Analysis of BRCA1 and RAD51C Promoter Methylation in Italian Families at High-Risk of Breast and Ovarian Cancer
Abstract
1. Introduction
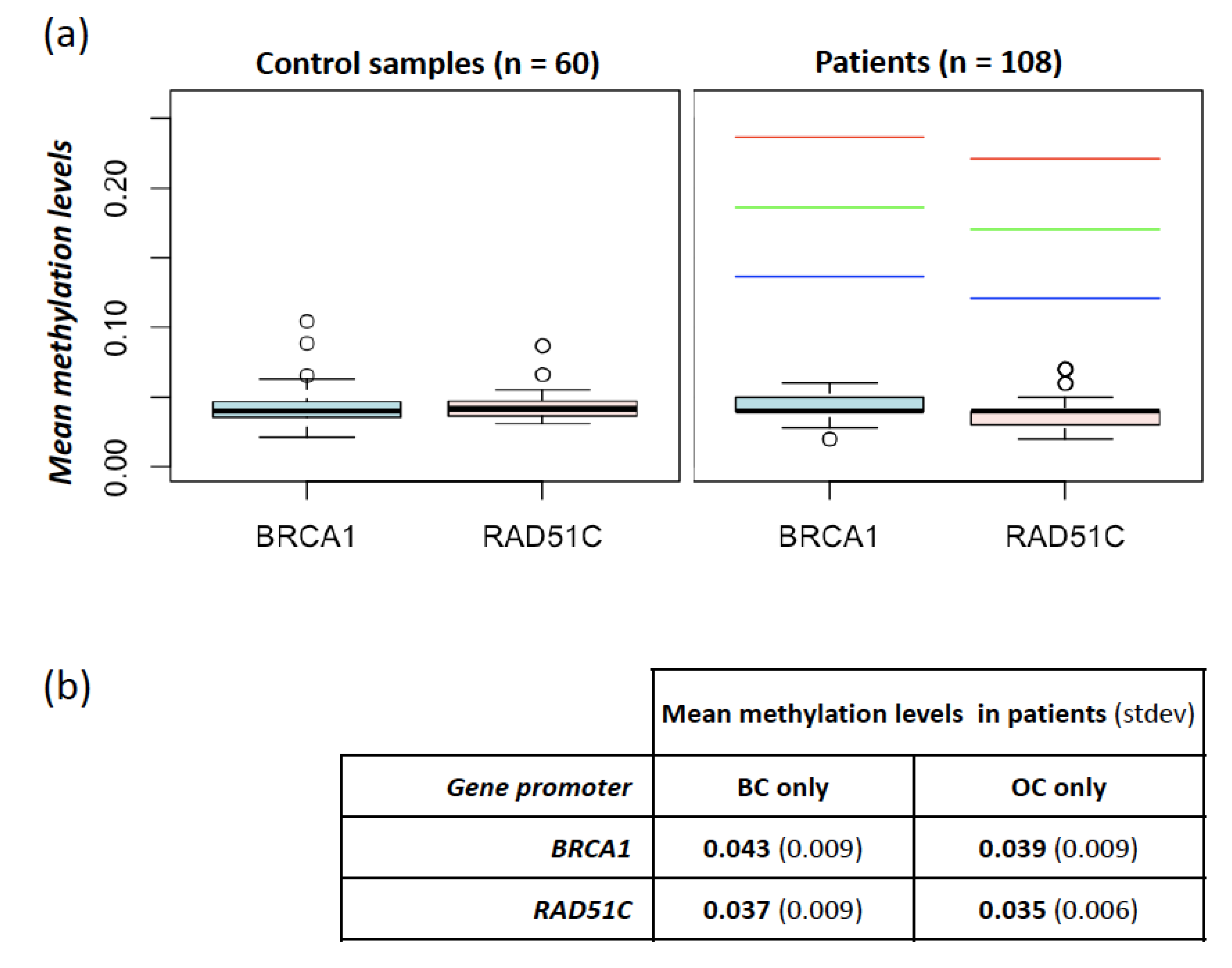
2. Results
BRCA1 and RAD51 Methylation Levels
3. Discussion
4. Materials and Methods
4.1. Study Subjects
4.2. DNA Extraction and Bisulfite Conversion
4.3. Epityper Mass Spectrometry
4.4. Statistical Pipeline
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Peltomäki, P. Epigenetic mechanisms in the pathogenesis of Lynch syndrome. Clin. Genet. 2014, 85, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Castillejo, A.; Hernández-Illán, E.; Rodriguez-Soler, M.; Pérez-Carbonell, L.; Egoavil, C.; Barberá, V.M.; Castillejo, M.I.; Guarinos, C.; Martínez-de-Dueñas, E.; Juan, M.J.; et al. Prevalence of MLH1 constitutional epimutations as a cause of Lynch syndrome in unselected versus selected consecutive series of patients with colorectal cancer. J. Med. Genet. 2015, 52, 498–502. [Google Scholar] [CrossRef] [PubMed]
- Veeck, J.; Ropero, S.; Setien, F.; Gonzalez-Suarez, E.; Osorio, A.; Benitez, J.; Herman, J.G.; Esteller, M. BRCA1 CpG island hypermethylation predicts sensitivity to poly(adenosine diphosphate) ribose polymerase inhibitors. J. Clin. Oncol. 2010, 28, e563–e564. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Diao, L.; Chen, Y.; Liu, Y.; Wang, C.; Ouyang, T.; Li, J.; Wang, T.; Fan, Z.; Fan, T.; et al. Promoter methylation of BRCA1 in triple-negative breast cancer predicts sensitivity to adjuvant chemotherapy. Ann. Oncol. 2013, 24, 1498–1505. [Google Scholar] [CrossRef]
- Evans, D.G.R.; van Veen, E.M.; Byers, H.J.; Wallace, A.J.; Ellingford, J.M.; Beaman, G.; Santoyo-Lopez, J.; Aitman, T.J.; Eccles, D.M.; Lalloo, F.I.; et al. A Dominantly Inherited 5′ UTR Variant Causing Methylation-Associated Silencing of BRCA1 as a Cause of Breast and Ovarian Cancer. Am. J. Hum. Genet. 2018, 103, 213–220. [Google Scholar] [CrossRef]
- Matros, E.; Wang, Z.C.; Lodeiro, G.; Miron, A.; Iglehart, J.D.; Richardson, A.L. BRCA1 promoter methylation in sporadic breast tumors: Relationship to gene expression profiles. Breast Cancer Res. Treat. 2005, 91, 179–186. [Google Scholar] [CrossRef]
- Iwamoto, T.; Yamamoto, N.; Taguchi, T.; Tamaki, Y.; Noguchi, S. BRCA1 promoter methylation in peripheral blood cells is associated with increased risk of breast cancer with BRCA1 promoter methylation. Breast Cancer Res. Treat. 2011, 129, 69–77. [Google Scholar] [CrossRef]
- Kontorovich, T.; Cohen, Y.; Nir, U.; Friedman, E. Promoter methylation patterns of ATM, ATR, BRCA1, BRCA2 and p53 as putative cancer risk modifiers in Jewish BRCA1/BRCA2 mutation carriers. Breast Cancer Res. Treat. 2009, 116, 195–200. [Google Scholar] [CrossRef]
- Wong, E.M.; Southey, M.C.; Fox, S.B.; Brown, M.A.; Dowty, J.G.; Jenkins, M.A.; Giles, G.G.; Hopper, J.L.; Dobrovic, A. Constitutional methylation of the BRCA1 promoter is specifically associated with BRCA1 mutation-associated pathology in early-onset breast cancer. Cancer Prev. Res. (Phila) 2011, 4, 23–33. [Google Scholar] [CrossRef]
- Hansmann, T.; Pliushch, G.; Leubner, M.; Kroll, P.; Endt, D.; Gehrig, A.; Preisler-Adams, S.; Wieacker, P.; Haaf, T. Constitutive promoter methylation of BRCA1 and RAD51C in patients with familial ovarian cancer and early-onset sporadic breast cancer. Hum. Mol. Genet. 2012, 21, 4669–4679. [Google Scholar] [CrossRef]
- Azzollini, J.; Pesenti, C.; Pizzamiglio, S.; Fontana, L.; Guarino, C.; Peissel, B.; Plebani, M.; Tabano, S.; Sirchia, S.M.; Colapietro, P.; et al. Constitutive BRCA1 Promoter Hypermethylation Can Be a Predisposing Event in Isolated Early-Onset Breast Cancer. Cancers 2019, 11, 58. [Google Scholar] [CrossRef] [PubMed]
- Lønning, P.E.; Berge, E.O.; Bjørnslett, M.; Minsaas, L.; Chrisanthar, R.; Høberg-Vetti, H.; Dulary, C.; Busato, F.; Bjørneklett, S.; Eriksen, C.; et al. White Blood Cell BRCA1 Promoter Methylation Status and Ovarian Cancer Risk. Ann. Intern. Med. 2018, 168, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Al-Moghrabi, N.; Al-Showimi, M.; Al-Yousef, N.; Al-Shahrani, B.; Karakas, B.; Alghofaili, L.; Almubarak, H.; Madkhali, S.; Al Humaidan, H. Methylation of BRCA1 and MGMT genes in white blood cells are transmitted from mothers to daughters. Clin. Epigenetics. 2018, 10, 99. [Google Scholar] [CrossRef]
- Lønning, P.E.; Eikesdal, H.P.; Løes, I.M.; Knappskog, S. Constitutional Mosaic Epimutations—A hidden cause of cancer? Cell Stress 2019, 3, 118–135. [Google Scholar] [CrossRef]
- Meindl, A.; Hellebrand, H.; Wiek, C.; Erven, V.; Wappenschmidt, B.; Niederacher, D.; Freund, M.; Lichtner, P.; Hartmann, L.; Schaal, H.; et al. Germline mutations in breast and ovarian cancer pedigrees establish RAD51C as a human cancer susceptibility gene. Nat. Genet. 2010, 42, 410–414. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Dicks, E.; Ramus, S.J.; Tyrer, J.P.; Intermaggio, M.P.; Hayward, J.; Edlund, C.K.; Conti, D.; Harrington, P.; Fraser, L.; et al. Contribution of Germline Mutations in the RAD51B, RAD51C, and RAD51D Genes to Ovarian Cancer in the Population. J. Clin. Oncol. 2015, 33, 2901–2907. [Google Scholar] [CrossRef]
- Li, N.; McInerny, S.; Zethoven, M.; Cheasley, D.; Lim, B.W.X.; Rowley, S.M.; Devereux, L.; Grewal, N.; Ahmadloo, S.; Byrne, D.; et al. Combined Tumor Sequencing and Case-Control Analyses of RAD51C in Breast Cancer. J. Natl. Cancer Inst. 2019, 111, 1332–1338. [Google Scholar] [CrossRef]
- Manié, E.; Popova, T.; Battistella, A.; Tarabeux, J.; Caux-Moncoutier, V.; Golmard, L.; Smith, N.K.; Mueller, C.R.; Mariani, O.; Sigal-Zafrani, B.; et al. Genomic hallmarks of homologous recombination deficiency in invasive breast carcinomas. Int. J. Cancer. 2016, 138, 891–900. [Google Scholar] [CrossRef]
- Cunningham, J.M.; Cicek, M.S.; Larson, N.B.; Davila, J.; Wang, C.; Larson, M.C.; Song, H.; Dicks, E.M.; Harrington, P.; Wick, M.; et al. Clinical characteristics of ovarian cancer classified by BRCA1, BRCA2, and RAD51C status. Sci. Rep. 2014, 4, 4026. [Google Scholar] [CrossRef]
- Bernards, S.S.; Pennington, K.P.; Harrell, M.I.; Agnew, K.J.; Garcia, R.L.; Norquist, B.M.; Swisher, E.M. Clinical characteristics and outcomes of patients with BRCA1 or RAD51C methylated versus mutated ovarian carcinoma. Gynecol. Oncol. 2018, 148, 281–285. [Google Scholar] [CrossRef]
- Rodríguez-Balada, M.; Roig, B.; Melé, M.; Salvat, M.; Martorell, L.; Borràs, J.; Gumà, J. Germline promoter hypermethylation in BRCA1 and BRCA2 genes is not present in hereditary breast cancer patients. Clin. Transl. Oncol. 2018, 20, 1226–1231. [Google Scholar] [CrossRef] [PubMed]
- Staaf, J.; Glodzik, D.; Bosch, A.; Vallon-Christersson, J.; Reuterswärd, C.; Häkkinen, J.; Degasperi, A.; Amarante, T.D.; Saal, L.H.; Hegardt, C.; et al. Whole-genome sequencing of triple-negative breast cancers in a population-based clinical study. Nat. Med. 2019, 25, 1526–1533. [Google Scholar] [CrossRef] [PubMed]
- Berrino, J.; Berrino, F.; Francisci, S.; Peissel, B.; Azzollini, J.; Pensotti, V.; Radice, P.; Pasanisi, P.; Manoukian, S. Estimate of the penetrance of BRCA mutation and the COS software for the assessment of BRCA mutation probability. Fam. Cancer 2015, 14, 117–128. [Google Scholar] [CrossRef] [PubMed]
| Inclusion Criteria | n. Patients | ||
|---|---|---|---|
| (1) | BC < 50 years | + high mutation probability | 73 |
| (2) | OC any age | + high mutation probability | 19 |
| (3) | TNBC < 55 years | + high mutation probability | 10 |
| (4) | BC < 50 years/TNBC any age | + OC any age | 6 |
| Breast Cancer Features | BC-Only Patients | BC+OC Patients | |
|---|---|---|---|
| No. | 83 | 6 | |
| 1st BC age (years) | Mean ± SD | 35 ± 6 | 46 ± 13 |
| Median | 33 | 43 | |
| Range | 24–51 | 36–71 | |
| 1st BC Invasive | Yes | 77 (93%) | 5 (83%) |
| No (CDIS) | 2 (2%) | 1 (17%) | |
| N.A.* | 4 (5%) | 0 | |
| 1st BC Histotype | Ductal | 66 (79%) | 5 (83%) |
| Lobular | 4 (5%) | 0 | |
| Mixed | 3 (4%) | 1 (17%) | |
| Other | 7 (8%) | 0 | |
| N.A. | 3 (4%) | 0 | |
| 1st BC Grade | I | 7 (8%) | 0 |
| II | 18 (22%) | 1 (17%) | |
| III | 30 (36%) | 1 (17%) | |
| N.A. | 28 (34%) | 4 (67%) | |
| 1st BC pT | Is | 1 (1%) | 1 (17%) |
| 1 | 35 (42%) | 1 (17%) | |
| 2 | 15 (18%) | 3 | |
| 3 | 2 (2,5%) | 0 | |
| 4 | 2 (2,5%) | 0 | |
| N.A. | 28 (34%) | 1 (17%) | |
| 1st BC ER | Pos | 39 (47%) | 1 (17%) |
| Neg | 26 (31%) | 1 (17%) | |
| N.A. | 18 (22%) | 4 (67%) | |
| 1st BC PgR | Pos | 38 (46%) | 0 |
| Neg | 27 (32%) | 2 (33%) | |
| N.A. | 18 (22%) | 4 (67%) | |
| 1st BC HER2 | Pos | 9 (11%) | 0 |
| Neg | 29 (35%) | 2 (33%) | |
| N.A. | 45 (54%) | 4 (67%) | |
| 1st BC TN | Yes | 10 (12%) | 1 (17%) |
| Ovarian Cancer Features | OC-Only Patients | BC + OC Patients | |
|---|---|---|---|
| No. | 19 | 6 | |
| OC Age (Years) | Mean ± SD | 58 ± 12 | 57 ± 12 |
| Median | 61 | 54.5 | |
| Range | 32–76 | 40–78 | |
| OC Histotype | Serous | 12 (63%) | 3 (50%) |
| Endometrioid | 3 (16%) | 3 (50%) | |
| Undifferentiated | 3 (16%) | 0 | |
| Clear cell | 1 (5%) | 0 | |
| OC Grade | II | 5 (26%) | 4 (67%) |
| III | 14 (74%) | 2 (33%) | |
| OC Stage | 1 | 1 (5%) | 2 (33%) |
| 2 | 1 (5%) | 0 | |
| 3 | 5 (26%) | 2 (33%) | |
| 4 | 2 (11%) | 0 | |
| N.A. | 10 (53%) | 2 (33%) | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tabano, S.; Azzollini, J.; Pesenti, C.; Lovati, S.; Costanza, J.; Fontana, L.; Peissel, B.; Miozzo, M.; Manoukian, S. Analysis of BRCA1 and RAD51C Promoter Methylation in Italian Families at High-Risk of Breast and Ovarian Cancer. Cancers 2020, 12, 910. https://doi.org/10.3390/cancers12040910
Tabano S, Azzollini J, Pesenti C, Lovati S, Costanza J, Fontana L, Peissel B, Miozzo M, Manoukian S. Analysis of BRCA1 and RAD51C Promoter Methylation in Italian Families at High-Risk of Breast and Ovarian Cancer. Cancers. 2020; 12(4):910. https://doi.org/10.3390/cancers12040910
Chicago/Turabian StyleTabano, Silvia, Jacopo Azzollini, Chiara Pesenti, Sara Lovati, Jole Costanza, Laura Fontana, Bernard Peissel, Monica Miozzo, and Siranoush Manoukian. 2020. "Analysis of BRCA1 and RAD51C Promoter Methylation in Italian Families at High-Risk of Breast and Ovarian Cancer" Cancers 12, no. 4: 910. https://doi.org/10.3390/cancers12040910
APA StyleTabano, S., Azzollini, J., Pesenti, C., Lovati, S., Costanza, J., Fontana, L., Peissel, B., Miozzo, M., & Manoukian, S. (2020). Analysis of BRCA1 and RAD51C Promoter Methylation in Italian Families at High-Risk of Breast and Ovarian Cancer. Cancers, 12(4), 910. https://doi.org/10.3390/cancers12040910





