatin, and Fluorouracil (DCF) Versus DCF Plus Growth Factor Support in Patients With Metastatic Gastric Adenocarcinoma: A Study of the US Gastric Cancer Consortium. J. Clin. Oncol. 2015, 33, 3874–3879. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xu, R.; Li, J.; Bai, Y.; Liu, T.; Jiao, S.; Dai, G.; Xu, J.; Liu, Y.; Fan, N.; et al. Randomized multicenter phase III study of a modified docetaxel and cisplatin plus fluorouracil regimen compared with cisplatin and fluorouracil as first-line therapy for advanced or locally recurrent gastric cancer. Gastric Cancer 2016, 19, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Al-Batran, S.E.; Hartmann, J.T.; Hofheinz, R.; Homann, N.; Rethwisch, V.; Probst, S.; Stoehlmacher, J.; Clemens, M.R.; Mahlberg, R.; Fritz, M.; et al. Biweekly fluorouracil, leucovorin, oxaliplatin, and docetaxel (FLOT) for patients with metastatic adenocarcinoma of the stomach or esophagogastric junction: A phase II trial of the Arbeitsgemeinschaft Internistische Onkologie. Ann. Oncol. 2008, 19, 1882–1887. [Google Scholar] [CrossRef] [PubMed]
- Van Cutsem, E.; Boni, C.; Tabernero, J.; Massuti, B.; Middleton, G.; Dane, F.; Reichardt, P.; Pimentel, F.L.; Cohn, A.; Follana, P.; et al. Docetaxel plus oxaliplatin with or without fluorouracil or capecitabine in metastatic or locally recurrent gastric cancer: A randomized phase II study. Ann. Oncol. 2015, 26, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Boku, N.; Mizusawa, J.; Iwasa, S.; Kadowaki, S.; Nakayama, N.; Azuma, M.; Sakamoto, T.; Shitara, K.; Tamura, T.; et al. Docetaxel plus cisplatin and S-1 versus cisplatin and S-1 in patients with advanced gastric cancer (JCOG1013): An open-label, phase 3, randomised controlled trial. Lancet Gastroenterol. Hepatol. 2019, 4, 501–510. [Google Scholar] [CrossRef]
- Mayer, R.J.; Van Cutsem, E.; Falcone, A.; Yoshino, T.; Garcia-Carbonero, R.; Mizunuma, N.; Yamazaki, K.; Shimada, Y.; Tabernero, J.; Komatsu, Y.; et al. Randomized trial of TAS-102 for refractory metastatic colorectal cancer. N. Engl. J. Med. 2015, 372, 1909–1919. [Google Scholar] [CrossRef]
- Deng, N.; Goh, L.K.; Wang, H.; Das, K.; Tao, J.; Tan, I.B.; Zhang, S.; Lee, M.; Wu, J.; Lim, K.H.; et al. A comprehensive survey of genomic alterations in gastric cancer reveals systematic patterns of molecular exclusivity and co-occurrence among distinct therapeutic targets. Gut 2012, 61, 673–684. [Google Scholar] [CrossRef]
- Wilke, H.; Muro, K.; Van Cutsem, E.; Oh, S.C.; Bodoky, G.; Shimada, Y.; Hironaka, S.; Sugimoto, N.; Lipatov, O.; Kim, T.Y.; et al. Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): A double-blind, randomised phase 3 trial. Lancet Oncol. 2014, 15, 1224–1235. [Google Scholar] [CrossRef]
- Hecht, J.R.; Bang, Y.J.; Qin, S.K.; Chung, H.C.; Xu, J.M.; Park, J.O.; Jeziorski, K.; Shparyk, Y.; Hoff, P.M.; Sobrero, A.; et al. Lapatinib in Combination With Capecitabine Plus Oxaliplatin in Human Epidermal Growth Factor Receptor 2-Positive Advanced or Metastatic Gastric, Esophageal, or Gastroesophageal Adenocarcinoma: TRIO-013/LOGiC—A Randomized Phase III Trial. J. Clin. Oncol. 2016, 34, 443–451. [Google Scholar] [CrossRef]
- Satoh, T.; Xu, R.H.; Chung, H.C.; Sun, G.P.; Doi, T.; Xu, J.M.; Tsuji, A.; Omuro, Y.; Li, J.; Wang, J.W.; et al. Lapatinib plus paclitaxel versus paclitaxel alone in the second-line treatment of HER2-amplified advanced gastric cancer in Asian populations: TyTAN—A randomized, phase III study. J. Clin. Oncol. 2014, 32, 2039–2049. [Google Scholar] [CrossRef] [PubMed]
- Swain, S.M.; Baselga, J.; Kim, S.B.; Ro, J.; Semiglazov, V.; Campone, M.; Ciruelos, E.; Ferrero, J.M.; Schneeweiss, A.; Heeson, S.; et al. Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. N. Engl. J. Med. 2015, 372, 724–734. [Google Scholar] [CrossRef] [PubMed]
- Tabernero, J.; Hoff, P.M.; Shen, L.; Ohtsu, A.; Shah, M.A.; Cheng, K.; Song, C.; Wu, H.; Eng-Wong, J.; Kim, K.; et al. Pertuzumab plus trastuzumab and chemotherapy for HER2-positive metastatic gastric or gastro-oesophageal junction cancer (JACOB): Final analysis of a double-blind, randomised, placebo-controlled phase 3 study. Lancet Oncol. 2018, 19, 1372–1384. [Google Scholar] [CrossRef]
- Ruschoff, J.; Hanna, W.; Bilous, M.; Hofmann, M.; Osamura, R.Y.; Penault-Llorca, F.; van de Vijver, M.; Viale, G. HER2 testing in gastric cancer: A practical approach. Mod. Pathol. 2012, 25, 637–650. [Google Scholar] [CrossRef]
- Hofmann, M.; Stoss, O.; Shi, D.; Buttner, R.; van de Vijver, M.; Kim, W.; Ochiai, A.; Ruschoff, J.; Henkel, T. Assessment of a HER2 scoring system for gastric cancer: Results from a validation study. Histopathology 2008, 52, 797–805. [Google Scholar] [CrossRef]
- Verma, S.; Miles, D.; Gianni, L.; Krop, I.E.; Welslau, M.; Baselga, J.; Pegram, M.; Oh, D.Y.; Dieras, V.; Guardino, E.; et al. Trastuzumab emtansine for HER2-positive advanced breast cancer. N. Engl. J. Med. 2012, 367, 1783–1791. [Google Scholar] [CrossRef]
- Krop, I.E.; Kim, S.B.; Gonzalez-Martin, A.; LoRusso, P.M.; Ferrero, J.M.; Smitt, M.; Yu, R.; Leung, A.C.; Wildiers, H. Trastuzumab emtansine versus treatment of physician’s choice for pretreated HER2-positive advanced breast cancer (TH3RESA): A randomised, open-label, phase 3 trial. Lancet Oncol. 2014, 15, 689–699. [Google Scholar] [CrossRef]
- Thuss-Patience, P.C.; Shah, M.A.; Ohtsu, A.; Van Cutsem, E.; Ajani, J.A.; Castro, H.; Mansoor, W.; Chung, H.C.; Bodoky, G.; Shitara, K.; et al. Trastuzumab emtansine versus taxane use for previously treated HER2-positive locally advanced or metastatic gastric or gastro-oesophageal junction adenocarcinoma (GATSBY): An international randomised, open-label, adaptive, phase 2/3 study. Lancet Oncol. 2017, 18, 640–653. [Google Scholar] [CrossRef]
- Ogitani, Y.; Aida, T.; Hagihara, K.; Yamaguchi, J.; Ishii, C.; Harada, N.; Soma, M.; Okamoto, H.; Oitate, M.; Arakawa, S.; et al. DS-8201a, a Novel HER2-Targeting ADC with a Novel DNA Topoisomerase I Inhibitor, Demonstrates a Promising Antitumor Efficacy with Differentiation from T-DM1. Clin. Cancer Res. 2016, 22, 5097–5108. [Google Scholar] [CrossRef]
- Ogitani, Y.; Hagihara, K.; Oitate, M.; Naito, H.; Agatsuma, T. Bystander killing effect of DS-8201a, a novel anti-human epidermal growth factor receptor 2 antibody-drug conjugate, in tumors with human epidermal growth factor receptor 2 heterogeneity. Cancer Sci. 2016, 107, 1039–1046. [Google Scholar] [CrossRef]
- Shitara, K.; Iwata, H.; Takahashi, S.; Tamura, K.; Park, H.; Modi, S.; Tsurutani, J.; Kadowaki, S.; Yamaguchi, K.; Iwasa, S.; et al. Trastuzumab deruxtecan (DS-8201a) in patients with advanced HER2-positive gastric cancer: A dose-expansion, phase 1 study. Lancet Oncol. 2019, 20, 827–836. [Google Scholar] [CrossRef]
- Ohtsu, A.; Shah, M.A.; Van Cutsem, E.; Rha, S.Y.; Sawaki, A.; Park, S.R.; Lim, H.Y.; Yamada, Y.; Wu, J.; Langer, B.; et al. Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: A randomized, double-blind, placebo-controlled phase III study. J. Clin. Oncol. 2011, 29, 3968–3976. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Li, J.; Xu, J.; Pan, H.; Dai, G.; Qin, S.; Wang, L.; Wang, J.; Yang, Z.; Shu, Y.; et al. Bevacizumab plus capecitabine and cisplatin in Chinese patients with inoperable locally advanced or metastatic gastric or gastroesophageal junction cancer: Randomized, double-blind, phase III study (AVATAR study). Gastric Cancer 2015, 18, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, C.S.; Shitara, K.; Di Bartolomeo, M.; Lonardi, S.; Al-Batran, S.E.; Van Cutsem, E.; Ilson, D.H.; Alsina, M.; Chau, I.; Lacy, J.; et al. Ramucirumab with cisplatin and fluoropyrimidine as first-line therapy in patients with metastatic gastric or junctional adenocarcinoma (RAINFALL): A double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2019, 20, 420–435. [Google Scholar] [CrossRef]
- Yoshikawa, T.; Muro, K.; Shitara, K.; Oh, D.Y.; Kang, Y.K.; Chung, H.C.; Kudo, T.; Chin, K.; Kadowaki, S.; Hamamoto, Y.; et al. Effect of First-line S-1 Plus Oxaliplatin With or Without Ramucirumab Followed by Paclitaxel Plus Ramucirumab on Advanced Gastric Cancer in East Asia: The Phase 2 RAINSTORM Randomized Clinical Trial. JAMA Netw. Open 2019, 2, e198243. [Google Scholar] [CrossRef]
- Li, J.; Qin, S.; Xu, J.; Xiong, J.; Wu, C.; Bai, Y.; Liu, W.; Tong, J.; Liu, Y.; Xu, R.; et al. Randomized, Double-Blind, Placebo-Controlled Phase III Trial of Apatinib in Patients With Chemotherapy-Refractory Advanced or Metastatic Adenocarcinoma of the Stomach or Gastroesophageal Junction. J. Clin. Oncol. 2016, 34, 1448–1454. [Google Scholar] [CrossRef]
- Kang, Y.K.; Kang, W.K.; Di Bartolomeo, M.; Chau, I.; Yoon, H.H.; Cascinu, S.; Ryu, M.H.; Kim, J.G.; Lee, K.W.; Oh, S.C.; et al. Randomized phase III ANGEL study of rivoceranib (apatinib) + best supportive care (BSC) vs placebo + BSC in patients with advanced/metastatic gastric cancer who failed ≥ 2 prior chemotherapy regimens. Ann. Oncol. 2019, 30, v877–v878. [Google Scholar] [CrossRef]
- Bennouna, J.; Sastre, J.; Arnold, D.; Österlund, P.; Greil, R.; Van Cutsem, E.; von Moos, R.; Viéitez, J.M.; Bouché, O.; Borg, C.; et al. Continuation of bevacizumab after first progression in metastatic colorectal cancer (ML18147): A randomised phase 3 trial. Lancet Oncol. 2013, 14, 29–37. [Google Scholar] [CrossRef]
- Ellis, L.M.; Hicklin, D.J. Pathways mediating resistance to vascular endothelial growth factor-targeted therapy. Clin. Cancer Res. 2008, 14, 6371–6375. [Google Scholar] [CrossRef]
- Lordick, F.; Kang, Y.K.; Chung, H.C.; Salman, P.; Oh, S.C.; Bodoky, G.; Kurteva, G.; Volovat, C.; Moiseyenko, V.M.; Gorbunova, V.; et al. Capecitabine and cisplatin with or without cetuximab for patients with previously untreated advanced gastric cancer (EXPAND): A randomised, open-label phase 3 trial. Lancet Oncol. 2013, 14, 490–499. [Google Scholar] [CrossRef]
- Waddell, T.; Chau, I.; Cunningham, D.; Gonzalez, D.; Okines, A.F.C.; Wotherspoon, A.; Saffery, C.; Middleton, G.; Wadsley, J.; Ferry, D.; et al. Epirubicin, oxaliplatin, and capecitabine with or without panitumumab for patients with previously untreated advanced oesophagogastric cancer (REAL3): A randomised, open-label phase 3 trial. Lancet Oncol. 2013, 14, 481–489. [Google Scholar] [CrossRef]
- Kim, M.A.; Lee, H.S.; Lee, H.E.; Jeon, Y.K.; Yang, H.K.; Kim, W.H. EGFR in gastric carcinomas: Prognostic significance of protein overexpression and high gene copy number. Histopathology 2008, 52, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Catenacci, D.V.T.; Tebbutt, N.C.; Davidenko, I.; Murad, A.M.; Al-Batran, S.E.; Ilson, D.H.; Tjulandin, S.; Gotovkin, E.; Karaszewska, B.; Bondarenko, I.; et al. Rilotumumab plus epirubicin, cisplatin, and capecitabine as first-line therapy in advanced MET-positive gastric or gastro-oesophageal junction cancer (RILOMET-1): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2017, 18, 1467–1482. [Google Scholar] [CrossRef]
- Shah, M.A.; Bang, Y.J.; Lordick, F.; Alsina, M.; Chen, M.; Hack, S.P.; Bruey, J.M.; Smith, D.; McCaffery, I.; Shames, D.S.; et al. Effect of Fluorouracil, Leucovorin, and Oxaliplatin With or Without Onartuzumab in HER2-Negative, MET-Positive Gastroesophageal Adenocarcinoma: The METGastric Randomized Clinical Trial. JAMA Oncol. 2017, 3, 620–627. [Google Scholar] [CrossRef]
- Catenacci, D.V.; Liao, W.L.; Thyparambil, S.; Henderson, L.; Xu, P.; Zhao, L.; Rambo, B.; Hart, J.; Xiao, S.Y.; Bengali, K.; et al. Absolute quantitation of Met using mass spectrometry for clinical application: Assay precision, stability, and correlation with MET gene amplification in FFPE tumor tissue. PLoS ONE 2014, 9, e100586. [Google Scholar] [CrossRef]
- Ohtsu, A.; Ajani, J.A.; Bai, Y.X.; Bang, Y.J.; Chung, H.C.; Pan, H.M.; Sahmoud, T.; Shen, L.; Yeh, K.H.; Chin, K.; et al. Everolimus for previously treated advanced gastric cancer: Results of the randomized, double-blind, phase III GRANITE-1 study. J. Clin. Oncol. 2013, 31, 3935–3943. [Google Scholar] [CrossRef]
- Al-Batran, S.E.; Riera-Knorrenschild, J.; Pauligk, C.; Goetze, T.O.; Hegewisch-Becker, S.; Seraphin, J.; Thuss-Patience, P.C.; Kopp, H.G.; Dechow, T.N.; Vogel, A.; et al. A randomized, double-blind, multicenter phase III study evaluating paclitaxel with and without RAD001 in patients with gastric cancer who have progressed after therapy with a fluoropyrimidine/platinum-containing regimen (RADPAC). J. Clin. Oncol. 2017, 35, 4. [Google Scholar] [CrossRef]
- Bang, Y.J.; Xu, R.H.; Chin, K.; Lee, K.W.; Park, S.H.; Rha, S.Y.; Shen, L.; Qin, S.; Xu, N.; Im, S.A.; et al. Olaparib in combination with paclitaxel in patients with advanced gastric cancer who have progressed following first-line therapy (GOLD): A double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2017, 18, 1637–1651. [Google Scholar] [CrossRef]
- Shah, M.A.; Shitara, K.; Lordick, F.; Bang, Y.J.; Tebbutt, N.C.; Metges, J.P.; Muro, K.; Shen, L.; Tjulandin, S.; Hays, J.L.; et al. The BRIGHTER trial: A phase 3 randomized double-blind study of napabucasin (NAPA) plus paclitaxel (PTX) versus placebo (PBO) plus PTX in patients (pts) with pretreated advanced gastric and gastroesophageal junction (GEJ) adenocarcinoma. J. Clin. Oncol. 2018, 36, 4010. [Google Scholar] [CrossRef]
- Shah, M.A.; Ruiz, E.P.Y.; Bodoky, G.; Starodub, A.; Cunningham, D.; Yip, D.; Wainberg, Z.A.; Bendell, J.C.; Thai, D.; Bhargava, P.; et al. A phase III, randomized, double-blind, placebo-controlled study to evaluate the efficacy and safety of andecaliximab combined with mFOLFOX6 as first-line treatment in patients with advanced gastric or gastroesophageal junction adenocarcinoma (GAMMA-1). J. Clin. Oncol. 2019, 37, 4. [Google Scholar] [CrossRef]
- Schuler, M.; Al-Batran, S.; Zvirbule, Z.; Manikhas, G.; Lordick, F.; Rusyn, A.; Vinnyk, Y.; Vynnychenko, I.; Fadeeva, N.; Nechaeva, M.; et al. Final results of the FAST study, an international, multicenter, randomized, phase II trial of epirubicin, oxaliplatin, and capecitabine (EOX) with or without the anti-CLDN18.2 antibody IMAB362 as first-line therapy in patients with advanced CLDN18.2+ gastric and gastroesophageal junction (GEJ) adenocarcinoma. Ann. Oncol. 2016, 27, 207–242. [Google Scholar] [CrossRef]
- Pearson, A.; Smyth, E.; Babina, I.S.; Herrera-Abreu, M.T.; Tarazona, N.; Peckitt, C.; Kilgour, E.; Smith, N.R.; Geh, C.; Rooney, C.; et al. High-Level Clonal FGFR Amplification and Response to FGFR Inhibition in a Translational Clinical Trial. Cancer Discov. 2016, 6, 838–851. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, M.S.; Stojanov, P.; Polak, P.; Kryukov, G.V.; Cibulskis, K.; Sivachenko, A.; Carter, S.L.; Stewart, C.; Mermel, C.H.; Roberts, S.A.; et al. Mutational heterogeneity in cancer and the search for new cancer-associated genes. Nature 2013, 499, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Boger, C.; Behrens, H.M.; Mathiak, M.; Kruger, S.; Kalthoff, H.; Rocken, C. PD-L1 is an independent prognostic predictor in gastric cancer of Western patients. Oncotarget 2016, 7, 24269–24283. [Google Scholar] [CrossRef]
- Kim, J.W.; Nam, K.H.; Ahn, S.H.; Park, D.J.; Kim, H.H.; Kim, S.H.; Chang, H.; Lee, J.O.; Kim, Y.J.; Lee, H.S.; et al. Prognostic implications of immunosuppressive protein expression in tumors as well as immune cell infiltration within the tumor microenvironment in gastric cancer. Gastric Cancer 2014, 19, 42–52. [Google Scholar] [CrossRef]
- Takaya, S.; Saito, H.; Ikeguchi, M. Upregulation of Immune Checkpoint Molecules, PD-1 and LAG-3, on CD4+ and CD8+ T Cells after Gastric Cancer Surgery. Yonago Acta Med. 2015, 58, 39–44. [Google Scholar]
- Bang, Y.J.; Ruiz, E.Y.; Van Cutsem, E.; Lee, K.W.; Wyrwicz, L.; Schenker, M.; Alsina, M.; Ryu, M.H.; Chung, H.C.; Evesque, L.; et al. Phase III, randomised trial of avelumab versus physician’s choice of chemotherapy as third-line treatment of patients with advanced gastric or gastro-oesophageal junction cancer: Primary analysis of JAVELIN Gastric 300. Ann. Oncol. 2018, 29, 2052–2060. [Google Scholar] [CrossRef]
- Chung, H.C.; Arkenau, H.T.; Lee, J.; Rha, S.Y.; Oh, D.Y.; Wyrwicz, L.; Kang, Y.K.; Lee, K.W.; Infante, J.R.; Lee, S.S.; et al. Avelumab (anti-PD-L1) as first-line switch-maintenance or second-line therapy in patients with advanced gastric or gastroesophageal junction cancer: Phase 1b results from the JAVELIN Solid Tumor trial. J. Immunother. Cancer 2019, 7, 30. [Google Scholar] [CrossRef]
- Doi, T.; Iwasa, S.; Muro, K.; Satoh, T.; Hironaka, S.; Esaki, T.; Nishina, T.; Hara, H.; Machida, N.; Komatsu, Y.; et al. Phase 1 trial of avelumab (anti-PD-L1) in Japanese patients with advanced solid tumors, including dose expansion in patients with gastric or gastroesophageal junction cancer: The JAVELIN Solid Tumor JPN trial. Gastric Cancer 2019, 22, 817–827. [Google Scholar] [CrossRef]
- Janjigian, Y.Y.; Bendell, J.; Calvo, E.; Kim, J.W.; Ascierto, P.A.; Sharma, P.; Ott, P.A.; Peltola, K.; Jaeger, D.; Evans, J.; et al. CheckMate-032 Study: Efficacy and Safety of Nivolumab and Nivolumab Plus Ipilimumab in Patients With Metastatic Esophagogastric Cancer. J. Clin. Oncol. 2018, 36, 2836–2844. [Google Scholar] [CrossRef]
- Bang, Y.J.; Kang, Y.K.; Catenacci, D.V.; Muro, K.; Fuchs, C.S.; Geva, R.; Hara, H.; Golan, T.; Garrido, M.; Jalal, S.I.; et al. Pembrolizumab alone or in combination with chemotherapy as first-line therapy for patients with advanced gastric or gastroesophageal junction adenocarcinoma: Results from the phase II nonrandomized KEYNOTE-059 study. Gastric Cancer 2019, 22, 828–837. [Google Scholar] [CrossRef] [PubMed]
- Brahmer, J.R.; Lacchetti, C.; Schneider, B.J.; Atkins, M.B.; Brassil, K.J.; Caterino, J.M.; Chau, I.; Ernstoff, M.S.; Gardner, J.M.; Ginex, P.; et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 2018, 36, 1714–1768. [Google Scholar] [CrossRef] [PubMed]
- Shitara, K.; Özgüroğlu, M.; Bang, Y.J.; Di Bartolomeo, M.; Mandalà, M.; Ryu, M.H.; Fornaro, L.; Olesiński, T.; Caglevic, C.; Chung, H.C.; et al. Pembrolizumab versus paclitaxel for previously treated, advanced gastric or gastro-oesophageal junction cancer (KEYNOTE-061): A randomised, open-label, controlled, phase 3 trial. Lancet 2018, 392, 123–133. [Google Scholar] [CrossRef]
- Ramakrishnan, R.; Assudani, D.; Nagaraj, S.; Hunter, T.; Cho, H.I.; Antonia, S.; Altiok, S.; Celis, E.; Gabrilovich, D.I. Chemotherapy enhances tumor cell susceptibility to CTL-mediated killing during cancer immunotherapy in mice. J. Clin. Invest. 2010, 120, 1111–1124. [Google Scholar] [CrossRef] [PubMed]
- Tabernero, J.; Cutsem, E.V.; Bang, Y.J.; Fuchs, C.S.; Wyrwicz, L.; Lee, K.W.; Kudaba, I.; Garrido, M.; Chung, H.C.; Salguero, H.R.C.; et al. Pembrolizumab with or without chemotherapy versus chemotherapy for advanced gastric or gastroesophageal junction (G/GEJ) adenocarcinoma: The phase III KEYNOTE-062 study. J. Clin. Oncol. 2019, 37, LBA4007. [Google Scholar] [CrossRef]
- Shitara, K.; Van Cutsem, E.; Bang, Y.J.; Fuchs, C.S.; Wyrwicz, L.; Lee, K.W.; Kudaba, I.; Garrido, M.; Cheol Chung, H.; Castro, H.R.; et al. Pembrolizumab with or without chemotherapy vs chemotherapy in patients with advanced G/GEJ cancer (GC) including outcomes according to Microsatellite Instability-High (MSI-H) status in KEYNOTE-062. Ann. Oncol. 2019, 30, v878–v879. [Google Scholar] [CrossRef]
- Moehler, M.H.; Dvorkin, M.; Ozguroglu, M.; Ryu, M.H.; Muntean, A.S.; Lonardi, S.; Nechaeva, M.; Bragagnoli, A.S.C.; Coskun, H.S.; Gracián, A.C.; et al. Results of the JAVELIN Gastric 100 phase 3 trial: Avelumab maintenance following first-line (1L) chemotherapy (CTx) vs continuation of CTx for HER2− advanced gastric or gastroesophageal junction cancer (GC/GEJC). J. Clin. Oncol. 2020, 38, 278. [Google Scholar] [CrossRef]
- Buchbinder, E.I.; Desai, A. CTLA-4 and PD-1 Pathways: Similarities, Differences, and Implications of Their Inhibition. Am. J. Clin. Oncol. 2016, 39, 98–106. [Google Scholar] [CrossRef]
- Park, S.; Jiang, Z.; Mortenson, E.D.; Deng, L.; Radkevich-Brown, O.; Yang, X.; Sattar, H.; Wang, Y.; Brown, N.K.; Greene, M.; et al. The Therapeutic Effect of Anti-HER2/neu Antibody Depends on Both Innate and Adaptive Immunity. Cancer Cell 2010, 18, 160–170. [Google Scholar] [CrossRef]
- Rahma, O.E.; Hodi, F.S. The Intersection between Tumor Angiogenesis and Immune Suppression. Clin. Cancer Res. 2019, 25, 5449–5457. [Google Scholar] [CrossRef]
- Schmittnaegel, M.; De Palma, M. Reprogramming Tumor Blood Vessels for Enhancing Immunotherapy. Trends Cancer 2017, 3, 809–812. [Google Scholar] [CrossRef] [PubMed]
- Chau, I.; Bendell, J.; Soriano, A.; Arkenau, H.; Cultrera, J.; Santana-Davila, R.; Calvo, E.; Tourneau, C.L.; Zender, L.; Mi, G.; et al. Safety and antitumor activity from the phase Ib study of ramucirumab plus pembrolizumab in treatment-naive advanced gastric or gastroesophageal junction (G/GEJ) adenocarcinoma (JVDF). Ann. Oncol. 2019, 30, iv122. [Google Scholar] [CrossRef]
- Hara, H.; Shoji, H.; Takahari, D.; Esaki, T.; Machida, N.; Nagashima, K.; Aoki, K.; Honda, K.; Miyamoto, T.; Boku, N.; et al. Phase I/II study of ramucirumab plus nivolumab in patients in second-line treatment for advanced gastric adenocarcinoma (NivoRam study). J. Clin. Oncol. 2019, 37, 129. [Google Scholar] [CrossRef]
- Kadowaki, S.; Izawa, N.; Minashi, K.; Nishina, T.; Yamanaka, T.; Muro, K.; Sunakawa, Y.; Hironaka, S.; Kajiwara, T.; Kawakami, Y.; et al. Multicenter phase I/II study of nivolumab combined with paclitaxel plus ramucirumab as the second-line treatment in patients with advanced gastric cancer. Ann. Oncol. 2019, 30, iv122. [Google Scholar] [CrossRef]
- Arai, H.; Battaglin, F.; Wang, J.; Lo, J.H.; Soni, S.; Zhang, W.; Lenz, H.J. Molecular insight of regorafenib treatment for colorectal cancer. Cancer Treat. Rev. 2019, 81, 101912. [Google Scholar] [CrossRef]
- Fukuoka, S.; Hara, H.; Takahashi, N.; Kojima, T.; Kawazoe, A.; Asayama, M.; Yoshii, T.; Kotani, D.; Tamura, H.; Mikamoto, Y.; et al. Regorafenib plus nivolumab in patients with advanced gastric (GC) or colorectal cancer (CRC): An open-label, dose-finding, and dose-expansion phase 1b trial (REGONIVO, EPOC1603). J. Clin. Oncol. 2019, 37, 2522. [Google Scholar] [CrossRef]
- Kawazoe, A.; Fukuoka, S.; Nakamura, Y.; Kuboki, Y.; Mikamoto, Y.; Shima, H.; Fujishiro, N.; Higuchi, T.; Wakabayashi, M.; Nomura, S.; et al. An open-label phase II study of lenvatinib plus pembrolizumab in patients with advanced gastric cancer (EPOC1706). J. Clin. Oncol. 2020, 38, 374. [Google Scholar] [CrossRef]
| Study | Year | Region | Phase | Setting | N | Subject | Lymphadenectomy | Treatment Arm | PE | Result |
|---|---|---|---|---|---|---|---|---|---|---|
| INT0116 | 2001 | US | III | Post | 603 | GC after curative resection | D0: 54% D1: 36% D2: 10% | CRT (5-FU/FA) Surgery alone | RFS/OS | Positive |
| CALGB80101 | 2017 | US | III | Post | 546 | GC after curative resection | NA | CRT (ECF) CRT (5-FU/FA) | OS | Negative |
| MAGIC | 2006 | UK | III | Peri | 503 | Resectable GC (including the lower esophagus) | D1: 18% D2: 38% | ECF Surgery alone | OS | Positive |
| MRC ST03 | 2017 | UK | II/III | Peri | 1063 | Resectable GC (including the lower esophagus) | Not available | ECF + bevacizumab ECF | OS | Negative |
| FLOT4 | 2019 | Germany | II/III | Peri | 716 | Resectable GC | D1: 2% D2: 55% | FLOT ECF (ECX) | OS | Positive |
| CRITICS | 2018 | Netherlands | III | Peri | 788 | Resectable GC | D1+: 79% D2: 6% | Pre ECX (EOX) + Post CRT (XP) Peri ECX (EOX) | OS | Negative |
| ACTS-GC | 2007 | Japan | III | Post | 1059 | GC after curative resection | D2: 94% D3: 6% | S-1 Surgery alone | OS | Positive |
| START2 | 2019 | Japan | III | Post | 915 | GC after curative resection (Only stage III cases) | D2: 100% | S-1+DTX S-1 | RFS | Positive |
| CLASSIC | 2012 | Korea | III | Post | 1035 | GC after curative resection | D2: 100% | CAPOX Surgery alone | DFS | Positive |
| ARTIST | 2012 | Korea | III | Post | 458 | GC after curative resection | D2: 100% | XP XP + CRT (X) | DFS | Negative |
| ARTIST-II | 2019 | Korea | III | Post | 538 | GC after curative resection (Only LN-positive cases) | D2: 100% | SOX + CRT (S-1) SOX S-1 | DFS | Positive Negative |
| PRODIGY | 2019 | Korea | III | Peri | 530 | Resectable GC (cT2-3/N+, or T4) | D2: 98% | Pre DOS + Post S-1 Post S-1 | PFS | Positive |
| RESOLVE | 2019 | China | III | Peri/Post | 1022 | Resectable GC (cT4a/N+, or cT4b) | D2: 100% | Peri SOX Post SOX (non-inferiority) Post CAPOX | DFS | Positive |
| Study | Region | Phase | Setting | N | Subject | Treatment Arm | PE |
|---|---|---|---|---|---|---|---|
| INNOVATION (NCT02205047) | Europe | rII | Peri | 220 | HER2-positive resectable GC | XP (FP) + trastuzumab + pertuzumab XP (FP) + trastuzumab XP (FP) | Near pCR rate |
| TOPGEAR (NCT01924819) | Australia Canada Europe | II/III | Peri | 620 | Resectable GC | Peri ECF (ECX, EOX) or FLOT + Pre CRT (5-FU or X) Peri ECF (ECX, EOX) or FLOT | OS |
| PETRARCA (FLOT6) (NCT02581462) | Germany | II/III | Peri | 81 | HER2-positive resectable GC | FLOT + trastuzumab + pertuzumab FLOT | DFS |
| RAMSES (FLOT7) (NCT02661971) | Germany | II/III | Peri | 908 | HER2-negative resectable GC | FLOT + ramucirumab FLOT | OS |
| DANTE (NCT03421288) | Germany | rII | Peri | 295 | Resectable GC | FLOT + atezolizumab FLOT | DFS PFS |
| IMAGINE (NCT04062656) | Germany | rII | Peri | 88 | Resectable GC | Nivolumab Nivolumab + ipilimumab Nivolumab + relatlimab (anti-LAG3) FLOT | pCR rate |
| CRITICS-II (NCT02931890) | Netherlands | rII | Pre | 207 | Resectable GC | DOC DOC + CRT (CBDCA+PTX) CRT (CBDCA+PTX) | EFS |
| ATTRACTION-05 (NCT03006705) | Asia | III | Post | 700 | GC after curative resection (pStage III) | S-1 or CAPOX + nivolumab S-1 or CAPOX + placebo | RFS |
| NAGISA (JCOG1509) (UMIN000024065) | Japan | III | Peri | 470 | Resectable GC (cT3-4/N1-3) | Pre SOX + Post S-1 or DS Post S-1 or DS | OS |
| Trigger (JCOG1301C) (UMIN000016920) | Japan | rII | Peri | 130 | HER2-positive resectable GC with extensive LN metastasis | Pre SP + trastuzumab + Post S-1 Pre SP + Post S-1 | OS |
| Neo-CRAG (NCT01815853) | China | III | Peri | 620 | Resectable GC (cT3/N2-3, cT4a/N+, or cT4b) | Pre CRT (CAPOX) + Post CAPOX Peri CAPOX | DFS |
| PREACT (NCT03013010) | China | III | Peri | 682 | Resectable GC (cStage IIB and III) | Peri SOX + Pre CRT (S-1) Peri SOX | DFS |
| RESCUE-GC (NCT02867839) | China | III | Post | 564 | GC after curative resection (pStage II and IIIA) | SOX S-1 | DFS |
| KEYNOTE-585 (NCT03221426) | International | III | Peri | 860 | Resectable GC | XP (FP) + pembrolizumab XP (FP) + placebo FLOT + pembrolizumab FLOT + placebo | OS EFS pCR rate |
| Study | Year | Region | Phase | Target | Drug | Line | Subject | N | TREATMENT ARM | PE | Result |
|---|---|---|---|---|---|---|---|---|---|---|---|
| ToGA | 2010 | International | III | HER2 | Trastuzumab | 1L | HER2 positive (IHC3+, FISH+) | 594 | XP (FP) + trastuzumab XP (FP) | OS | Positive |
| TRIO-013/LOGiC | 2016 | International | III | HER1/2 | Lapatinib | 1L | HER2 positive (FISH+) | 545 | CAPOX + lapatinib CAPOX + placebo | OS | Negative |
| TyTAN | 2014 | Asia | III | HER1/2 | Lapatinib | 2L | HER2 positive (FISH+) | 261 | PTX + lapatinib PTX | OS | Negative |
| JACOB | 2018 | International | III | HER2 | Pertuzumab | 1L | HER2 positive (IHC3+, IHC2+/FISH+) | 780 | XP (FP) + trastuzumab + pertuzumab XP (FP) + trastuzumab + placebo | OS | Negative |
| GATSBY | 2017 | International | II/III | HER2 | T-DM1 | 2L | HER2 positive (IHC3+, IHC2+/FISH+) | 345 | T-DM1 Taxane (DTX or PTX) | OS | Negative |
| ASLAN001-012 (NCT03130790) | Ongoing | Mainly Asia | II/III | HER1/2/4 | Varlitinib | 1L | HER1/2 co-expressing | 400 | mFOLFOX6 + varlitinib mFOLFOX6 + placebo | OS | Not yet |
| EXPAND | 2013 | International | III | HER1 | Cetuximab | 1L | All | 904 | XP + cetuximab XP | PFS | Negative |
| REAL3 | 2013 | UK | III | HER1 | Panitumumab | 1L | All | 553 | mEOC + panitumumab EOC | OS | Negative |
| RILOMET-1 | 2017 | International without Asia | III | HGF | Rilotumumab | 1L | MET positive HER2 negative | 609 | ECX + rilotumumab ECX + placebo | OS | Negative |
| METGastric | 2017 | International | III | MET | Onartuzumab | 1L | MET positive HER2 negative | 562 | mFOLFOX6 + onartuzumab mFOLFOX6 + placebo | OS | Negative |
| FIGHT (NCT03694522) | Ongoing | International | III | FGFR2b | Bemarituzumab (FPA144) | 1L | FGFR2b overexpression or FGFR2 amplification HER2 negative | 548 | mFOLFOX6+bemarituzumab mFOLFOX6 + placebo | OS | Not yet |
| GRANITE-1 | 2013 | International | III | mTOR | Everolimus | 2L/3L | All | 656 | Everolimus + BSC Placebo + BSC | OS | Negative |
| RADPAC | 2017 | Germany | III | mTOR | Everolimus | 2L/3L /4L | All | 300 | PTX + everolimus PTX + placebo | OS | Negative |
| GOLD | 2017 | Asia | III | PARP | Olaparib | 2L | All | 525 | PTX + olaparib PTX + placebo | OS | Negative |
| PARALLEL 303 (NCT03427814) | Ongoing | International | III | PARP | Pamiparib (BGB-290) | 1L | All | 540 | Pamiparib (maintenance) Placebo (maintenance) | PFS | Not yet |
| BRIGHTER | 2018 | International | III | STAT3 | Napabucasin | 2L | All | 714 | PTX + napabucasin PTX + placebo | OS | Negative |
| GAMMA-1 | 2019 | International without Asia | III | MMP9 | Andecaliximab | 1L | HER2 negative | 432 | mFOLFOX6 + andecaliximab mFOLFOX6 + placebo | OS | Negative |
| SPOTLIGHT (NCT03504397) | Ongoing | International | III | Claudin 18.2 | Zolbetuximab (IMAB362) | 1L | Claudin 18.2 positive HER2 negative | 550 | mFOLFOX6 + zolbetuximab mFOLFOX + placebo | PFS | Not yet |
| GLOW (NCT03653507) | Ongoing | International | III | Claudin 18.2 | Zolbetuximab (IMAB362) | 1L | Claudin 18.2 positive HER2 negative | 500 | CAPOX + zolbetuximab CAPOX + placebo | PFS | Not yet |
| AVAGAST | 2011 | International | III | VEGFA | Bevacizumab | 1L | All | 774 | XP (FP) + bevacizumab XP (FP) + placebo | OS | Negative |
| AVATAR | 2015 | China | III | VEGFA | Bevacizumab | 1L | All | 202 | XP + bevacizumab XP + placebo | OS | Negative |
| RAINBOW | 2014 | International | III | VEGFR2 | Ramucirumab | 2L | All | 665 | PTX + ramucirumab PTX + placebo | OS | Positive |
| REGARD | 2014 | International | III | VEGFR2 | Ramucirumab | 2L | All | 355 | Ramucirumab + BSC Placebo + BSC | OS | Positive |
| RAINFALL | 2019 | International | III | VEGFR2 | Ramucirumab | 1L | HER2 negative | 645 | XP (FP) + ramucirumab XP (FP) + placebo | PFS | Positive |
| RINDBeRG (UMIN000023065) | Ongoing | Japan | III | VEGFR2 | Ramucirumab | 3L | All | 400 | IRI + ramucirumab (beyond progression) IRI | OS | Not yet |
| ARMANI (NCT02934464) | Ongoing | Italy | III | VEGFR2 | Ramucirumab | 1L | HER2 negative | 280 | PTX + ramucirumab (switch maintenance) FOLFOX4, mFOLFOX6. or CAPOX | PFS | Not yet |
| HENGRUI 20101208 | 2016 | China | III | VEGFR2 | Apatinib | ≥ 3L | All | 267 | Apatinib Placebo | OS PFS | Positive |
| ANGEL | 2019 | International | III | VEGFR2 | Apatinib | ≥ 3L | All | 460 | Apatinib + BSC Placebo + BSC | OS | Negative |
| TJCC006 (NCT03598348) | Ongoing | China | III | VEGFR2 | Apatinib | 1L | HER2 negative | 288 | Apatinib + X (maintenance after CAPOX) Apatinib (maintenance after CAPOX) Observation (after CAPOX) | PFS | Not yet |
| FRUTIGA (NCT03223376) | Ongoing | China | III | VEGFR1/2/3 | Fruquintinib | 2L | All | 544 | PTX + fruquintinib PTX + placebo | OS | Not yet |
| INTEGRATE II (NCT02773524) | Ongoing | International | III | Multi-target | Regorafenib | ≥ 3L | All | 350 | Regorafenib Placebo | OS | Not yet |
| Study | Region | Phase | Drugs (Target) | Line | N | Subject | Treatment Arm | PE |
|---|---|---|---|---|---|---|---|---|
| AK104 (NCT03852251) | China | I/II | AK104 (PD-1/CTLA-4) | 1L | 112 | HER2 negative | AK104 CAPOX+AK104 | ORR |
| CP-MGAH22-05 (NCT02689284) | International | I/II | Pembrolizumab (PD-1) Margetuximab (HER2) | ≥ 2L | 95 | HER2 positive | Margetuximab + pembrolizumab | ORR DOR |
| KEYNOTE-659 (NCT03382600) | Japan | II | Pembrolizumab (PD-1) | 1L | 90 | PD-L1 positive HER2 negative | SOX + pembrolizumab (Cohort 1) SP + pembrolizumab (Cohort 2) | ORR |
| EPOC1706 (NCT03609359) | Japan | II | Pembrolizumab (PD-1) Lenvatinib (multi-target) | - | 29 | All | Lenvatinib + pembrolizumab | ORR |
| ESR-15-11655 (NCT03579784) | Korea | II | Durvalumab (PD-1) Olaparib (PARP) | 2L | 40 | All | PTX + olaparib + durvalumab | DCR |
| NCC2070 (NCT04140318) | China | II | Sintilimab (PD-1) | 2L | 60 | All | Nab-PTX + sintilimab | ORR |
| ASGARD (NCT04089657) | China | II | Sintilimab (PD-1)Apatinib (VEGFR2) | ≥ 3L | 40 | All | Apatinib + sintilimab | DCR |
| RiME (NCT03995017) | US | II | Nivolumab (PD-1) Rucaparib (PARP) Ramucirumab (VEGFR2) | 2L/3L | 61 | All | Rucaparib + ramucirumab + nivolumab Rucaparib + ramucirumab | ORR |
| 16-937 (NCT02954536) | US | II | Pembrolizumab (PD-1) Trastuzumab (HER2) | 1L | 37 | HER2 positive | XP (CAPOX) + trastuzumab + pembrolizumab | PFS |
| RAP (AIO-STO-0218) (NCT03966118) | Germany | II | Avelumab (PD-1) Ramucirumab (VEGFR2) | 2L | 59 | All | PTX + ramucirumab + avelumab | OS |
| INTEGA (AIO-STO-0217) (NCT03409848) | Germany | rII | Nivolumab (PD-1) Ipilimumab (CTLA-4) Trastuzumab (HER2) | 1L | 97 | HER2 positive | Nivolumab + ipilimumab + trastuzumab mFOLFOX6 + trastuzumab + nivolumab | OS |
| MOONLIGHT (AIO-STO-0417) (NCT03647969) | Germany | rII | Nivolumab (PD-1) Ipilimumab (CTLA-4) | 1L | 118 | HER2 negative | mFOLFOX6 + nivolumab + ipilimumab mFOLFOX6 | PFS |
| DURIGAST (PRODIGE59-FFCD1707) (NCT03959293) | France | rII | Durvalumab (PD-L1) Tremelimumab (CTLA-4) | 2L | 105 | All | FOLFIRI + durvalumab + tremelimumab FOLFIRI + durvalumab | PFS |
| SEQUEL (NCT04069273) | US | rII | Pembrolizumab (PD-1) Ramucirumab (VEGFR2) | ≥ 2L | 58 | All | PTX + ramucirumab + pembrolizumab (with patient-tailored algorithm) PTX + ramucirumab + pembrolizumab | ORR |
| CA224-060 (NCT03662659) | International without Asia | rII | Nivolumab (LAG-3) Relatlimab (PD-1) | 1L | 250 | HER2 negative | CAPOX (FOLFOX, SOX) + nivolumab + relatlimab CAPOX (FOLFOX, SOX) + nivolumab | ORR |
| FRACTION-GC (NCT02935634) | International without Asia | rII | Nivolumab (PD-1) Ipilimumab (CTLA-4) Relatlimab (LAG-3) BMS-986205 (IDO1) Rucaparib (PARP) | - | 600 | All | Nivolumab + relatlimab Nivolumab + BMS-986205 Nivolumab + rucaparib Ipilimumab + rucaparib Nivolumab + ipilimumab + rucaparib Nivolumab + ipilimumab | ORR DOR PFS |
| ATTRACTION-04 (NCT02746796) | Asia | II/III | Nivolumab (PD-1) | 1L | 680 | HER2 negative | CAPOX (SOX ) + nivolumab CAPOX (SOX) + placebo | OS PFS |
| MAHOGANY (NCT04082364) | US | II/III | MGA012 (PD-1) MGD013 (PD-1/LAG-3) Margetuximab (HER2) | 1L | 850 | Cohort A: HER2/PD-L1 positive Cohort B: HER2 positive | Margetuximab + MGA012 CAPOX (mFOLFOX6) + margetuximab + MGA012 CAPOX (mFOLFOX6) + margetuximab + MGD013 CAPOX (mFOLFOX6) + margetuximab CAPOX (mFOLFOX6) + trastuzumab | Cohort A: ORR Cohort B: OS |
| KEYNOTE-063 (NCT03019588) | Asia | III | Pembrolizumab (PD-1) | 2L | 360 | PD-L1 positive | Pembrolizumab PTX | OS PFS |
| GEMSTONE-303 (NCT03802591) | China | III | CS1001 (PD-L1) | 1L | 480 | HER2 negative | CAPOX + CS1001 CAPOX + placebo | OS PFS |
| SHR-1210-III-311 (NCT03813784) | China | III | SHR-1210 (PD-1) Apatinib (VEGFR2) | 1L | 568 | HER2 negative | CAPOX + SHR-1210 followed by apatinib + SHR-1210 CAPOX | OS |
| CIBI308E301 (NCT03745170) | China | III | Sintilimab (PD-1) | 1L | 650 | HER2 negative | CAPOX + sintilimab CAPOX + placebo | OS |
| CheckMate 649 (NCT02872116) | International | III | Pembrolizumab (PD-1) Ipilimumab (CTLA-4) | 1L | 2005 | HER2 negative | Nivolumab + ipilimumab CAPOX (FOLFOX) + nivolumab CAPOX (FOLFOX) | OS PFS |
| KEYNOTE-811 (NCT03615326) | International | III | Pembrolizumab (PD-1) Trastuzumab (HER2) | 1L | 732 | HER2 positive | FP (CAPOX, SOX) + trastuzumab + pembrolizumab FP (CAPOX, SOX) + trastuzumab + placebo | OS PFS |
| KEYNOTE-859 (NCT03675737) | International | III | Pembrolizumab (PD-1) | 1L | 780 | HER2 negative | FP (CAPOX) + pembrolizumab FP (CAPOX)+placebo | OS PFS |
| BGB-A317-305 (NCT03777657) | International | III | Tislelizumab (PD-1) | 1L | 720 | HER2 negative | CAPOX (FP) + tislelizumab CAPOX (FP)+placebo | OS PFS |
| Variation | East | West |
|---|---|---|
| Incidence (percentage in the world) | Asia: 48.4% | Europe: 23.4% North America: 13.2% |
| Localization of the primary legion | Distal third of the stomach | Proximal third of the stomach |
| Stage at the diagnosis | Early stage | Late stage |
| Overall survival | Better | Worse |
| Nationwide screening program | Present (Japan, South Korea, Matsu Islands in Taiwan) | Absent |
| Endoscopic resection for early stage GC | Common | Uncommon |
| Prevalence of Helicobacter pylori infection | Asia: 54.7% | Europe: 47.0% North America: 37.1% |
| Immune profiles |
|
|
| Surgery for resectable GC | D2 gastrectomy | D1 gastrectomy |
| Adjuvant treatment | Asia: post-operative chemotherapy (CAPOX for 6M, S-1 for 1Y, S-1+DTX for Stage III in Japan) | Europe: peri-operative chemotherapy (FLOT) US: post-operative chemoradiotherapy |
| 1L treatment for advanced GC | HER2-negative GC: platinum + fluoropyrimidine HER2-positive GC: platinum + fluoropyrimidine + trastuzumab | |
| 2L treatment for advanced GC |
| |
| 3L treatment for advanced GC |
|
|
| Post-progression treatment | 1L to 2L: 69–85% (in Japan) 2L to 3L: 58–90% (in Japan) | 1L to 2L: 11–59% 2L to 3L: 8–29% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arai, H.; Nakajima, T.E. Recent Developments of Systemic Chemotherapy for Gastric Cancer. Cancers 2020, 12, 1100. https://doi.org/10.3390/cancers12051100
Arai H, Nakajima TE. Recent Developments of Systemic Chemotherapy for Gastric Cancer. Cancers. 2020; 12(5):1100. https://doi.org/10.3390/cancers12051100
Chicago/Turabian StyleArai, Hiroyuki, and Takako Eguchi Nakajima. 2020. "Recent Developments of Systemic Chemotherapy for Gastric Cancer" Cancers 12, no. 5: 1100. https://doi.org/10.3390/cancers12051100
APA StyleArai, H., & Nakajima, T. E. (2020). Recent Developments of Systemic Chemotherapy for Gastric Cancer. Cancers, 12(5), 1100. https://doi.org/10.3390/cancers12051100




